Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Honestabe13, so you know, only 'ADMIN' (not Mods) can Ban a poster.
AZN publishes their Partnering Brochure each MAY. Why do you think it wouldn't be easy for them to change it (ie, remove Peregrine) when they updated & issued the new Brochure PDF for 5-2017?
5-2017: AZN’s 5-2017 “Partnering For Scientific Leadership” Brochure
https://www.astrazeneca.com/content/dam/az/PDF/2017/2017_18_AZ_BD%20&%20Partnering%20Brochure_FINAL.pdf
MORE: https://www.astrazeneca.com/our-focus-areas/oncology.html

Large Ownership now ~13.9mm shares, 31.0% of 45mm O/S. (Ronin/SW, E.Cap/Dart, Institutions)
7-14-17/13D: Group Ronin Trading/SWInvest (John Stafford III+Stephen White) acquires 8.8% stake (3,952,446sh.) in PPHM http://tinyurl.com/y7ezqvm9
...3,801,139 COMMON - 8.4% of 45,069,188 common O/S at 7-10-17 (total beneficial=3,952,446 if Pref. conv. x1.19 to Common, 8.8%)
...127,099 PREFERRED – 7.7% of 1,647,760 preferred O/S at 1-31-17
......As a Group, ie, “people that share the furtherance of a common objective/concerted action”; 13D’s are reserved for ACTIVE INVESTORS who may be “interested in agitating for some kind of a change at the company”.
...7-13-17: Ronin/SW-Invest Letter to Stockholders; PPHM Comments http://tinyurl.com/ybr8ycbp - Ronin nominates Gregory Sargen, Brian Scanlan, Saiid Zarrabian for election to PPHM's board at next ASM ~10-12-17.
- - - - - - - - - - -
10-30-15: Kenneth Dart (Eastern Capital) acquires 9.6% stake (4,300,992sh.) in PPHM http://tinyurl.com/y95yskck
...3,777,183 COMMON - 8.4% of 45,069,188 common O/S at 7-10-17 (total beneficial=4,300,992 if Pref. conv. x1.19 to Common, 9.6%)
...440,000 PREFERRED - 26.7% of 1,647,760 preferred O/S at 1-31-17
- - - - - - - - - - - - - - -
Plus, INSTITUTIONS a/o 3-31-17: 5,690,888sh. = 12.6% (of 45mm) http://www.nasdaq.com/symbol/pphm/institutional-holdings
TOP5:
Kennedy Capital Mgt. 1,247,224 +164,952
Tappan Street Partners 914,304 +914,304
Vanguard Group 882,964 +267,438
Blackrock (Larry Fink, CEO) 855,455 +177,856
Renaissance Technologies 439,673 +6,259
AZN lists the AZN-PPHM "Partnership" on their 5-2017 brochure. So, is AZN "unbelievable" too, Peregr?
5-2017: AZN’s 5-2017 “Partnering For Scientific Leadership” Brochure
https://www.astrazeneca.com/content/dam/az/PDF/2017/2017_18_AZ_BD%20&%20Partnering%20Brochure_FINAL.pdf
MORE: https://www.astrazeneca.com/our-focus-areas/oncology.html

PPHM’s 10K Notes re: Avid FY18 & Beyond (FY18 Guidance announced 7-14-17: $50-55mm - http://tinyurl.com/yb4wulvu )
From 4-30-17 10K iss. 7-14-17 - http://tinyurl.com/ycxu4l5n
Pg.2: “With respect to our CDMO business, FY17 was a record year for revenues, topping $57mm, a 30% revenue growth over FY16 FY. While we are pleased at the continued yr-over-yr revenue growth, we have also recently seen unanticipated decreases in mfg. demand from our largest customer [Halozyme] and a recent regulatory filing delay from our 2nd largest customer which will have some impact on our ability to grow the revenues from our CDMO business in FY18 and could impact our ability to achieve overall profitability by q/e July 31, 2018. However, we believe this to be temporary delay in revenue growth during FY18 and have recently secured 4 new customers and are continuing to focus on securing addl. customer business in order to better diversify our customer base. Our goal is to maintain profitability for Avid over the short term while positioning the business for long-term growth and attracting the resources necessary to continue to advance our promising research and development efforts.”
Pg.3: “FY18 Key Objectives - Our CDMO Business
* Expand our mfg. capacity through the installation & validation of two 2,000L single-use bioreactors in our Myford Facility to support the anticipated needs of a current customer
* Continue to diversify our customer base by securing addl. customers to support our future revenue growth beyond FY18.”
Pg.31: “We have been developing and mfg. biologics since 1993 in our Franklin biomfg. facility (the “Franklin Facility”) located at our current headquarters in Tustin and formed Avid in 2002 to offer these services to 3rd-party customers using. In March 2016, we expanded our mfg. capacity through the launch of our Myford biomfg. facility (“Myford Facility”), which doubled our mfg. capacity. The 42,000sf facility, which is our 2nd biomfg. facility, can accommodate single-use bioreactors up to the 2,000-liter mfg. scale. The Myford Facility was designed to accommodate a fully disposable biomfg. process for products in late stage clinical dev. to commercial. To date, Myford Facility has been utilized to complete a number of process validation runs for our 3rd-party customers, which may lead to future commercial production, and has supported the process validation of our internal product, bavituximab. The Myford Facility is located adjacent to our Franklin Facility. As we look to expand our CDMO business, in Feb. 2017, we leased an addl. 42,000sf of vacant warehouse space within the same building as our existing Myford Facility. The proximity of this space will allow us to utilize existing mfg. infrastructure that we believe should enhance our mfg. efficiencies and reduce the overall cost and timeframe to construct a 3rd biomfg. facility. Although we previously anticipated that the new mfg. facility would be constructed and ready for mfg. activities by mid-calendar year 2018, due to unanticipated changes in and/or timing of customer demand (as discussed above), we have decided to defer construction of this 3rd facility until demand from existing or potential new customers is expected to exceed the current mfg. capacity at our Franklin Facility & Myford Facility. Additionally, commencement of construction is also subject to our ability to raise sufficient addl. capital to support this expansion effort. As a result, we presently do not expect to commence construction of this 3rd facility prior to April 30, 2018.”
Pg33: “Excluding any future potential new business, we expect Avid revs for FY18 to slightly decline vs. FY17. Part of this decline is due to lower anticipated commitments from Halozyme (our largest customer) based on their most recent committed forecast (covering the 3 qtrs ending March 2018), which amount is expected to be partially offset by $10mm revenue that was expected to be recognized in FY17, but has been shifted to FY18 due to a delay in shipping product that was complete and ready for shipment as of fye 4-30-17.”
= = = = = = = = = = = = = = = = = = = = = = =
AVID BIOSERVICES, Inc. (Peregrine's Mfg. Subsidiary): http://www.avidbio.com
7-14-17: PPHM's Revs & Burns By Qtr Table, FY'07/Q1 thru FY'17/Q4 (q/e 4-30-17): http://tinyurl.com/yb4wulvu (since 5-2006: Avid=$231.2mm, Total=$257.8mm, incl.Govt)
......Avid FY18 (fye 4-30-18) revs guidance: $50-55mm; committed backlog=$58mm. Recently Leased +42,000sf in same bldg. as MYFORD for future expansion."
7-14-17: Avid Scientists develop “Antibody Discovery & Characterization Platform” http://tinyurl.com/ycr3erft
...S.King: “through which we can generate antibodies against virtually any target. These capabilities are state-of-the-art and meant for rapid screening for high affinity antibodies as drug candidates.”
5-2017: Avid II (Myford) adds 2 MilliporeSigma Mobius 2,000L single-use bioreactors; total mfg. capacity now ">11,000L". http://tinyurl.com/ky7bmu4
5-10-17: Halozyme comments on Avid II(Myford) expansion in their 3-31-17/10Q pub. 5-9-17: http://tinyurl.com/mrl34uk
..."validation of the new facility is scheduled to end in Q2/2017… Once this new facility is approved, it will become the primary source for Roche of bulk rHuPH20.”
NCI Scientist Speaking “PS” 10-2-17 at AACR/Immunotherapy Conf. in WashDC. Traces back to same author’s 6-1-17 Mol.Cell article with basically the same name. Interesting: Both the Lead+Senior authors (NCI) list MSKCC in their work declarations.
Oct1-4 2017: “AACR’s Tumor Immunology & Immunotherapy Conf.”, Boston
http://www.aacr.org/Meetings/Pages/MeetingDetail.aspx?EventItemID=122
Pgm: http://www.aacr.org/Meetings/Pages/MeetingDetail.aspx?EventItemID=122&DetailItemID=651
10-2-17 3:45-5:30pm: Plenary Session 4: A Systems Approach to Immuno-Oncology
“Long-Lived Disruption of Inflammation Stems from the Catch-and-Release of Cytokines Mediated by Surface Phosphatidylserine in Tumors”, Gregoire Altan-Bonnet, NCI, Bethesda
G. Altan-Bonnet profile: https://ccr.cancer.gov/Cancer-and-Inflammation-Program/gr%C3%A9goire-altan-bonnet
- - - - -
Related Pub. 6-1-17/MolCell: “Catch & Release of Cytokines Mediated by Tumor Phosphatidylserine Converts Transient Exposure into Long-Lived Inflammation”, Senior Author: Gregoire Altan-Bonnet **
https://www.ncbi.nlm.nih.gov/pubmed/28575659
NCI Followup PR: “Cancer cells with more PS on their surfaces could be more vulnerable to these treatments because they will maintain high IFNy levels in the surrounding tissue.”
https://ccr.cancer.gov/news/article/catch-and-release-a-weak-point-on-cancer-cells
**INTERESTING: Memorial Sloan Kettering CC (MSKCC) is listed in the author-info. section for both Dr. G.Altan-Bonnet and the lead author, J. Oyler-Yaniv.
COMPLETED BAVI PH.1/2 TRIALS ARCHIVED From iBox 7-16-17
Now linked to from within iBox for perpetuity: http://investorshub.advfn.com/boards/show_ibox.aspx?boardid=2076
BAVITUXIMAB SOLID CANCERS PHASE1+2 TRIALS:
==> PR=Partial-Resp(30-99% Red.), SD=Stable-Disease(29% Red.-19% Incr.), OR=Obj-Resp(PR+CR)
6-2012: FTM's charts of MOS Data from CtlArms(chemo) of Comp.Trials for 1NSCLC 2NSCLC PANCRE trials http://tinyurl.com/757plm7
11-25-11: Comp. of Bavi+Chemo vs. Avastin & Chemo-Only in 3 completed Ph.2 single-arm trials (ABC/2 & NSCLC): http://tinyurl.com/79b3jcj
O. 6th IST Trial: Bavi+Ipilimumab(Yervoy) vs. Adv.Melanoma (Ph1b, random, open-label, 2arms, n=24)
Protocol (UTSW): http://www.clinicaltrials.gov/ct2/show/NCT01984255 (PI: Dr. Arthur Frankel - see "Researching for Cures" http://youtu.be/0zLAxjFny5Q )
UTSW's listing: http://www.utsouthwestern.edu/research/fact/detail.html?studyid=STU%20102013-007
…Note: Ipilimumab = BMS’s “Yervoy” (anti-CTLA-4) http://www.yervoy.com
...12-10-15: “Due to newly-appr. therapies & chgs in SOC [ex: Keytruda/Opdivo: less-side-effects], enrollment recently stopped by UTSW.” http://tinyurl.com/jkp885g
...4-23-14: Bavi+Yervoy IST trial initiated: http://tinyurl.com/km7krcm ;
N. 5th IST Trial (Bavi+Capecitabine+RAD) vs. Rectal Cancer (Ph1, open-label, 1arm, n=18)
Protocol: http://www.clinicaltrials.gov/ct2/show/NCT01634685
...Note: Capecitabine (prodrug of 5-FU) = Roche/Genentech's Xeloda - see http://www.xeloda.com
...11-1-15/RadiationOncology – prelim. results/8 evaluables http://tinyurl.com/q3wey2b
…...”Of the 7 pts that proceeded to surgery, 1 demonstrated a pathologic complete response.”
...7-16-12: IST (Rectal) initiated at UTSW (PI=Jeffrey Meyer), ~18 patients - http://tinyurl.com/cr29l6k
M. Tumor Imaging & Dosimetry trial of I124-PGN650 (FH-Bavi) in Adv. Solid Tumors (Ph0, open-label, 1arm, n=12)
Protocol: http://clinicaltrials.gov/ct2/show/NCT01632696
..."I124-PGN650 is the Fab end of PS-targeting mab PGN635 (FH-Bavi) joined to the PET imaging radioisotope iodine-124, a new approach to imaging cancer."
5-1-15: Interim Results/8pts (Jrnl Nuclear Med, vol56 suppl#3) http://jnm.snmjournals.org/content/56/supplement_3/1033
…”124I-PGN650 is safe and results in acceptable dosimetry and needs further optimization as an imaging agent based on the level of increased tumor uptake on PET/CT”
...6-28-12: Trial added to Trials.gov, 1st site "recruiting" (Wash.UnivSM/St.Louis)
…4-3-12: Peregrine Launches PS-Targeting Clinical Imaging Pgm (AACR'12 #2452) http://tinyurl.com/7p7jovt & http://tinyurl.com/7yrwqm7
L. 4th IST Trial (Bavi+Cabazitaxel vs. 2nd-Line PROSTATE(CRPC) Cancer, open-label Ph.1B/IIA) *CANCELLED due to chg. in SOC Drugs*
Protocol: http://clinicaltrials.gov/show/NCT01335204 (CRPC=Castration-Resistant Prostate Cancer)
...Note: Cabazitaxel = Sanofi-Aventis' Jevtana - see http://www.jevtana.com
...3-12-13: CRPC IST cancelled (Slow-Enroll/2 New SOC Drugs): http://tinyurl.com/c48osut
...5-25-11: IST (Prostate) initiated at UCI -> moved ot Med.Univ.SCar (PI: Michael Lilly), ~31 patients - http://tinyurl.com/3mtvdvl
K. 3rd IST Trial (Bavi+PemCarbo vs. Frontline NSCLC, open-label Ph.1B, n=25)
Protocol: http://clinicaltrials.gov/ct2/show/NCT01323062
…10-30-14: Interim data n=23, MSTO’14: MOS=12.2mos (hist-ctl ~10mos), PFS=4.8mos, ORR=35% http://tinyurl.com/mll62c6
...9-9-14: Stage IV NSCLC IST Enrollment complete. http://tinyurl.com/ktrfswj
...4-2012: Clinicaltrials.gov shows 2nd site added: Univ. of Pittsburg (PI=Liza Villaruz, MD)
...4-2-12 AACR'12: "5pts. to-date, 3 have PR's" http://tinyurl.com/7yrwqm7 (see #1744)
...3-8-11: IST (NSCLC) initiated at UNC (PI= J.Grilley-Olson), ~25 patients - http://tinyurl.com/6b926ku
J. 2nd IST Trial (Bavi+Paclitaxel(Taxol) vs. Her2- Met. Breast Cancer, open-label Ph.1, n=14)
Protocol: http://clinicaltrials.gov/ct2/show/NCT01288261
...3-31-15/A.Stopeck article(Ph1 data) “Cancer-Medicine” N=13: PFS=7.3mos, ORR=85%, 2 CR's http://tinyurl.com/nm5oog4
…6-3-13: ASCO’13/interim data, n=14: ORR=85%, 2 CR’s (15%) http://tinyurl.com/kq3uv4e
...4-29-13: Enrollment complete. http://tinyurl.com/cqrup9e
...4-3-12 AACR'12: "5pts. to-date, 2 CR's, 1 PR" http://tinyurl.com/7yrwqm7 (see #4404)
...1-19-11: IST (Her2- MBC) initiated at Arizona CC (PI=A.Stopeck), ~14 patients - http://tinyurl.com/5t7zomn
I. Phase II Bavi+GEM vs. Front-Line Adv. PANCREATIC (randomized, unblinded, n=70)
Protocol: http://www.clinicaltrials.gov/ct2/show/NCT01272791 (15 U.S. + 4 Ukraine = 19 as of 6-7-2012)
…1-22-13: FTM's post of 13 Ph3 Gem+Treatment Pancreatic Trials ('02-'13) - Mean MOS: GEM=6.4mos., GEM+TR=7.3mos. http://tinyurl.com/al99hx9
......Another FTM Pancreatic Phase3 trials table showing HR's, P-Values, and ORR% stats: http://tinyurl.com/btzkw4l
…6-3-13 ASCO’13/final data: n=70, Ctl=>Bavi MOS 5.2=>5.6mos, ORR 13%=>28% HR=.75 http://tinyurl.com/kq3uv4e
......Promising ‘immuno-indicative’ 1yr SURVIVAL results: GEM-Only(n=31): 0%, Bavi+GEM(n=32): 24.5% - see: http://tinyurl.com/lz5yg4f
...2-13-13 Topline Data: “Bavi+Gem resulted in more than a doubling of ORR” http://tinyurl.com/aqny7ny
...6-25-12: Enrollment complete. http://tinyurl.com/72tvnfj
...6-20-12: Early data (cutoff=6/6/12 bavi=15 ctl=17) presented at AACR Pancreatic Conf. http://tinyurl.com/77m9fw2
...1-5-11: U.S. Ph.2 randomized trial initiated http://tinyurl.com/26hnuzv "up to 70 front-line patients at ~10 clinical sites."
H. 1st Investigator-Sponsored (IST) Ph.I/II Trial (Bavi+Sorafenib vs. Liver Cancer/HCC, open-label, n=9+38=47)
...Note: Sorafenib = Onyx/Bayer's Nexavar - see http://www.nexavar.com
Protocol: http://clinicaltrials.gov/ct2/show/NCT01264705 UTSW: http://tinyurl.com/mwdc2ql (5 sites: 3/UTSW, Parkland-Hosp, Dallas/VA, PI=Dr. Adam Yopp)
...3-25-15: Dr.Adam.Yopp(UTSW) Oral-pres./SSO (Liver-IST/Ph2-data) http://tinyurl.com/opkh5qy N=38(79%HepC, ECOG/0=34%): MTTP=6.7mos, MOS=6.1, MDSS=8.7, DCR=58%
...1-16-15 ASCO Gastro-Symposium: Ph.2 data/n=38 (Adam Yopp), “These clinical outcomes of TTP=6.7/DCR=58%/PFS=4mo are quite encouraging…” http://tinyurl.com/m9uz9mo
...11-8-14 SITC'14: Ph.2 Correlative Studies data (biopsies B4/After) on 6pts, incl. KOL Dimitry Gabrilovich’s comments: http://tinyurl.com/pchzr6h
...9-9-14 Enrollment complete. http://tinyurl.com/ktrfswj (ph2=38 Ph1=9)
...4-4-12 AACR'12: Dr. Adam Yopp, "promising safety profile to-date" http://tinyurl.com/7yrwqm7 (see #5591)
...Feb'12-Sep'14 10+ times: CEO Steve King hints of future ex-US partner-driven Bavi+Sorafenib/LIVER trial in Asia-Pacific: http://tinyurl.com/nkaxtcc
......Articles & Data describe Liver Cancer challenges in Asian populations: http://tinyurl.com/7z7o8j9 & http://tinyurl.com/7z99cy4
...12-1-10: PPHM's 1st IST (Liver Cancer) initiated at UTSW, ~56 patients - http://tinyurl.com/3xd3e6c
…Per S.King, 5-18-10/R&R, "We've had a lot of interest in running clinical trials with the compound from investigators who have either had prior experience with the drug or would like to study the drug in various settings. Potential IST indications include all the major solid tumor types. Of particular interest is Liver Cancer, in which we have a natural tie-in with our HCV program, Ovarian Cancer & Pancreatic Cancer, also very nicely supported by the prior data."
G. Phase IIb Bavi+PC vs. Front-Line NSCLC (randomized, unblinded, 'confirmatory', n=86)
Protocol: http://clinicaltrials.gov/ct2/show/NCT01160601 (17 U.S. + 9 India + 2 RepGA + 7 RussianFED + 5 Ukraine = 40 as of 8-12-11)
...Also listed in: India's CTRI registry http://tinyurl.com/ljg7aad and WHO's registry http://tinyurl.com/kcp4z89
...6-27-13: Bavi+CP MOS>14mos (“with < 60% of survival events”) http://tinyurl.com/pmcgsgp
…”with less than 60% of survival events, while the Bavi+CP arm currently demonstrates a MOS > 14mos, there was not a meaningful enough diff. in survival between the 2 arms that would support the advancement of this combination. Full results will be presented at a future sci. meeting or thru pub.”
...5-7-13: FTM's Table of 20 prev. Ph.3 Trials in 1st-Line NSCLC (ctl=CP), MOS & HR results (Note Avastin improved CP-Only by 19%): http://tinyurl.com/cho7o29
...3-9-12: Topline ORR & PFS Data (Bavi+PC vs.PC-only) http://tinyurl.com/7m9r6ya
…...LOCAL reads: ORR/32%-31% PFS/5.8-4.6mos , CENTRAL reads: ORR/25%-23% PFS/6.7-6.4mos
...12-6-11 Prelim. Data (n=86, 100% Stage IV's) => ORR=39%, PC/alone=25%: http://tinyurl.com/7ph4tty
......Comp. vs. Avastin+PC/Ph3/n=417(74% Stage IV's): ORR=35% (Sandler/E4599/2006 http://www.nejm.org/doi/pdf/10.1056/NEJMoa061884 )
...9-8-11: Enrollment complete. http://tinyurl.com/3vv9zfx
...7-14-11/CC: Enrollment was taking longer than expected; have amended protocol; expanding to 30+ sites, expect enroll. comp. "in coming weeks", interim data by Yr-end'11. http://tinyurl.com/6k6y2as
…7-14-10/CC, J.Shan (VP/Clin+RegAffairs): "This trial is intended to confirm in a randomized setting the results from our Ph.2 signal-seeking NSCLC trial which showed 43% ORR, more than double the generally accepted chemo ORR of under 20% in numerous publications. Favorable results could then lead to an end of Ph.2 meeting with the FDA, with possibly a pivotal Ph/3 trial for front-line lung cancer, our 2nd potential regulatory pathway for bavituximab."
...7-14-10: U.S. Ph.2b randomized trial initiated http://tinyurl.com/27kxksl
……up to 86 front-line patients at ~20 clinical sites; goal: enrollment comp. by mid'11.
F. LEAD IND: Phase IIb Bavi+Doce vs. 2nd-Line NSCLC (randomized, double-blinded, placebo-ctl'd, n=120, 'registrational')
Protocol: http://clinicaltrials.gov/ct2/show/NCT01138163 (24 U.S. + 15 India + 2 RepGA + 7 RussianFED + 5 Ukraine = 53 as of 8-12-11)
Enrolled Oct2010 - Oct2011 at 40 global sites (per J.Shan 9-7-12 webcast (http://tinyurl.com/8cn87la )
8-2012: Compare Bavi+Doce's MOS=11.7mos (Bavi/3mg) to the 4 Curr-Approved 2Line/NSCLC Drugs http://tinyurl.com/cgnkvpa
• Taxotere/docetaxel => MOS=6.3mos (meta-analysis of 5 trials, 865 pts)
• Altima/pemetrexed => No diff. vs. Docetaxel (Ph.3 non-inferiority vs. Doce, 571 pts)
• Tarceva/erlotinib => MOS=5.3mos (TITAN Ph.III n=424 trial - see http://tinyurl.com/8w8lo93 )
• Iressa/gefitinib => "Iressa does not improve OS"
2-19-16/Clin-Lung-Cancer-Jrnl: Ph2 Final Data, Dr. David Gerber et al http://tinyurl.com/z5a7fwu
9-8-15: CMS SETTLEMENT EXECUTED (CSM pays PPHM only $600k for their “breach of contract, negligence & constructive fraud”, due to “limitation of liability contract clauses”) http://tinyurl.com/pemub47 (10Q/pg.17)
6-23-14: PPHM files Opposition to CSM’s Motion For Partial Summary Judgment - excerpts: http://tinyurl.com/q8xwd4v
. . .Declaration of Joseph Shan (VP/Clin+RegAffairs): http://tinyurl.com/kdgllxn
. . .Declaration of Jeffery Masten (VP/Quality): http://tinyurl.com/oru9p5q #6: “up to 25%" of CTL had 1mg, and “up to 25%" of 1mg had CTL(DoceOnly).
3-28-14: Peregrine files 1st Amended Complaint vs. CSM (13pgs) http://tinyurl.com/lsgf5lz
...Pg6: ”[as of 4-15-10], CSM had already secretly & unilaterally swapped the A & B arms so that those patients that were randomized in the A arm (CTL) and supposed to receive placebo treatments, were actually receiving 1MG Bavi treatments, and vice-versa. Peregrine’s Fall’12 investigation revealed that CSM committed other labeling & distribution errors affecting the A & B arms above & beyond the swap of the A& B arms noted above.”
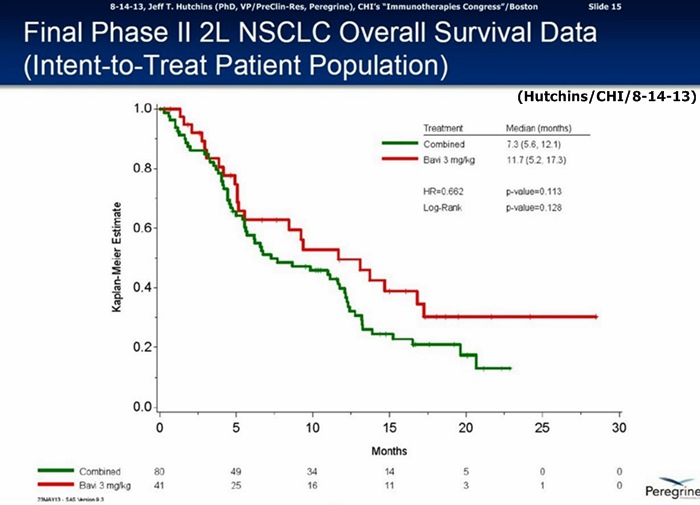
6-3-13/ASCO’13: Final Data Ph.II 2L/NSCLC http://tinyurl.com/my8qxw7
…60% improvement in MOS: Bavi/3mg=11.7mos. vs. 7.3mos. for CTL-arm(combined Bavi/1mg + DoxyOnly arms), HR=.662, P=.113
5-20-13: FDA Approves Bavituximab Ph.III Design for 2L/NSCLC; 600-pt trial to begin by y/e’13 http://tinyurl.com/n3dxtm6
...S.King: “We will now focus on starting the Ph.III trial while continuing ongoing partnering discussions.”
…R.Garnick: “This was a highly collaborative effort with the FDA; this trial, when combined with Bavi’s supporting data to date, could be sufficient to support a future BLA submission."
2-19-13: Topline Data Update from 2nd-Line NSCLC Trial after data discrepancies review http://tinyurl.com/ansqcea
…60% improvement in MOS: 3mg=11.7mos. vs. 7.3mos. for CTL-arm(combined 1mg & Doxy+placebo arms), HR=.73, p=.217
6-5-13: FTM's table of MOS data in 15 prior Doxy 2nd-Line NSCLC trials (Bavi's 60% MOS Improvement is Tops) http://tinyurl.com/m886ctb

1-25-13: MLV's George Zavoico recaps 2ndLine/NSCLC data errors & current status of PPHM's review http://tinyurl.com/b9u4pk8
...GZ: "This means that patients randomized into the high dose arm were administered Bavi correctly, whereas some of the patients in the placebo arm were administered low dose Bavi and some in the low dose Bavi arm were administered placebo. More importantly, the findings suggested that the MOS of 13.1 mos. in the high dose arm was likely to be valid. Even by historical measures, this is a remarkable result, since docetaxel's product insert lists the MOS of NSCLC patients receiving this widely used drug as 2nd-Line therapy in 2 trials as 5.7 & 7.5 mos. In effect, adding Bavi doubled the MOS. In our view, this was an extraordinary stroke of luck. If the high dose arm had been affected by the coding discrepancy, Peregrine would have been in a completely different & unfortunate position… Moreover, Peregrine must determine how best to present its case to the FDA. Will the historical controls be sufficient to justify moving Bavi into a Phase III pivotal trial, or will Peregrine have to pool the results of the placebo & low-dose arms and use that as a comparator to argue for moving ahead? A simple average of the placebo & low-dose arms results in a new control MOS of about 8.4 mos., still several months less than that of the high dose arm. This quick analysis results in about a 5-mo. survival advantage, a substantial prolongation for patients with second-line NSCLC and likely to justify moving Bavi into a pivotal Phase III trial in 2013, in our view."
1-7-13 PPHM PR - Review Update: "discrepancies are isolated to the placebo and 1 mg/kg arms; no evidence of discrepancies in the 3 mg/kg arm… Peregrine is taking a very conservative approach toward analyzing the results by combining the placebo & 1mg/kg arms into one treatment arm (control arm), and comparing to the 3mg/kg arm. This analysis indicates that the 3 mg/kg arm continues to show favorable TRR's, PFS, and OS over the new combined control arm. Peregrine expects to announce more detailed results from the analysis in the near term when it is completed." http://tinyurl.com/asup54d
9-24-12: Major Discrepancies found in 2nd-Line NSCLC Ph.2B Treatment Group Coding by Indep. 3rd-Party Vendor CMS/FargoND http://tinyurl.com/8r9zcqy
…"Investors should not rely on clinical data that the company disclosed on or before Sept. 7, 2012 from its Ph.2 Bavi trial in patients with 2nd-Line NSCLC or any presentations or other documents related to this Ph.2 trial."
9-24-12: Peregrine sues CSM Over Bavi Ph.2B 2nd-Line NSCLC Clinical Trial Mix-Up http://tinyurl.com/8fpgngu
…CSM = Clinical Supplies Management Inc., Fargo ND http://www.csmondemand.com
...1-17-13: Peregrine's lawsuit against CSM for "breach of contract & negligence" SERVED http://tinyurl.com/a7zrgys
…9-10-12 CEO Steve King, QtlyCC ( http://tinyurl.com/8nkwrml )
……"These are truly remarkable results (statistically doubling MOS) that are not only great for the pgm… but also great news for the NSCLC patients in the trial…"
…9-10-12 Robert Garnick (Head/Reg), QtlyCC ( http://tinyurl.com/8nkwrml )
……"The NSCLC data we announced 9-7-12 has far exceeded our expectations, and I hope that you're as excited as I am with bavituximab's potential. I feel strongly that Peregrine should be recognized for having the corporate courage to conduct the rigorous, randomized placebo-controlled Phase II trial that provided these robust data and that provide the basis for us to plan for a pivotal Phase III program."
...9-7-12: PPHM Press Release about Dr. Gerber's plenary at ASTRO/Thoracic/Chicago: http://tinyurl.com/96wrrso
…"The interim data showed a statistically significant improvement in OS (Hazard Ratio 0.524, p-value .0154) and a doubling of MOS (11.1/13.1mos. vs. 5.6mos.) in the Bavituximab-containing arms compared to the [Docetaxel] ctl-arm."
......VP Joe Shan's 15min. Webcast & Slideshow recapping Dr. David Gerber's 9-7-12 ASTRO/Chicago Plenary: http://tinyurl.com/96wrrso
…8-15-12 CEO Steve King, Wedbush/NYC ( http://tinyurl.com/8mhrtld )
......"As we're sitting here today, we have still not reached the # of events for MOS in either of the Bavituximab arms - and, in fact, we still have patients that are on treatments." Q&A: "it's going to be a very positive MOS result, it's just a matter now of magnitude."
…7-16-12 CEO Steve King, QtlyCC ( http://tinyurl.com/cs7spbz )
......"The strength of this 2nd-Line NSCLC data (esp. MOS trends) in this large area of high unmet medical need has also sparked a surge in partnering discussions that has included over 15 in-person partnering meetings since that time with major players in oncology, with all discussions ongoing and addl. parties showing interest. Our goal for the program is to position ourselves, along with a potential partner, to initiate Ph3 by mid-2013, which means an EOP2 meeting by yr-end'12. It would be ideal to have a partner on board to participate in the EOP2 meeting, and we have communicated this to interested parties and they agree."
…7-16-12 Robert Garnick (Head/Reg), QtlyCC ( http://tinyurl.com/cs7spbz )
……"We've been working very hard and very actively on the next steps in our Bavi 2nd-Line NSCLC pgm, given the favorable data that we've seen. As you can imagine, with data like this, there are many things that we need to consider. One consideration is that, should the data continue to trend the way it is, particularly in survival, this opens a door for potential discussions around a pathway for Accelerated Approval. At this point, all options are being considered, with Peregrine working towards the most efficient path forward from a regulatory standpoint." Q&A: "…all in all, I think the data is extremely compelling and I think it makes a really good case. Certainly, I think, I've seen a lot of Ph2 & Ph3 data, and this is as compelling Ph2 data as I've ever seen. So, I'm very comfortable proposing a meeting with the FDA for Q4'12."
…7-12-12 CEO Steve King, JMP-Conf/NYC ( http://tinyurl.com/csdclwb )
……"Re: 2nd-Line/NSCLC trial, the most thrilling thing is the fact that, even though we'd reached MOS for the ctl-arm(Doce) at end of Apr'12 of LESS THAN 6MOS, the majority of patients are still alive (today) in both Bavi arms, and we expect that to continue for some period of time still. Ph3 planning is underway already; our goal is to start this Ph3 by mid'13, meaning an EOP2 meeting with the FDA in Q4'12; our goal is to bring a partner on board, ideally in time for that EOP2 meeting, certainly before the beg. of the Ph3 trial."
…5-21-12: TopLine data n=117 for Bavi/3mg+Doce arm: ORR=17.9%/PFS=4.5mos (vs. CTL 7.9%/3mos) http://tinyurl.com/73aeyxj
......Importantly, MOS for CTL-arm "< 6 mos", but not yet reached in both Bavi arms.
...10-6-11: Enrollment complete. http://tinyurl.com/3m9re39
...7-14-11/CC: Enrollment was taking longer than expected; have amended protocol; expanding to ~45 sites, expect enroll. comp. "early in Q4/2011", data unblinding 1H'12. http://tinyurl.com/6k6y2as
…3-17-10/Roth, CEO S.King: "We refer to this trial as a Registrational Phase II Study, because we believe that if we have results anywhere near approaching what we saw in the earlier [India] study, it could be a conduit for Accelerated Approval."
...6-4-10: Ph.2b randomized reg. trial Open for enrollment: http://tinyurl.com/25v22qk
……"up to 120 refractory patients at ~30 clinical sites; goal: fully-enroll by mid'11, topline data by y/e'11."
1st 5 COMPLETED BAVI CANCER TRIALS:
These 5 completed Ph.1 & Ph.2 Bavituximab Cancer Trials archived to: http://tinyurl.com/72dnkfg
1. Ph.1A Bavi/Mono vs. Solid Cancers (USA n=26, 6/2005-6/2009)
2. Ph.1B Bavi+Chemo vs. Solid Cancers (India n=12, 11/2006-5/2007)
3. Ph.2 Bavi+Doce/BREAST/Refractory (RepGA n=46, 1/2008-5/2009): ORR=61%(Her2+=86%), PFS=7.4mos, MOS=20.7mos *
4. Ph.2 Bavi+PC/BREAST/Frontline (India n=46, 8/2008-9/2009): ORR=74%(Her2+=100%), PFS=6.9mos, MOS=23.2mos %
5. Ph.2 Bavi+PC/NSCLC/Frontline (India n=49, 6/2008-10/2009): ORR=43%, PFS=6.1mos, MOS=12.4mos #
......* BAVI+DOCE/Refract-MBC N=46: Compare *MOS=20.7mos to 11.4mos for Doce/alone (Nabholtz/JCO1999 Ph3/n=203 http://tinyurl.com/3rxqqtk )
......% BAVI+PC/FrontLine-MBC N=46: Compare %MOS=23.2mos to 16.0mos for P+C/alone (Loesch/JCO2002 Ph2/n=95 http://tinyurl.com/6wazs9p )
......# BAVI+PC/Frontline-NSCLC N=49: Compare #MOS=12.4mos to P+C/alone=10.3, Avastin+PC=12.3 (E4599/n=434), achieved using less Chemo (175-v-200 & AUC5-v-AUC6), treating 16% (8/49) more-difficult Squamous in Bavi trial (excluded totally from E4599), and treating higher % of sicker ECOG1 pts than in E4599 (96%-v-60%). See 6-15-11/PR http://tinyurl.com/3fcz5ok , ASCO'10 http://tinyurl.com/2g5cqof , and a discussion of differentiating factors between patient demographics & baselines treated in the 2 trials: http://tinyurl.com/6k5uuf7 .
BAVI HEPC TRIALS (2005-2011) ARCHIVED From iBox 7-16-17
Now linked to from within iBox for perpetuity: http://investorshub.advfn.com/boards/show_ibox.aspx?boardid=2076
4 COMPLETED BAVITUXIMAB HEP-C PHASE1+2 TRIALS (2005-2011):
9-9-11: PPHM is positioning Bavi as potential Interferon replacement in HCV cocktails: http://tinyurl.com/3novgzk
12-2011: Nature Medicine article lists Peregrine among 11 co's dev. interferon-free treatments for HCV: http://tinyurl.com/87ltkmt
7-2011: Nature Biotechnology article quotes Dr. Thorpe on Bavi's goal to elim. IFN-A: http://tinyurl.com/3n8qluq
D. Phase II Bavi+Riba vs. Frontline-HEPC (randomized, open-label):
Protocol (init=1-2011): http://clinicaltrials.gov/ct2/show/NCT01273948
12-29-11: HCV Ph.2 Trial Update http://tinyurl.com/coq2969
……SK: " this is a good time to seek partners for the antiviral pgm. which has shown promise in this study."
9-26-11: Enrollment Complete in Bavi+Riba/HCV trial http://tinyurl.com/3j9bba8
1-10-11: Bavi+Ribavirin/HepC Ph.2 trial initiated http://tinyurl.com/4uz97tv
......"~66 patients with previously untreated genotype-1 chronic HCV, 1 of 3 arms (.3Bavi+R / 3.0Bavi+R / PEGIFN+R) for 12 wks, testing for safety & antiviral activity."
C. Phase 1B HCV-HIV Co-Infected's Mono Repeat-Dose Trial:
Protocol (init=7-2007): http://clinicaltrials.gov/ct/show/NCT00503347
…4-2-11: Ph.1B trial data presented at EASL-2011/Berlin on 4-2-11 http://tinyurl.com/3mmwy5d
…1-31-11: Enrollment Complete in HCV-HIV trial http://tinyurl.com/4tagpz2
...7-10-07: Enrollment begins at St.Michaels (Peter Ho Mem. Clinic, NJ) http://tinyurl.com/2g7rdp
......Ascending dose levels of Bavi weekly for 8 wks; ~24 pts; designed to assess Safety & PK, but "HCV & HIV viral titers & other biomarkers will be evaluated".
...5-17-07: New Clinical Protocol filed with FDA: http://tinyurl.com/2lpacm
These 2 completed Ph.1 Bavituximab Hep-C Trials archived to: http://tinyurl.com/27f58fc
1. Phase 1A Bavi/Mono Single-Dose HCV Trial: (init=8-2005 comp=2-2006 n=30)
2. Phase 1B Bavi/Mono Repeat-Dose HCV Trial: (init=6-2006 comp=1-2007 n=24)
COTARA & ANTI-VEGF/2C3 PGMS ARCHIVED From iBox 7-16-17
Now linked to from within iBox for perpetuity: http://investorshub.advfn.com/boards/show_ibox.aspx?boardid=2076
TUMOR NECROSIS THERAPY (TNT/COTARA/VEA's) NEWS:
Highlights of Cotara/GBM Trials History 1998-2009: http://tinyurl.com/y8fyl2r
12-5-12: FDA OK's Ph3 Pivotal Trial Design for Cotara/GBM (single-infustion, dual catheters) http://tinyurl.com/czjy6mf
…SK: "We can now escalate our business development activities to secure a partnership, recognizing the great interest by companies in drug candidates within the orphan and rare disease space."
5-3-10: PPHM Licenses TNT rights in "Certain APEC Countries" to STASON(Stonsa Biopharm) http://tinyurl.com/29x433b
Stonsa's website: http://stonsabio.com
...Harry Fan/CEO/Stason, "This agreement represents a unique opportunity to acquire a novel, cutting-edge pharmaceutical technology package and bring innovative & promising treatments to millions of cancer patients in Asia & the Pacific Rim. We will also continue developing new applications & bioproducts based on TNT technologies to complement Peregrine's activities in the glioblastoma arena and will aggressively pursue new, diverse biotechnology markets. To advance these efforts, a new spin-off biopharmaceutical company (Stonsa Biopharm Inc.) headed by Dr. Eugene Mechetner will be formed which will focus on commercializing TNT technologies."
7-20-11: Dr. Missag Parseghian now VP/R&D at Stonsa Biopharm; pub's article in Jrnl/Chromatography http://tinyurl.com/m3c69w
…6-2010: More on PPHM-Stason Collab., "TNT For Tumors", Lloyd Dunla, Drug Disc. News http://tinyurl.com/28vmkzm
…5-27-10: Stonsa's CEO Dr. Eugene Mechetner presents TNT at Taiwanese Bio-Seminar http://tinyurl.com/29r7one
...12-16-10: "Stason Inks Deal to use Lonza's GS Gene Expression System with TNT Antibody Platform" http://tinyurl.com/2dzz4b5
7-1-09 Eur. Patent #1638989 for 'In-Line Labeling' (used in Cotara GBM trials) & JNM Article: http://tinyurl.com/mlgz8p
India (expanded Jan'10 to U.S.) Cotara/Brain Ph.2 Trial (40 patients, 1st relapse):
India's DCGI protocol (init=7-2007): http://clinicaltrials.gov/ct2/show/NCT00677716
7-2011 BRIT (India Radiolabelling co.) says: "(Cotara) Ph2 completed; another order is expected from Peregrine to enable them to go ahead with Ph3" http://tinyurl.com/43vb4ax
10-11-11: SIRO Clinpharm is Peregrine's Indian CRO for GBM/Cotara Ph.2 Trial: http://tinyurl.com/3eobdca
6-3-11 ASCO'11: Dr. William Shapiro (Barrow) presents Cotara N=41 Data http://tinyurl.com/3g7oz55
…"Interim MOS=8.8mos(38wks); we are eager to meet with the FDA in Q4'11 to determine the optimal registration pathway for Cotara."
...NOTE: In 12-12-2011 QtlyCC, S.King reported MOS at 9.3 mos., up from in "interim" 8.8mos reported at ASCO'11. http://tinyurl.com/75rfwrv
…Dr. Shapiro's Cotara Poster #2035 (PDF & Images) http://tinyurl.com/3wetx5r
5-19-11: ASCO'11 preview; Dr. Vladimir Evilevitch (ex-Novartis) hired as Med.Dir. "to execute Cotara Reg&Clin. strategy" http://tinyurl.com/3av4red
12-20-10: Treatment Complete in Cotara/GMB Ph.2 Trial http://tinyurl.com/2844wrq
…J.Shan: "We expect top-line data by mid-year 2011 and plan to meet with the FDA to define the optimal registration pathway for Cotara."
10-18-10: Interim Ph.2 update (n=14) at CNS'10: http://tinyurl.com/349kvca
...PI Dr. Deepak Gupta, ”MOS of 86 weeks far exceeded our expectations in this very difficult to treat patient pop. where treatment options are few and rarely extend med.survival beyond 6mos."
…Roth's Joe Pantginis: "Physicians know how long these patients are expected to survive on avg. and that's why these data are very, very promising. Because there is a huge unmet medical need for new treatments for this type of brain cancer, Peregrine may seek an accelerated approval that could avoid lengthy Phase III clinical trials."
...Link to Dr. Gupta's CNS'10 Poster (6pg PDF): http://tinyurl.com/26b8sp9
...6-3-10/ASCO: Cotara/Ph.2 Trial 75% enrolled; 3 more U.S. sites added: http://tinyurl.com/33z8ggo
...1-28-10 U.S. site (Barrow/Phoenix) added formerly India-Only Ph.2 trial: http://tinyurl.com/yk565jy
…Per 3-11-10 QtlyCC, VP/ClinAffairs J.Shan said also exp. UPenn & Univ. of S.Car. http://tinyurl.com/yl4befh
…9-2-09 Interim Ph.2 data (10 pts) presented at AANS Annual Mtg/Boston: http://tinyurl.com/mxzbzm
……P.I. Dr. A.K. Mahapatra: "Most importantly, Cotara has demonstrated promising signs of efficacy."
...8-2-07 1st Patient Dosed in Indian Cotara/Brain Ph.2 Trial: http://tinyurl.com/296mcj
...The obvious desire is to compare Cotara vs. SOC Temodar for GBM therapy: http://tinyurl.com/yttt99
USA Cotara/Brain 'Dosimetry & Dose Confirmation Trial' (originally funded by NABTT):
U.S.A. Cotara Brain Cancer trial protocol (added 8-14-07): http://clinicaltrials.gov/show/NCT00509301
6-6-10/ASCO: Final Results of Cotara/GBM Ph.1/Dosimetry trial (n=12): http://tinyurl.com/24qjxxh
......"Final data confirm Cotara's targeting capabilities, delivering 300-fold higher radiation levels to the tumor than to normal organs."
...2-11-10: "Current Cancer Therapy Reviews" article on Cotara/GMB http://tinyurl.com/yg2on8f
......the cases of "2 patients who have survived more than 9 years" are also reviewed.
...12-2-09: U.S. Trial Enrollment Complete http://tinyurl.com/yez7lzd
...6-16-09: Cotara/Brain Oral-Pres. at SNM Annual Meeting http://tinyurl.com/lmhkw2
......Dr. Sui Shen (U-Alabama), "With a mean dose ratio showing 300-fold greater delivery of radiation to the tumor as compared to other organs, Cotara represents a potentially valuable new therapy for GBM patients."
…9-23-08: Article in Cleveland paper - comments by P.I. Dr. Andrew Sloan (Case Western/CLEV) http://tinyurl.com/3mkmas
...5-31-08: Cotara USA Ph.1B data presented at ASCO/2008: http://tinyurl.com/68apro
...8-29-05: NABTT Initiates Cotara/Brain Trial (28 patients/4 sites): http://tinyurl.com/9w3cr
...Orig. NABTT protocol, "6-2007, completed": http://www.clinicaltrials.gov/ct/show/NCT00128635
...The Phil (Marfuta) Bannister Story (NABTT Cotara/GBM patient #1, diag. 2-4-06): http://tinyurl.com/24gkml
......4-22-10: Phil reports on YASG that his GBM was misdiagnosed; he has a GrIII Oligodendroglioma, which carries a MLE of 12yrs! http://tinyurl.com/23mhboa
Previous USA Cotara/Brain Ph.1-2 Trials, completed in 2003:
...Slides showing correlation between Cotara dosage levels and MST vs. Temodar(curr.SOC): http://tinyurl.com/26s265
...4-27-08 update: The Jerod Swan Cotara/Brain Success Story (10 years after diag.): http://tinyurl.com/68ofsv
...5-22-07 update: The Freddie Sanford Cotara/Brain Success Story (7 years after diag.): http://tinyurl.com/2du2e5
...6-1-05: Cotara w/CED Brain Delivery pub. in Neurosurgery Jrnl: http://tinyurl.com/anmaa
..."Cotara Holds Promise for Treating Brain Cancer - P1/P2 Data Suggests Extended Survival in a Number of Patients"
PRIOR COTARA/GBM PH.3 FDA NEGOTIATIONS (2 INFUSIONS, SINGLE-CATHETER)
…12-13-01: FDA OK's Ph3 Design for Cotara/GBM (2 doses, single interstitial catheter) http://tinyurl.com/a7m3xjw
…7-1-02: Peregrine Provides Update on Status of Cotara Phase III http://tinyurl.com/bac9rtr
......"Peregrine FDA have been in close consultation regarding the Cotara Ph3 trial. Peregrine is confident that the Ph3 will adequately address all regulatory, drug safety & efficacy requirements for such a large multi-national trial."
…11-19-02: Peregrine Provides Update on Status of Cotara Phase III http://tinyurl.com/a4mw5yj
......ELegere: "Although many issues have been finalized, there are several important issues that remain to be negotiated prior to Ph3 approval. We will request an expedited meeting with the FDA so we can clarify & finalize the remaining issues…"
…11-25-02: Peregrine Receives Orphan Drug Designation for Cotara in Europe http://tinyurl.com/b7wr9at
…2-24-03: Peregrine Receives FDA Approval for its Cotara Phase III Registration Trial Design for GBM http://tinyurl.com/bjowgh6
......"Peregrine has received approval from the FDA to start its Cotara Ph3 registration clinical study in GBM."
TNT3 (Vivatuxin) Lung Cancer Approval/Launch in China:
...Shanghai MediPharm Biotech's TNT/China website: http://www.vivatuxin.com
...8-2010 Profile of Shanghai MediPharm Biotech by WallStreetResearch (Alan Stone): http://tinyurl.com/2fn3nvk
...3-2010: New Vivatuxin+MicrowaveAblation NSCLC trial (n=2000) http://tinyurl.com/2ccghvr
...1-16-07: Medipharm Launches TNT in China for Lung Cancer http://tinyurl.com/ttlne
...6-19-02: TNT Interim Lung Data from China: http://tinyurl.com/ggcba & http://tinyurl.com/em6da
Peregrine v. CTL Lawsuit - Settled 6-19-09:
...6-19-09: Form 8-K, Peregrine & CTL Settle: http://tinyurl.com/mhgcg7
...3-29-07 CTL countersues PPHM over TNT/China licensing dispute: http://tinyurl.com/39k8sx
...3-30-07 PPHM comments on CTL's 3-29-07 countersuit: http://tinyurl.com/33xzzq
...1-12-07: New China Subsidiary; Suit Filed Against Cancer Therapeutics Labs (CTL) http://tinyurl.com/y4vzbj
Eur. Licensee Merck-KGaA's TNT/Cytokine Fusion Protein Ph.1 Trial:
...4-3-12: NHS-IL2-LT (EMD 521873) poster at AACR'12 http://tinyurl.com/7bm4lpx
...4-2009: 2nd SELECTIKINE Ph.1 trial added: SELECTIKINE+RAD/NSCLC: http://tinyurl.com/pzl9j8
...5-2007: 2nd SELECTIKINE Ph.1 site added, Univ. of Lausanne, Switz: http://tinyurl.com/2l933s
...Ph.1 Trial Protocol (Merck KGaA/Germany, PI=Dr. R.Stupp): http://tinyurl.com/2f4u2t
...2-22-07: TNT-based European Cancer Trial initiated by 'Licensee': http://tinyurl.com/2zcjqr
...Clearly, it's Merck-KGaA's 'SELECTIKINE' (NHS-IL2-LT/EMD521873): http://tinyurl.com/yud7pw
VEAs:
...5-1-08 Dr. Missag Parseghian (Dir/R&D) presents VEAs at PEGS/2008: http://tinyurl.com/66onj2
AFFITECH-PPHM Collab: Fully-Human Mabs, Anti-PS & Anti-VEGF/2C3
• AT001 is Fully-Human 2C3 = R84 = PGN311
• AT004 is Fully-Human BAVI=PGN635=1N11
• AT005 is the Duke-PPHM-HIV candidate PGN632=11.31
4-28-09 Affitech comments on PGN635 & PGN632 presented at AACR'09: http://tinyurl.com/cxkcb4
...All Affitech Press Releases: http://www.affitech.com/investors-news/press-releases-english
ANTI-ANGIOGENESIS AGENT '2C3' [Fhu: R84=At001] ANTI-VEGF MAB:
8-20-12: 1st patient dosed in R84 (AT001) Ph.1 "Various Cancers" Trial in Russia/CIS http://tinyurl.com/8ahaw3l
3-12-12 Affitech's Board rec's S/H's approve Trans Nova's buyout offer (7.85mm Euros for rem. 60%) http://tinyurl.com/6qwh4zb
6-16-11: Affitech partners with IBC Generium for R84 dev. in Russia/CIS http://tinyurl.com/6hezkux
…Affitech rec. $2.5mmEURO from IBC; PPHM re-invests $1.6mm payment into "further advancement of R84."
5-26-11 Affitech update on R84 progress: "trial matl. mfg. (by Avid)" http://tinyurl.com/3hu5nqe
4-28-11: Affitech's Annual SHM (32pg. PDF) - much on R84 progress/plans: http://tinyurl.com/3h47nqa
..Affitech's 12-31-10 10-K iss. 4-28-11 - much info on R84 as well: http://tinyurl.com/3dcnefa
…$1mm milestone to PPHM for pre-clin-pkg forthcoming? Also, Avid "1st-GMP-Mfg" => http://tinyurl.com/3hc6bw9
4-28-11: Affitech signs with UTSW/Dr. Rolf Brekken for further AT001/R84 research: http://tinyurl.com/4ybh2l2
...Dr. Rolf Brekken's research at UTSW: http://www4.utsouthwestern.edu/brekkenlab/research.htm
2-18-11 Interview with Affitech CEO Dr. Martin Welschof discusses R84 Progress/Plans http://tinyurl.com/5rhmlog
Nov/Dec'10: Affitech Reiterates R84 (FH-2C3) Dev. Project Plans http://tinyurl.com/2fhyzxz
9-30-10: PPHM & Affitech Amend 7-2009 R84 Licensing Agreement http://tinyurl.com/2cn77c2
…following Affitech/NTS+ Collab for dev. in Russia/Brazil. Also, Avid gets contract to mfg. R84 for clinical studies.
8-6-10/PLoS: PPHM-Brekken/UTSW/Affitech article on pre-clin. R84=AT001 (Fhu 2C3): http://tinyurl.com/2unp4j3
…"Extended r84 therapy controls tumor growth without induction of toxicity"
6-30-10: NTS-Plus to run 2C3/R84 clinical trial(s) in Russia: http://tinyurl.com/26hhln4
…"AT001/r84 differs from Avastin in a number of ways… more selective… may have a better side effect profile."
4-21-10: Affitech sub-licenses Anti-VEGF mabs (incl. R84) to Russian partner NTS-Plus: http://tinyurl.com/29tzen9
11-3-09/PLoS: R.Brekken (PPHM SRB) on R84, increases "immune cell infiltration" in Breast Cancer: http://tinyurl.com/y8ooxah
7-22-09: Peregrine Licenses Anti-VEGF mabs (incl. R84) to Affitech: http://tinyurl.com/n67v57
6-30-09: R.Brekken article in Mol-Cancer-Ther on 2C3/R84+Avastin vs. Breast Cancer: http://tinyurl.com/mqv5ol
12-11-08: Dr. Brekken (PPHM SRB) presents F.H. 2C3 (R84/PGN311) at TX Breast-Cancer Conf. http://tinyurl.com/63utya
11-3-07: Dr. Brekken presents 'R84' (F.H. 2C3, aka PGN311) data at IBC-Conf: http://tinyurl.com/yp459d
4-16-07 AACR2007: Brekken [PPHM SRB] presents fully-human 2C3 ('R3') http://tinyurl.com/2udkgr
"The anti-tumor activity of 2C3, which only blocks VEGFR2 compared favorably to that of Avastin, which blocks both VEGFR1+2"
2-14-05: Humanized 2C3 Pre-Clin. data presented at "Anti-Angio. Symposium" http://tinyurl.com/85qy3
2-10-05: VEGF Pioneer Dr. Donald Senger joins PPHM's SRB to assist with 2C3 pgm: http://tinyurl.com/yul72w
OTHER PEREGRINE DRUG CANDIDATES:
5-6-12: Dr. Phil Thorpe's poster on PGN632 vs. AMD/CNV at ARVO-2012/FtLaud http://tinyurl.com/6q6kuwa
1-9-07 PR: VTA Microbubbles as Contrast Agents (AACR/CCR article) http://tinyurl.com/v7mf9 & http://tinyurl.com/w4pe9
9-26-06: VTA+Combo Patent Granted; Expands VTA Licensing Opportunities http://tinyurl.com/er4hh & http://tinyurl.com/ry5a4
8-25-05: PPHM Licenses Conjugated Anti-PSMA VTA Technolody to Medarex: http://tinyurl.com/dv6rt
"These types of deals should add considerable value to the company as our partners move their VTA dev. programs forward."
4-19-05: VEGF121/rGel presented at AACR (50% Survival vs. Prostate Cancer): http://tinyurl.com/8gazv
###SUPG moving VEGF121/rGel (lic. from PPHM) fwd. via Targa/Rosenblum: http://tinyurl.com/ylevod
...Rosenblum 11-15-06, "Ph.I human trials of VEGF121/rGel are expected to open shortly at M.D.A."
How do I convert iBox formatting back to “posting” format? I need to archive a bunch of really old stuff to iHub posts and then link to them in iBox with something like:
“ARCHIVE of OLD Trials: http://investorshub.advfn.com/boards/read_msg.aspx?message_id=15555408 “
Is there a tool to allow me to CONVERT already marked-up iBox text like this to “iHub Post Suitable” format that retains the original BOLDS and ITALICS etc and the highlighted URL’s?
EXAMPLE – say this is in iBox:
7-14-17/CEO: “Another part of our effort to attract new customers is to expand our service offerings. Our scientists have developed a state-of-the-art Antibody Discovery & Characterization Platform through…
“The Antibody Discovery Platform discussed earlier [Bioservices] could also be instrumental on the R&D side of the business, allowing us to rapidly identify antibodies against already validated targets, as well as the potential to identify novel targets.
http://tinyurl.com/yb4wulvu
You can expect to hear more about this new capability over the coming months on both sides of our business.”
I want to somehow convert this to the following, so I can then create a new iHub post that retains the original markups and urls as in this:

Once I have this, I can then easily copy/paste to a new “Archive iBox” post that looks exactly like the iBox stuff did.
I need a tool to do this. I can’t be unique!!!
7-14-17: Updates on the PS Exosomes Diagnostic Platform – from the Q4/FY17 earnings PR, the CC, and the 10K...
7-14-17/CEO Steve King: “On the R&D side, we are pleased to report that the company continues to make progress with its PS-targeting Exosome Diagnostic Technology that is designed to detect & monitor cancer. Our scientists have successfully optimized the assay and we are currently preparing to generate addl. data, testing the diagnostic with an expanded set of human samples. Such data will be important to partnering discussions and we’ll keep you posted on progress. http://tinyurl.com/yb4wulvu
- - - - - - - - - -
**6-22-17: Nature/BJoC article, Dr. Alan Schroit/UTSW etal: "Detection of PS+ Exosomes for the Diagnosis of Early-Stage Breast & Pancreatic [Using Betabody KL15C]" http://tinyurl.com/ycrx9672 <= This article has not been PR’d or mentioned as yet by PPHM, to my knowledge.
**2-9-17/PR: PS+ Exosomes Proof-of-Concept Data (N=44, Ovarian, Dr. Alan Schroit/UTSW etal) pub. 1-22-17 in OncoTarget http://tinyurl.com/jhv57ua
From the 7-14-17 PR: http://tinyurl.com/yb4wulvu
PS EXOSOME TECHNOLOGY HIGHLIGHTS:
The company continues to make progress with its PS exosome diagnostic technology that is designed to detect and monitor cancer. The assay has been successfully optimized and we are currently preparing to generate addl. data by testing human samples. Such data will be important to partnering discussions.
4-30-17 10-K pub. 7-14-17 pg.6: http://tinyurl.com/ycxu4l5n
Exosome Technology
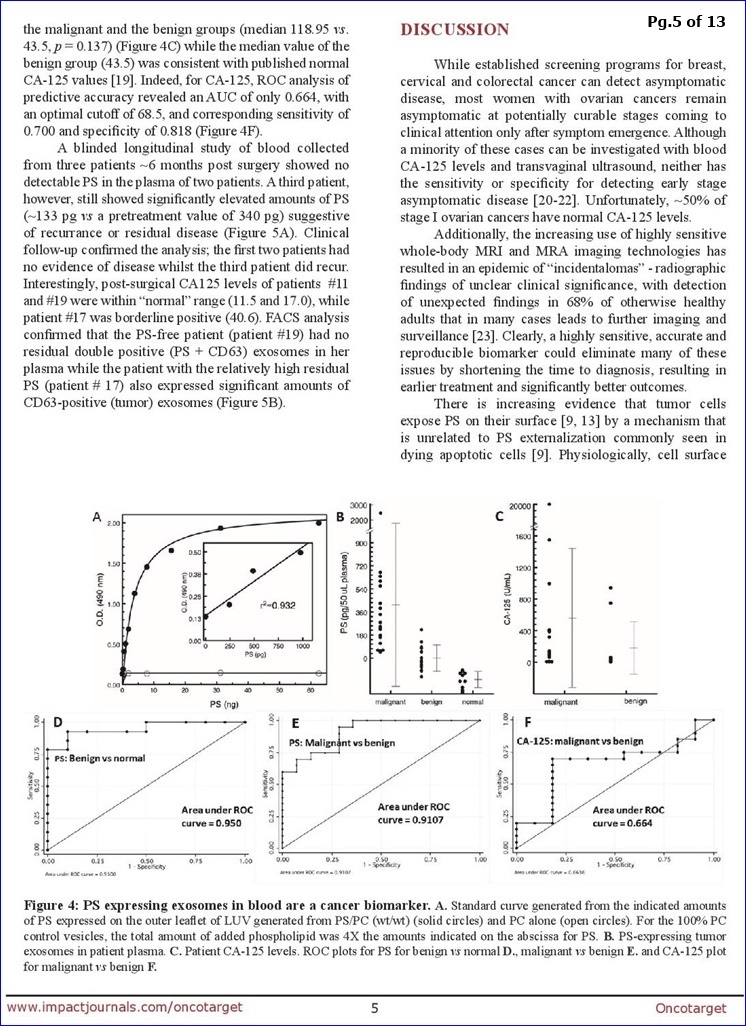
We licensed from UTSWMC in July 2016, proprietary Exosome-Based Cancer Diagnostic Technology, with the goal of developing an optimized test for further clinical testing in collaboration with a potential partner. The platform is based on the diagnostic potential of tumor derived exosomes, which are small vesicles from tumor cells that are released into the blood as tumors grow. Tumor derived exosomes have PS on their surface as a potentially detectable marker. It is believed that even small tumors begin to release PS-positive exosomes and thus the ability to detect these exosomes in the blood may be an indicator of the presence of a tumor. In the study published by Oncotarget [1-22-17: http://tinyurl.com/yb4wulvu ], plasma samples from 34 patients with ovarian tumors and 10 healthy subjects were analyzed for the presence of PS-expressing exosomes in a blinded test. Results demonstrated that those patients with malignant ovarian cancer displayed significantly higher blood PS exosome levels than those with benign tumors (median 0.237 vs. -0.027, p=0.0001) and the malignant and benign groups displayed significantly higher blood PS exosome levels than the healthy subjects (median 0.237 vs -0.158, p < 0.0001 and -0.027 vs -0.158, p=0.0002, respectively).
.
.
= = = = = = = = = = = = = = = = = = = = = = = = = = =
PPHM’s Exosomes Diag. Platform (incl. 6-22-2017 NATURE/BJC article)
...Known POC Data Summary (see below):
1-22-17/OncoTarget: Ovarian – senior author Alan Schroit (UTSW/PPHM SAB)
6-22-17/Nature(BJC): Breast & Pancreatic – senior author Alan Schroit (UTSW/PPHM SAB) *Using Betabody KL15C*
PPHM’s PS+ EXOSOME-BASED CANCER DETECTION & MONITORING TECHNOLOGY ("Liquid Biopsy") – Reverse Chron. History
...Excellent Exosome (aka microparticles, microvesicles) info: http://www.exosome-rna.com
6-22-17: Nature (British Jrnl of Cancer): ”Detection of Phosphatidylserine-Positive Exosomes for the Diagnosis of Early-Stage Malignancies [Using Betabody KL15C]”
Raghava Sharma, Xianming Huang, Rolf A Brekken (PPHM SAB), Alan J Schroit
….Dr. Schroit: PPHM SAB, UTSW profile: http://tinyurl.com/yaqnvcvq ; pubs: http://tinyurl.com/lqbd8kt
https://www.nature.com/bjc/journal/vaop/ncurrent/pdf/bjc2017183a.pdf
ABSTRACT
Background: There has been increasing interest in the detection of tumor exosomes in blood for cancer diagnostics. Most studies have focused on miRNA and protein signatures that are surrogate markers for specific tumor types. Because tumor cells and tumor-derived exosomes display phosphatidylserine (PS) in their outer membrane leaflet, we developed a highly sensitive ELISA-based system that detects picogram amounts of exosomal phospholipid in plasma as a cancer biomarker.
Methods: This report describes the development of a highly specific and sensitive ELISA for the capture of PS-expressing tumor exosomes in the blood of tumor-bearing mice. To monitor the relationship between tumor burden & tumor exosome plasma concentrations, plasma from one transplantable breast cancer model (MDA-MB-231) and 3 genetic mouse models (MMTV-PyMT; breast and KIC and KPC; pancreatic) were screened for captured exosomal phospholipid.
Results: We show that quantitative assessment of PS-expressing tumor exosomes detected very early-stage malignancies before clinical evidence of disease in all 4 model systems. tumor exosome levels showed significant increases by day 7 after tumor implantation in the MDA-MB-231 model while palpable tumors appeared only after day 27. For the MMTV-PyMT and KIC models, tumor exosome levels increased significantly by day 49 (P<=0.0002) and day 21 (P<=0.001) while tumors developed only after days 60 & 40, respectively. For the KPC model, a significant increase in blood exosome levels was detected by day 70 (P=0.023) when only preinvasive lesions are microscopically detectable.
Conclusions: These data indicate that blood PS exosome levels is a specific indicator of cancer and suggest that blood PS is a biomarker for early-stage malignancies.
- - - - -
Addl. article info (Fig.1) extracted by Bamboozler762 6-23-17 iHub #300359 http://investorshub.advfn.com/boards/read_msg.aspx?message_id=132436726
Fig1: Schematic diagram of betabody, KL15C
- - - - - - - - - -
Note: BETABODIES (Clipped/Nicked B2GPI - ex: KL15, “2nd-gen. PS-Targeting”) - bind to PS directly, are smaller in size (100 vs. 250KDa) and have a longer serum half-life (~5days) than natural antibodies (Bavi=~1day) – see http://tinyurl.com/khopa3d …2/17/15: UTSW/PPHM’s BetaBodies patent#8,956,616 Awarded(Granted) http://tinyurl.com/p75uyfu

Fig.1: Schematic diagram of betabody, KL15C
Domains 1 & 5 of the plasma PS-binding protein, B2GP1, were genetically fused to the C-terminus of the CH3 domains of the Fc fragment of human IgG1. A Gly4Ser linker was inserted between the CH3 domains and domains 1 and 5 of B2GP1. The recombinant betabody was expressed as a dimer.
- - - - - - - - - -
...Glimpse of beg. of 7pg. 6-22-17 BJC article: http://www.readcube.com/articles/10.1038/bjc.2017.183

2-23-17/StockNewsUnion.com: “What Next For Peregrine Pharmaceuticals After Exosomes Proof of Concept?”
Peregrine has said that its sight is on developing the technology into an optimized diagnostic platform for clinical testing of cancer. To reach that goal, Peregrine said it is seeking strategic partners to help it develop & commercialize the platform. Those partners would be expected to inject money into the project for an equity stake in PPHM or a cut of revenue from the sale of the platform. There are several large pharmaceutical companies developing their own ovarian cancer diagnostic systems are in need of such technology, so PPHM could pitch to them to join its exosomes-based diagnostic efforts.
POTENTIAL PARTNERS:
* AstraZeneca plc (NYSE:AZN https://www.astrazeneca.com - http://finance.yahoo.com/quote/AZN MktCap=$72B)
* Tesaro Inc. (NASDAQ:TSRO http://www.tesarobio.com - http://finance.yahoo.com/quote/TSRO MktCap=$9.5B)
* Clovis Oncology Inc. (NASDAQ:CLVS http://clovisoncology.com - http://finance.yahoo.com/quote/CLVS MktCap=$2.6B)
...are some of the major pharma brands that could be a fit for PPHM. The pharma giants are at various stages in the development of their ovarian cancer assets.”
http://stocknewsunion.com/what-next-for-peregrine-pharmaceuticals-nasdaqpphm-after-proof-of-concept/2043/
2-9-17/PR: ”Recent Publication Highlights Proof-of-Concept Data Supporting the Diagnostic Potential of Phosphatidylserine-Positive Exosomes in Ovarian Cancer”
http://ir.peregrineinc.com/releasedetail.cfm?ReleaseID=1011223
TUSTIN, Feb. 9, 2017: Peregrine Pharmaceuticals, Inc. (NASDAQ:PPHM/PPHMP), a biopharmaceutical company committed to improving patient lives by manufacturing high quality products for biotechnology and pharmaceutical companies and advancing its proprietary R&D pipeline, today announced the publication of positive proof-of-concept data for a novel exosome-based cancer detection platform. Results of the study, conducted at University of Texas (UT) Southwestern Medical Center, showed researchers were able to distinguish between healthy subjects and patients with ovarian tumors based on the levels of exosomes containing phosphatidylserine (PS) found in their plasma. Furthermore, analysis of the PS-positive exosome levels allowed researchers to distinguish between malignant and benign tumors. These data were recently published online by the peer-reviewed journal, Oncotarget, in a paper titled, "Detection of Phosphatidylserine-Positive Exosomes as a Diagnostic Marker for Ovarian Malignancies: A Proof-Of-Concept Study."
Peregrine is currently advancing the proprietary exosome-based cancer diagnostic technology, licensed from UT Southwestern Medical Center in July 2016 [7-14-16: http://tinyurl.com/zszd4fj ], with the goal of developing an optimized test for further clinical testing. As part of these efforts, the company is in the process of seeking a strategic partner for collaboration on developing and commercializing the technology. The platform is based on the diagnostic potential of tumor exosomes, which are small vesicles from tumor cells that are released into the blood as tumors grow. Tumor derived exosomes have PS on their surface as a detectable marker. It is believed that even small tumors begin to release PS-positive exosomes and thus the ability to detect these exosomes in the blood may be an indicator of the presence of a tumor.
In the study published by Oncotarget [1-22-17 FULL article: http://tinyurl.com/hlc68tq ], plasma samples from 34 patients with ovarian tumors and 10 healthy subjects were analyzed for the presence of PS-expressing exosomes in a blinded test. Results demonstrated that those patients with malignant ovarian cancer displayed significantly higher blood PS exosome levels than those with benign tumors (median 0.237 vs. -0.027, p=0.0001) and the malignant and benign groups displayed significantly higher blood PS exosome levels than the healthy subjects (median 0.237 vs -0.158, p < 0.0001 and -0.027 vs -0.158, p=0.0002, respectively).
"These initial proof-of-concept results are encouraging as they appear to support the underlying concept that the measurement of PS-positive exosome levels in blood could be a simple way to detect and monitor cancer. While the work is still early, we think these data serve as an important first step in highlighting the diagnostic potential of this platform," said Steven W. King, President and CEO of Peregrine. "This type of diagnostic technology is particularly important in an area such as ovarian cancer, in which screening options are limited and the ability to detect the disease at an early stage is inadequate. We look forward to continuing to explore the potential of the technology platform in ovarian as well as other types of cancer."
"There is a significant and growing interest in the healthcare industry around the ability to detect cancer and monitor its progression with more readily accessible blood tests. With this area being one of the fastest growing segments of the oncology diagnostics market, we believe that our exosome-based technology represents a significant product development and licensing opportunity," stated Stephen Worsley, VP of Business Development at Peregrine. "Based on the fact that PS is a marker associated with a broad range of cancer types, we believe our platform has potential applications in several solid tumors beyond ovarian cancer. With that in mind, we look forward to aligning with a partner to help explore the potential of this promising technology."
- - - - - - - - - - - - -
1-22-17/OncoTarget Article: PS-Exosomes: PPHM SAB’r Dr. Alan Schroit: Proof-of-Concept Datasupports the “high diagnostic power” of PS-Positive Exosomes in Ovarian Malignancies. ...Basically, Dr. Schroit (cell membrane guru) & his UTSW team used a variant of fully-human Bavi (1N11) to detect the PS on the exosomes…
FROM PG.8: “The data summarized in Fig. 4 [N=44] show that quantification of PS-exosomes in blood distinguishes, with 100% accuracy, healthy tumor-free individuals from patients with ovarian malignancies.”
1-22-17 Oncotarget:
“Detection of Phosphatidylserine-Positive Exosomes as a Diagnostic Marker for Ovarian Malignancies: A Proof of Concept Study”
Lea J 1, Sharma R 2, Yang F 2, Zhu H 1, Ward ES 3,4, Alan J. Schroit (PPHM SAB, UTSW Profile: http://tinyurl.com/yaqnvcvq ) 1,3
1 Harold Simmons Comprehensive CC, UTSW-MC/Dallas
2 Hamon Center for Therapeutic Oncology Res., UTSW-MC/Dallas
3 Dept of Immunology, UTSW-MC/Dallas
4 Dept of Molecular & Cellular Medicine, Texas A&M Univ. Health Science Ctr
https://www.ncbi.nlm.nih.gov/pubmed/28122335 Rec.1-4-17, Acc.1-11-17, Pub.1-22-17
ABSTRACT:
There are no suitable screening modalities for ovarian carcinomas (OC) and repeated imaging and CA-125 levels are often needed to triage equivocal ovarian masses. Definitive diagnosis of malignancy, however, can only be established by histologic confirmation. Thus, the ability to detect OC at early stages is low, and most cases are diagnosed as advanced disease. Since tumor cells expose phosphatidylserine (PS) on their plasma membrane, we predicted that tumors might secrete PS-positive exosomes into the bloodstream that could be a surrogate biomarker for cancer. To address this, we developed a highly stringent ELISA that detects picogram quantities of PS in patient plasma. Blinded plasma from 34 suspect ovarian cancer patients and 10 healthy subjects were analyzed for the presence of PS-expressing vesicles. The nonparametric Wilcoxon rank sum test showed the malignant group had significantly higher PS values than the benign group (median 0.237 vs. -0.027, p=0.0001) and the malignant & benign groups had significantly higher PS values than the healthy group (median 0.237 vs -0.158, p<0.0001 and -0.027 vs -0.158, p=0.0002, respectively). ROC analysis of the predictive accuracy of PS-expressing exosomes/vesicles in predicting malignant against normal, benign against normal & malignant against benign revealed AUCs of 1.0, 0.95 and 0.911, respectively. This study provides proof-of-concept data that supports the high diagnostic power of PS detection in the blood of women with suspect ovarian malignancies.
FULL ARTICLE (13pgs):
HTML: http://www.impactjournals.com/oncotarget/index.php?journal=oncotarget&page=article&op=view&path%5B%5D=14795&path%5B%5D=47251
PDF: http://www.impactjournals.com/oncotarget/index.php?journal=oncotarget&page=article&op=download&path%5B%5D=14795&path%5B%5D=47248
EXCERPTS FROM 1-22-17 ONCOTARGET ARTICLE:
**From DISCUSSION (pg. 8): “The data summarized in Fig. 4 show that quantification of PS-exosomes in blood distinguishes, with 100% accuracy, healthy tumor-free individuals from patients with ovarian malignancies… In summary, this study provides proof-of-concept data that supports the high diagnostic power of PS-expressing tumor exosome detection in blood from women with suspect ovarian malignancies. Ultimately, these studies could lead to earlier stage diagnosis, substantial cost savings, reduced patient exposure to radiation and invasive procedures, and improved clinical outcomes. The assay might also find utility in patients with radiographic abnormalities, even before clinical detection. Indeed, an accurate biomarker predicting the likelihood of malignancy would be extremely beneficial to such a population since they often face long periods of anxiety and uncertainty inherent to a “wait & watch” approach. Finally, if PS-exosome diagnostics are confirmed in a large study to be an accurate and reproducible biomarker of ovarian malignancies, the assay could be applied to the early detection of other visceral malignancies."
**From METHODS (pg.9): “Expression of an engineered tetravalent antibody for PS-detection Monoclonal 1N11 is a human IgG1^ that binds PS through the PS-specific plasma protein B2GP1. A tetravalent variant of 1N11 (1N11-T), with 4 binding sites per molecule was designed to generate a high avidity PS binding agent (Fig.1).”
……...[NOTE: 1N11 is Fully-Human Bavituximab (aka PGN635=AT004), B2GPI-dep. Binding]
**From FINANCIAL SUPPORT (pg.11): “Supported by Cancer Prevention & Res. Inst. of TX (CPRIT) Grant #RP110441, and a Simmons CC Support Grant 5P30 CA142543.”
**EDITORIAL NOTE (pg.11): “This paper has been accepted based in part on peer-review conducted by another journal and the authors’ response & revisions as well as expedited peer-review in Oncotarget."
.
PDF EXCERPTS 1-22-17/OncoTarget/A.Schroit/PPHM-Exosomes (5 pgs of the 13):




NOTE/2-10-17: Exosomes-RNA.com posted the news of the 2-9-17 PPHM Exosomes POC PR...
http://www.exosome-rna.com/proof-of-concept-data-for-a-novel-exosome-based-cancer-detection-platform-published/

10-13-16/ASM REPORT BY ATTENDEE COPPER888: “...I think there is a shift on how the company execs and BOD view the business. As mentioned by multiple posters, SK said that they are still looking to hit a "homerun" with Bavi. But I think that they are now doing that in a framework of risk avoidance, and profitability as their primary goals. With every initiative mentioned, SK would talk about partnering in the next sentence. Exosome testing with a partner; potential of exploring the utility of Beta Bodies - would "advance aggressively with a partner"; If Sunrise data warrants a small study to confirm, they would "partner the next step", etc. I think that for good or bad...the new company directive is the march toward profitability. He also said that the company is worth multiples of its current market cap and that they want to delay the RS as much as they can. "I am focused on getting the Share price over a dollar" He mentioned that there may be many events between now and April that may get us there...” http://tinyurl.com/jx7ouay

9-8-16/CC J.Hutchins PREPARED: “1st, an an update on our PS Exosome Program. As we announced in July, we executed a licensing agreement with UT Southwestern Medical Center for a novel exosome technology designed to detect PS-positive exosomes in a patient blood sample. It is our belief that this technology can provide clinicians with detection & monitoring information regarding the presence and the prevalence of cancer. Preliminary indep. studies have demonstrated that the levels of PS-positive exosomes present in the blood of cancer patients are higher than levels found in the blood of healthy volunteers. Furthermore, study findings also suggest that there is a correlation between the level of PS positive exosomes detected in the blood of cancer patients and the severity and extent of their disease burden. Given our in-house expertise in PS targeting, we believe that we are uniquely qualified to advance this technology. We believe there are significant opportunities to use this technology as both a complementary tool in bavituximab's ongoing development as well as more broadly as the basis for a novel cancer detection & monitoring test kit that will be the focus of partnering efforts. It is our goal to develop, optimize and validate a functional screening assay capable of detecting PS positive exosomes in a blood sample and to initiate partnering discussions for commercialization of the pgm in 2017. We are on track to achieve this goal and we look forward to providing further updates.” http://tinyurl.com/jydtkoy
9-8-16-PR/FY17Q1: “Peregrine in-licensed a novel exosome technology from UTSW that has potential for cancer detection and monitoring applications. This technology aligns directly with the company's expertise, its proprietary PS-targeting platform and the bavituximab dev. program. As such, there are opportunities to use this technology as both a complementary tool in bavituximab's ongoing development, as well as more broadly as the basis for novel cancer detection & monitoring tests that can be the focus of partnering efforts.” http://tinyurl.com/jydtkoy
7-14-16/PR: Peregrine Licenses Exosome-based technology from UTSW (Inventors: Alan Schroit/Philip Thorpe) http://tinyurl.com/zszd4fj
...“relates to assays that are able to detect small amts of PS+ Exosomes in a patient's blood sample as a way to detect cancer at a very early stage of development.”
VP JEFF HUTCHINS: “We are excited to enter into this licensing agreement with our long-term collaborators at UTSW. This technology offers a promising product development opportunity and aligns directly with the company's expertise with our proprietary PS-targeting platform and our longstanding CDMO capabilities around the development, qualification, and validation of in vitro analytical assays. As such, there are significant opportunities to use this technology as both a complementary tool in bavituximab's ongoing development, as well as more broadly as the basis for novel cancer detection & monitoring tests that can be the focus of partnering efforts. It is important to note that this dev. program will require minimal capital investment and has the potential to create significant value over the next 18mos., including potential partnering opportunities. As a result, we feel that today's licensing deal provides yet another important driver in our ongoing efforts to achieve profitability."
7-14-16/CC S.King PREPARED: “...In addition to bavituximab program which continues to have tremendous potential value, the company announced earlier today that it has licensed-in a Novel PS Exosome Technology, with the potential to detect & monitor cancer at an early stage through a simple blood test [ 7-14-16: http://tinyurl.com/zszd4fj ]. I recognize that some of you might think this theme is contrary to controlling spending as we move toward profitability, but in fact we believe that given our already existing knowledge base in Targeting PS and our already available infrastructure for developing & validating tests, that so a very modest capital investment we can quickly reach proof-of-concept with a goal of partnering this technology, which could then bring in addl. revenue, with another potential upside of the technology being that it can possibly be useful in the continued dev. of bavituximab. Jeff will talk more about this during his prepared remarks. Given the strategy of R&D targeted toward early partnering, will allow the company to continue its R&D activities with significant upside coming from partnering as we move to our profitability.” http://tinyurl.com/h8eqtg5
7-14-16/CC J.Hutchins PREPARED: “I'm very happy to be able to discuss a number of exciting developments in our preclinical group. First, as Steve discussed, we have executed the licensing agreement with UT Southwestern Medical Center, for novel exosome technology [ 7-14-16: http://tinyurl.com/zszd4fj ]. While many of you are familiar with exosomes, I’ll provide a brief overview for those who are not. Exosomes are cell-secreted vesicles, or mini-cells if you will, that are present in nearly all bodily fluids, including blood. Likewise, tumor-derived exosomes represent small pieces of tumor cells that are released into the blood as tumors grow. As well, these tumor-derived exosomes have Phosphatidylserine or PS on their surface as a marker and can also contain DNA, RNA and proteins as markers of malignant disease. It is believed that even small tumors begin to release PS-positive exosomes, and thus the ability to detect these exosomes in the blood may be an indicator of presence or progression of a tumor. The licensed technology is designed to detect & monitor PS-positive exosomes in a patient's blood sample, providing clinicians with detection & monitoring information regarding the presence & prevalence of cancer. These exosomes have PS flipped to the outside of the surface and demonstrate immunosuppressive activity, just as we find with tumor cells. Preliminary studies have demonstrated that the levels of PS-positive exosomes present in the blood of cancer patients are higher than levels found in the blood of healthy volunteers. Furthermore, study findings also suggest that there is a correlation between the level of PS-positive exosomes that are detected in the blood of cancer patients and the severity or extent of their disease burden. Given our in-house expertise in PS-targeting, we believe that we are uniquely qualified to advance this technology. As Steve stated, there are significant opportunities to use this technology as both a complimentary tool in bavituximab's ongoing development, which Joe will address later, as well as more broadly as the basis for a novel cancer detection & monitoring test kit that will be the focus of our partnering efforts. It is our goal to develop, optimize, and validate a functional detection & monitoring assay capable of detecting PS-positive exosomes from a simple blood sample, and, given the company's extensive experience in developing assays of this type, we do not anticipate the need for added personnel or any specialized equipment for this project. Once we have successfully validated this assay, we plan to establish proof-of-concept through an efficient preclinical & clinical testing program. We have no intention of conducting further development work beyond the proof-of-concept stage. Rather, we expect to initiate partnering discussions for commercialization of this program in 2017. We're very excited to begin this work on this new program and we'll have more details to offer in the coming months.”
...SUMMARY: “And lastly, we've in-licensed a new exosome technology for a minimal cost that leverages our existing in-house expertise and provides us with another opportunity for us to create value to product development. Together, we believe the strategy will provide success, as it will allow us to focus the majority of our resources on achieving our primary corporate goal, future sustainable profitability within 24mos. At same time, we will focus our R&D efforts on small early stage trials and development of the exosome technology in an effort to attract partners. We believe this strategy will allow us to build near-term revenues through Avid, while maintaining the potential for significant addl. value creation associated with our R&D efforts.” http://tinyurl.com/h8eqtg5
7-14-16/CC J.Shan PREPARED: “I'd first like to comment on our new exosome program. One of the most exciting aspects of this technology is the potential synergy that it offers with our bavituximab clinical dev. program. Through our ongoing work with bavituximab, we have gained significant understanding of PS-mediated immuno-suppression in cancer. The availability of the PS specific biomarker, which can be implemented in our planned future bavituximab clinical trials, aligns nicely with our refocused bavituximab dev. strategy aimed at generating the most meaningful data possible from small, early stage clinical trials to support partnering efforts. We are very anxious to bring this new technology to Peregrine and we look forward to the value it brings to our bavituximab program.” http://tinyurl.com/h8eqtg5
7-14-16/CC Q&A/Pantginis – SK: “(On the Exosome pgm), …it fits right in; we’re not having to hire addl. people, we’re not having to bring in addl. equipment. This really fits in with everything we are doing on both studying PS, as well as on the assay development side of the business. We think it’s actually going to be complementary. There are other technologies out there looking at exosomes; they're are all taking a very different approach to what we’re doing and we actually think they could be very complementary to each other. We also see a need, even as interest in exosomes begins to pick up, to actually utilize this in conjunction with other things that are in development.” http://tinyurl.com/h8eqtg5
7-14-16/CC Q&A/T.Yip – SK: “(On the Exosome pgm), the next step will be to validate that through patient samples. The beauty of this is that while you do need IRB approval of course, you’re not running really clinical trials, so this can be done in conjunction with either our ongoing trials or partners' trials, or there are many other sources of just receiving these types of blood samples. This gives us the ability to very quickly go through and test hundreds or thousands of patient samples as part of the validation process. At that point, we can zero in on what are the potential applications of the technology, outside of what we might do with our own PS Targeting programs – what would be the potential utility of this for patients. Our goal is not to become a diagnostics company, but to put this in the hands of a good organization that's already established in the diagnostics area and then have them finish up the commercialization and expansion of the utility of the actual assay itself. Our benefit at that point would become, hopefully, some residual royalties, milestones and what have you, which feeds back into our revenue goals of becoming profitable. So, we thought that this is a very attractive technology that just fits right in with what we’re doing and requires almost no addl. resources whatsoever…
Q2:/TY: ”when should we expect to see more preclin. data on this front?”… SK: “Our goal is, probably towards for the end of this year, to be in a position to have data that we can present. That will come in a lot of different formats, so it will be in conjunction with other ongoing studies that maybe taking place already to be standalone just on the diagnostic itself. So, you'll be hearing a lot about this, and one of the reasons when to get this news out there is because what you think sooner than later we will build a talk about this technology.” http://tinyurl.com/h8eqtg5
6-30-16: UTSW WEBSITE - “AVAILABLE TECHNOLOGIES” (#2932):

“A Blood-based Diagnostic Test for the Early Detection of Cancer” (Alan Schroit, S.Ward, A.Gazdar)
ALL: http://www.utsouthwestern.edu/about-us/administrative-offices/technology-development/industry/available-technologies.html
SCHROIT/EXOSOMES #2932: http://www.utsouthwestern.edu/edumedia/edufiles/about_us/admin_offices/technology_development/available_technologies/utsd-2932.pdf
This invention is a highly sensitive, blood-based assay system for the diagnosis of cancer. Tumors secret large quantities of cell membrane-derived exosomes into the blood stream of cancer patients. While all exosomes, whether from tumor cells or normal cells, share membrane epitopes similar to those found on the parent cell membrane, only tumor cell-derived exosomes express the membrane phospholipid, phosphatidylserine (PS), in the outer leaflet of the particle membrane. Based on data clearly showing that PS exposure is a unique signature of tumor-derived exosomes, the investigators developed a discovery platform for the screening & detection of tumor-derived exosomal PS signatures in patient blood. This novel tool provides:
1) A diagnostic marker that could dramatically improve the early detection of cancer.
2) Predicts whether a tumor is benign or malignant.
3) Could provide a predictive marker for response to therapy. Importantly, early detection of indolent neoplastic disease could significantly improve patient survival.
Currently, there are very few blood tests for cancer. These include alpha-fetoprotein for hepatocellular cancer, CA125 for ovarian cancer, and prostate specific antigen for prostate cancer. Although, high levels of these proteins might indicate the presence of neoplastic disease, none are diagnostic. Because our system detects a shared universal marker of neoplastic disease, it provides a viable method for detecting cancer of any type, in any organ.
6-9-16/WORLD-PATENT: Pub# WO/2016/201064, Filed 6-9-16, pub. 12-15-16 (A.Schroit, S.Ward, A.Gazdar)
Title: “Diagnostic Test For Early Stage Cancer”
URL (patentscope.wipo.int): http://tinyurl.com/gqvwkga
ABSTRACT:
“Disclosed are methods of identifying tumor-derived exosomes as an early cancer diagnostic, as well as for staging, assessing progression, and assessing therapy of cancer.”
Inventors: Alan J. Schroit [UTSW, PPHM SAB]; Adi Gazdar [UTSW]; E. Sally Ward [Texas A&M]
Applicant: Board of Regents, The Univ. of Texas System, Peregrine Pharmaceuticals, Inc.
EXCERPT from SUMMARY:
“Detecting may comprise contacting the sample of step (b) with an exosome binding agent that may be the same or other phosphatidylserine binding agent than the one used in step (b), or a non-phosphatidylserine binding agent. Given that only PS-positive tumor exosomes are captured and available at this stage (after step b), the detecting agent may be an agent that binds to all exosomes regardless of their source… In general, normal tumor-free individuals have undetectable levels of PS-exosomes in an unconcentrated sample. In contrast, patients with tumors will typically exhibit values above 100 pg/50 \xh of sample in an unconcentrated sample. Thus, values greater than 50 pg/50 ui, plasma (or serum) are indicative of a malignancy. The method may further comprise obtaining said sample from the subject prior to step (a). The method may further comprise diagnosing the subject from which the sample was obtained as having cancer. The method may further comprise staging the cancer or classifying the cancer type in the subject from which the sample was obtained. The method may further comprise performing steps (a)-(c) a 2nd time, and comparing the results from the 1st & 2nd times, thereby assessing cancer progression or regression. The method may further comprise performing steps (a)-(c) a 2nd time, wherein the subject has received a cancer therapy after the 1st performance of steps (a)-(c) and before the 2nd performance of steps (a)-(c), and comparing the results from the 1st & 2nd times, thereby assessing the efficacy of cancer therapy. The method may further comprise treating the subject with a cancer therapy… [0096] In addition to PS, exosomes exhibit a number of unique & non-unique markers. Some of the exosome-specific markers include biomarkers that can help distinguish the nature of the type of cancer from which the exosome originated. Some exemplary exosome biomarkers can be found above in Table 1. Global exosome biomarkers that can be detected with appropriately labeled agents include cd9, cd63, cd81, ALIX, HSP70, TSG101, duramycin, and heparin.”

- - - - - - - - -
Here is the corresponding U.S. PATENT APPLIC: Pub# 20170146542, Filed 6-9-16, pub. 5-25-17 (A.Schroit, S.Ward, A.Gazdar)
USPTO.gov: http://tinyurl.com/y7clom9w “DIAGNOSTIC TEST FOR EARLY STAGE CANCER”
- - - - - - - - - -
NOTE: 2-21-17: Exosomes-RNA.com posts the Exosomes PPHM/Schroit World-Patent App today.
http://www.exosome-rna.com/new-patent-application-published-diagnostic-test-for-early-stage-cancer/

2-27-15/U.S.PATENT: App#20150241431, filed 2-27-15 (Schroit/Thorpe)
Title: “Methods & Compositions for Isolating Exosomes”
http://tinyurl.com/Schroit-Thorpe20150241431
ABSTRACT:
“Disclosed are surprising new methods & compositions for isolating extracellular microvesicles such as exosomes, particularly disease-related and phosphatidylserine (PS)-positive extracellular microvesicles as exemplified by tumor- and viral-derived exosomes. The methods of the invention are rapid, efficient, cost-effective and, importantly, are suitable for use with large volumes of biological fluids and produce antigenically intact extracellular microvesicles & exosomes. The methods & compositions are based on the surprising use of acetate buffers to isolate large quantities of extracellular microvesicles, particularly tumor-derived exosomes, from solution, without damaging their morphological or functional properties or antigenicity.”
Inventors: Alan J. Schroit; Philip E. Thorpe; Shelley Fussey
Applicant: Board of Regents, The Univ. of Texas System, Peregrine Pharmaceuticals, Inc.
May2014/Article: J Immunol Methods, Thorpe/Schroit/etal, “A Novel ‘Salting-Out’ Procedure for the Isolation of Tumor-Derived Exosomes”
https://www.ncbi.nlm.nih.gov/pubmed/24735771
ABSTRACT:
“The last decade has seen an exponential growth in the number of exosome-related publications. Although many of these studies have used exosomes from biological fluids (blood, and ascites or pleural effusions) the vast majority employed vesicles isolated from large volumes of tissue culture supernatants. While several techniques are available for their isolation, all require a significant reduction in volume to obtain sufficient concentrations for study. One approach is to concentrate the medium before proceeding with their isolation, however, these procedures are very time consuming and require specialized laboratory equipment. Here we provide a new & effective method for the isolation of tumor-derived exosomes based on "charge neutralization" with acetate. We show that titration of tissue culture supernatants with 0.1M acetate to pH4.75 results in immediate precipitation of virtually all the exosomes. The precipitated exosomes can be washed to remove residual media and are readily "resolubilized" upon resuspension in acetate-free buffer at neutral pH. This simple cost effective method significantly increases the yield of exosomes from an unlimited quantity of culture supernatants. Exosomes isolated by this technique are indistinguishable from exosomes recovered by direct ultracentrifugation.”
7-14-17: Avid Scientists have developed “Antibody Discovery Platform” - applicable to Avid’s customer offerings and Peregrine’s drug dev. R&D side. Looks like good potential – SK calls it a “platform”, and it hit the 10-K. Most interesting (to me).
7-14-17/CEO Steve King: “Another part of our effort to attract new customers is to expand our service offerings. Our scientists have developed a state-of-the-art Antibody Discovery & Characterization Platform through which we can generate antibodies against virtually any target. These capabilities are meant to allow rapid screening for high affinity antibodies that are developable. These capabilities are a natural extension of the services we already offer through Avid and it represents an attractive way to bringing customers at a much earlier stage of development with the potential to move them quickly into process developments and cGMP manufacturing.”...
“The Antibody Discovery Platform discussed earlier [Avid] could also be instrumental on the R&D side of the business, allowing us to rapidly identify antibodies against already validated targets, as well as the potential to identify novel targets. As we think about diversifying our development pipeline to include lower risk, already validated targets, this new capability could help us to identify developable antibodies that could have short-term value, as the need for such antibodies is on the rise. And this approach fits very well into our core capabilities and experience in the development of monoclonal antibodies. You can expect to hear more about this new capability over the coming months on both sides of our business.” http://tinyurl.com/yb4wulvu
4-30-17 10-K pub. 7-14-17 pg.5:
Antibody Discovery Technology
“Recently, our scientists have developed an Antibody Discovery & Characterization Platform through which we can generate antibodies against virtually any target. These capabilities are state-of-the-art and meant for rapid screening for high affinity antibodies as drug candidates. These capabilities are a natural extension of the services we already offer through Avid and it may represent a way to bring in customers at a much earlier stage of development with the potential to move them quickly into process development and cGMP manufacturing. Continuing to diversify our customer base is one of our key initiatives at Avid and we believe the antibody discovery capabilities will help attract new customers. The antibody discovery capabilities can also be used in our R&D business to rapidly identify antibodies against known targets as well as to identify novel targets. The antibody discovery platform is now fully functional and we are in the process of evaluating the technology through identifying several known targets and thus far, the results have looked promising. Once this process is completed, the technology platform will be marketed to potential customers as part of our CDMO service offerings.” http://tinyurl.com/ycxu4l5n
7-14-17 Qtly CC-Transcript, PR(Financials Q4FY17/fye4-30-17), Avid Revs History Table
=> Total Revs May06-Apr17: $231.2mm/Avid + $24.1mm/Govt + $2.4mm/Lic. = $257.8mm.
Cash at 4-30-17: $46.8mm (Op. Cash Burn for q/e 4-30-17 was $3.9mm – see below).
As of Jul. 10, 2017, there were 45,069,188 shares outstanding. (10K 4-30-17 iss. 7-14-17 http://tinyurl.com/ycxu4l5n )
...NOTE: PPHM shares were 1:7 Reverse Split eff. 7-10-17 (315mm/$.606=>45mm/$4.24) http://tinyurl.com/ycohqn6j
This large post has 4 sections:
I. 7-14-17 Q4/FY17 Qtly. Earnings Conf. Call TRANSCRIPT (fy/e 4-30-17)
II. 7-14-17 PPHM Press Release: Q4/FY17 Earnings & Developments
III. A link to “O/S Shares & ATM Sales History – 2006-curr.” ( http://tinyurl.com/yagmu2on )
IV. Updated Table of Avid Revenues By Quarter (May’06-Current)
…Recall: Peregrine’s FY runs May-Apr, so FY’17 = May’16-Apr’17.
((( Orig. transcript from SeekingAlpha.com [http://tinyurl.com/y7ae7cqj ], with numerous corrections made. )))
Link to webcast replay: http://ir.peregrineinc.com/events.cfm => http://edge.media-server.com/m/p/yx85d7e7
FULL TRANSCRIPT… 7-14-17 FY’17/Q4 Earnings Conf. Call (fy/e 4-30-17) (King/Shan/Lytle)
WELCOME & FWD-LOOKING STATEMENTS: Tim Brons, Vida Strategic Partners (IR) http://www.peregrineinc.com
CEO STEVE KING – OPENING COMMENTS:
Thanks to all of you who have dialed in and all of you who are participating via webcast today. As most of you’re aware, Peregrine has a diverse business with a common goal of improving the lives of patients with serious diseases. We do this by manufacturing high-quality pharmaceutical products through our contract manufacturing business, Avid Bioservices, and by advancing cancer therapies & diagnostics, led by bavituximab, our novel immunotherapy product, through our R&D company, Peregrine. We continue to believe there are significant opportunities for both of these businesses, but also recognize they are very distinct businesses with very different roads to success.
I will first discuss recent highlights with the 2 companies and then more about potential plans to ensure the success of both companies. We’re very pleased to announce that Avid Bioservices recorded its highest annual revenues to-date during FY17, topping $57mm, representing 30% growth over the prior FY. Revenues could have been even higher if not for the need to delay shipment of several already released lots of materials due to the needs of the customer. While we’re pleased at the continued yr-over-yr revenue growth, we are projecting relatively flat revenues this FY due to recent changes in a large customer’s [Halozyme] forecasts and a delayed regulatory filing for another significant customer. We expect both of these factors to be temporary and remain confident that Avid will continue to achieve growth in the future, as these customers are successful and as we add new customers to diversify our client base. And on that front, we have recently signed 4 new customers that we expect to contribute significantly to top line revenues in the future.
In addition, we successfully completed 3 process validation campaigns for existing third-party customers, which again, we believe can have a significant revenue impact as the customers make regulatory filings and begin to move toward inventory builds for potential launch, followed of course by the potential for commercial supply. In addition to securing new customers and setting the stage for commercial production, we are continuing to make other important steps to ensure Avid’s growth in the coming years. As part of this effort, we have recently installed 2 new 2,000L bioreactors in our Myford facility, and we have already secured commitments for this capacity. Due to its state-of-the-art modular design, there is the potential to install addl. bioreactors in our Myford facility, which will allow us to continue to meet the increasing demand and grow the future business with our current facilities. Another part of our effort to attract new customers is to expand our service offerings. Our scientists have developed a state-of-the-art antibody discovery & characterization platform through which we can generate antibodies against virtually any target. These capabilities are meant to allow rapid screening for high affinity antibodies that are developable. These capabilities are a natural extension of the services we already offer through Avid and it represents an attractive way to bringing customers at a much earlier stage of development with the potential to move them quickly into process developments and cGMP manufacturing.
Switching gears to our R&D activities. While meeting our goal of reducing overall R&D spending by over 50% during FY17, we were also able to generate some of the most compelling data to-date supporting the combination of bavituximab & checkpoint inhibitors. We are working with some of the best institutions in the world that have been involved in the development of cancer immunotherapies. Perhaps the most important recent preclinical findings came through original research from our collaborators at Memorial Sloan Kettering Cancer Center (MSKCC). The researchers at MSKCC [Jedd Wolchok’s Lab] presented promising preclinical data [4-3-17/AACR17: http://tinyurl.com/lxlltd6 ] combining PS-targeting antibodies with adoptive T cell transfer therapy [ACT: CAR-T], which may support the combination of bavituximab with CAR-T cell immunotherapy in the future.
On the clinical front, we presented data from an analysis of the maturing SUNRISE Phase III trial [4-3-17/AACR17: #CT159/25, “IFN-y Analysis In Blood & Tissue as a Potential Prognostic and/or Predictive Biomarker” (Sunrise Biomarker #3) http://tinyurl.com/ktzr782 ], showing that patients in the arm that received bavituximab and subsequent immune checkpoint inhibitors lived significantly longer than those on the cohorts that received placebo and subsequently went on to immune checkpoint inhibitors. The data was impressive. It has been very well received by KOLs, and we believe strongly supports advancing bavituximab in combination with immune checkpoint inhibitors, such as PD-1 & PD-L1 inhibitors. Joe will talk more about this data shortly.
The data presented at AACR by the researchers at MSKCC and Peregrine collectively provide growing validation for the potential bavituximab in combination with immune stimulating therapies. Such as those we are exploring our collaboration with the National Comprehensive Cancer Network (NCCN), which will evaluate novel bavituximab combinations in Glioblastoma, Head & Neck cancer, and Hepatocellular Carcinoma, including a PD-1 combination. This collaboration is advancing well and again Joe will provide more details on the 3 trials during his prepared remarks.
The Antibody Discovery Platform discussed earlier [Avid] could also be instrumental on the R&D side of the business, allowing us to rapidly identify antibodies against already validated targets, as well as the potential to identify novel targets. As we think about diversifying our development pipeline to include lower risk, already validated targets, this new capability could help us to identify developable antibodies that could have short-term value, as the need for such antibodies is on the rise. And this approach fits very well into our core capabilities and experience in the development of monoclonal antibodies. You can expect to hear more about this new capability over the coming months on both sides of our business.
Lastly, on the R&D side, we are pleased to report that the company continues to make progress with its PS-targeting Exosome Diagnostic Technology that is designed to detect & monitor cancer. Our scientists have successfully optimized the assay and we are currently preparing to generate addl. Data, testing the diagnostic with an expanded set of human samples. Such data will be important to partnering discussions and we’ll keep you posted on progress.
[**6-22-17: Nature/BJC article, Dr. Alan Schroit/UTSW etal: "Detection of PS+ Exosomes for the Diagnosis of Early-Stage Breast & Pancreatic" http://tinyurl.com/ycrx9672
**2-9-17/PR: PS+ Exosomes Proof-of-Concept Data (N=44, Ovarian, Dr. Alan Schroit/UTSW etal) pub. 1-22-17 in OncoTarget http://tinyurl.com/jhv57ua ]
Both Peregrine & Avid have achieved important milestones in recent quarters. We are proud of both businesses and believe each has strong potential. However, we also recognize that they have very different business models and pathways to growth. As a result, we understand that some of our shareholders have divergent goals for the company’s future, depending on if they’re more focused on Avid or our R&D business [7-13-17: Ronin/SW-Invest Letter to Stockholders; PPHM Comments http://tinyurl.com/ybr8ycbp ]. So, as part of our continuous evaluation of various strategic options that we believe they enhance value for all of our stockholders, we are seriously considering the possibility of separating our 2 distinct businesses. We’re still at the early stages of evaluating this strategic opportunity, but are diligently moving forward with the analysis. I’ll now turn the call over to the other members of our team, who will give a detailed overview of our clinical, corporate, and CDMO activities.
JOE SHAN (VP/Clin.&Reg. Affairs) – CLINICAL TRIALS:
As Steve mentioned just a few moments ago, despite a significant reduction in R&D spending over the past FY, we were able to continue patient treatment & follow-up in the Phase III SUNRISE trial thus allowing the clinical data to further mature.
At the AACR annual meeting in April, we presented the most important and compelling data to-date supporting bavituximab’s potential to improve the outcome of anti-PD-1 or PD-L1 immune checkpoint inhibitors [4-3-17/AACR17: #CT159/25, “IFN-y Analysis In Blood & Tissue as a Potential Prognostic and/or Predictive Biomarker” (Sunrise Biomarker #3) http://tinyurl.com/ktzr782 ]. Among a subgroup of 91 patients who are treated with checkpoint inhibitors subsequent to the assigned SUNRISE treatment, MOS was not yet reached for the 45 patients who received docetaxel+bavituximab compared to a MOS of 13mos. for the 46 pts. who received docetaxel+placebo, with a hazard ratio of 0.43 and p=0.005. The statistically significant difference in survival provides strong clinical rationale for combining bavituximab with checkpoint inhibitors. We are encouraged by the positive response received from investigators & thought leaders regarding this subgroup analysis and exploring ways to advance the program as resources permit. In the meantime, as we focus the biomarker analysis on this subgroup of patients, we are seeing some intriguing results and plan to present our findings at key conferences later in the year.
At ASCO in June, we presented addl. supportive data [6-3-17/ASCO'17: "Prelim. Correlative Analysis of PD-L1 Expression from the Sunrise Study” http://tinyurl.com/y93upatl ] demonstrating that patients in the bavituximab containing arm who had low PD-L1 baseline expression on tumor cells, who typically experience poor response to PD-1 or PD-L1 checkpoint inhibitors, led significantly longer than patients with high baseline PD-L1 expression. These data further support the hypothesis that bavituximab may modulate the tumor microenvironment to complement enhance the anti-tumor activity of checkpoint inhibitors.
So the SUNRISE trial database was locked at the end of March of this year. Patients who were still receiving bavituximab at that time were transitioned to compassionate use program and some continue to remain on treatment. We believe bavituximab is active, as some patients have been on treatment for as long as up to 2.5 years and has a good safety profile, making it a useful candidate for combining with other therapies and we remain deeply committed to advancing the growth.
Now, let me provide a brief update in our collaborations, starting with the NCCN trials. At present, 2 of the 3 trials are now opened for enrollment. The 3rd trial at The Sidney Kimmel Comprehensive CC at Johns Hopkins is a Phase II Study of Pembrolizumab & Bavituximab for Recurrent or Metastatic Head & Neck cancer that has progressed on anti-PD-1 therapy, and this trial is expected to be initiated by the end of the year.
----------NCCN:
[NCCN Bavituximab Trials Announced 9-6-16 http://tinyurl.com/gutgwb5
...#1: Ph1/HepC-Related Hepatocellular(Liver) (Bavi+RAD+Bayer’s Nexavar=Sorafenib), MOFFITT CC (N=18)
. . . . . . .PI: Jessica Frakes, MD https://clinicaltrials.gov/ct2/show/NCT02989870 <=Recruiting a/o 3-27-17
...#2: Ph2/Newly Diag. Glioblastoma (Bavi+RAD+Merck’s Temodar), MASS-GEN. & DANA FARBER (N=36)
. . . . . . .PI: Elizabeth Gerstner, MD https://clinicaltrials.gov/ct2/show/NCT0313991 <=Recruiting a/o 6-16-17
...#3: Ph2/Progressive Squamous Head+Neck (Bavi+Merck’s Keytruda), JOHNS-HOPKINS(Sidney Kimmel CC)
. . . . . . .PI: Ranee Mehra, MD https://clinicaltrials.gov/ct2/show/XXXXXXXXX - See Dr. R.Mehra Jan'17/IFN-y Biomarker: http://tinyurl.com/h8gzkww
Last but certainly not least, our collaborators at MSKCC [Jedd Wolchok Lab] presented a preclinical study at the AACR Annual Meeting which evaluated and compared the anti-tumor activity and side effects of adoptive T cell transfer therapy [“ACT”] in combination with either PS-targeting or anti-OX40 antibodies in mice with advanced melanomas. While PS-targeting and anti-OX40 demonstrated comparable tumor regression when administered in combination with adoptive T cells, only the PS-targeting combination achieved these results without any off-target toxicities. In contrast, the anti-OX40 treatment combination elicited off-target inflammatory side effects. Toxicities have limited the clinical potential of adoptive T cell transfer and CAR-T approaches to-date. The absence of toxicity with PS-targeting in adoptive T cell transfer in this initial mouse melanoma experiment may have important future implications for combination strategies and solid tumors.
-----------
[4-3-17/AACR’17 #1651, Joint PPHM & Memorial Sloan Kettering, “Targeting Phosphatidylserine in Combination with Adoptive T Cell Transfer Eliminates Advanced Tumors without Off-Target Toxicities in a Melanoma Preclinical Model”, Daniel Hirschhorn-Cymerman, Jedd Wolchok, Taha Merghoub, etal http://tinyurl.com/lxlltd6 ]
I’ll wrap up by saying how excited we are about these recent clinical & preclinical findings, which really set the stage for combining bavituximab with other immunotherapies and we look forward to providing updates on our progress over the coming months.
PAUL LYTLE (CFO): [4-30-17 10K iss. 7-14-17: http://tinyurl.com/ycxu4l5n ]
Let me switch gears now to discuss our financial results for the qtr and full FY17. Our overall guidance this past FY focused on increasing revenue from our contract mfg. business, Avid, while also reducing our spending on R&D, as we seek to position the company closer to overall profitability. During FY17, we achieved both a substantial increase in revenue and a significant reduction in R&D expenses. Let me first talk about top line revenues. Contract mfg. revenue for Q4/FY17 was $17.9mm and $57.6mm for the full FY, representing 30% yr-over-yr growth. And while we missed the low-end of our revenue guidance of $60mm, we were ready to ship approx. $10mm in mfg. revenue at the end of the FY that was unfortunately delayed due to events outside Avid’s control. This revenue will now be shifted from FY17 into FY18.
Regarding our revenue guidance, as Steve mentioned earlier, we currently expect revenue in FY18 to remain consistent with FY17. As temporary production roles are a common part of the CDMO industry, it emphasizes the need to continually expand & diversify our customer base. During FY16 and prior to the Myford facility being built, we had minimal available production slots in our Franklin facility to offer to new customers, and therefore, we had historically limited abilities to diversify our customer base. And as we built and commissioned the state-of-the-art Myford facility in March of 2016, diversifying our customer base was a key focus. We immediately secured one new customer that has completed process validation at Myford and we have recently signed project work for 4 new customers, including one project that is in late-stage development. Based on our current book of business, we are providing FY18 revenue guidance of $50-55mm, which is consistent with the revenue projections we made at this time last year. This projection is based on current information and is supported by current revenue backlog of approx. $58mm under signed contracts, most of which we expect to recognize during FY18.
I’ll now address our efforts to reduce overall spending on R&D. As we announced last June, our internal drug development strategy has shifted away from large late-stage clinical trials to small, cost effective, early-stage clinical trials designed to attract potential partners to further advance our products. Our goal in adopting the strategy was to further reduce R&D spending by 50% in FY17 vs. FY16. We’re pleased to report that our R&D expenses declined 52% over the same prior year period exceeding our targeted reduction. Although we have generated some of the most promising bavituximab data to-date as Steve and Joe just covered, we’re still expecting to further reduce R&D spending by 40% or more in FY18, unless we can secure addl. funding to further advance this program in connection with exploring the various strategic options, including a possible separation of our 2 distinct businesses. In summary, the increase in mfg. Revenue, combined with a decrease in R&D spending, has translated into a reduction in our net loss by 56% for the qtr and 49% for the full FY17 vs. the same prior year period.
Switching gears now, I’d like to provide a quick update on the recently effected reverse stock split [1:7 eff. 7-10-17]. As I outlined during the Q3 earnings call, Peregrine was committed to taking the necessary steps to preserve its NASDAQ listing. While we executed this R/S as a last resort, we firmly believe that doing so was in the long-term interest of our stockholders. To maintain our continued listing on NASDAQ Capital Market, our min. closing bid price has to close at or above a $1 for 10 consecutive trading days. As of the close today, we are now 5 days into that process with a min. closing bid of $1 or higher. This concludes my financial overview.
Q&A: [beg. 20:30]
1. Kumara Raja - Noble Life Science Partners http://noblelsp.com/research
KR: ”Given that revenues are going to be flat comparatively yr-over-yr, how should we think about expenses, ways of reducing R&D expenses? And also costs were higher. When do you expect margins to start getting better?”
Steve King: As revenues fluctuate, we have to watch the costs associated with both the biomanufacturing business as well as our R&D expenditures. If you look at this year’s R&D budget, it is even significantly down from last year. So that that’s going to be further cost savings on the R&D side. Of course, that’s kind of counterintuitive, given the fact we have some really exciting data coming out. Thats one of the reasons we’re looking at the strategic alternatives is really feel like we should be putting more money into R&D, not taking more money out of it, but we also have to deal with financial realities of today, and so we’ll make those adjustments as we go along. But, our goal is to find a strategic alternative that will allow us to move the R&D sort of forward independently of the Avid business. On the Avid side as far as cost of goods go, part of that was bringing on board the Myford facility. So last year, even though we did obviously some nice business in the facility and it’s been busy since day one, it’s still not running at full capacity when you do the process validations, they’re a little bit more cumbersome, a little bit more complex, so there’s more costs associated with those runs. As we get into more full utilization of the facility in kind of campaign mode, where you’re running multiple runs of the same product simultaneously, we’ll see those cost of goods come down, because they simply get spread out over more runs. We’re constantly looking at the business adjusting the expenses in order to continue to have a profitable biomanufacturing business and also to watch your overall corporate finances.
KR: And in terms of making a strategic decision in terms of separating the mfg. vs. the clinical trials business, what are you all thinking in terms of the timeline for making such a decision? And what kind of challenges and opportunities do you see there? Also, on on the clinical data presented at ASCO, obviously, you guys are seeing that patients who had low PD-1 expression, they are surviving longer. For these patients, what treatments are there on after moving from bavituximab? Are there on any other treatments, or are they just continuing on bavituximab?”
Joe Shan: The ASCO presentation you referred to was analysis done on a subset of patients selected by whether or not we had tumor tissue to evaluate the PD-L1, and that ended up being relatively small percentage of the total number of patients enrolled in the SUNRISE trial. We’ve not dissected that small number of patients further by what treatment they went on after SUNRISE trial. So…
Steve King: Most of the patients that did receive the subsequent immunotherapy, it was all primarily PD-1 inhibitors, probably all primarily Opdivo, given the timing as far as that subset, but the rest of the data, as Joe said is – can certainly [ph] as small numbers. On the opportunities for the businesses as far as the kind of the process, I think, it is a process. Obviously, there’s a lot of considerations that you can imagine that need to be taken into account, as we’re looking at different structures that might work and thinking about all those avenues. There’s things such as taxes, where we have to think about. There’s the overall net operating losses of the company that we want to make sure we get full utilization of. So there’s just a lot of complexities. So we don’t currently have a particular timeline we’re marching to. I would say, we will diligently move forward the thinking process and at the appropriate time, we’ll update as we get closer and closer to be in a position to have a clear direction.
Paul Lytle: And Kumar, looking at a number of potential opportunities, so which type of opportunity that we move forward with will basically dictate the timeline.
2. Andrew De Silva (B. Riley & Co.): http://brileyco.com/research
AS: ”On Avid’s committed contracts thus far to-date, do you have a sense as when some of these higher revenue periods should hit? Will they smooth out or be lumpy like it was last year in your opinion? And, what needs to take place for you to get closer to the 40-50% GM% range?”
Paul Lytle: Overall, the CDMO business generally has lumpy revenue streams, because you have a mix of customers, sometimes they’re in an earlier stage and they do a few mfg. runs and then they go away for a while and they come back for the next set of runs. And then you have commercial supply, where it can go up and down depending on the product and the launch and the timelines of your commercial customers. With respect to our particular lumpiness, we’ve had issues with our third-party vendors, where we have delays in testing over the last FY year that caused some lumpiness in our revenue streams. So our goal is to expand & diversify our customer base. The more customers we have in the mix, the less lumpy the revenue streams would be. We also want to make sure we secure good commercial customers, like the ones we have today, that that do provide kind of a consistent consistent need for manufacturing. The more diversified you can get and more commercial you can get, the less lumpiness you’ll see in future revenues and that’s our kind of our goal moving forward. I think we’re going to see some lumpiness in terms of FY18, but as our customers submit their BLAs and go through the commercial launch & inventory builds that Steve mentioned earlier, I think you’ll see less lumpiness going forward in the FY19 time period.
AS: Is it still too early for you guys to get a sense of what the quarterly impact might be this year from a seasonality standpoint?”
Paul Lytle: Yes, it’s a quite complex in terms of timing and lot release. As we mentioned in the script, we had $10mm in revenue that was expected to ship by the end of April, and due to unforeseen events that were outside our control, that now has shifted into FY18, so that’s a large lumpy number.
AS: ”re: R&D, you mentioned that that should continue to decline yr-over-yr. I’m just kind of curious how does that work now that you’re expected to start a Phase II trial [NCCN/Head+Neck] in the back-half of this FY?”
Steve King: At this point we haven’t committed to starting new studies. The only new studies that we’ve committed to are the NCCN studies, which were all funded under a $2mm grant. Those are the only ones we are really committed to, and in order to start new studies beyond that, it’s one of the reasons that we’re looking at the strategic alternatives for the company, because I think in order to do that, we need to raise dedicated funding for that. At the same time, we recognize that for the Avid business continue to grow, we should raise dedicated funding for that business, in order to add on to our capabilities to upgrade some of our capabilities and then to add on more capacity at the right time. So, right now it’s sort of status quo as we move forward on the R&D front, and as we’re able to really have a clearly defined pathway forward for the R&D business with the appropriate funding, then I know our group already has some plans of what could be the next study designs.
Paul Lytle: The Phase II that you alluded to that that does fall under our $2mm NCCN grant, some of which was paid last FY year and then there’s some being paid this FY. But that covers 3 trials being run by this NCCN consortium.
AS: ”So there shouldn’t be too much of an impact then as we go through the year unless you guys decide to be more aggressive or able to get the adequate funds?”
Paul Lytle: Correct.
AS: ”You talked about potential strategic relationships and you have a preclinical collaboration with Sloan Kettering. Can you elaborate how that collaboration gets extended into a clinical work? And then how it works when you tie into a strategic partner? Does that all have to be rehashed out, or are there some covenants in there that make that a simple process?”
Steve King: One of the goals of the group is that Sloan Kettering is to directly translate what they’re doing on the preclinical front into clinical studies at the institution. And so, they typically only want to work on things that they have that opportunity for. If we think about the possibility of bavi in combination with CAR-T therapy, then we would expect that once they’ve generated the right amount of data then we would have a discussion with obviously one of the CAR-T companies about collaborating on moving that forward under the umbrella of the group there at Sloan Kettering. We’re working with probably the top immunology lab [Jedd Wolchok’s Lab] when it comes to the development of immune checkpoint inhibitors there, and they have very, very tight clinical connections. Jedd Wolchok obviously is the head of the lab there and he’s very well connected. So, we would expect actually that some doors can open through the collaboration and through the introductions, because we know the clinicians at Sloan Kettering already work with all the major pharma, and so there are already good connections there that can help really hopefully expedite those into clinical development.
AS: ”Does it have to be CAR-T, or other immuno-oncology optionalities open as well, or are you kind of just focused strictly on that vertical right now?”
Steve King: That’s [CAR-T] is one of their favorite projects, so they’ve really been excited about the data, because if you look at the results were presented, it’s really the ability of bavituximab to enhance the activity of the adoptive T cell therapy vs. the lack of any additional toxicity. And, when they compared it with an agent like OX40 that actually also increased activity, but also had more safety events, then they really like that approach. But no, this could extend to PD-1/PD-L1 probably quite a number of other molecules that there’s an interest in. So, there’s a lot of interest across the board, because as the field is maturing and more results are coming out, you’re running into two things. One is, drugs just don’t work well together either from a safety standpoint or there’s no enhanced efficacy, and we’re seeing more & more of that reported. I think there’s a real window of opportunity here. In addition, you have patients that are on treatments that develop a resistance to the therapies, and that opens up another door for, based on the mechanism of action for bavi, to potentially reinvigorate the ability of the PD-1/PD-L1 inhibitors. The Sloan Kettering Group, there are quite a number of actual researchers that are working on it, each of which are looking at different aspect. CAR-T and adoptive T cell is just simply one of those.
MR. KING’S CLOSING COMMENTS:
I’d like to thank you all again for participating in today’s phone call. As always, I want to thank you, our stockholders, for your continued support, and I would like to specially thank our patients, their families and the investigators that are participating in our bavituximab clinical trials. With that, we will conclude the call.
= = = = = = = = = = = = = = = = = = = = = = = = = = = = = == = = =
7-14-17/PR: Peregrine Pharmaceuticals Reports Financial Results for Quarter and FY Ended April 30, 2017 and Recent Developments
**Avid Achieves Year-Over-Year Topline Revenue Growth of 30% Exceeding $57 Million
**Overall Survival in Phase III SUNRISE Subgroup Receiving Subsequent Immune Checkpoint Inhibitors
**Recent Presentation Supports Bavituximab's Potential to Improve Clinical Outcome for Immune Checkpoint Inhibitors
http://ir.peregrineinc.com/releasedetail.cfm?ReleaseID=1033237
TUSTIN, July 14, 2017: Peregrine Pharmaceuticals, Inc. (NASDAQ:PPHM/PPHMP), a biopharmaceutical company committed to improving patient lives by manufacturing high quality products for biotechnology and pharmaceutical companies, and advancing its proprietary R&D pipeline, today announced financial results for the fourth quarter and fiscal year (FY) ended April 30, 2017, and provided an update on its contract manufacturing operations, research and development programs, and other corporate highlights.
Highlights Since January 31, 2017
"We are very pleased to announce that the business recorded its highest annual revenue to date during fiscal 2017," stated Steven W. King, President & CEO of Peregrine, and President of Avid Bioservices. "While we are happy to report continued year-over-year revenue growth, we are projecting revenues for FY 2018 to be similar with FY 2017 due to recent changes in the forecast of a large customer and a delayed regulatory filing for another customer. We believe this is a temporary lull and remain confident that Avid is in a strong position for continued growth in the future." Mr. King continued, "An important part of continuing revenue growth and reducing risk for the business is to attract new customers. On that front, we have recently signed four new customers that we expect to contribute to top-line revenue in FY 2018 and into the future. In addition, we successfully completed three process validation campaigns this fiscal year for existing third-party customers which we believe may significantly contribute to future revenue as these customers move toward building inventory for their potential launch, and commercial supply.
"We are also continuing to take other important steps to ensure Avid's growth in the coming years. As part of this effort, we have recently installed two new 2,000 liter bioreactors in our Myford facility, and we already have commitments for part of this capacity. Due to its state-of-the-art and modular design, there remains potential to install additional bioreactors in Myford, which will allow us to continue growing the business within our current facilities.
"Turning to the Peregrine business, we were able to generate some of the most compelling bavituximab data to date, further supporting the combination of bavituximab and checkpoint inhibitors even while reducing R&D spending by over 50% in FY 2017. The clinical findings came from the comprehensive analysis of maturing Phase III SUNRISE data while impressive preclinical results came from our collaborators at Memorial Sloan Kettering Cancer Center (MSKCC). The researchers at MSKCC presented promising preclinical data combining PS-targeting with adoptive T cell transfer therapy which may support the combination of bavituximab with CAR-T cell immunotherapy in the future.
"Both Peregrine and Avid have achieved important milestones in recent quarters. The success of Avid and its continued revenue growth with the compelling data we have seen from the Phase III SUNRISE trial have led us to explore various strategic options that we believe will enable us to enhance stockholder value for all stockholders, including the possible separation of these two distinct businesses."
Research and Development Highlights
"The most compelling data to date from the Phase III SUNRISE trial was presented at AACR and together with the PD-L1 results presented at ASCO this year add to the growing body of data supporting the further development of bavituximab with checkpoint inhibitors," said Joseph Shan, VP of Clinical & Regulatory affairs at Peregrine.
AACR Highlights:
In a subgroup analysis, Peregrine researchers looked at the outcome of 91 patients that were enrolled in the Phase III SUNRISE trial that were subsequently treated with anti-PD-1/PD-L1 immune checkpoint inhibitors ("ICI's") post study treatments. The results from this analysis demonstrated that the patients who received docetaxel plus bavituximab (Doc+Bavi) and subsequent ICI had not yet reached median overall survival ("mOS") compared to mOS of 13.0 months for patients who received docetaxel plus placebo (Doc+Placebo) (hazard ratio [HR], 0.43; p=0.005). The statistically significant difference between the two arms in the trial provides strong rationale for combining bavituximab with ICI's and supports the hypothesis that bavituximab may modulate the tumor microenvironment to enhance the anti-tumor activity of ICI's.
A photo accompanying this announcement is available at http://www.globenewswire.com/NewsRoom/AttachmentNg/428c1a61-a599-4976-8a92-d65148671bcd
ASCO Highlights:
Peregrine researchers presented additional supportive data demonstrating that patients in the bavituximab containing arm who had low baseline PD-L1 expression on tumor cells (i.e., patients typically with poorer response to PD-1/PD-L1 checkpoint inhibitors) lived significantly longer than patients with high baseline PD-L1 expression. These data further support the hypothesis that bavituximab may modulate the tumor microenvironment to complement and enhance the anti-tumor activity of ICI's.
NCCN Highlights:
The three clinical trials under the collaboration with the NCCN are advancing as planned.
Massachusetts General Hospital Cancer Center - Phase I/II Clinical Trial of Bavituximab with Radiation and Temozolomide for Patients with Newly Diagnosed Glioblastoma. This trial is open for enrollment.
Moffitt Cancer Center - A Phase I Trial of Sorafenib and Bavituximab Plus Stereotactic Body Radiation Therapy for Unresectable Hepatitis C Associated Hepatocellular Carcinoma. This trial is open for enrollment.
The Sidney Kimmel Comprehensive Cancer Center at Johns Hopkins - Phase II Study of Pembrolizumab and Bavituximab for Progressive Recurrent/Metastatic Squamous Cell Carcinoma of the Head and Neck. This trial is expected to be initiated by the end of the calendar year 2017.
Preclinical Highlights:
Researchers from Memorial Sloan Kettering Cancer Center (MSKCC) presented a preclinical study evaluating the anti-tumor activity and toxicities of adoptive T cell transfer therapy in combination with either PS-targeting antibodies or anti-OX40 antibodies in mice with advanced melanomas. Whereas PS-targeting and anti-OX40 demonstrated comparable tumor regression when administered in combination with transferred adoptive T cells, only the PS-targeting combination achieved these results without any off-target toxicities. In contrast, the anti-OX40 treatment combination triggered off-target side effects.
PS Exosome Technology Highlights:
The company continues to make progress with its PS exosome diagnostic technology that is designed to detect and monitor cancer. The assay has been successfully optimized and we are currently preparing to generate additional data by testing human samples. Such data will be important to partnering discussions.
Avid Bioservices Highlights
"FY 2017 was a strong year for Avid Bioservices with year-over-year revenue growth of 30% compared to FY 2016. The company recognized revenue of $17.9 million for the fourth quarter and $57.6 million for the full fiscal year," stated Paul Lytle, CFO of Peregrine. "While we missed our revenue guidance of $60-65 million, we were ready to ship a number of process validation runs which were delayed due to events outside our control. This delay has caused approximately $10 million in manufacturing revenue to shift from fiscal year 2017 to fiscal year 2018."
The company is providing manufacturing revenue guidance for the full FY 2018 of $50-55 million.
Avid's current manufacturing revenue backlog is $58 million, representing estimated future manufacturing revenue to be recognized under committed contracts. Most of the backlog is expected to be recognized during FY 2018.
Financial Highlights and Results
During the fourth quarter of FY 2017, we recorded total revenues of $17,904,000 as compared to $18,783,000 in the fourth quarter of the prior FY. For FY 2017, we achieved total revenues of $57,630,000 as compared to $44,686,000 for FY 2016. This represents total revenue growth of 29% for FY 2017 compared to the same prior year period.
Contract manufacturing revenue from Avid's clinical and commercial biomanufacturing services was $17,904,000 for the fourth quarter of FY 2017 compared to $18,783,000 for the fourth quarter of FY 2016. For the year, revenue increased 30% to $57,630,000 for FY 2017 compared to $44,357,000 for FY 2016. The fiscal year increase was primarily attributed to an increase in demand for contract manufacturing services associated with process validation activities. Current committed manufacturing backlog for Avid is approximately $58 million, covering services to be provided during FY 2018 and into FY 2019. Based on this current backlog, Peregrine expects contract manufacturing revenue for FY 2018 to be between $50 and $55 million.
Total costs and expenses for the fourth quarter of FY 2017 were $23,208,000, compared to $30,698,000 for the fourth quarter of FY 2016. For FY 2017, total costs and expenses were $85,890,000 compared to $101,046,000 for FY 2016. For the fourth quarter of FY 2017, research and development expenses decreased 59% to $6,717,000, compared to $16,265,000 for the fourth quarter of FY 2016. For FY 2017, research and development expenses decreased 52% to $28,297,000 compared to $59,529,000 for FY 2016.
Cost of contract manufacturing increased to $11,782,000 in the fourth quarter of FY 2017 compared to $9,721,000 for the fourth quarter of FY 2016, and to $38,259,000 for the full FY 2017 as compared to $22,966,000 for the full FY 2016. These increases are primarily due to an increase in the cost of contract manufacturing associated with higher reported revenue. Also contributing to this increase and impacting gross margins for the period is the higher overhead cost of operating the new Myford facility as well as higher labor cost associated with performing process validation runs combined with lower utilization of available capacity. For the fourth quarter of FY 2017, selling, general and administrative expenses decreased slightly to $4,709,000 compared to $4,712,000 for FY 2016. For FY 2017 selling, general and administrative expenses increased to $19,334,000 compared to $18,551,000 for FY 2016. The full-year increase is primarily due to the company's growing manufacturing business.
Peregrine's consolidated net loss attributable to common stockholders was $6,714,000 or $0.16 per share, for the fourth quarter of FY 2017, compared to a net loss attributable to common stockholders of $13,264,000, or $0.40 per share, for the same prior year quarter. For FY 2017, net loss attributable to common stockholders was $32,799,000, or $0.88 per share, compared to $60,136,000, or $1.95 per share, for FY 2016.
Peregrine reported $46,799,000 in cash and cash equivalents as of April 30, 2017, compared to $61,412,000 at fiscal year ended April 30, 2016.
More detailed financial information and analysis may be found in Peregrine's Annual Report on Form 10-K, which will be filed with the Securities and Exchange Commission today. [ http://tinyurl.com/ycxu4l5n ]
CONFERENCE CALL
Peregrine will host a conference call and webcast this afternoon, July 14, 2017, at 4:30 PM EDT (1:30 PM PDT). To listen to the conference call, please dial (877) 312-5443 or (253) 237-1126 and request the Peregrine Pharmaceuticals conference call. To listen to the live webcast, or access the archived webcast, please visit: http://ir.peregrineinc.com/events.cfm .
About Peregrine Pharmaceuticals, Inc.
Peregrine Pharmaceuticals, Inc. is a biopharmaceutical company committed to improving the lives of patients by delivering high quality pharmaceutical products through its contract development and manufacturing organization (CDMO) services and through advancing and licensing its investigational immunotherapy and related products. Peregrine's in-house CDMO services, including cGMP manufacturing and development capabilities, are provided through its wholly-owned subsidiary Avid Bioservices, Inc. ( http://www.avidbio.com ), which provides development and biomanufacturing services for both Peregrine and third-party customers. The company is also working to evaluate its lead immunotherapy candidate, bavituximab, in combination with immune stimulating therapies for the treatment of various cancers, and developing its proprietary exosome technology for the detection and monitoring of cancer. For more information, please visit http://www.peregrineinc.com .
About Avid Bioservices
Avid Bioservices provides a comprehensive range of process development, high quality cGMP clinical and commercial manufacturing services for the biotechnology and biopharmaceutical industries. With over 15 years of experience producing monoclonal antibodies and recombinant proteins in batch, fed-batch and perfusion modes, Avid's services include cGMP clinical and commercial product manufacturing, purification, bulk packaging, stability testing and regulatory strategy, submission and support. The company also provides a variety of process development activities, including cell line development and optimization, cell culture and feed optimization, analytical methods development and product characterization. For more information about Avid, please visit http://www.avidbio.com .
PEREGRINE PHARMACEUTICALS, INC.
CONSOLIDATED STATEMENTS OF OPERATIONS AND COMPREHENSIVE LOSS
THREE MONTHS ENDED
APRIL 30, TWELVE MONTHS ENDED
APRIL 30,
2017 2016 2017 2016
Unaudited Unaudited
REVENUES:
Contract manufacturing revenue $ 17,904,000 $ 18,783,000 $ 57,630,000 $ 44,357,000
License revenue — — — 329,000
Total revenues 17,904,000 18,783,000 57,630,000 44,686,000
COSTS AND EXPENSES:
Cost of contract manufacturing 11,782,000 9,721,000 38,259,000 22,966,000
Research and development 6,717,000 16,265,000 28,297,000 59,529,000
Selling, general and administrative 4,709,000 4,712,000 19,334,000 18,551,000
Total costs and expenses 23,208,000 30,698,000 85,890,000 101,046,000
LOSS FROM OPERATIONS (5,304,000 ) (11,915,000 ) (28,260,000 ) (56,360,000 )
OTHER INCOME (EXPENSE):
Interest and other income 37,000 31,000 108,000 722,000
Interest and other expense (5,000 ) — (7,000 ) (14,000 )
NET LOSS $(5,272,000) $ (11,884,000 ) $ (28,159,000 ) $ (55,652,000 )
COMPREHENSIVE LOSS $ (5,272,000 ) $ (11,884,000 ) $ (28,159,000 ) $ (55,652,000 )
Series E preferred stock accumulated dividends (1,442,000 ) (1,380,000 ) (4,640,000 ) (4,484,000 )
NET LOSS ATTRIBUTABLE TO COMMON STOCKHOLDERS $ (6,714,000
) $ (13,264,000 ) $ (32,799,000 ) $ (60,136,000)
WEIGHTED AVERAGE COMMON SHARES OUTSTANDING:
Basic and Diluted (1) 42,141,720 33,478,863 37,109,493 30,895,089
BASIC AND DILUTED LOSS PER COMMON SHARE (1) $ (0.16 ) $ (0.40 ) $ (0.88 ) $ (1.95 )
(1) All share and per share amounts of our common stock issued and outstanding for all periods have been retroactively adjusted to reflect the one-for-seven reverse stock split which took effect with the opening of trading on July 10, 2017.
PEREGRINE PHARMACEUTICALS, INC.
CONSOLIDATED BALANCE SHEETS
AS OF APRIL 30, 2017 AND 2016
2017 2016
ASSETS
CURRENT ASSETS:
Cash and cash equivalents $ 46,799,000 $ 61,412,000
Trade and other receivables 7,742,000 2,859,000
Inventories 33,099,000 16,186,000
Prepaid expenses 1,460,000 1,351,000
Total current assets 89,100,000 81,808,000
PROPERTY AND EQUIPMENT:
Leasehold improvements 20,098,000 19,610,000
Laboratory equipment 10,777,000 10,257,000
Furniture, fixtures, office equipment and software 4,499,000 4,045,000
35,374,000 33,912,000
Less accumulated depreciation and amortization (11,700,000 ) (9,610,000 )
Property and equipment, net 23,674,000 24,302,000
Restricted cash 1,150,000 600,000
Other assets 4,188,000 2,333,000
TOTAL ASSETS $ 118,112,000 $ 109,043,000
PEREGRINE PHARMACEUTICALS, INC.
CONSOLIDATED BALANCE SHEETS
AS OF APRIL 30, 2017 AND 2016 (continued)
2017 2016
LIABILITIES AND STOCKHOLDERS' EQUITY
CURRENT LIABILITIES:
Accounts payable $ 5,779,000 $ 8,429,000
Accrued clinical trial and related fees 4,558,000 7,594,000
Accrued payroll and related costs 6,084,000 5,821,000
Deferred revenue 28,500,000 10,030,000
Customer deposits 17,017,000 24,212,000
Other current liabilities 993,000 1,488,000
Total current liabilities 62,931,000 57,574,000
Deferred rent, less current portion 1,599,000 1,395,000
Commitments and contingencies
STOCKHOLDERS' EQUITY (1):
Preferred stock - $.001 par value; authorized 5,000,000 shares;
issued and outstanding - 1,647,760 and 1,577,440, respectively 2,000 2,000
Common stock - $.001 par value; authorized 500,000,000
shares; issued and outstanding - 44,014,040 and 33,847,213,
respectively 44,000 34,000
Additional paid-in-capital 590,971,000 559,314,000
Accumulated deficit (537,435,000 ) (509,276,000 )
Total stockholders' equity 53,582,000 50,074,000
TOTAL LIABILITIES AND STOCKHOLDERS' EQUITY $ 118,112,000 $ 109,043,000
(1) All share and per share amounts of our common stock issued and outstanding for all periods have been retroactively adjusted to reflect the one-for-seven reverse stock split which took effect with the opening of trading on July 10, 2017.
Safe Harbor *snip*
CONTACTS:
• Stephanie Diaz (Investors) Vida Strategic Partners 415-675-7401 sdiaz@vidasp.com
• Tim Brons (Media) Vida Strategic Partners 415-675-7402 tbrons@vidasp.com
- - - - - - - - -
From 10-Q header: “As of Jul. 10, 2017, there were 45,069,188 shares outstanding.”
- - - - - - - - - - - - - - - - -
Latest 10K 4-30-17 iss. 7-14-17 http://tinyurl.com/ycxu4l5n PR: http://tinyurl.com/yb4wulvu (Cash 4-30-17=$46.8mm)
Latest 10Q 1-31-17 iss. 3-13-17 http://tinyurl.com/zlozrv4 PR: http://tinyurl.com/grhwjvy (Cash 1-31-17=$41.5mm)
ALL SEC filings for PPHM: http://tinyurl.com/6d4jw8
7-14-17: O/S Shares & ATM Sales History (’06–curr.) http://tinyurl.com/yagmu2on
**NOTES:
1. PPHM shares were 1:7 Reverse Split eff. 7-10-17 (~315mm/$.606=>~45mm/$4.24) http://tinyurl.com/ycohqn6j
2. Per the 4-30-17 10-K, ALL ATM Agreements have been fully used – none remain. http://tinyurl.com/ycxu4l5n (F-20 & F-28)
= = = = = = = = = = = = = = = = = = = = = = = = = = = =
Updated PPHM REVS-BY-QTR TABLE, now thru FY17'Q4(fye 4-30-17), per the 10-K ( http://tinyurl.com/ycxu4l5n ) issued 7-14-17.
• Total Revs since May’06: ($231.2mm/Avid + $24.1mm/Govt + $2.5mm/Lic.) = $257.8mm
• 7-14-17: FY'18 (May'17-Apr'18) Avid revs guidance $50-55mm (committed B/L=$58mm). **
• Deferred-Revs at 4-30-17 total $28.5mm, UP from $26.4mm at 1-31-17.
• Cust.Deposits at 4-40-17 total $17.0mm, DOWN from $26.2mm at 1-31-17.
• Inventories at 4-30-17 total $33.1mm, DOWN from $33.8mm at 1-31-17.
• Avid’s Gross-Profit over last 4 qtrs: $19.4mm on revs of $57.6mm (GP%=34%)
• Recall, Avid Rev$ from Gov’t DTRA Contract work (6/30/08 – 4/15/11, totaling $24.15mm), went into GOVT-REVS, not AVID-REVS, in the Financials.
**4-30-17 10K/p.33: “Excluding any future potential new business, we expect Avid revs for FY18 to slightly decline vs. FY17. Part of this decline is due to lower anticipated commitments from Halozyme (our largest customer) based on their most recent committed forecast (covering the 3 qtrs ending March 2018), which amount is expected to be partially offset by $10mm revenue that was expected to be recognized in FY17, but has been shifted to FY18 due to a delay in shipping product that was complete and ready for shipment as of fye 4-30-17. http://tinyurl.com/ycxu4l5n
Avid’s website: http://www.avidbio.com
AVID PROFITABILITY (GROSS*) BY QTR:
QTR Avid-Rev$ CostofMfg$ Gross-Profit$ GP%
FY13Q1 7-31-12 4,135,000 2,024,000 2,111,000 51%
FY13Q2 10-31-12 6,061,000 3,703,000 2,358,000 39%
FY13Q3 1-31-13 6,961,000 3,651,000 3,310,000 47%
FY13Q4 4-30-13 4,176,000 3,217,000 959,000 23%
FY14Q1 7-31-13 4,581,000 2,670,000 1,911,000 42%
FY14Q2 10-31-13 7,354,000 4,195,000 3,159,000 43%
FY14Q3 1-31-14 3,885,000 2,416,000 1,469,000 38%
FY14Q4 4-30-14 6,474,000 3,829,000 2,645,000 41%
FY15Q1 7-31-14 5,496,000 3,583,000 1,913,000 35%
FY15Q2 10-31-14 6,263,000 4,139,000 2,124,000 34%
FY15Q3 1-31-15 5,677,000 3,113,000 2,564,000 45%
FY15Q4 4-30-15 9,308,000 4,758,000 4,550,000 49%
FY16Q1 7-31-15 9,379,000 4,608,000 4,771,000 51%
FY16Q2 10-31-15 9,523,000 4,741,000 4,782,000 50%
FY16Q3 1-31-16 6,672,000 3,896,000 2,776,000 42%
FY16Q4 4-30-16 18,783,000 9,721,000 9,062,000 48%
FY17Q1 7-31-16 5,609,000 3,062,000 2,547,000 45%
FY17Q2 10-31-16 23,370,000 15,441,000 7,929,000 34%
FY17Q3 1-31-17 10,747,000 7,974,000 2,773,000 26%
FY17Q4 4-30-17 17,904,000 11,782,000 6,122,000 34%
FY13 TOTAL: 21,333,000 12,595,000 8,738,000 41%*
FY14 TOTAL: 22,294,000 13,110,000 9,184,000 41%*
FY15 TOTAL: 26,744,000 15,393,000 11,151,000 42%*
FY16 TOTAL: 44,357,000 22,966,000 21,391,000 48%*
FY17 TOTAL: 57,630,000 38,259,000 19,371,000 34%*
*Avid Net-Profit (ie, incl. Selling, G&A) not split out from PPHM-Corp. in the financials.
.
PPHM REVENUES (in thousands) DEFERRED
-------REVENUES------- REVENUES INVEN-
Quarter Avid Govt Lic. TOTAL Avid Govt TORIES
FY07Q1 7-31-06 398 0 23 421 317 0 971
FY07Q2 10-31-06 636 0 48 684 1388 0 1899
FY07Q3 1-31-07 347 0 16 363 2202 0 1325
FY07Q4 4-30-07 2111 0 129 2240 1060 0 1916
FY08Q1 7-31-07 1621 0 4 1625 1820 0 2363
FY08Q2 10-31-07 1863 0 29 1892 1338 0 3500
FY08Q3 1-31-08 1662 0 13 1675 1434 0 2394
FY08Q4 4-30-08 751 0 150 901 2196 0 2900
FY09Q1 7-31-08 1193 324 0 1517 4021 980 4628
FY09Q2 10-31-08 983 958 0 1941 6472 1701 6700
FY09Q3 1-31-09 5778 1048 0 6826 4805 3262 5547
FY09Q4 4-30-09 5009 2683 175 7867 3776 3871 4707
FY10Q1 7-31-09 2070 4671 9 6750 5755 2332 6177
FY10Q2 10-31-09 5308 1510 78 6896 4260 3989 5850
FY10Q3 1-31-10 2945 6854 78 9877 3052 76 3861
FY10Q4 4-30-10 2881 1461 78 4420 2406 78 3123
FY11Q1 7-31-10 983 2111 115 3209 3719 47 4692
FY11Q2 10-31-10 3627 966 78 4671 2447 35 3555
FY11Q3 1-31-11 1922 882 79 2883 4300 40 3915
FY11Q4 4-30-11 1970 681 78 2729 5617 0 5284
FY12Q1 7-31-11 5439 0 216 5655 4145 0 4481
FY12Q2 10-31-11 4154 0 78 4232 2012 0 3178
FY12Q3 1-31-12 3203 0 78 3281 2552 0 2722
FY12Q4 4-30-12 1987 0 78 2065 3651 0 3611
FY13Q1 7-31-12 4135 0 116 4251 6056 0 5744
FY13Q2 10-31-12 6061 0 78 6139 6221 0 5426
FY13Q3 1-31-13 6961 0 78 7039 5061 0 4635
FY13Q4 4-30-13 4176 0 78 4254 4171 0 4339
FY14Q1 7-31-13 4581 0 107 4688 4164 0 5679
FY14Q2 10-31-13 7354 0 0 7354 3468 0 4033
FY14Q3 1-31-14 3885 0 0 3885 4329 0 5224
FY14Q4 4-30-14 6474 0 0 6474 5241 0 5530
FY15Q1 7-31-14 5496 0 0 5496 4670 0 5998
FY15Q2 10-31-14 6263 0 37 6300 3612 0 5379
FY15Q3 1-31-15 5677 0 0 5677 5752 0 6148
FY15Q4 4-30-15 9308 0 0 9308 6630 0 6148
FY16Q1 7-31-15 9379 0 292 9671 8291 0 10457
FY16Q2 10-31-15 9523 0 0 9523 9688 0 12554
FY16Q3 1-31-16 6672 0 37 6709 15418 0 15189
FY16Q4 4-30-16 18783 0 0 18783 15418 0 15189
FY17Q1 7-31-16 5609 0 0 5609 21531 0 25274
FY17Q2 10-31-16 23370 0 0 23370 21531 0 25274
FY17Q3 1-31-17 10747 0 0 10747 26367 0 33829
FY17Q4 4-30-17 17904 0 0 17904 28500 0 33099
Totals: 231199 24149 2453 257801 <=since5/1/2006
.
TOTAL REV’s BY YEAR (Avid+Gov’t+Lic):
FY04 4-30-04 3,314 …Avid(CMO)= 3,039 (Avid-Revs don’t incl. Govt-SVCS)
FY05 4-30-05 4,959 …Avid(CMO)= 4,684
FY06 4-30-06 3,193 …Avid(CMO)= 3,005
FY07 4-30-07 3,708 …Avid(CMO)= 3,492
FY08 4-30-08 6,093 …Avid(CMO)= 5,897
FY09 4-30-09 18,151 …Avid(CMO)= 12,963
FY10 4-30-10 27,943 …Avid(CMO)= 13,204
FY11 4-30-11 13,492 …Avid(CMO)= 8,502
FY12 4-30-12 15,233 …Avid(CMO)= 14,783
FY13 4-30-13 21,683 …Avid(CMO)= 21,333
FY14 4-30-14 22,401 …Avid(CMO)= 22,294
FY15 4-30-15 26,781 …Avid(CMO)= 26,744
FY16 4-30-16 44,686 …Avid(CMO)= 44,357
FY17 4-30-17 57,630 …Avid(CMO)= 57,630
...Total Gov’t Revs from 7-2008 inception thru FY11Q1(Apr’11): $24.15mm
.
PPHM’S QTLY. NET LOSS BY QTR:
FY08Q1 7-31-07 4,656,000
FY08Q2 10-31-07 6,207,000
FY08Q3 1-31-08 6,154,000
FY08Q4 4-30-08 6,159,000
FY09Q1 7-31-08 5,086,000
FY09Q2 10-31-08 4,497,000
FY09Q3 1-31-09 3,332,000
FY09Q4 4-30-09 3,609,000
FY10Q1 7-31-09 2,428,000
FY10Q2 10-31-09 2,787,000
FY10Q3 1-31-10 1,538,000
FY10Q4 4-30-10 7,741,000
FY11Q1 7-31-10 7,695,000
FY11Q2 10-31-10 7,513,000
FY11Q3 1-31-11 8,929,000
FY11Q4 4-30-11 10,014,000
FY12Q1 7-31-11 8,092,000
FY12Q2 10-31-11 12,055,000
FY12Q3 1-31-12 11,090,000
FY12Q4 4-30-12 10,882,000
FY13Q1 7-31-12 7,664,000
FY13Q2 10-31-12 8,753,000
FY13Q3 1-31-13 4,914,000
FY13Q4 4-30-13 8,449,000
FY14Q1 7-31-13 7,600,000
FY14Q2 10-31-13 7,790,000
FY14Q3 1-31-14 9,724,000
FY14Q4 4-30-14 10,248,000
FY15Q1 7-31-14 13,129,000
FY15Q2 10-31-14 12,100,000
FY15Q3 1-31-15 12,994,000
FY15Q4 4-30-15 12,135,000
FY16Q1 7-31-15 13,723,000
FY16Q2 10-31-15 13,198,000
FY16Q3 1-31-16 16,847,000
FY16Q4 4-30-16 11,884,000
FY17Q1 7-31-16 11,057,000
FY17Q2 10-31-16 4,056,000
FY17Q3 1-31-17 7,774,000
FY17Q4 4-30-17 5,272,000
= = = = = = = =
OPER. CASH BURNS* BY QTR(FROM THE 10-Q/K’S):
FY10Q1 7-31-09 2,024,000 (from 10Q pg.25)
FY10Q2 10-31-09 2,351,000 (Q1+Q2: 4,375,000 pg.28)
FY10Q3 1-31-10 1,158,000 (Q1+Q2+Q3: 5,533,000 pg.30)
FY10Q4 4-30-10 6,375,000 (FY’10: 11,908,000 10K pg.58)
FY11Q1 7-31-10 6,567,000 (from 10Q pg.24)
FY11Q2 10-31-10 6,167,000 (Q1+Q2: $12,734,000 pg.25)
FY11Q3 1-31-11 7,736,000 (Q1+Q2+Q3: $20,470,000 pg.26)
FY11Q4 4-30-11 8,961,000 (FY’11: 29,431,000 10K pg.54)
FY12Q1 7-31-11 6,984,000 (from 10Q pg.25)
FY12Q2 10-31-11 11,668,000 (Q1+Q2: 18,652,000 pg.25)
FY12Q3 1-31-12 8,490,000 (Q1+Q2+Q3: 27,142,000 pg.25)
FY12Q4 4-30-12 11,265,000 (FY’12: 38,407,000 10K pg.55)
FY13Q1 7-31-12 6,742,000 (from 10Q pg.21)
FY13Q2 10-31-12 6,162,000 (Q1+Q2: 12,904,000 pg.23)
FY13Q3 1-31-13 3,597,000 (Q1+Q2+Q3: 16,501,000 pg.23)
FY13Q4 4-30-13 7,053,000 (FY’13: 23,554,000 10K pg.60)
FY14Q1 7-31-13 5,750,000 (from 10Q pg.23)
FY14Q2 10-31-13 5,834,000 (Q1+Q2: 11,584,000 10Q pg.24)
FY14Q3 1-31-14 7,875,000 (Q1+Q2+Q3: 19,459,000 10Q pg.26)
FY14Q4 4-30-14 8,706,000 (FY’14: 28,165,000 10K pg.55)
FY15Q1 7-31-14 11,076,000 (from 10Q pg.23)
FY15Q2 10-31-14 9,947,000 (Q1+Q2: 21,023,000 10Q pg.25)
FY15Q3 1-31-15 11,116,000 (Q1+Q2+Q3: 32,139,000 10Q pg.26)
FY15Q4 4-30-15 10,474,000 (FY’15: 42,613,000 10K pg.54)
FY16Q1 7-31-15 12,306,000 (from 10Q pg.25)
FY16Q2 10-31-15 11,701,000 (Q1+Q2: 24,007,000 10Q pg.26)
FY16Q3 1-31-16 15,086,000 (Q1+Q2+Q3: 39,093,000 10Q pg.27)
FY16Q4 4-30-16 10,112,000 (FY'16: 49,205,000 10K pg.39)
FY17Q1 7-31-16 9,607,000 (from 10Q pg.22)
FY17Q2 10-31-16 2,565,000 (Q1+Q2: 12,172,000 10Q pg.24)
FY17Q3 1-31-17 6,274,000 (Q1+Q2+Q3: 18,446,000 10Q pg.24)
FY17Q4 4-30-17 3,886,000 (FY'17: 22,332,000 10K pg.40)
FY’09 total Op-Burn: $14,715,000
FY’10 total Op-Burn: $11,908,000
FY’11 total Op-Burn: $29,431,000
FY’12 total Op-Burn: $38,407,000
FY’13 total Op-Burn: $23,554,000
FY’14 total Op-Burn: $28,165,000
FY’15 total Op-Burn: $42,613,000
FY’16 total Op-Burn: $49,205,000
FY’17 total Op-Burn: $22,332,000
Period Halozyme Cust-A Other-Custs
FYE 4-30-14 91% 1% 8%
FYE 4-30-15 79% 12% 9%
FYE 4-30-16 69% 26% 5%
Q/E 7-31-16 65% 29% 6%
Q/E 10-31-16 77% 10% 13%
Q/E 1-31-17 29% 56% 15%
FYE 4-30-17 58% 26% 16%
Updated PPHM REVS-BY-QTR TABLE, now thru FY17'Q4(fye 4-30-17), per the 10-K ( http://tinyurl.com/ycxu4l5n ) issued 7-14-17.
• Total Revs since May’06: ($231.2mm/Avid + $24.1mm/Govt + $2.5mm/Lic.) = $257.8mm
• 7-14-17: FY'18 (May'17-Apr'18) Avid revs guidance $50-55mm (Committed B/L=$58mm).
• Deferred-Revs at 4-30-17 total $28.5mm, UP from $26.4mm at 1-31-17.
• Cust.Deposits at 4-40-17 total $17.0mm, DOWN from $26.2mm at 1-31-17.
• Inventories at 4-30-17 total $33.1mm, DOWN from $33.8mm at 1-31-17.
• Avid’s Gross-Profit over last 4 qtrs: $19.4mm on revs of $57.6mm (GP%=34%)
• Recall, Avid Rev$ from Gov’t DTRA Contract work (6/30/08 – 4/15/11, totaling $24.15mm), went into GOVT-REVS, not AVID-REVS, in the Financials.
Avid’s website: http://www.avidbio.com
AVID PROFITABILITY (GROSS*) BY QTR:
QTR Avid-Rev$ CostofMfg$ Gross-Profit$ GP%
FY13Q1 7-31-12 4,135,000 2,024,000 2,111,000 51%
FY13Q2 10-31-12 6,061,000 3,703,000 2,358,000 39%
FY13Q3 1-31-13 6,961,000 3,651,000 3,310,000 47%
FY13Q4 4-30-13 4,176,000 3,217,000 959,000 23%
FY14Q1 7-31-13 4,581,000 2,670,000 1,911,000 42%
FY14Q2 10-31-13 7,354,000 4,195,000 3,159,000 43%
FY14Q3 1-31-14 3,885,000 2,416,000 1,469,000 38%
FY14Q4 4-30-14 6,474,000 3,829,000 2,645,000 41%
FY15Q1 7-31-14 5,496,000 3,583,000 1,913,000 35%
FY15Q2 10-31-14 6,263,000 4,139,000 2,124,000 34%
FY15Q3 1-31-15 5,677,000 3,113,000 2,564,000 45%
FY15Q4 4-30-15 9,308,000 4,758,000 4,550,000 49%
FY16Q1 7-31-15 9,379,000 4,608,000 4,771,000 51%
FY16Q2 10-31-15 9,523,000 4,741,000 4,782,000 50%
FY16Q3 1-31-16 6,672,000 3,896,000 2,776,000 42%
FY16Q4 4-30-16 18,783,000 9,721,000 9,062,000 48%
FY17Q1 7-31-16 5,609,000 3,062,000 2,547,000 45%
FY17Q2 10-31-16 23,370,000 15,441,000 7,929,000 34%
FY17Q3 1-31-17 10,747,000 7,974,000 2,773,000 26%
FY17Q4 4-30-17 17,904,000 11,782,000 6,122,000 34%
FY13 TOTAL: 21,333,000 12,595,000 8,738,000 41%*
FY14 TOTAL: 22,294,000 13,110,000 9,184,000 41%*
FY15 TOTAL: 26,744,000 15,393,000 11,151,000 42%*
FY16 TOTAL: 44,357,000 22,966,000 21,391,000 48%*
FY17 TOTAL: 57,630,000 38,259,000 19,371,000 34%*
*Avid Net-Profit (ie, incl. Selling, G&A) not split out from PPHM-Corp. in the financials.
.
PPHM REVENUES (in thousands) DEFERRED
-------REVENUES------- REVENUES INVEN-
Quarter Avid Govt Lic. TOTAL Avid Govt TORIES
FY07Q1 7-31-06 398 0 23 421 317 0 971
FY07Q2 10-31-06 636 0 48 684 1388 0 1899
FY07Q3 1-31-07 347 0 16 363 2202 0 1325
FY07Q4 4-30-07 2111 0 129 2240 1060 0 1916
FY08Q1 7-31-07 1621 0 4 1625 1820 0 2363
FY08Q2 10-31-07 1863 0 29 1892 1338 0 3500
FY08Q3 1-31-08 1662 0 13 1675 1434 0 2394
FY08Q4 4-30-08 751 0 150 901 2196 0 2900
FY09Q1 7-31-08 1193 324 0 1517 4021 980 4628
FY09Q2 10-31-08 983 958 0 1941 6472 1701 6700
FY09Q3 1-31-09 5778 1048 0 6826 4805 3262 5547
FY09Q4 4-30-09 5009 2683 175 7867 3776 3871 4707
FY10Q1 7-31-09 2070 4671 9 6750 5755 2332 6177
FY10Q2 10-31-09 5308 1510 78 6896 4260 3989 5850
FY10Q3 1-31-10 2945 6854 78 9877 3052 76 3861
FY10Q4 4-30-10 2881 1461 78 4420 2406 78 3123
FY11Q1 7-31-10 983 2111 115 3209 3719 47 4692
FY11Q2 10-31-10 3627 966 78 4671 2447 35 3555
FY11Q3 1-31-11 1922 882 79 2883 4300 40 3915
FY11Q4 4-30-11 1970 681 78 2729 5617 0 5284
FY12Q1 7-31-11 5439 0 216 5655 4145 0 4481
FY12Q2 10-31-11 4154 0 78 4232 2012 0 3178
FY12Q3 1-31-12 3203 0 78 3281 2552 0 2722
FY12Q4 4-30-12 1987 0 78 2065 3651 0 3611
FY13Q1 7-31-12 4135 0 116 4251 6056 0 5744
FY13Q2 10-31-12 6061 0 78 6139 6221 0 5426
FY13Q3 1-31-13 6961 0 78 7039 5061 0 4635
FY13Q4 4-30-13 4176 0 78 4254 4171 0 4339
FY14Q1 7-31-13 4581 0 107 4688 4164 0 5679
FY14Q2 10-31-13 7354 0 0 7354 3468 0 4033
FY14Q3 1-31-14 3885 0 0 3885 4329 0 5224
FY14Q4 4-30-14 6474 0 0 6474 5241 0 5530
FY15Q1 7-31-14 5496 0 0 5496 4670 0 5998
FY15Q2 10-31-14 6263 0 37 6300 3612 0 5379
FY15Q3 1-31-15 5677 0 0 5677 5752 0 6148
FY15Q4 4-30-15 9308 0 0 9308 6630 0 6148
FY16Q1 7-31-15 9379 0 292 9671 8291 0 10457
FY16Q2 10-31-15 9523 0 0 9523 9688 0 12554
FY16Q3 1-31-16 6672 0 37 6709 15418 0 15189
FY16Q4 4-30-16 18783 0 0 18783 15418 0 15189
FY17Q1 7-31-16 5609 0 0 5609 21531 0 25274
FY17Q2 10-31-16 23370 0 0 23370 21531 0 25274
FY17Q3 1-31-17 10747 0 0 10747 26367 0 33829
FY17Q4 4-30-17 17904 0 0 17904 28500 0 33099
Totals: 231199 24149 2453 257801 <=since5/1/2006
.
TOTAL REV’s BY YEAR (Avid+Gov’t+Lic):
FY04 4-30-04 3,314 …Avid(CMO)= 3,039 (Avid-Revs don’t incl. Govt-SVCS)
FY05 4-30-05 4,959 …Avid(CMO)= 4,684
FY06 4-30-06 3,193 …Avid(CMO)= 3,005
FY07 4-30-07 3,708 …Avid(CMO)= 3,492
FY08 4-30-08 6,093 …Avid(CMO)= 5,897
FY09 4-30-09 18,151 …Avid(CMO)= 12,963
FY10 4-30-10 27,943 …Avid(CMO)= 13,204
FY11 4-30-11 13,492 …Avid(CMO)= 8,502
FY12 4-30-12 15,233 …Avid(CMO)= 14,783
FY13 4-30-13 21,683 …Avid(CMO)= 21,333
FY14 4-30-14 22,401 …Avid(CMO)= 22,294
FY15 4-30-15 26,781 …Avid(CMO)= 26,744
FY16 4-30-16 44,686 …Avid(CMO)= 44,357
FY17 4-30-17 57,630 …Avid(CMO)= 57,630
...Total Gov’t Revs from 7-2008 inception thru FY11Q1(Apr’11): $24.15mm
.
PPHM’S QTLY. NET LOSS BY QTR:
FY08Q1 7-31-07 4,656,000
FY08Q2 10-31-07 6,207,000
FY08Q3 1-31-08 6,154,000
FY08Q4 4-30-08 6,159,000
FY09Q1 7-31-08 5,086,000
FY09Q2 10-31-08 4,497,000
FY09Q3 1-31-09 3,332,000
FY09Q4 4-30-09 3,609,000
FY10Q1 7-31-09 2,428,000
FY10Q2 10-31-09 2,787,000
FY10Q3 1-31-10 1,538,000
FY10Q4 4-30-10 7,741,000
FY11Q1 7-31-10 7,695,000
FY11Q2 10-31-10 7,513,000
FY11Q3 1-31-11 8,929,000
FY11Q4 4-30-11 10,014,000
FY12Q1 7-31-11 8,092,000
FY12Q2 10-31-11 12,055,000
FY12Q3 1-31-12 11,090,000
FY12Q4 4-30-12 10,882,000
FY13Q1 7-31-12 7,664,000
FY13Q2 10-31-12 8,753,000
FY13Q3 1-31-13 4,914,000
FY13Q4 4-30-13 8,449,000
FY14Q1 7-31-13 7,600,000
FY14Q2 10-31-13 7,790,000
FY14Q3 1-31-14 9,724,000
FY14Q4 4-30-14 10,248,000
FY15Q1 7-31-14 13,129,000
FY15Q2 10-31-14 12,100,000
FY15Q3 1-31-15 12,994,000
FY15Q4 4-30-15 12,135,000
FY16Q1 7-31-15 13,723,000
FY16Q2 10-31-15 13,198,000
FY16Q3 1-31-16 16,847,000
FY16Q4 4-30-16 11,884,000
FY17Q1 7-31-16 11,057,000
FY17Q2 10-31-16 4,056,000
FY17Q3 1-31-17 7,774,000
FY17Q4 4-30-17 5,272,000
= = = = = = = =
OPER. CASH BURNS* BY QTR(FROM THE 10-Q/K’S):
FY10Q1 7-31-09 2,024,000 (from 10Q pg.25)
FY10Q2 10-31-09 2,351,000 (Q1+Q2: 4,375,000 pg.28)
FY10Q3 1-31-10 1,158,000 (Q1+Q2+Q3: 5,533,000 pg.30)
FY10Q4 4-30-10 6,375,000 (FY’10: 11,908,000 10K pg.58)
FY11Q1 7-31-10 6,567,000 (from 10Q pg.24)
FY11Q2 10-31-10 6,167,000 (Q1+Q2: $12,734,000 pg.25)
FY11Q3 1-31-11 7,736,000 (Q1+Q2+Q3: $20,470,000 pg.26)
FY11Q4 4-30-11 8,961,000 (FY’11: 29,431,000 10K pg.54)
FY12Q1 7-31-11 6,984,000 (from 10Q pg.25)
FY12Q2 10-31-11 11,668,000 (Q1+Q2: 18,652,000 pg.25)
FY12Q3 1-31-12 8,490,000 (Q1+Q2+Q3: 27,142,000 pg.25)
FY12Q4 4-30-12 11,265,000 (FY’12: 38,407,000 10K pg.55)
FY13Q1 7-31-12 6,742,000 (from 10Q pg.21)
FY13Q2 10-31-12 6,162,000 (Q1+Q2: 12,904,000 pg.23)
FY13Q3 1-31-13 3,597,000 (Q1+Q2+Q3: 16,501,000 pg.23)
FY13Q4 4-30-13 7,053,000 (FY’13: 23,554,000 10K pg.60)
FY14Q1 7-31-13 5,750,000 (from 10Q pg.23)
FY14Q2 10-31-13 5,834,000 (Q1+Q2: 11,584,000 10Q pg.24)
FY14Q3 1-31-14 7,875,000 (Q1+Q2+Q3: 19,459,000 10Q pg.26)
FY14Q4 4-30-14 8,706,000 (FY’14: 28,165,000 10K pg.55)
FY15Q1 7-31-14 11,076,000 (from 10Q pg.23)
FY15Q2 10-31-14 9,947,000 (Q1+Q2: 21,023,000 10Q pg.25)
FY15Q3 1-31-15 11,116,000 (Q1+Q2+Q3: 32,139,000 10Q pg.26)
FY15Q4 4-30-15 10,474,000 (FY’15: 42,613,000 10K pg.54)
FY16Q1 7-31-15 12,306,000 (from 10Q pg.25)
FY16Q2 10-31-15 11,701,000 (Q1+Q2: 24,007,000 10Q pg.26)
FY16Q3 1-31-16 15,086,000 (Q1+Q2+Q3: 39,093,000 10Q pg.27)
FY16Q4 4-30-16 10,112,000 (FY'16: 49,205,000 10K pg.39)
FY17Q1 7-31-16 9,607,000 (from 10Q pg.22)
FY17Q2 10-31-16 2,565,000 (Q1+Q2: 12,172,000 10Q pg.24)
FY17Q3 1-31-17 6,274,000 (Q1+Q2+Q3: 18,446,000 10Q pg.24)
FY17Q4 4-30-17 3,886,000 (FY'17: 22,332,000 10K pg.40)
FY’09 total Op-Burn: $14,715,000
FY’10 total Op-Burn: $11,908,000
FY’11 total Op-Burn: $29,431,000
FY’12 total Op-Burn: $38,407,000
FY’13 total Op-Burn: $23,554,000
FY’14 total Op-Burn: $28,165,000
FY’15 total Op-Burn: $42,613,000
FY’16 total Op-Burn: $49,205,000
FY’17 total Op-Burn: $22,332,000
Period Halozyme Cust-A Other-Custs
FYE 4-30-14 91% 1% 8%
FYE 4-30-15 79% 12% 9%
FYE 4-30-16 69% 26% 5%
Q/E 7-31-16 65% 29% 6%
Q/E 10-31-16 77% 10% 13%
Q/E 1-31-17 29% 56% 15%
FYE 4-30-17 58% 26% 16%
7-14-17/13D: Group Ronin/SWPartners add 34,891/Common & 3,600/Pref. Jun29-Jul7
That brings total ownership of the Group to:
3,801,139 COMMON, 8.4% of 45,069,188 common O/S at 7-10-17.
127,099 PREFERRED, 7.7% of 1,647,760 preferred O/S at 1-31-17
...Total “beneficial”=3,952,446 (3,310,651 + 641,795) if Preferred conv. to Common (x1.19), 8.8% of 45,069,188 O/S.
NOTE: ALL historical Common #’s adjusted for the 1:7 R/S eff. 7-10-17.
13D TransDates COMMON-CHG PREF-CHG ENDING-COMMON ENDING-PREF.
3-2-17 1/20/17-3/1/17 +2,947,425 +51,364 2,947,425 51,364 http://tinyurl.com/jr42u23
3-10-17 3/2/17-3/9/17 +433,509 +25,661 3,380,934 77,025 http://tinyurl.com/ydxra96u
4-17-17 3/28/17-4/10/17 0 +23,334 3,380,934 100,359 http://tinyurl.com/lanjddc
5-19-17 5/1/17-5/17/17 0 +23,140 3,380,934 123,499 http://tinyurl.com/mgnn92x
6-20-17 3/10/17-6/16/17 +378,170 0 3,759,105 123,499 http://tinyurl.com/y76q5rqu
6-29-17 6/21/17 +7,143 0 3,766,248 123,499 http://tinyurl.com/y9sp8bfv
7-14-17 6/29/17-7/7/17 +34,891 +3,600 3,801,139 127,099 http://tinyurl.com/ybra4s69
SPLITOUT 13D GROUP into Ronin(John Stafford III) and SW-Partners(Stephen White):
Ronin Trading (Stafford) 3,173,391 115,299
SW-Partners (White) 627,748 11,800
O/S Shares & ATM Sales History (’06–curr.)
Shares O/S as of 7-10-17: 45,069,188
**NOTES:
1. PPHM shares were 1:7 Reverse Split eff. 7-10-17 (~315mm/$.606=>~45mm/$4.24) http://tinyurl.com/ycohqn6j
2. Per the 4-30-10 10-K, ALL ATM Agreements have been fully used – none remain. http://tinyurl.com/ycxu4l5n (F-20 & F-28)
PPHM - O/S Shares History (’06–curr.)
4-30-06 35,876,438
1-31-07 39,222,440
4-30-07 39,222,440
7-6-07 45,233,123
7-31-07 45,242,123
10-31-07 45,242,123
1-31-08 45,242,123
4-30-08 45,242,123
7-31-08 45,242,123
10-31-08 45,242,123
1-31-09 45,242,123
4-30-09 45,537,711
7-10-09 47,392,883
7-31-09 47,393,783
**NOTE: PPHM shares were 1:5 Reverse Split eff. 10-19-09 (~237mm/$.64=>~47.4mm/$3.20) http://tinyurl.com/ykuw588
10-31-09 48,869,563 +1,475,780
1-31-10 50,903,404 +2,033,841
4-30-10 53,094,894 +2,191,490
6-21-10 54,388,917 +1,294,023 (6-22-10 ATM/mlv Form424)
7-9-10 55,069,449 +475,987 (4-30-10 10K iss. 7-14-10)
7-31-10 55,784,955 +715,506
10-31-10 59,220,742 +3,435,787
11-30-10 63,932,353 +4,711,611 (10-31-10 10Q iss. 12-9-10)
12-15-10 64,404,097 +471,744 (12-17-10 S-3: $75M Shelf Reg.)
1-31-11 66,813,419 +2,409,322
2-28-11 67,885,811 +1,072,392 (1-31-11 10Q iss. 3-11-11)
4-30-11 69,837,142 +1,951,331
7-8-11 71,069,858 +1,232,716 (4-30-11 10K iss. 7-14-11)
8-22-11 72,704,647 +1,634,789 (Proxy iss. 8-26-11)
8-31-11 73,284,016 +579,369 (424B5 iss. 9-2-11)
9-8-11 79,536,268 +6,252,252 (Roth Sale to 3 Inst’s @ $1.11/sh.)
10-31-11 82,638,201 +3,101,933
12-9-11 86,788,817 +4,150,616 (10-31-11 10Q iss. 12-12-11)
1-31-12 93,146,226 +6,357,409
2-29-12 98,873,172 +5,726,946 (1-31-12 10Q iss. 3-9-12)
4-30-12 101,421,365 +2,548,193
7-13-12 104,174,056 +2,752,691 (4-30-12 10K iss. 7-16-12)
7-31-12 104,178,431 +4,375 (7-31-12 10Q iss. 9-10-12)
8-16-12 104,191,176 +12,745 (prelim. proxy 14A http://tinyurl.com/c48bvof )
9-7-12 104,191,176 nochg (7-31-12 10Q iss. 9-10-12)
10-31-12 123,310,188 +19,119,012
12-7-12 132,539,783 +9,229,595 (10-31-12 10Q iss. 12-10-12)
1-31-13 133,770,614 +1,230,831
3-12-13 137,110,758 +3,340,144 (1-31-13 10Q iss. 3-12-13)
4-30-13 143,768,946 +6,658,188 (4-30-13 10K iss. 7-11-13)
7-5-13 151,602,765 +7,833,819 (4-30-13 10K iss. 7-11-13)
7-31-13 153,506,811 +1,904,046
9-5-13 156,461,114 +2,954,303 (7-31-13 10K iss. 9-9-13)
10-31-13 160,248,742 +3,781,628
12-6-13 160,325,891 +77,149 (10-31-13 10K iss. 12-10-13)
1-31-14 176,453,261 +16,127,370
3-3-14 176,481,054 +27,793 (1-31-14 10Q iss. 3-7-14)
4-30-14: 178,871,164 +2,390,110
7-7-14: 179,209,458 +338,294 (4-30-14 10-K cover page, iss. 7-14-14)
7-31-14: 179,216,032 +6,574 (7-31-14 10Q iss. 9-9-14)
8-22-14: 179,226,424 +10,392 (8-28-14 Proxy/Def14A)
9-5-14: 179,505,424 +279,000 (7-31-14 10Q iss. 9-9-14)
10-31-14: 182,000,583 +2,495,159 (10-31-14 10Q iss. 12-10-14)
12-5-14: 182,081,234 +80,651 (10-31-14 10Q cover pg., iss. 12-10-14)
12-19-14: 182,081,234 -0- (12-23-14 S-3)
1-31-15: 184,244,698 +2,163,464 (1-31-15 10Q iss. 3-12-15)
3-12-15: 188,332,872 +4,088,174 (“ “ “)
4-30-15: 193,346,627 +5,013,755 (4-30-15 10-K iss. 7-14-15)
7-10-15: 199,934,918 +6,588,291 (4-30-15 10-K/cover-pg, iss. 7-14-15)
7-31-15: 200,983,948 +1,049,030 (7-31-15 10Q iss. 9-9-15)
9-4-15: 202,124,031 +1,140,083 (“ “ “)
10-31-15: 225,824,551 +23,700,520 (10-31-15 10Q iss. 12-10-15)<=Incl. 18.5mm sh. Dart/EastCAP @1.08
12-9-15: 229,701,808 +3,877,257 (10-31-15 10Q iss. 12-10-15)
1-31-16: 232,231,242 +2,529,434 (1-31-16 10Q iss. 3-9-16)
3-8-16: 233,738,426 +1,507,184 (“ “ “)
4-30-16: 236,930,485 +3,192,059 (4-30-16 10K iss. 7-14-16)
7-11-16: 241,456,721 +4,526,236 (“ “ “)
7-31-16: 241,456,721 -0- (7-31-16 10Q iss. 9-8-16)
9-2-16: 242,381,850 +925,129 (“ “ “)
10-31-16: 251,765,279 +9,383,429 (10-31-16 10Q iss. 12-12-16)
12-8-16: 257,141,534 +5,376,255 (“ “ “)
1-31-17: 271,068,464 +13,926,930 (1-31-17 10Q iss. 3-13-17)
3-10-17: 297,709,478 +26,641,014 (“ “ “)
4-30-17: 44,014,040(x7)=308,098,280 +10,388,802 (4-30-17 10K iss. 7-14-17)
7-10-17: 1:7 R/S (~315mm/$.606=>~45mm/$4.24) http://tinyurl.com/ycohqn6j
7-10-17: 45,069,188 +1,055,148 (“ “ “)
= = = = = = = = = = = = = = = = = =
ATM Sales Summary (3/2009–3/13/2017). Also, PPHM O/S Shares History Table (’06–curr.) at the bottom of this post. At 7-10-17, common shares O/S = 45,069,188sh. (following 1:7 R/S eff. 7-10-17)
ATM = “At-The-Market Sales Issuance”
**NOTE: Per the 4-30-10 10-K, ALL ATM Agreements have been fully used – none remain. http://tinyurl.com/ycxu4l5n (F-20 & F-28)
I. WM-SMITH 3-2009:
• $7.5mm ATM/Wm.SMITH 3-26-09: $7,500,000gr. / 2,150,759sh. = $3.49/sh. (commiss: 3%)
• $25mm ATM/Wm.SMITH 7-14-09: $25,000,000gr. / 7,569,314sh. = $3.30/sh. (commiss: 3%/1st$15mm, then 2%)
*Total Raised via WmSmith ATM Sales thru 7-31-10:
. . .Before 7-10-17 1:7RS: $32,500,000gr. / 9,720,073sh. = $3.34/sh.
. . .After 7-10-17 1:7RS: $32,500,000gr. / 1,388,582sh. = $23.41/sh.
II. MLV 6-2010: http://www.mlvco.com
$15mm ATM/MLV 6-22-10 (commiss: 2%) Form424: http://tinyurl.com/24txkxb
• Sold 6/22/10–10/31/10: $6,840,000gr. / 4,031,018sh. = $1.70/sh.
• Sold 11/1/10–11/30/10: $7,407,000gr. / 4,711,611sh. = $1.57/sh.
• Sold 12/1/10–1/31/11: $753,000gr. / 471,744sh. = $1.60/sh.
*Total Raised via MLV June’10 ATM Sales thru 1-31-11:
. . .Before 7-10-17 1:7RS: $15,000,000gr. / 9,214,373 = $1.63/sh.
. . .After 7-10-17 1:7RS: $15,000,000gr. / 1,316,339 = $11.40/sh.
III. MLV 12-2010: “Dec’10 AMI Agreement” http://www.mlvco.com
$75mm ATM/MLV 12-29-10 (commiss: max=5%) Form8K: http://tinyurl.com/2a6w76g
(pursuant to $75mm S-3 Shelf Reg. filed 12-17-10: http://tinyurl.com/2469b2d )
• Sold 12/29/10-1/31/11: $6,460,000gr. / 2,385,862sh. = $2.71/sh.
• Sold 2/1/11-2/28/11: $2,358,000gr. / 998,142sh. = $2.36/sh.
• Sold 3/1/11-4/30/11: $4,470,000gr. / 1,840,487sh. = $2.43/sh.
• Sold 5/1/11-7/31/11: $3,713,000gr. / 1,912,576sh. = $1.94/sh.
• Sold 8/1/11-10/31/11: $5,582,000gr. / 4,727,840sh. = $1.18/sh.
• Sold 9-2-12 Roth Direct: $6,940,000gr./ 6,252,252sh. = $1.11/sh.
• Sold 11/1/11-1/31/12: $10,961,000gr. / 10,308,025sh. = $1.06/sh.
• Sold 2/1/12-2/29/12: $5,871,000gr. / 5,726,946sh. = $1.03/sh.
• Sold 3/1/12-4/30/12: $1,263,000gr. / 2,198,543sh. = $.57/sh.
• Sold 5/1/12-6/30/12: $1,496,000gr. / 2,752,691sh. = $.54/sh.
• Sold 7/1/12-9/26/12: none**
• Sold 9/27/12-10/31/12: $16,719,000gr./ 18,557,928 = $.90/sh.
• Sold 11/1/12-11/30/12: $7,296,000gr./ 9,220,313 = $.79
• Sold 12/1/12-1/31/13: $1,540,000gr./ 1,131,282 = $1.36
• Sold 2/1/13-3/12/13: $330,000gr./ 201,154 = $1.64
*Total Raised via MLV Dec’10 ATM Sales thru 3-12-2013:
. . .Before 7-10-17 1:7RS: $75,000,000gr. / 68,214,041 = $1.10sh.
. . .After 7-10-17 1:7RS: $75,000,000gr. / 9,744,863 = $7.70sh.
IV. MLV 12-2012: “Dec’12 AMI Agreement” http://www.mlvco.com
$75mm ATM/MLV 12-29-12 (commiss: max=5%) Form8K: http://tinyurl.com/2a6w76g
(pursuant to $75mm S-3 Shelf Reg. filed 3-9-12: http://tinyurl.com/7dl7pjm )
• Sold 2/1/13-3/12/13: $4,475,000gr. / 3,132,402sh. = $1.43/sh.
• Sold 3/13/13-4/30/13: $8,897,000gr. / 6,188,273sh. = $1.44/sh.
• Sold 5/1/13-7/11/13: $12,729,000gr. / 7,927,016sh. = $1.61/sh.
• Sold 7/12/13-7/31/13: $2,468,000gr. / 1,690,864sh. = $1.46/sh.
• Sold 8/1/13-9/9/13: $4,372,000gr. / 3,057,431sh. = $1.43/sh.
• Sold 9/10/13-10/31/13: $4,708,000gr. / 3,262,958sh. = $1.44/sh.
• Sold 11/1/13-12/6/13: NONE – see 10Q note below.
• Sold 12/7/13-1/31/14: $28,130,000gr. / 16,045,717sh. = $1.75/sh.
• Sold 2/1/14-4/30/14: $3,017,000gr. / 1,543,383sh. = $1.95/sh.
• Sold 5/1/14-7/31/14: $425,000gr. / 226,700sh. = $1.92/sh.
• Sold 8/1/14-10/31/14: $3,891,000gr. / 2,494,835sh. = $1.56/sh.
• Sold 11/1/14-1/31/15: $1,878,000gr. / 1,261,825sh. = $1.49/sh.
*Total Raised via MLV Dec’12 ATM Sales thru 10-31-2014:
. . .Before 7-10-17 1:7RS: $75,000,000gr. / 46,831,404 = $1.60sh.
. . .After 7-10-17 1:7RS: $75,000,000gr. / 6,690,200 = $1.21sh.
V. MLV 6-2014: “Jun’14 AMI Agreement – up to $25mm” http://www.mlvco.com
• Sold 11/1/14-1/31/15: $1,193,000gr. / 869,504sh. = $1.43/sh.
• Sold 2/1/15-3/12/15: $6,204,000gr. / 4,354,954sh. = $1.44/sh.
• Sold 3/13/15-4/30/15: $6,147,000gr. / 4,457,299sh. = $1.38/sh.
• Sold 5/1/15-7/14/15: $8,896,000gr. / 6,534,400sh. = $1.36/sh.
• Sold 7/15/15-7/31/15: $1,270,000gr. / 1,003,830sh. = $1.27/sh.
• Sold 8/1/15-9/9/15: $1,290,000gr. / 1,091,508sh. = $1.18/sh.
*Total Raised via MLV Jun’14 ATM Sales thru 9-9-2015:
. . .Before 7-10-17 1:7RS: $25,000,000gr. / 18,311,495 = $1.37/sh.
. . .After 7-10-17 1:7RS: $25,000,000gr. / 2,615,928 = $9.56/sh.
VI. MLV 8-2015: “Aug’15 AMI Agreement – up to $30mm” http://www.mlvco.com
• Sold 8/7/15-9/9/15: $892,000gr. / 1,091,000sh. = $1.18/sh.
• Sold 9/10/15-10/31/15: $4,294,000gr. / 4,059,478sh. = $1.06/sh.
• Sold 11/1/15-12/10/15: $2,261,000gr. / 1,939,413sh. = $1.17/sh.
• Sold 12/11/15-1/31/16: *NO ATM SALES*, per 1-31-16 10Q iss. 3-9-16
• Sold 2/2/16-4/30/16: *NO ATM SALES*, per 4-30-16 10Q iss. 7-14-16
• Sold 5/1/16-7/14/16: $937,000gr. / 1,876,918sh. = $.50/sh.
• Sold 7/15/16-9/8/16: *NO ATM SALES*, per 7-31-16 10Q iss. 9-8-16
• Sold 9/9/16-10/31/16: $2,513,000gr. / 6,432,439sh. = $.39/sh.
• Sold 11/1/16-12/12/16: $1,174,000gr. / 3,788,346sh. = $.31/sh.
• Sold 12/13/16-1/31/17: $3,404,000gr. / 11,069,646sh. = $.31/sh.
• Sold 2/1/17-3/13/17: $7,335,000gr. / 15,308,300sh. = $.48/sh.
• Sold 3/14/17-4/30/17: $2,883,000gr. / 4,486,182 = $.64/sh.
• Sold 5/1/17-6/30/17: $4,307,000gr. / 7,358,806 = $0.59/sh.
. . .Before 7-10-17 1:7RS: $30,000,000gr. / 57,072,288 = $.53/sh.
. . .After 7-10-17 1:7RS: $30,000,000gr. / 8,153,184 = $3.68/sh.
VII. NOBLE 8-2015: “Aug’15 Eq.Dist. Agreement – up to $20mm” http://www.noblelsp.com
• Sold 11/1/15-12/10/16: $2,371,000gr. / 1,925,844sh. = $1.23/sh.
• Sold 12/11/15-1/31/16: $2,857,000gr. / 2,529,434sh. = $1.13/sh.
• Sold 2/1/16-4/30/16: $1,741,000gr. / 4,017,010sh. = $.43/sh.
• Sold 5/1/16-7/14/16: $1,233,000gr. / 2,649,318sh. = $.47/sh.
• Sold 7/15/16-9/8/16: *NO ATM SALES*, per 7-31-16 10Q iss. 9-8-16
• Sold 9/9/16-10/31/16: $1,283,000gr. / 2,988,086sh. = $.43/sh.
• Sold 11/1/16-12/12/16: $496,000gr. / 1,587,909sh. = $.31/sh.
• Sold 12/13/16-1/31/17: $904,000gr. / 2,857,284sh. = $.32/sh.
• Sold 2/1/17-3/13/17: $6,006,000gr. / 11,273,296sh. = $.53/sh.
• Sold 3/14/17-4/30/17: $3,109,000gr. / 4,896,376sh. = $.63/sh.
. . .Before 7-10-17 1:7RS:$20,000,000gr. / 34,724,557 = $.58/sh.
. . .After 7-10-17 1:7RS:$20,000,000gr. / 4,960,651 = $4.03/sh.
TOTAL ALL A-T-M SALES – INCEPTION (3-2009) THRU 6-30-2017:
. . .Before 7-10-17 1:7RS: => $272,500,000gr. / 244,088,231sh. = $1.12/sh.
. . .After 7-10-17 1:7RS: => $272,500,000gr. / 34,869,747sh. = $7.81/sh.
= = = = = = = = = = = = = = = = = =
O/S WARRANTS & STOCK-OPTIONS A/O 4-30-2017 (10K pg.F23,25):
• WARRANTS: As of 4-30-17, warrants to purchase 39,040 shares at an exercise price of $17.29 were outstanding and are exercisable through Aug30 2018. These warrants were issued in FY’13 in connection with the Aug.2012 [Oxford] loan agreement, which was paid in full Sept.2012.
• STOCK OPTIONS OUTSTANDING A/O 4-30-17: 4,081,548 shares at a wgt.avg. exercise price of $8.77.
- - - - - - - - - - - - - - - - -
Latest 10K 4-30-17 iss. 7-14-17 http://tinyurl.com/ycxu4l5n PR: http://ir.peregrineinc.com/releasedetail.cfm?ReleaseID=1033237 (Cash 4-30-17=$46.8mm)
Latest 10Q 1-31-17 iss. 3-13-17 http://tinyurl.com/zlozrv4 PR: http://ir.peregrineinc.com/releasedetail.cfm?ReleaseID=1017192 (Cash 1-31-17=$41.5mm)
ALL SEC filings for PPHM: http://tinyurl.com/6d4jw8
= = = = = = = = = = =PREFERRED STOCK:
...2-5-14: PPHM Announces Public Offering of 10.5% Series E Convertible Preferred Stock http://tinyurl.com/lkxsna6
...2-11-14: PPHM Raises net $16.2mm selling 700k Preferred Shares with 10.5% div. at $25/sh, convertible to common at $3/sh http://tinyurl.com/jwmsnsk (8-K)
...6/14/14-7/14/13: PPHM Raises net $9.5mm selling 400k Preferred Shares with 10.5% div. at $25/sh, convertible to common at $3/sh http://tinyurl.com/mhva3k3 (4-30-14 10-K pg. F-34; $20mm gross remaining)
PREFERRED SHARES:
4-30-15 10K/p.F26: On 6-13-14, we entered into an At Market Issuance Sales Agreement with MLV, pursuant to which we may issue & sell shares of our Series E Preferred Stock through MLV, as agent, for gross proceeds of up to $30,000,000, in registered transactions from our Jan2014 Shelf. During FY’2015, we sold 799,764 shares of our Series E Preferred Stock at mkt prices for gross $19,205,000 before deducting commissions/costs of $1,002,000. As of 4-30-15, gross proceeds of up to $10,795,000 remained available under the Series E AMI Agreement.
NOTE: One of the holders of Preferred is KENNETH DART (EASTERN CAPITAL), who in a 2-13-15 SG14G ( http://tinyurl.com/k4nsfuu ) reported their 240,000sh. Preferred holdings as 2,000,000 common shares (SHARED VOTING POWER: 9,921,760,% OF CLASS REPRESENTED: 5.4%).
“which has a liquidation preference of $25.00/sh. and a conversion price of $3.00/sh.” 2,000,000 / 240,000 = 8.333
10Q: http://tinyurl.com/j2u2bjk
**7-31-16 10Q(iss. 9-8-16): 8/1/16-9/8/16, we sold 68,910sh. for gross=$1,601,000; a/o 9-8-16, $9,134,000 remained available.
EasternCap/K.Dart & Ronin-SWIM(Stafford-White) Ownership: #1 & #2
8-2016: One 5% Stockholder: Eastern Capital Limited (Kenneth Dart), Grand Cayman Islands
...from Form14A ASM Proxy http://tinyurl.com/ybu8nr7l
Owned:
3,777,183 COMMON - 8.4% of ~45,000,000 common O/S at 7-7-17
440,000 PREFERRED - 26.7% of 1,647,760 preferred O/S at 1-31-17
(total beneficial=4,300,992 if Preferred conv. to Common, 9.6% of 45mm)
- - - - - - - -
8-2016: “The info. set forth herein is based solely on a Schd. 13G/A filed 11-2-15 by Eastern Capital Ltd. The number of beneficial shares owned (30,106,945sh.) includes 3,666,667 shares of common stock that could be acquired upon conversion of the 440,000 shares of Series E Preferred Stock held by Eastern Capital.”
11-2-15/E.Cap 13G & Form3: http://tinyurl.com/pexs62e & http://tinyurl.com/ns2s9p6
= = = = = = = = = = = = = = = = = = = =
6-29-17/13D: Ronin Trading(John Stafford III) & SWPartners(Stephen White), a 13D “Group”, adds 7,143 COMMON on June 21, 2017.
That brings total ownership of the Group to:
3,766,248 COMMON, 8.4% of ~45,000,000 common O/S at 7-7-17.
123,499 PREFERRED, 7.5% of 1,647,760 preferred O/S at 1-31-17 (convertible to 147,023 Common @8.333/7).
(total beneficial=3,913,271 if Preferred conv. to Common, 8.7% of 45mm)
>NOTE: ALL historical Common #’s adjusted for the 1:7 R/S eff. 7-10-17.
13D TransDates COMMON-CHG PREF-CHG ENDING-COMMON ENDING-PREF.
3-2-17 1/20/17-3/1/17 +2,947,425 +51,364 2,947,425 51,364 http://tinyurl.com/jr42u23
3-10-17 3/2/17-3/9/17 +433,509 +25,661 3,380,934 77,025 http://tinyurl.com/ydxra96u
4-17-17 3/28/17-4/10/17 0 +23,334 3,380,934 100,359 http://tinyurl.com/lanjddc
5-19-17 5/1/17-5/17/17 0 +23,140 3,380,934 123,499 http://tinyurl.com/mgnn92x
6-20-17 3/10/17-6/16/17 +378,170 0 3,759,105 123,499 http://tinyurl.com/y76q5rqu
6-29-17 6/21/17 +7,143 0 3,766,248 123,499 http://tinyurl.com/y9sp8bfv
SPLITOUT 13D GROUP into Ronin(John Stafford III) and SW-Partners(Stephen White):
(since the COMMON 13D filed on 3-10-17)
Ronin Trading (Stafford) +1,997,194 +39,974 22,213,734 111,699
SW-Partners (White) +700,000 +6,500 4,150,000 11,800
Thx BB762: A.Schroit’s 6-22-17 Nature/BJOC Exosomes testing Betabodies
PPHM’S EXOSOMES DIAG. PLATFORM (incl. 6-22-2017 NATURE/BJC article)
...Known POC Data Summary (see below):
1-22-17/OncoTarget: Ovarian – senior author Alan Schroit (UTSW/PPHM SAB)
6-22-17/Nature(BJC): Breast & Pancreatic – senior author Alan Schroit (UTSW/PPHM SAB) *Using Betabody KL15C*
PPHM’s PS+ EXOSOME-BASED CANCER DETECTION & MONITORING TECHNOLOGY ("Liquid Biopsy")
...Excellent Exosome (aka microparticles, microvesicles) info: http://www.exosome-rna.com
6-22-17: Nature (British Jrnl of Cancer): ”Detection of Phosphatidylserine-Positive Exosomes for the Diagnosis of Early-Stage Malignancies [Using Betabody KL15C]”
Raghava Sharma, Xianming Huang, Rolf A Brekken (PPHM SAB), Alan J Schroit
….Dr. Schroit: PPHM SAB, UTSW profile: http://tinyurl.com/yaqnvcvq ; pubs: http://tinyurl.com/lqbd8kt
https://www.nature.com/bjc/journal/vaop/ncurrent/pdf/bjc2017183a.pdf
ABSTRACT
Background: There has been increasing interest in the detection of tumor exosomes in blood for cancer diagnostics. Most studies have focused on miRNA and protein signatures that are surrogate markers for specific tumor types. Because tumor cells and tumor-derived exosomes display phosphatidylserine (PS) in their outer membrane leaflet, we developed a highly sensitive ELISA-based system that detects picogram amounts of exosomal phospholipid in plasma as a cancer biomarker.
Methods: This report describes the development of a highly specific and sensitive ELISA for the capture of PS-expressing tumor exosomes in the blood of tumor-bearing mice. To monitor the relationship between tumor burden & tumor exosome plasma concentrations, plasma from one transplantable breast cancer model (MDA-MB-231) and 3 genetic mouse models (MMTV-PyMT; breast and KIC and KPC; pancreatic) were screened for captured exosomal phospholipid.
Results: We show that quantitative assessment of PS-expressing tumor exosomes detected very early-stage malignancies before clinical evidence of disease in all 4 model systems. tumor exosome levels showed significant increases by day 7 after tumor implantation in the MDA-MB-231 model while palpable tumors appeared only after day 27. For the MMTV-PyMT and KIC models, tumor exosome levels increased significantly by day 49 (P<=0.0002) and day 21 (P<=0.001) while tumors developed only after days 60 & 40, respectively. For the KPC model, a significant increase in blood exosome levels was detected by day 70 (P=0.023) when only preinvasive lesions are microscopically detectable.
Conclusions: These data indicate that blood PS exosome levels is a specific indicator of cancer and suggest that blood PS is a biomarker for early-stage malignancies.
- - - - -
Addl. article info (Fig.1) extracted by Bamboozler762 6-23-17 iHub #300359 http://investorshub.advfn.com/boards/read_msg.aspx?message_id=132436726
Fig1: Schematic diagram of betabody, KL15C
- - - - - - - - - -
Note: BETABODIES (Clipped/Nicked B2GPI - ex: KL15, “2nd-gen. PS-Targeting”) - bind to PS directly, are smaller in size (100 vs. 250KDa) and have a longer serum half-life (~5days) than natural antibodies (Bavi=~1day) – see http://tinyurl.com/khopa3d …2/17/15: UTSW/PPHM’s BetaBodies patent#8,956,616 Awarded(Granted) http://tinyurl.com/p75uyfu

Fig.1: Schematic diagram of betabody, KL15C
Domains 1 & 5 of the plasma PS-binding protein, B2GP1, were genetically fused to the C-terminus of the CH3 domains of the Fc fragment of human IgG1. A Gly4Ser linker was inserted between the CH3 domains and domains 1 and 5 of B2GP1. The recombinant betabody was expressed as a dimer.
- - - - - - - - - -
...Glimpse of beg. of 7pg. 6-22-17 the Schroit/Brekken/etal BJC article: http://www.readcube.com/articles/10.1038/bjc.2017.183

7-13-17: Ronin/SW-Invest Letter to Stockholders; PPHM Comments
7-13-17: Ronin & SW-Invest Issue Letter to Stockholders of Peregrine Pharmaceuticals
* Believe Change is Desperately Needed to Peregrine's Board Given Current Strategy, Poor Corporate Governance, Apparent Misalignment of Interests with Stockholders, and Constant Dilution
* Announces Nomination of Gregory P. Sargen, Brian W. Scanlan, and Saiid Zarrabian for Election at Upcoming 2017 Annual Meeting
CHICAGO, July 13, 2017, PR Newswire:
Ronin Trading, LLC [John Stafford III] and SW Investment Mgt. LLC [Stephen White] (together with the other participants in their solicitation, "Ronin"), collectively the 2nd largest stockholder of Peregrine Pharmaceuticals, Inc. (NASDAQ: PPHM), with aggregate beneficial ownership of approx. 8.8% of the Company's outstanding shares of common stock, today issued a letter to Peregrine's stockholders.
In the letter, Ronin announced that it has formally nominated 3 independent, highly-qualified candidates, Gregory P. Sargen, Brian W. Scanlan, and Saiid Zarrabian, for election to the Company's Board of Directors at the Company's upcoming 2017 annual meeting of stockholders [~Oct. 12, 2017].
As explained in the letter, Ronin believes that there are opportunities to increase stockholder value; however, Ronin is concerned that stockholders will continue to suffer unless the Board is reconstituted with directors who will represent stockholders' best interests. The full text of the letter follows. . .
FULL LETTER w/Charts: http://tinyurl.com/y96wtrdb (PRNewswire)
7-13-17/Schd14A Proxy: http://tinyurl.com/y7tx3mv3 (14A)
= = = = = = = = = = = = = = = =
7-13-17: Peregrine Pharmaceuticals Issues Statement Regarding Ronin Trading & SW Investment Management's Letter to Stockholders
http://ir.peregrineinc.com/releasedetail.cfm?ReleaseID=1033161
TUSTIN, July 13, 2017: Peregrine Pharmaceuticals, Inc. (NASDAQ:PPHM) today confirmed that Ronin Trading, LLC and SW Investment Management LLC (together, "Ronin/SWIM") submitted 3 candidates for election to the Peregrine Board of Directors at the Company's Annual Meeting of Stockholders. The Company issued the following statement on Ronin/SWIM's nomination of directors and its letter to the Peregrine stockholders:
"Peregrine welcomes the input of our stockholders and is committed to maintaining a highly qualified Board to lead the company forward. We respect the right of stockholders to nominate directors and our Board follows a defined process to evaluate any potential nominees. Our Nominating Committee will carefully evaluate Ronin/SWIM's nominees consistent with that process. Following the Committee's review, it will make a recommendation to the Board that is in the best interests of Peregrine and all of our stockholders. Stockholders need take no action at this time.
"We are actively engaged in an ongoing dialogue with our stockholders and welcome their constructive input on how we can further strengthen the company. We have engaged in various discussions with representatives of Ronin Trading and SW Investment Management and welcome a constructive dialogue toward enhancing value.
"Peregrine's Board, which has a deep understanding of the Company's R&D and CDMO business lines, has been actively involved in setting and overseeing a strategy that has delivered meaningful growth over the past five years in our CDMO business and promising progress in our R&D efforts while substantially reducing R&D spend.
"Our Board is committed to maximizing value for all stockholders. Peregrine will continue to evaluate stockholder input, assess opportunities and make decisions to achieve this objective."
ABOUT PEREGRINE PHARMACEUTICALS
Peregrine Pharmaceuticals, Inc. is a biopharmaceutical company committed to improving the lives of patients by delivering high quality pharmaceutical products through its contract development and manufacturing organization (CDMO) services and through advancing and licensing its investigational immunotherapy and related products. Peregrine's in-house CDMO services, including cGMP manufacturing and development capabilities, are provided through its wholly-owned subsidiary Avid Bioservices, Inc. (http://www.avidbio.com ), which provides development and biomanufacturing services for both Peregrine and third-party customers. The company is also working to evaluate its lead immunotherapy candidate, bavituximab, in combination with immune stimulating therapies for the treatment of various cancers, and developing its proprietary exosome technology for the detection and monitoring of cancer. For more information, please visit http://www.peregrineinc.com .
MEDIA CONTACTS:
John Christiansen / Matt Reid, Sard Verbinnen & Co, (415) 618-8750 / (310) 201-2040
INVESTOR CONTACTS:
Stephanie Diaz, Vida Strategic Partners, (415) 675-7401
= = = = = = = = = = = =
6-29-17/13D: Ronin Capital+SWInvest (John Stafford III+Stephen White) add 2,697,193 shs. COMMON during Mar10-June21.
...Their 13D 'Group' stake now 8.9% of COMMON (26,363,734sh.) & 7.5% of PREFERRED (123,499sh.) - see: http://tinyurl.com/ycqsps62
...NOTES: As a Group, ie, “people that share the furtherance of a common objective/concerted action”; 13D’s are reserved for ACTIVE INVESTORS who may be “interested in agitating for some kind of a change at the company”.
7-13-17: Ronin & SW-Invest Issue Letter to Stockholders of Peregrine Pharmaceuticals
* Believe Change is Desperately Needed to Peregrine's Board Given Current Strategy, Poor Corporate Governance, Apparent Misalignment of Interests with Stockholders, and Constant Dilution
* Announces Nomination of Gregory P. Sargen, Brian W. Scanlan, and Saiid Zarrabian for Election at Upcoming 2017 Annual Meeting
CHICAGO, July 13, 2017, PR Newswire:
Ronin Trading, LLC [John Stafford III] and SW Investment Mgt. LLC [Stephen White] (together with the other participants in their solicitation, "Ronin"), collectively the 2nd largest stockholder of Peregrine Pharmaceuticals, Inc. (NASDAQ: PPHM), with aggregate beneficial ownership of approx. 8.8% of the Company's outstanding shares of common stock, today issued a letter to Peregrine's stockholders.
In the letter, Ronin announced that it has formally nominated 3 independent, highly-qualified candidates, Gregory P. Sargen, Brian W. Scanlan, and Saiid Zarrabian, for election to the Company's Board of Directors at the Company's upcoming 2017 annual meeting of stockholders [~Oct. 12, 2017].
As explained in the letter, Ronin believes that there are opportunities to increase stockholder value; however, Ronin is concerned that stockholders will continue to suffer unless the Board is reconstituted with directors who will represent stockholders' best interests. The full text of the letter follows. . .
FULL LETTER w/Charts: http://tinyurl.com/y96wtrdb (PRNewswire)
= = = = = = = = = = = =
6-29-17/13D: Ronin Capital+SWInvest (John Stafford III+Stephen White) add 2,697,193 shs. COMMON during Mar10-June21.
...Their 13D 'Group' stake now 8.9% of COMMON (26,363,734sh.) & 7.5% of PREFERRED (123,499sh.) - see: http://tinyurl.com/ycqsps62
...NOTES: As a Group, ie, “people that share the furtherance of a common objective/concerted action”; 13D’s are reserved for ACTIVE INVESTORS who may be “interested in agitating for some kind of a change at the company”.
TDAmeritrade didn't charge me a fee - very strange. I've had nothing but great service from them for about 20yrs now.
They still haven't rounded up yet, but a friend told me he called and they said that would happen soon, when PPHM's transfer agent sends them their "round up" shares and they get it processed.
I've seen that .-01 Q4/est. many times before.
I think that's -.01x7=-.07 DELETE
JD, Semper found the 5-2016 the AZN-Partnership Brochure last Monday, and then I found the 5-2017 update, “Partnering For Scientific Leadership”…
MORE: http://investorshub.advfn.com/boards/read_msg.aspx?message_id=132699419

PPHM will 1:7 R/S Shares eff. 7-10-17 (315mm/.606=>45mm/4.24)
7-7-17/PR: http://ir.peregrineinc.com/releasedetail.cfm?ReleaseID=1032464
ALSO:
Jul14/Fri.(after close): FY'17Q4(fye 4-30-17) Financials & Conf. Call/4:30pmET
http://ir.peregrineinc.com/events.cfm
Known Upcoming Events
Jul14/Fri.(after close): FY'17Q4(fye 4-30-17) Financials & Conf. Call/4:30pmET - http://ir.peregrineinc.com/events.cfm
Jul25: Neo-Synth’s "Precision: Lung Cancer - World R&D Summit", Boston http://tinyurl.com/ydcxlkja
...12:00pm: Bruce Freimark (Res.Dir/PreClin-Oncol), “Overriding PS-Mediated Tumor Immune Suppression to Enhance Immune Checkpoint Therapies”
Jul27: Final Settlement Hearing for 2013 Shareholder Derivative CA Lawsuit vs. BOD http://tinyurl.com/y982h3rt
Aug15-16: CBI’s MES 2017 (Mfg. Execution Systems), Orlando http://tinyurl.com/ya4twmbj
...Aug16/8:45am: Mehron Mirian (SeniorMgr./QA Validation), "MES AUDIT DEEP DIVE Computerized System Audit"
Aug21-25/Avid Booth #403: CHI's 9th Annual BioProcessing Summit, Boston http://www.bioprocessingsummit.com
~Sep7: FY'18Q1 (qe 7-31-17) Financials & Conf. Call - http://ir.peregrineinc.com/events.cfm
Sep25-28/Avid Exhibiting: Informa’s BPI’17: BioProcess Intl. Conf. & Exhibition, Boston https://lifesciences.knect365.com/bioprocessinternational
~Oct12: Peregrine's Annual Shareholder’s Meeting (2016 attendee reports: http://tinyurl.com/jx7ouay )
The 2017 Russell/3000 Rank Day was 5-12-17, JDM.
If a company didn't have the MktCap ($125mm or whatever it was this year) or meet $1+ on MAY 12 2017, they didn't make it into the Add/Delete list for 6-27-17 activation. It didn't matter if on May 15th they shot up to $500mm mktCap or had solved their $1 min. problem – they did not then get then ADDED to the May 12th list. It was frozen, except for “style & stability” issues.
Holler if I'm wrong and I'll fix!!
http://investorshub.advfn.com/boards/read_msg.aspx?message_id=132440645
http://www.ftserussell.com/files/press-releases/ftse-russell-announces-schedule-annual-russell-us-index-reconstitution
JDM, re: R3000, I don’t think you’re right. Read this post of mine from 6-23-17 and let me know if you think I’m wrong. I believe that the Russell/3000 list for 2017 was DONE on 5-12-17. Didn’t matter anything after that.
http://investorshub.advfn.com/boards/read_msg.aspx?message_id=132440645
That's from 10-2009: temp 20day Symbol, PPHMD, after 10-19-2009 1:5.
The closing price on 11-13-2009 was 3.15.
http://ir.peregrineinc.com/releasedetail.cfm?ReleaseID=416393
PPHMD iHub Quote: $3.15 (Closing price of temp 20day symbol 11-13-2009):
https://ih.advfn.com/stock-market/NASDAQ/peregrine-pharmaceuticals-inc-mm-PPHMD/stock-price
Historical quotes: https://finance.yahoo.com/quote/PPHM/history?p=PPHM
Semper, that NCCN announcement is from last Sept
NCCN Bavituximab Trials Announced 9-6-16 http://tinyurl.com/gutgwb5
#1: Ph1/HepC-Related Hepatocellular(Liver) (Bavi+RAD+Bayer’s Nexavar=Sorafenib), MOFFITT CC (N=18)
. . . . . . .PI: Jessica Frakes, MD https://clinicaltrials.gov/ct2/show/NCT02989870 <=Recruiting a/o 3-27-17
#2: Ph2/Newly Diag. Glioblastoma (Bavi+RAD+Merck’s Temodar), MASS-GEN. & DANA FARBER (N=36)
. . . . . . .PI: Elizabeth Gerstner, MD https://clinicaltrials.gov/ct2/show/NCT0313991 <=Recruiting a/o 6-16-17
#3: Ph2/Progressive Squamous Head+Neck (Bavi+Merck’s Keytruda), JOHNS-HOPKINS(Sidney Kimmel CC)
. . . . . . .PI: Ranee Mehra, MD https://clinicaltrials.gov/ct2/show/XXXXXXXXX - See Dr. R.Mehra Jan'17/IFN-y Biomarker: http://tinyurl.com/h8gzkww
That AZN-Partnership Brochure was 5-2016. Here's the 5-2017 one…
5-2017: “Partnering For Scientific Leadership”
https://www.astrazeneca.com/content/dam/az/PDF/2017/2017_18_AZ_BD%20&%20Partnering%20Brochure_FINAL.pdf
...MORE: https://www.astrazeneca.com/our-focus-areas/oncology.html

= = = = = = = =
10-15-15: Peregrine & AstraZeneca Expand Collab. w/Ph2/2ndLine-NSCLC Trial, Bavi+durvalumab(MEDI4736, brand-name "Imfinzi"), squamous or non-squamous. http://tinyurl.com/q79bkam
8-24-15: AstraZeneca & Peregrine Collaborate on Bavi+Durvalumab(“Imfinzi”) Ph1/1B Trial for “multiple solid tumors” http://tinyurl.com/owlxpsf
...Durvalumab=Imfinzi=MEDI4736(anti-PD-L1 immune checkpoint inhibitor). AZN’s Head/I-O(Robert Iannone): “Our partnership with Peregrine provides the opportunity to explore an exciting, novel combination that could deliver important clinical benefit to patients across a range of cancers."
SB, note the “approx sign”(~) before ~Jul13. That’s just what it will be if it corresponds to last year’s Q4/CC, which was on Thursday, July 14, 2016.
It could certainly NOT be July 13th (Thursday) this year – just an estimate.
~Jul13: FY'17Q4 (fye 4-30-17) Financials & Conf. Call - http://ir.peregrineinc.com/events.cfm
Known Upcoming Events
Jul7: PPS must be $1+ by July7, then 10 trading days at $1+ by July21 to avoid a R/S of up to 1:7. http://tinyurl.com/ya69tlso
~Jul13: FY'17Q4 (fye 4-30-17) Financials & Conf. Call - http://ir.peregrineinc.com/events.cfm
Jul25: Neo-Synth’s "Precision: Lung Cancer - World R&D Summit", Boston http://tinyurl.com/ydcxlkja
...12:00pm: Bruce Freimark (Res.Dir/PreClin-Oncol), “Overriding PS-Mediated Tumor Immune Suppression to Enhance Immune Checkpoint Therapies”
Jul27: Final Settlement Hearing for 2013 Shareholder Derivative CA Lawsuit vs. BOD http://tinyurl.com/y982h3rt
Aug15-16: CBI’s MES 2017 (Mfg. Execution Systems), Orlando http://tinyurl.com/ya4twmbj
...Aug16/8:45am: Mehron Mirian (SeniorMgr./QA Validation), "MES AUDIT DEEP DIVE Computerized System Audit"
Aug21-25/Avid Booth #403: CHI's 9th Annual BioProcessing Summit, Boston http://www.bioprocessingsummit.com
~Sep7: FY'18Q1 (qe 7-31-17) Financials & Conf. Call - http://ir.peregrineinc.com/events.cfm
Sep25-28/Avid Exhibiting: Informa’s BPI’17: BioProcess Intl. Conf. & Exhibition, Boston https://lifesciences.knect365.com/bioprocessinternational
~Oct12: Peregrine's Annual Shareholder’s Meeting (2016 attendee reports: http://tinyurl.com/jx7ouay )
7-25-17: Bruce Freimark (Res.Dir/PreClin-Oncol), Lung Cancer Conf., Boston
Jul25-26 2017: “Neo-Synth’s Precision: Lung Cancer - World R&D Summit”, Boston
“Precision: Lung Cancer (A Global Pharma R&D Summit) will bring together scientists and business leaders from pharma, biotech & academia, using extensive networking sessions to forge meaningful collaborations.”
http://precisionlungcancer.com ( http://neo-synth.com/#aboutus )
AGENDA: http://precisionlungcancer.com/agenda--day-one.html , http://precisionlungcancer.com/agenda--day-two.html
7-25-17 12:00pm (Day1):
“Overriding Phosphatidylserine-Mediated Tumor Immune Suppression to Enhance Immune Checkpoint Therapies”
Bruce Freimark (Research Dir., Preclinical Oncology), Peregrine Pharmaceuticals

= = = = = = = =
BAVI MOA 5-11-16 Breast Cancer Res. article, B.Freimark/M.Gray/CW.Hughes-et-al, “PS-Targeting/Bavi Combo w/Anti-PD1/PDL1 in Triple.Neg-MBC” http://tinyurl.com/zxu882y
...”our observations demonstrate that including PS-targeting antibodies such as bavituximab can enhance the anti-tumor activity of anti-PD-1/PD-L1 treatments, not only by increasing TIL responses but also by inhibiting cytokines stimulated by single-agent anti-PD-1 therapy that serve to suppress the immune response & promote tumor progression.”
BAVI MOA 2-9-15: PPHM's Dr. Bruce Freimark’s presentation at KEYSTONE "Tumor Immunology Meeting", Banff/CN – PR & Slides: http://tinyurl.com/q6cx4w6
BAVI MOA 12-12-14 San Antomio Breast Cancer Symposium, Dr. Bruce Freimark, Bavi+anti-PD-1 vs. Breast Cancer http://tinyurl.com/p5ng6vs
BAVI MOA 11-2014 SITC’14: 3 posters on preclin. Bavi+aCTLA4/aPD1 combo data (Hutchins Freimark Brekken Huang etal) http://tinyurl.com/pchzr6h
6-29-17/13D: Group Ronin & SW-Partners add 50,000sh. COMMON/June21 (since last COMMON 13D filed 6-20-17). The triggering event of this 13D is this: On 6-27-17, Mr. Stafford (sole, indirect beneficial owner) transferred all his 23,144,559sh./Common+Preferred from Ronin Capital LLC to Ronin Trading LLC.
6-29-17/13D: Ronin Trading(John Stafford III) & SWPartners(Stephen White), a 13D “Group”, adds 50,000 COMMON on June 21, 2017.
That brings total ownership of the Group to:
26,363,734 COMMON, 8.9% of 297,709,478 common O/S at 3-10-17.
123,499 PREFERRED, 7.5% of 1,647,760 preferred O/S at 1-31-17 (convertible to 1,029,158 Common @8.333).
13D TransDates COMMON-CHG PREF-CHG ENDING-COMMON ENDING-PREF.
3-2-17 1/20/17-3/1/17 +20,631,977 +51,364 20,631,977 51,364 http://tinyurl.com/jr42u23
3-10-17 3/2/17-3/9/17 +3,034,564 +25,661 23,666,541 77,025 http://tinyurl.com/ydxra96u
4-17-17 3/28/17-4/10/17 0 +23,334 23,666,541 100,359 http://tinyurl.com/lanjddc
5-19-17 5/1/17-5/17/17 0 +23,140 23,666,541 123,499 http://tinyurl.com/mgnn92x
6-20-17 3/10/17-6/16/17 +2,647,193 0 26,313,734 123,499 http://tinyurl.com/y76q5rqu
6-29-17 6/21/17 +50,000 0 26,363,734 123,499 http://tinyurl.com/y9sp8bfv
SPLITOUT 13D GROUP into Ronin(John Stafford III) and SW-Partners(Stephen White):
(since the COMMON 13D filed on 3-10-17)
Ronin Trading (Stafford) +1,997,194 +39,974 22,213,734 111,699
SW-Partners (White) +700,000 +6,500 4,150,000 11,800
6-29-17/13D: Group Ronin & SW-Partners add 50,000sh. COMMON/June21 (since last COMMON 13D filed 6-20-17)
6-29-17/13D: Ronin Trading (John Stafford III) & SWPartners(Stephen White), a 13D “Group”, adds 50,000 COMMON on June 21, 2017.
That brings total ownership of the Group to:
26,363,734 COMMON, 8.9% of 297,709,478 common O/S at 3-10-17.
123,499 PREFERRED, 7.5% of 1,647,760 preferred O/S at 1-31-17 (convertible to 1,029,158 Common @8.333).
13D TransDates COMMON-CHG PREF-CHG ENDING-COMMON ENDING-PREF.
3-2-17 1/20/17-3/1/17 +20,631,977 +51,364 20,631,977 51,364 http://tinyurl.com/jr42u23
3-10-17 3/2/17-3/9/17 +3,034,564 +25,661 23,666,541 77,025 http://tinyurl.com/ydxra96u
4-17-17 3/28/17-4/10/17 0 +23,334 23,666,541 100,359 http://tinyurl.com/lanjddc
5-19-17 5/1/17-5/17/17 0 +23,140 23,666,541 123,499 http://tinyurl.com/mgnn92x
6-20-17 3/10/17-6/16/17 +2,647,193 0 26,313,734 123,499 http://tinyurl.com/y76q5rqu
6-29-17 6/21/17 +50,000 0 26,363,734 123,499 http://tinyurl.com/y9sp8bfv
SPLITOUT 13D GROUP into Ronin(John Stafford III) and SW-Partners(Stephen White):
(since the COMMON 13D filed on 3-10-17)
Ronin Trading (Stafford) +1,997,194 +39,974 22,213,734 111,699
SW-Partners (White) +700,000 +6,500 4,150,000 11,800
Avid’s M.Mirian (SeniorMgr./QA Validation) speaks 8-16-17 at MES2017. Mehron Mirian joined Avid 2-2017; formerly 10yrs w/Bbraun Medical.
Aug15-16 2017: “CBI’s MES 2017 (Mfg. Execution Systems)”, Orlando
“12th Life Sciences Forum on Mfg. Execution Systems: Beyond Theory — Drive the Facility of the Future Utilizing MES & Continuous Manufacturing”
http://www.cbinet.com/conference/pc17144
CASE STUDIES FEATURING REAL, QUANTIFIABLE BUSINESS VALUE...
Mehron Mirian, Senior Mgr./QA Validation, Avid Bioservices (joined Avid 2-2017; formerly 10yrs w/Bbraun Medical)
https://www.linkedin.com/in/mehron-mirian-9bab2520

8-15-17 3:30pm: ADVANCED APPLICATION OF MES
Validation of Manufacturing Execution Systems (MES)
ELITE EXCHANGE Trailblazer Talk - The Journey to the Facility of the Future
FACILITATOR: Mehron Mirian (Avid, QA Validation)
----------
8-16-17 8:45-9:45am: Mehron Mirian (Avid Bioservices, QA Validation)
MES AUDIT DEEP DIVE Computerized System Audit - What Are the Considerations for MES?
I. What does an Audit mean to Life Science Industries?
II. What Are references for Computerized Systems Audit?
III. What Are the Agency Audit Considerations for a Computerized System? MES?
IV. What Does the “Survey” Say?
PPHM’s Exosomes Diag. Platform (incl. 6-22-2017 NATURE/BJC article)
...Known POC Data Summary (see below):
1-22-17/OncoTarget: Ovarian – senior author Alan Schroit (UTSW/PPHM SAB)
6-22-17/Nature(BJC): Breast & Pancreatic – senior author Alan Schroit (UTSW/PPHM SAB)
PPHM’s PS+ EXOSOME-BASED CANCER DETECTION & MONITORING TECHNOLOGY ("Liquid Biopsy") – Reverse Chron. History
...Excellent Exosome (aka microparticles, microvesicles) info: http://www.exosome-rna.com
6-22-17: Nature (British Jrnl of Cancer): ”Detection of Phosphatidylserine-Positive Exosomes for the Diagnosis of Early-Stage Malignancies””
Raghava Sharma, Xianming Huang, Rolf A Brekken (PPHM SAB), Alan J Schroit (PPHM SAB, UTSW Profile: http://tinyurl.com/yaqnvcvq )
https://www.nature.com/bjc/journal/vaop/ncurrent/pdf/bjc2017183a.pdf
ABSTRACT
Background: There has been increasing interest in the detection of tumor exosomes in blood for cancer diagnostics. Most studies have focused on miRNA and protein signatures that are surrogate markers for specific tumor types. Because tumor cells and tumor-derived exosomes display phosphatidylserine (PS) in their outer membrane leaflet, we developed a highly sensitive ELISA-based system that detects picogram amounts of exosomal phospholipid in plasma as a cancer biomarker.
Methods: This report describes the development of a highly specific and sensitive ELISA for the capture of PS-expressing tumor exosomes in the blood of tumor-bearing mice. To monitor the relationship between tumor burden & tumor exosome plasma concentrations, plasma from one transplantable breast cancer model (MDA-MB-231) and 3 genetic mouse models (MMTV-PyMT; breast and KIC and KPC; pancreatic) were screened for captured exosomal phospholipid.
Results: We show that quantitative assessment of PS-expressing tumor exosomes detected very early-stage malignancies before clinical evidence of disease in all 4 model systems. tumor exosome levels showed significant increases by day 7 after tumor implantation in the MDA-MB-231 model while palpable tumors appeared only after day 27. For the MMTV-PyMT and KIC models, tumor exosome levels increased significantly by day 49 (P<=0.0002) and day 21 (P<=0.001) while tumors developed only after days 60 & 40, respectively. For the KPC model, a significant increase in blood exosome levels was detected by day 70 (P=0.023) when only preinvasive lesions are microscopically detectable.
Conclusions: These data indicate that blood PS exosome levels is a specific indicator of cancer and suggest that blood PS is a biomarker for early-stage malignancies.
...Glimpse of beg. of 7pg. article: http://www.readcube.com/articles/10.1038/bjc.2017.183

2-23-17/StockNewsUnion.com: “What Next For Peregrine Pharmaceuticals After Exosomes Proof of Concept?”
Peregrine has said that its sight is on developing the technology into an optimized diagnostic platform for clinical testing of cancer. To reach that goal, Peregrine said it is seeking strategic partners to help it develop & commercialize the platform. Those partners would be expected to inject money into the project for an equity stake in PPHM or a cut of revenue from the sale of the platform. There are several large pharmaceutical companies developing their own ovarian cancer diagnostic systems are in need of such technology, so PPHM could pitch to them to join its exosomes-based diagnostic efforts.
POTENTIAL PARTNERS:
* AstraZeneca plc (NYSE:AZN https://www.astrazeneca.com - http://finance.yahoo.com/quote/AZN MktCap=$72B)
* Tesaro Inc. (NASDAQ:TSRO http://www.tesarobio.com - http://finance.yahoo.com/quote/TSRO MktCap=$9.5B)
* Clovis Oncology Inc. (NASDAQ:CLVS http://clovisoncology.com - http://finance.yahoo.com/quote/CLVS MktCap=$2.6B)
...are some of the major pharma brands that could be a fit for PPHM. The pharma giants are at various stages in the development of their ovarian cancer assets.”
http://stocknewsunion.com/what-next-for-peregrine-pharmaceuticals-nasdaqpphm-after-proof-of-concept/2043/
2-9-17/PR: ”Recent Publication Highlights Proof-of-Concept Data Supporting the Diagnostic Potential of Phosphatidylserine-Positive Exosomes in Ovarian Cancer”
http://ir.peregrineinc.com/releasedetail.cfm?ReleaseID=1011223
TUSTIN, Feb. 9, 2017: Peregrine Pharmaceuticals, Inc. (NASDAQ:PPHM/PPHMP), a biopharmaceutical company committed to improving patient lives by manufacturing high quality products for biotechnology and pharmaceutical companies and advancing its proprietary R&D pipeline, today announced the publication of positive proof-of-concept data for a novel exosome-based cancer detection platform. Results of the study, conducted at University of Texas (UT) Southwestern Medical Center, showed researchers were able to distinguish between healthy subjects and patients with ovarian tumors based on the levels of exosomes containing phosphatidylserine (PS) found in their plasma. Furthermore, analysis of the PS-positive exosome levels allowed researchers to distinguish between malignant and benign tumors. These data were recently published online by the peer-reviewed journal, Oncotarget, in a paper titled, "Detection of Phosphatidylserine-Positive Exosomes as a Diagnostic Marker for Ovarian Malignancies: A Proof-Of-Concept Study."
Peregrine is currently advancing the proprietary exosome-based cancer diagnostic technology, licensed from UT Southwestern Medical Center in July 2016 [7-14-16: http://tinyurl.com/zszd4fj ], with the goal of developing an optimized test for further clinical testing. As part of these efforts, the company is in the process of seeking a strategic partner for collaboration on developing and commercializing the technology. The platform is based on the diagnostic potential of tumor exosomes, which are small vesicles from tumor cells that are released into the blood as tumors grow. Tumor derived exosomes have PS on their surface as a detectable marker. It is believed that even small tumors begin to release PS-positive exosomes and thus the ability to detect these exosomes in the blood may be an indicator of the presence of a tumor.
In the study published by Oncotarget [1-22-17 FULL article: http://tinyurl.com/hlc68tq ], plasma samples from 34 patients with ovarian tumors and 10 healthy subjects were analyzed for the presence of PS-expressing exosomes in a blinded test. Results demonstrated that those patients with malignant ovarian cancer displayed significantly higher blood PS exosome levels than those with benign tumors (median 0.237 vs. -0.027, p=0.0001) and the malignant and benign groups displayed significantly higher blood PS exosome levels than the healthy subjects (median 0.237 vs -0.158, p < 0.0001 and -0.027 vs -0.158, p=0.0002, respectively).
"These initial proof-of-concept results are encouraging as they appear to support the underlying concept that the measurement of PS-positive exosome levels in blood could be a simple way to detect and monitor cancer. While the work is still early, we think these data serve as an important first step in highlighting the diagnostic potential of this platform," said Steven W. King, President and CEO of Peregrine. "This type of diagnostic technology is particularly important in an area such as ovarian cancer, in which screening options are limited and the ability to detect the disease at an early stage is inadequate. We look forward to continuing to explore the potential of the technology platform in ovarian as well as other types of cancer."
"There is a significant and growing interest in the healthcare industry around the ability to detect cancer and monitor its progression with more readily accessible blood tests. With this area being one of the fastest growing segments of the oncology diagnostics market, we believe that our exosome-based technology represents a significant product development and licensing opportunity," stated Stephen Worsley, VP of Business Development at Peregrine. "Based on the fact that PS is a marker associated with a broad range of cancer types, we believe our platform has potential applications in several solid tumors beyond ovarian cancer. With that in mind, we look forward to aligning with a partner to help explore the potential of this promising technology."
- - - - - - - - - - - - -
1-22-17/OncoTarget Article: PS-Exosomes: PPHM SAB’r Dr. Alan Schroit: Proof-of-Concept Datasupports the “high diagnostic power” of PS-Positive Exosomes in Ovarian Malignancies. ...Basically, Dr. Schroit (cell membrane guru) & his UTSW team used a variant of fully-human Bavi (1N11) to detect the PS on the exosomes…
FROM PG.8: “The data summarized in Fig. 4 [N=44] show that quantification of PS-exosomes in blood distinguishes, with 100% accuracy, healthy tumor-free individuals from patients with ovarian malignancies.”
1-22-17 Oncotarget:
“Detection of Phosphatidylserine-Positive Exosomes as a Diagnostic Marker for Ovarian Malignancies: A Proof of Concept Study”
Lea J 1, Sharma R 2, Yang F 2, Zhu H 1, Ward ES 3,4, Alan J. Schroit (PPHM SAB, UTSW Profile: http://tinyurl.com/yaqnvcvq ) 1,3
1 Harold Simmons Comprehensive CC, UTSW-MC/Dallas
2 Hamon Center for Therapeutic Oncology Res., UTSW-MC/Dallas
3 Dept of Immunology, UTSW-MC/Dallas
4 Dept of Molecular & Cellular Medicine, Texas A&M Univ. Health Science Ctr
https://www.ncbi.nlm.nih.gov/pubmed/28122335 Rec.1-4-17, Acc.1-11-17, Pub.1-22-17
ABSTRACT:
There are no suitable screening modalities for ovarian carcinomas (OC) and repeated imaging and CA-125 levels are often needed to triage equivocal ovarian masses. Definitive diagnosis of malignancy, however, can only be established by histologic confirmation. Thus, the ability to detect OC at early stages is low, and most cases are diagnosed as advanced disease. Since tumor cells expose phosphatidylserine (PS) on their plasma membrane, we predicted that tumors might secrete PS-positive exosomes into the bloodstream that could be a surrogate biomarker for cancer. To address this, we developed a highly stringent ELISA that detects picogram quantities of PS in patient plasma. Blinded plasma from 34 suspect ovarian cancer patients and 10 healthy subjects were analyzed for the presence of PS-expressing vesicles. The nonparametric Wilcoxon rank sum test showed the malignant group had significantly higher PS values than the benign group (median 0.237 vs. -0.027, p=0.0001) and the malignant & benign groups had significantly higher PS values than the healthy group (median 0.237 vs -0.158, p<0.0001 and -0.027 vs -0.158, p=0.0002, respectively). ROC analysis of the predictive accuracy of PS-expressing exosomes/vesicles in predicting malignant against normal, benign against normal & malignant against benign revealed AUCs of 1.0, 0.95 and 0.911, respectively. This study provides proof-of-concept data that supports the high diagnostic power of PS detection in the blood of women with suspect ovarian malignancies.
FULL ARTICLE (13pgs):
HTML: http://www.impactjournals.com/oncotarget/index.php?journal=oncotarget&page=article&op=view&path%5B%5D=14795&path%5B%5D=47251
PDF: http://www.impactjournals.com/oncotarget/index.php?journal=oncotarget&page=article&op=download&path%5B%5D=14795&path%5B%5D=47248
EXCERPTS FROM 1-22-17 ONCOTARGET ARTICLE:
**From DISCUSSION (pg. 8): “The data summarized in Fig. 4 show that quantification of PS-exosomes in blood distinguishes, with 100% accuracy, healthy tumor-free individuals from patients with ovarian malignancies… In summary, this study provides proof-of-concept data that supports the high diagnostic power of PS-expressing tumor exosome detection in blood from women with suspect ovarian malignancies. Ultimately, these studies could lead to earlier stage diagnosis, substantial cost savings, reduced patient exposure to radiation and invasive procedures, and improved clinical outcomes. The assay might also find utility in patients with radiographic abnormalities, even before clinical detection. Indeed, an accurate biomarker predicting the likelihood of malignancy would be extremely beneficial to such a population since they often face long periods of anxiety and uncertainty inherent to a “wait & watch” approach. Finally, if PS-exosome diagnostics are confirmed in a large study to be an accurate and reproducible biomarker of ovarian malignancies, the assay could be applied to the early detection of other visceral malignancies."
**From METHODS (pg.9): “Expression of an engineered tetravalent antibody for PS-detection Monoclonal 1N11 is a human IgG1^ that binds PS through the PS-specific plasma protein B2GP1. A tetravalent variant of 1N11 (1N11-T), with 4 binding sites per molecule was designed to generate a high avidity PS binding agent (Fig.1).”
……...[NOTE: 1N11 is Fully-Human Bavituximab (aka PGN635=AT004), B2GPI-dep. Binding]
**From FINANCIAL SUPPORT (pg.11): “Supported by Cancer Prevention & Res. Inst. of TX (CPRIT) Grant #RP110441, and a Simmons CC Support Grant 5P30 CA142543.”
**EDITORIAL NOTE (pg.11): “This paper has been accepted based in part on peer-review conducted by another journal and the authors’ response & revisions as well as expedited peer-review in Oncotarget."
.
PDF EXCERPTS 1-22-17/OncoTarget/A.Schroit/PPHM-Exosomes (5 pgs of the 13):





NOTE/2-10-17: Exosomes-RNA.com posted the news of the 2-9-17 PPHM Exosomes POC PR...
http://www.exosome-rna.com/proof-of-concept-data-for-a-novel-exosome-based-cancer-detection-platform-published/

10-13-16/ASM REPORT BY ATTENDEE COPPER888: “...I think there is a shift on how the company execs and BOD view the business. As mentioned by multiple posters, SK said that they are still looking to hit a "homerun" with Bavi. But I think that they are now doing that in a framework of risk avoidance, and profitability as their primary goals. With every initiative mentioned, SK would talk about partnering in the next sentence. Exosome testing with a partner; potential of exploring the utility of Beta Bodies - would "advance aggressively with a partner"; If Sunrise data warrants a small study to confirm, they would "partner the next step", etc. I think that for good or bad...the new company directive is the march toward profitability. He also said that the company is worth multiples of its current market cap and that they want to delay the RS as much as they can. "I am focused on getting the Share price over a dollar" He mentioned that there may be many events between now and April that may get us there...” http://tinyurl.com/jx7ouay

9-8-16/CC J.Hutchins PREPARED: “1st, an an update on our PS Exosome Program. As we announced in July, we executed a licensing agreement with UT Southwestern Medical Center for a novel exosome technology designed to detect PS-positive exosomes in a patient blood sample. It is our belief that this technology can provide clinicians with detection & monitoring information regarding the presence and the prevalence of cancer. Preliminary indep. studies have demonstrated that the levels of PS-positive exosomes present in the blood of cancer patients are higher than levels found in the blood of healthy volunteers. Furthermore, study findings also suggest that there is a correlation between the level of PS positive exosomes detected in the blood of cancer patients and the severity and extent of their disease burden. Given our in-house expertise in PS targeting, we believe that we are uniquely qualified to advance this technology. We believe there are significant opportunities to use this technology as both a complementary tool in bavituximab's ongoing development as well as more broadly as the basis for a novel cancer detection & monitoring test kit that will be the focus of partnering efforts. It is our goal to develop, optimize and validate a functional screening assay capable of detecting PS positive exosomes in a blood sample and to initiate partnering discussions for commercialization of the pgm in 2017. We are on track to achieve this goal and we look forward to providing further updates.” http://tinyurl.com/jydtkoy
9-8-16-PR/FY17Q1: “Peregrine in-licensed a novel exosome technology from UTSW that has potential for cancer detection and monitoring applications. This technology aligns directly with the company's expertise, its proprietary PS-targeting platform and the bavituximab dev. program. As such, there are opportunities to use this technology as both a complementary tool in bavituximab's ongoing development, as well as more broadly as the basis for novel cancer detection & monitoring tests that can be the focus of partnering efforts.” http://tinyurl.com/jydtkoy
7-14-16/PR: Peregrine Licenses Exosome-based technology from UTSW (Inventors: Alan Schroit/Philip Thorpe) http://tinyurl.com/zszd4fj
...“relates to assays that are able to detect small amts of PS+ Exosomes in a patient's blood sample as a way to detect cancer at a very early stage of development.”
VP JEFF HUTCHINS: “We are excited to enter into this licensing agreement with our long-term collaborators at UTSW. This technology offers a promising product development opportunity and aligns directly with the company's expertise with our proprietary PS-targeting platform and our longstanding CDMO capabilities around the development, qualification, and validation of in vitro analytical assays. As such, there are significant opportunities to use this technology as both a complementary tool in bavituximab's ongoing development, as well as more broadly as the basis for novel cancer detection & monitoring tests that can be the focus of partnering efforts. It is important to note that this dev. program will require minimal capital investment and has the potential to create significant value over the next 18mos., including potential partnering opportunities. As a result, we feel that today's licensing deal provides yet another important driver in our ongoing efforts to achieve profitability."
7-14-16/CC S.King PREPARED: “...In addition to bavituximab program which continues to have tremendous potential value, the company announced earlier today that it has licensed-in a Novel PS Exosome Technology, with the potential to detect & monitor cancer at an early stage through a simple blood test [ 7-14-16: http://tinyurl.com/zszd4fj ]. I recognize that some of you might think this theme is contrary to controlling spending as we move toward profitability, but in fact we believe that given our already existing knowledge base in Targeting PS and our already available infrastructure for developing & validating tests, that so a very modest capital investment we can quickly reach proof-of-concept with a goal of partnering this technology, which could then bring in addl. revenue, with another potential upside of the technology being that it can possibly be useful in the continued dev. of bavituximab. Jeff will talk more about this during his prepared remarks. Given the strategy of R&D targeted toward early partnering, will allow the company to continue its R&D activities with significant upside coming from partnering as we move to our profitability.” http://tinyurl.com/h8eqtg5
7-14-16/CC J.Hutchins PREPARED: “I'm very happy to be able to discuss a number of exciting developments in our preclinical group. First, as Steve discussed, we have executed the licensing agreement with UT Southwestern Medical Center, for novel exosome technology [ 7-14-16: http://tinyurl.com/zszd4fj ]. While many of you are familiar with exosomes, I’ll provide a brief overview for those who are not. Exosomes are cell-secreted vesicles, or mini-cells if you will, that are present in nearly all bodily fluids, including blood. Likewise, tumor-derived exosomes represent small pieces of tumor cells that are released into the blood as tumors grow. As well, these tumor-derived exosomes have Phosphatidylserine or PS on their surface as a marker and can also contain DNA, RNA and proteins as markers of malignant disease. It is believed that even small tumors begin to release PS-positive exosomes, and thus the ability to detect these exosomes in the blood may be an indicator of presence or progression of a tumor. The licensed technology is designed to detect & monitor PS-positive exosomes in a patient's blood sample, providing clinicians with detection & monitoring information regarding the presence & prevalence of cancer. These exosomes have PS flipped to the outside of the surface and demonstrate immunosuppressive activity, just as we find with tumor cells. Preliminary studies have demonstrated that the levels of PS-positive exosomes present in the blood of cancer patients are higher than levels found in the blood of healthy volunteers. Furthermore, study findings also suggest that there is a correlation between the level of PS-positive exosomes that are detected in the blood of cancer patients and the severity or extent of their disease burden. Given our in-house expertise in PS-targeting, we believe that we are uniquely qualified to advance this technology. As Steve stated, there are significant opportunities to use this technology as both a complimentary tool in bavituximab's ongoing development, which Joe will address later, as well as more broadly as the basis for a novel cancer detection & monitoring test kit that will be the focus of our partnering efforts. It is our goal to develop, optimize, and validate a functional detection & monitoring assay capable of detecting PS-positive exosomes from a simple blood sample, and, given the company's extensive experience in developing assays of this type, we do not anticipate the need for added personnel or any specialized equipment for this project. Once we have successfully validated this assay, we plan to establish proof-of-concept through an efficient preclinical & clinical testing program. We have no intention of conducting further development work beyond the proof-of-concept stage. Rather, we expect to initiate partnering discussions for commercialization of this program in 2017. We're very excited to begin this work on this new program and we'll have more details to offer in the coming months.”
...SUMMARY: “And lastly, we've in-licensed a new exosome technology for a minimal cost that leverages our existing in-house expertise and provides us with another opportunity for us to create value to product development. Together, we believe the strategy will provide success, as it will allow us to focus the majority of our resources on achieving our primary corporate goal, future sustainable profitability within 24mos. At same time, we will focus our R&D efforts on small early stage trials and development of the exosome technology in an effort to attract partners. We believe this strategy will allow us to build near-term revenues through Avid, while maintaining the potential for significant addl. value creation associated with our R&D efforts.” http://tinyurl.com/h8eqtg5
7-14-16/CC J.Shan PREPARED: “I'd first like to comment on our new exosome program. One of the most exciting aspects of this technology is the potential synergy that it offers with our bavituximab clinical dev. program. Through our ongoing work with bavituximab, we have gained significant understanding of PS-mediated immuno-suppression in cancer. The availability of the PS specific biomarker, which can be implemented in our planned future bavituximab clinical trials, aligns nicely with our refocused bavituximab dev. strategy aimed at generating the most meaningful data possible from small, early stage clinical trials to support partnering efforts. We are very anxious to bring this new technology to Peregrine and we look forward to the value it brings to our bavituximab program.” http://tinyurl.com/h8eqtg5
7-14-16/CC Q&A/Pantginis – SK: “(On the Exosome pgm), …it fits right in; we’re not having to hire addl. people, we’re not having to bring in addl. equipment. This really fits in with everything we are doing on both studying PS, as well as on the assay development side of the business. We think it’s actually going to be complementary. There are other technologies out there looking at exosomes; they're are all taking a very different approach to what we’re doing and we actually think they could be very complementary to each other. We also see a need, even as interest in exosomes begins to pick up, to actually utilize this in conjunction with other things that are in development.” http://tinyurl.com/h8eqtg5
7-14-16/CC Q&A/T.Yip – SK: “(On the Exosome pgm), the next step will be to validate that through patient samples. The beauty of this is that while you do need IRB approval of course, you’re not running really clinical trials, so this can be done in conjunction with either our ongoing trials or partners' trials, or there are many other sources of just receiving these types of blood samples. This gives us the ability to very quickly go through and test hundreds or thousands of patient samples as part of the validation process. At that point, we can zero in on what are the potential applications of the technology, outside of what we might do with our own PS Targeting programs – what would be the potential utility of this for patients. Our goal is not to become a diagnostics company, but to put this in the hands of a good organization that's already established in the diagnostics area and then have them finish up the commercialization and expansion of the utility of the actual assay itself. Our benefit at that point would become, hopefully, some residual royalties, milestones and what have you, which feeds back into our revenue goals of becoming profitable. So, we thought that this is a very attractive technology that just fits right in with what we’re doing and requires almost no addl. resources whatsoever…
Q2:/TY: ”when should we expect to see more preclin. data on this front?”… SK: “Our goal is, probably towards for the end of this year, to be in a position to have data that we can present. That will come in a lot of different formats, so it will be in conjunction with other ongoing studies that maybe taking place already to be standalone just on the diagnostic itself. So, you'll be hearing a lot about this, and one of the reasons when to get this news out there is because what you think sooner than later we will build a talk about this technology.” http://tinyurl.com/h8eqtg5
6-30-16: UTSW WEBSITE - “AVAILABLE TECHNOLOGIES” (#2932):

“A Blood-based Diagnostic Test for the Early Detection of Cancer” (Alan Schroit, S.Ward, A.Gazdar)
ALL: http://www.utsouthwestern.edu/about-us/administrative-offices/technology-development/industry/available-technologies.html
SCHROIT/EXOSOMES #2932: http://www.utsouthwestern.edu/edumedia/edufiles/about_us/admin_offices/technology_development/available_technologies/utsd-2932.pdf
This invention is a highly sensitive, blood-based assay system for the diagnosis of cancer. Tumors secret large quantities of cell membrane-derived exosomes into the blood stream of cancer patients. While all exosomes, whether from tumor cells or normal cells, share membrane epitopes similar to those found on the parent cell membrane, only tumor cell-derived exosomes express the membrane phospholipid, phosphatidylserine (PS), in the outer leaflet of the particle membrane. Based on data clearly showing that PS exposure is a unique signature of tumor-derived exosomes, the investigators developed a discovery platform for the screening & detection of tumor-derived exosomal PS signatures in patient blood. This novel tool provides:
1) A diagnostic marker that could dramatically improve the early detection of cancer.
2) Predicts whether a tumor is benign or malignant.
3) Could provide a predictive marker for response to therapy. Importantly, early detection of indolent neoplastic disease could significantly improve patient survival.
Currently, there are very few blood tests for cancer. These include alpha-fetoprotein for hepatocellular cancer, CA125 for ovarian cancer, and prostate specific antigen for prostate cancer. Although, high levels of these proteins might indicate the presence of neoplastic disease, none are diagnostic. Because our system detects a shared universal marker of neoplastic disease, it provides a viable method for detecting cancer of any type, in any organ.
6-9-16/WORLD-PATENT: Pub# WO/2016/201064, Filed 6-9-16, pub. 12-15-16 (A.Schroit, S.Ward, A.Gazdar)
Title: “Diagnostic Test For Early Stage Cancer”
URL (patentscope.wipo.int): http://tinyurl.com/gqvwkga
ABSTRACT:
“Disclosed are methods of identifying tumor-derived exosomes as an early cancer diagnostic, as well as for staging, assessing progression, and assessing therapy of cancer.”
Inventors: Alan J. Schroit [UTSW, PPHM SAB]; Adi Gazdar [UTSW]; E. Sally Ward [Texas A&M]
Applicant: Board of Regents, The Univ. of Texas System, Peregrine Pharmaceuticals, Inc.
EXCERPT from SUMMARY:
“Detecting may comprise contacting the sample of step (b) with an exosome binding agent that may be the same or other phosphatidylserine binding agent than the one used in step (b), or a non-phosphatidylserine binding agent. Given that only PS-positive tumor exosomes are captured and available at this stage (after step b), the detecting agent may be an agent that binds to all exosomes regardless of their source… In general, normal tumor-free individuals have undetectable levels of PS-exosomes in an unconcentrated sample. In contrast, patients with tumors will typically exhibit values above 100 pg/50 \xh of sample in an unconcentrated sample. Thus, values greater than 50 pg/50 ui, plasma (or serum) are indicative of a malignancy. The method may further comprise obtaining said sample from the subject prior to step (a). The method may further comprise diagnosing the subject from which the sample was obtained as having cancer. The method may further comprise staging the cancer or classifying the cancer type in the subject from which the sample was obtained. The method may further comprise performing steps (a)-(c) a 2nd time, and comparing the results from the 1st & 2nd times, thereby assessing cancer progression or regression. The method may further comprise performing steps (a)-(c) a 2nd time, wherein the subject has received a cancer therapy after the 1st performance of steps (a)-(c) and before the 2nd performance of steps (a)-(c), and comparing the results from the 1st & 2nd times, thereby assessing the efficacy of cancer therapy. The method may further comprise treating the subject with a cancer therapy… [0096] In addition to PS, exosomes exhibit a number of unique & non-unique markers. Some of the exosome-specific markers include biomarkers that can help distinguish the nature of the type of cancer from which the exosome originated. Some exemplary exosome biomarkers can be found above in Table 1. Global exosome biomarkers that can be detected with appropriately labeled agents include cd9, cd63, cd81, ALIX, HSP70, TSG101, duramycin, and heparin.”

- - - - - - - - -
Here is the corresponding U.S. PATENT APPLIC: Pub# 20170146542, Filed 6-9-16, pub. 5-25-17 (A.Schroit, S.Ward, A.Gazdar)
USPTO.gov: http://tinyurl.com/y7clom9w “DIAGNOSTIC TEST FOR EARLY STAGE CANCER”
- - - - - - - - - -
NOTE: 2-21-17: Exosomes-RNA.com posts the Exosomes PPHM/Schroit World-Patent App today.
http://www.exosome-rna.com/new-patent-application-published-diagnostic-test-for-early-stage-cancer/

2-27-15/U.S.PATENT: App#20150241431, filed 2-27-15 (Schroit/Thorpe)
Title: “Methods & Compositions for Isolating Exosomes”
http://tinyurl.com/Schroit-Thorpe20150241431
ABSTRACT:
“Disclosed are surprising new methods & compositions for isolating extracellular microvesicles such as exosomes, particularly disease-related and phosphatidylserine (PS)-positive extracellular microvesicles as exemplified by tumor- and viral-derived exosomes. The methods of the invention are rapid, efficient, cost-effective and, importantly, are suitable for use with large volumes of biological fluids and produce antigenically intact extracellular microvesicles & exosomes. The methods & compositions are based on the surprising use of acetate buffers to isolate large quantities of extracellular microvesicles, particularly tumor-derived exosomes, from solution, without damaging their morphological or functional properties or antigenicity.”
Inventors: Alan J. Schroit; Philip E. Thorpe; Shelley Fussey
Applicant: Board of Regents, The Univ. of Texas System, Peregrine Pharmaceuticals, Inc.
May2014/Article: J Immunol Methods, Thorpe/Schroit/etal, “A Novel ‘Salting-Out’ Procedure for the Isolation of Tumor-Derived Exosomes”
https://www.ncbi.nlm.nih.gov/pubmed/24735771

ABSTRACT:
“The last decade has seen an exponential growth in the number of exosome-related publications. Although many of these studies have used exosomes from biological fluids (blood, and ascites or pleural effusions) the vast majority employed vesicles isolated from large volumes of tissue culture supernatants. While several techniques are available for their isolation, all require a significant reduction in volume to obtain sufficient concentrations for study. One approach is to concentrate the medium before proceeding with their isolation, however, these procedures are very time consuming and require specialized laboratory equipment. Here we provide a new & effective method for the isolation of tumor-derived exosomes based on "charge neutralization" with acetate. We show that titration of tissue culture supernatants with 0.1M acetate to pH4.75 results in immediate precipitation of virtually all the exosomes. The precipitated exosomes can be washed to remove residual media and are readily "resolubilized" upon resuspension in acetate-free buffer at neutral pH. This simple cost effective method significantly increases the yield of exosomes from an unlimited quantity of culture supernatants. Exosomes isolated by this technique are indistinguishable from exosomes recovered by direct ultracentrifugation.”
The 2017 Russell/3000 Rank Day was 5-12-17. DONE.
If you don’t have the MktCap ($125mm or whatever this year) or meet $1+ on MAY 12 2017, you don’t make it into the Add/Delete list for 6-27-17 activation. PERIOD. It doesn’t matter if on May 15th a company shoots up to $500mm mktCap or solves their $1 min. problem – they do not get then ADDED to the May 12th list – it’s frozen, except for “style & stability” issues – see below.
What happens AFTER MAY 12th doesn’t matter re: the 2017 Russell/3000 reshuffle (and that includes us solving the $1 problem after May 12).
If I’m wrong about this let me know and I’ll correct, but I don’t think I am.
May 12 2017 = “Rank Day” – Russell US Index membership eligibility for 2017 reconstitution determined from constituent market capitalization at market close.
...Rank date, the date used to determine Russell US Index membership, usually in late May, will be on Friday, May 12 to facilitate the scheduled June alignment of shares outstanding & free float across the FTSE & Russell indexes. Fundamental data used to determine index constituent characteristics such as style & stability will still continue to be based on May month-end data.
http://www.ftserussell.com/files/press-releases/ftse-russell-announces-schedule-annual-russell-us-index-reconstitution
Glimpse of beg. of 7pg. Nature/BJC Exosomes article
http://www.readcube.com/articles/10.1038/bjc.2017.183

PPHM’s Exosomes Diag. Platform overview http://tinyurl.com/yacaw8xn (a/o 6-22-17)