Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
“Slated to begin production in 2021, the Caucharí-Olaroz project is approximately 50% complete and represents one of the few large-scale lithium operations currently in development globally.”
Musk secured a contract with PLL TO SECURE A FUTURE SUPPLY FOR 2023 AND BEYOND. THAT DEAL IS DONE. NOW HE’S GOING TO SECURE A DEAL WITH LAC FOR 2021 RIGHT INTO 2023. MUSK IS SUPER SMART. HE HAS A LOCK ON 2023 ALREADY AND PLL DOESN’T HAVE ANY LITHIUM YET.
YOU’LL SEE, I AM RIGHT. MUSK JUST BOUGHT THE FUTURE. WAIT UNTIL HE BUYS THE PRESENT. MUSK AND MANY OTHER COMPANIES NEED THE LITHIUM NOW. MUSK IS SHOPPING AROUND TO SECURE ALL THE LITHIUM HE CAN GET NOW. AND HE STARTED WITH A FUTURE CONTRACT THROUGH PLL FIRST. WHEN HE SECURES THE DEAL WITH LAC, THAT WILL BE FOR THE NEAR TERM.
Tesla and other companies cannot afford to ignore LAC. You will see. MUSK IS SMART AND ENTERED INTO AGREEMENT WITH PLL, KNOWING THAT THEY WILL PRODUCE LATER THAN LAC. THIS WAS A SUPER SMART MOVE THAT INDICATES TESLA IS LOOKING FOR LONG TERM CONTRACTS. PLL PRODUCTION BEGINS IN 2023. LAC PRODUCTION BEGINS IN 2021. THINK ABOUT THIS CAREFULLY.
WHY WOULD TESLA ENTER INTO AGREEMENT FOR 2023 WITH PLL AND NOT ENTER A CONTRACT WITH LAC THAT WILL PRODUCE SOONER. ELON MUSK IS MAKING A VERY POWERFUL MOVE THAT HAS TELL TALE SIGNS OF A MASSIVE LAC CONTRACT. MUSK NEEDS ALL THE LITHIUM HE CAN GET. MUSK HAS LAC IN HIS SIGHTS AND SO DO OTHERS.
YOU WILL SEE HOW THIS PLAYS OUT. NIO IS ALSO GOING TO GET INVOLVED. LAC HAS A MASSIVE SUPPLY THAT CANNOT BE IGNORED. $35.00. SET A GTC-EXT AND RIDE IT OUT.
LAC HAS A MASSIVE SUPPLY AND RIGHTS TO THE LITHIUM IN THEIR PRE FEASIBILITY AND DEFINITIVE FEASIBILITY STUDIES. PEOPLE WANT TO BUY THE SHARES AND THEY ARE MESSING WITH THE PRICES. LAC IS WORTH MUCH MORE. LAC IS NUMBER 3 IN THE WORLD.
“Argentina Lithium reserves: 1,700,000 MT
Argentina is the fourth largest lithium producer in the world, and last year it put out 6,400 MT of the metal, the same amount as in 2018. It also ranks third in lithium reserves in the world, at 1,700,000 MT. Putting that into perspective, the Financial Post notes that the country has enough lithium reserves to feed global demand for roughly 300 years.
It’s worth noting that Chile, Argentina and Bolivia comprise the “Lithium Triangle,” which hosts more than half of the world’s lithium reserves. However, while there is a great deal of lithium in the ground in Argentina, lithium extraction is another story. Only time will tell if the country will be able to harvest it economically as the lithium industry continues to heat up.”
https://investingnews.com/daily/resource-investing/battery-metals-investing/lithium-investing/lithium-reserves-country/
“There’s only one thing standing between Tesla and world domination, the global supply of lithium. And that’s good news for lithium producers. Right now, the world mines roughly 400,000 tons of lithium a year, enough to power 2 million to 3 million electric vehicles, though only a third of that goes to EVs right now. That number will have to increase perhaps as much as tenfold to meet Musk’s goal, and that doesn’t take into account other automakers.
In addition, global governments are providing a big catalyst for lithium.”
https://www.nasdaq.com/articles/3-lithium-stocks-with-deal-making-potential-2020-10-07?amp
“Success during present low prices would align to producer size, Bacanora’s Secker said, stressing that large investments were still flowing into the lithium supply chain from established majors. He also argued that companies with partnerships or joint ventures would be more resilient.”
https://www.spglobal.com/en/research-insights/articles/lithium-supply-is-set-to-triple-by-2025-will-it-be-enough
“South America is also set to see supply growth of around 199%, new brine lakes are beginning production and most existing salars are expected to increase output. With longer lead times, the oncoming brine projects were probably in the same batch of funding as the hard rock mines that have come online in Australia since 2016.
With the Authier, Rose and Whabouchi mines set to come online before 2025, North America’s share of lithium supply is set to increase to over 5%, potentially allaying some fears around North America’s present minor role in the EV battery chain.
Europe, however, is expected to add only one new lithium source by 2025, marginally increasing its share of global supply. With potential for 25 battery gigafactories to be operational across the continent by 2025, Europe looks set to remain dependent on non-domestic raw material supply – even as the European Commission has thrown its weight behind developing the sector and ensuring security of raw material supply
Keeping up with EVs
Despite the present oversupply, and near threefold supply growth expected by 2025, there is a strong argument that further out, as momentum builds, demand could outweigh supply.
“If forecasts for EV penetration are to be believed – along with the billions of dollars car companies have sunk or will sink into EV development and production – then lithium demand is set to increase 10-fold over the next decade.” Bridle said.
https://www.spglobal.com/en/research-insights/articles/lithium-supply-is-set-to-triple-by-2025-will-it-be-enough
“Based on current plans, it is expected that lithium supply will keep up with demand for the next five years. There is a growing concern for after that, that there will be an undersupply to the battery market.”
https://www.mining-technology.com/features/are-mining-companies-jumping-the-gun-on-lithium/
LAC is the next best thing to Coca Cola and the Emancipation Proclamation. LAC will produce more Lithium than the roast pork fried rice in all the Chinese food restaurants in China Town, New York City.
Get ready for lift off: https://www.lithiumamericas.com/_resources/presentations/LAC-Cauchari-DFS-and-Thacker-Pass.pdf. There’s more Lithium in Cuchi-Cuchi than New York City and Alaska combined. We are good.
Get ready for the dump.
Easy short here.
Scam company without an ounce of Lithium.
This pos will never make it past 15.
We will short this one and keep profiting from this no Lithium scam company.
LITHIUM ION BATTERIES ARE HAZARDOUS: http://www.idealind.mx/pdf/msds/lithiumionbattery_msds.pdf
I HEARD THE DEPARTMENT OF HEALTH AND ENVIRONMENTAL PROTECTION ARE INVESTIGATING LAC.
Tesla wouldn’t touch this company with a 10 foot pole.
Going out of business equipment liquidation.
LAC DUMPS HAZARDOUS WASTE PRODUCTS INTO THE ENVIRONMENT.
LAC USES ENVIRONMENTALLY HAZARDOUS ACID LEACHING PROCESSES.
MASSIVE DUMP INBOUND; IMMINENT.
LAC ONLY HAS LIABILITIES AND EXPENSES.
LAC IS NOT PRODUCING ANY LITHIUM.
We are shorting this to the bottom.
LAC doesn’t have any sales, earnings or revenue from any Lithium transactions.
This company is going down right now and failing to make any presence in the Lithium market. They don’t have any sales or earnings or revenue from any Lithium transaction. Go check if you don’t believe me.
Yale researchers will begin a clinical trial at Yale New Haven Hospital to test the effectiveness of a drug called ibudilast (MN-166) for treating acute respiratory distress syndrome (ARDS), a life-threatening lung condition developed by some of the most seriously ill COVID-19 patients. The researchers are part of Yale’s Advanced Therapies Group, which was formed in response to the pandemic and is identifying and advancing possible treatments for the disease.
The drug — which has been approved for years in Japan and Korea for the treatment of asthma — has shown promising results for reducing inflammation associated with ARDS in mouse models. The researchers have partnered with the California company MediciNova to launch the trial. MediciNova had been developing ibudilast as a treatment for multiple sclerosis and neuroinflammation before shifting its attention to COVID-19.
The trials will be controlled studies that test the safety and efficacy of ibudilast for use in patients with COVID-19.
The researchers anticipate FDA approval soon “to treat patients with COVID-19 pneumonia before they develop severe respiratory distress and require mechanical ventilation,” said Dr. Geoffrey Chupp, professor of medicine (pulmonary), director of the Yale Center for Asthma and Airways Disease, and principal investigator of the clinical trial.
The most severely ill COVID-19 patients develop ARDS, which causes hyper-inflammation and fluid buildup in the lungs and leads to a sharp drop in oxygen levels. These patients need ventilators to support their breathing, and their prognosis for survival has been poor. The researchers hope that the drug will reduce damaging inflammation and mitigate the progression of COVID-19-related ARDS, diminish lung injury, and allow patients to recover.
“COVID-19 causes an intense inflammation reaction known as a ‘cytokine storm,’” said Chupp. In patients, this presents as COVID-19-related pneumonia or ARDS.
“If you can modulate that,” he said, “you can dial back the severity of the illness.”
“We have designed a placebo-controlled trial to see if blocking the effects of macrophage inhibitory factor, or MIF, can protect patients with COVID-19 from developing ARDS,” said Dr. Maor Sauler, assistant professor of medicine (pulmonary) and co-investigator in the study, who researches the effects of MIF on inflammatory diseases in the lung.
Ibudilast is a type of drug known as a MIF inhibitor. MIF is a gene that regulates the immune response — a driver of inflammation and the so-called “cytokine storm.” Over-expression of the gene has been found to play a key role in a number of diseases, including ARDS, asthma, rheumatoid arthritis, lupus, and multiple sclerosis.
Dr. Richard Bucala, chief of rheumatology, allergy, and immunology at Yale School of Medicine and rheumatologist-in-chief at Yale New Haven Health, is a pioneer in the discovery of drugs that interfere with the MIF pathway to target disease. He was the first to clone MIF and its receptor, and he has developed small-molecule MIF inhibitors to treat autoimmune diseases and cancer. MN-166’s inhibition of MIF activity was discovered by Elias Lolis, professor of pharmacology. Bucala, who co-directs the Advanced Therapies Group with Dr. Naftali Kaminski, led the partnership with MediciNova. Kaminski is the Boehringer Ingelheim Pharmaceuticals, Inc. professor of medicine (pulmonary).
“We are excited for this partnership opportunity to initiate this study, given the high morbidity and mortality of COVID-19, and the present lack of any evidence-based therapies,” said Bucala. “We additionally hope that insights from this trial will help us to better understand how ARDS develops, irrespective of its underlying cause.”
COVID-19 patients who are on ventilators for ARDS currently have few treatment options.
“Once a patient is in respiratory failure on a ventilator, there is no specific treatment for reducing lung injury and minimizing ARDS,” Chupp said. “All approaches are just to support the patient until the lung heals.”
The effort is one of many by Yale’s Advanced Therapies Group to find new treatments, diagnostics, and targets for combatting COVID-19. Recently, the group helped to lay the groundwork for clinical trials of the novel drug sobetirome to treat patients with ARDS, also at Yale New Haven Hospital.
https://news.yale.edu/2020/04/23/yale-launches-clinical-trial-drug-treat-severe-covid-19-patients
Earnings looking great:
“MNOV reported its earnings at -$0.06 per share in the fiscal quarter closing of 3/30/2020. The Analysts for Wall Street were expecting to report its earnings at -$0.08/share signifying the difference of 0.02 and 25.00% surprise value. Comparing the previous quarter ending of 12/30/2019, the stated earnings were -$0.05 calling estimates for -$0.08/share with the difference of 0.03 depicting the surprise of 37.50%.”
About MN-001 Positive Phase 2 Results
“MN-001 (tipelukast) is a novel, orally bioavailable small molecule compound which exerts its effects through several mechanisms to produce its anti-fibrotic and anti-inflammatory activity in preclinical models, including leukotriene (LT) receptor antagonism, inhibition of phosphodiesterases (PDE) (mainly 3 and 4), and inhibition of 5-lipoxygenase (5-LO). The 5-LO/LT pathway has been postulated as a pathogenic factor in fibrosis development and MN-001’s inhibitory effect on 5-LO and the 5-LO/LT pathway is considered to be a novel approach to treat fibrosis. MN-001 has been shown to down-regulate expression of genes that promote fibrosis including LOXL2, Collagen Type 1 and TIMP-1. MN-001 has also been shown to down-regulate expression of genes that promote inflammation including CCR2 and MCP-1. In addition, histopathological data shows that MN-001 reduces fibrosis in multiple animal models.
Previously, MediciNova evaluated MN-001 for its potential clinical efficacy in asthma and had positive Phase 2 results. MN-001 has been exposed to more than 600 subjects and considered generally safe and well-tolerated.”
https://medicinova.com/clinical-development/core/mn-001-nash/
Development Plans
Recent preclinical results provide compelling evidence that MN-001 warrants further evaluation for the treatment of NASH in humans. MediciNova has an open IND at FDA. Due to safety data from previous clinical studies of MN-001, FDA has approved the protocol for a Phase 2 study as the first clinical trial of MN-001 in NASH.
https://medicinova.com/clinical-development/core/mn-001-nash/liver-fibrosis/
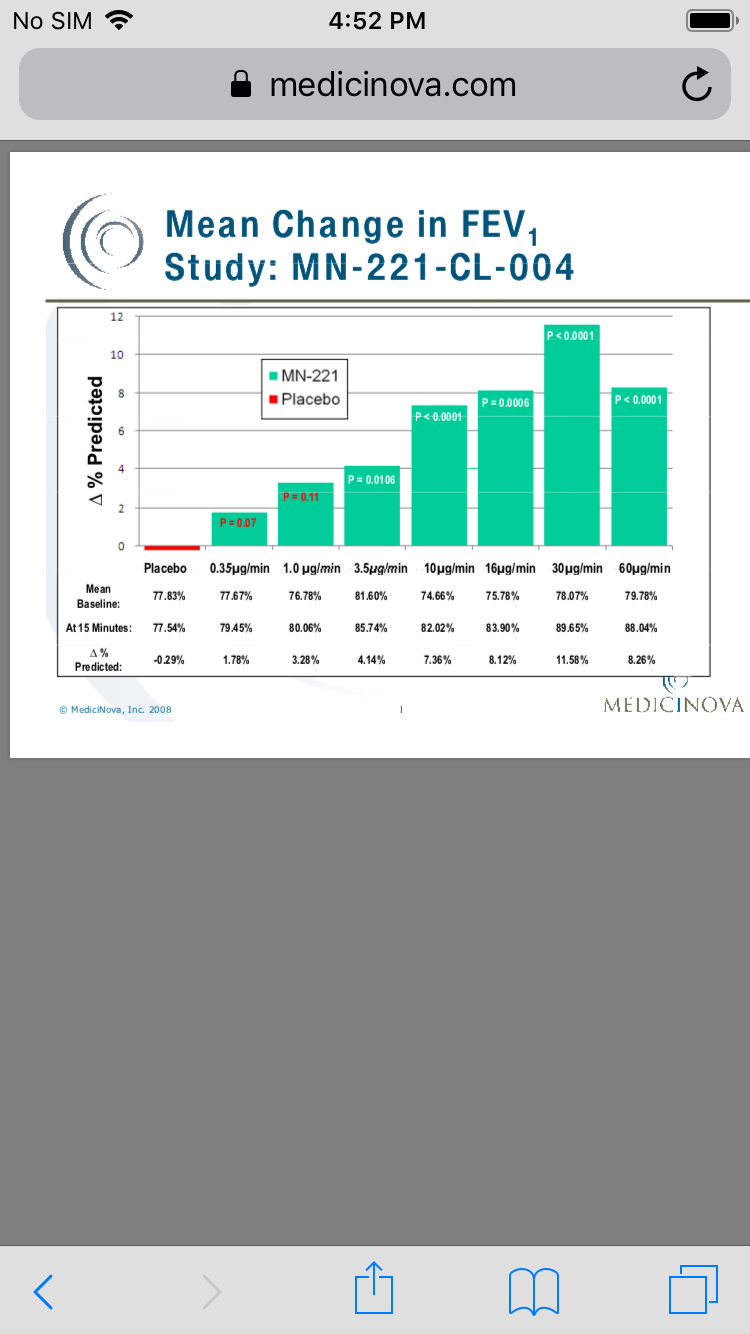
Phase II Study – MN-221-CL-004
This clinical trial achieved statistical significance in its primary endpoint of mean change in forced expiratory volume in one second, or FEV1, from baseline to measurement at 15 minutes (the end of the infusion) at doses of 10, 16, 30 and 60 micrograms per minute of MN-221 (p-value less than or equal to 0.0006) compared to placebo.
There were no clinically significant cardiovascular, electrocardiogram, or ECG, or vital sign changes observed at any dose tested. In addition, no serious adverse effects were observed in this clinical trial.
“Ibudilast is a relatively non-selective phosphodiesterase inhibitor which has been marketed for almost 20 years in Japan for treating asthma. More recently it has been found to have anti-inflammatory activity in both the peripheral immune system and in the central nervous system via glial cell attenuation. This CNS-directed anti-inflammatory activity is of potential use in the treatment of multiple sclerosis, neuropathic pain, and in the improved efficacy and safety of opioids by decreasing opioid tolerance, withdrawal and reinforcement. Its suitable pharmacokinetics and generally good tolerability make it a promising potential treatment for these conditions.”
https://rsds.org/wp-content/uploads/2015/02/Rolan_Ibudilast_Review.pdf
FDA Gives OK to Medicinova’s Phase 3 Clinical Trial Plan for Ibudilast
https://alsnewstoday.com/news-posts/2018/09/27/fda-approves-phase-3-plan-ibudilast-potential-als-treatment/
FDA Approves Pivotal Phase 2b/3 Trial for Ibudilast in ALS Patient
https://alsnewstoday.com/news-posts/2019/04/17/fda-approves-pivotal-phase-2b-3-trial-ibudilast-als-patients/
