Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
$NRXP - Company to be initially owned by NRx and current NRx shareholders via a planned tax-free dividend
https://finance.yahoo.com/news/nrx-pharmaceuticals-inc-nasdaq-nrxp-114800404.html
$NRXP "Our Asia Pacific partner, Lotus Pharmaceuticals has identified enrollment completion of the #NRX101 trial in suicidal bipolar depression as a material event in their Q4 filing."
Lotus Reported Unaudited January Revenues of NT$1,996 Million, surging 45.4% MoM.
#bipolar #$NRXP #Nasdaq
Link:
Our Asia Pacific partner, Lotus Pharmaceuticals has identified enrollment completion of the #NRX101 trial in suicidal bipolar depression as a material event in their Q4 filing.
— NRx Pharmaceuticals (@NRxPharma) February 8, 2024
Lotus Reported Unaudited January Revenues of NT$1,996 Million, surging 45.4% MoM. #bipolar #$NRXP
$NRXP Going green! NRx Pharmaceutucals is focused on the high unmet need for lifesaving treatments for people with severe bipolar depression & PTSD in the presence of suicidality.
https://t.co/dQEshQQkQb
NRx Pharmaceutucals is focused on the high unmet need for lifesaving treatments for people with severe bipolar depression & PTSD in the presence of suicidality.
— NRx Pharmaceuticals (@NRxPharma) February 13, 2024
Follow our journey. https://t.co/dQEshQQkQb#mentalhealth #depression #bipolar #PTSD #NRX101 #NRX100 #ketamine $NRXP
The second portion of the milestone will be $4 million and, as before, be triggered by a positive response to the Company's planned end of phase 2 meeting with FDA.
$NRXP
$NRXP NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) Announces Receipt of Positive Nasdaq Listing Determination https://finance.yahoo.com/news/nrx-pharmaceuticals-inc-nasdaq-nrxp-114800600.html @Nasdaq $AAPL $NVDA $MSFT $PATH @NRxPharma #pharma #bipolar #depression
Great point. Those are financial incentives to investing here. $NRXP
$NRXP .50 +4.17% NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) Announces Advance of $5 Million Milestone Payment from Partners Alvogen, Inc. and Lotus Pharma https://finance.yahoo.com/news/nrx-pharmaceuticals-inc-nasdaq-nrxp-133000063.html?soc_src=social-sh&soc_trk=tw&tsrc=twtr via @YahooFinance @Nasdaq $AAPL $NVDA $MSFT $PATH @NRxPharma #pharma #bipolar #depression
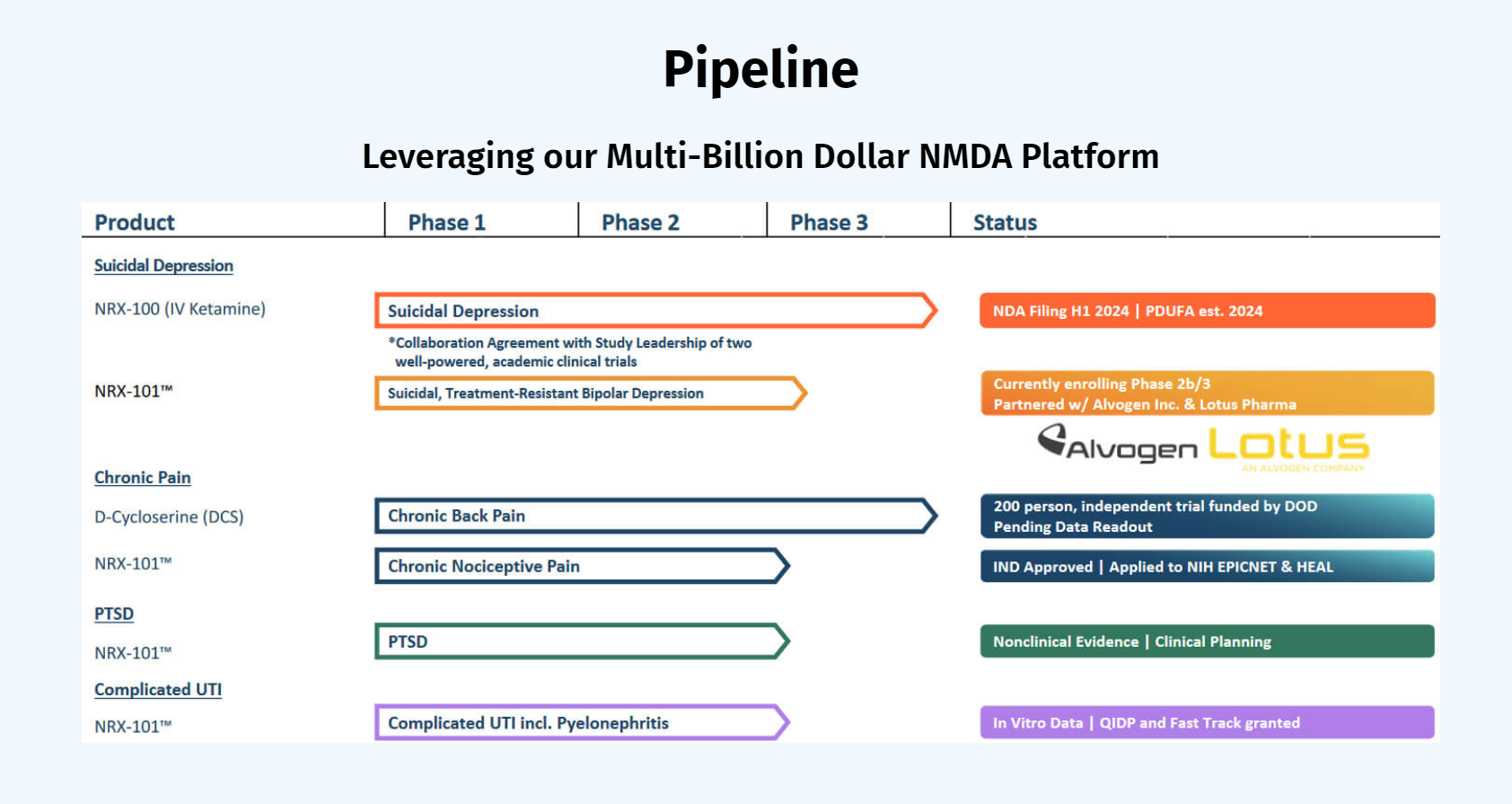
NRx has recently announced plans to submit a New Drug Application for NRX-100 (IV ketamine) in the treatment of suicidal depression, based on results of well-controlled clinical trials conducted under the auspices of the US National Institutes of Health and newly obtained data from French health authorities, licensed under a data sharing agreement. NRx was awarded Fast Track Designation for development of ketamine (NRX-100) by the US FDA as part of a protocol to treat patients with acute suicidality.
"In a market environment where biotech companies have been increasingly challenged to fund clinical development, we appreciate the confidence of partners that share our view that NMDA-antagonist drugs potentially represent a new and potent class of medicines to treat bipolar depression with suicidality. Over the past six months, NRx, Lotus, and Alvogen have formed a close working relationship and we share a joint commitment to bringing this potentially life-saving drug to patients." said Stephen Willard, Chief Executive Officer of NRx Pharmaceuticals.
NRx remains eligible for an additional $324 million in development and sales milestones, as well as tiered double-digit royalties upon approval and commercialization of NRX-101.
$NRXP
Companies continue to work collaboratively to advance NRX-101 through registrational trials
$NRXP - Payment materially extends the Company's cash runway
This is a significant amount of money...
NRx remains eligible for an additional $324 million in development and sales milestones, as well as tiered double-digit royalties upon approval and commercialization of NRX-101.
$NRXP
Awesome News today! Here's the Yahoo Finance link.
$NRXP News February 12, 2024
NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) Announces Advance of $5 Million Milestone Payment from Partners Alvogen, Inc. and Lotus Pharmaceutical Co. Ltd. (1975.TW)
https://finance.yahoo.com/news/nrx-pharmaceuticals-inc-nasdaq-nrxp-133000063.html
She will bust soon.
Impressive news for NRXP today on major funding received for development of the company's breakthrough CNS bio-pharma treatments...
NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) Announces Advance of $5 Million Milestone Payment from Partners Alvogen, Inc. and Lotus Pharmaceutical Co. Ltd. (1975.TW)
Companies continue to work collaboratively to advance NRX-101 through registrational trials
NRx remains eligible for an additional $324 million in development and sales milestones, as well as tiered double-digit royalties upon approval and commercialization of NRX-101.
Payment materially extends the Company's cash runway
RADNOR, Pa., Feb. 12, 2024 /PRNewswire/ -- NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) ("NRx Pharmaceuticals", the "Company"), a clinical-stage biopharmaceutical company, today announced the advance of the first $5 million milestone payment based on the Company's partnership agreement with Alvogen, Inc. and Lotus Pharmaceutical Co. Ltd. (1975.TW).
Under the terms of the updated agreement, the Company will immediately receive $5 million of the first milestone, which NRx will use to fund development of NRX-101 through the phase 2 meeting with FDA. As compensation for advancing the milestone, Alvogen and Lotus will receive 4.1 million warrants to purchase the Company's common stock, at a strike price of $0.40 with a three (3) year term. The second portion of the milestone will be $4 million and, as before, be triggered by a positive response to the Company's planned end of phase 2 meeting with FDA.
NRx then remains eligible to receive up to $320 million in future development and sales milestones, as well as royalty payments escalating to mid-teen percentages on Net Sales, subject to achievement of certain sales volumes. Additionally, Alvogen and Lotus will be responsible for future development and commercialization costs for NRX-101 in treatment of bipolar depression with suicidality.
"In a market environment where biotech companies have been increasingly challenged to fund clinical development, we appreciate the confidence of partners that share our view that NMDA-antagonist drugs potentially represent a new and potent class of medicines to treat bipolar depression with suicidality. Over the past six months, NRx, Lotus, and Alvogen have formed a close working relationship and we share a joint commitment to bringing this potentially life-saving drug to patients." said Stephen Willard, Chief Executive Officer of NRx Pharmaceuticals.
About NRx Pharmaceuticals
NRx Pharmaceuticals is a clinical-stage biopharmaceutical company developing therapeutics based on its NMDA platform for the treatment of central nervous system disorders, specifically suicidal bipolar depression, chronic pain and PTSD. The Company is developing NRX-101, an FDA-designated investigational Breakthrough Therapy for suicidal treatment-resistant bipolar depression and chronic pain. NRx has partnered with Alvogen and Lotus around the development and marketing of NRX-101 for the treatment of suicidal bipolar depression. NRX-101 additionally has potential to act as a non-opioid treatment for chronic pain, as well as a treatment for complicated UTI.
NRx has recently announced plans to submit a New Drug Application for NRX-100 (IV ketamine) in the treatment of suicidal depression, based on results of well-controlled clinical trials conducted under the auspices of the US National Institutes of Health and newly obtained data from French health authorities, licensed under a data sharing agreement. NRx was awarded Fast Track Designation for development of ketamine (NRX-100) by the US FDA as part of a protocol to treat patients with acute suicidality.
Cautionary Note Regarding Forward-Looking Statements
This announcement of NRx Pharmaceuticals, Inc. includes "forward-looking statements" within the meaning of the "safe harbor" provisions of the U.S. Private Securities Litigation Reform Act of 1995, which may include, but are not limited to, statements regarding our financial outlook, product development, business prospects, and market and industry trends and conditions, as well as the Company's strategies, plans, objectives, and goals. These forward-looking statements are based on current beliefs, expectations, estimates, forecasts, and projections of, as well as assumptions made by, and information currently available to, the Company's management.
The Company assumes no obligation to revise any forward-looking statement, whether as a result of new information, future events or otherwise. Accordingly, you should not place reliance on any forward-looking statement, and all forward-looking statements are herein qualified by reference to the cautionary statements set forth above.
https://c212.net/c/img/favicon.png?sn=CL34218&sd=2024-02-12 View original content to download multimedia:https://www.prnewswire.com/news-releases/nrx-pharmaceuticals-inc-nasdaq-nrxp-announces-advance-of-5-million-milestone-payment-from-partners-alvogen-inc-and-lotus-pharmaceutical-co-ltd-1975tw-302059098.html
SOURCE NRx Pharmaceuticals
$NRXP NEWS! NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) Announces Advance of $5 Million Milestone Payment from Partners Alvogen, Inc. and Lotus Pharmaceutical Co. Ltd.
Read More - https://ir.nrxpharma.com/2024-02-12-NRx-Pharmaceuticals,-Inc-Nasdaq-NRXP-Announces-Advance-of-5-Million-Milestone-Payment-from-Partners-Alvogen,-Inc-and-Lotus-Pharmaceutical-Co-Ltd-1975-TW
#NRX101 #PharmaNews #suicide #mentalhealth #depression #stress #PTSD #bipolar $NRXP
$NRXP .5011 +8.00% Great corporate update today!
NRx Pharmaceuticals, Inc. (Nasdaq: $NRXP) Reports Comments by Strategic Partner Lotus Pharmaceutical Co. Ltd. (https://t.co/PKVQC2OL7V) in Recent Financial Report.
— NRx Pharmaceuticals (@NRxPharma) February 9, 2024
Read More - https://t.co/ml7RyYSE0F#bipolar #depression #suicide #mentalhealth #NRX101
NEWS: NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) Reports Comments by Strategic Partner Lotus Pharmaceutical Co. Ltd. (1975.TW) in Recent Financial Report
Fri, February 9, 2024 at 8:30 AM EST
In this article: NRXP
+14.20%
Companies continue to work collaboratively to advance NRX-101 in bipolar depression with suicidality
*Lotus notes that NRx has "achieved a significant milestone by completing enrollment for its phase 2b/3 trial of NRX-101 in suicidal treatment-resistant bipolar depression. The readout for this trial is anticipated in Q2 of this year."
*Productive partnership aims to accelerate NRX-101 to patients in need
*Lotus further reported revenues of NT$1996, a 45% month over month increase.
RADNOR, Pa., Feb. 9, 2024 /PRNewswire/ -- NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) ("NRx Pharmaceuticals", the "Company"), a clinical-stage biopharmaceutical company, today announced that its Asia Pacific strategic partner Lotus Pharmaceutical Co. Ltd (1975.TW) identified enrollment completion of the NRx clinical trial in bipolar depression as a material event in is February 7 financial filing.[1] Lotus provides the Asia Pacific component of NRx's global partnership with Alvogen, Inc.
Lotus reported in the accompanying press release that "Lotus Pharmaceutical's strategic partner, NRX Pharmaceuticals, has achieved a significant milestone by completing enrollment for its phase 2b/3 trial of NRX-101 in suicidal treatment-resistant bipolar depression. The readout for this trial is anticipated in Q2 of this year."
"We at NRx are honored that Lotus has focused its resources on helping us advance NRX-101 for the benefit of patients in the Asia Pacific region. As reported by the World Health Organization, suicidal depression is a significant public health crisis in Pacific rim countries[2], just as it is in the US," said Stephen Willard, Chief Executive Officer of NRx Pharmaceuticals. "We recently held very productive meetings at the JP Morgan conference in San Francisco; we share a mutual commitment to bringing a lifesaving treatment to patients who have been excluded from the clinical trials of all known antidepressants and whose only approved treatment today is electroshock therapy."
NRx anticipates further updates regarding its global partnerships in the coming days.
About NRx Pharmaceuticals
NRx Pharmaceuticals is a clinical-stage biopharmaceutical company developing therapeutics based on its NMDA platform for the treatment of central nervous system disorders, specifically suicidal bipolar depression, chronic pain and PTSD. The Company is developing NRX-101, an FDA-designated investigational Breakthrough Therapy for suicidal treatment-resistant bipolar depression and chronic pain. NRx has partnered with Alvogen and Lotus around the development and marketing of NRX-101 for the treatment of suicidal bipolar depression. NRX-101 additionally has potential to act as a non-opioid treatment for chronic pain, as well as a treatment for complicated UTI.
NRx has recently announced plans to submit a New Drug Application for NRX-100 (IV ketamine) in the treatment of suicidal depression, based on results of well-controlled clinical trials conducted under the auspices of the US National Institutes of Health and newly obtained data from French health authorities, licensed under a data sharing agreement. NRx was awarded Fast Track Designation for development of ketamine (NRX-100) by the US FDA as part of a protocol to treat patients with acute suicidality.
Cautionary Note Regarding Forward-Looking Statements
This announcement of NRx Pharmaceuticals, Inc. includes "forward-looking statements" within the meaning of the "safe harbor" provisions of the U.S. Private Securities Litigation Reform Act of 1995, which may include, but are not limited to, statements regarding our financial outlook, product development, business prospects, and market and industry trends and conditions, as well as the Company's strategies, plans, objectives, and goals. These forward-looking statements are based on current beliefs, expectations, estimates, forecasts, and projections of, as well as assumptions made by, and information currently available to, the Company's management.
The Company assumes no obligation to revise any forward-looking statement, whether as a result of new information, future events or otherwise. Accordingly, you should not place reliance on any forward-looking statement, and all forward-looking statements are herein qualified by reference to the cautionary statements set forth above.
1 Lotus Reported Unaudited January Revenues of NT$1,996 Million, surging 45.4% MoM - Lotus (lotuspharm.com)
2 https://apps.who.int/gho/data/node.main.MHSUICIDEASDR?lang=en
Cision
Cision
View original content to download multimedia:https://www.prnewswire.com/news-releases/nrx-pharmaceuticals-inc-nasdaq-nrxp-reports-comments-by-strategic-partner-lotus-pharmaceutical-co-ltd-1975tw-in-recent-financial-report-302058404.html
SOURCE NRx Pharmaceuticals, Inc.
$NRXP .51 +9.91% Premarket as NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) Announces the Incorporation of HOPE Therapeutics, Inc., and Planned Share Dividend/Royalty Coupon https://finance.yahoo.com/news/nrx-pharmaceuticals-inc-nasdaq-nrxp-114800404.html
$NRXP "Our Asia Pacific partner, Lotus Pharmaceuticals has identified enrollment completion of the #NRX101 trial in suicidal bipolar depression as a material event in their Q4 filing."
Lotus Reported Unaudited January Revenues of NT$1,996 Million, surging 45.4% MoM.
#bipolar #$NRXP #Nasdaq
Link:
Our Asia Pacific partner, Lotus Pharmaceuticals has identified enrollment completion of the #NRX101 trial in suicidal bipolar depression as a material event in their Q4 filing.
— NRx Pharmaceuticals (@NRxPharma) February 8, 2024
Lotus Reported Unaudited January Revenues of NT$1,996 Million, surging 45.4% MoM. #bipolar #$NRXP
$NRXP NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) Announces the Incorporation of HOPE Therapeutics, Inc., and Planned Share Dividend/Royalty Coupon https://finance.yahoo.com/news/nrx-pharmaceuticals-inc-nasdaq-nrxp-114800404.html?soc_src=social-sh&soc_trk=tw&tsrc=twtr via @YahooFinance
NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) Announces Completion of Enrollment of its Phase 2b/3 Trial of NRX-101 in Suicidal Treatment Resistant Bipolar Depression
January 22 2024 - 06:48AM
PR Newswire (US)
https://ih.advfn.com/stock-market/NASDAQ/nrx-pharmaceuticals-NRXP/stock-news/93100410/nrx-pharmaceuticals-inc-nasdaq-nrxp-announces-c
PR Newswire
Wed, January 17, 2024 at 3:48 AM PST
RADNOR, Pa., Jan. 17, 2024 /PRNewswire/ -- NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) ("NRx" or the "Company"), a clinical-stage biopharmaceutical company developing therapies for suicidal depression, chronic pain, and PTSD, today announced that on January 16, 2024, the Company was notified by The Nasdaq Stock Market LLC ("Nasdaq") that the Nasdaq Hearings Panel (the "Panel") granted the Company's request for the transfer of its listing to The Nasdaq Capital Market, subject to the Company evidencing compliance with all applicable criteria for continued listing on the Capital Market tier, including the $35 million in market value of listed securities (MVLS) and the $1.00 bid price requirements. The Company believes it has already satisfied the market value of listed securities requirement and is working to evidence compliance with the minimum bid price requirement by April 16, 2024, as required by the Panel's decision. An issuer must evidence compliance with the minimum threshold for at least ten consecutive business days to be deemed by Nasdaq to have complied with the applicable requirement.
"NRx is pleased to share this encouraging news with our shareholders as we continue to meet our drug development milestones as set outlined in our recent communications (NRx Corporate Presentation). This quarter we aim to file our first New Drug Application for NRX-100 (ketamine) for treatment of acute suicidal depression and look forward to announcing top line results from our phase 2b/3 trial of NRX-101 (D-cycloserine/lurasidone) in the treatment of suicidal bipolar depression. These milestones, together with additional updates, are anticipated in advance of the Nasdaq compliance date," said Stephen Willard, JD, CEO of NRx.
NRX received the FDA QUIDP for the NRX-101 in Complicated Urinary Tract Infection and Pyelonephritis
From the FDA: III. QIDP DESIGNATION DEFINED
47
48 Section 505E(g) of the FD&C Act provides for the designation by FDA of certain
49 antimicrobial products as QIDPs. A QIDP is defined in section 505E(g) as: "an
50 antibacterial or antifungal drug for human use intended to treat serious or life-threatening
51 infections, including those caused by –
52
53 (1) an antibacterial or antifungal resistant pathogen, including novel or emerging
54 infectious pathogens; or
55
56 (2) qualifying pathogens listed by the Secretary under" section 505E(f) of the
57 FD&C Act.
58
59 The Agency has codified the list of qualifying pathogens at 21 CFR 317.2.
60
61 For a drug product to be designated a QIDP, the sponsor is required to demonstrate that the drug
62 is an "antibacterial or antifungal drug for human use intended to treat serious or life-threatening
infections."3 63 A sponsor requesting a QIDP designation may also include documentation that the
64 product is intended to treat an "antibacterial or antifungal resistant pathogen, including novel or
emerging infectious pathogens"4 65 or a qualifying pathogen as part of the designation request;
66 however, such documentation is not required.
NRx Pharmaceuticals Announces Signing of a Data and Technical Information Agreement with Columbia University Accessing Key Data Demonstrating Efficacy and Safety of Intravenous Ketamine for the Treatment of Suicidal Depression
https://ir.nrxpharma.com/2023-12-19-NRx-Pharmaceuticals-Announces-Signing-of-a-Data-and-Technical-Information-Agreement-with-Columbia-University-Accessing-Key-Data-Demonstrating-Efficacy-and-Safety-of-Intravenous-Ketamine-for-the-Treatment-of-Suicidal-Depression
NRx Pharmaceuticals Announces FDA Clearance of its Investigational New Drug (IND) Application for NRX-101 in the Treatment of Complicated Urinary Tract Infections
https://ir.nrxpharma.com/2023-12-18-NRx-Pharmaceuticals-Announces-FDA-Clearance-of-its-Investigational-New-Drug-IND-Application-for-NRX-101-in-the-Treatment-of-Complicated-Urinary-Tract-Infections
always good info here.. arqt jumped in thanks to whoever fisrt posted
News Release Issued: November 6, 2023 (6:48am EST)
https://ir.nrxpharma.com/2023-11-06-NRx-Pharmaceuticals-and-Nephron-Pharmaceuticals-Announce-Joint-Agreement-to-Develop-Intravenous-Ketamine-to-Treat-Suicidal-Depression
NRx Pharmaceuticals (NRXP) Enters Development Agreement for the Manufacture of Ketamine to Treat Suicidality and Depression
https://www.google.com/url?rct=j&sa=t&url=https://www.streetinsider.com/Corporate%2BNews/NRx%2BPharmaceuticals%2B%2528NRXP%2529%2BEnters%2BDevelopment%2BAgreement%2Bfor%2Bthe%2BManufacture%2Bof%2BKetamine%2Bto%2BTreat%2BSuicidality%2Band%2BDepression/22335240.html&ct=ga&cd=CAEYACoTMjEzNzQyMTU5MTEzNDcwMTg0NzIaNWMyNTMyOTNiYTRkNjY3Mjpjb206ZW46VVM&usg=AOvVaw24Sh_M_p_xsQry2Kr-cHf1
https://www.google.com/url?rct=j&sa=t&url=https://beststocks.com/nrx-pharmaceuticals-obtains-fda-clearance-for/&ct=ga&cd=CAEYASoUMTA4NTcxMjE2NTM3NTEzNTI1ODQyGjVjMjUzMjkzYmE0ZDY2NzI6Y29tOmVuOlVT&usg=AOvVaw3I13qCUdkzlu8VSOMer8TF
NRx Pharmaceuticals Obtains FDA Clearance for Human Trials of NRX101 A Groundbreaking Treatment for Chronic Pain
RADNOR, Pa., Oct. 2, 2023 /PRNewswire/ -- NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) ("NRx Pharmaceuticals", the "Company"), a clinical-stage biopharmaceutical company, today announced that FDA has given the company clearance to proceed with human trials to treat Chronic Pain under the Investigational New Drug (IND) application filed with the U.S. Food and Drug Administration (FDA) for the use of NRX-101. The IND application leverages pioneering research on the use of D-cycloserine (a key ingredient of NRX-101) in the treatment of chronic pain and the recent licensure by NRx of a US Patent for the use of D-cycloserine in the treatment of pain.
NRX Pharmaceuticals Announces Data Sharing Agreement Demonstrating Efficacy and Safety of Intravenous Ketamine for the Treatment of Suicidal Bipolar Depression
September 15 2023 - 06:46AM
PR Newswire (US)
Ketamine, an NMDA blocker, was highly effective in Bipolar subgroup (p<0.001)
NRx plans to present the data to FDA with a goal of identifying a path to an NDA
RADNOR, Pa., Sept. 15, 2023 /PRNewswire/ -- NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) ("NRx Pharmaceuticals", the "Company"), a clinical-stage biopharmaceutical company, today announced that it has signed a data sharing agreement with the study leadership of a randomized, placebo-controlled trial of 156 patients hospitalized for Acute Suicidality and Depression in 7 French Government Hospitals.
This trial, conducted under International Good Clinical Practices and Helsinki standards demonstrated a dramatic and statistically-significant reduction in suicidal ideation and depression (P<.0001) among patients randomized to intravenous racemic ketamine, compared to those randomized to placebo. The trial reached its primary endpoint for all patients and demonstrated the largest effect among the subgroup with bipolar depression (84% vs 28% remission, drug vs. placebo; p<0.0001; Odds ratio 14).
The top line data from this trial were published in the British Medical Journal (BMJ 2022; 376 doi: https://doi.org/10.1136/bmj-2021-067194) and additional data reports are planned. The efficacy demonstrated confirms earlier, smaller trials conducted in the United States and elsewhere.
As disclosed previously, NRx met with the FDA in January 2023 at which time the FDA requested randomized, placebo-controlled data in support of intravenous ketamine for acute suicidality in the inpatient setting. Such trials are extraordinarily complex to organize and generally require Government support. In this case, NRx approached the Fondation FundaMental, led by Prof. Marion Leboyer, MD, PhD, who served on the NRx Scientific Advisory Board and facilitated establishing the current Data Sharing Agreement.
Under this agreement, NRx has translated the clinical study report, which will be submitted to FDA and is converting the electronic, patient level data files to a form suitable for FDA review. NRx plans to meet with FDA in the coming months to discuss a regulatory path for the use of ketamine to treat patients with Bipolar Depression and Acute Suicidal Ideation and Behavior.
"We at NRx are honored that the study leadership has agreed to share these landmark data so that they may be submitted to and reviewed by the US FDA," said Dr. Jonathan Javitt, Founder and Chief Scientist of NRx Pharmaceuticals. "We look forward to a fruitful ongoing collaboration with Prof. Abbar and his study team leadership for the benefit of patients everywhere."
Looks like they knocked out of the park with the French study news
NRx Pharmaceuticals Announces Agreement with LifeSci Associates to Provide Financial Services and Support
Industry veteran Richard Narido joins the NRx Pharmaceuticals management team
RADNOR, Pa., Sept. 14, 2023 /PRNewswire/ -- NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) ("NRx Pharmaceuticals" or the "Company"), a clinical-stage biopharmaceutical company, today announced that it has entered into an agreement (the "Agreement") with LS Associates, a division of LifeSci Advisors, LLC ("LSA"), pursuant to which LSA will provide certain consulting services to the Company, including but not limited to, arranging for the provision of the Company's Interim Chief Financial Officer. In connection with the Agreement, the Company appointed Richard Narido to serve as Interim Chief Financial Officer of the Company. As Interim Chief Financial Officer, Mr. Narido will serve as the Company's principal financial officer and principal accounting officer.
"I am pleased to welcome Richard to the team at NRx Pharmaceuticals. He brings great experience and energy to our dynamic company," said Stephen Willard, Chief Executive Officer of NRx Pharmaceuticals. "Additionally, we are pleased to have immediate access to LSA's broad team of financial professionals, who will be available to us as needed," he continued.
Prior to his appointment as the Company's Interim Chief Financial Officer, Mr. Narido served as the Chief Financial Officer of Lucira Health ("Lucira") until Pfizer Inc.'s acquisition of Lucira in April 2023. From July 2018 to March 2021, Mr. Narido served in various roles at Assembly Biosciences, Inc., including most recently as Executive Director, Finance, Controllership and Treasury. From June 2014 to June 2018, Mr. Narido served in various roles at Bio-Rad Laboratories, Inc., including as Americas Head of Finance, Global Commercial Operations. Prior to June 2018, Mr. Narido held various finance roles, including Global Head Finance Reporting and Accounting for Novartis Vaccines and Diagnostics and several industry-related positions, including Business Unit Controller for McKesson Corporation. Mr. Narido started his career with PricewaterhouseCoopers's Financial Audit and Assurance practice. Mr. Narido holds a Bachelor of Science degree from the University of San Francisco and a Master of Science degree from the Pepperdine Graziadio Business School.
The Company also today announced that it accepted the resignation of Seth Van Voorhees, Ph.D., former Chief Financial Officer of the Company, which was tendered on September 11, 2023, as he has decided to pursue other professional opportunities. His resignation will be effective as of September 30, 2023, in order to facilitate a smooth transition. "We thank Seth for his many contributions to the Company, and we wish him well in his future endeavors," Mr. Willard commented.
About NRX-101
NRX-101, a fixed dose combination of D-cycloserine and lurasidone, has been granted Fast Track Designation, Breakthrough Therapy Designation, a Special Protocol Agreement, and a Biomarker Letter of Support from the FDA for S-TRBD. Additionally, the product is being developed in chronic pain and PTSD.
I signed up for Chaikin Power Pulse and here is what they say about NRXP .. I rarely join these services, but it was a free trial. I did not expect that rating. My META and TSLA are just listed as bullish.. But they are not recommending a buy yet on NRXP just listing it as a "wait".. So we shall see.
NRXP - VERY BULLISH
NRx Pharmaceuticals, Inc.
$0.25
0.01
(+4.17%)
As of 09/11/23 00:28AM EDT
INDUSTRY
Pharmaceuticals
EARNINGS
POWER GAUGE RATING
As of 09/11/23
NRXP is Very Bullish
Financials
Earnings
Technicals
Experts
The Chaikin Power Gauge Rating for NRXP is Very Bullish due to very positive expert activity, attractive financial metrics and strong earnings performance. The stock also has weak price/volume activity.
Expert activity for NRXP is very positive which is evidenced by insiders purchasing stock and relative strength of the stock's industry.
PRE-TRADE CHECKLIST
Strength
2/3
Power Gauge Rating Very Bullish
Rel. Strength vs SPY Weak
Industry Strength Strong
So they are claiming that NRX will do three things.. Miracle drug? or... are they just trying to get something to stick to the wall? I do not trust the FDA to keep their profit agenda and or politics out of all this.
If just one of these options works and is approved then it could be a good thing.
NRx Pharmaceuticals Announces the Licensure of a US Patent to Support Use of NRX-101™ for Chronic Pain
NRx Pharmaceuticals Reports Minutes of Recent U.S. Food and Drug Administration (FDA) Meeting on the Development of NRX-101 to Treat Severe Bipolar Depression in Patients with Suicidality
NRx Pharmaceuticals Announces Potent Antibacterial Activity of NRX-101 Against Common, Antibiotic Resistant Urinary Pathogens
This turned out to be real POS didn't it. Geez. Moon market never mentioned it again.
|
Followers
|
52
|
Posters
|
|
|
Posts (Today)
|
1
|
Posts (Total)
|
1116
|
|
Created
|
12/15/20
|
Type
|
Free
|
| Moderators ProfitScout jedijazz | |||












Both drugs demonstrated > 50% response for treating depression. NRX-101 demonstrated a mean 76% reduction in symptoms of akathisia compared to lurasidone that was sustained over 42 days (Effect...

New data demonstrates no impact of NRX-101 on gut or vaginal flora – considered primary causes of pseudomembranous colitis due to C difficile and vaginal yeast infections NRX-101 previously...
Formulation based on prior patents by NRx founder Achieved pH neutral formulation of ketamine, potentially enabling both intravenous (IV) and subcutaneous (SQ) administration Company expects...
Data transferred for independent statistical analysis Top-line data expected in April 2024 RADNOR, Pa., April 8, 2024 /PRNewswire/ -- NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) ("NRx...
Four potential near-term milestones, including data from two clinical trials, an NDA filing and an upcoming share dividend -- 50% reduction in corporate overhead and 25% reduction in overall net...
Four potential near-term milestones, including data from two clinical trials, an NDA filing and a share dividend RADNOR, Pa., March 28, 2024 /PRNewswire/ -- NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) ("
NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) ("NRx Pharmaceuticals", the "Company"), a clinical-stage biopharmaceutical company, today announced that it will release its fourth quarter and full year...
NRx Pharmaceuticals, Inc. (Nasdaq:NRXP) ("NRx Pharmaceuticals", the "Company"), a clinical-stage biopharmaceutical company, today announced that Dr. Jonathan Javitt, Chairman and Chief Scientist...
NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) ("NRx Pharmaceuticals", the "Company"), a clinical-stage biopharmaceutical company, today announced that it will release its fourth quarter and full year...
61.4% (56,781,354) of eligible shares voted 94.4% of votes were cast in favor of the resolution RADNOR, Pa., March 21, 2024 /PRNewswire/ -- NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) ("NRx...
NRx Pharmaceuticals Board of Directors has authorized its Chairman, CEO, and management to take all necessary steps to affect the Dividend and Royalty Rights to NRXP Shareholders and applicable...
Initiative is intended to protect shareholder value through continued compliance with Nasdaq listing rules and elimination of naked short sales positions in the Company's securities The Company is...
Company to receive first allocation of ketamine for sale by month end Partners preparing to ship IV Ketamine to full range of customers via 503a and 503b pharmacies NRx Pharmaceuticals and HOPE...
Marks a major step in the development of what could be the first drug approved for Suicidal Bipolar Depression The study database is being cleaned and locked; statistical analysis and top-line...
NRx Pharmaceuticals received approximately $1.0 million in cash from an existing investor Shares were sold at $0.38, a 26.7% premium the recent share offering, along with one common 5-year warrant...
In the news release, NRx Pharmaceuticals, Inc. Announces Pricing of $1.5 Million Underwritten Public Offering of Common Stock, issued 27-Feb-2024 by NRx Pharmaceuticals, Inc. over PR Newswire, we...
NRx Pharmaceuticals, Inc. (Nasdaq: NRXP), ("NRx Pharmaceuticals" or the "Company"), a clinical-stage biopharmaceutical company, today announced that it intends to offer to sell shares of its...
Presentation available on NRx Pharmaceuticals website at https://www.nrxpharma.com/hope-therapeutics/ KEY UPDATES ARE AS FOLLOWS: NRx management is proposing to award 50% of founding shares in...
DISCLAIMER:
Nothing in the contents transmitted on this board should be construed as an investment advisory, nor should it be used to make investment decisions.
There is no express or implied solicitation to buy or sell securities.
The author(s) may have positions in the stocks or financial relationships with the company or companies discussed and may trade in the stocks mentioned.
Readers are advised to conduct their own due diligence prior to considering buying or selling any stock. All information should be considered for information purposes only.
No stock exchange has approved or disapproved of the information here.
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
