Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
COMPLETED BAVI PH.1/2 TRIALS ARCHIVED FROM iBox 7-16-17, and updated 2-3-18.
Now linked to from within iBox for perpetuity: http://investorshub.advfn.com/boards/show_ibox.aspx?boardid=2076
BAVITUXIMAB SOLID CANCERS PHASE1+2 TRIALS:
==> PR=Partial-Resp(30-99% Red.), SD=Stable-Disease(29% Red.-19% Incr.), OR=Obj-Resp(PR+CR)
6-2012: FTM's charts of MOS Data from CtlArms(chemo) of Comp.Trials for 1NSCLC 2NSCLC PANCRE trials http://tinyurl.com/757plm7
11-25-11: Comp. of Bavi+Chemo vs. Avastin & Chemo-Only in 3 completed Ph.2 single-arm trials (ABC/2 & NSCLC): http://tinyurl.com/79b3jcj
O. 6th IST Trial: Bavi+Ipilimumab(Yervoy) vs. Adv.Melanoma (Ph1b, random, open-label, 2arms, n: 24=>3)
Protocol (UTSW): http://www.clinicaltrials.gov/ct2/show/NCT01984255 (PI: Dr. Arthur Frankel - see "Researching for Cures" http://youtu.be/0zLAxjFny5Q )
UTSW's listing: http://www.utsouthwestern.edu/research/fact/detail.html?studyid=STU%20102013-007
…Note: Ipilimumab = BMS’s “Yervoy” (anti-CTLA-4) http://www.yervoy.com
...11-10-15: UTSW chgd. ClinicalTrials to NO LONGER RECRUITING, Actual-Pts: 3
...12-10-15: “Due to newly-appr. therapies & chgs in SOC [ex: Keytruda/Opdivo: less-side-effects], enrollment recently stopped by UTSW.”; http://tinyurl.com/jkp885g
...4-23-14: Bavi+Yervoy IST trial initiated: http://tinyurl.com/km7krcm
N. 5th IST Trial (Bavi+Capecitabine+RAD) vs. Rectal Cancer (Ph1, open-label, 1arm, n=18)
Protocol: http://www.clinicaltrials.gov/ct2/show/NCT01634685
...Note: Capecitabine (prodrug of 5-FU) = Roche/Genentech's Xeloda - see http://www.xeloda.com
...7-31-17/Jrnl-Clin-Oncol(UTSW): Phase I update http://www.redjournal.org/article/S0360-3016(15)01602-8/fulltext
“There is interest in improving the tumoricidal effects of preoperative radiotherapy for rectal carcinoma by studying new radiosensitizers. The safety & toxicity profile of these combination regimens needs rigorous clinical evaluation. The primary objective of this study was to evaluate the toxicity of combining bavituximab, an antibody that targets exposed phosphatidylserine, with capecitabine, and radiation therapy. Patients with stage II/III rectal adenocarcinoma were enrolled on a phase I study combining radiation therapy, capecitabine, and bavituximab. A std. 3+3 trial designed was used. In general, bavituximab was safe & well tolerated in combination with radiation therapy & capecitabine in the treatment of rectal adenocarcinoma. To date, there are 8 evaluable patients, 3 at each of the 1st two dose levels and 2 at the 3rd and highest dose level. One patient at the highest dose level experienced a grade III infusion reaction related to the bavituximab. Of the 7 patients who have proceeded to surgery, 1 patient's tumor demonstrated a pathologic complete response. Bavituximab is safe in combination with capecitabine & radiation therapy at the doses selected for the study. Further clinical investigation would be necessary to better define the efficacy of this combination… Trial enrollment continues at the 3 mg/kg dose level. Toxicities have been relatively mild to date...”
...11-1-15/RadiationOncology – prelim. results/8 evaluables http://tinyurl.com/q3wey2b
…...”Of the 7 pts that proceeded to surgery, 1 demonstrated a pathologic complete response.”
...7-16-12: IST (Rectal) initiated at UTSW (PI=Jeffrey Meyer), ~18 patients - http://tinyurl.com/cr29l6k
M. Tumor Imaging & Dosimetry trial of I124-PGN650 (FH-Bavi) in Adv. Solid Tumors (Ph0, open-label, 1arm, n: 12=>11)
Protocol: http://clinicaltrials.gov/ct2/show/NCT01632696
..."I124-PGN650 is the Fab end of PS-targeting mab PGN635 (FH-Bavi) joined to the PET imaging radioisotope iodine-124, a new approach to imaging cancer."
...10-2017/Mol-Imag: Results of Human Testing (n=11) of 124I-PGN650 PET Imaging http://tinyurl.com/y9ofyu8o
...”Tumor targeting in Pts was less than prev. observed in animal studies… tumor uptake was quite low & not sufficient for clinical studies.”
...5-1-15: Interim Results/8pts (Jrnl Nuclear Med, vol56 suppl#3) http://jnm.snmjournals.org/content/56/supplement_3/1033
…”124I-PGN650 is safe and results in acceptable dosimetry and needs further optimization as an imaging agent based on the level of increased tumor uptake on PET/CT”
...6-28-12: Trial added to Trials.gov, 1st site "recruiting" (Wash.UnivSM/St.Louis)
…4-3-12: Peregrine Launches PS-Targeting Clinical Imaging Pgm (AACR'12 #2452) http://tinyurl.com/7p7jovt & http://tinyurl.com/7yrwqm7
L. 4th IST Trial (Bavi+Cabazitaxel vs. 2nd-Line PROSTATE(CRPC) Cancer, open-label Ph.1B/IIA) *CANCELLED due to chg. in SOC Drugs*
Protocol: http://clinicaltrials.gov/show/NCT01335204 (CRPC=Castration-Resistant Prostate Cancer)
...Note: Cabazitaxel = Sanofi-Aventis' Jevtana - see http://www.jevtana.com
...3-12-13: CRPC IST cancelled (Slow-Enroll/2 New SOC Drugs): http://tinyurl.com/c48osut
...5-25-11: IST (Prostate) initiated at UCI -> moved ot Med.Univ.SCar (PI: Michael Lilly), ~31 patients - http://tinyurl.com/3mtvdvl
K. 3rd IST Trial (Bavi+PemCarbo vs. Frontline NSCLC, open-label Ph.1B, n=25)
Protocol: http://clinicaltrials.gov/ct2/show/NCT01323062
…10-30-14: Interim data n=23, MSTO’14: MOS=12.2mos (hist-ctl ~10mos), PFS=4.8mos, ORR=35% http://tinyurl.com/mll62c6
...9-9-14: Stage IV NSCLC IST Enrollment complete. http://tinyurl.com/ktrfswj
...4-2012: Clinicaltrials.gov shows 2nd site added: Univ. of Pittsburg (PI=Liza Villaruz, MD)
...4-2-12 AACR'12: "5pts. to-date, 3 have PR's" http://tinyurl.com/7yrwqm7 (see #1744)
...3-8-11: IST (NSCLC) initiated at UNC (PI= J.Grilley-Olson), ~25 patients - http://tinyurl.com/6b926ku
J. 2nd IST Trial (Bavi+Paclitaxel(Taxol) vs. Her2- Met. Breast Cancer, open-label Ph.1, n=14)
Protocol: http://clinicaltrials.gov/ct2/show/NCT01288261
...3-31-15/A.Stopeck article(Ph1 data) “Cancer-Medicine” N=13: PFS=7.3mos, ORR=85%, 2 CR's http://tinyurl.com/nm5oog4
…6-3-13: ASCO’13/interim data, n=14: ORR=85%, 2 CR’s (15%) http://tinyurl.com/kq3uv4e
...4-29-13: Enrollment complete. http://tinyurl.com/cqrup9e
...4-3-12 AACR'12: "5pts. to-date, 2 CR's, 1 PR" http://tinyurl.com/7yrwqm7 (see #4404)
...1-19-11: IST (Her2- MBC) initiated at Arizona CC (PI=A.Stopeck), ~14 patients - http://tinyurl.com/5t7zomn
I. Phase II Bavi+GEM vs. Front-Line Adv. PANCREATIC (randomized, unblinded, n=70)
Protocol: http://www.clinicaltrials.gov/ct2/show/NCT01272791 (15 U.S. + 4 Ukraine = 19 as of 6-7-2012)
…1-22-13: FTM's post of 13 Ph3 Gem+Treatment Pancreatic Trials ('02-'13) - Mean MOS: GEM=6.4mos., GEM+TR=7.3mos. http://tinyurl.com/al99hx9
......Another FTM Pancreatic Phase3 trials table showing HR's, P-Values, and ORR% stats: http://tinyurl.com/btzkw4l
…6-3-13 ASCO’13/final data: n=70, Ctl=>Bavi MOS 5.2=>5.6mos, ORR 13%=>28% HR=.75 http://tinyurl.com/kq3uv4e
......Promising ‘immuno-indicative’ 1yr SURVIVAL results: GEM-Only(n=31): 0%, Bavi+GEM(n=32): 24.5% - see: http://tinyurl.com/lz5yg4f
...2-13-13 Topline Data: “Bavi+Gem resulted in more than a doubling of ORR” http://tinyurl.com/aqny7ny
...6-25-12: Enrollment complete. http://tinyurl.com/72tvnfj
...6-20-12: Early data (cutoff=6/6/12 bavi=15 ctl=17) presented at AACR Pancreatic Conf. http://tinyurl.com/77m9fw2
...1-5-11: U.S. Ph.2 randomized trial initiated http://tinyurl.com/26hnuzv "up to 70 front-line patients at ~10 clinical sites."
H. 1st Investigator-Sponsored (IST) Ph.I/II Trial (Bavi+Sorafenib vs. Liver Cancer/HCC, open-label, n=9+38=47)
...Note: Sorafenib = Onyx/Bayer's Nexavar - see http://www.nexavar.com
Protocol: http://clinicaltrials.gov/ct2/show/NCT01264705 UTSW: http://tinyurl.com/mwdc2ql (5 sites: 3/UTSW, Parkland-Hosp, Dallas/VA, PI=Dr. Adam Yopp)
...3-25-15: Dr.Adam.Yopp(UTSW) Oral-pres./SSO (Liver-IST/Ph2-data) http://tinyurl.com/opkh5qy N=38(79%HepC, ECOG/0=34%): MTTP=6.7mos, MOS=6.1, MDSS=8.7, DCR=58%
...1-16-15 ASCO Gastro-Symposium: Ph.2 data/n=38 (Adam Yopp), “These clinical outcomes of TTP=6.7/DCR=58%/PFS=4mo are quite encouraging…” http://tinyurl.com/m9uz9mo
...11-8-14 SITC'14: Ph.2 Correlative Studies data (biopsies B4/After) on 6pts, incl. KOL Dimitry Gabrilovich’s comments: http://tinyurl.com/pchzr6h
...9-9-14 Enrollment complete. http://tinyurl.com/ktrfswj (ph2=38 Ph1=9)
...4-4-12 AACR'12: Dr. Adam Yopp, "promising safety profile to-date" http://tinyurl.com/7yrwqm7 (see #5591)
...Feb'12-Sep'14 10+ times: CEO Steve King hints of future ex-US partner-driven Bavi+Sorafenib/LIVER trial in Asia-Pacific: http://tinyurl.com/nkaxtcc
......Articles & Data describe Liver Cancer challenges in Asian populations: http://tinyurl.com/7z7o8j9 & http://tinyurl.com/7z99cy4
...12-1-10: PPHM's 1st IST (Liver Cancer) initiated at UTSW, ~56 patients - http://tinyurl.com/3xd3e6c
…Per S.King, 5-18-10/R&R, "We've had a lot of interest in running clinical trials with the compound from investigators who have either had prior experience with the drug or would like to study the drug in various settings. Potential IST indications include all the major solid tumor types. Of particular interest is Liver Cancer, in which we have a natural tie-in with our HCV program, Ovarian Cancer & Pancreatic Cancer, also very nicely supported by the prior data."
G. Phase IIb Bavi+PC vs. Front-Line NSCLC (randomized, unblinded, 'confirmatory', n=86)
Protocol: http://clinicaltrials.gov/ct2/show/NCT01160601 (17 U.S. + 9 India + 2 RepGA + 7 RussianFED + 5 Ukraine = 40 as of 8-12-11)
...Also listed in: India's CTRI registry http://tinyurl.com/ljg7aad and WHO's registry http://tinyurl.com/kcp4z89
...6-27-13: Bavi+CP MOS>14mos (“with < 60% of survival events”) http://tinyurl.com/pmcgsgp
…”with less than 60% of survival events, while the Bavi+CP arm currently demonstrates a MOS > 14mos, there was not a meaningful enough diff. in survival between the 2 arms that would support the advancement of this combination. Full results will be presented at a future sci. meeting or thru pub.”
...5-7-13: FTM's Table of 20 prev. Ph.3 Trials in 1st-Line NSCLC (ctl=CP), MOS & HR results (Note Avastin improved CP-Only by 19%): http://tinyurl.com/cho7o29
...3-9-12: Topline ORR & PFS Data (Bavi+PC vs.PC-only) http://tinyurl.com/7m9r6ya
…...LOCAL reads: ORR/32%-31% PFS/5.8-4.6mos , CENTRAL reads: ORR/25%-23% PFS/6.7-6.4mos
...12-6-11 Prelim. Data (n=86, 100% Stage IV's) => ORR=39%, PC/alone=25%: http://tinyurl.com/7ph4tty
......Comp. vs. Avastin+PC/Ph3/n=417(74% Stage IV's): ORR=35% (Sandler/E4599/2006 http://www.nejm.org/doi/pdf/10.1056/NEJMoa061884 )
...9-8-11: Enrollment complete. http://tinyurl.com/3vv9zfx
...7-14-11/CC: Enrollment was taking longer than expected; have amended protocol; expanding to 30+ sites, expect enroll. comp. "in coming weeks", interim data by Yr-end'11. http://tinyurl.com/6k6y2as
…7-14-10/CC, J.Shan (VP/Clin+RegAffairs): "This trial is intended to confirm in a randomized setting the results from our Ph.2 signal-seeking NSCLC trial which showed 43% ORR, more than double the generally accepted chemo ORR of under 20% in numerous publications. Favorable results could then lead to an end of Ph.2 meeting with the FDA, with possibly a pivotal Ph/3 trial for front-line lung cancer, our 2nd potential regulatory pathway for bavituximab."
...7-14-10: U.S. Ph.2b randomized trial initiated http://tinyurl.com/27kxksl
……up to 86 front-line patients at ~20 clinical sites; goal: enrollment comp. by mid'11.
F. LEAD IND: Phase IIb Bavi+Doce vs. 2nd-Line NSCLC (randomized, double-blinded, placebo-ctl'd, n=120, 'registrational')
Protocol: http://clinicaltrials.gov/ct2/show/NCT01138163 (24 U.S. + 15 India + 2 RepGA + 7 RussianFED + 5 Ukraine = 53 as of 8-12-11)
Enrolled Oct2010 - Oct2011 at 40 global sites (per J.Shan 9-7-12 webcast (http://tinyurl.com/8cn87la )
8-2012: Compare Bavi+Doce's MOS=11.7mos (Bavi/3mg) to the 4 Curr-Approved 2Line/NSCLC Drugs http://tinyurl.com/cgnkvpa
• Taxotere/docetaxel => MOS=6.3mos (meta-analysis of 5 trials, 865 pts)
• Altima/pemetrexed => No diff. vs. Docetaxel (Ph.3 non-inferiority vs. Doce, 571 pts)
• Tarceva/erlotinib => MOS=5.3mos (TITAN Ph.III n=424 trial - see http://tinyurl.com/8w8lo93 )
• Iressa/gefitinib => "Iressa does not improve OS"
2-19-16/Clin-Lung-Cancer-Jrnl: Ph2 Final Data, Dr. David Gerber et al http://tinyurl.com/z5a7fwu
9-8-15: CMS SETTLEMENT EXECUTED (CSM pays PPHM only $600k for their “breach of contract, negligence & constructive fraud”, due to “limitation of liability contract clauses”) http://tinyurl.com/pemub47 (10Q/pg.17)
6-23-14: PPHM files Opposition to CSM’s Motion For Partial Summary Judgment - excerpts: http://tinyurl.com/q8xwd4v
. . .Declaration of Joseph Shan (VP/Clin+RegAffairs): http://tinyurl.com/kdgllxn
. . .Declaration of Jeffery Masten (VP/Quality): http://tinyurl.com/oru9p5q #6: “up to 25%" of CTL had 1mg, and “up to 25%" of 1mg had CTL(DoceOnly).
3-28-14: Peregrine files 1st Amended Complaint vs. CSM (13pgs) http://tinyurl.com/lsgf5lz
...Pg6: ”[as of 4-15-10], CSM had already secretly & unilaterally swapped the A & B arms so that those patients that were randomized in the A arm (CTL) and supposed to receive placebo treatments, were actually receiving 1MG Bavi treatments, and vice-versa. Peregrine’s Fall’12 investigation revealed that CSM committed other labeling & distribution errors affecting the A & B arms above & beyond the swap of the A& B arms noted above.”
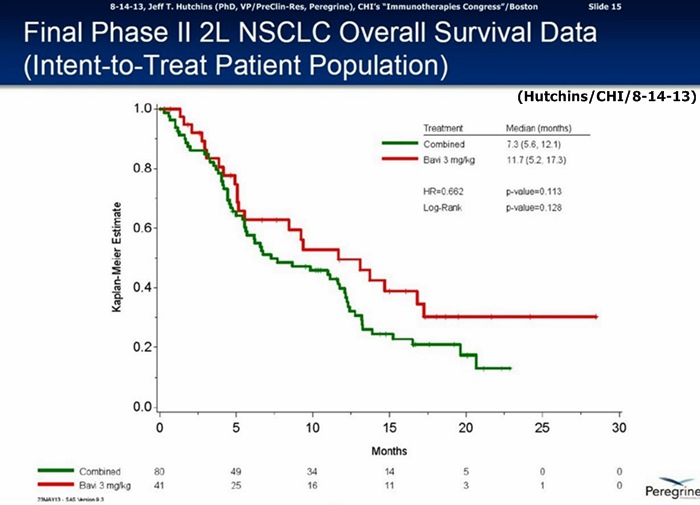
6-3-13/ASCO’13: Final Data Ph.II 2L/NSCLC http://tinyurl.com/my8qxw7
…60% improvement in MOS: Bavi/3mg=11.7mos. vs. 7.3mos. for CTL-arm(combined Bavi/1mg + DoxyOnly arms), HR=.662, P=.113
5-20-13: FDA Approves Bavituximab Ph.III Design for 2L/NSCLC; 600-pt trial to begin by y/e’13 http://tinyurl.com/n3dxtm6
...S.King: “We will now focus on starting the Ph.III trial while continuing ongoing partnering discussions.”
…R.Garnick: “This was a highly collaborative effort with the FDA; this trial, when combined with Bavi’s supporting data to date, could be sufficient to support a future BLA submission."
2-19-13: Topline Data Update from 2nd-Line NSCLC Trial after data discrepancies review http://tinyurl.com/ansqcea
…60% improvement in MOS: 3mg=11.7mos. vs. 7.3mos. for CTL-arm(combined 1mg & Doxy+placebo arms), HR=.73, p=.217
6-5-13: FTM's table of MOS data in 15 prior Doxy 2nd-Line NSCLC trials (Bavi's 60% MOS Improvement is Tops) http://tinyurl.com/m886ctb

1-25-13: MLV's George Zavoico recaps 2ndLine/NSCLC data errors & current status of PPHM's review http://tinyurl.com/b9u4pk8
...GZ: "This means that patients randomized into the high dose arm were administered Bavi correctly, whereas some of the patients in the placebo arm were administered low dose Bavi and some in the low dose Bavi arm were administered placebo. More importantly, the findings suggested that the MOS of 13.1 mos. in the high dose arm was likely to be valid. Even by historical measures, this is a remarkable result, since docetaxel's product insert lists the MOS of NSCLC patients receiving this widely used drug as 2nd-Line therapy in 2 trials as 5.7 & 7.5 mos. In effect, adding Bavi doubled the MOS. In our view, this was an extraordinary stroke of luck. If the high dose arm had been affected by the coding discrepancy, Peregrine would have been in a completely different & unfortunate position… Moreover, Peregrine must determine how best to present its case to the FDA. Will the historical controls be sufficient to justify moving Bavi into a Phase III pivotal trial, or will Peregrine have to pool the results of the placebo & low-dose arms and use that as a comparator to argue for moving ahead? A simple average of the placebo & low-dose arms results in a new control MOS of about 8.4 mos., still several months less than that of the high dose arm. This quick analysis results in about a 5-mo. survival advantage, a substantial prolongation for patients with second-line NSCLC and likely to justify moving Bavi into a pivotal Phase III trial in 2013, in our view."
1-7-13 PPHM PR - Review Update: "discrepancies are isolated to the placebo and 1 mg/kg arms; no evidence of discrepancies in the 3 mg/kg arm… Peregrine is taking a very conservative approach toward analyzing the results by combining the placebo & 1mg/kg arms into one treatment arm (control arm), and comparing to the 3mg/kg arm. This analysis indicates that the 3 mg/kg arm continues to show favorable TRR's, PFS, and OS over the new combined control arm. Peregrine expects to announce more detailed results from the analysis in the near term when it is completed." http://tinyurl.com/asup54d
9-24-12: Major Discrepancies found in 2nd-Line NSCLC Ph.2B Treatment Group Coding by Indep. 3rd-Party Vendor CMS/FargoND http://tinyurl.com/8r9zcqy
…"Investors should not rely on clinical data that the company disclosed on or before Sept. 7, 2012 from its Ph.2 Bavi trial in patients with 2nd-Line NSCLC or any presentations or other documents related to this Ph.2 trial."
9-24-12: Peregrine sues CSM Over Bavi Ph.2B 2nd-Line NSCLC Clinical Trial Mix-Up http://tinyurl.com/8fpgngu
…CSM = Clinical Supplies Management Inc., Fargo ND http://www.csmondemand.com
...1-17-13: Peregrine's lawsuit against CSM for "breach of contract & negligence" SERVED http://tinyurl.com/a7zrgys
…9-10-12 CEO Steve King, QtlyCC ( http://tinyurl.com/8nkwrml )
……"These are truly remarkable results (statistically doubling MOS) that are not only great for the pgm… but also great news for the NSCLC patients in the trial…"
…9-10-12 Robert Garnick (Head/Reg), QtlyCC ( http://tinyurl.com/8nkwrml )
……"The NSCLC data we announced 9-7-12 has far exceeded our expectations, and I hope that you're as excited as I am with bavituximab's potential. I feel strongly that Peregrine should be recognized for having the corporate courage to conduct the rigorous, randomized placebo-controlled Phase II trial that provided these robust data and that provide the basis for us to plan for a pivotal Phase III program."
...9-7-12: PPHM Press Release about Dr. Gerber's plenary at ASTRO/Thoracic/Chicago: http://tinyurl.com/96wrrso
…"The interim data showed a statistically significant improvement in OS (Hazard Ratio 0.524, p-value .0154) and a doubling of MOS (11.1/13.1mos. vs. 5.6mos.) in the Bavituximab-containing arms compared to the [Docetaxel] ctl-arm."
......VP Joe Shan's 15min. Webcast & Slideshow recapping Dr. David Gerber's 9-7-12 ASTRO/Chicago Plenary: http://tinyurl.com/96wrrso
…8-15-12 CEO Steve King, Wedbush/NYC ( http://tinyurl.com/8mhrtld )
......"As we're sitting here today, we have still not reached the # of events for MOS in either of the Bavituximab arms - and, in fact, we still have patients that are on treatments." Q&A: "it's going to be a very positive MOS result, it's just a matter now of magnitude."
…7-16-12 CEO Steve King, QtlyCC ( http://tinyurl.com/cs7spbz )
......"The strength of this 2nd-Line NSCLC data (esp. MOS trends) in this large area of high unmet medical need has also sparked a surge in partnering discussions that has included over 15 in-person partnering meetings since that time with major players in oncology, with all discussions ongoing and addl. parties showing interest. Our goal for the program is to position ourselves, along with a potential partner, to initiate Ph3 by mid-2013, which means an EOP2 meeting by yr-end'12. It would be ideal to have a partner on board to participate in the EOP2 meeting, and we have communicated this to interested parties and they agree."
…7-16-12 Robert Garnick (Head/Reg), QtlyCC ( http://tinyurl.com/cs7spbz )
……"We've been working very hard and very actively on the next steps in our Bavi 2nd-Line NSCLC pgm, given the favorable data that we've seen. As you can imagine, with data like this, there are many things that we need to consider. One consideration is that, should the data continue to trend the way it is, particularly in survival, this opens a door for potential discussions around a pathway for Accelerated Approval. At this point, all options are being considered, with Peregrine working towards the most efficient path forward from a regulatory standpoint." Q&A: "…all in all, I think the data is extremely compelling and I think it makes a really good case. Certainly, I think, I've seen a lot of Ph2 & Ph3 data, and this is as compelling Ph2 data as I've ever seen. So, I'm very comfortable proposing a meeting with the FDA for Q4'12."
…7-12-12 CEO Steve King, JMP-Conf/NYC ( http://tinyurl.com/csdclwb )
……"Re: 2nd-Line/NSCLC trial, the most thrilling thing is the fact that, even though we'd reached MOS for the ctl-arm(Doce) at end of Apr'12 of LESS THAN 6MOS, the majority of patients are still alive (today) in both Bavi arms, and we expect that to continue for some period of time still. Ph3 planning is underway already; our goal is to start this Ph3 by mid'13, meaning an EOP2 meeting with the FDA in Q4'12; our goal is to bring a partner on board, ideally in time for that EOP2 meeting, certainly before the beg. of the Ph3 trial."
…5-21-12: TopLine data n=117 for Bavi/3mg+Doce arm: ORR=17.9%/PFS=4.5mos (vs. CTL 7.9%/3mos) http://tinyurl.com/73aeyxj
......Importantly, MOS for CTL-arm "< 6 mos", but not yet reached in both Bavi arms.
...10-6-11: Enrollment complete. http://tinyurl.com/3m9re39
...7-14-11/CC: Enrollment was taking longer than expected; have amended protocol; expanding to ~45 sites, expect enroll. comp. "early in Q4/2011", data unblinding 1H'12. http://tinyurl.com/6k6y2as
…3-17-10/Roth, CEO S.King: "We refer to this trial as a Registrational Phase II Study, because we believe that if we have results anywhere near approaching what we saw in the earlier [India] study, it could be a conduit for Accelerated Approval."
...6-4-10: Ph.2b randomized reg. trial Open for enrollment: http://tinyurl.com/25v22qk
……"up to 120 refractory patients at ~30 clinical sites; goal: fully-enroll by mid'11, topline data by y/e'11."
1st 5 COMPLETED BAVI CANCER TRIALS:
These 5 completed Ph.1 & Ph.2 Bavituximab Cancer Trials archived to: http://tinyurl.com/72dnkfg
1. Ph.1A Bavi/Mono vs. Solid Cancers (USA n=26, 6/2005-6/2009)
2. Ph.1B Bavi+Chemo vs. Solid Cancers (India n=12, 11/2006-5/2007)
3. Ph.2 Bavi+Doce/BREAST/Refractory (RepGA n=46, 1/2008-5/2009): ORR=61%(Her2+=86%), PFS=7.4mos, MOS=20.7mos *
4. Ph.2 Bavi+PC/BREAST/Frontline (India n=46, 8/2008-9/2009): ORR=74%(Her2+=100%), PFS=6.9mos, MOS=23.2mos %
5. Ph.2 Bavi+PC/NSCLC/Frontline (India n=49, 6/2008-10/2009): ORR=43%, PFS=6.1mos, MOS=12.4mos #
......* BAVI+DOCE/Refract-MBC N=46: Compare *MOS=20.7mos to 11.4mos for Doce/alone (Nabholtz/JCO1999 Ph3/n=203 http://tinyurl.com/3rxqqtk )
......% BAVI+PC/FrontLine-MBC N=46: Compare %MOS=23.2mos to 16.0mos for P+C/alone (Loesch/JCO2002 Ph2/n=95 http://tinyurl.com/6wazs9p )
......# BAVI+PC/Frontline-NSCLC N=49: Compare #MOS=12.4mos to P+C/alone=10.3, Avastin+PC=12.3 (E4599/n=434), achieved using less Chemo (175-v-200 & AUC5-v-AUC6), treating 16% (8/49) more-difficult Squamous in Bavi trial (excluded totally from E4599), and treating higher % of sicker ECOG1 pts than in E4599 (96%-v-60%). See 6-15-11/PR http://tinyurl.com/3fcz5ok , ASCO'10 http://tinyurl.com/2g5cqof , and a discussion of differentiating factors between patient demographics & baselines treated in the 2 trials: http://tinyurl.com/6k5uuf7 .
EMAIL ADDY you PM'd doesn't work: RESEND pls.
DELETE
5 Events Added to AvidBio.com (March-Sept)
Mar 19–22 2018 - BioProcess Intl. West Conf., SanFran
Mar 19–22 2018 - DCAT Week, NYC
Jun 4–7 2018 - BIO Intl. Convention, Boston
Aug 13–17 2018 - The Bioprocessing Summit, Boston
Sep 4–7 2018 - BioProcess Intl. Conf. & Exhibition, Boston
https://avidbio.com = > http://ir.avidbio.com/events.cfm

= = = = = = = = =
EARLIER R.LIAS PRESENTATIONS:
1-29-18/Roger Lias: NobleCon14, Ft.Laud. - Webcast Replay: https://tinyurl.com/yanwk9yo
...Slide4/2:26 COMPLETED KEY ELEMENTS OF TRANSITION: “...We are currently seeking to monetize the R&D assets, the remainder, some residual value we believe, and we hope to have news on that front very soon, and obviously that represents potential upside for stockholders over the short term.”
...Slide15/10:13 NEAR-TERM PLANS FOR GROWING THE BUSINESS: “...We are expanding our sales & marketing. As I mentioned before, we haven’t been, for several years really, selling at all; there hasn’t been a need for it. So suddenly we’ve had to take that on, and I’m pleased to report that we’re already showing, really, tremendous progress in that area, and I hope we’ll have some announcements soon about several new clients.”
1-18-18: ASM2017 (@Myford Facility) - Roger Lias’ Slideshow & Attendee Reports https://tinyurl.com/yca6enbr PROXY/14A: https://tinyurl.com/y7qprpg9
1-8-18/Roger Lias: EBD's Biotech Showcase 2018 (parallel w/JPM Conf.), SanFran - Slideshow: https://tinyurl.com/ya6tgxxa
12-11-17: Qtly. Conf. Call (Lias/Lytle) Transcript https://tinyurl.com/ybycb2s6
...Dr. Lias, "the company is undergoing a broad-scale transformation, the goals of which are to shift complete focus to the Avid Bioservices CDMO business and the complete divestiture of all of Peregrine's legacy R&D assets, which include bavituximab."
FULL HISTORY of PPHM-Ronin PR’s, Letters, 13-D’s, Form4’s, Proxy’s, etc: https://tinyurl.com/ycb3fpfm
1-29-18/NobleCon14: Roger Lias Presentation Webcast Link (20mins/24 Slides)
Jan29 2018: “NobleCon14 – Noble Financial’s 14th Annual Inv. Conf.”, Ft.Laud.
Conf. website: http://nobleconference.com
11:30amET: Roger J. Lias, PhD: Corporate presentation
WEBCAST Link - 20mins/24 Slides:
http://noble.mediasite.com/mediasite/Play/a4bbd41fda0d41ab9be7504f2a8eb7901d
http://ir.avidbio.com/events.cfm
Slide4 “Completed Key Elements of Transition”
2:26/RL: “...We are currently seeking to monetize the R&D assets, the remainder, some residual value we believe, and we hope to have news on that front very soon, and obviously that represents potential upside for stockholders over the short term.”

Slide15 “Near-term Plans for Growing the Business”
10:13/RL: “...We are expanding our sales & marketing. As I mentioned before, we haven’t been, for several years really, selling at all; there hasn’t been a need for it. So suddenly we’ve had to take that on, and I’m pleased to report that we’re already showing, really, tremendous progress in that area, and I hope we’ll have some announcements soon about several new clients.”

= = = = = = = = =EARLIER R.LIAS PRESENTATIONS:
1-18-18: ASM2017 (@Myford Facility) - Roger Lias’ Slideshow & Attendee Reports https://tinyurl.com/yca6enbr PROXY/14A: https://tinyurl.com/y7qprpg9
1-8-18/Roger Lias: EBD's Biotech Showcase 2018 (parallel w/JPM Conf.), SanFran - Slideshow: https://tinyurl.com/ya6tgxxa
12-11-17: Qtly. Conf. Call (Lias/Lytle) Transcript https://tinyurl.com/ybycb2s6
...Dr. Lias, "the company is undergoing a broad-scale transformation, the goals of which are to shift complete focus to the Avid Bioservices CDMO business and the complete divestiture of all of Peregrine's legacy R&D assets, which include bavituximab."
FULL HISTORY of PPHM-Ronin PR’s, Letters, 13-D’s, Form4’s, Proxy’s, etc: https://tinyurl.com/ycb3fpfm
CEO Roger Lias 11:30amET/NobleCon14: VIDEO TOMORROW
Jan29 2018: “NobleCon14 – Noble Financial’s 14th Annual Inv. Conf.”, Ft.Laud.
Conf. website: http://nobleconference.com
11:30amET: Roger J. Lias, PhD: Corporate presentation
Delayed Webcast (24hrs later):
=> http://noble.mediasite.com/mediasite/Play/a4bbd41fda0d41ab9be7504f2a8eb7901d
http://ir.avidbio.com/events.cfm
Received by DovePress Sept. 6, 2017
Pub. 1-23-18/DovePress: “Antibody Targeting of Phosphatidylserine for the Detection & Immunotherapy of Cancer”
Rec: 9-6-17, Acc: 10-27-17, Pub: 1-23-18 (14 pgs.)
Olivier Belzile 1 Xianming Huang 2,3 Jian Gong 2,3 Jay Carlson 2,3 Alan J Schroit 1 Rolf A Brekken 1 Bruce Freimark 2,3
1 Hamon Center for Therapeutic Oncology Res.s, UTSW/MC/Dallas
2 Dept of Preclinical Res., Peregrine Pharm.
3 Dept of Antibody Discovery, Peregrine Pharm.
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=137906812
Updated w/Table3, History of Bavi Human Cancer Trials
1-23-18: New Dovepress article by UTSW+PPHM on PS-Targeting immunotherapy for cancer, concluding that, “targeting exposed PS in the tumor microenvironment may be a novel approach to enhance immune responses to cancer”. Gives a history of PS-Targeting (Bavituximab), both pre-clinical studies and human trials to date. For SUNRISE, for Results (Table 3), it says, “Manuscript in preparation”.
1-23-18/DovePress: “Antibody Targeting of Phosphatidylserine for the Detection & Immunotherapy of Cancer”
Rec: 9-6-17, Acc: 10-27-17, Pub: 1-23-18 (14 pgs.)
Olivier Belzile 1 Xianming Huang 2,3 Jian Gong 2,3 Jay Carlson 2,3 Alan J Schroit 1 Rolf A Brekken 1 Bruce Freimark 2,3
1 Hamon Center for Therapeutic Oncology Res.s, UTSW/MC/Dallas
2 Dept of Preclinical Res., Peregrine Pharm.
3 Dept of Antibody Discovery, Peregrine Pharm.
https://www.dovepress.com/antibody-targeting-of-phosphatidylserine-for-the-detection-and-immunot-peer-reviewed-article-ITT
ABSTRACT:
Phosphatidylserine (PS) is a negatively charged phospholipid in all eukaryotic cells that is actively sequestered to the inner leaflet of the cell membrane. Exposure of PS on apoptotic cells is a normal physiological process that triggers their rapid removal by phagocytic engulfment under noninflammatory conditions via receptors primarily expressed on immune cells. PS is aberrantly exposed in the tumor microenvironment and contributes to the overall immunosuppressive signals that antagonize the development of local and systemic antitumor immune responses. PS-mediated immunosuppression in the tumor microenvironment is further exacerbated by chemotherapy & radiation treatments that result in increased levels of PS on dying cells & necrotic tissue.
Antibodies targeting PS localize to tumors and block PS-mediated immunosuppression.
Targeting exposed PS in the tumor microenvironment may be a novel approach to enhance immune responses to cancer.
LINK TO FULL PDF (14pgs): https://www.dovepress.com/getfile.php?fileID=40225
CONCLUSION:
PS is well-recognized as a cell surface marker of apoptotic cells which provides signals to specific receptors for noninflammatory efferocytosis by phagocytes. The same signals are usurped in the tumor microenvironment by the exposure of PS on tumor blood vessel endothelium and tumor cells, contributing to immunosuppression and tolerance of tumor growth. Specific receptors that bind PS, including TIMs and TAMs, on immune cells and tumors, trigger these immunosuppressive pathways. The uptake of PS-targeting antibodies by tumors is readily demonstrated in preclinical models and initial clinical studies. Multiple preclinical studies serve as proof of concept that the antibody-mediated blockade of PS in tumors can reactivate innate & adaptive immune responses in the tumor microenvironment. A combination of PS-targeting antibodies with approved immune activating therapies such as chemotherapy, radiation, and immune checkpoint inhibitors (including antibodies targeting CTLA-4, PD-1, and PD-L1) and with novel therapies such as oncolytic viruses has the potential to treat a variety of different tumor types. These data support clinical trial evaluation of the PS-targeting antibody, bavituximab, in multiple oncology indications.
ACKNOWLEDGMENTS
We acknowledge the Dept’s of Clinical & Regulatory Affairs and Process Sciences at Peregrine Pharmaceuticals, for current insight on the clinical development of bavituximab and for providing antibodies for preclinical studies. We also thank Steve King for helpful technical discussions and support of preclinical studies and Dave Primm for editorial assistance.

1-18-18/2017 ASM: Roger Lias’ Slideshow & Attendee Reports
...Attendee Reports are after the Slides. I will update & re-post as more come in over the next few days. Thanks N40K for your addl. feedback – hopefully more to come.
1-18-2018: Avid Bioservices’ 2017 Annual Shareholder’s Mtg http://ir.avidbio.com/events.cfm
…10amPT at Avid’s Myford Facility, 14191 Myford Rd, Tustin, CA 72780 MAP: https://tinyurl.com/y9kpqesd
Webcast https://edge.media-server.com/m6/p/ij9i8ypx
PROXY/14A: https://tinyurl.com/y7qprpg9 VotingRecDate=11-27-17
CEO Roger Lias’ ASM Presentation Slides (1-18-18):














ATTENDEE REPORTS (2017 ASM 1-18-18):
By: Hawkfan1 (Michael) 1-18-18 9:41pmET #322974
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=137796412
Here are some of my notes taken at the ASM. They are very brief paraphrases, since I never took shorthand in school.
As usual, Brandon Cox was the first to take the microphone with a long list of questions, that at least this shareholder very much appreciated.
Q. You are burning $3-4mm/qtr, with no cash and no back log. How do you plan to raise capital?
A. Your estimate of our burn rate is in the right ball park. We do have $33mm in backlog, but that is quite conservative. Most companies recognize all of an order when it is placed, but traditionally, PPHM has only recognized backlog as work orders for that order are placed. But clearly, we need more capital. We must eliminate the 'going concern' clause. While it may not be ideal for a small biotech, it is probably something they can live with. But for a manufacturing company, it is a death knell. Customers need to know that you are going to be around for the long term.
Q. How dilutive will it be?
A. We don't know yet. We don't know how much we will need yet.
Q. Is anyone from the old management team working to help sell the R&D technology?
A. Yes, Shelly. And others are working as consultants.
Q. Most CDMO's are private companies. Are there any plans to take the company private?
A. Not currently. (He did mention, kind of jokingly, that it would be nice).
Q. Who are the major institutional investors?
A. Still Ronan, Tappan, and Eastern Capital.
Q. Will they be involved in the capital raise?
A. Hopefully.
Q. What is the cost of being a public company?
A. About $2-3mm/year.
Q. As a strictly mfg. company, do we really need 7 board members?
A. Right now, the experience that they all bring is extremely helpfull. Down the road, maybe not.
Q. How much cash do we currently have?
A. $27.7mm. (Not quite sure I got this right, but I think that $10mm was in customer deposits). [NOTE: the $27.7mm is a/o 10-31-17, the last 10-Q (Deposits were $13.1mm): https://tinyurl.com/ybycb2s6 ]
Q. Will Avid expand beyond the earthquake prone Tustin area?
A. It could be done relatively easily. Down the road, it will be evaluated.
Then Brandon relinquished the mike and North40k gave a long and impassioned commentary about various trials and people (such as Dr. Wolchok) that the company should contact about Bavituximab. He mentioned Wolchok's comment that his next trial "depends on the company".
Dr. Lias stated that Dr. Wolchok has not contacted the company. He mentioned that an associate of Dr. Wolchok had said that it will be 18 mos. before the next trial proceeds. (This was met with incredulity from several shareholders in attendance).
Dr. Lias stated that they have numerous discussions regarding CAR-T, but there was no interest. In some other discussions, they found our technology interesting, but then said "prove it in a trial".
N40k then asked if the recent Halozyme trial failure would affect Avid. It will not.
Brandon Cox then resumed his questioning. He asked a question regarding the IP patent clock, which I previously reported. For continuity, Dr. Lias's answer was that as patents start to expire, and a BP realizes that it will take about $70mm and 3-4 years to bring a product to market, a 5-year patent life is not that exciting to them.
Q. How much Bavi do we have in storage?
A. Enough for a couple of small trials, nothing big.
Q. Client #1 is Halozyme. We don't know the name of client #2, but are they gone for good?
A. No, but I don't expect them to be hugely successful. They will be years down the road.
Q. Question regarding the interest in the Exosome test kit.
A. Not much commercial interest.
Q. Are you trying to sell the IP in bits and pieces or as a package deal?
A. While a package deal would be ideal, not gonna happen.
Then Greg got up and asked about the process of sell the IP. Dr. Lias commented that there was not much interest. The previous board has talked to everyone - it is not a secret - just not much interest.
Then N40k asked the board to contact Precision for Medicine, which concluded the Q&A.
Again, this is very roughly paraphrased from my notes. Other members that were in attendance, please correct anything that I may have inadvertently gotten wrong.
As I decided to take the tour, I was not able to talk to the board members one on one after the meeting.
BY: HAWKFAN1 (MICHAEL)
... 1-18-18 7:54pmET #322954 (INITIAL POST)
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=137794640
Dr. Lias comment about the lack of interest in the IP by BP was in response to a question by Brandon Cox about the declining IP clock. His example was that if BP knows that it will take about $70M and 3-4 years of trials to get a drug to market, then a 5-year patent life is just not that exciting to them. This doesn't address the question of getting a patent extension, however. If this is true, then Dr. Lias needs to be made aware of it.
FU #323010:
Just a few more general impressions from the meeting today. Like the rest of you, I was very disappointed in the approach to marketing the IP. On the one hand, Dr. Lias said that there were on-going discussions that he could not talk about, but he continually down played any expectation that we would receive much value for it. It was almost like they have already given up and have a 'Hey, we tried, but nobody is interested' attitude. They certainly didn't spell out any detailed marketing plan.
On the CDMO front, they generally impressed me as qualified, and will probably do a fine job, but I almost choked when Lias said that Tracy Kinjersky "hit the ground running". Hopefully, she will not emulate Mary Boyd.
The tour of the Myford facility was as impressive as ever. The 2 new 2000L reactors are installed, tested and ready to go, but have not yet been used in production. Someone asked how many employees we have now and we were told about 200, of which 30 something (I don't remember the exact number) are in production. I asked, if only 30 something are in production, what do the others do? I was told quality control, testing, etc., which I would consider as part of production. This brought the number up to about 75. Unfortunately, we were interrupted, and I wasn't able to pursue this breakdown any further.
It seems reasonable to me that we can get to $200mm in sales, but it will be at least 2-3 years down the road. They already have space in the Myford building to build out a duplicate of the existing facility, except that I believe it will use all 2000L reactors. But this space is still raw warehouse space, and will take a minimum of a year to build. And they won't start building it until they have reached significant profits from the exiting facility. So it all hinges on how fast they can attract new clients.
FU#323064:
re: How many in meeting? I didn't actually count, but I was going to guess that there were probably about 20 shareholders, and we were matched, if not outnumbered, by company employees. My impression was that it was a bit smaller attendance by shareholders than in years past.
By: Holotawoopas 1/18/18 4:49pmET #322902
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=137790910
For those who have pinned their hopes on the value of the IP it is time to mourn your loss and move on as there was nothing concrete about any potential monetizing extrapolated at the ASM. N40 gave a valiant effort to plead the case of value of the IP but IMO it fell on deaf ears! The CEO said no BP interest has been received!
FU#322909:
The CEO stated that the biggest down fall to monetizing the IP has nothing to do with leverage but has everything to do with patent expiration!!!
By: Eb0783 1/18/18 1:45pmET #322864
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=137783413
ASM votes: #1 all re-elected, #2 for 36.4mm, #3 for 15.2 v. against 3.6, #4 1 year
FU#322879:
Per Lias, there is no formal process, group, or investment bank in place to sell the IP.
FU#322934:
Lias said he wasn't a patent expert when made those comments about short patent life being an issue. It was obvious he wasn't aware that patents could be extended due to years lost in development. Some of us took that short patent life excuse with a grain of salt.
FU#323063:
re: How many in meeting? I counted about 40 at the start of the meeting. I would guess 50%-60% were shareholders. That is about normal from my experience. I remember counting 40 & 50 in previous ASMs.
FU#323066:
The thing is, if they are really not having a process, group, or IB in place to evaluate/sell our IP, they don't have to spend anything to do it. At the very least, give the task to some IB or 3rd party expert on contingency: you can keep 50% of whatever you can sell it for. (You lawyers often take cases on the same basis and accept 30, 40, 50% quite often). I said that to Lias after the meeting, with Ziebell & Lytle standing there - no comment from them. I find their stance on this incredulous, if true!
By: Djohn 1-19-18 2:18pmET #323067
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=137818937
MY THOUGHTS AFTER THE ASM: I missed the fancy danish, the cream cheese and bagels also missed the fancy hotel backdrop making me think we were on the verge of something astronomical. What did not miss was my thoughts of how many shares had to be sold to pay for me to bite on my bagel, sip on my fine coffee while looking at a beautiful swimming pool at a posh hotel.
It’s amazing what can be done with a couple of cases of bottled water, one pot of coffee and a room with chairs. That alone convinced me our new leadership is serious about how they spend the shareholders money!
For me the ASM went as I expected. It just confirmed to me that we are no longer chasing a dream. We no longer have a questionable cast of characters spending our money diluting our shares. It’s pretty easy, we are a manufacturing company now with basic straight forward business model. We have people in place who are qualified to grow this business and IMO recoup the total amount I invested in PPHM maybe even more.
I have given up the dream! I will awake daily and put on a BaviDermCQ patch. I will develop a 12-step program to wean myself from astronomical thoughts. One of those steps will be to not visit this board as often. Another step will be to fully investigate the members of the BOD in any company I ever invest in again. One other will be to take full responsibility for my investment decisions and accept how they turn out.
I bugged out right after the ASM ended and didn’t stick around for the tour of Avid. I had a flight to catch. Only one flight I could get to make it home that day. Maybe some of you there could gather more insight in talking with BOD or management during the tour. I went on the Avid tour last year and was impressed with the facilities.
FWIW This is what I gathered and is my opinion about the current situation. I saw a group of highly qualified individuals both on the new BOD and new leadership for Avid. These individuals are try to make lemonade out of lemons. Face it folks, after years and years of being told we had a bag of diamonds but we ended up with a bag of lemons! I am not saying that PS/Bavi/IP is worthless. Nobody really knows! Yet! but because the leadership of legacy PPHM did not do the correct things to prove it’s worth, we are left with an IP in shambles. After spending several hundreds of millions they left us with no value for the IP and the only way to prove value of the IP is to spend many more millions. ( A Bag of Lemons) Basically, after Sunrise failure the PPHM leadership decided to spend millions more to search around in the bag of lemons to see if they could just one diamond. We can all speculate on why legacy PPHM leadership continued down their path.
Fact to me is last year PPHM had two things. An R&D division with the IP and faced with spending millions more to prove some worth with low chance of success, even lower based on past successes. A CDMO business that with the right leadership and direction could become a nice profitable business in a few years. The chance of success with CDMO is very high IMO and I think valued much higher than R&D portion.
I think Ronin saw the CDMO business as a good development opportunity and took it. Real business people made real business decisions with maximum potential and maximum returns in mind. C'est la vie PPHM HELLO CDMO
FU#323070:
Don't get me wrong. I am not saying IP does not have value! I bought TCLN because I believed it did have value. I could hear the enthusiasm in Dr Thorpe's voice when he presented it. That was powerful! I just think the IP has been squandered all these years and nobody knows how much more money must be spent to realize it's real worth and with failed PIII no one is willing to take a chance on it. Hence, It's not worth much at this point in time. I think best path forward is CDMO to recoup invested money.
By: Aikifredicist 1-18-18 8:16pmET #322961
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=137795041
I got up at the meeting and asked about Exosomes, but Lias just fudged on the answer. He doesn’t know what he has.
By: North40000 1-22-18 11:33amET #323194 (Initial Main Report)
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=137869635
...Hawkfan posted that my comments to the CDMO BOD at the ASM last Thursday morning seemed to him impassioned. I was not aware of that quality in my remarks. I did try to portray facts, as I knew them, to the BOD, with suggestions as to what that BOD should do to bring increased value to CDMO, PPHM and SH assets/share value or $$$price.
Frankly, those facts & remarks seemed to me to fall on deaf ears - the stolid, expressionless faces of BOD members while I spoke made me feel I was talking to an army or array of Chinese stone soldiers. I was pleased to see one BOD member take notes during my presentation (I hope it was not a grocery list for his wife when he returned home). It was like delivering remarks to a judicial bench of judges who were not sufficiently informed of the record [history] before them to know what questions to ask.
I began explaining that we had invested in past years some $550k in PPHM therapy technology, and once had ~ 500k shares of PPHM with a break-even point of $1.10/share. Now, with a reverse split behind us together with a change in focus and name of PPHM to CDMO, we hold ~73k shares CDMO with a break-even point of ~ $7.70/share. I expressed my own opinion that the individual new BOD members had the qualifications necessary to bring CDMO share price/value well beyond the above $7.70 eventually. I was a bit dismayed to see from the slides that revenues might increase to $200mm by 2022 from >$100mm in 2018. The identity of further customers signed by Avid/CDMO since Jan.2017 was not further explained.
For now, I will not belabor my previous comments as to what Dr. Jedd Wolchok and other MSKCC personnel have told me in face-to-face conversations at SITC 2016, Wistar (Dec. 7, 2016), AACR 2017 or JHU (Johns Hopkins) in Baltimore(Dec. 7, 2017). Nor will I expand my comments re: conversations had with Dr. Nikoletta Kallinteris (PPHM) or Dr. Thomas O’Kleen (Precision for Medicine) at SITC in Nov.2017. My remarks are already recorded here, and my comments last week to the CDMO BOD contain basically the same information, including the fact that Precision for Medicine has acquired $200mm in addl. funds and has >1000 employees world-wide...
EARLIER #323153:
When I asked PL at ASM what SK is doing these days, PL told me he is occupying his time acting as a kind of entrepreneur… marketing PPHM platform.
FU#323169:
No mention by any of 6 new BOD members of what you post (Biopharm: GORE Protein Capture Device) at least that I heard. Indeed, but for Dr. Lias, I heard none of the other 5 new BOD members speak at all about anything - all 6 that were there at the front table [one could not attend the ASM at all due to snow in N.Car.] had secreted themselves out of our sight until shortly before 10am, when they all filed in from a heretofore closed door, sat down in chairs with name placards in front of them, were introduced by either the new BOD chairman [who welcomed us and called the meeting to order] or Dr. Lias [I forget which]. The new BOD chair & Dr. Lias had evidently decided who should thereafter speak and run the meeting, and it was all Dr. Lias after that who stepped to the microphone to speak.
After the meeting was adjourned post Q&A session, we were in a large group of SHs and employees who lined up in front of that same door mentioned above for an Avid/Myford facility tour. I do not know where the BOD members went, or whether any remaining SHs had an opportunity to speak to them.
In a curious way, I personally felt I was back in the courtroom at 717 Madison Place in WashDC, where I had argued many appellate cases before a 5-judge tribunal, sitting at a more elegant bench with placards identifying them by name in front of them. A major difference - at least those judges asked questions.
= = = = = = = = = = = = = = =RECALL:
1-8-17: EBD's Biotech Showcase 2018 (parallel w/JPM Conf.), SanFran https://ebdgroup.knect365.com/biotech-showcase
...1-2-18 PR: https://tinyurl.com/ydcc9agv
...3:30pmPT: CEO Roger Lias, Corporate Presentation
...1-8-18 Lias BioShowcase’18 Presentation PDF (24 slides): https://tinyurl.com/ya6tgxxa
FULL HISTORY of PPHM-Ronin PR’s, Letters, 13-D’s, Form4’s, Proxy’s, etc: https://tinyurl.com/ycb3fpfm
12-26-17: Roger Lias replaces Steven King as Pres./CEO; in process of changing name to Avid Bio. and new Nasdaq Ticker ("early 2018") https://tinyurl.com/yb34e2t8
12-11-17: Qtly. Conf. Call (Lias/Lytle) Transcript https://tinyurl.com/ybycb2s6
...Dr. Lias, "the company is undergoing a broad-scale transformation, the goals of which are to shift complete focus to the Avid Bioservices CDMO business and the complete divestiture of all of Peregrine's legacy R&D assets, which include bavituximab."
Thx much, North40k. Please do me a favor before I re-post the Attendee reports compilation with your latest one included…
For me, and others I’m sure, would you translate these abbreviations that you used (or give me hints so I can look them up and expand them for clarity)…
THANK YOU!
JHU = xxx
UPDATE – I think I’ve got these 3:
Dr. NK = xxx => I know now: PPHM’s Dr. Nikoletta Kallinteris
Dr. TK (s/b TG ?) = Tobias Guennel (Precision for Medicine)
PFM = Precision for Medicine, Frederick, MD
THIS SECTION:
N40K: “For now, I will not belabor my previous comments as to what Dr. Jedd Wolchok and other MSKCC personnel have told me in face-to-face conversations at SITC 2016, Wistar (Dec. 7, 2016), AACR 2017 or JHU in Baltimore(Dec. 7, 2017). Nor will I expand my comments re: conversations had with Dr NK(PPHM/CDMO) or Dr. TK(PFM) at SITC in Nov.2017. My remarks are already recorded here, and my comments last week to the CDMO BOD contain basically the same information, including the fact that PFM has acquired $200mm in additional funds and has >1000 employees world-wide.”
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=137869635
Keep 'em coming, Attendees - much thanks! DELETE
1-18-18/2017 ASM: Roger Lias’ Slideshow & Attendee Reports
...Attendee Reports are after the Slides. I will update & re-post as more come in over the next few days.
1-18-2018: Avid Bioservices’ 2017 Annual Shareholder’s Mtg http://ir.avidbio.com/events.cfm
…10amPT at Avid’s Myford Facility, 14191 Myford Rd, Tustin, CA 72780 MAP: https://tinyurl.com/y9kpqesd
Webcast https://edge.media-server.com/m6/p/ij9i8ypx
PROXY/14A: https://tinyurl.com/y7qprpg9 VotingRecDate=11-27-17
CEO Roger Lias’ ASM Presentation Slides (1-18-18):














ATTENDEE REPORTS (2017 ASM 1-18-18):
By: Hawkfan1 (Michael) 1-18-18 9:41pmET #322974
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=137796412
Here are some of my notes taken at the ASM. They are very brief paraphrases, since I never took shorthand in school.
As usual, Brandon Cox was the first to take the microphone with a long list of questions, that at least this shareholder very much appreciated.
Q. You are burning $3-4mm/qtr, with no cash and no back log. How do you plan to raise capital?
A. Your estimate of our burn rate is in the right ball park. We do have $33mm in backlog, but that is quite conservative. Most companies recognize all of an order when it is placed, but traditionally, PPHM has only recognized backlog as work orders for that order are placed. But clearly, we need more capital. We must eliminate the 'going concern' clause. While it may not be ideal for a small biotech, it is probably something they can live with. But for a manufacturing company, it is a death knell. Customers need to know that you are going to be around for the long term.
Q. How dilutive will it be?
A. We don't know yet. We don't know how much we will need yet.
Q. Is anyone from the old management team working to help sell the R&D technology?
A. Yes, Shelly. And others are working as consultants.
Q. Most CDMO's are private companies. Are there any plans to take the company private?
A. Not currently. (He did mention, kind of jokingly, that it would be nice).
Q. Who are the major institutional investors?
A. Still Ronan, Tappan, and Eastern Capital.
Q. Will they be involved in the capital raise?
A. Hopefully.
Q. What is the cost of being a public company?
A. About $2-3mm/year.
Q. As a strictly mfg. company, do we really need 7 board members?
A. Right now, the experience that they all bring is extremely helpfull. Down the road, maybe not.
Q. How much cash do we currently have?
A. $27.7mm. (Not quite sure I got this right, but I think that $10mm was in customer deposits). [NOTE: the $27.7mm is a/o 10-31-17, the last 10-Q (Deposits were $13.1mm): https://tinyurl.com/ybycb2s6 ]
Q. Will Avid expand beyond the earthquake prone Tustin area?
A. It could be done relatively easily. Down the road, it will be evaluated.
Then Brandon relinquished the mike and North40k gave a long and impassioned commentary about various trials and people (such as Dr. Wolchok) that the company should contact about Bavituximab. He mentioned Wolchok's comment that his next trial "depends on the company".
Dr. Lias stated that Dr. Wolchok has not contacted the company. He mentioned that an associate of Dr. Wolchok had said that it will be 18 mos. before the next trial proceeds. (This was met with incredulity from several shareholders in attendance).
Dr. Lias stated that they have numerous discussions regarding CAR-T, but there was no interest. In some other discussions, they found our technology interesting, but then said "prove it in a trial".
N40k then asked if the recent Halozyme trial failure would affect Avid. It will not.
Brandon Cox then resumed his questioning. He asked a question regarding the IP patent clock, which I previously reported. For continuity, Dr. Lias's answer was that as patents start to expire, and a BP realizes that it will take about $70mm and 3-4 years to bring a product to market, a 5-year patent life is not that exciting to them.
Q. How much Bavi do we have in storage?
A. Enough for a couple of small trials, nothing big.
Q. Client #1 is Halozyme. We don't know the name of client #2, but are they gone for good?
A. No, but I don't expect them to be hugely successful. They will be years down the road.
Q. Question regarding the interest in the Exosome test kit.
A. Not much commercial interest.
Q. Are you trying to sell the IP in bits and pieces or as a package deal?
A. While a package deal would be ideal, not gonna happen.
Then Greg got up and asked about the process of sell the IP. Dr. Lias commented that there was not much interest. The previous board has talked to everyone - it is not a secret - just not much interest.
Then N40k asked the board to contact Precision for Medicine, which concluded the Q&A.
Again, this is very roughly paraphrased from my notes. Other members that were in attendance, please correct anything that I may have inadvertently gotten wrong.
As I decided to take the tour, I was not able to talk to the board members one on one after the meeting.
BY: HAWKFAN1 (MICHAEL)
... 1-18-18 7:54pmET #322954 (INITIAL POST)
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=137794640
Dr. Lias comment about the lack of interest in the IP by BP was in response to a question by Brandon Cox about the declining IP clock. His example was that if BP knows that it will take about $70M and 3-4 years of trials to get a drug to market, then a 5-year patent life is just not that exciting to them. This doesn't address the question of getting a patent extension, however. If this is true, then Dr. Lias needs to be made aware of it.
FU #323010:
Just a few more general impressions from the meeting today. Like the rest of you, I was very disappointed in the approach to marketing the IP. On the one hand, Dr. Lias said that there were on-going discussions that he could not talk about, but he continually down played any expectation that we would receive much value for it. It was almost like they have already given up and have a 'Hey, we tried, but nobody is interested' attitude. They certainly didn't spell out any detailed marketing plan.
On the CDMO front, they generally impressed me as qualified, and will probably do a fine job, but I almost choked when Lias said that Tracy Kinjersky "hit the ground running". Hopefully, she will not emulate Mary Boyd.
The tour of the Myford facility was as impressive as ever. The 2 new 2000L reactors are installed, tested and ready to go, but have not yet been used in production. Someone asked how many employees we have now and we were told about 200, of which 30 something (I don't remember the exact number) are in production. I asked, if only 30 something are in production, what do the others do? I was told quality control, testing, etc., which I would consider as part of production. This brought the number up to about 75. Unfortunately, we were interrupted, and I wasn't able to pursue this breakdown any further.
It seems reasonable to me that we can get to $200mm in sales, but it will be at least 2-3 years down the road. They already have space in the Myford building to build out a duplicate of the existing facility, except that I believe it will use all 2000L reactors. But this space is still raw warehouse space, and will take a minimum of a year to build. And they won't start building it until they have reached significant profits from the exiting facility. So it all hinges on how fast they can attract new clients.
FU#323064:
re: How many in meeting? I didn't actually count, but I was going to guess that there were probably about 20 shareholders, and we were matched, if not outnumbered, by company employees. My impression was that it was a bit smaller attendance by shareholders than in years past.
By: Holotawoopas 1/18/18 4:49pmET #322902
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=137790910
For those who have pinned their hopes on the value of the IP it is time to mourn your loss and move on as there was nothing concrete about any potential monetizing extrapolated at the ASM. N40 gave a valiant effort to plead the case of value of the IP but IMO it fell on deaf ears! The CEO said no BP interest has been received!
FU#322909:
The CEO stated that the biggest down fall to monetizing the IP has nothing to do with leverage but has everything to do with patent expiration!!!
By: Eb0783 1/18/18 1:45pmET #322864
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=137783413
ASM votes: #1 all re-elected, #2 for 36.4mm, #3 for 15.2 v. against 3.6, #4 1 year
FU#322879:
Per Lias, there is no formal process, group, or investment bank in place to sell the IP.
FU#322934:
Lias said he wasn't a patent expert when made those comments about short patent life being an issue. It was obvious he wasn't aware that patents could be extended due to years lost in development. Some of us took that short patent life excuse with a grain of salt.
FU#323063:
re: How many in meeting? I counted about 40 at the start of the meeting. I would guess 50%-60% were shareholders. That is about normal from my experience. I remember counting 40 & 50 in previous ASMs.
FU#323066:
The thing is, if they are really not having a process, group, or IB in place to evaluate/sell our IP, they don't have to spend anything to do it. At the very least, give the task to some IB or 3rd party expert on contingency: you can keep 50% of whatever you can sell it for. (You lawyers often take cases on the same basis and accept 30, 40, 50% quite often). I said that to Lias after the meeting, with Ziebell & Lytle standing there - no comment from them. I find their stance on this incredulous, if true!
By: Djohn 1-19-18 2:18pmET #323067
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=137818937
MY THOUGHTS AFTER THE ASM: I missed the fancy danish, the cream cheese and bagels also missed the fancy hotel backdrop making me think we were on the verge of something astronomical. What did not miss was my thoughts of how many shares had to be sold to pay for me to bite on my bagel, sip on my fine coffee while looking at a beautiful swimming pool at a posh hotel.
It’s amazing what can be done with a couple of cases of bottled water, one pot of coffee and a room with chairs. That alone convinced me our new leadership is serious about how they spend the shareholders money!
For me the ASM went as I expected. It just confirmed to me that we are no longer chasing a dream. We no longer have a questionable cast of characters spending our money diluting our shares. It’s pretty easy, we are a manufacturing company now with basic straight forward business model. We have people in place who are qualified to grow this business and IMO recoup the total amount I invested in PPHM maybe even more.
I have given up the dream! I will awake daily and put on a BaviDermCQ patch. I will develop a 12-step program to wean myself from astronomical thoughts. One of those steps will be to not visit this board as often. Another step will be to fully investigate the members of the BOD in any company I ever invest in again. One other will be to take full responsibility for my investment decisions and accept how they turn out.
I bugged out right after the ASM ended and didn’t stick around for the tour of Avid. I had a flight to catch. Only one flight I could get to make it home that day. Maybe some of you there could gather more insight in talking with BOD or management during the tour. I went on the Avid tour last year and was impressed with the facilities.
FWIW This is what I gathered and is my opinion about the current situation. I saw a group of highly qualified individuals both on the new BOD and new leadership for Avid. These individuals are try to make lemonade out of lemons. Face it folks, after years and years of being told we had a bag of diamonds but we ended up with a bag of lemons! I am not saying that PS/Bavi/IP is worthless. Nobody really knows! Yet! but because the leadership of legacy PPHM did not do the correct things to prove it’s worth, we are left with an IP in shambles. After spending several hundreds of millions they left us with no value for the IP and the only way to prove value of the IP is to spend many more millions. ( A Bag of Lemons) Basically, after Sunrise failure the PPHM leadership decided to spend millions more to search around in the bag of lemons to see if they could just one diamond. We can all speculate on why legacy PPHM leadership continued down their path.
Fact to me is last year PPHM had two things. An R&D division with the IP and faced with spending millions more to prove some worth with low chance of success, even lower based on past successes. A CDMO business that with the right leadership and direction could become a nice profitable business in a few years. The chance of success with CDMO is very high IMO and I think valued much higher than R&D portion.
I think Ronin saw the CDMO business as a good development opportunity and took it. Real business people made real business decisions with maximum potential and maximum returns in mind. C'est la vie PPHM HELLO CDMO
FU#323070:
Don't get me wrong. I am not saying IP does not have value! I bought TCLN because I believed it did have value. I could hear the enthusiasm in Dr Thorpe's voice when he presented it. That was powerful! I just think the IP has been squandered all these years and nobody knows how much more money must be spent to realize it's real worth and with failed PIII no one is willing to take a chance on it. Hence, It's not worth much at this point in time. I think best path forward is CDMO to recoup invested money.
By: Aikifredicist 1-18-18 8:16pmET #322961
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=137795041
I got up at the meeting and asked about Exosomes, but Lias just fudged on the answer. He doesn’t know what he has.
= = = = = = = = = = = = = = =RECALL:
1-8-17: EBD's Biotech Showcase 2018 (parallel w/JPM Conf.), SanFran https://ebdgroup.knect365.com/biotech-showcase
...1-2-18 PR: https://tinyurl.com/ydcc9agv
...3:30pmPT: CEO Roger Lias, Corporate Presentation
...1-8-18 Lias BioShowcase’18 Presentation PDF (24 slides): https://tinyurl.com/ya6tgxxa
FULL HISTORY of PPHM-Ronin PR’s, Letters, 13-D’s, Form4’s, Proxy’s, etc: https://tinyurl.com/ycb3fpfm
12-26-17: Roger Lias replaces Steven King as Pres./CEO; in process of changing name to Avid Bio. and new Nasdaq Ticker ("early 2018") https://tinyurl.com/yb34e2t8
12-11-17: Qtly. Conf. Call (Lias/Lytle) Transcript https://tinyurl.com/ybycb2s6
...Dr. Lias, "the company is undergoing a broad-scale transformation, the goals of which are to shift complete focus to the Avid Bioservices CDMO business and the complete divestiture of all of Peregrine's legacy R&D assets, which include bavituximab."
I do hope we'll get more Attendee Reports/Followups.
I'll keep adding them to the compilation, and re-posting as makes sense.
LATEST ASM Post:
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=137805841
1-18-18/2017 ASM: Roger Lias’ Slideshow & Attendee Reports
...Attendee Reports are after the Slides. I will update & re-post as more come in over the next few days.
1-18-2018: Avid Bioservices’ 2017 Annual Shareholder’s Mtg http://ir.avidbio.com/events.cfm
…10amPT at Avid’s Myford Facility, 14191 Myford Road, Tustin, CA 72780
Webcast https://edge.media-server.com/m6/p/ij9i8ypx
...PROXY/14A: https://tinyurl.com/y7qprpg9 VotingRecDate=11-27-17
CEO Roger Lias’ ASM Presentation Slides (1-18-18):














ATTENDEE REPORTS (2017 ASM 1-18-18):
By: Hawkfan1 (Michael) 1-18-18 9:41pmET #322974
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=137796412
Here are some of my notes taken at the ASM. They are very brief paraphrases, since I never took shorthand in school.
As usual, Brandon Cox was the first to take the microphone with a long list of questions, that at least this shareholder very much appreciated.
Q. You are burning $3-4mm/qtr, with no cash and no back log. How do you plan to raise capital?
A. Your estimate of our burn rate is in the right ball park. We do have $33mm in backlog, but that is quite conservative. Most companies recognize all of an order when it is placed, but traditionally, PPHM has only recognized backlog as work orders for that order are placed. But clearly, we need more capital. We must eliminate the 'going concern' clause. While it may not be ideal for a small biotech, it is probably something they can live with. But for a manufacturing company, it is a death knell. Customers need to know that you are going to be around for the long term.
Q. How dilutive will it be?
A. We don't know yet. We don't know how much we will need yet.
Q. Is anyone from the old management team working to help sell the R&D technology?
A. Yes, Shelly. And others are working as consultants.
Q. Most CDMO's are private companies. Are there any plans to take the company private?
A. Not currently. (He did mention, kind of jokingly, that it would be nice).
Q. Who are the major institutional investors?
A. Still Ronan, Tappan, and Eastern Capital.
Q. Will they be involved in the capital raise?
A. Hopefully.
Q. What is the cost of being a public company?
A. About $2-3mm/year.
Q. As a strictly mfg. company, do we really need 7 board members?
A. Right now, the experience that they all bring is extremely helpfull. Down the road, maybe not.
Q. How much cash do we currently have?
A. $27.7mm. (Not quite sure I got this right, but I think that $10mm was in customer deposits). [NOTE: the $27.7mm is a/o 10-31-17, the last 10-Q (Deposits were $13.1mm): https://tinyurl.com/ybycb2s6 ]
Q. Will Avid expand beyond the earthquake prone Tustin area?
A. It could be done relatively easily. Down the road, it will be evaluated.
Then Brandon relinquished the mike and North40k gave a long and impassioned commentary about various trials and people (such as Dr. Wolchok) that the company should contact about Bavituximab. He mentioned Wolchok's comment that his next trial "depends on the company".
Dr. Lias stated that Dr. Wolchok has not contacted the company. He mentioned that an associate of Dr. Wolchok had said that it will be 18 mos. before the next trial proceeds. (This was met with incredulity from several shareholders in attendance).
Dr. Lias stated that they have numerous discussions regarding CAR-T, but there was no interest. In some other discussions, they found our technology interesting, but then said "prove it in a trial".
N40k then asked if the recent Halozyme trial failure would affect Avid. It will not.
Brandon Cox then resumed his questioning. He asked a question regarding the IP patent clock, which I previously reported. For continuity, Dr. Lias's answer was that as patents start to expire, and a BP realizes that it will take about $70mm and 3-4 years to bring a product to market, a 5-year patent life is not that exciting to them.
Q. How much Bavi do we have in storage?
A. Enough for a couple of small trials, nothing big.
Q. Client #1 is Halozyme. We don't know the name of client #2, but are they gone for good?
A. No, but I don't expect them to be hugely successful. They will be years down the road.
Q. Question regarding the interest in the Exosome test kit.
A. Not much commercial interest.
Q. Are you trying to sell the IP in bits and pieces or as a package deal?
A. While a package deal would be ideal, not gonna happen.
Then Greg got up and asked about the process of sell the IP. Dr. Lias commented that there was not much interest. The previous board has talked to everyone - it is not a secret - just not much interest.
Then N40k asked the board to contact Precision for Medicine, which concluded the Q&A.
Again, this is very roughly paraphrased from my notes. Other members that were in attendance, please correct anything that I may have inadvertently gotten wrong.
As I decided to take the tour, I was not able to talk to the board members one on one after the meeting.
BY: HAWKFAN1 (MICHAEL)
... 1-18-18 7:54pmET #322954 (INITIAL POST)
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=137794640
Dr. Lias comment about the lack of interest in the IP by BP was in response to a question by Brandon Cox about the declining IP clock. His example was that if BP knows that it will take about $70M and 3-4 years of trials to get a drug to market, then a 5-year patent life is just not that exciting to them. This doesn't address the question of getting a patent extension, however. If this is true, then Dr. Lias needs to be made aware of it.
FU #323010:
Just a few more general impressions from the meeting today. Like the rest of you, I was very disappointed in the approach to marketing the IP. On the one hand, Dr. Lias said that there were on-going discussions that he could not talk about, but he continually down played any expectation that we would receive much value for it. It was almost like they have already given up and have a 'Hey, we tried, but nobody is interested' attitude. They certainly didn't spell out any detailed marketing plan.
On the CDMO front, they generally impressed me as qualified, and will probably do a fine job, but I almost choked when Lias said that Tracy Kinjersky "hit the ground running". Hopefully, she will not emulate Mary Boyd.
The tour of the Myford facility was as impressive as ever. The 2 new 2000L reactors are installed, tested and ready to go, but have not yet been used in production. Someone asked how many employees we have now and we were told about 200, of which 30 something (I don't remember the exact number) are in production. I asked, if only 30 something are in production, what do the others do? I was told quality control, testing, etc., which I would consider as part of production. This brought the number up to about 75. Unfortunately, we were interrupted, and I wasn't able to pursue this breakdown any further.
It seems reasonable to me that we can get to $200mm in sales, but it will be at least 2-3 years down the road. They already have space in the Myford building to build out a duplicate of the existing facility, except that I believe it will use all 2000L reactors. But this space is still raw warehouse space, and will take a minimum of a year to build. And they won't start building it until they have reached significant profits from the exiting facility. So it all hinges on how fast they can attract new clients.
By: Holotawoopas 1/18/18 4:49pmET #322902
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=137790910
For those who have pinned their hopes on the value of the IP it is time to mourn your loss and move on as there was nothing concrete about any potential monetizing extrapolated at the ASM. N40 gave a valiant effort to plead the case of value of the IP but IMO it fell on deaf ears! The CEO said no BP interest has been received!
FU#322909:
The CEO stated that the biggest down fall to monetizing the IP has nothing to do with leverage but has everything to do with patent expiration!!!
By: Eb0783 1/18/18 1:45pmET #322864
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=137783413
ASM votes: #1 all re-elected, #2 for 36.4mm, #3 for 15.2 v. against 3.6, #4 1 year
FU#322879:
Per Lias, there is no formal process, group, or investment bank in place to sell the IP.
FU#322934:
Lias said he wasn't a patent expert when made those comments about short patent life being an issue. It was obvious he wasn't aware that patents could be extended due to years lost in development. Some of us took that short patent life excuse with a grain of salt.
By: Aikifredicist 1-18-18 8:16pmET #322961
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=137795041
I got up at the meeting and asked about Exosomes, but Lias just fudged on the answer. He doesn’t know what he has.
= = = = = = = = = = = = = = =RECALL:
1-8-17: EBD's Biotech Showcase 2018 (parallel w/JPM Conf.), SanFran https://ebdgroup.knect365.com/biotech-showcase
...1-2-18 PR: https://tinyurl.com/ydcc9agv
...3:30pmPT: CEO Roger Lias, Corporate Presentation
...1-8-18 Lias BioShowcase’18 Presentation PDF (24 slides): https://tinyurl.com/ya6tgxxa
FULL HISTORY of PPHM-Ronin PR’s, Letters, 13-D’s, Form4’s, Proxy’s, etc: https://tinyurl.com/ycb3fpfm
12-26-17: Roger Lias replaces Steven King as Pres./CEO; in process of changing name to Avid Bio. and new Nasdaq Ticker ("early 2018") https://tinyurl.com/yb34e2t8
12-11-17: Qtly. Conf. Call (Lias/Lytle) Transcript https://tinyurl.com/ybycb2s6
...Dr. Lias, "the company is undergoing a broad-scale transformation, the goals of which are to shift complete focus to the Avid Bioservices CDMO business and the complete divestiture of all of Peregrine's legacy R&D assets, which include bavituximab."
1-18-18/2017 ASM: R.Lias’ Presentation (Slideshow)
PS: Attendee Reports will be added as they come in.
1-18-2018: Avid Bioservices’ 2017 Annual Shareholder’s Mtg http://ir.avidbio.com/events.cfm
…10amPT at Avid’s Myford Facility, 14191 Myford Road, Tustin, CA 72780
11-13-17/PR: http://tinyurl.com/y9tdcob5
Webcast https://edge.media-server.com/m6/p/ij9i8ypx
...PROXY/14A: https://tinyurl.com/y7qprpg9 VotingRecDate=11-27-17
CEO Roger Lias’ ASM Presentation Slides (1-18-18):














= = = = = = = = = = = = = = =FYI:
1-8-17: EBD's Biotech Showcase 2018 (parallel w/JPM Conf.), SanFran https://ebdgroup.knect365.com/biotech-showcase
...1-2-18 PR: https://tinyurl.com/ydcc9agv
...3:30pmPT: CEO Roger Lias, Corporate Presentation
...1-8-18 Lias BioShowcase’18 Presentation PDF (24 slides): https://tinyurl.com/ya6tgxxa
ASM Webcast in 10mins or so, after they get everybody settled in and the opening formalities out of the way.
Hoping for positive vibes about the IP Sale and building Avid business...
Jan18: Avid Bioservices’ 2017 Annual Shareholder’s Mtg
11-13-17/PR: http://tinyurl.com/y9tdcob5 - http://ir.avidbio.com/events.cfm
10amPT(1pmET) at Avid’s Myford Facility, 14191 Myford Road, Tustin, CA 72780
Webcast https://edge.media-server.com/m6/p/ij9i8ypx
...PROXY/14A: https://tinyurl.com/y7qprpg9 VotingRecDate=11-27-17 Vote here: https://www.proxyvote.com
= = = = = = = = = = = =
1-8-17: EBD's Biotech Showcase 2018 (parallel w/JPM Conf.), SanFran
3:30pmPT: CEO Roger Lias, Corporate Presentation
...Presentation PDF (24 slides): https://tinyurl.com/ya6tgxxa
CEO Roger Lias’s 1-8-18 Presentation PDF (24 Slides) added to the Events website ( http://ir.avidbio.com/events.cfm )
Jan8-10 2018: “EBD’s Biotech Showcase 2018”, SanFran
“Biotech Showcase is an investor & networking conference devoted to providing private and public biotechnology & life sciences companies with an opportunity to present to, and meet with, investors, and pharmaceutical exec’s in one place during the course of one of the industry's largest annual healthcare investor conferences (JPM's HC Conf. '18).
https://ebdgroup.knect365.com/biotech-showcase
1-8-18 3:30pmPT: Roger Lias (Avid’s CEO), Corp. Presentation
**Presentation PDF (24 slides):
http://files.shareholder.com/downloads/PPHM/5841800040x0x968677/04F775C3-682A-414B-94E0-FE7C2622AF7B/Avid_Corporate_Presentation_Final_1.8.18.pdf
New iHub Link: http://investorshub.advfn.com/Avid-Bioservices-Inc-CDMO-2076/
Update your browser Favorite/Bookmark!
We’ll get it all fixed on Monday after it goes Live. No worries now, N40k. DELETE
"An updated corporate website featuring a new logo will be available at http://www.avidbio.com as of market open on Monday, Jan. 8, 2018."
This from the end of the "ABOUT AVID" section of today's PR.
https://tinyurl.com/y8vhjow4
Link to Jan18 ASM Webcast added to website (Events). I don't see anything about CEO Roger Lias' Jan8/Monday presentation at "Biotech Showcase 2018" being webcast.
KNOWN UPCOMING:
Jan8/Monday: EBD's Biotech Showcase 2018 (parallel w/JPM Conf.), SanFran https://ebdgroup.knect365.com/biotech-showcase
...1-2-18 PR: https://tinyurl.com/ydcc9agv
...3:30pmPT: CEO Roger Lias, Corporate Presentation
Jan18: Avid Bioservices’ 2017 Annual Shareholder’s Mtg 11-13-17/PR: http://tinyurl.com/y9tdcob5
…10amPT at Avid’s Myford Facility, 14191 Myford Road, Tustin, CA 72780 http://ir.peregrineinc.com/events.cfm
…….Webcast https://edge.media-server.com/m6/p/ij9i8ypx
...PROXY/14A: https://tinyurl.com/y7qprpg9 VotingRecDate=11-27-17 Vote here: https://www.proxyvote.com
= = = = = = = = =
1-8-18: Name=>AVID, Tickers=CDMO/CDMOP, History of CDMO Transition
“...Effective at market open on Monday, Jan. 8, 2018, trading for Avid Bioservices will begin under the symbol “CDMO”… Preferred stock will begin trading under “CDMOP”… The corp. name change to Avid Bioservices does not affect the rights of the company’s stockholders and no action is required by stockholders… Outstanding stock certificates are not affected by the name change and will not need to be exchanged.” https://tinyurl.com/y8vhjow4
- - - - - - - -
Full History of CDMO Transition & Ronin: PR’s, Letters, 13-D’s, Form4’s, and Proxy’s https://tinyurl.com/ycb3fpfm
1-8-18: Name=>AVID, Tickers=CDMO/CDMOP, History of CDMO Transition
“...Effective at market open on Monday, Jan. 8, 2018, trading for Avid Bioservices will begin under the symbol “CDMO”… Preferred stock will begin trading under “CDMOP”… The corp. name change to Avid Bioservices does not affect the rights of the company’s stockholders and no action is required by stockholders… Outstanding stock certificates are not affected by the name change and will not need to be exchanged.” https://tinyurl.com/y8vhjow4 (More: See Below)
- - - - - - - -
See below for full PPHM-RONIN History of PR’s, Letters, 13-D’s, Form4’s, and Proxy’s (sorted: newest=>oldest).
KNOWN UPCOMING:
Jan8: EBD's Biotech Showcase 2018 (parallel w/JPM Conf.), SanFran https://ebdgroup.knect365.com/biotech-showcase
...1-2-18 PR: https://tinyurl.com/ydcc9agv
...3:30pmPT: CEO Roger Lias, Corporate Presentation
Jan18: Avid Bioservices’ 2017 Annual Shareholder’s Mtg 11-13-17/PR: http://tinyurl.com/y9tdcob5
…10amPT at Avid’s Myford Facility, 14191 Myford Road, Tustin, CA 72780 http://ir.peregrineinc.com/events.cfm
…….Webcast https://edge.media-server.com/m6/p/ij9i8ypx
...PROXY/14A: https://tinyurl.com/y7qprpg9 VotingRecDate=11-27-17 Vote here: https://www.proxyvote.com
= = = = = = = = = = = = = = = = = = = = = = = = = = = = = = =
11-13-17: Large Ownership Summary(40.2%) - now 18,163,171shs., 40.2% of 45,210,608 O/S at 11-27-17.
(Ronin+SW/Stafford+Farley+White, Eastern Cap./K.Dart, Institutions incl. Tappan’s 11-13-17/13G)
#1: 10-27-17/PR: Group Ronin Trading/SWInvest (John Stafford III+R.Farley+Stephen White) now has 9.6% stake (4,325,889sh.) in PPHM
...4,173,391 COMMON – 9.2% of 45,210,608 common O/S at 11-27-17 (total beneficial=4,325,889 if Pref. conv. x1.1905 to Common)
...128,099 PREFERRED – 7.8% of 1,647,760 preferred O/S at 11-27-17
NOTE: ALL historical common #’s adjusted for the 1:7 R/S eff. 7-10-17.
#2: 10-30-15: Kenneth Dart (Eastern Capital) acquires 9.5% stake (4,300,992sh.) in PPHM http://tinyurl.com/y95yskck
...3,777,183 COMMON – 8.4% of 45,210,608 common O/S at 11-27-17 (total beneficial=4,300,992 if Pref. conv. x1.1905 to Common, 9.5%)
...440,000 PREFERRED – 26.7% of 1,647,760 preferred O/S at 11-27-17
…...9-29-17/Form4: http://tinyurl.com/y9oadloa <=No chg. In Ownership; status update?
RONIN/SW 13D SUMMARY:
13D TransDates COMMON-CHG PREF-CHG ENDING-COMMON ENDING-PREF.
3-2-17 1/20/17-3/1/17 +2,947,425 +51,364 2,947,425 51,364 http://tinyurl.com/jr42u23
3-10-17 3/2/17-3/9/17 +433,509 +25,661 3,380,934 77,025 http://tinyurl.com/ydxra96u
4-17-17 3/28/17-4/10/17 0 +23,334 3,380,934 100,359 http://tinyurl.com/lanjddc
5-19-17 5/1/17-5/17/17 0 +23,140 3,380,934 123,499 http://tinyurl.com/mgnn92x
6-20-17 3/10/17-6/16/17 +378,170 0 3,759,105 123,499 http://tinyurl.com/y76q5rqu
6-29-17 6/21/17 +7,143 0 3,766,248 123,499 http://tinyurl.com/y9sp8bfv
7-14-17 6/29/17-7/7/17 +34,891 +3,600 3,801,139 127,099 http://tinyurl.com/ybra4s69
8-29-17 8/4/17-8/15/17 +27,252 0 3,828,391 127,099 http://tinyurl.com/yay55u3p (14A)
10-10-17 8/16/17-10/10/17 +45,000 0 3,873,391 127,099 http://tinyurl.com/ybvkjd3e (14A)
10-17-17 10/13/17-10/16/17 +109,573 +1,000 3,982,964 128,099 http://tinyurl.com/y983myco
10-27-17 10/17/17-10/27/17 +190,427 0 4,173,391 128,099 http://tinyurl.com/y7hslequ (14A)
SPLITOUT 13D GROUP into Ronin(John Stafford III, R.Farley) & SW-Partners(Stephen White):
Ronin Trading (Stafford) 3,173,391 115,299 (beneficial=3,310,652)
Ronin’s Roger Farley 300,000 1,000 (beneficial=301,190)
SW-Partners (White) 700,000 11,800 (beneficial=714,047)
- - - - - - - - - - - - - - -
Plus, INSTITUTIONS a/o *9-30-17: 9,536,290sh. = 21.1% <=*incl. Tappan’s 11-13-17/13G
...8,491,679 + 1,044,611(Tappan adds after 9-30-17) = 9,536,290
http://www.nasdaq.com/symbol/pphm/institutional-holdings
TOP13:
Tappan St. Partners 3,915,611 +2,259,359 <=per 11-13-17/13G(see below**)
Vanguard Group 1,518,110 +488,355
Blackrock (Larry Fink) 761,302 -44,720
Bandera Partners 555,000 +351,996
Renaissance Technologies 547,190 +66,030
Geode Capital Mgt. 225,676 -3
Fondren Mgt. 205,000 NEW
Stifel Financial 204,305 +200,762
Eqis Capital Mgt. 143,595 -6,804
Wells Fargo & Co. 117,829 +69,488
Connor Clark & Lunn 98,772 NEW
Brown Advisory Sec. 82,858 -1
Pdt Partners 82,800 NEW
And this big Q3 drop:
#18 Kennedy Capital Mgt. 69,820 -1,324,246
**11-13-17/13G: Tappan Street (Prasad Phatak) http://tinyurl.com/y85pz4fs
2 Funds + P.Phatak’s personal shares:
...Partners LLC, Tappan St. Fund L.P. 1,540,000 0
...Tappan St. Partners Ideas Fund L.P. 2,289,504 +1,597,927 since 8-14-17
...Prasad Phatak (Principle Owner) 86,107 +19,000 since 8-14-17
Total TAPPAN: 3,915,611 (8.7%)
Note: Tappan Street (total) Holdings history:
a/o 3-31-17: 914,304
a/o 6-30-17: 1,656,252
a/o 8-14-17: 2,298,684 5.1% (of 45,069,188 O/S as of 7-11-17)
a/o 9-30-17: 2,871,000 6.4% (of 45,096,081 O/S as of 9-11-17)
a/o 11-6-17: 3,915,611 8.7% (of 45,096,081 O/S as of 9-11-17)
Final Qtly Inst. Holdings from Nasdaq:
3-31-17: 5,690,888
6-30-17: 6,834,425
9-30-17: 8,491,679

Lias Presenting MONDAY, not Wednesday. DELETE
Map of The Avid & Peregrine Campus (Tustin)
...To the best of my knowledge (with Eb0783 & Hawkfan1’s help).
Avid’s Myford Facility: 14191 Myford Rd., Tustin, CA 92780 <=1-18-18 ASM here
Michelle Offices: 2642 Michelle Dr., Suite 200, Tustin, CA 92780
PPHM’s Hqtrs & Avid I(Franklin): 14282 Franklin Ave., Tustin, CA 92780
http://avidbio.com <= http://www.peregrineinc.com

1-18-18: Peregrine's 2017 Annual Shareholder’s Meeting
11-13-17/PR: http://tinyurl.com/y9tdcob5 VotingRecDate=11-27-17
…10am at Avid’s Myford Facility, 14191 Myford Rd., Tustin, CA 72780
Vote here: https://www.proxyvote.com

Map of The Avid & Peregrine Campus (Tustin)
...To the best of my knowledge (with Eb0783 & Hawkfan1’s ASM'16 help)!
Avid’s Myford Facility: 14191 Myford Rd., Tustin, CA 92780 <=1-18-18 ASM here
Michelle Offices: 2642 Michelle Dr., Suite 200, Tustin, CA 92780
PPHM’s Hqtrs & Avid I(Franklin): 14282 Franklin Ave., Tustin, CA 92780
http://avidbio.com <= http://www.peregrineinc.com


12-26-17 R.Lias Appointed Pres./CEO, History of CDMO Transition
“...Dr. Roger Lias succeeds Steven W. King, who resigned as Pres./CEO of Peregrine to pursue other professional interests… Peregrine is in the process of officially changing the company's name to Avid Bioservices and adopting a new NASDAQ ticker. The company expects this process to be completed in early 2018...” (More: See Below)
- - - - - - - -
See below for full PPHM-RONIN History of PR’s, Letters, 13-D’s, Form4’s, and Proxy’s (sorted: newest=>oldest).
KNOWN UPCOMING:
Jan18: Peregrine's 2017 Annual Shareholder’s Meeting – 11-13-17/PR: http://tinyurl.com/y9tdcob5 VotingRecDate=11-27-17; PROXY/14A: https://tinyurl.com/y7qprpg9
…10am at Avid’s Myford Facility, 14191 Myford Rd., Tustin, CA 72780
= = = = = = = = = = = = = = = = = = = = = = = = = = = = = = =
11-13-17: Large Ownership Summary(40.2%) - now 18,163,171shs., 40.2% of 45,210,608 O/S at 11-27-17.
(Ronin+SW/Stafford+Farley+White, Eastern Cap./K.Dart, Institutions incl. Tappan’s 11-13-17/13G)
#1: 10-27-17/PR: Group Ronin Trading/SWInvest (John Stafford III+R.Farley+Stephen White) now has 9.6% stake (4,325,889sh.) in PPHM
...4,173,391 COMMON – 9.2% of 45,210,608 common O/S at 11-27-17 (total beneficial=4,325,889 if Pref. conv. x1.1905 to Common)
...128,099 PREFERRED – 7.8% of 1,647,760 preferred O/S at 11-27-17
NOTE: ALL historical common #’s adjusted for the 1:7 R/S eff. 7-10-17.
#2: 10-30-15: Kenneth Dart (Eastern Capital) acquires 9.5% stake (4,300,992sh.) in PPHM http://tinyurl.com/y95yskck
...3,777,183 COMMON – 8.4% of 45,210,608 common O/S at 11-27-17 (total beneficial=4,300,992 if Pref. conv. x1.1905 to Common, 9.5%)
...440,000 PREFERRED – 26.7% of 1,647,760 preferred O/S at 11-27-17
…...9-29-17/Form4: http://tinyurl.com/y9oadloa <=No chg. In Ownership; status update?
RONIN/SW 13D SUMMARY:
13D TransDates COMMON-CHG PREF-CHG ENDING-COMMON ENDING-PREF.
3-2-17 1/20/17-3/1/17 +2,947,425 +51,364 2,947,425 51,364 http://tinyurl.com/jr42u23
3-10-17 3/2/17-3/9/17 +433,509 +25,661 3,380,934 77,025 http://tinyurl.com/ydxra96u
4-17-17 3/28/17-4/10/17 0 +23,334 3,380,934 100,359 http://tinyurl.com/lanjddc
5-19-17 5/1/17-5/17/17 0 +23,140 3,380,934 123,499 http://tinyurl.com/mgnn92x
6-20-17 3/10/17-6/16/17 +378,170 0 3,759,105 123,499 http://tinyurl.com/y76q5rqu
6-29-17 6/21/17 +7,143 0 3,766,248 123,499 http://tinyurl.com/y9sp8bfv
7-14-17 6/29/17-7/7/17 +34,891 +3,600 3,801,139 127,099 http://tinyurl.com/ybra4s69
8-29-17 8/4/17-8/15/17 +27,252 0 3,828,391 127,099 http://tinyurl.com/yay55u3p (14A)
10-10-17 8/16/17-10/10/17 +45,000 0 3,873,391 127,099 http://tinyurl.com/ybvkjd3e (14A)
10-17-17 10/13/17-10/16/17 +109,573 +1,000 3,982,964 128,099 http://tinyurl.com/y983myco
10-27-17 10/17/17-10/27/17 +190,427 0 4,173,391 128,099 http://tinyurl.com/y7hslequ (14A)
SPLITOUT 13D GROUP into Ronin(John Stafford III, R.Farley) & SW-Partners(Stephen White):
Ronin Trading (Stafford) 3,173,391 115,299 (beneficial=3,310,652)
Ronin’s Roger Farley 300,000 1,000 (beneficial=301,190)
SW-Partners (White) 700,000 11,800 (beneficial=714,047)
- - - - - - - - - - - - - - -
Plus, INSTITUTIONS a/o *9-30-17: 9,536,290sh. = 21.1% <=*incl. Tappan’s 11-13-17/13G
...8,491,679 + 1,044,611(Tappan adds after 9-30-17) = 9,536,290
http://www.nasdaq.com/symbol/pphm/institutional-holdings
TOP13:
Tappan St. Partners 3,915,611 +2,259,359 <=per 11-13-17/13G(see below**)
Vanguard Group 1,518,110 +488,355
Blackrock (Larry Fink) 761,302 -44,720
Bandera Partners 555,000 +351,996
Renaissance Technologies 547,190 +66,030
Geode Capital Mgt. 225,676 -3
Fondren Mgt. 205,000 NEW
Stifel Financial 204,305 +200,762
Eqis Capital Mgt. 143,595 -6,804
Wells Fargo & Co. 117,829 +69,488
Connor Clark & Lunn 98,772 NEW
Brown Advisory Sec. 82,858 -1
Pdt Partners 82,800 NEW
And this big Q3 drop:
#18 Kennedy Capital Mgt. 69,820 -1,324,246
**11-13-17/13G: Tappan Street (Prasad Phatak) http://tinyurl.com/y85pz4fs
2 Funds + P.Phatak’s personal shares:
...Partners LLC, Tappan St. Fund L.P. 1,540,000 0
...Tappan St. Partners Ideas Fund L.P. 2,289,504 +1,597,927 since 8-14-17
...Prasad Phatak (Principle Owner) 86,107 +19,000 since 8-14-17
Total TAPPAN: 3,915,611 (8.7%)
Note: Tappan Street (total) Holdings history:
a/o 3-31-17: 914,304
a/o 6-30-17: 1,656,252
a/o 8-14-17: 2,298,684 5.1% (of 45,069,188 O/S as of 7-11-17)
a/o 9-30-17: 2,871,000 6.4% (of 45,096,081 O/S as of 9-11-17)
a/o 11-6-17: 3,915,611 8.7% (of 45,096,081 O/S as of 9-11-17)
Final Qtly Inst. Holdings from Nasdaq:
3-31-17: 5,690,888
6-30-17: 6,834,425
9-30-17: 8,491,679

Huts, the Bavi+Durva Ph2/Trial listed in that 11-2017 presentation was the originally planned PPHM/AZN trial from late 2015, that was withdrawn after Ph3/SUNRISE was stopped for Futility in 2-2016.
“Ph2 Trial of Durvalumab(“Imfinzi”)+Bavituximab in Patients With Prev. Treated Metastatic NSCLC”
7-30-16: “This study has been withdrawn prior to enrollment.”
https://www.clinicaltrials.gov/ct2/show/NCT02673814
10-15-15: Peregrine & AstraZeneca Expand Collab. w/Ph2/2ndLine-NSCLC Trial, Bavi+durvalumab(“Imfinzi”) http://tinyurl.com/q79bkam
= = = = = = =
11-2017 Pg.13: “How Big Data Will Disrupt Business Models”
http://www.himss.eu/sites/himsseu/files/community/community_presentations/impact17_Carl_Janssen.pdf
Hayward, DEC.2017 Company Update - "Transitioning to a Pure-Play CDMO, Avid Bioservices, Inc."
12-11-17 10-Q for q/e 10-31-17: https://tinyurl.com/y75kwojs
Business Description – We are a company committed to improving the lives of patients by manufacturing pharmaceutical products through our CDMO, Avid Bioservices, Inc.
"Over the next 60 days, we plan to complete the transition from a R&D company to a dedicated CDMO company focused on dev. & mfg. of biopharmaceutical products derived from mammalian cell culture.
We plan to take ADDL. STEPS steps over the near term, including but not limited to:
1. Rebranding the company as Avid Bioservices, Inc.
2. Changing our ticker symbol on the NASDAQ Capital Market to align with our rebranding efforts
3. Broadening our sales force
4. Increasing our marketing efforts to support our rebranding & vision
5. Completing the wind down of all R&D activities and the potential licensing/divestiture of our assets related to our R&D operations.”
$51,000,000, Loofman. I need that surPRISE!
2 Jobs added: Dir/BusDev/EASTCOAST, VP/Proc.Sciences
https://peregrineinc.applicantpro.com/jobs
Manager/Director, Business Development - East Coast Based
13-Dec-2017 – 28-Feb-2018
ESSENTIAL DUTIES AND RESPONSIBILITIES INCLUDE THE FOLLOWING:
Other duties may be assigned:
Grow revenues & increase the company's market share and book of business within the biopharmaceutical/biotechnology CDMO industry by targeting and meeting with prospective clients to sell the advantages of partnering with Avid. . .
Vice President, Process Sciences
13-Dec-2017 – 28-Feb-2018
SUMMARY: This role is the top executive overseeing the process sciences function, which includes design and scale-up processes. . .
= = = = =
Avid President Dr. Lias said this 12-11-17 in his opening CC remarks: https://tinyurl.com/ybycb2s6
“In her new role, Tracy Kinjerski (VP/Bus.Operations) is charged with expanding the company's client base and driving growth at Avid. In addition to her responsibilities in business dev., including strengthening our presence in the Eastern United States, Tracy will strengthen Avid's project mgt. and client relationship operations and systems and will direct our marketing & promotional activities...”
http://avidbio.com/team
Then why didn't you say He IMPLIED it?
PL did not say ANYTHING about using ATM in the CC.
PL 12-11-17/CC: “...we plan to raise addl. capital within the next 6 months to support our continued operations and other initiatives that will enhance our CDMO operations. The amount of capital we need to raise will depend on a number of factors, including, the amount of new business we can secure over the near term, the anticipated changes in our operations as we wind down R&D operations and align our cost structure to meet the future needs of the CDMO business, and the capital needed to support our other initiatives within our near term strategic plans that will enhance our operations. Due to some of these uncertainties, we cannot quantify the amount of capital we need to raise today, but we are focused on minimizing dilution and only raising sufficient capital to achieve these goals.”
https://tinyurl.com/ybycb2s6
DEC.2017 Company Update: "Transitioning to a Pure-Play CDMO, Avid Bioservices, Inc."
12-11-17 10-Q for q/e 10-31-17: https://tinyurl.com/y75kwojs
Business Description – We are a company committed to improving the lives of patients by manufacturing pharmaceutical products through our CDMO, Avid Bioservices, Inc.
"Over the next 60 days, we plan to complete the transition from a R&D company to a dedicated CDMO company focused on dev. & mfg. of biopharmaceutical products derived from mammalian cell culture.
We plan to take ADDL. STEPS steps over the near term, including but not limited to:
1. Rebranding the company as Avid Bioservices, Inc.
2. Changing our ticker symbol on the NASDAQ Capital Market to align with our rebranding efforts
3. Broadening our sales force
4. Increasing our marketing efforts to support our rebranding & vision
5. Completing the wind down of all R&D activities and the potential licensing/divestiture of our assets related to our R&D operations.”
= = = = = = = = = =
12-11-17: Qtly. Conf. Call (Lias/Lytle) PR & Transcript https://tinyurl.com/ybycb2s6
...Dr. Lias, ”The company is undergoing a broad-scale transformation, the goals of which are to shift complete focus to the Avid Bioservices CDMO business and the complete divestiture of all of Peregrine's legacy R&D assets, which include bavituximab."
11-28-17: PPHM+RONIN Settle - Reconstituted 7-Member BOD https://tinyurl.com/y7ou7ssb
...Full Agreement(8-K): Header: https://tinyurl.com/y8m35nax Complete Agreement: https://tinyurl.com/y7b3gkc2
...Profiles of all 7 BOD members & Compensation as of 11-28-17: https://tinyurl.com/y9lkl4q2
.....(Joseph Carleone/Chair, Richard Hancock, Gregory Sargen, Joel McComb, Roger Lias, Mark Bamforth, Patrick Walsh)

12-11-17 Qtly CC-Transcript, PR(Fin’s Q2FY18/qe10-31-17), Avid Revs History Table
=> Total Revs May06-Oct17: $271.1mm/Avid + $24.1mm/Govt + $2.5mm/Lic. = $297.7mm.
*Cash at 10-31-17: $27.7mm
*As of Dec. 6, 2017, 45,212,760 shares o/s. (1:7 R/S eff. 7-10-17: 315mm/$.606=>45mm/$4.24 http://tinyurl.com/ycohqn6j )
*10Q/10-31-17 iss. 12-11-17: https://tinyurl.com/y75kwojs
...Pg.4: “Business Description – We are a company committed to improving the lives of patients by manufacturing pharmaceutical products through our CDMO, Avid Bioservices, Inc. Over the next 60 days, we plan to complete the transition from a R&D company to a dedicated CDMO company focused on dev. & mfg. of biopharmaceutical products derived from mammalian cell culture…” Pg.16: “We plan to take ADDL. STEPS steps over the near term, including but not limited to:
1. Rebranding the company as Avid Bioservices, Inc.
2. Changing our ticker symbol on the NASDAQ Capital Market to align with our rebranding efforts
3. Broadening our sales force
4. Increasing our marketing efforts to support our rebranding & vision
5. Completing the wind down of all R&D activities and the potential licensing/divestiture of our assets related to our R&D operations.”
This large post has 4 sections:
I. 12-11-17 Q2/FY18 Qtly. Earnings Conf. Call TRANSCRIPT (q/e 10-31-17)
II. 12-11-17 PPHM Press Release: Q2/FY18 Earnings & Developments
III. A link to “O/S Shares & ATM Sales History – 2006-curr.” (https://tinyurl.com/y76cbyt5 )
IV. Updated Table of Avid Revenues By Quarter (May’06-Current)
…Recall: Peregrine’s FY runs May-Apr, so FY’18 = May’17-Apr’18.
((( Orig. transcript from SeekingAlpha.com [https://tinyurl.com/ya7qxk8w ], with numerous corrections made. )))
Link to webcast replay: http://ir.peregrineinc.com/events.cfm => https://edge.media-server.com/m6/p/4euqa8wh
FULL TRANSCRIPT… 12-11-17 FY’18/Q2 Earnings Conf. Call (q/e 10-31-17) (Lias/Lytle)
WELCOME & FWD-LOOKING STATEMENTS: Tim Brons, Vida Strategic Partners (IR) http://www.peregrineinc.com
AVID’s PRESIDENT ROGER LIAS – OPENING COMMENTS:
Thanks to all of you who've dialed in and all of you who are participating today via webcast. As discussed, during our Q1 earnings call last September (9-11-17), the company is undergoing a broad-scale transformation, the goals of which are to shift complete focus to the Avid Bioservices contract development and mfg. business [CDMO] and the complete divestiture of all of Peregrine's legacy R&D assets, which include bavituximab. As previously discussed, the current biologics contract services market allows us to take advantage of our installed cGMP mfg. capacity and existing capabilities and expertise. Our exemplary track record in releasing commercial products to many global markets, including the United States and European Union, catalyzes the transition to a focused, revenue-generating services business with a potential for consistent & sustainable growth at less risk. We initiated a plan during Q1/FY18 (q/e 7-31-17) to effect the transformation of the business and we continue to execute this transition. During Q2, we have achieved several important milestones that we believe will facilitate a successful outcome. I will give an overview addressing our corporate & organizational progress, and then we'll turn the call over to Paul to address the company's financial results for Q2.
Before discussing advances and executing our new corporate strategy however, I would like to first address the fact that I am beginning today's call instead of Steve King. Today, we'll be discussing our work to transition to a dedicated CDMO business and our progress in that effort. And for that reason, as President of Avid Bioservices, which represents the future of the company, I'm very pleased to be able to address you all today.
Next, I would like to address the proxy matters that have been publicly deliberated in recent months. As many of you know, several of Peregrine's investors have collectively lobbied for the new direction that the company is taking and a new Board composition [Ronin/SWIN, full history: https://tinyurl.com/y7ou7ssb ]. We recently entered into a settlement agreement with these investors under which 4 existing Board members have resigned and 4 new indep. Board members have been appointed. More specifically, on 11-27-17, directors Steven King, Carlton Johnson, Eric Swartz, and David Pohl each resigned from Peregrine's BOD and from the BOD of Avid Bioservices. The vacancies created by these resignations were immediately filled by Richard Hancock, Joel McComb, and Gregory Sargen, each of whom were nominated by the investor group for election at Peregrine's upcoming 2017 Annual Meeting of Stockholders [1-18-18]. In addition, Joseph Carleone, who is indep. of both the investors and of Peregrine, joined the Board and is serving as Chairman. Each of these appointees has very significant experience in the biomfg. industry, and the newly seated Board has already demonstrated that they will add great value as we work to establish Avid as a leading CDMO. In addition to the newly appointed members, Mark Bamforth, Patrick Walsh, and I will continue to serve on the Board. We've recently announced that we will hold our 2017 ASM on Jan. 18, 2018. We encourage all shareholders to review the biographies and accomplishments of these Board appointees, and we request your support for them in your proxy voting. We believe each new member will continue to bring a wealth of experience and the industry leadership required to take Avid to the next level. I am personally very pleased that we have avoided further distraction and are now able to focus fully on the execution of our strategic plan and the complete transition of the company to a dedicated CDMO.
I would now like to discuss in more detail some of the advances we've made with respect to our corporate strategy. In recent months, we have made substantial progress towards establishing the company as a dedicated CDMO with the potential to achieve significant growth. During Q1, we recorded $12.8M in revenue, and on a y-t-d basis, we reported revenues of approx. $40M. This is in line with the guidance provided earlier in the year, and we continue to expect revenue for FY18 to be consistent with revenue from last year, with most revenue recognized in the 1st & 2nd quarters. Though we achieved a 6-yr. compound annual growth rate of ~38% between FY11 & FY17, we are projecting relatively flat revenue this year due to a previously announced decrease in demand from our 2 largest customers. We expect this lull in growth to be temporary, as these customers sell through current inventory and work through a regulatory filing delay. This situation, however, points to the urgent need to access new growth opportunities for Avid to diversify our client base and to broaden Avid's service offering. To that end, during my first 2 months with the company, I've worked closely with the Avid team to establish a sustainable growth strategy for the company for the near term and into the future. This strategy is comprised of 3 primary components:
1. Firstly, we will diversify and broaden our customer base & project mix.
2. Next, we will expand & strengthen Avid's current service offerings, taking advantage of current capabilities and available space to increase revenue-generating potential. Activities will include optimization of production capacity within existing facilities and the addition of process dev. capabilities and strengthening of our commercial terms.
3. Finally, to further increase growth potential & profitability, we will, in future, introduce new service offerings into the biologics contract services market.
As we effect this strategy, Avid will no longer need to compete for resources with the company's internal R&D programs or be impacted by mgt. decisions related to the other side of the legacy business. There certainly remains some issues that need to be addressed in order for Avid to operate more efficiently as a dedicated CDMO, but we expect these to be short term, and we are already working actively to make the necessary changes. The fact that Avid has been able to grow to a $50M excess capacity business despite Peregrine's focus on dev. of R&D assets, and with extremely limited marketing visibility, demonstrates, I believe, both the strength and the potential of the company. Our target market remains strong & dynamic. The broad pharmaceutical industry remains highly dependent on the shift in focus from traditional small molecule drugs to biologics. In addition, we see acceleration in biosimilar dev. for global markets. Biologic sales are projected to reach greater than $250B by 2020 at a compound annual growth rate of around 9%. This represents very nearly half of the top 100 pharmaceutical products, and a significant majority of these products are derived from mammalian cell culture and require the type of process development and cGMP mfg. capabilities that we offer here at Avid. The explosive growth in the biologics market has necessitated considerable focus on the mfg. technologies & process economics. Avid's state-of-the-art Myford facility, featuring single-use bioreactors and other modern process innovations in custom-designed clean rooms, has been widely praised by current & future customers, including pharmaceutical multinationals, and this provides the opportunity to deliver highly efficient and cost-effective manufacturing. The demand for biologics CDMO services remains very strong and is widely predicted that, while there is no one-size-fits-all mfg. approach to biologics, we are seeing considerable growth in investment in new facilities & capacity, there will remain an imbalance between supply & demand for the foreseeable future.
I will now provide some addl. information on the 3 aspects of Avid Bioservices' strategy at a high level and how we intend to take advantage of this market opportunity:
First, we are working to expand and, importantly, diversify our customer base as a broader customer portfolio will help mitigate the risks associated with being reliant on a few large clients. Again, while we believe the current decrease in demand from 2 of our largest clients is temporary, we are working to obtain significant addl. visibility into their future production needs based on excellent existing relationships and improved communications. We must clearly bring in new customer projects as soon as possible to increase utilization rates for our existing capacity. In support of this important objective, we are executing our marketing & promotional plan, including some re-branding, to greatly increase market visibility and commercial reach and to position Avid Bioservices for the first time ever in its history as an expert and reliable, dedicated service provider. Our long experience and available state-of-the-art cGMP mfg. capacity, capable of producing both clinical-stage and commercial-stage biopharmaceuticals, position us extremely well to take advantage of the considerable market opportunity. We've recently hired Tracy Kinjerski as VP of Business Operations to lead this effort [11-29-17: https://tinyurl.com/yc4zenkc ]. Tracy is a senior business dev. executive with over 17 years of experience in the biopharmaceutical industry, almost all of which have been focused in contract development and mfg. of biologics. Tracy most recently served as Senior Director of Global Business Dev. at CMC Biologics, a global contract mfg. organization. There she achieved rapid success in her role spearheading business dev. & client relationship management, which resulted in significant new business growth. Prior to CMC Biologics, Tracy held senior corp. development positions with other leading CDMOs, including Fujifilm Diosynth, Althea Technologies, and Avecia Biologics. In her new role, Tracy is charged with expanding the company's client base and driving growth at Avid. In addition to her responsibilities in business dev., including strengthening our presence in the Eastern United States, Tracy will strengthen Avid's project mgt. and client relationship operations and systems and will direct our marketing & promotional activities. We're thrilled to have Tracy on board in this important role, and we believe that she will make significant contributions to Avid's continued growth, strengthening of the revenue backlog and profitability. We are actively engaging with numerous potential new customers with products at all stages of development and are confident that we will be able to announce additions to our client base in the near future. Concurrent with our ramp up in business development efforts, we continue to work closely with the 4 new customers that were signed earlier in 2017. While we expect to recognize some revenue from these relationships in this FY18, as we kick off projects and effect technology transfers, it will be FY19 before we see significant revenues derived from these new customers. These projects, ranging from early-stage development to commercialization, have the potential to make meaningful contribution to the top line in the future. While we continue to focus on opportunities to transfer any existing larger scale & later stage mfg. projects over the near term, it is important that we also continue to secure addl. early stage process development and clinical mfg. projects that will deliver the opportunity for significant incremental mfg. revenue in the future. As a final part of the first component of our strategy, we are taking immediate action to strengthen our contractual mechanisms and related terms and conditions with focus on pricing, forecasting, customer commitment, and revenue recognition.
The 2nd arm of Avid's new growth strategy is the expansion & strengthening of current service offerings. This includes the addition of process dev. capabilities and optimizing current mfg. operations. As a dedicated contract manufacturer, we need to both broaden & strengthen our process dev. capabilities. Not only does process dev. work represent an attractive & profitable standalone business opportunity, but, as just mentioned, it also generates future mfg. opportunities and allows development of strong client relationships with visibility & access to additional future projects. Process dev. laboratories & capabilities are used as shop fronts by our peers in the biologics contract mfg. space. And as we diversify both the client base and the project mix, we need to develop competitive capabilities in this area capable of delivering a strong project pipeline. Necessary expansions include addl. modern and well equipped laboratory space and strengthened service offerings in areas such as analytical development, cell line development, and formulation. The addition of new equipment and technologies will allow us to greatly increase throughput & efficiency and to deliver first-in-class process dev. services to our customers. Planning for these additions commenced during Q2 (q/e 10-31-17). On the mfg. side, we continue to ensure that Avid's facilities are state-of-the-art and capable of meeting the highest industry standards. The sophistication of our Myford facility clearly provides an advantage in this respect, but there are a number of areas for improvement. In recent months, we have expanded capacity by installing & validating 2 new 2,000L bioreactors in this facility, and we've already secured some commitments for this capacity. We also continue to work on changes necessary to improve efficiency & throughput when operating this facility in multi-product mode. Due to its modular design, the use of single-use mfg. technologies and already operational supporting quality laboratories, warehousing, and utilities, there's the potential to relatively quickly and cost-effectively install addl. mfg. capacity in available shelf space within the Myford facility. This provides the opportunity to organically & significantly grow our current drug substance mfg. business within our existing facilities.
Finally, beyond growth, strengthening, and extension of our current service offering, there's tremendous opportunity in areas that Avid is not currently active. This represents the 3rd facet of the growth strategy. At present, Avid is focused on the production of antibodies and various classes of recombinant proteins derived from mammalian cell culture. Opportunities exist to manufacture other biologics derived from microbial expression systems, and advances in gene, cellular and immunotherapies, antibody drug conjugates, and multi-specific antibody products feed a growing demand for manufacture of newer classes of products, including viral vectors and vaccines, which are quite similar but distinct mfg. processes to those currently offered at Avid. The available space in the Myford mfg. facility also provides us with the opportunity to consider addl. vertically integrated, high-value mfg. services, such as fill-and-finish drug product manufacture in support of customers' clinical requirements. While incorporating such offerings into the business will undoubtedly deliver technical and regulatory challenges, they offer considerable growth opportunity, and they ultimately contribute to the transformation of Avid into a global and leading CDMO. We continue to evaluate these strategic opportunities and we'll report our conclusions as they are reached. Given today's strong biologics market, the growing demand and limited production capacity available globally, it is clear that there is a tremendous opportunity for Avid in the coming years. We have established a strategy to take full advantage of promising market conditions and have launched a number of efforts to position Avid to successfully achieve stable growth and leadership in this sector.
Before turning over the call to Paul, I'd like to address 2 addl. steps that must be completed in order to finalize & formalize this transition: First, as discussed previously, we are in active discussions to divest the legacy Peregrine R&D assets and have taken action to formally wind down all R&D related activities not required to effect the transaction. We will keep you updated on progress. Lastly, we are taking steps to transition Peregrine and Avid into a single entity and will change the name of the company to Avid Bioservices Inc. Through this process, there will be no change to the company's shares outstanding or to the shares held by individual shareholders. We are currently taking steps to effect this transition, and we expect to have the entire process complete in early 2018. I look forward to completing the transition to a dedicated CDMO, to focusing on execution and to keeping you updated on progress. With that, I’ll hand over to Paul, who will cover the qtly financial results.
PAUL LYTLE (CFO): [10-31-17 10Q iss. 12-11-17: https://tinyurl.com/y75kwojs ]
I'll now discuss our financial results for Q2/FY18, starting with revenue. During Q2, we recognized $12.8M in contract mfg. revenue compared to $23.4M for Q2/FY17. While the current qtr revenue declined compared to the same prior year period, we have achieved higher revenue to date for the current 6mo. period of almost $40M vs. the same prior year period of $29M. In addition, as Roger mentioned, we are still on track to achieve the full FY’18 revenue guidance of $50-55M. This means though, we expect revenue for the remainder of FY18 year to be relatively light due to the decrease in demand from our 2 largest customers, combined with the normal dev. and mfg. time lines of new customer projects. As a backdrop, it's important to note that new customer projects generally commence with either a technology transfer of a known or defined mfg. process, or we can develop a new mfg. process for them, which can take up to 1 year before the higher value mfg. revenue is generated. Therefore, customers that we secure in FY18 will generally contribute more meaningfully to top line revenue in FY19, and reinforces our need to secure new business over the near term. This brings me to our revenue backlog. As of today, our existing customers have signed and committed to $33M in future mfg. services, of which we expect to recognize $10-15M over the remainder of FY18. In addition to our committed backlog, it's important to highlight that our customers have historically committed to services that cover a relatively short period of time as their project advances. Therefore, the committed backlog does not generally include all mfg. services required to fully execute on their programs. As a result, we do have a soft backlog of business on top of what has been committed to, and our goal over the near term is to continue to convert the soft backlog to a committed backlog. Let me shift gears now to discuss our gross margins on contract mfg. revenue. During Q2, our gross margin declined to a negative 27%. This was mostly driven by mfg. capacity that was not utilized during the quarter, also referred to as idle capacity. During Q2, idle capacity negatively impacted gross margin by 39 percentage points. As we look to improve these margins, in addition to focusing on our book of business and growing our customer base & backlog, we are also actively evaluating our overall cost structure as a dedicated CDMO, and we plan to align our cost structure to match the future needs of this new dedicated CDMO business. Turning now to operating expenses. Total operating expenses for Q2/FY18 were $9.2M compared to $12M for Q2/FY17. Included in operating expenses this qtr was $1.6M in restructuring charges associated with workforce reduction pursuant to our restructuring plan implemented last August 2017. R&D expenses decreased to $3.7M in Q2/FY18 compared to $7M for Q2/FY17, in line with our ongoing wind-down of R&D activities. It's important to note that over the next few months, we will continue to rapidly wind down all R&D costs to zero, supporting only those efforts needed to pursue the license or sale of our R&D assets. And for Q2/FY18, SG&A expenses decreased to $3.9M compared to $5M for Q2/FY17. The company's consolidated net loss attributable to common stockholders was $14.1M or $.31/sh. for Q2/FY18 compared to $5.5M or $.16/sh. for the same prior year qtr. Cash & cash equivalents as of Oct. 31, 2017, were $27.7M compared to $46.8M at FYE April 30, 2017.
As further discussed in the company's 10-Q [https://tinyurl.com/y75kwojs ], which was filed today, we plan to raise addl. capital within the next 6 months to support our continued operations and other initiatives that will enhance our CDMO operations. The amount of capital we need to raise will depend on a number of factors, including, the amount of new business we can secure over the near term, the anticipated changes in our operations as we wind down R&D operations and align our cost structure to meet the future needs of the CDMO business, and the capital needed to support our other initiatives within our near term strategic plans that will enhance our operations. Due to some of these uncertainties, we cannot quantify the amount of capital we need to raise today, but we are focused on minimizing dilution and only raising sufficient capital to achieve these goals. This concludes my financial overview. I will now open the call up for questions.
Q&A: [beg. 23:01]
1. Tarun Aswani (for Caroline Palomeque) - Noble Life Science Partners http://noblelsp.com/research
TA: ”This is Tarun Aswani calling in on behalf of Caroline. For Avid, can you specifically outline what you'll be looking for if the strategy is based on potential M&A? And likewise, if the company plans to grow organically, what can we expect to see specifically moving forward for Avid?”
Roger Lias: Hi, Tarun. This is Roger. Thanks for your question. And thanks for your standing in for Caroline. We're still looking at that. One the problems we face, which is not necessarily a problem in the big picture, is we have a lot of opportunity. I'm very keen to make sure that we're running before we walk and focusing on our slightly nearer term opportunities to start off with. With respect to M&A, there are certainly opportunities out there, but I see probably in that regard, short term focus would be on some add-on adjunct services perhaps in areas such as cell line development or analytical. With the organic growth, again as outlined, certainly for the foreseeable, reasonably near future, we certainly need to consolidate what we have. We have tremendous opportunity occasioned to us by the available capacity, but we also have tremendous opportunity to bring on new capacity within our existing infrastructure. So, I think without a doubt, that has to be our short-term focus. We, I believe, will be opportunistic with respect to other services. I think, for instance, fill-finish, drug product work is relatively easily conceptualized as a bolt-on, as an add-on, to our current services. If we start talking about viral vectors and other such products, clearly these involve addl. capital and so on and so forth. so I believe we have to be more cautious and more opportunistic in those areas.
TA: ”Great. Thank you very much for answering the question.”
DR. LIAS’S CLOSING COMMENTS:
Thank you everybody, again, for participating in today's call. I'd certainly like to take the opportunity to welcome our new Board of Directors and to thank them for their input and guidance to date. And of course, as always, I'd like to thank our stockholders for their continued support. Thank you, and have a great afternoon. And with that, I will conclude the call. Thank you.
= = = = = = = = = = = = = = = = = = = = = = = = = = = = = == = = =
12-11-17/PR: Peregrine Pharmaceuticals Reports Financial Results for Q2/FY2018 and Recent Developments
* Avid Bioservices Records Revenues of $12.8 Million in the Second Quarter of FY2018
* Transition to a Dedicated CDMO Business Nearing Completion
http://ir.peregrineinc.com/releasedetail.cfm?ReleaseID=1051443
TUSTIN, Dec. 11, 2017: Peregrine Pharmaceuticals, Inc. (NASDAQ:PPHM/PPHMP), a company committed to improving patient lives by manufacturing and delivering high quality biologics, today announced financial results for the second quarter of fiscal year (FY) 2018 ended October 31, 2017, and provided an update on its contract manufacturing operations, and other corporate highlights.
HIGHLIGHTS SINCE JULY 31, 2017
"Today, we are pleased to report that the company has made great progress in its transition from an R&D focused business to a dedicated contract development and manufacturing organization (CDMO)," stated Roger J. Lias, PhD, President of Avid Bioservices. "In late November, the company came to an agreement with an investor group [Ronin/SWIM, see: https://tinyurl.com/y7ou7ssb ], appointing a highly qualified new board of directors consisting of 3 new independent members from this investor group and one mutually designated independent member in addition to myself and the 2 independent members previously appointed. We have now added 6 highly qualified and independent board members since October. In addition, we are focused on hiring experienced and successful CDMO professionals who are dedicated to revenue growth through the expansion and diversification of Avid's client base, as evidenced by the recently announced hiring of Tracy Kinjerski as VP of Business Operations. We are actively planning to expand Avid's service offerings and enhance our manufacturing infrastructure to ensure that we are offering the highest quality services, and state-of-the-art facilities to our customers. We are also taking steps to officially change the name of the entire organization to Avid Bioservices, Inc. to formalize this transition. Lastly, we are in continued discussions with third parties regarding the divestiture of the company's remaining R&D assets and we will keep you apprised on our progress as we advance the process."
= = = = = = = = = = = = = = =
[FULL HISTORY of PPHM-Ronin PR’s, Letters, 13-D’s, Form4’s, Proxy’s, etc: https://tinyurl.com/y7ou7ssb ]
RECENT DEVELOPMENTS AT AVID BIOSERVICES
Established a dedicated CDMO management infrastructure with the hiring of Roger J. Lias, PhD, as the President of Avid Bioservices and director. [9-11-17: http://tinyurl.com/yd3eh3uv ]
• Dr. Lias brings more than 20 years of experience in the industry having held senior management positions at several leading CDMOs including Cytovance Biologics, KBI BioPharma, Diosynth RTP (formerly Covance Biotechnology Services) and Lonza Biologics.
Strengthened Avid's sales and business development function with the hiring of Tracy Kinjerski as VP of Business Operations. [11-29-17: https://tinyurl.com/yc4zenkc ]
• Ms. Kinjerski brings more than 17 years of experience with a focus in contract development and manufacturing. She is charged with driving Avid's growth through the strategic expansion and diversification of the company's commercial and clinical client base.
Reconstituted the board of directors to include 6 independent directors, all with significant CDMO experience.
• In October 2017, Mark R. Bamforth was appointed as an independent member of the board of directors. Mr. Bamforth has 30 years of biologics leadership experience including founding two CDMOs, Brammer Bio, where he is currently the president and CEO, and Gallus BioPharmaceuticals, which was acquired by DPx Holdings B.V., the parent company of Patheon. Additionally, he served for more than 20 years in key roles at Genzyme Corporation, including 10 years as a corporate officer responsible for running global manufacturing.
• In October 2017, Patrick Walsh was appointed as an independent member of the board of directors. Mr. Walsh has a record of leading successful, high-growth CDMOs and he has also led complex laboratory and pharmaceutical manufacturing operations including parenteral and active pharmaceutical ingredients (API) on a global scale.
• In November 2017, the company entered into a settlement agreement with its largest shareholder (Ronin/SWIM) regarding the composition of Peregrine's board of directors. Under the terms of the Agreement, on November 27, 2017, directors Steven W. King, Carlton M. Johnson, Jr., Eric S. Swartz and David H. Pohl each tendered his resignation, effective immediately, from Peregrine's board of directors, and from the board of directors of Avid Bioservices. The vacancies created by these resignations were immediately filled by three individuals who were nominated by Ronin/SWIM for election at Peregrine's upcoming 2017 Annual Meeting of Stockholders (Richard B. Hancock, Gregory P. Sargen and Joel McComb), and one director (Joseph Carleone, Ph.D.) who is independent of Ronin/SWIM and new to Peregrine.
• Joseph Carleone, Ph.D. (independent appointee): Dr. Carleone is Chairman of the Board of AMPAC Fine Chemicals LLC, a leading manufacturer of pharmaceutical active ingredients. Prior to this position, Dr. Carleone was President, Chief Executive Officer and director of American Pacific Corporation, a leading custom manufacturer of fine and specialty chemicals and propulsion products.
• Richard B. Hancock (Ronin/SWIM appointee): Richard (Rick) B. Hancock has worked in the biologic CDMO industry for over 30 years in various operational and executive roles, serving most recently as President and CEO of Althea Technologies, Inc., a large molecule CDMO producing a wide range of biologics, vaccines and parenteral products.
• Joel McComb (Ronin/SWIM appointee): Joel McComb is the CEO, Chairman and Co-Founder of BioSpyder Technologies, Inc. Prior to BioSpyder, Mr. McComb served as Senior Vice President and General Manager of Illumina, Inc., President of GE Healthcare's Life Sciences and Discovery Systems division, and President of GE Healthcare's Interventional Medicine division.
• Gregory P. Sargen (Ronin/SWIM appointee): Gregory P. Sargen currently serves as Executive Vice President - Corporate Development and Strategy of Cambrex Corporation ("Cambrex"), a global manufacturer and provider of services to life sciences companies. Prior to his current role, Mr. Sargen served as Executive Vice President and Chief Financial Officer of Cambrex.
Expanded production capacity in the Myford facility to allow organic and significant growth using existing facilities.
• In recent months, the company expanded its capacity in its Myford facility by installing two new 2,000 liter single-use bioreactors.

FINANCIAL HIGHLIGHTS AND RESULTS
The company maintains its manufacturing revenue guidance for the full FY 2018 of $50-55 million.
Contract manufacturing revenue from Avid's clinical and commercial biomanufacturing services was $12.8 million for the second quarter of FY 2018 compared to $23.4 million for the second quarter of FY 2017.
Avid's current manufacturing revenue backlog is $33.0 million, representing estimated future manufacturing revenue to be recognized under committed contracts. Most of the backlog is expected to be recognized during the remainder of FY 2018 and into FY 2019.
Total operating expenses for the second quarter of FY 2018 were $9.2 million, compared to $12.0 million for the second quarter of FY 2017. For the second quarter of FY 2018, total operating expenses included restructuring charges of $1.6 million associated with termination benefits including severance and other employee related costs related to a workforce reduction pursuant to a restructuring plan implemented in August 2017. The company is also actively evaluating its overall operating expenses and cost structure as a dedicated CDMO and plans to align its cost structure to match the future needs of the business.
Research and development expenses decreased to $3.7 million in the second quarter of FY 2018 compared to $7.0 million for the second quarter of FY 2017. Over the next 60 or fewer days, the Company will continue to rapidly wind down all research and development costs to zero and plans to support only those efforts needed to pursue the license or sale of its research and development assets.
Cost of contract manufacturing increased to $16.2 million in the second quarter of FY 2018 compared to $15.4 million for the second quarter of FY 2017.
For the second quarter of FY 2018, selling, general and administrative expenses decreased to $3.9 million compared to $5.0 million for FY 2017.
Peregrine's consolidated net loss attributable to common stockholders was $14.1 million or $0.31 per share, for the second quarter of FY 2018, compared to a net loss attributable to common stockholders of $5.5 million, or $0.16 per share, for the same prior year quarter.
Peregrine reported $27.7 million in cash and cash equivalents as of October 31, 2017, compared to $46.8 million at fiscal year ended April 30, 2017. As further discussed in the Company's Quarterly Report on Form 10-Q, the Company plans to raise additional capital within the next 6 months to support its continued operations and other initiatives that will enhance its CDMO operations.
More detailed financial information and analysis may be found in Peregrine's Quarterly Report on Form 10-Q [https://tinyurl.com/y75kwojs ], which will be filed with the Securities and Exchange Commission today.
CONFERENCE CALL
Peregrine will host a conference call and webcast this afternoon, December 11, 2017, at 4:30PM EST (1:30PM PST). To listen to the conference call, please dial (877) 312-5443 or (253) 237-1126 and request the Peregrine Pharmaceuticals conference call. To listen to the live webcast, or access the archived webcast, please visit: http://ir.peregrineinc.com/events.cfm .
REPLAY: https://edge.media-server.com/m6/p/4euqa8wh
ABOUT PEREGRINE PHARMACEUTICALS, INC.
Peregrine Pharmaceuticals, Inc. is a company transitioning from an R&D focused business to a pure play contract development and manufacturing organization (CDMO). Peregrine's in-house CDMO services, including cGMP manufacturing and development capabilities, are provided through its wholly-owned subsidiary Avid Bioservices, Inc. ( http://www.avidbio.com ). Peregrine is pursuing the licensing or sale of its proprietary R&D assets, including its lead immunotherapy candidate, bavituximab, which is currently being evaluated in clinical trials in combination with immune stimulating therapies for the treatment of various cancers. For more information, please visit http://www.peregrineinc.com .
ABOUT AVID BIOSERVICES, INC.
Avid Bioservices, a wholly owned subsidiary of Peregrine Pharmaceuticals, provides a comprehensive range of process development, high quality cGMP clinical and commercial manufacturing services for the biotechnology and biopharmaceutical industries. With nearly 25 years of experience producing monoclonal antibodies and recombinant proteins in batch, fed-batch and perfusion modes, Avid's services include cGMP clinical and commercial product manufacturing, purification, bulk packaging, stability testing and regulatory strategy, submission and support. The company also provides a variety of process development activities, including cell line development and optimization, cell culture and feed optimization, analytical methods development and product characterization. For more information about Avid, please visit http://www.avidbio.com .
Safe Harbor *snip*
CONTACTS:
• Stephanie Diaz (Investors) Vida Strategic Partners 415-675-7401 sdiaz@vidasp.com
• Tim Brons (Media) Vida Strategic Partners 415-675-7402 tbrons@vidasp.com
PEREGRINE PHARMACEUTICALS, INC.
CONDENSED CONSOLIDATED STATEMENTS OF OPERATIONS AND COMPREHENSIVE LOSS (UNAUDITED)
Three Months Ended
October 31, Six Months Ended October 31,
2017 2016 2017 2016
Contract manufacturing revenue $ 12,782,000 $ 23,370,000 $ 39,859,000 $ 28,979,000
Cost of contract manufacturing 16,242,000 15,441,000 36,690,000 18,503,000
Gross profit (loss) (3,460,000 ) 7,929,000 3,169,000 10,476,000
Operating expenses:
Selling, general and administrative 3,867,000 4,984,000 8,080,000 10,044,000
Research and development 3,722,000 7,022,000 7,367,000 15,591,000
Restructuring charges 1,588,000 — 1,588,000 —
Total operating expenses 9,177,000 12,006,000 17,035,000 25,635,000
Operating loss (12,637,000 ) (4,077,000 ) (13,866,000 ) (15,159,000 )
Other income (expense):
Interest and other income 14,000 21,000 41,000 46,000
Interest and other expense (1,000 ) — (4,000 ) —
Net loss $ (12,624,000 ) $ (4,056,000 ) $ (13,829,000 ) $ (15,113,000 )
Comprehensive loss $ (12,624,000 ) $ (4,056,000 ) $ (13,829,000 ) $ (15,113,000 )
Series E preferred stock
accumulated dividends (1,442,000 ) (1,442,000 ) (2,523,000 ) (2,477,000 )
Net loss attributable to common stockholders
$ (14,066,000 ) $ (5,498,000) $(16,352,000) $(17,590,000)
Weighted average common shares outstanding:
Basic and Diluted (1) 45,097,474 34,973,681 44,935,600 34,600,776
Basic and diluted loss per common share (1) $ (0.31 ) $ (0.16 ) $ (0.36 ) $ (0.51 )
(1) All share and per share amounts of our common stock for all prior fiscal year periods presented have been retroactively adjusted to reflect the 1:7 reverse stock split of our issued and outstanding common stock, which took effect on July 10, 2017 (Note 1).
PEREGRINE PHARMACEUTICALS, INC.
CONDENSED CONSOLIDATED BALANCE SHEETS
October 31, 2017 April 30, 2017 Unaudited
ASSETS
Current assets:
Cash and cash equivalents $ 27,727,000 $ 46,799,000
Trade and other receivables 3,508,000 7,742,000
Inventories 16,518,000 33,099,000
Prepaid expenses 1,223,000 1,460,000
Total current assets 48,976,000 89,100,000
Property and equipment, net 27,148,000 26,515,000
Restricted cash 1,150,000 1,150,000
Other assets 1,353,000 1,347,000
Total assets $ 78,627,000 $ 118,112,000
LIABILITIES AND STOCKHOLDERS' EQUITY
Current liabilities:
Accounts payable $ 2,739,000 $ 5,779,000
Accrued clinical trial and related fees 5,392,000 4,558,000
Accrued payroll and related costs 4,063,000 6,084,000
Deferred revenue 7,473,000 28,500,000
Customer deposits 13,138,000 17,017,000
Other current liabilities 745,000 993,000
Total current liabilities 33,550,000 62,931,000
Deferred rent, less current portion 2,171,000 1,599,000
Commitments and contingencies
Stockholders' equity: Preferred stock—$0.001 par value; authorized 5,000,000 shares;
1,647,760 issued and outstanding at October 31, 2017 and April 30,
2017, respectively 2,000 2,000
Common stock—$0.001 par value; authorized 500,000,000 shares;
45,172,632 and 44,014,040 issued and outstanding at October 31,
2017 and April 30, 2017, respectively 45,000 44,000
Additional paid-in capital 594,004,000 590,971,000
Accumulated deficit (551,145,000 ) (537,435,000 )
Total stockholders' equity 42,906,000 53,582,000
Total liabilities and stockholders' equity $ 78,627,000 $ 118,112,000
- - - - - - - -
From 10-Q header: “As of Dec. 6, 2017, there were 45,212,760 shares outstanding.”
- - - - - - - - - - - - - - - - -
Latest 10K 4-30-17 iss. 7-14-17 http://tinyurl.com/ycxu4l5n PR: http://tinyurl.com/yb4wulvu (Cash 4-30-17=$46.8mm); Amended 8-25-17: http://tinyurl.com/yb5jq7vc
Latest 10Q 10-31-17 iss. 12-11-17 https://tinyurl.com/y75kwojs PR: http://ir.peregrineinc.com/releasedetail.cfm?ReleaseID=1051443 (Cash 10-31-17=$27.7mm)
ALL SEC filings for PPHM: http://tinyurl.com/6d4jw8
12-11-17: O/S Shares & ATM Sales History (’06–curr.) https://tinyurl.com/y76cbyt5
**NOTES:
1. PPHM shares were 1:7 Reverse Split eff. 7-10-17 (~315mm/$.606=>~45mm/$4.24) http://tinyurl.com/ycohqn6j
2. Per the 4-30-10 10-K (pub. 7-14-17), ALL ATM Agreements have been fully used – none remain (F-20 & F-28). If any new ATM’s are opened (and reported via Form 424B5), they go against the $67mm remaining on the orig. $150mm shelf filed via the S-3 filed 4-23-14. 10K: http://tinyurl.com/ycxu4l5n
3. The last ATM sales made (a/o the 10-31-17 10Q iss 12-11-17) were these: • Sold 5/1/17-6/30/17: $4,307,000gr./1,051,258sh. = $4.10/sh.
= = = = = = = = = = = = = = = = = = = = = = = = = = = =
Updated PPHM REVS-BY-QTR TABLE, now thru FY18'Q2(qe 10-31-17), per the 10-Q (https://tinyurl.com/y75kwojs ) issued 12-11-17.
• Total Revs since May’06: ($271.1mm/Avid + $24.1mm/Govt + $2.5mm/Lic.) = $297.7mm
• 12-11-17: FY'18 (May'17-Apr'18) Avid revs guidance $50-55mm (committed B/L=$33mm). **
• Deferred-Revs at 10-31-17 total $7.5mm, DOWN from $15.1mm at 7-31-17.
• Cust.Deposits at 10-31-17 total $13.1mm, UP from $2.7mm at 7-31-17.
• Inventories at 10-31-17 total $16.5mm, DOWN from $24.2mm at 7-31-17.
• Avid’s Gross-Profit over last 4 qtrs: $12.1mm on revs of $68.5mm (GP%=17.6)
• Recall, Avid Rev$ from Gov’t DTRA Contract work (6/30/08 – 4/15/11, totaling $24.15mm), went into GOVT-REVS, not AVID-REVS, in the Financials.
**10-31-17 10Q/p.18: “For FY18 (fye 4-30-18) we have secured 4 new cust’s since Jan.2017. However, as previously disclosed, during FY18/Q1 we experienced unanticipated decreases in mfg. demand from our largest cust. [Halozyme] and a regulatory filing delay from our 2nd largest cust., both of which will impact our ability to increase revenue during the remainder of FY’18 and potentially beyond.”
Avid’s website: http://www.avidbio.com
AVID PROFITABILITY (GROSS*) BY QTR:
QTR Avid-Rev$ CostofMfg$ Gross-Profit$ GP%
FY13Q1 7-31-12 4,135,000 2,024,000 2,111,000 51%
FY13Q2 10-31-12 6,061,000 3,703,000 2,358,000 39%
FY13Q3 1-31-13 6,961,000 3,651,000 3,310,000 47%
FY13Q4 4-30-13 4,176,000 3,217,000 959,000 23%
FY14Q1 7-31-13 4,581,000 2,670,000 1,911,000 42%
FY14Q2 10-31-13 7,354,000 4,195,000 3,159,000 43%
FY14Q3 1-31-14 3,885,000 2,416,000 1,469,000 38%
FY14Q4 4-30-14 6,474,000 3,829,000 2,645,000 41%
FY15Q1 7-31-14 5,496,000 3,583,000 1,913,000 35%
FY15Q2 10-31-14 6,263,000 4,139,000 2,124,000 34%
FY15Q3 1-31-15 5,677,000 3,113,000 2,564,000 45%
FY15Q4 4-30-15 9,308,000 4,758,000 4,550,000 49%
FY16Q1 7-31-15 9,379,000 4,608,000 4,771,000 51%
FY16Q2 10-31-15 9,523,000 4,741,000 4,782,000 50%
FY16Q3 1-31-16 6,672,000 3,896,000 2,776,000 42%
FY16Q4 4-30-16 18,783,000 9,721,000 9,062,000 48%
FY17Q1 7-31-16 5,609,000 3,062,000 2,547,000 45%
FY17Q2 10-31-16 23,370,000 15,441,000 7,929,000 34%
FY17Q3 1-31-17 10,747,000 7,974,000 2,773,000 26%
FY17Q4 4-30-17 17,904,000 11,782,000 6,122,000 34%
FY18Q1 7-31-17 27,077,000 20,448,000 6,629,000 24%
FY18Q2 10-31-17 12,782,000 16,242,000 -3,460,000 -27%
FY13 TOTAL: 21,333,000 12,595,000 8,738,000 41%*
FY14 TOTAL: 22,294,000 13,110,000 9,184,000 41%*
FY15 TOTAL: 26,744,000 15,393,000 11,151,000 42%*
FY16 TOTAL: 44,357,000 22,966,000 21,391,000 48%*
FY17 TOTAL: 57,630,000 38,259,000 19,371,000 34%*
*Avid Net-Profit (ie, incl. Selling, G&A) not split out from PPHM-Corp. in the financials.
.
PPHM REVENUES (in thousands) DEFERRED
-------REVENUES------- REVENUES INVEN-
Quarter Avid Govt Lic. TOTAL Avid Govt TORIES
FY07Q1 7-31-06 398 0 23 421 317 0 971
FY07Q2 10-31-06 636 0 48 684 1388 0 1899
FY07Q3 1-31-07 347 0 16 363 2202 0 1325
FY07Q4 4-30-07 2111 0 129 2240 1060 0 1916
FY08Q1 7-31-07 1621 0 4 1625 1820 0 2363
FY08Q2 10-31-07 1863 0 29 1892 1338 0 3500
FY08Q3 1-31-08 1662 0 13 1675 1434 0 2394
FY08Q4 4-30-08 751 0 150 901 2196 0 2900
FY09Q1 7-31-08 1193 324 0 1517 4021 980 4628
FY09Q2 10-31-08 983 958 0 1941 6472 1701 6700
FY09Q3 1-31-09 5778 1048 0 6826 4805 3262 5547
FY09Q4 4-30-09 5009 2683 175 7867 3776 3871 4707
FY10Q1 7-31-09 2070 4671 9 6750 5755 2332 6177
FY10Q2 10-31-09 5308 1510 78 6896 4260 3989 5850
FY10Q3 1-31-10 2945 6854 78 9877 3052 76 3861
FY10Q4 4-30-10 2881 1461 78 4420 2406 78 3123
FY11Q1 7-31-10 983 2111 115 3209 3719 47 4692
FY11Q2 10-31-10 3627 966 78 4671 2447 35 3555
FY11Q3 1-31-11 1922 882 79 2883 4300 40 3915
FY11Q4 4-30-11 1970 681 78 2729 5617 0 5284
FY12Q1 7-31-11 5439 0 216 5655 4145 0 4481
FY12Q2 10-31-11 4154 0 78 4232 2012 0 3178
FY12Q3 1-31-12 3203 0 78 3281 2552 0 2722
FY12Q4 4-30-12 1987 0 78 2065 3651 0 3611
FY13Q1 7-31-12 4135 0 116 4251 6056 0 5744
FY13Q2 10-31-12 6061 0 78 6139 6221 0 5426
FY13Q3 1-31-13 6961 0 78 7039 5061 0 4635
FY13Q4 4-30-13 4176 0 78 4254 4171 0 4339
FY14Q1 7-31-13 4581 0 107 4688 4164 0 5679
FY14Q2 10-31-13 7354 0 0 7354 3468 0 4033
FY14Q3 1-31-14 3885 0 0 3885 4329 0 5224
FY14Q4 4-30-14 6474 0 0 6474 5241 0 5530
FY15Q1 7-31-14 5496 0 0 5496 4670 0 5998
FY15Q2 10-31-14 6263 0 37 6300 3612 0 5379
FY15Q3 1-31-15 5677 0 0 5677 5752 0 6148
FY15Q4 4-30-15 9308 0 0 9308 6630 0 6148
FY16Q1 7-31-15 9379 0 292 9671 8291 0 10457
FY16Q2 10-31-15 9523 0 0 9523 9688 0 12554
FY16Q3 1-31-16 6672 0 37 6709 15418 0 15189
FY16Q4 4-30-16 18783 0 0 18783 15418 0 15189
FY17Q1 7-31-16 5609 0 0 5609 21531 0 25274
FY17Q2 10-31-16 23370 0 0 23370 21531 0 25274
FY17Q3 1-31-17 10747 0 0 10747 26367 0 33829
FY17Q4 4-30-17 17904 0 0 17904 28500 0 33099
FY18Q1 7-31-17 27077 0 0 27077 13433 0 24235
FY18Q2 10-31-17 12782 0 0 12782 7473 0 16518
Totals: 271058 24149 2453 297660 <=since5/1/2006
.
TOTAL REV’s BY YEAR (Avid+Gov’t+Lic):
FY04 4-30-04 3,314 …Avid(CMO)= 3,039 (Avid-Revs don’t incl. Govt-SVCS)
FY05 4-30-05 4,959 …Avid(CMO)= 4,684
FY06 4-30-06 3,193 …Avid(CMO)= 3,005
FY07 4-30-07 3,708 …Avid(CMO)= 3,492
FY08 4-30-08 6,093 …Avid(CMO)= 5,897
FY09 4-30-09 18,151 …Avid(CMO)= 12,963
FY10 4-30-10 27,943 …Avid(CMO)= 13,204
FY11 4-30-11 13,492 …Avid(CMO)= 8,502
FY12 4-30-12 15,233 …Avid(CMO)= 14,783
FY13 4-30-13 21,683 …Avid(CMO)= 21,333
FY14 4-30-14 22,401 …Avid(CMO)= 22,294
FY15 4-30-15 26,781 …Avid(CMO)= 26,744
FY16 4-30-16 44,686 …Avid(CMO)= 44,357
FY17 4-30-17 57,630 …Avid(CMO)= 57,630
...Total Gov’t Revs from 7-2008 inception thru FY11Q1(Apr’11): $24.15mm
.
PPHM’S QTLY. NET LOSS BY QTR:
FY08Q1 7-31-07 4,656,000
FY08Q2 10-31-07 6,207,000
FY08Q3 1-31-08 6,154,000
FY08Q4 4-30-08 6,159,000
FY09Q1 7-31-08 5,086,000
FY09Q2 10-31-08 4,497,000
FY09Q3 1-31-09 3,332,000
FY09Q4 4-30-09 3,609,000
FY10Q1 7-31-09 2,428,000
FY10Q2 10-31-09 2,787,000
FY10Q3 1-31-10 1,538,000
FY10Q4 4-30-10 7,741,000
FY11Q1 7-31-10 7,695,000
FY11Q2 10-31-10 7,513,000
FY11Q3 1-31-11 8,929,000
FY11Q4 4-30-11 10,014,000
FY12Q1 7-31-11 8,092,000
FY12Q2 10-31-11 12,055,000
FY12Q3 1-31-12 11,090,000
FY12Q4 4-30-12 10,882,000
FY13Q1 7-31-12 7,664,000
FY13Q2 10-31-12 8,753,000
FY13Q3 1-31-13 4,914,000
FY13Q4 4-30-13 8,449,000
FY14Q1 7-31-13 7,600,000
FY14Q2 10-31-13 7,790,000
FY14Q3 1-31-14 9,724,000
Net loss attributable to common stockholders (incl. PREF Div’s):
2-11-14: PPHM Raises $16.2M selling 700k Pref. Shares w/10.5% DIV)
FY14Q4 4-30-14 10,649,000
FY15Q1 7-31-14 14,157,000
FY15Q2 10-31-14 13,131,000
FY15Q3 1-31-15 14,027,000
FY15Q4 4-30-15 13,513,000
FY16Q1 7-31-15 15,101,000
FY16Q2 10-31-15 14,578,000
FY16Q3 1-31-16 18,227,000
FY16Q4 4-30-16 13,264,000
FY17Q1 7-31-16 12,437,000
FY17Q2 10-31-16 4,498,000
FY17Q3 1-31-17 9,216,000
FY17Q4 4-30-17 6,714,000
FY18Q1 7-31-17 2,647,000
FY18Q2 10-31-17 14,066,000
Period Halozyme Cust-A Other-Custs
FYE 4-30-14 91% 1% 8%
FYE 4-30-15 79% 12% 9%
FYE 4-30-16 69% 26% 5%
Q/E 7-31-16 65% 29% 6%
Q/E 10-31-16 77% 10% 13%
Q/E 1-31-17 29% 56% 15%
FYE 4-30-17 58% 26% 16%
Q/E 7-31-17 78% 16% 16%
Q/E 10-31-17 Cust%’s section Removed from this 10-Q)
“And a regulatory filing-delay from our 2nd largest-cust”
7-31-17 10Q/pg.19 (iss. 9-11-17): “We have recently secured 4 new cust’s since Jan.2017. However, as previously disclosed, we have seen unanticipated decreases in mfg. demand from our largest customer and a regulatory filing delay from our 2nd largest customer which will impact our ability to grow the revenues from our CDMO business in FY18 and potentially beyond.”
SK, 9-11-17: “And while we are projecting a relatively flat revenue year, based on the decr. FY18 forecasts from our 2 largest custs, we do believe this decrease will be temporary and expect that Avid will continue to thrive despite lower revenue from these 2 clients in the short term.”
PL, 9-11-17/CC: “While we had an excellent Q1, we have also seen decreases in mfg. from our 2 largest customers.”
PR 9-11-17: “Avid Bioservices Highlights: ...we have seen a lower mfg. demand from our top 2 cust’s; we are still expecting to generate $50-55M in revs.”
http://tinyurl.com/y9y8qdac
O/S Shares & ATM Sales History (’06–curr.)
Shares O/S as of 12-6-17: 45,212,760
**NOTES:
1. PPHM shares were 1:7 Reverse Split eff. 7-10-17 (~315mm/$.606=>~45mm/$4.24) http://tinyurl.com/ycohqn6j
2. Per the 4-30-10 10-K (iss. 7-14-17), ALL ATM Agreements had been fully used – none remained (F-20 & F-28). If any new ATM’s are opened (and reported via Form 424B5), they go against the $67mm remaining on the orig. $150mm shelf filed via the S-3 filed 4-23-14. 10K: http://tinyurl.com/ycxu4l5n
-------
NOTE: the 10-31-17 10Q (iss. 12-11-17 https://tinyurl.com/y75kwojs ) confirmed this: NO ATM SALES OCCURRED AFTER THOSE REPORTED IN THE “SUBSEQUENT EVENTS” SECTION OF THE 4-30-17 10K (iss. 7-14-17) – these were the last ATM’s sold:
• Sold 5/1/17-6/30/17: $4,307,000gr. / 7,358,806(1,051,258 post/RS)= $.59/sh.($4.10/sh. Post/RS)
PPHM - O/S Shares History (’06–curr.)
4-30-06 35,876,438
1-31-07 39,222,440
4-30-07 39,222,440
7-6-07 45,233,123
7-31-07 45,242,123
10-31-07 45,242,123
1-31-08 45,242,123
4-30-08 45,242,123
7-31-08 45,242,123
10-31-08 45,242,123
1-31-09 45,242,123
4-30-09 45,537,711
7-10-09 47,392,883
7-31-09 47,393,783
**NOTE: 1:5 R/S eff. 10-19-09 (~237mm/$.64=>~47.4mm/$3.20) http://tinyurl.com/ykuw588
10-31-09 48,869,563 +1,475,780
1-31-10 50,903,404 +2,033,841
4-30-10 53,094,894 +2,191,490
6-21-10 54,388,917 +1,294,023 (6-22-10 ATM/mlv Form424)
7-9-10 55,069,449 +475,987 (4-30-10 10K iss. 7-14-10)
7-31-10 55,784,955 +715,506
10-31-10 59,220,742 +3,435,787
11-30-10 63,932,353 +4,711,611 (10-31-10 10Q iss. 12-9-10)
12-15-10 64,404,097 +471,744 (12-17-10 S-3: $75M Shelf Reg.)
1-31-11 66,813,419 +2,409,322
2-28-11 67,885,811 +1,072,392 (1-31-11 10Q iss. 3-11-11)
4-30-11 69,837,142 +1,951,331
7-8-11 71,069,858 +1,232,716 (4-30-11 10K iss. 7-14-11)
8-22-11 72,704,647 +1,634,789 (Proxy iss. 8-26-11)
8-31-11 73,284,016 +579,369 (424B5 iss. 9-2-11)
9-8-11 79,536,268 +6,252,252 (Roth Sale to 3 Inst’s @ $1.11/sh.)
10-31-11 82,638,201 +3,101,933
12-9-11 86,788,817 +4,150,616 (10-31-11 10Q iss. 12-12-11)
1-31-12 93,146,226 +6,357,409
2-29-12 98,873,172 +5,726,946 (1-31-12 10Q iss. 3-9-12)
4-30-12 101,421,365 +2,548,193
7-13-12 104,174,056 +2,752,691 (4-30-12 10K iss. 7-16-12)
7-31-12 104,178,431 +4,375 (7-31-12 10Q iss. 9-10-12)
8-16-12 104,191,176 +12,745 (prelim. proxy 14A http://tinyurl.com/c48bvof )
9-7-12 104,191,176 nochg (7-31-12 10Q iss. 9-10-12)
10-31-12 123,310,188 +19,119,012
12-7-12 132,539,783 +9,229,595 (10-31-12 10Q iss. 12-10-12)
1-31-13 133,770,614 +1,230,831
3-12-13 137,110,758 +3,340,144 (1-31-13 10Q iss. 3-12-13)
4-30-13 143,768,946 +6,658,188 (4-30-13 10K iss. 7-11-13)
7-5-13 151,602,765 +7,833,819 (4-30-13 10K iss. 7-11-13)
7-31-13 153,506,811 +1,904,046
9-5-13 156,461,114 +2,954,303 (7-31-13 10K iss. 9-9-13)
10-31-13 160,248,742 +3,781,628
12-6-13 160,325,891 +77,149 (10-31-13 10K iss. 12-10-13)
1-31-14 176,453,261 +16,127,370
3-3-14 176,481,054 +27,793 (1-31-14 10Q iss. 3-7-14)
4-30-14: 178,871,164 +2,390,110
7-7-14: 179,209,458 +338,294 (4-30-14 10-K cover page, iss. 7-14-14)
7-31-14: 179,216,032 +6,574 (7-31-14 10Q iss. 9-9-14)
8-22-14: 179,226,424 +10,392 (8-28-14 Proxy/Def14A)
9-5-14: 179,505,424 +279,000 (7-31-14 10Q iss. 9-9-14)
10-31-14: 182,000,583 +2,495,159 (10-31-14 10Q iss. 12-10-14)
12-5-14: 182,081,234 +80,651 (10-31-14 10Q cover pg., iss. 12-10-14)
12-19-14: 182,081,234 -0- (12-23-14 S-3)
1-31-15: 184,244,698 +2,163,464 (1-31-15 10Q iss. 3-12-15)
3-12-15: 188,332,872 +4,088,174 (“ “ “)
4-30-15: 193,346,627 +5,013,755 (4-30-15 10-K iss. 7-14-15)
7-10-15: 199,934,918 +6,588,291 (4-30-15 10-K/cover-pg, iss. 7-14-15)
7-31-15: 200,983,948 +1,049,030 (7-31-15 10Q iss. 9-9-15)
9-4-15: 202,124,031 +1,140,083 (“ “ “)
10-31-15: 225,824,551 +23,700,520 (10-31-15 10Q iss. 12-10-15)<=Incl. 18.5mm sh. Dart/EastCAP @1.08
12-9-15: 229,701,808 +3,877,257 (10-31-15 10Q iss. 12-10-15)
1-31-16: 232,231,242 +2,529,434 (1-31-16 10Q iss. 3-9-16)
3-8-16: 233,738,426 +1,507,184 (“ “ “)
4-30-16: 236,930,485 +3,192,059 (4-30-16 10K iss. 7-14-16)
7-11-16: 241,456,721 +4,526,236 (“ “ “)
7-31-16: 241,456,721 -0- (7-31-16 10Q iss. 9-8-16)
9-2-16: 242,381,850 +925,129 (“ “ “)
10-31-16: 251,765,279 +9,383,429 (10-31-16 10Q iss. 12-12-16)
12-8-16: 257,141,534 +5,376,255 (“ “ “)
1-31-17: 271,068,464 +13,926,930 (1-31-17 10Q iss. 3-13-17)
3-10-17: 297,709,478 +26,641,014 (“ “ “)
4-30-17: 44,014,040(x7)=308,098,280 +10,388,802 (4-30-17 10K iss. 7-14-17)
7-10-17: 1:7 R/S (~315mm/$.606=>~45mm/$4.24) http://tinyurl.com/ycohqn6j
7-10-17: 45,069,188 +1,055,148 (“ “ “)
7-31-16: 45,094,154 +24,966 (7-31-17 10Q iss. 9-11-17)
8-25-17: 45,096,081 +1,927 (8-25-17 Amended 10K http://tinyurl.com/yb5jq7vc )
9-6-17: 45,096,081 nochg (7-31-17 10Q iss. 9-11-17)
10-31-16: 45,172,632 +76,551 (10-31-17 10Q iss. 12-11-17)
11-27-17: 45,210,608 +37,976 (14A/Proxy iss. 12-7-17 https://tinyurl.com/y7qprpg9 )
12-6-17: 10-31-16: 45,212,760 +2,152 (10-31-17 10Q iss. 12-11-17)
= = = = = = = = = = = = = = = = = =
ATM Sales Summary (3/2009–6/30/2017). Also, PPHM O/S Shares History Table (’06–curr.) at the bottom of this post. At 12-6-17, common shares O/S = 45,212,760sh. (recall, 1:7 R/S was eff. 7-10-17)
ATM = “At-The-Market Sales Issuance”
-------
The 10-31-17 10Q (iss. 12-11-17 https://tinyurl.com/y75kwojs ) confirmed that NO ATM SALES OCCURRED AFTER THOSE REPORTED IN THE “SUBSEQUENT EVENTS” SECTION OF THE 4-30-17 10K (iss. 7-14-17) – these were the last ATM’s sold:
• Sold 5/1/17-6/30/17: $4,307,000gr. / 7,358,806(1,051,258 post/RS)= $.59/sh.($4.10/sh. Post/RS)
I. WM-SMITH 3-2009:
• $7.5mm ATM/Wm.SMITH 3-26-09: $7,500,000gr. / 2,150,759sh. = $3.49/sh. (commiss: 3%)
• $25mm ATM/Wm.SMITH 7-14-09: $25,000,000gr. / 7,569,314sh. = $3.30/sh. (commiss: 3%/1st$15mm, then 2%)
*Total Raised via WmSmith ATM Sales thru 7-31-10:
. . .Before 7-10-17 1:7RS: $32,500,000gr. / 9,720,073sh. = $3.34/sh.
. . .After 7-10-17 1:7RS: $32,500,000gr. / 1,388,582sh. = $23.41/sh.
II. MLV 6-2010: http://www.mlvco.com
$15mm ATM/MLV 6-22-10 (commiss: 2%) Form424: http://tinyurl.com/24txkxb
• Sold 6/22/10–10/31/10: $6,840,000gr. / 4,031,018sh. = $1.70/sh.
• Sold 11/1/10–11/30/10: $7,407,000gr. / 4,711,611sh. = $1.57/sh.
• Sold 12/1/10–1/31/11: $753,000gr. / 471,744sh. = $1.60/sh.
*Total Raised via MLV June’10 ATM Sales thru 1-31-11:
. . .Before 7-10-17 1:7RS: $15,000,000gr. / 9,214,373 = $1.63/sh.
. . .After 7-10-17 1:7RS: $15,000,000gr. / 1,316,339 = $11.40/sh.
III. MLV 12-2010: “Dec’10 AMI Agreement” http://www.mlvco.com
$75mm ATM/MLV 12-29-10 (commiss: max=5%) Form8K: http://tinyurl.com/2a6w76g
(pursuant to $75mm S-3 Shelf Reg. filed 12-17-10: http://tinyurl.com/2469b2d )
• Sold 12/29/10-1/31/11: $6,460,000gr. / 2,385,862sh. = $2.71/sh.
• Sold 2/1/11-2/28/11: $2,358,000gr. / 998,142sh. = $2.36/sh.
• Sold 3/1/11-4/30/11: $4,470,000gr. / 1,840,487sh. = $2.43/sh.
• Sold 5/1/11-7/31/11: $3,713,000gr. / 1,912,576sh. = $1.94/sh.
• Sold 8/1/11-10/31/11: $5,582,000gr. / 4,727,840sh. = $1.18/sh.
• Sold 9-2-12 Roth Direct: $6,940,000gr./ 6,252,252sh. = $1.11/sh.
• Sold 11/1/11-1/31/12: $10,961,000gr. / 10,308,025sh. = $1.06/sh.
• Sold 2/1/12-2/29/12: $5,871,000gr. / 5,726,946sh. = $1.03/sh.
• Sold 3/1/12-4/30/12: $1,263,000gr. / 2,198,543sh. = $.57/sh.
• Sold 5/1/12-6/30/12: $1,496,000gr. / 2,752,691sh. = $.54/sh.
• Sold 7/1/12-9/26/12: none**
• Sold 9/27/12-10/31/12: $16,719,000gr./ 18,557,928 = $.90/sh.
• Sold 11/1/12-11/30/12: $7,296,000gr./ 9,220,313 = $.79
• Sold 12/1/12-1/31/13: $1,540,000gr./ 1,131,282 = $1.36
• Sold 2/1/13-3/12/13: $330,000gr./ 201,154 = $1.64
*Total Raised via MLV Dec’10 ATM Sales thru 3-12-2013:
. . .Before 7-10-17 1:7RS: $75,000,000gr. / 68,214,041 = $1.10sh.
. . .After 7-10-17 1:7RS: $75,000,000gr. / 9,744,863 = $7.70sh.
IV. MLV 12-2012: “Dec’12 AMI Agreement” http://www.mlvco.com
$75mm ATM/MLV 12-29-12 (commiss: max=5%) Form8K: http://tinyurl.com/2a6w76g
(pursuant to $75mm S-3 Shelf Reg. filed 3-9-12: http://tinyurl.com/7dl7pjm )
• Sold 2/1/13-3/12/13: $4,475,000gr. / 3,132,402sh. = $1.43/sh.
• Sold 3/13/13-4/30/13: $8,897,000gr. / 6,188,273sh. = $1.44/sh.
• Sold 5/1/13-7/11/13: $12,729,000gr. / 7,927,016sh. = $1.61/sh.
• Sold 7/12/13-7/31/13: $2,468,000gr. / 1,690,864sh. = $1.46/sh.
• Sold 8/1/13-9/9/13: $4,372,000gr. / 3,057,431sh. = $1.43/sh.
• Sold 9/10/13-10/31/13: $4,708,000gr. / 3,262,958sh. = $1.44/sh.
• Sold 11/1/13-12/6/13: NONE – see 10Q note below.
• Sold 12/7/13-1/31/14: $28,130,000gr. / 16,045,717sh. = $1.75/sh.
• Sold 2/1/14-4/30/14: $3,017,000gr. / 1,543,383sh. = $1.95/sh.
• Sold 5/1/14-7/31/14: $425,000gr. / 226,700sh. = $1.92/sh.
• Sold 8/1/14-10/31/14: $3,891,000gr. / 2,494,835sh. = $1.56/sh.
• Sold 11/1/14-1/31/15: $1,878,000gr. / 1,261,825sh. = $1.49/sh.
*Total Raised via MLV Dec’12 ATM Sales thru 10-31-2014:
. . .Before 7-10-17 1:7RS: $75,000,000gr. / 46,831,404 = $1.60sh.
. . .After 7-10-17 1:7RS: $75,000,000gr. / 6,690,200 = $1.21sh.
V. MLV 6-2014: “Jun’14 AMI Agreement – up to $25mm” http://www.mlvco.com
Form 424B5 filed 6-13-15: http://tinyurl.com/ycxje6pj
• Sold 11/1/14-1/31/15: $1,193,000gr. / 869,504sh. = $1.43/sh.
• Sold 2/1/15-3/12/15: $6,204,000gr. / 4,354,954sh. = $1.44/sh.
• Sold 3/13/15-4/30/15: $6,147,000gr. / 4,457,299sh. = $1.38/sh.
• Sold 5/1/15-7/14/15: $8,896,000gr. / 6,534,400sh. = $1.36/sh.
• Sold 7/15/15-7/31/15: $1,270,000gr. / 1,003,830sh. = $1.27/sh.
• Sold 8/1/15-9/9/15: $1,290,000gr. / 1,091,508sh. = $1.18/sh.
*Total Raised via MLV Jun’14 ATM Sales thru 9-9-2015:
. . .Before 7-10-17 1:7RS: $25,000,000gr. / 18,311,495 = $1.37/sh.
. . .After 7-10-17 1:7RS: $25,000,000gr. / 2,615,928 = $9.56/sh.
VI. MLV 8-2015: “Aug’15 AMI Agreement – up to $30mm” http://www.mlvco.com
Form 424B5 filed 8-7-15: http://tinyurl.com/oc53ugn
• Sold 8/7/15-9/9/15: $892,000gr. / 1,091,000sh. = $1.18/sh.
• Sold 9/10/15-10/31/15: $4,294,000gr. / 4,059,478sh. = $1.06/sh.
• Sold 11/1/15-12/10/15: $2,261,000gr. / 1,939,413sh. = $1.17/sh.
• Sold 12/11/15-1/31/16: *NO ATM SALES*, per 1-31-16 10Q iss. 3-9-16
• Sold 2/2/16-4/30/16: *NO ATM SALES*, per 4-30-16 10Q iss. 7-14-16
• Sold 5/1/16-7/14/16: $937,000gr. / 1,876,918sh. = $.50/sh.
• Sold 7/15/16-9/8/16: *NO ATM SALES*, per 7-31-16 10Q iss. 9-8-16
• Sold 9/9/16-10/31/16: $2,513,000gr. / 6,432,439sh. = $.39/sh.
• Sold 11/1/16-12/12/16: $1,174,000gr. / 3,788,346sh. = $.31/sh.
• Sold 12/13/16-1/31/17: $3,404,000gr. / 11,069,646sh. = $.31/sh.
• Sold 2/1/17-3/13/17: $7,335,000gr. / 15,308,300sh. = $.48/sh.
• Sold 3/14/17-4/30/17: $2,883,000gr. / 4,486,182 = $.64/sh.
• Sold 5/1/17-6/30/17: $4,307,000gr. / 7,358,806 = $0.59/sh.
. . .Before 7-10-17 1:7RS: $30,000,000gr. / 57,072,288 = $.53/sh.
. . .After 7-10-17 1:7RS: $30,000,000gr. / 8,153,184 = $3.68/sh.
VII. NOBLE 8-2015: “Aug’15 Eq.Dist. Agreement – up to $20mm” http://www.noblelsp.com
Form 424B5 filed 8-7-15: http://tinyurl.com/yazonjyw
• Sold 11/1/15-12/10/16: $2,371,000gr. / 1,925,844sh. = $1.23/sh.
• Sold 12/11/15-1/31/16: $2,857,000gr. / 2,529,434sh. = $1.13/sh.
• Sold 2/1/16-4/30/16: $1,741,000gr. / 4,017,010sh. = $.43/sh.
• Sold 5/1/16-7/14/16: $1,233,000gr. / 2,649,318sh. = $.47/sh.
• Sold 7/15/16-9/8/16: *NO ATM SALES*, per 7-31-16 10Q iss. 9-8-16
• Sold 9/9/16-10/31/16: $1,283,000gr. / 2,988,086sh. = $.43/sh.
• Sold 11/1/16-12/12/16: $496,000gr. / 1,587,909sh. = $.31/sh.
• Sold 12/13/16-1/31/17: $904,000gr. / 2,857,284sh. = $.32/sh.
• Sold 2/1/17-3/13/17: $6,006,000gr. / 11,273,296sh. = $.53/sh.
• Sold 3/14/17-4/30/17: $3,109,000gr. / 4,896,376sh. = $.63/sh.
. . .Before 7-10-17 1:7RS: $20,000,000gr. / 34,724,557 = $.58/sh.
. . .After 7-10-17 1:7RS: $20,000,000gr. / 4,960,651 = $4.03/sh.
TOTAL ALL A-T-M SALES – INCEPTION (3-2009) THRU 6-30-2017:
. . .Before 7-10-17 1:7RS: => $272,500,000gr. / 244,088,231sh. = $1.12/sh.
. . .After 7-10-17 1:7RS: => $272,500,000gr. / 34,869,747sh. = $7.81/sh.
= = = = = = = = = = = = = = = = = =
O/S WARRANTS & STOCK-OPTIONS A/O 10-31-2017 (10Q pgs. 13-14):
• WARRANTS: As of 7-31-17, warrants to purchase 39,040 shares at an exercise price of $17.29 were outstanding and are exercisable thru Aug30 2018. These warrants were issued in FY’13 in connection with the Aug.2012 [Oxford] loan agreement, which was paid in full Sept.2012.
• STOCK OPTIONS OUTSTANDING A/O 10-31-17: 4,123,054 shares at a wgt.avg. exercise price of $8.96.
- - - - - - - - - - - - - - - - -
Latest 10K 4-30-17 iss. 7-14-17 http://tinyurl.com/ycxu4l5n PR: http://tinyurl.com/yb4wulvu (Cash 4-30-17=$46.8mm); Amended 8-25-17: http://tinyurl.com/yb5jq7vc
Latest 10Q 10-31-17 iss. 12-11-17 https://tinyurl.com/y75kwojs PR: http://ir.peregrineinc.com/releasedetail.cfm?ReleaseID=1051443 (Cash 10-31-17=$27.7mm)
ALL SEC filings for PPHM: http://tinyurl.com/6d4jw8
10-13-16 ASM Voting Results: http://tinyurl.com/ycrrrb43
Per Proxy, SK has options to buy 425,161sh@$9.29
Outstanding Equity Awards at FY/e 4-30-17 (pg. 39 of Proxy https://tinyurl.com/y7qprpg9 )
Steven W. King
7,143 @13.65 exp. 1/11/2018
31,571 @20.51 2/1/2020
20,357 @17.08 5/2/2021
37,519 @6.65 2/17/2022
35,714 @3.22 5/4/2022 **
28,571 @8.26 12/27/2022
57,143 @9.87 5/6/2023
35,714 @9.73 10/15/2023
57,143 @12.25 5/6/2024
37,500 @ 9.17 5/11/2025 – plus 5,357 not yet vested (qtly over 2yrs beg. 8-11-15) = 42,857
26,787 @3.50 6/2/2026 – plus 44,642 not yet vested (qtly over 2yrs beg. 9-2-16) = 71,429 **
= = = = = =
Total: 425,161sh. X Avg=$9.29/sh. = $3,947,718 out of pocket to exercise them all.
** 107,143sh. currently in the money (once all vested)
12-7-17/PROXY; 11-28-17 PPHM+RONIN Settle(4 New Directors)
THESE UPDATES:
I. 12-7-17: Peregrine’s 14A DEF Proxy Statement: https://tinyurl.com/y7qprpg9
……At the 2017 Annual Meeting (1-18-18, 10am), the Company asks you to vote on 4 proposals…*SEE BELOW*
II. 11-28-17: PPHM+RONIN Settle, NEW BOD(7): Joseph Carleone (Chair), Richard Hancock, Gregory Sargen, Joel McComb, Roger Lias, Mark Bamforth, Patrick Walsh (King, Johnson, Swartz, Pohl resign). Complete Agreement: https://tinyurl.com/y7b3gkc2
- - - - - - -
See below for full PPHM-RONIN History of PR’s, Letters, 13-D’s, Form4’s, and Proxy’s (sorted: newest=>oldest).
KNOWN UPCOMING:
Dec11 After Mkt: FY'18Q2 (qe 10-31-17) Financials & Conf. Call - http://ir.peregrineinc.com/events.cfm
Dec11-15/Avid Booth #311: KNect365’s Antibody Eng. & Therapeutics Conf., SanDiego https://lifesciences.knect365.com/antibody-engineering-therapeutics
Jan18: Peregrine's 2017 Annual Shareholder’s Meeting – 11-13-17/PR: http://tinyurl.com/y9tdcob5 VotingRecDate=11-27-17; PROXY/14A: https://tinyurl.com/y7qprpg9
= = = = = = = = = = = = = = = = = = = = = = = = = = = = = = =
11-13-17: Large Ownership Summary(40.2%) - now 18,163,171shs., 40.2% of 45,210,608 O/S at 11-27-17.
(Ronin+SW/Stafford+Farley+White, Eastern Cap./K.Dart, Institutions incl. Tappan’s 11-13-17/13G)
#1: 10-27-17/PR: Group Ronin Trading/SWInvest (John Stafford III+R.Farley+Stephen White) now has 9.6% stake (4,325,889sh.) in PPHM
...4,173,391 COMMON – 9.2% of 45,210,608 common O/S at 11-27-17 (total beneficial=4,325,889 if Pref. conv. x1.1905 to Common)
...128,099 PREFERRED – 7.8% of 1,647,760 preferred O/S at 7-31-17
NOTE: ALL historical common #’s adjusted for the 1:7 R/S eff. 7-10-17.
#2: 10-30-15: Kenneth Dart (Eastern Capital) acquires 9.5% stake (4,300,992sh.) in PPHM http://tinyurl.com/y95yskck
...3,777,183 COMMON – 8.4% of 45,210,608 common O/S at 11-27-17 (total beneficial=4,300,992 if Pref. conv. x1.1905 to Common, 9.5%)
...440,000 PREFERRED – 26.7% of 1,647,760 preferred O/S at 7-31-17
…...9-29-17/Form4: http://tinyurl.com/y9oadloa <=No chg. In Ownership; status update?
RONIN/SW 13D SUMMARY:
13D TransDates COMMON-CHG PREF-CHG ENDING-COMMON ENDING-PREF.
3-2-17 1/20/17-3/1/17 +2,947,425 +51,364 2,947,425 51,364 http://tinyurl.com/jr42u23
3-10-17 3/2/17-3/9/17 +433,509 +25,661 3,380,934 77,025 http://tinyurl.com/ydxra96u
4-17-17 3/28/17-4/10/17 0 +23,334 3,380,934 100,359 http://tinyurl.com/lanjddc
5-19-17 5/1/17-5/17/17 0 +23,140 3,380,934 123,499 http://tinyurl.com/mgnn92x
6-20-17 3/10/17-6/16/17 +378,170 0 3,759,105 123,499 http://tinyurl.com/y76q5rqu
6-29-17 6/21/17 +7,143 0 3,766,248 123,499 http://tinyurl.com/y9sp8bfv
7-14-17 6/29/17-7/7/17 +34,891 +3,600 3,801,139 127,099 http://tinyurl.com/ybra4s69
8-29-17 8/4/17-8/15/17 +27,252 0 3,828,391 127,099 http://tinyurl.com/yay55u3p (14A)
10-10-17 8/16/17-10/10/17 +45,000 0 3,873,391 127,099 http://tinyurl.com/ybvkjd3e (14A)
10-17-17 10/13/17-10/16/17 +109,573 +1,000 3,982,964 128,099 http://tinyurl.com/y983myco
10-27-17 10/17/17-10/27/17 +190,427 0 4,173,391 128,099 http://tinyurl.com/y7hslequ (14A)
SPLITOUT 13D GROUP into Ronin(John Stafford III, R.Farley) & SW-Partners(Stephen White):
Ronin Trading (Stafford) 3,173,391 115,299 (beneficial=3,310,652)
Ronin’s Roger Farley 300,000 1,000 (beneficial=301,190)
SW-Partners (White) 700,000 11,800 (beneficial=714,047)
- - - - - - - - - - - - - - -
Plus, INSTITUTIONS a/o *9-30-17: 9,536,290sh. = 21.1% <=*incl. Tappan’s 11-13-17/13G
...8,491,679 + 1,044,611(Tappan adds after 9-30-17) = 9,536,290
http://www.nasdaq.com/symbol/pphm/institutional-holdings
TOP13:
Tappan St. Partners 3,915,611 +2,259,359 <=per 11-13-17/13G(see below**)
Vanguard Group 1,518,110 +488,355
Blackrock (Larry Fink) 761,302 -44,720
Bandera Partners 555,000 +351,996
Renaissance Technologies 547,190 +66,030
Geode Capital Mgt. 225,676 -3
Fondren Mgt. 205,000 NEW
Stifel Financial 204,305 +200,762
Eqis Capital Mgt. 143,595 -6,804
Wells Fargo & Co. 117,829 +69,488
Connor Clark & Lunn 98,772 NEW
Brown Advisory Sec. 82,858 -1
Pdt Partners 82,800 NEW
And this big Q3 drop:
#18 Kennedy Capital Mgt. 69,820 -1,324,246
**11-13-17/13G: Tappan Street (Prasad Phatak) http://tinyurl.com/y85pz4fs
2 Funds + P.Phatak’s personal shares:
...Partners LLC, Tappan St. Fund L.P. 1,540,000 0
...Tappan St. Partners Ideas Fund L.P. 2,289,504 +1,597,927 since 8-14-17
...Prasad Phatak (Principle Owner) 86,107 +19,000 since 8-14-17
Total TAPPAN: 3,915,611 (8.7%)
Note: Tappan Street (total) Holdings history:
a/o 3-31-17: 914,304
a/o 6-30-17: 1,656,252
a/o 8-14-17: 2,298,684 5.1% (of 45,069,188 O/S as of 7-11-17)
a/o 9-30-17: 2,871,000 6.4% (of 45,096,081 O/S as of 9-11-17)
a/o 11-6-17: 3,915,611 8.7% (of 45,096,081 O/S as of 9-11-17)
Final Qtly Inst. Holdings from Nasdaq:
3-31-17: 5,690,888
6-30-17: 6,834,425
9-30-17: 8,491,679

Common O/S update given in Proxy: 45,210,608 as of 11-27-17, up from 45,096,081 as 9-6-17.
7-10-17: 45,069,188 +1,055,148
7-31-16: 45,094,154 +24,966 (7-31-17 10Q iss. 9-11-17)
8-25-17: 45,096,081 +1,927 (8-25-17 Amended 10K http://tinyurl.com/yb5jq7vc )
9-6-17: 45,096,081 nochg (7-31-17 10Q iss. 9-11-17)
11-27-17: 45,210,608 +114,454 (14A/Proxy iss. 12-7-17 https://tinyurl.com/y7qprpg9 )
12-4-17/OCBJ: “Peregrine Full Speed Ahead In Shift To CDMO - Board To Add Mfg. Experience”
By: Sherry Hsieh
“Less than 6 months since investors issued a public statement calling for Peregrine Pharmaceuticals Inc. to cease clinical dev. activities of its cancer drug and focus on growing its biologics manufacturing business, the company is well on its way to completing the transition...” (must subscribe)
http://www.ocbj.com/news/2017/dec/04/peregrine-full-speed-ahead-shift-cdmo/
If anybody subscribes to this, maybe they could share a paraphrase or a couple of quotes.