Watching alerts...
Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Nice news!
there is no news... its all BS.
MNMD & ATAI - FDA Issues First Draft Guidance on Clinical Trials with Psychedelic Drugs
Agency Recommendations Aim to Inform Psychedelic Drug Development
https://www.fda.gov/news-events/press-announcements/fda-issues-first-draft-guidance-clinical-trials-psychedelic-drugs
For Immediate Release:
June 23, 2023
Today, the U.S. Food and Drug Administration published a new draft guidance to highlight fundamental considerations to researchers investigating the use of psychedelic drugs for potential treatment of medical conditions, including psychiatric or substance use disorders. This is the first FDA draft guidance that presents considerations to industry for designing clinical trials for psychedelic drugs.
There has been growing interest in the therapeutic potential of psychedelic drugs in recent years. They are being evaluated for use in the potential treatment of conditions such as depression, post-traumatic stress disorder, substance use disorders and other conditions. However, designing clinical studies to evaluate the safety and effectiveness of these compounds presents a number of unique challenges that require careful consideration.
“Psychedelic drugs show initial promise as potential treatments for mood, anxiety and substance use disorders. However, these are still investigational products. Sponsors evaluating the therapeutic potential of these drugs should consider their unique characteristics when designing clinical studies,” said Tiffany Farchione, M.D., director of the Division of Psychiatry in the FDA’s Center for Drug Evaluation and Research. “By publishing this draft guidance, the FDA hopes to outline the challenges inherent in designing psychedelic drug development programs and provide information on how to address these challenges. The goal is to help researchers design studies that will yield interpretable results that will be capable of supporting future drug applications.”
The purpose of the draft guidance is to advise researchers on study design and other considerations as they develop medications that contain psychedelics. Within the draft guidance, the term psychedelics refers to “classic psychedelics,” typically understood to be drugs such as psilocybin and lysergic acid diethylamide (LSD) that act on the brain’s serotonin system, as well as “entactogens” or “empathogens” such as methylenedioxymethamphetamine (MDMA).
The document describes basic considerations throughout the drug development process including trial conduct, data collection, subject safety and new drug application requirements. For example, psychedelic drugs may produce psychoactive effects such as mood and cognitive changes, as well as hallucinations. As a result, there is the potential for abuse of these drugs, which is a drug safety issue that requires careful consideration and putting sufficient safety measures in place for preventing misuse throughout clinical development. For psychedelics that are currently Schedule I controlled substances, the draft guidance notes that activities associated with investigations under an Investigational New Drug Application must comply with applicable Drug Enforcement Administration regulatory requirements.
The evidentiary standard for establishing effectiveness of psychedelic drugs is the same as for all other drugs. However, there are unique factors investigators may need to consider when designing their clinical trials if those trials are to be considered adequate and well-controlled. The draft guidance also addresses the role of psychotherapy in psychedelic drug development, considerations for safety monitoring and the importance of characterizing dose-response and the durability of any treatment effect.
The FDA is encouraging the public to provide comments on the draft guidance. Comments should be submitted within 60 days to ensure the agency considers them.
You will be sorry
I keep my mark on buyout in 1-2 years from right now. Get your tickets.
25.00 PT!!
Mind Medicine has a huge upside, says Roth
The stock remains well off its highs, but investors will want to be thinking of Mind Medicine (Mind Medicine Stock Quote, Charts, News, Analysts, Financials NASDAQ:MNMD), according to Roth Capital Partners analyst Jonathan Aschoff, who reported on the biopharma company on Wednesday and reiterated a “Buy” rating.
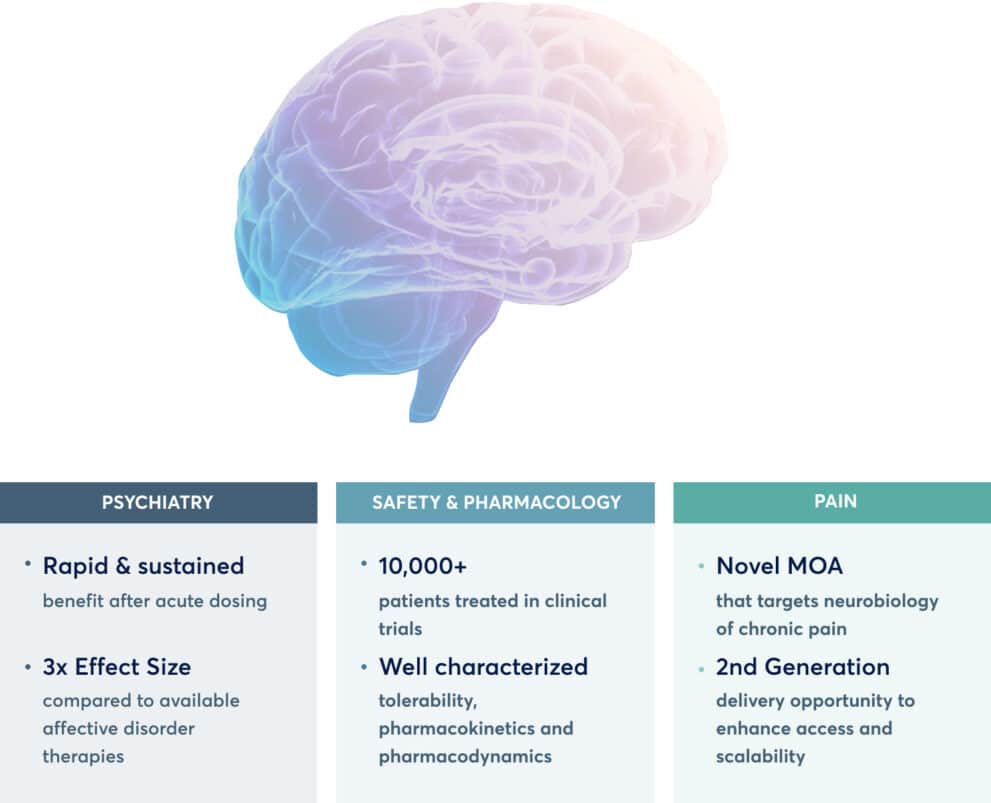
Mind Medicine is a clinical-stage developer of candidates to treat brain health disorders, with a pipeline of assets targeting neurotransmitter pathways playing a role in brain health disorders. The company held on Monday an Investor Day, where it provided an update on MM-120 (lysergide D-tartrate), in trials for generalized anxiety disorder (GAD).
MindMed said the current Phase 2b trial for MM-120 remains on track for a late 2023 topline data readout. The trial has participants receiving a single administration of the drug at various dose strengths, with the aim being to measure reduction in anxiety symptoms four weeks after the single administration. Secondary objectives will be measuring results up to 12 weeks after dosing.
MM-120 is a pharmacologically optimized form of LSD being developed for GAD and other brain health disorders. Mind Medicine received clearance from the US FDA for its Investigational New Drug (IND) application in January 2022, allowing it to proceed with the Phase 2b trial.
Aschoff commented on the discussions from four experts who spoke at the Investor Day, starting with Dr. Maria Oquendo, who said anxiety, depression and alcohol use disorder are indications in which LSD has seen clinical success, which somewhat reduces the clinical risk for MM-120, in Aschoff’s view, at least for the GAD trial to read out by the year end. Dr. Oquendo put the US GAD prevalence at about 2.9 per cent of the population, 76 per cent of which have a moderate to severe condition that disrupts th patient’s ability to function effectively either socially or at work.
Next, Dr. David Feifel said that one in five people in the US takes psychiatric drugs and that the crisis is the low number of new drugs in the space. Dr. Feifel’s view of using psychedelics for treating disease is highly favourable compared to his view of SSRIs for many of the same conditions and that the large increase in behavioural health conditions in recent years has pushed the FDA to be more permissive than ever in allowing clinical trials with what were once viewed as purely recreational drugs.
Aschoff said CEO Rob Barrow’s closing remarks included comments on MM-120’s rapid and durable effect and how 65 per cent of patients taking LSD in other trials had clinical benefit versus nine per cent of placebo.
“We look forward to Phase 2b and 2a MM-120 data in GAD and ADHD, respectively by YE23, followed by an EOP2 meeting that should nail down a Phase 3 plan,” Aschoff wrote.
With the update, Aschoff maintained a 12-month target on MNMD of $25.00, which at press time represented a projected return of 593 per cent.
I keep buying. FDA approval of MM-120 will be one of the most iconic decisions. It will be unreal. You thought the NASDAQ run was nuts... just hang on for a few years.
Waters fine. Come in.
Getting feisty in here mick
I'm fully convinced we will see it happen this summer. Any time now.
I'm not in it... I'm just watching the shit show.
I don't think so
It's coming!
Going back down...
So good
This is so ready to 10X
Pot of sauce on the stove Sunday... crusty bread dragged through it. :)
good news!
This will sink
I'm confident we get our run. Official news and revenues will help drive that I believe.
Coming back down...
Nice! This is still a good play to be in. Big run in the works.
False pump... do not chase this you will be sorry.
Yes they have the cash on hand and they themselves have stated they have the funds to see all milestones into mid 2025. We should be good.
I expect a buy out prior to 2025.
This one and M!KP should be good through the next few months. Really interest to see you DD pack
What i see is that a lot of folks bought this when it last ran, chased it and are now seeing the spikes down here as hopeful and as a possible reversal. To which I say is utter none sense. This ticker is dead.
Still not convinced.
I believe this is going to see some crazy fast movement up soon.
LSD for anxiety and psychedelic clinical trials with MindMed CMO, Dr. Daniel Karlin
https://www.youtube.com/watch?v=cdhS8SUCUVU
LSD for anxiety and psychedelic clinical trials with MindMed CMO, Dr. Daniel Karlin
https://www.youtube.com/watch?v=cdhS8SUCUVU
I smell something big brewing. All my holdings are turning green. Market sentiment is getting better. Ride the wave.
Cedar Clinical Research, by Numinus, the top enrolling clinical research site for MindMed's Phase 2B study evaluating MM-120
June 05 2023 - 07:00AM
PR Newswire (US)
VANCOUVER, BC, June 5, 2023 /PRNewswire/ - Numinus Wellness Inc. ("Numinus" or the "Company") (TSX: NUMI) (OTCQX: NUMIF), a mental health care company advancing innovative treatments and safe, evidence-based psychedelic-assisted therapies, is pleased to announce Cedar Clinical Research (CCR) is a top research site for MindMed's Phase 2b study evaluating MM-120 (lysergide D-tartrate) for General Anxiety Disorder.
CCR's research clinic in Draper, Utah, is the top enrolling site for the research study – having enrolled and dosed 19 clinical trial participants for the study to date. CCR also recently began enrolling volunteers for this trial at a second research clinic – in Murray, Utah.
"We're excited to support MindMed in their clinical trial work, by evaluating new important treatments and protocols to address mental health disorders," said Paul Thielking, Chief Science Officer, Numinus, and Principal Investigator at CCR Murray. "The fact that our Draper research clinic is the top enrolling site for the study underscores the quality of our clinical research capabilities and recruiting efforts."
The MindMed study aims to enroll up to 200 participants who will receive a single administration of 25 µg, 50 µg, 100 µg or 200 µg of MM-120 or placebo. Topline results are expected to be announced by MindMed in late 2023.
To learn more about the study, and find out if you qualify to participate in it, please visit: https://www.anxietyresearchstudy.com/
About Numinus
Numinus Wellness Inc. (TSX: NUMI) helps people to heal and be well through the development and delivery of innovative mental health care and access to safe, evidence-based psychedelic-assisted therapies. The Numinus model - including psychedelic production, research and clinic care - is at the forefront of a transformation aimed at healing rather than managing symptoms for depression, anxiety, trauma, pain and substance use. At Numinus, we are leading the integration of psychedelic-assisted therapies into mainstream clinical practice and building the foundation for a healthier society.
Learn more at www.numinus.com and follow us on LinkedIn, Facebook, Twitter, and Instagram.
About MindMed
MindMed (NASDAQ: MNMD) (NEO: MMED) is a clinical stage biopharmaceutical company developing novel products to treat brain health disorders, with a particular focus on psychiatry, addiction, pain and neurology. The company is developing a pipeline of innovative drug candidates, with and without acute perceptual effects, targeting the serotonin, dopamine and acetylcholine systems.
Looks like its getting ready for a nice burst.
I probably have not been in as long as you, but this should be one of the harder runners this year.
Going to be one hell of a ride!
here she comes... back down - way down
We leaving the trips this week :)