Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
I am just guessing here. Compensation and annual bonus based upon TTM sales?
Moreover, doubling the amount of active toxin administered to a fixed region of the face would be expected to produce a higher rate of ptosis, which EOLS’ dataset did not show—a red flag that something is amiss.
it’s pretty obvious to me that double-dose Jeuveau is a ploy that EOLS engaged not to further its own business, but rather to create confusion in the marketplace about Daxxify and thereby impede Daxxify’s commercial uptake. On this score alone, EOLS may have been successful in accomplishing what it hoped to accomplish, but the impeding of Daxxify’s uptake will be transient. A year from now, nobody will be talking about double-dose Jeuveau.
What did PACB gain by "hiding" the revenue problem for one quarter?
Re: Double-dose Jeuveau
Thanks for the correction. However, the fact that EOLS published the double-dose Jeuveau data in a little known journal doesn’t change my contention that the dataset has been doctored.
Jeuveau is simply a Korean knockoff of Botox, and there is no mechanistic explanation for why it ought to have a longer duration than Botox or other Botox knockoffs. Reducing the relative amount of diluent, which EOLS did the double-dose Jeuveau study, is something that Botox injectors have experimented with for decades without being able to show a material benefit in duration.
Moreover, doubling the amount of active toxin administered to a fixed region of the face would be expected to produce a higher rate of ptosis, which EOLS’ dataset did not show—a red flag that something is amiss.
IF EOLS’ double-dose Jeuveau data were real, it would be a big breakthrough that would comprise the cornerstone of EOLS’ go-to-market strategy. Instead, EOLS treats the double-dose data as an afterthought and acknowledges that few of its customers will want it.
All told, it’s pretty obvious to me that double-dose Jeuveau is a ploy that EOLS engaged not to further its own business, but rather to create confusion in the marketplace about Daxxify and thereby impede Daxxify’s commercial uptake. On this score alone, EOLS may have been successful in accomplishing what it hoped to accomplish, but the impeding of Daxxify’s uptake will be transient. A year from now, nobody will be talking about double-dose Jeuveau.
Based upon the way PACB shares acted, Q4 numbers were massaged heavily to hide the drop off in sales. Don't understand why ILMN wanted PACB years ago and why FTC objected. NP.
The remaining 93% is the CVD market for which no generic has an FDA-approved ANDA or sANDA.
The total triglyceride-lowering market is ~7% of the total market that Amarin is aiming at. The remaining 93% is the CVD market for which no generic has an FDA-approved ANDA or sANDA.
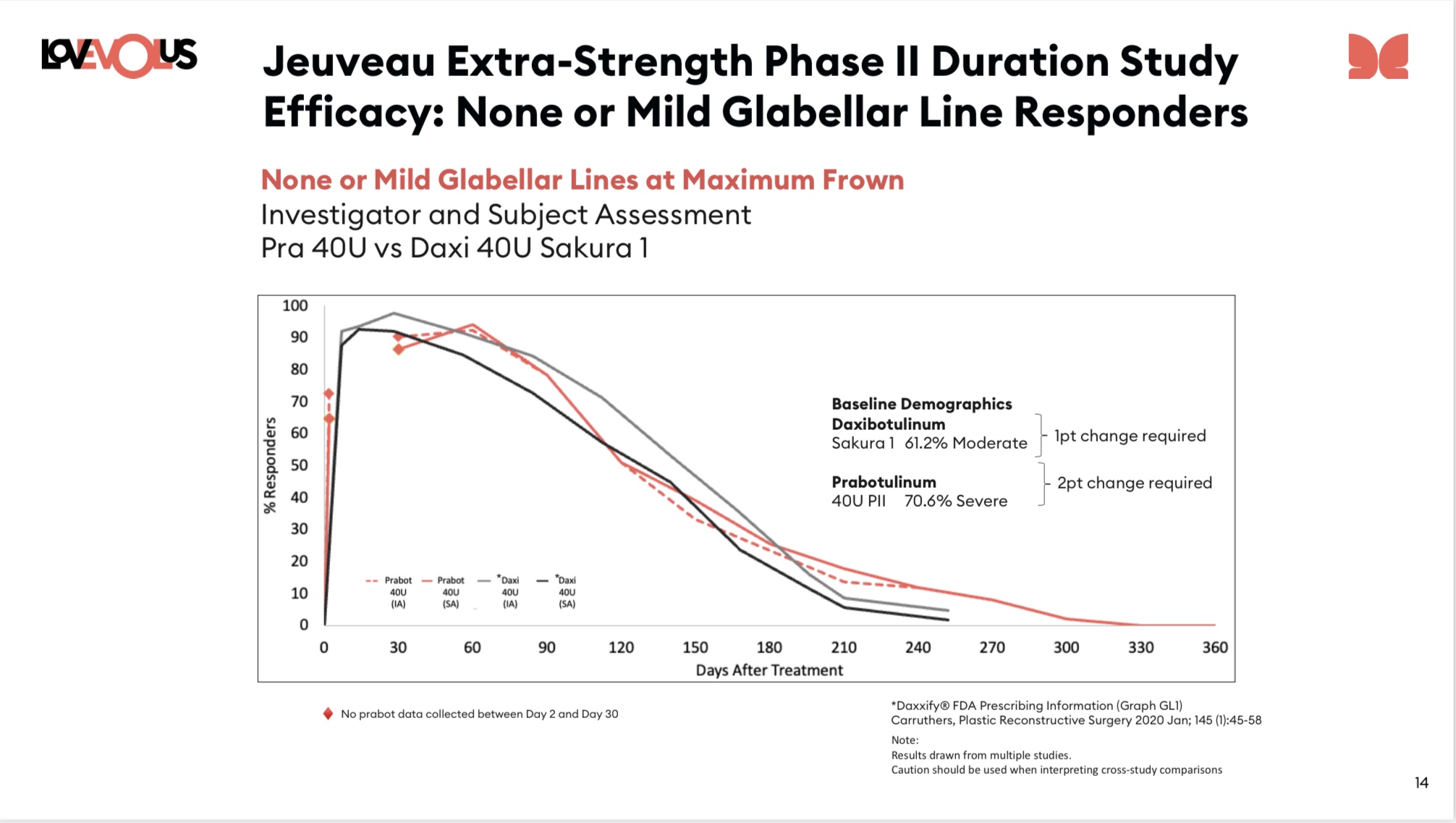
EOLS - Here are the pictures from the paper. I've now done more than a 'quick check' for *one* of the Jeuveau 40 unit curves (the Investigator curve - but not the Patient curve).
For the investigator curves given below (left curve) it is, as best it's possible for me to measure, identical to the 23Q3 version of the same (investigator) curve out through 120 days (which is also the median - about 15 days later than 20 unit dose of Jeuveau, and about 30 days later than botox dose and 23 days earlier than Sakura 1). But from there on the formally published paper is meaningfully lower than the 23Q3 Jeuveau 40 unit dose curve, although still above the Sakura 1 dose from day 190 on.
Essentially: Sakura 1 has a better median by about 23 days (but beware the caveats below), but the last quarter of the population seems to be about the same for Daxxy and Jeuveau 40 unit dose
And the caveats:
a) The comparison to Sakura is a cross trial comparison... inherently noisy. See next point.
b) The Jeuveau 40 unit population is a meaningfully tougher population in which to get no-or-mild-lines than either of the Sakura populations (Jeuveau is 70% Severe, Sakura closer to only 40% Severe).
with patient’s insurance permitting Amarin to maintain a 57% market share against 5-6 generics who have no sANDA approved by FDA for Vascepa treatment of the much broader CVD indication.
And some corrections for which I ran out of edit time:
A) A link to the EOLS paper that, hopefully, won’t time out.
B) I mistakenly said Figures 8 and 9 (the EOLS figures in the above link that are comparable to Sakura figures) were the last figures. They aren’t. There are three more figures in the EOLS paper (10, 11 and 12.. obviously). Another ‘my bad’, although not really pertinent to any argument. This is just an administrative clean up by me. C’est la vie.
EOLS and RVNC
In other words, EOLS declined to publish what they said they would, which is consistent with what I posited in #msg-173201637.
To re-iterate: Double-dose Jeuveau is a gimmick; it won't move the needle for Jeuveau to any material degree.
In other words, EOLS declined to publish what they said they would, which is consistent with what I posited in #msg-173201637.
I stand by everything I said in the above-linked post. To re-iterate: Double-dose Jeuveau is a gimmick; it won't move the needle for Jeuveau to any material degree.
Right, EOLS is using an different and easier metric to fool uninformed people. Thanks for pointing that out.
So far C has been right on his calls re AAPL and NVDA: Buy at a lower price(whatever that price is), hold and do not sell(unless you bought at the all time high and need a tax loss). I will abide by that recommendation.
He stuck a Pitchfork in AMRN last year: “Too speculative, stay away, do not buy.” He may now be missing a recovery with Amarin’s Vascepa now approved and reimbursed in all Canadian provinces; approved in China, Australia and New Zealand; approved by EU(EMEA) with reimbursement in UK, SPAIN, and being negotiated in remaining countries of EU; FDA approved in U.S., with patient’s insurance permitting Amarin to maintain a 57% market share against 5-6 generics who have no sANDA approved by FDA for Vascepa treatment of the much broader CVD indication. Take a look at the posters/abstracts featuring Vascepa that were presented at ACC earlier this month.
Ahhh Thx . I had pitched UNCY some time ago . Reduced my position today as stock sold off on almost 3 X normal vol , after a presentation .
Not something I like to see .
Will hold remainder thru to data readout in June.
Kiwi
Pigs get fed, hogs get slaughtered
This proverb teaches you to be satisfied with enough because being too greedy or too ambitious will be your ruin.
Just as the pig that has been too greedy in eating, it has gotten big and fat and becomes a candidate for slaughter at hog-killing time. The pig that has eaten just enough will continue to be fed.
(. Ala bulls make $ but pigs get slaughtered ...or something to that effect )
I wonder if she’ll adhere to privacy agreements customers initially signed up for after it becomes a private company?
Part of their sales pitch was that you could agree to share your genetic data anonymously with scientists in order to advance research. In my opinion, their lax security practices breached the privacy assurances that explicitly went along with that altruistic agreement.
Data mining is great for medical research, but is even more valuable to advertisers willing to sell anything under the sun if they can target the right (or most vulnerable) audience.
RZLT. ..follow up to my Feb 20th pitch
My guess is that they have completed the P 2 DME trial and will publish results in May 2024 .
I'm up over 100% on this position so have sold half (. Ala bulls make $ but pigs get slaughtered ...or something to that effect )
Will hold the remaining 50% thru read out .
Kiwi
Dew. thx for the update re Parkinson's
Kiwi
Then why didn't RVNC use 3 steps going from NONE to SERVERE?
You've really worked yourself into a corner. RVNC has 6 months with 2 points of improvement. EOLS has 6 months with 1 point improvement. I know you got burned on RVNC but it's a shame you've turned into a RVNCQ clown who jumps on every opportunity to spread FUD.
VIR adds two luminaries to BoD:
https://www.businesswire.com/news/home/20240418098064/en
The capsule bios in the PR speak for themselves.
vinny
For some reason you don't seem to think it is.
CERE—(being acquired by ABBV)—reports phase-3 Parkinson’s data:
https://www.globenewswire.com/news-release/2024/04/18/2865216/0/en/Cerevel-Therapeutics-Announces-Positive-Topline-Results-for-Tavapadon-in-Phase-3-Adjunctive-Trial-for-People-Living-with-Parkinson-s-Disease.html
23andMe probably getting acquired by CEO/founder, Anne Wojcicki:
https://www.globenewswire.com/news-release/2024/04/18/2865332/0/en/23andMe-announces-CEO-s-intention-to-pursue-a-potential-take-private.html
https://www.sec.gov/Archives/edgar/data/1804591/000134100424000072/sc13da1.htm
On April 13, 2024, Ms. Wojcicki notified members of the special committee (the “Special Committee”) of the Board that she is considering making a proposal to acquire the Issuer in a potential go-private transaction. Ms. Wojcicki indicated that she was working with advisors and intended to begin speaking to potential partners and financing sources. Ms. Wojcicki stated that any proposal by her would be conditioned irrevocably upon the approval of the Special Committee and a majority of the unaffiliated stockholders of the Issuer. Ms. Wojcicki also indicated that she wishes to maintain control of the Issuer and, therefore, will not be willing to support any alternative transaction.
You seem to think going from None to Moderate (2 Steps) is a stricter parameter than going from None to Mild.
How's revance handling the news?
Vinny
Right, EOLS is using an different and easier metric to fool uninformed people
The GLS system of testing comprises of 4 numbers. The numbers correspond to a word, not a scale.
0= NO Lines
1= Mild Lines
EOLS: looks like the market is not as dumb as jbog 🤣
https://stocktwits.com/pablosplace13 great dd of industry insider. In beauty, i believe Jeuveau will persist.
He knew before filings that Deerfield was unwinding a big position in summer/fall 23 and that EOLS would beat earnings
edcool and Vinny
The GLS system of testing comprises of 4 numbers. The numbers correspond to a word, not a scale.
0= NO Lines
1= Mild Lines
2= Moderate Lines
4= Severe Lines
So isn't measuring to MILD LINES Versus Moderate Lines A higher hill to climb?
Do your homework
Price them both the same and office makes $34 more profit with Daxxify.
It's a 1 point improvement and 150 patients. It's a farce. As a RVNC shareholder there are plenty of things to worry about but double dose Jeuveau is near the bottom of the list.
They could compare. Based on EOLS using a weaker metric, Daxxify would be preferred.
Vin
And who's eyes beside the customer could define the difference between mild and moderate. What is acceptable.
Data showed duration effect of 26 weeks, or 6 months, based on a ≥ 1-point Glabellar Line Scale (GLS) improvement and return to baseline value using the Global Aesthetic Improvement Scale
40 units of Jeuveau at $3.60/unit = best price: $144 COGS
40 units of Daxxify at $2.75/unit = best price: $110 COGS
Price them both the same and office makes $34 more profit with Daxxify.
I also feel strongly that 40U Daxxify will last longer than 40U Jeuveau.
Vin
After the fiasco with Revance I won't accept any beauty results from a clinical trial whereas the results are solely the decision of the patient. Revances #1 mistake was the promise of superiority.
If they promised little and delivered more they'd be king of the hill.
VNDA - the official version of the 'poison pill' has been posted...
https://capedge.com/filing/1347178/0001193125-24-099263/VNDA-8A12B
Is anyone able to make sense of this legalese mumbo jumbo? Ahhh, the promiscuity of the legal profession...
Evolus Announces Publication of Safety and Duration Data from Phase 2 Study for "Extra-Strength" 40U Formulation of Jeuveau®
Business Wire
Wed, Apr 17, 2024, 4:05 PM EDT9 min read
In This Article:
EOLS
-3.40%
The article published in Aesthetic Surgery Journal supports safety and duration of effect for temporary improvement in the appearance of moderate to severe glabellar lines presented at the 2023 ASDS Annual Meeting
Data showed duration effect of 26 weeks, or 6 months, based on a ≥ 1-point Glabellar Line Scale (GLS) improvement and return to baseline value using the Global Aesthetic Improvement Scale
NEWPORT BEACH, Calif., April 17, 2024--(BUSINESS WIRE)--Evolus, Inc. (NASDAQ: EOLS), a performance beauty company with a focus on building an aesthetic portfolio of consumer brands, today announced that the Aesthetic Surgery Journal has published the safety and duration of effect results from the Phase 2 study of 40U Jeuveau® (prabotulinumtoxinA-xvfs) for the treatment of moderate to severe glabellar lines in adult patients.
The "extra-strength" glabellar line study is a multicenter, double-blind, randomized, Phase 2 trial following 150 patients for up to 12 months or until the patient loses their correction. The study has three arms: Jeuveau® Extra-Strength 40U and two active controls, BOTOX® 20U and Jeuveau® 20U.
Efficacy results demonstrated 26 weeks, or 6 months duration across the multiple metrics presented, including the time it took for patients to return to their baseline GLS score after their treatment, the duration of effect for a patient with at least a one-point GLS improvement, and the time it took a patient to return to their baseline using the Global Aesthetic Improvement Scale. The safety profile was similar across all three arms and overall, 88.9% of adverse events were rated as mild and no serious adverse events were identified.
"This publication provides the details of the study design along with the efficacy and safety data from the Phase 2 Jeuveau extra-strength study and makes it accessible to all," said Dr. Rui Avelar, MD, Chief Medical Officer and Head of R&D of Evolus. "The study confirms the correlation between increasing dose and increasing duration, while maintaining a similar safety profile, with the majority of adverse events rated as mild."
Thanks, Doc (eom).
I believe its entirely or almost entirely due to the actual weight loss. The same phenomenon was seen in the bariatric surgery literature with similar results and seen across the board for different surgical techniques (slight exception with Lap Band showing less weight loss and less AHI reduction). Blinding is of course difficult with bariatric surgery and many of these are uncontrolled studies. Other weight loss drugs have also seen good OSA data. Though results were pretty much a foregone conclusion, the main positive for the GLP-1's is that this data will help to get insurance to cover patients who do not have DM/hyperglycemia
Here's a review
https://www.sciencedirect.com/science/article/pii/S217351151630046X?via%3Dihub
As you stated:
If the (placebo-adjusted) OSA benefit is seen early in the two one-year trials—i.e. before patients have lost a significant amount of weight—we may surmise that Zepbound is having a direct effect on symptoms, and people will want to know what the MoA is. On the other hand, if the OSA benefit kicks in mostly during the latter months of these trials, we may presume that the OSA benefit stems, at least in large part, from the weight loss.
It's not a chicken-and-egg question, IMO. After we see the full dataset, we can form reasonable suppositions as to whether Zepbound has a direct effect on OCA, or merely an indirect one. For the direct effect to have credibility, there has to be a plausible MoA (i.e. something other than weight loss).
How_Much_Does Weight Loss_Help_with Sleep Apnea?
https://www.healthline.com/health/sleep-apnea/sleep-apnea-weight-loss
Weight loss tends to reduce problems with sleep apnea (and snoring,) so I’m not sure how you can disentangle the two questions.
I.e., which came first, the chicken or the egg? If weight loss reduces sleep apnea, and weight loss drugs reduce weight, are they specifically reducing sleep apnea or is that a beneficial side effect?
LLY/sleep-apnea—I couldn't tell from the article whether OSA symptoms were reduced concomitant with weight loss -which would be expected - or were a direct result of the exposure to the drug?
Net bad news for the stock need for a booster would have far outweighed any new barrier to competing vaccines that this creates
Good news/bad news for GSK’s Shingrix:
https://www.gsk.com/en-gb/media/press-releases/new-long-term-data-show-shingrix-continues-to-provide-high-protection-against-shingles-in-adults-aged-50-and-over-for-more-than-a-decade/#
Shingrix’s protection is longer lasting than most people expected. Corollary: It’s unlikely that patients will need booster shots (beyond the initial two-dose regimen).
|
Followers
|
1524
|
Posters
|
|
|
Posts (Today)
|
10
|
Posts (Total)
|
252422
|
|
Created
|
11/14/02
|
Type
|
Free
|
| Moderator DewDiligence | |||
| Assistants Biowatch | |||
Biotech Values is a forum for discussing
all facets of biotech investing with an emphasis
on fundamental analysis and avoiding scams.
We generally do not discuss microcap stocks.
Compilations
Biotech Buyouts/Premiums
Oral Weight-Loss Drugs
Biotech-Newbie Misconceptions (see Reply chain)
Biotech Abbreviations and Acronyms
'Way Back' Archive (Greatest Hits 2003-2009)
|
Posts Today
|
10
|
|
Posts (Total)
|
252422
|
|
Posters
|
|
|
Moderator
|
|
|
Assistants
|
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
