Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
I'm getting tired of...
...reiterating the posts. I wish we could just sticky them.
The propper way to say this...
...is 2.2 mil Buys at or above the Ask price and 1.4 mil Buys below the Ask price.
Oh, it's going to move up...
...flippers and daytraders bought in yesterday at the end of the day...around .0091
In the past, they've waited for an early morning spike...then sold off, caused panic, drove the price way down and bought in again at a lower price. They've been doing this for days.
No spike this morning, just nice healthy trading. If this pace continues, we're going to move up nicely. The flippers can sell responsibly and the buyers can buy with confidence.
Still, with everything in Genta's pipeline, anything in these levels is absolutely absurd and undervalued.
Keep an eye on GNTA...
...showed clear reversal with an inside bar yesterday and it's being confirmed in this mornings trading session.
The reversal continues...
...looking good early from yesterday's inside bar formation. Moving up.
http://stockcharts.com/h-sc/ui?s=gnta
Genta has met all of the requests from the FDA in order to receive approval of Genasense for the treatment of CLL, as specified by the FDA.
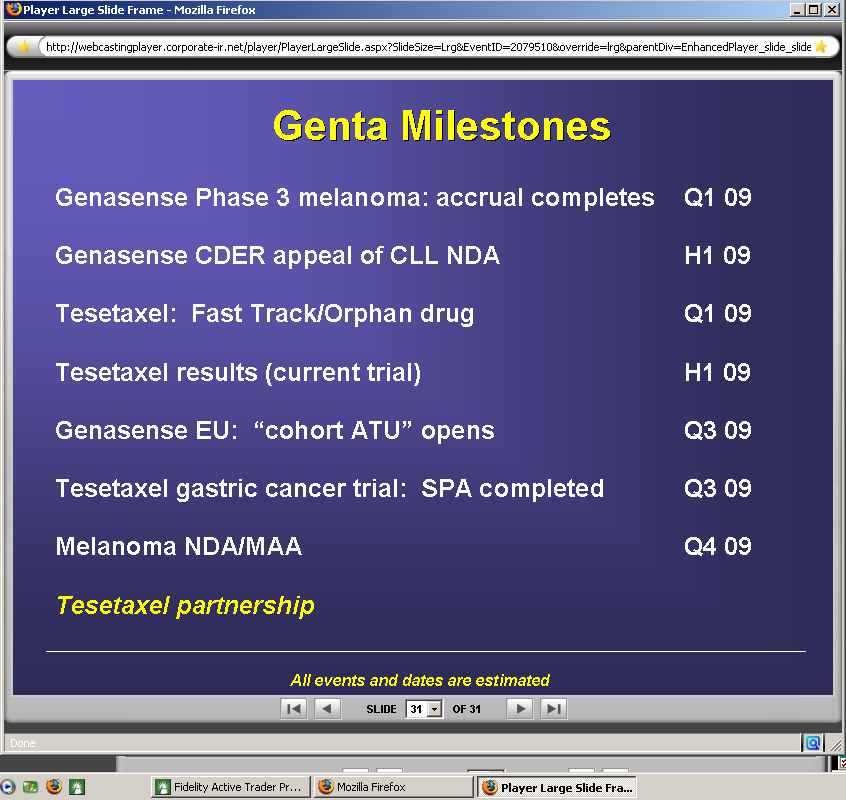
Please read slides 18 and 19 from the following link to get yourself up to speed on why the appeal was submitted by Genta leadership. Genta chose the second option from slide 18 and resubmitted the NDA. Start the media player on the left at 11 minutes *may have to copy/paste link*
http://webcastingplayer.corporate-ir.net/player/LrgPlayer.aspx?EventId=2079510&StreamId=1260249&TI%20K={8CEC9688-9089-484E-ABC1-DBB417AFD647}&RGS=1&override=lrg
Not only this, but the Agenda Trial is producing incredible confirmatory data of a prior phase III clinical study. It's the company's #1 focus and is going to support world-wide application of Genasense in the treatment of Melanoma.
That's incredible
ENOUGH REACHING ON THE WEBSITE.
Seriously, let's stick to the facts here.
Genasense is providing incredible confirmatory data of a prior phase III trial in its late phase III Agenda Trial.
Now THAT is something to be excited about. I mean, really effin' excited. The data is and will continue to exceed expectations with respect to confirming prior positive results and will ULTIMATELY lead to the approval and world-wide application of Genasense in the treatment of Melanoma.
I don't know and don't care about the website and why it's down. It likely has little to absolutely nothing to do with anything we'd be interested in. What does interest me is the data from the Agenda trial.
The menu navigation buttons are different.
Thanks, Bioman. It's the reason I'm excited about the future of the company, especially in 2009. It's not based on hype, or pumping, or any type of dreampt up prospects. It's deeply rooted in data and facts.
If Genasense gets approved for the treatment of CLL, that's just an added bonus for me. My eyes are on the prize, the Agenda Trial results and Tesetaxel partnerships.
Best of luck.
Nate...
...an independent group is doing the analytical data for both trials, as specified from the FDA.
The data speaks for itself, Nate. There's really no need for the CEO from Genta to say anything at all.
Are you suggesting that the independent group is fabricating the results and the Genta CEO has them in his back pocket? Surely you're not.
Look at the results, Nate.
I'm just posting the facts as they currently stand with this company, I know you're a fact man.
Fact is, Genasense has exceeded all expectations of the EMEA and FDA with respect to the treatment of Melanoma and the treatment of CLL.
If only one application is successful, this easily hits 50 cents. Easily.
I think you can clearly see that the Agenda trial is about to provide concrete, overwhelming, confirmatory data that both the FDA and EMEA requested in order to gain approval for world-wide application.
You need to start asking yourself how much longer you're going to sit on the sideline.
Nate, merger or not, here are the facts:
You want to talk facts and valuation, let's talk about it. Less than a penny? Are you seriously on drugs or are you trying to mislead people?
The CEO of Genta has clearly stated on several occasions that Genasense in Melanoma continues to be their #1 priority, the AGENDA Clinical Trial.
In 2007, the EMEA (European Medicines Agency ) asked Genta for an additional clinical study on the effectiveness of Genasense in Melanoma in order for approval.
"In its review, the EMEA rapporteurs indicated that positive
findings observed in the Phase 3 trial of Genasense should be confirmed in
an additional study."
http://www.prnewswire.com/cgi-bin/stories.pl?ACCT=104&STORY=/www/story/07-19-2007/0004629263&EDATE=
In the same link above it goes on to say:
"Genta has previously announced its intent to conduct this study, known as AGENDA, which is expected to initiate in the near future."
So Genta says, "That's fine...not only are we going to give you an additional clinical study, we're going to give you a world-wide (self explanatory), double-blind (the patient doesn't know what they're receiving and the administor of the drug doesn't know what they're giving), placebo-controlled(I'm assuming you know what a placebo is), completely randomized, parallel-group (A trial that compares two groups of people, one of which receives the intervention of interest and one of which is a control group)study...and not only that, we're going to let an outside group do our analytical data to remove any kind of bias with respect to how the results are calculated. Sound like a plan? Good."
As we all know, it's now 2009. The Agenda trial is well underway and is expecting to complete enrollment by the end of March, 2009. The company released preliminary data from the Agenda trial in January, 2009.
Here are the interim results from the study released in January, 2009.

That is a 188.88% increase in overall response compared to chemotherapy alone (DTIC). If you think that's not significant action, then there is something wrong with you. 188.88% increase in overall response, people. EMEA asked for confirmatory evidence, with a second trial, and this is MORE than confirmatory. It's the FDA's Golden Standard with respect to how clinical trials are conducted. Completely unbiased and now Genasense has been administered to well over 2,000 patients showing similar results.
I'd say this gives everyone something to be excited about, if they are a Genta shareholder. As spoken by the CEO of Genta, the Agenda trial is producing "overwhelming" results and will easily confirm the prior results submitted to the EMEA.
Not only this, but Genta has a pending FDA appeal and should hear on it within the next 30 days. Data the FDA was requesting on the long term survival was submitted in the appeal after Genta leadership met with the FDA on their requirements...the data is positive.
Genta has met all of the requests from the FDA in order to receive approval of Genasense for the treatment of CLL, as specified by the FDA.
Please read slides 18 and 19 from the following link to get yourself up to speed on why the appeal was submitted by Genta leadership:
http://webcastingplayer.corporate-ir.net/player/LrgPlayer.aspx?EventId=2079510&StreamId=1260249&TIK={8CEC9688-9089-484E-ABC1-DBB417AFD647}&RGS=1&override=lrg
Argue that, pal, and check your attitude at the door.
Nice GNTA reversal...
The CEO of Genta has clearly stated on several occasions that Genasense in Melanoma continues to be their #1 priority, the AGENDA Clinical Trial.
In 2007, the EMEA (European Medicines Agency ) asked Genta for an additional clinical study on the effectiveness of Genasense in Melanoma in order for approval.
"In its review, the EMEA rapporteurs indicated that positive
findings observed in the Phase 3 trial of Genasense should be confirmed in
an additional study."
http://www.prnewswire.com/cgi-bin/stories.pl?ACCT=104&STORY=/www/story/07-19-2007/0004629263&EDATE=
In the same link above it goes on to say:
"Genta has previously announced its intent to conduct this study, known as AGENDA, which is expected to initiate in the near future."
So Genta says, "That's fine...not only are we going to give you an additional clinical study, we're going to give you a world-wide (self explanatory), double-blind (the patient doesn't know what they're receiving and the administor of the drug doesn't know what they're giving), placebo-controlled(I'm assuming you know what a placebo is), completely randomized, parallel-group (A trial that compares two groups of people, one of which receives the intervention of interest and one of which is a control group)study...and not only that, we're going to let an outside group do our analytical data to remove any kind of bias with respect to how the results are calculated. Sound like a plan? Good."
As we all know, it's now 2009. The Agenda trial is well underway and is expecting to complete enrollment by the end of March, 2009. The company released preliminary data from the Agenda trial in January, 2009.
Here are the interim results from the study released in January, 2009.

That is a 188.88% increase in overall response compared to chemotherapy alone (DTIC). If you think that's not significant action, then there is something wrong with you. 188.88% increase in overall response, people. EMEA asked for confirmatory evidence, with a second trial, and this is MORE than confirmatory. It's the FDA's Golden Standard with respect to how clinical trials are conducted. Completely unbiased and now Genasense has been administered to well over 2,000 patients showing similar results.
I'd say this gives everyone something to be excited about, if they are a Genta shareholder. As spoken by the CEO of Genta, the Agenda trial is producing "overwhelming" results and will easily confirm the prior results submitted to the EMEA.
Not only this, but Genta has a pending FDA appeal and should hear on it within the next 30 days. Data the FDA was requesting on the long term survival was submitted in the appeal after Genta leadership met with the FDA on their requirements...the data is positive.
Genta has met all of the requests from the FDA in order to receive approval of Genasense for the treatment of CLL, as specified by the FDA.
Please read slides 18 and 19 from the following link to get yourself up to speed on why the appeal was submitted by Genta leadership:
http://webcastingplayer.corporate-ir.net/player/LrgPlayer.aspx?EventId=2079510&StreamId=1260249&TIK={8CEC9688-9089-484E-ABC1-DBB417AFD647}&RGS=1&override=lrg
It's good DD...
...I'm sure most people who are considering taking a position appreciate the roundups.
Not only that, they also have now met all of the requests from the FDA in order to receive approval of Genasense for the treatment of CLL, as specified by the FDA. The wording from the FDA was "not approvable in its current form" reads like it was an issue that could be addressed. Genta leadership met with FDA leadership to discuss the issue(s), then submitted the appeal.
The FDA has a 30 day response performance metric for submitted appeals. If it was a slam dunk NO, we would've certainly heard the response by now. The appeal was submitted in mid-December 2008. We're now at the end of February.
Please read slides 18 and 19 from the following link to get yourself up to speed on why the appeal was submitted by Genta leadership:
http://webcastingplayer.corporate-ir.net/player/LrgPlayer.aspx?EventId=2079510&StreamId=1260249&TIK={8CEC9688-9089-484E-ABC1-DBB417AFD647}&RGS=1&override=lrg
HERE IS THE BOTTOM LINE..READ
The CEO of Genta has clearly stated on several occasions that Genasense in Melanoma continues to be their #1 priority, the AGENDA Clinical Trial.
In 2007, the EMEA (European Medicines Agency ) asked Genta for an additional clinical study on the effectiveness of Genasense in Melanoma in order for approval.
"In its review, the EMEA rapporteurs indicated that positive
findings observed in the Phase 3 trial of Genasense should be confirmed in
an additional study."
http://www.prnewswire.com/cgi-bin/stories.pl?ACCT=104&STORY=/www/story/07-19-2007/0004629263&EDATE=
In the same link above it goes on to say:
"Genta has previously announced its intent to conduct this study, known as AGENDA, which is expected to initiate in the near future."
So Genta says, "That's fine...not only are we going to give you an additional clinical study, we're going to give you a world-wide (self explanatory), double-blind (the patient doesn't know what they're receiving and the administor of the drug doesn't know what they're giving), placebo-controlled(I'm assuming you know what a placebo is), completely randomized, parallel-group (A trial that compares two groups of people, one of which receives the intervention of interest and one of which is a control group)study...and not only that, we're going to let an outside group do our analytical data to remove any kind of bias with respect to how the results are calculated. Sound like a plan? Good."
As we all know, it's now 2009. The Agenda trial is well underway and is expecting to complete enrollment by the end of March, 2009. The company released preliminary data from the Agenda trial in January, 2009.
Here are the interim results from the study released in January, 2009.

That is a 188.88% increase in overall response compared to chemotherapy alone (DTIC). If you think that's not significant action, then there is something wrong with you. 188.88% increase in overall response, people. EMEA asked for confirmatory evidence, with a second trial, and this is MORE than confirmatory. It's the FDA's Golden Standard with respect to how clinical trials are conducted. Completely unbiased and now Genasense has been administered to well over 2,000 patients showing similar results.
I'd say this gives everyone something to be excited about, if they are a Genta shareholder. As spoken by the CEO of Genta, the Agenda trial is producing "overwhelming" results and will easily confirm the prior results submitted to the EMEA.
You people are selling out of your position for under a penny?! Are you out of your mind? It doesn't GET any better than this.
I'll bet you 100 dollars it does.
This is all you need to know:
The CEO of Genta has clearly stated on several occasions that Genasense in Melanoma continues to be their #1 priority, the AGENDA Clinical Trial.
To get everyone caught up, The Agenda trial is a confirmation of a very successful Phase 3 trial.
"In its initial Phase 3 trial, Genta performed the largest randomized controlled trial that has ever been conducted in patients with advanced melanoma. In that trial, 771 patients were randomly assigned to receive chemotherapy with DTIC alone or in combination with Genasense. A
scientific article that describes efficacy and safety results from this study can be accessed at:
http://jco.ascopubs.org/cgi/content/abstract/JCO.2006.06.0483v1
In that trial, patients were prospectively stratified according to levels of a blood enzyme known as LDH. Further analysis of this study showed that maximum benefit accrued to patients whose LDH biomarker did not exceed 80% of the upper limit of normal. Efficacy results in this group of 274 patients, which are the focus of the confirmatory AGENDA trial, are displayed in the table below. (The hazard ratios indicate that the addition of Genasense to DTIC was associated with a 42% reduction in the risk of disease progression and a 36% reduction in risk of death.)"
http://www.drugs.com/clinical_trials/genasense-data-melanoma-presented-opening-session-first-worldwide-melanoma-center-meeting-european-1869.html
In 2007, the EMEA (European Medicines Agency ) asked Genta for an additional clinical study on the effectiveness of Genasense in Melanoma in order for approval.
"In its review, the EMEA rapporteurs indicated that positive
findings observed in the Phase 3 trial of Genasense should be confirmed in
an additional study."
http://www.prnewswire.com/cgi-bin/st...4629263&EDATE=
In the same link above it goes on to say:
"Genta has previously announced its intent to conduct this study, known as AGENDA, which is expected to initiate in the near future."
So Genta says, "That's fine...not only are we going to give you an additional clinical study, we're going to give you a world-wide (self explanatory), double-blind (the patient doesn't know what they're receiving and the administor of the drug doesn't know what they're giving), placebo-controlled(I'm assuming you know what a placebo is), completely randomized, parallel-group (A trial that compares two groups of people, one of which receives the intervention of interest and one of which is a control group)study using a biomarker...and not only that, we're going to let an outside group do our analytical data to remove any kind of bias with respect to how the results are calculated. Sound like a plan? Good."
As we all know, it's now 2009. The Agenda trial is well underway and is expecting to complete enrollment by the end of March, 2009. The company released preliminary data from the Agenda trial in January, 2009.
Here are the interim results from the study released in January, 2009.

That is a 188.88% increase in overall response compared to chemotherapy alone (DTIC). If you think that's not significant action, then there is something wrong with you. 188.88% increase in overall response, people. EMEA asked for confirmatory evidence, with a second trial, and this is MORE than confirmatory. It's the FDA's Golden Standard with respect to how clinical trials are conducted. Completely unbiased and now Genasense has been administered to well over 2,000 patients showing similar results.
I'd say this gives everyone something to be excited about, if they are a Genta shareholder. As spoken by the CEO of Genta, the Agenda trial is producing "overwhelming" results and will easily confirm the prior results submitted to the EMEA.
You people are selling out of your position for under a penny?! Are you out of your mind? It doesn't GET any better than this.
Anything under 5 cents is a deal with everything on the horizon for Genta.
The CEO of Genta has clearly stated on several occasions that Genasense in Melanoma continues to be their #1 priority, the AGENDA Clinical Trial.
To get everyone caught up, The Agenda trial is a confirmation of a very successful Phase 3 trial.
"In its initial Phase 3 trial, Genta performed the largest randomized controlled trial that has ever been conducted in patients with advanced melanoma. In that trial, 771 patients were randomly assigned to receive chemotherapy with DTIC alone or in combination with Genasense. A
scientific article that describes efficacy and safety results from this study can be accessed at:
http://jco.ascopubs.org/cgi/content/abstract/JCO.2006.06.0483v1
In that trial, patients were prospectively stratified according to levels of a blood enzyme known as LDH. Further analysis of this study showed that maximum benefit accrued to patients whose LDH biomarker did not exceed 80% of the upper limit of normal. Efficacy results in this group of 274 patients, which are the focus of the confirmatory AGENDA trial, are displayed in the table below. (The hazard ratios indicate that the addition of Genasense to DTIC was associated with a 42% reduction in the risk of disease progression and a 36% reduction in risk of death.)"
http://www.drugs.com/clinical_trials/genasense-data-melanoma-presented-opening-session-first-worldwide-melanoma-center-meeting-european-1869.html
In 2007, the EMEA (European Medicines Agency ) asked Genta for an additional clinical study on the effectiveness of Genasense in Melanoma in order for approval.
"In its review, the EMEA rapporteurs indicated that positive
findings observed in the Phase 3 trial of Genasense should be confirmed in
an additional study."
http://www.prnewswire.com/cgi-bin/st...4629263&EDATE=
In the same link above it goes on to say:
"Genta has previously announced its intent to conduct this study, known as AGENDA, which is expected to initiate in the near future."
So Genta says, "That's fine...not only are we going to give you an additional clinical study, we're going to give you a world-wide (self explanatory), double-blind (the patient doesn't know what they're receiving and the administor of the drug doesn't know what they're giving), placebo-controlled(I'm assuming you know what a placebo is), completely randomized, parallel-group (A trial that compares two groups of people, one of which receives the intervention of interest and one of which is a control group)study using a biomarker...and not only that, we're going to let an outside group do our analytical data to remove any kind of bias with respect to how the results are calculated. Sound like a plan? Good."
As we all know, it's now 2009. The Agenda trial is well underway and is expecting to complete enrollment by the end of March, 2009. The company released preliminary data from the Agenda trial in January, 2009.
Here are the interim results from the study released in January, 2009.

That is a 188.88% increase in overall response compared to chemotherapy alone (DTIC). If you think that's not significant action, then there is something wrong with you. 188.88% increase in overall response, people. EMEA asked for confirmatory evidence, with a second trial, and this is MORE than confirmatory. It's the FDA's Golden Standard with respect to how clinical trials are conducted. Completely unbiased and now Genasense has been administered to well over 2,000 patients showing similar results.
I'd say this gives everyone something to be excited about, if they are a Genta shareholder. As spoken by the CEO of Genta, the Agenda trial is producing "overwhelming" results and will easily confirm the prior results submitted to the EMEA.
You people are selling out of your position for under a penny?! Are you out of your mind? It doesn't GET any better than this.
Genta has now met all of the requests from the FDA in order to receive approval of Genasense for the treatment of CLL, as specified by the FDA. The wording from the FDA was "not approvable in its current form" reads like it was an issue that could be addressed. Genta leadership met with FDA leadership to discuss the issue(s), then submitted the appeal.
The FDA has a 30 day response performance metric for submitted appeals. If it was a slam dunk NO, we would've certainly heard the response by now. The appeal was submitted in mid-December 2008. We're now at the end of February.
Please read slides 18 and 19 from the following link to get yourself up to speed on why the appeal was submitted by Genta leadership (may have to copy and paste):
http://webcastingplayer.corporate-ir.net/player/LrgPlayer.aspx?EventId=2079510&StreamId=1260249&TIK={8CEC9688-9089-484E-ABC1-DBB417AFD647}&RGS=1&override=lrg
That's not totally true...
...Genta is working with Loyolla University on the application of antisense drugs in Genta's pipeline. Loyola receives grants for that work, so Genta also benefits.
That's not here nor there.
The fact is, the president of the United States spoke specifically tonight in an address to the entire nation on seeking treatments, and ultimately, finding a cure for cancer. I challenge ANYONE to find a better treatment for Melanoma, the most deadly skin cancer accounting for 79% of skin cancer deaths, than Genta's Genasense application.
You think the FDA isn't under pressure to give drugs like Genasense, with proven efficacy and safety in the treatment of some of the most deadly forms of cancer approval?
Sir, I DVR'd it. Should I upload a video for you?
He most certainly did say what was written below about funding treatments for cancer, and ultimately, finding a cure in our time.
Those in the stems would've killed an endorsement like the one that was given for cancer research and treatment tonight by the president.
In the address to the nation, Obama said:
"Our recovery plan will launch a new effort to conquer a disease that has touched the life of nearly every american, including me, by seeking a cure for cancer in our time."
Get on board folks. The FDA is under some serious pressure this time around.
Melanoma is the least common but the most deadly skin cancer, accounting for 79% of skin cancer deaths.
Melanoma is currently the sixth most common cancer in American men and the seventh most common in American women. The median age at diagnosis is between 45 and 55, although 25% of cases occur in individuals before age 40. It is the second most common cancer in women between the ages of 20 and 35, and the leading cause of cancer death in women ages 25 to 30.
For everyone who is/was waiting for similar stem cell news...well...there it is live and direct from the presidents lips for cancer treatment and I challenge ANYONE to find a better treatment for Melanoma than Genta's Genasense application.
Barack Obama in tonights address to the nation:
"Our recovery plan will launch a new effort to conquer a disease that has touched the life of nearly every american, including me, by seeking a cure for cancer in our time."
Get on board folks. The FDA is under some serious pressure this time around.
I just want people to continue to see the forest through the trees. Some people get blinded by the ebbs and flows of the shareprice and lose sight of what the prize is.
We're going to do it either way, so get in or get out.
HERE IS THE BOTTOM LINE
The CEO of Genta has clearly stated on several occasions that Genasense in Melanoma continues to be their #1 priority, the AGENDA Clinical Trial.
This is my whole reason for being in this stock. If you can talk me out of why I bought in (using facts to back up your position), then I'll sell my entire position. No questions asked.
In 2007, the EMEA (European Medicines Agency ) asked Genta for an additional clinical study on the effectiveness of Genasense in Melanoma in order for approval.
"In its review, the EMEA rapporteurs indicated that positive
findings observed in the Phase 3 trial of Genasense should be confirmed in
an additional study."
http://www.prnewswire.com/cgi-bin/stories.pl?ACCT=104&STORY=/www/story/07-19-2007/0004629263&EDATE=
In the same link above it goes on to say:
"Genta has previously announced its intent to conduct this study, known as AGENDA, which is expected to initiate in the near future."
So Genta says, "That's fine...not only are we going to give you an additional clinical study, we're going to give you a world-wide (self explanatory), double-blind (the patient doesn't know what they're receiving and the administor of the drug doesn't know what they're giving), placebo-controlled(I'm assuming you know what a placebo is), completely randomized, parallel-group (A trial that compares two groups of people, one of which receives the intervention of interest and one of which is a control group)study...and not only that, we're going to let an outside group do our analytical data to remove any kind of bias with respect to how the results are calculated. Sound like a plan? Good."
As we all know, it's now 2009 and the Agenda trial is well underway and is expecting to complete enrollment by the end of March, 2009. The company released preliminary data from the Agenda trial in January, 2009.
Here are the interim results from the study released in January, 2009.

That is a 188.88% increase in overall response compared to chemotherapy alone (DTIC). If you think that's not significant action, then there is something wrong with you. 188.88% increase in overall response, people. EMEA asked for confirmatory evidence, with a second trial, and this is MORE than confirmatory. It's the FDA's Golden Standard with respect to how clinical trials are conducted. Completely unbiased and now Genasense has been administered to well over 2,000 patients showing similar results.
I'd say this gives everyone something to be excited about, if they are a Genta shareholder. As spoken by the CEO of Genta, the Agenda trial is producing "overwhelming" results and will easily confirm the prior results submitted to the EMEA.
You people are selling out of your position for under a penny?! Are you out of your mind? It doesn't GET any better than this people!
Oh yeah?
What was this new information that you stumbled upon that curbed your expectations?
Figures...
...I was wondering why you were poor mouthing this stock since a few days back. You better hope approval doesn't come while you sit on the sideline like a cheapskate.
Did anyone just see that????
TALK ABOUT blatant manipulation. Good lord!
That is the same with every single company that's openly traded on the markets. If you (not you specifically, but people in general) can't accept that known true, then they shouldn't be in the stock market.
I'm saying they met all of the requests from the FDA....
...in order to receive approval of Genasense for the treatment of CLL, as specified by the FDA. The wording from the FDA was "not approvable in its current form" reads like it was an issue that could be addressed. Genta leadership met with FDA leadership to discuss the issue(s), then submitted the appeal.
The FDA has a 30 day response performance metric for submitted appeals. If it was a slam dunk NO, we would've certainly heard the response by now. The appeal was submitted in mid-December 2008. We're now at the end of February.
A company with late stage anticancer drugs in their pipeline like Genta and everything on the horizon for 2009...this is a STEAL at one penny. Are you kidding me?
Chances looking good...
Genta Incorporated (Nasdaq: GNTA) announced that the Food and Drug Administration's (FDA) Center for Drug Evaluation and Research (CDER) has decided that available data are not adequate to support approval of Genasense® (oblimersen sodium) Injection for treatment of patients with relapsed or refractory chronic lymphocytic leukemia (CLL). In a decision issued in response to an appeal filed by Genta in October 2007, CDER acknowledged that complete response, which was the primary endpoint in the pivotal trial, was an appropriate endpoint for assessing efficacy. FDA also agreed that this endpoint was achieved, and that those results supported the efficacy of the drug. However, CDER concluded that at present there was insufficient "confirmatory evidence" in the New Drug Application (NDA) to approve the drug.
CDER recommended two alternatives for exploring the efficacy of Genasense that could provide such confirmatory evidence. One option is to conduct an additional clinical trial. The other option is to collect additional information regarding the clinical course and progression of disease in patients from the previous pivotal trial in order to ascertain whether those data contain sufficient confirmatory evidence. The Company currently plans to pursue both of these options.
__________________________________________________ _______________
Alright, didn't we just hear in the investors conference that they obtained new data on the progression of disease in patients from the previous clinical trial? I certainly think we did and it was very eye opening. It was very significant.
So it acheived it's primary endpoint AND has new data on the progression of disease in patients from the previous trial that were undeniable. This is EXACTLY what the FDA asked for, was it not? You can read just as well as I can. Now this from Genta's site:
Phase 3/Chronic Lymphocytic Leukemia (CLL)/NDA Status: Genta has appealed a prior “non-approvable” decision on the Genasense® New Drug Application (NDA) for patients with relapsed/refractory CLL. The appeal is now pending a decision by FDA’s Center for Drug Evaluation and Research (CDER). The pivotal randomized trial achieved its primary endpoint, which was to significantly increase the proportion of patients who achieved complete remission by adding Genasense® to standard chemotherapy compared with patients treated with chemotherapy alone. With 5-years of followup, all patients who achieved a major response (either complete or partial remission) on the Genasense® treatment arm achieved superior survival compared with responders treated with chemotherapy alone. A decision from CDER is expected in the current quarter.
http://www.genta.com/index.php?optio...23&SHID=&COID=
What was the real reason for non-approval on December 3, 2008? Genta clearly met both requests from the FDA. I think the wording from the FDA is important here. "Not approvable in its current form" very well could be as simple as a clerical error that was addressed by Genta and FDA leadership prior to the appeal being submitted.
Genasense in CLL may stand a better chance for approval this time around than I previously thought.
New clinical studies popping up for Genta drugs!
Sponsors and Collaborators:
Loyola University
Genta Incorporated
http://clinicaltrials.gov/ct2/show/N...=genta&rank=19
Yessir...if the FDA gets what they wanted out of the ammended NDA and appeal submitted in December we're going to see a major pop here. I'm not talking about a few hundred percent either.
Here is the real question....
Genta Incorporated (Nasdaq: GNTA) announced that the Food and Drug Administration's (FDA) Center for Drug Evaluation and Research (CDER) has decided that available data are not adequate to support approval of Genasense® (oblimersen sodium) Injection for treatment of patients with relapsed or refractory chronic lymphocytic leukemia (CLL). In a decision issued in response to an appeal filed by Genta in October 2007, CDER acknowledged that complete response, which was the primary endpoint in the pivotal trial, was an appropriate endpoint for assessing efficacy. FDA also agreed that this endpoint was achieved, and that those results supported the efficacy of the drug. However, CDER concluded that at present there was insufficient "confirmatory evidence" in the New Drug Application (NDA) to approve the drug.
CDER recommended two alternatives for exploring the efficacy of Genasense that could provide such confirmatory evidence. One option is to conduct an additional clinical trial. The other option is to collect additional information regarding the clinical course and progression of disease in patients from the previous pivotal trial in order to ascertain whether those data contain sufficient confirmatory evidence. The Company currently plans to pursue both of these options.
__________________________________________________ _______________
Alright, didn't we just hear in the investors conference that they obtained new data on the progression of disease in patients from the previous clinical trial? I certainly think we did and it was very eye opening. It was very significant.
So it acheived it's primary endpoint AND has new data on the progression of disease in patients from the previous trial that were undeniable. This is EXACTLY what the FDA asked for, was it not? You can read just as well as I can. Now this from Genta's site:
Phase 3/Chronic Lymphocytic Leukemia (CLL)/NDA Status: Genta has appealed a prior “non-approvable” decision on the Genasense® New Drug Application (NDA) for patients with relapsed/refractory CLL. The appeal is now pending a decision by FDA’s Center for Drug Evaluation and Research (CDER). The pivotal randomized trial achieved its primary endpoint, which was to significantly increase the proportion of patients who achieved complete remission by adding Genasense® to standard chemotherapy compared with patients treated with chemotherapy alone. With 5-years of followup, all patients who achieved a major response (either complete or partial remission) on the Genasense® treatment arm achieved superior survival compared with responders treated with chemotherapy alone. A decision from CDER is expected in the current quarter.
http://www.genta.com/index.php?optio...23&SHID=&COID=
What was the real reason for non-approval on December 3, 2008? Genta clearly met both requests from the FDA. I think the wording from the FDA is important here. "Not approvable in its current form" very well could be as simple as a clerical error that was addressed by Genta and FDA leadership prior to the appeal being submitted.
Genasense in CLL may stand a better chance for approval this time around than I previously thought.
You need to do more reasearch.
The approval of Genasense for world wide applications and Tesetaxel will easily push this stock pretty close to what you're talking about there.
You're talking BLOCKBUSTER anticancer drugs...worth billions and billions of dollars.
Saying that they have no means to get to that pps is simply an uninformed statement.
