Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
$NRXP NRx Pharmaceuticals (NASDAQ:NRXP) Initiates Strategy to Combat Short Sales
"Initiative is intended to protect shareholder value through continued compliance with Nasdaq listing rules and elimination of naked short sales positions in the Company's securities" https://finance.yahoo.com/news/nrx-pharmaceuticals-nasdaq-nrxp-initiates-114900937.html
$NRXP @elonmusk Elon Musk said that his prescribed use of ketamine alleviates periods of low mood and is in the best interest of investors in Tesla Inc.
[ @gravyman) is superior. https://bloomberg.com/news/articles/2024-03-18/musk-says-ketamine-prescription-is-in-investors-best-interests #TSLA #FDA #News
$NRXP .4899 HOD .54 NRx Pharma Announces Plan to Distribute Shares of HOPE Therapeutics and Royalty Rights on Ketamine Sales to Existing NRx Shareholders
Love the NRXP dividend news. This is the best way to reward investors who are supporting the company and also a pressure to drive short sellers out. When a company issues a dividend, shorts are liable to pay the value of that dividend rather than receiving it as they are not the true owners of the stock.
NRx Pharmaceuticals (NASDAQ:NRXP) Announces Plan to Distribute Shares of HOPE Therapeutics and Royalty Rights on Ketamine Sales to Existing NRx Shareholders
https://finance.yahoo.com/news/nrx-pharmaceuticals-nasdaq-nrxp-announces-123000925.html?soc_src=social-sh&soc_trk=tw&tsrc=twtr
Any thoughts on the vote for RS? I know about the theory behind it, but it seams like if they had confidence over the future of their three candidate drugs then the SP would easily and soon enough pop over the edge of a buck.
$CBDW @CBDWInc 1606 Corp. Building on success of its first marketing campaign
$NRXP .54 +4.85% nice move pre-market may open higher!
Elon Musk Says His Ketamine Prescription Matters to Investors
https://www.bloomberg.com/news/articles/2024-03-18/musk-says-ketamine-prescription-is-in-investors-best-interests
Elon Musk Says His Ketamine Prescription Matters to Investors Interesting article...
Elon Musk said that his prescribed use of ketamine alleviates periods of low mood and is in the best interest of investors in Tesla Inc. and the other companies he runs.
For Wall Street, “what matters is execution,” Musk said in an interview with former CNN anchor Don Lemon streamed Monday on YouTube. “From an investor standpoint, if there is something I’m taking, I should keep taking it,” he said referring to Tesla’s success.
Musk said he takes the drug as prescribed periodically to treat what he described as “chemical tides” that lead to depression-like symptoms. Doctors can prescribe ketamine to treat pain and depression and at higher doses, the drug acts as a sedative. It can also cause hallucinations, and overdosing can lead to unconsciousness and dangerously slowed breathing, according to the US Drug Enforcement Administration.
An article in the Wall Street Journal in January said that executives in the Tesla and SpaceX chief executive officer’s companies had grown concerned about his recreational drug use. Musk responded that he hadn’t failed drug tests and, in a post on his X social network, that “whatever I’m doing, I should obviously keep doing it!”
$NRXP NEWS! NRx Pharmaceuticals (NASDAQ:NRXP) Announces Plan to Distribute Shares of HOPE Therapeutics and Royalty Rights on Ketamine Sales to Existing NRx Shareholders @NRxPharma #news #dividends #announced https://finance.yahoo.com/news/nrx-pharmaceuticals-nasdaq-nrxp-announces-123000925.html?soc_src=social-sh&soc_trk=tw&tsrc=twtr via @YahooFinance
$NRXP More DD: #nasdaq "NRX-100 - A Beacon of Light in the Darkness of Acute Suicidality and Depression" https://axecapitalusa.com/nrxp/
$NRXP Short Interest https://www.nasdaq.com/market-activity/stocks/nrxp/short-interest #shortsqueeze
$NRXP 0.475 +2.59% HOD .4859 Looks to be moving back into the .50 cent range!
$NRXP - Company has retained former SEC enforcement leadership to notify leading brokerages of the need to close all naked short positions in the Company's securities and to prevent future accumulation of naked short positions in the Company's new security
https://finance.yahoo.com/news/nrx-pharmaceuticals-nasdaq-nrxp-initiates-114900937.html
$NRXP .4699 +6.94% Resilient After yesterdays Dip - NRX Pharmaceuticals Inc NASDAQ
https://finance.yahoo.com/news/nrx-pharmaceuticals-nasdaq-nrxp-initiates-114900937.html
Excellent News today... Let's see how this initiative goes!
$NRXP
$NRXP Agreed! With the Nasdaq they run on news. Shorts need to close positions but yeah longs may benefit if it does well. It's different when larger hedge funds can invest at higher prices as well. FDA news and it explodes. RS may be frowned upon in OTC but this is Nasdaq.
$NRXP News: NRx Pharmaceuticals (NASDAQ:NRXP) Initiates Strategy to Combat Short Sales
PR Newswire
Tue, Mar 12, 2024, 7:49 AM EDT
Initiative is intended to protect shareholder value through continued compliance with Nasdaq listing rules and elimination of naked short sales positions in the Company's securities
The Company is contemplating a reverse split, if needed to maintain NASDAQ listing compliance, to be accompanied by change in corporate name and CUSIP number
Company has retained former SEC enforcement leadership to notify leading brokerages of the need to close all naked short positions in the Company's securities and to prevent future accumulation of naked short positions in the Company's new security
RADNOR, Pa., March 12, 2024 /PRNewswire/ -- NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) ("NRx Pharmaceuticals", the "Company") today initiated actions to combat short sellers in the Company's stock.
The Company announced a proposal to simultaneously change the CUSIP under which the Company's shares are traded, together with changing the name of the Company to NRx Therapeutics, Inc. The Company plans to accompany these actions with a required exchange of the underlying stock certificates. This certificate change is expected to be seamless for those investors holding NRXP shares in electronic form and similarly for investors who may have established short positions in NRXP by borrowing the underlying shares from a registered shareholder. Any party holding a short position in the Company's shares who has not complied with the legal requirement to borrow the underlying shares of stock (i.e. "a naked short") may be unable to exchange that position for a position in the new security.
The Company is working with attorneys who formerly served in leadership positions at the SEC Division of Enforcement to correspond with corporate counsel and compliance heads at leading brokerages to emphasize the current legal prohibitions against naked short sales. As identified in previous announcements, the Company was advised by ShareIntel in September 2023 that substantial naked short positions in the Company's securities had been identified at major brokerage firms.
"We believe it is in the best interests of our shareholders to have fully-compliant trading in the markets," said Janet Rehnquist, Esq., who chairs the Company's Compliance Committee. "As we focus on the development of potentially life-saving drugs to combat suicidal depression, PTSD, and Chronic Pain, it is critical that we take all steps possible to maximize the value of those who have invested in our Company and provided the resources to address these critical unmet medical needs."
About NRx Pharmaceuticals
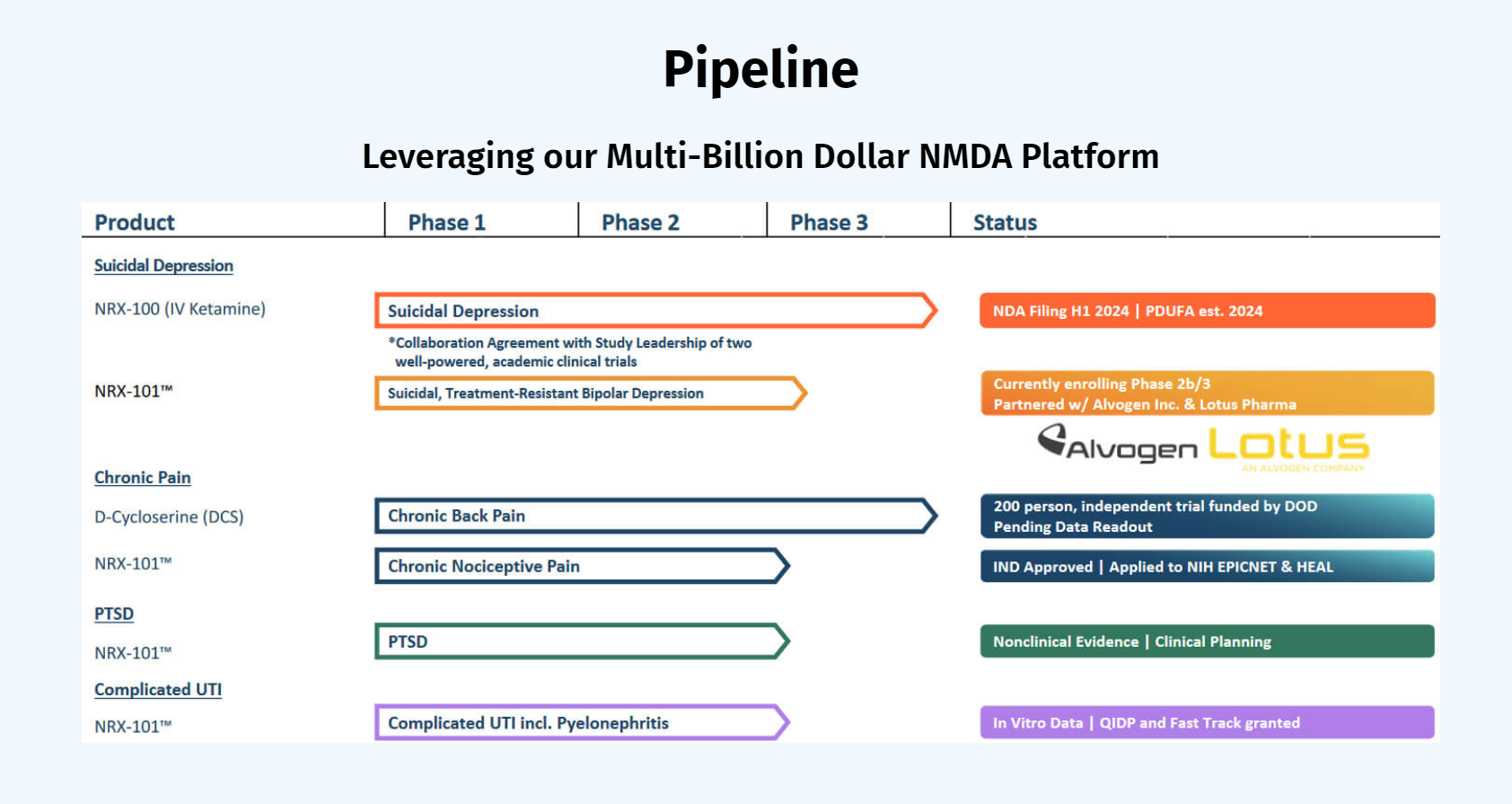
NRx Pharmaceuticals is a clinical-stage biopharmaceutical company developing therapeutics based on its NMDA platform for the treatment of central nervous system disorders, specifically suicidal bipolar depression, chronic pain and PTSD. The Company is developing NRX-101, an FDA-designated investigational Breakthrough Therapy for suicidal treatment-resistant bipolar depression and chronic pain. NRx has partnered with Alvogen and Lotus around the development and marketing of NRX-101 for the treatment of suicidal bipolar depression. NRX-101 additionally has potential to act as a non-opioid treatment for chronic pain, as well as a treatment for complicated UTI.
NRx has recently announced plans to submit a New Drug Application for HTX-100 (IV ketamine), through Hope Therapeutics, in the treatment of suicidal depression, based on results of well-controlled clinical trials conducted under the auspices of the US National Institutes of Health and newly obtained data from French health authorities, licensed under a data sharing agreement. NRx was awarded Fast Track Designation for development of ketamine (NRX-100) by the US FDA as part of a protocol to treat patients with acute suicidality.
Notice Regarding Forward-Looking Statements
The information contained herein includes forward-looking statements within the meaning of Section 21E of the Securities Exchange Act of 1934, as amended, and Section 27A of the Securities Act of 1933, as amended. These statements include, among others, statements regarding the proposed public offering and the timing and the use of the proceeds from the offering. Forward-looking statements generally include statements that are predictive in nature and depend upon or refer to future events or conditions, and include words such as "may," "will," "should," "would," "expect," "plan," "believe," "intend," "look forward," and other similar expressions among others. These statements relate to future events or to the Company's future financial performance, and involve known and unknown risks, uncertainties and other factors that may cause the Company's actual results to be materially different from any future results, levels of activity, performance or achievements expressed or implied by these forward-looking statements. You should not place undue reliance on forward-looking statements since they involve known and unknown risks, uncertainties and other factors which are, in some cases, beyond the Company's control and which could, and likely will, materially affect actual results, levels of activity, performance or achievements. Any forward-looking statement reflects the Company's current views with respect to future events and is subject to these and other risks, uncertainties and assumptions relating to the Company's operations, results of operations, growth strategy and liquidity. More detailed information about the Company and the risk factors that may affect the realization of forward-looking statements is set forth in the Company's most recent Annual Report on Form 10-K and other filings with the Securities and Exchange Commission. Investors and security holders are urged to read these documents free of charge on the SEC's website at http://www.sec.gov. Except as may be required by applicable law, The Company assumes no obligation to publicly update or revise these forward-looking statements for any reason, or to update the reasons actual results could differ materially from those anticipated in these forward-looking statements, whether as a result of new information, future events or otherwise.
CisionCision
Cision
View original content to download multimedia:https://www.prnewswire.com/news-releases/nrx-pharmaceuticals-nasdaqnrxp-initiates-strategy-to-combat-short-sales-302086668.html
SOURCE NRx Pharmaceuticals, Inc.
The SEC filing is on the website describing the special meeting and reverse split.
DEF 14A Official notification to shareholders of matters to be brought to a vote ("Proxy") filed on March 11, 2024
https://ir.nrxpharma.com/sec-filings
NASDAQ listing requires 10 consecutive days above $1.00 to remain listed so suspect something in the 1:5 to 1:10 range although 1:2 would work if it holds. Probably be safe and go to a higher ratio. TWT.
$NRXP .6825 +11.27% in power hour! Great day for NRX Pharma as NRx Pharmaceuticals (NASDAQ:NRXP) Completes Memorandum of Understanding and Collaborations with Distribution Partners for HTX-100 (IV Ketamine) https://ir.nrxpharma.com/2024-03-11-NRx-Pharmaceuticals-NASDAQ-NRXP-Completes-Memorandum-of-Understanding-and-Collaborations-with-Distribution-Partners-for-HTX-100-IV-Ketamine
$NRXP @NRxPharma NRx Pharmaceuticals Announces Last Patient, Last Visit in Its Phase 2B/3 Trial of NRX-101 in Suicidal Treatment Resistant Bipolar Depression #MarketScreener https://www.marketscreener.com/quote/stock/NRX-PHARMACEUTICALS-INC-38908842/news/NRX-Pharmaceuticals-Announces-Last-Patient-Last-Visit-in-Its-Phase-2B-3-Trial-of-NRX-101-in-Suicida-46092753/?utm_source=twitter&utm_medium=social&utm_campaign=share
$NRXP looking good on today's News, UP almost 11% on nearly 20% of its 10-day average volume within the first hour.
Company to receive first allocation of ketamine for sale by month end
https://finance.yahoo.com/news/nrx-pharmaceuticals-nasdaq-nrxp-completes-123000827.html
$NRXP .685 +11.67% Based on 2 Wall Street analysts offering 12 month price targets for NRX Pharmaceuticals in the last 3 months. The average price target is $3.75 with a high forecast of $5.50 and a low forecast of $2.00. The average price target represents a 459.70% change! huge #NEWS #OUT
$NRXP News: NRx Pharmaceuticals (NASDAQ:NRXP) Completes Memorandum of Understanding and Collaborations with Distribution Partners for HTX-100 (IV Ketamine)
PR Newswire
Mon, Mar 11, 2024, 8:30 AM EDT
- Company to receive first allocation of ketamine for sale by month end
- Partners preparing to ship IV Ketamine to full range of customers via 503a and 503b pharmacies
- NRx Pharmaceuticals and HOPE Therapeutics aim to provide highest quality product available to those in need and assure ongoing supply
- Important step toward goal of providing full suite of supply options, to ultimately include an FDA approved, and widely reimbursed, solution for patients and clinics, subject to board approval
RADNOR, Pa., March 11, 2024 /PRNewswire/ -- NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) ("NRx Pharmaceuticals", the "Company") and its wholly owned subsidiary HOPE Therapeutics, clinical-stage biopharmaceutical companies, today announced completion of a Memorandum of Understanding with Conversio Health, a national 503a pharmacy, and a strong, ongoing partnership with Nephron Pharmaceuticals, a manufacturer of Ketamine and 503b pharmacy to provide IV Ketamine to patients and clinics across the country, subject to board approval. These are important steps to providing a full suite of ketamine options for customers across the country, and preparation for potential approval of HTX-100 by regulatory authorities worldwide.
The Company has further entered into a business relationship with an organization that currently serves the business needs of more than 100 ketamine clinics nationwide. The Company has been given a target delivery date prior to the end of this month (March 2024) for initial stock of ketamine for sale to licensed end users. Sales are planned to commence once technical and logistical elements are finalized. Details for ordering will be available on our website as soon as they are available. www.hopetherapeutics.com
In contrast to NRx Pharmaceuticals, which is structured as a Biotechnology research and development company, HOPE Therapeutics is structured as a Specialty Pharmaceutical company that aims to supply a variety of therapeutic products to clinics that are treating suicidal depression and PTSD, including both traditional and digital therapeutics.
"We are pleased to be working with two outstanding organizations in Conversio Health and Nephron Pharmaceuticals to provide this important medication to a market in need of a consistent supply of high-quality, FDA- and DEA- compliant IV ketamine," said Matthew Duffy Chief Business Officer of NRx Pharmaceuticals and co-CEO of HOPE Therapeutics. "Importantly, these efforts will provide a strong foundation for expanding our product offering with our planed filing a New Drug Application with the FDA in the coming months, and the potential launch of HTX-100 in early 2025." The NDA filing currently awaits completion of an additional three months of product stability.
About NRx Pharmaceuticals
NRx Pharmaceuticals is a clinical-stage biopharmaceutical company developing therapeutics based on its NMDA platform for the treatment of central nervous system disorders, specifically suicidal bipolar depression, chronic pain and PTSD. The Company is developing NRX-101, an FDA-designated investigational Breakthrough Therapy for suicidal treatment-resistant bipolar depression and chronic pain. NRx has partnered with Alvogen and Lotus around the development and marketing of NRX-101 for the treatment of suicidal bipolar depression. NRX-101 additionally has potential to act as a non-opioid treatment for chronic pain, as well as a treatment for complicated UTI.
NRx has recently announced plans to submit a New Drug Application for HTX-100 (IV ketamine), through HOPE Therapeutics, in the treatment of suicidal depression, based on results of well-controlled clinical trials conducted under the auspices of the US National Institutes of Health and newly obtained data from French health authorities, licensed under a data sharing agreement. NRx was awarded Fast Track Designation for development of ketamine (NRX-100) by the US FDA as part of a protocol to treat patients with acute suicidality.
About HOPE Therapeutics, Inc.
HOPE Therapeutics, Inc. (www.hopetherapeutics.com) is a Specialty Pharmaceutical Company, wholly-owned by NRX Pharmaceuticals focused on development and marketing of an FDA-approved form of intravenous ketamine for the treatment of acute suicidality and depression together with a digital therapeutic-enabled platform designed to augment and preserve the clinical benefit of NMDA-targeted drug therapy.
Notice Regarding Forward-Looking Statements
The information contained herein includes forward-looking statements within the meaning of Section 21E of the Securities Exchange Act of 1934, as amended, and Section 27A of the Securities Act of 1933, as amended. These statements include, among others, statements regarding the proposed public offering and the timing and the use of the proceeds from the offering. Forward-looking statements generally include statements that are predictive in nature and depend upon or refer to future events or conditions, and include words such as "may," "will," "should," "would," "expect," "plan," "believe," "intend," "look forward," and other similar expressions among others. These statements relate to future events or to the Company's future financial performance, and involve known and unknown risks, uncertainties and other factors that may cause the Company's actual results to be materially different from any future results, levels of activity, performance or achievements expressed or implied by these forward-looking statements. You should not place undue reliance on forward-looking statements since they involve known and unknown risks, uncertainties and other factors which are, in some cases, beyond the Company's control and which could, and likely will, materially affect actual results, levels of activity, performance or achievements. Any forward-looking statement reflects the Company's current views with respect to future events and is subject to these and other risks, uncertainties and assumptions relating to the Company's operations, results of operations, growth strategy and liquidity. More detailed information about the Company and the risk factors that may affect the realization of forward-looking statements is set forth in the Company's most recent Annual Report on Form 10-K and other filings with the Securities and Exchange Commission. Investors and security holders are urged to read these documents free of charge on the SEC's website at http://www.sec.gov. Except as may be required by applicable law, The Company assumes no obligation to publicly update or revise these forward-looking statements for any reason, or to update the reasons actual results could differ materially from those anticipated in these forward-looking statements, whether as a result of new information, future events or otherwise.
HOPE Therapeutics, Inc. (PRNewsfoto/NRx Pharmaceuticals, Inc.)
HOPE Therapeutics, Inc. (PRNewsfoto/NRx Pharmaceuticals, Inc.)
CisionCision
Cision
View original content to download multimedia:https://www.prnewswire.com/news-releases/nrx-pharmaceuticals-nasdaqnrxp-completes-memorandum-of-understanding-and-collaborations-with-distribution-partners-for-htx-100-iv-ketamine-302085271.html
SOURCE NRx Pharmaceuticals, Inc.
https://finance.yahoo.com/news/nrx-pharmaceuticals-nasdaq-nrxp-completes-123000827.html
NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) ("NRx Pharmaceuticals", the "Company") and its wholly owned subsidiary HOPE Therapeutics, clinical-stage biopharmaceut...
$NRXP With a Novel Breakthrough Drug, This Little-Known Biotech Company is on the verge of significant FDA results.
Of the thousands of drugs approved by FDA, only 544 have been designated as “Breakthrough Therapy.” NRX-101 is the first Breakthrough Therapy for Suicidal Depression https://axecapitalusa.com/nrxp/
$NRXP .62 HOD .65 Huge Volume! @NRxPharma
NRx Pharmaceuticals Hits Milestone in Suicidal Bipolar Depression Treatment; Potential $329 Million Deal with Partners: NRXP https://www.einpresswire.com/article/693114260/trial-endpoint-for-1st-suicidal-bipolar-ptsd-pain-drug-partners-can-provide-329-million-royalties-nasdaq-nrxp via @ein_news
$NRXP .6885 +3.85% Trial Endpoint Reached for First Suicidal Bipolar Depression, Chronic Pain and PTSD Drug; Agreement with Partners Can Provide up to $329 Million and Royalties: NRx Pharmaceuticals (Nasdaq: NRXP) https://www.benzinga.com/pressreleases/24/03/ab37472614/trial-endpoint-reached-for-first-suicidal-bipolar-depression-chronic-pain-and-ptsd-drug-agreement via @Benzinga
$NRXP .6599 +26.90% Great interview out! The Big Biz Show interviews Jonathan C. Javitt, M.D., M.P.H./Chief Scientist, Director, and Founder, NRx Pharmaceuticals.
Watch Interview - https://nrxpharma.com/the-big-biz-show-interviews-jonathan-c-javitt-m-d-m-p-h-chief-scientist-director-and-founder-nrx-pharmaceuticals-3/
#mentalhealth #depression #suicide #bipolar #PTSD #NRX100 #ketamine #NRXPharma #PharmaNews $NRXP
$NRXP up pre-market .5414 +4.12% as NRx Pharma marks a major step in the development of what could be the first drug approved for Suicidal Bipolar Depression.
2x Phase 3 readouts expected in early Q2 while valuation is still super low at $44 million
NRXP MOVING UP NOW
$NRXP Positive data and FDA comment would trigger the next $4 million milestone payment from partners Alvogen and Lotus and their assumption of development costs; agreement provides for up to $329 million in milestone payments and a royalty on Net Sales in the mid-teens
$NRXP PR Newswire: NRx Pharmaceuticals (NASDAQ: NRXP) Announces Last Patient, Last Visit in its Phase 2b/3 Trial of NRX-101 in Suicidal Treatment Resistant Bipolar Depression
Mon, Mar 4, 2024, 8:30 AM EST
In This Article:
NRXP
+1.28%
NRXPW
+2.50%
Marks a major step in the development of what could be the first drug approved for Suicidal Bipolar Depression
The study database is being cleaned and locked; statistical analysis and top-line data to follow shortly thereafter
Study maintained 95% concordance rate between study sites and central raters on primary endpoint. No unexpected Serious Adverse Events were reported.
Positive data and FDA comment would trigger the next $4 million milestone payment from partners Alvogen and Lotus and their assumption of development costs; agreement provides for up to $329 million in milestone payments and a royalty on Net Sales in the mid-teens
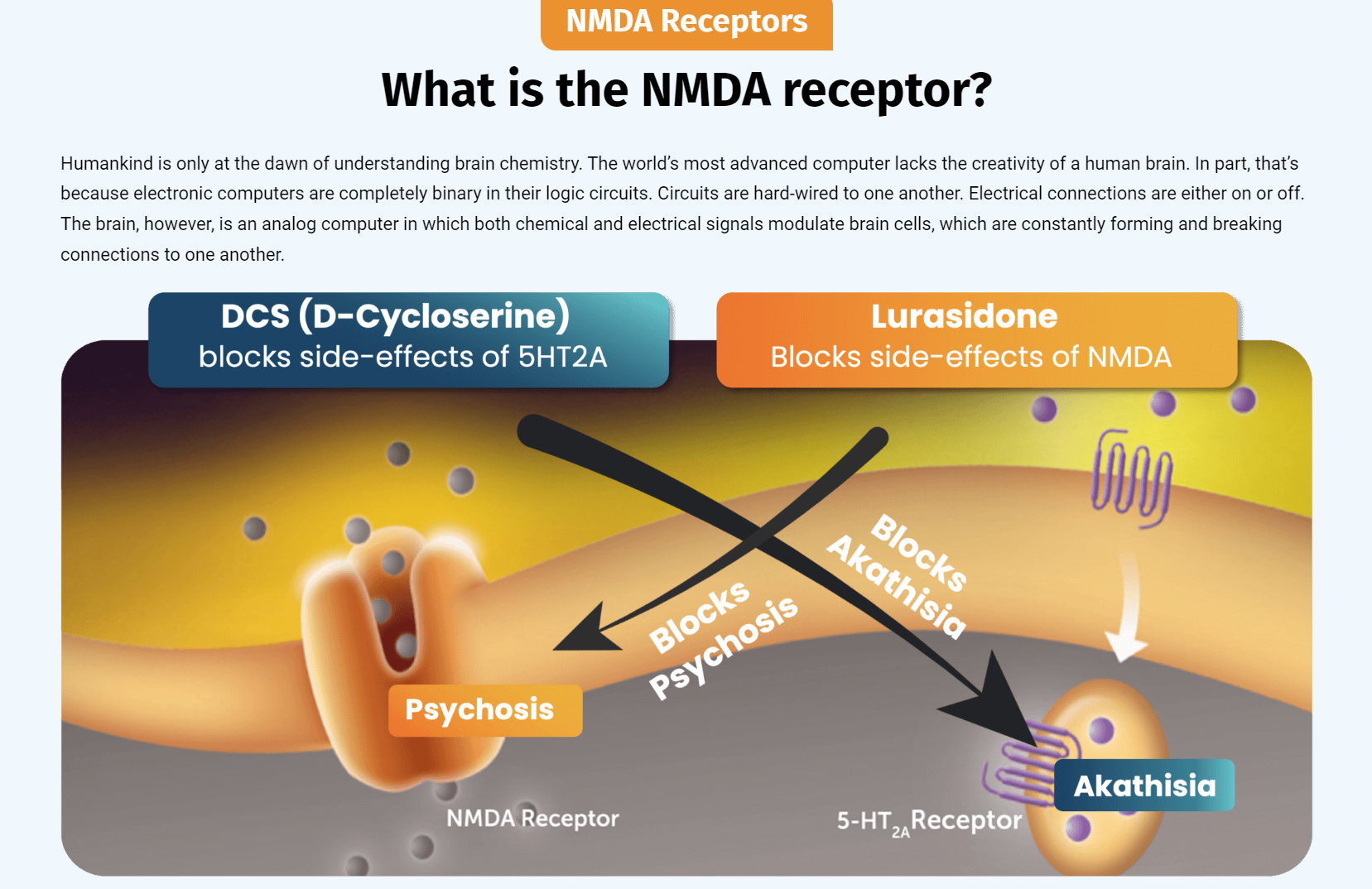
RADNOR, Pa., March 4, 2024 /PRNewswire/ -- NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) ("NRx Pharmaceuticals", the "Company"), a clinical-stage biopharmaceutical company, today announced that the 74th and last evaluable patient has completed their day 42 visit in its Phase 2b/3 study of NRX-101, the Company's patented combination of the NMDA antagonist D-cycloserine and lurasidone, in Suicidal Treatment Resistant Bipolar Depression. The database is being cleaned, finalized, and locked; statistical analysis will then be performed, with top-line data to follow shortly thereafter. As previously disclosed, positive data from this trial triggers a milestone payment from Alvogen. Alvogen will then be responsible for further development and commercialization costs for this program.
NRX-101 has been awarded Breakthrough Therapy Designation, Fast Track Designation, a Biomarker Letter of Support, and a Special Protocol Agreement by the FDA for treatment of suicidal bipolar depression. It is the only oral medication to have demonstrated reduced suicidal ideation in patients with bipolar depression, a lethal disease that claims the lives of one in five who live with it.
"This is the first clinical trial, to the company's knowledge, conducted among patients with suicidal bipolar depression in the outpatient setting. Our previous trial measured the ability of NRX-101 to maintain the anti-depressant and anti-suicidal effect of ketamine administered in the hospital setting. These patients, whose clinical need is urgent and extraordinary have routinely been excluded from the clinical trials of all previously-known anti-depressant drugs. said Dr. Jonathan Javitt, Founder, Chairman and Chief Scientist of NRx Pharmaceuticals. Although there were patients whose depression worsened and required hospitalization (we don't yet know whether they were on NRX-101 or comparator), patient safety was maintained, and no trial participant suffered a serious unexpected adverse outcome. Our thanks go out to our investigators, clinics, partners and, most importantly, our amazing patients and their families for seeing this study through to this important milestone," "
The Phase 2b/3 trial (www.clinicaltrials.gov NCT 03395392) is a randomized, prospective, multicenter, double-blind study comparing NRX-101 to lurasidone over six weeks. The Principal Investigator is Prof. Andrew Nierenberg of Harvard Massachusetts General Hospital. The primary efficacy endpoint is reduction in depression as measured on the MADRS scale and the secondary endpoint is reduction of suicidal ideation as measured by the Clinical Global Impression Suicidality Scale (CGI-SS). As previously disclosed, treatment compliance and concordance of local raters to central raters scores was in excess of 94%, well above the industry standard that is normally seen in CNS trials.
Top-line results are expected around the end of this quarter.
About NRx Pharmaceuticals
NRx Pharmaceuticals is a clinical-stage biopharmaceutical company developing therapeutics based on its NMDA platform for the treatment of central nervous system disorders, specifically suicidal bipolar depression, chronic pain and PTSD. The Company is developing NRX-101, an FDA-designated investigational Breakthrough Therapy for suicidal treatment-resistant bipolar depression and chronic pain. NRx has partnered with Alvogen and Lotus around the development and marketing of NRX-101 for the treatment of suicidal bipolar depression. NRX-101 additionally has potential to act as a non-opioid treatment for chronic pain, as well as a treatment for complicated UTI.
NRx has recently announced plans to submit a New Drug Application for HTX-100 (IV ketamine), through Hope Therapeutics, in the treatment of suicidal depression, based on results of well-controlled clinical trials conducted under the auspices of the US National Institutes of Health and newly obtained data from French health authorities, licensed under a data sharing agreement. NRx was awarded Fast Track Designation for development of ketamine (NRX-100) by the US FDA as part of a protocol to treat patients with acute suicidality.
About HOPE Therapeutics, Inc.
HOPE Therapeutics, Inc. (www.hopetherapeutics.com) is a Specialty Pharmaceutical Company, wholly-owned by NRX Pharmaceuticals focused on development and marketing of an FDA-approved form of intravenous ketamine for the treatment of acute suicidality and depression together with a digital therapeutic-enabled platform designed to augment and preserve the clinical benefit of NMDA-targeted drug therapy.
Notice Regarding Forward-Looking Statements
The information contained herein includes forward-looking statements within the meaning of Section 21E of the Securities Exchange Act of 1934, as amended, and Section 27A of the Securities Act of 1933, as amended. These statements include, among others, statements regarding the proposed public offering and the timing and the use of the proceeds from the offering. Forward-looking statements generally include statements that are predictive in nature and depend upon or refer to future events or conditions, and include words such as "may," "will," "should," "would," "expect," "plan," "believe," "intend," "look forward," and other similar expressions among others. These statements relate to future events or to the Company's future financial performance, and involve known and unknown risks, uncertainties and other factors that may cause the Company's actual results to be materially different from any future results, levels of activity, performance or achievements expressed or implied by these forward-looking statements. You should not place undue reliance on forward-looking statements since they involve known and unknown risks, uncertainties and other factors which are, in some cases, beyond the Company's control and which could, and likely will, materially affect actual results, levels of activity, performance or achievements. Any forward-looking statement reflects the Company's current views with respect to future events and is subject to these and other risks, uncertainties and assumptions relating to the Company's operations, results of operations, growth strategy and liquidity. More detailed information about the Company and the risk factors that may affect the realization of forward-looking statements is set forth in the Company's most recent Annual Report on Form 10-K and other filings with the Securities and Exchange Commission. Investors and security holders are urged to read these documents free of charge on the SEC's website at http://www.sec.gov. Except as may be required by applicable law, The Company assumes no obligation to publicly update or revise these forward-looking statements for any reason, or to update the reasons actual results could differ materially from those anticipated in these forward-looking statements, whether as a result of new information, future events or otherwise.
View original content to download multimedia:https://www.prnewswire.com/news-releases/nrx-pharmaceuticals-nasdaq-nrxp-announces-last-patient-last-visit-in-its-phase-2b3-trial-of-nrx-101-in-suicidal-treatment-resistant-bipolar-depression-302078041.html
SOURCE NRx Pharmaceuticals, Inc. https://finance.yahoo.com/news/nrx-pharmaceuticals-nasdaq-nrxp-announces-133000763.html?soc_src=social-sh&soc_trk=tw&tsrc=twtr via @YahooFinance
$NRXP big news just out: NRx Pharmaceuticals (Nasdaq:NRXP) Launches HOPE Therapeutics, Inc. at the BIO CEO & Investor Conference 2024 https://finance.yahoo.com/news/nrx-pharmaceuticals-nasdaq-nrxp-launches-133000517.html?soc_src=social-sh&soc_trk=tw&tsrc=twtr via @YahooFinance
$NRXP NEWS: NRx Pharmaceuticals (Nasdaq:NRXP) Launches HOPE Therapeutics, Inc. at the BIO CEO & Investor Conference 2024
PR Newswire
Mon, Feb 26, 2024, 8:30 AM EST
In This Article: NRXP
Presentation available on NRx Pharmaceuticals website at https://www.nrxpharma.com/hope-therapeutics/
KEY UPDATES ARE AS FOLLOWS:
NRx management is proposing to award 50% of founding shares in HOPE Therapeutics to current shareholders together with a royalty coupon with an expected ex-dividend date in the near future, all subject to board approval
Dividend to be available to all shareholders who sign a covenant not to engage in short sales of NRx stock, subject to board approval
HOPE anticipates having manufactured ketamine supplies for shipment under 503a pharmacy regulations by July 1, 2024
RADNOR, Pa., Feb. 26, 2024 /PRNewswire/ -- NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) ("NRx Pharmaceuticals," the "Company"), a clinical-stage biopharmaceutical company, today announced that Dr. Jonathan Javitt, Chairman and Chief Scientist of NRx Pharmaceuticals, will present a corporate overview at the BIO CEO & Investor Conference, which is scheduled to take place February 26-27, 2024, at the Marriott Marquis in New York City. Presenting with him will be Matthew Duffy, the newly-appointed co-CEO of HOPE Therapeutics. The presentation will be available on the NRx Pharmaceuticals website at https://www.nrxpharma.com/hope-therapeutics/.
"Subsequent to our announcement last week, we are in the process of completing a Memorandum of Understanding that may enable us to begin sales of IV ketamine to qualified clinics in partnership with a holder of a nationwide pharmacy license. Additionally, we will be updating investors with regard to our plans to add a digital therapeutic component to our ketamine offering that has potential to extend the clinical benefit to patients." said Dr. Javitt. "We thank our investors for their support and look forward to updating the public on our progress towards building a lifesaving therapy."
Presentation Date: Monday, February 26th, 2024
Time: 2:30 PM ET
Location: The Royale Room, Marriot Marquis, New York City
Registration: https://bcic.bio.org/registration
About NRx Pharmaceuticals
NRx Pharmaceuticals is a clinical-stage biopharmaceutical company developing therapeutics based on its NMDA platform for the treatment of central nervous system disorders, specifically suicidal bipolar depression, chronic pain and PTSD. The Company is developing NRX-101, an FDA-designated investigational Breakthrough Therapy for suicidal treatment-resistant bipolar depression and chronic pain. NRx has partnered with Alvogen and Lotus around the development and marketing of NRX-101 for the treatment of suicidal bipolar depression. NRX-101 additionally has potential to act as a non-opioid treatment for chronic pain, as well as a treatment for complicated UTI.
NRx has recently announced plans to submit a New Drug Application for HTX-100 (IV ketamine), through Hope Therapeutics, in the treatment of suicidal depression, based on results of well-controlled clinical trials conducted under the auspices of the US National Institutes of Health and newly obtained data from French health authorities, licensed under a data sharing agreement. NRx was awarded Fast Track Designation for development of ketamine (NRX-100) by the US FDA as part of a protocol to treat patients with acute suicidality.
About HOPE Therapeutics, Inc.
HOPE Therapeutics, Inc. (www.hopetherapeutics.com) is a wholly-owned subsidiary of NRX Pharmaceuticals focused on development and marketing of an FDA-approved form of intravenous ketamine for the treatment of acute suicidality and depression together with a digital therapeutic-enabled platform designed to augment and preserve the clinical benefit of NMDA-targeted drug therapy.
View original content to download multimedia:https://www.prnewswire.com/news-releases/nrx-pharmaceuticals-nasdaqnrxp-launches-hope-therapeutics-inc-at-the-bio-ceo--investor-conference-2024-302070687.html
SOURCE NRx Pharmaceuticals, Inc.
Do I understand correctly that NRXP has until mid-April 2024 to be above $1.00 for 10 days or be de-listed from NASDAQ? It shows up red flagged on my Fidelity ATP account is why I ask. Will this require a reverse split of 4:1? I appreciate thoughts. TIA.
$NRXP SHARE DIVIDEND TO NRXP Shareholders! https://www.prnewswire.com/news-releases/nrx-pharmaceuticals-nasdaqnrxp-to-launch-hope-therapeutics-inc-at-the-bio-ceo--investor-conference-2024-302065827.html
$NRXP HOPE plans to initiate a 506(c) pre-IPO offering of pre-IPO shares for qualified investors that is structured to potentially yield both capital appreciation and a royalty on sales of ketamine https://finance.yahoo.com/news/nrx-pharmaceuticals-nasdaq-nrxp-launch-133000426.html?soc_src=social-sh&soc_trk=tw&tsrc=twtr via @YahooFinance
$NRXP NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) The Company Announces Advance of $5 Million Milestone Payment from Partners Alvogen, Inc. and Lotus Pharmaceutical Co. Ltd. (1975.TW) https://finance.yahoo.com/news/nrx-pharmaceuticals-inc-nasdaq-nrxp-133000063.html?soc_src=social-sh&soc_trk=tw&tsrc=twtr via @YahooFinance
Breaking NEWS! NRXP with planned share dividend to existing NRXP shareholders.
$NRXP Premarket News Out! NRx Pharmaceuticals (Nasdaq:NRXP) to Launch HOPE Therapeutics, Inc. at the BIO CEO & Investor Conference 2024 https://finance.yahoo.com/news/nrx-pharmaceuticals-nasdaq-nrxp-launch-133000426.html?soc_src=social-sh&soc_trk=tw&tsrc=twtr via @YahooFinance
$NRXP News: NRx Pharmaceuticals (Nasdaq:NRXP) to Launch HOPE Therapeutics, Inc. at the BIO CEO & Investor Conference 2024
PR Newswire
Tue, Feb 20, 2024, 8:30 AM EST
In This Article: NRXP
NRXPW +52.04%
HOPE shares currently owned by NRXP with planned share dividend to existing NRXP shareholders
HOPE has now completed initial manufacture of Ketamine for IV infusion and plans to file FDA New Drug Application for treatment of acute suicidality upon demonstration of 2-year shelf stability (expected Q2 2024)
HOPE plans to file patient-level data from two well-controlled clinical trials comparing ketamine to placebo in patients with acute suicidality and depression, which demonstrated a statistically significant benefit (P<.001)
HOPE plans to initiate a 506(c) pre-IPO offering of pre-IPO shares for qualified investors that is structured to potentially yield both capital appreciation and a royalty on sales of ketamine
RADNOR, Pa., Feb. 20, 2024 /PRNewswire/ -- NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) ("NRx Pharmaceuticals", the "Company"), a clinical-stage biopharmaceutical company, today announced that Dr. Jonathan Javitt, Chairman and Chief Scientist of NRx Pharmaceuticals, will present a corporate overview at the BIO CEO & Investor Conference, which is scheduled to take place February 26-27, 2024, at the Marriott Marquis in New York City. Presenting with him will be Matthew Duffy, the newly-appointed co-CEO of HOPE Therapeutics.
Concurrent with the presentation, NRx plans to file a proxy statement, subject to board approval, for Nasdaq:NRXP shareholders outlining the share structure and seeking a shareholder advisory vote to support the planned share dividend for HOPE Therapeutics. The Company has received consistent advice from public shareholders that the planned share dividend and royalty coupon be distributed only to shareholders and warrant-holders who execute a covenant not to participate in short sales of the Company's stock.
"Subsequent to our initial announcement of HOPE Therapeutics in December 2023, we have focused on establishing HOPE's basic corporate and board structure, securing our manufacturing partnerships and manufacturing initial productions batches of ketamine, establishing nationwide distribution, pharmacovigilance, and medical liaison partnerships, and initiating plans to pair ketamine therapy with a novel digital therapeutic that has potential to reinforce the effect of NMDA-targeted drug therapy in reducing suicidal ideation and depression," said Dr. Javitt. "We thank our investors for their support and look forward to updating the public on our progress towards building a lifesaving therapy."
Presentation Date: Monday, February 26th, 2024
Time: 2:30 PM ET
Location: The Royale Room, Marriott Marquis, New York City
Registration: https://bcic.bio.org/registration
SIMULCAST: BIZTV
Information will be available on the NRx website at https://ir.nrxpharma.com/events prior to the conference.
About NRx Pharmaceuticals
NRx Pharmaceuticals is a clinical-stage biopharmaceutical company developing therapeutics based on its NMDA platform for the treatment of central nervous system disorders, specifically suicidal bipolar depression, chronic pain, and PTSD. The Company is developing NRX-101, an FDA-designated investigational Breakthrough Therapy for suicidal treatment-resistant bipolar depression and chronic pain. NRx has partnered with Alvogen and Lotus around the development and marketing of NRX-101 for the treatment of suicidal bipolar depression. NRX-101 additionally has potential to act as a non-opioid treatment for chronic pain, as well as a treatment for complicated UTI.
NRx has recently announced plans to submit a New Drug Application for HTX-100 (IV ketamine), through Hope Therapeutics, in the treatment of suicidal depression, based on results of well-controlled clinical trials conducted under the auspices of the US National Institutes of Health and newly obtained data from French health authorities, licensed under a data sharing agreement. NRx was awarded Fast Track Designation for development of ketamine (NRX-100) by the US FDA as part of a protocol to treat patients with acute suicidality.
About HOPE Therapeutics, Inc.
HOPE Therapeutics, Inc. (www.hopetherapeutics.com) is a wholly-owned subsidiary of NRX Pharmaceuticals focused on development and marketing of an FDA-approved form of intravenous ketamine for the treatment of acute suicidality and depression together with a digital therapeutic-enabled platform designed to augment and preserve the clinical benefit of NMDA-targeted drug therapy.
View original content to download multimedia:https://www.prnewswire.com/news-releases/nrx-pharmaceuticals-nasdaqnrxp-to-launch-hope-therapeutics-inc-at-the-bio-ceo--investor-conference-2024-302065827.html
SOURCE NRx Pharmaceuticals
$NRXP - Shareholder meeting planned for February 7, 2024, is cancelled, and will be rescheduled, if necessary; Company expects to convene a new meeting to vote on the HOPE transaction and share dividend, subject to Board approval
https://finance.yahoo.com/news/nrx-pharmaceuticals-inc-nasdaq-nrxp-114800404.html
$NRXP Barchart Technical Opinion rating is a 88% Buy with a Average short term outlook on maintaining the current direction.
https://www.barchart.com/stocks/quotes/NRXP/overview
|
Followers
|
53
|
Posters
|
|
|
Posts (Today)
|
2
|
Posts (Total)
|
1100
|
|
Created
|
12/15/20
|
Type
|
Free
|
| Moderators ProfitScout jedijazz | |||












Both drugs demonstrated > 50% response for treating depression. NRX-101 demonstrated a mean 76% reduction in symptoms of akathisia compared to lurasidone that was sustained over 42 days (Effect...

New data demonstrates no impact of NRX-101 on gut or vaginal flora – considered primary causes of pseudomembranous colitis due to C difficile and vaginal yeast infections NRX-101 previously...
Formulation based on prior patents by NRx founder Achieved pH neutral formulation of ketamine, potentially enabling both intravenous (IV) and subcutaneous (SQ) administration Company expects...
Data transferred for independent statistical analysis Top-line data expected in April 2024 RADNOR, Pa., April 8, 2024 /PRNewswire/ -- NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) ("NRx...
Four potential near-term milestones, including data from two clinical trials, an NDA filing and an upcoming share dividend -- 50% reduction in corporate overhead and 25% reduction in overall net...
Four potential near-term milestones, including data from two clinical trials, an NDA filing and a share dividend RADNOR, Pa., March 28, 2024 /PRNewswire/ -- NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) ("
NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) ("NRx Pharmaceuticals", the "Company"), a clinical-stage biopharmaceutical company, today announced that it will release its fourth quarter and full year...
NRx Pharmaceuticals, Inc. (Nasdaq:NRXP) ("NRx Pharmaceuticals", the "Company"), a clinical-stage biopharmaceutical company, today announced that Dr. Jonathan Javitt, Chairman and Chief Scientist...
NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) ("NRx Pharmaceuticals", the "Company"), a clinical-stage biopharmaceutical company, today announced that it will release its fourth quarter and full year...
61.4% (56,781,354) of eligible shares voted 94.4% of votes were cast in favor of the resolution RADNOR, Pa., March 21, 2024 /PRNewswire/ -- NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) ("NRx...
NRx Pharmaceuticals Board of Directors has authorized its Chairman, CEO, and management to take all necessary steps to affect the Dividend and Royalty Rights to NRXP Shareholders and applicable...
Initiative is intended to protect shareholder value through continued compliance with Nasdaq listing rules and elimination of naked short sales positions in the Company's securities The Company is...
Company to receive first allocation of ketamine for sale by month end Partners preparing to ship IV Ketamine to full range of customers via 503a and 503b pharmacies NRx Pharmaceuticals and HOPE...
Marks a major step in the development of what could be the first drug approved for Suicidal Bipolar Depression The study database is being cleaned and locked; statistical analysis and top-line...
NRx Pharmaceuticals received approximately $1.0 million in cash from an existing investor Shares were sold at $0.38, a 26.7% premium the recent share offering, along with one common 5-year warrant...
In the news release, NRx Pharmaceuticals, Inc. Announces Pricing of $1.5 Million Underwritten Public Offering of Common Stock, issued 27-Feb-2024 by NRx Pharmaceuticals, Inc. over PR Newswire, we...
NRx Pharmaceuticals, Inc. (Nasdaq: NRXP), ("NRx Pharmaceuticals" or the "Company"), a clinical-stage biopharmaceutical company, today announced that it intends to offer to sell shares of its...
Presentation available on NRx Pharmaceuticals website at https://www.nrxpharma.com/hope-therapeutics/ KEY UPDATES ARE AS FOLLOWS: NRx management is proposing to award 50% of founding shares in...
DISCLAIMER:
Nothing in the contents transmitted on this board should be construed as an investment advisory, nor should it be used to make investment decisions.
There is no express or implied solicitation to buy or sell securities.
The author(s) may have positions in the stocks or financial relationships with the company or companies discussed and may trade in the stocks mentioned.
Readers are advised to conduct their own due diligence prior to considering buying or selling any stock. All information should be considered for information purposes only.
No stock exchange has approved or disapproved of the information here.
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
