Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Veronica Francardo, the Dr. and author of the therapeutics poster, used that very same title some years ago.
“Pharmacological stimulation of sigma-1 receptors has neurorestorative effects in experimental parkinsonism”
Interesting read to see how she thought of sigma-1 back in 2014.
In the last years several studies also reported anti-amnesic and neuroprotective effects of Sig-1R ligands in animal models of AD (Maurice and Su, 2009). Clinical trials in patients with AD are now ongoing (further informations can be found at www.anavex.com), and the aim for the future is to accelerate the steps from preclinical to clinical trials also on PD patients.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4281425/
Actually, it could very well be there this time next year. Eternity avoided.
A little myelin goes a long way to restore nervous system function
Nice read if you have the time.
https://medicalxpress.com/news/2017-10-myelin-nervous-function.html
The purpose of the study, says Duncan, was to confirm that thin myelin sheaths persisted and supported normal neurologic function.
To expand on the results, Duncan also looked at a condition in cats, another long-lived species that has been shown to fully recover nervous system function after demyelination. In particular, Duncan's team was interested in remyelination of the optic nerves.
That element of the study, looking at remyelination two years after the onset of the condition, Duncan notes, is an example of "true demyelination and remyelination. We found that nearly every optic nerve fiber was remyelinated with a thin myelin sheath, which is important for understanding human disease because in multiple sclerosis, the optic nerve is often the first to be demyelinated."
So we have oktoberinvestfest tomorrow, followed by a 3rd party, Wayne State, presentation of A-273 as a potential candidate for MS on Friday.
Wait a minute, why would there be research of a-273 and MS, when MS is no longer on the a-273 trials pipeline?
Then CTAD the following weekend.
Hmm... I wonder where in the next 9 weeks we see 3 trials beginning.
Ahh.... way to keep pressure on the shorts. Eventually THEY will take our pps higher.
Whats up with the questioning of grants? It helped the company get to where they currently are today. Curious how the p2 and p2/3 trials will be funded? just wait like everyone else and we will find out at the same time.
Way to ASSume Australian funding was for p2/3.
Didn't they help fund this p2a/b trial.
par·a·digm shift - a fundamental change in approach or underlying assumptions.
Perfect word to do some DD on when trying to reference timelines using the "old way" things got done.
Thinking a-273 would take 3 more years to gain approval?
I laugh at that assumption.
That was my take. That it was written earlier and only now posted. Maybe the author needs to see the Wayne State abstract for a complimentary article to this.
"How this stops the big boys that are NOT part of the Gentlemans Club from taking a stake. .. I will never understand."
Sure you do, they would rather back a hostile takeover than risk being on the losing side.
These 4 words strike fear in any money manager.
"You will regret it"
You think he cares about the share price? It'll take care of itself. Doesn't matter what he releases, it'll always be used against him because 32,now 25, patients are unimaginable in the previous ways of doing trials.
The days of 1000,2000,3000 patient trials are soon to be extinct.
Just watch when the FDA releases the info
I'm not saying that nothing will happen once its done.
Oh no, Me thinks the Dr. has his plan ready to execute and just needed more time to get through the dog days of fall.
Once we get to that point, there's no looking back.
I'd see it as the end of his purchases, nothing more. Begin and end with a 750 purchase.
What's the purchase plan shares up to now
So he too dared to think different.
Let's suppose we get stabilizing data for the 104 week read out. Eventually you would think patients in Australia would start using the "Special Access Scheme",SAS, to gain access to a-273 from their pcp. SAS, being a program that allows Australian patients access to drugs that isn't approved by the (ARTG)Australian Registry of Therapeutic Goods yet.
Missling, having first hand experience of a family member having Alzheimer's, would gladly allow a-273 be used if it helped the patient, family, and anyone else involved.
Looks like it's been updated sept 27,2017
https://www.tga.gov.au/form/special-access-scheme
Hmmm.....
Ehh, I'd be pretty hesitant to take a pill that the bottle says "made in china"
Then there's the counterfeiting issue
This company is imho way too small to deal with the three upcoming trials on its own, and I doubt they have sufficient cash to support all three trials without a partner or partners.
Whats intriguing is how Australia will treat Anavex. They see their citizens in the trial. What does it take for Australia to approve or better question is would they approve before the FDA does.
Nice reminder of what Missling has said, and how he is executing. Sticky for "unsophisticated investors" to discover.
Que Emmanuel O. Fadiran hiring which occurred on May 1, 2017.
With regulatory experience with the FDA and leadership positions at CDER, we have someone that can help guide Anavex in the right direction.
Thanks investor, shows how silly shorts are getting. Twisting Dr perry quote to align with their agenda. Then again, what else is there to attack?
I had no such cringe, just another validation that p2/3 will be something I look forward to.
Then again, Rett and Parkinson's should have data sooner.
That doesn't look like a slight decline, it looks like a downward spiral after 12 weeks.
For future reference, you should use this cached link as not everyone has an account to view what you are posting.
http://webcache.googleusercontent.com/search?q=cache:YeBsOKwjG4gJ:www.medscape.org/viewarticle/481063_5+&cd=1&hl=en&ct=clnk&gl=us
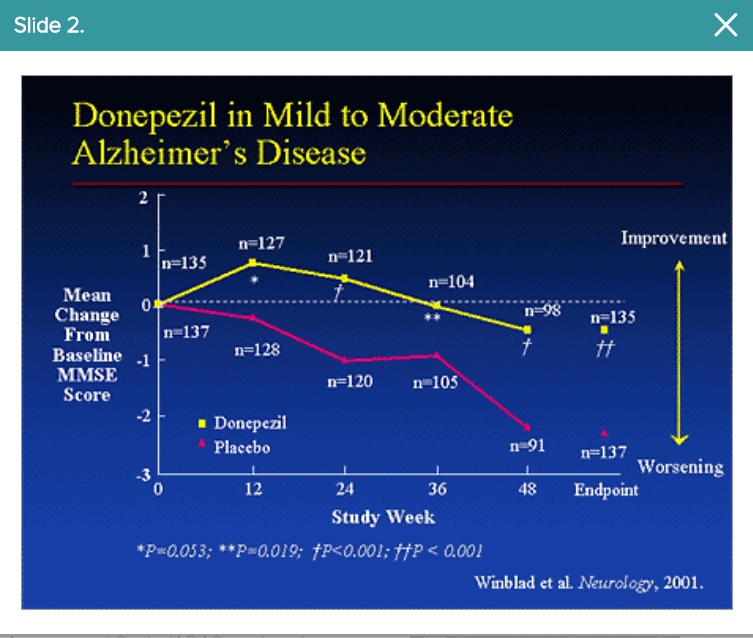
Major difference between Donepezil and A-273 is the side effects. If the two were hypothetically equal, just on less side effects, we would have a new SOC if p2/3 repeats its p2a/b results.
However, we all know Donepezil doesn't work for a year, more like a month or two before the decline starts again.
Plus, we don't know exactly HOW much a-273 does benefit, or as the naysayers say, if it benefits at all, the patient because the dosage isn't optimized across the board. Some need to understand that and not treat this data as "as good as it gets".
We are yet to see just how good a-273 works when given with the correct dosage.
Can't wait to see what an optimized dosage for everyone would look like on the chart.
Drum roll.... p2/3 start
The only signal is that he is done with his purchases and everyone had their chance to cover or grab more shares.
On with the show now.
I believe Missling presented in this investment conference back in oct 13 of 2015 also.
Which we then surprise up listed to the daq 2 weeks after
Mycroft, that was a great book to read and I quoted it a couple of days back.
Something's brewing and we should soon know what that is.
It's not that he didn't find water. It's that according to all previous wells, the water was murky and brown. This go around, with Anavex digging their own well, the water was clear and filtered. So to figure out how/why it happened this way, a third party was hired to crunch the numbers and figure out what happened.
It's why most small bio either sell or go belly up. I'm sure it was the goal of the manipulators, but thanks to his background and previous jobs in finance, he knew what to do. Something many take for granted.
Missling has done a great job with what he was given. I'm sure part of the reason he decided on these recent purchases is because he WANTS retail to hold rather than sell and have some entity hold a too close for comfort share amount. Remember, over 50 percent of the shares is also end game for Anavex too.
Frontotemporal degeneration (FTD) -- the most common dementia for people under age 60 -- inflicts a significantly higher economic burden on both patients and their caregivers, a new study shows. While the social and economic cost of Alzheimer's is well documented, the study found that the average annual costs associated with FTD to total $119,654, nearly two times the reported annual cost of Alzheimer's.source
Right behind a-273, is a-371, which we cant forget the pr released april 8, 2016 reading:
U.S. FDA Grants Orphan Drug Designation to ANAVEX 3-71 for the Treatment of Frontotemporal Dementia
FULL PR
So much upside potential from here. I sure hope Missling wouldn't sell out for a measly $2-$5B
I'll bet he underestimated how greedy BP can be and decided to just keep quiet since any PR was being taken advantage of by the shorts.
Who would of thought that we are only a couple cents higher today, than we were back in the OTC days.
I mean $1 and some change pre-split price today seems ludicrous with the amount of money being dumped in other known failed drug candidates.
Dementia has become the leading cause of death among Australian women and remains the second leading cause of all Australians, according to new statistics released by the Australian Bureau of Statistics today.
As of Sept 27 2017
FULL STORY
Do ponder if Australia would be the first to approve a-273 being that they desperately need to find something to slow it down.
Here we go again. 99.6% when you target amyloid plaque. So far there is no shots on goal for them
For those not familiar with adaptive trials. Know that Fadiran was a contributor to the book which can be found HERE



So here's a question I have.
Let's say depression is an endpoint for a trial. Under the 21cca, if a-273 gets approved for something and depression was an endpoint, could a-273 be used for depression as well if it ends up being approved or am I understanding the master protocol, multiple targets wrong?
Biotech stocks drop after FDA makes it easier for public to search for drug side effects
Imagine what'll happen when/if a-273 gets added on that database.
Nuplazid, for patients with Parkinson's, had 1,343 serious case reports, including 493 deaths, FAERS reveals.
TERRIBLE!!!!
https://www.cnbc.com/2017/09/29/biotech-stocks-drop-after-fda-makes-drug-side-effects-more-searchable.html
Perspective as we go unrecognized by the "wall"
I'd also like to add that the author is using old FDA trial design. The FDA will introduce the new procedures soon enough and should benefit baby bio nicely.
With that reasoning, why keep Fragile X, Infantile Spasms, Angelmans up there too?
