Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
haha - I think a Robot removed me as Mod due to not enough posts. I've asked to be put back in, to take care of iBox and post the Quarterlies, etc. Good luck everybody.
12-2-20 Qtly CC-Transcript, PR(qe10-31-20), Avid Revs History Table
*Revs Guidance (FY’21 fye 4-30-21): $84-88M. 10-31-20 Backlog=$67M
*Cash: 10-31-20: $35.7M
*As of 11-23-20: 56,726,334 shares o/s
*10Q/10-31-20 iss. 12-2-20: https://tinyurl.com/y3ournzr
*10K/4-30-20 iss. 6-30-20: https://tinyurl.com/yak25nco
*Avid Total Revs May03-Jul20: $455.3M
*Avid’s website: https://avidbio.com (A/o 4-30-20, 222 full-time & 5 part-time emps)
This large post has 4 sections:
I. 12-2-20 Qtly. Earnings Conf. Call TRANSCRIPT (FY21/Q2 qe 10-31-20)
II. 12-2-20 CDMO Press Release: Q2/FY21 Earnings & Developments
IV. Updated Table of Avid Revenues By Quarter (May’06-Current)
III. Updated O/S Shares History Table – 2006-curr.
…Recall: Avid’s FY runs May-Apr, so FY’21 = May’20-Apr’21.
TRANSCRIPT 12-2-20 FY21/Q2 Earnings Conf. Call (qe 10-31-20):
Nick Green(CEO), Daniel Hart(CFO), Tim Compton(CCO)
Q&A Analysts:
Matt Hewitt - Craig-Hallum Capital
Paul Knight – KeyBanc
Jacob Johnson – Stephens Inc.
Link to webcast replay: http://ir.avidbio.com/events-and-presentations => https://edge.media-server.com/mmc/p/6XXXeurd
CC TRANSCRIPT EXTRACTS – PREPARED REMARKS BY CEO NICK GREEN:
CEO NICK GREEN OPENING:
Thank you, Tim and thank you to everyone who has dialed in and to those who are participating today via webcast. I am pleased to report that Q2 was highly productive for Avid. From a financial perspective, we again beat revenue expectations and had a strong showing in other key financial metrics. In business development, we added a new process development customer as well as another new manufacturing project from an existing customer. The Company had a strong operational performance during the quarter during which we successfully completed our annual maintenance program. More importantly, we finalized our review of expansion options and we look forward to proceeding with this important effort. Tim and I will provide additional details on business development and operations following an overview of our second quarter financial results. And for that, I will turn the call over to Dan.”
CEO NICK GREEN - AFTER HART/COMPTON OPENING:
During Q2, Avid's operations continue to manufacture to plan. As we reported last quarter, we initiated our scheduled annual preventative maintenance shutdown at the end of July. And this was brought to a successful conclusion during the period. As I indicated last quarter, one of my first target was to review the Company's expansion plans as well as other ancillary requirements ahead of making our final decision with respect to the best path forward. We have now completed this review, and I am happy to report that the Company is already moving forward with our expansion using a phased approach. We recently developed plans for the 2-phase expansion of our Myford facility. The first phase expands the production capacity of our existing Myford North facility by the addition of a second downstream processing suite. The second phase further expands the capacity through the build out of a second manufacturing train including both upstream and downstream processing suites within Myford South.
Due to an anticipated increase in customer demand, we have commenced the first phase of expansion, which we estimate will take approximately 12 to 15 months to complete at an estimated cost of approximately $15 million. We expect that the first phase of expansion could increase our annual revenue generating capacity by up to $50 million, bringing the combined annual revenue generating capacity of our Franklin and Myford North facilities up to $170 million. The decision to commence the second phase of expansion will be dictated by revenue growth and projected customer demand. Based on preliminary conceptual plans, we estimate that the Myford South expansion will take 18 to 24 months to complete at a cost of approximately $45 million to $55 million. We estimate that the addition of the future Myford South facility will increase our annual revenue generating capacity by up to $100 million. To complete these anticipated expansions, we expect to raise external capital at the appropriate time. Accessing the form of capital that we determined is the most appropriate, considering the markets available to us and their respective cost of capital. In closing, we are happy to report a very positive quarter during which we achieved strong revenues and margins beating estimates for both revenue and earnings per share as well as generating operating cash flow and income from our operations. Further increasing our backlog from $60 million to $67 million, while delivering the above is testament to the excellent work of our business development team and the many people behind the scenes to support them. As a result, we have been able to increase guidance and also initiate our expansion plans with the phased approach, which we believe provides capacity, well aligned with demand, while being cognizant of both time and cost. This concludes my prepared remarks for today, and we can now open up the call for questions.”
CEO NICK GREEN - CLOSING (After Q&A):
“Thank you to everyone participating on today's call. In closing, I'd like to thank all our employees at Avid. Not only have they been effective and efficient in improving the business performance, but they continue to do this in the challenging environment as a result of COVID-19. Avid’s success is dependent upon our incredible employees. And I wish to thank them for their continued efforts in adapting to the challenges which are impacting both their professional, but also their personal lives. Thank you again for participating on today's call and for your continued support of Avid Bioservices.”
READ the entire CC Transcript here:
https://www.fool.com/earnings/call-transcripts/2020/12/02/avid-bioservices-inc-cdmo-q2-2021-earnings-call-tr/
= = = = = = = = = = = = = = = = = = = = = == = = =
12-2-20/PR: Avid Bioservices Reports Financial Results for Q2/FY21 Ended Oct. 31, 2020, and Recent Developments
https://ir.avidbio.com/news-releases/news-release-details/avid-bioservices-reports-financial-results-second-quarter-1
-- Recorded Second Quarter Revenue of $21.1 Million
-- Signed $28 Million in Business Orders and Ended the Quarter with a Backlog of $67 Million
-- Initiated First Phase of Myford Expansion
-- Increasing Revenue Guidance for Fiscal 2021 to $84 to $88 Million
TUSTIN, Dec. 2, 2020: Avid Bioservices, Inc. (NASDAQ:CDMO/CDMOP), a dedicated biologics contract development and manufacturing organization (CDMO) working to improve patient lives by providing high quality development and manufacturing services to biotechnology and pharmaceutical companies, today announced financial results for the second quarter and first six months of fiscal 2021, ended October 31, 2020.
HIGHLIGHTS SINCE JULY 31, 2020
“During the second quarter, we recorded strong revenues, expanded our customer base and project pipeline, and advanced the company’s expansion plans,” stated Nicholas Green, president and chief executive officer of Avid Bioservices. “Driven by growth in customer demand, the company achieved higher-than-expected revenues and margins, and generated operating cash flow and income from operations during the period. In consideration of these results combined with our substantial backlog and our visibility into customer demand, we are raising revenue guidance for fiscal 2021 from between $76 and $81 million to between $84 and $88 million.
“On the business development front, our team continues to execute, signing new business orders and project expansion orders with existing customers for $28 million during the quarter and increasing backlog to $67 million, our highest level since becoming a pure-play CDMO.
“With respect to operations, we have completed a comprehensive review of our options and have initiated a phased approach plan for expansion. Phase 1, which is currently underway, is focused on the streamlining of existing facilities. We are confident that this work will allow us to optimize capacity, increase revenue, minimize near-term expense, and best align our expansion with growth in customer demand.
“And finally, it is important to note that we continue to execute our business and achieve growth without interruption to our operations as a result of the COVID-19 pandemic. This resilience is due largely to the diligence and dedication of our employees. Despite these challenging times, Avid’s incredible workforce remains committed to excellence to ensure the highest quality product for our clients.”
FINANCIAL HIGHLIGHTS AND GUIDANCE
* The company is increasing revenue guidance for the full fiscal year 2021 from between $76 million and $81 million to between $84 million and $88 million.
* Revenues for the second quarter of fiscal 2021 were $21.1 million, a 15% increase compared to revenues of $18.3 million recorded during the second quarter of fiscal 2020. The year-over-year increase in revenue was primarily attributable to the growth in the number and scope of in-process and/or completed manufacturing runs during the quarter. In addition, the increase in manufacturing revenues included the recognition of $1.7 million from changes in estimated variable revenue consideration as a result of completing performance obligations for certain projects during the quarter, therefore increasing revenue recognized for those projects during the period. For the first six months of fiscal 2021, revenues were $46.5 million, a 38% increase as compared to revenues of $33.6 million in the prior year period. The increase in revenues can be attributed to a $13.6 million increase in manufacturing revenues primarily due to an increase in the number and scope of in-process and/or completed manufacturing runs during the first six months of fiscal 2021, partially offset by a $0.7 million decrease in process development revenues.
* As of October 31, 2020, revenue backlog was $67 million, an increase of 12% compared to $60 million at the end of the first quarter of fiscal 2021, and an increase of 3% compare to $65 million at the end of last fiscal year. The company expects to recognize the majority of this backlog over the next twelve months.
* Gross margin for the second quarter of fiscal 2021 was 30%, a significant increase compared to a gross margin of 18% for the second quarter of fiscal 2020. The increase in gross margin for the 2021 quarter was primarily attributable to the growth in manufacturing revenues, including the $1.7 million in additional manufacturing revenue recognized, as previously discussed. Excluding the $1.7 million in additional variable revenue consideration, gross margin for the second quarter was approximately 24%. Gross margin for the first six months of fiscal 2021 was 32%, a significant increase compared to 13% in the prior year period. This increase was also primarily due to the growth in manufacturing revenues.
* Selling, general and administrative expenses (“SG&A”) for the second quarter of fiscal 2021 were $4.2 million, an increase compared to $3.5 million recorded for the second quarter of fiscal 2020. The increase during the 2021 quarter was due primarily to increases in payroll related costs, including stock-based compensation. For the first six months of fiscal 2021, SG&A expenses were $8 million, consistent with $8 million for the prior year period.
* For the second quarter of fiscal 2021, the company recorded a consolidated net income attributable to common stockholders of $0.8 million or $0.01 per basic and diluted share, as compared to a consolidated net loss attributable to common stockholders of $1.9 million or $0.03 per basic and diluted share, for the second quarter of fiscal 2020. For the first six months of fiscal 2021, the company recorded a consolidated net income attributable to common stockholders of $4.5 million or $0.08 per basic and diluted share, compared to a consolidated net loss attributable to common stockholders of $6.1 million or $0.11 per basic and diluted share, for fiscal 2020.
* Avid reported $35.7 million in cash and cash equivalents as of October 31, 2020, an increase of $7.5 million compared to cash of $28.2 million at the end of the first quarter of fiscal 2021, and consistent with $36.3 million in cash as of the prior fiscal year ended April 30, 2020. The company also generated cash flows from operating activities of $8.1 million during the six months ended October 31. 2020.
More detailed financial information and analysis may be found in Avid Bioservices’ Quarterly Report on Form 10-Q, which will be filed with the SEC today. [https://tinyurl.com/y3ournzr ]
RECENT CORPORATE DEVELOPMENTS
* Signed orders for $28 million during the quarter with new and existing customers, driving Avid’s backlog to its highest level since transitioning to a dedicated CDMO.
* Developed plans for a two-phased expansion of our Myford facility. The first phase, which has commenced, expands the production capacity of our existing Myford North facility by adding a second downstream processing suite. The second phase, the timing of which will be dictated by revenue growth and projected customer demand, will further expand capacity through the build out of a second manufacturing train, including both upstream and downstream processing suites within Myford South.
* The company estimates the first phase will take approximately 12 to 15 months to complete at an estimated cost of approximately $15 million and may increase the company’s annual revenue generating capacity by up to $50 million, bringing the combined annual revenue generating capacity of our Franklin and Myford North facilities to up to $170 million.
CONFERENCE CALL
Avid will host a conference call and webcast this afternoon, December 2, 2020, at 4:30 PM EST (1:30 PM PST).
To listen to the conference call, please dial (877) 312-5443 or (253) 237-1126 and request the Avid Bioservices conference call. To listen to the live webcast, or access the archived webcast, please visit: https://ir.avidbio.com/investor-events
ABOUT AVID BIOSERVICES, INC.
Avid Bioservices is a dedicated contract development and manufacturing organization (CDMO) focused on development and CGMP manufacturing of biopharmaceutical drug substances derived from mammalian cell culture. The company provides a comprehensive range of process development, CGMP clinical and commercial manufacturing services for the biotechnology and biopharmaceutical industries. With 27 years of experience producing monoclonal antibodies and recombinant proteins, Avid's services include CGMP clinical and commercial drug substance manufacturing, bulk packaging, release and stability testing and regulatory submissions support. For early-stage programs the company provides a variety of process development activities, including upstream and downstream development and optimization, analytical methods development, testing and characterization. The scope of our services ranges from standalone process development projects to full development and manufacturing programs through commercialization. www.avidbio.com.
Forward-Looking *SNIP*
AVID BIOSERVICES, INC.
CONDENSED CONSOLIDATED STATEMENTS OF OPERATIONS AND COMPREHENSIVE INCOME (LOSS)
(Unaudited) (In thousands, except per share information)
Three Months Ended
October 31, Six Months Ended
October 31,
2020 2019 2020 2019
Revenues $ 21,064 $ 18,313 $ 46,456 $ 33,567
Cost of revenues 14,646 14,953 31,494 29,121
Gross profit
6,418 3,360 14,962 4,446
Operating expenses:
Selling, general and administrative 4,166 3,534 7,991 7,993
Loss on lease termination — 355 — 355
Total operating expenses 4,166 3,889 7,991 8,348
Operating income (loss) 2,252 (529 ) 6,971 (3,902 )
Interest and other income, net 32 99 43 308
Net income (loss) $ 2,284 $ (430 ) $ 7,014 $ (3,594 )
Comprehensive income (loss) $ 2,284 $ (430 ) $ 7,014 $ (3,594 )
Series E preferred stock accumulated dividends (1,442 ) (1,442 ) (2,523 ) (2,523 )
Net income (loss) attributable to common stockholders $ 842 $ (1,872 ) $ 4,491 $ (6,117 )
Net income (loss) per share attributable to common stockholders:
Basic $ 0.01 $ (0.03 ) $ 0.08 $ (0.11 )
Diluted $ 0.01 $ (0.03 ) $ 0.08 $ (0.11 )
Weighted average common shares outstanding:
Basic 56,660 56,253 56,592 56,210
Diluted 57,248 56,253 57,073 56,210
AVID BIOSERVICES, INC.
CONDENSED CONSOLIDATED BALANCE SHEETS
(Unaudited) (In thousands, except par value)
October 31,
2020 April 30,
2020
ASSETS
Current assets:
Cash and cash equivalents $ 35,664 $ 36,262
Accounts receivable 11,568 8,606
Contract assets 5,343 3,300
Inventory 9,723 10,883
Prepaid expenses 828 712
Total current assets 63,126 59,763
Property and equipment, net 30,232 27,105
Operating lease right-of-use assets 19,408 20,100
Restricted cash 350 350
Other assets 479 302
Total assets $ 113,595 $ 107,620
LIABILITIES AND STOCKHOLDERS’ EQUITY
Current liabilities:
Accounts payable $ 5,654 $ 5,926
Accrued payroll and related costs 4,850 3,019
Contract liabilities 31,450 29,120
Current portion of operating lease liabilities 1,326 1,228
Note payable — 4,379
Other current liabilities 379 808
Total current liabilities 43,659 44,480
Operating lease liabilities, less current portion 20,550 21,244
Total liabilities 64,209 65,724
Commitments and contingencies
Stockholders’ equity:
Preferred stock, $0.001 par value; 5,000 shares authorized; 1,648 shares issued and outstanding at October 31, 2020 and April 30, 2020, respectively 2 2
Common stock, $0.001 par value; 150,000 shares authorized; 56,722 and 56,483 shares issued and outstanding at October 31, 2020 and April 30, 2020, respectively 57 56
Additional paid-in capital 613,384 612,909
Accumulated deficit (564,057 ) (571,071 )
Total stockholders’ equity 49,386 41,896
Total liabilities and stockholders’ equity $ 113,595 $ 107,620
CONTACTS:
• Stephanie Diaz (Investors) Vida Strategic Partners 415-675-7401 sdiaz@vidasp.com
• Tim Brons (Media) Vida Strategic Partners 415-675-7402 tbrons@vidasp.com
- - - - - - - -
From 10-Q header: “As of Nov 23, 2020, there were 56,726,334 shares outstanding.”
- - - - - - - - - - - - - - - - -
Latest 10Q 10-31-20 iss. 12-2-20 https://tinyurl.com/y3ournzr (Cash 10-31-20=$35.7mm)
Latest 10K 4-30-20 iss. 6-30-20 https://tinyurl.com/yak25nco (Cash 4-30-20=$36.3mm)
Latest DEF14A/Proxy iss. 8-27-20 (re: 10-20-20 ASM): https://tinyurl.com/y46ga9el
ALL SEC filings for PPHM: http://tinyurl.com/6d4jw8
10-K: “As of 4-30-20, we employed 222 full-time & 5 part-time emps.” (2018: 185/1, 2019: 211/4)
= = = = = = = = = = = = = = = = = = = = = = = = = = = =
Updated PPHM REVS-BY-QTR TABLE, now thru FY21Q2(qe 10-31-20), per the 10Q issued 12-2-20.
• Total Avid Revs since May’03: $455.3M
• 12-2-20: FY'21 (May'20-Apr'21) Avid revs guidance $84-88M (committed B/L=$67 at 10-31-20).
• Inventories at 10-31-20 total $9.7M, UP from $9.6M at 7-31-20.
Avid’s website: http://www.avidbio.com
AVID GROSS PROFITABILITY BY QTR: CONTRACT
QTR (1000’s) Rev$ COGS$ Prof$ GP% INVEN$ LIABILITIES*
FY13Q1 7-31-12 4,135 2,024 2,111 51% 5,744 16,280
FY13Q2 10-31-12 6,061 3,703 2,358 39% 5,426 14,721
FY13Q3 1-31-13 6,961 3,651 3,310 47% 4,635 11,790
FY13Q4 4-30-13 4,176 3,217 959 23% 4,339 12,230
FY14Q1 7-31-13 4,581 2,670 1,911 42% 5,679 12,692
FY14Q2 10-31-13 7,354 4,195 3,159 43% 4,033 11,126
FY14Q3 1-31-14 3,885 2,416 1,469 38% 5,224 12,975
FY14Q4 4-30-14 6,474 3,829 2,645 41% 5,530 11,001
FY15Q1 7-31-14 5,496 3,583 1,913 35% 5,998 10,896
FY15Q2 10-31-14 6,263 4,139 2,124 34% 5,379 11,161
FY15Q3 1-31-15 5,677 3,113 2,564 45% 6,148 14,063
FY15Q4 4-30-15 9,308 4,758 4,550 49% 7,354 17,993
FY16Q1 7-31-15 9,379 4,608 4,771 51% 10,457 17,890
FY16Q2 10-31-15 9,523 4,741 4,782 50% 12,554 24,623
FY16Q3 1-31-16 6,672 3,896 2,776 42% 15,189 37,851
FY16Q4 4-30-16 18,783 9,721 9,062 48% 15,189 39,630
FY17Q1 7-31-16 5,609 3,062 2,547 45% 25,274 43,262
FY17Q2 10-31-16 23,370 15,441 7,929 34% 25,924 44,908
FY17Q3 1-31-17 10,747 7,974 2,773 26% 33,829 52,577
FY17Q4 4-30-17 17,904 11,782 6,122 34% 33,099 45,517
FY18Q1 7-31-17 27,077 20,448 6,629 24% 24,235 27,755
FY18Q2 10-31-17 12,782 16,242 -3,460 -27% 16,518 20,611
FY18Q3 1-31-18 6,819 10,951 -4,132 -61% 14,218 24,235
FY18Q4 4-30-18 6,943 8,904 -1,961 -28% 16,129 27,935
FY19Q1 7-31-18 12,589 11,397 1,192 9% 9,168 17,994
FY19Q2 10-31-18 10,178 9,844 334 3% 9,736 17,307
FY19Q3 1-31-19 13,781 11,731 2,050 15% 8,660 14,620
FY19Q4 4-30-19 17,055 13,407 3,648 21% 6,557 14,651
FY20Q1 7-31-19 15,254 14,168 1,086 7% 8,031 18,104
FY20Q2 10-31-19 18,313 14,953 3,360 18% 7,809 22,199
FY20Q3 1-31-20 13,585 12,800 785 6% 9,565 26,355
FY20Q4 4-30-20 12,550 13,849 -1,299 -10% 10,883 29,120
FY21Q1 7-31-20 25,392 16,848 8,544 34% 9,610 27,123
FY21Q2 10-31-20 21,064 14,646 6,418 30% 9,723 31,450
*7-31-18 10Q: “prior-yr amts related to (deferred revenue
+ cust deposits) now reclass’d as contract liabilities.”
Rev$ COGS$ Prof$ GP%
FY13 TOTAL: 21,333 12,595 8,738 41%*
FY14 TOTAL: 22,294 13,110 9,184 41%*
FY15 TOTAL: 26,744 15,393 11,151 42%*
FY16 TOTAL: 44,357 22,966 21,391 48%*
FY17 TOTAL: 57,630 38,259 19,371 34%*
FY18 TOTAL: 53,621 56,545 -2,924 -5%*
FY19 TOTAL: 53,603 46,379 7,224 13%*
FY20 TOTAL: 59,702 55,770 3,932 7%*
*Avid Net-Profit(Selling/G&A) not split out from PPHM-Corp. in the fin’s.
AVID TOTAL REV’s BY YEAR):
FY04 4-30-04 3,039 (Avid-Revs didn’t incl. Avid’s Gov’t work)
FY05 4-30-05 4,684
FY06 4-30-06 3,005
FY07 4-30-07 3,492
FY08 4-30-08 5,897
FY09 4-30-09 12,963
FY10 4-30-10 13,204
FY11 4-30-11 8,502
FY12 4-30-12 14,783
FY13 4-30-13 21,333
FY14 4-30-14 22,294
FY15 4-30-15 26,744
FY16 4-30-16 44,357
FY17 4-30-17 57,630
FY18 4-30-18 53,621
FY19 4-30-19 53,603
FY20 4-30-20 59,702
FY21 4-30-21 46,456 (thru Q2)
**TOTAL: 455,309 (5/1/2003–7/31/20)
.
QTLY. NET PROFIT/LOSS BY QTR:
(“attributable to common stockholders”; ie, incl. PREF Div’s**)
**2-11-14: PPHM Raises $16.2M, 700k Pref. Shares w/10.5% DIV.
FY16Q1 7-31-15 -15,101,000
FY16Q2 10-31-15 -14,578,000
FY16Q3 1-31-16 -18,227,000
FY16Q4 4-30-16 -13,264,000
FY17Q1 7-31-16 -12,437,000
FY17Q2 10-31-16 -4,498,000
FY17Q3 1-31-17 -9,216,000
FY17Q4 4-30-17 -6,714,000
FY18Q1 7-31-17 -2,647,000
FY18Q2 10-31-17 -14,066,000
FY18Q3 1-31-18 -12,446,000
FY18Q4 4-30-18 +1,578,000 <=includes $9,154,000 income from disc. operations.
FY19Q1 7-31-18 -3,403,000
FY19Q2 10-31-18 -2,893,000
FY19Q3 1-31-19 -2,581,000
FY19Q4 4-30-19 -1,106,000
FY20Q1 7-31-19 -4,606,000
FY20Q2 10-31-19 -1,872,000
FY20Q3 1-31-20 -3,546,000
FY20Q4 4-30-20 -6,210,000
FY21Q1 7-31-20 +3,288,000
FY21Q2 10-31-20 +842,000
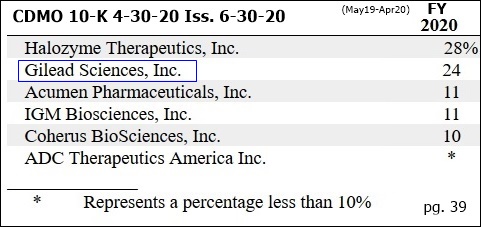
CUSTOMER SPLITS - 4-30-20 10-K pg.39 (iss. 6-30-20):
Period Halozyme Gilead Acumen IGM Coherus ADC-Ther. Others
FYE 4-30-14 91% 8%
FYE 4-30-15 79% 9%
FYE 4-30-16 69% 26% 5%
FYE 4-30-17 58% 16% 26% 16%
FYE 4-30-18 55% 22% 9% 14%
FYE 4-30-19 30% <10% <10% 13% 21% 36%
FYE 4-30-20 28% 24% 11% 11% 10% <10% --
...(cust. splits not given in 7-31-18+ 10Q’s)
Yes, I'm seeing IsZo Capital 3,459,888 6.1% -88,144 a/o 9-30-20 Nasdaq Inst.
https://www.nasdaq.com/market-activity/stocks/cdmo/institutional-holdings
Thanks, I'll fix and repost... They won't let me remove my own post.
Total INST.+LARGE Holdings now 41,617,965 73.5% (9-30-20)
Up from 38,627,460 68.4% at 6-30-20.
33,031,973 47.9% INSTITUTIONS (a/o 9-30-20)
https://www.nasdaq.com/symbol/cdmo/institutional-holdings
+ 8,585,992 15.2% LARGE SHAREHOLDERS via Forms13/14 (EasternCap/Dart8-2019, Tappan/12-2019) – ie, not in Nasdaq Inst. List.
------------
= 41,617,965 73.5%
Ownership %’s based on 56,602,792 shares O/S at 8-24-20 (10Q: https://tinyurl.com/y5puv3b7 )
13 LARGEST SHAREHOLDERS:
1. Eastern Capital (Kenneth Dart): 4,300,992 7.6% (a/o 8-21-19 14A/Proxy: https://tinyurl.com/y9c972fa – orig. 13G filed 11-2015)
2. Tappan Street (Prasad Phatak): 4,285,000 7.6% (a/o 12-31-19 13G: https://tinyurl.com/wlcwnnv )
3. Blackrock Inc. (Larry Fink): 3,834,816 6.8% (-111,172 a/o 9-30-20 Nasdaq Inst.)
4. IsZo Capital Mgt. (Brian Sheehy): 3,459,888 6.1% (-88,144 a/o 9-30-20 Nasdaq Inst.)
5. Millennium Mgt. 2,678,984 4.7% (+633,918 a/o 9-30-20 Nasdaq Inst.)
6. Vanguard Group 2,659,738 4.7% (-19,553 q/e 9-30-20 Nasdaq Inst.)
7. Snyder Capital 2,302,621 4.1% (-2,258 a/o 9-30-20 Nasdaq Inst.)
8. Altravue Capital 1,973,349 3.5% (+39,534 a/o 9-30-20 Nasdaq Inst.)
9. Wellington Mgt. 1,503,162 2.7% (+236,947 a/o 9-30-20 Nasdaq Inst.)
10. Sargent Invest. Grp. 1,052,099 1.9% (-11,000 a/o 9-30-20 Nasdaq Inst.)
11. State Street 992,280 1.8% (-65,922 a/o 9-30-20 Nasdaq Inst.)
12. Portolan Capital 963,374 1.7% (+528,662 a/o 9-30-20 Nasdaq Inst.)
13. Silvercrest Asset Mgt. 878,805 1.6% (-16,746 a/o 9-30-20 Nasdaq Inst.)
BIGGEST DROPS q/e 9-30-20:
Oxford Asset Mgt. -264,077
Kennedy Capital -234,631
= = = = = = = = = = = =
Shares O/S as of 8-24-20=56,602,792 - history since 4-2006: https://tinyurl.com/y329llow (at bottom)
...A/O 7-31-20: 3,616,000 stock options outstanding at a wgt.avg. exercise price of $6.36 (pg.12 10Q).
9-1-20: Qtly. Conf. Call (Green/Hart/Compton) PR & Transcript https://tinyurl.com/y329llow
10-20-20: Avid’s ASM Replay 9:45 (Control# not needed)
https://www.virtualshareholdermeeting.com/CDMO2020 (Control# not needed)
…14A Proxy iss. 8-27-20: https://tinyurl.com/y46ga9el

*Revs Guidance (FY’21 fye 4-30-21): $76-81M. Backlog 7-31-20=$60M
*Cash: 7-31-20: $28.2M
*As of 8-24-20: 56,602,792 shares o/s.
*Total INST.+LARGE Holdings: 38,627,460 68.4% a/o 6-30-20 https://tinyurl.com/y3jxd4oh
*10Q/7-31-20 iss. 9-1-20: https://tinyurl.com/y5puv3b7
*10K/4-30-20 iss. 6-30-20: https://tinyurl.com/yak25nco
*Avid Total Revs May03-Jul20: $434.2M
*Avid’s website: https://avidbio.com (A/o 4-30-20, 222 full-time & 5 part-time emps)
9-1-20 Qtly CC-Transcript, PR(qe7-31-20), Avid Revs History Table
*Revs Guidance (FY’21 fye 4-30-21): $76-81M. 7-31-20 Backlog=$60M
*Cash: 7-31-20: $28.2M
*As of 8-24-20: 56,602,792 shares o/s
*10Q/7-31-20 iss. 9-1-20: https://tinyurl.com/y5puv3b7
*10K/4-30-20 iss. 6-30-20: https://tinyurl.com/yak25nco
*Avid Total Revs May03-Jul20: $434.2M
*Avid’s website: https://avidbio.com (A/o 4-30-20, 222 full-time & 5 part-time emps)
This large post has 4 sections:
I. 9-1-20 Qtly. Earnings Conf. Call TRANSCRIPT (FY21/Q1 qe 7-31-20)
II. 9-1-20 CDMO Press Release: Q1/FY21 Earnings & Developments
IV. Updated Table of Avid Revenues By Quarter (May’06-Current)
III. Updated O/S Shares History Table – 2006-curr.
…Recall: Avid’s FY runs May-Apr, so FY’21 = May’20-Apr’21.
TRANSCRIPT 9-1-20 FY21/Q1 Earnings Conf. Call (qe 7-31-20):
Nick Green(CEO), Daniel Hart(CFO), Tim Compton(CCO)
Q&A Analysts:
Matt Hewitt - Craig-Hallum Capital
Jacob Johnson – Stephens Inc.
Link to webcast replay: http://ir.avidbio.com/events-and-presentations => https://edge.media-server.com/mmc/p/64ateurd
CC TRANSCRIPT EXTRACTS – PREPARED REMARKS BY CEO NICK GREEN:
CEO NICK GREEN OPENING:
Thank you to everybody who has dialed in and also to those who are participating today via webcast. I officially joined Avid in late July and have truly enjoyed my first month on the job. Despite the challenges presented by the pandemic, my family and I have successfully relocated to Orange County and are settling into our new community well. My first few weeks have done nothing but confirm my view that I believe Avid to be a strong and state-of-the-art company with significant opportunity for growth. This is especially true given our focus on biologics, which is growing faster than many other sectors in the industry. As I return to the USA from my time abroad, I can't help but feel that Avid is well positioned to benefit from some of the industry trends we have seen developing recently. Given the broad and deep talent of our employees, our quality facilities, systems, expertise and commitment to excellence that fuels our work here at Avid, I am confident that the company will continue to thrive.
As I begin what I believe will be an exciting and successful time at Avid, I would like to thank my predecessor Rick Hancock for his contributions over the last year. Under Rick's leadership, Avid has evolved into the growth organization it is today and I thank him and the Board for entrusting me with the future of the company.
I'll now address our financial business dev. and operational achievements for the period. From a financial perspective, Q1 was particularly strong as we significantly exceeded revenue expectations as well as other key metrics. In business development, we expanded our customer base with the addition of 3 new customers, each of which brings an exciting new project to the manufacturing portfolio. Lastly, Avid's operational activities include the completion of multiple production runs that validated the remediation activities we undertook in late FY2020, as well as the continued development of our plans to provide additional capacity. Tim and I will provide additional details on the business dev. and operations following an overview of our Q1 financial results. And for that, I turn the call over to Dan.”
CEO NICK GREEN - AFTER HART/COMPTON OPENING:
“I'd like to expand on Tim's final comments to address Avid's broader operations within the context of COVID-19. Despite the disruption caused globally by the pandemic, we are fortunate that we have not experienced any interruption to our operations. To date, we have observed no material impact to our production programs or our supply chain, and our employees remain healthy and productive. Management continues to take every precaution and follow state and local guidelines to ensure the continued safety and well-being of our team members, and we are hopeful that our operations will remain un-impacted.
During and subsequent to the quarter, we made progress with a number of important operational projects. I'd first like to address the equipment issue that interrupted several production runs during Q3 & Q4 FY2020. As we reported last quarter, the specific piece of equipment in question is now operational. Following efforts to investigate and remediate the problem during Q4, we have now validated these efforts by completing multiple successful revenue generating production campaigns using this equipment. Importantly, as Dan reported earlier on, all of the delayed FY2020 have now been completed. At the very end of July, Avid also initiated its annual preventative maintenance shutdown. As in prior years, this process temporarily reduces available capacity, which as usual we expect to have some impact on our Q2 FY2021 financial results.
Lastly, we continue to make progress with our expansion plans, as we continue to see growth in customer demand, the ability to access additional capacity grows increasingly important. As I am new to the team and considering the importance of this in the future of the business, I'm in the process of reviewing the design plans and other ancillary requirements ahead of making our final decision with respect to the best path forward for the business. An expansion of this nature typically takes up to 24 months to complete, and as such we are keen to not only move swiftly, but also thoughtfully.
In closing, I can say that I believe the accomplishments of Q1 have established a good momentum for the rest of the year to come. During the quarter, our business dev. team signed $20 million in project orders with new customers as well as with existing customers. Looking ahead, we see growth in manufacturing demand and expect to continue to expand our production pipeline. Financially, we believe this demand will continue to drive our top line growth with margins improving in line. While we are not currently in a position to project sustainable quarter-to-quarter profitability, we are focused on extending this performance across our fiscal year.”
CEO NICK GREEN - CLOSING (After Q&A):
“Thank you to everyone participating on today's call. I am pleased to be at Avid at this exciting time in the company's evolution, and have a great deal of optimism for the future of the business, and I'm very happy to be working alongside our exceptional employees. In closing, I'd like to thank our dedicated employees, many of whom are at Avid daily, working through the challenges presented by COVID-19, both at home and at work. They work to ensure that we are able to deliver commercial and clinical products we need to supply to patients in need. None of this success would be possible without the hard work, commitment to our employees and I am grateful for their dedication. I thank you again for participating in today's call and thank you for your continued support of Avid Bioservices.”
READ the entire CC Transcript here:
https://www.fool.com/earnings/call-transcripts/2020/09/01/avid-bioservices-inc-cdmo-q1-2021-earnings-call-tr
= = = = = = = = = = = = = = = = = = = = = == = = =
9-1-20/PR: Avid Bioservices Reports Financial Results for Q1/FY21 Ended July 31, 2020, and Recent Developments
http://ir.avidbio.com/news-releases/news-release-details/avid-bioservices-reports-financial-results-first-quarter-ended
-- Recorded Q1 Revenue of $25.4 Million
-- Signed Three New Customers and Project Expansion Orders with Current Customers for $20 Million
-- Backlog of $60 Million at Quarter-End
TUSTIN: Avid Bioservices, Inc. (NASDAQ:CDMO/CDMOP), a dedicated biologics contract development and manufacturing organization (CDMO) working to improve patient lives by providing high quality development and manufacturing services to biotechnology and pharmaceutical companies, today announced financial results for the Q1 of FY2021, ended July 31, 2020.
HIGHLIGHTS SINCE APRIL 30, 2020
“FY2021 is off to a strong start as a result of the accomplishments of the Q1,” stated Nicholas Green, President and CEO of Avid Bioservices. “During the quarter, the company achieved revenues of $25.4 million and net income of $4.7 million. And though revenue for the quarter was supplemented as a result of our completion of runs deferred from the third and fourth quarters of fiscal 2020, we are optimistic that future demand will support our growth trajectory. During the period, our business development team signed three new customers as well as project expansion orders with several existing customers. The visibility we currently have into the industry, as well as our customer projections point to continued growth in production demand throughout the year, and it is our priority to be well-positioned to support that demand.
“In recent weeks, the company also made progress with several important operational projects. As reported previously, the company experienced an equipment issue that interrupted several production runs during the third and fourth quarters of fiscal 2020. I am pleased to report that the remediation activities conducted were validated during the Q1 of FY2021 by completing multiple successful revenue-generating production campaigns using this equipment. Avid also recently initiated its planned annual preventative facility maintenance program. This effort is proceeding well, with work largely completed in the Franklin facility and work on our Myford facility well underway. Lastly, during the Q1, we made further progress with our expansion plans. As we continue to see growth in customer demand, this anticipated expansion will be important to sustaining strong future growth. We continue to dedicate significant time and resources to design, review and pre-engineering activities and look forward to updating you on our plans in the near future.
“I was very pleased to officially join the company in late July, and my first month has only strengthened my belief that Avid is a state-of-the art business with significant opportunity for growth. This is especially true given our focus on biologics, which is one of the fastest growing sectors of the industry. As I return to the United States from my time abroad, I can’t help but feel that Avid is well positioned to benefit from some of the industry trends we have seen developing recently. Given the broad and deep talent of our employees, our quality facilities and systems, and the expertise and commitment to excellence that fuels our work here at Avid, I am confident that the company will continue to thrive. As I begin what I believe will be an exciting and successful time at Avid, I wish to thank my predecessor, Rick Hancock, for his contributions over the last year. Under Rick’s leadership, Avid has evolved into the growth organization it is today, and I thank him and the board for entrusting me with the future of the company.”
FINANCIAL HIGHLIGHTS AND GUIDANCE
The company is confirming revenue guidance for the full FY 2021 of $76-$81 million.
Revenues for the Q1 of FY2021 were $25.4 million, a 66% increase compared to revenues of $15.3 million recorded during the Q1 of FY2020. The increase in revenue was primarily attributable to growth in the number and scale of in-process and/or completed manufacturing runs during the quarter, including $4.3 million from the completion of all batches that had been deferred from the third and fourth quarters of FY2020 due to a previously reported equipment issue, and $3.1 million in fees received from a customer that reached its inventory requirements with fewer than expected runs, therefore not utilizing all their reserved capacity that had been scheduled for third quarter of FY2021. Avid expects demand for high quality biologics capacity to continue, and it remains on track to achieve the stated revenue guidance for FY2021. However, the company cautions against using its Q1 revenues as a new benchmark, as the deferred batches from the third and fourth quarters of FY2020 and the fees associated with a customer’s unused capacity contributed to the top line during the period.
As of July 31, 2020, revenue backlog was $60 million, compared to $65 million at the end of Q4 of FY2020. The company expects to recognize the majority of this backlog during FY2021.
Gross margin for the Q1 of FY2021 was 34%, a significant increase compared to a gross margin of 7% for the Q1 of FY2020. The increase in gross margin for the FY2021 quarter was primarily attributable to increased manufacturing revenue from the growth in the number and scale of manufacturing runs and the aforementioned fees associated with a customer’s unused capacity.
Selling, general and administrative expenses (“SG&A”) for the Q1 of FY2021 were $3.8 million versus $4.5 million recorded for the Q1 of FY2020. The decrease in SG&A was primarily attributed to a decrease in separation related expenses and other general SG&A expenses, partially offset by a net increase in payroll and benefits costs.
For the Q1 of FY2021, the company recorded a consolidated net income attributable to common stockholders of $3.3 million or $.06 per basic and diluted share, as compared to a consolidated net loss attributable to common stockholders of $4.6 million or $.08 per basic and diluted share, for the Q1 of FY2020.
Avid reported $28.2 million in cash and cash equivalents as of July 31, 2020, compared to $36.3 million as of the prior FY ended April 30, 2020.
More detailed financial information and analysis may be found in Avid Bioservices’ Quarterly Report on Form 10-Q, which will be filed with the SEC today. [https://tinyurl.com/y5puv3b7 ]
RECENT CORPORATE DEVELOPMENTS
Expanded the company’s customer base with the addition of three new customers and executed multiple project expansion orders with existing customers representing additional net revenue backlog of $20 million during the Q1. The company’s new customers include Iovance Biotherapeutics, Inc., Oragenics, Inc., and Mapp Biopharmaceutical, Inc. These new customers will take advantage of the range of capabilities at Avid including technical transfer, process development and scale-up, all with a view to future GMP manufacture.
Entered into a co-marketing agreement with Argonaut Manufacturing Services to support drug product manufacturing. This partnership is designed to offer customers Avid’s upstream and downstream process development and drug substance manufacturing services in tandem with Argonaut’s parenteral drug product fill-finish services, to support the efficient delivery of CGMP parenteral drug products for use in clinical studies. Through the collaborations signed with Aragen Bioscience during Q4 FY2020, and with Argonaut in Q1 FY2021, Avid has strategically established an end-to-end option for customers seeking support from early cell line development through drug product manufacturing.
Continued the pre-engineering, design and permitting work required to allow the company to break ground on a facility expansion at the appropriate time. While a specific kick-off date has not yet been established for this expansion, the company believes that continued strong customer demand will require additional capacity and Avid is proactively working to prepare for this growth.
CONFERENCE CALL
Avid will host a conference call and webcast this afternoon, September 1, 2020, at 4:30 PM EDT (1:30 PM PDT). To listen to the conference call, please dial (877) 312-5443 or (253) 237-1126 and request the Avid Bioservices conference call. To listen to the live webcast, or access the archived webcast, please visit: http://ir.avidbio.com/investor-events .
ABOUT AVID BIOSERVICES, INC.
Avid Bioservices is a dedicated contract development and manufacturing organization (CDMO) focused on development and CGMP manufacturing of biopharmaceutical drug substances derived from mammalian cell culture. The company provides a comprehensive range of process development, CGMP clinical and commercial manufacturing services for the biotechnology and biopharmaceutical industries. With 27 years of experience producing monoclonal antibodies and recombinant proteins, Avid's services include CGMP clinical and commercial drug substance manufacturing, bulk packaging, release and stability testing and regulatory submissions support. For early-stage programs the company provides a variety of process development activities, including upstream and downstream development and optimization, analytical methods development, testing and characterization. The scope of our services ranges from standalone process development projects to full development and manufacturing programs through commercialization. http://www.avidbio.com
AVID BIOSERVICES, INC.
CONDENSED CONSOLIDATED STATEMENTS OF OPERATIONS AND COMPREHENSIVE INCOME (LOSS)
(Unaudited) (In thousands, except per share information)
Three Months Ended July 31,
2020 2019
Revenues $ 25,392 $ 15,254
Cost of revenues 16,848 14,168
Gross profit 8,544 1,086
Operating expenses:
Selling, general and administrative 3,825 4,459
Operating income (loss) 4,719 (3,373 )
Interest and other income, net 11 209
Net income (loss) $ 4,730 $ (3,164 )
Comprehensive income (loss) $ 4,730 $ (3,164 )
Series E preferred stock accumulated dividends (1,442 ) (1,442 )
Net income (loss) attributable to common stockholders $ 3,288 $ (4,606 )
Net income (loss) per share attributable to common stockholders:
Basic $ 0.06 $ (0.08 )
Diluted $ 0.06 $ (0.08 )
Weighted average common shares outstanding:
Basic 56,523 56,167
Diluted 56,892 56,167
AVID BIOSERVICES, INC.
CONDENSED CONSOLIDATED BALANCE SHEETS
(Unaudited) (In thousands, except par value)
July 31, 2020 April 30, 2020
ASSETS
Current assets:
Cash and cash equivalents $ 28,211 $ 36,262
Accounts receivable 14,311 8,606
Contract assets 4,317 3,300
Inventory 9,610 10,883
Prepaid expenses 569 712
Total current assets 57,018 59,763
Property and equipment, net 28,134 27,105
Operating lease right-of-use assets 19,757 20,100
Restricted cash 350 350
Other assets 302 302
Total assets $ 105,561 $ 107,620
LIABILITIES AND STOCKHOLDERS’ EQUITY
Current liabilities:
Accounts payable $ 5,724 $ 5,926
Accrued payroll and related costs 3,405 3,019
Contract liabilities 27,123 29,120
Operating lease liabilities 1,268 1,228
Note payable — 4,379
Other current liabilities 591 808
Total current liabilities 38,111 44,480
Operating lease liabilities, less current portion 20,911 21,244
Total liabilities 59,022 65,724
Commitments and contingencies
Stockholders’ equity:
Preferred stock, $0.001 par value; 5,000 shares authorized;
1,648 shares issued and outstanding at July 31, 2020 and April 30,
2020, respectively 2 2
Common stock, $0.001 par value; 150,000 shares authorized;
56,601 and 56,483 shares issued and outstanding at July 31, 2020 and
April 30, 2020, respectively 56 56
Additional paid-in capital 612,822 612,909
Accumulated deficit (566,341 ) (571,071 )
Total stockholders’ equity 46,539 41,896
Total liabilities and stockholders’ equity $ 105,561 $ 107,620
CONTACTS:
• Stephanie Diaz (Investors) Vida Strategic Partners 415-675-7401 sdiaz@vidasp.com
• Tim Brons (Media) Vida Strategic Partners 415-675-7402 tbrons@vidasp.com
- - - - - - - -
From 10-Q header: “As of Aug 24, 2020, there were 56,602,792 shares outstanding.”
- - - - - - - - - - - - - - - - -
Latest 10Q 7-31-20 iss. 9-1-20 https://tinyurl.com/y5puv3b7 (Cash 7-31-20=$28.2mm)
Latest 10K 4-30-20 iss. 6-30-20 https://tinyurl.com/yak25nco (Cash 4-30-20=$36.3mm)
Latest DEF14A/Proxy iss. 8-27-20 (re: 10-20-20 ASM): https://tinyurl.com/y46ga9el
ALL SEC filings for PPHM: http://tinyurl.com/6d4jw8
10-K: “As of 4-30-20, we employed 222 full-time & 5 part-time emps.” (2018: 185/1, 2019: 211/4)
= = = = = = = = = = = = = = = = = = = = = = = = = = = =
Updated PPHM REVS-BY-QTR TABLE, now thru FY21Q1(qe 7-31-20), per the 10Q issued 9-1-20.
• Total Avid Revs since May’03: $434.2M
• 9-1-20: FY'21 (May'20-Apr'21) Avid revs guidance $76-81M (committed B/L=$60 at 7-31-20).
• Inventories at 7-31-20 total $9.6M, DOWN from $10.9M at 4-30-20.
Avid’s website: http://www.avidbio.com
AVID GROSS PROFITABILITY BY QTR: CONTRACT
QTR (1000’s) Rev$ COGS$ Prof$ GP% INVEN$ LIABILITIES*
FY13Q1 7-31-12 4,135 2,024 2,111 51% 5,744 16,280
FY13Q2 10-31-12 6,061 3,703 2,358 39% 5,426 14,721
FY13Q3 1-31-13 6,961 3,651 3,310 47% 4,635 11,790
FY13Q4 4-30-13 4,176 3,217 959 23% 4,339 12,230
FY14Q1 7-31-13 4,581 2,670 1,911 42% 5,679 12,692
FY14Q2 10-31-13 7,354 4,195 3,159 43% 4,033 11,126
FY14Q3 1-31-14 3,885 2,416 1,469 38% 5,224 12,975
FY14Q4 4-30-14 6,474 3,829 2,645 41% 5,530 11,001
FY15Q1 7-31-14 5,496 3,583 1,913 35% 5,998 10,896
FY15Q2 10-31-14 6,263 4,139 2,124 34% 5,379 11,161
FY15Q3 1-31-15 5,677 3,113 2,564 45% 6,148 14,063
FY15Q4 4-30-15 9,308 4,758 4,550 49% 7,354 17,993
FY16Q1 7-31-15 9,379 4,608 4,771 51% 10,457 17,890
FY16Q2 10-31-15 9,523 4,741 4,782 50% 12,554 24,623
FY16Q3 1-31-16 6,672 3,896 2,776 42% 15,189 37,851
FY16Q4 4-30-16 18,783 9,721 9,062 48% 15,189 39,630
FY17Q1 7-31-16 5,609 3,062 2,547 45% 25,274 43,262
FY17Q2 10-31-16 23,370 15,441 7,929 34% 25,924 44,908
FY17Q3 1-31-17 10,747 7,974 2,773 26% 33,829 52,577
FY17Q4 4-30-17 17,904 11,782 6,122 34% 33,099 45,517
FY18Q1 7-31-17 27,077 20,448 6,629 24% 24,235 27,755
FY18Q2 10-31-17 12,782 16,242 -3,460 -27% 16,518 20,611
FY18Q3 1-31-18 6,819 10,951 -4,132 -61% 14,218 24,235
FY18Q4 4-30-18 6,943 8,904 -1,961 -28% 16,129 27,935
FY19Q1 7-31-18 12,589 11,397 1,192 9% 9,168 17,994
FY19Q2 10-31-18 10,178 9,844 334 3% 9,736 17,307
FY19Q3 1-31-19 13,781 11,731 2,050 15% 8,660 14,620
FY19Q4 4-30-19 17,055 13,407 3,648 21% 6,557 14,651
FY20Q1 7-31-19 15,254 14,168 1,086 7% 8,031 18,104
FY20Q2 10-31-19 18,313 14,953 3,360 18% 7,809 22,199
FY20Q3 1-31-20 13,585 12,800 785 6% 9,565 26,355
FY20Q4 4-30-20 12,550 13,849 -1,299 -10% 10,883 29,120
FY21Q1 7-31-20 25,392 16,848 8,544 34% 9,610 27,123
*7-31-18 10Q: “prior-yr amts related to (deferred revenue
+ cust deposits) now reclass’d as contract liabilities.”
Rev$ COGS$ Prof$ GP%
FY13 TOTAL: 21,333 12,595 8,738 41%*
FY14 TOTAL: 22,294 13,110 9,184 41%*
FY15 TOTAL: 26,744 15,393 11,151 42%*
FY16 TOTAL: 44,357 22,966 21,391 48%*
FY17 TOTAL: 57,630 38,259 19,371 34%*
FY18 TOTAL: 53,621 56,545 -2,924 -5%*
FY19 TOTAL: 53,603 46,379 7,224 13%*
FY20 TOTAL: 59,702 55,770 3,932 7%*
*Avid Net-Profit(Selling/G&A) not split out from PPHM-Corp. in the fin’s.
AVID TOTAL REV’s BY YEAR):
FY04 4-30-04 3,039 (Avid-Revs didn’t incl. Avid’s Gov’t work)
FY05 4-30-05 4,684
FY06 4-30-06 3,005
FY07 4-30-07 3,492
FY08 4-30-08 5,897
FY09 4-30-09 12,963
FY10 4-30-10 13,204
FY11 4-30-11 8,502
FY12 4-30-12 14,783
FY13 4-30-13 21,333
FY14 4-30-14 22,294
FY15 4-30-15 26,744
FY16 4-30-16 44,357
FY17 4-30-17 57,630
FY18 4-30-18 53,621
FY19 4-30-19 53,603
FY20 4-30-20 59,702
FY21 4-30-21 25,392 (thru Q1)
**TOTAL: 434,245 (5/1/2003–7/31/20)
.
QTLY. NET PROFIT/LOSS BY QTR:
(“attributable to common stockholders”; ie, incl. PREF Div’s**)
**2-11-14: PPHM Raises $16.2M, 700k Pref. Shares w/10.5% DIV.
FY16Q1 7-31-15 -15,101,000
FY16Q2 10-31-15 -14,578,000
FY16Q3 1-31-16 -18,227,000
FY16Q4 4-30-16 -13,264,000
FY17Q1 7-31-16 -12,437,000
FY17Q2 10-31-16 -4,498,000
FY17Q3 1-31-17 -9,216,000
FY17Q4 4-30-17 -6,714,000
FY18Q1 7-31-17 -2,647,000
FY18Q2 10-31-17 -14,066,000
FY18Q3 1-31-18 -12,446,000
FY18Q4 4-30-18 +1,578,000 <=includes $9,154,000 income from disc. operations.
FY19Q1 7-31-18 -3,403,000
FY19Q2 10-31-18 -2,893,000
FY19Q3 1-31-19 -2,581,000
FY19Q4 4-30-19 -1,106,000
FY20Q1 7-31-19 -4,606,000
FY20Q2 10-31-19 -1,872,000
FY20Q3 1-31-20 -3,546,000
FY20Q4 4-30-20 -6,210,000
FY21Q1 7-31-20 +3,288,000
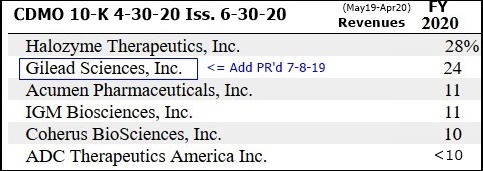
CUSTOMER SPLITS - 4-30-20 10-K pg.39 (iss. 6-30-20):
Period Halozyme Gilead Acumen IGM Coherus ADC-Ther. Others
FYE 4-30-14 91% 8%
FYE 4-30-15 79% 9%
FYE 4-30-16 69% 26% 5%
FYE 4-30-17 58% 16% 26% 16%
FYE 4-30-18 55% 22% 9% 14%
FYE 4-30-19 30% <10% <10% 13% 21% 36%
FYE 4-30-20 28% 24% 11% 11% 10% <10% --
...(cust. splits not given in 7-31-18+ 10Q’s)

In June2019, MappBio got a BARDA contract for $16.5M, w/option for $30M more, for dev. of MBP091 to neutralize Marburg virus though the completion of a Phase 1 clinical trial.
6-11-19: “MappBio Announces Contract with BARDA for MBP091 as a pan-Marburg Medical Countermeasure”
…awarded an advanced R&D contract for $16.5M with options for an addl. $30M by BARDA… MBP091 is a single human monoclonal antibody (mAb) that is being developed as a pan-Marburg virus therapeutic for the treatment of hemorrhagic fevers caused by Marburg and the closely related Ravn virus. The mAb was isolated in the laboratory of Dr. James Crowe at Vanderbilt University Medical Center from a survivor or Marburg virus infection and subsequently licensed by Mapp. MBP091 has been evaluated in numerous preclinical experiments and has demonstrated the ability to neutralize Marburg virus in vitro and protect from lethal challenge of Marburg in vivo…
"We are pleased to continue and expand our productive relationship with BARDA, which represents a good example of a public-private partnership that advances much needed therapeutics for infected individuals and the Strategic National Stockpile," said Dr. Kevin J. Whaley, Mapp’s CEO.
About Mapp Biopharmaceutical:
Mapp Biopharmaceutical was founded in 2003 to develop novel pharmaceuticals for the prevention and treatment of infectious diseases, focusing on unmet needs in global health and biodefense. https://www.mappbio.com
This project has been funded in whole or in part with Federal funds from the Dept of HHS; Office of the Assistant Secretary for Preparedness and Response; BARDA, under Contract Nos. HHSO100201900018C and HHSO100201700023C, a NIH grant #2SB1 AI082744 and Medical CBRN Defense Consortium Base Agreement 2018-843 under OTA W15QKN-16-9-1002.
= = = = = =
8-26-20 Avid’s Known Customers Mkt-Caps
Pg.39 of 4-30-20 10-K (iss. 6-30-20):
1. Halozyme Therapeutics https://finance.yahoo.com/quote/HALO 3.9B
2. Gilead Sciences https://finance.yahoo.com/quote/GILD 82.7B
3. Acumen Pharmaceuticals PRIV: https://www.acumenpharm.com
4. IGM Biosciences https://finance.yahoo.com/quote/IGMS 1.5B
5. Coherus BioSciences https://finance.yahoo.com/quote/CHRS 1.4B
6. ADC Therapeutics America https://finance.yahoo.com/quote/ADCT 2.9B
NEW 8-2020:
7. Iovance Biotherapeutics https://finance.yahoo.com/quote/IOVA 4.2B
8. Oragenics https://finance.yahoo.com/quote/OGEN 58M
9. Mapp Biopharmaceutical (Private) https://mappbio.com BARDA #HHSO100201900018C
Aug2020 Adds:
8-6-20: IOVANCE Biotherapeutics signs w/Avid(+Aragen) to Dev+Mfg. IOV-3001 (IL-2 Analog) https://tinyurl.com/y2lgzh6x
BioProcIntl: “Iovance said it would focus on GMP manufacturing of IOV-3001 during 2020 and may initiate IND-enabling activities as early as 2021” https://tinyurl.com/y4ko9j7t
8-20-20: ORAGENICS Signs Dev/Mfg Agreement with Avid for COVID-19 Vaccine “TerraCoV2” https://tinyurl.com/yxqg3w4v
8-26-20: MAPP BIOPHARMACEUTICAL signs w/Avid for Clin. Dev. of Novel Antiviral Antibody https://tinyurl.com/yxwvr949 BARDA #HHSO100201900018C
So ElSid18, let me try to unscramble what you posted yesterday at 2:45pmET, after reading your explanations and corrections… Yes, most definitely, the corrected post makes sense v. the original. Thanks for clarifying and correcting.
Aug24/12:09pmET: “look for a move into the $6.20's and then a return to the teens…”
Note: at 12:09pm, CDMO price was 8.17. https://finance.yahoo.com/quote/CDMO?p=CDMO
Final Corrected: “look for a move into the $8.20's and then a return to the teens(as in $8.15 $8.17, to the right of the decimal point)…”
8-21-20 Avid’s Known Customers Mkt-Caps
Pg.39 of 4-30-20 10-K (iss. 6-30-20):
1. Halozyme Therapeutics https://finance.yahoo.com/quote/HALO 3.9B
2. Gilead Sciences https://finance.yahoo.com/quote/GILD 82.7B
3. Acumen Pharmaceuticals PRIV: https://www.acumenpharm.com
4. IGM Biosciences https://finance.yahoo.com/quote/IGMS 1.5B
5. Coherus BioSciences https://finance.yahoo.com/quote/CHRS 1.4B
6. ADC Therapeutics America https://finance.yahoo.com/quote/ADCT 2.9B
NEW 8-2020:
7. Iovance Biotherapeutics https://finance.yahoo.com/quote/IOVA 4.2B
8. Oragenics https://finance.yahoo.com/quote/OGEN 58M
Aug2020 Adds:
8-6-20: IOVANCE Biotherapeutics signs w/Avid(+Aragen) to Dev+Mfg. IOV-3001 (IL-2 Analog) https://tinyurl.com/y2lgzh6x
BioProcIntl: “Iovance said it would focus on GMP manufacturing of IOV-3001 during 2020 and may initiate IND-enabling activities as early as 2021” https://tinyurl.com/y4ko9j7t
8-20-20: ORAGENICS Signs Dev/Mfg Agreement with Avid for COVID-19 Vaccine “TerraCoV2” https://tinyurl.com/yxqg3w4v
Pg. 39 of CDMO’s 4-30-20 10-K (iss. 6-30-20):
Period Halozyme Gilead Acumen IGM Coherus ADC-Ther. Others
FYE 4-30-14 91% 8%
FYE 4-30-15 79% 9%
FYE 4-30-16 69% 26% 5%
FYE 4-30-17 58% 16% 26% 16%
FYE 4-30-18 55% 22% 9% 14%
FYE 4-30-19 30% <10% <10% 13% 21% 36%
FYE 4-30-20 28% 24% 11% 11% 10% <10% --

Now we have 2 Adds possibly boosting FY21/Guidance
8-20-20: Oragenics Signs Dev/Mfg Agreement with Avid for COVID-19 Vaccine “TerraCoV2” https://tinyurl.com/yxqg3w4v
8-6-20: Iovance Biotherapeutics signs w/Avid(+Aragen) to Dev+Mfg. IOV-3001 (IL-2 Analog) https://tinyurl.com/y2lgzh6x
BioProcIntl: “Iovance said it would focus on GMP manufacturing of IOV-3001 during 2020 and may initiate IND-enabling activities as early as 2021” https://tinyurl.com/y4ko9j7t
= = = = =
6-30-20: Revs Guidance (FY’21 fye 4-30-21): $76-81M
https://tinyurl.com/y9zbgmos
Note: Avid’s FY runs May-Apr, so FY’21 = May’20-Apr’21
~Sep3: Q1/FY21 (qe 7-31-20) CDMO Financials & Conf. Call
Total INST.+LARGE Holdings now 38,627,460 68.4% (6-30-20*)
Up from 36,882,771 65.3% at 3-31-20.
27,049,083 47.9% INSTITUTIONS (a/o 6-30-20*)
https://www.nasdaq.com/symbol/cdmo/institutional-holdings
+ 11,578,377 20.5% LARGE SHAREHOLDERS via Forms13/14 (EasternCap/Dart8-2019, Tappan/12-2019, Millennium/7-2020) – ie, not in Nasdaq Inst. List.
------------
= 38,627,460 68.4%
NOTE1: *Except I picked up Millennium’s 7-15-20 13G +947,319, moving them to 5.3%.
NOTE2: I pulled 2,045,066/Millennium from 6-30-20 Nasdaq Inst. since now 13G/5.3%, so not double-counted (29,094,149 – 2,045,066).
Ownership %’s based on 56,511,294 shares O/S at 6-19-20 (10K: https://tinyurl.com/yak25nco )
12 LARGEST SHAREHOLDERS:
1. Eastern Capital (Kenneth Dart): 4,300,992 7.6% (a/o 8-21-19 14A/Proxy: https://tinyurl.com/y9c972fa – orig. 13G filed 11-2015)
2. Tappan Street (Prasad Phatak): 4,285,000 7.6% (a/o 12-31-19 13G: https://tinyurl.com/wlcwnnv )
3. Blackrock Inc. (Larry Fink): 3,945,988 7.0% (+64,467 a/o 6-30-20 Nasdaq Inst.)
4. IsZo Capital Mgt. (Brian Sheehy): 3,548,032 6.3% (-2,097,669 a/o 6-30-20 Nasdaq Inst.)
5. Millennium Mgt. 2,992,385 5.3% (+2,292,216 a/o 7-15-20 13G: https://tinyurl.com/yydztxkv ; 6-30-20Naz=2,045,066)
6. Vanguard Group 2,679,291 4.7% (+41,246 q/e 6-30-20 Nasdaq Inst.)
7. Snyder Capital 2,304,879 4.2% (+23,974 a/o 6-30-20 Nasdaq Inst.)
8. Altravue Capital 1,933,815 3.4% (-16,540 a/o 6-30-20 Nasdaq Inst.)
9. Wellington Mgt. 1,266,215 2.2% (+561,300 a/o 6-30-20 Nasdaq Inst.)
10. Sargent Invest. Grp. 1,063,099 1.9% (-102,898 a/o 6-30-20 Nasdaq Inst.)
11. State Street 1,058,202 1.9% (+19,529 a/o 6-30-20 Nasdaq Inst.)
12. Silvercrest Asset Mgt. 895,551 1.6% (+159,408 a/o 6-30-20 Nasdaq Inst.)
BIGGEST DROPS q/e 6-30-20:
IsZo Capital Mgt. -2,097,669
Essex Invest. Mgt. -142,763
Sargent Invest. Grp. -102,898
= = = = = = = = = = = =
Shares O/S as of 6-19-20=56,511,294 - history since 4-2006: https://tinyurl.com/yak25nco (at bottom)
...A/O 4-30-20: 1,530,000 stock options outstanding at a wgt.avg. exercise price of $6.77 (pg.50 10K).
6-30-20: Qtly. Conf. Call (Hancock/Hart/Compton) PR & Transcript https://tinyurl.com/y9zbgmos
The big adder is Millennium
7-22-20/13G: Millennium adds +2,292,216sh since 3-31-20, eff. July16 moves to 5.3% stake @2,992,385 shares:
3-31-20: 700,169 Nasdaq Inst.
6-30-20: 2,045,066 +1,344,897 Nasdaq Inst.
7-16-20: 2,992,385 +947,319 13G: 5.3% stake (+2,292,216sh since 3-31-20)
#5. Millennium Mgt. 2,992,385 5.3% (+2,292,216 a/o 7-21-20 13G: https://tinyurl.com/yydztxkv ; in 6-30-20 Nasdaq Inst @2,045,066)
Yes, R2, Eastern Cap.(K.Dart) summarizes to 4,300,992 7.6% per the 14A/Proxy issued 8-19-19. Thanks. Nice that this year's 14A/Proxy is due out soon.
#2. Eastern Capital (Kenneth Dart): 4,300,992 7.6% (a/o 8-21-19 14A/Proxy: https://tinyurl.com/y9c972fa - based on 13G filed 11-2015)
Latest DEF14A/Proxy iss. 8-21-19 (re: 10-9-19 ASM): https://tinyurl.com/y9c972fa
Eastern Capital (Kenneth Dart) 13D/G’s. I assume/haha that if he ever sells his CDMO, we’ll see it here.
https://fintel.io/i/eastern-capital
I think if his CDMO falls below 5%, he’ll have to file a 13D/G. That’s what caused his 8-13-20 IBIO 13D. “On Aug. 12, 2020, the Reporting Persons ceased to be the beneficial owner of more than 5% of the (IBIO) shares of Common Stock of the Issuer.”
#2. Eastern Capital (Kenneth Dart): 4,300,992 CDMO 7.6% (a/o 8-21-19 14A: https://tinyurl.com/y9c972fa - based on 13G filed 11-2015)
I'd remove that, but they told me I can't remove my own posts.
Thanks. Actually, that's the 13G link I have in my post - it's a/o 12-31-19, filed Feb2020.
#3. Tappan Street (Prasad Phatak): 4,285,000 7.6% (a/o 12-31-19 13G: https://tinyurl.com/wlcwnnv )
re: Large+INST, these are the 2 that for me are tough to track/confirm. They are via 13G’s, and not via Nasdaq 13F’s. I honestly am never totally sure we can rely on the latest, often dated, 13G’s that we see. Always something with these things that drive you crazy. If anybody has any insight, please help me.
#2. Eastern Capital (Kenneth Dart): 4,300,992 7.6% (a/o 8-21-19 14A: https://tinyurl.com/y9c972fa - based on 13G filed 11-2015)
#3. Tappan Street (Prasad Phatak): 4,285,000 7.6% (a/o 12-31-19 13G: https://tinyurl.com/wlcwnnv )
================
12 LARGEST SHAREHOLDERS:
1. IsZo Capital Mgt. (Brian Sheehy): 5,645,701 10.0% (+1,425,553 a/o 3-31-20 Nasdaq Inst.)
2. Eastern Capital (Kenneth Dart): 4,300,992 7.6% (a/o 8-21-19 14A: https://tinyurl.com/y9c972fa - based on 13G filed 11-2015)
3. Tappan Street (Prasad Phatak): 4,285,000 7.6% (a/o 12-31-19 13G: https://tinyurl.com/wlcwnnv )
4. Blackrock Inc. (Larry Fink): 3,881,521 6.9% (-52,910 a/o 3-31-20 Nasdaq Inst.)
=>5. Millennium Mgt. (Israel Englander) 2,992,385 5.3% (+2,292,216 a/o 7-21-20 13G: https://tinyurl.com/yydztxkv )
6. Vanguard Group 2,638,045 4.7% (+57,499 q/e 3-31-20 Nasdaq Inst.)
7. Snyder Capital 2,280,905 4.0% (+98,519 a/o 3-31-20 Nasdaq Inst.)
8. Altravue Capital 1,950,355 3.5% (-261,087 a/o 3-31-20 Nasdaq Inst.)
9. Sargent Invest. Grp. 1,165,997 2.1% (+204,415 a/o 3-31-20 Nasdaq Inst.)
10. State Street 1,038,673 1.8% (-21,871 a/o 3-31-20 Nasdaq Inst.)
11. Geode Capital 740,689 1.3% 42,493 (+42,493 a/o 3-31-20 Nasdaq Inst.)
12. Silvercrest Asset Mgt. 895,551 1.3% (+159,408 a/o 6-30-20 Nasdaq Inst.)
=======
UPDATE 7-21-20/13G: Millennium Mgt.(Israel Englander) adds +2,292,216sh, now 5.3% stake w/2,992,385sh. This moves INST.+LARGE to 39,174,987 69.3%. (7-22-2013G: https://tinyurl.com/yydztxkv )
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=157096216
Silvercrest Asset Mgt. added +159,408 to 895,551 @6-30-20, after adding +173,411 during the 3-31-20 qtr. 13F: https://fintel.io/so/us/cdmo
Large+INST: https://investorshub.advfn.com/boards/read_msg.aspx?message_id=157096216
What would a cv19 contract do now? @June30, they were talking with “most of the people”, and “looking at contracts”, and “anticipating hearing back from some of the opportunities”… I still think that’s at least 3. Click on just ONE, St. Nick...
6-30-20 Qtly CC-Transcript https://tinyurl.com/y9zbgmos
Joe Pantginis (H.C. Wainwright), “...going back to the potential COVID contracts…”
RICK HANCOCK (Int. CEO): So the contracts we're looking at currently, exclusivity is not really a part of it. The main drivers to get something through proof of concept as quickly as possible and I think one of the things that makes Avid an attractive partner for these kinds of things is, while we don't have the largest scale, some of them are requiring very small doses or very small amounts of material per dose, but the fact that we could take something very rapidly from proof of concept to commercial production with our many years of commercial capability. Just my opinion, I think the strategy of most of the people that we're talking to, based on where they are getting their funding from, the goal is to identify something that works either as a vaccine or therapeutic and then make it as widely available as possible.
TIM COMPTON (COO): It's very much timeline driven and certainly the exclusivity in those types of legal agreements have not been engaged or discussed on. It's really about delivering and timeline to deliver.
6-30-20: Matt Hewitt (Craig-Hallum), ”but given these COVID opportunities, how quickly would you anticipate hearing back…”
TIM COMPTON (COO): ...every opportunity is different, every timeline is different, but we certainly anticipate hearing back from some of the opportunities that we have in the queue for COVID probably in the next - probably next 90 days, but there are several opportunities in various stages of the sales cycle.
8-11-20/BioProcIntl: “Iovance Hires Avid To Make IL-2 Receptor Antagonist Cancer Candidate [IOV-3001]”
Followup recap to Avid’s 8-6-20 Iovance announcement...
https://bioprocessintl.com/bioprocess-insider/facilities-capacity/iovance-hires-avid-to-make-il-2-receptor-antagonist-cancer-candidate/
By Gareth Macdonald, BioProcess Intl. (div. of Informa)
Avid Bioservices will make IOV-3001, Iovance Therapeutics’ candidate interleukin-2 receptor agonist intended for the treatment of cancer. Avid will commence analytical activities, upstream and downstream process development, and pilot-scale non-GMP manufacturing for IOV-3001.
The firm will also work with Aragen Biosciences, which is carrying out cell line development activities. The next stage will see Avid manufacture the drug at its Myford facility in Tustin, CA. News of the contract comes just a month after Avid said it planned to increase mfg. capacity at the facility over the next few years [6-30-20: https://tinyurl.com/y9zbgmos ].
IND PLAN
Iovance licensed development rights to IOV-3001 from Novartis in January. The protein is an engineered IL-2 CDR graft that targets IL2R beta-gamma-expressing cells and limits IL2R alpha-beta-gamma-dependent Treg activation. At the time said it would focus on GMP manufacturing of IOV-3001 during 2020 and may initiate IND-enabling activities as early as 2021.
ALLIANCES
Avid CCO Timothy Compton welcomed the deal. He also said, “This new contract award also illustrates the value of Avid’s efforts to establish alliances with key providers of complementary CDMO services such as Aragen.”
Avid and Aragen teamed up to offer combined cell line and process development in May [5-6-20:https://tinyurl.com/ycuu2m5r ] . Under the non-exclusive deal, the companies will offer customers Aragen’s cell line development expertise integrated with Avid’s upstream and downstream process development and analytical services.
At the time an Avid spokesman said, “Many of our prospect customers are requesting expedited timelines to GLP-Tox pilot batch and cGMP drug substance deliveries for their early stage programs. “Those early-stage prospects are also requesting cell line development bundled with process development, pilot manufacturing and cGMP manufacturing. Likewise, Aragen tells us that their customers have similar requests for later phase process development and manufacturing services. For those reasons, we believe it makes sense to team up with Aragen to co-promote an integrated offering.”
*end*
= = = = = = = =
8-6-20: Iovance Biotherapeutics signs w/Avid(+Aragen) to Dev+Mfg. IOV-3001 (IL-2 Analog) https://tinyurl.com/y2lgzh6x
Iovance(IOVA) mktcap=$4.5B, licensed IOV-3001 from Novartis.
1-13-20: “Iovance Biotherapeutics Secures IOV-3001 (Novel IL-2 Analog) Licensing From Novartis”
https://www.globenewswire.com/news-release/2020/01/12/1969205/0/en/Iovance-Biotherapeutics-Obtains-License-to-Develop-and-Commercialize-a-Novel-IL-2-Analog.html
https://finance.yahoo.com/quote/IOVA?p=IOVA
5-6-20: Avid Teams with Aragen to speed up drug dev./delivery timelines https://tinyurl.com/ycuu2m5r
7-28-20: Avid Teams with Argonaut to add “Parenteral Fill-Finish Services” https://tinyurl.com/y2lgzh6x
Well, I suspected that Yahoo Float=39.5M was wrong. I found Marketwatch.com that says OS=56.51M FLOAT=56.05M. That makes a lot more sense.
https://www.marketwatch.com/investing/stock/cdmo
Float is (OS – Restricted); ie, the #shares publicly available for trading.
BAD YAHOO SAYS Float=39.52M: https://finance.yahoo.com/quote/CDMO/key-statistics?p=CDMO
= = = = = = = = = = = = =
10K/4-30-20 iss. 6-30-20: https://tinyurl.com/yak25nco
Total INST.+LARGE Holdings: 36,882,771 65.3% a/o 3-31-20 – See https://investorshub.advfn.com/boards/read_msg.aspx?message_id=155675008
**UPDATE 7-22-20/13G: Millennium Mgt.(Israel Englander) adds +2,292,216sh, now 5.3% stake w/2,992,385sh.
....This moves INST.+LARGE to 39,174,987 69.3%
Note: Ownership %’s based on 56,511,294 shares o/s. common O/S at 6-19-20 (10K)
Iovance(IOVA) mktcap=$4.5B, licensed IOV-3001 from Novartis.
1-13-20: “Iovance Biotherapeutics Secures IOV-3001 (Novel IL-2 Analog) Licensing From Novartis”
https://www.globenewswire.com/news-release/2020/01/12/1969205/0/en/Iovance-Biotherapeutics-Obtains-License-to-Develop-and-Commercialize-a-Novel-IL-2-Analog.html
https://finance.yahoo.com/quote/IOVA?p=IOVA
CDMO Float = 39.52M shares. I know that Float is (OS – Restricted); ie, the #shares publicly available for trading. But, I never saw it listed anywhere, and I honestly can’t figure it out from the 10Q/K’s.
Well today, I found a Yahoo page that lists it – presumably(?) it’s right...
https://finance.yahoo.com/quote/CDMO/key-statistics?p=CDMO
It’s showing:
Shares OS: 56.51M
Float: 39.52M
So, Restricted = 17M. Where they came up with that I don’t know.
= = = = = = = = = = = = =
10K/4-30-20 iss. 6-30-20: https://tinyurl.com/yak25nco
Total INST.+LARGE Holdings: 36,882,771 65.3% a/o 3-31-20 – See https://investorshub.advfn.com/boards/read_msg.aspx?message_id=155675008
**UPDATE 7-22-20/13G: Millennium Mgt.(Israel Englander) adds +2,292,216sh, now 5.3% stake w/2,992,385sh.
....This moves INST.+LARGE to 39,174,987 69.3%
St.Nick Green is Avid’s CEO today, his LinkedIN was updated yesterday.
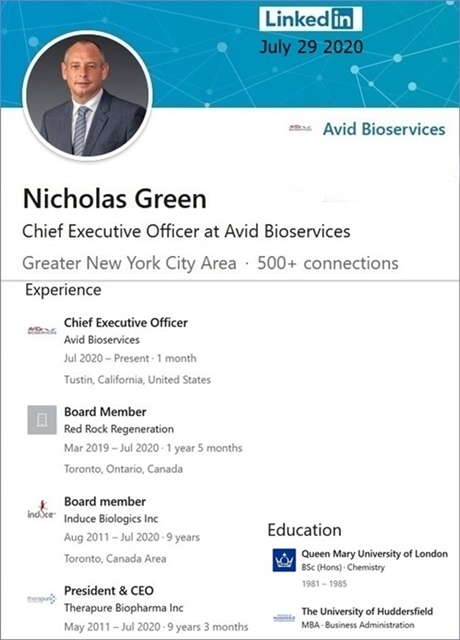
6-23-20: Nicholas Green (ex-Therapure Biopharma) to become President/CEO of Avid Bioservices (eff. 7-30-2020)
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=156481310
7-30-20 updated BOD pic:
Nick Green 2014 Interview. He becomes Avid’s CEO on 7-30-20.
March 2014: CMO Leadership Awards 2014 - Therapure Biopharma
7-22-20/13G: Millennium adds +2,292,216sh, eff. July16 moves to 5.3% stake @2,992,385 shares. Keeping the Nasqaq Inst. figures at 3-31-20 (most won’t update to 6-30-20 until Aug15), these are the latest known Large Ownership figures:
Total INST.+LARGE Holdings now 39,174,987 69.3% (3-31-20 + 7-22-20/Millenium 13G)
28,296,779 50.1% INSTITUTIONS (a/o 3-31-20)
https://www.nasdaq.com/symbol/cdmo/institutional-holdings
+ 10,878,208 19.2% LARGE SHAREHOLDERS via Forms13/14 (EasternCap/Dart8-2019, Tappan/12-2019, Millennium/7-2020) – ie, not in Nasdaq Inst. List.
------------
= 39,174,987 69.3%
Note: Ownership %’s based on 56,511,294 shares o/s. common O/S at 6-19-20 (10K: https://tinyurl.com/yak25nco )
12 LARGEST SHAREHOLDERS:
1. IsZo Capital Mgt. (Brian Sheehy): 5,645,701 10.0% (+1,425,553 a/o 3-31-20 Nasdaq Inst.)
2. Eastern Capital (Kenneth Dart): 4,300,992 7.6% (a/o 8-21-19 14A: https://tinyurl.com/y9c972fa - based on 13G filed 11-2015)
3. Tappan Street (Prasad Phatak): 4,285,000 7.6% (a/o 12-31-19 13G: https://tinyurl.com/wlcwnnv )
4. Blackrock Inc. (Larry Fink): 3,881,521 6.9% (-52,910 a/o 3-31-20 Nasdaq Inst.)
=>5. Millennium Mgt. (Israel Englander) 2,992,385 5.3% (+2,292,216 a/o 7-21-20 13G: https://tinyurl.com/yydztxkv )
6. Vanguard Group 2,638,045 4.7% (+57,499 q/e 3-31-20 Nasdaq Inst.)
7. Snyder Capital 2,280,905 4.0% (+98,519 a/o 3-31-20 Nasdaq Inst.)
8. Altravue Capital 1,950,355 3.5% (-261,087 a/o 3-31-20 Nasdaq Inst.)
9. Sargent Invest. Grp. 1,165,997 2.1% (+204,415 a/o 3-31-20 Nasdaq Inst.)
10. State Street 1,038,673 1.8% (-21,871 a/o 3-31-20 Nasdaq Inst.)
11. Geode Capital 740,689 1.3% 42,493 (+42,493 a/o 3-31-20 Nasdaq Inst.)
12. Silvercrest Asset Mgt. 736,143 1.3% (+173,411 a/o 3-31-20 Nasdaq Inst.)
= = = = = = = = = = =
6-30-20: Qtly. Conf. Call (Hancock/Hart/Compton) PR & Transcript https://tinyurl.com/y9zbgmos
...RickH: "Based on our customers' growing demand and our expanding business development activity, we believe that we will significantly increase capacity utilization in 2021 and beyond. Accordingly, we have entered into a new phase of planning for the expansion that will support our growing business in the years to come.”
...”Top10 Pharma customer added 7-2019 (https://tinyurl.com/yyq8zgb9) named in 10-K: GILEAD SCIENCES.”
New Avid job add. About 3 weeks ago 14, then down to 9. Now, this new one, so 10 openings:

https://avidbio.applicantpro.com/jobs/
Of course, I had to Google ELISA.
Somebody explain it to me please.
Probably just a normal CDMO-type job – I’m just killing time...
*What is an ELISA (enzyme-linked immunosorbent assay)?
ELISA (enzyme-linked immunosorbent assay) is a plate-based assay technique designed for detecting and quantifying soluble substances such as peptides, proteins, antibodies, and hormones. In an ELISA, the antigen (target macromolecule) is immobilized on a solid surface (microplate) and then complexed with an antibody that is linked to a reporter enzyme. Detection is accomplished by measuring the activity of the reporter enzyme via incubation with the appropriate substrate to produce a measurable product. The most crucial element of an ELISA is a highly specific antibody-antigen interaction.
Oops, Three, I forget they didn't name GILEAD in the 7-8-2019 new customer PR. They just called them a "Top10 Global Pharma".
We didn't learn that it was Gilead until the 4-30-20 10-K, issued 6-30-20. So, I see your point - I guess it could have been that Gilead said, don't PR our name. ??
= = = = = = = = =
7-8-19/PR: Avid Signs New Top10 Global Pharma [GILEAD] https://tinyurl.com/yyq8zgb9
They PR’d the Gilead add pretty solidly July2019, but yes, I know that there are many new customer adds that they don’t PR. I think some customers will not let them PR it, but not sure how prevalent that is.
7-8-19/PR: Avid Signs New Top10 Global Pharma [GILEAD] https://tinyurl.com/yyq8zgb9

Again, Gilead was 24% (v. 28% Halo) for 12mos FY20 (May19-Apr20), not for Q4.
But since the Gilead add was PR’d 7-8-19, and normally new customers don’t gen. Revs for at least 6 mos. (correct me if I’m wrong - that’s what I remember them saying), then Gilead’s 24% was only for 4-5 months of the 12.
...No telling what Gilead’s % for the last 3 or 6 months v. Halo would be.
I wish they would post Cust splits in the 10Q’s - they used to, but stopped 3-4 years ago.
3’s, Gilead was 24% (v. 28% Halo) for 12mos FY20 (May19-Apr20), not for Q4.
But since the Gilead add was PR’d 7-18-19, and normally new customers don’t gen. Revs for at least 6 mos. (correct me if I’m wrong - that’s what I remember them saying), then Gilead’s 24% was only for 4-5 months of the 12.
...No telling what Gilead’s % for the last 3 or 6 months v. Halo would be.
I wish they would
post Cust splits in the 10Q’s - they used to, but stopped 3-4 years ago.
7-1-20/PharmasAlmanac: “Accelerating COVID-19 Programs with Comprehensive Support from Cell Line to Commercial Production at a Single Site”
by Magnus Schroeder, PhD, VP/ProcessDev, Avid Bioservices
https://www.pharmasalmanac.com/articles/accelerating-covid-19-programs-with-comprehensive-support-from-cell-line-to-commercial-production-at-a-single-site
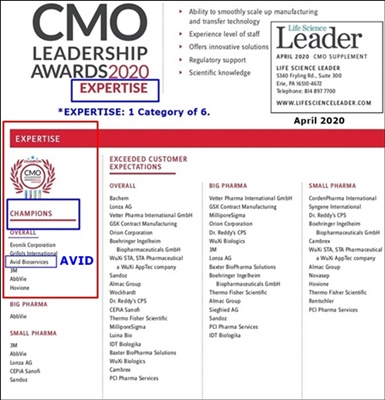
Excerpt:
“In March 2020, Avid Bioservices was honored to receive 2020 Contract Manufacturing Organization (CMO) Leadership Awards (https://tinyurl.com/yb5cnh6m ) for Capabilities, Expertise, Reliability, Compatibility, and Service based on market research and surveys conducted by Industry Std. Research (ISR) and presented by industry publication Life Science Leader. We were also named a 2020 CMO Leadership Award Champion in the categories of Expertise & Service.
These awards are truly gratifying, because Avid’s entire business philosophy is built upon the concept of doing whatever it takes to deliver for our customers. We work to an incredibly high standard, with our teams taking real ownership of client programs and treating them as if they were internal projects.
They also underscore the key attributes of Avid as a CDMO well-positioned to help companies responding to the COVID-19 pandemic. We can support the process development and manufacture of traditional and novel potential therapies. We can rapidly advance candidates all the way to commercialization from a single site, eliminating complex and time-consuming site transfers…”

= = = = = =
5-18-20: Avid Listed In Top CMO Awards Article https://tinyurl.com/yb5cnh6m
7-8-19/PR: Avid Signs New Top10 Global Pharma [GILEAD] https://tinyurl.com/yyq8zgb9

7-3-20/BioProcINTL: “Avid Preps To Kick-Off $30-40m Mfg. Expansion”
by Dan Stanton (Editor - BioProcess Insider, part of Informa Life Sciences' BioProcess Intl.)
https://bioprocessintl.com/bioprocess-insider/facilities-capacity/avid-preps-to-kick-off-30-40m-manufacturing-expansion/
= = = = = = =6-30-20 CC re: cv19 https://tinyurl.com/y9zbgmos
6-30-20: Joe Pantginis (H.C. Wainwright), “...going back to the potential COVID contracts…”
RICK HANCOCK (Int. CEO): So the contracts we're looking at currently, exclusivity is not really a part of it. The main drivers to get something through proof of concept as quickly as possible and I think one of the things that makes Avid an attractive partner for these kinds of things is, while we don't have the largest scale, some of them are requiring very small doses or very small amounts of material per dose, but the fact that we could take something very rapidly from proof of concept to commercial production with our many years of commercial capability. Just my opinion, I think the strategy of most of the people that we're talking to, based on where they are getting their funding from, the goal is to identify something that works either as a vaccine or therapeutic and then make it as widely available as possible.
TIM COMPTON (COO): It's very much timeline driven and certainly the exclusivity in those types of legal agreements have not been engaged or discussed on. It's really about delivering and timeline to deliver.
6-30-20: Matt Hewitt (Craig-Hallum), ”but given these COVID opportunities, how quickly would you anticipate hearing back…”
TIM COMPTON (COO): ...every opportunity is different, every timeline is different, but we certainly anticipate hearing back from some of the opportunities that we have in the queue for COVID probably in the next - probably next 90 days, but there are several opportunities in various stages of the sales cycle.
7-3-20/BioProcINTL: “Avid Preps To Kick-Off $30-40m Mfg. Expansion”
by Dan Stanton (Editor - BioProcess Insider, part of Informa Life Sciences' BioProcess Intl.)
https://bioprocessintl.com/bioprocess-insider/facilities-capacity/avid-preps-to-kick-off-30-40m-manufacturing-expansion/
. . .RICHARD HANCOCK (Int. CEO): “Our customer relationships are strong and forecasted demand is growing. Combined with our expanded business development activity, which we expect to generate an increasing number of projects from both existing and new customers, we believe that we are on a path to significantly increased capacity utilization. Accordingly, we are readying to kick-off an expansion that will allow us to meet the increasing demand of our ever-growing customer list. This concludes my prepared remarks for today.” . . .
. . .CFO DANIEL HART added that these initial steps require a low outlay of capital expenditure but does allow the CDMO to pull the trigger on construction at any moment. “Once we find the right time and move forward toward building out, at that time, we’ll look at any financing options that we feel are appropriate when the time presents itself.” . . .
. . .CCO TIMOTHY COMPTON: “the overall build-out would be somewhere around $30-40M all-in” and would take 18-24 months to be completed.
= = = = = = = = = =
6-30-20: Qtly. Conf. Call (Hancock/Hart/Compton) PR & Transcript https://tinyurl.com/yak25nco
...”Top10 Pharma customer added 7-2019 named in 10-K: GILEAD SCIENCES.”
BTW, Gilead add PR’d 7-8-2019. https://tinyurl.com/yyq8zgb9
...I’ve always heard that min. 6 mos. for revenues from a new cust. Thus, Gilead’s 24% of total FYE 4-30-20 business only included them for 3-4 months.

Thanks for finding Matt Hewitt’s(Craig-Hallum) Q4 comments, BB1.
ANALYST COVERAGE: http://ir.avidbio.com/analyst-coverage
Also see: https://www.marketbeat.com/stocks/NASDAQ/CDMO/price-target
HOPEFULLY THESE ARE ACCURATE:
Janney Montgomery Scott - Paul Knight PT=$10
H.C. Wainwright & Co. - Joseph Pantginis PT=$9
Craig-Hallum Capital Group - Matt G. Hewitt PT=$10
First Analysis Securities - Joseph Munda PT=$7.50
Stephens Inc. - Jacob Johnson PT=10
= = = = = =CRAIG-HALLUM (Matt G. Hewitt) - PT=$10
7-2-20: CDMO Reit. Buy - PT=$10
https://www.tipranks.com/news/article/analysts-say-these-3-stocks-are-their-top-picks-for-post-pandemic-gains-2/
(thanks to iHub poster Bigbro1 for finding)
= = = = = =STEPHENS (Jacob Johnson) - PT=$10
7-1-20: CDMO 4Q20 Wrap Up: Momentum Building, Reit. OW - PT=$10
https://www.stephens.com/institutional-equities-and-research/research/industries-and-companies/all-companies/avid-bioservices-inc/
= = = = = =H.C. WAINWRIGHT (Joseph Pantginis) - PT=$9:
7-1-20: HC Wainwright Reiterates Buy Rating for Avid Bioservices
https://www.marketbeat.com/instant-alerts/nasdaq-cdmo-a-buy-or-sell-right-now/
(thanks to iHub poster Cheynew for finding)
Re: cv19 (I don’t capitalize things I hate), I think that “most of the people that we're talking to” and “several opportunities” means at least 3. I don’t think you put those ways if it’s 2 (it’s def. not one!). I'd like to see what just one cv19 strike would do to $76-81 and margins and profitability for FY21.
6-30-20: Joe Pantginis (H.C. Wainwright), “...going back to the potential COVID contracts…”
RICK HANCOCK (Int. CEO): So the contracts we're looking at currently, exclusivity is not really a part of it. The main drivers to get something through proof of concept as quickly as possible and I think one of the things that makes Avid an attractive partner for these kinds of things is, while we don't have the largest scale, some of them are requiring very small doses or very small amounts of material per dose, but the fact that we could take something very rapidly from proof of concept to commercial production with our many years of commercial capability. Just my opinion, I think the strategy of most of the people that we're talking to, based on where they are getting their funding from, the goal is to identify something that works either as a vaccine or therapeutic and then make it as widely available as possible.
TIM COMPTON (COO): It's very much timeline driven and certainly the exclusivity in those types of legal agreements have not been engaged or discussed on. It's really about delivering and timeline to deliver.
6-30-20: Matt Hewitt (Craig-Hallum), ”but given these COVID opportunities, how quickly would you anticipate hearing back…”
TIM COMPTON (COO): ...every opportunity is different, every timeline is different, but we certainly anticipate hearing back from some of the opportunities that we have in the queue for COVID probably in the next - probably next 90 days, but there are several opportunities in various stages of the sales cycle.
https://tinyurl.com/y9zbgmos
DJohn, you're a funny man.
Stephens (Jacob Johnson) Reits OW Target=$10
7-1-20: CDMO 4Q20 Wrap Up: Momentum Building, Reit. OW-Vol./$10 PT
https://www.stephens.com/institutional-equities-and-research/research/industries-and-companies/all-companies/avid-bioservices-inc/
ANALYST COVERAGE: http://ir.avidbio.com/analyst-coverage
Also see: https://www.marketbeat.com/stocks/NASDAQ/CDMO/price-target
HOPEFULLY THESE ARE ACCURATE:
Janney Montgomery Scott - Paul Knight PT=$10
H.C. Wainwright & Co. - Joseph Pantginis PT=$9
Craig-Hallum Capital Group - Matt G. Hewitt PT=$11
First Analysis Securities - Joseph Munda PT=$7.50
Stephens Inc. - Jacob Johnson PT=10
= = = = = =
TRANSCRIPT 6-30-20 FY20/Q4 Earnings Conf. Call (fye 4-30-20):
Rick Hancock(Interim CEO), Daniel Hart(CFO), Tim Compton(CCO)
Q&A Analysts:
Matt Hewitt - Craig-Hallum Capital
Joe Pantginis - H.C. Wainwright
JACOB JOHNSON – STEPHENS
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=156637714
I got a kick out of Rick Hancock’s sense of humor yesterday. Responding to Joe Pantginis’ asking about the selection process of hiring Nick Green: “So, obviously, this wasn't a lightening fast process...” - haha.
Int. CEO Rick Hancock – 6-30-20 Q&A
“You bet, Joe. Great question. So, obviously, this wasn't a lightening fast process, but I really think at the end of the day, we wound up with just the exact right person to lead Avid for the future based on Nick's tremendous experience both in the commercial realm, operational excellence, strategic growth. So we're very lucky to get Nick and very happy that he's joining us, and I spoke with him just today and he expressed again how excited he is to get here. I think his vision is really a continuation of what the senior management team here has been doing, making sure that we capitalize on the very unusually exciting asset that we have here. Not many independent CDMOs in our space that can do what we can do. Again, going from proof of concept, all the way through to commercialization and post-commercial support.
So I think he's delighted to get here to a fast-moving nimble company and as we pointed out, he's built up and expanded many companies over his career. So, I think his first focus is going to be on operational excellence throughout the organization, making sure that we're running lean, looking to build and strengthen with Tim Compton, our relationships with existing customers and bringing on some more marquee clients and then focusing on the build out and just making sure we capitalize on all the growth potential that we have.”
https://www.fool.com/earnings/call-transcripts/2020/06/30/avid-bioservices-inc-cdmo-q4-2020-earnings-call-tr.aspx
= = = = = =
6-23-20: Nicholas Green (ex-Therapure Biopharma) becomes President/CEO of Avid Bioservices (eff. 7-30-2020) https://tinyurl.com/yczapcc7
Thanks, WH. Doing good. Even better after yesterday.
6-30-20 Qtly CC-Transcript, PR(fye4-30-20), Avid Revs History Table
*Revs Guidance (FY’21 fye 4-30-21): $76-81M. 4-30-20 Backlog=$65M
*Cash: 4-30-20: $36.3M
*As of 6-19-20: 56,511,294 shares o/s.
*10K/4-30-20 iss. 6-30-20: https://tinyurl.com/yak25nco
*Avid Total Revs May03-Apr20: $408.9M
*Avid’s website: https://avidbio.com (A/o 4-30-20, 222 full-time & 5 part-time emps)
This large post has 4 sections:
I. 6-30-20 Qtly. Earnings Conf. Call TRANSCRIPT (FY20/Q4 fye 4-30-20)
II. 6-30-20 CDMO Press Release: Q4/FY20 Earnings & Developments
IV. Updated Table of Avid Revenues By Quarter (May’06-Current)
III. Updated O/S Shares History Table – 2006-curr.
…Recall: Avid’s FY runs May-Apr, so FY’21 = May’20-Apr’21.
TRANSCRIPT 6-30-20 FY20/Q4 Earnings Conf. Call (fye 4-30-20):
Rick Hancock(Interim CEO), Daniel Hart(CFO), Tim Compton(CCO)
Q&A Analysts:
Matt Hewitt - Craig-Hallum Capital
Joe Pantginis - H.C. Wainwright
Jacob Johnson - Stephens
Link to webcast replay: http://ir.avidbio.com/events-and-presentations => https://edge.media-server.com/mmc/p/bt2399kr
CC TRANSCRIPT EXTRACTS – PREPARED REMARKS BY INT. CEO RICK HANCOCK:
OPENING:
“Thank you to everyone who has dialed in and to those who are participating today via webcast. Before addressing the company's financial and operational performance, I'd first like to welcome Nicholas Green to the Avid team. Nick has recently been appointed Avid's new CEO [6-23-20/PR: https://tinyurl.com/yczapcc7 ] and we are thrilled to have him onboard. Nick has more than 30 years of experience in the global pharmaceutical and healthcare services industry with significant expertise in the contract manufacturing of novel pharmaceutical products. His global pharmaceutical experience spans four continents having run 31 facilities in 9 countries in North America, South America, Europe, and Asia. Throughout his career, Nick has held a number of senior management roles for several contract manufacturing organizations and life science companies where he is credited with successfully building and expanding those businesses and delivering significant value for customers, patients, employees, and other stakeholders. We are very pleased to have Nick join Avid and we believe his experience will be highly valuable as the company grows both commercially and operationally. Nick's start date is July 30th, 2020. With his arrival, I will no longer be part of the company's day-to-day operations, but I will continue to support Avid as the Director on the company's Board and I look forward to working with Nick and this team going forward.
I'll now address the quarter. Avid's performance during Q4 partially offset the lower than expected revenues recorded during Q3. As discussed on our last earnings call, during Q3, the company experienced a problem with a specific piece of equipment. This resulted in a production interruption during Q3 and a deferral of some revenue for that period. While the interruption extended into Q4, additional unplanned revenue generating projects were initiated during the period, allowing the company to exceed its restated annual revenue guidance and end the year with a strong backlog. We are pleased to report that during Q4, we identified and mitigated the cause of the equipment problem responsible for this production interruption and we are currently in the process of confirming the successful remediation. During Q4, we also expanded our business development team and entered into a co-marketing partnership that we believe will provide another channel from which to fill capacity. Based on our customers' growing demand and our expanding business development activity, we believe that we will significantly increase capacity utilization in 2021 and beyond. Accordingly, we have entered into a new phase of planning for the expansion that will support our growing business in the years to come.”
POST HART/COMPTON:
“As a continuation of Tim's comments, I'd first like to address Avid's operations in context of the COVID-19 pandemic. Since the beginning of the pandemic, Avid management has closely monitored our production pipeline and the health of our workforce. We are pleased that to date, we have observed no material impact to our production programs for our supply chain and our employees remain healthy and highly productive. Management is taking every precaution and following state and local guidelines to ensure the continued safety and well-being of our team members and we are hopeful that our operations will remain uninterrupted. I'd like to reiterate that no work has been canceled or delayed due to the pandemic and we currently have no expectations that, that will change.
I will now comment further regarding last quarter's equipment issue and production interruption. Let me first confirm that the specific piece of equipment in question is now operational. However, this project is being executed in 3 phases: investigation, remediation, and confirmation. To date, we believe that we have successfully completed the first 2 stages by identifying and remediating the source of the problem. The company is currently progressing through the confirmation stage during which it is running multiple revenue generating production campaigns. While we are optimistic, the company will not be able to confirm the full resolution until it completes the confirmation stage, which we expect will be completed in the coming months.
As we look ahead, we believe that growing customer demand and our expanding business development activity will result in a need for additional capacity. In recent quarters, we have done much of the ancillary work that is required to support a more comprehensive expansion in the future. As we've addressed in prior calls, this work has included the enhancement of key systems and upgrades to general infrastructure. However, we are now taking additional steps to prepare for the growth opportunities we see ahead. While specific kick-off date has not been set for this expansion, we recognize that construction may take 18-24 months to complete. For that reason, we are undertaking the pre-engineering, design, and permitting work required to allow us to break ground on this project at the appropriate time.
Given this backdrop, I'm very pleased to announce that for FY2021, we expect to record revenue of $76-$81 million, representing growth of approx. 27-36% over FY2020 and as we continue to achieve revenue growth, we expect the improvement in margins to track accordingly.
In closing, I'd like to communicate our strong optimism for FY2021 and beyond. Despite the challenges of the third and fourth quarters of fiscal 2020, we finished strong with higher revenues than expected and a record-breaking backlog of business. We are preparing to welcome our new CEO, Nick Green, and we are confident that he will provide the vision and execution strategy to elevate Avid to the next level.
Our customer relationships are strong and forecasted demand is growing. Combined with our expanded business development activity, which we expect to generate an increasing number of projects from both existing and new customers, we believe that we are on a path to significantly increased capacity utilization. Accordingly, we are readying to kick-off an expansion that will allow us to meet the increasing demand of our ever-growing customer list. This concludes my prepared remarks for today. We can now open the call up to questions.”
READ the entire CC Transcript here:
https://www.fool.com/earnings/call-transcripts/2020/06/30/avid-bioservices-inc-cdmo-q4-2020-earnings-call-tr.aspx
= = = = = = = = = = = = = = = = = = = = = == = = =
6-30-20/PR: Avid Bioservices Reports Financial Results for Q4 FYE April 30, 2020, and Recent Developments
https://www.globenewswire.com/news-release/2020/06/30/2055808/0/en/Avid-Bioservices-Reports-Financial-Results-for-Fourth-Quarter-and-Fiscal-Year-Ended-April-30-2020-and-Recent-Developments.html
-- Appointed Nicholas Green as President & Chief Executive Officer
-- Recorded Q4 Revenue of $12.6 Million and Annual Revenue of $59.7 Million
-- Signed $23 Million in Business Orders and Achieved Record Backlog of $65 Million During Q4
-- Project Fiscal 2021 Revenue of $76-$81 Million
TUSTIN, June 30, 2020: Avid Bioservices, Inc. (NASDAQ:CDMO/CDMOP), a dedicated biologics contract development and manufacturing organization (CDMO) working to improve patient lives by providing high quality development and manufacturing services to biotechnology and pharmaceutical companies, today announced financial results for Q4 and full FY 2020, ended April 30, 2020.
HIGHLIGHTS SINCE JANUARY 31, 2020
“Q4 of fiscal 2020 was a very productive period, generating higher than anticipated revenues,” said Rick Hancock, Interim President and CEO of Avid. “As discussed previously, the production interruption and revenue deferral that took place during the third quarter of fiscal 2020 required the company to lower revenue guidance for the year. While this interruption caused us to fall short of our originally stated revenue guidance, the productivity during Q4 allowed us to exceed our re-stated guidance for the FY, sign $23 million in project expansion orders with existing customers during Q4, and end the FY with a strong backlog.
“Avid also significantly strengthened its management team in recent weeks. First, we were pleased to announce the appointment of Nicholas Green, as the company’s new President and CEO. Nick, who will join the team on July 30, comes to Avid with many years of experience driving growth and expansion for multiple key CDMOs around the world, and we are very pleased to have him leading the company’s future strategy.
[SEE: 6-23-20/PR: https://tinyurl.com/yczapcc7, and 2014 interview while CEO at Therapure: https://youtu.be/c3eRQ5M05i4 ]
“During Q4, the company also added two senior directors to our business development team with proven track records in growing CDMOs. They are responsible for driving business growth in North America, Europe and Asia. This expanded and highly experienced business development team has quickly elevated our level of engagement with both existing customers and prospective customers. Additionally, our new team is in discussions with multiple parties regarding potential novel programs to combat COVID-19, and we hope to have the opportunity to support the fight against this global pandemic. Based on this sharp increase in business development activity, combined with the growth of existing customer forecasts, we expect to significantly increase our capacity utilization in the quarters ahead.
“To accommodate this anticipated increase in demand, during Q4, the company initiated the pre-engineering, design and permitting work required to allow us to break ground on a facility expansion at the appropriate time. While a specific kick-off date has not yet been established for this expansion, we believe that customer demand will require additional capacity and we are actively moving forward to prepare for this growth opportunity.
“Regarding the equipment issue and production interruption that we reported last quarter, I can confirm that the specific piece of equipment in question is now operational. However, this project is being executed in three phases: investigation, remediation, and confirmation. We are currently progressing though the confirmation stage during which we are running multiple revenue-generating production campaigns to confirm the successful remediation of the equipment issue. We expect this work to be completed in the coming months.
“While we faced a number of challenges in fiscal 2020, we are very encouraged by the events of, and subsequent to, Q4. In particular, we are pleased to welcome a new and highly experienced CEO to lead Avid into its next phase of growth. During Q4, we also significantly strengthened our business development team, signed multiple project expansion orders with existing customers, and finished the year with a record $65 million backlog. The company continues to have strong relationships with its customers as evidenced by their forecasted growth in demand for fiscal 2021 and beyond, and we expect the number of projects from new customers to grow nicely moving forward. Finally, in the era of COVID-19, it is important to note that Avid, as a critical supplier, has lost no signed business during this period and continues to operate at full-scale while maintaining social distancing requirements. This is due in large part to our exceptional employees who have been diligent in their daily work and who remain productive and committed to excellence despite the hardships posed by the pandemic. We expect each of these factors to contribute to meaningful growth in fiscal 2021.”
FINANCIAL HIGHLIGHTS AND GUIDANCE
The company is providing revenue guidance for the full FY 2021 of $76 to $81 million.
Revenues for Q4 of fiscal 2020 were $12.6 million, a 26% decrease compared to revenues of $17.1 million recorded during Q4 of fiscal 2019. The year-over-year decline in revenue was primarily attributable to the interruption of production which began during the third quarter and continued into Q4 of fiscal 2020, as a result of a problem with a specific piece of equipment. Despite this decline, fourth quarter 2020 revenues were stronger than expected based on our revised guidance for the quarter due primarily to an increase in customer projects. For the full FY 2020, revenues were $59.7 million, an 11% increase as compared to revenues of $53.6 million in the prior year period. This increase was primarily the result of continued growth in the number and scope of customer projects.
As of April 30, 2020, revenue backlog was $65 million, an increase of 12% compared to $58 million at the end of the third quarter of fiscal 2020, and an increase of 41% compared to $46 million at the end of last FY. The company expects to recognize the majority of this backlog during fiscal 2021.
Gross margin for Q4 of fiscal 2020 was a negative 10% compared to a gross margin of 21% for Q4 of fiscal 2019. The decrease in gross margin for the 2020 quarter was primarily attributed to the costs associated with the production interruption described earlier, an increase in depreciation expense from the acquisition of new equipment, and a net decrease in revenue. Gross margin for the full FY 2020 was 7%, a decrease compared to 13% in the prior year period. This decrease was primarily due to higher facility and equipment related costs primarily associated with the production interruption during the second half of fiscal 2020, the planned growth costs associated with payroll and related costs, and an increase in depreciation expense from the acquisition of new equipment.
Selling, general and administrative expenses (“SG&A”) for Q4 of fiscal 2020 were $3.5 million, a slight decline compared to $3.6 million recorded for Q4 of fiscal 2019. For the full FY 2020, SG&A expenses were $14.5 million, a 13% increase compared to $12.8 million for the prior year. The increase in SG&A for the year was primarily attributed to employee separation-related expenses, recruiting fees and increased stock-based compensation. When excluding the separation-related expenses, SG&A increased 3% during fiscal 2020 as compared to the prior year.
For Q4 of fiscal 2020, the company recorded a consolidated net loss attributable to common stockholders of $6.2 million or $0.11 per share, as compared to a consolidated net loss attributable to common stockholders of $1.1 million or $.02 per share, for Q4 of fiscal 2019. For the full FY 2020, the company recorded a consolidated net loss attributable to common stockholders of $15.2 million or $.27 per share, compared to a consolidated net loss attributable to common stockholders of $8.9 million or $.16 per share, for fiscal 2019.
Avid reported $36.3 million in cash and cash equivalents as of April 30, 2020, a 12% increase as compared to $32.4 million as of the prior FY ended April 30, 2019. The company also achieved positive free cash flow, measured as the change in cash from operations net of capital expenditures, of $2.0 million for the full FY 2020.
More detailed financial information and analysis may be found in Avid Bioservices’ Annual Report on Form 10-K, which will be filed with the SEC today. [https://tinyurl.com/yak25nco ]
RECENT CORPORATE DEVELOPMENTS
* Appointed Nicholas (Nick) Green as President and CEO. Mr. Green has more than 30 years of experience in the industry. He has held a number of senior management roles, most recently serving as president and CEO of Therapure Biopharma, with prior positions as managing director of Nipa Laboratories Ltd., head of the Life Science Division of Clariant International Ltd. in the USA, President and CEO of Rhodia Pharma Solutions Ltd. and President of Codexis, Inc.’s Pharma Division. Mr. Green holds a BSc (Hons) in Chemistry from Queen Mary College in London and an MBA from the University of Huddersfield. [SEE 6-23-20/PR: https://tinyurl.com/yczapcc7 ]
* Significantly strengthened the business development team with the appointment of two senior directors with responsibility for driving CDMO business growth within North America, Europe and Asia. Jason C. Brady, Ph.D. serves as senior director of business development for eastern North America, as well as Europe. Sylvia Hinds serves as senior director of business development for western North America in addition to the Asia Pacific region.
* Signed project expansion orders for $23 million during the quarter with existing customers. During fiscal 2020, the company signed new business orders for $80 million as compared to $55 million during fiscal 2019.
* Entered into a co-marketing agreement with Aragen Bioscience, a leading contract research organization (CRO) focused on accelerating pre-clinical biologics product development to provide clients with an integrated “sequence-to-manufacturing” service. Under terms of the non-exclusive agreement, the companies will offer customers Aragen’s cell line development expertise integrated with Avid’s upstream and downstream process development and analytical services to drive efficiencies and reduce overall timelines for delivering CGMP bulk drug substances.
* Avid was named a recipient of five 2020 Contract Manufacturing Organization (CMO) Leadership Awards in the following categories: Capabilities, Expertise, Reliability, Compatibility and Service. Presented by the industry publication Life Science Leader and based on market research and surveys conducted by Industry Standard Research (ISR), these awards are intended to honor those companies in the contract manufacturing space that provide their customers with the industry’s highest level of service.
* Restored operation of the specific piece of equipment that caused an interruption of production in the third and fourth quarters of fiscal 2020. This project is being executed in three phases: investigation; remediation; and, confirmation. To date, Avid believes it has successfully completed the first two stages by identifying and remediating the source of the problem. The company is currently progressing through the confirmation stage, during which it is running multiple revenue-generating production campaigns. The company will not be able to confirm a full resolution until it completes the confirmation stage. It is expected that this work will be completed in the coming months.
* Initiated the pre-engineering, design and permitting work required to allow the company to break ground on a facility expansion at the appropriate time. While a specific kick-off date has not yet been established for this expansion, the company believes that customer demand will require additional capacity and Avid is proactively working to prepare for this growth.
CONFERENCE CALL
Avid will host a conference call and webcast this afternoon, June 30, 2020, at 4:30PM EDT (1:30PM PDT). To listen to the conference call, please dial (877) 312-5443 or (253) 237-1126 and request the Avid Bioservices conference call. To listen to the live webcast, or access the archived webcast, please visit: http://ir.avidbio.com/investor-events .
ABOUT AVID BIOSERVICES, INC.
Avid Bioservices is a dedicated contract development and manufacturing organization (CDMO) focused on development and CGMP manufacturing of biopharmaceutical drug substances derived from mammalian cell culture. The company provides a comprehensive range of process development, CGMP clinical and commercial manufacturing services for the biotechnology and biopharmaceutical industries. With 27 years of experience producing monoclonal antibodies and recombinant proteins, Avid's services include CGMP clinical and commercial drug substance manufacturing, bulk packaging, release and stability testing and regulatory submissions support. For early-stage programs the company provides a variety of process development activities, including upstream and downstream development and optimization, analytical methods development, testing and characterization. The scope of our services ranges from standalone process development projects to full development and manufacturing programs through commercialization. https://www.avidbio.com
Forward-Looking *snip*
AVID BIOSERVICES, INC.
CONSOLIDATED STATEMENTS OF OPERATIONS AND COMPREHENSIVE LOSS
(in thousands, except per share information)
Three Months Ended Twelve Months Ended
April 30, April 30,
2020 2019 2020 2019
Revenues $ 12,550 $ 17,055 $ 59,702 $ 53,603
Cost of revenues 13,849 13,407 55,770 46,379
Gross (loss) profit (1,299 ) 3,648 3,932 7,224
Operating expenses:
Selling, general and administrative 3,528 3,573 14,517 12,846
Loss on lease termination — — 355 —
Total operating expenses 3,528 3,573 14,872 12,846
Operating (loss) income (4,827 ) 75 (10,940 ) (5,622 )
Interest and other income, net 59 92 474 282
(Loss) income from continuing operations before income taxes $ (4,768 ) $ 167 $ (10,466 ) $ (5,340 )
Income tax benefit — 67 — 284
(Loss) income from continuing operations, net of tax (4,768 ) 234 (10,466 ) (5,056 )
Income from discontinued operations, net of tax — 102 — 841
Net (loss) income $ (4,768 ) $ 336 $ (10,466 ) $ (4,215 )
Comprehensive (loss) income $ (4,768 ) $ 336 $ (10,466 ) $ (4,215 )
Series E preferred stock accumulated dividends (1,442 ) (1,442 ) (4,686 ) (4,686 )
Net loss attributable to common stockholders $ (6,210 ) $ (1,106 ) $ (15,152 ) $ (8,901 )
Basic and diluted net (loss) income per common share attributable to common stockholders:
Continuing operations $ (0.11 ) $ (0.02 ) $ (0.27 ) $ (0.17 )
Discontinued operations — — — 0.01
Net loss per share attributable to common stockholders $ (0.11 ) $ (0.02 ) $ (0.27 ) $ (0.16 )
Weighted average basic and diluted shares outstanding 56,482 56,080 56,326 55,981
AVID BIOSERVICES, INC.
CONSOLIDATED BALANCE SHEETS
(In thousands, except par value)
April 30, April 30,
2020 2019
ASSETS
Current assets:
Cash and cash equivalents $ 36,262 $ 32,351
Accounts receivable 8,606 7,374
Contract assets 3,300 4,327
Inventory 10,883 6,557
Prepaid expenses 712 709
Total current assets 59,763 51,318
Property and equipment, net 27,105 25,625
Operating lease right-of-use assets 20,100 —
Restricted cash 350 1,150
Other assets 302 302
Total assets $ 107,620 $ 78,395
LIABILITIES AND STOCKHOLDERS’ EQUITY
Current liabilities:
Accounts payable $ 5,926 $ 4,352
Accrued payroll and related costs 3,019 3,540
Note payable 4,379 —
Contract liabilities 29,120 14,651
Operating lease liabilities 1,228 —
Other current liabilities 808 619
Total current liabilities 44,480 23,162
Operating lease liabilities, less current portion 21,244 —
Deferred rent, less current portion — 2,072
Other long-term liabilities — 93
Total liabilities 65,724 25,327
Commitments and contingencies
Stockholders’ equity:
Preferred stock, $0.001 par value; 5,000 shares authorized;
1,648 shares issued and outstanding at respective dates 2 2
Common stock, $0.001 par value; 150,000 shares authorized;
56,483 and 56,135 shares issued and outstanding at respective dates 56 56
Additional paid-in capital 612,909 613,615
Accumulated deficit (571,071 ) (560,605 )
Total stockholders’ equity 41,896 53,068
Total liabilities and stockholders’ equity $ 107,620 $ 78,395
CONTACTS:
• Stephanie Diaz (Investors) Vida Strategic Partners 415-675-7401 sdiaz@vidasp.com
• Tim Brons (Media) Vida Strategic Partners 415-675-7402 tbrons@vidasp.com
- - - - - - - -
From 10-Q header: “As of June 19, 2020, there were 56,511,294 shares outstanding.”
- - - - - - - - - - - - - - - - -
Latest 10K 4-30-20 iss. 6-30-20 https://tinyurl.com/yak25nco (Cash 4-30-20=$36.3mm)
Latest 10Q 1-31-20 iss. 3-10-20 https://tinyurl.com/vkocrsg (Cash 1-31-20=$30.7mm)
ALL SEC filings for PPHM: http://tinyurl.com/6d4jw8
10-K: “As of 4-30-20, we employed 222 full-time & 5 part-time emps.” (2018: 185/1, 2019: 211/4)
= = = = = = = = = = = = = = = = = = = = = = = = = = = =
Updated PPHM REVS-BY-QTR TABLE, now thru FY20Q4(fye 4-30-20), per the 10K issued 6-30-20.
• Total Avid Revs since May’03: $408.9M
• 6-30-20: FY'21 (May'20-Apr'21) Avid revs guidance $76-81M (committed B/L=$65 at 4-30-20).
• Inventories at 4-30-20 total $10.9M, UP from $9.6M at 4-30-20.
Avid’s website: http://www.avidbio.com
AVID GROSS PROFITABILITY BY QTR: CONTRACT
QTR (1000’s) Rev$ COGS$ Prof$ GP% INVEN$ LIABILITIES*
FY13Q1 7-31-12 4,135 2,024 2,111 51% 5,744 16,280
FY13Q2 10-31-12 6,061 3,703 2,358 39% 5,426 14,721
FY13Q3 1-31-13 6,961 3,651 3,310 47% 4,635 11,790
FY13Q4 4-30-13 4,176 3,217 959 23% 4,339 12,230
FY14Q1 7-31-13 4,581 2,670 1,911 42% 5,679 12,692
FY14Q2 10-31-13 7,354 4,195 3,159 43% 4,033 11,126
FY14Q3 1-31-14 3,885 2,416 1,469 38% 5,224 12,975
FY14Q4 4-30-14 6,474 3,829 2,645 41% 5,530 11,001
FY15Q1 7-31-14 5,496 3,583 1,913 35% 5,998 10,896
FY15Q2 10-31-14 6,263 4,139 2,124 34% 5,379 11,161
FY15Q3 1-31-15 5,677 3,113 2,564 45% 6,148 14,063
FY15Q4 4-30-15 9,308 4,758 4,550 49% 7,354 17,993
FY16Q1 7-31-15 9,379 4,608 4,771 51% 10,457 17,890
FY16Q2 10-31-15 9,523 4,741 4,782 50% 12,554 24,623
FY16Q3 1-31-16 6,672 3,896 2,776 42% 15,189 37,851
FY16Q4 4-30-16 18,783 9,721 9,062 48% 15,189 39,630
FY17Q1 7-31-16 5,609 3,062 2,547 45% 25,274 43,262
FY17Q2 10-31-16 23,370 15,441 7,929 34% 25,924 44,908
FY17Q3 1-31-17 10,747 7,974 2,773 26% 33,829 52,577
FY17Q4 4-30-17 17,904 11,782 6,122 34% 33,099 45,517
FY18Q1 7-31-17 27,077 20,448 6,629 24% 24,235 27,755
FY18Q2 10-31-17 12,782 16,242 -3,460 -27% 16,518 20,611
FY18Q3 1-31-18 6,819 10,951 -4,132 -61% 14,218 24,235
FY18Q4 4-30-18 6,943 8,904 -1,961 -28% 16,129 27,935
FY19Q1 7-31-18 12,589 11,397 1,192 9% 9,168 17,994
FY19Q2 10-31-18 10,178 9,844 334 3% 9,736 17,307
FY19Q3 1-31-19 13,781 11,731 2,050 15% 8,660 14,620
FY19Q4 4-30-19 17,055 13,407 3,648 21% 6,557 14,651
FY20Q1 7-31-19 15,254 14,168 1,086 7% 8,031 18,104
FY20Q2 10-31-19 18,313 14,953 3,360 18% 7,809 22,199
FY20Q3 1-31-20 13,585 12,800 785 6% 9,565 26,355
FY20Q4 4-30-20 12,550 13,849 -1,299 -10% 10,883 29,120
*7-31-18 10Q: “prior-yr amts related to (deferred revenue
+ cust deposits) now reclass’d as contract liabilities.”
Rev$ COGS$ Prof$ GP%
FY13 TOTAL: 21,333 12,595 8,738 41%*
FY14 TOTAL: 22,294 13,110 9,184 41%*
FY15 TOTAL: 26,744 15,393 11,151 42%*
FY16 TOTAL: 44,357 22,966 21,391 48%*
FY17 TOTAL: 57,630 38,259 19,371 34%*
FY18 TOTAL: 53,621 56,545 -2,924 -5%*
FY19 TOTAL: 53,603 46,379 7,224 13%*
FY20 TOTAL: 59,702 55,770 3,932 7%*
*Avid Net-Profit(Selling/G&A) not split out from PPHM-Corp. in the fin’s.
AVID TOTAL REV’s BY YEAR):
FY04 4-30-04 3,039 (Avid-Revs didn’t incl. Avid’s Gov’t work)
FY05 4-30-05 4,684
FY06 4-30-06 3,005
FY07 4-30-07 3,492
FY08 4-30-08 5,897
FY09 4-30-09 12,963
FY10 4-30-10 13,204
FY11 4-30-11 8,502
FY12 4-30-12 14,783
FY13 4-30-13 21,333
FY14 4-30-14 22,294
FY15 4-30-15 26,744
FY16 4-30-16 44,357
FY17 4-30-17 57,630
FY18 4-30-18 53,621
FY19 4-30-19 53,603
FY20 4-30-20 59,702
**TOTAL: 408,853 (5/1/2003–4/30/20)
.
QTLY. NET PROFIT/LOSS BY QTR:
(“attributable to common stockholders”; ie, incl. PREF Div’s**)
**2-11-14: PPHM Raises $16.2M, 700k Pref. Shares w/10.5% DIV.
FY16Q1 7-31-15 -15,101,000
FY16Q2 10-31-15 -14,578,000
FY16Q3 1-31-16 -18,227,000
FY16Q4 4-30-16 -13,264,000
FY17Q1 7-31-16 -12,437,000
FY17Q2 10-31-16 -4,498,000
FY17Q3 1-31-17 -9,216,000
FY17Q4 4-30-17 -6,714,000
FY18Q1 7-31-17 -2,647,000
FY18Q2 10-31-17 -14,066,000
FY18Q3 1-31-18 -12,446,000
FY18Q4 4-30-18 +1,578,000 <=includes $9,154,000 income from disc. operations.
FY19Q1 7-31-18 -3,403,000
FY19Q2 10-31-18 -2,893,000
FY19Q3 1-31-19 -2,581,000
FY19Q4 4-30-19 -1,106,000
FY20Q1 7-31-19 -4,606,000
FY20Q2 10-31-19 -1,872,000
FY20Q3 1-31-20 -3,546,000
FY20Q4 4-30-20 -6,210,000
Period(10-K) Halozyme Gilead Acumen IGM Coherus ADC-Ther. Others
FYE 4-30-14 91% 8%
FYE 4-30-15 79% 9%
FYE 4-30-16 69% 26% 5%
FYE 4-30-17 58% 16% 26% 16%
FYE 4-30-18 55% 22% 9% 14%
FYE 4-30-19 30% <10% <10% 13% 21% 36%
FYE 4-30-20 28% 24% 11% 11% 10% <10% --
...(cust. splits not given in 7-31-18+ 10Q’s)
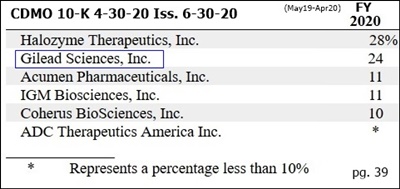
I found the Gilead Announcement (7-8-2019) – they just called the new customer “A Top 10 Global Pharma”.
Gilead Sciences: Revenue=$22.5B(2019), Headquarters: Foster City, CA
7-8-19/PR: Avid Signs New Top10 Global Pharma and Expands Existing Cust. Relationship
...today announced that it has signed 2 new contract manufacturing service agreements to support the development of novel drug candidates. The agreements include the addition of one the world’s Top 10 pharmaceutical companies by revenue to Avid’s growing list of customers, as well as the expansion of the relationship with one of the company’s existing biotechnology customers…
…Under terms of the agreement, Avid will provide process transfer and clinical manufacturing services to the large pharmaceutical company to support development of a novel therapeutic candidate. Avid and its client will immediately commence process transfer of cell line & bench scale information for the program, followed by initiation of 2000L CGMP clinical manufacture of the compound.
“These 2 new manufacturing contracts represent key business development successes which serve to maintain the momentum we have established at Avid toward our dual goals of revenue growth and customer base diversification. Importantly, we have established a high-value relationship with one of the world’s largest pharmaceutical companies, while continuing the expansion of one of our key existing client relationships,” said Rick Hancock, interim President and CEO of Avid. “Taken together, the signing of these agreements highlights the ability of Avid to leverage our CDMO expertise and track record of success to meet the varying needs of drug developers of all types and sizes. We are pleased to be trusted with the responsibility of delivering exceptional contract manufacturing services to these clients and look forward to long, productive relationships with both companies.”...
http://ir.avidbio.com/news-releases/news-release-details/avid-bioservices-enters-contract-manufacturing-agreements-new
= = = = = =
Pg. 39 of CDMO’s 4-30-20 10-K (iss. 6-30-20):
Period Halozyme Gilead Acumen IGM Coherus ADC-Ther. Others
FYE 4-30-14 91% 8%
FYE 4-30-15 79% 9%
FYE 4-30-16 69% 26% 5%
FYE 4-30-17 58% 16% 26% 16%
FYE 4-30-18 55% 22% 9% 14%
FYE 4-30-19 30% <10% <10% 13% 21% 36%
FYE 4-30-20 28% 24% 11% 11% 10% <10% --