Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
...IMO "fair value" at this time: $.75 !
Unilife and Stason Pharmaceuticals Sign Asian Distribution Agreement for the Unitract(TM) 1mL Safety Syringe
Date : 03/10/2010 @ 8:00AM
Source : PR Newswire
Stock : Unilife (MM) (UNIS)
http://ih.advfn.com/p.php?pid=nmona&cb=1268227212&article=41908671&symbol=N^UNIS
LEWISBERRY, Pa., March 10 /PRNewswire-FirstCall/ --
Unilife Corporation ("Unilife" or "Company") today announced that it has signed an exclusive five-year agreement with Stason Pharmaceuticals Inc ("Stason"), a U.S.-based pharmaceutical company, to market the Unitract(TM) 1mL safety syringe ("Product") in Japan, China and Taiwan ("designated territories"). Stason's Taiwan-based strategic alliance affiliate, Standard Chem. and Pharm. Co., LTD ("Standard"), will act as the primary distributor of the product within these designated territories.
This agreement includes a requirement for Stason to purchase a minimum of one million units of the Unitract(TM) 1mL syringe per year during the term of the contract (subject to annual review). Stason is placing an immediate annual order with Unilife for the purchase of one million units of the Unitract(TM) 1mL syringe. Other countries within South-East Asia may be added to the list of designated territories where Stason has exclusive rights to sell the Product if the parties reach agreement on minimum orders for those countries.
Stason CEO Harry Fan stated: "We believe strongly in the market for the Unitract(TM) safety syringe and its potential to help decrease needlestick injuries around the world. We are excited to be one of the first distributors of this revolutionary device and to have the opportunity to exclusively distribute the Unitract(TM) syringe to our customers in Asia. In particular, we believe there are significant opportunities for pharmaceutical companies to gain a competitive market advantage by supplying their injectable drugs in kits containing the drug vial and units of the Unitract(TM) 1mL Syringe."
Stason Vice President, Corporate Development Diana Wood, added: "This strategic alliance with Unilife also represents our joint vision where drugs and devices come together to deliver medications more comfortably to patients and more safely for hospital staff."
Unilife CEO Alan Shortall stated: "With manufacturing of our Unitract(TM) 1mL syringes underway at our FDA-certified facility in Pennsylvania, we are excited to move forward with this initial distribution agreement and initial order for one million Unitract(TM) safety syringes with Stason Pharmaceuticals and Standard Chem. and Pharma. We believe there is a significant market opportunity for our safety syringes throughout the Asia-Pacific region, especially within fast-growing nations where there is both an appreciation for innovative US-manufactured technology and a desire to use premium safety medical devices which can minimize the transmission of blood-borne pathogens such as HIV and hepatitis C within healthcare facilities.
"We look forward to working closely with Stason and Standard to develop these markets and secure sustained rates of growth for the sale of our products across Asia. We are also continuing to negotiate with other pharmaceutical and healthcare companies across the world for distribution rights for the Unitract(TM) 1mL syringes. We expect that additional agreements will be signed in conjunction with the continued global rollout of the Unitract(TM) 1mL syringes," Mr. Shortall said.
The Unitract(TM) range of 1mL syringes is the world's first and only known syringe that allows operators to control the speed of automatic needle retraction directly from the patient's body into the barrel of the syringe where it is locked in place. The products are well positioned to help prevent the transmission of blood-borne diseases such as HIV and hepatitis C via aerosol dispersal, needlestick injuries and syringe reuse. Primary target markets of the products include healthcare facilities, pharmaceutical companies and patients who self-administer prescription medication.
Also, while Unilife continues to have a relationship with Shanghai Kindly Enterprise Development Group Company Ltd ("KDL") for the assembly of its Unitract(TM) 1mL syringes within China on a per order basis, other agreements between Unilife and KDL relating to the distribution of the product within China and other agreed territories have expired. Stason has advised the Company of a preference to receive units of the Unitract(TM) 1mL syringe that are assembled at Unilife's FDA-registered facilities in the U.S. State of Pennsylvania.
About Unilife Corporation
Unilife Corporation is a U.S.-based medical device company focused on the design, development, manufacture and supply of a proprietary range of retractable syringes. Primary target customers for Unilife products include pharmaceutical manufacturers, suppliers of medical equipment to healthcare facilities and patients who self-administer prescription medication. These patent-protected syringes incorporate automatic and fully-integrated safety features which are designed to protect those at risk of needlestick injuries and unsafe injection practices. Unilife is ISO 13485 certified and has FDA-registered medical device manufacturing facilities in Pennsylvania.
About Stason Pharmaceuticals Inc
Stason Pharmaceuticals, Inc is a California-based pharmaceutical company involved in drug development, manufacturing, importation exportation and the licensing and marketing of both generic and branded products. The company's primary area of development is oncology, as well supportive products for the treatment of side effects related to cancer therapy. Additional therapeutic areas include cardiovascular, central nervous system, autoimmune and endocrine disorders. Stason's most important strategic alliance affiliation in Asia is with Standard Chem. And Pharm. Co., LTD. Standard was established in Taiwan in 1967 and has a number of subsidiaries active across North America and Asia which are focused on ethical and OTC drugs, APIs, medical devices and health food supplements.
This press release contains forward-looking statements. All statements that address operating performance, events or developments that we expect or anticipate will occur in the future are forward-looking statements. These forward-looking statements are based on management's beliefs and assumptions and on information currently available to our management. Our management believes that these forward-looking statements are reasonable as and when made. However, you should not place undue reliance on any such forward-looking statements because such statements speak only as of the date when made. We do not undertake any obligation to publicly update or revise any forward-looking statements, whether as a result of new information, future events or otherwise, except as required by law. In addition, forward-looking statements are subject to certain risks and uncertainties that could cause actual results, events and developments to differ materially from our historical experience and our present expectations or projections. These risks and uncertainties include, but are not limited to, those described in "Item 1A. Risk Factors" and elsewhere in our registration statement on Form 10 and those described from time to time in other reports which we file with the Securities and Exchange Commission.
General: UNIS-G
Investor Contacts (US):
Todd Fromer / Garth Russell Stuart Fine
KCSA Strategic Communications Carpe DM Inc
Phone + 1 212-682-6300 Phone + 1 908-469-1788
Investor Contacts (Australia):
Jeff Carter
Unilife Corporation
Phone + 61 2 8346 6500
DATASOURCE: Unilife Corporation
CONTACT: Investor Contacts (US): Todd Fromer or Garth Russell, both ofKCSA Strategic Communications, +1-212-682-6300 or Stuart Fine of Carpe DM Inc,+1-908-469-1788 or Investor Contacts (Australia): Jeff Carter of UnilifeCorporation, +61-2-8346-6500
Discovery Labs Reports Fourth Quarter 2009 Financial Results and Provides Business Update
Date : 03/09/2010 @ 11:45PM
Source : GlobeNewswire Inc.
Stock : Discovery Laboratories, Inc. (DSCO)
http://ih.advfn.com/p.php?pid=nmona&article=41907968&symbol=DSCO
Discovery Labs Reports Fourth Quarter 2009 Financial Results and Provides Business Update
WARRINGTON, Pa., March 10, 2010
GLOBE NEWSWIRE
Discovery Laboratories, Inc. (Nasdaq:DSCO) today announces financial results for the fourth quarter ended December 31, 2009 and provides a business update.
For the quarter ended December 31, 2009, the Company reported a net loss of $6.1 million (or $0.05 per share) on 125.6 million weighted average common shares outstanding compared to a net loss of $8.5 million (or $0.08 per share) on 100.5 million weighted average common shares outstanding for the same period in 2008. Net cash burn for the fourth quarter of 2009 was $1.9 million as a result of $6.0 million used for operating activities and $0.2 million used for debt service, partially offset by aggregate proceeds of $4.3 million from the issuance of 4.6 million shares of common stock pursuant to financings under the Company's Committed Equity Financing Facilities (CEFFs).
For the year ended December 31, 2009, the Company reported a net loss of $30.2 million (or $0.26 per share) on 115.2 million weighted average common shares outstanding compared to a net loss of $39.1 million (or $0.40 per share) on 98.1 million weighted average common shares outstanding for the same period in 2008.
As of December 31, 2009, the Company had cash and marketable securities of $15.7 million. In February 2010 the Company completed a public offering of common stock and warrants resulting in net proceeds of $15.1 million. Additionally, the Company currently has two CEFFs that, subject to certain conditions, including price and volume limitations, may allow the Company in the future to raise additional capital to support its business plans. Relevant details regarding the recent financing and the CEFF arrangements are included in the "Additional Financial Information" section of this release. The Company had 126.4 and 153.9 million common shares outstanding as of December 31, 2009 and March 10, 2010 respectively.
W. Thomas Amick, the Company's Chairman and interim Chief Executive Officer, commented, "We believe that our proprietary KL4 surfactant technology has the potential to significantly improve the medical outcomes of patients, from premature infants to adults, suffering from debilitating respiratory disorders. Our near term priorities are to satisfy the FDA's remaining requirements and potentially gain approval of Surfaxin for RDS in 2011, advance the development of Aerosurf®, which we believe holds the promise to significantly advance neonatal respiratory medicine, and continue to strengthen the long-term financial position of our company. We continue to be engaged in discussions with potential strategic and financial partners that, if successful, will provide the financial resources needed to potentially advance the development our KL4 surfactant pipeline and maximize shareholder value."
Selected key initiatives and anticipated milestones include:
Surfaxin for RDS: In response to written guidance recently received from the U.S. Food and Drug Administration (FDA), the Company will now focus on a pathway that would entail solely performing additional preclinical work to potentially address the sole remaining Chemistry, Manufacturing & Control issue regarding the final validation of a fetal rabbit Biological Activity Test (BAT, an important quality control release and stability test) necessary for Surfaxin approval. A key component of this approach is to first satisfactorily optimize and revalidate the BAT and those efforts are currently underway and presently meeting all pre-specified acceptance criteria. The Company believes that the optimization and revalidation of the BAT will be completed in the second quarter of 2010. Upon successful completion, the Company anticipates conducting a series of prospectively-designed, side-by-side preclinical studies employing the optimized BAT and a well-established preterm lamb model of RDS. The Company plans to seek FDA advice regarding important aspects of the preclinical program, including study design and appropriate success criteria. We believe a Complete Response could be submitted to the FDA in the first quarter of 2011.
Our neonatal pipeline programs, Surfaxin®, Surfaxin LS™ and Aerosurf®, have the potential to greatly improve the management of RDS and represent the opportunity, over time, to expand the current RDS estimated worldwide annual market of $200 million to a $1 billion opportunity. Surfaxin LS is a lyophilized (dry powder) formulation of KL4 surfactant intended to improve product ease of use for healthcare practitioners, eliminate the need for cold-chain storage, and potentially further improve product clinical performance. Aerosurf, our aerosolized KL4 surfactant, holds the promise to significantly expand the use of surfactant therapy in pediatric respiratory medicine by providing neonatologists with a means of delivering KL4 surfactant while potentially avoiding the risks associated with invasive endotracheal intubation and mechanical ventilation. The Company is currently advancing its preclinical development activities and preparing to further engage the FDA and foreign regulators with respect to a planned Phase 3 clinical program for Surfaxin LS and a Phase 2 clinical program for Aerosurf and intends to initiate these programs upon final determination of the regulatory strategy and after securing appropriate strategic alliances and necessary capital.
Discovery Labs is conducting a Phase 2 clinical trial to determine whether Surfaxin improves lung function and reduces the duration and related risk-exposure of mechanical ventilation in children up to two years of age diagnosed with Acute Respiratory Failure (ARF). ARF is a severe respiratory disorder associated with lung injury, often entailing surfactant dysfunction. ARF occurs after patients have been exposed to serious respiratory infections, such as influenza (including the type A serotype referred to as H1N1) or respiratory syncytial virus (RSV). Hospitalization following influenza or other viral infection is associated with high morbidity and significant healthcare costs. Enrollment is expected to be complete in March 2010 with top-line results becoming available in the second quarter of 2010.
Aerosolized KL4 surfactant is being evaluated in an investigator-initiated Phase 2a clinical trial in Cystic Fibrosis (CF) patients. The trial has been designed to assess the safety, tolerability and short-term effectiveness (via improvement in mucociliary clearance) of aerosolized KL4 surfactant in CF patients. The trial is being conducted at The University of North Carolina and supported by the Cystic Fibrosis Foundation. Results are anticipated in the second quarter of 2010.
An important business initiative for the Company in 2010 is to strengthen its long-term strategic and financial position in order to meaningfully advance its promising KL4 surfactant pipeline and maximize shareholder value. The Company continues to assess various strategic and financial alternatives to secure the necessary resources to potentially advance its development programs. Although a key priority for the Company is to secure strategic partners and capital to potentially support its ongoing research and development activities and its future financial condition, there can be no assurance that any strategic alliance or other financing alternatives will be successfully concluded.
Additionally, as of December 31, 2009 the Company had $10.5 million outstanding under its loan with Quintiles (formerly Novaquest). The outstanding principal and all accrued interest is due and payable on April 30, 2010. The Company's plans include pursuing a potential strategic restructuring of this loan with Quintiles and assessing alternative means of financing its payment; however, there can be no assurance that any such restructuring will occur or financing alternatives will be obtained.
Readers are referred to, and encouraged to read in its entirety, the Company's Annual Report on Form 10-K for the fiscal year ended December 31, 2009, which includes full details on the Company's business plans and operations, financial condition and results of operations. The Company's audited financial statements for the year ending December 31, 2009, are accompanied by an unqualified audit opinion from Ernst & Young LLP, the Company's independent public accounting firm, which includes a "going concern" explanatory statement. The Company is providing this information to comply with Nasdaq Marketplace Rule 4350(b)(1)(B), which requires an issuer that receives an audit report containing a going concern explanatory statement to make a public announcement through the news media disclosing the receipt of such statement.
Additional Selected Financial Information
On February 23, 2010, the Company completed a public offering of common stock and warrants resulting in gross proceeds of $16,500,000 from the issuance of 27,500,000 shares of common stock and warrants to purchase 13,750,000 shares. The shares and warrants were priced at $0.60 per unit and the warrants are exercisable for cash, except in certain circumstances provided therein, for a period of five years at an exercise price of $0.85 per share of common stock. Net proceeds, after underwriting discounts and commissions and other fees and expenses, were $15.1 million.
The Company currently has two CEFFs that (subject to certain conditions, including price and volume limitations) may allow the Company to raise additional capital to support its business plans. Under the December 2008 CEFF, currently there are approximately 7.1 million shares (not to exceed an aggregate $17.7 million) available for issuance, provided that the volume-weighted average price per share on each trading day in the draw-down period must be at least equal to the greater of $0.60 or 90% of the closing market price on the trading day immediately preceding the draw-down period. The expiration date of the December 2008 CEFF is February 2011. Under the May 2008 CEFF, currently there are approximately 12.8 million shares (not to exceed an aggregate of $51.7 million) available for issuance, provided that the average price on each trading day in the draw-down period must be at least equal to the greater of $1.15 or 90% of the closing market price on the trading day immediately preceding the draw-down period. The expiration date of the May 2008 CEFF is June 2011.
About Discovery Labs
Discovery Laboratories, Inc. is a biotechnology company developing Surfactant Therapies for respiratory diseases. Surfactants are produced naturally in the lungs and are essential for breathing. Discovery Labs' novel proprietary KL4 Surfactant Technology produces a synthetic, peptide-containing surfactant that is structurally similar to pulmonary surfactant and is being developed in liquid, aerosol or lyophilized formulations. In addition, Discovery Labs' proprietary Capillary Aerosolization Technology produces a dense aerosol, with a defined particle size that is capable of potentially delivering aerosolized KL4 surfactant to the deep lung without the complications currently associated with liquid surfactant administration. Discovery Labs believes that its proprietary technology platform makes it possible, for the first time, to develop a significant pipeline of surfactant products to address a variety of respiratory diseases for which there frequently are few or no approved therapies. For more information, please visit our website at www.Discoverylabs.com.
Forward-Looking Statements
To the extent that statements in this press release are not strictly historical, all such statements are forward-looking, and are made pursuant to the safe harbor provisions of the Private Securities Litigation Reform Act of 1995. These forward-looking statements are subject to certain risks and uncertainties that could cause actual results to differ materially from the statements made. Examples of such risks and uncertainties are: risks relating to the rigorous regulatory requirements required for approval of any drug or drug-device combination products that Discovery Labs may develop, including that: (a) Discovery Labs and the U.S. Food and Drug Administration (FDA) or other regulatory authorities will not be able to agree on the matters raised during regulatory reviews, or Discovery Labs may be required to conduct significant additional activities to potentially gain approval of its product candidates, if ever, (b) the FDA or other regulatory authorities may not accept or may withhold or delay consideration of any of Discovery Labs' applications, or may not approve or may limit approval of Discovery Labs' products to particular indications or impose unanticipated label limitations, and (c) changes in the national or international political and regulatory environment may make it more difficult to gain FDA or other regulatory approval; risks relating to Discovery Labs' research and development activities, including (i) time-consuming and expensive pre-clinical studies, clinical trials and other efforts, which may be subject to potentially significant delays or regulatory holds, or fail, and (ii) the need for sophisticated and extensive analytical methodologies, including an acceptable biological activity test, if required, as well as other quality control release and stability tests to satisfy the requirements of the regulatory authorities; risks relating to Discovery Labs' ability to develop and manufacture drug products and capillary aerosolization systems for clinical studies, and, if approved, for commercialization of drug and combination drug-device products, including risks of technology transfers to contract manufacturers and problems or delays encountered by Discovery Labs, its contract manufacturers or suppliers in manufacturing drug products, drug substances and capillary aerosolization systems on a timely basis or in an amount sufficient to support Discovery Labs' development efforts and, if approved, commercialization; the risk that Discovery Labs may be unable to identify potential strategic partners or collaborators to develop and commercialize its products, if approved, in a timely manner, if at all; the risk that Discovery Labs will not be able in a changing financial market to raise additional capital or enter into strategic alliances or collaboration agreements, or that the ongoing credit crisis will adversely affect the ability of Discovery Labs to fund its activities, or that additional financings could result in substantial equity dilution; the risk that Discovery Labs will not be able to access credit from its committed equity financing facilities (CEFFs), or that the minimum share price at which Discovery Labs may access the CEFFs from time to time will prevent Discovery Labs from accessing the full dollar amount potentially available under the CEFFs; the risk that Discovery Labs or its strategic partners or collaborators will not be able to retain, or attract, qualified personnel; the risk that Discovery Labs will be unable to regain compliance with The Nasdaq Global Market listing requirements prior to the expiration of the grace period currently in effect, which could cause the price of Discovery Labs' common stock to decline; the risk that recurring losses, negative cash flows and the inability to raise additional capital could threaten Discovery Labs' ability to continue as a going concern; the risks that Discovery Labs may be unable to maintain and protect the patents and licenses related to its products, or other companies may develop competing therapies and/or technologies, or health care reform may adversely affect Discovery Labs; risks of legal proceedings, including securities actions and product liability claims; risks relating to health care reform; and other risks and uncertainties described in Discovery Labs' filings with the Securities and Exchange Commission including the most recent reports on Forms 10-K, 10-Q and 8-K, and any amendments thereto.
...
Technology Stock Alert for Zoom Technologies Inc. Issued by Beacon Equity
Zoom Technologies Inc
ZOOM | 3/10/2010 6:52:29 AM
http://www.stockhouse.com/News/USReleasesDetail.aspx?n=7658661
DALLAS, Mar 10, 2010 (GlobeNewswire via COMTEX News Network) --
BeaconEquity.com announces an investment report featuring Zoom Technologies Inc. (Nasdaq:ZOOM). The report includes financial, comparative and investment analyses, and pertinent industry information you need to know to make an educated investment decision.
The full report is available at: http://www.beaconequity.com/i/ZOOM
Get our alerts BEFORE the rest of the market. Follow us on Twitter: http://twitter.com/BeaconEquity
Zoom Technologies Inc. (ZOOM), through its wholly owned holding company subsidiaries, Gold Lion Holding Ltd., owns 51.03% of TCB Digital and 100% of Profit Harvest Corporation Ltd. ZOOM is comprised of the businesses of TCB Digital, Jiangsu Leimone Electronics Co. Ltd. and Profit Harvest. TCB Digital and Jiangsu Leimone manufacture, research and develop, and sell electronic components for third generation mobile phones, wireless communication circuitry, global positioning system (GPS) equipment, and related software products.
Message Board Search for ZOOM: http://www.boardcentral.com/boards/ZOOM
In the report, the analyst notes:
"ZOOM reported a net loss of $0.9 million or 44 cents per share for its third quarter ended September 30, 2009, a slight improvement over its net loss of $1.0 million or 50 cents per share for Q3 2008. More than half of the Q3 2009 loss was due to expenses relating to the previously announced September 2009 merger involving ZOOM and the simultaneous spin-off of Zoom Telephonics. Zoom Telephonics reported net sales of $2.5 million for Q3 2009, down 34.8% from $3.9 million for the third quarter of 2008.
"ZOOM manufactures routers with 802.11-N wireless and Ethernet to distribute, manage and secure both a mobile and a wired broadband connection. The mobile broadband connection is obtained by simply plugging in a USB modem into a USB interface on the routers. A wide range of modems and services are supported."
To read the entire report visit: http://www.beaconequity.com/i/ZOOM
See what investors are saying about ZOOM at penny stock forum
BeaconEquity.com is one of the industry's largest small-cap report providers. Beacon strives to provide a balanced view of many promising small-cap companies that would otherwise fall under the radar of the typical Wall Street investor. We provide investors with an excellent first step in their research and due diligence by providing daily trading ideas, and consolidating the public information available on them. For more information on Beacon Research, please visit http://www.BeaconEquity.com
Beacon Equity Disclosure
DO NOT BASE ANY INVESTMENT DECISION UPON ANY MATERIALS FOUND ON THIS REPORT. We are not registered as a securities broker-dealer or an investment adviser either with the U.S. Securities and Exchange Commission (the "SEC") or with any state securities regulatory authority. We are neither licensed nor qualified to provide investment advice. Beacon Equity Research nor its affiliates have a beneficial interest in the mentioned company; nor have they received compensation of any kind for any of the companies listed in this communication. The information contained in our report is not an offer to buy or sell securities. We distribute opinions, comments and information free of charge exclusively to individuals who wish to receive them.
This news release was distributed by GlobeNewswire, www.globenewswire.com
SOURCE: Beacon Equity
CONTACT: Beacon Equity Research Jeff Bishop (469)-252-3505 press@beaconequity.com
(C) Copyright 2010 GlobeNewswire, Inc. All rights reserved.
Annual Report (10-K)
03/10/2010
http://ih.advfn.com/p.php?pid=nmona&cb=1268221221&article=41905812&symbol=N%5EDSCO
RT-Link Frankfurt @ IBOX !
Scrip For Scrip Rollover Relief Under Redomiciliation Confirmed by Australian Taxation Office
Date : 03/09/2010 @ 8:00AM
Source : PR Newswire
Stock : Unilife (MM) (UNIS)
http://ih.advfn.com/p.php?pid=nmona&article=41888054&symbol=UNIS
LEWISBERRY, Pa., March 9 /PRNewswire-FirstCall/ --
Unilife Corporation ("Unilife" or "Company") is pleased to advise shareholders that the Australian Taxation Office (ATO) has now issued Class Ruling CR 2010/6 (Class Ruling) in connection with the redomiciliation of the Unilife group in the United States.
Under the Class Ruling, the ATO has provided confirmation that Australian security holders of Unilife Medical Solutions Limited (UMSL) (as described in the Class Ruling) will be eligible for scrip for scrip capital gains tax roll-over relief with respect to the exchange of their shares and standalone options in UMSL for replacement securities in the Company as part of the redomiciliation. This ruling is consistent with the Company's request to the ATO and also the expectations as described in the Information Memorandum that was sent to all security holders for the Scheme Meetings.
A copy of the Class Ruling is available on Unilife's website (http://ir.unilife.com/tax.cfm) and from the ATO (http://law.ato.gov.au/atolaw/search.htm).
Further details of the tax implications arising in relation to the exchange of shares and options in UMSL for equivalent securities in Unilife are set out in section 9 of the Information Memorandum which was prepared in connection with the redomiciliation. A copy of the Information Memorandum is available on Unilife's website at http://ir.unilife.com/tax.cfm.
Security holders should seek their own independent tax advice having regard to their individual circumstances prior to relying on the tax information contained in the Class Ruling and Information Memorandum.
About Unilife Corporation
Unilife Corporation is a U.S.-based medical device company focused on the design, development, manufacture and supply of a proprietary range of retractable syringes. Primary target customers for Unilife products include pharmaceutical manufacturers, suppliers of medical equipment to healthcare facilities and patients who self-administer prescription medication. These patent-protected syringes incorporate automatic and fully-integrated safety features which are designed to protect those at risk of needlestick injuries and unsafe injection practices. Unilife is ISO 13485 certified and has FDA-registered medical device manufacturing facilities in Pennsylvania.
This press release contains forward-looking statements. All statements that address operating performance, events or developments that we expect or anticipate will occur in the future are forward-looking statements. These forward-looking statements are based on management's beliefs and assumptions and on information currently available to our management. Our management believes that these forward-looking statements are reasonable as and when made. However, you should not place undue reliance on any such forward-looking statements because such statements speak only as of the date when made. We do not undertake any obligation to publicly update or revise any forward-looking statements, whether as a result of new information, future events or otherwise, except as required by law. In addition, forward-looking statements are subject to certain risks and uncertainties that could cause actual results, events and developments to differ materially from our historical experience and our present expectations or projections. These risks and uncertainties include, but are not limited to, those described in "Item 1A. Risk Factors" and elsewhere in our registration statement on Form 10 and those described from time to time in other reports which we file with the Securities and Exchange Commission.
General: UNIS-G
Investor Contacts
Investor Contacts (US): (Australia)
Todd Fromer /
Garth Russell Stuart Fine Jeff Carter
KCSA Strategic
Communications Carpe DM Inc Unilife Corporation
Phone + 1 212-682-6300 Phone +1 908 469 1788 Phone + 61 2 8346 6500
DATASOURCE: Unilife Corporation
CONTACT: Investor Contacts (US): Todd Fromer or Garth Russell, KCSAStrategic Communications, +1-212-682-6300; Stuart Fine, Carpe DM Inc,+1-908-469-1788; Investor Contacts (Australia): Jeff Carter, UnilifeCorporation, +61-2-8346-6500
Web Site: http://ir.unilife.com/tax.cfm
Cardium Reports on Registered Direct Offering
09.03.2010 15:11
http://www.finanznachrichten.de/nachrichten-2010-03/16329937-cardium-reports-on-registered-direct-offering-008.htm
SAN DIEGO, March 9 /PRNewswire-FirstCall/ -- Cardium Therapeutics, Inc. (NYSE Amex: CXM) today announced that it has entered into agreements with certain institutional investors to sell $5.0 million out of its proposed $10 million registered direct offering. The balance of the registered direct offering is expected to be confirmed by additional investors on or around March 12, 2010 resulting in aggregate gross offering proceeds to Cardium of up to approximately $10 million, before deducting placement agent fees and offering expenses. Cardium expects to use the proceeds from the offering for general corporate purposes.
(Logo: http://www.newscom.com/cgi-bin/prnh/20051018/CARDIUMLOGO)
In the offering, Cardium is selling 2 million Units at a per Unit purchase price of $5.00. Each Unit consists of 10 shares of common stock and a common stock purchase warrant to purchase 5 shares of common stock. The common stock purchase warrants are exercisable at an exercise price of $0.64 per share, are exercisable at any time after six months from the date of closing and have a term of exercise equal to five years.
The offering was made pursuant to a shelf registration statement that was filed by Cardium Therapeutics with the Securities and Exchange Commission (the "SEC") and declared effective by the SEC on December 19, 2007. A preliminary prospectus supplement related to the public offering was filed with the SEC on February 17, 2010. Copies of the final prospectus and accompanying preliminary prospectus supplement relating to the offering may be obtained from the SEC's web-site at http://www.sec.gov/.
This press release shall not constitute an offer to sell or the solicitation of an offer to buy, nor shall there be any sale of these securities in any jurisdiction in which such offer, solicitation or sale would be unlawful. Any offer will be made only by means of a prospectus, including a prospectus supplement, forming a part of the effective registration statement.
About Cardium
Cardium is focused on the acquisition and strategic development of new and innovative bio-medical product opportunities and businesses that have the potential to address significant unmet medical needs and definable pathways to commercialization, partnering and other economic monetizations. Cardium's investment portfolio includes the Tissue Repair Company and Cardium Biologics, medical technology companies primarily focused on the development of innovative therapeutic products for tissue repair and cardiovascular indications. News from Cardium is located at http://www.cardiumthx.com/.
Forward-Looking Statements
Except for statements of historical fact, the matters discussed in this press release are forward looking and reflect numerous assumptions and involve a variety of risks and uncertainties, many of which are beyond our control and may cause actual results to differ materially from stated expectations. For example, there can be no assurance that the registered direct offering can be completed as proposed, that the company's human clinical trials can be conducted and completed in an efficient and successful manner, that product formulation enhancements will be successful or will effectively simplify or expand the use of product candidates or technologies, that the company's product candidates will prove to be sufficiently safe and effective, that results or trends observed in one clinical study or procedure will be reproduced in subsequent studies or procedures, that clinical studies even if successful will lead to product advancement or partnering, that our products or product candidates will not be unfavorably compared to competitive products that may be regarded as safer, more effective, easier to use or less expensive, that FDA or other regulatory clearances or other certifications, or other commercialization efforts will be successful or will effectively enhance our businesses or their market value, that our products or product candidates will prove to be sufficiently safe and effective after introduction into a broader patient population, or that third parties on whom we depend will perform as anticipated.
Actual results may also differ substantially from those described in or contemplated by this press release due to risks and uncertainties that exist in our operations and business environment, including, without limitation, risks and uncertainties that are inherent in the development of complex biologics and in the conduct of human clinical trials, including the timing, costs and outcomes of such trials, our ability to obtain necessary funding, regulatory approvals and expected qualifications, our dependence upon proprietary technology, our history of operating losses and accumulated deficits, our reliance on collaborative relationships and critical personnel, and current and future competition, as well as other risks described from time to time in filings we make with the Securities and Exchange Commission. We undertake no obligation to release publicly the results of any revisions to these forward-looking statements to reflect events or circumstances arising after the date hereof.
Copyright 2010 Cardium Therapeutics, Inc. All rights reserved. For Terms of Use Privacy Policy, please visit http://www.cardiumthx.com/. Cardium Therapeutics(TM) and Generx(R) are trademarks of Cardium Therapeutics, Inc. Tissue Repair(TM), Gene Activated Matrix(TM), GAM(TM), Excellarate(TM), Excellagen(TM), MedPodium(TM) and Osteorate(TM)are trademarks of Tissue Repair Company. Other trademarks are the property of their respective owners.
Photo: http://www.newscom.com/cgi-bin/prnh/20051018/CARDIUMLOGO
http://photoarchive.ap.org/
PRN Photo Desk, photodesk@prnewswire.com
Cardium Therapeutics
CONTACT: Bonnie Ortega, Director, Investor/Public Relations of Cardium
Therapeutics, Inc., +1-858-436-1018, InvestorRelations@cardiumthx.com
Web Site: http://www.cardiumthx.com/
© 2010 PR Newswire
Analyst Blog - Good News for Cell Therapeutics
March 08, 2010
http://www.zacks.com/stock/news/31373/Good+News+for+Cell+Therapeutics
In a positive development, Cell Therapeutics (CTIC) announced that the US Food and Drug Administration (FDA) has completed its inspection of the company’s manufacturing facility in Italy and found it to be compliant with all the required norms. The facility manufactures Cell Therapeutics’ Pixuvri (pixantrone), which is currently under FDA review.
Cell Therapeutics is seeking FDA approval of pixantrone for the treatment of relapsed or refractory aggressive non-Hodgkin’s Lymphoma (NHL) in patients who have not responded to other treatment options. A final decision on the drug should be taken by April 23.
Pixantrone was supposed to be reviewed by the Oncologic Drugs Advisory Committee in a meeting scheduled for February 10, which had to be postponed due to bad weather conditions in the Washington region. A new review date of March 22 has been decided upon.
Although we are pleased with the approval of the manufacturing site, we remain concerned about pixantrone’s approval due to certain issues raised by the FDA reviewers related to the study design. The study that opened in June 2004 sought to enroll 320 patients. However, the enrollment was challenging and was stopped in March 2008 after enrolling 140 patients, less than half the original plan.
Overall, the reviewers are of the view that pixantrone was well-tolerated, with manageable toxicities. The pivotal study successfully met its primary efficacy endpoint. It was observed that 20% of patients receiving the drug achieved a complete remission, compared to 6% who were treated with another drug. While pixantrone could be toxic to the heart, conclusions could not be drawn about its risk compared to other cancer drugs called anthracyclines.
The committee has to decide on the effectiveness of pixantrone and if risks associated with it outweigh the benefits. At present, investor focus is primarily on the outcome of the FDA advisory panel meeting, and a favorable decision should help in the final approval process.
Cell Therapeutics depends heavily on the approval of pixantrone. We are also concerned about the company’s liquidity position. Operating expenses are on an uptrend as the company prepares for the potential launch of pixantrone. We have a Neutral recommendation on the stock.
Video -
Annamaria interviews Branislav Vajdic, NewCardio (NWCI)
Posted on 07. Mar, 2010 by admin in Videos
http://www.corporateprofile.com/?p=655
NewCardio CEO Interviewed on CorporateProfile.com
Mar 08, 2010 14:01 ET
http://www.marketwire.com/press-release/NewCardio-CEO-Interviewed-on-CorporateProfilecom-1128340.htm
NEW YORK, NY--(Marketwire - March 8, 2010) - NewCardio, Inc. (OTCBB: NWCI) announced today that CEO, Dr. Branislav Vajdic, was interviewed on CorporateProfile.com.
The video can be viewed at: www.corporateprofile.com.
About NewCardio, Inc.
NewCardio is a cardiac diagnostic and services company focused on the development of a proprietary platform technology to provide higher accuracy to, and increase the value of, the standard 12-lead electrocardiogram (ECG). NewCardio's development-stage software and hardware products and services are intended to improve the diagnosis and monitoring of cardiovascular disease (CVD), as well as cardiac safety assessment of drugs under development. NewCardio's three-dimensional ECG platform is designed to reduce the time and expense involved in assessing cardiac status while increasing the ability to diagnose clinically significant conditions which were previously difficult to detect. For more information, visit www.newcardio.com.
About CorporateProfile.com
CorporateProfile.com is a broadcasting website where Fashion meets Finance. Merging two mainstream industries results in the unique platform for investors to receive today's hottest tips and market info.
From New York's top models straight to Wall Street, Corporate Profile's financial models are on the street and in the markets, interviewing CEOs, investment managers, and market gurus about what really drives returns.
Safe Harbor Disclaimer
Under The Private Securities Litigation Reform Act of 1995: Except for historical information contained herein, the statements in this news release are forward-looking statements that are made pursuant to the safe harbor provisions of the Private Securities Act of 1995. Forward-looking statements involve known and unknown risks and uncertainties, which may cause a company's actual results, performance and achievement in the future to differ materially from forecasted results, performance, and achievement. These risks and uncertainties are described in the Company's periodic filings with the Securities and Exchange Commission. The Company undertakes no obligation to publicly release the results of any revisions to these forward-looking statements that may be made to reflect events or circumstances after the date hereof, or to reflect the occurrence of unanticipated events or changes in the Company's plans or expectation.
Zoom Technologies (ZOOM)
03/08/10 - 11:59 AM EST
http://www.thestreet.com/_yahoo/story/10697561/1/cell-therapeutics-zoom-technologies-midday-volume-plays.html?cm_ven=YAHOO&cm_cat=FREE&cm_ite=NA
...surged by $1.88, or 30.8%, to $7.98 after the company filed with the Securities and Exchange Commission a presentation it plans to present at upcoming conferences. In the presentation, Zoom makes reference to Google's (GOOG) Android operating system, an indication it could incorporate the software for its own Leimone brand of mobile devices. Volume topped 922,000 shares, compared to the 50-day average daily volume of 50,000.
Cardium's Exchange Listing Compliance Plan Accepted by NYSE AMEX
08.03.2010 18:06
http://www.finanznachrichten.de/nachrichten-2010-03/16320702-cardium-s-exchange-listing-compliance-plan-accepted-by-nyse-amex-008.htm
SAN DIEGO, March 8 /PRNewswire-FirstCall/ -- Cardium Therapeutics (NYSE AMEX: CXM) reported that its exchange listing compliance plan submitted on January 26, 2010 has been accepted by the NYSE AMEX LLC (formerly the American Stock Exchange).
(Logo: http://www.newscom.com/cgi-bin/prnh/20051018/CARDIUMLOGO)
As previously reported on January 4, 2010, the company received notification from the staff of its current listing exchange indicating that the company was considered to be noncompliant with certain listing requirements of the NYSE AMEX. Based on the company's quarterly report on Form 10-Q, which was filed on November 9, 2009, noncompliance was noted with respect to the requirements for minimum stockholders' equity under section 1003(a)(i) of the exchange's company guide with stockholders' equity of less than $2,000,000 and losses from continuing operations and net losses in two of its three most recent fiscal years and section 1003(a)(ii) of the exchange's company guide with stockholders' equity of less than $4,000,000 and losses from continuing operations and net losses in three of its four most recent fiscal years.
The notification received from NYSE AMEX LLC had no current effect on the listing of the company's shares on the exchange. Rather, the company was afforded the opportunity to submit a supplement to its compliance plan, pursuant to which the company would seek to establish compliance with the requirements of section 1003(a)(i) and 1003(a)(ii) by June 23, 2010, as called for by the exchange. The company submitted its supplement to the compliance plan to the exchange on January 26, 2010.
On March 5, 2010, the exchange notified Cardium that, based on its review of information provided by the company, Cardium had made a reasonable demonstration of its ability to regain compliance with the requirements of sections 1003(a)(i) and 1003(a)(ii) of the NYSE AMEX company guide by June 23, 2010.
The company will be subject to periodic review by the exchange staff during the extension period covered by the plan. Failure to make progress consistent with the plan or to regain compliance with the continued listing standards by the end of the applicable extension periods could result in the company's shares being delisted from the exchange. If the company's common stock was ultimately delisted from the exchange, it would be expected to trade on the OTC Bulletin Board, a regulated quotation service that provides quotes, sale prices and volume information in over-the-counter equity securities. The company's common stock was traded on the OTC Bulletin Board until July 2007, when the company elected to instead list its shares on the American Stock Exchange.
About Cardium
Cardium is focused on the acquisition and strategic development of new and innovative bio-medical product opportunities and businesses that have the potential to address significant unmet medical needs and definable pathways to commercialization, partnering and other economic monetizations. Cardium's investment portfolio includes the Tissue Repair Company and Cardium Biologics, medical technology companies primarily focused on the development of innovative therapeutic products for wound healing, bone repair, and cardiovascular indications. In July 2009, Cardium completed the sale of its InnerCool Therapies medical device business to Royal Philips Electronics, the first asset monetization from the Company's biomedical investment portfolio. News from Cardium is located at http://www.cardiumthx.com/.
Forward-Looking Statements
Except for statements of historical fact, the matters discussed in this press release are forward looking and reflect numerous assumptions and involve a variety of risks and uncertainties, many of which are beyond our control and may cause actual results to differ materially from stated expectations. For example, there can be no assurance that the company can achieve and maintain compliance with the requirements of its listing exchange or that its shares can continue to be listed on national exchange, that results observed in one study or using one type of product or procedure will be replicated in subsequent studies or in studies using newly-developed products or procedures, that planned product development efforts and clinical studies can be performed in an efficient and effective manner, that regulatory approvals can be obtained in a timely manner or at all, that partnering, distribution or other commercialization efforts can be achieved, that product modifications or launches will be successful or that the resulting products will be favorably received in the marketplace, that our products or proposed products will prove to be sufficiently safe and effective, that our products or product candidates will not be unfavorably compared to competitive products that may be regarded as safer, more effective, easier to use or less expensive, that results or trends observed in one clinical study will be reproduced in subsequent studies, that third parties on whom we depend will behave as anticipated, or that necessary regulatory approvals will be obtained. Actual results may also differ substantially from those described in or contemplated by this press release due to risks and uncertainties that exist in our operations and business environment, including, without limitation, risks and uncertainties that are inherent in the development, testing and marketing of therapeutic hypothermia devices and the conduct of human clinical trials, including the timing, costs and outcomes of such trials, whether our efforts to launch new devices and systems and expand our markets will be successful or completed within the time frames contemplated, our dependence upon proprietary technology, our ability to obtain necessary funding, regulatory approvals and qualifications, our history of operating losses and accumulated deficits, our reliance on collaborative relationships and critical personnel, and current and future competition, as well as other risks described from time to time in filings we make with the Securities and Exchange Commission. We undertake no obligation to release publicly the results of any revisions to these forward-looking statements to reflect events or circumstances arising after the date hereof.
Copyright 2010 Cardium Therapeutics, Inc. All rights reserved. For Terms of Use Privacy Policy, please visit http://www.cardiumthx.com/.
Cardium Therapeutics(TM) and Generx(TM) are trademarks of Cardium Therapeutics, Inc.
Tissue Repair(TM), Gene Activated Matrix(TM), GAM(TM), Excellagen(TM) and Excellarate(TM) are trademarks of Tissue Repair Company.
Photo: http://www.newscom.com/cgi-bin/prnh/20051018/CARDIUMLOGO
http://photoarchive.ap.org/
PRN Photo Desk, photodesk@prnewswire.com
Cardium Therapeutics
CONTACT: Press/Investors, Bonnie Ortega, Director, Investor/Public
Relations of Cardium Therapeutics, Inc., +1-858-436-1018,
InvestorRelations@cardiumthx.com
Web Site: http://www.cardiumthx.com/
© 2010 PR Newswire
Mindspeed Debuts Optical Physical Media Device (PMD) Chipsets for 3G/4G Wireless Base Stations; Collaborates with EMCORE on Subsystems
08.03.2010 12:04
http://www.finanznachrichten.de/nachrichten-2010-03/16316455-mindspeed-debuts-optical-physical-media-device-pmd-chipsets-for-3g-4g-wireless-base-stations-collaborates-with-emcore-on-subsystems-004.htm
Low-Power Chipsets Support Common Public Radio Interface (CPRI)-Based Solutions That Connect Base Station Servers to Remote Radio Heads at High Data Rates Over Long-Distance Fiber Links
Mindspeed Technologies, Inc. (NASDAQ: MSPD), a leading supplier of semiconductor solutions for network infrastructure applications, today announced a high-performance, low-power physical media device (PMD) chipset that includes all necessary components required for CPRI-based optical transceivers that economically connect 3G/4G wireless base station servers to one or more remote radio heads (RRHs) over optical fiber.
Mindspeed® has collaborated with EMCORE'S Fiber Optics Division to develop a family of transmit and receive optical subassembly solutions based on Mindspeed's PMD devices. This family of products will allow base station servers to transmit over longer distances and at significantly higher data rates than can be supported with copper links.
"The wireless infrastructure is a rapidly growing market and we are excited to work with Mindspeed to develop a family of optical components for next-generation base station platforms," said Chris Wiggins, director of sales and marketing for EMCORE. "By leveraging our broad optoelectronic device portfolio, this collaboration effort provides key components with optimized performance to our customers. This will significantly reduce the time to market of their integrated products."
"The industry's migration to a 3G/4G mobile infrastructure is creating the need for significantly improved base station performance - not just for baseband processing, but also for the links between the main base station server and one or more distributed RRH units," said Gary Shah, executive director of product marketing with Mindspeed's high-performance analog business unit. "Mindspeed is an industry leader in high-speed PMD solutions for fiber-access networks, and now we are applying that same expertise to a new generation of power-efficient CPRI-enabled optical PMD solutions for 3G/4G base stations. These PMDs join our recently announced Transcede™ multi-core baseband processing SoCs to address key challenges in the next-generation mobile infrastructure."
Mindspeed's CPRI optical PMD chipsets fully support the latest CPRI V4.1 2009-02-18 specification and are optimized for 4.915Gbps and 6.144Gbps operation with lower power consumption compared to the traditional 10Gbps PMD chipsets. They support data rates from 614.4Mbps to 6144 Mbps over 20km Single-Mode-Fiber (SMF) and 500m Multi-Mode-Fiber (MMF). The receiver solution includes the M02020 transimpedance amplifier (TIA) combined with the M02049 limiting amplifier, optimized for 4.915 Gigabits/second (Gbps) or the M02129 TIA, combined with the M02142 limiting amplifier for 6.144Gbps. The limiting amplifiers each feature selectable band-width for optimal receiver sensitivity across CPRI rates. On the transmit side, solutions include the M02061 laser driver for long-wave laser diodes and the M02069 driver for short-wave vertical-cavity surface-emitting lasers (VCSELs).
Mindspeed's CPRI optical PMD chipsets are available now, along with a small-form-factor reference design for up to 6.144Gbps data rates. By offering seamless chipset solutions, Mindspeed's CPRI optical PMD family enables optical transceiver vendors to get to market quickly with cost competitive solutions that deliver industry leading performance.
About Mindspeed Technologies
Mindspeed Technologies, Inc. designs, develops and sells semiconductor solutions for communications applications in the wireline and wireless network infrastructure, which includes today's separate but interrelated and converging enterprise, broadband access, metropolitan and wide area networks. Our products are classified into three focused product families: multiservice access, high-performance analog and wide area networking communications. Our products are sold to original equipment manufacturers (OEMs) for use in a variety of network infrastructure equipment, including voice and media gateways, high-speed routers, switches, access multiplexers, cross-connect systems, add-drop multiplexers, digital loop carrier equipment, IP private branch exchanges (PBXs), optical modules, broadcast video systems and wireless basestation equipment.
To learn more, visit us at www.mindspeed.com.
About EMCORE
EMCORE Corporation offers a broad portfolio of compound semiconductor-based products for the broadband, fiber optics, satellite and solar power markets. EMCORE's Fiber Optics segment offers optical components, subsystems and systems for high speed data and telecommunications networks, cable television (CATV) and fiber-to-the-premises (FTTP). EMCORE's Photovoltaics segment provides products for both satellite and terrestrial applications. For satellite applications, EMCORE offers high efficiency gallium arsenide (GaAs) solar cells, covered interconnected cells (CICs) and panels. For terrestrial applications, EMCORE is adapting its high-efficiency GaAs solar cells for use in solar concentrator systems. For further information about EMCORE, visit http://www.emcore.com.
Safe Harbor Statement
This press release contains forward-looking statements within the meaning of Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Securities Exchange Act of 1934, as amended. Such statements include statements regarding the company's expectations, goals or intentions, including, but not limited to, product features and their benefits. These forward-looking statements are based on management's current expectations, estimates, forecasts and projections about the company and are subject to risks and uncertainties that could cause actual results and events to differ materially from those stated in the forward-looking statements. These risks and uncertainties include, but are not limited to: cash requirements and terms and availability of financing; future operating losses; worldwide political and economic uncertainties, and specific conditions in the markets we address; fluctuations in the price of our common stock and our operating results; loss of or diminished demand from one or more key customers or distributors; our ability to attract and retain qualified personnel; constraints in the supply of wafers and other product components from our third-party manufacturers; pricing pressures and other competitive factors; successful development and introduction of new products; doing business internationally and our ability to successfully and cost effectively establish and manage operations in foreign jurisdictions; industry consolidation; order and shipment uncertainty; our ability to obtain design wins and develop revenues from them; lengthy sales cycles; the expense of and our ability to defend our intellectual property against infringement claims by others; product defects and bugs; business acquisitions and investments; and our ability to utilize our net operating loss carryforwards and certain other tax attributes. Risks and uncertainties that could cause the company's actual results to differ from those set forth in any forward-looking statement are discussed in more detail under "Risk Factors" and "Management's Discussion and Analysis of Financial Condition and Results of Operations" in the company's Quarterly Report on Form 10-Q for the quarter ended January 1, 2010, as well as similar disclosures in the company's subsequent SEC filings. Forward-looking statements contained in this press release are made only as of the date hereof, and the company undertakes no obligation to update or revise the forward-looking statements, whether as a result of new information, future events or otherwise.
Contacts:
Editorial Contact:
Magnet PR Group
Lisa Briggs
949-305-5131
lisab@magnetprgroup.com
or
Investor Relations Contact:
Mindspeed Technologies, Inc.
Andrea D. Williams
949-579-3111
© 2010 Business Wire
FDA Approves NerPharMa to Manufacture Cell Therapeutics' Drug Pixantrone
Date : 03/08/2010 @ 1:30AM
Source : PR Newswire
Stock : Cell Therapeutics (MM) (CTIC)
http://ih.advfn.com/p.php?pid=nmona&cb=1268034751&article=41863686&symbol=CTIC
FDA Approves NerPharMa to Manufacture Cell Therapeutics' Drug Pixantrone
SEATTLE, March 8 /PRNewswire-FirstCall/ --
Cell Therapeutics, Inc. ("CTI") (Nasdaq and MTA: CTIC) today announced that the U.S. Food and Drug Administration ("FDA") has completed its inspection of the facility at NerPharMa (a pharmaceutical manufacturing company belonging to Nerviano Medical Sciences Srl, in Nerviano, Italy), which manufactures the CTI's drug pixantrone and has found the site in compliance and acceptable for continued manufacturing of the drug product. CTI has a New Drug Application ("NDA") under review at the FDA for pixantrone to treat relapsed/refractory aggressive non-Hodgkin's lymphoma. As previously announced, the FDA's Oncologic Drugs Advisory Committee ("ODAC") will review the NDA for pixantrone on March 22, 2010 and the FDA is expected to make a final decision on approval by April 23, 2010.
"FDA approval of the NerPharMa facility to manufacture our drug product is a major milestone in the drug approval process and we are pleased that our manufacturing partner is prepared to provide commercial supplies when pixantrone is approved," said Craig W. Philips, President of CTI.
About Nerviano Medical Sciences (NMS)
Nerviano Medical Sciences is the largest pharmaceutical R&D facility in Italy and one of the largest oncology-focused, integrated discovery and development companies in Europe.
About Cell Therapeutics, Inc.
Headquartered in Seattle, CTI is a biopharmaceutical company committed to developing an integrated portfolio of oncology products aimed at making cancer more treatable. For additional information, please visit http://www.celltherapeutics.com/.
This press release includes forward-looking statements that involve a number of risks and uncertainties, the outcome of which could materially and/or adversely affect actual future results and the trading price of CTI's securities. Specifically, the risks and uncertainties that could affect the development of pixantrone include risks associated with preclinical and clinical developments in the biopharmaceutical industry in general, and with pixantrone in particular, including, without limitation, the potential failure of pixantrone to prove safe and effective for the treatment of relapsed or refractory, aggressive NHL as determined by the FDA (including ODAC), that the FDA may postpone the ODAC meeting again, that the FDA may not approve the NDA for pixantrone, CTI's ability to continue to raise capital as needed to fund its operations, competitive factors, technological developments, costs of developing, producing and selling pixantrone, and the risk factors listed or described from time to time in CTI's filings with the Securities and Exchange Commission including, without limitation, CTI's most recent filings on Forms 10-K, 10-Q and 8-K. Except as may be required by law, CTI does not intend to update or alter its forward-looking statements whether as a result of new information, future events, or otherwise.
Media Contact:
Dan Eramian
T: 206.272.4343
C: 206.854.1200
F: 206.272.4434
E: deramian@ctiseattle.com
http://www.celltherapeutics.com/press_room
Investors Contact:
Ed Bell
T: 206.272.4345
Lindsey Jesch Logan
T: 206.272.4347
F: 206.272.4434
E: invest@ctiseattle.com
http://www.celltherapeutics.com/investors
DATASOURCE: Cell Therapeutics, Inc.
CONTACT: Media, Dan Eramian, +1-206-272-4343, cell, +1-206-854-1200,fax, +1-206-272-4434, deramian@ctiseattle.com, or Investors, Ed Bell,+1-206-272-4345, or Lindsey Jesch Logan, +1-206-272-4347, fax,+1-206-272-4434, invest@ctiseattle.com, all of Cell Therapeutics, Inc.
Web Site: http://www.celltherapeutics.com/
ZOOM PRESENTATION - March 2010 (Current_Business_and_Acquisition_Strategy)
http://www.sec.gov/Archives/edgar/data/822708/000113626110000064/exh99-1.htm
http://www.sec.gov/Archives/edgar/data/822708/000113626110000064/exhibit99-1.pdf
























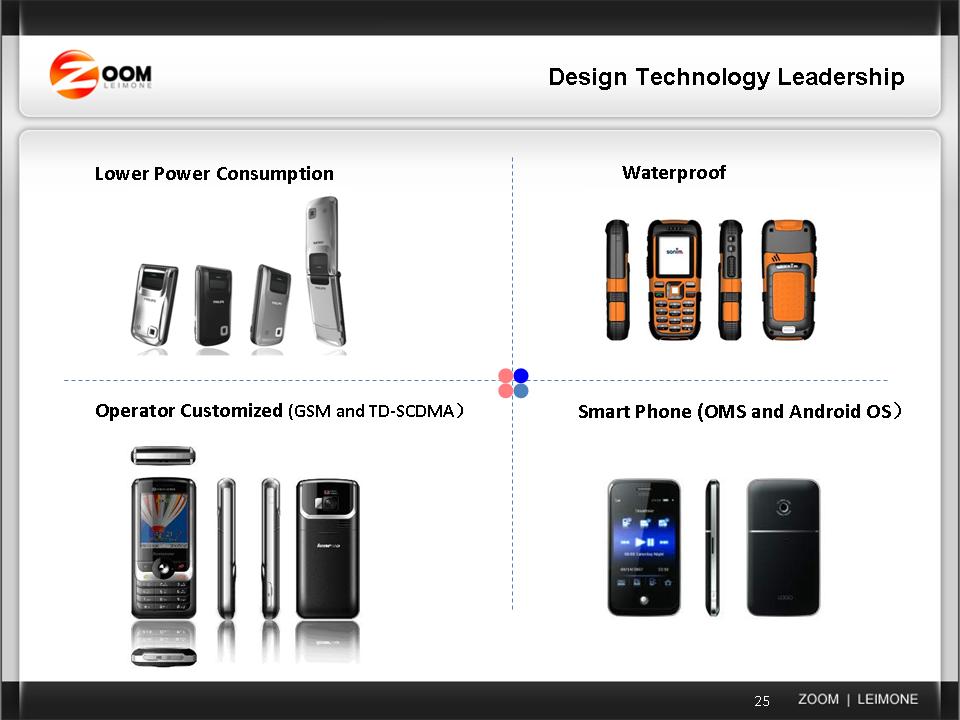













ZOOM PRESENTATION - March 2010 (Current_Business_and_Acquisition_Strategy)
http://www.sec.gov/Archives/edgar/data/822708/000113626110000064/exh99-1.htm
http://www.sec.gov/Archives/edgar/data/822708/000113626110000064/exhibit99-1.pdf
IHUB: http://investorshub.advfn.com/boards/read_msg.aspx?message_id=47497876
PRESENTATION - March 2010 (Current_Business_and_Acquisition_Strategy)
http://www.sec.gov/Archives/edgar/data/822708/000113626110000064/exh99-1.htm
http://www.sec.gov/Archives/edgar/data/822708/000113626110000064/exhibit99-1.pdf






































The Hartford Launches National Campaign To Cheer Team USA To Victory At Vancouver Paralympic Winter Games
Date : 03/04/2010 @ 10:45AM
Source : Business Wire
Stock : The Hartford Financial Services Group, Inc. (HIG)
http://ih.advfn.com/p.php?pid=nmona&cb=1267813436&article=41832369&symbol=NY^HIG
The Hartford Financial Services Group, Inc. (NYSE: HIG), the founding sponsor of U.S. Paralympics, today launched a national campaign to showcase the abilities of the U.S. Paralympic Team competing at the 2010 Paralympic Winter Games from March 12-21 and recruit more fans of elite athletes with physical disabilities.
The Hartford’s campaign kicks off with a new Website, (www.TheUSRocks.com) where Americans can send encouraging messages to U.S. Paralympic athletes.
“We congratulate the athletes who have been chosen to represent our country at the 2010 Paralympic Winter Games in Vancouver, and we are honored to have helped them reach their dreams,” said Robert Reiff, senior vice president of distribution, field service, association and product for The Hartford’s Group Benefits Division. “The abilities of U.S. Paralympic athletes are amazing, and their achievements are inspiring. We hope all Americans will join us in wishing the entire team the best of luck in Vancouver.”
The US Rocks website also features a clock counting down to the start of the 2010 Paralympic Winter Games, a Paralympic crossword challenge, and biographies and fun facts about U.S. Paralympic athletes competing in Vancouver, as well as details about their sports.
“We want everyone to experience the 2010 Paralympic Winter Games,” Reiff said. “We hope Americans check our website daily after March 12 to get up to speed on the medal tally.”
Athlete Adoptions
As part its campaign, more than 60 teams of The Hartford employees have “adopted” Paralympic athletes who are competing at the 2010 Paralympic Winter Games. Employees have expressed their heart-felt support for their “adopted” athlete through a good luck video and “care packages.” The Hartford recently held send-off parties for one of its “adopted” athletes – U.S. Paralympian Augusto “Goose” Perez – at its offices in Simsbury, Conn., and Syracuse, N.Y.
On Friday, March 12, The Hartford will hold an opening day celebration at its Simsbury office. The event will kick off at 11 a.m. with a flag-raising ceremony followed by a torch relay by employees on each floor of the four-floor building. The torch will handed off virtually to employees in offices throughout the country.
In addition, The Hartford is sending a group of employees to Vancouver to personally cheer on the U.S. team. “We can’t wait to see first-hand our athletes competing against the best of the best from around the globe,” said Reiff, who will be among the spectators at the Paralympic Winter Games. “You can also follow The Hartford on Twitter and Facebook for updates from Vancouver on Team USA.”
Gold medalist Stephani Victor, who will be competing in alpine skiing in Vancouver, her third Paralympic Games, said The Hartford’s support has been essential.
“The Hartford has been a vital part of my development as a Paralympian, and I am most grateful for all of the support,” said Victor, who was named the USOC's 2009 Paralympic SportsWoman of the Year. “The Hartford's involvement has positively moved Paralympic sport forward in the U.S.”
Role Models
The Hartford has engaged its business partners in its campaign. The Hartford created a 2010 calendar, posters, note cards and postage stamps featuring stylized photos of U.S. Paralympic athletes. The calendar, posters and stationery are being given to The Hartford’s employer clients and benefits professionals.
Also, callers who dial into The Hartford about their group disability insurance benefits and are placed on hold will hear a message inviting them to tune into the 2010 Paralympic Winter Games.
“The elite athletes of U.S. Paralympics are powerful, positive role models for our disability claimants. They are the true embodiment of our Ability Philosophy – that success is possible when you focus on abilities, not limitations,” said Reiff. “We believe it’s important for all Americans to support these incredible athletes as they represent our country on a world stage.”
Last year, The Hartford received the Paralympic Amazing Investor Award from U.S. Paralympics, in recognition of its long-time support of disabled sports organizations and athletes.
“Our sponsorship of U.S. Paralympics would not be possible if not for employees’ enthusiastic support,” Reiff said. “We appreciate the time and effort that they’ve volunteered for our Paralympic events through the years.”
About U.S. Paralympics
U.S. Paralympics, a Division of the U.S. Olympic Committee, is dedicated to becoming the world leader in the Paralympic movement and is dedicated to promoting excellence in the lives of people with physical disabilities. Visit the U.S. Paralympics Web site at usparalympics.org.
About The Hartford
Celebrating nearly 200 years, The Hartford (NYSE: HIG) is an insurance-based financial services company that serves households, businesses and employees by helping to protect their assets and income from risks, and by managing wealth and retirement needs. A Fortune 500 company, The Hartford is recognized widely for its service expertise and as one of the world’s most ethical companies. More information on the company and its financial performance is available at www.thehartford.com.
HIG-L
Some of the statements in this release may be considered forward-looking statements as defined in the Private Securities Litigation Reform Act of 1995. We caution investors that these forward-looking statements are not guarantees of future performance, and actual results may differ materially. Investors should consider the important risks and uncertainties that may cause actual results to differ. These important risks and uncertainties include those discussed in our Quarterly Reports on Form 10-Q, our 2009 Annual Report on Form 10-K and the other filings we make with the Securities and Exchange Commission. We assume no obligation to update this release, which speaks as of the date issued.
The Hartford Launches National Campaign To Cheer Team USA To Victory At Vancouver Paralympic Winter Games
Date : 03/04/2010 @ 10:45AM
Source : Business Wire
Stock : The Hartford Financial Services Group, Inc. (HIG)
http://ih.advfn.com/p.php?pid=nmona&cb=1267813436&article=41832369&symbol=NY^HIG
The Hartford Financial Services Group, Inc. (NYSE: HIG), the founding sponsor of U.S. Paralympics, today launched a national campaign to showcase the abilities of the U.S. Paralympic Team competing at the 2010 Paralympic Winter Games from March 12-21 and recruit more fans of elite athletes with physical disabilities.
The Hartford’s campaign kicks off with a new Website, (www.TheUSRocks.com) where Americans can send encouraging messages to U.S. Paralympic athletes.
“We congratulate the athletes who have been chosen to represent our country at the 2010 Paralympic Winter Games in Vancouver, and we are honored to have helped them reach their dreams,” said Robert Reiff, senior vice president of distribution, field service, association and product for The Hartford’s Group Benefits Division. “The abilities of U.S. Paralympic athletes are amazing, and their achievements are inspiring. We hope all Americans will join us in wishing the entire team the best of luck in Vancouver.”
The US Rocks website also features a clock counting down to the start of the 2010 Paralympic Winter Games, a Paralympic crossword challenge, and biographies and fun facts about U.S. Paralympic athletes competing in Vancouver, as well as details about their sports.
“We want everyone to experience the 2010 Paralympic Winter Games,” Reiff said. “We hope Americans check our website daily after March 12 to get up to speed on the medal tally.”
Athlete Adoptions
As part its campaign, more than 60 teams of The Hartford employees have “adopted” Paralympic athletes who are competing at the 2010 Paralympic Winter Games. Employees have expressed their heart-felt support for their “adopted” athlete through a good luck video and “care packages.” The Hartford recently held send-off parties for one of its “adopted” athletes – U.S. Paralympian Augusto “Goose” Perez – at its offices in Simsbury, Conn., and Syracuse, N.Y.
On Friday, March 12, The Hartford will hold an opening day celebration at its Simsbury office. The event will kick off at 11 a.m. with a flag-raising ceremony followed by a torch relay by employees on each floor of the four-floor building. The torch will handed off virtually to employees in offices throughout the country.
In addition, The Hartford is sending a group of employees to Vancouver to personally cheer on the U.S. team. “We can’t wait to see first-hand our athletes competing against the best of the best from around the globe,” said Reiff, who will be among the spectators at the Paralympic Winter Games. “You can also follow The Hartford on Twitter and Facebook for updates from Vancouver on Team USA.”
Gold medalist Stephani Victor, who will be competing in alpine skiing in Vancouver, her third Paralympic Games, said The Hartford’s support has been essential.
“The Hartford has been a vital part of my development as a Paralympian, and I am most grateful for all of the support,” said Victor, who was named the USOC's 2009 Paralympic SportsWoman of the Year. “The Hartford's involvement has positively moved Paralympic sport forward in the U.S.”
Role Models
The Hartford has engaged its business partners in its campaign. The Hartford created a 2010 calendar, posters, note cards and postage stamps featuring stylized photos of U.S. Paralympic athletes. The calendar, posters and stationery are being given to The Hartford’s employer clients and benefits professionals.
Also, callers who dial into The Hartford about their group disability insurance benefits and are placed on hold will hear a message inviting them to tune into the 2010 Paralympic Winter Games.
“The elite athletes of U.S. Paralympics are powerful, positive role models for our disability claimants. They are the true embodiment of our Ability Philosophy – that success is possible when you focus on abilities, not limitations,” said Reiff. “We believe it’s important for all Americans to support these incredible athletes as they represent our country on a world stage.”
Last year, The Hartford received the Paralympic Amazing Investor Award from U.S. Paralympics, in recognition of its long-time support of disabled sports organizations and athletes.
“Our sponsorship of U.S. Paralympics would not be possible if not for employees’ enthusiastic support,” Reiff said. “We appreciate the time and effort that they’ve volunteered for our Paralympic events through the years.”
About U.S. Paralympics
U.S. Paralympics, a Division of the U.S. Olympic Committee, is dedicated to becoming the world leader in the Paralympic movement and is dedicated to promoting excellence in the lives of people with physical disabilities. Visit the U.S. Paralympics Web site at usparalympics.org.
About The Hartford
Celebrating nearly 200 years, The Hartford (NYSE: HIG) is an insurance-based financial services company that serves households, businesses and employees by helping to protect their assets and income from risks, and by managing wealth and retirement needs. A Fortune 500 company, The Hartford is recognized widely for its service expertise and as one of the world’s most ethical companies. More information on the company and its financial performance is available at www.thehartford.com.
HIG-L
Some of the statements in this release may be considered forward-looking statements as defined in the Private Securities Litigation Reform Act of 1995. We caution investors that these forward-looking statements are not guarantees of future performance, and actual results may differ materially. Investors should consider the important risks and uncertainties that may cause actual results to differ. These important risks and uncertainties include those discussed in our Quarterly Reports on Form 10-Q, our 2009 Annual Report on Form 10-K and the other filings we make with the Securities and Exchange Commission. We assume no obligation to update this release, which speaks as of the date issued.
Keryx Biopharmaceuticals to Present at the Cowen and Company 30th Annual Health Care Conference
Date : 03/05/2010 @ 8:30AM
Source : PR Newswire
Stock : Keryx Biopharmaceuticals (MM) (KERX)
http://ih.advfn.com/p.php?pid=nmona&article=41848306&symbol=KERX
NEW YORK, March 5 /PRNewswire-FirstCall/ --
Keryx Biopharmaceuticals, Inc. today announced that Ron Bentsur, the Company's Chief Executive Officer, will be presenting at the Cowen and Company 30th Annual Health Care Conference, being held at The Boston Marriott Copley Place Hotel in Boston, MA. Mr. Bentsur's presentation will take place on Thursday March 11, 2010 at 11:05 a.m. Eastern Time.
A live webcast of Mr. Bentsur's presentation will be accessible from the Investor Information page of the Company's Website at http://investors.keryx.com/. An archived version of the webcast will be available following the conclusion of the live presentation.
About Keryx Biopharmaceuticals, Inc.
Keryx Biopharmaceuticals is focused on the acquisition, development and commercialization of medically important pharmaceutical products for the treatment of life-threatening diseases, including cancer and renal disease. Keryx is developing KRX-0401 (perifosine), a novel, potentially first-in-class, oral anti-cancer agent that inhibits the phosphoinositide 3-kinase (PI3K)/Akt pathway, a key signaling cascade that has been shown to induce cell growth and cell transformation. KRX-0401 has demonstrated both safety and clinical efficacy in several tumor types, both as a single agent and in combination with novel therapies. KRX-0401 also modulates a number of other key signal transduction pathways, including the JNK pathway, which are pathways associated with programmed cell death, cell growth, cell differentiation and cell survival. KRX-0401 is currently in a Phase 3 trial, under Special Protocol Assessment (SPA), in multiple myeloma, with a Phase 3 trial in refractory metastatic colorectal cancer, under SPA, pending commencement, and in Phase 2 clinical development for several other tumor types. Keryx is also developing Zerenex(TM) (ferric citrate), an oral, iron-based compound that has the capacity to bind to phosphate and form non-absorbable complexes. The Phase 3 clinical program of Zerenex in the treatment for hyperphosphatemia (elevated phosphate levels) in patients with end-stage renal disease is pending commencement under an SPA agreement with the FDA. Keryx is headquartered in New York City.
KERYX CONTACT:
Lauren Fischer
Director of Investor Relations
Keryx Biopharmaceuticals
Tel: 212 531 5962
E-mail: lfischer@keryx.com
DATASOURCE: Keryx Biopharmaceuticals, Inc.
CONTACT: Lauren Fischer, Director of Investor Relations, KeryxBiopharmaceuticals, +1-212-531-5962, lfischer@keryx.com
Web Site: http://www.keryx.com/
Gynecologic Oncology Group (GOG) Notifies CTI That Continuation of GOG-212 Pivotal Trial of OPAXIO Maintenance Therapy in Front Line Ovarian Cancer Remains High Priority
GOG-218 Bevacizumab Results Do Not Influence Importance of GOG-212
Press Release Source: Cell Therapeutics, Inc.
Thursday March 4, 2010, 1:30 am EST
http://finance.yahoo.com/news/Gynecologic-Oncology-Group-prnews-1211275465.html?x=0&.v=1
SEATTLE, March 4 /PRNewswire-FirstCall/ -- Cell Therapeutics, Inc. ("CTI") (Nasdaq and MTA: CTIC) announced today that CTI received a statement on March 1, 2010 from the Gynecologic Oncology Group (GOG) leadership that the phase III GOG-212 clinical trial of CTI's OPAXIO™ used as maintenance therapy for ovarian cancer remains a high priority and enrollment will continue. The GOG made the statement to clarify that the recent results of the GOG-218 clinical trial bevacizumab in maintenance therapy for ovarian cancer has not influenced the importance of completing the GOG-212 clinical trial. The Gynecologic Oncology Group (GOG) is one of the National Cancer Institute's (NCI) funded cooperative cancer research groups. The GOG is a multidisciplinary cooperative clinical trial research group focused on the study of gynecologic malignancies. The GOG is conducting phase III trials in ovarian cancer and other gynecologic cancers and has established standard treatments for these diseases in the U.S.
GOG leadership noted that, "GOG-218 and GOG-212 differ in the type of patients under study. It is important to note that some of the patients who completed the initial 6 cycles of chemotherapy in GOG-218 had clinical evidence of persistent tumor and where randomized to either placebo (no treatment) or bevacizumab. Thus a subset of GOG-218 patients received no therapy, despite the presence of persistent tumor. This is not the typical setting of using maintenance or consolidation therapy and it is not the setting for patients enrolled in GOG-212. In GOG-212, only patients who have achieved a complete clinical response are considered candidates for enrollment in the trial."
"Reliance upon the data from GOG-218 to establish the "standard of care" must take into consideration the actual treatment effect (i.e. duration of benefit), the cost of the treatment, and the associated toxicity... [in GOG-212] the toxicity of the intervention may have less associated mortality and the incremental cost-effectiveness ratio may be more acceptable to patients and the health care economists. Thus the GOG has no intention to discontinue enrollment in GOG 212 as they feel that the study is addressing a different scientific question and the primary outcome study goal is survival, not progression free survival, an outcome of greater importance to both physicians and patients," the statement added.
The Data Monitoring Committee is scheduled to conduct an interim analysis of overall survival when 130 events are recorded among patients in the no maintenance treatment arm. The statistical analysis plan utilizes pre-specified boundaries for early stopping for success. Based on current enrollment and study duration, the interim analysis could be conducted as early as 2011. If successful, the Company could utilize those results to form the basis of its New Drug Application for OPAXIO.
About Cell Therapeutics, Inc.
Headquartered in Seattle, CTI is a biopharmaceutical company committed to developing an integrated portfolio of oncology products aimed at making cancer more treatable. For additional information, please visit www.celltherapeutics.com.
Blockbuster Moving Beyond Reliance on DVDs: CEO
Published: Thursday, 4 Mar 2010 | 1:51 PM ET
By: CNBC.com
http://www.cnbc.com/id/35709880/site/14081545?__source=yahoo|headline|quote|text|&par=yahoo
In the new digital arena, Blockbuster faces many challenges against a growing field of competitors. While its stock is down some 70 percent over the past year, the company's CEO sees a "bright future," he told CNBC.
"We're in the middle of a dramatic transformation," said James Keyes, Blockbuster's CEO. "During the course of this transformation, we're moving away from a total reliance on DVDs and now a different form of distribution for both DVDs and digital content. We're building new channels."
The chain plans to shutter 1,000 stores, and its restructuring includes adding 10,000 points of purchase through Blockbuster Express.
In the near term, the company looks to compete with Netflix's
by-mail service with its own a-la-carte offerings. To challenge Redbox's vending system, Blockbuster kiosks are built to enable digital downloads and physical distributions.
As for its brick and mortar locations, the company last year launched a prototype of its "Rock the Block" stores, which give customers access to games, movies, and a self-service Coca-Cola bar.
In the long term, Keyes believes the Blockbuster brand remains its legacy selling point.
"When you're driving down main street and you see the Blockbuster brand, you'll know it's the place to rent movies," Keyes said. "The same experience, we think, will be true for the internet. There will be a vast array of people offering movies, but Blockbuster is a very reliable brand."
© 2010 CNBC.com
WaMu shareholders urge for an annual meeting: court filing
Thu Mar 4, 2010 1:09am EST
http://www.reuters.com/article/idUSTRE6230PX20100304?
(Reuters) - Shareholders of Washington Mutual Inc (WAMUQ.PK) have filed a complaint against the savings bank holding company for not convening an annual shareholders' meeting for nearly two years, court documents show.
The Official Committee of Equity Security Holders, representing shareholders of Washington Mutual, wants the bank to convene and hold an annual shareholders' meeting for the nomination and election of directors, court papers showed.
In the complaint, the committee said the bank held its last annual shareholder meeting on or about April 20, 2008.
Also, the bank has not set a date for an annual meeting of shareholders in 2010, which in line with its bylaws, would normally be held on April 20, according to the court filings.
Washington Mutual has issued about 3 million shares of Class R preferred stock, about 20 million shares of Class K preferred stock, and about 1.7 billion shares of common stock, according to the filing.
In 2008, Washington Mutual had filed for Chapter 11 bankruptcy protection. On January 28, a judge refused to disband the shareholders' committee, which Washington Mutual said would complicate the case.
The U.S. Trustee, which plays an oversight role in bankruptcy, appointed the committee earlier this month after being petitioned by 3,500 shareholders. The company immediately asked the court to disband it.
The case is In re Washington Mutual Inc, et al, U.S. Bankruptcy Court, District of Delaware, No. 08-12229. (Reporting by Sakthi Prasad in Bangalore; Editing by Valerie Lee )
Unilife and sanofi-aventis Agree to Exclusivity List for Unifill(TM) Ready-to-Fill Syringe
March 2, 2010
http://ir.unilife.com/releasedetail.cfm?ReleaseID=448725
LEWISBERRY, Pa., March 2, 2010 /PRNewswire via COMTEX News Network/ -- Unilife Corporation ("Unilife" or "Company") (Nasdaq: UNIS; ASX: UNS), today announced that it has agreed to a list of therapeutic drug classes ("Exclusivity List") within which sanofi-aventis has the exclusive right to purchase the Unifill(TM) ready-to-fill syringe.
Sanofi-aventis has secured exclusivity for the Unifill(TM) syringe ("Product") within the full therapeutic classes of antithrombotic agents and vaccines until June 30, 2014 ("Period of Exclusivity"). These two therapeutic classes together represent the majority of all prefilled syringes consumed globally. Sanofi-aventis has also secured Product exclusivity in an additional six smaller sub-groups that fall within other therapeutic classes that Unilife believes represent new market opportunities in the pharmaceutical use of prefilled syringes.
The scope of the Exclusivity List allows Unilife to commence formal discussions with other pharmaceutical companies relating to the potential use of the Unifill(TM) syringe within a number of significant therapeutic classes that fall outside of those areas retained by sanofi-aventis.
In accordance with the Exclusive Agreement, sanofi-aventis will receive a ten year extension ("Additional Period") on its Period of Exclusivity within a designated therapeutic class should sanofi-aventis purchase commercial quantities of the product prior to July 1, 2014. This extension will be reduced on a per therapeutic class basis to five years in the event that sanofi-aventis does not sell a minimum of 20 million units of the Unifill(TM) syringe for use with an injectable drug product to be marketed for this therapeutic class in at least one of the first five years of the Additional Period.
During the Period of Exclusivity, sanofi-aventis may also nominate additional therapeutic sub-groups to be placed onto the Exclusivity List should the Company not have already signed a commercial arrangement within this sub-group with a third party. Before an additional therapeutic class can be added to the Exclusivity List, both parties will need to be reasonably satisfied that a target drug suitable for use with the Unifill(TM) syringe is likely to generate a commercial order.
Unilife CEO Alan Shortall stated: "The agreement of an exclusive list of therapeutic drug classes with sanofi-aventis for the purchase of the Unifill(TM) syringe is a significant business milestone for Unilife. The confined nature of the therapeutic sectors defined within the Exclusivity List considerably expands our commercial opportunities with additional pharmaceutical companies. In return, sanofi-aventis retains the opportunity to nominate the placement of additional therapeutic drugs onto the Exclusivity list provided they are commercially favorable and do not infringe upon any future agreements we may sign with other pharmaceutical companies. This is indicative of the strong collaborative relationship that has been established between both parties. We look forward to commencing supply of the Unifill(TM) syringe after the scheduled completion of the industrialization program."
The Unifill(TM) Syringe
The Unifill(TM) syringe is targeted for use by pharmaceutical manufacturers who utilize prefilled (ready-to-fill) syringes as a preferred drug delivery device for injectable drugs and vaccines. We are aware of more than 50 drug products used within healthcare facilities, or by patients who self-administer prescription medication, that are currently available in a prefilled syringe format. Unilife has designed the Unifill(TM) syringe so that it is compatible with the drug validation and manufacturing systems currently used by target pharmaceutical customers to fill and package standard prefilled syringes.
To Unilife's knowledge, the Unifill(TM) syringe is the only known product of its kind with automatic safety features which are integrated inside the glass barrel. The compact size, intuitive use, functionality and automatic safety features of the Unifill(TM) syringe may help pharmaceutical companies extend product lifecycles, increase levels of market differentiation in competitive therapeutic areas, and expand the marketability of some drugs for convenient self-administration by patients outside of the healthcare setting. Sanofi-aventis has paid Unilife a euro 10 million exclusivity fee and committed to pay the Company up to an additional euro 17 million to fund the industrialization program for the Unifill syringe. Upon the scheduled completion of the industrialization program in late 2010, Unilife expects to commence the supply and sale of the Unifill syringe to sanofi-aventis.
About Unilife Corporation
Unilife Corporation is a U.S.-based medical device company focused on the design, development, manufacture and supply of a proprietary range of retractable syringes. Primary target customers for Unilife products include pharmaceutical manufacturers, suppliers of medical equipment to healthcare facilities and patients who self-administer prescription medication. These patent-protected syringes incorporate automatic and fully-integrated safety features which are designed to protect those at risk of needlestick injuries and unsafe injection practices. Unilife is ISO 13485 certified and has FDA-registered medical device manufacturing facilities in Pennsylvania.
This press release contains forward-looking statements. All statements that address operating performance, events or developments that we expect or anticipate will occur in the future are forward-looking statements. These forward-looking statements are based on management's beliefs and assumptions and on information currently available to our management. Our management believes that these forward-looking statements are reasonable as and when made. However, you should not place undue reliance on any such forward-looking statements because such statements speak only as of the date when made. We do not undertake any obligation to publicly update or revise any forward-looking statements, whether as a result of new information, future events or otherwise, except as required by law. In addition, forward-looking statements are subject to certain risks and uncertainties that could cause actual results, events and developments to differ materially from our historical experience and our present expectations or projections. These risks and uncertainties include, but are not limited to, those described in "Item 1A. Risk Factors" and elsewhere in our registration statement on Form 10 and those described from time to time in other reports which we file with the Securities and Exchange Commission.
General: UNIS-G
Investor Contacts Investor Contacts
(US): (Australia)
Todd Fromer / Garth Russell Stuart Fine Jeff Carter
KCSA Strategic Communications Carpe DM Inc Unilife Corporation
Phone Phone Phone
+ 1 212-682-6300 + 1 908 469 1788 + 61 2 8346 6500
SOURCE Unilife Corporation
Copyright (C) 2010 PR Newswire. All rights reserved
XTXI - new 52wk-high
Crosstex Energy Companies Swing To Profit In Q4 - Update
http://www.rttnews.com/ArticleView.aspx?Id=1225978
FDA Sets March 22 for ODAC Meeting to Review CTI's New Drug Application for Pixantrone
Date : 03/01/2010 @ 12:02PM
Source : PR Newswire
Stock : Cell Therapeutics (MM) (CTIC)
http://ih.advfn.com/p.php?pid=nmona&cb=1267464739&article=41771466&symbol=N^CTIC
SEATTLE, March 1 /PRNewswire-FirstCall/ --
Cell Therapeutics, Inc. ("CTI") (Nasdaq and MTA: CTIC) announced today that the U.S. Food and Drug Administration's ("FDA") Oncologic Drugs Advisory Committee ("ODAC") will review CTI's New Drug Application ("NDA") for pixantrone for the treatment of relapsed/refractory aggressive non-Hodgkin's lymphoma ("NHL") on March 22, 2010. The meeting was to take place on February 10, 2010, but the FDA postponed the meeting because of severe weather conditions in the Washington, D.C. area. Following the vote at the rescheduled ODAC meeting, the FDA is expected to make a final decision on approval of CTI's NDA for pixantrone by April 23, 2010.
"Our team is well-prepared to deliver an evidenced-based presentation to the ODAC panel, which we expect will provide a persuasive argument to the clinicians on the panel for the approval of pixantrone," said James A. Bianco, M.D., CEO of CTI. "The ODAC meeting is an important milestone in the review process, and we look forward to discussing the efficacy and safety data for pixantrone with the members of the panel and the FDA review team." Dr. Owen O'Connor, Chief of Hematology Oncology at NYU Cancer Institute, and Dr. John Leonard, Chief of Lymphoma Myeloma at Weill Corner Cancer Center will be presenting on behalf of CTI.
ODAC is an independent panel of experts that evaluates data concerning the efficacy and safety of marketed and investigational products for use in the treatment of cancer and makes appropriate recommendations to the FDA. The FDA regulations indicate that although the FDA will consider the recommendation of the panel, the final decision regarding the approval of the product is made by the FDA.
About Cell Therapeutics, Inc.
Headquartered in Seattle, CTI is a biopharmaceutical company committed to developing an integrated portfolio of oncology products aimed at making cancer more treatable. For additional information, please visit http://www.celltherapeutics.com/.
This press release includes forward-looking statements that involve a number of risks and uncertainties, the outcome of which could materially and/or adversely affect actual future results and the trading price of the securities of CTI. Specifically, the risks and uncertainties that could affect the development of pixantrone include risks associated with preclinical and clinical developments in the biopharmaceutical industry in general, and with pixantrone in particular, including, without limitation, the potential failure of pixantrone to prove safe and effective for the treatment of relapsed or refractory, aggressive NHL as determined by the FDA (including ODAC), that the FDA may postpone the ODAC meeting again, CTI's ability to continue to raise capital as needed to fund its operations, competitive factors, technological developments, costs of developing, producing and selling pixantrone, and the risk factors listed or described from time to time in CTI's filings with the Securities and Exchange Commission including, without limitation, CTI's most recent filings on Forms 10-K, 10-Q and 8-K. Except as may be required by law, CTI does not intend to update or alter its forward-looking statements whether as a result of new information, future events, or otherwise.
Media Contact: Dan Eramian T: 206.272.4343 C: 206.854.1200 F: 206.272.4434 E: deramian@ctiseattle.com http://www.celltherapeutics.com/press_room
Investors Contact: Ed Bell T: 206.272.4345 Lindsey Jesch Logan T: 206.272.4347 F: 206.272.4434 E: invest@ctiseattle.com http://www.celltherapeutics.com/investors
DATASOURCE: Cell Therapeutics, Inc.
CONTACT: Media, Dan Eramian, +1-206-272-4343, Cell, +1-206-854-1200,Fax, +1-206-272-4434, deramian@ctiseattle.com, or Investors, Ed Bell,+1-206-272-4345, or Lindsey Jesch Logan, +1-206-272-4347, Fax,+1-206-272-4434, invest@ctiseattle.com, all of Cell Therapeutics, Inc.
Web Site: http://www.celltherapeutics.com/
Crosstex Energy Reports Fourth-Quarter and Full-Year 2009 Results
Date : 03/01/2010 @ 6:30AM
Source : Business Wire
Stock : (XTXI)
http://ih.advfn.com/p.php?pid=nmona&cb=1267458334&article=41763990&symbol=N^XTXI
Crosstex Energy Reports Fourth-Quarter and Full-Year 2009 Results
The Crosstex Energy companies, Crosstex Energy, L.P. (NASDAQ: XTEX) (the Partnership) and Crosstex Energy, Inc. (NASDAQ: XTXI) (the Corporation), today reported earnings for the fourth-quarter and full-year 2009.
Fourth-Quarter 2009 – Crosstex Energy, L.P. Financial Results The Partnership realized adjusted cash flow of $42.3 million in the fourth quarter of 2009 compared with $61.3 million in the fourth quarter of 2008. Adjusted cash flow is a non-GAAP financial measure and is explained in greater detail under “Non-GAAP Financial Information.” There is a reconciliation of this non-GAAP measure to net income (loss) in the tables at the end of this news release. Fourth-quarter 2008 adjusted cash flow included other income of $20.0 million associated with the assignment of certain contract rights to a nonaffiliated third party. Additionally, assets sold during 2009 contributed $18.3 million to fourth-quarter 2008 realized adjusted cash flow.
The Partnership reported net income of $55.9 million in the fourth quarter of 2009, compared with a net loss of $9.4 million in the fourth quarter of 2008. Fourth-quarter 2009 results included an $86.3 million gain on the sale of the Partnership’s Treating assets, while fourth-quarter 2008 results included a $49.8 million gain on the sale of the Partnership’s interest in the Seminole gas processing plant. Fourth-quarter 2009 results also included a $6.1 million loss from discontinued operations that was mainly related to the write-off of debt issuance costs and senior note make whole expense due to the repayment of notes with the proceeds from asset sales.
“We are extremely pleased with the significant progress we made executing our plan in 2009,” said Barry E. Davis, Crosstex President and Chief Executive Officer. “We have focused on optimizing results from our core assets in north Texas and Louisiana while investing in high-return projects, realigning our cost structure, lowering our business risks and significantly reducing our leverage.
“We recently completed our long-term recapitalization and reducing our leverage will continue to be a priority as we conservatively manage our business. We have established financial guidelines that we will follow when making decisions regarding the restoration of our distribution and dividend. Based on our current assumptions, distributions could be paid from the results of operations of the fourth quarter of 2010,” added Davis.
The Partnership’s gross margin from continuing operations for the fourth quarter of 2009 increased to $80.6 million from $65.2 million in the fourth quarter of 2008. This improvement was related to higher margins from the Partnership’s gathering and transmission business and increases in the natural gas processing business as a result of a more favorable natural gas liquids (NGL) market in the 2009 period. Gross margin from the south Louisiana processing and NGL business increased by $10.7 million, as a result of improvements in the NGL marketing business and increased plant inlet volumes. Gross margin from LIG gathering, transmission and processing increased by $7.9 million due to higher margins and a more favorable NGL market. Gross margin derived from north Texas operations declined $7.1 million compared with the fourth-quarter 2008. The decrease was primarily related to a $3.7 million charge associated with a final arbitration decision in a producer lawsuit and declines in throughput. Fourth-quarter 2009 north Texas volumes declined from the third quarter of 2009 mainly due to the renegotiation of a key producer contact, which reduced gathering volumes and increased transmission volumes.
Fourth-quarter 2009 operating expenses declined $6.4 million, or 20 percent, compared with the fourth quarter of 2008, as a result of the Partnership’s continued focus on expense reduction. Fourth-quarter 2009 general and administrative expense was $16.2 million, including a one-time charge of $1.0 million for severance expenses related to asset sales, a decline of $4.7 million versus the same period a year ago. Depreciation and amortization expense of $29.2 million in the fourth quarter of 2009 rose slightly over the fourth quarter of 2008 amount of $28.3 million. Interest expense declined to $28.0 million in the fourth quarter of 2009 from $41.0 million in the fourth quarter of 2008, primarily the result of the debt paydown associated with asset sales during the year.
The net income per limited partner common unit in the fourth quarter of 2009 was $1.09 basic and $1.07 diluted compared with a net loss per limited partner common unit of $0.18 basic and diluted in the fourth quarter of 2008.
Full-Year 2009 – Crosstex Energy, L.P. Financial Results The Partnership realized adjusted cash flow of $203.8 million in 2009 compared with $245.1 million in 2008. The reduction in realized adjusted cash flow is mainly due to the sale of assets during 2009. The Partnership reported net income of $104.5 million in 2009, compared with $11.1 million in 2008. Results for 2009 included a $183.7 million gain on the sale of Partnership assets compared with a gain of $49.8 million recorded in 2008. The Partnership’s net income in 2009 also included a loss from discontinued operations related to the asset sales of $1.8 million compared to income of $25.0 million in 2008.
The Partnership’s gross margin from continuing operations for 2009 increased to $311.2 million from $307.8 million in 2008. This improvement was primarily related to higher margins on the Partnership’s gathering and transmission assets, which was partially offset by a decline in the natural gas processing business due to a less favorable NGL market. The LIG system’s gross margin increased by $14.0 million year over year primarily due to improved pricing and higher volumes on the northern portion of the system, which was offset by a $15.1 million decline in processing margins in Louisiana. Gross margin derived from north Texas operations rose $10.2 million compared with 2008. The growth was primarily related to increased gathering and plant inlet volumes versus the previous year, which was partially offset by the previously mentioned arbitration charge.
The Partnership’s 2009 operating expense declined $15.4 million, or 12 percent, to $110.4 million versus $125.8 million in 2008. General and administrative expense for 2009 decreased by $9.0 million from 2008, including the effect of a one-time charge of $1.9 million for severance expenses. Depreciation and amortization expense increased $11.6 million in 2009 compared with 2008 due to the Partnership’s investment in its north Texas and Louisiana assets. Interest expense rose to $95.1 million in 2009 from $75.0 million 2008. The increase in interest expense results primarily from an increase in interest rates pursuant to the February 2009 amendments to the Partnership’s debt agreements.
The net income per limited partner common unit for 2009 was $1.44 basic and $1.40 diluted compared with a net loss per limited partner common unit of $3.19 basic and diluted in 2008.
Crosstex Energy, Inc. Financial Results The Corporation reported net income of $12.1 million in the fourth quarter of 2009 compared with a loss of $4.5 million in the comparable 2008 period. The Corporation’s loss from continuing operations before income taxes (which includes interest of non-controlling partners in the net income of the Partnership) was $24.3 million in the fourth quarter of 2009, compared with $64.3 million in the fourth quarter of 2008.
Net income for the year 2009 was $15.6 million compared with net income of $24.2 million in 2008. The Corporation’s loss from continuing operations before income taxes (which includes interest of non-controlling partners in the net income of the Partnership) was $78.4 million in 2009, compared with $65.6 million in 2008.
The Corporation had $9.9 million of cash on hand and no debt at the end of the fourth-quarter 2009.
In accordance with U.S. accounting standards, the Partnership and Corporation classified the results of their operations for assets sold as discontinued operations for all accounting periods presented. Tables of selected financial data in which amounts have been reclassified as discontinued operations for each period presented are included in this news release.
Crosstex Provides Preliminary 2010 Guidance The following is the Partnership’s low and high estimates of 2010 adjusted cash flow and distributable cash flow based upon two commodity price scenarios. The Partnership has established financial guidelines that will govern its considerations as to the timing and amount of any future distribution payments, including achieving a ratio of total debt to adjusted cash flow of less than 4.5 to 1 (pro forma for the payment of any distribution), strong coverage from cash flow to continue to allow the Partnership to delever and a positive outlook for its business. Assuming actual results are within the range of guidance, the Partnership expects it could generate sufficient distributable cash flow to pay up to $0.30 per unit in distributions relating to the fourth quarter of 2010 with strong coverage from cash flows generated in such quarter; and the Corporation expects it could pay up to $0.10 per share in dividends, assuming the receipt of a $0.30 per unit distribution from the Partnership. The payment and amount of any distributions and dividends will be subject to approval by the respective Boards of Directors and to economic conditions and other factors existing at the time of determination.
Crosstex Energy, L.P. Forecast for 2010 Net Income Reconciliation to Distributable Cash Flow* (In millions except prices and ratios) Total Year 2010 Low High Net income (loss) $ (41 ) $ (10 ) Depreciation and amortization 113 113 Stock-based compensation 6 6 Interest 80 79 Taxes and other 2 2 Adjusted cash flow * $ 160 $ 190 Interest (80 ) (79 ) Taxes and other (3 ) (3 ) Maintenance capital expenditures (15 ) (12 ) Distributable cash flow * $ 62 $ 96 Growth Capital $ 25 $ 30 Key Assumptions for Forecast Weighted Average Liquids Price ($/gallon) $ 0.80 $ 1.09 Crude ($/Bbl) $ 69.37 $ 94.52 Natural Gas ($/MMBtu) $ 6.00 $ 5.00 Natural Gas Liquids to Gas Ratio 149.9 % 245.0 % * Adjusted cash flow and Distributable cash flow are non-GAAP financial measures and are explained in greater detail under "Non-GAAP Financial Information." Processing Sensitivities: Impact Percent of Liquids Contracts - $0.10 change in Weighted Average Liquids Price $2.9 million Processing Margin Contracts - 5% change in Natural Gas Liquids to Gas Ratio (1) $1.8 million (1) Assumes constant gas price of $5.79/MMBtu.
Crosstex to Hold Earnings Conference Call Today The Partnership and the Corporation will hold their quarterly conference call to discuss fourth-quarter and full- year 2009 results today, March 1, at 10:00 a.m. Central time (11:00 a.m. Eastern time). The dial-in number for the call is 1-888-679-8034. Callers outside the United States should dial 1-617-213-4847. The passcode for all callers is 88621274. Investors are advised to dial in to the call at least 10 minutes prior to the call time to register. Participants may preregister for the call at https://www.theconferencingservice.com/prereg/key.process?key=PUBWL748L. Preregistrants will be issued a pin number to use when dialing in to the live call, which will provide quick access to the conference by bypassing the operator upon connection. Interested parties also can access a live Web cast of the call on the Investors page of Crosstex’s Web site at www.crosstexenergy.com.
After the conference call, a replay can be accessed until June 8, 2010, by dialing 1-888-286-8010. International callers should dial 1-617-801-6888 for a replay. The passcode for all callers listening to the replay is 91226576. Interested parties also can visit the Investors page of Crosstex’s Web site to listen to a replay of the call.
About the Crosstex Energy Companies Crosstex Energy, L.P., a midstream natural gas company headquartered in Dallas, operates approximately 3,300 miles of pipeline, nine processing plants and three fractionators. The Partnership currently provides services for 3.2 billion cubic feet per day of natural gas, or approximately six percent of marketed U.S. daily production.
Crosstex Energy, Inc. owns the two percent general partner interest, a 25 percent limited partner interest and the incentive distribution rights of Crosstex Energy, L.P.
Additional information about the Crosstex companies can be found at www.crosstexenergy.com.
Non-GAAP Financial Information This press release contains non-generally accepted accounting principle financial measures that the Partnership refers to as Distributable Cash Flow and Adjusted Cash Flow. Distributable Cash Flow is defined as earnings before certain noncash charges, less maintenance capital. Adjusted Cash Flow is defined as net income before interest, income taxes, depreciation, amortization and impairments, stock-based compensation, noncash mark-to-market items gain on the sale of assets and other miscellaneous noncash items. The amounts included in the calculation of these measures are computed in accordance with generally accepted accounting principles (GAAP), with the exception of maintenance capital expenditures. Maintenance capital expenditures are capital expenditures made to replace partially or fully depreciated assets in order to maintain the existing operating capacity of the assets and to extend their useful lives.
The Partnership believes these measures are useful to investors because they may provide users of this financial information with meaningful comparisons between current results and prior-reported results and a meaningful measure of the Partnership’s cash flow after it has satisfied the capital and related requirements of its operations.
Distributable Cash Flow and Adjusted Cash Flow are not measures of financial performance or liquidity under GAAP. They should not be considered in isolation or as an indicator of the Partnership’s performance. Furthermore, they should not be seen as measures of liquidity or a substitute for metrics prepared in accordance with GAAP. A reconciliation of these measures to net income (loss) is included among the preceding and following tables.
This press release contains forward-looking statements within the meaning of the federal securities laws. These statements are based on certain assumptions made by the Partnership and the Corporation based upon management's experience and perception of historical trends, current conditions, expected future developments and other factors the Partnership and the Corporation believe are appropriate in the circumstances. These statements include, but are not limited to, statements with respect to the Partnership’s and the Corporation’s guidance and future outlook, distribution and dividend guidelines and future estimates, financing plans, financial condition, liquidity and results of operations. Such statements are subject to a number of assumptions, risks and uncertainties, many of which are beyond the control of the Partnership and the Corporation, which may cause the Partnership's and the Corporation's actual results to differ materially from those implied or expressed by the forward-looking statements. These risks include the following: (1) the Partnership’s profitability is dependent upon prices and market demand for natural gas and NGL’s; (2) the Partnership’s substantial indebtedness could limit its flexibility and adversely effect its financial health (3) the Partnership may not be able to obtain funding due to the deterioration of the credit and capital markets and current economic conditions; (4) the Partnership and the Corporation do not have diversified assets; (5) drilling levels may decrease due to deterioration in the credit and commodity markets; (6) the Partnership's credit risk management efforts may fail to adequately protect against customer nonpayment; (7) the Partnership’s use of derivative financial instruments does not eliminate its exposure to fluctuations in commodity prices and interest rates; (8) the Partnership may not be successful in balancing its purchases and sales; (9) the amount of natural gas transported in the Partnership's gathering and transmission lines may decline as a result of reduced drilling by producers, competition for supplies, reserve declines and reduction in demand from key customers and markets; (10) the level of the Partnership's processing operations may decline for similar reasons; (11) operational, regulatory and other asset-related risks, including weather conditions such as hurricanes, exist because a significant portion of the Partnership’s assets are located in southern Louisiana; and (12) other factors discussed in the Partnership's and the Corporation's Annual Reports on Form 10-K for the year ended December 31, 2009, and other filings with the Securities and Exchange Commission. The Partnership and the Corporation have no obligation to publicly update or revise any forward-looking statement, whether as a result of new information, future events, or otherwise.
...
Unilife Sticking To Growth Plan
NEW YORK (TheStreet) - Alan Shortall, CEO of Unilife Corporation, says his newly Nasdaq listed company has built a better and safer syringe. And with the help of drug giant Sanofi-Aventis, he plans on taking a big slice of the market.
Mon 03/01/10 16:00 PM EST -- Gregg Greenberg
http://www.thestreet.com/video/10691705/unilife-sticking-to-growth-plan.html?puc=_tscrss&s=1
Aeterna's blood cancer drug gets European nod
http://www.reuters.com/article/idCAN0124223120100301?rpc=44
* Phase 3 trial multiple myeloma set to begin
* U.S. FDA announced similar approval last year
TORONTO, March 1 (Reuters) - Canadian cancer drugmaker Aeterna Zentaris Inc (AEZ.TO) said on Monday that European health regulators granted its perifosine blood cancer treatment orphan drug status, marking the second such designation for the drug.
Last year, the drug received a similar designation from the U.S. FDA, which is reserved for new treatments that are being developed to treat diseases or conditions that affect fewer than 200,000 people in the United States, and which grants the drug developers seven years of market exclusivity.
Perifosine is currently in Phase 2 mid-stage clinical development for multiple tumor types, and a Phase 3 late-stage trial for multiple myeloma is set to begin.
Aeterna is developing the drug with partner and licensee Keryx Biopharmaceuticals (KERX.O) in the United States, Canada and Mexico. ($1=$1.05 Canadian) (Reporting by Scott Anderson, editing by Dave Zimmerman)
