Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
I was wrong. I only hold a small amount of warrants with them. GLTA
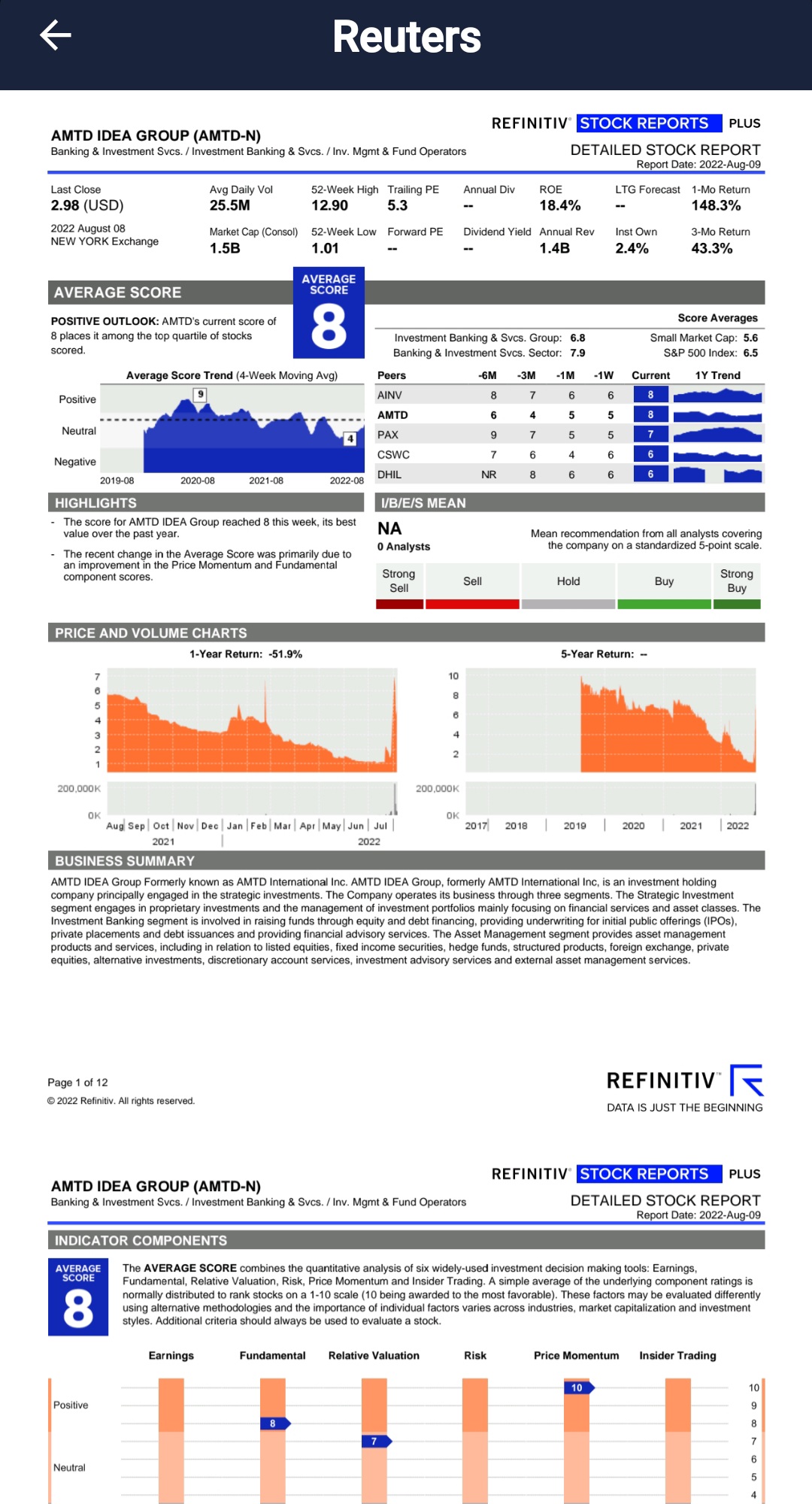
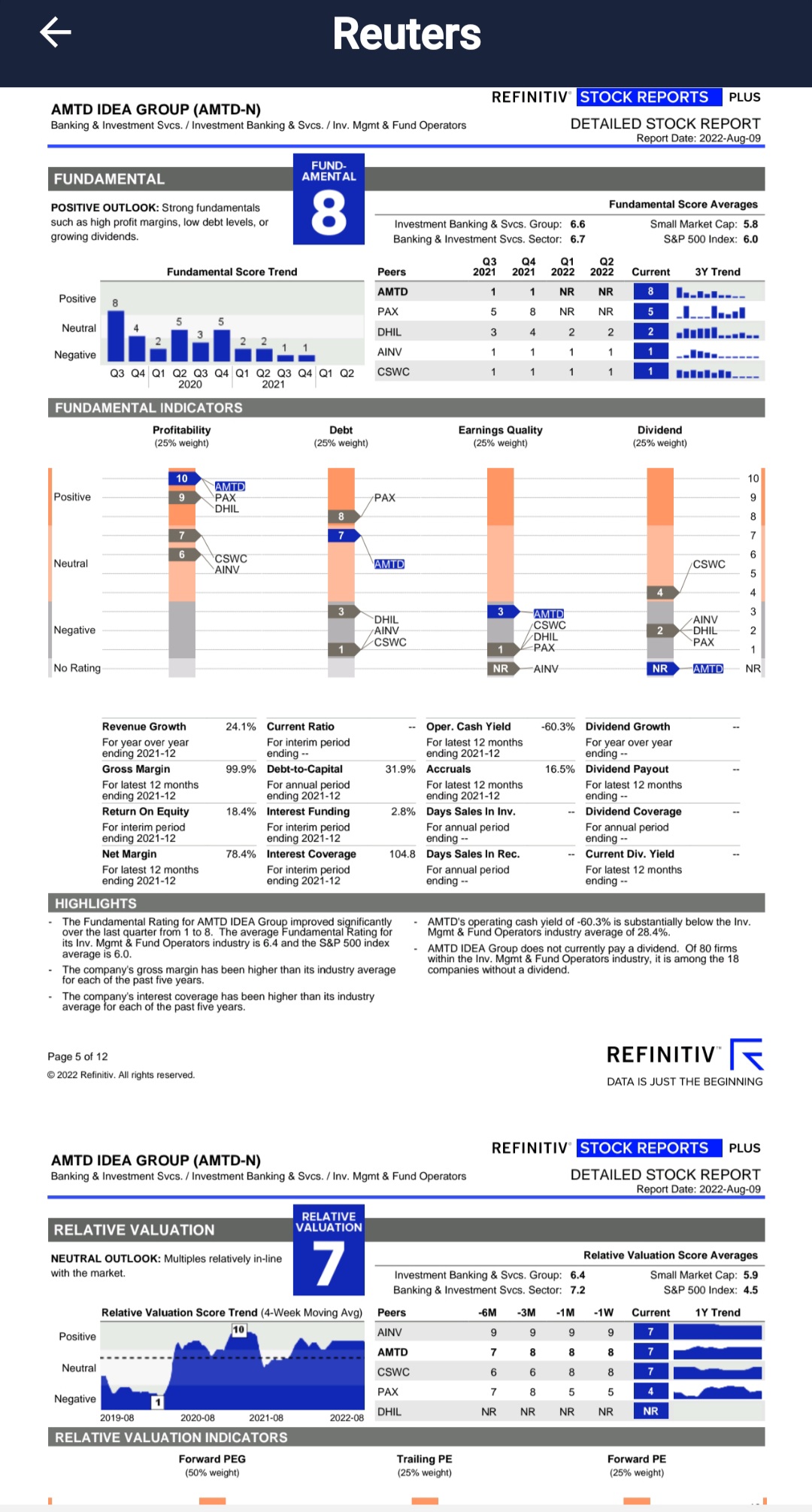
Thomson Reuters recently upgraded AMTD in Etrade Analyst Research section on Aug 9th Report

Morgan Stanley has over a million shares unless they sold them during the spike.
https://fintel.io/so/us/amtd
I thought that alone would send this to $30+ with current share structure. Imagine the devastation, heartache and financial burden an Ebola outbreak would cause to a major metropolitan area. GeoVax Labs Inc. has a solution. $GOVX
Impact of Ebola on the Economy
Aside from the devastating health effects, the Ebola epidemic also had a pronounced socio-economic impact in Guinea, Liberia, and Sierra Leone.
According to 2014 projections from the World Bank, an estimated $2.2 billion was lost in 2015 in the gross domestic product (GDP) of the three countries.
The outbreak resulted in lower investment and a substantial loss in—
Private sector growthAgricultural production leading to concerns about food securityCross-border trade as restrictions on movement, goods, and services increased
https://www.cdc.gov/vhf/ebola/history/2014-2016-outbreak/cost-of-ebola.html
Took a loss here. Sold at $1.67 Median price target of $15+ Will try and get back in if i can catch part of a positive wave. They are suppose to give an update soon on when they will release earnings. Good Luck!
$GOVX
As I reported in a previous article, the company’s CEO, David Dodd, wrote in May that the firm’s “vaccines have been validated and registered for [the] prevention of Monkeypox virus infection.” And, in the same post, Dodd specified that one of the company’s shots may stop humans from contracting monkeypox.
Elaborating in June, the CEO reported that: “We already have published in peer-reviewed journals that our MVA vaccines, in addition [to] protecting animal models against HIV and other infections, also prevents monkeypox,” He vowed that his company would “address the monkeypox virus.”
https://investorplace.com/2022/07/why-is-geovax-govx-stock-up-90-today/
https://investorplace.com/author/larryramerdos/
Bought in during AH. 560 shares @2.03
GLTA
Borrow Rate over 600% with zero shares available to short.
If this gets volume and doesn't go up, then there is nothing right, left in the stock market.
https://fintel.io/ss/us/usea
The Company has 8,862,004 Common Shares issued and outstanding as of the date hereof, reflecting the exercise of certain of the Pre-Funded Warrants issued in the Offering.
https://sec.report/Document/0001140361-22-026528/#ny20004194x16_6k.htm
Healthy Volume returns to ENSC. Time to rebuild support over $2. Lots of boxes for the company to check along with steps to complete in getting greater value for investors and institutions. ENSC has to regain Nasdaq Compliance. Upcoming Trial results must be positively reported without allowing any room for a financial adviser/broker like Morgan Stanley to tear apart the data and downgrade the Stock. Looking forward to how we will forge profitable partnerships with drug manufacturers using our proprietary prodrug technologies: abuse resistant TAAP™ and overdose resistant MPAR™
June 23 2022 Annual Virtual Stockholders Meeting 9am PT
Register online 15 minutes prior to webcast
https://agm.issuerdirect.com/ensc
Proxy Statement
https://sec.report/Document/0001493152-22-011736/
May 2, 2022
Dear Fellow Stockholder:
On behalf of the Board of Directors and management of Ensysce Biosciences, Inc., I cordially invite you to attend our annual meeting of stockholders on Thursday, June 23, 2022 at 9:00 a.m. (Pacific Time). The 2022 Annual Meeting will be a virtual meeting of stockholders. You will be able to attend the 2022 Annual Meeting, vote your shares electronically and submit your questions during the meeting via live webcast by visiting https://agm.issuerdirect.com/ensc. Stockholders will be able to listen, vote, and submit questions from their home or any location with internet connectivity. To participate in the meeting, you must have the 16-digit number that is shown on your Notice of Internet Availability of Proxy Materials or on your proxy card if you elected to receive proxy materials by mail. The notice of meeting and proxy statement that follow describe the business that we will consider at the meeting.
We hope that you will be able to attend the meeting via our live webcast. However, regardless of whether you attend the meeting, your vote is very important. We are pleased to offer multiple options for voting your shares. You may vote via the internet, by mail or through our live webcast of the Annual Meeting, as described beginning on page two of the proxy statement.
Thank you for your continued support of Ensysce Biosciences, Inc.
Sincerely, /s/ Dr. Lynn Kirkpatrick Dr. Lynn Kirkpatrick President and Chief Executive Officer
ENSC Chairman Bob G. Gower PhD (original founder and largest shareholder) said this in an interview with Southern Illinois University Foundation reporter Rebecca Renshaw recently.
One might think that with all of the success he achieved in his life, Gower would sit back and take it easy. Not so.
One of his current passions is the field of nanotechnology, which he has used to help develop a therapeutic, abuse-resistant opioid pain drug.
“It is currently in clinical trials, and the FDA recently gave Ensysce Biosciences a fast-track designation, which means we won’t have such a lengthy process to approval,” he said.
Gower said that opioid drugs are the only way to handle certain types of severe pain, so simply banning them from use is not the answer.
“The problem is that people quickly become addicted to opioid drugs,” he said. “We need to prevent abuse. What we have done with our drug is to modify it so that the drug is not active when taken. Only when it is in the digestive system and it meets an enzyme known as trypsin does it become activated via a two-step reaction. If a person snorts it or dissolves it and injects the drug, it will not be activated. If chewed, the reaction still will not take place until it gets to the digestive system.
https://foreversiu.org/news/bob-gower.php
CEO Lynn Kirkpatrick purchased shares, 70000 @ $0.4869 on May 25th and 27000 @$0.5265 on May 26th.
https://ih.advfn.com/stock-market/NASDAQ/ensysce-biosciences-ENSC/stock-news/88234139/statement-of-changes-in-beneficial-ownership-4
$ENSC
BlackRock Inc. reports 41,654.01% increase in ownership of ENSC / Ensysce Biosciences, Inc.
2022-05-12 - BlackRock Inc. has filed a 13F-HR form disclosing ownership of 234,240 shares of Ensysce Biosciences, Inc. (US:ENSC) with total holdings valued at $267,000 USD as of 2022-03-31. BlackRock Inc. had filed a previous 13F-HR on 2022-02-10 disclosing 561 shares of Ensysce Biosciences, Inc. at a value of $3,000 USD. This represents a change in shares of 41,654.01 percent and a change in value of 8,800.00 percent during the quarter.
https://fintel.io/so/us/ensc/blackrock
This stock will reach $1 plus again without needing a RS. Only 35 Million Outstanding. No doubt the company is receiving offers from companies like Maxim Group LLC to help them get back over a dollar for a price... Hope they are smart and avoid the shenanigans from the ones who shorted/gapped this down. IMO. GLTA Longs
If Ensysce achieves its mission and keeps the share structure low and stable, this will be a 3 figure stock IMO. If they fail, it'll likely accept toxic financing and balloon the outstanding shares and destroy shareholder value. Only time will tell. The world needs a painkiller that won't kill them. Traders/investors need a stock worth investing in.
June 17th Event.
On June 17, 2022, the Company received a notice in the form of a letter (“Deficiency Letter”) from the Listing Qualifications Staff of the Nasdaq stating that the Company was not in compliance with Nasdaq Listing Rule 5550(a)(2) because the bid price for the Company’s common stock had closed below $1.00 per share for the previous 30 consecutive business days.
https://ih.advfn.com/stock-market/NASDAQ/ensysce-biosciences-ENSC/stock-news/88398039/current-report-filing-8-k
$ENSC
$ENSC Underappreciated Stock With Big Potential #opioidaddiction #opioidabuse - https://t.co/VGJjPxtb6X pic.twitter.com/ssBVqQwGkT
— Zacks SCR (@ZacksSmallCap) March 29, 2022
If that prophecy does pan out, this land is worth 75 Billion.
Stock trading over 7 when Gold was in 1700s
SPAC opened at 10 i believe, gotta verify that.
44 Billion in Paydirt for a company with a market cap of 114 Million, and that's just the territory we know... I'm willing to bet there might be some Palladium and Platinum in that location waiting to be found.
I think it is Darkpool shorting at the moment... difficult to say. We need this to be the end of consolidation at these rock bottom levels.
HYMC Venezuela Gold lowest in 50 years
https://www.reuters.com/world/americas/gold-reserves-venezuelan-central-bank-down-new-50-year-low-2022-02-15/
Production: Gold production for the year ended December 31, 2021, of 55,668 ounces exceeded the high end of the guidance range as the process team continued to improve equipment, process control and costs. Silver production of 355,967 ounces
https://ih.advfn.com/stock-market/NASDAQ/hycroft-mining-HYMC/stock-news/87348872/hycroft-provides-preliminary-2021-operating-result
There’s gold in them thar hills
Math
600,000,000 oz silver x $25 oz spot silver = $15 billion... and I believe silver may double this year
15,000,000 oz gold x $1920 oz spot gold today = $28.8 billion... and I think we will go over $2300 oz this year
Wonder how much they process/produce every quarter and how they plan on improving production numbers...
Waiting to see what current outstanding shares are to get an accurate Market Cap estimate
AMC is playing on offense again with a bold diversification move. We just purchased 22% of Hycroft Mining (NASDAQ: HYMC) of northern Nevada. It has 15 million ounces of gold resources! And 600 million ounces of silver resources! Our expertise to help them bolster their liquidity. pic.twitter.com/LihqZguwnd
— Adam Aron (@CEOAdam) March 15, 2022
I believe it could double and go past $50 oz
The all time record silver price was hit on April 28, 2011 with Silver spot prices exceeding $49.50 oz intraday.
https://sdbullion.com/silver-prices-2011
Spot Gold
https://www.kitco.com/charts/livegold.html
https://goldprice.org/live-gold-price.html
Spot Silver
https://www.kitco.com/charts/livesilver.html
undervalued IMO
I'm wishing for over $40. I'm expecting $10-25.
And I'm hoping for $25-35. I don't believe the 148 million in share volume today was all retail.
I disagree 100 percent... A parent who has a child with cancer will seek a FDA approved cure/treatment for their child. What fund doesn't want a pediatric saving drug in their portfolio for their current and future shareholders. Shortbots have been latched to this stock all day. Its only a short matter of time before this goes double digits... Form 8-K still hasn't hit newswires yet.
FORM 8-K
https://www.immixbio.com/sec-filings/
Item 8.01 Other Events.
On January 3, 2022, Immix Biopharma, Inc. announced that the U.S. Food and Drug Administration (FDA) has granted Rare Pediatric Disease (RPD) designation for IMX-110 for the treatment of a life-threatening form of pediatric cancer in children, rhabdomyosarcoma. IMX-110, an investigational product, is currently being evaluated in a Phase 1b/2a clinical trial. A copy of the press release is attached hereto as Exhibit 99.1 and is incorporated herein by reference.
Item 9.01 Financial Statements and Exhibits.
(d) Exhibits.
Exhibit No. Description99.1 Press release dated January 3, 2022
Exhibit 99.1
U.S. Food and Drug Administration Approves Immix Biopharma Rare Pediatric Disease Designation for IMX-110 as a Treatment for Life-Threatening Pediatric Cancer in Children
Rare Pediatric Disease Designation (“RPDD”) qualifies Immix Biopharma to receive fast track review, and a priority review voucher (“PRV”) at the time of marketing approval of IMX-110.
PRV holders can benefit from an expedited six-month review of a new drug application for any disease by the FDA.
While their future value is uncertain, PRVs are transferable to other companies and have historically sold for $67 to $350 million according to a January 2020 report on drug development by the Government Accountability Office.
LOS ANGELES, January 3, 2021 (GLOBE NEWSWIRE) — Immix Biopharma, Inc. (Nasdaq: IMMX) (“ImmixBio” or the “Company”), a biopharmaceutical company pioneering Tissue-Specific Therapeutics (TSTx)TM targeting oncology and immuno-dysregulated diseases, announced today that the U.S. Food and Drug Administration (FDA) has granted Rare Pediatric Disease (RPD) designation for IMX-110 for the treatment of a life-threatening form of pediatric cancer in children, rhabdomyosarcoma. IMX-110, an investigational product, is currently being evaluated in a Phase 1b/2a clinical trial.
The FDA grants Rare Pediatric Disease designation for serious and life-threatening diseases that primarily affect children aged 18 years or younger and impact fewer than 200,000 people in the United States.
If a New Drug Application in the United States for IMX-110 is approved, ImmixBio may be eligible to receive a Priority Review Voucher (PRV) from the FDA, which can be redeemed to obtain priority review for any subsequent marketing application, or may be sold or transferred.
“We are pleased by FDA’s acknowledgment of the urgent need for a safe and effective treatment for children with this devastating disease,” stated ImmixBio’s Chief Executive Officer Ilya Rachman, M.D., PhD. “We are encouraged by our Phase 1b/2a clinical data in soft tissue sarcoma. IMX-110 is a tissue-specific therapeutic that simultaneously attacks all 3 components of the tumor micro-environment, severing the critical lifelines between the tumor and its metabolic and structural support. We believe our SMARxT platform generating Tissue-Specific Therapeutics represents a distinct alternative to the traditional ‘single target, single mutation’ development model.”
Rhabdomyosarcoma (“RMS”) is a high-grade, malignant neoplasm, the most common soft tissue sarcoma in pediatric and adolescent populations and which rarely occurs in adults. The prevalence of RMS in the United States is approximately 20,000 children of all ages. The five-year survival rate ranges from 20% to 30% for children in the high-risk group where cancer spreads widely in the body.
IMX-110 is the first clinical-stage product of ImmixBio’s SMARxT Tissue-Specific™ Platform, which produces Tissue-Specific Therapeutics that accumulate at intended therapeutic sites at 3 to 5 times the rate of conventional medicines. The FDA has already granted orphan drug designation (ODD) to IMX-110 for the treatment of soft tissue sarcoma.
ImmixBio recently shared clinical data across multiple soft tissue sarcoma subtypes in several heavily pretreated patients demonstrating median progression-free survival (PFS) of 4 months with zero drug-related severe adverse events and zero dose interruptions due to toxicity. The data can be viewed in the Immix Biopharma Corporate Presentation at http://www.immixbio.com/pres
Dr. Ilya Rachman IMMX CEO gave a brilliant response to the Benzinga interviewer who may have a short interest based on his questions... CEO stated they don't need to raise cash for the next year which would mean no public offering... Happy New Year!
CEO interview around 12:45pm East Coast Time today according to IMMX Twitter
https://mobile.twitter.com/immixbio
$IMMX
Could see a big move similar to LGVN
GLTA