Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
19s printed....
Modern Technology Corp Releases 2008 Interim Fourth Quarter Financial Results Reporting Sales of $29,000,000
http://finance.yahoo.com/news/Modern-Technology-Corp-iw-14208303.html
sales of 29 mill
Modern Technology Corp Releases 2008 Interim Fourth Quarter Financial Results Reporting Sales of $29,000,000
http://finance.yahoo.com/news/Modern-Technology-Corp-iw-14208303.html
THIS
Modern Technology Corp Releases 2008 Interim Fourth Quarter Financial Results Reporting Sales of $29,000,000
* Friday January 30, 2009, 11:05 am EST
* Yahoo! Buzz
* Print
OXFORD, MS--(MARKET WIRE)--Jan 30, 2009 -- Modern Technology Corporation (Other OTC:MODC.PK - News) announced today it released its interim financial results for the period ending December 31, 2008. The company's complete financial statements will be posted on or before 15 February 2009.
The company's revenues for 2008 were approximately $29,000,000 with net profits of approximately 3% of gross. The company's gross revenues and net profits are somewhat lower than projections due to the sudden and dramatic downturn in the worldwide economy. This downturn has affected all sectors including and notably electronics and distribution. Sales and demand appear stable at this point and the company is confident sales will remain strong and anticipates remaining profitable. The company believes its new products and technology, particularly its hydrogen generator systems, will facilitate strong revenue growth in 2009. The Company has 2,000,000,000 shares authorized with approximately 650,000,000 shares issued and outstanding.
Financials and Disclosure information will be posted pursuant to paragraphs (a)(5)(i) to (xiv), inclusive, and paragraph (a)(5)(xvi) of Rule 240.15c2-11. Additional disclosures will be made on a frequent and ongoing basis.
Investors can expect timely updates on the following:
-- Sales projections for 2009 will be announced
-- Various international distributors for the Gen-H Hydrogen Injector
System will be announced
-- Additional portfolio acquisitions are under review. Should these
acquisitions occur, revenues will be increased another $5-$15 million
dollars.
-- The company will announce a new cancer detection system design
incorporating significant advances in both patient test throughputs and
cell sample scan speeds.
-- New marketing initiatives to pathologists based on revenue sharing
models for cancer detection scans.
-- Details on new distribution, management services and new energy-
related initiatives will be released.
MODC
Modern Technology Corp Releases 2008 Interim Fourth Quarter Financial Results Reporting Sales of $29,000,000
Modern Technology Corp Releases 2008 Interim Fourth Quarter Financial Results Reporting Sales of $29,000,000
it has very good reason too!!!
MODC is looking good!!!
now 320K!
well it depends on how you look at it. I dont think shares seem plenty. In fact the opposite. I sat on the bid of .0118 then moved it up to .0119 for about 20 minutes this afternoon. As soon as I took it off they moved it down. Better for me I got them at .0117.
LEHJQ is looking good!
LEHJQ is rockin today. Bid and ask is tight.
when the ask was at .0006 it took me longer than usual to get filled at the ask.
MODC is looking nice. It took longer than usual for me to get filled on the ask.
GNTA is also one to take a look at.
Form 8-K for GENTA INC DE/
11-Feb-2009
Other Events
Item 8.01 Other Events.
On February 11, 2009, Genta Incorporated, (the Company), announced that it has submitted a proposal to the Food and Drug Administration for a randomized clinical trial of tesetaxel, an oral taxane chemotherapy compound, for Special Protocol Assessment (SPA). A SPA is intended to secure agreement on the design, size, and endpoints of clinical trials that are intended to form the primary basis of an efficacy claim in a New Drug Application (NDA). Genta also expects to seek Scientific Advice from the European Medicines Agency (EMEA) for this study to support a Marketing Authorization Application (MAA). The Company believes tesetaxel is the leading oral taxane currently in clinical development. The proposed protocol will examine the safety and efficacy of tesetaxel in patients with advanced gastric cancer whose disease has progressed after receiving first-line chemotherapy. Further details of the study will be announced after final agreement is secured with both regulatory agencies. FDA has previously granted Orphan Drug designation to tesetaxel for treatment of patients with advanced gastric cancer
Item 9.01 Financial Statements and Exhibits.
NEWSWIRES
Genta Submits Randomized Study of Tesetaxel in Patients with Advanced Gastric Cancer to FDA for Special Protocol Assessment
Date : 02/11/2009 @ 8:05AM
Source : Business Wire
Stock : Genta Incorporated (GNTA)
Quote : 0.0149 0.0008 (5.67%) @ 9:36AM
Genta Submits Randomized Study of Tesetaxel in Patients with Advanced Gastric Cancer to FDA for Special Protocol Assessment
Genta Incorporated (OTCBB: GNTA) announced today that the Company has submitted a proposal to the Food and Drug Administration for a randomized clinical trial of tesetaxel, an oral taxane chemotherapy compound, for Special Protocol Assessment (SPA). A SPA is intended to secure agreement on the design, size, and endpoints of clinical trials that are intended to form the primary basis of an efficacy claim in a New Drug Application (NDA). Genta also expects to seek Scientific Advice from the European Medicines Agency (EMEA) for this study to support a Marketing Authorization Application (MAA). The Company believes tesetaxel is the leading oral taxane currently in clinical development.
The proposed protocol will examine the safety and efficacy of tesetaxel in patients with advanced gastric cancer whose disease has progressed after receiving first-line chemotherapy. Further details of the study will be announced after final agreement is secured with both regulatory agencies. FDA has previously granted Orphan Drug designation to tesetaxel for treatment of patients with advanced gastric cancer
About Tesetaxel
Tesetaxel is a novel, orally absorbed, semi-synthetic taxane that is in the same class of drugs as paclitaxel and docetaxel. However, both prototype agents suffer from serious safety issues, particularly hypersensitivity reactions related to intravenous infusions that are occasionally fatal and that require careful premedication and observation. Other prominent side-effects of this drug class include myelosuppression (low blood counts) and peripheral neuropathy (disabling nerve damage).
With administration as an oral capsule, tesetaxel was developed to maintain the high antitumor activity of the taxane drug class while eliminating infusion reactions, reducing neuropathy, and increasing patient convenience. The oral route also enables development of novel schedules that may expand dosing options when tesetaxel is used alone or in combination with other anticancer drugs. Preclinically, tesetaxel has demonstrated substantially higher activity against cell lines that were resistant to paclitaxel and docetaxel, since acquired resistance is not mediated by the multidrug-resistant p-glycoprotein.
Tesetaxel has demonstrated anticancer activity in several Phase 2 clinical trials. The drug has not been associated with severe infusion reactions that are linked with other taxanes. Moreover, unlike other oral taxanes, nerve damage has not been a prominent side effect of tesetaxel. Thus, the drug offers substantial opportunities to improve patient convenience, safety, and anticancer activity. More than 250 patients worldwide have been treated with oral tesetaxel in Phase 1 and Phase 2 clinical trials.
Tesetaxel in Advanced Gastric Cancer
In the completed Phase 2 study, 35 patients with advanced gastric cancer were treated with tesetaxel at doses ranging from 27 to 35 mg/m2 once every three weeks. All patients had received extensive prior treatment, having failed a combination regimen that included cisplatin plus 5-fluorouracil or capecitabine (Xeloda®; Hoffmann-La Roche, Inc.), and all but 2 patients had received a third chemotherapy drug with this regimen. Final intent-to-treat analysis, including all patients enrolled in the study, showed that 5 patients achieved a partial response, 2 patients achieved a partial response unconfirmed by CT scan, and 14 patients achieved stable disease, for an overall major response rate of 20% and a disease-control rate of 60%. The most serious adverse reaction was Grade 3-4 neutropenia, which occurred in 57% of patients. Six patients failed to complete the first course of treatment. Five patients died on study from differing causes that included intestinal perforation, pneumonia, hepatic failure, hemorrhagic shock, and rapid disease progression. One patient withdrew before receiving the first treatment dose.
gap filled. time to go upppp....
yeah
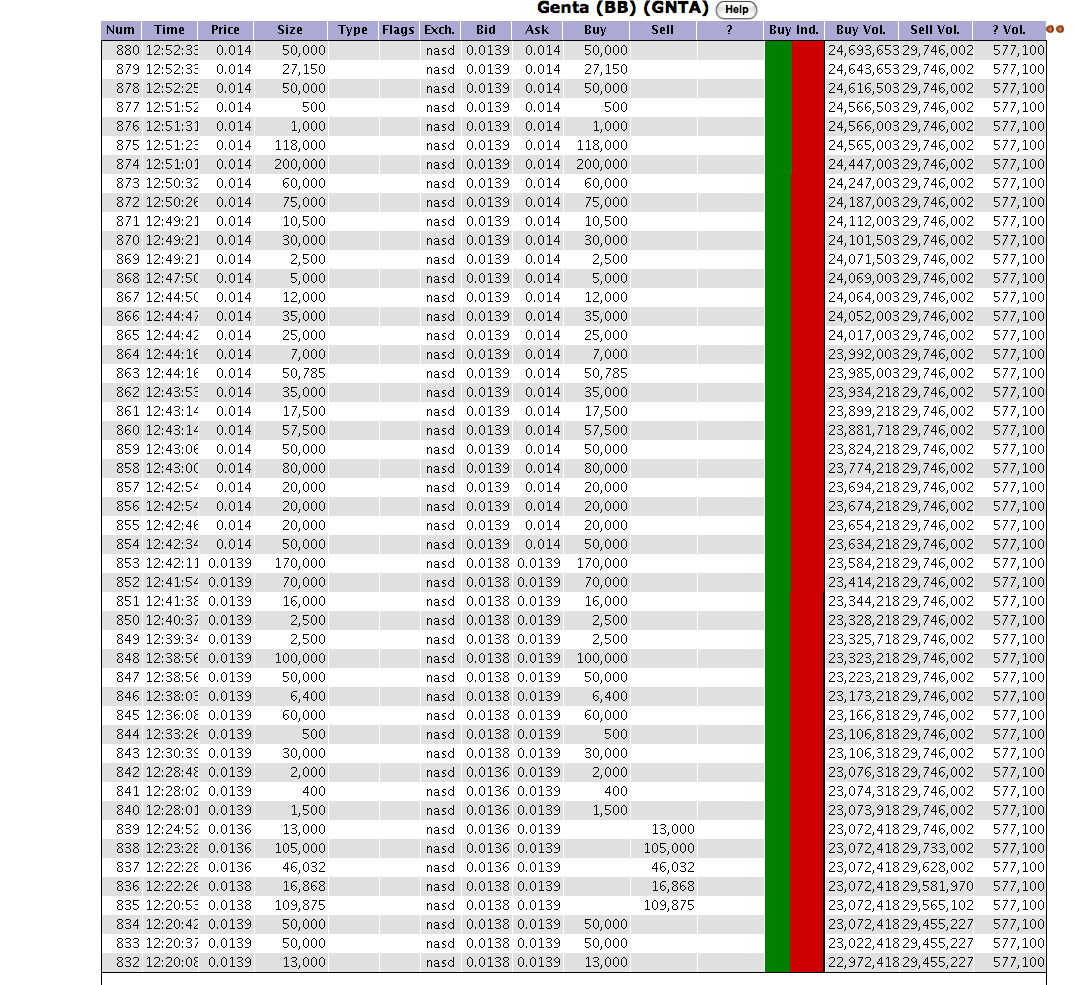
yeah I thought it seemed a bit baffling
ahh thanks. too bad I cant be there...
trying to catch up on some info here that I must have missed?... so there is a meeting today @ 12:30 for investors
I like that chart!
II. "Naked" Short Sales
In a "naked" short sale, the seller does not borrow or arrange to borrow the securities in time to make delivery to the buyer within the standard three-day settlement period. 3 As a result, the seller fails to deliver securities to the buyer when delivery is due (known as a "failure to deliver" or "fail").
Failures to deliver may result from either a short or a long sale. There may be legitimate reasons for a failure to deliver. For example, human or mechanical errors or processing delays can result from transferring securities in physical certificate rather than book-entry form, thus causing a failure to deliver on a long sale within the normal three-day settlement period. A fail may also result from naked short selling. For example, market makers who sell short thinly traded, illiquid stock in response to customer demand may encounter difficulty in obtaining securities when the time for delivery arrives.
Naked short selling is not necessarily a violation of the federal securities laws or the Commission's rules. Indeed, in certain circumstances, naked short selling contributes to market liquidity. For example, broker-dealers that make a market in a security4 generally stand ready to buy and sell the security on a regular and continuous basis at a publicly quoted price, even when there are no other buyers or sellers. Thus, market makers must sell a security to a buyer even when there are temporary shortages of that security available in the market. This may occur, for example, if there is a sudden surge in buying interest in that security, or if few investors are selling the security at that time. Because it may take a market maker considerable time to purchase or arrange to borrow the security, a market maker engaged in bona fide market making, particularly in a fast-moving market, may need to sell the security short without having arranged to borrow shares. This is especially true for market makers in thinly traded, illiquid stocks such as securities quoted on the OTC Bulletin Board,5 as there may be few shares available to purchase or borrow at a given time.
http://www.sec.gov/spotlight/keyregshoissues.htm
among the giants....
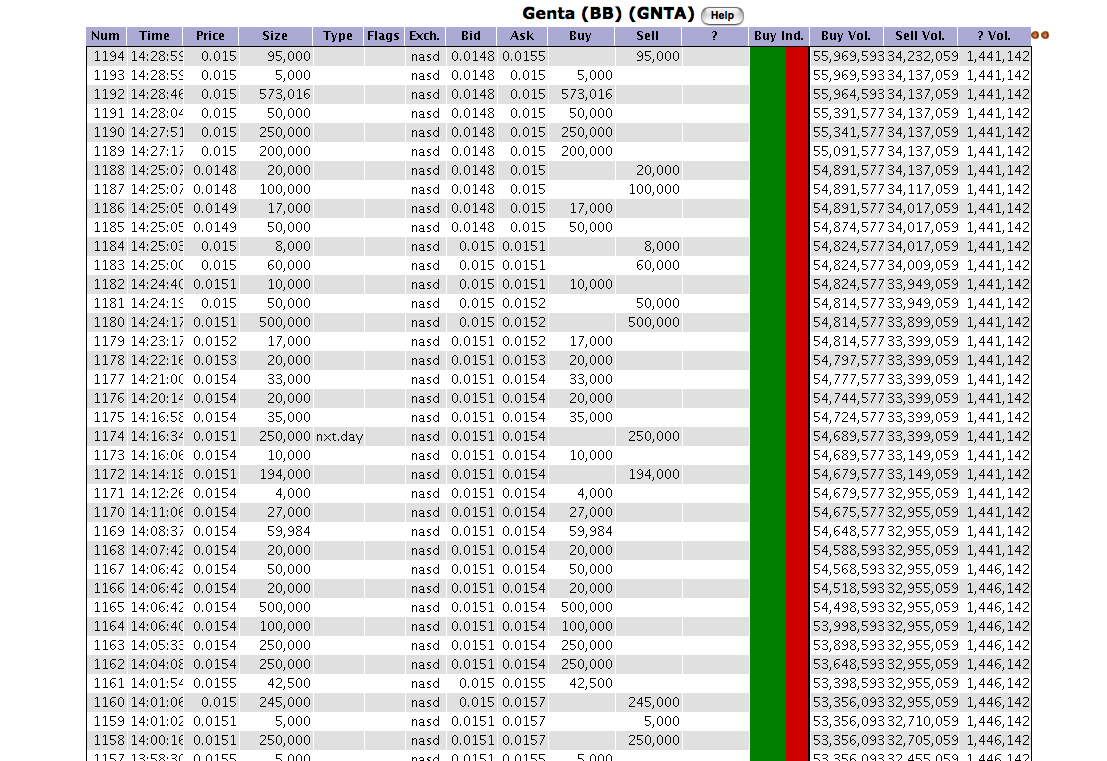
does anyone know what a nxt.day trade is? I seen this the other day too...
Court Documents link for everyone.
http://chapter11.epiqsystems.com/Documents.aspx
yes it will trade probably at least until march 31st

LOL not hating here. I want everyone to make mad cash. In fact I had some earlier before bk. I have had this stock on my watch list for months (and keep following). Just pointing out info.
I was going to grab some earlier but at the time of this announcement from the company, common shareholders will get nothing. http://www.apexsilver.com/pdf/Restructure/Investor%20Frequently%20Asked%20Questions_.pdf

http://www.apexsilver.com/home.html
not bad, its Friday, how about u?
CBAI looking good!
Moving up!
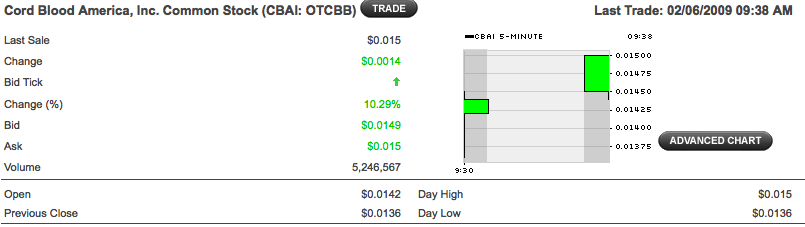
Over 3 million more buys and only 200 thousand sells in about 1 hour

Buys outnumbering sells 2:1

what do these dollar amounts mean by each company?
about 36 mill more buys than sells....
I am pretty sure I seen at least one over $25,000
look at all the buys outnumbering the sells.
can someone do a screenshot of L2 thanks
moving back up now...