Making $$ in the Market
Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
CEO Tepper @ YouTube Interview
November 2016
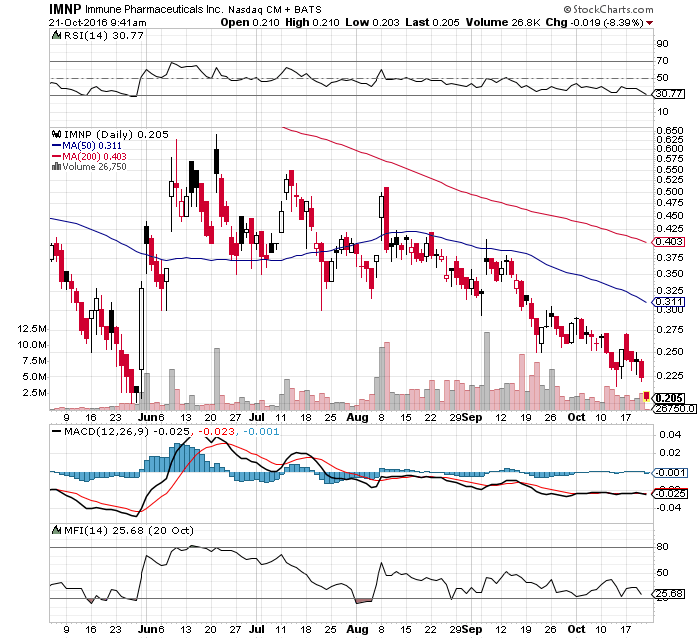
Explosive AH @ IMNP
.27 / up 6.33% / 548K shares traded
Santa delivering some good news...hmmm
Investor 100
Recap PR @ IMNP
Immune Pharmaceuticals Secures Up to $11 Million in New Financing to be Deployed With a Focus on Bertilimumab Clinical Development
November 17th, 2016
http://finance.yahoo.com/news/immune-pharmaceuticals-secures-11-million-141500951.html
NEW YORK, Nov. 17, 2016 /PRNewswire/ -- Immune Pharmaceuticals (IMNP) (Immune) announced today that it has secured up to $11 million in new financing from a single family office that is an existing investor. The financing includes the sale of a $1 million convertible note and up to $10 million in future equity sales at prevailing market prices in increments of at least $1 million per month.
Dr. Daniel Teper, Immune's CEO commented: "We appreciate the continuous commitment of our investors to support this financing of Immune, which will be dedicated to the advancement of bertilimumab clinical development. We are also implementing a strategy to unlock the value of other assets through the establishment of new, privately-funded companies which will be partially owned by Immune."
Immune continues to accrue patients in two phase 2 clinical trials for bertilimumab, a first-in-class monoclonal antibody targeting eotaxin-1: an open label 10 patient trial in the treatment of bullous pemphigoid, an orphan auto-immune dermatological condition, and a double-blind, placebo-controlled, 42 patient trial in moderate-to-severe ulcerative colitis, a gastrointestinal inflammatory condition. In both conditions, a significant number of patients have elevated levels of eotaxin-1, which is also considered a biomarker of disease severity. Targeting patients with elevated eotaxin-1 offers the potential to select patients who may be more likely to respond to treatment. Additionally in 2017, Immune plans to initiate a phase 2 trial in severe atopic dermatitis, a condition where eotaxin–1 plays an important role, and to explore the role of eotaxin-1 in patients with nonalcoholic steatohepatitis (NASH).
Immune is also moving forward with forming three private companies separate from the development of bertilimumab, focusing on Immuno-Oncology, Dermatology, and Pain & Neurology respectively. The Immuno-Oncology subsidiary will focus on Ceplene, which recently received guidance from the FDA for a pivotal phase 3 Overall Survival study in remission maintenance for Acute Myeloid Leukemia, and novel bispecific antibodies targeting immune check points and other novel targets. The Dermatology subsidiary will focus on the application of nanotechnology to dermatology. The lead program is a topical cyclosporine for the treatment of psoriasis and atopic dermatitis. The Pain and Neurology subsidiary will focus on AmiKet® for the treatment of peripheral neuropathic pain with the subsidiary's new management looking at complementary assets.
About Immune Pharmaceuticals Inc.:
Immune Pharmaceuticals Inc. (IMNP) applies a personalized approach to treating and developing novel, highly targeted antibody therapeutics to improve the lives of patients with inflammatory diseases and cancer. Immune's lead product candidate, bertilimumab, is in Phase II clinical development for moderate-to-severe ulcerative colitis as well as for bullous pemphigoid, an orphan autoimmune dermatological condition. Other indications being considered for development include atopic dermatitis, Crohn's disease, severe asthma and Non-Alcoholic Steato-Hepatitis (NASH), an inflammatory liver disease. Immune recently expanded its portfolio in immuno-dermatology with topical nano-formulated cyclosporine-A for the treatment of psoriasis and atopic dermatitis. Immune's oncology pipeline includes Ceplene® which is in late stage clinical development for maintenance remission in Acute Myeloid Leukemia (AML) in combination with IL-2. Additional oncology pipeline includes Azixa® and crolibulin, Phase II clinical stage vascular disrupting agents, and novel technology platforms; bispecific antibodies and NanomAbs™. Maxim Pharmaceuticals Inc., Immune's pain and neurology subsidiary is developing AmiKet™ and AmiKet™ Nano™ for the treatment of neuropathic pain. For more information, visit Immune's website at www.immunepharma.com, the content of which is not a part of this press release.
Insider Monkey @ IMNP
The companies in focus are Immune Pharmaceuticals Inc (NASDAQ:IMNP), AEterna Zentaris Inc. (USA) (NASDAQ:AEZS) and Biodel Inc (NASDAQ:BIOD).
October 28,2016.
http://www.insidermonkey.com/blog/here-are-three-big-biotech-movers-and-our-take-on-whats-next-483757/?yptr=yahoo
Immune Pharmaceuticals Inc (NASDAQ:IMNP) announced yesterday that it had received feedback from the FDA concerning its lead asset program, an investigation of its drug Ceplene in combination with low dose IL-2 for the maintenance of remission in patients with Acute Myeloid Leukemia (AML).
This one’s an interesting one. Basically, the announcement was negative, but markets have regarded it as extremely positive. Well, negative is probably a little strong. It’s a step forward in the sense that the company knows what it needs to do to put together a protocol for the FDA that will get accepted, and once accepted, it’s got a clear path to a pivotal initiation. However, that the agency has come back with some recommendations and requirements suggests that the initial proposed trial had some deficiencies, and at core, this is a delay. Delays cost money, and at this end of the biotech space anything that costs more money that initially expected can be crippling.
We don’t expect the company to falter too much, and the phase III will almost certainly go ahead base don a solid protocol application, but there’s probably a near term correction on the cards for Immune Pharmaceuticals Inc (NASDAQ:IMNP) as markets reevaluate what we see as a bit of an overzealous response to the news.
Immune Pharmaceuticals Announces Immuno-Oncology R&D Update on December 8, 2016
Have my seat with popcorn!
http://finance.yahoo.com/news/immune-pharmaceuticals-announces-immuno-oncology-140000904.html
NEW YORK, Nov. 22, 2016 /PRNewswire/ -- Immune Pharmaceuticals (IMNP) (Immune) announced today that it will hold an Immuno-Oncology R&D Update for investors on December 8, 2016 in New York City. Immune management and several Oncology Key Opinion leaders will review development plans and milestones for Ceplene® and other pipeline assets to be funded separately in the recently announced Immune subsidiary, Cytovia Oncology.
Investor 100
Shareholders @ IMNP
http://www.nasdaq.com/symbol/imnp/institutional-holdings
Investor 100
Immune Pharmaceuticals Announces Immuno-Oncology R&D Update on December 8, 2016
Confirms plans for pivotal phase 3 Overall Survival study with Ceplene in AML
http://www.prnewswire.com/news-releases/immune-pharmaceuticals-announces-immuno-oncology-rd-update-on-december-8-2016-300367275.html
NEW YORK, Nov. 22, 2016 /PRNewswire/ -- Immune Pharmaceuticals (NASDAQ: IMNP) (Immune) announced today that it will hold an Immuno-Oncology R&D Update for investors on December 8, 2016 in New York City. Immune management and several Oncology Key Opinion leaders will review development plans and milestones for Ceplene® and other pipeline assets to be funded separately in the recently announced Immune subsidiary, Cytovia Oncology.
Prior to the Immuno-Oncology R&D Update, the Ceplene Scientific Advisory Board (SAB) will meet on December 4, 2016 in San Diego where the American Society of Hematology is holding its annual meeting. Following the guidance from the Food and Drug Administration, the SAB will review the proposed clinical protocol for a pivotal phase 3 Overall Survival clinical study of Ceplene in combination with low dose IL2 in the remission maintenance of patients with Acute Myeloid Leukemia (AML).
About Immune Pharmaceuticals Inc.:
Immune Pharmaceuticals Inc. (NASDAQ: IMNP) applies a personalized approach to treating and developing novel, highly targeted antibody therapeutics to improve the lives of patients with inflammatory diseases and cancer. Immune's lead product candidate, bertilimumab, is in Phase II clinical development for moderate-to-severe ulcerative colitis as well as for bullous pemphigoid, an orphan autoimmune dermatological condition. Other indications being considered for development include atopic dermatitis, Crohn's disease, severe asthma and Non-Alcoholic Steato-Hepatitis (NASH), an inflammatory liver disease. Immune recently expanded its portfolio in immuno-dermatology with topical nano-formulated cyclosporine-A for the treatment of psoriasis and atopic dermatitis. Immune's oncology pipeline includes Ceplene® which is in late stage clinical development for maintenance remission in Acute Myeloid Leukemia (AML) in combination with IL-2. Additional oncology pipeline includes Azixa® and crolibulin, Phase II clinical stage vascular disrupting agents, and novel technology platforms; bispecific antibodies and NanomAbs™. Maxim Pharmaceuticals Inc., Immune's pain and neurology subsidiary is developing AmiKet™ and AmiKet™ Nano™ for the treatment of neuropathic pain. For more information, visit Immune's website at www.immunepharma.com, the content of which is not a part of this press release.
Forward-Looking Statements:
This news release and any oral statements made with respect to the information contained in this news release contain forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. You are urged to consider statements that include the words "may," "will," "would," "could," "should," "believes," "estimates," "projects," "potential," "expects," "plans," "anticipates," "intends," "continues," "forecast," "designed," "goal" or the negative of those words or other comparable words to be uncertain and forward-looking. Such forward-looking statements include statements that express plans, anticipation, intent, contingency, goals, targets, future development and are otherwise not statements of historical fact. These statements are based on our current expectations and are subject to risks and uncertainties that could cause actual results or developments to be materially different from historical results or from any future results expressed or implied by such forward-looking statements. Factors that may cause actual results or developments to differ materially include, but not limited to: the risks associated with the adequacy of our existing cash resources and our ability to continue as a going concern; the risks associated with our ability to continue to meet our obligations under our existing debt agreements; the risk that clinical trials for bertilimumab, Ceplene, Azixa, AmiKet, AmiKet Nano, LidoPain or NanoCyclo will not be successful; the risk that bertilimumab, AmiKet or compounds arising from our NanomAbs program will not receive regulatory approval or achieve significant commercial success; the risk that we will not be able to find a partner to help conduct the Phase III trials for AmiKet on attractive terms, on a timely basis or at all; the risk that our other product candidates that appeared promising in early research and clinical trials do not demonstrate safety and/or efficacy in larger-scale or later-stage clinical trials; the risk that we will not obtain approval to market any of our product candidates; the risks associated with dependence upon key personnel; the risks associated with reliance on collaborative partners and others for further clinical trials, development, manufacturing and commercialization of our product candidates; the cost, delays and uncertainties associated with our scientific research, product development, clinical trials and regulatory approval process; our history of operating losses since our inception; the highly competitive nature of our business; risks associated with litigation; and risks associated with our ability to protect our intellectual property; risks associated with the contemplated transaction with NPT. These factors and other material risks are more fully discussed in our periodic reports, including our reports on Forms 8-K, 10-Q and 10-K and other filings with the U.S. Securities and Exchange Commission. You are urged to carefully review and consider the disclosures found in our filings, which are available at www.sec.gov or at www.immunepharma.com. You are cautioned not to place undue reliance on any forward-looking statements, any of which could turn out to be wrong due to inaccurate assumptions, unknown risks or uncertainties or other risk factors. We expressly disclaim any obligation to publicly update any forward-looking statements contained herein, whether as a result of new information, future events or otherwise, except as required by law.
For further information: Anna Baran-Djokovic, anna.baran@immunepharma.com; Audrey Rebibo, audrey.rebibo@immunepharma.com; Immune Pharmaceuticals Inc., 646-481-5058, investors@immunepharma.com
SOURCE Immune Pharmaceuticals Inc.
Investor 100
Highly speculative play with this company with a high degree of risk and reward! 2017 will be a pivotal year for IMNP!
GLTA
Investor 100
Well it closed out at .1691 /2M shares / +2.24% or .0037.
Tax selling season is full swing so we'll see where we go this month!
-Holding shares - will double up on bigger dip-
Investor 100
That is a bold statement without any supportive back-up so I assume that is pure opinion on your part!
Investor 100
This investors may have tried that tactic and got burned back in September...paper loss the moment..but more than likely sold for a huge loss.
http://www.marketwatch.com/story/immune-pharmaceuticals-receives-2-million-investment-in-common-stock-at-050-per-share-2016-09-07
Holding my shares..waiting for a big dip to back-up the truck.
Investor 100
That happens at small bio tech companies buy the broader picture looks interesting for 2017.
Investor 100
Immune Pharmaceuticals Secures Up to $11 Million in New Financing to be Deployed With a Focus on Bertilimumab Clinical Development
Looking forward to 2017....
http://finance.yahoo.com/news/immune-pharmaceuticals-secures-11-million-141500951.html
Dr. Daniel Teper, Immune's CEO commented: "We appreciate the continuous commitment of our investors to support this financing of Immune, which will be dedicated to the advancement of bertilimumab clinical development. We are also implementing a strategy to unlock the value of other assets through the establishment of new, privately-funded companies which will be partially owned by Immune."
Investor 100
Immune Pharmaceuticals, Inc. (NASDAQ:IMNP) lost about -56.1 percent in value since last earnings
http://theindependentrepublic.com/2016/11/07/immune-pharmaceuticals-inc-nasdaqimnp-lost-about-56-1-percent-in-value-since-last-earnings/
Immune Pharmaceuticals, Inc. (NASDAQ:IMNP) is projected to declare fiscal third quarter financial results right before the stock market’s official open on November 16, 2016. The stock lost about -56.1 percent in price since last results when it was at $0.41 a share. Based on the most relevant past-periods data, there is an 80 percent probability for this firm’s share price to go down following next quarterly results. Earnings reaction history tells us that the equity price moved down 4 times out of last 5 reported quarters. It has beaten earnings-per-share estimates 33% of the time in its last 12 earnings reports. It fell short of earnings estimates on 6 occasions, and it has met expectations 1 time.
Here’s how traders responded to IMNP earnings announcements over the past few quarters.
Immune Pharmaceuticals, Inc. (IMNP) Earnings Surprises & Reaction
Given its history, the average earnings announcement surprise was -865.4 percent over the past four quarters. Back on August 15, 2016, it posted earnings per-share earnings at $-0.13 which beat the consensus $-0.14 projection (positive surprise of7.14%. For the quarter, revenue came in at 0M versus consensus estimate of 0M. The stock dropped -4.88 percent the session following the earnings reports were released, and on 7th day price change was -7.32 percent.
On May 16, 2016, it reported earnings at $-0.17 a share compared with the consensus estimate of $-0.17 per share (negative surprise of 0%). Revenue of 0M for that quarter was below the $0M analysts had expected. The stock climbed 11.11% the day following the earnings announcement, and on 7th day price change was 0%.
On November 12, 2015, it announced earnings per share at $-0.33 versus the consensus estimate of $0.01 per share (negative surprise of -3400%). That came on revenues of $0M for that period. Analysts had expected $0M in revenue.
Immune Pharmaceuticals, Inc. Earnings Estimates
As Q3 earnings announcement date approaches, Wall Street is expecting earnings per share of $-0.13. The analysts’ present consensus range is $-0.15-$-0.11 for EPS. The market consensus range for revenue is between $0M and $0M, with an average of $0M.
Immune Pharmaceuticals, Inc. (NASDAQ:IMNP) last ended at $0.18, sending the company’s market cap near $15.3M. The consensus 12-month price target from analysts covering the stock is $2. The share price has declined -88.05% from its top level in 52 weeks and advanced -74.79% this year. It recently traded at a volume of 1162574 shares. The recent trading ended with the price nearly -17.45 lower for the last 5 trading days, rebounding 10.25% from its 52-week low.
investor 100
Immune Pharmaceuticals Inc. (IMNP) Set to Announce Earnings on Thursday
mmune Pharmaceuticals Inc. (NASDAQ:IMNP) is scheduled to post its quarterly earnings results on Thursday, November 10th. Analysts expect Immune Pharmaceuticals to post earnings of ($0.14) per share for the quarter.
Immune Pharmaceuticals (NASDAQ:IMNP) last released its quarterly earnings results on Tuesday, August 16th. The biopharmaceutical company reported ($0.13) earnings per share (EPS) for the quarter, hitting the Zacks’ consensus estimate of ($0.13). On average, analysts expect Immune Pharmaceuticals to post $-0.58 EPS for the current fiscal year and $-0.17 EPS for the next fiscal year.
Shares of Immune Pharmaceuticals Inc. (NASDAQ:IMNP) traded down 2.9950% during trading on Tuesday, reaching $0.1749. The company had a trading volume of 2,103,717 shares. The company’s 50 day moving average is $0.25 and its 200-day moving average is $0.35. Immune Pharmaceuticals Inc. has a 1-year low of $0.17 and a 1-year high of $1.54. The stock’s market capitalization is $13.85 million.
Separately, FBR & Co reaffirmed a “buy” rating on shares of Immune Pharmaceuticals in a report on Sunday, September 18th.
Immune Pharmaceuticals Company Profile
Immune Pharmaceuticals Inc is a clinical-stage biopharmaceutical company. The Company is engaged in the development and commercialization of targeted therapeutics in the fields of immuno-inflammation and immuno-oncology. The Company’s segment is acquiring, developing and commercializing prescription drug products.
http://www.com-unik.info/2016/11/08/immune-pharmaceuticals-inc-imnp-set-to-announce-earnings-on-thursday.html
investor 100
IMNP @ Stockcharts
Without a doubt the whole market is in limbo but with that some buying opportunities...bought more shares today as I look forward to 2017!
http://www.marketwatch.com/story/immune-pharmaceuticals-receives-fda-guidance-for-low-dose-il-2-in-combination-with-phase-iii-pivotal-trial-of-its-aml-therapy-ceplene-2016-10-27

Investor 100
IMNP @ Boston BioTech Conference
https://bbbiotechconference.com/conference-agenda.php?id=43
Investor 100
Immune Pharmaceuticals (IMNP) Receives FDA Guidance for Phase III Pivotal Trial
POisitve interview with CMO & Principal Owner...positive feedback from FDA...look forward to 2017 for more updates!
IMNP @ Stockcharts
Market has been choppy for weeks and IMNP not immune.
Proxy Statement - Notice of Shareholders Meeting (preliminary) (pre 14a)
http://ih.advfn.com/p.php?pid=nmona&article=72719816&symbol=IMNP
At the annual meeting, we will ask our holders of Common Stock to:
1. ratify the selection of BDO USA, LLP as our independent registered public accounting firm for our fiscal year ending December 31, 2016;
2. approve an increase to the amount of shares authorized under the Company’s 2015 Equity Incentive Plan from 5,000,000 to 15,000,000;
3. re-elect each of Mr. Daniel G. Teper and Mr. John Neczesny, nominees for Class III directors, as members of our Board of Directors; and
4. authorize an amendment to our Certificate of Incorporation to effect a reverse stock split of our common stock at a specific ratio, within a range of 1-for- 2 and 1-for- 25, to be determined by our Board of Directors in its sole discretion and effected, if at all, on or before December 16, 2017.
The Board of Directors recommends the approval of each of these proposals.
Investor 100
The Long Case For Immune Pharmaceuticals Inc. 2 comments
Oct 9, 2016 8:51 AM | about stocks: IMNP
Elevator Pitch
Ceplene new data from a post hoc analysis of EpiCept's Phase 3 clinical study with Ceplene has shown positive data non-FAB-M2 subtype AML. Non-FAB-M2 AML making up roughly 70% of the total AML market. If Phase 3 trial initiated and successful IMNP expected value will increase dramatically.
Thesis & Catalyst For Immune Pharmaceuticals Inc. (NASDAQ:IMNP)
IMNP is undervalued because at the AACR annual meeting, researchers out of Sweden presented data on the Phase 4 MISSION trial, designed to assess the immunomodulatory properties of Ceplene + IL-2, relating potential biomarkers with clinical outcome. Investigators found a reduction in the frequency of CD8+ T effector memory TEM cells during the first cycle of Ceplene + IL-2 prognosticated leukemia-free survival LFS HR 0.25, P=0.001 and overall survival OS HR 0.24, P=0.009. Induction of T effector cells TEFF during the first cycle also impacted favorably on patient outcome P=0.048 for OS.
A reduction in TEM cells during the first cycle of Ceplene + IL-2 led to an improvement in both LFS and OS in all patients. The results were particularly well in patients over the age of 60. Similarly, an induction of TEFF cells during the first cycle of Ceplene + IL-2 led to an improvement in both LFS and OS in all patients, again, with excellent results in patients over the age of 60.
The increase in TEFF cells and a decrease in TEM cells was the strongest predictor of complete response and improvement in LFS HR 0.19, P=0.001 and OS HR 0.13, P=0.008 in older patients, over 60 years with high risk relapsed AML.
Published data indicates that targeting different subtypes of AML for a clinical trial - all non-FAB-M2 subtypes has shown great results. It also indicates that Immune's explanation on how Ceplene + IL-2 was able to demonstrate the strongest improvement in LFS in non-FAB-M2 AML patients is due to the mechanism of action and not by chance.
Non-FAB-M2 AML making up roughly 70% of the total AML market so you can imagine market potential.
Once Ceplene + IL-2 is being used to treat AML patients, oncologists can analyze T cell phenotypic expression before and after the first cycle looking for an increase in TEFF cells and a reduction in TEM cells. With confirmed response, patients can be kept on Ceplene + IL-2 therapy. Lack of response can also be identified if the T cell expression does not relate to the altered distribution, and those patients can be moved to other medications.
Valuation
Immune Pharma is currently working with principal investigators to design a Phase 3 trial in line with the FDA's guidance issued to EpiCept in 2011 and if talks with financier for Phase 3 successful present shareholders will be rewarded with expected share price of around 2$ Similar share price is indicated by analysts following the company. Also would like to be noted that after Ceplene data published insiders loaded heavily and one institutional - present investor has purchased 4M share @ 0.5$ with no warrants, no restrictive covenants, and no restrictions on use of proceeds. If you add IMNP pipeline with above described Ceplene potential even average investor can see 300-400% gain in share price.
Disclosure: I/we have no positions in any stocks mentioned, but may initiate a long position in IMNP over the next 72 hours.
Additional disclosure: All data presented are available and publicly published
Stocks: IMNP
http://seekingalpha.com/instablog/7947381-golfbag/4923206-long-case-immune-pharmaceuticals-inc
Added @IMUC
Took this opportunity on the dip and added on this play which requires patience.
High Rusk High Reward!
Investor 100
AH @ IMNP
You are correct over 114k shares traded closing at .31
Next week could be interesting ( IMO ).
Investor 100
Immune Forms Pain & Neurology Spin-Off Called Maxim Pharma
Outstanding summary opportunity at IMNP!
http://www.bionapcfa.com/2016/09/immune-forms-pain-neurology-spin-off.html
Last week, Immune Pharmaceuticals, Inc. (NASDAQ: IMNP) announced the formation of a pain and neurology focused subsidiary that will own worldwide rights to AmiKet™ and Amiket™ Nano for the treatment of neuropathic pain, along with all the intellectual property around both key assets. The new company, called Maxim Pharmaceuticals, Inc., is wholly-owned by Immune Pharma at this time; however, Immune has also entered into a binding agreement with a syndicate of experienced healthcare investors to purchase up to $20 million Maxim capital stock, with an initial investment of $5 million expected in the next 30 days.
The creation of a pain and neurology focused subsidiary is an important step towards realizing what Immune believes is the significant value of AmiKet for shareholders. Below is a quick review of AmiKet, the transaction, and what to expect over the next several months at Maxim Pharma.
Background On AmiKet
AmiKet is a topical formulation containing 4% amitriptyline and 2% ketamine, both U.S. FDA approved compounds, designed for the treatment of neuropathic pain. AmiKet is applied twice daily and should offer several advantages for the treatment of neuropathic pain, including a potent analgesic effect and lack of systemic adverse events. This type of topical delivery of a non-opioid is the preferred mode of administration by many patients and physicians.
Immune has conducted a total of nine Phase 1 and Phase 2 clinical studies with AmiKet in over 700 patients. A multicenter, double-blind, randomized, placebo-controlled, parallel group Phase 2 non-inferiority study examined AmiKet compared to oral gabapentin (1,800 mg/day) for the treatment of post-herpetic neuralgia (PHN) in 360 patients over four weeks (1). The primary endpoint was the change in mean pain score, measured on an 11-point scale, from baseline to final week of treatment. The data are presented below (source: Immune Pharma).
Results show AmiKet to be non-inferior to gabapentin for the relief of pain associated with PHN. Additionally, the data demonstrated statistically superior pain relief compared to placebo, as well as a significantly greater reduction from baseline in average sleep disturbance score compared to placebo (average reduction: 1.87 vs. 1.31, p=0.0265) (2). Gabapentin, marketed by Pfizer as Neurontin®, posted worldwide sales at peak of over $2.7 billion until generics arrived in 2004.
In 2010, the FDA granted AmiKet orphan drug status for the treatment PHN. As such, if approved in PHN, AmiKet will have seven years of market exclusivity. AmiKet Nano is an improved topical nanoparticle formulation of AmiKet that will provide better penetration of the two drugs, along with intellectual property protection to 2036. AmiKet also received Fast Track designation from the U.S. FDA in 2012 for chemotherapy-induced peripheral neuropathy (CIPN).
If successfully commercialized in PHN, the label expansion for AmiKet and AmiKet Nano is substantial. There are several neuropathic pain indications that make sense beyond PHN, including the aforementioned CIPN, as well as HIV-related neuropathy and diabetic neuropathy (DPN).
Maxim Pharma Valuation & Future
Immune has established the pre-money valuation for Maxim at $15 million. As noted above, Immune shareholders currently own 100% right now. Upon the initial investment of $5 million from the syndicate of experienced healthcare investors, Immune will own 75% of Maxim, with the other 25% owned by the syndicate. The binding agreement allows for the future investment of up to an additional $15 million, which would result in Immune owning approximately 43% of the company upon full investment. I expect the first tranche to close in the next 30 days.
The initial $5 million investment is enough to allow Maxim to push AmiKet into a Phase 3 trial by the middle of 2017. Maxim likely will be engaging with both the U.S. and EU regulatory authorities over the next several months. I expect a second Phase 3 trial will be required and that Maxim is likely 24 months from filing the first application on AmiKet once the Phase 3 trial begins. A total investment of $20-25 million likely is required to file, with an additional $5 million necessary for pre-commercialization activities.
At this time, not much is known about the syndicate of experienced healthcare investors, only that part includes a prominent New York-based family office, and may include a broader group of investors which will be secured through an investment bank to be selected by both parties. Pursuant to the binding agreement, immediately following the $5 million initial funding, Immune will expand the Board of Directors of Maxim to five members, which will include one representative of the syndicate investors. A newly appointed Chief Executive Officer of Maxim is also expected to be announced in the coming weeks (3).
Maxim's goal is to become a fully-integrated specialty pharma company with a focus on pain and neurology. Companies that are successful with a similar model include Depomed Inc. (DEPO), Endo Pharmaceuticals (ENDP), Mallinckrodt plc (MNK), and Purdue Pharma. I believe Maxim will be pursuing additional pain or neurology assets in 2017, one of which may be Immune's LidoPain product (4). Maxim will use reasonable efforts to become publicly traded on a national stock exchange within 18 months of the effective date of the agreement.
In the meantime, Immune is eligible to receive up to $25 million in development milestones and up to $120 million in commercial milestones, as well as product sales royalties from AmiKet or AmiKet Nano from Maxim. Immune will also be eligible to receive 25% and up to 50% of sublicense fees received by Maxim. The deal is not too far off from what I predicted back in April 2015.
Conclusion
Chronic pain affects 100 million people in the U.S., more than the total affected by heart disease, cancer, and diabetes combined. IMS Health reported over 328 million prescriptions for pain medications and sales of prescription pain products totaled $13 billion in 2014. Lidoderm®, a 5% topical lidocaine, posted sales of $1.2 billion at Endo Pharmaceuticals prior to the patent expiration in 2013. A topical product such as AmiKet offers patients a safer and more effective approach for the treatment of neuropathic pains, such as PHN, could be a $350 million product in the U.S. Label expansion into CIPH and DPN put the peak market opportunity for AmiKet and AmiKet Nano over $1 billion.
I think this is a good deal for shareholders of Immune Pharma. AmiKet is now in the hands of Maxim Pharma and Immune can focus more attention on the two ongoing Phase 2 trials with bertilimumab, one in bullous pemphigoid and the other in ulcerative colitis. Positive data from these programs have the potential to turn around the stock. In the meantime, as Maxim progresses with AmiKet into Phase 3, Immune has the opportunity to receive increased equity in Maxim or cash milestones to drive bertilimumab forward. I look forward to seeing a similar transaction for the oncology assets perhaps in 2017.
Recap @ IMNP
For those of you investigating a purchase here you might consider the fact that management has purchased shares well above this weeks closing prices and the most recent developments are listed below are positive in my opinion.
http://openinsider.com/search?q=imnp
Immune Pharmaceuticals Signs Agreement to Fund Maxim Pharmaceuticals, Inc., its Pain and Neurology subsidiary
NEW YORK, Sept. 16, 2016 /PRNewswire/ -- Immune Pharmaceuticals Inc. (IMNP) ("Immune" or the "Company"), a biopharmaceutical company focused on the development of targeted therapeutics for the treatment of inflammatory diseases and cancer, announced today that the Board of Directors has approved to designate Maxim Pharmaceuticals Inc. ("Maxim"), one of the Company's existing subsidiaries, as the dedicated entity to develop and commercialize, and hold the intellectual property related to, AmiKet® and other related pain and neurology assets. In addition, the Company has entered into a binding agreement with NPT (the "Binding Agreement"), a syndicate of experienced healthcare investors, pursuant to which NPT or its designees have agreed to purchase up to $20 million of the capital stock of Maxim, with an initial funding of $5 million to occur within thirty days. Previously, the Company entered into an option agreement with NPT, dated May 15, 2016, as amended on July 18, 2016. Pursuant to the Binding Agreement, immediately following the $5 million initial funding by NPT, Immune will expand the Board of Directors of Maxim, to five members which will include one NPT representative, and a newly hired Chief Executive Officer of Maxim.
For full disclosure please refer to the Form 8-K filed on September 16, 2016
Immune Pharmaceuticals Receives $2 Million Investment in Common Stock at $0.50 per Share
Immune Pharmaceuticals Inc. (IMNP) ("Immune"), a biopharmaceutical company focused on the development of targeted therapeutics for the treatment of inflammatory diseases and cancer, announced today that it has entered into a stock purchase agreement with an existing stockholder for the sale of 4,000,000 shares of the Company's common stock at a fixed price of $0.50 per share, for gross proceeds of $2,000,000. There are no warrants, no restrictive covenants, and no restrictions on use of proceeds.
For full disclosure, please refer to the Form 8-K filed on September 7, 2016.
Immune Pharmaceuticals Provides Business and R&D Update and Announces Second Quarter 2016 Financial Results
Immune Pharmaceuticals Inc. (IMNP) ("Immune" or the "Company") announced financial results for the second quarter and six months ended June 30, 2016 as well as provided pipeline highlights and a business update.
"Immune has continued to make significant R&D and operational progress, while we seek to unlock the potential value of our pipeline through institutional financing and corporate transaction opportunities," said Dr. Daniel Teper, CEO of Immune Pharmaceuticals Inc.
Pipeline Highlights
Immuno-inflammation
Bertilimumab continues to accrue patients in its two phase 2a clinical trials in bullous pemphigoid (BP) and ulcerative colitis (UC). The BP trial is expanding to six US centers in addition to the two Israeli centers. The first US center was initiated in August 2016 and started to screen patients with others to follow shortly. The UC trial is expanding to Eastern Europe with site initiations to be completed in the fourth quarter of 2016. An additional phase 2a trial in Atopic Dermatitis (AD) is in final planning stages in Canada.
In the second quarter of 2016, new pre-clinical data was generated in AD and a new provisional patent was filed in partnership with Hadasit, the technology transfer of Hadassah hospital, for oral use of anti-eotaxin antibodies in Non-Alcoholic Steato-Hepatitis (NASH).
Immune also advanced process development for its new cell line for the production of bertilimumab. Immune expects to bridge to the new cell line starting in mid-2017. Immune is assessing options for development of a sub-cutaneous formulation of bertilimumab.
Immune advanced the development of NanoCyclo, a topical formulation of cyclosporine A for the potential treatment of AD and psoriasis. We are currently focused on the GMP manufacturing process and pre-clinical regulatory studies toward filing of an Investigational New Drug application in 2017.
Immuno-oncology
We are continuing to advance our plans to focus on our two clinical stage assets in oncology, Ceplene® and Azixa® and have made significant progress in the past quarter with both of these assets.
Ceplene is approved in Europe for remission maintenance and prevention of relapse in adults with Acute Myeloid Leukemia (AML), an orphan indication with poor survival prognosis, for which no effective therapy is available to patients.
Presented new positive biomarker driven clinical data from the European Phase IV study at the American Association of Cancer Research (AACR) Annual meeting in New Orleans in April 2016. The phase IV RE:MISSION trial was designed to assess the immuno-modulatory properties of Ceplene/IL-2, and to correlate potential biomarkers with clinical outcome. Results indicated that outcomes were strongly predicted with a specific T-cell biomarker including leukemia-free survival (LFS) (HR 0.25, P=0.001) and overall survival (OS) (HR 0.24, P=0.009).
Reported new data showing that Ceplene enhanced response to PD-1 and PD-L1 checkpoint inhibitors in lymphoma and solid tumor models. This data is supporting the provisional patent application filed in the first quarter of 2016 for use of Ceplene in combination with check point inhibitors.
Presented new data at the Conference "Regulatory Myeloid Suppressor Cells: From Basic Discovery to Therapeutic Application" held in Philadelphia, PA providing further mechanistic evidence to explain the promising efficacy of Ceplene in combination with low dose IL-2 observed clinically in the myelomonocytic M4 and M5 AML subtypes in both Phase III and the recently completed Phase IV clinical trials.
Immune now plans to leverage recent Ceplene/IL-2 data on predictive bio-markers and recent phase IV data results to design and reach an agreement with the FDA for a pivotal study in AML supporting a new drug application in the U.S.
Presented new positive biomarker driven clinical data from the European Phase IV study at the American Association of Cancer Research (AACR) Annual meeting in New Orleans in April 2016. The phase IV RE:MISSION trial was designed to assess the immuno-modulatory properties of Ceplene/IL-2, and to correlate potential biomarkers with clinical outcome. Results indicated that outcomes were strongly predicted with a specific T-cell biomarker including leukemia-free survival (LFS) (HR 0.25, P=0.001) and overall survival (OS) (HR 0.24, P=0.009).
Reported new data showing that Ceplene enhanced response to PD-1 and PD-L1 checkpoint inhibitors in lymphoma and solid tumor models. This data is supporting the provisional patent application filed in the first quarter of 2016 for use of Ceplene in combination with check point inhibitors.
Presented new data at the Conference "Regulatory Myeloid Suppressor Cells: From Basic Discovery to Therapeutic Application" held in Philadelphia, PA providing further mechanistic evidence to explain the promising efficacy of Ceplene in combination with low dose IL-2 observed clinically in the myelomonocytic M4 and M5 AML subtypes in both Phase III and the recently completed Phase IV clinical trials.
Immune now plans to leverage recent Ceplene/IL-2 data on predictive bio-markers and recent phase IV data results to design and reach an agreement with the FDA for a pivotal study in AML supporting a new drug application in the U.S.
Azixa is a novel microtubule destabilizer that has a dual mode of action, acting as a vascular disrupting agent (VDA) and a potent cytotoxin with a unique ability to penetrate the blood brain barrier and reach high concentrations in the brain.
Generated and reported new data demonstrating that the combination of Azixa and immune checkpoint inhibitors such as anti-CTLA-4 antibody resulted in enhanced activity compared to the activity elicited by the single agents alone.
Filed a provisional patent application with the USPTO relating to the combination of Azixa and immune checkpoint inhibitors such as an anti-CTLA-4 antibody and anti-PD1 monoclonal antibodies in the treatment of cancer.
Generated and reported new data demonstrating that the combination of Azixa and immune checkpoint inhibitors such as anti-CTLA-4 antibody resulted in enhanced activity compared to the activity elicited by the single agents alone.
Filed a provisional patent application with the USPTO relating to the combination of Azixa and immune checkpoint inhibitors such as an anti-CTLA-4 antibody and anti-PD1 monoclonal antibodies in the treatment of cancer.
Business Update
On July 18, 2016, the Company signed an amendment to the May 15, 2016 Option Agreement with Novel Pain Therapeutics, LLC (NPT), a syndicate of experienced healthcare investors. Immune agreed to designate a subsidiary that will hold all of the Intellectual Property for Immune's pain assets (Amiket®, Amiket Nano™ and LidoPain®). Under the terms of the Amended Option Agreement, the parties have agreed to a pre-money valuation of $15 million for Immune's equity stake in the pain subsidiary with a target closing date of September 15, 2016 for the first tranche of dedicated financing by NPT and other investors. In addition, subject to the exercise of the Option Agreement and the execution of the definitive License Agreement, as well as the ultimate development, sale and/or licensing of the products, Immune will be eligible to receive up to $145 million in milestone payments as well as sublicensing fees and royalties.
Second Quarter and Six Months Ended June 30, 2016 Financial Discussion
Immune reported a loss attributable to common stockholders of $5.7 million, or $0.13 per share, for the quarter ended June 30, 2016, compared to a loss attributable to common stockholders of $2.9 million, or $0.12 per share, for the quarter ended June 30, 2015. For the six months ended June 30, 2016, Immune reported a loss attributable to common stockholders of $11.7 million, or $0.30 per share, compared to a loss attributable to common stockholders of $8.2 million, or $0.34 per share, for the six months ended June 30, 2015.
Research and Development ("R&D") expenses increased by $0.8 million during the quarter ended June 30, 2016 to $1.9 million compared with $1.1 million during the quarter ended June 30, 2015. For the six months ended June 30, 2016 R&D expenses increased by $1.7 million to $4.0 million from $2.3 million. The increase in R&D expenses for the quarter and six months ended June 30, 2016 was primarily driven by higher salaries, employee benefits and share-based compensation as a result of higher R&D headcount. In addition, the increase in R&D expenses was driven by higher clinical trial expenses as the Company continues to ramp up its clinical trials related to bertilimumab in both BP and UC.
General and Administrative ("G&A") expenses decreased $0.8 million during the quarter ended June 30, 2016 to $0.9 million compared with $1.7 million during the quarter ended June 30, 2015, due to lower share based compensation expense and lower consulting and business development expenses. For the six months ended June 30, 2016 G&A expenses decreased by $0.6 million to $3.4 million from $4.0 million during the six months ended June 30, 2015 primarily due to lower share based compensation expense and lower legal fees.
Non-operating expense was $0.7 million during the three months ended June 30, 2016 compared with non-operating expense of $0.1 million during the three months ended June 30, 2015. For the six months ended June 30, 2016, non-operating expense was $1.4 million compared with $0.2 million for the six months ended June 30, 2015. Non-operating expense increased for the three and six months ended June 30, 2016 primarily relating to cash interest expense and amortization of debt issuance costs for the Company's loan agreement and the loss on the change in fair value of derivative liability instrument.
Investor 100
Hopefully todays positive movements numbs the pain and gets up up and about soon!
Investor 100
