| Followers | 128 |
| Posts | 5480 |
| Boards Moderated | 0 |
| Alias Born | 06/09/2020 |
Thursday, April 21, 2022 6:25:00 AM
I'm at a loss at "people" - like AF - still trying to sow doubt about the DCVax-L and Keytruda trial.
I guess they know, that this is the big Tzar bomba, that could valuate NWBO to the stratosphere, so they have to somehow still question this, even though it seems like rational seppuku.
It IS quite easy.
There are 3 UCLA SPORE projects.
https://trp.cancer.gov/spores/abstracts/ucla_brain.htm
The first one is the combo trial between DCvax-L and Keytruda
https://trp.cancer.gov/spores/abstracts/ucla_brain.htm#h03
Project 1: Active immunotherapy combined with checkpoint modulation for glioblastoma
Project Co-Leaders:
Robert M. Prins, PhD (Basic Co-Leader)
Linda M. Liau, MD, PhD, MBA (Clinical Co-Leader)
It says directly:
"... in conjunction with tumor lysate-pulsed DC vaccination (DCVax-L)"
This is an old description. Look at aim 3
Aim 3: To develop predictive immunological and imaging biomarkers of response in recurrent glioblastoma patients enrolled in a Phase II clinical trial of DCVax-L +/- Nivolumab.
This is the original clinical trial that was setup with BMYs Opdivo (Nivolumab)
"A Phase II Clinical Trial Evaluating Combination Therapy Using DCVax-L (Autologous Dendritic Cells Pulsed With Tumor Lysate Antigen) and Nivolumab (an Anti-PD-1 Antibody) for Subjects With Recurrent Glioblastoma Multiforme"
https://clinicaltrials.gov/ct2/show/NCT03014804
But that was withdrawn,
If we look at Linda Liaus most recent slides for the SPORE PROJECT 1, we can see, that DCVax is mentioned here
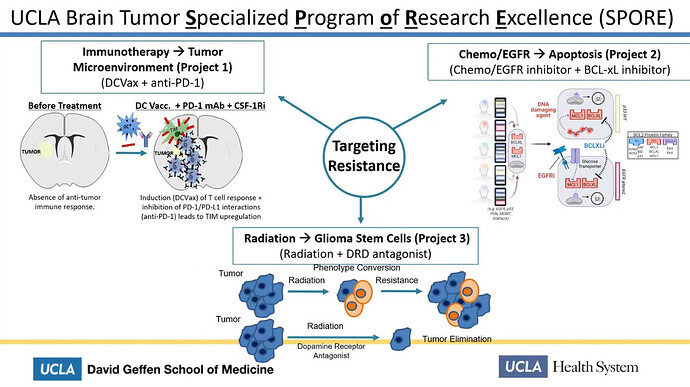
in a later slide we can see the word PEMBRO (short for Pembrolizumab aka Keytruda) is mentioned for the PD-1 inhibitor
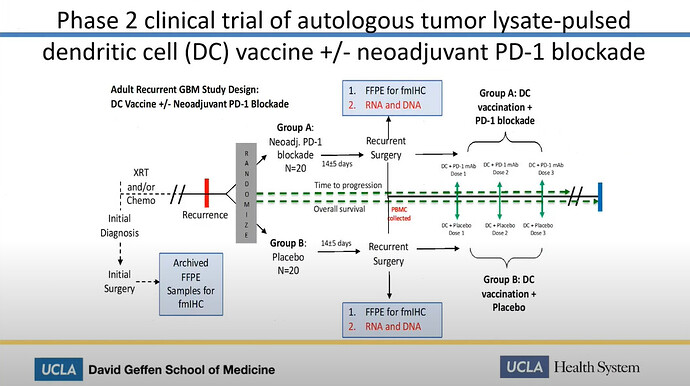
So the combo trial with DCVax-L and Keytruda exchanged the combo trial with Nivolumab (Opdivo) and that equals the SPORE 1 PROJECT at UCLA.
If we look at the patent
"COMBINATIONS OF CHECKPOINT INHIBITORS AND THERAPEUTICS TO TREAT CANCER"
We can see that the last two in the "inventors list" are the same two UCLA persons Liau and Prins that are the SPORE 1 PROJECT UCLA collaborators.
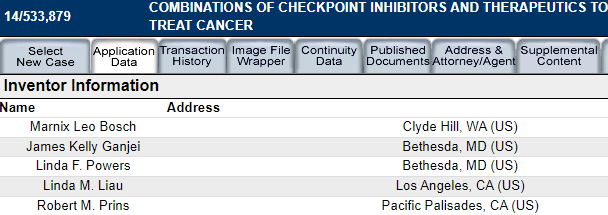
Look at the patent

The original inventors include key NWBO personnel
“Inventor: Marnix Leo BOSCH, James Kelly GANJEI, Linda F. POWERS, Linda M. Liau, Robert M. PRINS”
The rights don’t expire. The patent is assigned and that is that. There has been no reassignment back to UCLA.
The definitive assignment is what is in the patent database maintained by the government.
Invention means these parties all hold equal rights in the invention. We’d have knowledge if the application, which has not been granted yet, were to change the assignees, and there is likely a clear agreement between all the parties as to who has what exact rights from the INVENTORS:
“Current Assignee: RevImmune Inc University of California Northwest Biotherapeutics LLC Cognate Bioservices Inc”
The University retains research rights, as does NWBO, and improvements likely accrue according to the way the original assignment agreement stipulates. None of the other parties has ever represented that it has the rights to market DCVax as a commercial product with the FDA or other regulators except NWBO. There is no indication that any of this has changed.
The patent is global

Annual shareholders meeting 2018.
Linda Powers talk about the combo trial NWBO will do with Keytruda.
"That trial, which we did announce publicly and has been approved by Merck, and those agreements were signed and all that ..."
NWBO has all rights.
I like flippers comment here

What can I say, but
GAME OVER MAN!
I can't lie about shorters chances, but they have my sympathies.
Recent NWBO News
- Form 4 - Statement of changes in beneficial ownership of securities • Edgar (US Regulatory) • 09/03/2024 08:01:40 PM
- Form 4 - Statement of changes in beneficial ownership of securities • Edgar (US Regulatory) • 08/13/2024 08:01:24 PM
- Form 10-Q - Quarterly report [Sections 13 or 15(d)] • Edgar (US Regulatory) • 08/09/2024 09:06:07 PM
- Form 4 - Statement of changes in beneficial ownership of securities • Edgar (US Regulatory) • 08/08/2024 08:30:09 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 08/02/2024 02:42:28 PM
- Form 4 - Statement of changes in beneficial ownership of securities • Edgar (US Regulatory) • 07/30/2024 11:49:38 PM
- Biophma Announces Exclusive In License for Dendritic Cell Technology, Sending Shares Higher • AllPennyStocks.com • 06/17/2024 04:40:00 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 06/04/2024 09:11:16 PM
- Form DEF 14A - Other definitive proxy statements • Edgar (US Regulatory) • 06/03/2024 09:22:55 PM
- Form PRE 14A - Other preliminary proxy statements • Edgar (US Regulatory) • 05/22/2024 08:13:36 PM
- Form 10-Q - Quarterly report [Sections 13 or 15(d)] • Edgar (US Regulatory) • 05/10/2024 09:04:57 PM
- Form NT 10-K - Notification of inability to timely file Form 10-K 405, 10-K, 10-KSB 405, 10-KSB, 10-KT, or 10-KT405 • Edgar (US Regulatory) • 03/01/2024 10:04:38 PM
- Form 4 - Statement of changes in beneficial ownership of securities • Edgar (US Regulatory) • 12/02/2023 01:31:35 AM
- Form 8-K - Current report • Edgar (US Regulatory) • 11/16/2023 10:11:54 PM
- Epazz, Inc. (OTC Pink: EPAZ) ZenaDrone Demonstration to Defense Departments of UAE and Saudi Arabia • InvestorsHub NewsWire • 11/15/2023 12:19:31 PM
- Form 10-Q - Quarterly report [Sections 13 or 15(d)] • Edgar (US Regulatory) • 11/09/2023 09:30:39 PM
- Epazz, Inc. (OTC Pink: EPAZ) US Navy Collaboration ZenaDrone 1000 • InvestorsHub NewsWire • 11/09/2023 01:00:34 PM
- Epazz, Inc. (OTC Pink: EPAZ) US Navy Collaboration ZenaDrone 1000 Extreme Weather Demo • InvestorsHub NewsWire • 11/07/2023 12:29:43 PM
ZenaTech, Inc. (NASDAQ: ZENA) Launchs IQ Nano Drone for Commercial Indoor Use • HALO • Oct 10, 2024 8:09 AM
CBD Life Sciences Inc. (CBDL) Targets Alibaba as the Next Retail Giant for Wholesale Expansion of Top-Selling CBD Products • CBDL • Oct 10, 2024 8:00 AM
Foremost Lithium Announces Option Agreement with Denison on 10 Uranium Projects Spanning over 330,000 Acres in the Athabasca Basin, Saskatchewan • FAT • Oct 10, 2024 5:51 AM
Element79 Gold Corp. Reports Significant Progress in Community Relations and Development Efforts in Chachas, Peru • ELEM • Oct 9, 2024 10:30 AM
Unitronix Corp Launches Share Buyback Initiative • UTRX • Oct 9, 2024 9:10 AM
BASANITE INDUSTRIES, LLC RECEIVES U.S. PATENT FOR ITS BASAFLEX™ BASALT FIBER COMPOSITE REBAR AND METHOD OF MANUFACTURING • BASA • Oct 9, 2024 7:30 AM







