Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
LC,
We will add that to the list of great article recommendations from LC—thank you for highlighting the beauty and potentially limitless value of the DCVax-L platform technology—cooperation and combination are key features of how this novel technology might be implemented in a clinical setting during this emerging era of targeted cell-based combination therapy. The JCI article link that you posted a couple weeks ago was very much appreciated too!
https://academic.oup.com/ajhp/advance-article-abstract/doi/10.1093/ajhp/zxad251/7289179
https://www.jci.org/articles/view/169314
https://www.raps.org/News-and-Articles/News-Articles/2023/9/FDA%E2%80%99s-Califf-Expect-to-see-more-RWE-based-regulato
https://clinicaltrials.gov/study/NCT04201873
Combo is King!
LC,
https://x.com/mc17595091/status/1722795659546304877?s=46&t=BbaVVAHSNyBEm-BUTWiU6w
The publisher has a lot precious and significant data to prepare…
Thanks again for the JCI link BTW…
Exciting times … Combo is King!

https://www.raps.org/news-and-articles/news-articles/2023/8/fda-finalizes-guidance-on-real-world-evidence-in-d
https://www.raps.org/News-and-Articles/News-Articles/2023/9/FDA%E2%80%99s-Califf-Expect-to-see-more-RWE-based-regulato
https://www.fda.gov/media/171667/download
https://www.uclahealth.org/cancer/researchers/spores/ucla-spore-brain-cancer/research-projects
https://finance.yahoo.com/news/northwest-biotherapeutics-announces-dr-linda-140000962.html
https://www.merck.com/news/daiichi-sankyo-and-merck-announce-global-development-and-commercialization-collaboration-for-three-daiichi-sankyo-dxd-adcs/




https://www.jci.org/articles/view/169314
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
https://hotb.org/wp-content/uploads/2022/05/spore-article-for-donors-4.18.22.pdf
https://www.uclahealth.org/news/brain-cancer-discovery-clinical-trials

https://www.fda.gov/drugs/news-events-human-drugs/50-years-progress-treating-patients-cancer

LC, This study found that autologous tumor lystate-loaded dendritic cell vaccination plus standard of care (SOC) was associated with improved survival in both newly diagnosed & recurrent glioblastoma compared with matched controls who received SOC treatment. https://t.co/Uso7Pkh1Kt
https://x.com/mc17595091/status/1722786151629480429?s=46&t=BbaVVAHSNyBEm-BUTWiU6w
I’ve been reading your posts and it sounds like you’re misinformed and need more reliable information. Perhaps consider looking beyond social media to seek trustworthy full-context sources…
For a reliable appraisal of the trial, its design, and the significance of the resulting survival data, it is advisable to refer to the analyses of qualified physicians. One such analysis can be found on the JAMA Oncology website where the link to the independent peer review and supplementary materials were published.


https://finance.yahoo.com/news/northwest-biotherapeutics-announces-dr-linda-140000962.html
https://www.jci.org/articles/view/169314
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
https://hotb.org/wp-content/uploads/2022/05/spore-article-for-donors-4.18.22.pdf
https://www.uclahealth.org/news/brain-cancer-discovery-clinical-trials
learningcurve2020,
Your frequently posted “guesses” are baseless innuendo, but I would be concerned if you ceased to spread rumors—your desire to muddy the waters only reaffirms the existence of a treasure—there would exist no reason to spend time obstructing the view if there were nothing to see on the horizon…
Surprisingly, it was your post of the JCI link that really reaffirmed my strong belief in the possibility of a sky-high price tag … The implications of the JCI article really enlighten the invaluable impact that the DCVax-L platform could have in combination with other technologies for a number of cancers well into the future…
A picture is worth 100 billion words.
https://clinicaltrials.gov/study/NCT04201873
You have painted a very captivating picture with the investment of your time and energy … insightful inferences can be drawn from statements that were perhaps intended to obstruct clear understanding—there are hidden precious gems interwoven into your 30 daily posts.
Like the morning sunlight that we perceive vividly because its rays are not darkness, the relentless attempt to deceive with darkness might define the truth sharply in light of the persistent contrast to what it is not.
Combo is King!
https://www.jci.org/articles/view/169314



learningcurve2020,
Thank you for reposting your speculation about other DC therapies again and for challenging us to identify the differentiating features of the DCVax-L novel technology.
From the perspectives of patients and investors worldwide, we all would love to hear more about those other DC technologies—please post links from regulatory agencies and independently-reviewed clinical studies so that we can all perform further research.
In the U.S. and the U.K., the DCVax-L platform technology has years of combo data, NWBO has built the capacity to scale production to serve a global market, the NIH is continuing their support, and the clinical evidence-supported implications beyond GBM are promising …
Yesterday, ex highlighted the NIH which is a highly relevant topic to raise—The vast significance of Liau’s work and the further development of the DCVax-L cell-based platform technology in combination with other technologies was/is funded in large part by public dollars awarded through NIH peer-reviewed grants. DCVax-L is already supported by peer-reviewed clinical evidence, but there is much more to the story that is yet to unfold.
If you do some research into the NIH and the FDA, you will discover that not every investigational technology receives the same level of support and not every researcher’s work is recognized by consistent support spanning many consecutive years.
The NCI division of the NIH develops oncology drugs, and FDA approves drugs. There is some connection, and their historical and continuing support is not insignificant …
https://www.bentley.edu/news/new-study-shows-nih-investment-new-drug-approvals-comparable-investment-pharmaceutical
https://www.bentley.edu/news/nih-funded-research-related-every-new-cancer-drug-approved-2010-2016
https://ashpublications.org/ashclinicalnews/news/3819/NIH-Funding-Tied-to-FDA-Approval-of-210-Drugs
Targeted individualized combo therapies are emerging as the defining feature of a new era of oncology. Just look at the impact that PD1 blockade technology has had on the field of oncology in recent years!
https://clinicaltrials.gov/ct2/show/NCT04201873
The U.S. NIH has supported the DCVax-L platform for years, and the NIH renewed its support in 2022 to continue to fund the development of this technology and to fund research into combination therapies.
NIH-funded DCVax-L research is ongoing at UCLA today.
NIH grants are peer-reviewed and the research that NIH funds is highly scrutinized in advance of the award and also intermittently for the term of the various research projects.

https://www.uclahealth.org/cancer/researchers/spores/ucla-spore-brain-cancer/research-projects
https://www.raps.org/news-and-articles/news-articles/2023/8/fda-finalizes-guidance-on-real-world-evidence-in-d

https://www.raps.org/News-and-Articles/News-Articles/2023/9/FDA%E2%80%99s-Califf-Expect-to-see-more-RWE-based-regulato
https://www.fda.gov/media/171667/download
https://www.merck.com/news/daiichi-sankyo-and-merck-announce-global-development-and-commercialization-collaboration-for-three-daiichi-sankyo-dxd-adcs/

Mr. Newirth is another DCVax-L survivor well surpassing 10 years following a 2012 diagnosis! Congratulations!
https://www.hawaii.edu/news/2017/03/30/newirth-laker-for-a-day/
2023 Interview:
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/












ex,
Those excerpts that you posted are all accurate:
1. FDA does not allow trial participants to be used as lab rats.
2. Patients demanded crossover option.
3. Crossover was required to recruit and retain participants because nobody would join otherwise.
Number 1 aligns with common sense—FDA’s job includes protecting trial participants.
Number 2 also reflects common sense—wouldn’t you want to have the crossover option after becoming aware of progression?
Number 3 is obviously common sense too—who would want to be included in a placebo group after developing progressive disease without having the option to receive an efficacious treatment?
Conclusion: the P3 was exemplary—clinically appropriate and compassionate.
Interestingly, the PD1 combo study does have a placebo group—the reason why the pembrolizumab combo study that is currently underway at UCLA does not require a crossover is because the placebo group participants have access to an efficacious rGBM treatment.
Skip to timestamp 8:01. Dr. Liau again confirms that the crossover was mandated by the FDA.
https://edhub.ama-assn.org/jn-learning/audio-player/18738384

https://www.raps.org/news-and-articles/news-articles/2023/8/fda-finalizes-guidance-on-real-world-evidence-in-d

https://www.raps.org/News-and-Articles/News-Articles/2023/9/FDA%E2%80%99s-Califf-Expect-to-see-more-RWE-based-regulato
https://www.fda.gov/media/171667/download
https://jamanetwork.com/journals/jamaoncology/fullarticle/2798847
HyGro,
Thanks for the reply.
The externally controlled P3 was exemplary, but if you prefer placebo trials, I would recommend that you look at the small but mighty pembrolizumab combo study that is currently underway at UCLA with the support of NCI and Merck…
The interim PD1 combo data are brilliant, and the trial is significant for a number of reasons relating to FDA guidelines and NIH peer-reviewed grant funding.

https://clinicaltrials.gov/ct2/show/NCT04201873
https://www.fda.gov/media/120721/download
In the combo trial, patients who receive DCVax-L + placebo are being compared to patients who receive DCVax-L + pembrolizumab.
The control arm is receiving DCVax-L. While DCVax-L is not the current SOC, it is highly significant and notable that DCVax-L is being permitted to fill the role of SOC as the best available therapy within this combo study for rGBM. The combo trial also follows the P3 in sequence which in itself validates the findings of the P3 study—the placebo group in the PD1 study would not be receiving DCVax-L today if the preceding P3 trial had not proven its efficacy.

HyGo,
Thanks for reposting your opinions again, but I believe that you need better sources of credible and verifiable information…
The clinical data in total include data beyond the trial with the ECA, and the ECA included over a thousand well-matched contemporaneous patients.
Regarding the ECA/crossover design and the SAP, please review the video recording of Dr. Ashkan at ASCO 2022 and the JAMA Oncology podcast recording of Dr. Liau. Both recordings are included at the links below.
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=171254048;
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=171254756;
The P3 data were peer reviewed by independent physicians and qualified statisticians—please refer to JAMA Oncology. You might consider researching credible full-context sources to gather reliable information.


https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=173154324
learningcurve2020,
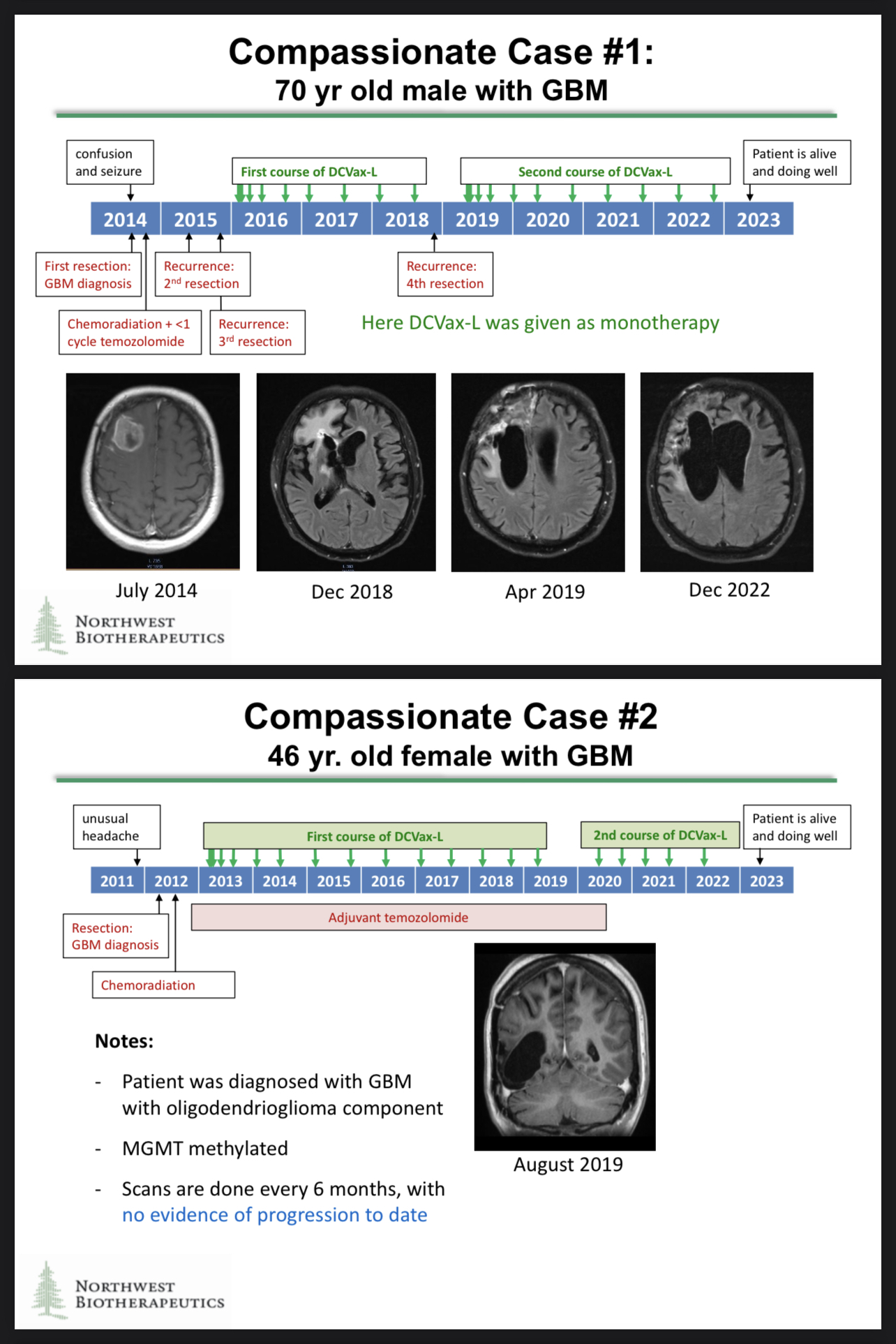
Great point! Thank you for reminding us about the brilliant compassionate use results. The compassionate use cases generated some astonishing data, and those data could be included in the regulatory filings!


https://t.co/082urSTQ0s

https://www.braintumourresearch.org/media/news/news-item/2022/11/18/astonishing-results-for-brain-tumour-vaccine-trial
learningcurve2020,
Reading your recent posts and ex’s recent posts remind us of some of the differentiating features of the DCVax-L novel technology and NWBO—years of combo data, capacity to scale production to serve a global market, continuing NIH support, and the implications for other cancers/diseases beyond GBM…
Earlier this evening, ex highlighted the NIH which is a highly relevant topic to raise—The vast significance of Liau’s work and the further development of the DCVax-L cell-based platform technology in combination with other technologies was/is funded in large part by public dollars awarded through NIH peer-reviewed grants. DCVax-L is already supported by peer-reviewed clinical evidence, but there is much more to the story that is yet to unfold.
If you do some research into the NIH and the FDA, you will discover that not every investigational technology receives the same level of support and not every researcher’s work is recognized by consistent support spanning many consecutive years.
The NCI division of the NIH develops oncology drugs, and FDA approves drugs. There is some connection, and their historical and continuing support of DCVax-L is not insignificant …
https://www.bentley.edu/news/new-study-shows-nih-investment-new-drug-approvals-comparable-investment-pharmaceutical
https://www.bentley.edu/news/nih-funded-research-related-every-new-cancer-drug-approved-2010-2016
https://ashpublications.org/ashclinicalnews/news/3819/NIH-Funding-Tied-to-FDA-Approval-of-210-Drugs
Targeted individualized combo therapies are emerging as the defining feature of a new era of oncology. Just look at the impact that PD1 blockade technology has had on the field of oncology in recent years!
https://clinicaltrials.gov/ct2/show/NCT04201873
The U.S. NIH has supported the DCVax-L platform for years, and the NIH renewed its support in 2022 to continue to fund the development of this technology and to fund research into combination therapies.
NIH-funded DCVax-L research is ongoing at UCLA today.
NIH grants are peer-reviewed and the research that NIH funds is highly scrutinized in advance of the award and also intermittently for the term of the various research projects.

https://www.uclahealth.org/cancer/researchers/spores/ucla-spore-brain-cancer/research-projects
https://www.raps.org/news-and-articles/news-articles/2023/8/fda-finalizes-guidance-on-real-world-evidence-in-d

https://www.raps.org/News-and-Articles/News-Articles/2023/9/FDA%E2%80%99s-Califf-Expect-to-see-more-RWE-based-regulato
https://www.fda.gov/media/171667/download
https://www.merck.com/news/daiichi-sankyo-and-merck-announce-global-development-and-commercialization-collaboration-for-three-daiichi-sankyo-dxd-adcs/

Mr. Newirth is another DCVax-L survivor well surpassing 10 years following a 2012 diagnosis! Congratulations!
https://www.hawaii.edu/news/2017/03/30/newirth-laker-for-a-day/
2023 Interview:
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/












iclight, Britain's Medicines and Healthcare products Regulatory Agency (MHRA) will officially join #ProjectOrbis on January 1 and work with regulators in the US, Canada, Australia, Singapore and Switzerland on assessing new treatments. https://t.co/JjaXoq6yfh
Thanks for reposting that comment again—I hear you. It sounds like you’re misinformed and need more reliable information. Look beyond social media and seek trustworthy full-context sources…
For a reliable appraisal of the trial, its design, and the significance of the resulting survival data, it is advisable to refer to the analyses of qualified physicians. One such analysis can be found on the JAMA Oncology website where the independent peer review was published.
The forthcoming regulatory advisory from MHRA will further enlighten the significance the clinical data, and an assessment from MHRA may be available in advance of advisory decisions from FDA and regulators in other nations.
Additionally, I recommend that you review the forthcoming NICE appraisal of the DCVax-L cell-based technology when it is published.

https://www.nice.org.uk/guidance/indevelopment/gid-ta10143



https://www.fda.gov/about-fda/oncology-center-excellence/project-orbis




https://www.kcl.ac.uk/people/keyoumars-ashkan
The MIA was approved recently and is evidence of a regulatory filing plan that has begun to unfold. Further RA applications have been under construction for a significant period of time and include a voluminous catalog of clinical data and analyses. The announcements of regulatory filings are forthcoming.
The recent MIA approval is an important step in the regulatory process, and this commercial manufacturing license will allow DCVax-L to be exported globally from Sawston. The MIA is a prerequisite of the MAA application.
“Companies applying for a marketing authorisation (also known as a product licence) need to have a manufacturer licence first. The manufacturer licence will be granted first providing the product is in the process of being approved.” —MHRA
https://www.gov.uk/guidance/apply-for-manufacturer-or-wholesaler-of-medicines-licences
https://finance.yahoo.com/news/northwest-biotherapeutics-advent-bioservices-announce-234600744.html
https://finance.yahoo.com/news/northwest-biotherapeutics-announces-approval-pediatric-133900403.html
Another great article recommendation from LC—this time on the expanding universe of cell-based technologies … the JCI article link that you posted a couple weeks ago was very much appreciated too!
https://academic.oup.com/ajhp/advance-article-abstract/doi/10.1093/ajhp/zxad251/7289179
https://www.jci.org/articles/view/169314
https://www.raps.org/News-and-Articles/News-Articles/2023/9/FDA%E2%80%99s-Califf-Expect-to-see-more-RWE-based-regulato
https://clinicaltrials.gov/study/NCT04201873
Combo is King!
Grip, This study found that autologous tumor lystate-loaded dendritic cell vaccination plus standard of care (SOC) was associated with improved survival in both newly diagnosed & recurrent glioblastoma compared with matched controls who received SOC treatment. https://t.co/Uso7Pkh1Kt
I think that the Dane has some real foresight … let’s watch as more unfolds throughout the forthcoming regulatory phase of the process!
Three years ago was before this happened:
https://www.jci.org/articles/view/169314
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers




https://www.pharmaceutical-technology.com/news/mhra-revamps-uk-clinical-trial-regulation-with-the-promise-of-faster-timelines/
https://finance.yahoo.com/amphtml/news/northwest-biotherapeutics-announces-dr-linda-140000962.html
HyGro,
Thank you for highlighting FDA guidance. We are now in the era of RWD and immunotherapy!
Exciting times … Combo is King!

https://www.raps.org/news-and-articles/news-articles/2023/8/fda-finalizes-guidance-on-real-world-evidence-in-d
https://www.raps.org/News-and-Articles/News-Articles/2023/9/FDA%E2%80%99s-Califf-Expect-to-see-more-RWE-based-regulato
https://www.fda.gov/media/171667/download
https://www.uclahealth.org/cancer/researchers/spores/ucla-spore-brain-cancer/research-projects
https://finance.yahoo.com/news/northwest-biotherapeutics-announces-dr-linda-140000962.html
https://www.merck.com/news/daiichi-sankyo-and-merck-announce-global-development-and-commercialization-collaboration-for-three-daiichi-sankyo-dxd-adcs/




https://www.jci.org/articles/view/169314
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
https://hotb.org/wp-content/uploads/2022/05/spore-article-for-donors-4.18.22.pdf
https://www.uclahealth.org/news/brain-cancer-discovery-clinical-trials

https://www.fda.gov/drugs/news-events-human-drugs/50-years-progress-treating-patients-cancer

HyGro,
Thanks for the reply. That’s an interesting new study! UCLA is doing invaluable work and the NIH is wise to have supported their research for so many consecutive years.
UCLA developed the DCVax-L cell-based platform technology, and it will be exciting to see their further research involving other cancers beyond GBM and with other combination therapies!
The Nobel Prize-winning work of Dr. Steinman lives on at UCLA!
https://www.livescience.com/16354-nobel-prize-medicine-2011-immune-research.html
“I will respect the hard-won scientific gains of those physicians in whose steps I walk, and gladly share such knowledge as is mine with those who are to follow.”
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=172987533
https://clinicaltrials.gov/study/NCT03546361



HyGro,
Thanks for posting! UCLA is doing invaluable work and the NIH is wise to have supported their research for so many consecutive years.
UCLA developed the DCVax-L cell-based platform technology, and it will be exciting to see their further research!
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=172987533
“I will respect the hard-won scientific gains of those physicians in whose steps I walk, and gladly share such knowledge as is mine with those who are to follow.”
https://clinicaltrials.gov/study/NCT03546361

ex, Britain's Medicines and Healthcare products Regulatory Agency (MHRA) will officially join #ProjectOrbis on January 1 and work with regulators in the US, Canada, Australia, Singapore and Switzerland on assessing new treatments. https://t.co/JjaXoq6yfh
For a reliable appraisal of the trial, its design, and the significance of the resulting survival data, it is advisable to refer to the analyses of qualified physicians. Such an analysis can be found on the JAMA Oncology website where the independent peer review was published.
The forthcoming regulatory advisory from MHRA will further enlighten the significance the clinical data, and an assessment from MHRA may be available in advance of advisory decisions from FDA and regulators in other nations.
Additionally, I recommend that you review the forthcoming NICE appraisal of the DCVax-L cell-based technology when it is published.

https://www.nice.org.uk/guidance/indevelopment/gid-ta10143



https://www.fda.gov/about-fda/oncology-center-excellence/project-orbis




https://www.kcl.ac.uk/people/keyoumars-ashkan
The MIA was approved recently and is evidence of a regulatory filing plan that has begun to unfold. Further RA applications have been under construction for a significant period of time and include a voluminous catalog of clinical data and analyses. The announcements of regulatory filings are forthcoming.
The recent MIA approval is an important step in the regulatory process, and this commercial manufacturing license will allow DCVax-L to be exported globally from Sawston. The MIA is a prerequisite of the MAA application.
“Companies applying for a marketing authorisation (also known as a product licence) need to have a manufacturer licence first. The manufacturer licence will be granted first providing the product is in the process of being approved.” —MHRA
https://www.gov.uk/guidance/apply-for-manufacturer-or-wholesaler-of-medicines-licences
https://finance.yahoo.com/news/northwest-biotherapeutics-advent-bioservices-announce-234600744.html
https://finance.yahoo.com/news/northwest-biotherapeutics-announces-approval-pediatric-133900403.html
ex,
I hear you—controls are an interesting topic with regard to DCVax-L. Safety and efficacy data have been gathered for DCVax-L from the P3 clinical study that had external controls and from clinical studies having placebo controls. The data record that exists in total reflects DCVax-L as a monotherapy and DCVax-L in combination with other agents…
The placebo-controlled pembrolizumab combo study has significant interim survival data.
https://clinicaltrials.gov/ct2/show/NCT04201873
In the combo trial, patients who receive DCVax-L + placebo are being compared to patients who receive DCVax-L + pembrolizumab.
The control arm is receiving DCVax-L. While DCVax-L is not the current SOC, it is highly significant and notable that DCVax-L is being permitted to fill the role of SOC as the best available therapy within this combo study for rGBM. The combo trial also follows the P3 in sequence which in itself validates the findings of the P3 study—the placebo group in the PD1 study would not be receiving DCVax-L today if the preceding P3 trial had not proven its efficacy.
https://www.jci.org/articles/view/169314
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
https://finance.yahoo.com/news/northwest-biotherapeutics-announces-dr-linda-140000962.html
LC, #CancerResearch #Cancers #cancermoonshot #GBM #Immunotherapy #oncology $nwbo #murcidencel #DCVax #CellTherapy Explore Professor Keyoumars Ashkan's passion for neurosurgery and his love for @KingsCollegeNHS below as part of their #BrilliantPeople series🧠👇@KingsNeuro @KingsIoPPN https://t.co/yGKt2681Ie
I hear you—exciting times! I agree that UCLA deserves a lot of recognition for their role, but the NIH and King’s College deserve recognition too—the DCVax-L cell-based platform technology was developed with the support of public dollars and with the scientific contributions of many physicians.
Dr. Steinman’s invaluable work is also part of the DCVax-L story!
The Nobel Prize was a harbinger of the DCVax-L cell-based platform technology! https://t.co/YW8un0Kujq pic.twitter.com/W4R8rcXVOS
https://www.livescience.com/16354-nobel-prize-medicine-2011-immune-research.html
https://finance.yahoo.com/news/northwest-biotherapeutics-announces-dr-linda-140000962.html
The U.S. NIH has supported the DCVax-L platform for years, and the NIH renewed its support in 2022 to continue to fund the development of this technology and to fund research into combination therapies.
NIH-funded DCVax-L research is ongoing at UCLA today.
NIH grants are peer-reviewed and the research that NIH funds is highly scrutinized in advance of the award and also intermittently for the term of the various research projects.

https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers



https://www.uclahealth.org/news/brain-cancer-discovery-clinical-trials
https://www.uclahealth.org/news/ucla-received-590-million-in-nih-funding-second-highest-total-for-academic-medical-centers-in-2020
https://www.fda.gov/science-research/advancing-regulatory-science/fda-nih-joint-leadership-council-charter



LC,
Yes, the independent peer review of the P3 was published in JAMA Oncology!
https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/


https://ceoworld.biz/2022/04/05/top-5-medical-journals-in-the-world-everyone-should-know-about/
HyGro,
Dr. Liau has joined the SAB at NWBO! UCLA has reported the further development of the platform technology in combination with other technologies and for indications beyond GBM!
Combo is King!
https://www.jci.org/articles/view/169314
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
https://hotb.org/wp-content/uploads/2022/05/spore-article-for-donors-4.18.22.pdf
https://www.uclahealth.org/news/brain-cancer-discovery-clinical-trials


https://finance.yahoo.com/news/northwest-biotherapeutics-announces-dr-linda-140000962.html
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=172749240
HyGro,
Dr. Liau has joined the SAB at NWBO!
Combo is King!
https://www.jci.org/articles/view/169314
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
https://hotb.org/wp-content/uploads/2022/05/spore-article-for-donors-4.18.22.pdf
https://www.uclahealth.org/news/brain-cancer-discovery-clinical-trials


https://finance.yahoo.com/news/northwest-biotherapeutics-announces-dr-linda-140000962.html
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=172749240
Yes, that’s a good idea! Thanks HyGro!
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
https://hotb.org/wp-content/uploads/2022/05/spore-article-for-donors-4.18.22.pdf
https://www.uclahealth.org/news/brain-cancer-discovery-clinical-trials



https://finance.yahoo.com/news/northwest-biotherapeutics-announces-dr-linda-140000962.html
HyGro,
Thanks for the reply. Dr. Linda Liau is now on the NWBO SAB! Exciting times!
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=172875571
learningcurve2020,
I have been recommending the JCI article that you provided for us recently. It is highly relevant and has further enlightened our understanding of the significance of the DCVax-L cell-based technology as a platform.
Combo is King!
https://www.jci.org/articles/view/169314
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
HyGro, This study found that autologous tumor lystate-loaded dendritic cell vaccination plus standard of care (SOC) was associated with improved survival in both newly diagnosed & recurrent glioblastoma compared with matched controls who received SOC treatment. https://t.co/Uso7Pkh1Kt Britain's Medicines and Healthcare products Regulatory Agency (MHRA) will officially join #ProjectOrbis on January 1 and work with regulators in the US, Canada, Australia, Singapore and Switzerland on assessing new treatments. https://t.co/JjaXoq6yfh #CancerResearch #Cancers #cancermoonshot #GBM #Immunotherapy #oncology $nwbo #murcidencel #DCVax #CellTherapy https://t.co/BnJHM3myP9 Explore Professor Keyoumars Ashkan's passion for neurosurgery and his love for @KingsCollegeNHS below as part of their #BrilliantPeople series🧠👇@KingsNeuro @KingsIoPPN https://t.co/yGKt2681Ie
The DCVax-L cell-based platform technology is a fascinating medical innovation to research and read about. In combination with other technologies, it is even more intriguing!
Combo is King!
https://www.jci.org/articles/view/169314


https://doi.org/10.3171/2020.12.FOCUS20954
https://finance.yahoo.com/news/northwest-biotherapeutics-announces-dr-linda-140000962.html
The U.S. NIH has supported the DCVax-L platform for years, and the NIH renewed its support in 2022 to continue to fund the development of this technology and to fund research into combination therapies.
NIH-funded DCVax-L research is ongoing at UCLA today.
NIH grants are peer-reviewed and the research that NIH funds is highly scrutinized in advance of the award and also intermittently for the term of the various research projects.

https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers


https://www.uclahealth.org/news/brain-cancer-discovery-clinical-trials
https://www.uclahealth.org/news/ucla-received-590-million-in-nih-funding-second-highest-total-for-academic-medical-centers-in-2020

https://www.fda.gov/science-research/advancing-regulatory-science/fda-nih-joint-leadership-council-charter



FDA is of course the focus, but I think that the U.K. will prove vastly significant too! King’s College and the Specials Program and MHRA developments are all very significant!
Additionally, I recommend that you review the forthcoming NICE appraisal of the DCVax-L cell-based technology. Stay tuned!

https://www.nice.org.uk/guidance/indevelopment/gid-ta10143



https://www.fda.gov/about-fda/oncology-center-excellence/project-orbis




https://www.kcl.ac.uk/people/keyoumars-ashkan
The MIA was approved recently and is evidence of a regulatory filing plan that has begun to unfold. Further RA applications have been under construction for a significant period of time and include a voluminous catalog of clinical data and analyses. The announcements of regulatory filings are forthcoming.
The recent MIA approval is an important step in the regulatory process, and this commercial manufacturing license will allow DCVax-L to be exported globally from Sawston. The MIA is a prerequisite of the MAA application.
“Companies applying for a marketing authorisation (also known as a product licence) need to have a manufacturer licence first. The manufacturer licence will be granted first providing the product is in the process of being approved.” —MHRA
https://www.gov.uk/guidance/apply-for-manufacturer-or-wholesaler-of-medicines-licences
https://finance.yahoo.com/news/northwest-biotherapeutics-advent-bioservices-announce-234600744.html
https://finance.yahoo.com/news/northwest-biotherapeutics-announces-approval-pediatric-133900403.html
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=172639755%5B/color%5D

https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/
Seek trustworthy full-context sources and independently verify facts. Consider statements published online from those who disclose their conflicts, and weigh opinions critically.


ex,
Thanks for the reply. Yes, it is exciting to see immunotherapy combos emerging from clinical trials with brilliant efficacy—Combo is King!
Nobody seems to ever mention the DCVax-L P3 without also mentioning the UCLA combo!
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/
HyGro, Explore Professor Keyoumars Ashkan's passion for neurosurgery and his love for @KingsCollegeNHS below as part of their #BrilliantPeople series🧠👇@KingsNeuro @KingsIoPPN https://t.co/yGKt2681Ie
You are again reposting disinformation from Reddit and your assertions are mixed up and misleading, but thank you for again highlighting the combo study that is ongoing at UCLA today …
Combo is King!
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=172749240
Seek verifiable sources and consider refocusing on what matters. I believe that you are distracted from the vast significance of Liau’s work and the further development of this technology in combination with other technologies. DCVax-L is already supported by peer-reviewed clinical evidence, but there is much more to the story that is yet to unfold, and the SP will follow the regulatory developments worldwide.
Targeted individualized combo therapies are emerging as the defining feature of a new era of oncology. Just look at the impact that PD1 blockade technology has had on the field of oncology in recent years!

https://www.fda.gov/drugs/news-events-human-drugs/50-years-progress-treating-patients-cancer
The P3 study of the DCVax-L cell-based platform technology is a landmark trial.
The P3 data were peer reviewed by independent physicians and qualified statisticians—please refer to JAMA Oncology. You might consider researching credible full-context sources to gather reliable information.
The DCVax-L cell-based technology has been under clinical investigation for a significant period of time, and the overall clinical data in total include three trials spanning many years. Safety and efficacy data have been gathered from trials with external controls and trials with placebo controls. The data include DCVax-L as a monotherapy and in combination with other agents.
The interim PD1 combo data are brilliant, and the trial is significant for a number of reasons relating to FDA guidelines and NIH peer-reviewed grant funding.

https://clinicaltrials.gov/ct2/show/NCT04201873
https://www.fda.gov/media/120721/download
In the combo trial, patients who receive DCVax-L + placebo are being compared to patients who receive DCVax-L + pembrolizumab.
The control arm is receiving DCVax-L. While DCVax-L is not the current SOC, it is highly significant and notable that DCVax-L is being permitted to fill the role of SOC as the best available therapy within this combo study for rGBM. The combo trial also follows the P3 in sequence which in itself validates the findings of the P3 study—the placebo group in the PD1 study would not be receiving DCVax-L today if the preceding P3 trial had not proven its efficacy.

The NIH is the most significant player in this game—If they had any doubt about the P3, the NIH would not continue their support and the FDA would not have permitted the combo trial to commence or continue.
https://clinicaltrials.gov/ct2/show/NCT04201873
The U.S. NIH has supported the DCVax-L platform for years, and the NIH renewed its support in 2022 to continue to fund the development of this technology and to fund research into combination therapies.
NIH-funded DCVax-L research is ongoing at UCLA today.
NIH grants are peer-reviewed and the research that NIH funds is highly scrutinized in advance of the award and also intermittently for the term of the various research projects.

Mr. Newirth is another DCVax-L survivor well surpassing 10 years following a 2012 diagnosis! Congratulations!
https://www.hawaii.edu/news/2017/03/30/newirth-laker-for-a-day/
June 13, 2023 Interview:
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers



https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/















LC,
You are 100% correct—that would be stupid!

Seek trustworthy full-context sources and verify statements posted online. Do your own research.
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=172848430


https://jamanetwork.com/journals/jamaoncology/fullarticle/2798847
LC,
Thanks for the reply. Anyone may email the company, and there is nothing that needs to be settled.
https://doi.org/10.3171/2020.12.FOCUS20954
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
https://finance.yahoo.com/news/northwest-biotherapeutics-announces-dr-linda-140000962.html


HyGro, Explore Professor Keyoumars Ashkan's passion for neurosurgery and his love for @KingsCollegeNHS below as part of their #BrilliantPeople series🧠👇@KingsNeuro @KingsIoPPN https://t.co/yGKt2681Ie
Thanks for the reply, but you are again reposting disinformation from Reddit.
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=172749240
Seek verifiable sources and consider refocusing on what matters. I believe that you are distracted from the vast significance of Liau’s work and the further development of this technology in combination with other technologies. DCVax-L is already supported by peer-reviewed clinical evidence, but there is much more to the story that is yet to unfold, and the SP will follow the regulatory developments worldwide.
Targeted individualized combo therapies are emerging as the defining feature of a new era of oncology. Just look at the impact that PD1 blockade technology has had on the field of oncology in recent years!

https://www.fda.gov/drugs/news-events-human-drugs/50-years-progress-treating-patients-cancer
The P3 study of the DCVax-L cell-based platform technology is a landmark trial.
The P3 data were peer reviewed by independent physicians and qualified statisticians—please refer to JAMA Oncology. You might consider researching credible full-context sources to gather reliable information.
The DCVax-L cell-based technology has been under clinical investigation for a significant period of time, and the overall clinical data in total include three trials spanning many years. Safety and efficacy data have been gathered from trials with external controls and trials with placebo controls. The data include DCVax-L as a monotherapy and in combination with other agents.
The interim PD1 combo data are brilliant, and the trial is significant for a number of reasons relating to FDA guidelines and NIH peer-reviewed grant funding.

https://clinicaltrials.gov/ct2/show/NCT04201873
https://www.fda.gov/media/120721/download
In the combo trial, patients who receive DCVax-L + placebo are being compared to patients who receive DCVax-L + pembrolizumab.
The control arm is receiving DCVax-L. While DCVax-L is not the current SOC, it is highly significant and notable that DCVax-L is being permitted to fill the role of SOC as the best available therapy within this combo study for rGBM. The combo trial also follows the P3 in sequence which in itself validates the findings of the P3 study—the placebo group in the PD1 study would not be receiving DCVax-L today if the preceding P3 trial had not proven its efficacy.

The NIH is the most significant player in this game—If they had any doubt about the P3, the NIH would not continue their support and the FDA would not have permitted the combo trial to commence or continue.
https://clinicaltrials.gov/ct2/show/NCT04201873
The U.S. NIH has supported the DCVax-L platform for years, and the NIH renewed its support in 2022 to continue to fund the development of this technology and to fund research into combination therapies.
NIH-funded DCVax-L research is ongoing at UCLA today.
NIH grants are peer-reviewed and the research that NIH funds is highly scrutinized in advance of the award and also intermittently for the term of the various research projects.

Mr. Newirth is another DCVax-L survivor well surpassing 10 years following a 2012 diagnosis! Congratulations!
https://www.hawaii.edu/news/2017/03/30/newirth-laker-for-a-day/
June 13, 2023 Interview:
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers



https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/















HyGro, Explore Professor Keyoumars Ashkan's passion for neurosurgery and his love for @KingsCollegeNHS below as part of their #BrilliantPeople series🧠👇@KingsNeuro @KingsIoPPN https://t.co/yGKt2681Ie
Thank you for highlighting the NIH and the vast significance of Liau’s work and the further development of this technology in combination with other technologies. DCVax-L is already supported by peer-reviewed clinical evidence, but there is much more to the story that is yet to unfold.
Targeted individualized combo therapies are emerging as the defining feature of a new era of oncology. Just look at the impact that PD1 blockade technology has had on the field of oncology in recent years!

https://www.fda.gov/drugs/news-events-human-drugs/50-years-progress-treating-patients-cancer
The P3 study of the DCVax-L cell-based platform technology is a landmark trial.
The P3 data were peer reviewed by independent physicians and qualified statisticians—please refer to JAMA Oncology. You might consider researching credible full-context sources to gather reliable information.
The DCVax-L cell-based technology has been under clinical investigation for a significant period of time, and the overall clinical data in total include three trials spanning many years. Safety and efficacy data have been gathered from trials with external controls and trials with placebo controls. The data include DCVax-L as a monotherapy and in combination with other agents.
The interim PD1 combo data are brilliant, and the trial is significant for a number of reasons relating to FDA guidelines and NIH peer-reviewed grant funding.

https://clinicaltrials.gov/ct2/show/NCT04201873
https://www.fda.gov/media/120721/download
In the combo trial, patients who receive DCVax-L + placebo are being compared to patients who receive DCVax-L + pembrolizumab.
The control arm is receiving DCVax-L. While DCVax-L is not the current SOC, it is highly significant and notable that DCVax-L is being permitted to fill the role of SOC as the best available therapy within this combo study for rGBM. The combo trial also follows the P3 in sequence which in itself validates the findings of the P3 study—the placebo group in the PD1 study would not be receiving DCVax-L today if the preceding P3 trial had not proven its efficacy.

The NIH is the most significant player in this game—If they had any doubt about the P3, the NIH would not continue their support and the FDA would not have permitted the combo trial to commence or continue.
https://clinicaltrials.gov/ct2/show/NCT04201873
The U.S. NIH has supported the DCVax-L platform for years, and the NIH renewed its support in 2022 to continue to fund the development of this technology and to fund research into combination therapies.
NIH-funded DCVax-L research is ongoing at UCLA today.
NIH grants are peer-reviewed and the research that NIH funds is highly scrutinized in advance of the award and also intermittently for the term of the various research projects.

Mr. Newirth is another DCVax-L survivor well surpassing 10 years following a 2012 diagnosis! Congratulations!
https://www.hawaii.edu/news/2017/03/30/newirth-laker-for-a-day/
June 13, 2023 Interview:
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers



https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/















HyGro,
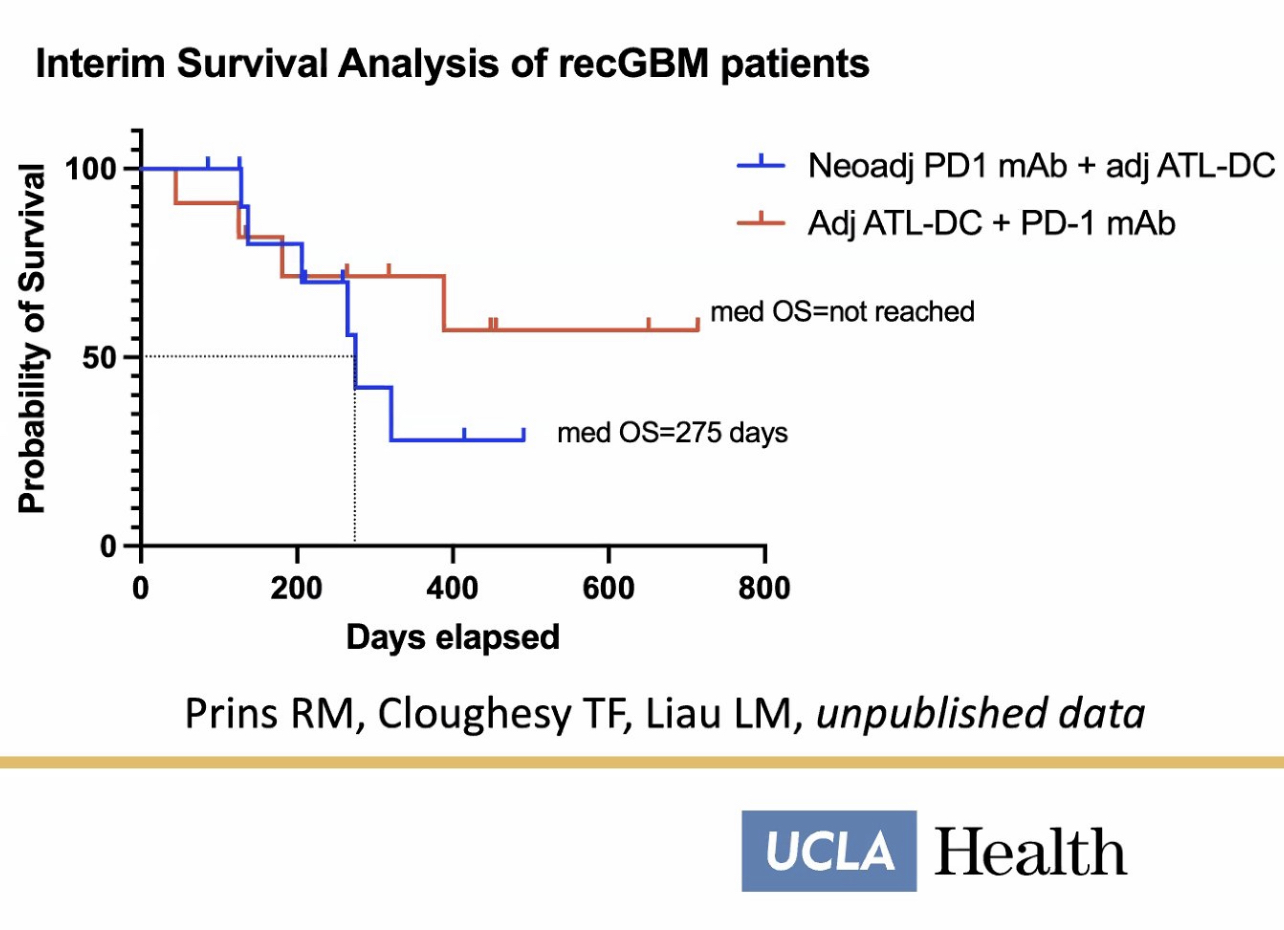
What do you think about the interim survival data of the UCLA combo study?
Merck is doing the Pragmatica trial that is described in the NCI interview linked below.
https://www.urotoday.com/video-lectures/asco-gu-2023/video/3244-new-nci-fda-unit-aims-to-streamline-drug-development-for-cancer-patients-michael-morris.html
https://www.cancer.gov/types/lung/research/pragmatica-lung-cancer-trial
Exactly.
Yes, the original study took many years because they captured OS data and participants lived long, but any studies following commercialization could be much more swift.
DCVax-L has such a solid safety profile—I think that it can move rapidly through clinical investigations for other cancers once it is initially approved for GBM.
This was another insightful idea that was sparked by learningcurve2020’s comment yesterday about the possibility of future combinations with
DCVax-L
https://www.urotoday.com/video-lectures/asco-gu-2023/video/3244-new-nci-fda-unit-aims-to-streamline-drug-development-for-cancer-patients-michael-morris.html


https://www.jci.org/articles/view/169314
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
brazen22,
You are never going to see the FDA or MHRA allow trials to treat patients like lab rats.
If you prefer a randomized placebo design, then you’ll love the pembrolizumab PD1 blockade combo study that is ongoing at UCLA today. The study has a placebo design because the placebo participants are receiving DCVax-L as the best available therapy for rGBM.
https://classic.clinicaltrials.gov/ct2/show/NCT04201873
https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/
brazen22,
Yes, I read that one. In my view, it is very similar to your other posted citations that all make the case for phase 4 investigation. Data should be gathered in support of all newly commercialized drugs, and I think that any physician would support further data collection — phase 4 can also lead to further development and broader application across oncology or perhaps beyond oncology.
The reason why we cannot expect a placebo design is because GBM is somewhat unique, and a placebo design would require trial physicians to treat participants without clinically appropriate compassion.
The UCLA combo study is using a placebo design because the placebo participants are receiving a treatment that is efficacious for rGBM.
https://www.jci.org/articles/view/169314
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
brazen22,
I highly recommend that you study the article that learningcurve2020 provided for us yesterday. It is highly relevant and will help enlighten your understanding of the significance of the DCVax-L cell-based platform technology.
Combo is King!
https://www.jci.org/articles/view/169314
HyGro,
You posted a hundred times referring to the Keytruda combo with “DCVax-L” Now you want us to believe that it’s not DCVax-L?


https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers