Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Please provide the case/ docket number for the cases you are referring to.
Which jurisdiction is it?
Are you able to articulate the damages sought?
Any word on the mediation ??
So far its been a one way street,
ALL GIVE by RLF, and ALL TAKE by Neuro
yes, TB, It must be like all the other numerous law firms
that have filed suit against Neuro for fraud.
Suppose none of these numerous law firms have any clients either ???
Especially considering all the losses on almost every shareholder
or former shareholder....
Interesting interpretation. Looks like they are still looking for the first client
Hot News Out Today On NRXP..........The Elephant in the Court Room
https://www.prnewswire.com/news-releases/nrxp-investors-have-opportunity-to-lead-nrx-pharmaceuticals-inc-securities-fraud-lawsuit-301486155.html
Another Fraud Lawsuit Against Neuro and Co
47% of NRXP stock is owned by insiders. Obviously it is in there best interest for the stock price to go up AS SOON AS POSSIBLE!!!!
https://finance.yahoo.com/news/much-nrx-pharmaceuticals-inc-nasdaq-122113410.html
Good luck and GOD bless,
"God Save The Queen"
Send her some aviptadil and through in some zyesami too.
I'll pay the shipping.
Fox's Neil Cavuto said a second case of Covid-19 nearly killed him
https://www.cnn.com/2022/02/21/media/neil-cavuto-covid-19-pneumonia/index.html
The whole world needs ZYESAMI® (Avipdatil) ASAP (As Soon as possible)
Good luck and GOD bless,
The whole market tanked.... Considering what is going on with everything...it is no surprise..
Big Dump today on light volume.
That does not look very bullish to me.
From another place where posts are often deleted much later.
Seems this jj is very active again............
........................................
Jonathan Javitt 13 hours ago @MERICA
So you are referring to a document stating that a hospital brought in an a doctor who was not a licensed investigator without telling the sponsor and we reported that to FDA. Also, that we published the statistically significant difference on primary endpoint and on survival if you control for site of care (tertiary care vs. regional hospitals). Everyone who looks at the published data can see that two regional hospitals had zero survival in randomized patients. In the midst of the December 2020 surge. The data obtained in the study to which you refer were the same data that NIH used to select ZYESAMI for ACTIV-3b in which ZYESAMI is the only remaining investigational medicine. Now NIH is taking the medicine we have successfully manufactured to The EU, the UK, Brazil, and and Scandinavia with the active participation of those governments. As Dr. Francis Collins said to the public, NIH started with a pool of 600 candidate medicines. ZYESAMI was one of a handful selected for ACTIV.
This week you saw an important report from the NIH DSMB based on enrollment of 448 patients, funded by the US Government. We continue to provide FDA with additional data in support of safety and efficacy and are delighted by this week’s news of the appointment of a Senate-confirmed FDA commissioner.
We are likely the smallest biotech company ever to make it to the finals. You should expect us to keep advancing our investigational medicines in the interests of patients and shareholders. Don’t expect 100% pass completion. It doesn’t happen in the Superbowl and it doesn’t happen in biotech.
......................................
AND BY THAT...................
Jonathan Javitt 20 hours ago
@MERICA
Unlicensed people?
What are you talking about?
.........................................
Some Great posters replying to these comments also....
Has jjs legal team let him out of the box to speak
on public BBs. ????? Is this cause the stock is dropping again ?
FDA Exec on Camera Reveals Future COVID Policy
by Project Veritas.
Apparently The FDA will approve a drug if they get paid enough to do so! Perhaps that is why we are waiting so long. This is disgusting!
Millions of people worldwide have died of COVID 19 so far and nearly 10,000 people are still dying each day!!!'
There were over 278,000 deaths worldwide During the last 28 days including over 67,000 deaths in the USA during the last 28 days
Those are the TRUE FACTS!!!
https://coronavirus.jhu.edu/map.html
The USA and the entire world needs NRx Pharmaceuticals drugs and vaccines.
Good luck and GOD bless,
Ahhh yes- When you see the lawyers (especially vultures from several different firms)sniffing around, you know this thing is about to get EUA and rocket up.
https://www.nrxpharma.com/nrx-pharmaceuticals-announces-us-national-institutes-of-health-study-of-zyesami-aviptadil-in-critical-covid-19-is-cleared-to-complete-full-enrollment/
RADNOR, Pa., Feb. 15, 2022 /PRNewswire/ — NRx Pharmaceuticals (Nasdaq: NRXP), a clinical-stage, biopharmaceutical company, today announced results of a review conducted by the Therapeutics and Prevention Data Safety and Monitoring Board (DSMB) of the National Institute of Allergy and Infectious Diseases (NIAID) of the National Institutes of Health (NIH) on February 14, 2022. The DSMB reviewed data on 448 ICU patients with Critical COVID-19 Respiratory Failure who were enrolled in the ACTIV-3b (TESICO) trial1. The TESICO protocol was submitted by NIH and cleared by the US Food and Drug Administration (FDA) as a Phase 3 trial that, if positive, may be used in the submission of a New Drug Application for ZYESAMI®.
ACTIV-3b is a randomized, placebo-controlled clinical trial testing ZYESAMI and Veklury® (remdesivir; Gilead Sciences:Nasdaq:GILD) — alone and in combination — in hospitalized patients with acute respiratory failure due to COVID-19. The patients enrolled in TESICO are critically-ill patients in the ICU who have exhausted other approved therapies and require high-flow nasal oxygen, mechanical ventilation, or extracorporeal membrane oxygenation to maintain blood oxygen.
At a September 2021 Reagan Udall Foundation conference chaired by Dr. Francis Collins, former Director of the NIH and Dr. Janet Woodcock, Acting Commissioner of the FDA, Dr. Collins identified ZYESAMI® as one of a handful of investigational compounds selected by the NIH for Phase 3 study from among 600 candidate compounds initially considered.2 Last week, the NIH announced the discontinuation of the PF-07304814 arm of the ACTIV-3 trial on account of futility3 According to an NIH spokesperson, aviptadil is the sole remaining investigational medicine being offered within the ACTIV-3 trial, which targets the most critically-ill patients with COVID-19.4
While the continuation of this trial certainly does not guarantee success in proving the primary endpoint, NRx is encouraged to reach this level of enrollment without identifying either a safety or futility stopping point. Some of the underlying experimental hypotheses in the trial and the statistical power available at the projected study enrollment to prove those hypotheses will be presented in future corporate updates. Next week, NRx investigators will present the first peer-reviewed findings from the Company’s Phase 2b/3 clinical trial, completed in March 2021.5 These data were reviewed by the NIH as part of its decision to select ZYESAMI for inclusion in ACTIV-3b.
NRx further confirms that it has received no reports of drug-related Serious Adverse Events from the NIH and is forwarding this safety information to the FDA for inclusion in its ongoing review. These findings increase the safety database of patients treated with ZYESAMI to more than 800 patients.
At the DSMB meeting, it was noted that recruitment into the aviptadil arms of the study is ahead of recruitment into the other arms and may potentially be completed in a matter of months patients to the. The Company views this recruitment by physicians at the TESICO study sites as an encouraging sign.The ACTIV-3b trial has now enrolled 448 Critical COVID-19 patients, representing more than 80% of the targeted recruitment. NIH is now moving forward to bring the ACTIV-3b protocol to Brazil, the European Union, the United Kingdom, and Scandinavia. NRx has manufactured investigational medicine to the standards required in those jurisdictions and reported in September 2021 that it passed a European Qualified Person (QP) audit related to that drug supply. NRx expects to complete regulatory requirements to export ZYESAMI to those regions for investigational use in the coming weeks.
“With more than 80% of the trial enrolled, we are highly encouraged that the Data Safety and Monitoring Board has continued enrollment and that trial investigators have continued to express enthusiasm for enrolling patients into the ZYESAMI arm of the trial at some of the nation’s most advanced hospitals,” said Prof. Jonathan Javitt, MD, MPH, Chairman and CEO of NRx. “So far, we have received no reports of drug-related Serious Adverse Events in ZYESAMI-treated patients and are sharing those data with the FDA. We are indebted to the NIH leadership for its decision to include our investigational medicine in the ACTIV program alongside investigational medicines from major pharmaceutical companies and hope that ZYESAMI will soon become an important therapeutic option that enables physicians to offer a renewed chance at life to patients who have exhausted all approved therapies.”
2/12 Robbins Geller Rudman & Dowd Lawfirm Update....
https://www.marketscreener.com/quote/stock/NRX-PHARMACEUTICALS-INC-38908842/news/NOTICE-Investors-in-NRx-Pharmaceuticals-Inc-with-Substantial-Losses-Have-Opportunity-to-Lead-Clas-38272928/
Good Luck and God Bless
NRx Aims to Bring Zyesami to Market Based on Real-World Data in Treating Seriously Ill COVID-19 Patients
https://biobuzz.io/nrx-aims-to-bring-zyesami-to-market-based-on-real-world-data-in-treating-seriously-ill-covid-19-patients/
Good luck and GOD bless,
LISTEN, regarding the treatment of COVID-19 and it's variants
New COVID-19 Antiviral Pills: How Do They Work?
https://www.sciencefriday.com/segments/covid-antiviral-pills/
******************************************************************
ZYESAMI®
ZYESAMI® (AVIPTADIL®) ZYESAMI® (Aviptadil acetate), a synthetic form of human Vasoactive Intestinal Peptide (VIP), is a substance produced by the body, that amongst other functions, helps protect cells against inflammatory conditions. VIP has been granted Fast Track Designation by the U.S. Food and Drug Administration (FDA) for the treatment of patients suffering from Critical COVID-19 with respiratory failure, and is now in clinical trials. The FDA has also provided an Expanded Access Protocol for ZYESAMI®, a destination that an investigational medicine is intended to treat serious or life-threatening diseases or conditions, and has shown potential to address unmet medical needs. Emerging data indicates that VIP binds uniquely to receptors on Alveolar Type II cells in the lung, the same cells that bind the SARS-CoV-2 virus. VIP is believed to protect those cells and the surrounding pulmonary epithelium by blocking cytokines, preventing cytopathy (cell death), and upregulating the production of surfactant, the loss of which is increasingly implicated in COVID-19 respiratory failure. Ongoing trials show VIP may also inhibit the replication of the SARS-CoV2 virus. In the most recent Phase 3 trial for ZYESAMI®, data showed ZYESAMI® was more effective than placebo in treating patients suffering from Critical COVID-19 with respiratory failure, reducing their hospital stay, and increasing their likelihood of survival. The most common adverse event noted in the data was diarrhea.
Uh-oh, the rats are leaving the sinking ship!
https://ih.advfn.com/stock-market/NASDAQ/nrx-pharmaceuticals-NRXP/stock-news/87258595/current-report-filing-8-k
walker.....
Great Find....all the thanks to you for sharing....
Thanks, Joe, for your analysis.
When I read it, I wasn't too impressed, either. Nothing definitive for the drug.
JJ will create a fluff PR out of nothing.
quick summary.......
...."may have better neutralizing ability"......
Yep, it MIGHT WORK...
.........."has unique potential"..........
Yes, ....it MIGHT WORK..........
Sounds like a desperation bit of buts, mights, maybes, news to me..............
No mention of the elephant in the court room.
NRx Reports Remarks by Israel Institute for Biological Research on the future of the BriLife™ Covid-19 Investigational Vaccine
A friend of mine sent me this... It mentions Aviptadil as one of the last standing studies. Big pharm has failed to compete..
Sorry if already posted!
https://www.fiercebiotech.com/biotech/pfizer-a-rare-covid-19-setback-dumps-paxlovid-s-intravenous-sibling-to-leave-activ-3-future
Pfizer, in a rare COVID-19 setback, dumps Paxlovid's intravenous sibling in further blow to ACTIV-3
by Nick Paul Taylor | Feb 8, 2022 8:20am
Add Pfizer to the long list of victims of ACTIV-3. While the Big Pharma has enjoyed unparalleled success in COVID-19, it was unable to buck the trend in the National Institutes of Health (NIH) study of hospitalized patients and has stopped development of Paxlovid’s intravenous sibling PF-07304814.
The NIH set up ACTIV-3 to test anti-SARS-CoV-2 monoclonal antibodies and other therapies in patients hospitalized with COVID-19. Brii Biosciences, Eli Lilly, GlaxoSmithKline and Novartis are among the drug developers to contribute candidates to the study. All those candidates failed to move the needle in hospitalized patients with severe COVID-19, resulting in a persistent unmet need even as the broader pandemic armory has gone from strength to strength.
PF-07304814 represented a different answer to the question of how to treat severe COVID-19. Like one of the components of Pfizer’s oral COVID-19 drug Paxlovid, PF-07304814 is a SARS-CoV-2 main protease inhibitor. The idea was to expand into patients with severe COVID-19 through intravenous delivery.
That plan, like others tested by Pfizer’s peers, came unstuck in NIH’s ACTIV-3 trial. Dosing of the intravenous antiviral in the trial has stopped, and Pfizer has discontinued the global clinical development. Pfizer said the decision “was made based on a totality of information, including a careful review of early data and a thorough assessment of the candidate’s potential to successfully fulfill patient needs.”
RELATED: Novartis-partnered DARPin flames out in COVID-19 study
The discontinuation means ACTIV-3 is no longer enrolling patients. In a recent update to the listing on ClinicalTrials.gov, the sponsor changed the status from “recruiting” to “active, not recruiting” and added “Suspended: Participants are not currently being randomized to this intervention” to the description of the PF-07304814 arm. Patients aren’t being randomized into any of the other arms, either.
ACTIV-3 has been a rare graveyard for COVID-19 programs. The global vaccine race and push to create therapies for mild to moderate COVID-19 each featured multiple successes. But efforts to improve on Gilead Sciences' Veklury in hospitalized COVID-19 patients have floundered, potentially because by that point in the disease pathway elements other than the virus are at play. The NIH is still pushing ahead, though.
"The ACTIV-3 program has not ended. The ACTIV-3 inpatient protocol is no longer enrolling participants to receive PF-07304814. However, ACTIV-3b, also known as the ACTIV-3 critical care protocol, continues to randomize participants to receive aviptadil. Looking ahead, ACTIV-3 partners are considering ways to evolve to evaluate different treatment strategies based on different stages of disease," a spokesperson for the National Institute of Allergy and Infectious Diseases said.
Pfizer disclosed the discontinuation of PF-07304814 in its quarterly pipeline update. The Big Pharma also used the update to reveal the end of work on PRMT5 inhibitor PF-06939999 in solid tumor patients and PF-07059013 in sickle cell disease. Both programs were in phase 1.
Editor's note: This story was updated at 05:10 a.m. ET on Feb. 9 to include information from the NIAID.
A friend of mine sent me this... It mentions Aviptadil as one of the last standing studies. Big pharm has failed to compete..
Sorry if already posted!
https://www.fiercebiotech.com/biotech/pfizer-a-rare-covid-19-setback-dumps-paxlovid-s-intravenous-sibling-to-leave-activ-3-future
Pfizer, in a rare COVID-19 setback, dumps Paxlovid's intravenous sibling in further blow to ACTIV-3
by Nick Paul Taylor | Feb 8, 2022 8:20am
Add Pfizer to the long list of victims of ACTIV-3. While the Big Pharma has enjoyed unparalleled success in COVID-19, it was unable to buck the trend in the National Institutes of Health (NIH) study of hospitalized patients and has stopped development of Paxlovid’s intravenous sibling PF-07304814.
The NIH set up ACTIV-3 to test anti-SARS-CoV-2 monoclonal antibodies and other therapies in patients hospitalized with COVID-19. Brii Biosciences, Eli Lilly, GlaxoSmithKline and Novartis are among the drug developers to contribute candidates to the study. All those candidates failed to move the needle in hospitalized patients with severe COVID-19, resulting in a persistent unmet need even as the broader pandemic armory has gone from strength to strength.
PF-07304814 represented a different answer to the question of how to treat severe COVID-19. Like one of the components of Pfizer’s oral COVID-19 drug Paxlovid, PF-07304814 is a SARS-CoV-2 main protease inhibitor. The idea was to expand into patients with severe COVID-19 through intravenous delivery.
That plan, like others tested by Pfizer’s peers, came unstuck in NIH’s ACTIV-3 trial. Dosing of the intravenous antiviral in the trial has stopped, and Pfizer has discontinued the global clinical development. Pfizer said the decision “was made based on a totality of information, including a careful review of early data and a thorough assessment of the candidate’s potential to successfully fulfill patient needs.”
RELATED: Novartis-partnered DARPin flames out in COVID-19 study
The discontinuation means ACTIV-3 is no longer enrolling patients. In a recent update to the listing on ClinicalTrials.gov, the sponsor changed the status from “recruiting” to “active, not recruiting” and added “Suspended: Participants are not currently being randomized to this intervention” to the description of the PF-07304814 arm. Patients aren’t being randomized into any of the other arms, either.
ACTIV-3 has been a rare graveyard for COVID-19 programs. The global vaccine race and push to create therapies for mild to moderate COVID-19 each featured multiple successes. But efforts to improve on Gilead Sciences' Veklury in hospitalized COVID-19 patients have floundered, potentially because by that point in the disease pathway elements other than the virus are at play. The NIH is still pushing ahead, though.
"The ACTIV-3 program has not ended. The ACTIV-3 inpatient protocol is no longer enrolling participants to receive PF-07304814. However, ACTIV-3b, also known as the ACTIV-3 critical care protocol, continues to randomize participants to receive aviptadil. Looking ahead, ACTIV-3 partners are considering ways to evolve to evaluate different treatment strategies based on different stages of disease," a spokesperson for the National Institute of Allergy and Infectious Diseases said.
Pfizer disclosed the discontinuation of PF-07304814 in its quarterly pipeline update. The Big Pharma also used the update to reveal the end of work on PRMT5 inhibitor PF-06939999 in solid tumor patients and PF-07059013 in sickle cell disease. Both programs were in phase 1.
Editor's note: This story was updated at 05:10 a.m. ET on Feb. 9 to include information from the NIAID.
Thinking about it a little, and with jjs self admitted employment all over the map, at a bunch of unnamed "successful" biopharma startups, and named BP companies, like Merck, Allergan, Pharmacia, Novartis, and Pfizer, not to mention other such specifically named like, IT, etc and unnamed awards/accomplishments....
I gotta wonder why a guy can not remain in one place with any number of great companies for any length of time, especially if he is such a fantastic genius hot shot, .....at least in his own mind. Surely one of those companies would have a top slot for a guy like that. So, Gotta Ask Why not ???
Sure seems like to me and that with neuro, and the large number of lawsuits, the CA violations, the family payment involvement, the stock price disaster, and the failed FDA approvals, along with still no mediation or desire for it, ....that there is something very very different than all the other fabulous professed success stories.
Yeah, I'm thinking JJ might be the perfect candidate for NRXP's new drug NRX-101 currently in development. NRX-101 is an investigational medicine to treat suicidal bipolar depression. I hope not, but things are getting really really strange with him.(IMO)
PW / VF, thank you both for the clarity and the link.
Greatly appreciated again and very helpful.
To me, the NRXP story seems to keep spinning wilder and almost into the world of the bizzaro and unbelievable, except its real.
Someone is pretty far out in space and I do not think its RLF.
Joe, If you click this link and scroll to the bottom where the 2/3/2022 entries are. Then click the 'exhibits' links. A lot of new information in those attachments.
https://iapps.courts.state.ny.us/webcivil/FCASeFiledDocsDetail?county_code=Mx2C3WUA2I1olvkdMyJtIA%3D%3D&txtIndexNo=Ya8t3VIslUimFDAk9KSeKA%3D%3D&showMenu=no&isPreRji=N&civilCase=elf6mvw0TRa5CzNnOKuHIQ%3D%3D
** TWO TRIAL SITES RUN BY UNQUALIFIED PEOPLE**
And Javitt claims trial wasn’t “botched” or “tainted”
In his own words he says all Regional Hospitals could have been thrown out due to issues but furthers that
Specifically site 13&14 HAD UNLICENSED PEOPLE RUNNING HIS TRIAL!!!!
Based on the blinded analysis, we immediately sent a demand letter for investigation to site 14, in coordination with the Criminal Investigation Division of FDA, whom we notified of the 100% mortality. The investigation revealed that the licensed, trained investigators who signed the 1572 were not present to render care as per our clinical trial agreement – in one case apparently because of the effects of the pandemic and in the other because of a military deployment. The site brought in a non GCP-trained unqualified investigator without notifying us. That physician decided to exercise ad hoc “triage” in order to clear ICU beds. He therefore, over the objections of the onsite study coordinator, summoned the palliative care team and convinced the families of six subjects to terminate ICU care and ventilatory support with the expected fatal consequences. The investigation into site 14’s behavior is ongoing and FDA has involved other branches of law enforcement.
THE EMAIL BELOW WAS DATED JUNE 5th, 2021!!! DAYS AFTER SUBMISSION OF SECOND EUA!! ALL THIS WAS KNOWN, NOT COMMUNICATED TO US/PUBLIC, INSTEAD WE WERE TOLD
TO EXPECT EUA!!!!
https://iapps.courts.state.ny.us/nyscef/ViewDocument?docIndex=AX5pqPZYdEoE0SG7jcTw7A==&TSPD_101_R0=08533cd43fab20001faaa963620707b5d2a35a22829226644d99d3fc2743f76a94729171ea2b9c950862cb51bb14300019f46c96877780a68ab14d8fb7e60425e449964fbc5e74e74273b95bff546163a3dc5a4788529cc7c27121fc082802db
PW, I am with you on this. There was nothing published anywhere that I read or saw about the 25 Million in new stock issued as being with restricted usage. Unless it is restricted and not publicly shared, which might or could be some type of SEC violation.
VF, you are an intelligent person, but your constant cryptic short screaming statements in all caps and without links leave a lot to the be desired in the way of good communication.
I understand this is your opinion and we all have one.
It would be fantastic if you would share the specific links to the new court filings you refer to.
I believe that the 25K can be used for most forms of 'cost of doing business' (including lawsuits against the company). We may need an CPA or lawyer chime in on that question. But ask yourself...where else would the $$ to defend themselves come from? JJ's own back pocket? NO WAY! That would happen in a 'sole proprietorship', not a formal corporation.
I DONT THINK THE PLACEMENT $ CAN BE USED FOR LEGAL COSTS
*** WOW READ NEW COURT DOCS, RIGHT AGAIN!***
You make a solid point….he points fault at others and owns none of it.
Is it a case of perhaps that there might be something there besides just a bit of arrogance ?
Especially when it comes to completely denying any responsibility for any problems .........
https://www.verywellmind.com/what-is-a-psychopath-5025217
Lol….I’m from Texas…lol. We’re a bit basic. Seriously though JJ’s arrogance will be of detriment to him unless he can self reflect and make a change. Imho
An astute observation.
Definition of comeuppance (“come up-in’s”)
: a deserved rebuke or penalty : deserts One of these days, he'll get his comeuppance for treating people so arrogantly.
https://www.merriam-webster.com/
Share price indicates JJ is getting his “come up-in’s”
I suspect (as others have posted too) that Dr JJ needs the 25M to pay for legal fees to defend NRXP in the courts. He's going for broke on this bet of his. JMHO
Yeah, I'm kind of wondering what that money will be used for too. Unless I'm way off base with my perception of JJ, I just don't see anyone willing to take a chance working with NRXP as long as he's in charge. I mean if he is willing to flagrantly violate the CA between RLFTF and NRXP what company would ever trust him to comply with a future CA. Maybe I'm wrong about JJ, after all, it's just my opinion and everybody has one, but I smell a rat and the odor seems to be coming from the direction of NRXP.
I guess we will see how all this plays out in court. Such a sad outcome so far for both RLFTF and NRXP.
choo choo, yes agreed. Suspect there are very few that
have made money on neuro, except for possibly jj and son, or a very few that bailed in the upper double digits and ran away.
Tempting to buy back in, but difficult to do so with the murky track
record thats anything but clear, and numerous serious legal open issues, very questionable PRs, and the like. Not good for a very young new company that was just hatched.
Gotta wonder if the 25 mill is for managmt/ee payroll, a lengthy legal battle, legal fees, fines, and settlements, to fund some other new unrelated project, or for a bigger legitimate cooperative aviptadil/zyesami study in conjunction with RLF.
Yes, I sold off long ago. It seems that both RLF & JJ started off putting the cart before the horse, their egos grew too big, and then the greed factor took over.
|
Followers
|
53
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
1102
|
|
Created
|
12/15/20
|
Type
|
Free
|
| Moderators ProfitScout jedijazz | |||





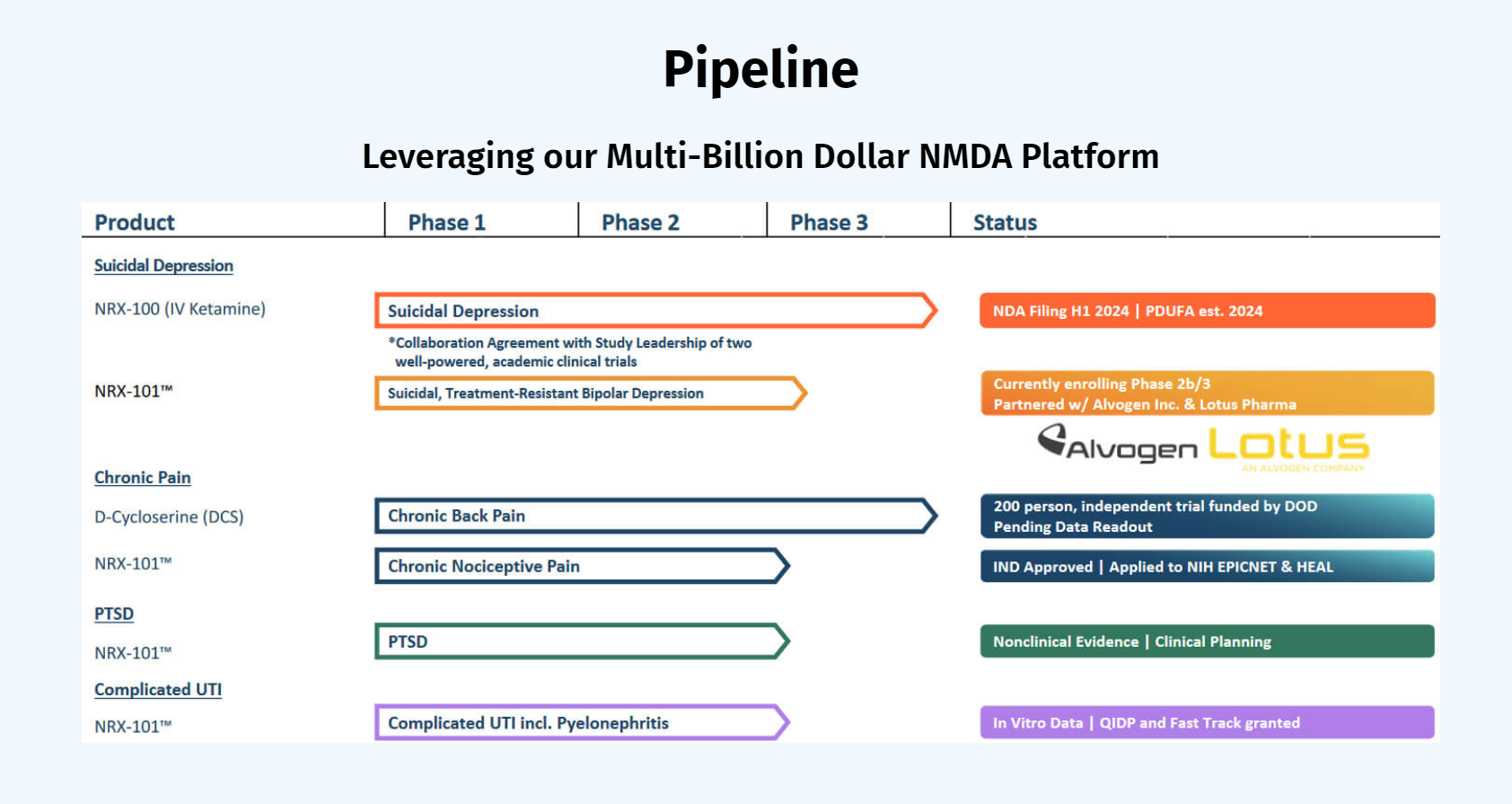
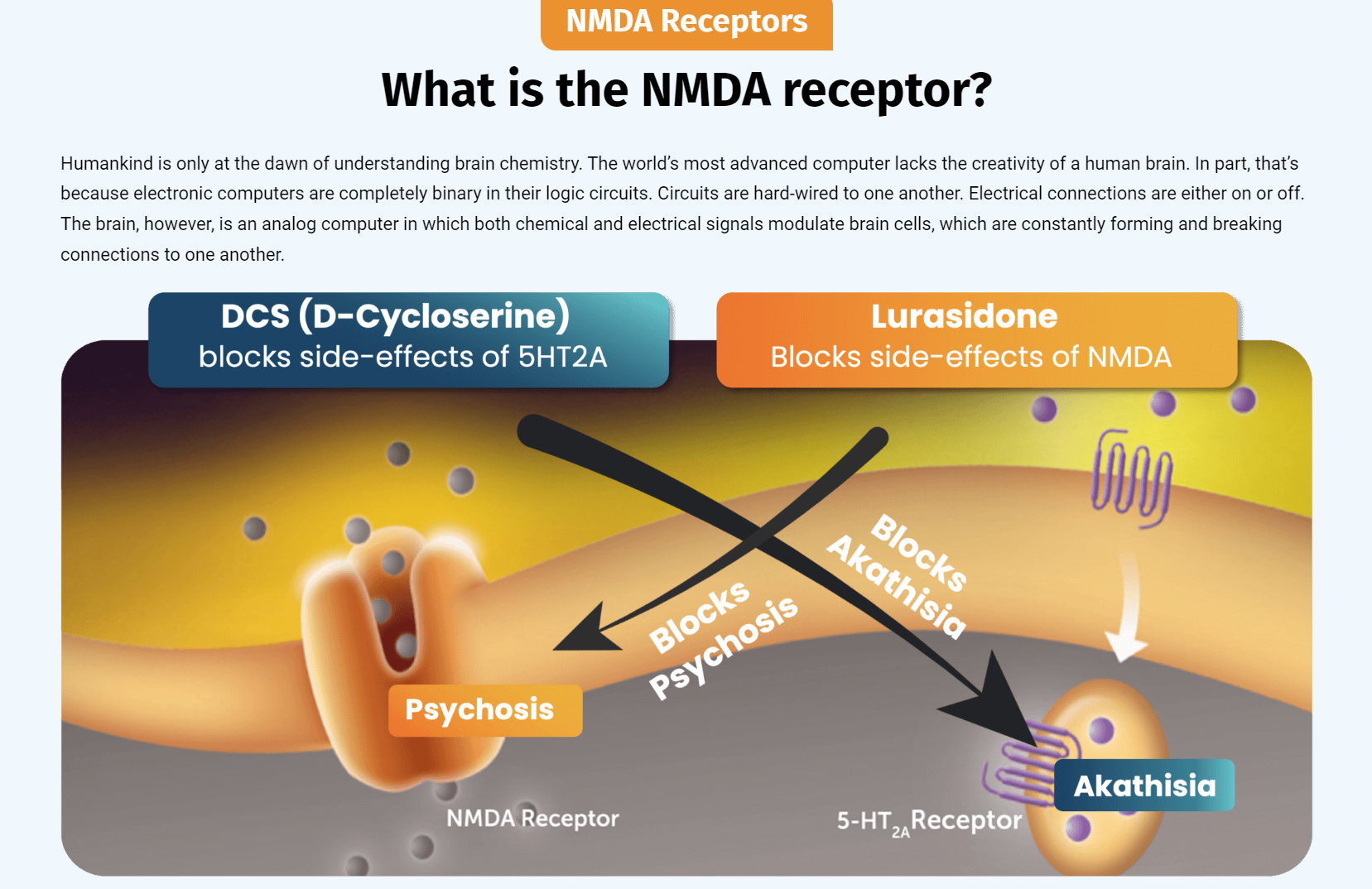







Both drugs demonstrated > 50% response for treating depression. NRX-101 demonstrated a mean 76% reduction in symptoms of akathisia compared to lurasidone that was sustained over 42 days (Effect...

New data demonstrates no impact of NRX-101 on gut or vaginal flora – considered primary causes of pseudomembranous colitis due to C difficile and vaginal yeast infections NRX-101 previously...
Formulation based on prior patents by NRx founder Achieved pH neutral formulation of ketamine, potentially enabling both intravenous (IV) and subcutaneous (SQ) administration Company expects...
Data transferred for independent statistical analysis Top-line data expected in April 2024 RADNOR, Pa., April 8, 2024 /PRNewswire/ -- NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) ("NRx...
Four potential near-term milestones, including data from two clinical trials, an NDA filing and an upcoming share dividend -- 50% reduction in corporate overhead and 25% reduction in overall net...
Four potential near-term milestones, including data from two clinical trials, an NDA filing and a share dividend RADNOR, Pa., March 28, 2024 /PRNewswire/ -- NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) ("
NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) ("NRx Pharmaceuticals", the "Company"), a clinical-stage biopharmaceutical company, today announced that it will release its fourth quarter and full year...
NRx Pharmaceuticals, Inc. (Nasdaq:NRXP) ("NRx Pharmaceuticals", the "Company"), a clinical-stage biopharmaceutical company, today announced that Dr. Jonathan Javitt, Chairman and Chief Scientist...
NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) ("NRx Pharmaceuticals", the "Company"), a clinical-stage biopharmaceutical company, today announced that it will release its fourth quarter and full year...
61.4% (56,781,354) of eligible shares voted 94.4% of votes were cast in favor of the resolution RADNOR, Pa., March 21, 2024 /PRNewswire/ -- NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) ("NRx...
NRx Pharmaceuticals Board of Directors has authorized its Chairman, CEO, and management to take all necessary steps to affect the Dividend and Royalty Rights to NRXP Shareholders and applicable...
Initiative is intended to protect shareholder value through continued compliance with Nasdaq listing rules and elimination of naked short sales positions in the Company's securities The Company is...
Company to receive first allocation of ketamine for sale by month end Partners preparing to ship IV Ketamine to full range of customers via 503a and 503b pharmacies NRx Pharmaceuticals and HOPE...
Marks a major step in the development of what could be the first drug approved for Suicidal Bipolar Depression The study database is being cleaned and locked; statistical analysis and top-line...
NRx Pharmaceuticals received approximately $1.0 million in cash from an existing investor Shares were sold at $0.38, a 26.7% premium the recent share offering, along with one common 5-year warrant...
In the news release, NRx Pharmaceuticals, Inc. Announces Pricing of $1.5 Million Underwritten Public Offering of Common Stock, issued 27-Feb-2024 by NRx Pharmaceuticals, Inc. over PR Newswire, we...
NRx Pharmaceuticals, Inc. (Nasdaq: NRXP), ("NRx Pharmaceuticals" or the "Company"), a clinical-stage biopharmaceutical company, today announced that it intends to offer to sell shares of its...
Presentation available on NRx Pharmaceuticals website at https://www.nrxpharma.com/hope-therapeutics/ KEY UPDATES ARE AS FOLLOWS: NRx management is proposing to award 50% of founding shares in...
DISCLAIMER:
Nothing in the contents transmitted on this board should be construed as an investment advisory, nor should it be used to make investment decisions.
There is no express or implied solicitation to buy or sell securities.
The author(s) may have positions in the stocks or financial relationships with the company or companies discussed and may trade in the stocks mentioned.
Readers are advised to conduct their own due diligence prior to considering buying or selling any stock. All information should be considered for information purposes only.
No stock exchange has approved or disapproved of the information here.
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
