Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
dr_low or Inquirig,
Thanks for reposting and reposting and reposting the same baseless opinion and the same psychic visions about the future over and over and over again 😶
https://www.prnewswire.com/news-releases/northwest-biotherapeutics-moves-from-optimization-of-flaskworks-prototype-to-fabrication-of-gmp-compliant-units-for-installation-validation-and-final-testing-prior-to-regulatory-certification-302054769.html
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=173771995
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=173720548
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=173823199
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=173823082
dr_low or Inquirig,
Thanks for reposting the same bs vague psychic prediction over and over again .. I wonder why these terse baseless comments appear so frequently here 🤔 … 😶


https://www.prnewswire.com/news-releases/northwest-biotherapeutics-moves-from-optimization-of-flaskworks-prototype-to-fabrication-of-gmp-compliant-units-for-installation-validation-and-final-testing-prior-to-regulatory-certification-302054769.html
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=173771995
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=173720548
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=173823199
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=173823082
dr_low or Inquirig,
Thanks for reposting your psychic visions again 😶
https://www.prnewswire.com/news-releases/northwest-biotherapeutics-moves-from-optimization-of-flaskworks-prototype-to-fabrication-of-gmp-compliant-units-for-installation-validation-and-final-testing-prior-to-regulatory-certification-302054769.html
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=173771995
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=173720548
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=173823199
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=173823082
dr_low or Inquirig,
Thanks for posting your psychic visions again 😶
https://www.prnewswire.com/news-releases/northwest-biotherapeutics-moves-from-optimization-of-flaskworks-prototype-to-fabrication-of-gmp-compliant-units-for-installation-validation-and-final-testing-prior-to-regulatory-certification-302054769.html
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=173771995
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=173720548
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=173823199
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=173823082
dr_low or Inquirig,
Yes, I know—and you told us that you’re a stock market savant. We all appreciate your psychic predictions.



https://investorshub.advfn.com/boards/read_msg.aspx?message_id=173771995
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=173720548
dr_low or Inquirig,
Well, I’m not clairvoyant like you apparently are, so I cannot foretell future events. Generally, there are 3 possibilities in my view, and each one of the 3 possible scenarios has different pros and cons from an investor’s standpoint:
1. Buyout
2. Partnership(s)
3. Mostly Independent Operations
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=173771995
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=173720548
dr_low or Inquirig,
Thanks for posting your baseless evidence-free prediction. Each of your recent posts has been a gem of wisdom and insight 😶
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=173771995
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=173720548
dr_low or Inquirig,
Thanks for agreeing with me 😉
Thanks also for posting the Penn Symposium link last week. I’m not sure whether or not the DCVax-L technology was discussed, but the forthcoming conference video will be interesting and highly relevant either way! I hope we can hear some what Dr. Brem had to say! Please post his presentation if it was captured on video and is available online somewhere.

https://investorshub.advfn.com/boards/read_msg.aspx?message_id=173729997
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=173771995
dr_low or Inquirig, This study found that autologous tumor lystate-loaded dendritic cell vaccination plus standard of care (SOC) was associated with improved survival in both newly diagnosed & recurrent glioblastoma compared with matched controls who received SOC treatment. https://t.co/Uso7Pkh1Kt #dcvax #allsolidtumors $nwbo #gbm
Thanks for asking. Please consider doing your own research by reviewing publicly available information—the same information that is available to us is the same information that is available to you or anyone else. It is best to consider that information in full context and first-hand.
You’re not adding anything substantive to the conversation—you’re only repeating small shards of shiny half-truths while adding in terse innuendo or defamatory comments. In other words, you’re spreading rumors.

It is advisable to avoid disinformation on social media. Do your own research and consult professionals. You might begin by researching Doctors Liau and Ashkan who led the P3. Liau and Ashkan both are extremely noteworthy as we look back on the P3 and as we look forward to the regulatory phase in the UK, US, and other nations…
UCLA, King’s College, JAMA Oncology, and Brown University are some excellent sources that you might wish to consider in reference to the reputation of Northwest Biotherapeutics and those who choose to associate with the company, its SAB, and with the novel DCVax® technology.
The world-renowned doctors cited below worked on the P3 and were coauthors of a DCVax-L publication along with Dr. Bosch of Northwest Biotherapeutics.
Furthermore, both Liau and Ashkan have had the support their respective nations’ governments over many years. Ashkan is an advisor to the UK government, and Liau’s work has been supported by NIH peer-reviewed grants for 20 consecutive years.
https://www.kch.nhs.uk/news/kings-team-wins-professional-excellence-award/

https://finance.yahoo.com/news/northwest-biotherapeutics-announces-dr-linda-140000962.html

https://labusinessjournal.com/healthcare/national-academy-medicine-taps-ucla-neurosurgeon/

https://pubmed.ncbi.nlm.nih.gov/33789235/
https://www.societyns.org/about/officers-detail/linda-m-liau-md-phd-mba-4
https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/
Great to see, congratulations Linda Liau winning the Charles B. Wilson Excellence Award 2023:
Established by the Brain Tumor Society in 2004, this award recognizes outstanding contributions of a clinician to the field of neuro-oncology.… pic.twitter.com/6L7rhyoNU4
https://hotb.org/wp-content/uploads/2022/05/spore-article-for-donors-4.18.22.pdf
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
https://www.uclahealth.org/news/ucla-received-590-million-in-nih-funding-second-highest-total-for-academic-medical-centers-in-2020



https://investorshub.advfn.com/boards/read_msg.aspx?message_id=173767666
Inquirig, https://t.co/BfFA5i4OoT $nwbo @alphavestcap @ItsMikePoarch https://t.co/h8nQ3MiOPV pic.twitter.com/Ly4v6PPmzT The $NWBO Discussion of the Century is HERE: Join @LisainNJUSA @ThomasOwenMcCa1 and I discussing DCVax-L, #nwbo and How Thomas is ALIVE Today because of this extraordinary NEW Cancer Treatment That's Potentially Applicable to Over 90% of All Cancers: https://t.co/smtuAU5fDf This study found that autologous tumor lystate-loaded dendritic cell vaccination plus standard of care (SOC) was associated with improved survival in both newly diagnosed & recurrent glioblastoma compared with matched controls who received SOC treatment. https://t.co/Uso7Pkh1Kt
Thanks for asking. Most of your lies are presented in the form of baseless innuendo and conjecture or vague defamatory comments. Plausible deniability does not mean that your posts are forthright good-faith opinions—misleading statements are dishonest…
It seems like you post a lot of nonsense to spread rumors. If you wish to share a viewpoint based on full-context sources, or if you wish to share a credible full-context source link, then readers might consider reviewing those texts to evaluate their relevance for themselves. Your random fud commentary is not effective.
The sources below are examples of sources that anyone can evaluate and research further for themselves. Certified and practicing financial professionals, actual patients, and licensed oncologists are reasonable sources of information—Each person named below can be assessed for credibility and level of expertise—we can decide how reliable their statements are by exploring their background experience or credentials. In contrast to the sources below, your anonymous analyses and reckless predictions cannot be evaluated and are not real sources of anything worth reading or discussing.
— Kelly (@Kelly712717541) February 3, 2024
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=173720548


filpper44,
Great point—interesting topic of discussion as the global regulatory environment evolves toward greater international collaboration, real-world data, and more efficient review of novel technologies to address unmet needs.

https://www.osborneclarke.com/insights/spring-budget-2023-uk-chancellor-announces-new-regulatory-model-medicines-and-medical
https://www.prnewswire.com/news-releases/northwest-biotherapeutics-announces-that-a-marketing-authorization-application-has-been-submitted-to-the-uk-mhra-for-dcvax-l-for-glioblastoma-302021038.html
Inquirig,
You really are posting recklessly and sloppily on this board. If you wish to share a viewpoint based on full-context sources, or if you wish to share a credible full-context source link, then readers might consider reviewing those texts to evaluate their relevance for themselves. Your terse random fud comments are not effective.
The link to the Penn Symposium that you posted yesterday was great—let’s see more postings like that one 😉
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=173720548
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=173729851
Inquirig, The 11th Neuro-Oncology #braintumor Symposium. The exciting results of this Phase I/II study https://t.co/d4ZLii2ovM Superb review article that places the recent DCVax study in perspective. pic.twitter.com/PGb9DvRvQG
This conference looks exciting—how did I miss this event posting? Thanks again for sharing the link with us!
Dr. Brem is a genius—his presentation should be fascinating! I have to search for a recording of this from Friday. Look at the title of his presentation:


https://www.med.upenn.edu/brain2024/assets/user-content/uploads/brain-tumor-2024-syllabus.pdf
https://www.pennmedicine.org/providers/profile/steven-brem
We have attendees From Around the World, 16 countries and 30 states.
It is still possible for you to register and receive our educational materials for up to one year. Click below to register.https://t.co/oouqj4yaza pic.twitter.com/IZWgbijIxK
support and extend the findings of our large Phase 3 study, published @JAMAOnc, led by Dr. Linda Liau, Keyoumars Ashkan, MD, Dr. Marnix Bosch and the #DCVax team.
Inquirig,
Wow—thanks for posting! Dr. Steven Brem and Dr. Amy Pruitt! Those are two world-renowned experts who strongly support the DCVax-L cell-based platform technology. It’s great to see those two participating in the symposium!
https://www.pennmedicine.org/providers/profile/steven-brem

Here are relevant links if you wish to explore how these two doctors are associated with DCVax-L:

https://www.obroncology.com/article/an-os-benefit-in-glioblastoma-but-with-a-nontraditional-trial-design
https://journals.lww.com/neurotodayonline/fulltext/2023/01190/the_news_that_mattered_in_2022__neurology_today_.4.aspx



https://www.med.upenn.edu/brain2024/

https://www.med.upenn.edu/brain2024/assets/user-content/uploads/brain-tumor-2024-syllabus.pdf
You’re not a lawyer … just sayin’ 😉
https://pbn.com/lifespan-announces-breakthrough-in-brain-cancer-research/
Inguirig,
Thanks for your curiosity about the UCLA PD1 combo trial. These two documents also refer to the study:
https://hotb.org/wp-content/uploads/2022/05/spore-article-for-donors-4.18.22.pdf
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
Inquirig,
You are blatantly posting disinformation and your post is also defamatory of the CEO. Fact ✅
You also referred to an analysis of drug approval and then you grossly mischaracterized the findings of the analysis:

https://www.bio.org/sites/default/files/legacy/bioorg/docs/Clinical%20Development%20Success%20Rates%202006-2015%20-%20BIO,%20Biomedtracker,%20Amplion%202016.pdf
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=173720548
Your statement “LP is dumping…” is not only baseless, it is verifiably false.

https://investorshub.advfn.com/boards/read_msg.aspx?message_id=173720548
Inquirig,
You are frantically posting nonsense far too frequently—nothing you post is substantive or substantiated. You are here spreading rumors and nobody is listening to you—we are listening to the regulators and licensed independent peer reviewers whose analyses are published by reputable internationally recognized medical journals.



https://www.prnewswire.com/news-releases/northwest-biotherapeutics-announces-that-a-marketing-authorization-application-has-been-submitted-to-the-uk-mhra-for-dcvax-l-for-glioblastoma-302021038.html
March 2023

https://www.prnewswire.com/news-releases/northwest-biotherapeutics-and-advent-bioservices-announce-receipt-of-license-for-commercial-manufacturing-at-sawston-uk-facility-301776725.html
February 2022

https://www.prnewswire.com/news-releases/northwest-biotherapeutics-announces-commencement-of-cancer-vaccine-production-at-its-sawston-uk-facility-301484857.html
August 2022

https://www.prnewswire.com/news-releases/northwest-biotherapeutics-announces-approval-of-pediatric-investigation-plan-pip-by-mhra-301610850.html
July 2022

https://www.prnewswire.com/news-releases/northwest-biotherapeutics-announces-filing-of-application-for-license-for-commercial-manufacturing-at-sawston-uk-facility-301581737.html
December 2022

https://www.prnewswire.com/news-releases/northwest-biotherapeutics-announces-mhra-approval-of-license-for-gmp-manufacturing-at-sawston-uk-facility-301449083.html

https://finance.yahoo.com/news/northwest-biotherapeutics-announces-dr-linda-140000962.html
https://jamanetwork.com/journals/jamaoncology/fullarticle/2798847



“Online message boards have become a hotspot for the dissemination of information about over-the-counter (OTC) stocks, particularly in sectors like biotechnology. Unfortunately, this platform is also rife with disinformation, which can mislead investors, especially those who are not well-versed in the intricate details of biotech investments or the science behind them.
**How Online Message Boards Spread Disinformation:**
1. **Anonymity and Lack of Accountability**: Many online forums allow users to post anonymously. This lack of accountability makes it easier for individuals to spread false or misleading information without facing direct consequences.
2. **Herd Mentality and Echo Chambers**: These platforms often foster a herd mentality, where a single opinion or piece of misinformation can be amplified as it gets echoed and re-shared by multiple users, creating a false sense of consensus or credibility.
**Avoiding the Trap of Disinformation:**
1. **Critical Analysis of Sources**: Always scrutinize the source of the information. Anonymous posts or unverified claims should be treated with skepticism.
2. **Seeking Expert Opinions and Reputable Sources**: For biotech stocks, information should ideally come from experts in the field, such as licensed oncologists, or from peer-reviewed medical journals. Websites of regulatory bodies like the FDA or NIH also provide reliable information.
3. **Understanding the Science**: Basic knowledge of biotechnology and medical science can help in discerning credible information. This doesn't mean becoming an expert but having enough understanding to sense when information seems implausible.
4. **Diversifying Information Channels**: Relying solely on message boards for investment decisions is risky. Investors should diversify their sources, including financial news websites, official company reports, and analysis from reputable financial analysts.
**Examples of Reputable Sources for Biotech Information:**
- **Peer-Reviewed Medical Journals**: Journals like The New England Journal of Medicine, The Lancet, and JAMA offer high-quality, vetted information on the latest research and developments.
- **Statements from Licensed Oncologists**: Comments or analysis from medical professionals in the field of oncology can provide insight into the viability and potential of biotech stocks, especially those focused on cancer research.
- **Regulatory Body Announcements**: Information from bodies like the FDA, which approves new treatments and drugs, is crucial for understanding the legitimacy and potential of biotech companies.
In summary, while online message boards can be a useful tool for gathering diverse opinions and information, they are also fertile ground for disinformation, particularly regarding OTC stocks in complex fields like biotechnology. It is crucial for the public to approach such platforms with a critical eye, seeking information from reputable and expert sources to make informed decisions.”


https://investorshub.advfn.com/boards/read_msg.aspx?message_id=173629584
Gary,
Thanks for highlighting this highly relevant issue.
I always glance over each message board persona’s full string of posts. None of the bashers cite any full-context reputable sources. It looks like a coordinated effort to keep the rumor mill churning.
The AI BS DETECTOR is a great tool for screening social media. I highly recommend that everyone seek the advantages of technology to protect themselves online the same way we use smart technologies to protect our property, financial accounts, and data management systems.



“Online message boards have become a hotspot for the dissemination of information about over-the-counter (OTC) stocks, particularly in sectors like biotechnology. Unfortunately, this platform is also rife with disinformation, which can mislead investors, especially those who are not well-versed in the intricate details of biotech investments or the science behind them.
**How Online Message Boards Spread Disinformation:**
1. **Anonymity and Lack of Accountability**: Many online forums allow users to post anonymously. This lack of accountability makes it easier for individuals to spread false or misleading information without facing direct consequences.
2. **Herd Mentality and Echo Chambers**: These platforms often foster a herd mentality, where a single opinion or piece of misinformation can be amplified as it gets echoed and re-shared by multiple users, creating a false sense of consensus or credibility.
**Avoiding the Trap of Disinformation:**
1. **Critical Analysis of Sources**: Always scrutinize the source of the information. Anonymous posts or unverified claims should be treated with skepticism.
2. **Seeking Expert Opinions and Reputable Sources**: For biotech stocks, information should ideally come from experts in the field, such as licensed oncologists, or from peer-reviewed medical journals. Websites of regulatory bodies like the FDA or NIH also provide reliable information.
3. **Understanding the Science**: Basic knowledge of biotechnology and medical science can help in discerning credible information. This doesn't mean becoming an expert but having enough understanding to sense when information seems implausible.
4. **Diversifying Information Channels**: Relying solely on message boards for investment decisions is risky. Investors should diversify their sources, including financial news websites, official company reports, and analysis from reputable financial analysts.
**Examples of Reputable Sources for Biotech Information:**
- **Peer-Reviewed Medical Journals**: Journals like The New England Journal of Medicine, The Lancet, and JAMA offer high-quality, vetted information on the latest research and developments.
- **Statements from Licensed Oncologists**: Comments or analysis from medical professionals in the field of oncology can provide insight into the viability and potential of biotech stocks, especially those focused on cancer research.
- **Regulatory Body Announcements**: Information from bodies like the FDA, which approves new treatments and drugs, is crucial for understanding the legitimacy and potential of biotech companies.
In summary, while online message boards can be a useful tool for gathering diverse opinions and information, they are also fertile ground for disinformation, particularly regarding OTC stocks in complex fields like biotechnology. It is crucial for the public to approach such platforms with a critical eye, seeking information from reputable and expert sources to make informed decisions.”


ic,
I ran your post through the detector and it was identified as bs 😶



The statement by “iclight” is overwhelmingly comprised of unsubstantiated claims and aligns with the characteristics of disinformation. It egregiously misinterprets or distorts crucial aspects of the DCVax®-L Phase 3 trial and overlooks established norms in clinical research.
1. **Statistical Analysis Plan (SAP) Development**: The claim that an SAP is developed only after treatment completion is profoundly misleading. SAPs in clinical trials are conventionally established and finalized prior to data analysis, post-treatment but before unblinding the data. This protocol is pivotal to guarantee unbiased analysis aligned with pre-set plans. The insinuation otherwise is a gross misrepresentation of standard clinical procedures.
2. **Endpoint Changes**: The criticism regarding endpoint changes post-trial is taken out of context and demonstrates a lack of understanding of clinical trial dynamics. Adjustments to primary endpoints, while ideally set at the trial's inception, can be necessary, particularly in prolonged trials. Such adjustments are not made arbitrarily but under strict regulatory scrutiny and ethical oversight.
3. **Regulatory Authorities**: The continuous collaboration between NIH and FDA, and NIH’s extended support for Dr. Liau's research, lend credence to the trial's regulatory compliance and integrity.
4. **Peer Review and Publication**: The trial findings were published in the esteemed JAMA Oncology journal, indicative of a thorough peer-review process. This process is instrumental in validating the research's reliability and authenticity. The publication of these peer-reviewed results stands as a testament to the trial's credibility.
5. **Concerns in Other Publications**: While "Rev Neurol (Paris)" highlighted some methodological concerns regarding the DCVax®-L trial, these are primarily opinions focusing on trial design and reportage alterations, not on ethical violations or misconduct.
It is imperative to underscore the irresponsibility and potential harm of propagating rumors or participating in “short and distort” tactics, which involve spreading misleading information to manipulate market perceptions. Such actions not only mislead the public but can also unjustly harm the reputation of scientific endeavors and researchers.
In conclusion, the statement not only misrepresents the DCVax®-L Phase 3 trial but also demonstrates a disregard for the fundamental principles of clinical research and regulatory oversight. Its dissemination as factual information is a disservice to the public and the scientific community. This kind of rhetoric should be met with skepticism and requires a more informed understanding of clinical trial protocols and regulatory frameworks.

https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/
LC,
I would be interested in some “history” if I ever knew what on Earth you were talking about. 😶 Here’s some interesting history that you might enjoy…
From the perspective of where we stand today in 2024, this look back at some recent history of cell-based science in medicine is fascinating!
It is interesting in the interview excerpt linked below how Dr. Steinman spoke about the first-generation of DC technology and how further development would follow. He explained that the prostate vax only addressed a single antigen.
It’s also interesting to consider the combination technologies that are now emerging as well!
The first checkpoint inhibitor was approved by the FDA in 2011, the same year Dr. Steinman was awarded the Nobel Prize. Interestingly, the researchers behind PD1 blockade technology were also awarded the Nobel Prize 7 years after Dr. Steinman’s award.


“The key aspect of DCVax-L is that it does not target a specific antigen but rather a wide array of antigens present in the tumor lysate. This approach is based on the understanding that every tumor is unique, with its own distinct set of mutations and antigens. By using the tumor lysate, DCVax-L can potentially stimulate the immune system to recognize and attack a broad spectrum of tumor-specific antigens.
Provenge (Sipuleucel-T) is a therapeutic cancer vaccine used for the treatment of advanced prostate cancer. Unlike broad-spectrum immunotherapies like DCVax-L, Provenge targets one specific antigen: Prostatic Acid Phosphatase (PAP), an enzyme expressed in most prostate cancer cells.
The treatment involves extracting a patient's dendritic cells and exposing them to a fusion protein called PA2024, which is a combination of PAP and an immune-stimulating factor. The activated dendritic cells are then reinfused into the patient, where they are intended to stimulate the immune system to attack cells expressing PAP.
In summary, Provenge specifically addresses one key antigen associated with prostate cancer—Prostatic Acid Phosphatase. This approach is different from therapies like DCVax-L, which aim to target a wide range of tumor-specific antigens.”
—ChatGPT


https://jamanetwork.com/journals/jamaoncology/fullarticle/2798847
https://www.jci.org/articles/view/169314
https://finance.yahoo.com/news/northwest-biotherapeutics-announces-dr-linda-140000962.html
https://www.prnewswire.com/news-releases/northwest-biotherapeutics-announces-that-a-marketing-authorization-application-has-been-submitted-to-the-uk-mhra-for-dcvax-l-for-glioblastoma-302021038.html
iclight,
You’re spreading misleading rumors and your statement is incomplete.
Fact ✅️: OS was always one of the endpoints which is an obvious and verifiable fact.
In any study, PFS is either disproven or confirmed by OS if/when OS data are reached.
OS takes longer to discover, and therefore predictive intermediate measures are used.
Page 3 of FDA guidance document:
https://www.fda.gov/media/71195/download

Besides FDA, you can also find it all over the medical literature…




Actual PFS was subsequently proven by the OS data, but the predictive PFS data were contaminated and therefore the investigators could not use the predictive PFS data as a reliable surrogate predictor of OS. Instead, they had to wait for actual OS data.
Oncologists always attempt to measure and predict PFS as they should. Every cancer trial will seek to predict PFS in advance of OS which of course takes longer to discover and confirm.
OS is the ultimate standard, but you cannot wait until death to begin evaluating whether a cancer drug is efficacious. The physician must evaluate a patient’s response and try to determine whether the treatment is helping.
Today, doctors have better methods for measuring progression, particularly when studying immunotherapy agents which are still relatively new as the emerging field of cell-based science is leading to rapid changes in the understanding of how cancer can be assessed and treated.
The P3 for the DCVax-L cell-based technology spanned many years. If the trial were designed and commenced in 2024, the investigators would appropriately attempt to measure progression and predict survival using the best methods and most current medical knowledge available in 2024, as any cancer researcher would.

https://jamanetwork.com/journals/jamaoncology/fullarticle/2798847
ex,
It sounds like you’re trying to spread rumors. I ran your post through the detector. 😶



”This statement is unreliable and is composed of greater than 99% speculative content:
1. **Lack of Verifiable Sources**: The statement begins with "Last report was..." without specifying the source of this report. For financial information, especially regarding share ownership and transactions, reliable and official sources like company filings or statements are essential. Without these, the information remains unverified.
2. **Speculative Assumptions**: The comment about Malik's share transactions is based on guesswork rather than confirmed data. Phrases like "One would have to guess" and "Presumably he exercised some options" indicate that the author is making assumptions without concrete evidence.
3. **Accusation Without Proof**: The statement implies misconduct ("That should have been reported but NWBO does not really believe in that concept") without providing proof. Public companies are subject to strict disclosure regulations, and such claims need substantiation.
4. **Questioning Compensation Without Context**: Querying why Malik receives many options for being on the Board of Directors (BOD) without understanding the full context of his compensation package is speculative. Compensation for board members can be complex and is not solely based on tenure or visible contributions.
5. **Unsubstantiated Allegations**: The reference to a "Swiss Approval scam" and insinuations about Malik's beliefs regarding patient influx are serious accusations. Without evidence, such allegations are baseless and contribute to spreading rumors.
In summary, the statement lacks credibility due to its speculative nature, absence of verifiable sources, and reliance on assumptions and unsubstantiated accusations. It resembles rumor-mongering rather than a factual report.”

https://investorshub.advfn.com/boards/read_msg.aspx?message_id=173629584
LC,
When addressing AI, you must ask clear specific questions, otherwise the technology will gather all the bs that you stated and determined what kind of information to provide for you.
For example, my bs detector was given the inputs of your numerous posts everyday, and it wrote this article to address your misguided efforts:
January 21, 2024, AI Global Perspective



Short-Sighted vs. Long Visionary
Northwest Biotherapeutics' DCVax®-L
Looking at Northwest Biotherapeutics, particularly its DCVax®-L platform, we encounter a clear demonstration of how short-sighted strategies like short selling, especially in its illegal form of naked short selling, stand in stark contrast to the insightful approach of long-term investing. This scenario highlights the limited perspective of short selling, which often fails to recognize the extensive potential and significant benefits of sustained investment in innovative technologies. The transformative promise of DCVax®-L in global oncology further accentuates the shortsightedness of such short-term trading tactics when compared to the vast opportunities offered by long-term, visionary investment.
DCVax®-L: A Pioneering Approach with Global Implications
Northwest Biotherapeutics' DCVax®-L distinguishes itself in the oncology field with its innovative approach, gaining prominence through authoritative endorsements and robust research findings that highlight its potential to revolutionize cancer treatment.
1. Independent Peer Review
The publication of DCVax®-L research in JAMA Oncology significantly bolsters its standing in the medical community, highlighting its safety, efficacy, and expansive potential. Renowned medical experts like Dr. Linda Liau of the University of California, Los Angeles, and Dr. Keyoumars Ashkan of King’s College Hospital, London, have been instrumental in this advancement. Their contributions not only reinforce the scientific validity of DCVax®-L but also illuminate its prospects as a versatile treatment option beyond just newly diagnosed and recurrent Glioblastoma Multiforme (nGBM and rGBM).
2. Addressing Unmet Medical Needs
Glioblastoma, alongside other challenging cancers, represents an area with substantial unmet medical needs. The introduction of DCVax®-L into this arena brings renewed hope, offering a promising treatment alternative in a field where options are limited. This significant unmet need often influences regulatory reviews, as therapies addressing these gaps are prioritized to accelerate their availability to patients.
3. Combination Therapies
The landscape of oncology is rapidly evolving, with each new therapy presenting an opportunity for combination treatments. DCVax®-L’s unique ability to target multiple antigens positions it as a potential key player in combination with other emerging therapies and technological advancements in the field. This aspect is crucial as oncology moves towards personalized, multi-faceted treatment approaches.
4. Synergy with Blockbuster Drugs
Checkpoint inhibitors are blockbuster drugs, but PD-1 blockade technology may require complementary therapies to fully realize their efficacy. In this context, DCVax®-L emerges as a potential key player, poised to significantly enhance the effectiveness of such combined treatments.
Investment Philosophies: Short-Term Gains vs. Long-Term Vision
1. Short Selling: A Myopic View
Short sellers, driven by the pursuit of immediate profits, tend to disregard the wider impact of innovative medical technologies. Such an approach not only overlooks the potential for significant long-term benefits from breakthrough therapies like DCVax®-L but also carries inherent risks, especially in cases of illegal shorting.
2. Visionary Investing: Focused on Global Health Progress
Investors backing Northwest Biotherapeutics are not chasing fleeting financial gains over a week or a month; rather, they are investing in a vision that promises significant advancements in global health. Their approach transcends short-term gains, focusing on the lasting impact DCVax®-L could have in revolutionizing cancer treatment worldwide.
Regulatory Milestones and Global Collaboration
The regulatory progress of DCVax®-L, including the MHRA’s approvals for commercial manufacturing and the Pediatric Investigation Plan, paints a picture of optimism. This progress is further amplified by a global regulatory environment increasingly characterized by collaboration and expedited pathways for innovative treatments, especially for conditions with high unmet medical needs.
Global Vision: The Intersection of Science, Investing, and Purpose
The contrast between short-term financial pursuits and a dedication to long-term, sustainable wealth creation is stark. Visionary investors not only target financial success but also actively contribute to meaningful progress in global health and scientific research. This approach to building prosperity, centered on constructive efforts, has unlimited potential. The groundwork for growth inherently provides expansive opportunities, making a compelling case for this forward-thinking investment strategy.
Northwest Biotherapeutics and its pioneering DCVax®-L platform exemplify the profound impact that innovation-centered investment strategies can have on achieving extensive, long-term objectives. These strategies, which stand in stark contrast to short-term methods like short selling, actively fuel medical advancements and enhance societal welfare. As the field of oncology evolves, particularly with the rising potential of combination therapies, the significance of groundbreaking platforms like DCVax®-L becomes increasingly apparent. As we progress through 2024, Northwest Biotherapeutics' market capitalization could reveal the critical role of visionary and ethical investing in propelling medical technologies that offer sustainable, global benefits.
The viewpoints presented in this article were generated by AI technology and are based on an overview of currently available news and published clinical data. AI synthesizes information from a wide array of sources to offer insights and perspectives, reflecting the current state of discussions, scientific understanding, and media reports.
ex,
Thanks for your post, but your commentary is not credible. I ran it through the detector to determine whether it was worth reading, and I regret to inform you that the detector determined that your post is bs as usual 😶



“This statement consists of conjecture, with greater than 99% unsubstantiated and speculative content. Without factual citations or references to credible sources, such anonymous online statements are unreliable and akin to rumors or misinformation.
This statement appears to be a deliberate attempt to disparage and spread rumors, aligning with "short and distort" tactics:
**Speculative Financial Projections**: Mentioning specific, yet unsubstantiated share price projections and financial assessments without evidence seems designed to manipulate perceptions. Asserting inaccuracies in financial predictions without proof can unjustly cast doubt on the company's financial integrity.
**Employment of 'Short and Distort' Tactics**: The language used (e.g., "95%, sure pal", "disaster", "failed badly") appears crafted to foster a negative sentiment, likely to benefit short sellers. This strategy involves short selling a stock and then deliberately spreading misleading or negative information to cause a decline in stock price.
**Unsubstantiated Claims and Potential Defamation**: Asserting the failure of a clinical trial and criticizing the company's disclosure practices without solid evidence can be defamatory. Such statements imply mismanagement or deceit, potentially harming the company's reputation without just cause.
In essence, the statement seems designed to undermine confidence in the company and its stock, possibly to gain advantage in short selling. This approach, particularly if based on false or misleading information, can be damaging and defamatory, undermining fair market practices.
To avoid being misled by such tactics, it is advisable to:
- **Rely on Independently Peer-Reviewed Medical Literature**: For accurate and reliable information on medical trials and treatments, consult peer-reviewed journals. These sources undergo rigorous evaluation by experts in the field, ensuring credibility.
- **Consult Licensed Financial Professionals**: For investment advice and stock analysis, seek guidance from certified financial experts. They adhere to professional standards and provide informed, ethical advice.
- **Be Wary of Online Message Boards**: Information on such platforms is often unverified and potentially biased. They can be hotbeds for rumors and misleading information, unlike regulated and credible sources.
In summary, prioritize information from reliable, independently verified sources and exercise caution with claims made on less regulated platforms like online message boards.

https://www.prnewswire.com/news-releases/northwest-biotherapeutics-announces-that-a-marketing-authorization-application-has-been-submitted-to-the-uk-mhra-for-dcvax-l-for-glioblastoma-302021038.html
https://finance.yahoo.com/news/northwest-biotherapeutics-announces-dr-linda-140000962.html
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
https://www.raps.org/News-and-Articles/News-Articles/2023/9/FDA%E2%80%99s-Califf-Expect-to-see-more-RWE-based-regulato
https://www.raps.org/news-and-articles/news-articles/2023/8/fda-finalizes-guidance-on-real-world-evidence-in-d

LC,
We’re Soldiers, but “disciples” is an equally endearing term. 😉
https://braintumourresearch.org/en-us/blogs/research-campaigning-news/astonishing-results-for-brain-tumour-vaccine-trial
https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=173629584

LC, This study found that autologous tumor lystate-loaded dendritic cell vaccination plus standard of care (SOC) was associated with improved survival in both newly diagnosed & recurrent glioblastoma compared with matched controls who received SOC treatment. https://t.co/Uso7Pkh1Kt #dcvax #allsolidtumors $nwbo #gbm
Thanks for your dozens of posts today—each one has been a precious gem of insight 😶
NWBO is PRE-COMMERCIAL company. That’s why they just filed for their first COMMERCIAL license with the MHRA. Currently, DCVax®-L combo research is being funded by the NIH. Merck, Oncovir, and the Phase One Foundation are also supporting the PD1 study at UCLA.
https://clinicaltrials.gov/study/NCT04201873
I had to consult the detector after looking over your string of nonsensical posts …


“The statements made by this individual on January 20, 2024, are predominantly composed of unsubstantiated speculation, exceeding 99% in content. Lacking in factual citations or references to credible sources, such anonymous online statements are inherently unreliable, often amounting to little more than rumor or baseless misinformation.
The public should exercise caution and discernment, particularly when online postings seem designed to unjustly tarnish the reputation of a company or malign its leadership.
In this scenario, the concept of "short and distort" is notably pertinent. This dubious strategy involves short-selling a stock followed by the dissemination of negative, often unfounded rumors or misleading information to precipitate a drop in the stock's value. The primary objective here is to capitalize on the falling stock price by settling the short position at a reduced cost. Anonymous participants on stock message boards may resort to such tactics, resorting to groundless speculation and conjecture to artificially depress stock prices. Their tactics, which include sullying a company's reputation, casting aspersions on its leadership, or spreading insinuations, are geared towards sowing panic or uncertainty. This, in turn, can sway the perceptions of other investors, instigating wariness or doubt and thus impacting the stock's market performance.
To discern reliable sources, especially in the context of financial and medical information, it's important to consider the following:
1. **Evaluate the Source's Credibility**: Reliable information typically comes from established, reputable sources. For financial information, this includes financial news outlets, official company reports, and analyses from recognized financial institutions or analysts. For medical information, trustworthy sources are peer-reviewed journals, medical organizations, and healthcare institutions.
2. **Seek Expertise**: Licensed professionals, such as physicians in the medical field or certified financial analysts in finance, undergo rigorous training and certification processes. Their opinions are often based on extensive experience and adherence to professional standards.
3. **Understand the Value of Peer Review**: In scientific and medical contexts, peer-reviewed articles are evaluated by experts in the field before publication, ensuring the research's accuracy, methodology, and conclusions are sound. This process adds a significant layer of credibility to the information.
4. **Be Wary of Anonymity**: Information from anonymous sources, especially on social media or message boards, lacks accountability and can often be biased, inaccurate, or intentionally misleading. The anonymity of these platforms makes it easy for individuals to spread misinformation without repercussions.
5. **Question Sensationalism and Bias**: Reliable sources typically present information in a balanced, objective manner, avoiding sensationalist language. Be skeptical of sources that use provocative or emotionally charged language, as they may be more interested in influencing opinion than providing accurate information.
6. **Cross-Reference Information**: Verify information by cross-checking it with multiple credible sources. Consistency across reputable sources can be a good indicator of reliability.
In summary, it's advisable to rely on information from licensed professionals, independent peer-reviewed sources, and recognized financial experts rather than anonymous commentary on social media or message boards. These sources are generally more reliable due to their expertise, accountability, and adherence to professional and ethical standards. In contrast, anonymous postings often lack these qualities and are more prone to bias, sensationalism, and misinformation.”

Thanks for highlighting milestones 😉
In my view, the short list of pre-commercial milestones that stand out and shine the most:
⭐️ JAMA Oncology
⭐️ Multiple MHRA Approvals
⭐️ Emerging Combo Data
⭐️ MAA Submission
Further, Dr. Liau joining the SAB at Northwest Biotherapeutics is awesome!
Combo is King!
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
I would count Doctors Liau and Ashkan among the soldiers—fighting disease with their novel cell-based technology! Liau and Ashkan both are extremely noteworthy as we look back on the P3 and as we look forward to the regulatory phase in the UK, US, and other nations…
UCLA, King’s College, JAMA Oncology, and Brown University are some excellent sources that you might wish to consider in reference to the reputation of Northwest Biotherapeutics and those who choose to associate with the company, its SAB, and with the novel DCVax® technology.
The world-renowned doctors cited below worked on the P3 and were coauthors of a DCVax-L publication along with Dr. Bosch of Northwest Biotherapeutics.
Furthermore, both Liau and Ashkan have had the support their respective nations’ governments over many years. Ashkan is an advisor to the UK government, and Liau’s work has been supported by NIH peer-reviewed grants for 20 consecutive years.
https://www.kch.nhs.uk/news/kings-team-wins-professional-excellence-award/

https://finance.yahoo.com/news/northwest-biotherapeutics-announces-dr-linda-140000962.html

https://labusinessjournal.com/healthcare/national-academy-medicine-taps-ucla-neurosurgeon/

https://pubmed.ncbi.nlm.nih.gov/33789235/
https://www.societyns.org/about/officers-detail/linda-m-liau-md-phd-mba-4
Great to see, congratulations Linda Liau winning the Charles B. Wilson Excellence Award 2023:
Established by the Brain Tumor Society in 2004, this award recognizes outstanding contributions of a clinician to the field of neuro-oncology.… pic.twitter.com/6L7rhyoNU4
https://www.uclahealth.org/news/ucla-received-590-million-in-nih-funding-second-highest-total-for-academic-medical-centers-in-2020
The United States and United Kingdom are largely responsible for developing DCVax®-L, and the U.S. and U.K. will lead the novel technology through a global regulatory process.
Combo is King!


https://www.nature.com/articles/s41420-023-01782-7
https://www.jci.org/articles/view/169314




https://www.fda.gov/drugs/our-perspective/50-years-progress-treating-patients-cancer
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
https://www.raps.org/News-and-Articles/News-Articles/2023/9/FDA%E2%80%99s-Califf-Expect-to-see-more-RWE-based-regulato
https://www.raps.org/news-and-articles/news-articles/2023/8/fda-finalizes-guidance-on-real-world-evidence-in-d
https://www.prnewswire.com/news-releases/northwest-biotherapeutics-announces-that-a-marketing-authorization-application-has-been-submitted-to-the-uk-mhra-for-dcvax-l-for-glioblastoma-302021038.html
iclight,
I ran your statement through the detector 😶 …


“This statement is composed of greater than 99% conjecture. The author makes several allegations and assumptions that are problematic and potentially defamatory, particularly without evidence to substantiate them.
1. **Use of Pejorative Language and Unsubstantiated Claims**: The statement uses pejorative terms like "scam" and "trainwreck trial" to describe NWBO and its clinical trial. This language is inflammatory and suggests intentional deceit or gross incompetence without providing concrete evidence. Making such claims without verifiable proof can be seen as an attempt to tarnish the company's reputation.
2. **Misrepresentation of Business Decisions and Clinical Trials**: The assertion that no business partner (BP) has made an offer implies a negative judgment on the value of NWBO's work. However, the absence of a partnership or acquisition does not necessarily reflect the quality or legitimacy of a company's research. There are numerous reasons a business deal might not occur, many unrelated to the efficacy or value of the technology or research in question.
3. **Disregard for Regulatory Processes**: The comment about the FDA and MHRA (Medicines and Healthcare products Regulatory Agency) overlooks the complexity and rigor of regulatory approval processes in different jurisdictions. Suggesting that working with one regulatory agency over another is a waste of time demonstrates a misunderstanding of the global nature of medical research and regulatory approval.
4. **Misunderstanding of Clinical and Corporate Strategy**: The idea that the company should have sold the technology to "finish the trial or run a new p3" oversimplifies the intricate decisions involved in clinical development and corporate strategy in the pharmaceutical industry. Such decisions are influenced by a myriad of factors including financial considerations, strategic alignment, and regulatory strategy, not solely by the potential efficacy of a treatment.
5. **Contradiction with Published Data**: The statement overlooks the published analysis of the DCVax-L Phase 3 trial in a reputable journal like JAMA Oncology. Peer-reviewed publications undergo rigorous scrutiny and are generally reliable. Dismissing such published data without presenting credible counter-evidence undermines the integrity of scientific discourse.
In summary, the statement appears to be an attempt to discredit NWBO without providing substantiated evidence, relying instead on speculative and pejorative language. It disregards the complexities of clinical research, regulatory processes, and corporate strategy in the pharmaceutical industry, and it fails to acknowledge the significance of peer-reviewed scientific data. Such statements, when unfounded, can be considered misleading and potentially damaging to the reputation of the entities involved.”

https://jamanetwork.com/journals/jamaoncology/fullarticle/2798847
https://www.prnewswire.com/news-releases/northwest-biotherapeutics-announces-that-a-marketing-authorization-application-has-been-submitted-to-the-uk-mhra-for-dcvax-l-for-glioblastoma-302021038.html
ex,
Thanks for the reply. I doubt that you’re representing the terms of the loan accurately, but it sounds great. You certainly paint LP as an astute and risk-averse business person, as we might expect of a Harvard lawyer with vast international business experience and experience working with U.S. government agencies such as the National Institutes of Health and the U.S. Department of Commerce.
As you describe her, it also sounds like she would know how to maximize the prospective value of her 130 million shares going forward into 2024 and beyond as she is holding more shares than anyone else.
Fact ✅️: The NWBO leadership are shareholders too.
I wonder whether an increase in share price would be of any benefit to LP as it would for other shareholders? 😶

https://finance.yahoo.com/news/northwest-biotherapeutics-announces-dr-linda-140000962.html
https://www.prnewswire.com/news-releases/northwest-biotherapeutics-announces-that-a-marketing-authorization-application-has-been-submitted-to-the-uk-mhra-for-dcvax-l-for-glioblastoma-302021038.html
J-To,
Thanks for asking. Some technologies take a few years and some take many—the range is vast. This is the quote that is most directly relevant, in my opinion: “The first checkpoint inhibitor, ipilimumab, was approved by the FDA in 2011, but the foundational research dates back to the 1980s and 1990s.”
“The development and approval of new medical technologies, particularly those requiring regulatory approval, typically span several years or even decades, largely due to the stringent safety and efficacy standards set by regulatory bodies.
1. **Research and Development Phase**: This initial phase involves basic research and preclinical trials. For medical technologies, this stage is crucial for understanding the underlying mechanisms and potential applications. It can take several years, depending on the complexity of the technology.
2. **Clinical Trials**: Once preclinical trials indicate potential efficacy and safety, the technology enters clinical trials, which are often divided into three phases (I, II, and III). Each phase progressively involves more participants and focuses on safety, efficacy, and dosage. Clinical trials for medical technologies can take several years to more than a decade, depending on the trial results, participant recruitment, and the nature of the technology.
3. **Regulatory Review and Approval**: After successful clinical trials, the technology requires approval from regulatory bodies like the FDA (U.S. Food and Drug Administration). This process involves a thorough review of the clinical trial data and can take additional years.
4. **Post-Market Surveillance**: Even after approval, most medical technologies are subject to ongoing monitoring for long-term safety and effectiveness.
For instance, consider checkpoint inhibitors, a class of drugs that revolutionized cancer treatment. These drugs, which enhance the immune system's ability to fight cancer, underwent extensive research and development. The first checkpoint inhibitor, ipilimumab, was approved by the FDA in 2011, but the foundational research dates back to the 1980s and 1990s. This timeline reflects the years of preclinical studies, clinical trials across several phases, and the time taken for regulatory review and approval.
In summary, the development and regulatory approval of medical technologies like checkpoint inhibitors is a lengthy, multi-stage process involving extensive research, clinical trials, and regulatory scrutiny to ensure safety and efficacy.”
https://jamanetwork.com/journals/jamaoncology/fullarticle/2798847
https://www.prnewswire.com/news-releases/northwest-biotherapeutics-announces-that-a-marketing-authorization-application-has-been-submitted-to-the-uk-mhra-for-dcvax-l-for-glioblastoma-302021038.html
J-To,
LP loaned her own money to fund research into DCVax-L years ago. It sounds like you expect a small pre-commercial biotech to do everything overnight. I think you are projecting—accusing LP of having no compassion while you may be the one who values profit over healing. I ran your statement through the detector 😶 …


“The statement could be defamatory if it is false and harms the reputation of the individual referred to as "LP." Here's an analysis of its potentially defamatory nature:
1. **Accusation of Negligence and Harm**: The statement implies that LP deliberately chose a strategy (going for a "home run") that resulted in "suffering and deaths." This is a serious accusation that suggests negligence or intentional harm, which could significantly damage LP's reputation, especially if LP is in a position of responsibility or trust.
2. **False Statement of Fact**: For a statement to be defamatory, it must be false. If LP never made the statement, then the claim is factually incorrect. The key is that the harmful aspect of the statement is presented as a fact, not an opinion.
3. **Lack of Context or Evidence**: The statement lacks context or evidence to support its serious accusations. It doesn't provide details about the supposed strategy or the resulting harm, which can lead to misinterpretation and unjust damage to LP's reputation.
4. **Public Communication**: For a statement to be defamatory, it needs to be communicated to others besides the person it's about. Posting this statement on a public forum or message board meets this criterion.
However, whether a statement is legally defamatory depends on jurisdictional laws, including how they interpret and enforce defamation, and the specific circumstances of the case. Typically, the person claiming defamation (LP in this case) must prove that the statement was made publicly, is false, and caused harm to their reputation.”

https://jamanetwork.com/journals/jamaoncology/fullarticle/2798847
https://www.prnewswire.com/news-releases/northwest-biotherapeutics-announces-that-a-marketing-authorization-application-has-been-submitted-to-the-uk-mhra-for-dcvax-l-for-glioblastoma-302021038.html
LC,
AI is great for screening false or baseless claims. The best part—you gave us the idea of using AI to detect manipulative bs! 😉
AI makes an awesome bs detector! Hopefully, the SEC is beginning to use AI more to identify illegal activity or to provide clues about such activities.
“Hedge funds and short sellers sometimes use a tactic known as "short and distort" to profit from the decline in a company's stock price. This strategy involves first short selling a stock, which means borrowing shares and selling them with the intention of buying them back later at a lower price. The "distort" part comes into play when these investors use various platforms, including message boards, to spread baseless rumors or misleading information about the company.
The goal of spreading these rumors is to create panic or uncertainty among other investors, leading to a drop in the stock price. As the stock price falls, the short seller can buy back the shares at a lower price, return them to the lender, and pocket the difference.
However, identifying such deceptive practices can be challenging. This is where Artificial Intelligence (AI) comes into play. AI algorithms can analyze vast amounts of data from social media, forums, and message boards to detect patterns of misinformation or manipulation. These algorithms can look for specific indicators of deceit, such as the repetition of certain unfounded claims, the use of sensationalist language, or coordinated posting patterns.
By flagging potential cases of "short and distort," AI helps in identifying bad-faith actors who intend to damage the reputation of a company and its leadership. This tool is valuable for regulatory bodies, companies, and individual investors, as it aids in maintaining the integrity of financial markets and protects against market manipulation.”

iclight,
Thank you for again reminding us about the brilliant compassionate use results. The compassionate use cases generated some astonishing data, and those data may be included in the regulatory filings!


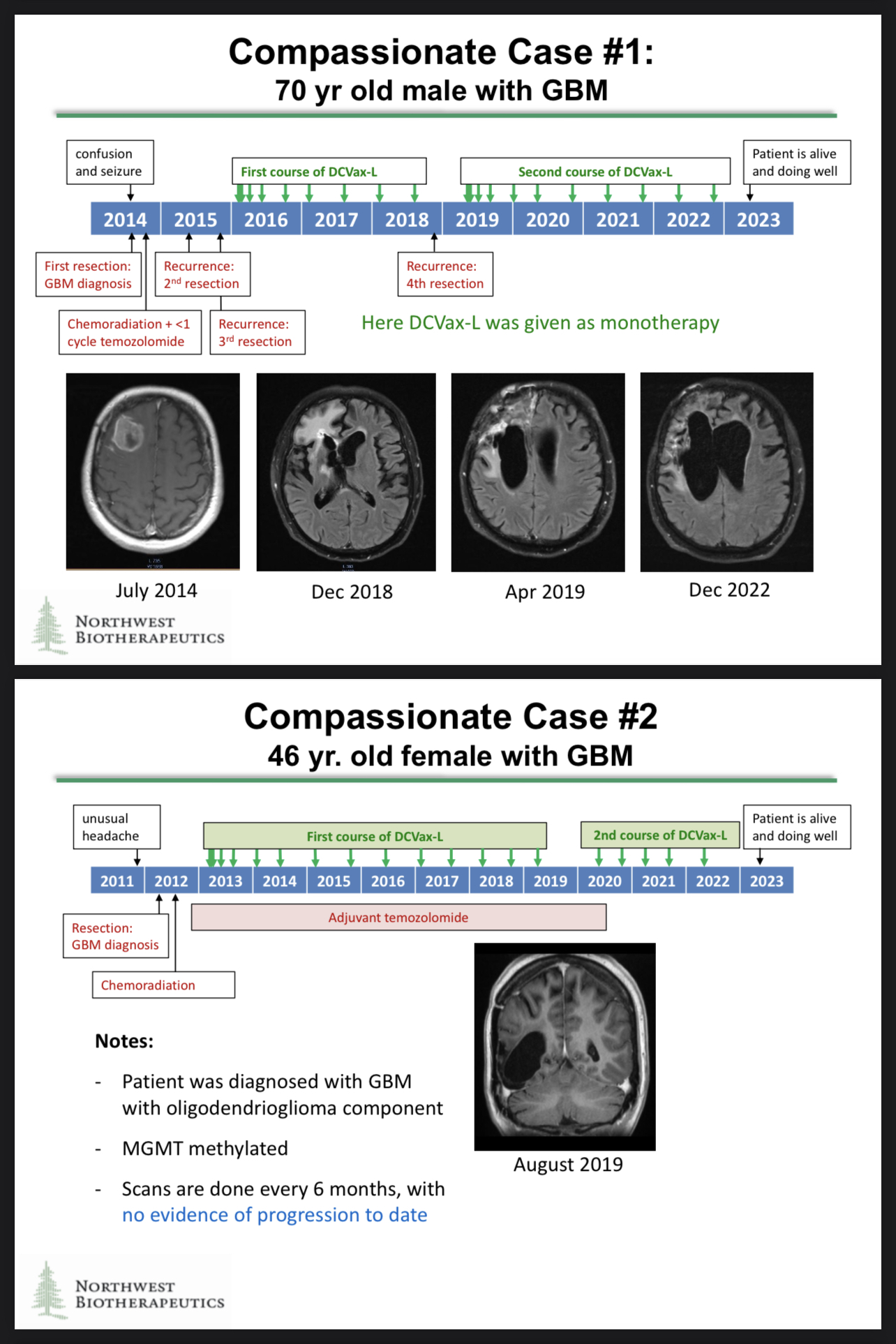
https://t.co/082urSTQ0s

https://www.braintumourresearch.org/media/news/news-item/2022/11/18/astonishing-results-for-brain-tumour-vaccine-trial
HyGro,
Thanks for reposting your baseless opinion again. I ran your statement through the detector 😶 …


“The statement makes several claims that are speculative and lack a robust foundation, which can contribute to the spread of unfounded rumors. Here's an analysis:
1. **Generalization about Big Pharma (BP) Investments**: Suggesting that BPs universally invest in biotechnology without scrutiny oversimplifies the complex decision-making process in these industries. Investment decisions are typically based on a thorough evaluation of scientific data, financial stability, market potential, and regulatory considerations.
2. **Assumptions about Company Viability and Trial Data**: The statement assumes that a specific company is insolvent and has questionable clinical trial data, without providing concrete evidence. It's important to avoid making such serious accusations without substantial proof. In fact, NWBO's DCVax-L Phase 3 trial was independently peer-reviewed and published in JAMA Oncology, which is a reputable source. This publication suggests that the trial underwent a rigorous scientific review process, countering the claim of "dubious" data.
3. **Misinterpretation of Clinical Trial Adjustments**: While the statement alleges that NWBO inappropriately altered its trial for DCVax-L, it's critical to understand that changes in clinical trials can occur for various valid reasons and are subject to strict regulatory scrutiny. The independent peer review and publication in a respected journal like JAMA Oncology lend credibility to the trial's methodology and results.
4. **Stereotyping BP Business Development Professionals**: Asserting that no BP business development professional would support a project deemed laughable by their clinical peers is speculative. Business decisions in biotech often involve weighing risks and potentials, and are not solely based on peer perception.
5. **Contribution to Rumor Spreading**: Statements like these, lacking in specific evidence and grounded in speculation, can contribute to the spread of rumors. In industries like biotechnology, where information is complex and nuanced, it's particularly important to rely on verified data and credible sources.
In summary, it is crucial to avoid generalizations and unfounded assumptions, especially when discussing complex sectors like biotech. The acknowledgment of NWBO's DCVax-L Phase 3 trial being independently peer-reviewed and published adds a crucial element of credibility in contrast to the baseless claims offered by “HyGro.”

https://jamanetwork.com/journals/jamaoncology/fullarticle/2798847
https://www.prnewswire.com/news-releases/northwest-biotherapeutics-announces-that-a-marketing-authorization-application-has-been-submitted-to-the-uk-mhra-for-dcvax-l-for-glioblastoma-302021038.html
iclight,
I think it’s back up—you should be able to sleep tonight with one less worry on your mind. 😴
https://nwbio.com/
I included another website that you might enjoy!
https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/
Reefrad,
That sounds like a request for the link. Thanks for asking! I cannot offer any insight into a forthcoming publication, but there is a Nature article available. I also included some related or otherwise currently relevant links.
https://www.nature.com/articles/s41420-023-01782-7
Combo is King!


https://www.jci.org/articles/view/169314




https://www.fda.gov/drugs/our-perspective/50-years-progress-treating-patients-cancer
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
https://www.raps.org/News-and-Articles/News-Articles/2023/9/FDA%E2%80%99s-Califf-Expect-to-see-more-RWE-based-regulato
https://www.raps.org/news-and-articles/news-articles/2023/8/fda-finalizes-guidance-on-real-world-evidence-in-d
iclight,
That’s absurd. You know the reason for the outage? Please enlighten us 🙄. Google any zillion-dollar company and you will find headlines about all sorts of outages for all sorts of reasons.
I also included a link to all the major headlines for NWBO so you don’t miss anything during the outage. 😉
https://9to5mac.com/2023/02/20/apple-support-website-invalid-url/
https://amp.cnn.com/cnn/2023/01/25/tech/microsoft-cloud-outage-worldwide-trnd/index.html
https://www.cnbc.com/amp/2023/12/14/chatgpt-back-online-after-major-outage-openai-says.html
https://amp.smh.com.au/national/major-optus-outage-affects-millions-of-customers-20231108-p5eics.html
https://www.securityweek.com/pharmaceutical-giant-eisai-takes-systems-offline-following-ransomware-attack/amp/
https://www.theverge.com/2023/2/24/23613737/dish-outage-internal-system-issue-boost-mobile-genesis
https://www.informationweek.com/it-infrastructure/google-outage-impacts-search-maps
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=173629105
LC,
That’s not true. I’m a soldier and I like clean and simple facts—the science and substance are much more important than the superficial styling of a website. I think that’s what you meant to say.
✅️ I want to see the independent peer review posted.
✅️ I want to see regulatory developments posted.
Maybe you’ll get your wish and see a new fancy website soon. 😉
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=173629105
I love you, Papito Curvito!
Thanks for more sage advice. I do respect the wisdom of my elders, especially when they’re also accomplished and wealthy. For example, the leadership of NWBO were well educated, experienced in international business, and they built vast personal wealth before they joined NWBO.
Also—Robert Hefner, whom you highlighted recently, is quite elderly and was wise enough to amass a personal fortune of over a billion dollars!
https://www.belfercenter.org/person/robert-hefner-iii

https://ghkco.com/wp-content/uploads/2022/06/2022-06-07_Statement-by-Robert-Hefner-on-NWBOs-DCVax-L-Phase-III-Trial.pdf