Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
** I WAS RIGHT ALL ALONG... APOLOGIES ACCEPTED **
Until they get rid of JJ and those on NRXP's board who were culpable in creating this mess I don't think things will get much better for the company's long-term stability. This has been a disaster. It's the second SPAC I've been invested in, and both had high and lofty goals with a seemingly stellar business plan, but the end result was a huge loss for most all who bought into the story.
I sold my position in NRXP several weeks ago, but I seriously wish everyone who is still holding the very best of luck. Maybe some way this mess can be cleaned up and the company can make a turnaround.
IF EUA WAS CLOSE WOULDNT THEY WAIT TO DILUTE
Walker, yes, it sure looks like neuro failed to deliver
on all the big talk and pr, on following the CA, and even SEC rules.
Yes, its been one disappointment and failure after another with lack of FDA approvals, law suits galore, and until there are some real FDA approvals and or solid working agreements/real cooperation with RLF, settlement of the law suits, these lower lows many continue, IMHO.
I will be surprised if any meaningful mediation occurs and I think neuro has been backed into a corner of its own making, with no escape. All IMHO.
Bree Life looks DOA after the deal with yet unapproved NVAX for up to 10 mil units in Israel, and neuro seems unwilling or unable to end its standoff with RLF. Perhaps way down the road they may get some orders, if Covid is even still around.
I think we investors have all been very disappointed, and I wish good luck to everyone especially RLF and even the mistake plagued NEURO.
I hope NRX makes lotsa $$$$ on something cause they might need it to pay large potential judgements and avoid CH 7 or 11.
Joe, this has been a disaster for those of us that believed the influence level that JJ and Neuro claimed in its lead up to the SPAC deal.
While we made nice gains pre-deal, it has been not so good since, but still up 5-figures ... and have stopped the bleeding.
Peace,
powerwalker
P.S. Still watching for a re-entry point after some good news in both companies.
Walker, thanks for sharing...
Gotta wonder how much of this 25 mil will be for legal fees, SEC judgements, and the like.
Interesting that Neuro STILL does not acknowledge any partnership agreement with RLF in their news releases, like they never violated the agreement in so many ways, or the agreement does not even exist.
Also no public mention by neuro of all the lawsuits by all the various law firms, like the legal / SEC problems do not exist.
At the moment, the stock is back to a 52 week low of $2.80,
Not good sign.
Red...You may have lots of opportunity to add, and good luck, as
even Israel is NOT buying jjs Brilife , but yet UNAPPROVED Novavax.
https://www.jpost.com/breaking-news/article-694872
The Israeli government has agreed a deal with US-based biotech company Novavax to buy five million COVID-19 vaccine doses, the Health Ministry announced on Friday.
The Novavax vaccines, expected to arrive in Israel over the next few months, are yet to receive the nod of approval from the US Food and Drug Administration (FDA). As part of the deal, Israel has the option to purchase an extra five million vaccine doses.
Personally, I feel the winds are more favorable behind RLF, instead of NRX with all its legal issues.
Definitely a lot more of a lotto than it used to be.
Honestly, this is the first time I've been involved with warrants. I expected they'd go lower since there's an 11.50 executing fee. I'm not a chart guy, but I'd expect warrants to end up near 1.00-1.10 if commons go much lower.
Lotto or not, I'm going to ride this one to the end. So, lower price just means adding more to my position.
Red, I really have no expectations on the warrants, and I have not followed them, thank goodness. To me, at least, right now, to answer your question, it seems like it could be a lotto ticket, all IMHO.
What are your expectations might be a better question ??
What do you expect warrants to go to? I thought, with limited experience, that they would have dropped further by now. There's just $1.50 separating them.
https://stockcharts.com/h-sc/ui?s=NRXP&p=D&b=5&g=0&id=p45922685032
Technically this does not look good, IMHO. ....Back in the 2's.
https://ir.nrxpharma.com/news-events/press-releases/detail/115/nrx-announces-publication-of-initial-findings-of-brilife
Phase 2 test results of, lucky number, 13 whole people, may and might be the luck needed to roll this out to BILLIONS in profits for a company caught up in plenty of legal problems.
NRAnnounces Publication of Initial Findings of BriLife® Vaccine-produced Antibodies Against Omicron Variant
JANUARY 27, 2022 6:51AM EST
Download as PDF
- Israel Institute for Biological Research files initial scientific results of BriLife vaccine effectiveness in producing neutralizing antibody against Omicron Variant
- Neutralizing antibody level against Omicron is approximately 1/3 of the comparable antibody level against the original Corona Virus
- Ten of 13 tested patients vaccinated during phase 2 trials demonstrated measurable level of Omicron neutralizing antibody.
- Detected evidence that BriLife vaccine spontaneously acquired Omicron mutation suggests that vaccine will continue to evolve immunity towards future variants
RADNOR, Pa., Jan. 27, 2022 /PRNewswire/ -- NRx Pharmaceuticals (NASDAQ: NRXP), a clinical-stage, biopharmaceutical company has received scientific evidence from the Israel Institute for Biological Research (IIBR) that the BriLife® vaccine may produce effective levels of neutralizing antibody against the Omicron variant of the SARS-CoV-2 virus.1 More importantly, many of the mutations (alterations) that cause the Omicron variant spike protein to differ from the spike protein of the original SARS-CoV-2 virus that causes COVID-19 have been identified in the spike protein of the BriLife vaccine. This natural evolution of the BriLife vaccine suggests that the vaccine may continue to evolve to address future Variants of Concern (VOCs). Unlike the current mRNA vaccines and attenuated virus vaccines, the BriLife vaccine is a live, viral vector vaccine in which the spike protein of the SARS-CoV-2 virus has been added to a benign virus, called VSV. A similar viral vector was used to create the successful, FDA-approved vaccine against Ebola virus.
(PRNewsfoto/NRx Pharmaceuticals)
The findings released yesterday were based on blood samples (sera) of patients vaccinated during the phase 2 trial of the BriLife vaccine conducted in Israel. These sera indicated a mean neutralization titer (NT50) of 53 against Omicron vs. a titer of 152 against the original wild-type virus and 131 against the Delta variant. This 3-fold change against the wild-type virus may be compared to the 20-fold decrement associated with mRNA vaccines, which helps explain the high infectivity of Omicron in currently vaccinated individuals.2 The IIBR report documents that 10 of 13 tested sera demonstrated clinically detectable levels of Omicron-neutralizing antibody.
The IIBR report concludes that, "Taken together, our data indicates that BriLife®-induced antibodies maintain neutralizing potential against all tested variants, and most importantly against delta, and the recently emerged omicron VOCs. We suggest that spontaneously-acquired mutations that occurred during BriLife® development and correspond to naturally-occurring mutations of SARSCoV-2 variants, may increase the potential of BriLife® to maintain effectiveness against current SARS-CoV-2 variants, and potentially against future VOCs."
"We are enormously encouraged by the recent findings of the IIBR," said Prof. Jonathan Javitt, MD, MPH, CEO and Chairman of NRx. "At a time when the majority of western populations have been vaccinated with mRNA vaccines, there is a clear need for a booster vaccine that can broaden immunity against current and future variants of concern. The BriLife vaccine has now demonstrated potential to continue to evolve in a manner that has the potential to protect against variants that are not yet known, and is planned to be tested as a booster in a phase 2 trial that will include Israel and other partner countries,"
Good luck and GOD bless,
NRx Announces Publication of Initial Findings of BriLife® Vaccine-produced Antibodies Against Omicron Variant
https://ir.nrxpharma.com/news-events/press-releases/detail/115/nrx-announces-publication-of-initial-findings-of-brilife
- Israel Institute for Biological Research files initial scientific results of BriLife vaccine effectiveness in producing neutralizing antibody against Omicron Variant
- Neutralizing antibody level against Omicron is approximately 1/3 of the comparable antibody level against the original Corona Virus
- Ten of 13 tested patients vaccinated during phase 2 trials demonstrated measurable level of Omicron neutralizing antibody.
- Detected evidence that BriLife vaccine spontaneously acquired Omicron mutation suggests that vaccine will continue to evolve immunity towards future variants
RADNOR, Pa., Jan. 27, 2022 /PRNewswire/ -- NRx Pharmaceuticals (NASDAQ: NRXP), a clinical-stage, biopharmaceutical company has received scientific evidence from the Israel Institute for Biological Research (IIBR) that the BriLife® vaccine may produce effective levels of neutralizing antibody against the Omicron variant of the SARS-CoV-2 virus.1 More importantly, many of the mutations (alterations) that cause the Omicron variant spike protein to differ from the spike protein of the original SARS-CoV-2 virus that causes COVID-19 have been identified in the spike protein of the BriLife vaccine. This natural evolution of the BriLife vaccine suggests that the vaccine may continue to evolve to address future Variants of Concern (VOCs). Unlike the current mRNA vaccines and attenuated virus vaccines, the BriLife vaccine is a live, viral vector vaccine in which the spike protein of the SARS-CoV-2 virus has been added to a benign virus, called VSV. A similar viral vector was used to create the successful, FDA-approved vaccine against Ebola virus.
The findings released yesterday were based on blood samples (sera) of patients vaccinated during the phase 2 trial of the BriLife vaccine conducted in Israel. These sera indicated a mean neutralization titer (NT50) of 53 against Omicron vs. a titer of 152 against the original wild-type virus and 131 against the Delta variant. This 3-fold change against the wild-type virus may be compared to the 20-fold decrement associated with mRNA vaccines, which helps explain the high infectivity of Omicron in currently vaccinated individuals.2 The IIBR report documents that 10 of 13 tested sera demonstrated clinically detectable levels of Omicron-neutralizing antibody.
The IIBR report concludes that, "Taken together, our data indicates that BriLife®-induced antibodies maintain neutralizing potential against all tested variants, and most importantly against delta, and the recently emerged omicron VOCs. We suggest that spontaneously-acquired mutations that occurred during BriLife® development and correspond to naturally-occurring mutations of SARSCoV-2 variants, may increase the potential of BriLife® to maintain effectiveness against current SARS-CoV-2 variants, and potentially against future VOCs."
"We are enormously encouraged by the recent findings of the IIBR," said Prof. Jonathan Javitt, MD, MPH, CEO and Chairman of NRx. "At a time when the majority of western populations have been vaccinated with mRNA vaccines, there is a clear need for a booster vaccine that can broaden immunity against current and future variants of concern. The BriLife vaccine has now demonstrated potential to continue to evolve in a manner that has the potential to protect against variants that are not yet known, and is planned to be tested as a booster in a phase 2 trial that will include Israel and other partner countries,"
About NRx Pharmaceuticals
NRx Pharmaceuticals (NRx) draws upon more than 300 years of collective, scientific, and drug-development experience to bring improved health to patients. The Company is developing the BriLife™ Covid vaccine, developed by the Israel Institute for Biological Research, under an exclusive license from the Israel Ministry of Defense. NRx is additionally developing ZYESAMI® (aviptadil) for patients with COVID-19, and has been granted Fast Track designation by the US Food and Drug Administration (FDA), and is currently undergoing phase 3 trials funded by the US National Institutes of Health, the Biomedical Advanced Research and Development Authority (BARDA) of the US Department of Health and Human Services, and the Medical Countermeasures program, part of the US Department of Defense. The FDA has additionally granted Breakthrough Therapy Designation, a Special Protocol Agreement, and a Biomarker Letter of Support to NRx for NRX-101, an investigational medicine to treat suicidal bipolar depression. NRX-101 is currently in Phase 3 trials, with readouts expected in 2022.
NRx is led by executives who have held senior roles at Allergan, J&J, Lilly, Novartis, Pfizer, and the US FDA. NRx is chaired by Prof Jonathan Javitt, MD, MPH, who has held leadership roles in six biotechnology startup companies with public exits and been appointed to advisory roles in four US Presidential Administrations. The NRx board includes Dr. Sherry Glied, former US Assistant Secretary for Health (ASPE), Daniel E. Troy, JD, former Chief Counsel of the US FDA, Chaim Hurvitz, former director of Teva and President of the Teva International Group, and General H.R. McMaster, Ph.D. (US Army, Ret.) the 26th United States National Security Advisor.
Cautionary Note Regarding Forward-Looking Statements
This announcement of NRx Pharmaceuticals, Inc. includes "forward-looking statements" within the meaning of the "safe harbor" provisions of the US Private Securities Litigation Reform Act of 1995, which may include, but are not limited to, statements regarding our financial outlook, product development, business prospects, and market and industry trends and conditions, as well as the Company's strategies, plans, objectives, and goals. These forward-looking statements are based on current beliefs, expectations, estimates, forecasts, and projections of, as well as assumptions made by, and information currently available to, the Company's management.
The Company assumes no obligation to revise any forward-looking statement, whether as a result of new information, future events, or otherwise. Accordingly, you should not place reliance on any forward-looking statement, and all forward-looking statements are herein qualified by reference to the cautionary statements set forth above.
CORPORATE CONTACT:
INVESTOR RELATIONS
Jack Hirschfield –
Eric Goldstein
Head of Corporate Communications, NRx
Managing Director – LifeSci Advisors
jhirschfield@nrxpharma.com
egoldstein@lifesciadvisors.com
Available data on ACTIV-3b recruitment shows 1.6 patients added per day over five months.
We are at approx 427 patients.
At this enrollment, it should take 133 days to add the remaining 213 patients, estimated June 8 2022.
The study is looking for 180 day results on some things, which means it should be about 373 days until this study can be available for an NDA application, or the first week of February 2023.
08/18/21 140 Patients Added Days Patients/day
09/30/21 232 92 43 2.1395348837
11/02/21 300 68 33 2.0606060606
11/22/21 320 20 20 1
12/14/21 349 29 22 1.3181818182
01/26/22 427 78 43 1.8139534884
Some PRs say "approx" x patients, "over" x patients. I chose PRs that I believe make this a correct estimate.
NRx Receives Initial Report of Patient Safety and Survival from Right to Try use of ZYESAMI® (aviptadil) during Omicron Surge
JANUARY 26, 2022 6:51AM EST
Download as PDF
-A Southwestern hospital has reported safety data collected under the Federal Right to Try law indicating that 16 of 19 patients (84%) with COVID-19 respiratory failure treated with ZYESAMI® by Dec 31, 2021 have survived the ICU.
-No Serious Adverse Events were reported
-Patients were treated during the Omicron Surge
-Patients were treated at first onset of respiratory failure after exhausting remdesivir and other approved therapies
-Data are being included provided by NRx to FDA in support of ongoing application to FDA for Emergency Use Authorization
RADNOR, Pa., Jan. 26, 2022 /PRNewswire/ -- NRx Pharmaceuticals (NASDAQ: NRXP), a clinical-stage, biopharmaceutical company, has received a first safety report from a Southwestern hospital where physicians have administered ZYESAMI® (Aviptadil) to patients with COVID-19 respiratory failure. These patients were treated under the Federal Right to Try Law that gives access to investigational medicines for patients who have been diagnosed with life-threatening diseases or conditions, who have tried all approved treatment options, and who are unable to participate in a clinical trial to access certain unapproved treatments. This Right to Try use of ZYESAMI occurred during the current Omicron surge, although patients were not necessarily tested for the specific COVID variant that caused their ICU admission.
NRx Logo
The safety update report received from the hospital indicated that of the first 19 patients treated by Dec. 31, 2021, three had died and 16 (84%) were reported to be alive by Jan. 22, 2021. At the time of the report, 14 of these 16 patients had been discharged to a rehabilitation center or home and two remained in the hospital. No Serious Adverse Events (SAEs) related to ZYESAMI were reported. These data were included in an application to FDA for Emergency Use Authorization for the treatment of patients with critical COVID-19 who are at immediate risk of death from respiratory failure despite treatment with approved therapy including remdesivir. NRx will continue to update the safety database collected under the Right to Try law on an ongoing basis.
The data being received from hospitals and patients treated under the Right to Try law do not involve a control group and are not part of a research study designed to test efficacy.
ZYESAMI continues to be tested in the ongoing NIH-sponsored ACTIV-3b (TESICO) trial that has now accrued two-thirds of its targeted enrollment.
NRx Receives Initial Report of Patient Safety and Survival from Right to Try use of ZYESAMI® (aviptadil) during Omicron Surge
https://ir.nrxpharma.com/news-events/press-releases/detail/114/nrx-receives-initial-report-of-patient-safety-and-survival
The root of that lawsuit:
The NRx Pharma class action lawsuit alleges that, throughout the Class Period,
defendants made false and misleading statements and failed to disclose that:
(i) the ZYESAMI EUA application contained insufficient data regarding the
potential benefits and risks of ZYESAMI;
Is going to be very difficult to prove in court.
The EUA app contained "insufficient data?" How is that going to be proven that NRX knew that prior to submitting the EUA app?
Hindsight is always crystal clear and investing in the stock market is always at your own risk with no guarantees of success.
The only way that lawsuit is successful is if they have more money to pour into the case than NRX does, and NRX simply caves and pays up to make the lawsuit go away.
This lawsuit isn't going anywhere.
On the bigger picture...
I'm pissed too that this stock is tanking.
But that isn't fault of NRX.
This is clearly, 100% the fault of the FDA and the countless government functionaries who are all vax all the time. They've absolutely neglected therapeutics, unless they are tied to their big pharma donors.
It's not like NRX is asking for full licensing approval at this point. There is no reason at all, given the known data for about a year and a half now, that Zyesami should be continued to be denied an Emergency Use Authorization.
This is all about Political Science, and not actual Science.
The people who should be facing lawsuits are the FDA, the CDC, Fraudchi et. al. for withholding viable therapeutics from getting the same EUA treatment that the brand-new vax's got.
I would really like to see jj put his own money into a significant amount of NRX stock bought at market prices.
Interesting analogy with the "hat trick" prsentation.
It might even help a few hockey players to buy in, but
its difficult not to consider with all the
EXPERIENCED people on the management page, and all the
EXPERIENCED people listed on the board, as to how one
company could so quickly accumulate so many lawsuits
and lawyers probing into NRX for legal issues.
**** PROVEN RIGHT YET AGAIN.. APOLOGIES ACCEPTED ****
https://stockcharts.com/h-sc/ui?s=NRXP&p=D&b=5&g=0&id=p38879565319
technicals not lookin very good here right at the moment, another new low. Could the 2's be in the cards ?????
Sure would be nice if NRX would break out the two lines of business
between the VAX and AVIPTADIL/ZYESAMI on the F/S.
Very difficult/impossible to follow what is going on with that in the F/S for the two completely different lines of business.
Lawsuits are Turning into Get in Line and take a number..
Now Another.... Robbins Geller Rudman & Dowd
https://www.bloomberg.com/press-releases/2022-01-20/alert-nrx-pharmaceuticals-inc-investors-with-substantial-losses-have-opportunity-to-lead-class-action-lawsuit-nrxp-nrxpw
https://www.lawyer-monthly.com/2022/01/alert-nrx-pharmaceuticals-inc-investors-with-substantial-losses-have-opportunity-to-lead-class-action-lawsuit-nrxp-nrxpw/
Gotta wonder if DENIAL is the main defense here also.
So far not a word from the company.
Posting this to both boards..
If they would take their disagreements behind closed doors via conflict resolution... put egos aside... then they both stand much more to gain working together rather than publicly bitch slapping each other. They both owe that to all us shareholders.
And still... as a safety fuse... If you look at the past price of NRX going back from the IPO in 2018 all the way up to Dec 2020, it averaged 10 to 11 bucks per share. RLFTF was also higher going back last year till the FDA did not act on approval. So to me there is a possibility that these two positions still hold some old inherent value aside from the current therapeutics at hand. There is also Brilife and other products on the table waiting in the wings.
Not sure if short selling and manipulating is happening... but for sure there is a lack of confidence... for now.
Been invested in NRXP and RLFTF since Aug '20 and still bullish.
Looking pretty grim today for NRX. New lows again.
Short of a quick and compete settlement with RLF, and quick approval
by the FDA for AVIPTADIL / ZYESAMI, it looks like NRX is in for
some heavy stormy hurricane weather.
Perhaps Brilife can manage a big win to NRX and a miraculous come back for NRX stock price. It continues down, even at this level.
Those days of double digit prices sure look long gone now.
NRx Pharmaceuticals Announces Expansion of ZYESAMI® (aviptadil) US Expanded Access and Right to Try Programs for Patients with COVID-19 Respiratory Failure who have Exhausted All Approved Treatments
Good luck and GOD bless,
There is a new Dr Joey Johnson video--LOTS of NRXP info
Looks like BriLife could potentially be the path for getting the virus behind us.
NRx Responds to Relief’s Allegations of January 14, 2022
https://ir.nrxpharma.com/news-events/press-releases/detail/112/nrx-responds-to-reliefs-allegations-of-january-14-2022
Neither NRx nor Jonathan Javitt accused current Relief management or board of a criminal past. The lawsuit identifies FINRA actions against Dr. Sevelraju, prior securities investigation, and civil fines against Dr. John Paul Waymack, and a prior securities fraud conviction, incarceration, and SEC fines against Adam Gottbetter.
NRx provided all financial records to Relief on December 6, 2021, yet Relief advised the SEC on December 16, 2021, that it received no records from NRx.
NRx looks forward to meeting Relief in mediation on February 22, 2022.
RADNOR, Pa., Jan. 14, 2022 (GLOBE NEWSWIRE) -- NRx Pharmaceuticals (NASDAQ: NRXP), a clinical-stage biopharmaceutical company, responded to today’s press release issued by Relief Therapeutics.
Allegations of securities violations and criminal behavior
Relief has accused NRx and its CEO of issuing false and misleading statements about the past actions of Relief’s Board and Management. Relief has clearly misread those statements. At no time has NRx accused any member of Relief’s board of current or past criminal activity. NRx has documented past history of FINRA disciplinary action against Dr. Raghuram Selvaraju, which may readily be verified on the FINRA Brokercheck website1. At least one of those disciplinary events may be connected to Selvaraju’s former association in his role with TYME Technologies with Adam Gottbetter, who was ultimately convicted of securities fraud, incarcerated, fined by the SEC, and banned from the penny stock industry for life.23 Mr. Gottbetter introduced himself to NRx as Relief’s head of mergers and acquisitions. The complaint filed by NRx also identifies Relief’s Regulatory Consultant, John Paul Waymack, MD, ScD, as having been investigated by the Israel Securities Agency (ISA), settled for civil fines with the ISA, and having pled his fifth amendment rights before the US SEC in connection with Kitov Pharmaceuticals.45 NRx further notes that Peter DeSvastich, who was a founding director of Relief’s board, similarly served as Chairman of TYME Technologies and worked with Mr. Gottbetter and Dr. Selvaraju in that capacity.
Allegations of failure to deliver financial records
Relief has alleged that NRx failed to deliver promised detailed financial records in time for Relief to attend the mediation on January 5, 2022, to which Relief acknowledges it committed to attend. Relief acknowledged receipt on December 6, 2021, of more than 1000 pages of detailed financial information to support research and development costs associated with the development of ZYESAMI. This information was delivered more than 30 days in advance of the January 5, 2022 mediation date. Yet, inexplicably on December 16, 2021, Relief filed a form 20F with the US SEC in which Relief stated (P. 66) over the signatures of Mr. Weinstein and Dr. Selvaraju that “we note that we have not received invoices from NeuroRx that are anywhere close to the amounts that NeuroRx claims are allegedly due. Relief continues to have no idea of how the amount allegedly owed that is set forth in NRx’s SEC filings are calculated.”
The crux of the matter
Relief persists in issuing false and misleading press releases in order to attempt to obscure several basic truths, as outlined in NRx’s complaint before the New York State Supreme Court.
Rather than fund the development of ZYESAMI (aviptadil) as required by the collaboration agreement, Relief breached and repudiated its obligations under the agreement and resorted to malicious defamation of NRx and its CEO in order to distract from a simple breach of contract matter.
NRx looks forward to mediating this matter on February 22, 2022.
Good luck and GOD bless,
CEO Update: BriLife Investigational Vaccine for COVID-19 as a Booster
At this week’s 2022 H.C. Wainwright BioConnect Virtual Conference, NRx identified the plans to move forward with a vanguard trial of the BriLife investigational vaccine for COVID-19 as a heterologous booster for those already vaccinated with mRNA vaccines.
https://www.nrxpharma.com/ceo-update-brilife-investigational-vaccine-for-covid-19-as-a-booster/
This newly-announced direction was likely a surprise, given our initial focus on unvaccinated patients. However, as the pool of unvaccinated people in Western countries shrinks, scientists and policymakers are rapidly recognizing the fragility of the immunity created with first-generation vaccines. The Omicron wave has created a sudden focus on the importance of using more than one type of vaccine to create true herd immunity, particularly if the next-generation vaccines are able to provide mucosal immunity, which mRNA vaccines do not.
It is well recognized that mRNA vaccines protect the vaccinated individual from some of the worst effects of COVID-19 but do not create an immune response in the mucosal cells that line the nose, throat, and lungs. As we have seen with this latest Omicron wave, vaccinated individuals incubate the Corona Virus in their nasal cavity and then can spread it to others.
There is reason to believe that a viral vector vaccine that binds to cells that express angiotensin converting enzyme 2 (ACE2) receptors, such as the BriLife investigational vaccine, has the potential to create true mucosal immunity and provide a different type of immunity than the mRNA vaccines. Generally speaking, our ability to treat and protect patients only grows with the addition of diverse new tools and science.
A path forward that involves a heterologous vaccination strategy (i.e., several types of vaccination) for each person is rapidly gaining favor, and this path forward has now been validated by leading experts in the European Medicines Agency and the World Health Organization. Those wishing to understand the science behind this should read the attached briefing documents. We know mRNA vaccines provide superior immediate immunity to the specific antigens they target compared to viral vector vaccines but also rapidly hit an immunologic ceiling and are unable to provide effective, long-lasting immunity against COVID-19 and its variants. The infection rate among vaccinated people throughout the world demonstrates that breakthrough COVID-19 infections are almost becoming common in those vaccinated with mRNA vaccines.
Israel was not an ideal country for the next phase of BriLife research as a primary vaccine, given the extensive vaccination rate with mRNA vaccines. However, Israel and its citizens, who are vaccinated predominately with mRNA vaccines, represent a prime location for the launch of our BriLife heterologous booster program. NRx is currently designing the trial according to guidelines that are emerging in Europe and from the World Health Organization. The world is rapidly realizing that a single vaccination approach against COVID-19 is likely to be insufficient to meet the challenge.
In the past weeks, government representatives from the United States, Hungary, France, and other nations have approached NRx regarding updates involving the BriLife investigational vaccine. As data becomes available, NRx will share that information with those governments.
We expect to post the initial protocol on Clinicaltrials.gov within the next few weeks.

Good luck and GOD bless,
I am surprised this did not move the stock price in a positive direction.
A MUST READ
NRx Pharmaceuticals Files Counterclaim Against its Former Collaboration Partner, Relief Therapeutics
JANUARY 12, 2022 5:48PM EST
- Documents NRx's development of a potentially lifesaving drug for treatment of Covid-19 despite Relief's breach and repudiation of the collaboration agreement
- Details Relief's false claims of worldwide rights to Aviptadil and to ongoing phase 3 research, misleading NRx and the public
- Identifies Raghuram Selvaraju's history of collaborating with Relief principals and associates on prior matters that have led to sanctions, fines, and incarceration
- Documents Relief's attempt to extort NRx into using Relief's ineffective and potentially harmful formulation of Aviptadil
- Debunks the libelous claim that NRx caused harm to patients in Europe by withholding patient data
https://ir.nrxpharma.com/news-events/press-releases/detail/111/nrx-pharmaceuticals-files-counterclaim-against-its-former
Good luck and GOD bless,
Jan. 11, 2022 (GLOBE NEWSWIRE) -- NRx Pharmaceuticals (NASDAQ: NRXP), a clinical-stage, biopharmaceutical company, expanded on the information provided Monday at the H.C. Wainwright BioConnect Virtual Conference regarding the BriLife™ investigational vaccine for COVID-19.
Last week, NRx met with experts from the Israel Institute for Biological Research (IIBR) to review data and research related to the ability of the BriLife vaccine to induce neutralizing antibodies against the Omicron variant. Based on the preliminary findings, NRx is currently designing a phase 2b/3 study of the BriLife vaccine as a booster to protect against COVID-19 variants of concern including the Omicron variant. Patients in the study will be fully vaccinated with mRNA vaccines. It is anticipated that the study will begin in the first quarter of 2021 in Israel and will be expanded in coordination with the health ministries of several countries. The IIBR previously published initial serological findings documenting a neutralizing antibody response against the Delta variant that was comparable to the response against the wild-type SARS-CoV-2 virus.
“We have seen the deadly impact caused by COVID-19 and its increasing number of variants, and we are eager to determine the immunity-building impact the BriLife vaccine may offer on top of that already conferred by baseline mRNA vaccination,” said Prof Jonathan Javitt, MD, MPH, Chairman and CEO of NRx Pharmaceuticals. “We are working closely with the experts at the IIBR to design a study that we hope increases BriLife’s accelerated path to regulatory approval.”
NRx was contacted in late December by representatives of the US Department of Health and Human Services and several European Governments with a request to present Omicron findings.
In further news, the Israel Ministry of Health recently approved a study investigating the NanoPass MicronJet™ intradermal injection system for the BriLife vaccine. The NanoPass system, invented in Israel, uses a patented microneedle system to deliver the vaccine into the skin with minimal discomfort.
This approach is especially promising for the BriLife vaccine because it binds to angiotensin-converting enzyme 2 (ACE2) receptors, which are present in significantly larger quantities in human skin cells than in muscle cells where traditional vaccines are injected. In addition, early data with other vaccines suggests that intradermal delivery of BriLife may result in a more robust immune response at substantially lower vaccine dosing.
As these studies are moving towards initiation, NRx continues technology transfer and scale-up activities in anticipation of commercial scale manufacture by Q4 2022
Good luck and GOD bless,
george, it is live at 9:00 AM per HCW's web site:
H.C. Wainwright BIOCONNECT Virtual Conference - January 10 - 13, 2022
NRx Pharmaceuticals, Inc.
Live on Monday, 1/10 at 9:00 AM(Eastern Standard Time)
NRx Pharmaceutical’s Chairman and Chief Executive Officer Jonathan Javitt to Present Company Business Update at H.C. Wainwright BioConnect Virtual Conference
RADNOR, Pa., Jan. 07, 2022 (GLOBE NEWSWIRE) — NRx Pharmaceuticals (NASDAQ: NRXP), a clinical-stage, biopharmaceutical company, today announced its Chairman of the Board and Chief Executive Officer, Prof Jonathan Javitt, MD, MPH, will be presenting updates to the Company’s business at the H.C. Wainwright BioConnect Virtual Conference.
Details of the presentation can be found below:
Date: Monday, January 10, 2022
Time: 7:00 AM EST
Link to Presentation: https://journey.ct.events/view/1464b340-bd79-4a96-ae16-572a470d569b
About NRx Pharmaceuticals
NRx Pharmaceuticals (NRx) draws upon more than 300 years of collective, scientific, and drug-development experience to bring improved health to patients. The Company is developing the BriLife™ Covid vaccine, developed by the Israel Institute for Biological Research, under an exclusive license from the Israel Ministry of Defense. NRx is additionally developing ZYESAMI® (aviptadil) for patients with COVID-19, and has been granted Fast Track designation by the US Food and Drug Administration (FDA), and is currently undergoing phase 3 trials funded by the US National Institutes of Health, the Biomedical Advanced Research and Development Authority (BARDA) of the US Department of Health and Human Services, and the Medical Countermeasures program, part of the US Department of Defense. The FDA has additionally granted Breakthrough Therapy Designation, a Special Protocol Agreement, and a Biomarker Letter of Support to NRx for NRX-101, an investigational medicine to treat suicidal bipolar depression. NRX-101 is currently in Phase 3 trials, with readouts expected in 2022.
NRx is led by executives who have held senior roles at Allergan, J&J, Lilly, Novartis, Pfizer, and the US FDA. NRx is chaired by Prof Jonathan Javitt, MD, MPH, who has held leadership roles in six biotechnology startup companies with public exits and been appointed to advisory roles in four US Presidential Administrations. The NRx board includes Dr. Sherry Glied, former US Assistant Secretary for Health (ASPE), Daniel E. Troy, JD, former Chief Counsel of the US FDA, Chaim Hurvitz, former director of Teva and President of the Teva International Group, and General H.R. McMaster, Ph.D. (US Army, Ret.) the 26th United States National Security Advisor.
Good luck and GOD bless,
Georgie I feel good about this one. When is Anvs getting news check out bivi May be better than Sava and Anvs super low float
NRx Pharmaceuticals Submits Emergency Use Authorization Application to US Food and Drug Administration for ZYESAMI® (aviptadil) to Treat Patients at Immediate Risk of Death from COVID-19 Despite Treatment with Remdesivir and Other Approved Therapies
Requested indication focuses on patients whose respiratory failure has progressed despite treatment with Remdesivir or other approved therapies
Patients treated with ZYESAMI vs. placebo demonstrated statistically significant (P=.03), 2.8-fold increased odds of being alive and free of respiratory failure at day 28 and day 60 and a 4-fold increased odds of surviving to 60 days (P=.006)
Patients at highest risk – those on ventilators at time of randomization – demonstrated a 10-fold increased odds of survival (P=.03)
US National Institutes of Health-sponsored trial comparing ZYESAMI and Remdesivir individually and in combination continues to demonstrate safety and has enrolled more than 350 patients
RADNOR, Pa., Jan. 5, 2022 /PRNewswire/ — NRx Pharmaceuticals (Nasdaq: NRXP) announced today that it has submitted an application for Emergency Use Authorization (EUA) to the US Food and Drug Administration (FDA) for the use of ZYESAMI® (aviptadil) in patients with Critical COVID-19 who are at immediate risk of death from Respiratory Failure despite treatment with approved therapy including Remdesivir and who are ineligible for enrollment into the ongoing ACTIV-3b NIH-sponsored trial.
“We appreciate the detailed feedback and learnings from our previous EUA submission, and applied them to this narrower request to the FDA,” said Prof Jonathan Javitt, MD, MPH, Chairman and CEO NRx Pharmaceuticals. “As the Omicron surge continues to push our hospitals far past capacity, and with more than 1200 Americans dying every day, we continue to believe ZYESAMI can provide an option to the sickest of COVID patients who have no other options and significantly increase their chances at recovering.”
The filing comes with a new analysis of the Phase 2b/3 data of ZYESAMI in patients treated with Remdesivir or other approved or authorized medicines for critical COVID-19. The new analysis was conducted by Prof. David Schoenfeld, PhD, an expert statistician with unique expertise in life-threatening diseases of the lung. The analysis, requested by the FDA, demonstrated that the odds of meeting the original primary endpoint of the trial, being “alive and free of respiratory failure at 28 days” were 2.8-fold higher on ZYESAMI than on placebo (P=.03). There was a 4 fold increased odds of surviving to 60 days (P=.006). Patients at highest risk – those on ventilators at time of randomization – demonstrated a 10-fold increased odds of survival (P=.03) after doctors administered ZYESAMI. The most common side effects of ZYESAMI noted were mild to moderate diarrhea and systemic hypotension (low blood pressure).
ZYESAMI (aviptadil) is a synthetic form of Vasoactive Intestinal Polypeptide (VIP) and binds specifically to the alveolar type II cell (ATII) in the air sac (alveolus) of the lung, where it has been shown to have potent anti-inflammatory and anti-cytokine activity in animal models of respiratory distress, acute lung injury, and inflammation. VIP stimulates ATII cells to make the surfactant that must coat the lining of the lungs for them to exchange oxygen with the blood. Loss of surfactant causes respiratory failure and alveolar collapse, hallmarks of COVID-19.
NRx looks forward to working with the FDA and providing further information regarding the EUA submission upon FDA request.
In 2021, the FDA declined to issue EUA for ZYESAMI to treat patients suffering Critical COVID-19 with respiratory failure. This new EUA submission is a narrower, proposed indication for patients with no other available therapy and is supported by data requested by the FDA. ZYESAMI was selected by NIH from among 600 candidate drugs for study in the ongoing Phase 3 ACTIV-3b trial. The revised indication is designed to ensure that enrollment in this trial is not compromised.
https://www.nrxpharma.com/nrx-pharmaceuticals-submits-emergency-use-authorization-application-to-us-food-and-drug-administration-for-zyesami/
Good luck and GOD bless,
I am a bit puzzled though as to WHY today's PR doesn't make any mention of how well Zyesami performed as compared to Remdesivir. Unless I'm missing something, those facts were left out. Isn't that what the FDA was asking for? It only references Zyesami as compared to a "placebo".
georgejj - we have a couple of trials that may soon report out an update.
Clinical Trials... What's Next !!!........
Inhaled Zyesami - 144 participants
Inhaled ZYESAMI™ (Aviptadil Acetate) for the Treatment of Severe COVID-19 (AVICOVID-2)
Estimated Primary Completion: Dec 15, 2021
Estimated Study Completion: Dec 31, 2021
https://clinicaltrials.gov/ct2/show/NCT04360096
Inhaled Aviptadil - 82 participants
Inhaled Aviptadil for the Treatment of COVID-19 in Patients at High Risk for ARDS
Estimated Primary Completion: Dec 2021
Estimated Study Completion: Apr 2022
https://clinicaltrials.gov/ct2/show/NCT04536350
The following Phase 2/3 trial should be completed within days or weeks (imminent)
Inhaled ZYESAMI™ (Aviptadil Acetate) for the Treatment of Severe COVID-19 (AVICOVID-2)
https://clinicaltrials.gov/ct2/show/NCT04360096
Good luck and GOD bless,
The entire world needs ZYESAMI® (aviptadil) FDA and EMA approval early in 2022.
Gooo luck and GOD bless,
|
Followers
|
52
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
1125
|
|
Created
|
12/15/20
|
Type
|
Free
|
| Moderators ProfitScout jedijazz | |||





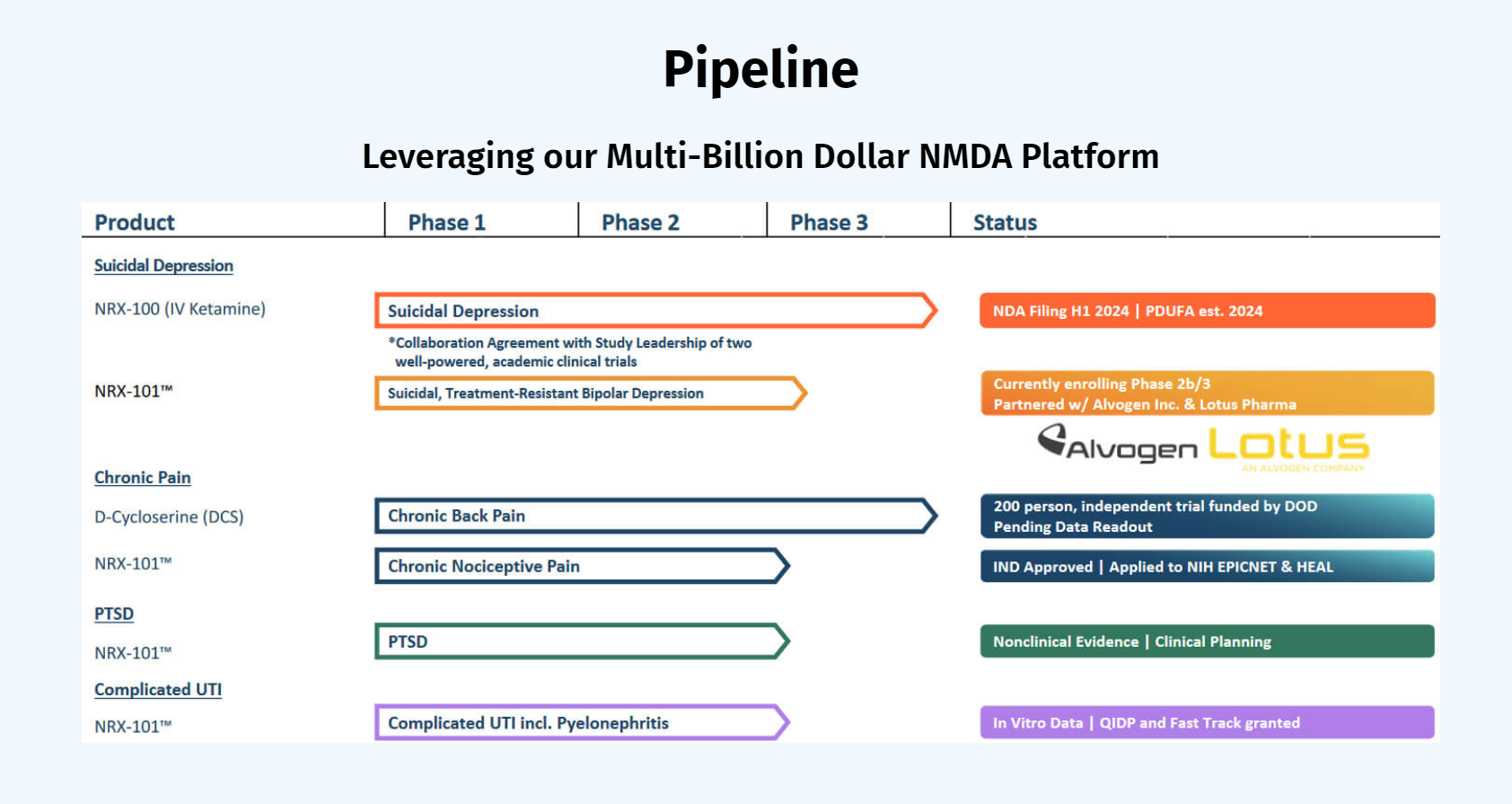
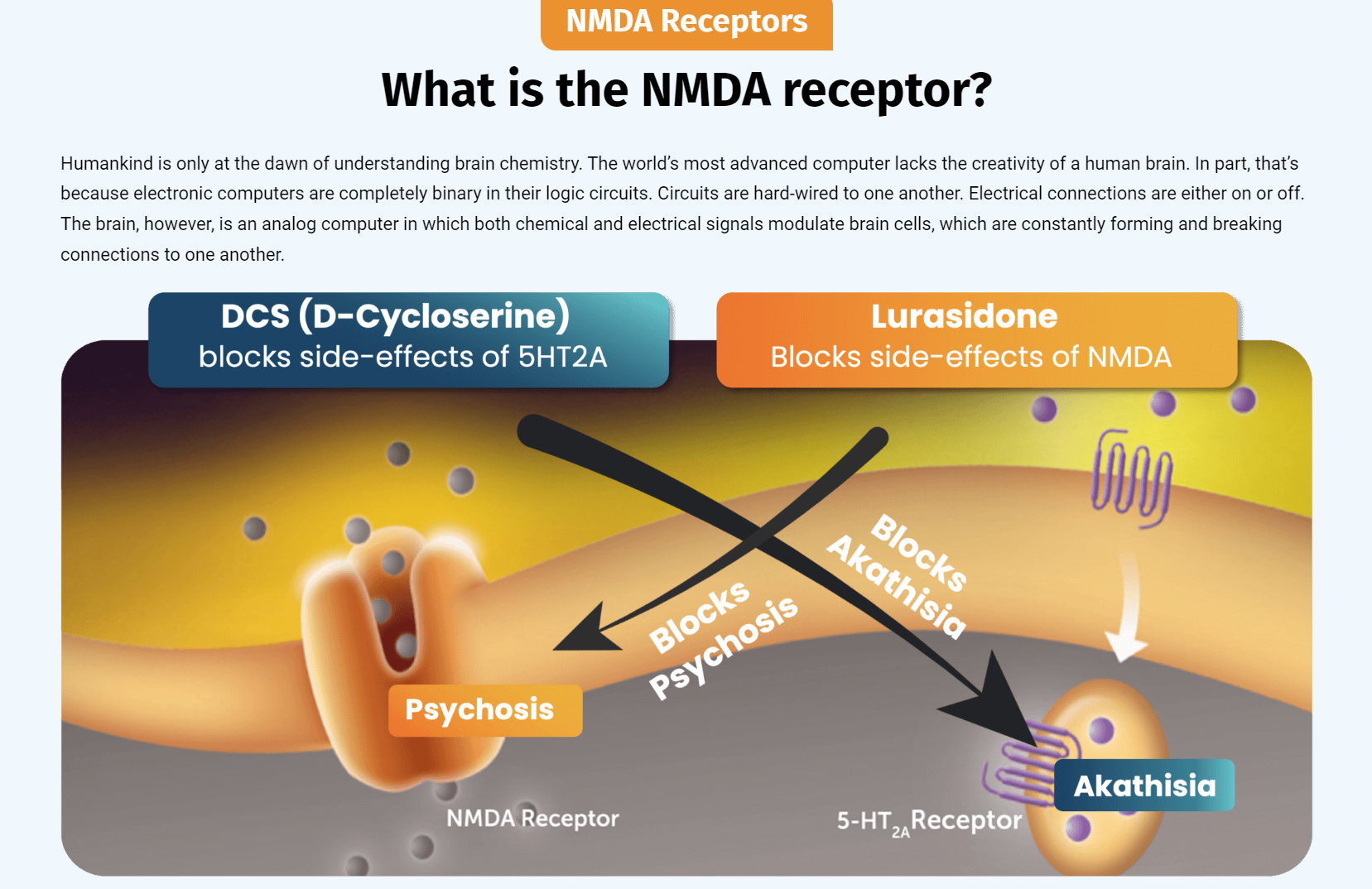







Both drugs demonstrated > 50% response for treating depression. NRX-101 demonstrated a mean 76% reduction in symptoms of akathisia compared to lurasidone that was sustained over 42 days (Effect...

New data demonstrates no impact of NRX-101 on gut or vaginal flora – considered primary causes of pseudomembranous colitis due to C difficile and vaginal yeast infections NRX-101 previously...
Formulation based on prior patents by NRx founder Achieved pH neutral formulation of ketamine, potentially enabling both intravenous (IV) and subcutaneous (SQ) administration Company expects...
Data transferred for independent statistical analysis Top-line data expected in April 2024 RADNOR, Pa., April 8, 2024 /PRNewswire/ -- NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) ("NRx...
Four potential near-term milestones, including data from two clinical trials, an NDA filing and an upcoming share dividend -- 50% reduction in corporate overhead and 25% reduction in overall net...
Four potential near-term milestones, including data from two clinical trials, an NDA filing and a share dividend RADNOR, Pa., March 28, 2024 /PRNewswire/ -- NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) ("
NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) ("NRx Pharmaceuticals", the "Company"), a clinical-stage biopharmaceutical company, today announced that it will release its fourth quarter and full year...
NRx Pharmaceuticals, Inc. (Nasdaq:NRXP) ("NRx Pharmaceuticals", the "Company"), a clinical-stage biopharmaceutical company, today announced that Dr. Jonathan Javitt, Chairman and Chief Scientist...
NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) ("NRx Pharmaceuticals", the "Company"), a clinical-stage biopharmaceutical company, today announced that it will release its fourth quarter and full year...
61.4% (56,781,354) of eligible shares voted 94.4% of votes were cast in favor of the resolution RADNOR, Pa., March 21, 2024 /PRNewswire/ -- NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) ("NRx...
NRx Pharmaceuticals Board of Directors has authorized its Chairman, CEO, and management to take all necessary steps to affect the Dividend and Royalty Rights to NRXP Shareholders and applicable...
Initiative is intended to protect shareholder value through continued compliance with Nasdaq listing rules and elimination of naked short sales positions in the Company's securities The Company is...
Company to receive first allocation of ketamine for sale by month end Partners preparing to ship IV Ketamine to full range of customers via 503a and 503b pharmacies NRx Pharmaceuticals and HOPE...
Marks a major step in the development of what could be the first drug approved for Suicidal Bipolar Depression The study database is being cleaned and locked; statistical analysis and top-line...
NRx Pharmaceuticals received approximately $1.0 million in cash from an existing investor Shares were sold at $0.38, a 26.7% premium the recent share offering, along with one common 5-year warrant...
In the news release, NRx Pharmaceuticals, Inc. Announces Pricing of $1.5 Million Underwritten Public Offering of Common Stock, issued 27-Feb-2024 by NRx Pharmaceuticals, Inc. over PR Newswire, we...
NRx Pharmaceuticals, Inc. (Nasdaq: NRXP), ("NRx Pharmaceuticals" or the "Company"), a clinical-stage biopharmaceutical company, today announced that it intends to offer to sell shares of its...
Presentation available on NRx Pharmaceuticals website at https://www.nrxpharma.com/hope-therapeutics/ KEY UPDATES ARE AS FOLLOWS: NRx management is proposing to award 50% of founding shares in...
DISCLAIMER:
Nothing in the contents transmitted on this board should be construed as an investment advisory, nor should it be used to make investment decisions.
There is no express or implied solicitation to buy or sell securities.
The author(s) may have positions in the stocks or financial relationships with the company or companies discussed and may trade in the stocks mentioned.
Readers are advised to conduct their own due diligence prior to considering buying or selling any stock. All information should be considered for information purposes only.
No stock exchange has approved or disapproved of the information here.
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
