Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
5-3-17 BioPhamaREP quotes Avid re: MilliporeSigma 2,000L Expansion
5-3-17 BioPharma-Reporter: “Latest Avid investment to turn bioreactor capacity up to 11,000L”
Avid Bioservices will add multiple MilliporeSigma 2,000L bioreactors at its Californian single-use production plant in an effort to its increase annual revenue to over $100m.
The Myford facility [Avid II] came online in Dec.2015, but continued demand for clinical & commercial biomanufacturing has led to a capacity upgrade.
While an Avid spokesperson told Biopharma-Reporter the firm has experience with a “wide number of different single-use systems across multiple vendors,” this is the CMO’s first foray into the 2,000L scale...
The investment will “bring total capacity within the Myford facility to over 9,000L of capacity at the 1,000 & 2,000L scale and the total capacity at our 2 facilities to over 11,000L,” we were told.
Avid’s other facility is the Franklin plant, also located in Tustin, CA.
“With the installation of the new bioreactors at Myford, we expect that facility, when operating in campaign mode, can exceed more than $75m annually. This would bring our total potential annual revenue across both of manufacturing facilities to well over $100m.”
http://www.biopharma-reporter.com/Upstream-Processing/Avid-selects-MilliporeSigma-2-000L-single-use-bioreactors-at-CA-plant
= = = = = = = = = =
5-1-17/MPS-PR: MilliporeSigma Signs Biomanufacturing Deal with Avid Bioservices
Avid Bioservices, a wholly-owned subsidiary of Peregrine Pharmaceuticals, reports that it will upgrade its Myford clinical and commercial manufacturing facility with multiple Mobius 2000L single-use bioreactors from MilliporeSigma. The new bioreactors, to be installed in mid-2017, will expand the company’s mfg. Capacity...
“We ran a competitive process and chose the MilliporeSigma Mobius 2,000L single-use bioreactor system because it offers technical and ease of use advantages over competing single-use systems,” said Steven W. King, President, Avid Bioservices. “The confidence in the choice has only been further reinforced by our experience to date with the Mobius system up to the 1000L scale as part of a pilot program. Adding the Mobius technology to expand Avid’s Myford biomanufacturing capacity highlights our commitment to offer customers the latest single-use equipment and flexible solutions. The planned installation of MilliporeSigma’s Mobius 2000L bioreactors opens new opportunities for current and future Avid customers who require greater manufacturing capacity. We have already secured customer commitments for the 2,000L capacity as soon as it is installed in our Myford facility [Avid II] and believe the advanced technology it offers will be a key factor in continuing to grow our contract development and mfg. organization business.” King added that the first 2,000L pilot batch is planned for later this month.
http://www.genengnews.com/gen-news-highlights/milliporesigma-signs-biomanufacturing-deal-with-avid-bioservices/81254281
3-13-17 CC, CEO Steve King: “...An important component of our Avid growth strategy is capacity expansion within our Myford facility. We are currently on track to install two 2,000-liter bioreactors [Mobius 2000L single-use] in the facility within the next few months, with a book of business for the reactors already in place. We believe the total capacity potential of the facility, when operating in campaign mode, can exceed more than $75mm annually, bringing us to well over $100mm in total potential revenue between our 2 mfg. facilities, providing us with adequate capacity to continue Avid revenue growth through FY’18. As we look beyond this FY and into the future, based on current operations & projected demand from our clients, we have also recently secured addl. space adjacent to our Myford facility, which we already have the use for as part of our growing operations. And additionally, would also allow us to further expand mfg. capacity based on committed business. While we will only begin converting space into mfg. capacity once client commitments and other necessary financing is in place, this puts us in an excellent position for continuing to grow the business beyond the coming FY.” http://tinyurl.com/grhwjvy
10-13-16 ASM: Shareholder Attendee Reports http://tinyurl.com/z3nv3g9

2nd NCCN Trial (GBM Bavi+RAD+Temodar) hits ClinicalTrials.gov. “Not Yet Recruiting”, Start=6/30/17. Note it’s a Phase II N=36, not Ph.I-II as originally stated in the 9-6-16 NCCN PR. 2 Sites: Mass Gen & Dana Farber Cancer Institute. (both Boston).
NCCN Bavituximab Trials Announced 9-6-16 - To Begin "Early 2017" http://tinyurl.com/gutgwb5
...#1: Ph1/HepC-Related Hepatocellular(Liver) (Bavi+RAD+Bayer’s Nexavar=Sorafenib), MOFFITT CC (N=18)
. . . . . . .PI: Jessica Frakes, MD https://clinicaltrials.gov/ct2/show/NCT02989870 <=Recruiting a/o 3-27-17
...#2: Ph2/Newly Diag. Glioblastoma (Bavi+RAD+Merck’s Temodar), MASS-GEN. & DANA FARBER (N=36)
. . . . . . .PI: Elizabeth Gerstner, MD https://clinicaltrials.gov/ct2/show/NCT03139916 <=Added 5-2-17
...#3: Ph2/Progressive Squamous Head+Neck (Bavi+Merck’s Keytruda), JOHNS-HOPKINS(Sidney Kimmel CC)
. . . . . . .PI: Ranee Mehra, MD https://clinicaltrials.gov/ct2/show/XXXXXXXXX - See Dr. R.Mehra Jan'17/IFN-y Biomarker: http://tinyurl.com/h8gzkww
= = = = = = = = = = = = = =
3-13-17/CC JOE SHAN (VP/Clin.&Reg. Affairs) http://tinyurl.com/grhwjvy
“…Now turning to new trials, I’m happy to report that the 3 clinical trials to be funded through our collaboration with the NCCN [http://tinyurl.com/gutgwb5 ] are advancing as expected. The first study, expected to begin patient enrollment shortly, is being conducted by Dr. Jessica Frakes, at the Moffitt Cancer Center. This study, which builds upon a prior investigator sponsor trial [3-25-15/Dr. Adam. Yopp(UTSW) http://tinyurl.com/opkh5qy ], will evaluate the combination of Stereotactic Body Radiation Therapy, sorafenib, and bavituximab, for the treatment of Unresectable Hepatocellular [Liver] Carcinoma. We are interested in this trial design as radiation has been shown to increased PS expression on the surface of tumor cells, induce immunogenic tumor cell depth, and increase tumor specific T-cell activity. In addition, NCCN has recently communicated that both the study of bavituximab, temozolomide, and radiation in newly diagnosed Glioblastoma and the trial of bavituximab & temozolomide in recurrent Head & Neck cancer, which has progressed on checkpoint inhibitor treatment, are on track to be initiated by mid-year.”
N40 re: The Rosenberg w/Wolchok ACT Article... Try this:
Do this Google: https://www.google.com/#q=rosenberg+wolchok
Select the one that says, "Immune Therapy Steps Up the Attack".
When I do it this way, I can see the whole article - it's like Google overrides the subscription requirement in some way. ??
Does this work for you like it does for me?
Good find; Rosenberg="Father of ACT" (w/Wolchok etal). Here's a better link that allows you to see the 2010 article, "Immune Therapy Steps Up the Attack" (via Google search - lets you in, I THINK): http://tinyurl.com/n87tuvv
"ACT has taught us a great deal, says Wolchok at Sloan-Kettering - — for example, that people with heavy cancer burdens can be helped just by expanding and reintroducing their T cells. But ACT faces daunting hurdles in “being able to produce the cell product for every patient who needs it,” he says. Even in Rosenberg's lab, the cells don't always grow, says Wolchok, who has referred patients to the program..."
======
Oh, 5-2014 Video: "Dr. Rosenberg on Dr. Jedd Wolchok's Accomplishments"
http://www.giantsofcancercare.com/news/dr-rosenberg-on-dr-jedd-wolchoks-accomplishments
NCI’s Steven Rosenberg speaking 5-5-17 on “Improving ACT” at an AACR workshop in Bethesda, whose chair is PPHM collaborator, Duke’s Herbert K. Lyerly. I just wonder if Dr. Rosenberg is aware of the Jedd Wolchok Lab study, “PS Targeting Improves ACT, Eliminates Advanced Tumors” presented 4-3-17 at AACR’17?
May3-5 2017: “AACR’s Accelerating Anticancer Agent Dev. & Validation Workshop”, Bethesda
"AAADV is the only workshop held in collaboration with the U.S. FDA designed specifically to help participants understand and negotiate the drug development approval process to get effective cancer treatments to patients more quickly."
https://www.acceleratingworkshop.org/2017/workshop
Workshop Chair: Herbert K. Lyerly, Duke Univ. MC (PPHM Collaborator** – see: http://tinyurl.com/zzryfok )
5-5-17 AAADV Workshop Plenary Session III: “New Developments in Cancer Immunotherapy: Generating Prolonged Anti-Cancer Immunity”
Moderators: Ke Liu, FDA; Jeffrey Weber, New York Univ.
**”Improving Adaptive Cell Therapy (ACT) for Cancer” - Steven Rosenberg, NCI
**”Designing New CARs for Cancer Treatment” - Marcela Maus, MassGen
**”Beyond Checkpoint Inhibitors: New Approaches to Altering the Tumor Environment” - Roy S. Herbst, Yale Univ.
---------
**The Duke/H. Kim Lyerly collab: ex: “Anti-PS + Anti-PD-L1 Leads to Greater Anti-Tumor Responses in TNBC” - latest is 10-22-16/AACR I-O Conf. – see: http://tinyurl.com/zzryfok
---------
Is NCI’s Dr. Steven Rosenberg aware of the MSKCC Jedd Wolchok Lab “PS-Targeting Improves ACT Therapy” study presented 4-3-17 at AACR’17?
...AACR17 MSKCC+PPHM: 4-3-17 #1651, “PSTargeting+Adoptive TCell Transfer (ACT) Eliminates Adv. Tumors w/o Off-Target Toxicities, Melanoma” <=NEW(2nd) Joint MemSloan/PPHM study SEE: http://tinyurl.com/lxlltd6
P.Lytle’s entire 3-13-17/CC Delist/Appeal statement so people can see put the one sentence extracted in context to his entire statement and make their own minds up...
3-13-17 Qtly. Conf. Call (King/Shan/Worsley/Lytle) Transcript http://tinyurl.com/grhwjvy
PL: Switching gears now, I’d like to address our plans to preserve our NASDAQ listing. At a backdrop, last April, we were notified by the NASDAQ that our share price did not meet the $1 min. bid price for 30 consecutive days, which is a requirement for continued listing on the NASDAQ market. At that time, we were automatically afforded an initial compliance period of 180 days or until Oct. 10, 2016 to regain compliance. At the end of the initial compliance period, we requested and were granted a 180 day extension to regain compliance. This 2nd extension period will end on April 10, 2017. Let me discuss our strategy and future steps we plan to take over the next few months. First & foremost, we have determined that it is not in the best interest of our stockholders to affect a reverse stock split prior to April 10, 2017. If our share price does not trade above $1 for 10 consecutive trading days by April 10th, we expect to receive a notice of delisting. If we received this notice, we will appeal this decision within the required 7 days and this appeal will stay any delisting actions by NASDAQ. At that point, NASDAQ will schedule a hearing, which is typically held within 45 days, and we will present our plan to regain compliance. We are considering several paths to regain compliance, including having addl. time to continue to allow the market to adjust, to reflect the current value of Avid as evidence with the recent movement in our share price. The hearing panel could afford us up to 180 addl. days to regain compliance, although this addl. time is not guaranteed and this decision is at the discretion of the NASDAQ’s hearing panel. If we are not afforded addl. time from this panel, it is important to note that our stockholders approved the Board’s ability to implement a reverse stock split at a ratio of up to 1:7 as a means to regain compliance and to preserve our listing on NASDAQ. During this entire process, Peregrine will continue to be listed on that NASDAQ capital market as it does today. We will keep you informed on any material developments through our SEC filings on Form 8-K as we execute on this plan.
3-13-17 Qtly. Conf. Call (King/Shan/Worsley/Lytle) Transcript http://tinyurl.com/grhwjvy
Dr.Raymond.Birge will speak on 5-19-17 at the ICHC’17/May18-21/Turkey (delayed from June 2016 due to terrorist attack). The ICHC website just added Dr. Birge’s talk title: “PS Sensing by TAM Receptors Regulates AKT-dependent Chemo-Resistance & PD-L1 Expression”
May18-21 2017: “ICHC’17: 15th Intl. Congress of Histochemistry & Cytochemistry - from Molecules to Diseases”, Antalya Turkey
“The main goal of ICHC 2017 is to bring the worldwide histochemists together and provide an environment for close cooperation, exchange of information, and collaborations.”
http://www.ichc2017.com Pgm: http://www.ichc2017.com/default1.asp?p=scientific
Speakers: http://www.ichc2017.com/default1.asp?p=invited-chairs
Track: “Cellular Aging & Cell Death” (sponsored by: Intl. Cell Death Society):
5-19-17/12pm: Dr. Raymond Birge (Rutgers Univ. http://birgelab.org ), “Phosphatidylserine Sensing by TAM Receptors Regulates AKT-dependent Chemo-Resistance & PD-L1 Expression”
= = = = = = = = = = = = = = = = = = =
Also Speaking 6-20-16 in ISRAEL:
Jun18-20 2017: “ICDS’17: Annual ICDS Conf. on Cell Death in Development & Disease”, Israel
“The conference will be under the umbrella of the Intl. Cell Death Society (ICDS). The ICDS is a organization attempting to promulgate basic research & translational studies on the broad topic of cell death.”
http://www.weizmann.ac.il/conferences/CDDD2017
ICDS = Intl. Cell Death Society
Pgm: http://www.weizmann.ac.il/conferences/CDDD2017/program
6-20-17 Session IX: Immunity & Clearance of Dying Cells II
...Chair: Simone Fulda (Goethe-University; DKFZ, Germany)
11:30-12:00pm: Raymond Birge, Rutgers Univ.: “Regulation of Efforocytosis by Phosphatidyserine Receptor”
...Def. Efferocytosis: “the process by which dying/dead cells (e.g. apoptotic or necrotic) are removed by phagocytic cells (neutrophils, macrophages, dendritic cells, etc.).”
= = = = = = = = = = = = = = = = = = = = = = = = =
2016: ICHC’16 Delayed until May 2017...
Rutgers' Dr. Raymond Birge, “PS: a Global Immunosuppressive Signal...”. Spoke at Peregrine's 11-2015 “Sci. Session” at SITC'15/Maryland; was the chair in June'16 of the ICDS'16 session where Dr. Rolf Brekken (PPHM SAB) spoke; and now also speaking in late June'16 at ICHC'16/Istanbul on, “Phosphatidylserine is a Global Immunosuppressive Signal in Cell Death & Cancer”… (Rutgers Univ. http://birgelab.org “The Birge laboratory conducts basic science focused on the eradication of cancer.”)
-----
Jun19-22 2016: *Delayed till May'17 due to Unfortunate World Events* “ICHC'16 - From Molecules To Diseases”, Istanbul Turkey http://tinyurl.com/h4zjvo4 - ….Rutgers' Dr. Raymond Birge ( http://birgelab.org ) “Phosphatidylserine is a Global Immunosuppressive Signal in Cell Death & Cancer” - see http://tinyurl.com/j3whbx3
= = = = = = = = = = = = =RECALL:
June2-4 2016: “ICDS 2016: Translational Implications in Cell Death", Cork, Ireland
ICDS = Intl. Cell Death Society
http://www.celldeath-apoptosis.org/extensions/templates/next-meeting-html/tentative-program-cork.html
*Chair (1 of 9): Ray Birge** (USA)
6-3-16 Session: Cell Death & Cancer Therapy
Dr. Rolf Brekken (USA, UTSW, PPHM SAB): “Blockade of Phosphatidylserine & Immune Activation in Cancer”
- - - - - - -
2-26-16/CellDeath+Differentiation(PDF): “Phosphatidylserine Is A Global Immunosuppressive Signal In Efferocytosis, Infectious Disease, And Cancer”
RB Birge*,1,10, S Boeltz*,2,10, S Kumar1,10, J Carlson3,10, J Wanderley4,10, D Calianese1,10, M Barcinski5,10, RA Brekken6,7,10, X Huang6,7,10, JT Hutchins3,10, B Freimark3,10, C Empig3,10, J Mercer8,10, AJ Schroit9,10, G Schett2,10 and M Herrmann*,2,10
–----
Also Recall… 10-29-15: Rutgers' Dr. Raymond Birge, John-Hopkins-Univ/Seminar: “PS is a Global-Immune-Checkpoint In Cancer” (12-1pm) http://tinyurl.com/oks5uo6
...Dr.Birge: PhD, Vice Chair, Professor, Dept of Molecular, Biochemistry & Molecular Genetics, Rutgers Univ. ( http://birgelab.org )
VIDEO(2:54): “Apoptosis – PS Exposed on Cell Surface”
Stoney, very smart friend rec’s you call DUKE – says Duke is the leader in fighting Brain Cancer… I quickly put this together…. Just call them - find out about the NEW Temodar+Vaccine trial (Ph.2 I think – starts soon).
4-14-17: “Immunotherapy For Glioblastoma Well Tolerated; Survival Gains Observed
Small, Phase I (DUKE) Trial Of A Dendritic Cell Vaccine Supports Further Study In Larger Trials”
...Batich and colleagues -- including senior author John Sampson, MD, PhD, chair of Duke's Department of Neurosurgery -- treated 11 patients as part of a single arm study to test the safety of using a dose-intensified regimen of temozolomide [Merck’s Temodar] along with a dendritic cell vaccine therapy that selectively targets a cytomegalovirus (CMV) protein. CMV proteins are abundant in glioblastoma tumors, but are absent in surrounding brain cells… "These are surprisingly promising clinical outcomes," Sampson said. "However, it is important to emphasize that this was a very small study that used historical comparisons rather than randomizing patients to two different treatments, but the findings certainly support further study of this approach in larger, controlled clinical trials." The research team has received approval to launch a new study that will compare the standard dose of temozolomide vs. the dose-intensified regimen along with the vaccine in glioblastoma patients.
https://www.sciencedaily.com/releases/2017/04/170414105842.htm
Also: https://www.oncology-central.com/2017/04/18/promising-results-reported-glioblastoma-vaccine/
Also: http://blog.braintumor.org/national-brain-tumor-society-funding-and-support-of-innovative-research-helps-bring-about-an-immunotherapy-clinical-trial-with-encouraging-results/
4-21-17: “Cancer breakthrough: Duke Clinical Trial Destroys Woman’s Brain Tumor”
http://www.healthnutnews.com/cancer-breakthrough-duke-clinical-trial-destroys-sc-womans-brain-tumor/
Duke Clinical Trials: http://www.dukecancerinstitute.org/clinical-trials
Duke Cancer Institute: http://www.dukecancerinstitute.org
Ref. Guide:
http://www.dukecancerinstitute.org/sites/default/files/ncprg_i.pdf Duke consultation and referral center - For more information on services offered at the Duke Cancer Institute, contact the Duke Consultation and Referral Center at 888-275-DUKE (888.275.3853).
JCNJ, ASCO titles pub. yesterday: Analysis/Sunrise PD-L1 Expression
Jun2-6: ASCO 2017, Chicago https://am.asco.org (Abstracts: Titles/Apr20, Full=May17)
6-3-17 1:15-4:45pm, Abstract #11603, Track: Tumor Biology
“Preliminary Correlative Analysis of PD-L1 Expression from the Sunrise Study”, Nikoletta Lea Kallinteris (1st Author), Peregrine Pharmaceuticals
PPHM Exhibiting – booth #13124 (next to Moffitt CC)
NOTE: Known Sunrise Biomarkers thru 4-2017(AACR): B2GPI, Complement+IL10, IFN-y – see http://tinyurl.com/ktzr78
= = = = =ASCO'17:
Abstract Titles: rel. 4-20-17 http://iplanner.asco.org/am2017
Full Abstracts: rel. 5-17-17 5pmET http://abstracts.asco.org
Known Upcoming Events (incl. ASCO’17: Analysis/Sunrise PD-L1 Expression)
May18-21: ICHC’17, 15th Intl. Congress of Histo/Cyto Chemistry - from Molecules to Diseases, Turkey http://tinyurl.com/m4gyh2y
...May19: Raymond Birge (Rutgers), “PS is a Global Immunosuppressive Signal in Cell Death & Cancer”
Jun2-6: ASCO 2017, Chicago https://am.asco.org (Abstracts: Titles/Apr20, Full=May17)
6-3-17 1:15-4:45pm, Abstract #11603, Track: Tumor Biology
“Preliminary Correlative Analysis of PD-L1 Expression from the Sunrise Study”, Nikoletta Lea Kallinteris (1st Author), Peregrine Pharmaceuticals
…...NOTE: Known Sunrise Biomarkers thru 4-2017(AACR): B2GPI, Complement+IL10, IFN-y – see http://tinyurl.com/ktzr78
PPHM Exhibiting – booth #13124 (next to Moffitt CC)
Jun18-20: ICDS’17, Annual ICDS Conf. on Cell Death in Development & Disease, Israel http://www.weizmann.ac.il/conferences/CDDD2017
...Jun20 11:30-12:00pm: Raymond Birge (Rutgers), “Regulation of Efforocytosis by Phosphatidyserine Receptor”
Jun19-22/Avid Booth #1411: BIO Intl. Convention, SanDiego http://www.convention.bio.org/2017
Aug21-25/Avid Booth #403: CHI's 9th Annual BioProcessing Summit, Boston http://www.bioprocessingsummit.com
Sep25-28/Avid Exhibiting: Informa’s BPI’17: BioProcess Intl. Conf. & Exhibition, Boston https://lifesciences.knect365.com/bioprocessinternational
~Jul13: FY'17Q4 (fye 4-30-17) Financials & Conf. Call - http://ir.peregrineinc.com/events.cfm
~Sep7: FY'18Q1 (qe 7-31-17) Financials & Conf. Call - http://ir.peregrineinc.com/events.cfm
Yes, Rutgers' Dr.Birge is speaking 6-20-17/Israel on PS. I found it, "ICDS 2017"...
Jun18-20 2017: “Annual ICDS Conf. on Cell Death in Dev. & Disease”, Israel
“The conference will be under the umbrella of the Intl. Cell Death Society (ICDS). The ICDS is a organization attempting to promulgate basic research & translational studies on the broad topic of cell death.”
http://www.weizmann.ac.il/conferences/CDDD2017
ICDS = Intl. Cell Death Society
Pgm: http://www.weizmann.ac.il/conferences/CDDD2017/program
6-20-17 Session IX: Immunity & Clearance of Dying Cells II
...Chair: Simone Fulda (Goethe-University; DKFZ, Germany)
11:30-12:00pm: Raymond Birge, Rutgers Univ.: “Regulation of Efforocytosis by Phosphatidyserine Receptor”
Bio, Dr. Raymond Birge will speak at ICHC’17/May18-21(Turkey), delayed from June 2016 (due to terrorist attack), where he was scheduled to speak on, “Phosphatidylserine is a Global Immunosuppressive Signal in Cell Death & Cancer”. Dr. Birge is now scheduled to speak on 5-19-17, but the title is not currently listed in the progam – see below.
May18-21 2017: “ICHC’17: 15th Intl. Congress of Histochemistry & Cytochemistry - from Molecules to Diseases”, Antalya Turkey
“The main goal of ICHC 2017 is to bring the worldwide histochemists together and provide an environment for close cooperation, exchange of information, and collaborations.”
http://www.ichc2017.com Pgm: http://www.ichc2017.com/default1.asp?p=scientific
Speakers: http://www.ichc2017.com/default1.asp?p=raymond
5-19-17: INVITED SPEAKER – Dr. Raymond Birge (Rutgers Univ. http://birgelab.org ): Intl. Cell Death Society Sponsored Session: “Cellular Aging & Cell Death”
= = = = = = = = = = = = = = = = = = = = = = = = =
2016: ICHC’16 Delayed until May 2017...
Rutgers' Dr. Raymond Birge, “PS: a Global Immunosuppressive Signal...”. Spoke at Peregrine's 11-2015 “Sci. Session” at SITC'15/Maryland; was the chair in June'16 of the ICDS'16 session where Dr. Rolf Brekken (PPHM SAB) spoke; and now also speaking in late June'16 at ICHC'16/Istanbul on, “Phosphatidylserine is a Global Immunosuppressive Signal in Cell Death & Cancer”… (Rutgers Univ. http://birgelab.org “The Birge laboratory conducts basic science focused on the eradication of cancer.”)
-----
Jun19-22 2016: *Delayed till May'17 due to Unfortunate World Events* “ICHC'16 - From Molecules To Diseases”, Istanbul Turkey http://tinyurl.com/h4zjvo4 - ….Rutgers' Dr. Raymond Birge ( http://birgelab.org ) “Phosphatidylserine is a Global Immunosuppressive Signal in Cell Death & Cancer” - see http://tinyurl.com/j3whbx3
= = = = = = = = = = = = =RECALL:
June2-4 2016: “ICDS 2016: Translational Implications in Cell Death", Cork, Ireland
ICDS = Intl. Cell Death Society
http://www.celldeath-apoptosis.org/extensions/templates/next-meeting-html/tentative-program-cork.html
*Chair (1 of 9): Ray Birge** (USA)
6-3-16 Session: Cell Death & Cancer Therapy
Dr. Rolf Brekken (USA, UTSW, PPHM SAB): “Blockade of Phosphatidylserine & Immune Activation in Cancer”
–----
Also Recall… 10-29-15: Rutgers' Dr. Raymond Birge, John-Hopkins-Univ/Seminar: “PS is a Global-Immune-Checkpoint In Cancer” (12-1pm) http://tinyurl.com/oks5uo6
...Dr.Birge: PhD, Vice Chair, Professor, Dept of Molecular, Biochemistry & Molecular Genetics, Rutgers Univ. ( http://birgelab.org )
VIDEO(2:54): “Apoptosis – PS Exposed on Cell Surface”
No PM, don't really understand your question, but all this Ronin+SWInvest (John Stafford III+Stephen White) purchase activity has been reported via 13D's - no 13G's.
= = = = = = = = =
* 13D Group: “people that share the furtherance of a common objective/concerted action”.
* 13D’s are reserved for ACTIVE INVESTORS who may be “interested in agitating for some kind of a change at the company”.
4-17-17/13D: Ronin adds 23,334 PREFERRED(~194,449 COMMON); Group=>25,502,864sh./8.6%.
(8.6% stake based on Common shares equivalent for all Preferred shares, x 8.333)
I think I did this right – I took the 23,334 Preferred PURCHASED from Mar28-Apr10 from this latest 4-17-17 13D (required because Ronin’s Preferred stake crossed over 5% to 5.8%), and ADDED them to the totals from the next prior 3-10-17 13D (Mar2-9 purchases). Also, the 8.6% total stake that “group” Ronin+SWInvest (John Stafford III+Stephen White) now holds is based on 297,709,478 common O/S at 3-10-17. At 1-31-17 (10-Q), there were 1,647,760 Preferred O/S, but for the overall Stake% calc, best to convert Preferred to equiv. Common based on the 8.333 conversion factor. Let me know if any errors and I’ll correct.
= = = = = = = = = = = = = = =
3-2-17/13D: Ronin/17,565,843(Feb3-Mar1) + SWInvestMgt/3,494,166(Jan20-Feb16) = 21,060,009sh. http://tinyurl.com/jr42u23
3-10-17/13D: Ronin adds 3,248,405sh.(Mar2-9) http://tinyurl.com/huundwu
4-17-17/13D(PREFERRED): Ronin adds 23,334 (Mar28-Apr10), bringing total Preferred to 95,059, 5.8% of Preferred O/S. 23,334 x 8.333=194,449 Common equiv. http://tinyurl.com/lanjddc (13D filed Apr17)
…...On Mar28, 2017, Ronin Capital pur. 1,807 sh. Preferred @$21.60
…...On Apr6, 2017, Ronin Capital pur. 5,817 sh. Preferred @$22.16
…...On Apr7, 2017, Ronin Capital pur. 10,510 sh. Preferred @22.06
…...On Apr10, 2017, Ronin Capital pur. 5,200 sh. Preferred @$21.96
……...NOTE/13D: "Each share of Preferred Stock is convertible into a #shares of Common determined by dividing the liquidation preference of $25/sh. by the conv. price, currently $3.00/sh. (8.333). The Group for purposes of 13D also holds 5% or more of the Common Stock of Peregrine as reported in its separate 13D filed on March 2, 2017, amended March 8, 2017.”
NOTES:
* 13D Group: “people that share the furtherance of a common objective/concerted action”.
* 13D’s are reserved for ACTIVE INVESTORS who may be “interested in agitating for some kind of a change at the company”.
* See John Stafford III/XENCOR BOD(18yrs service): http://tinyurl.com/hcmsv8p
* Ronin Capital LLC: http://www.ronin-capital.com https://www.linkedin.com/company/ronin-capital
* Item3(13D 3-2-17) shows that SWIM Partners (Stephen White) also owns 2,533,333sh. pur. on or before 1-5-17. For some reason Item5 DOES NOT include these in The GROUP’s total stake, and there ARE NOT in the 25,502,864 group total above.
* Stake%’s based on 3-10-17 O/S=297,709,478sh. per latest 10-Q.
* For calc’ing total stake (Common+Preferred), using curr. conv. rate of 8.333 sh./Common per one share of Series E Convertible Preferred.
ALL SEC filings for PPHM: http://tinyurl.com/6d4jw8
Inst. Holdings (Nasdaq.com) - updated qtly, 45-days after each q/e cutoff: http://tinyurl.com/ys7fed
Tend to think Ignorance v. Intentional, but could_be_wrong.
Ronin’s 95,059 PREFERRED/1,647,760 PreferredO/S = 5.8% of PREFERRED O/S.
...When you look at TOTAL COMMON+PREFERRED, the Ronin+SwInvest Group (John Stafford III+Stephen White) now owns the equiv. of 25,502,864 of COMMON (incl. their Preferred x 8.333), or 8.6% of 297,709,478 common O/S at 3-10-17.
See: http://tinyurl.com/m9atpa6
AACR 2017 (WashDC) – All 5 PPHM Poster Images
5 PPHM ABSTRACTS: 2/MemSloan, 1/Immunovaccine, 1/PPHM-Only. There’s also a 5th one (PPHM, Vanderbilt, Precision-for-Medicine, Providence CC) in the “Ph2/3 Clinical Trials” session: “CT159/25: IFN-y Analysis in Blood & Tissue as a Potential Prognostic and/or Predictive Biomarker” – this is our new (3rd) Sunrise Biomaker analysis - see below.
...Add ImmunoVaccine Inc. (Halifax https://www.imvaccine.com ) to the list of collaborators.
...#1651(Apr3) is the newly revealed 2nd joint PPHM+Mem.Sloan/Wolchok preclin. study: “PS Targeting + Adoptive T Cell Transfer Eliminates Advanced Tumors without Off-Target Toxicities in a Melanoma Model”. IMO, the Wolchok Lab is saying in #1651 that the effectiveness of either type of Adoptive TCell Transfer (ACT), be it TCRs or CAR T (ie, the newer gene-modified T cells approaches), is improved by combining with Bavi and makes for a “highly desirable strategy.” - see more below.
...Memorial Sloan’s Dr. Jedd. Wolchok is co-author of 9 AACR’17 abstracts. 2 of the 9 are w/PPHM re: PS-Targeting. With biotechs, he has one each with Genocea Biosciences(ex-vivo screening), Infinity Pharm(Ph1), Leap Ther.(Ph1), and two with BMS(Ph1+Ph3). The 8th is MSKCC ONLY, and the 9th is jointly w/Cedars-Sinai/LA. http://www.abstractsonline.com/pp8/#!/4292 .
= = = = = = = = = = = = = = = = = = = = = = = = = =
AACR’17: http://www.aacr.org/Meetings/Pages/MeetingDetail.aspx?EventItemID=105
Abstracts: http://www.abstractsonline.com/pp8/#!/4292
SUMMARY of PPHM’s 5 AACR’17 ABSTRACTS: ...Details below.
1. MSKCC+PPHM: 4-2-17/1pm #574, “PSTargeting+RAD+AntiPD1 Promotes Anti-Tumor Activity, Melanoma” (same as SITC'16/11-14-16 http://tinyurl.com/js3fca4 )
2. MSKCC+PPHM: 4-3-17/8am #1651, “PSTargeting+Adoptive TCell Transfer (ACT) Eliminates Adv. Tumors w/o Off-Target Toxicities, Melanoma” <=NEW(2nd) Joint MemSloan/PPHM study SEE: http://tinyurl.com/lxlltd6
3. PPHM: 4-4-17/8am #3652, “PSTargeting+LAG3+AntiPD1 Significantly Enhances Anti-Tumor Activity, Triple- MBC”
4. IMMUNOVACCINE+PPHM: 4-4-17/8am #3657, ”PSTargeting Enhances Anti-Tumor Activity of a Tumor Vaccine(DepoVax), HPV-Induced Tumor” https://www.imvaccine.com
5. PPHM+VANDERBILT+PRECISIONMEDICINE+PROVIDENCECC: 4-3-17/1pm #CT159/25 (Session: Phase II/III Clinical Trials in Progress), “IFN-y Analysis In Blood & Tissue as a Potential Prognostic and/or Predictive Biomarker” (Sunrise Biomarker #3) SEE: http://tinyurl.com/ktzr782
ALSO: Peregrine Exhibiting – booth #3312.
AACR’17 DETAILS (5 PPHM ABSTRACTS)...
= = = = = = = = = = = = = = = = = = =
1. 4-2-17/1pm #574 - Session: CHECKPOINTS 1 [Joint PPHM & Memorial Sloan Kettering]
“Phosphatidylserine Targeting Antibody in Combination with Tumor Radiation & Immune Checkpoint Blockade Promotes Anti-Tumor Activity in Mouse B16 Melanoma”
=> Sadna Budhu 1, Olivier De Henau 1, Roberta Zappasodi 1, Rachel Giese 1, Luis F. Campesato 1, Christopher Barker 1, Bruce Freimark 2, Jeff Hutchins 2, Jedd D. Wolchok 1, Taha Merghoub [Memorial Sloan Kettering]
1=Memorial Sloan Kettering CC, NYC [ http://www.mskcc.org/research-areas/labs/jedd-wolchok ]
2=Peregrine Pharmaceuticals
NOTE: SAME TITLE AS WAS PRESENTED BY Mem.Sloan 11-14-16 SITC’16: see http://tinyurl.com/js3fca4
ABSTRACT:
Phosphatidylserine (PS) is a phospholipid that is exposed on surface of apoptotic cells, tumor cells and tumor endothelium. PS has been shown to promote immunosuppressive signals in the tumor microenvironment. Antibodies that target PS have been shown to reactivate anti-tumor immunity by polarizing tumor associated macrophages into a pro-inflammatory M1 phenotype, reducing the number of MDSCs in tumors and promoting the maturation of dendritic cells into functional APCs. In a mouse B16 melanoma model, targeting PS in combination with immune checkpoint blockade promoted greater anti-tumor activity than either agent alone. This combination was shown to enhance CD4+ and CD8+ T cell infiltration and activation in the tumors of treated animals. Radiation therapy (RT) is an effective focal treatment of primary solid tumors, but is less effective in treating metastatic solid tumors as a monotherapy. There is evidence that RT induces immunogenic tumor cell death and enhances tumor-specific T cell infiltration in treated tumors. The abscopal effect, a phenomenon in which tumor regression occurs outside the site of RT, has been observed in both preclinical and clinical trials when RT is combined with immunotherapy. In this study, we show that irradiation treatment of B16 melanoma causes an increase in PS expression on the surface of viable tumor and immune infiltrates. We subsequently examined the effects of combining RT with an antibody that targets PS (mch1N11) [“Mouse version of Bavituximab”] and immune checkpoint blockade (anti-PD-1) in B16 melanoma. We found that treatment with mch1N11 synergizes with RT to improve anti-tumor activity and overall survival in tumor bearing mice. In addition, the triple combination of mch1N11, RT and anti-PD-1 treatment displayed even greater anti-tumor and survival benefit. Analysis of local immune responses in the tumors of treated animals revealed an increase in tumor-associated macrophages with a shift towards a pro-inflammatory M1 phenotype after treatment with RT & mch1N11. In addition, analysis of the systemic immune responses in the spleen and tumor draining lymph nodes revealed an increase in CD8 T cell activation, effector cytokine production and differentiation into effector memory cells in the triple combination. This finding highlights the potential of combining these 3 agents to improve outcome in patients with advanced-stage melanoma and other cancers and may inform the design of clinical studies combining PS-targeting antibodies with RT and/or checkpoint blockade.
POSTER #574 IMAGE: http://www.peregrineinc.com/images/stories/pdfs/aacr2017budhu.pdf

= = = = = = = = = = = = = = = =
2. 4-3-17/8am #1651 - Session: TUMOR MICROENVIRONMENT & CHECKPOINTS [Joint PPHM & Memorial Sloan Kettering]
“Targeting Phosphatidylserine in Combination with Adoptive T Cell Transfer Eliminates Advanced Tumors without Off-Target Toxicities in a Melanoma Preclinical Model” <=NEW(2nd) MSK STUDY
=> Daniel Hirschhorn-Cymerman 1, Sara Sara Schad 1, Sadna Budhu 1, Zhong Hong 1, Xia Yang 1, Hutchins T. Jeff 2, Bruce D. Freimark 2, Michael J. Gray 2, Jedd Wolchok 1, Taha Merghoub [Memorial Sloan Kettering]
1=Memorial Sloan Kettering CC, NYC [ http://www.mskcc.org/research-areas/labs/jedd-wolchok ]
2=Peregrine Pharmaceuticals
---------------------
FROM PPHM’s 4-3-17 PR: http://ir.peregrineinc.com/releasedetail.cfm?ReleaseID=1019762
…”Latest Findings from Ongoing Collaboration with Memorial Sloan Kettering (MSK) Support Potential Applications for Combining CAR T & Anti-PS in Treatment of Solid Tumors”… For this study, a team of MSK researchers led by cancer immunotherapy thought-leaders, Taha Merghoub, Ph.D. and Jedd D. Wolchok, M.D., Ph.D., evaluated and compared the anti-tumor activity and off-target toxicities of adoptive T cell transfer therapy in combination with either PS-targeting antibodies or anti-OX40 antibodies in mice with advanced melanomas. Whereas PS-targeting and anti-OX40 demonstrated comparable tumor regression when administered in combination with transferred adoptive T cells, only the PS-targeting combination achieved these results without any off-target toxicities. By contrast, the anti-OX40 treatment combination triggered off-target inflammatory destruction of healthy tissues. Additional study results demonstrated that the PS-targeting antibodies decreased tumor-induced immunosuppression as evidenced by a decrease in immunosuppressive regulatory T cells (Tregs) and M2 macrophages. This finding is consistent with Peregrine’s belief that bavituximab may modulate the immunosuppressive tumor microenvironment and enhance the activity of immunotherapy agents.
Taha Merghoub, Ph.D., Co-Dir. of the Ludwig Collaborative Laboratory at MSK:
“While adoptive T cell transfer remains one of the most exciting new approaches to treating cancer, to date the toxicity associated with the treatment has limited its potential. We are encouraged that these study results showed that the combination of anti-PS and adoptive T cell treatment led to enhanced anti-tumor effect without any evidence of additional off-target side effects. “We believe that these findings may support potential applications for this combination in solid tumors in the future.”
Joseph Shan, Peregrine’s VP of Clinical & Regulatory Affairs:
“These study results provide further support for our belief that anti-PS agents such as bavituximab can play an important role as part of combination cancer treatments. This is directly tied to the agents’ ability to modulate the tumor microenvironment to combat the immunosuppression that limits the activity of CAR T and immunotherapies. Importantly, we are now also seeing evidence that this targeted modulation of the tumor microenvironment by anti-PS allows for enhanced activity of these other treatments without triggering any off-target toxicities. This is opposed to other conventional immunotherapies such as anti-OX40 with systemic mechanisms of action. We believe this advantageous tolerability profile will be a key benefit in positioning anti-PS agents for inclusion in optimal combination cancer regimens.”
-----------
ABSTRACT #1651:
A viable strategy to treat advanced cancers includes transferring of tumor-specific T cells. T cells that recognize tumor antigens can be expanded and reinvigorated ex-vivo. Furthermore, autologous T cells can be genetically modified to express anti-tumor T cell receptors [TCRs] or chimeric antigen receptors [CARs]. Although the potency and specificity of tumor-specific T cells can be manipulated ex-vivo, once re-infused into patients, the T cells are subjected to immunosuppressive mechanisms established by the tumor. An important immune checkpoint regulator within tumors is phosphatidylserine (PS). Innate immune cells exposed to PS secrete suppressive cytokines and chemokines that can significantly impair the function and activation of anti-tumor T cells. Therefore, monoclonal antibodies that block PS activity can increase the anti-tumor potency of transferred T cells to treat aggressive cancers. Here we show that a PS targeting monoclonal antibody in combination with CD4+ T cells that recognize the melanoma antigen Trp1 can regress very advanced melanomas in all treated mice. Combination of anti-Trp1 CD4+ T cells with other immunomodulatory modalities such as anti-OX40 antibodies, can achieve equivalent treatment rates but these are typically accompanied by severe immune related adverse events. In contrast, in this setting, PS blockade did not show any off-target toxicities. Flow cytometry analysis revealed lower levels of CD206 expression concomitant with higher activation markers in macrophages and neutrophils in tumors from anti-PS treated mice. These results suggest that diminishing suppressive mechanisms locally in adoptive transfer protocols is a highly desirable strategy that can eliminate tumors while minimizing related adverse events.
POSTER #1651 IMAGE: http://www.peregrineinc.com/images/stories/pdfs/aacr2017hirschhorn.pdf

---------------------
8-2015: Presently there are 3 types of Adoptive Cell Transfer (ACT) using effector T cells that are advancing on a path towards regulatory approval:
1. TILs (tumor infiltrating lymphocytes) have been developed with slow but continuing progress over several decades.
2. CARs (chimeric antibody receptors) – newer gene-modified T cells strategy
3. TCRs (T-cell receptors) – newer gene-modified T cells strategy
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4507286/

...“Adoptive T cell transfer is like ‘giving patients a living drug’,” states Renier J. Brentjens, MD/PhD, of Memorial Sloan Kettering Cancer Center (MSKCC).
… https://www.cancer.gov/about-cancer/treatment/research/car-t-cells
...“Adoptive T cell therapy (ACT) is one stone in this new pillar, a potentially powerful approach to cancer treatment that relies on the infusion of tumor-specific T cells.”
… https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4327320
MSK Tweet 4-3-17 https://twitter.com/sloan_kettering :
...See bottom right: “SWIM” - Swim Across America http://www.swimacrossamerica.org

= = = = = = = = =
Interesting 3-31-17 Sloan Kettering Tweet re: AACR’17
3-31-17 9:20amET “Getting ready for #AACR17? #Immunotherapy will be a hot topic. Learn more about how it works (video). @AACR”
https://twitter.com/sloan_kettering
Points to this 4-2016 Video (1:20):
“Immunotherapy – How It Works” https://www.youtube.com/watch?v=COQ1AeoGyco
Beg@:39:
“Immunotherapy drugs release this brake and empower immune cells to fight the cancer.
Sometimes the immune system needs a tune-up before it can fight cancer.
Immune cells can be removed from the body, armed with new proteins that can target cancer cells, and given back to a patient in large numbers.
Once inside the body, the modified immune cells recognize and attack the cancer.
This approach is called CAR T cell therapy. [an Adoptive Cell Transfer “ACT” strategy]
Despite the promise of immunotherapy, not everybody responds.
MSKCC scientists are exploring ways to improve immunotherapy.”

=============AND, THIS 3-23-17 PARKER FOUNDATION RELEASE ON AACR’17:
3-23-17: “Parker Institute for Cancer Immunotherapy Scientists to Present Research on Checkpoint Inhibitors, Adoptive Cell Therapy, and Other Advances in Immuno-oncology at AACR 2017”
Investigators affiliated with the Parker Institute for Cancer Immunotherapy will present some of the most anticipated immuno-oncology research at the 2017 AACR Annual Meeting. The event takes place at the Walter E. Washington Convention Center in Washington, DC, April 1-5 2017. . .
Other adoptive cell therapy abstracts of interest:
• ”Targeting Phosphatidylserine in Combination with Adoptive T Cell Transfer Eliminates Advanced Tumors without Off-Target Toxicities in a Melanoma Preclinical Model” [see http://tinyurl.com/nx5q5os ]
The principal investigator is Taha Merghoub, PhD, Parker Institute member researcher at Memorial Sloan Kettering Cancer Center. Co-authors include Parker Institute Center Director Jedd Wolchok, MD, PhD, at Memorial Sloan Kettering Cancer Center.
http://www.parkerici.org/media/2017/parker-institute-for-cancer-immunotherapy-scientists-to-present-research-on
----------ACT NOTES:
NOTE1:
From 3-2014 Immunotherapy article: “Cancer immunotherapy, particularly adoptive cell transfer (ACT), has shown great promise in the treatment of patients with late-stage disease, including those who are refractory to std. Therapies” https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4372895
NOTE2:
The Parker Foundation: “The Immunotherapy Dream Team”
Through the support from SEAN PARKER, Stand Up To Cancer (SU2C) and the Cancer Research Institute (CRI) formed an Immunology Research Dream Team dedicated to cancer immunology: “Immunologic Checkpoint Blockade & Adoptive Cell Transfer in Cancer Therapy.” The Immunotherapy Dream Team is focused on 2 approaches for this translational cancer research project, which will unite laboratory & clinical efforts towards the immunological treatment, control, and prevention of cancer.
The 1st is investigating blockades (by inhibitory molecules called “checkpoints”) of T lymphocytes’ inhibitory receptors, which block immune responses; and the use of antibodies to remove the checkpoints, once again allowing white blood cells called T lymphocytes to kill the cancer cells.
Second, the Immunotherapy Dream Team is pursuing multiple Adoptive Cell Transfer (ACT) approaches, which increase immunity.”
DREAM TEAM LEADERSHIP: 10 scientists, incl. MSKCC’s Jedd D. Wolchok & Michel Sadelain.
http://parker.org/initiatives/immunotherapy
= = = = = = = = = = = = = = = =
3. 4-4-17/8am #3652 - Session: BITES BISPECIFICS & CHECKPOINTS
“Combinational Activity of LAG3 & PD-1 Targeted Therapies is Significantly Enhanced by the Addition of Phosphatidylserine Targeting Antibodies and Establishes an Anti-Tumor Memory Response in Murine Triple Negative Breast Cancer”
=> Michael J. Gray, Jian Gong, Jeff Hutchins, Bruce Freimark (Peregrine Pharmaceuticals)
ABSTRACT:
Previous studies utilizing NanoString immune profile analysis demonstrated that intratumoral levels of LAG3 (lymphocyte activation gene 3) mRNA increased in response to phosphatidylserine (PS) and PD-1 targeting antibodies in murine triple negative breast cancers (TNBC). This suggests LAG3 acts to attenuate immune system activation during I/O therapies - and that PD-1 and LAG3 function cooperatively in suppressing immune system activation. Here we show that adding PS targeting antibodies can further enhance the effectiveness of antibodies targeting LAG3 and/or LAG3+PD-1. We first examined expression of LAG3 and PD-1 in the murine TNBC model E0771 and found that tumor associated T-cells (CD4+ and CD8+) have expression of both markers. Mice implanted with TNBC tumors were next treated with antibodies targeting PS, PD-1, and LAG3 alone and in combination with each other. Interestingly, the addition of PS targeting antibodies not only increased the effectiveness anti-PD-1 effectiveness as previously observed, but also enhanced anti-LAG3 treatment, showing that PS targeting antibodies are capable of augmenting additional I/O therapeutic regimens. Comparison of anti-PD-1+LAG3 combination vs. single anti-PD-1 or anti-LAG3 treatments showed moderately more anti-tumor activity than single treatments, however the addition of PS targeting antibodies to either checkpoint inhibitor was as equally effective in inhibiting tumor growth as observed in the anti-LAG3+PD-1 treatment. Further comparison of antibody treatments targeting PD-1+LAG3 vs. PS+PD-1+LAG3 demonstrated that the addition of PS targeting antibodies resulted in a significant decrease in tumor growth with complete tumor regression in 80% of the animals (along with the ability to completely reject secondary TNBC challenge) compared to 0% in the anti-PD-1+LAG3 treatment group. Immunoprofiling showed that the addition of PS targeting antibodies to these checkpoint therapies, including the combination of anti-PD-1+LAG3, resulted in a phenotype associated with enhanced immune system activation and immune-surveillance including increased tumor infiltrating lymphocytes (TILs) with upregulation of T-cell associated activation pathways, increased Th1 to Th2 profile, and enhanced antigen presentation processing /presentation mechanisms along with cytokines associated with immune system activation. Overall our data demonstrate that adding PS targeting antibodies to clinically relevant therapies, including PD-1 and LAG3, may significantly enhance their ability to activate and redirect the host immune system into recognition and elimination of tumor cells compared to single and combinational treatments that lack PS targeting antibodies.
POSTER #3652 IMAGE: http://www.peregrineinc.com/images/stories/pdfs/aacr2017gray.pdf
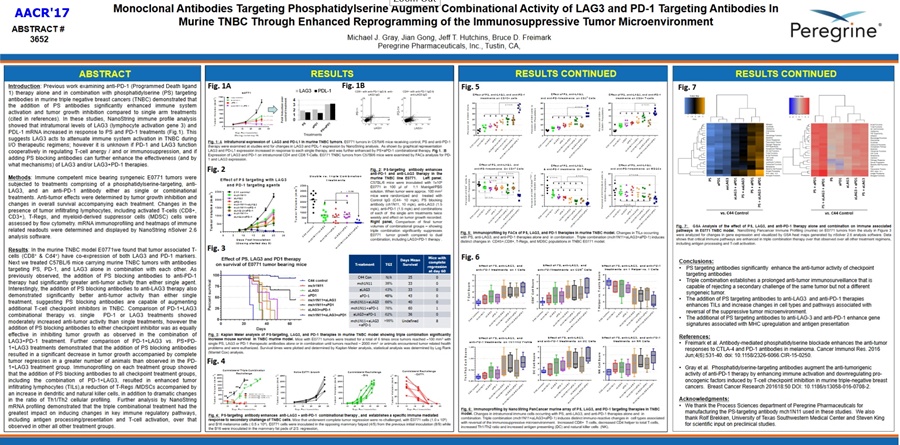
= = = = = = = = = = = = = = = =
4. 4-4-17/8am #3657 - Session: BITES BISPECIFICS & CHECKPOINTS
”Phosphatidylserine-Targeting Antibodies Enhance Anti-Tumor Activity of a Tumor Vaccine in a HPV-Induced Tumor Model”
=> Genevieve Weir 1, Tara Quinton 1, Jeff T. Hutchins 2, Bruce D. Freimark 2, Marianne Stanford (VP/Res., Immunovaccine)
1=Immunovaccine, Inc., Halifax, NS, Canada [ https://www.imvaccine.com ]
2=Peregrine Pharmaceuticals
[Note: clearly, this study is combining PPHM’s Anti-PS with ImmunoVaccine’s DepoVax Vaccine Adjuvanting Platform https://www.imvaccine.com/depovax.php ]
ABSTRACT:
Antibodies targeting phosphatidylserine (PS) have been shown to induce anti-tumor responses by induction of tumor-specific T cells. Based on this observation, we evaluated the responses of PS and PD-1 targeting antibody therapy to enhance anti-tumor responses of a HPV16 peptide vaccine formulated in DepoVax (DPX) in mice bearing HPV-transformed C3 mouse tumors. The addition of PS-targeting antibody (mch1N11) [“Mouse version of Bavituximab”] to DPX/metronomic cyclophosphamide (mCPA) immunotherapy prolonged survival in comparison to mice receiving an isotype control in combination with DPX/mCPA. When anti-PD-1 was added to mch1N11 + mCPA, there was no increase in survival. The addition of mch1N11 to DPX/mCPA immunotherapy had no effect on tumor growth or survival in the aggressive B16-F10 model. TIL analysis revealed an increase in CD8+ T cells, antigen specific CD8+ T cells and PD-1+ T cells in the tumor with mch1N11 treatment. The expression of surface markers for macrophages (CD68high, F4/80) and dendritic cells (CD11c) were also increased in the tumors of mice treated with mch1N11. RT-qPCR analysis of the tumor confirmed higher mRNA expression of T cells markers (CD8, Granzyme B, PD-1) and antigen presenting cell markers (F4/80, CD74). In the spleen, expression of cell surface markers for monocytes (CD11b) and PD-1+ T cells (CD8) were elevated in groups treated with mch1N11 in combination with anti-PD-1. Combined, these findings indicate that in this model, PS-targeting antibodies can enhance the activity of phagocytic cells involved in antigen presentation. We have found that PD-1 expression increases as anti-tumor activity increases, therefore these results also provide an indication that antibodies targeting PS enhance the anti-tumor immune response induced by DPX/mCPA therapy. The observations suggest that PS-targeting antibodies may enhance therapeutic vaccines for the treatment of cancer.
POSTER #3657 IMAGE: (from ImmunoVaccine.com)
https://www.imvaccine.com/userfiles/IMMUNOVACCINE%20AACR%20Poster%202017MAR14%20GW(2).pdf

-------
4-5-17 Immunovaccine PR: “...Immunovaccine Presents Preclinical Research at AACR’17... Part of Ongoing Effort to Identify Novel Combinations of DepoVax-based Immuno-oncology Candidates to Improve the Responses of Other Novel Immunotherapy Agents… preclin. data presented demonstrated that phosphatidylserine (PS) targeting antibodies can enhance the anti-cancer activity of its DepoVax-based therapeutic vaccine platform. In the study, researchers combined a Peregrine Pharmaceuticals’ PS-targeting antibody compound (mch1N11) with a DepoVax-based HPV16 peptide vaccine & metronomic cyclophosphamide (mCPA). This combined immunotherapy prolonged survival in C3 mouse models as compared to mice receiving an isotope control in combination with DepoVax/mCPA. Addl. analysis also demonstrated an increase in T cells in the tumor following treatment. Taken together, researchers believe that the data suggests that the antibody targeting PS can increase the anti-tumor immune response induced by a DepoVax-based cancer immunotherapy. Marianne Stanford, PhD, VP/Res., ”This study is another step in our exploration of combining DepoVax-based cancer vaccines & other promising immuno-modulatory compounds. Our process of generating supportive preclin. data to guide a potential clinical path forward has been effective in identifying these novel combinations in the past. We now look forward to continued work with our partners to advance combination candidates into and through the clinic, with the goal of expanding treatment options for hard-to-treat cancers.” The study was designed to analyze the potential synergistic effects of combining DPX-based immunotherapies with bavituximab, Peregrine’s investigational chimeric monoclonal antibody that targets PS...” https://www.imvaccine.com/releases.php?releases_id=421
-----
...Ahhhhh, ImmunoVaccine & Dr. Wolchok – zero doubt IMO how & why PPHM & Immunovaccine have hooked up...
ImmunoVaccine & Jedd Wolchok go back to ASCO’13:
6-3-13: “Our poster (Ph1 data) is one of few selected for detailed discussion at ASCO by Dr. Jedd Wolchok, top cancer immunotherapy thought leader… Immunovaccine Inc. believes that these immune responses are consistent in profile to those necessary from a cancer vaccine to potentially impact disease progression. These study results were further discussed by Dr. Jedd Wolchok of Memorial Sloan-Kettering CC, a top thought leader in the area of cancer immunotherapy, at the poster discussion session that followed.”
https://www.imvaccine.com/communications.php?communications_id=11
= = = = = =THIS A NEW (3RD) KNOWN SUNRISE BIOMAKER ANALYSIS:
#5. 4-3-17/1pm #CT159/25 - Session: Phase III Clinical Trials & Phase II/III Clinical Trials in Progress
http://www.abstractsonline.com/pp8/#!/4292/presentation/12566
”IFN-y Analysis In Blood & Tissue as a Potential Prognostic and/or Predictive Biomarker”
=> Nikoletta Kallinteris 1, Leora Horn 2, Min Tang 1, Tobias Guennel 3, Shen Yin 1, Jennifer Lai 1, Joseph Shan 1, Rachel E. Sanborn [Providence CC, Dir./Thoracic-Oncology http://cancergrace.org/faculty/rachel-sanborn-md - Dr. Sanborn's Conflicts of interest: DNA, AZN]
1=Peregrine Pharmaceuticals
2=Vanderbilt-Ingram Cancer Center, Nashville, TN
3=Precision for Medicine, Frederick, MD
4=Providence Cancer Center, Portland, OR
---------------------
FROM PPHM’s 4-4-17 PR: http://ir.peregrineinc.com/releasedetail.cfm?ReleaseID=1020046
…”SUNRISE Data Analysis Demonstrates Stat. Significant Overall Survival (OS) Improvement in Patients Receiving Bavituximab+Docetaxel and Subsequent Immunotherapy Compared to Placebo+Docetaxel and Subsequent Immunotherapy”… AACR’17 presentation (#CT159/25) of results of a new analysis of the Phase III SUNRISE trial… Data demonstrated that for patients in the study's BAVI+DOCE treatment arm who received subsequent immunotherapy, the mOS was not reached, while mOS was 13.0mos. for patients in the study's DOCE+PLACEBO arm who received subsequent immunotherapy [HR=0.43; p=.005]. These are the first clinical results reported supporting the hypothesis that bavituximab may modulate the tumor microenvironment to enhance the anti-tumor activity of immunotherapy agents... The presentation highlighted an analysis in which the company evaluated the impact of subsequent immunotherapy treatment, as well as patients' pre-treatment interferon gamma (IFN-y) levels on overall survival. Overall, low peripheral IFN-y correlated with more favorable OS in the patients receiving BAVI+DOCE and is a biomarker of interest. Data were also analyzed by low vs. high IFN-y levels. For patients with low pre-treatment IFN-y levels who received subsequent immunotherapy, those in the BAVI+DOCE arm did not reach mOS compared to mOS of 12.1mos. for the DOCE+PLACEBO arm [HR=0.24; p<.001].
Joseph Shan, Peregrine’s VP of Clinical & Regulatory Affairs:
"We are extremely encouraged by the results of these exploratory analyses which provide further clinical rationale for combining bavituximab and checkpoint inhibitors. This will be the key focus for upcoming early phase clinical trials, which includes a study of bavituximab and pembrolizumab in head and neck cancer through our ongoing collaboration with the NCCN.”
...Bavituximab is an investigational chimeric monoclonal antibody that targets phosphatidylserine (PS). Signals from PS inhibit the ability of immune cells to recognize and fight tumors. PS-targeting antibodies have demonstrated an ability to shift the functions of immune cells in tumors, resulting in multiple signs of immune activation and anti-tumor immune responses. Bavituximab is believed to override PS immunosuppressive signaling by blocking the engagement of PS with its receptors and sending an alternate immune activating signal.
----------
ABSTRACT #159/25:
BACKGROUND: SUNRISE, a global, double-bind, Phase III trial of docetaxel (D) plus bavituximab (B) or D plus placebo (P) in previously treated non-squamous non-small cell lung cancer (NSCLC), demonstrated similar overall survival (OS) in both treatment arms. Immune correlate analyses including pre-treatment IFN-y levels in blood and tumor tissue were used to potentially identify prognostic and/or predictive correlation with clinical outcome.
METHODS: Serum was isolated from all randomized NSCLC patients at screening, periodically during treatment and at disease progression for evaluation of IFN-y levels using the Simoa TM assay (Myriad RBM, Austin, TX). Available archival tissue was also tested for 91- immune gene activation markers, including IFN-y by the Fluidigm-based gene-expression platform (Sirona Dx, Lake Oswego, OR). Kaplan-Meier statistical methods and Cox proportion hazards models were utilized to evaluate and contrast the correlation of peripheral and intratumoral IFN-y levels with OS. Patients were classified paradoxically as IFN-y "low" with a favorable disease prognosis vs. "high" associated with more aggressive disease based on the median.
RESULTS: Pretreatment serum results were available for 582 out of the 597 randomized patients. Each patient was classified to be pre-treatment IFN-y high or low (< cut-off) using cut-off 0.093 pg/ml, which is the median IFN-y value in the D+B group. Median overall survival (mOS) in all patients with IFN-y low is 11.3mos. (95% CI, 10.1-13.5) vs. 10.4mos. (95% CI, 8.4-11.3) in all IFN-y high; p=0.047. mOS of D+B arm is 11.6mos. (95% CI, 10.2-13.9) and 11.1mos. (95% CI, 9.1-14.7) in the D group; p=0.982 for IFN-y low. mOS of D+B arm is 9.0mos. (95% CI, 6.7-11.2) and 10.6mos. (95% CI, 8.9-13.0) in the D group; p=0.252 for IFN-y high. With the limited intratumoral IFN-y gene expression data (n=33), no statistically significant correlation with OS was observed.
CONCLUSIONS: Correlative approaches identified low peripheral low IFN-y at pretreatment as a biomarker of interest correlating with more favorable clinical outcomes and is consistent with the hypothesis that bavituximab may demonstrate more immunomodulatory effects in patients with “immune cold” tumors.
POSTER #159/25 IMAGE:
http://www.peregrineinc.com/images/stories/pdfs/aacr2017kallinteris.pdf
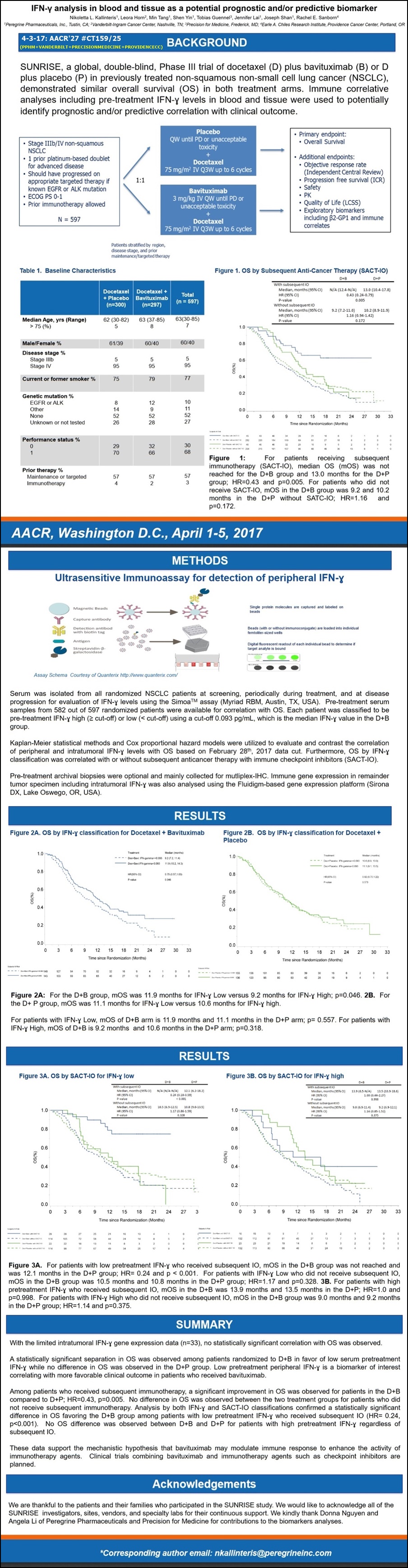
---------
**NOTE: Per JDM find, we know that 96 Sunrise Pts received “Subsequent Immunotherapy” - see 10-13-16 ASM SKing Slide#14: http://tinyurl.com/n2bajew
-------
This AACR’17 #CT159/25 IFN-y is certainly the 3rd Sunrise Biomarker.
Known Ph.3 Sunrise Biomarkers/UTSW’s Dr. David Gerber et al:
#1=B2GPI: 10-10-16 http://tinyurl.com/hp73njt
#2=Complement & IL-10 Pathways(12-7-16/IASLC delayed, “not done”, after prelim. abstract said, “Pts=193, within the 1st subgroup of N=104, a 2nd subgroup isolated: MOS 5.9=>12.5mos”
#3=IFN-y (this 4-3-17 AACR’17 one, #CT159/25)
- - - - - - -
***Maybe Dr. Ranee Mehra (Johns Hopkins, PPHM’s NCCN Bavi+Keytruda Head&Neck Ph2 P.I.) will stop by...
...Dr. Mehra’s work with Biomarker IFN-y seems to dovetail into PPHM’s newly revealed Sunrise Biomarker #3 to be presented 4-3-17 at AACR’17: #CT159/25, “IFN-y Analysis In Blood & Tissue as a Potential Prognostic and/or Predictive Biomarker” ( http://tinyurl.com/zaz525l ).

Note: DR. RANEE MEHRA was co-author of ASCO’16, “Biomarkers & Response to Pembro(Keytruda) in Recurrent/Metastatic Head & Neck Cancer” - Conclusion: “The IFN-y signature score was significantly associated with ORR, PFS, and OS (all, P< .001)… PD-L2 & IFN-y signature may be associated with clinical response to Pembro[Keytruda] and may offer addl. strategies to improve prediction of response.” http://meetinglibrary.asco.org/content/165708-176
MORE: http://tinyurl.com/h8gzkww


= = = = = = = = =
INTERESTING: Mem. Sloan’s Dr. Jedd. Wolchok is co-author of 9 AACR’17 abstracts. 2 of the 9 are w/PPHM re: PS-Targeting. With biotechs, he has one each with Genocea Biosciences(ex-vivo screening), Infinity Pharm(Ph1), Leap Ther.(Ph1), and two with BMS(Ph1+Ph3). The 8th is MSKCC ONLY, and the 9th is jointly w/Cedars-Sinai/LA. http://www.abstractsonline.com/pp8/#!/4292 .
PEREGRINE (Joint Mem.Sloan Kettering Wolchok Lab & PPHM):
1. 4-2-17 #574 “Phosphatidylserine Targeting Antibody in Combination with Tumor Radiation & Immune Checkpoint Blockade Promotes Anti-Tumor Activity in Mouse B16 Melanoma” <=Same as SITC’16 11-14-16.
4-3-17 #1651 “Targeting Phosphatidylserine in Combination With Adoptive T Cell Transfer Eliminates Advanced Tumors Without Off-Target Toxicities in a Melanoma Preclin. Model” <=NEW/2nd PPHM+MSK.
NON-PEREGRINE:
3. (MSK ONLY): 4-2-17 #874 “Lifting the iron curtain: Imaging cellular barriers to combination chelation-immune checkpoint therapy”
4. (MSK & Genocea Bio. http://www.genocea.com GNCA/$130mm MktCap) 4-2-17 #632 “Genome-scale neoantigen screening using ATLAS prioritizes candidate antigens for immunotherapy in a NSCLC patient”
5. (MSK & Cedars-Sinai/LA) 4-4-17 #4705 “CTLA4 blockade with HER2-directed therapy yields clinical benefit in women undergoing radiation therapy for HER2+ breast cancer brain metastases”
6. (MSK & Infinity Pharm.) 4-4-17 #CT089 “A Ph1, first-in-human study of IPI-549, a PI3K-y inhibitor, as monotherapy & in combo w/nivolumab in adv. solid tumors”
7. (MSK & Leap Ther.) 4-2-17 #CT018 “Intratumor & peripheral Treg modulation as a pharmacodynamic biomarker of the GITR agonist antibody TRX-518 in the 1st in-human trial”
8. (MSK & BMS) 4-3-17 #CT073 “(Ph1) Immunomodulatory effects of NIVO+IPI or NIVO/mono in adv. melanoma: CheckMate038”
9. (MSK & BMS) 4-3-17 #CT075 “OS results from a Ph3 of NIVO+IPI in treatment-naïve pts w/adv. Melanoma: CheckMate067”

= = = = = = = = = = = = =
2-28-17: Collabs with Mem.Sloan(Wolchok), Duke, MDA, Rutgers, ImmunoVaccine, UTSW… http://tinyurl.com/heg9t3v
BAVI MOA 11-14-16: SITC’16: Joint Memorial Sloan Kettering (Wolchok Lab) & PPHM poster on Triple Combo Rad+Bavi+aPD1 vs. Melanoma http://tinyurl.com/js3fca4
“PS Targeting Antibody in Combination with Checkpoint Blockade & Tumor Radiation Therapy Promotes Anti-Cancer Activity in Mouse Melanoma” (same as AACR’17 4-2-17 #574)
DR. JEDD WOLCHOK: "Based on these study results, we believe that the targeting of PS is having meaningful activity within the tumor microenvironment in the B16 melanoma model. It appears that this activity creates a more immune active environment in which other treatments, including radiation, are able to have a greater anti-tumor impact."
DR. TAHA MERGHOUB (Co-Dir., Ludwig Collaborative Lab at MSK): "We have noted that the combination of PS-targeting treatment and radiation, as well as triple combination of PS-targeting treatment, radiation and anti-PD-1, resulted in clear advantages in anti-tumor activity in the mouse B16 melanoma model. We believe that these findings suggest the potential benefit of combining these agents to improve the outcomes of patients with cancer. With this in mind, we think this research may play an important role in designing future clinical trials of PS-targeting agents in melanoma and other cancers."
5-29-15: Peregrine & Sloan Kettering Enter Collab. to “Investigate Novel PS-Targeting Immunotherapy Combos” http://tinyurl.com/o3k9ux8
...Dr. Wolchock states, ”The phosphatidylserine (PS) signaling pathway is a very interesting target for modulating the immune system's response to cancer. We look forward to exploring the potential of PS-targeting agents alone and with other immune modulators that may lead to novel advances in cancer therapy.”
...Dr. Taha Merghoub states, "A key focus of the Wolchok Lab's research is studying novel immunotherapy combinations that work together to enable the immune system to recognize & destroy cancer. This collaboration will allow us to focus on the role & contribution of PS blockade therapy in determining which combination of the current & next gen. of immune modulators is likely to increase the extent & amplitude of anti-tumor response. This important pre-clinical and translational work will potentially guide the design of the next gen. of clinical studies with bavituximab.”
POSSIBLE GENESIS OF IMMUNOVACCINE COLLAB???
11-9-15 SITC'15: New Bavi+Checkpoint Inhibitors preclin. data (UTSW/DUKE's Herbert K. Lyerly) http://tinyurl.com/pbof95w
...Also, collab. with Dr. Bernard Fox (Immunotherapist/Earle A. Chiles Res.Inst.) on new Immuno-Profiling Clinical Test (Opal 6-plex quantitative IF Assay), PPHM roundtable with Raymond Birge (Rutgers), Douglas Graham (Emory), Dmitry Gabrilovich (Wistar), Rolf Brekken (UTSW), Maria Karasarides (AstraZeneca) - ”Combining Bavi w/anti-PD-1 significantly enhanced O/S… significant incr. CD45+, CD8+ and CD3+ T-cells… led a prolonged anti-tumor immune response which protected the animals against a re-challenge w/same tumor.”
SITC’16 PPHM/Mem.Sloan Poster – same as AACR’17 4-2-17 #574…

4-11-17/J.Hutchins speaking at Immune Profiling World-Congress'17, WashDC http://tinyurl.com/zlr5cyj
Feb2017: Chgd. To HEAT BIO!!!!!!!!!!!
xxxApr10-12 2017: “Terrapinn’s Immune Profiling World Congress 2017”, WashDC
Part of “World Vaccine Congress Washington 2017” (1 of 8 Co-Conf’s)
“The Immune Profiling World Congress USA brings together experts to discuss profiling of the immune system, how using high throughput technologies can aid therapeutic design & treatment strategies in infectious & non-infectious diseases, including cancer.”
http://www.terrapinn.com/conference/world-vaccine-congress-washington/index.stm
http://www.terrapinn.com/conference/immune-profiling-usa/
4-11-17/Day1 12:10pm: Jeff Hutchins (VP/PreClinRes., PPHM)
“Increasing the Benefits of Immune Checkpoint Therapies Through Increased Tumor Infiltrating Activated T-Cells”
*Expanding the responding population
*Sharing pre-clinical & translation data
CHGD to: 12:10 “How Immuno-Oncology Combinations Utilizing A Multi-Antigen Vaccine Could Enhance Immune Checkpoint Therapy Benefits”
*Novel therapeutic vaccines drive antigen specific tumor-infiltrating lymphocytes to unlock clinical benefits in patients with poor checkpoint therapy prognosis
*Immune profiling methods on patient derived samples validate the mechanism of action for the combination approach
*Two platforms, 3 products focused on synergistic combination therapies in oncology
Dr Jeff Hutchins, CSO, VP/PreclinDEV, Heat Biologics, Inc.
- - - - - -
Chair: Dr Alison Deckhut Augustine, Branch Chief Basic Immunology, NIAID/NIH
DAY1 SPEAKERS 4-11-17(10):
...Bali Pulendran – Emory
...Dr Masahide Yano - FDA
...Dr Jeff Hutchins – Peregrine
...Dr Ali M. Harandi – Univ. of Gothenburg
...Dr Mahesh Yadav - Genentech
...Giuseppe Del Giudice - GSK Vaccines
...Mark M Davis - Stanford Univ.
...Alessandro Sette - La Jolla Infectious Disease Inst.
...Peter W. Che - Repertoire Genesis, Inc
...Dr Wayne C Koff - Human Vaccines Project, Intl. Aids Vaccine Initative
DAY2 SPEAKERS 4-12-17(4):
...Dr Roy Baynes (PD-1 Antibodies) – Merck
...Mr Bruno Gomes - iTeos Therapeutics
...Mr Lei Zheng - Johns Hopkins
...Dr Rakesh Dixit - MedImmune

Oops, missed the Immunovaccine AACR'17 #3657 Poster Image...
PPHM/MSKCC(Jedd Wolchok Lab) Collab: #1/Bavi+PD1+Rad, #2/Bavi+”ACT”
CHRONOLOGICAL HISTORY (May2015 – Apr2017):
APRIL 2017 AACR’17: Two Joint PPHM+MSKCC Posters Presented...
1. MSKCC+PPHM: 4-2-17/1pm #574, “PSTargeting+RAD+AntiPD1 Promotes Anti-Tumor Activity, Melanoma” (same as SITC'16/11-14-16 http://tinyurl.com/js3fca4 )
2. MSKCC+PPHM: 4-3-17/8am #1651, “PSTargeting+Adoptive TCell Transfer (ACT) Eliminates Adv. Tumors w/o Off-Target Toxicities, Melanoma”
AACR’17 DETAILS:
= = = = = = = = = = = = = = = = = = =
AACR’17 1. 4-2-17 #574 - Session: CHECKPOINTS 1 [Joint PPHM & Memorial Sloan Kettering]
“Phosphatidylserine Targeting Antibody in Combination with Tumor Radiation & Immune Checkpoint Blockade Promotes Anti-Tumor Activity in Mouse B16 Melanoma”
=> Sadna Budhu 1, Olivier De Henau 1, Roberta Zappasodi 1, Rachel Giese 1, Luis F. Campesato 1, Christopher Barker 1, Bruce Freimark 2, Jeff Hutchins 2, Jedd D. Wolchok 1, Taha Merghoub [Memorial Sloan Kettering]
1=Memorial Sloan Kettering CC, NYC [ http://www.mskcc.org/research-areas/labs/jedd-wolchok ]
2=Peregrine Pharmaceuticals
NOTE: SAME TITLE AS WAS PRESENTED BY Mem.Sloan 11-14-16 SITC’16: see http://tinyurl.com/js3fca4
ABSTRACT:
Phosphatidylserine (PS) is a phospholipid that is exposed on surface of apoptotic cells, tumor cells and tumor endothelium. PS has been shown to promote immunosuppressive signals in the tumor microenvironment. Antibodies that target PS have been shown to reactivate anti-tumor immunity by polarizing tumor associated macrophages into a pro-inflammatory M1 phenotype, reducing the number of MDSCs in tumors and promoting the maturation of dendritic cells into functional APCs. In a mouse B16 melanoma model, targeting PS in combination with immune checkpoint blockade promoted greater anti-tumor activity than either agent alone. This combination was shown to enhance CD4+ and CD8+ T cell infiltration and activation in the tumors of treated animals. Radiation therapy (RT) is an effective focal treatment of primary solid tumors, but is less effective in treating metastatic solid tumors as a monotherapy. There is evidence that RT induces immunogenic tumor cell death and enhances tumor-specific T cell infiltration in treated tumors. The abscopal effect, a phenomenon in which tumor regression occurs outside the site of RT, has been observed in both preclinical and clinical trials when RT is combined with immunotherapy. In this study, we show that irradiation treatment of B16 melanoma causes an increase in PS expression on the surface of viable tumor and immune infiltrates. We subsequently examined the effects of combining RT with an antibody that targets PS (mch1N11) [“Mouse version of Bavituximab”] and immune checkpoint blockade (anti-PD-1) in B16 melanoma. We found that treatment with mch1N11 synergizes with RT to improve anti-tumor activity and overall survival in tumor bearing mice. In addition, the triple combination of mch1N11, RT and anti-PD-1 treatment displayed even greater anti-tumor and survival benefit. Analysis of local immune responses in the tumors of treated animals revealed an increase in tumor-associated macrophages with a shift towards a pro-inflammatory M1 phenotype after treatment with RT & mch1N11. In addition, analysis of the systemic immune responses in the spleen and tumor draining lymph nodes revealed an increase in CD8 T cell activation, effector cytokine production and differentiation into effector memory cells in the triple combination. This finding highlights the potential of combining these 3 agents to improve outcome in patients with advanced-stage melanoma and other cancers and may inform the design of clinical studies combining PS-targeting antibodies with RT and/or checkpoint blockade.
POSTER #574 IMAGE: http://www.peregrineinc.com/images/stories/pdfs/aacr2017budhu.pdf

= = = = = = = = = = = = = = = =
AACR’17 2. 4-3-17/8am #1651 - Session: TUMOR MICROENVIRONMENT & CHECKPOINTS [Joint PPHM & Memorial Sloan Kettering]
“Targeting Phosphatidylserine in Combination with Adoptive T Cell Transfer Eliminates Advanced Tumors without Off-Target Toxicities in a Melanoma Preclinical Model” <=NEW(2nd) MSK STUDY
=> Daniel Hirschhorn-Cymerman 1, Sara Sara Schad 1, Sadna Budhu 1, Zhong Hong 1, Xia Yang 1, Hutchins T. Jeff 2, Bruce D. Freimark 2, Michael J. Gray 2, Jedd Wolchok 1, Taha Merghoub [Memorial Sloan Kettering]
1=Memorial Sloan Kettering CC, NYC [ http://www.mskcc.org/research-areas/labs/jedd-wolchok ]
2=Peregrine Pharmaceuticals
---------------------
FROM PPHM’s 4-3-17 PR: http://ir.peregrineinc.com/releasedetail.cfm?ReleaseID=1019762
…”Latest Findings from Ongoing Collaboration with Memorial Sloan Kettering (MSK) Support Potential Applications for Combining CAR T & Anti-PS in Treatment of Solid Tumors”… For this study, a team of MSK researchers led by cancer immunotherapy thought-leaders, Taha Merghoub, Ph.D. and Jedd D. Wolchok, M.D., Ph.D., evaluated and compared the anti-tumor activity and off-target toxicities of adoptive T cell transfer therapy in combination with either PS-targeting antibodies or anti-OX40 antibodies in mice with advanced melanomas. Whereas PS-targeting and anti-OX40 demonstrated comparable tumor regression when administered in combination with transferred adoptive T cells, only the PS-targeting combination achieved these results without any off-target toxicities. By contrast, the anti-OX40 treatment combination triggered off-target inflammatory destruction of healthy tissues. Additional study results demonstrated that the PS-targeting antibodies decreased tumor-induced immunosuppression as evidenced by a decrease in immunosuppressive regulatory T cells (Tregs) and M2 macrophages. This finding is consistent with Peregrine’s belief that bavituximab may modulate the immunosuppressive tumor microenvironment and enhance the activity of immunotherapy agents.
Taha Merghoub, Ph.D., Co-Dir. of the Ludwig Collaborative Laboratory at MSK:
“While adoptive T cell transfer remains one of the most exciting new approaches to treating cancer, to date the toxicity associated with the treatment has limited its potential. We are encouraged that these study results showed that the combination of anti-PS and adoptive T cell treatment led to enhanced anti-tumor effect without any evidence of additional off-target side effects. “We believe that these findings may support potential applications for this combination in solid tumors in the future.”
Joseph Shan, Peregrine’s VP of Clinical & Regulatory Affairs:
“These study results provide further support for our belief that anti-PS agents such as bavituximab can play an important role as part of combination cancer treatments. This is directly tied to the agents’ ability to modulate the tumor microenvironment to combat the immunosuppression that limits the activity of CAR T and immunotherapies. Importantly, we are now also seeing evidence that this targeted modulation of the tumor microenvironment by anti-PS allows for enhanced activity of these other treatments without triggering any off-target toxicities. This is opposed to other conventional immunotherapies such as anti-OX40 with systemic mechanisms of action. We believe this advantageous tolerability profile will be a key benefit in positioning anti-PS agents for inclusion in optimal combination cancer regimens.”
-----------
ABSTRACT #1651:
A viable strategy to treat advanced cancers includes transferring of tumor-specific T cells. T cells that recognize tumor antigens can be expanded and reinvigorated ex-vivo. Furthermore, autologous T cells can be genetically modified to express anti-tumor T cell receptors [TCRs] or chimeric antigen receptors [CARs]. Although the potency and specificity of tumor-specific T cells can be manipulated ex-vivo, once re-infused into patients, the T cells are subjected to immunosuppressive mechanisms established by the tumor. An important immune checkpoint regulator within tumors is phosphatidylserine (PS). Innate immune cells exposed to PS secrete suppressive cytokines and chemokines that can significantly impair the function and activation of anti-tumor T cells. Therefore, monoclonal antibodies that block PS activity can increase the anti-tumor potency of transferred T cells to treat aggressive cancers. Here we show that a PS targeting monoclonal antibody in combination with CD4+ T cells that recognize the melanoma antigen Trp1 can regress very advanced melanomas in all treated mice. Combination of anti-Trp1 CD4+ T cells with other immunomodulatory modalities such as anti-OX40 antibodies, can achieve equivalent treatment rates but these are typically accompanied by severe immune related adverse events. In contrast, in this setting, PS blockade did not show any off-target toxicities. Flow cytometry analysis revealed lower levels of CD206 expression concomitant with higher activation markers in macrophages and neutrophils in tumors from anti-PS treated mice. These results suggest that diminishing suppressive mechanisms locally in adoptive transfer protocols is a highly desirable strategy that can eliminate tumors while minimizing related adverse events.
POSTER #1651 IMAGE: http://www.peregrineinc.com/images/stories/pdfs/aacr2017hirschhorn.pdf

---------------------
8-2015: Presently there are 3 types of Adoptive Cell Transfer (ACT) using effector T cells that are advancing on a path towards regulatory approval:
1. TILs (tumor infiltrating lymphocytes) have been developed with slow but continuing progress over several decades.
2. CARs (chimeric antibody receptors) – newer gene-modified T cells strategy
3. TCRs (T-cell receptors) – newer gene-modified T cells strategy
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4507286/

...“Adoptive T cell transfer is like ‘giving patients a living drug’,” states Renier J. Brentjens, MD/PhD, of Memorial Sloan Kettering Cancer Center (MSKCC).
… https://www.cancer.gov/about-cancer/treatment/research/car-t-cells
...“Adoptive T cell therapy (ACT) is one stone in this new pillar, a potentially powerful approach to cancer treatment that relies on the infusion of tumor-specific T cells.”
… https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4327320
MSK Tweet 4-3-17 https://twitter.com/sloan_kettering :
...See bottom right: “SWIM” - Swim Across America http://www.swimacrossamerica.org

= = = = = = = = =
Interesting 3-31-17 Sloan Kettering Tweet re: AACR’17
3-31-17 9:20amET “Getting ready for #AACR17? #Immunotherapy will be a hot topic. Learn more about how it works (video). @AACR”
https://twitter.com/sloan_kettering
Points to this 4-2016 Video (1:20):
“Immunotherapy – How It Works” https://www.youtube.com/watch?v=COQ1AeoGyco
Beg@:39:
“Immunotherapy drugs release this brake and empower immune cells to fight the cancer.
Sometimes the immune system needs a tune-up before it can fight cancer.
Immune cells can be removed from the body, armed with new proteins that can target cancer cells, and given back to a patient in large numbers.
Once inside the body, the modified immune cells recognize and attack the cancer.
This approach is called CAR T cell therapy. [an Adoptive Cell Transfer “ACT” strategy]
Despite the promise of immunotherapy, not everybody responds.
MSKCC scientists are exploring ways to improve immunotherapy.”

=============AND, THIS 3-23-17 PARKER FOUNDATION RELEASE ON AACR’17:
3-23-17: “Parker Institute for Cancer Immunotherapy Scientists to Present Research on Checkpoint Inhibitors, Adoptive Cell Therapy, and Other Advances in Immuno-oncology at AACR 2017”
Investigators affiliated with the Parker Institute for Cancer Immunotherapy will present some of the most anticipated immuno-oncology research at the 2017 AACR Annual Meeting. The event takes place at the Walter E. Washington Convention Center in Washington, DC, April 1-5 2017. . .
Other adoptive cell therapy abstracts of interest:
• ”Targeting Phosphatidylserine in Combination with Adoptive T Cell Transfer Eliminates Advanced Tumors without Off-Target Toxicities in a Melanoma Preclinical Model” [see http://tinyurl.com/nx5q5os ]
The principal investigator is Taha Merghoub, PhD, Parker Institute member researcher at Memorial Sloan Kettering Cancer Center. Co-authors include Parker Institute Center Director Jedd Wolchok, MD, PhD, at Memorial Sloan Kettering Cancer Center.
http://www.parkerici.org/media/2017/parker-institute-for-cancer-immunotherapy-scientists-to-present-research-on
----------ACT NOTES:
NOTE1:
From 3-2014 Immunotherapy article: “Cancer immunotherapy, particularly adoptive cell transfer (ACT), has shown great promise in the treatment of patients with late-stage disease, including those who are refractory to std. Therapies” https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4372895
NOTE2:
The Parker Foundation: “The Immunotherapy Dream Team”
Through the support from SEAN PARKER, Stand Up To Cancer (SU2C) and the Cancer Research Institute (CRI) formed an Immunology Research Dream Team dedicated to cancer immunology: “Immunologic Checkpoint Blockade & Adoptive Cell Transfer in Cancer Therapy.” The Immunotherapy Dream Team is focused on 2 approaches for this translational cancer research project, which will unite laboratory & clinical efforts towards the immunological treatment, control, and prevention of cancer.
The 1st is investigating blockades (by inhibitory molecules called “checkpoints”) of T lymphocytes’ inhibitory receptors, which block immune responses; and the use of antibodies to remove the checkpoints, once again allowing white blood cells called T lymphocytes to kill the cancer cells.
Second, the Immunotherapy Dream Team is pursuing multiple Adoptive Cell Transfer (ACT) approaches, which increase immunity.”
DREAM TEAM LEADERSHIP: 10 scientists, incl. MSKCC’s Jedd D. Wolchok & Michel Sadelain.
http://parker.org/initiatives/immunotherapy
-------
NOTE: Mem. Sloan’s Dr. Jedd. Wolchok wasco-author of 9 AACR’17 abstracts. 2 of the 9 are w/PPHM re: PS-Targeting. With biotechs, he had one each with Genocea Biosciences(ex-vivo screening), Infinity Pharm(Ph1), Leap Ther.(Ph1), and two with BMS(P1+3). The 8th was MSKCC ONLY, and the 9th was jointly w/Cedars-Sinai/LA. http://www.abstractsonline.com/pp8/#!/4292 .
PEREGRINE (Joint Mem.Sloan Kettering Wolchok Lab & PPHM):
1. 4-2-17 #574 “Phosphatidylserine Targeting Antibody in Combination with Tumor Radiation & Immune Checkpoint Blockade Promotes Anti-Tumor Activity in Mouse B16 Melanoma” <=Same as SITC’16 11-14-16.
2. 4-3-17 #1651 “Targeting Phosphatidylserine in Combination With Adoptive T Cell Transfer Eliminates Advanced Tumors Without Off-Target Toxicities in a Melanoma Preclin. Model” <=NEW/2nd PPHM+MSK.
NON-PEREGRINE:
3. (MSK ONLY): 4-2-17 #874 “Lifting the iron curtain: Imaging cellular barriers to combination chelation-immune checkpoint therapy”
4. (MSK & Genocea Bio. http://www.genocea.com GNCA/$130mm MktCap) 4-2-17 #632 “Genome-scale neoantigen screening using ATLAS prioritizes candidate antigens for immunotherapy in a NSCLC patient”
5. (MSK & Cedars-Sinai/LA) 4-4-17 #4705 “CTLA4 blockade with HER2-directed therapy yields clinical benefit in women undergoing radiation therapy for HER2+ breast cancer brain metastases”
6. (MSK & Infinity Pharm.) 4-4-17 #CT089 “A Ph1, first-in-human study of IPI-549, a PI3K-y inhibitor, as monotherapy & in combo w/nivolumab in adv. solid tumors”
7. (MSK & Leap Ther.) 4-2-17 #CT018 “Intratumor & peripheral Treg modulation as a pharmacodynamic biomarker of the GITR agonist antibody TRX-518 in the 1st in-human trial”
8. (MSK & BMS) 4-3-17 #CT073 “(Ph1) Immunomodulatory effects of NIVO+IPI or NIVO/mono in adv. melanoma: CheckMate038”
9. (MSK & BMS) 4-3-17 #CT075 “OS results from a Ph3 of NIVO+IPI in treatment-naïve pts w/adv. Melanoma: CheckMate067”

FYI: JULY 2014: Mem. Sloan’s Jedd Wolchok Video: “Immunotherapy for Metastatic Melanoma - PD-1 Clinical Trials”… Dr. Wolchok (1:42), “We will not rest until every patient can have a durable response.” http://cyberspaceandtime.com/kvWeW8Zt0sU.video (1:49)
= = = = = = = = = = = = = = = = = = = = = = = = = = = = = = =
Nov9-13 2016: “(SITC) Society for Immunotherapy of Cancer 31st Annual Meeting”, Natl-Harbor MD
”The premier destination for scientific exchange, education, and networking in the Cancer Immunotherapy Field”
SITC = The Society for Immunotherapy of Cancer http://www.sitcancer.org
SITC 2016 Meeting: http://www.sitcancer.org/2016
--------
Abstracts #199 (11-11-16) is the joint MSKCC/PPHM one; its Senior author is Dr. Taha Merghoub (Jedd Wolchok Lab, Memorial Sloan Kettering), who said this in the 5-29-15 PPHM/MSK Collab. Announcement PR (see below): "A key focus of the Wolchok Lab's research is studying novel immunotherapy combinations that work together to enable the immune system to recognize & destroy cancer. This collaboration will allow us to focus on the role & contribution of PS blockade therapy in determining which combination of the current and next generation of immune modulators is likely to increase the extent and amplitude of anti-tumor response. This important pre-clinical & translational work will potentially guide the design of the next generation of clinical studies with bavituximab."
--------
11-14-16 – SITC’16: Joint MSKCC & PPHM Poster, “Phosphatidylserine Targeting Antibody in Combination with Checkpoint Blockade & Tumor Radiation Therapy Promotes Anti-Cancer Activity in Mouse Melanoma”) (#199) … See: http://tinyurl.com/js3fca4
11-14-16/PR: “New Study Demonstrates Anti-Tumor Advantages for Combination Treatment Featuring Peregrine Pharmaceuticals' PS-Targeting Antibodies in a Preclinical Melanoma Model”
* Promising Results of MSK Study Evaluating Combinations of PS-Targeting Treatment, Anti-PD-1 and Radiation in Mouse B16 Melanoma Model Presented at SITC 2016
* New Data from Second Study Conducted by Peregrine Shows Triple Combination of PS-Targeting Treatment, Anti-PD-1 and Anti-LAG3 Created Long-Term Immunity in Triple Negative Breast Cancer Model; Protected Animals when Re-Challenged with Breast Cancer Cells
http://ir.peregrineinc.com/releasedetail.cfm?ReleaseID=999202
TUSTIN, Nov. 14, 2016: Peregrine Pharmaceuticals, Inc. (NASDAQ:PPHM/PPHMP), a biopharmaceutical company committed to improving patient lives by manufacturing high quality products for biotechnology and pharmaceutical companies and advancing its proprietary R&D pipeline, today announced the presentation of positive data from multiple new preclinical studies of the company's phosphatidylserine (PS)-targeting antibodies. Study results highlight that PS-targeting antibodies similar to bavituximab synergize with checkpoint inhibitors and radiation to improve anti-tumor activity in various animal tumor models. Importantly, the improved anti-tumor activity seen in these studies was even greater when PS-targeting therapy was a part of triple combination treatment including anti-PD-1 and another therapy. Data were presented by Peregrine scientists, as well as researchers from Memorial Sloan Kettering Cancer Center (MSK), at the Society for Immunotherapy of Cancer (SITC) 2016 Annual Meeting, which was held November 9-13, 2016 in National Harbor, MD. [ http://www.sitcancer.org/2016 ]
Initial results from Peregrine's ongoing collaboration with MSK researchers were featured in a poster presented by Sadna Budhu, PhD, at SITC 2016. A team of MSK researchers led by cancer immunotherapy thought-leaders, Taha Merghoub, PhD and Jedd D. Wolchok, MD, PhD [ http://www.mskcc.org/research-areas/labs/jedd-wolchok ], evaluated the effects of combining PS-targeting, anti-PD-1 and radiation therapies in the mouse B16 melanoma model. Study data showed that PS-targeting antibodies synergize with both anti-PD-1 and radiation therapy to improve anti-cancer activity. PS-targeting treatment in combination with radiation, as well as triple combination of PS-targeting treatment, anti-PD-1 and radiation, led to a reduction in tumor burden. Median survival for the triple combination treatment still had not been reached at the end of the 80-day observation period with other arms in the study showing median survival that ranged from 24-70 days.
Researchers also evaluated the impact of the PS-targeting and radiation combination treatment on the level and type of immune activity. These results demonstrated that the combination led to a change in the tumor microenvironment, shifting it from immunosuppressive in which tumors are protected to immune active in which tumors are more susceptible to treatment. Analysis of local immune responses in the tumors of the treated animals showed that the combination treatment increased the number of tumor associated macrophages and shifted the macrophage polarization from the immunosuppressive M2 type to the immune active M1 type. When systemic immune responses were analyzed following triple combination of PS-targeting treatment, anti-PD-1 and radiation, researchers also saw evidence of increased immune activity. This was illustrated by key indicators of immune activity, including increases in CD8+ T-cell activation, effector cytokine production and differentiation into effector memory cells.
"Based on these study results, we believe that the targeting of PS is having meaningful activity within the tumor microenvironment in the B16 melanoma model," stated Dr. Jedd Wolchok. "It appears that this activity creates a more immune active environment in which other treatments, including radiation, are able to have a greater anti-tumor impact."
"We have noted that the combination of PS-targeting treatment and radiation, as well as triple combination of PS-targeting treatment, radiation and anti-PD-1, resulted in clear advantages in anti-tumor activity in the mouse B16 melanoma model," said Dr. Taha Merghoub, PhD, co-director of the Ludwig Collaborative Laboratory at MSK. "We believe that these findings suggest the potential benefit of combining these agents to improve the outcomes of patients with cancer. With this in mind, we think this research may play an important role in designing future clinical trials of PS-targeting agents in melanoma and other cancers."
[Note: FULL SITC’16 ABSTRACT #199: http://bit.ly/2dHTEVn
...Full Poster Image: http://peregrineinc.com/images/stories/pdfs/sitc_2016_budhu.pdf ]
A second study, conducted by Peregrine, evaluated the effects of combining PS-targeting, anti-PD-1 and anti-LAG3 therapies in the E0771 triple negative breast cancer (TNBC) model. Initial findings from this study were previously reported and demonstrated that eight of the ten (80%) animals receiving the PS-targeting, anti-PD-1 and anti-LAG3 treatment combination experienced complete tumor regressions, whereas there were no animals (0/10) in the anti-PD-1 and anti-LAG3 combination treatment arm that had a complete regression. New data presented for the first time at SITC demonstrated that the triple combination established a specific and prolonged anti-tumor immune response which protected those eight animals that achieved a complete tumor regression against a re-challenge with the same E0771 TNBC model tumor cells. This sustained anti-tumor response demonstrates the ability of the triple combination treatment to trigger immune system memory and support adaptive immune responses against reemerging disease in the E0771 TNBC model.
[Note: FULL SITC’16 ABSTRACT #213: http://bit.ly/2dpUy4C ]
...Full Poster Image: http://peregrineinc.com/images/stories/pdfs/sitc_2016_gray.pdf ]
Further highlighting the immune impact of the PS-targeting/anti-PD-1/anti-LAG3 treatment combination were initial results of a new analysis from this study using the nCounter ® PanCancer Immune Profiling Panel from NanoString Technologies ®. Data from the analysis demonstrated that the triple combination induced a greater shift in the tumor microenvironment from immunosuppressive to immune active as compared to all other treatment groups. This was evidenced by greater increases in the activity of several critical immune activating pathways, including presentation and processing of antigens and signaling and activation of T-cells, for the triple combination as compared to all other treatments.
"It is very encouraging to see the consistent increase in anti-tumor activity triggered by triple combination treatments that combine PS-targeting agents and anti-PD-1 with other cancer treatments. By demonstrating this activity across multiple studies in multiple tumor models, we are continuing to build scientific support for the therapeutic potential of adding PS-targeting therapies in combination with other cancer treatments, including checkpoint inhibitors such as anti-PD-1," said Jeff T. Hutchins, PhD, Peregrine's VP, Preclinical Research. "As cancer research continues to explore the potential of combination treatments that marry complementary mechanisms, we are pleased to see that our efforts continue to generate data supporting the role that PS-targeting agents such as bavituximab may play in this area."
Bavituximab is an investigational monoclonal antibody that targets PS. Signals from PS inhibit the ability of immune cells to recognize and fight tumors. Bavituximab is believed to override PS mediated immunosuppressive signaling by blocking the engagement of PS with its receptors as well as by sending an alternate immune activating signal. Previous studies demonstrated PS-targeting antibodies shift the functions of immune cells in tumors, resulting in multiple signs of immune activation and anti-tumor responses. Peregrine continues to support and guide clinical development through the evaluation of the preclinical equivalent of bavituximab, ch1N11, in animal model studies.
Peregrine's clinical development strategy for bavituximab currently focuses on small, early-stage, proof-of-concept trials evaluating the drug in combination with other cancer treatments. This approach includes the recently announced grants awarded by the National Comprehensive Cancer Network (NCCN) to support three different clinical trials of bavituximab treatment combinations [See: http://tinyurl.com/gutgwb5 ]. These trials will evaluate novel bavituximab combinations in glioblastoma, head and neck cancer, and hepatocellular carcinoma including an immunotherapy combination. Additionally, Peregrine continues to advance its pre-clinical collaboration with Memorial Sloan Kettering Cancer Center with the goal of evaluating combinations of bavituximab with other checkpoint inhibitors and immune stimulatory agents. The intent behind this strategy is to focus our research and development spending to further validate bavituximab's combination potential as we seek to advance the program though a pharmaceutical or biotechnology partner.
ABOUT PEREGRINE *SNIP*
CONCLUSION SECTION FROM THE JOINT MSKCC/PPHM SITC’16 POSTER:


ABSTRACT: #199/SITC’16/11-11-16(MSK+PPHM): “Phosphatidylserine Targeting Antibody in Combination with Checkpoint Blockade & Tumor Radiation Therapy Promotes Anti-Cancer Activity in Mouse Melanoma”
AUTHORS:
Sadna Budhu(MSKCC PRESENTING AUTHOR) 1, Olivier De Henau 2, Roberta Zappasodi 1, Kyle Schlunegger 3, Bruce Freimark 4, Jeff Hutchins 5, Christopher A. Barker 6, Jedd D. Wolchok 7, Taha Merghoub(Memorial Sloan Kettering SENIOR AUTHOR)
BACKGROUND:
Phosphatidylserine (PS) is a phospholipid that is exposed on the surface of apoptotic cells, some tumor cells and tumor endothelium. PS has been shown to promote anti-inflammatory and immunosuppressive signals in the tumor microenvironment. Antibodies that target PS have been shown to reactivate anti-tumor immunity by repolarizing tumor associated macrophages to a M1-like phenotype, reducing the number of MDSCs in tumors and promote the maturation of dendritic cells into functional APCs. In a B16 melanoma model, targeting PS in combination with immune checkpoint blockade has been shown to have a significantly greater anti-cancer effect than either agent alone. This combination was shown to enhance CD4+ and CD8+ T cell infiltration and activation in the tumors of treated animals. Radiation therapy is an effective focal treatment of primary solid tumors, but is less effective in treating metastatic solid tumors as a monotherapy. There is evidence that radiation induces immunogenic tumor cell death and enhances tumor-specific T cell infiltration in irradiated tumors. In addition, the abscopal effect, a phenomenon in which tumor regression occurs outside the site of radiation therapy, has been observed in both preclinical and clinical trials with the combination of radiation therapy and immunotherapy.
METHODS:
We examined the effects of combining tumor radiation therapy with an antibody that targets PS (1 N11) [Note: PGN635 (B2GPI-dep.) is Fully-Human Bavituximab=1N11=AT004; Genentech studying 89Zr-PGN635 as a Tumor Imaging Agent, “indep. of cancer type”] and an immune checkpoint blockade (anti-PD-1) using the mouse B16 melanoma model. Tumor surface area and overall survival of mice were used to determine efficacy of the combinations.
RESULTS:
We examined the expression of PS on immune cells infiltrating B16 melanomas. CD11b + myeloid cells expressed the highest levels of PS on their surface whereas T cells and B16 tumor cells express little to no PS. These data suggest that targeting PS in B16 melanoma would induce a pro-inflammatory myeloid tumor microenvironment. We hypothesize that therapies that induce apoptotic cell death on tumor cells would enhance the activity of PS-targeting antibodies. We therefore examined the effects of combining a PS-targeting antibody with local tumor radiation. We found that the PS-targeting antibody synergizes with both anti-PD-1 and radiation therapy to improve anti-cancer activity and overall survival. In addition, the triple combination of the PS-targeting antibody, tumor radiation and anti-PD-1 treatment displayed even greater anti-cancer and survival benefit.
CONCLUSIONS:
This finding highlights the potential of combining these 3 agents to improve outcome in patients with advanced-stage melanoma and may inform the design of future clinical trials with PS targeting in melanoma and other cancers *end*
...FULL POSTER IMAGE: http://peregrineinc.com/images/stories/pdfs/sitc_2016_budhu.pdf ]

= = = = = = = = = = = =
9-8-16/CC - JEFF HUTCHINS (VP/PreClinical Res.) http://tinyurl.com/jmy77g3
“...I would now like to provide an update on Peregrine's preclinical I-O focused internal efforts and our collaboration with Memorial Sloan Kettering Cancer Center. The goal of this work is to evaluate combinations of PS targeting with other checkpoint inhibitors and immune stimulatory agents for the purpose of developing new and increasingly effective anticancer treatments. These programs are advancing well and to-date, we have seen impressive signs of activity with new combinations of PS targeting and other treatment modalities such as checkpoint blockers, T-cell agonists and radiation. These new combinations are improving overall survival accompanied with increases in cyto-reactive T cells into the tumor tissue. This exciting new internal work will be presented at the AACR/CRI Immunotherapy Meeting in New York later this month [Sept25-28] and at ESMO in early October [Oct7-11: http://tinyurl.com/jxdppyo ]. We expect the first results from our collaboration with MSK’s Jedd Wolchok Lab investigators [See MSK Collab: http://tinyurl.com/zkvebh6 ] to be presented at SITC in November [Nov9-13: http://www.sitcancer.org/2016 Natl.Harbor MD] and we will provide more detailed information as that presentation becomes available.”
= = = = = = = = = = = = = = = = = = = = = = = = = = = = = = = =
5-29-15: Peregrine & Sloan Kettering Enter Collab. to “Investigate Novel PS-Targeting Immunotherapy Combos” http://tinyurl.com/o3k9ux8
...The studies at MSK will be performed under the direction of Taha Merghoub, PhD, [ http://www.mskcc.org/research-areas/labs/members/taha-merghoub-01 ] Associate Attending Biologist, Melanoma and Immunotherapeutics Service, Ludwig Collaborative and the Swim Across America Laboratory, a part of the laboratory of Jedd D. Wolchok, MD, PhD [ http://www.mskcc.org/research-areas/labs/jedd-wolchok ], a leader in the field of cancer immunotherapy. Dr. Wolchok serves as the Chief, Melanoma and Immunotherapeutics Service, Lloyd J. Old Chair for Clinical Investigation as well as an Associate Director of the Ludwig Center for Cancer Immunotherapy at MSK.
----
”The phosphatidylserine (PS) signaling pathway is a very interesting target for modulating the immune system's response to cancer. We look forward to exploring the potential of PS-targeting agents alone and with other immune modulators that may lead to novel advances in cancer therapy," said Dr. Jedd Wolchok.
–--
As part of the collaboration, researchers at MSK will conduct research to further explore the combination of PS-targeting agents, including bavituximab, that block a primary immunosuppressive pathway thereby allowing anti-tumor immune responses with other immuno-stimulatory agents that enhance immune responses. Specifically, MSK researchers will examine the combination of bavituximab alongside models of checkpoint blockade that are unresponsive to inhibition or co-stimulation given the ability of bavituximab to reprogram myeloid derived suppressor cells (MDSC) and increase tumoricidal T-cells in tumors, a mechanism of action that is complementary to checkpoint blockade and T-cell activation.
–--
"A key focus of the Wolchok Lab's research is studying novel immunotherapy combinations that work together to enable the immune system to recognize & destroy cancer. This collaboration will allow us to focus on the role & contribution of PS blockade therapy in determining which combination of the current & next gen. of immune modulators is likely to increase the extent & amplitude of anti-tumor response. This important pre-clinical and translational work will potentially guide the design of the next gen. of clinical studies with bavituximab," said Dr. Taha Merghoub.
–--
"We are delighted to be working with a world-renowned pioneer and leader in the immuno-oncology space, recognizing that there remains significant research in order for more cancer patients to realize the benefits of combination immune therapy," said Jeff T. Hutchins, PhD, VP of Preclinical Research at Peregrine. ”Our internal and collaborative research presented over the last year has established a robust foundation of PS-targeting activity on which to initiate this next chapter in PS research and development."
–--
"This collaboration is an important extension of our established research efforts to further explore and understand the potential of our PS-targeting platform including bavituximab our lead clinical candidate. This research will focus on better understanding how treatment with PS-targeting agents can assist other anti-tumor immunotherapies in order to work better," said Steven King, CEO of Peregrine. ”Our goal is to change the way cancer patients are treated by allowing their immune system to recognize and fight their disease. This collaboration will undoubtedly assist us in identifying potential new opportunities to better treat patients with cancer."
PPHM’s Sunrise Biomarkers (#1/B2GPI, #2/COMPL,IL-10, #3/IFN-y)
Summary of the 3 known Ph3/Sunrise Biomarker Data Presentations:
#1 10-10-16/ESMO’16: “B2GPI Biomarker(30%pts) StatSig OS 7.7=>13.2mos.”
David R. Spigel: LEAD AUTHOR: CSO/Dir. Lung Cancer Pgm/Sarah Cannon Res.
David E. Gerber: SENIOR AUTHOR: UTSW/Dallas (Sunrise PI)
CO-AUTHORS: R.Natale/CEDARS-SINAI, R.Sanborn/PROVCC, PPHM’s N.Kallinteris, J.Lai, M.Tang, J.Shan, and 7 Intl. Sunrise PI’s: Ger/2, Ukraine, Greece, Spain, Korea, Hungry.
RESULTS:
~200 of 600 pts (~30%) tested positive for B2GPI(200-240). Those 200 received Stat.Sig. Improvement in O/S 7.7=>13.2mos; ie, lived +70% longer.
MORE DETAILS: http://tinyurl.com/hp73njt
#2 12-7-16/WCLC’16(IASLC): “Complement & IL-10 Pathways Id Pts Benefiting from Bavi+Doce”
PRESENTATION CANCELLED – IR said,”Data analysis not completed in time.”
David E. Gerber: LEAD AUTHOR: UTSW/Dallas (Sunrise PI)
Rachael Sanborn: SENIOR AUTHOR: Dir./Thoracic-Oncology, Providence CC/Portland
CO-AUTHORS: L.Horn/VANDY, G.Losonczy/BUDAPEST, R.Natale/CEDARS-SINAI, H.Roder, J.Roder/BIODESIX, PPHM’s N.Kallinteris, M.Tang, J.Shan.
RESULTS:
104 of 193 pts tested high for complement activation, and from that, a 2nd subgrp isolated that tested low IL-10: O/S 5.9=>12.5mos. Remaining 397pts will be presented at WCLC’16.
* ”The Complement System is an enzyme cascade that is a collection of blood & cell surface proteins to help the abilities of antibodies to clear pathogens from an organism.”
* ”IL-10 is an anti-inflammatory TH2 cytokine that has a critical role in limiting the immune response to pathogens to prevent host damage.”
MORE DETAILS: http://tinyurl.com/z8cq8vx
. . . .
12-12-16 CEO/S.King: “We are actively evaluating addl. potential biomarkers and we hope to identify a profile for patients who will receive therapeutic benefit from treatment with bavituximab.” VP/JOE SHAN: ”Numerous addl. biomarkers are currently being evaluated.” http://tinyurl.com/hhn4gga
#3 4/3/17 AACR’17 CT159/25 (Session: Phase II/III Clinical Trials in Progress)
“IFN-y Analysis In Blood & Tissue as a Potential Prognostic and/or Predictive Biomarker”
Lead Author: Nikoletta Kallinteris (PPHM); Senior Author: Rachael Sanborn (Dir./Thoracic-Oncology, Providence CC); CO-AUTHORS: L.Horn/VANDY, T.Guennel/PRECISION-FOR-MEDICINE, PPHM’s N.Kallinteris, M.Tang, S.Yin, J.Lai, J.Shan.
NOTE: “IFNy, or type II interferon gamma, is a cytokine that is critical for innate & adaptive immunity against viral, some bacterial & protozoal infections.”
4-4-17/PR: ( http://ir.peregrineinc.com/releasedetail.cfm?ReleaseID=1020046 )
“SUNRISE Data Analysis Demonstrates Stat. Significant Overall Survival (OS) Improvement in Patients Receiving Bavituximab+Docetaxel and Subsequent Immunotherapy Compared to Placebo+Docetaxel and Subsequent Immunotherapy…”
Pts in the study's BAVI+DOCE arm who received subsequent immunotherapy, the mOS was not reached, while mOS was 13.0mos. for patients in the study's DOCE+PLACEBO arm who received subsequent immunotherapy [HR=0.43; p=.005]. These are the first clinical results reported supporting the hypothesis that bavituximab may modulate the tumor microenvironment to enhance the anti-tumor activity of immunotherapy agents... The presentation highlighted an analysis in which the company evaluated the impact of subsequent immunotherapy treatment, as well as patients' pre-treatment interferon gamma (IFN-y) levels on overall survival. Overall, low peripheral IFN-y correlated with more favorable OS in the patients receiving BAVI+DOCE and is a biomarker of interest. Data were also analyzed by low vs. high IFN-y levels. For patients with low pre-treatment IFN-y levels who received subsequent immunotherapy, those in the BAVI+DOCE arm did not reach mOS compared to mOS of 12.1mos. for the DOCE+PLACEBO arm [HR=0.24; p<.001].
Joseph Shan, Peregrine’s VP of Clinical & Regulatory Affairs (4-4-17/PR):
"We are extremely encouraged by the results of these exploratory analyses which provide further clinical rationale for combining bavituximab and checkpoint inhibitors. This will be the key focus for upcoming early phase clinical trials, which includes a study of bavituximab & pembrolizumab in Head & Neck cancer through our ongoing collaboration with the NCCN.”
---------
**NOTE: Per JDM find, we know that 96 Sunrise Pts received “Subsequent Immunotherapy” - see 10-13-16 ASM CEO S.King Slide#14: http://tinyurl.com/n2bajew
-----------
Nikoletta Kallinteris 1, Leora Horn 2, Min Tang 1, Tobias Guennel 3, Shen Yin 1, Jennifer Lai 1, Joseph Shan 1, Rachel E. Sanborn [Providence CC, Dir./Thoracic-Oncology http://cancergrace.org/faculty/rachel-sanborn-md - Dr. Sanborn's Conflicts of interest: DNA, AZN]
1=Peregrine Pharmaceuticals
2=Vanderbilt-Ingram Cancer Center, Nashville, TN
3=Precision for Medicine, Frederick, MD
4=Providence Cancer Center, Portland, OR
ORIG. AACR’17 ABSTRACT:
BACKGROUND: SUNRISE, a global, double-bind, Phase III trial of docetaxel (D) plus bavituximab (B) or D plus placebo (P) in previously treated non-squamous non-small cell lung cancer (NSCLC), demonstrated similar overall survival (OS) in both treatment arms. Immune correlate analyses including pre-treatment IFN-y levels in blood and tumor tissue were used to potentially identify prognostic and/or predictive correlation with clinical outcome.
METHODS: Serum was isolated from all randomized NSCLC patients at screening, periodically during treatment and at disease progression for evaluation of IFN-y levels using the Simoa TM assay (Myriad RBM, Austin, TX). Available archival tissue was also tested for 91- immune gene activation markers, including IFN-y by the Fluidigm-based gene-expression platform (Sirona Dx, Lake Oswego, OR). Kaplan-Meier statistical methods and Cox proportion hazards models were utilized to evaluate and contrast the correlation of peripheral and intratumoral IFN-y levels with OS. Patients were classified paradoxically as IFN-y "low" with a favorable disease prognosis vs. "high" associated with more aggressive disease based on the median.
RESULTS: Pretreatment serum results were available for 582 out of the 597 randomized patients. Each patient was classified to be pre-treatment IFN-y high or low (< cut-off) using cut-off 0.093 pg/ml, which is the median IFN-y value in the D+B group. Median overall survival (mOS) in all patients with IFN-y low is 11.3mos. (95% CI, 10.1-13.5) vs. 10.4mos. (95% CI, 8.4-11.3) in all IFN-y high; p=0.047. mOS of D+B arm is 11.6mos. (95% CI, 10.2-13.9) and 11.1mos. (95% CI, 9.1-14.7) in the D group; p=0.982 for IFN-y low. mOS of D+B arm is 9.0mos. (95% CI, 6.7-11.2) and 10.6mos. (95% CI, 8.9-13.0) in the D group; p=0.252 for IFN-y high. With the limited intratumoral IFN-y gene expression data (n=33), no statistically significant correlation with OS was observed.
CONCLUSIONS: Correlative approaches identified low peripheral low IFN-y at pretreatment as a biomarker of interest correlating with more favorable clinical outcomes and is consistent with the hypothesis that bavituximab may demonstrate more immunomodulatory effects in patients with “immune cold” tumors.
POSTER #159/25 IMAGE:
http://www.peregrineinc.com/images/stories/pdfs/aacr2017kallinteris.pdf



= = = = = = = =BIOMARKER #3 (IFN-y) AND PPHM/NCCN JOHNS-HOPKINS TRIAL TIE-IN (???):
Dr. Ranee Mehra’s (Johns-Hopkins/SidneyKimmelCC) work with Biomarker IFN-y seems to dovetail into PPHM’s newly revealed Sunrise Biomarker #3 that was presented 4-3-17 at AACR’17: #CT159/25, “IFN-y Analysis In Blood & Tissue as a Potential Prognostic and/or Predictive Biomarker”
-------
1-6-17 Dr. Ranee Mehra (Johns-Hopkins/SidneyKimmelCC), P.I. for the upcoming NCCN Ph2 Bavi+Keytruda Head&Neck trial. Excerpts from her 1-6-17 talk at GBMC/Greater Balt. MC “H+N Grand Rounds”… I do believe she views this trial is an important part of Johns-Hopkins future anti-cancer direction. Also, look at her Slide #36: “Interferon-y Signature”. Is that an exact tie-in to the newly-revealed AACR’17(4-3-17) Sunrise Biomarker #3 Abstract, “IFN-y Analysis In Blood & Tissue as a Potential Prognostic and/or Predictive Biomarker” (embargoed)? I still totally believe that PPHM has been working with the 3 NCCN Bavi Awardees (Moffitt, MassGEN, JohnsHopkins) to weave in the “chosen” Sunrise Biomarker that is associated with generating “improved outcomes for bavituximab-containing treatments” in future trials.
PDF Link(1-6-17): http://www.gbmc.org/workfiles/HeadNeck/Grand%20Rounds/IO_Therapy_SCCHN02017.pdf
-------
Note: DR. RANEE MEHRA was co-author of ASCO’16, “Biomarkers & Response to Pembro(Keytruda) in Recurrent/Metastatic Head & Neck Cancer” - Conclusion: “The IFN-y signature score was significantly associated with ORR, PFS, and OS (all, P< .001)… PD-L2 & IFN-y signature may be associated with clinical response to Pembro[Keytruda] and may offer addl. strategies to improve prediction of response.” http://meetinglibrary.asco.org/content/165708-176


PPHM’s NCCN#3: Ph2/Progressive Squamous Head+Neck (Bavi+Merck’s Keytruda), JOHNS-HOPKINS(Sidney Kimmel CC) - PI: Ranee Mehra, MD
”Phase II Study of Pembrolizumab[Keytruda] & Bavituximab for Progressive Recurrent/Metastatic Squamous Cell Carcinoma of the Head & Neck" http://tinyurl.com/gutgwb5
Ranee Mehra, MD: Dir., Head & Neck Oncology Therapeutics, Johns Hopkins Medicine https://www.linkedin.com/in/ranee-mehra-34a0467
RANEE MEHRA Disclosures(ASCO’16): GSK, Bayer, BMS, Genentech, Novartis, Mirati Ther.


NCCN Bavituximab Trials Announced 9-6-16 - To Begin "Early 2017" http://tinyurl.com/gutgwb5
...#1: Ph1/HepC-Related Hepatocellular(Liver) (Bavi+RAD+Sorafenib), MOFFITT CANCER CENTER - PI: Jessica Frakes, MD - https://clinicaltrials.gov/ct2/show/NCT02989870
...#2: P1-2/Newly Diag. Glioblastoma (Bavi+RAD+Merck’s Temodar), MASS-GEN. CANCER CENTER - PI: Elizabeth Gerstner, MD
...#3: Ph2/Progressive Squamous Head+Neck (Bavi+Merck’s Keytruda), JOHNS-HOPKINS(Sidney Kimmel CC) - PI: Ranee Mehra, MD

9-9-16/CC/JoeShan: “The 3rd award is for a Phase II study of pembrolizumab [Merck’s Keytruda, anti-PD-1] & bavituximab in Head & Neck Cancer. We are particularly excited about this project, as it will be the 1st clinical trial of bavituximab with a checkpoint inhibitor. In multiple previous preclin. studies, we have observed bavituximab's potential to work synergistically with PD1 inhibitors such as pembrolizumab (Merck’s Keytruda).” http://tinyurl.com/ktrfswj
- - - - - - - - - -
Steve King 9-9-16/CC: “Our collaboration with the NCCN has been an important part of our strategy for advancing the bavituximab clinical program in a cost effective way. We earlier provided NCCN with a $2mm grant to support bavituximab related clinical research with no further financial obligations, and these grant awards represent the outcome of a competitive selection process for the best proposals. These studies will evaluate novel bavituximab combinations in Glioblastoma, Head & Neck Cancer, and Hepatocellular Carcinoma, including an immunotherapy combination [Bavi + Merck’s Keytruda], which is a major focus for advancing the program.” http://tinyurl.com/ktrfswj
- - - - - - - - - -
Steve King 9-9-16/CC/Q&A: “I’m very excited about the combinations that were chosen because the Radiation combination is one that in preclinical studies, as was mentioned during the prepared remarks, has always shown a lot of promise. It’s great to be able to now see that put into a clinical setting in a couple of different clinical trials. And the I-O combinations, as Jeff mentioned during his prepared remarks, is a major focus of ours. So to see a Pembro [Merck’s Keytruda] combination picked as well, we are just really excited that these were the 3 winners out of the NCCN selection process.” http://tinyurl.com/ktrfswj
= = = = = = = = = =DETAILS ON SUNRISE BIOMARKERS #1(B2GPI) & #2(COMPLEMENT/IL-10):
#1 10-10-16/ESMO’16: “B2GPI Biomarker(30%pts) StatSig OS 7.7=>13.2mos.” http://tinyurl.com/hp73njt
10-10-16/PR:
“Peregrine Reports Top-Line and Initial Biomarker Data from Phase III SUNRISE Trial of Bavituximab in Oral Presentation at Eur. Society for Medical Oncology (ESMO) 2016 Congress”
-- Company Has Identified Beta-2 Glycoprotein-1 (B2GP1) as a Biomarker that Correlates with Statistically Significant Improvement in Overall Survival for Patients Receiving the Bavituximab Combination Compared to Chemotherapy Alone
-- Ongoing SUNRISE Trial Biomarker Analysis Expected to Identify Addl. Biomarkers Associated with Patients Benefiting from Bavituximab Treatment that Will Help Guide Program's Future Clinical Development. . .
** "With every clinical trial we conduct, we are constantly reminded of the difficulty involved in treating patients with NSCLC. This continues to prove to be a very challenging cancer to combat and the need for effective treatments remains high," David R. Spigel, MD, CSO and PgmDir. of Lung Cancer Res. at the Sarah Cannon Res. Inst. and one of the lead investigators in the SUNRISE trial. "The findings with regard to B2GP1 that have been collected as part of the ongoing SUNRISE trial data analysis are interesting and support further investigation."
** Peregrine intends to further evaluate the role of B2GP1 levels in response to bavituximab therapy in future clinical trials. The company has filed a new patent application directed to the use of this initial biomarker discovery. Addl. patient sample testing & analysis is ongoing and may result in other biomarkers of importance.
--------------
** Data presented at ESMO’16 demonstrated that patients with pre-treatment B2GP1 levels between 200 and 240 (representing approx. 30% of randomized patients) achieved a statistically significant, 5.5-mo. improvement (13.2 mos. vs. 7.7 mos.) in MOS as compared to patients in the ctl. group with the same range of B2GP1 levels [p = 0.049; HR=.67].
-------------
** "We would once again like to thank all of the patients, clinical investigators and scientists who participated in the SUNRISE trial and have made it possible for us to continue to collect and analyze a range of key data from the study. While we were disappointed with the trial being discontinued earlier in the year, we are excited by the fact that we are beginning to learn important information from the trial through the ongoing biomarker analysis program that will be critical in helping guide the future clinical development of bavituximab," said Joseph Shan, VP/Clin&Reg.Affairs at Peregrine. "It is encouraging that the initial biomarker analysis has identified an important biomarker early in the process and we are optimistic that additional biomarkers associated with improved outcomes for bavituximab-containing treatments will be identified as the analysis continues. We expect to be able to share the emerging data over the coming months at scientific & medical conferences as the more results become available.”
http://investorshub.advfn.com/boards/read_msg.aspx?message_id=125687447
= = = = = = = = = = = = = = = =
#2 12-7-16/WCLC’16(IASLC): “Complement & IL-10 Pathways Id Pts Benefitting from Bavi+Doce” <=CANCELLED/”Anal.Not.Done(IR)”
...Lead author (presenter) is UTSW’s Dr. David Gerber (previously presented Ph2/NSCLC data and Prelim. SUNRISE data at AACR’14). The Senior author is Dr. Rachel E. Sanborn, Co-Dir., Thoracic Oncology Pgm, Robert W. Franz Cancer Res. Center, Earle A. Chiles Res. Inst., Providence CC, Portland, OR. Interestingly, one co-author is Heinrich Roder, CTO of Biodesix, Boulder CO...
ABSTRACT CONCLUSION:
“...Proteomic & correlative approaches identified complement activation and low IL-10 levels as important pathways for predicting improved outcomes of patient treatment with Doce+Bavi, in line with preclinical work on Bavi’s MOA...” [Pts=193, within the 1st subgroup of N=104, a 2nd subgroup isolated: MOS 5.9mos => 12.5mos.]
Dec4-7 2016: “WCLC’16 - IASLC’s 17th World Conf. on Lung Cancer”, Vienna, Austria
http://wclc2016.iaslc.org Pgm: http://wclc2016.iaslc.org/wp-content/uploads/2016/10/WCLC-2016-Poster-Program.pdf
Poster Session with Presenters Present (ID 472) - Track: Advanced NSCLC
12/7/16 2:30-3:45pm David E. Gerber [UTSW], J. Roder, N.L. Kallinteris, L. Horn, G. Losonczy, R. Natale, M. Tang, Heinrich Roder [CTO, Biodesix http://www.biodesix.com/project/heinrichroder ], Joe S. Shan [VP/Clin+Reg], Rachel E. Sanborn [Providence Portland Medical Ctr]
”A Pre-Treatment Serum Test Based on Complement & IL-10 Pathways Identifies Patients Benefiting from the Addition of Bavituximab to Docetaxel”
ABSTRACT Book PDF: http://wclc2016.iaslc.org/wp-content/uploads/2016/12/WCLC2016-Abstract-Book_vF-WEB_revDec12.pdf
ABSTRACT: http://library.iaslc.org/virtual-library-search?product_id=6
BACKGROUND:
SUNRISE, a global, double-bind, Phase III trial of docetaxel (D) plus bavituximab (B) or D plus placebo (P) in previously treated non-squamous non-small cell lung cancer, demonstrated similar overall survival (OS) in both treatment arms. Mass spectrometry and correlative analysis were used to create a test able to identify a subgroup of patients benefitting from the addition of B to D.
METHODS:
Pre-treatment serum samples were available for 197 of the first 200 subjects enrolled in the trial. Mass spectra could be generated for 193 samples using the Deep MALDI method (Duncan et al, ASMS 2013), processed and features (peaks) identified. Mass spectral (MS) features associated with various biological functions were identified using a gene set enrichment analysis approach. Analysis of scores based on these MS feature, subsets indicated that in patients with High Complement Activation outcome depended on IL-10 activation in D+B but not in D+P. A test using the MS features associated with these functions was created to reliably identify a patient subgroup associated with clinical benefit using modern machine learning methods.
RESULTS:
Complement activation, as assessed by a classifier trained using related MS features, was a prognostic factor in both treatment arms, with high activation associated with poorer clinical outcome (OS HR = 0.54, log-rank p = 0.013 for D+B; OS HR = 0.60, log-rank p = 0.040 for D+P). Within the subgroup with high complement activation [N=50 (D+B); N=54 (D+P)], a second classifier using features related to IL-10 activation was able to isolate a subgroup of patients showing numerical benefit from the addition of B [Bavituximab] [median OS 5.9mos.(D+Placebo), 12.5mos.(D+Bavituximab)]. The remaining subgroup showed no benefit from addition of B [MOS 10.4mos.(D+P), 5.6mos.(D+B)]. Blinded validation of the test in the remainder 397 patients randomized in SUNRISE is will be presented.
CONCLUSION:
Proteomic and correlative approaches identified complement activation and low IL-10 levels as important pathways for predicting improved outcomes of patient treatment with D+B, in line with preclinical work on B’s mechanism of action. The test resulting from this work will undergo blinded indep. validation.
- - - - - - - - -
12-20-16/S.Diaz(per Cheynew post #282068): “There was no poster in Vienna. The team was working on a very tight timeframe and, despite their best efforts, couldn’t complete the data analysis in time. Given the crunch, we knew there was a possibility that we wouldn’t meet the timing, so we never issued a PR announcing that we would present. Hence no PR announcing that we did not present.” S.DIAZ/FOLLOWUP: ”Unfortunately, I don’t have a timeline for completion of the analysis or where/when it might be presented. We’ll certainly announce this as soon as we know for sure. Only Joe Shan attended the conference for Peregrine.”
= = = = = = = = = =NOTE:
Ann Oncol (11-8-16/suppl8): ESMO Symposium on Immuno-Oncology, Nov4-6 2016, Lausanne, Switzerland
#30P: “Proteomic Signature Analysis & Application in Clinical Development of the Novel Phosphatidylserine-Targeting Immunotherapy, Bavituximab”
http://annonc.oxfordjournals.org/content/27/suppl_8/mdw525.30
David E. Gerber [UTSW] 1, N.L. Kallinteris 2, L. Horn 3, G. Losonczy 4, R. Natale 5, Heinrich Roder 6 [CTO, Biodesix], M. Tang 7, J. Lai 2, J. Shan 8, Rachel E. Sanborn [9=Providence Portland Medical Ctr]
1 Oncology, UTSW-MC/Dallas
2 Clinical, Peregrine Pharmaceuticals Inc.
3 Oncology, Vanderbilt Ingram CC, Nashville, TN
4 Oncology, Semmelweis Univ., Budapest, Hungary
5 Oncology, Cedars-Sinai M/C, Los Angeles
6 Biodesix, Boulder, CO [CTO, Dr. Heinrich Roder: http://www.biodesix.com/project/heinrichroder ] “Founded in 2005, Biodesix discovers & commercializes cancer tests (diagnostics) that help patients & their doctors make more informed decisions about treatment based on a patient’s unique molecular profile.”
7 Biostatistics, Peregrine Pharmaceuticals Inc.
8 Clinical & Regulatory Affairs, Peregrine Pharmaceuticals, Inc.
9 Thoracic Oncology, Providence Cancer Care, Providence, OR
Aim/Background:
Understanding the multi-dimensional characteristics of cancer is essential to patient selection and treatment planning. Topline results from SUNRISE, a global double-blind Phase III trial of docetaxel + bavituximab (D+B) vs. docetaxel + placebo (D) in previously treated non-squamous NSCLC demonstrated mOS of 10.7mos. in the D+B group and 10.8mos. for the D group, which was unexpectedly different from the assumed 9.1mos. for D+B vs. 7.0mos. used for study powering. VeriStrat, a….[must subscribe]
- - - - - - - - - -
5-31-14 ASCO’14: David Gerber/Joe Shan Poster on Ph3/SUNRISE Trial (#TPS8129) http://tinyurl.com/nv4jloo
Next 2 posts updated with AACR'17 poster Images.
Vin, you said, “They lived because they were_given_Opdivo”. You need to re-read (read) the PR. The people that didn’t live longer also “were given Opdivo” (or whatever the I-O drug they subsequently went to). The point is, those that stat-sig. “lived longer” GOT BAVI FIRST vs. the others that DIDN’T GET BAVI FIRST.
FROM PPHM’s 4-4-17 PR: http://ir.peregrineinc.com/releasedetail.cfm?ReleaseID=1020046
…”SUNRISE Data Analysis Demonstrates Stat. Significant Overall Survival (OS) Improvement in Patients Receiving Bavituximab+Docetaxel and Subsequent Immunotherapy Compared to Placebo+Docetaxel and Subsequent Immunotherapy… AACR’17 presentation (#CT159/25) of results of a new analysis of the Phase III SUNRISE trial… Data demonstrated that for patients in the study's BAVI+DOCE treatment arm who received subsequent immunotherapy, the mOS was not reached, while mOS was 13.0mos. for patients in the study's DOCE+PLACEBO arm who received subsequent immunotherapy [HR=0.43; p=.005]. These are the first clinical results reported supporting the hypothesis that bavituximab may modulate the tumor microenvironment to enhance the anti-tumor activity of immunotherapy agents... The presentation highlighted an analysis in which the company evaluated the impact of subsequent immunotherapy treatment, as well as patients' pre-treatment interferon gamma (IFN-y) levels on overall survival. Overall, low peripheral IFN-y correlated with more favorable OS in the patients receiving BAVI+DOCE and is a biomarker of interest. Data were also analyzed by low vs. high IFN-y levels. For patients with low pre-treatment IFN-y levels who received subsequent immunotherapy, those in the BAVI+DOCE arm did not reach mOS compared to mOS of 12.1mos. for the DOCE+PLACEBO arm [HR=0.24; p<.001].
Joseph Shan, Peregrine’s VP of Clinical & Regulatory Affairs:
"We are extremely encouraged by the results of these exploratory analyses which provide further clinical rationale for combining bavituximab and checkpoint inhibitors. This will be the key focus for upcoming early phase clinical trials, which includes a study of bavituximab and pembrolizumab in head and neck cancer through our ongoing collaboration with the NCCN.”
...Bavituximab is an investigational chimeric monoclonal antibody that targets phosphatidylserine (PS). Signals from PS inhibit the ability of immune cells to recognize and fight tumors. PS-targeting antibodies have demonstrated an ability to shift the functions of immune cells in tumors, resulting in multiple signs of immune activation and anti-tumor immune responses. Bavituximab is believed to override PS immunosuppressive signaling by blocking the engagement of PS with its receptors and sending an alternate immune activating signal.
**NOTE: Per JDM find, we know that 96 Sunrise Pts received “Subsequent Immunotherapy” - see 10-13-16 ASM SK Slide#14: http://tinyurl.com/n2bajew


4-4-17 http://investorshub.advfn.com/boards/read_msg.aspx?message_id=130151723
WOW: 2 Stat. Sig. Improvements when Bavi added. “mOS was not reached” in both cases where the BAVI arm "received subsequent immunotherapy"...
FROM PPHM’s 4-4-17 PR: http://ir.peregrineinc.com/releasedetail.cfm?ReleaseID=1020046
…”SUNRISE Data Analysis Demonstrates Stat. Significant Overall Survival (OS) Improvement in Patients Receiving Bavituximab+Docetaxel and Subsequent Immunotherapy Compared to Placebo+Docetaxel and Subsequent Immunotherapy… AACR’17 presentation (#CT159/25) of results of a new analysis of the Phase III SUNRISE trial… Data demonstrated that for patients in the study's BAVI+DOCE treatment arm who received subsequent immunotherapy, the mOS was not reached, while mOS was 13.0mos. for patients in the study's DOCE+PLACEBO arm who received subsequent immunotherapy [HR=0.43; p=.005]. These are the first clinical results reported supporting the hypothesis that bavituximab may modulate the tumor microenvironment to enhance the anti-tumor activity of immunotherapy agents... The presentation highlighted an analysis in which the company evaluated the impact of subsequent immunotherapy treatment, as well as patients' pre-treatment interferon gamma (IFN-y) levels on overall survival. Overall, low peripheral IFN-? correlated with more favorable OS in the patients receiving bavituximab+docetaxel and is a biomarker of interest. Data were also analyzed by low versus high IFN-? levels. For patients with low pre-treatment IFN-? levels who received subsequent immunotherapy, those in the BAVI+DOCE arm did not reach mOS compared to mOS of 12.1mos. for the DOCE+PLACEBO arm [HR=0.24; p<.001].
Joseph Shan, Peregrine’s VP of Clinical & Regulatory Affairs:
"We are extremely encouraged by the results of these exploratory analyses which provide further clinical rationale for combining bavituximab and checkpoint inhibitors. This will be the key focus for upcoming early phase clinical trials, which includes a study of bavituximab and pembrolizumab in head and neck cancer through our ongoing collaboration with the NCCN.”
...Bavituximab is an investigational chimeric monoclonal antibody that targets phosphatidylserine (PS). Signals from PS inhibit the ability of immune cells to recognize and fight tumors. PS-targeting antibodies have demonstrated an ability to shift the functions of immune cells in tumors, resulting in multiple signs of immune activation and anti-tumor immune responses. Bavituximab is believed to override PS immunosuppressive signaling by blocking the engagement of PS with its receptors and sending an alternate immune activating signal.
Oops, sorry, Vin. “The people that lived longer also were given Opdivo” should have read “The people that didn’t live longer also were given Opdivo”. Will fix and repost. (Delete)
Immunovaccine PR’d the AACR’17 IMMUNOVACCINE+PPHM #3657, ”PSTargeting Enhances Anti-Tumor Activity of a Tumor Vaccine(DepoVax), HPV-Induced Tumor Model” today. See below.
Apr1-5 2017: “AACR 2017”, WashDC
http://www.aacr.org/Meetings/Pages/MeetingDetail.aspx?EventItemID=105
4-4-17/8am #3657 - Session: BITES BISPECIFICS & CHECKPOINTS
”Phosphatidylserine-Targeting Antibodies Enhance Anti-Tumor Activity of a Tumor Vaccine in a HPV-Induced Tumor Model”
=> Genevieve Weir 1, Tara Quinton 1, Jeff T. Hutchins 2, Bruce D. Freimark 2, Marianne Stanford (VP/Res., Immunovaccine)
1=Immunovaccine, Inc., Halifax, NS, Canada [ https://www.imvaccine.com ]
2=Peregrine Pharmaceuticals
[Note: clearly, this study is combining PPHM’s Anti-PS with ImmunoVaccine’s DepoVax Vaccine Adjuvanting Platform https://www.imvaccine.com/depovax.php ]
ABSTRACT:
Antibodies targeting phosphatidylserine (PS) have been shown to induce anti-tumor responses by induction of tumor-specific T cells. Based on this observation, we evaluated the responses of PS and PD-1 targeting antibody therapy to enhance anti-tumor responses of a HPV16 peptide vaccine formulated in DepoVax (DPX) in mice bearing HPV-transformed C3 mouse tumors. The addition of PS-targeting antibody (mch1N11) [“Mouse version of Bavituximab”] to DPX/metronomic cyclophosphamide (mCPA) immunotherapy prolonged survival in comparison to mice receiving an isotype control in combination with DPX/mCPA. When anti-PD-1 was added to mch1N11 + mCPA, there was no increase in survival. The addition of mch1N11 to DPX/mCPA immunotherapy had no effect on tumor growth or survival in the aggressive B16-F10 model. TIL analysis revealed an increase in CD8+ T cells, antigen specific CD8+ T cells and PD-1+ T cells in the tumor with mch1N11 treatment. The expression of surface markers for macrophages (CD68high, F4/80) and dendritic cells (CD11c) were also increased in the tumors of mice treated with mch1N11. RT-qPCR analysis of the tumor confirmed higher mRNA expression of T cells markers (CD8, Granzyme B, PD-1) and antigen presenting cell markers (F4/80, CD74). In the spleen, expression of cell surface markers for monocytes (CD11b) and PD-1+ T cells (CD8) were elevated in groups treated with mch1N11 in combination with anti-PD-1. Combined, these findings indicate that in this model, PS-targeting antibodies can enhance the activity of phagocytic cells involved in antigen presentation. We have found that PD-1 expression increases as anti-tumor activity increases, therefore these results also provide an indication that antibodies targeting PS enhance the anti-tumor immune response induced by DPX/mCPA therapy. The observations suggest that PS-targeting antibodies may enhance therapeutic vaccines for the treatment of cancer.
-------
4-5-17 Immunovaccine PR: “...Immunovaccine Presents Preclinical Research at AACR’17... Part of Ongoing Effort to Identify Novel Combinations of DepoVax-based Immuno-oncology Candidates to Improve the Responses of Other Novel Immunotherapy Agents… preclin. data presented demonstrated that phosphatidylserine (PS) targeting antibodies can enhance the anti-cancer activity of its DepoVax-based therapeutic vaccine platform. In the study, researchers combined a Peregrine Pharmaceuticals’ PS-targeting antibody compound (mch1N11) with a DepoVax-based HPV16 peptide vaccine & metronomic cyclophosphamide (mCPA). This combined immunotherapy prolonged survival in C3 mouse models as compared to mice receiving an isotope control in combination with DepoVax/mCPA. Addl. analysis also demonstrated an increase in T cells in the tumor following treatment. Taken together, researchers believe that the data suggests that the antibody targeting PS can increase the anti-tumor immune response induced by a DepoVax-based cancer immunotherapy. Marianne Stanford, PhD, VP/Res., ”This study is another step in our exploration of combining DepoVax-based cancer vaccines & other promising immuno-modulatory compounds. Our process of generating supportive preclin. data to guide a potential clinical path forward has been effective in identifying these novel combinations in the past. We now look forward to continued work with our partners to advance combination candidates into and through the clinic, with the goal of expanding treatment options for hard-to-treat cancers.” The study was designed to analyze the potential synergistic effects of combining DPX-based immunotherapies with bavituximab, Peregrine’s investigational chimeric monoclonal antibody that targets PS...” https://www.imvaccine.com/releases.php?releases_id=421
-----
...Ahhhhh, ImmunoVaccine & Dr. Wolchok – zero doubt IMO how & why PPHM & Immunovaccine have hooked up...
ImmunoVaccine & Jedd Wolchok go back to ASCO’13:
6-3-13: “Our poster (Ph1 data) is one of few selected for detailed discussion at ASCO by Dr. Jedd Wolchok, top cancer immunotherapy thought leader… Immunovaccine Inc. believes that these immune responses are consistent in profile to those necessary from a cancer vaccine to potentially impact disease progression. These study results were further discussed by Dr. Jedd Wolchok of Memorial Sloan-Kettering CC, a top thought leader in the area of cancer immunotherapy, at the poster discussion session that followed.”
https://www.imvaccine.com/communications.php?communications_id=11
Oh, most certainly I will add the Full_PPHM/IFN-y_Poster when it hits, My Friend! ...Plus any more comments about AACR'17 you add between now and then.
Very Welcome, N40K - owed it TO YOU for your excellent Live Reporting. I haven't seen the full Poster PDF for Peregrine's #CT159 ”IFN-y Analysis In Blood & Tissue as a Potential Prognostic and/or Predictive Biomarker” as yet. Will check http://www.peregrineinc.com/ each day. Try to get a pic of the Peregrine Booth (#3312) if you can.
Thx for finding my 12-7-17 typo, will fix!
AZN’s AACR #1773/IFN-y Biomarker; Compare to PPHM’s #CT159/IFN-y
Recall that North40K/292662 http://tinyurl.com/k58d9ow & Hutschi/292661 http://tinyurl.com/mtq2b9c were discussing AZN’s AACR’17 poster #1773, titled, “A Baseline IFNG (IFN-y) Gene Expression Signature Correlates with Clinical Outcomes in Durvalumab-Treated Advanced NSCLC Cancer Patients”, with N40K suggesting that AZN’s #1773 may be “quite complementary and not inconsistent” with PPHM’s 4-4-17 AACR/#CT159, “IFN-y Analysis In Blood & Tissue as a Potential Prognostic and/or Predictive Biomarker” that was PR’d today.
Now, on 4-4-17, N40K posted this in #293764 http://tinyurl.com/k3tnlno :
“I was told by Joe Shan (VP/Clin+Reg), as he looked at the 2 posts 292661 & 292662 from a printed sheet of paper I "happened" to have with me, that the respective companies, PPHM & AZN, had not seen each others data on those AACR abstracts until 4-3-17. Shan himself took a photo of AZN abstract #1773 that morning, but had not studied it himself. He forwarded his photo of that AZN abstract (PDF) by email to me. I then asked Shan what the status of AZN was. A smile & shrug of shoulders appeared as his answer. He then explained why the 2 abstracts are not necessarily inconsistent/contradictory, as I first thought. I also went to AZN booth at AACR yesterday afternoon to see if they had a printed handout of that abstract. They did not. I was referred to AZN website as a possible source for that abstract [it had been taken down from its location at AACR by noon yesterday]. I have asked 2 of our moderators to post the AZN abstract PDF here. I do not know if a published of PPHM's embargoed abstract from Sunrise (4-3-17 #CT159/IFN-y) has appeared anywhere yet.”
...So, here is the AZN/#1773/IFN-y poster image,
followed by what we know about the 4-3-17 PPHM
#CT159/IFN-y poster for comparison...
AACR’17 4-3-17: Poster Session - Predictive Biomarkers 1 #1773 (Medimmune/AstraZeneca)
4-3-17: “A Baseline IFNG Gene Expression Signature Correlates with Clinical Outcomes in Durvalumab-Treated Advanced NSCLC Cancer Patients”
http://www.abstractsonline.com/pp8/#!/4292/presentation/1304
ALL AZN/MEDI: Brandon W. Higgs, Chris A. Morehouse, Katie Streicher, Philp Z. Brohawn, Keith Steele, Marlon Rebelatto, Fernanda Pilataxi, Carlos Bais, Li Shi, Xiaoping Jin, Joyce Antal, Ashok Gupta, Koustubh Ranade
ABSTRACT
Durvalumab (D) is a human IgG1 monoclonal antibody which inhibits PDL1 binding to PD-1 and CD80, restoring antitumor immunity. In D-treated (tx) NSCLC patients (pts), we previously reported high baseline levels of tumoral PD-L1 protein and IFN? mRNA expression associated with improved ORRs, PFS and OS. Here, a gene expression signature of baseline tumors associates with improved outcomes on D. CP1108/NCT01693562 was a nonrandomized phase 1/2 trial evaluating D in advanced previously tx NSCLC or other solid tumors. By 29APR2016, 304 NSCLC pts received 10 mg/kg Q2W of D <=12 months with median 18.8 months follow up. RNA sequencing of frozen biopsies was conducted on 97 NSCLC tumors of sufficient quality with matched IHC for tumoral PD-L1 on 92 fresh or archival biopsies. Among 21 pre-identified immune-related genes, mRNAs for IFNG, LAG3, CXCL9, and PDL1 individually correlated best with outcomes in NSCLC after adjustment for sex, age, prior therapy, histology, ECOG and smoking. A signature was developed as mean mRNA levels of the 4 genes; signatures >upper tertile were IFNG signature positive (IFNGS+). Analysis was performed on NSCLC, then applied to 30 available urothelial bladder cancer (UBC) biopsies. NSCLC with >=25% tumor cells stained for PD-L1 at any intensity were PD-L1+. 29 NSCLC had pre/post-treatment tumors for mRNA analysis. KM and Cox PH models were used. IFNGS+ D-tx NSCLC pts had higher ORR, median PFS and OS compared to PDL1+, PDL1-, and IFNGS- pts (Table 1); IFNGS+ UBC D-tx pts also correlated with these outcomes. Following D treatment, IFNGS was induced in NSCLC pts (FC=2; p=0.0046) regardless of clinical response. High levels of pre-treatment IFNGS in NSCLC pts associated with greater benefit from D. D induces IFNGS within the tumor microenvironment. Observations from other tumor types will be presented.
Table 1. Clinical outcomes by IFNGS or PD-L1 status...



= = = = = =
Apr1-5: AACR 2017, WashDC – Peregrine’s #CT159/INF-y:
THIS A NEW (3RD) KNOWN SUNRISE BIOMAKER ANALYSIS:
PPHM’s #5. 4-3-17/1pm #CT159/25 - Session: Phase III Clinical Trials & Phase II/III Clinical Trials in Progress
http://www.abstractsonline.com/pp8/#!/4292/presentation/12566
”IFN-y Analysis In Blood & Tissue as a Potential Prognostic and/or Predictive Biomarker”
=> Nikoletta Kallinteris 1, Leora Horn 2, Min Tang 1, Tobias Guennel 3, Shen Yin 1, Jennifer Lai 1, Joseph Shan 1, Rachel E. Sanborn [Providence CC, Dir./Thoracic-Oncology http://cancergrace.org/faculty/rachel-sanborn-md - Dr. Sanborn's Conflicts of interest: DNA, AZN]
1=Peregrine Pharmaceuticals
2=Vanderbilt-Ingram Cancer Center, Nashville, TN
3=Precision for Medicine, Frederick, MD
4=Providence Cancer Center, Portland, OR
ABSTRACT:
BACKGROUND: SUNRISE, a global, double-bind, Phase III trial of docetaxel (D) plus bavituximab (B) or D plus placebo (P) in previously treated non-squamous non-small cell lung cancer (NSCLC), demonstrated similar overall survival (OS) in both treatment arms. Immune correlate analyses including pre-treatment IFN-y levels in blood and tumor tissue were used to potentially identify prognostic and/or predictive correlation with clinical outcome.
METHODS: Serum was isolated from all randomized NSCLC patients at screening, periodically during treatment and at disease progression for evaluation of IFN-y levels using the Simoa TM assay (Myriad RBM, Austin, TX). Available archival tissue was also tested for 91- immune gene activation markers, including IFN-y by the Fluidigm-based gene-expression platform (Sirona Dx, Lake Oswego, OR). Kaplan-Meier statistical methods and Cox proportion hazards models were utilized to evaluate and contrast the correlation of peripheral and intratumoral IFN-y levels with OS. Patients were classified paradoxically as IFN-y "low" with a favorable disease prognosis vs. "high" associated with more aggressive disease based on the median.
RESULTS: Pretreatment serum results were available for 582 out of the 597 randomized patients. Each patient was classified to be pre-treatment IFN-y high or low (< cut-off) using cut-off 0.093 pg/ml, which is the median IFN-y value in the D+B group. Median overall survival (mOS) in all patients with IFN-y low is 11.3mos. (95% CI, 10.1-13.5) vs. 10.4mos. (95% CI, 8.4-11.3) in all IFN-y high; p=0.047. mOS of D+B arm is 11.6mos. (95% CI, 10.2-13.9) and 11.1mos. (95% CI, 9.1-14.7) in the D group; p=0.982 for IFN-y low. mOS of D+B arm is 9.0mos. (95% CI, 6.7-11.2) and 10.6mos. (95% CI, 8.9-13.0) in the D group; p=0.252 for IFN-y high. With the limited intratumoral IFN-y gene expression data (n=33), no statistically significant correlation with OS was observed.
CONCLUSIONS: Correlative approaches identified low peripheral low IFN-y at pretreatment as a biomarker of interest correlating with more favorable clinical outcomes and is consistent with the hypothesis that bavituximab may demonstrate more immunomodulatory effects in patients with “immune cold” tumors.
---------------------
FROM PPHM’s 4-4-17 PR: http://ir.peregrineinc.com/releasedetail.cfm?ReleaseID=1020046
…”SUNRISE Data Analysis Demonstrates Stat. Significant Overall Survival (OS) Improvement in Patients Receiving Bavituximab+Docetaxel and Subsequent Immunotherapy Compared to Placebo+Docetaxel and Subsequent Immunotherapy… AACR’17 presentation (#CT159/25) of results of a new analysis of the Phase III SUNRISE trial… Data demonstrated that for patients in the study's BAVI+DOCE treatment arm who received subsequent immunotherapy, the mOS was not reached, while mOS was 13.0mos. for patients in the study's DOCE+PLACEBO arm who received subsequent immunotherapy [HR=0.43; p=.005]. These are the first clinical results reported supporting the hypothesis that bavituximab may modulate the tumor microenvironment to enhance the anti-tumor activity of immunotherapy agents... The presentation highlighted an analysis in which the company evaluated the impact of subsequent immunotherapy treatment, as well as patients' pre-treatment interferon gamma (IFN-y) levels on overall survival. Overall, low peripheral IFN-y correlated with more favorable OS in the patients receiving BAVI+DOCE and is a biomarker of interest. Data were also analyzed by low vs. high IFN-y levels. For patients with low pre-treatment IFN-y levels who received subsequent immunotherapy, those in the BAVI+DOCE arm did not reach mOS compared to mOS of 12.1mos. for the DOCE+PLACEBO arm [HR=0.24; p<.001].
Joseph Shan, Peregrine’s VP of Clinical & Regulatory Affairs:
"We are extremely encouraged by the results of these exploratory analyses which provide further clinical rationale for combining bavituximab and checkpoint inhibitors. This will be the key focus for upcoming early phase clinical trials, which includes a study of bavituximab and pembrolizumab in head and neck cancer through our ongoing collaboration with the NCCN.”
...Bavituximab is an investigational chimeric monoclonal antibody that targets phosphatidylserine (PS). Signals from PS inhibit the ability of immune cells to recognize and fight tumors. PS-targeting antibodies have demonstrated an ability to shift the functions of immune cells in tumors, resulting in multiple signs of immune activation and anti-tumor immune responses. Bavituximab is believed to override PS immunosuppressive signaling by blocking the engagement of PS with its receptors and sending an alternate immune activating signal.
---------
AACR’17 Attendee North40000 comments (iHub #293687): “I was told in response to a question I asked yesterday that PPHM does not know precisely how many patients in the Sunrise treatment arm are still alive.. Those pts are all over the world, and those who continued on maint. therapy remain uncounted. Best guess/ball park, as many as 6-12 pts in treatment arm who continue on maint. are still alive. After all, PPHM does continue to ship Bavi for use in that maint. Therapy. That surprised me that PPHM could not say how many are still alive. One would think that the CROs & PI’s would know. I made my views clear to at least 4-5 PPHM personnel on the convention floor yesterday that it would be useful to publish an accurate count as possible, with an "as of xx date " time attached. I was told that at least some of the above patients are back at work.”
-------
This AACR’17 #CT159/25 IFN-y is certainly the 3rd Sunrise Biomarker.
Known Ph.3 Sunrise Biomarkers/UTSW’s Dr. David Gerber et al:
#1=B2GPI: 10-10-16 http://tinyurl.com/hp73njt
#2=Complement & IL-10 Pathways(12-7-17/IASLC delayed, “not done”, after prelim. abstract said, “Pts=193, within the 1st subgroup of N=104, a 2nd subgroup isolated: MOS 5.9=>12.5mos”
#3=IFN-y (this 4-3-17 AACR’17 one, #CT159/25)
The results of th the Biomarkers analysis may drive into the design of the 3 planned NCCN human trials and the AZN Bavi+Durva ‘Mult. Solid Tumors’ trial, the design of which is currently “under evaluation”. See http://tinyurl.com/jbv3ms5
- - - - - - -
***Maybe Dr. Ranee Mehra (Johns Hopkins, PPHM’s NCCN Bavi+Keytruda Head&Neck Ph2 P.I.) will stop by!
...Dr. Mehra’s work with Biomarker IFN-y seems to dovetail into PPHM’s newly revealed Sunrise Biomarker #3 to be presented 4-3-17 at AACR’17: #CT159/25, “IFN-y Analysis In Blood & Tissue as a Potential Prognostic and/or Predictive Biomarker” ( http://tinyurl.com/zaz525l ).

Note: DR. RANEE MEHRA was co-author of ASCO’16, “Biomarkers & Response to Pembro(Keytruda) in Recurrent/Metastatic Head & Neck Cancer” - Conclusion: “The IFN-y signature score was significantly associated with ORR, PFS, and OS (all, P< .001)… PD-L2 & IFN-y signature may be associated with clinical response to Pembro[Keytruda] and may offer addl. strategies to improve prediction of response.” http://meetinglibrary.asco.org/content/165708-176
MORE: http://tinyurl.com/h8gzkww

Great find, JDM: 96 Sunrise Pts w/Subsequent Immunotherapy, per 10-13-16 ASM. You just don’t get Stat.Sig. as in the 2 cases from today’s AACR #1651 PR unless you’ve got a decent number of Pts in the comparison.
JDM 4-4-17 #293751: “Fyi per pg.14 of the 2016 ASM presentation, the number of subsequent immunotherapy patients was 96 (16% of total 597) and per the OS Forest plot, bavi + D was better than control. Overall hazard ratio for both groups was .844. They didn't break out the groups for HR. So assuming basically even split, approx 48 patients each arm. Enough to get some stat significance. And given the immunotherapy option was prob US based, better chance of follow up IMO.”

---------------------
FROM PPHM’s 4-4-17 PR: http://ir.peregrineinc.com/releasedetail.cfm?ReleaseID=1020046
…”SUNRISE Data Analysis Demonstrates Stat. Significant Overall Survival (OS) Improvement in Patients Receiving Bavituximab+Docetaxel and Subsequent Immunotherapy Compared to Placebo+Docetaxel and Subsequent Immunotherapy… AACR’17 presentation (#CT159/25) of results of a new analysis of the Phase III SUNRISE trial… Data demonstrated that for patients in the study's BAVI+DOCE treatment arm who received subsequent immunotherapy, the mOS was not reached, while mOS was 13.0mos. for patients in the study's DOCE+PLACEBO arm who received subsequent immunotherapy [HR=0.43; p=.005]. These are the first clinical results reported supporting the hypothesis that bavituximab may modulate the tumor microenvironment to enhance the anti-tumor activity of immunotherapy agents... The presentation highlighted an analysis in which the company evaluated the impact of subsequent immunotherapy treatment, as well as patients' pre-treatment interferon gamma (IFN-y) levels on overall survival. Overall, low peripheral IFN-y correlated with more favorable OS in the patients receiving BAVI+DOCE and is a biomarker of interest. Data were also analyzed by low vs. high IFN-y levels. For patients with low pre-treatment IFN-y levels who received subsequent immunotherapy, those in the BAVI+DOCE arm did not reach mOS compared to mOS of 12.1mos. for the DOCE+PLACEBO arm [HR=0.24; p<.001].
Joseph Shan, Peregrine’s VP of Clinical & Regulatory Affairs:
"We are extremely encouraged by the results of these exploratory analyses which provide further clinical rationale for combining bavituximab and checkpoint inhibitors. This will be the key focus for upcoming early phase clinical trials, which includes a study of bavituximab and pembrolizumab in head and neck cancer through our ongoing collaboration with the NCCN.”
...Bavituximab is an investigational chimeric monoclonal antibody that targets phosphatidylserine (PS). Signals from PS inhibit the ability of immune cells to recognize and fight tumors. PS-targeting antibodies have demonstrated an ability to shift the functions of immune cells in tumors, resulting in multiple signs of immune activation and anti-tumor immune responses. Bavituximab is believed to override PS immunosuppressive signaling by blocking the engagement of PS with its receptors and sending an alternate immune activating signal.
---------
AACR’17 Attendee North40000 comments (iHub #293687): “I was told in response to a question I asked yesterday that PPHM does not know precisely how many patients in the Sunrise treatment arm are still alive.. Those pts are all over the world, and those who continued on maint. therapy remain uncounted. Best guess/ball park, as many as 6-12 pts in treatment arm who continue on maint. are still alive. After all, PPHM does continue to ship Bavi for use in that maint. Therapy. That surprised me that PPHM could not say how many are still alive. One would think that the CROs & PI’s would know. I made my views clear to at least 4-5 PPHM personnel on the convention floor yesterday that it would be useful to publish an accurate count as possible, with an "as of xx date " time attached. I was told that at least some of the above patients are back at work.”
I didn't have a bit of trouble understanding it, and I'm not the smartest knife in the drawer...
"SUNRISE Data Analysis Demonstrates Stat. Significant Overall Survival (OS) Improvement in Patients Receiving BAVI+DOCE and Subsequent Immunotherapy..."
I do admit I know what "MOS has not yet been reached" means.
4-4-17/PR: http://investorshub.advfn.com/boards/read_msg.aspx?message_id=130151723
WOW: 2 Stat. Sig. Improvements when Bavi added. “mOS was not reached” in both cases where the BAVI arm "received subsequent immunotherapy"...
FROM PPHM’s 4-4-17 PR: http://ir.peregrineinc.com/releasedetail.cfm?ReleaseID=1020046
…”SUNRISE Data Analysis Demonstrates Stat. Significant Overall Survival (OS) Improvement in Patients Receiving Bavituximab+Docetaxel and Subsequent Immunotherapy Compared to Placebo+Docetaxel and Subsequent Immunotherapy… AACR’17 presentation (#CT159/25) of results of a new analysis of the Phase III SUNRISE trial… Data demonstrated that for patients in the study's BAVI+DOCE treatment arm who received subsequent immunotherapy, the mOS was not reached, while mOS was 13.0mos. for patients in the study's DOCE+PLACEBO arm who received subsequent immunotherapy [HR=0.43; p=.005]. These are the first clinical results reported supporting the hypothesis that bavituximab may modulate the tumor microenvironment to enhance the anti-tumor activity of immunotherapy agents... The presentation highlighted an analysis in which the company evaluated the impact of subsequent immunotherapy treatment, as well as patients' pre-treatment interferon gamma (IFN-y) levels on overall survival. Overall, low peripheral IFN-y correlated with more favorable OS in the patients receiving BAVI+DOCE and is a biomarker of interest. Data were also analyzed by low versus high IFN-y levels. For patients with low pre-treatment IFN-y levels who received subsequent immunotherapy, those in the BAVI+DOCE arm did not reach mOS compared to mOS of 12.1mos. for the DOCE+PLACEBO arm [HR=0.24; p<.001].
Joseph Shan, Peregrine’s VP of Clinical & Regulatory Affairs:
"We are extremely encouraged by the results of these exploratory analyses which provide further clinical rationale for combining bavituximab and checkpoint inhibitors. This will be the key focus for upcoming early phase clinical trials, which includes a study of bavituximab and pembrolizumab in head and neck cancer through our ongoing collaboration with the NCCN.”
...Bavituximab is an investigational chimeric monoclonal antibody that targets phosphatidylserine (PS). Signals from PS inhibit the ability of immune cells to recognize and fight tumors. PS-targeting antibodies have demonstrated an ability to shift the functions of immune cells in tumors, resulting in multiple signs of immune activation and anti-tumor immune responses. Bavituximab is believed to override PS immunosuppressive signaling by blocking the engagement of PS with its receptors and sending an alternate immune activating signal.
Jeff4, I'm not sure exactly what you're saying, but if Nasdaq approves the extension, it will be for another 6mos (180 days), starting from 4-10-17, so roughly 10-10-17, though PL said "up to 180 days" in the 3-13-17 CC..
CFO Paul Lytle, 3-13-17 CC:
"First & foremost, we have determined that it is not in the best interest of our stockholders to affect a reverse stock split prior to April 10, 2017. If our share price does not trade above $1 for 10 consecutive trading days by April 10th, we expect to receive a notice of delisting. If we received this notice, we will appeal this decision within the required 7 days and this appeal will stay any delisting actions by NASDAQ. At that point, NASDAQ will schedule a hearing, which is typically held within 45 days, and we will present our plan to regain compliance. We are considering several paths to regain compliance, including having addl. time to continue to allow the market to adjust, to reflect the current value of Avid as evidence with the recent movement in our share price. The hearing panel could afford us up to 180 addl. days to regain compliance, although this addl. time is not guaranteed and this decision is at the discretion of the NASDAQ’s hearing panel." http://tinyurl.com/grhwjvy
Thx N40K for Pic of PPHM+MSK/WolchokLab ”ACT” #1651
AACR’17 4-3-17/8am #1651, MSKCC+PPHM: “PSTargeting+Adoptive TCell Transfer (ACT) Eliminates Adv. Tumors w/o Off-Target Toxicities, Melanoma” <=NEW(2nd) Joint MemSloan/PPHM study
MORE: http://tinyurl.com/momrheh

T517, the 1pmCT one is the new 3rd_Sunrise_Biomarker_IFN-y
#5. PPHM+VANDERBILT+PRECISION MEDICINE+PROVIDENCE CC:
4-3-17/1pm #CT159/25 (Session: Ph2/3 Clinical Trials in Progress), “IFN-y Analysis In Blood & Tissue as a Potential Prognostic and/or Predictive Biomarker” (abstract embargoed, Sunrise Biomarker #3)
= = = = = = =THIS A NEW (3RD) KNOWN SUNRISE BIOMAKER ANALYSIS:
AACR’17 4-3-17/1pm #CT159/25 - Session: Phase III Clinical Trials & Phase II/III Clinical Trials in Progress
http://www.abstractsonline.com/pp8/#!/4292/presentation/12566
”IFN-y Analysis In Blood & Tissue as a Potential Prognostic and/or Predictive Biomarker”
=> Nikoletta Kallinteris 1, Leora Horn 2, Min Tang 1, Tobias Guennel 3, Shen Yin 1, Jennifer Lai 1, Joseph Shan 1, Rachel E. Sanborn (Providence CC) 4
1=Peregrine Pharmaceuticals
2=Vanderbilt-Ingram Cancer Center, Nashville, TN
3=Precision for Medicine, Frederick, MD
4=Providence Cancer Center, Portland, OR
Rachel Sanborn MD, Dir./Thoracic-Oncology, Providence CC http://cancergrace.org/faculty/rachel-sanborn-md - Dr. Sanborn's Conflicts of interest: DNA, AZN
ABSTRACT: Embargoed – assuming until 4/3/17.
-------
This AACR’17 #5 is certainly the 3rd Sunrise Biomarker.
Known Ph.3 Sunrise Biomarkers/UTSW’s Dr. David Gerber et al:
#1=B2GPI: 10-10-16 http://tinyurl.com/hp73njt
#2=Complement & IL-10 Pathways(12-7-17/IASLC delayed, “not done”, after prelim. abstract said, “Pts=193, within the 1st subgroup of N=104, a 2nd subgroup isolated: MOS 5.9=>12.5mos”
#3=IFN-y (this 4-3-17 AACR’17 one, #CT159/25)
The results of th the Biomarkers analysis may drive into the design of the 3 planned NCCN human trials and the AZN Bavi+Durva ‘Mult. Solid Tumors’ trial, the design of which is currently “under evaluation”. See http://tinyurl.com/jbv3ms5
- - - - - - -
***Maybe Dr. Ranee Mehra (Johns Hopkins, PPHM’s NCCN Bavi+Keytruda Head&Neck Ph2 P.I.) will stop by!
...Dr. Mehra’s work with Biomarker IFN-y seems to dovetail into PPHM’s newly revealed Sunrise Biomarker #3 to be presented 4-3-17 at AACR’17: #CT159/25, “IFN-y Analysis In Blood & Tissue as a Potential Prognostic and/or Predictive Biomarker” ( http://tinyurl.com/zaz525l ).

Note: DR. RANEE MEHRA was co-author of ASCO’16, “Biomarkers & Response to Pembro(Keytruda) in Recurrent/Metastatic Head & Neck Cancer” - Conclusion: “The IFN-y signature score was significantly associated with ORR, PFS, and OS (all, P< .001)… PD-L2 & IFN-y signature may be associated with clinical response to Pembro[Keytruda] and may offer addl. strategies to improve prediction of response.” http://meetinglibrary.asco.org/content/165708-176
MORE: http://tinyurl.com/h8gzkww


Added PR Notes to AACR’17/#1651 PPHM+MSK “Bavi+ACT/CAR T”
4-3-17/8am #1651 - Session: TUMOR MICROENVIRONMENT & CHECKPOINTS [Joint PPHM & Memorial Sloan Kettering]
“Targeting Phosphatidylserine in Combination with Adoptive T Cell Transfer Eliminates Advanced Tumors without Off-Target Toxicities in a Melanoma Preclinical Model” <=NEW(2nd) MSK STUDY
=> Daniel Hirschhorn-Cymerman 1, Sara Sara Schad 1, Sadna Budhu 1, Zhong Hong 1, Xia Yang 1, Hutchins T. Jeff 2, Bruce D. Freimark 2, Michael J. Gray 2, Jedd Wolchok 1, Taha Merghoub [Memorial Sloan Kettering]
1=Memorial Sloan Kettering CC, NYC [ http://www.mskcc.org/research-areas/labs/jedd-wolchok ]
2=Peregrine Pharmaceuticals
ABSTRACT:
A viable strategy to treat advanced cancers includes transferring of tumor-specific T cells. T cells that recognize tumor antigens can be expanded and reinvigorated ex-vivo. Furthermore, autologous T cells can be genetically modified to express anti-tumor T cell receptors [TCRs] or chimeric antigen receptors [CARs]. Although the potency and specificity of tumor-specific T cells can be manipulated ex-vivo, once re-infused into patients, the T cells are subjected to immunosuppressive mechanisms established by the tumor. An important immune checkpoint regulator within tumors is phosphatidylserine (PS). Innate immune cells exposed to PS secrete suppressive cytokines and chemokines that can significantly impair the function and activation of anti-tumor T cells. Therefore, monoclonal antibodies that block PS activity can increase the anti-tumor potency of transferred T cells to treat aggressive cancers. Here we show that a PS targeting monoclonal antibody in combination with CD4+ T cells that recognize the melanoma antigen Trp1 can regress very advanced melanomas in all treated mice. Combination of anti-Trp1 CD4+ T cells with other immunomodulatory modalities such as anti-OX40 antibodies, can achieve equivalent treatment rates but these are typically accompanied by severe immune related adverse events. In contrast, in this setting, PS blockade did not show any off-target toxicities. Flow cytometry analysis revealed lower levels of CD206 expression concomitant with higher activation markers in macrophages and neutrophils in tumors from anti-PS treated mice. These results suggest that diminishing suppressive mechanisms locally in adoptive transfer protocols is a highly desirable strategy that can eliminate tumors while minimizing related adverse events.
---------------------
FROM PPHM’s 4-3-17 PR: http://ir.peregrineinc.com/releasedetail.cfm?ReleaseID=1019762
…”Latest Findings from Ongoing Collaboration with Memorial Sloan Kettering (MSK) Support Potential Applications for Combining CAR T & Anti-PS in Treatment of Solid Tumors”… For this study, a team of MSK researchers led by cancer immunotherapy thought-leaders, Taha Merghoub, Ph.D. and Jedd D. Wolchok, M.D., Ph.D., evaluated and compared the anti-tumor activity and off-target toxicities of adoptive T cell transfer therapy in combination with either PS-targeting antibodies or anti-OX40 antibodies in mice with advanced melanomas. Whereas PS-targeting and anti-OX40 demonstrated comparable tumor regression when administered in combination with transferred adoptive T cells, only the PS-targeting combination achieved these results without any off-target toxicities. By contrast, the anti-OX40 treatment combination triggered off-target inflammatory destruction of healthy tissues. Additional study results demonstrated that the PS-targeting antibodies decreased tumor-induced immunosuppression as evidenced by a decrease in immunosuppressive regulatory T cells (Tregs) and M2 macrophages. This finding is consistent with Peregrine’s belief that bavituximab may modulate the immunosuppressive tumor microenvironment and enhance the activity of immunotherapy agents.
Taha Merghoub, Ph.D., Co-Dir. of the Ludwig Collaborative Laboratory at MSK:
“While adoptive T cell transfer remains one of the most exciting new approaches to treating cancer, to date the toxicity associated with the treatment has limited its potential. We are encouraged that these study results showed that the combination of anti-PS and adoptive T cell treatment led to enhanced anti-tumor effect without any evidence of additional off-target side effects. “We believe that these findings may support potential applications for this combination in solid tumors in the future.”
Joseph Shan, Peregrine’s VP of Clinical & Regulatory Affairs:
“These study results provide further support for our belief that anti-PS agents such as bavituximab can play an important role as part of combination cancer treatments. This is directly tied to the agents’ ability to modulate the tumor microenvironment to combat the immunosuppression that limits the activity of CAR T and immunotherapies. Importantly, we are now also seeing evidence that this targeted modulation of the tumor microenvironment by anti-PS allows for enhanced activity of these other treatments without triggering any off-target toxicities. This is opposed to other conventional immunotherapies such as anti-OX40 with systemic mechanisms of action. We believe this advantageous tolerability profile will be a key benefit in positioning anti-PS agents for inclusion in optimal combination cancer regimens.”
---------------------
8-2015: Presently there are 3 types of Adoptive Cell Transfer (ACT) using effector T cells that are advancing on a path towards regulatory approval:
1. TILs (tumor infiltrating lymphocytes) have been developed with slow but continuing progress over several decades.
2. CARs (chimeric antibody receptors) – newer gene-modified T cells strategy
3. TCRs (T-cell receptors) – newer gene-modified T cells strategy
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4507286/

...“Adoptive T cell transfer is like ‘giving patients a living drug’,” states Renier J. Brentjens, MD/PhD, of Memorial Sloan Kettering Cancer Center (MSKCC).
… https://www.cancer.gov/about-cancer/treatment/research/car-t-cells
...“Adoptive T cell therapy (ACT) is one stone in this new pillar, a potentially powerful approach to cancer treatment that relies on the infusion of tumor-specific T cells.”
… https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4327320
= = = = = = = = =
Interesting 3-31-17 Sloan Kettering Tweet re: AACR’17
3-31-17 9:20amET “Getting ready for #AACR17? #Immunotherapy will be a hot topic. Learn more about how it works (video). @AACR”
https://twitter.com/sloan_kettering
Points to this 4-2016 Video (1:20):
“Immunotherapy – How It Works” https://www.youtube.com/watch?v=COQ1AeoGyco
Beg@:39:
“Immunotherapy drugs release this brake and empower immune cells to fight the cancer.
Sometimes the immune system needs a tune-up before it can fight cancer.
Immune cells can be removed from the body, armed with new proteins that can target cancer cells, and given back to a patient in large numbers.
Once inside the body, the modified immune cells recognize and attack the cancer.
This approach is called CAR T cell therapy. [an Adoptive Cell Transfer “ACT” strategy]
Despite the promise of immunotherapy, not everybody responds.
MSKCC scientists are exploring ways to improve immunotherapy.”

=============AND, THIS 3-23-17 PARKER FOUNDATION RELEASE ON AACR’17:
3-23-17: “Parker Institute for Cancer Immunotherapy Scientists to Present Research on Checkpoint Inhibitors, Adoptive Cell Therapy, and Other Advances in Immuno-oncology at AACR 2017”
Investigators affiliated with the Parker Institute for Cancer Immunotherapy will present some of the most anticipated immuno-oncology research at the 2017 AACR Annual Meeting. The event takes place at the Walter E. Washington Convention Center in Washington, DC, April 1-5 2017. . .
Other adoptive cell therapy abstracts of interest:
• ”Targeting Phosphatidylserine in Combination with Adoptive T Cell Transfer Eliminates Advanced Tumors without Off-Target Toxicities in a Melanoma Preclinical Model” [see http://tinyurl.com/nx5q5os ]
The principal investigator is Taha Merghoub, PhD, Parker Institute member researcher at Memorial Sloan Kettering Cancer Center. Co-authors include Parker Institute Center Director Jedd Wolchok, MD, PhD, at Memorial Sloan Kettering Cancer Center.
http://www.parkerici.org/media/2017/parker-institute-for-cancer-immunotherapy-scientists-to-present-research-on
----------ACT NOTES:
NOTE1:
From 3-2014 Immunotherapy article: “Cancer immunotherapy, particularly adoptive cell transfer (ACT), has shown great promise in the treatment of patients with late-stage disease, including those who are refractory to std. Therapies” https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4372895
NOTE2:
The Parker Foundation: “The Immunotherapy Dream Team”
Through the support from SEAN PARKER, Stand Up To Cancer (SU2C) and the Cancer Research Institute (CRI) formed an Immunology Research Dream Team dedicated to cancer immunology: “Immunologic Checkpoint Blockade & Adoptive Cell Transfer in Cancer Therapy.” The Immunotherapy Dream Team is focused on 2 approaches for this translational cancer research project, which will unite laboratory & clinical efforts towards the immunological treatment, control, and prevention of cancer.
The 1st is investigating blockades (by inhibitory molecules called “checkpoints”) of T lymphocytes’ inhibitory receptors, which block immune responses; and the use of antibodies to remove the checkpoints, once again allowing white blood cells called T lymphocytes to kill the cancer cells.
Second, the Immunotherapy Dream Team is pursuing multiple Adoptive Cell Transfer (ACT) approaches, which increase immunity.”
DREAM TEAM LEADERSHIP: 10 scientists, incl. MSKCC’s Jedd D. Wolchok & Michel Sadelain.
http://parker.org/initiatives/immunotherapy
EVENTS Updated: more on AACR'17, esp. 4-3-17 #1651(PPHM+MSKCC "ACT") and added ASCO'17 Booth#.
Known Upcoming Events (incl. AACR’17 w/new MSK/Wolchok”ACT” & IFNy-Biomarker)
Apr1-5: AACR 2017, WashDC http://tinyurl.com/mxz23gz
SUMMARY of PPHM’s 5 AACR’17 ABSTRACTS: [See DETAILS below]
1. MSKCC+PPHM: 4-2-17/1pm #574, “PSTargeting+RAD+AntiPD1 Promotes Anti-Tumor Activity, Melanoma” (same as SITC'16/11-14-16 http://tinyurl.com/js3fca4 )
2. MSKCC+PPHM: 4-3-17/8am #1651, “PSTargeting+Adoptive TCell Transfer (ACT) Eliminates Adv. Tumors w/o Off-Target Toxicities, Melanoma” <=NEW(2nd) Joint MemSloan/PPHM study
3. PPHM: 4-4-17/8am #3652, “PSTargeting+LAG3+AntiPD1 Significantly Enhances Anti-Tumor Activity, Triple- MBC”
4. IMMUNOVACCINE+PPHM: 4-4-17/8am #3657, ”PSTargeting Enhances Anti-Tumor Activity of a Tumor Vaccine(DepoVax), HPV-Induced Tumor” https://www.imvaccine.com See 3-24-17:
5. PPHM+VANDERBILT+PRECISIONMEDICINE+PROVIDENCECC: 4-3-17/1pm #CT159/25 (Session: Ph2/3 Clinical Trials in Progress), “IFN-y Analysis In Blood & Tissue as a Potential Prognostic and/or Predictive Biomarker” (abstract embargoed, Sunrise Biomarker #3) - See below.
ALSO: Peregrine Exhibiting – booth #3312.
Jun2-6: ASCO 2017, Chicago https://am.asco.org (Abstracts: Titles/MidAPR, Full=May17)
...Peregrine will be presenting at ASCO’17, per CEO Steve King, 3-13-17 CC Q&A.
...PPHM Exhibiting – booth #13124 (next to Moffitt CC)
Jun19-22/Avid Booth #1411: BIO Intl. Convention, SanDiego http://www.convention.bio.org/2017
~Jul13: FY'17Q4 (fye 4-30-17) Financials & Conf. Call - http://ir.peregrineinc.com/events.cfm
.
.
.
======================AACR’17 DETAILS:
Apr1-5 2017: “AACR 2017”, WashDC
5 PPHM ABSTRACTS: 2/MemSloan, 1/Immunovaccine, 1/PPHM-Only. There’s also a 5th one (PPHM, Vanderbilt, Precision-for-Medicine, Providence CC) in the “Ph2/3 Clinical Trials” session, whose Abstract is still embargoed: “CT159/25: IFN-y Analysis in Blood & Tissue as a Potential Prognostic and/or Predictive Biomarker” – this is our new (3rd) Sunrise Biomaker analysis - see below.
...Add ImmunoVaccine Inc. (Halifax https://www.imvaccine.com ) to the list of collaborators.
...#1651(Apr3) is the newly revealed 2nd joint PPHM+Mem.Sloan/Wolchok preclin. study: “PS Targeting + Adoptive T Cell Transfer Eliminates Advanced Tumors without Off-Target Toxicities in a Melanoma Model”. IMO, the Wolchok Lab is saying in #1651 that the effectiveness of either type of Adoptive TCell Transfer (ACT), be it TCRs or CAR T (ie, the newer gene-modified T cells approaches), is improved by combining with Bavi and makes for a “highly desirable strategy.” - see more below.
...Memorial Sloan’s Dr. Jedd. Wolchok is co-author of 9 AACR’17 abstracts. 2 of the 9 are w/PPHM re: PS-Targeting. With biotechs, he has one each with Genocea Biosciences(ex-vivo screening), Infinity Pharm(Ph1), Leap Ther.(Ph1), and two with BMS(Ph1+Ph3). The 8th is MSKCC ONLY, and the 9th is jointly w/Cedars-Sinai/LA. http://www.abstractsonline.com/pp8/#!/4292 .
--------
AACR’17 HOME: http://www.aacr.org/Meetings/Pages/MeetingDetail.aspx?EventItemID=105
...Abstracts: http://www.abstractsonline.com/pp8/#!/4292
DETAILS OF 5 PPHM ABSTRACTS...
= = = = = = = = = = = = = = = = = = =
1. 4-2-17/1pm #574 - Session: CHECKPOINTS 1 [Joint PPHM & Memorial Sloan Kettering]
“Phosphatidylserine Targeting Antibody in Combination with Tumor Radiation & Immune Checkpoint Blockade Promotes Anti-Tumor Activity in Mouse B16 Melanoma”
=> Sadna Budhu 1, Olivier De Henau 1, Roberta Zappasodi 1, Rachel Giese 1, Luis F. Campesato 1, Christopher Barker 1, Bruce Freimark 2, Jeff Hutchins 2, Jedd D. Wolchok 1, Taha Merghoub [Memorial Sloan Kettering]
1=Memorial Sloan Kettering CC, NYC [ http://www.mskcc.org/research-areas/labs/jedd-wolchok ]
2=Peregrine Pharmaceuticals
NOTE: SAME TITLE AS WAS PRESENTED BY Mem.Sloan 11-14-16 SITC’16: see http://tinyurl.com/js3fca4
ABSTRACT:
Phosphatidylserine (PS) is a phospholipid that is exposed on surface of apoptotic cells, tumor cells and tumor endothelium. PS has been shown to promote immunosuppressive signals in the tumor microenvironment. Antibodies that target PS have been shown to reactivate anti-tumor immunity by polarizing tumor associated macrophages into a pro-inflammatory M1 phenotype, reducing the number of MDSCs in tumors and promoting the maturation of dendritic cells into functional APCs. In a mouse B16 melanoma model, targeting PS in combination with immune checkpoint blockade promoted greater anti-tumor activity than either agent alone. This combination was shown to enhance CD4+ and CD8+ T cell infiltration and activation in the tumors of treated animals. Radiation therapy (RT) is an effective focal treatment of primary solid tumors, but is less effective in treating metastatic solid tumors as a monotherapy. There is evidence that RT induces immunogenic tumor cell death and enhances tumor-specific T cell infiltration in treated tumors. The abscopal effect, a phenomenon in which tumor regression occurs outside the site of RT, has been observed in both preclinical and clinical trials when RT is combined with immunotherapy. In this study, we show that irradiation treatment of B16 melanoma causes an increase in PS expression on the surface of viable tumor and immune infiltrates. We subsequently examined the effects of combining RT with an antibody that targets PS (mch1N11) [“Mouse version of Bavituximab”] and immune checkpoint blockade (anti-PD-1) in B16 melanoma. We found that treatment with mch1N11 synergizes with RT to improve anti-tumor activity and overall survival in tumor bearing mice. In addition, the triple combination of mch1N11, RT and anti-PD-1 treatment displayed even greater anti-tumor and survival benefit. Analysis of local immune responses in the tumors of treated animals revealed an increase in tumor-associated macrophages with a shift towards a pro-inflammatory M1 phenotype after treatment with RT & mch1N11. In addition, analysis of the systemic immune responses in the spleen and tumor draining lymph nodes revealed an increase in CD8 T cell activation, effector cytokine production and differentiation into effector memory cells in the triple combination. This finding highlights the potential of combining these 3 agents to improve outcome in patients with advanced-stage melanoma and other cancers and may inform the design of clinical studies combining PS-targeting antibodies with RT and/or checkpoint blockade.
= = = = = = = = = = = = = = = =
2. 4-3-17/8am #1651 - Session: TUMOR MICROENVIRONMENT & CHECKPOINTS [Joint PPHM & Memorial Sloan Kettering]
“Targeting Phosphatidylserine in Combination with Adoptive T Cell Transfer Eliminates Advanced Tumors without Off-Target Toxicities in a Melanoma Preclinical Model” <=NEW(2nd) MSK STUDY
=> Daniel Hirschhorn-Cymerman 1, Sara Sara Schad 1, Sadna Budhu 1, Zhong Hong 1, Xia Yang 1, Hutchins T. Jeff 2, Bruce D. Freimark 2, Michael J. Gray 2, Jedd Wolchok 1, Taha Merghoub [Memorial Sloan Kettering]
1=Memorial Sloan Kettering CC, NYC [ http://www.mskcc.org/research-areas/labs/jedd-wolchok ]
2=Peregrine Pharmaceuticals
ABSTRACT:
A viable strategy to treat advanced cancers includes transferring of tumor-specific T cells. T cells that recognize tumor antigens can be expanded and reinvigorated ex-vivo. Furthermore, autologous T cells can be genetically modified to express anti-tumor T cell receptors [TCRs] or chimeric antigen receptors [CARs]. Although the potency and specificity of tumor-specific T cells can be manipulated ex-vivo, once re-infused into patients, the T cells are subjected to immunosuppressive mechanisms established by the tumor. An important immune checkpoint regulator within tumors is phosphatidylserine (PS). Innate immune cells exposed to PS secrete suppressive cytokines and chemokines that can significantly impair the function and activation of anti-tumor T cells. Therefore, monoclonal antibodies that block PS activity can increase the anti-tumor potency of transferred T cells to treat aggressive cancers. Here we show that a PS targeting monoclonal antibody in combination with CD4+ T cells that recognize the melanoma antigen Trp1 can regress very advanced melanomas in all treated mice. Combination of anti-Trp1 CD4+ T cells with other immunomodulatory modalities such as anti-OX40 antibodies, can achieve equivalent treatment rates but these are typically accompanied by severe immune related adverse events. In contrast, in this setting, PS blockade did not show any off-target toxicities. Flow cytometry analysis revealed lower levels of CD206 expression concomitant with higher activation markers in macrophages and neutrophils in tumors from anti-PS treated mice. These results suggest that diminishing suppressive mechanisms locally in adoptive transfer protocols is a highly desirable strategy that can eliminate tumors while minimizing related adverse events.
---------------------
8-2015: Presently there are 3 types of Adoptive Cell Transfer (ACT) using effector T cells that are advancing on a path towards regulatory approval:
1. TILs (tumor infiltrating lymphocytes) have been developed with slow but continuing progress over several decades.
2. CARs (chimeric antibody receptors) – newer gene-modified T cells strategy
3. TCRs (T-cell receptors) – newer gene-modified T cells strategy
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4507286/

...“Adoptive T cell transfer is like ‘giving patients a living drug’,” states Renier J. Brentjens, MD/PhD, of Memorial Sloan Kettering Cancer Center (MSKCC).
… https://www.cancer.gov/about-cancer/treatment/research/car-t-cells
...“Adoptive T cell therapy (ACT) is one stone in this new pillar, a potentially powerful approach to cancer treatment that relies on the infusion of tumor-specific T cells.”
… https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4327320
= = = = = = = = =
Interesting 3-31-17 Sloan Kettering Tweet re: AACR’17
3-31-17 9:20amET “Getting ready for #AACR17? #Immunotherapy will be a hot topic. Learn more about how it works (video). @AACR”
https://twitter.com/sloan_kettering
Points to this 4-2016 Video (1:20):
“Immunotherapy – How It Works” https://www.youtube.com/watch?v=COQ1AeoGyco
Beg@:39:
“Immunotherapy drugs release this brake and empower immune cells to fight the cancer.
Sometimes the immune system needs a tune-up before it can fight cancer.
Immune cells can be removed from the body, armed with new proteins that can target cancer cells, and given back to a patient in large numbers.
Once inside the body, the modified immune cells recognize and attack the cancer.
This approach is called CAR T cell therapy. [an Adoptive Cell Transfer “ACT” strategy]
Despite the promise of immunotherapy, not everybody responds.
MSKCC scientists are exploring ways to improve immunotherapy.”

=============AND, THIS 3-23-17 PARKER FOUNDATION RELEASE ON AACR’17:
3-23-17: “Parker Institute for Cancer Immunotherapy Scientists to Present Research on Checkpoint Inhibitors, Adoptive Cell Therapy, and Other Advances in Immuno-oncology at AACR 2017”
Investigators affiliated with the Parker Institute for Cancer Immunotherapy will present some of the most anticipated immuno-oncology research at the 2017 AACR Annual Meeting. The event takes place at the Walter E. Washington Convention Center in Washington, DC, April 1-5 2017. . .
Other adoptive cell therapy abstracts of interest:
• ”Targeting Phosphatidylserine in Combination with Adoptive T Cell Transfer Eliminates Advanced Tumors without Off-Target Toxicities in a Melanoma Preclinical Model” [see http://tinyurl.com/nx5q5os ]
The principal investigator is Taha Merghoub, PhD, Parker Institute member researcher at Memorial Sloan Kettering Cancer Center. Co-authors include Parker Institute Center Director Jedd Wolchok, MD, PhD, at Memorial Sloan Kettering Cancer Center.
http://www.parkerici.org/media/2017/parker-institute-for-cancer-immunotherapy-scientists-to-present-research-on
----------ACT NOTES:
NOTE1:
From 3-2014 Immunotherapy article: “Cancer immunotherapy, particularly adoptive cell transfer (ACT), has shown great promise in the treatment of patients with late-stage disease, including those who are refractory to std. Therapies” https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4372895
NOTE2:
The Parker Foundation: “The Immunotherapy Dream Team”
Through the support from SEAN PARKER, Stand Up To Cancer (SU2C) and the Cancer Research Institute (CRI) formed an Immunology Research Dream Team dedicated to cancer immunology: “Immunologic Checkpoint Blockade & Adoptive Cell Transfer in Cancer Therapy.” The Immunotherapy Dream Team is focused on 2 approaches for this translational cancer research project, which will unite laboratory & clinical efforts towards the immunological treatment, control, and prevention of cancer.
The 1st is investigating blockades (by inhibitory molecules called “checkpoints”) of T lymphocytes’ inhibitory receptors, which block immune responses; and the use of antibodies to remove the checkpoints, once again allowing white blood cells called T lymphocytes to kill the cancer cells.
Second, the Immunotherapy Dream Team is pursuing multiple Adoptive Cell Transfer (ACT) approaches, which increase immunity.”
DREAM TEAM LEADERSHIP: 10 scientists, incl. MSKCC’s Jedd D. Wolchok & Michel Sadelain.
http://parker.org/initiatives/immunotherapy
= = = = = = = = = = = = = = = =
3. 4-4-17/8am #3652 - Session: BITES BISPECIFICS & CHECKPOINTS
“Combinational Activity of LAG3 & PD-1 Targeted Therapies is Significantly Enhanced by the Addition of Phosphatidylserine Targeting Antibodies and Establishes an Anti-Tumor Memory Response in Murine Triple Negative Breast Cancer”
=> Michael J. Gray, Jian Gong, Jeff Hutchins, Bruce Freimark (Peregrine Pharmaceuticals)
ABSTRACT:
Previous studies utilizing NanoString immune profile analysis demonstrated that intratumoral levels of LAG3 (lymphocyte activation gene 3) mRNA increased in response to phosphatidylserine (PS) and PD-1 targeting antibodies in murine triple negative breast cancers (TNBC). This suggests LAG3 acts to attenuate immune system activation during I/O therapies - and that PD-1 and LAG3 function cooperatively in suppressing immune system activation. Here we show that adding PS targeting antibodies can further enhance the effectiveness of antibodies targeting LAG3 and/or LAG3+PD-1. We first examined expression of LAG3 and PD-1 in the murine TNBC model E0771 and found that tumor associated T-cells (CD4+ and CD8+) have expression of both markers. Mice implanted with TNBC tumors were next treated with antibodies targeting PS, PD-1, and LAG3 alone and in combination with each other. Interestingly, the addition of PS targeting antibodies not only increased the effectiveness anti-PD-1 effectiveness as previously observed, but also enhanced anti-LAG3 treatment, showing that PS targeting antibodies are capable of augmenting additional I/O therapeutic regimens. Comparison of anti-PD-1+LAG3 combination vs. single anti-PD-1 or anti-LAG3 treatments showed moderately more anti-tumor activity than single treatments, however the addition of PS targeting antibodies to either checkpoint inhibitor was as equally effective in inhibiting tumor growth as observed in the anti-LAG3+PD-1 treatment. Further comparison of antibody treatments targeting PD-1+LAG3 vs. PS+PD-1+LAG3 demonstrated that the addition of PS targeting antibodies resulted in a significant decrease in tumor growth with complete tumor regression in 80% of the animals (along with the ability to completely reject secondary TNBC challenge) compared to 0% in the anti-PD-1+LAG3 treatment group. Immunoprofiling showed that the addition of PS targeting antibodies to these checkpoint therapies, including the combination of anti-PD-1+LAG3, resulted in a phenotype associated with enhanced immune system activation and immune-surveillance including increased tumor infiltrating lymphocytes (TILs) with upregulation of T-cell associated activation pathways, increased Th1 to Th2 profile, and enhanced antigen presentation processing /presentation mechanisms along with cytokines associated with immune system activation. Overall our data demonstrate that adding PS targeting antibodies to clinically relevant therapies, including PD-1 and LAG3, may significantly enhance their ability to activate and redirect the host immune system into recognition and elimination of tumor cells compared to single and combinational treatments that lack PS targeting antibodies.
= = = = = = = = = = = = = = = =
4. 4-4-17/8am #3657 - Session: BITES BISPECIFICS & CHECKPOINTS
”Phosphatidylserine-Targeting Antibodies Enhance Anti-Tumor Activity of a Tumor Vaccine in a HPV-Induced Tumor Model”
=> Genevieve Weir 1, Tara Quinton 1, Jeff T. Hutchins 2, Bruce D. Freimark 2, Marianne Stanford (VP/Res., Immunovaccine)
1=Immunovaccine, Inc., Halifax, NS, Canada [ https://www.imvaccine.com ]
2=Peregrine Pharmaceuticals
[Note: clearly, this study is combining PPHM’s Anti-PS with ImmunoVaccine’s DepoVax Vaccine Adjuvanting Platform https://www.imvaccine.com/depovax.php ]
ABSTRACT:
Antibodies targeting phosphatidylserine (PS) have been shown to induce anti-tumor responses by induction of tumor-specific T cells. Based on this observation, we evaluated the responses of PS and PD-1 targeting antibody therapy to enhance anti-tumor responses of a HPV16 peptide vaccine formulated in DepoVax (DPX) in mice bearing HPV-transformed C3 mouse tumors. The addition of PS-targeting antibody (mch1N11) [“Mouse version of Bavituximab”] to DPX/metronomic cyclophosphamide (mCPA) immunotherapy prolonged survival in comparison to mice receiving an isotype control in combination with DPX/mCPA. When anti-PD-1 was added to mch1N11 + mCPA, there was no increase in survival. The addition of mch1N11 to DPX/mCPA immunotherapy had no effect on tumor growth or survival in the aggressive B16-F10 model. TIL analysis revealed an increase in CD8+ T cells, antigen specific CD8+ T cells and PD-1+ T cells in the tumor with mch1N11 treatment. The expression of surface markers for macrophages (CD68high, F4/80) and dendritic cells (CD11c) were also increased in the tumors of mice treated with mch1N11. RT-qPCR analysis of the tumor confirmed higher mRNA expression of T cells markers (CD8, Granzyme B, PD-1) and antigen presenting cell markers (F4/80, CD74). In the spleen, expression of cell surface markers for monocytes (CD11b) and PD-1+ T cells (CD8) were elevated in groups treated with mch1N11 in combination with anti-PD-1. Combined, these findings indicate that in this model, PS-targeting antibodies can enhance the activity of phagocytic cells involved in antigen presentation. We have found that PD-1 expression increases as anti-tumor activity increases, therefore these results also provide an indication that antibodies targeting PS enhance the anti-tumor immune response induced by DPX/mCPA therapy. The observations suggest that PS-targeting antibodies may enhance therapeutic vaccines for the treatment of cancer.
-------
3-24-17 Immunovaccine PR: “Immunovaccine to Present New Preclin. Combo Therapy Data at AACR’17” …Immunovaccine conducted this research in collaboration with Peregrine Pharm. with the goal of analyzing the potential enhanced anti-cancer activity of combining DepoVax-based cancer immunotherapies with Peregrine's lead clinical product candidate bavituximab… Immunovaccine Dir./Res. Genevieve Weir will present data analyzing the potential for enhanced anti-tumor responses of PS & PD-1 targeting antibody therapies when combined with an HPV16 peptide vaccine formulated in Immunovaccine's proprietary DepoVax technology. "Our work with Peregrine, and the research it has produced, fits squarely into our corporate objective of exploring clinical stage immunotherapies that may have synergistic effects when combined with our lead candidate, DPX-Survivac," said Marianne Stanford (PhD) VP/Res. for Immunovaccine. "We look forward to discussing the results of this study, and its potential implications, with our colleagues in the scientific community at this year's AACR meeting." https://www.imvaccine.com/releases.php?releases_id=418
...Ahhhhh, ImmunoVaccine & Dr. Wolchok – zero doubt IMO how & why PPHM & Immunovaccine have hooked up...
ImmunoVaccine & Jedd Wolchok go back to ASCO’13:
6-3-13: “Our poster (Ph1 data) is one of few selected for detailed discussion at ASCO by Dr. Jedd Wolchok, top cancer immunotherapy thought leader… Immunovaccine Inc. believes that these immune responses are consistent in profile to those necessary from a cancer vaccine to potentially impact disease progression. These study results were further discussed by Dr. Jedd Wolchok of Memorial Sloan-Kettering CC, a top thought leader in the area of cancer immunotherapy, at the poster discussion session that followed.”
https://www.imvaccine.com/communications.php?communications_id=11
= = = = = = =THIS A NEW (3RD) KNOWN SUNRISE BIOMAKER ANALYSIS:
#5. 4-3-17/1pm #CT159/25 - Session: Phase III Clinical Trials & Phase II/III Clinical Trials in Progress
http://www.abstractsonline.com/pp8/#!/4292/presentation/12566
”IFN-y Analysis In Blood & Tissue as a Potential Prognostic and/or Predictive Biomarker”
=> Nikoletta Kallinteris 1, Leora Horn 2, Min Tang 1, Tobias Guennel 3, Shen Yin 1, Jennifer Lai 1, Joseph Shan 1, Rachel E. Sanborn (Providence CC)*** 4
1=Peregrine Pharmaceuticals
2=Vanderbilt-Ingram Cancer Center, Nashville, TN
3=Precision for Medicine, Frederick, MD
4=Providence Cancer Center, Portland, OR
***Rachel Sanborn MD, Dir./Thoracic-Oncology, Providence CC http://cancergrace.org/faculty/rachel-sanborn-md - Dr. Sanborn's Conflicts of interest: DNA, AZN
ABSTRACT: Embargoed – assuming until 4/3/17.
-------
This AACR’17 #5 is certainly the 3rd Sunrise Biomarker.
Known Ph.3 Sunrise Biomarkers/UTSW’s Dr. David Gerber et al:
#1=B2GPI: 10-10-16 http://tinyurl.com/hp73njt
#2=Complement & IL-10 Pathways(12-7-17/IASLC delayed, “not done”, after prelim. abstract said, “Pts=193, within the 1st subgroup of N=104, a 2nd subgroup isolated: MOS 5.9=>12.5mos”
#3=IFN-y (this 4-3-17 AACR’17 one, #CT159/25)
The results of th the Biomarkers analysis may drive into the design of the 3 planned NCCN human trials and the AZN Bavi+Durva ‘Mult. Solid Tumors’ trial, the design of which is currently “under evaluation”. See http://tinyurl.com/jbv3ms5
- - - - - - -
***Maybe Dr. Ranee Mehra (Johns Hopkins, PPHM’s NCCN Bavi+Keytruda Head&Neck Ph2 P.I.) will stop by!
...Dr. Mehra’s work with Biomarker IFN-y seems to dovetail into PPHM’s newly revealed Sunrise Biomarker #3 to be presented 4-3-17 at AACR’17: #CT159/25, “IFN-y Analysis In Blood & Tissue as a Potential Prognostic and/or Predictive Biomarker” ( http://tinyurl.com/zaz525l ).

Note: DR. RANEE MEHRA was co-author of ASCO’16, “Biomarkers & Response to Pembro(Keytruda) in Recurrent/Metastatic Head & Neck Cancer” - Conclusion: “The IFN-y signature score was significantly associated with ORR, PFS, and OS (all, P< .001)… PD-L2 & IFN-y signature may be associated with clinical response to Pembro[Keytruda] and may offer addl. strategies to improve prediction of response.” http://meetinglibrary.asco.org/content/165708-176
MORE: http://tinyurl.com/h8gzkww


= = = = = = = = =
INTERESTING: Mem. Sloan’s Dr. Jedd. Wolchok is co-author of 9 AACR’17 abstracts. 2 of the 9 are w/PPHM re: PS-Targeting. With biotechs, he has one each with Genocea Biosciences(ex-vivo screening), Infinity Pharm(Ph1), Leap Ther.(Ph1), and two with BMS(Ph1+Ph3). The 8th is MSKCC ONLY, and the 9th is jointly w/Cedars-Sinai/LA. http://www.abstractsonline.com/pp8/#!/4292 .
PEREGRINE (Joint Mem.Sloan Kettering Wolchok Lab & PPHM):
1. 4-2-17 #574 “Phosphatidylserine Targeting Antibody in Combination with Tumor Radiation & Immune Checkpoint Blockade Promotes Anti-Tumor Activity in Mouse B16 Melanoma” <=Same as SITC’16 11-14-16.
4-3-17 #1651 “Targeting Phosphatidylserine in Combination With Adoptive T Cell Transfer Eliminates Advanced Tumors Without Off-Target Toxicities in a Melanoma Preclin. Model” <=NEW/2nd PPHM+MSK.
NON-PEREGRINE:
3. (MSK ONLY): 4-2-17 #874 “Lifting the iron curtain: Imaging cellular barriers to combination chelation-immune checkpoint therapy”
4. (MSK & Genocea Bio. http://www.genocea.com GNCA/$130mm MktCap) 4-2-17 #632 “Genome-scale neoantigen screening using ATLAS prioritizes candidate antigens for immunotherapy in a NSCLC patient”
5. (MSK & Cedars-Sinai/LA) 4-4-17 #4705 “CTLA4 blockade with HER2-directed therapy yields clinical benefit in women undergoing radiation therapy for HER2+ breast cancer brain metastases”
6. (MSK & Infinity Pharm.) 4-4-17 #CT089 “A Ph1, first-in-human study of IPI-549, a PI3K-y inhibitor, as monotherapy & in combo w/nivolumab in adv. solid tumors”
7. (MSK & Leap Ther.) 4-2-17 #CT018 “Intratumor & peripheral Treg modulation as a pharmacodynamic biomarker of the GITR agonist antibody TRX-518 in the 1st in-human trial”
8. (MSK & BMS) 4-3-17 #CT073 “(Ph1) Immunomodulatory effects of NIVO+IPI or NIVO/mono in adv. melanoma: CheckMate038”
9. (MSK & BMS) 4-3-17 #CT075 “OS results from a Ph3 of NIVO+IPI in treatment-naïve pts w/adv. Melanoma: CheckMate067”

= = = = = = = = = = = = =
2-28-17: Collabs with Mem.Sloan(Wolchok), Duke, MDA, Rutgers, ImmunoVaccine, UTSW… http://tinyurl.com/heg9t3v
BAVI MOA 11-14-16: SITC’16: Joint Memorial Sloan Kettering (Wolchok Lab) & PPHM poster on Triple Combo Rad+Bavi+aPD1 vs. Melanoma http://tinyurl.com/js3fca4
“PS Targeting Antibody in Combination with Checkpoint Blockade & Tumor Radiation Therapy Promotes Anti-Cancer Activity in Mouse Melanoma” (same as AACR’17 4-2-17 #574)
DR. JEDD WOLCHOK: "Based on these study results, we believe that the targeting of PS is having meaningful activity within the tumor microenvironment in the B16 melanoma model. It appears that this activity creates a more immune active environment in which other treatments, including radiation, are able to have a greater anti-tumor impact."
DR. TAHA MERGHOUB (Co-Dir., Ludwig Collaborative Lab at MSK): "We have noted that the combination of PS-targeting treatment and radiation, as well as triple combination of PS-targeting treatment, radiation and anti-PD-1, resulted in clear advantages in anti-tumor activity in the mouse B16 melanoma model. We believe that these findings suggest the potential benefit of combining these agents to improve the outcomes of patients with cancer. With this in mind, we think this research may play an important role in designing future clinical trials of PS-targeting agents in melanoma and other cancers."
5-29-15: Peregrine & Sloan Kettering Enter Collab. to “Investigate Novel PS-Targeting Immunotherapy Combos” http://tinyurl.com/o3k9ux8
...Dr. Wolchock states, ”The phosphatidylserine (PS) signaling pathway is a very interesting target for modulating the immune system's response to cancer. We look forward to exploring the potential of PS-targeting agents alone and with other immune modulators that may lead to novel advances in cancer therapy.”
...Dr. Taha Merghoub states, "A key focus of the Wolchok Lab's research is studying novel immunotherapy combinations that work together to enable the immune system to recognize & destroy cancer. This collaboration will allow us to focus on the role & contribution of PS blockade therapy in determining which combination of the current & next gen. of immune modulators is likely to increase the extent & amplitude of anti-tumor response. This important pre-clinical and translational work will potentially guide the design of the next gen. of clinical studies with bavituximab.”
POSSIBLE GENESIS OF IMMUNOVACCINE COLLAB???
11-9-15 SITC'15: New Bavi+Checkpoint Inhibitors preclin. data (UTSW/DUKE's Herbert K. Lyerly) http://tinyurl.com/pbof95w
...Also, collab. with Dr. Bernard Fox (Immunotherapist/Earle A. Chiles Res.Inst.) on new Immuno-Profiling Clinical Test (Opal 6-plex quantitative IF Assay), PPHM roundtable with Raymond Birge (Rutgers), Douglas Graham (Emory), Dmitry Gabrilovich (Wistar), Rolf Brekken (UTSW), Maria Karasarides (AstraZeneca) - ”Combining Bavi w/anti-PD-1 significantly enhanced O/S… significant incr. CD45+, CD8+ and CD3+ T-cells… led a prolonged anti-tumor immune response which protected the animals against a re-challenge w/same tumor.”
SITC’16 PPHM/Mem.Sloan Poster – same as AACR’17 4-2-17 #574…

Chey, it's ASCO'17 where PPHM's Booth is next_to_Moffitt's
Jun2-6: “ASCO 2017”, Chicago https://am.asco.org
MID-APRIL: Release of Abstract Titles (Accessible via ASCO's iPlanner website)
MAY 17 (5:00pmET): Abstracts Released on abstracts.asco.org
PPHM Exhibiting – booth #13124 (next to Moffitt CC)
http://events.jspargo.com/ASCO17/Public/Exhibitors.aspx?ID=60467
...Peregrine will be presenting at ASCO’17, per CEO Steve King, 3-13-17 CC Q&A. http://tinyurl.com/grhwjvy
PW, I’ve learned a lot about ACT. I have no doubt that the Wolchok Lab is saying in #1651 that the effectiveness either type (the latest of Adoptive TCell Transfer (ACT), be it TCRs or CAR T (ie, the newer gene-modified T cells approaches), is improved by combining with Bavi, and makes for a “highly desirable strategy.”
ABSTRACT (AACR'17 4-3-17 #1651):
“PS Targeting+Adoptive TCell Transfer (ACT) Eliminates Adv. Tumors w/o Off-Target Toxicities, Melanoma”
A viable strategy to treat advanced cancers includes transferring of tumor-specific T cells. T cells that recognize tumor antigens can be expanded and reinvigorated ex-vivo. Furthermore, autologous T cells can be genetically modified to express anti-tumor T cell receptors [TCRs] or chimeric antigen receptors [CARs]. Although the potency and specificity of tumor-specific T cells can be manipulated ex-vivo, once re-infused into patients, the T cells are subjected to immunosuppressive mechanisms established by the tumor. An important immune checkpoint regulator within tumors is phosphatidylserine (PS). Innate immune cells exposed to PS secrete suppressive cytokines and chemokines that can significantly impair the function and activation of anti-tumor T cells. Therefore, monoclonal antibodies that block PS activity can increase the anti-tumor potency of transferred T cells to treat aggressive cancers. Here we show that a PS targeting monoclonal antibody in combination with CD4+ T cells that recognize the melanoma antigen Trp1 can regress very advanced melanomas in all treated mice. Combination of anti-Trp1 CD4+ T cells with other immunomodulatory modalities such as anti-OX40 antibodies, can achieve equivalent treatment rates but these are typically accompanied by severe immune related adverse events. In contrast, in this setting, PS blockade did not show any off-target toxicities. Flow cytometry analysis revealed lower levels of CD206 expression concomitant with higher activation markers in macrophages and neutrophils in tumors from anti-PS treated mice. These results suggest that diminishing suppressive mechanisms locally in adoptive transfer protocols is a highly desirable strategy that can eliminate tumors while minimizing related adverse events.
MORE: http://tinyurl.com/l69gbyf

Learning more about “ACT”, MSKCC+PPHM 4-3-17 AACR’17 #1651
Apr1-5: AACR 2017, WashDC http://tinyurl.com/mxz23gz - SUMMARY of PPHM’s 5 AACR’17 ABSTRACTS:
1. MSKCC+PPHM: 4-2-17/1pm #574, “PSTargeting+RAD+AntiPD1 Promotes Anti-Tumor Activity, Melanoma” (same as SITC'16/11-14-16 http://tinyurl.com/js3fca4 )
2. MSKCC+PPHM: 4-3-17/8am #1651, “PSTargeting+Adoptive TCell Transfer (ACT) Eliminates Adv. Tumors w/o Off-Target Toxicities, Melanoma” <=NEW(2nd) Joint MemSloan/PPHM study
3. PPHM: 4-4-17/8am #3652, “PSTargeting+LAG3+AntiPD1 Significantly Enhances Anti-Tumor Activity, Triple- MBC”
4. IMMUNOVACCINE+PPHM: 4-4-17/8am #3657, ”PSTargeting Enhances Anti-Tumor Activity of a Tumor Vaccine(DepoVax), HPV-Induced Tumor” https://www.imvaccine.com See 3-24-17:
5. PPHM+VANDERBILT+PRECISIONMEDICINE+PROVIDENCECC: 4-3-17/1pm #CT159/25 (Session: Ph2/3 Clinical Trials in Progress), “IFN-y Analysis In Blood & Tissue as a Potential Prognostic and/or Predictive Biomarker” (abstract embargoed, Sunrise Biomarker #3) - See below.
ALSO: Peregrine Exhibiting – booth #3312.
= = = = = = = = = = = = = = = =
AACR’17 PPHM#2: 4-3-17/8am #1651 - Session: TUMOR MICROENVIRONMENT & CHECKPOINTS
“Targeting Phosphatidylserine in Combination with Adoptive T Cell Transfer Eliminates Advanced Tumors without Off-Target Toxicities in a Melanoma Preclinical Model” <=NEW(2nd) MSK STUDY$$$
=> Daniel Hirschhorn-Cymerman 1, Sara Sara Schad 1, Sadna Budhu 1, Zhong Hong 1, Xia Yang 1, Hutchins T. Jeff 2, Bruce D. Freimark 2, Michael J. Gray 2, Jedd Wolchok 1, Taha Merghoub [Memorial Sloan Kettering]
1=Memorial Sloan Kettering CC, NYC [ http://www.mskcc.org/research-areas/labs/jedd-wolchok ]
2=Peregrine Pharmaceuticals
ABSTRACT:
A viable strategy to treat advanced cancers includes transferring of tumor-specific T cells. T cells that recognize tumor antigens can be expanded and reinvigorated ex-vivo. Furthermore, autologous T cells can be genetically modified to express anti-tumor T cell receptors [TCRs] or chimeric antigen receptors [CARs]. Although the potency and specificity of tumor-specific T cells can be manipulated ex-vivo, once re-infused into patients, the T cells are subjected to immunosuppressive mechanisms established by the tumor. An important immune checkpoint regulator within tumors is phosphatidylserine (PS). Innate immune cells exposed to PS secrete suppressive cytokines and chemokines that can significantly impair the function and activation of anti-tumor T cells. Therefore, monoclonal antibodies that block PS activity can increase the anti-tumor potency of transferred T cells to treat aggressive cancers. Here we show that a PS targeting monoclonal antibody in combination with CD4+ T cells that recognize the melanoma antigen Trp1 can regress very advanced melanomas in all treated mice. Combination of anti-Trp1 CD4+ T cells with other immunomodulatory modalities such as anti-OX40 antibodies, can achieve equivalent treatment rates but these are typically accompanied by severe immune related adverse events. In contrast, in this setting, PS blockadedid not show any off-target toxicities. Flow cytometry analysis revealed lower levels of CD206 expression concomitant with higher activation markers in macrophages and neutrophils in tumors from anti-PS treated mice. These results suggest that diminishing suppressive mechanisms locally in adoptive transfer protocols is a highly desirable strategy that can eliminate tumors while minimizing related adverse events.
----------
NOTE1:
From 3-2014 Immunotherapy article: “Cancer immunotherapy, particularly adoptive cell transfer (ACT), has shown great promise in the treatment of patients with late-stage disease, including those who are refractory to std. Therapies” https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4372895
NOTE2:
Through the support from SEAN PARKER, Stand Up To Cancer (SU2C) and the Cancer Research Institute (CRI) formed an Immunology Research Dream Team dedicated to cancer immunology: “Immunologic Checkpoint Blockade & Adoptive Cell Transfer in Cancer Therapy.” The Immunotherapy Dream Team is focused on 2 approaches for this translational cancer research project, which will unite laboratory & clinical efforts towards the immunological treatment, control, and prevention of cancer.
The 1st is investigating blockades (by inhibitory molecules called “checkpoints”) of T lymphocytes’ inhibitory receptors, which block immune responses; and the use of antibodies to remove the checkpoints, once again allowing white blood cells called T lymphocytes to kill the cancer cells.
Second, the Immunotherapy Dream Team is pursuing multiple Adoptive Cell Transfer (ACT) approaches, which increase immunity.”
DREAM TEAM LEADERSHIP: 10 scientists, incl. MSKCC’s Jedd D. Wolchok & Michel Sadelain.
http://parker.org/initiatives/immunotherapy
------------------------------
8-2015: Presently there are 3 types of Adoptive Cell Transfer (ACT) using effector T cells that are advancing on a path towards regulatory approval:
1. TILs (tumor infiltrating lymphocytes) have been developed with slow but continuing progress over several decades.
2. CARs (chimeric antibody receptors) – newer gene-modified T cells strategy
3. TCRs (T-cell receptors) – newer gene-modified T cells strategy
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4507286/

...“Adoptive cell transfer is like ‘giving patients a living drug’,” states Renier J. Brentjens, MD/PhD, of Memorial Sloan Kettering Cancer Center (MSKCC).
… https://www.cancer.gov/about-cancer/treatment/research/car-t-cells
...“Adoptive T cell therapy (ACT) is one stone in this new pillar, a potentially powerful approach to cancer treatment that relies on the infusion of tumor-specific T cells.”
… https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4327320
= = = = = = = = =
Interesting 3-31-17 Sloan Kettering Tweet re: AACR’17
3-31-17 9:20amET “Getting ready for #AACR17? #Immunotherapy will be a hot topic. Learn more about how it works (video). @AACR”
https://twitter.com/sloan_kettering
Points to this 4-2016 Video (1:20):
“Immunotherapy – How It Works” https://www.youtube.com/watch?v=COQ1AeoGyco
Beg@:39:
“Immunotherapy drugs release this brake and empower immune cells to fight the cancer.
Sometimes the immune system needs a tune-up before it can fight cancer.
Immune cells can be removed from the body, armed with new proteins that can target cancer cells, and given back to a patient in large numbers.
Once inside the body, the modified immune cells recognize and attack the cancer.
This approach is called CAR T cell therapy. [an Adoptive Cell Transfer “ACT” strategy]
Despite the promise of immunotherapy, not everybody responds.
MSKCC scientists are exploring ways to improve immunotherapy.”

=============AND, THIS 3-23-17 PARKER FOUNDATION RELEASE ON AACR’17:
3-23-17: “Parker Institute for Cancer Immunotherapy Scientists to Present Research on Checkpoint Inhibitors, Adoptive Cell Therapy, and Other Advances in Immuno-oncology at AACR 2017”
Investigators affiliated with the Parker Institute for Cancer Immunotherapy will present some of the most anticipated immuno-oncology research at the 2017 AACR Annual Meeting. The event takes place at the Walter E. Washington Convention Center in Washington, DC, April 1-5 2017. . .
Other adoptive cell therapy abstracts of interest:
• ”Targeting Phosphatidylserine in Combination with Adoptive T Cell Transfer Eliminates Advanced Tumors without Off-Target Toxicities in a Melanoma Preclinical Model” [see http://tinyurl.com/nx5q5os ]
The principal investigator is Taha Merghoub, PhD, Parker Institute member researcher at Memorial Sloan Kettering Cancer Center. Co-authors include Parker Institute Center Director Jedd Wolchok, MD, PhD, at Memorial Sloan Kettering Cancer Center.
http://www.parkerici.org/media/2017/parker-institute-for-cancer-immunotherapy-scientists-to-present-research-on
INTERESTING: Mem. Sloan’s Dr. Jedd. Wolchok is co-author of 9 AACR’17 abstracts. 2 of the 9 are w/PPHM re: PS-Targeting. With biotechs, he has one each with Genocea Biosciences(ex-vivo screening), Infinity Pharm(Ph1), Leap Ther.(Ph1), and two with BMS(Ph1+Ph3). The 8th is MSKCC ONLY, and the 9th is jointly w/Cedars-Sinai/LA. http://www.abstractsonline.com/pp8/#!/4292 .
PEREGRINE (Joint Mem.Sloan Kettering Wolchok Lab & PPHM):
1. 4-2-17 #574 “Phosphatidylserine Targeting Antibody in Combination with Tumor Radiation & Immune Checkpoint Blockade Promotes Anti-Tumor Activity in Mouse B16 Melanoma” <=Same as SITC’16 11-14-16.
4-3-17 #1651 “Targeting Phosphatidylserine in Combination With Adoptive T Cell Transfer Eliminates Advanced Tumors Without Off-Target Toxicities in a Melanoma Preclin. Model” <=NEW/2nd PPHM+MSK.
NON-PEREGRINE:
3. (MSK ONLY): 4-2-17 #874 “Lifting the iron curtain: Imaging cellular barriers to combination chelation-immune checkpoint therapy”
4. (MSK & Genocea Bio. http://www.genocea.com GNCA/$130mm MktCap) 4-2-17 #632 “Genome-scale neoantigen screening using ATLAS prioritizes candidate antigens for immunotherapy in a NSCLC patient”
5. (MSK & Cedars-Sinai/LA) 4-4-17 #4705 “CTLA4 blockade with HER2-directed therapy yields clinical benefit in women undergoing radiation therapy for HER2+ breast cancer brain metastases”
6. (MSK & Infinity Pharm.) 4-4-17 #CT089 “A Ph1, first-in-human study of IPI-549, a PI3K-y inhibitor, as monotherapy & in combo w/nivolumab in adv. solid tumors”
7. (MSK & Leap Ther.) 4-2-17 #CT018 “Intratumor & peripheral Treg modulation as a pharmacodynamic biomarker of the GITR agonist antibody TRX-518 in the 1st in-human trial”
8. (MSK & BMS) 4-3-17 #CT073 “(Ph1) Immunomodulatory effects of NIVO+IPI or NIVO/mono in adv. melanoma: CheckMate038”
9. (MSK & BMS) 4-3-17 #CT075 “OS results from a Ph3 of NIVO+IPI in treatment-naïve pts w/adv. Melanoma: CheckMate067”