| Followers | 789 |
| Posts | 38953 |
| Boards Moderated | 14 |
| Alias Born | 06/20/2009 |
Tuesday, August 15, 2023 3:18:05 PM
Please note that, in general, Section 801 of the Food and Drug Administration Amendments Act (FDAAA 801) requires Applicable Clinical Trials to be registered within 21 days of enrollment of the first participant
ClinicalTrials.gov: Home https://clinicaltrials.gov
A structured online system, such as ClinicalTrials.gov, that provides the public with access to summary information about ongoing and completed clinical studies
Purpose of Clinical Trials gov
ClinicalTrials.gov is a Web-based resource that provides patients, their family members, health care professionals, researchers, and the public with easy access to information on publicly and privately supported clinical studies on a wide range of diseases and conditions.
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=171473628&txt2find=Ongoing
to verify Dixon's Ongoing ________(s) feel free to fill in the blank(s) click here https://investorshub.advfn.com/boards/read_msg.aspx?message_id=171019590&txt2find=check%20box
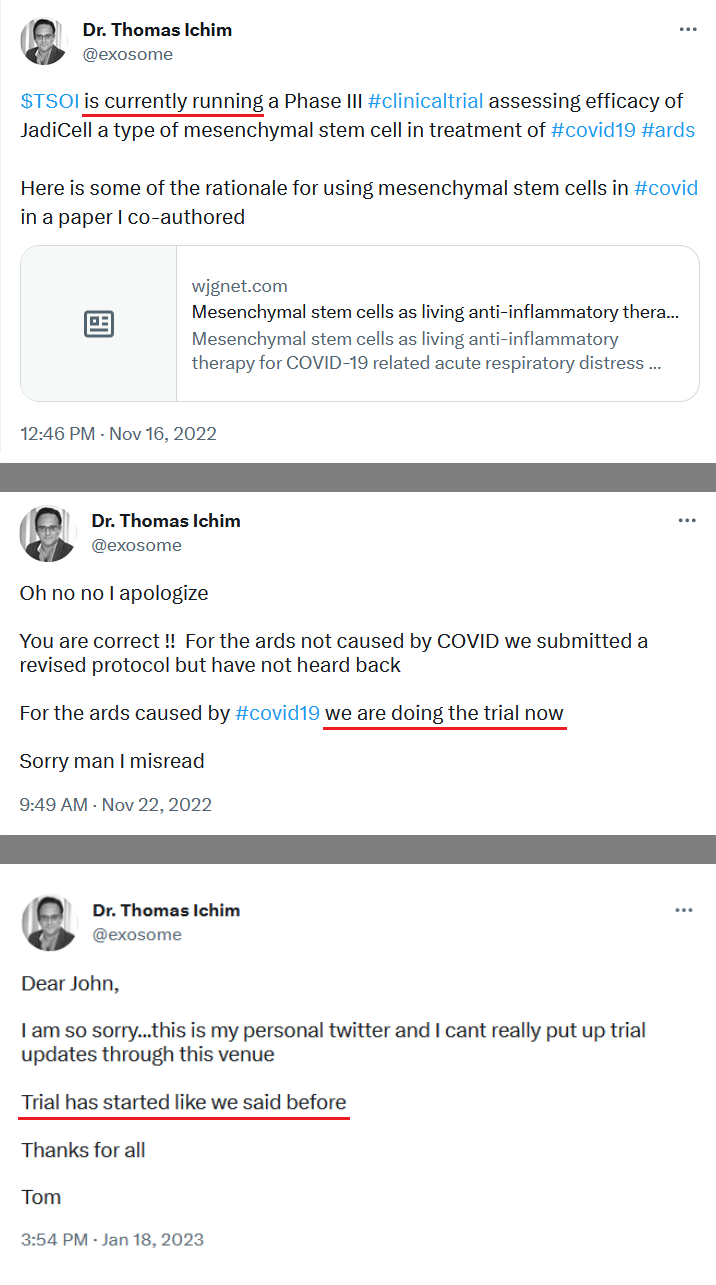
Re: JadiCells won't always be JadiCells
Post by TimGDixon » Wed Jul 20, 2022 6:17 pm
That's right everything was submitted and is sitting there but hasn't been cleared yet and when it is we're not going to check the box to make it visible. That's what I'm saying.
“...my pathway led by confusion boats, mutiny from stern to bow... Ah, but I was so much older then I’m younger than that now...”
https://forum.therapeuticsolutionsint.com/viewtopic.php?p=18989#p18989
by TimGDixon » Fri May 05, 2023 1:49 pm
what I am saying is intrinsically there is no monetary value to a listing of a clinical trial on a gov website - if there was we would have to account for it and write a footnote disclosure - way too much hype has ben placed on such things when I know as the operator its just not true - doesn't mean the trial isn't of value to investors but just a listing - btw, NIH says they don't stand behind any listing on their sites. we announced a new phase 3 ards and got .003 out of it for a day - its completely arbitrary and artificial - it was just an announcement - I've put out hundreds of "announcements" and you see what they do - nothing. No, what matters is getting through all 128 patients and closing the trial out, publishing the results and filing for a BLA. Then we will celebrate!
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=171676041&txt2find=check%20box
https://forum.therapeuticsolutionsint.com/viewtopic.php?t=16&hilit=even+press+releases&start=1290
8k https://www.otcmarkets.com/filing/html?id=16387200&guid=pfD-kn0KtMcHJth

https://www.nasdaq.com/market-activity/stocks/tsoi/press-releases
Recent TSOI News
- Form 8-K/A - Current report: [Amend] • Edgar (US Regulatory) • 05/31/2024 08:51:35 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 05/31/2024 04:47:07 PM
- Form 4 - Statement of changes in beneficial ownership of securities • Edgar (US Regulatory) • 05/31/2024 12:46:43 PM
- Form 4 - Statement of changes in beneficial ownership of securities • Edgar (US Regulatory) • 05/31/2024 12:44:01 PM
- Form 10-Q - Quarterly report [Sections 13 or 15(d)] • Edgar (US Regulatory) • 05/30/2024 06:46:26 PM
- Form NT 10-Q - Notification of inability to timely file Form 10-Q or 10-QSB • Edgar (US Regulatory) • 05/15/2024 04:01:05 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 05/06/2024 10:03:21 AM
- Form 4 - Statement of changes in beneficial ownership of securities • Edgar (US Regulatory) • 02/26/2024 07:15:22 PM
- Form 4 - Statement of changes in beneficial ownership of securities • Edgar (US Regulatory) • 02/26/2024 07:12:14 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 02/21/2024 10:25:13 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 02/20/2024 11:16:15 AM
- Form EFFECT - Notice of Effectiveness • Edgar (US Regulatory) • 02/08/2024 05:15:25 AM
- Form S-1/A - General form for registration of securities under the Securities Act of 1933: [Amend] • Edgar (US Regulatory) • 02/06/2024 11:34:40 AM
- Form 8-K - Current report • Edgar (US Regulatory) • 01/22/2024 08:42:55 PM
- Form S-1 - General form for registration of securities under the Securities Act of 1933 • Edgar (US Regulatory) • 01/17/2024 11:15:01 PM
- Form POS AM - Post-Effective amendments for registration statement • Edgar (US Regulatory) • 01/04/2024 09:03:26 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 12/22/2023 07:44:34 PM
- Form 10-Q - Quarterly report [Sections 13 or 15(d)] • Edgar (US Regulatory) • 11/14/2023 05:39:49 PM
- Form 4 - Statement of changes in beneficial ownership of securities • Edgar (US Regulatory) • 10/16/2023 11:17:29 PM
- Form 4 - Statement of changes in beneficial ownership of securities • Edgar (US Regulatory) • 10/16/2023 11:13:32 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 10/13/2023 06:50:35 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 09/13/2023 05:44:55 PM
- Form 10-Q - Quarterly report [Sections 13 or 15(d)] • Edgar (US Regulatory) • 08/21/2023 09:24:52 PM
- Form NT 10-Q - Notification of inability to timely file Form 10-Q or 10-QSB • Edgar (US Regulatory) • 08/15/2023 11:10:01 AM
North Bay Resources Announces Composite Assays of 0.53 and 0.44 Troy Ounces per Ton Gold in Trenches B + C at Fran Gold, British Columbia • NBRI • Jun 18, 2024 9:18 AM
VAYK Assembling New Management Team for $64 Billion Domestic Market • VAYK • Jun 18, 2024 9:00 AM
Fifty 1 Labs, Inc Announces Acquisition of Drago Knives, LLC • CAFI • Jun 18, 2024 8:45 AM
Hydromer Announces Attainment of ISO 13485 Certification • HYDI • Jun 17, 2024 9:22 AM
ECGI Holdings Announces LOI to Acquire Pacific Saddlery to Capitalize on $12.72 Billion Market Potential • ECGI • Jun 13, 2024 9:50 AM
Fifty 1 Labs, Inc. Announces Major Strategic Advancements and Shareholder Updates • CAFI • Jun 13, 2024 8:45 AM











