Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
VAPE is a California based Marijuana Co. Low OS/low AS....market cap under $5 million
20265 Ventura Blvd a
Woodland Hills, CA 91364
USA
Marijuana News Roundup: California’s Cannabis Crop Worth $23 Billion https://www.yahoo.com/news/marijuana-news-roundup-california-cannabis-130038582.html
$MNKD MannKind Successfully Completes Phase 1 Trial of Treprostinil Technosphere for Pulmonary Arterial Hypertension Advancing Development to Next Phase MNKD
https://www.nasdaq.com/press-release/mannkind-successfully-completes-phase-1-trial-of-treprostinil-technosphere-for-pulmonary-arterial-20180607-00582
$LVGI HEMPCORE HEALTH LVGI
?* RELAX HempCore Healths RELAX anxiety relief formula helps reduce stress and anxiety as well as promotes relaxation. RELAX bolsters a calm, focus, relaxed and positive mood. Relax during the day, sleep better at night. HempCore Healths RELAX contains Hemp, L-Theanine and B-vitamin that support relaxation and feelings of happiness. Hemp has long been used in Chinese medicine, to impart a calming effect by helping to reduce anxiety, it also promotes a more relaxed transition to evening activities. HempCore Healths RELAX relief is 100% drug free, formula with key nutrients to help reduce stress and anxiety. Together with the proper rest and nutrition, HempCore Healths RELAX promotes relaxation to help combat the effects of daily stress.
?* FIBERED UP Hemp Core Healths FIBERED UP is made with organic flax, oats, psyllium, flaxseed, oat bran, acacia and hemp. Hemp Core Fibered UP! helps remove toxins. Our organic fiber blend supports healthy digestion and detoxification. Getting plenty of fiber in the diet is essential for healthy bowel function. The blend of three organic fibers also helps absorb toxins in the digestive tract and eliminate them from the body. Get more of the natural fiber you need every day. Daily intake of fiber is an important part of your overall good health.
?* HEALTHY HEART Hemp Core Health’s Healthy Heart is a all natural product that promotes cardiovascular health and helps maintain healthy cholesterol levels . Gluten free plus heart healthy Ingredients: CoQ10 - Known as the hearts antioxidant, extensive clinical research supports CoQ10 as an essential tool for supporting healthy cardiovascular function. Hemp contains healthy plant chemicals, such as beta-sitosterol and campesterol, which have been linked to lower heart attack risk, and reduced LDL cholesterol (the bad kind). Sterols also lower inflammation and slow the progression of atherosclerosis. with proper diet and exercise will help you to achieve success in your program to maintain your cholesterol health.
?* BRAIN BOOST HempCore Healths BRAIN BOOST is the ultra neurotransmitter boosting formula that assists your central nervous system in delivering essential oxygen and nutrients to the brain. Abundant clinical research has shown Hemp which is high in Omega 6 and Omega 3 to be important in helping to optimize cognitive function, particularly in the aging brain. Omega 6 and Omega 3 are essential components of neuronal cell membranes and are shown to optimize cognitive and adrenal function and help optimize brain function. HempCore Healths BRAIN BOOST helps regulate cortisol levels during stress enhancing memory and focus. HempCore Healths BRAIN BOOST improves cell-to-cell communication in the brain.
?* JOINT RELEIF Hemp Core Anti inflammation joint Formula combines nutrients oils and botanical with the power of hemp to promote joint comfort. This powerful formula is designed to support healthy joints in three distinct ways:1) Rebuild The well-known ingredients of Glucosamine and Chondroitin are included in this formula to help rebuild cartilage. These nutrients promote healthy tissues and help to maintain joint structure and mobility. 2) Lubricate Glucosamine and Chondroitin enhance the lubricating synovial fluid that cushions and supports joints. These nutrients support joint comfort.3) Soothe - This formula is enhanced by the inclusion of Omega-3 fatty acids from Hemp oil. Hemp anti-inflammatory properties come from gamma-linolenic acid or GLA. GLA is a beneficial type of Omega-6 fat that builds anti-inflammatory hormones. Although GLA is not commonly found in foods, hemp are an abundant source. Hemp core anti inflammation can help to rebuild lubricate and soothe your aching joints. This advanced combination of nutrients can give you the support you need to promote long-term joint and connective tissue.
$WPWR Well Power, Inc.’s (WPWR) Licensed Technologies Aligned with Global Push to End Gas Flaring WPWR
Houston-based Well Power is targeting the oil and gas industry with a particular focus on commercializing a patented solution to convert flared, shut-in and stranded gas into clean power and engineered fuels. Using this mobile, high-yield technology licensed from ME Resources Corp. (“MEC”), Well Power aims to reduce waste emission and capitalize on this niche market of the oil and gas industry.
Each year, billions of cubic meters of natural gas are flared at oil production sites all over the world. According to the World Bank Group, gas flaring not only wastes a valuable energy resource, it also contributes to climate change by releasing more than 300 million tons of CO2 into the atmosphere.
In April, The World Bank announced that chief executives from major oil companies, including Royal Dutch Shell (NYSE:RDS-A, NYSE:RDS-B) and Statoil CEO (NYSE:STO), were convening with senior government officials from several oil-producing countries to commit to ending routine gas flaring. Already endorsed by nine countries, 10 oil companies and a handful of development institutions, United Nations Secretary-General Ban Ki-moon and World Bank Group President Jim Yong Kim concurrently launched the “Zero Routine Flaring by 2030” initiative to end routine gas flaring at oil production sites by the year 2030.
“Gas flaring is a visual reminder that we are wastefully sending CO2 into the atmosphere,” Jim Yong Kim stated in an earlier news release. “We can do something about this. Together we can take concrete action to end flaring and to use this valuable natural resource to light the darkness for those without electricity.”
Well Power is gearing up to participate in this broader movement through the commercialization of the licensed Micro-Refinery Unit (MRU), which processes raw natural gas into green fuel (such as diluents, drop-in diesel and pipeline quality synthetic crude) and clean power. MEC’s solution simultaneously reduces CO2 emissions while creating revenue streams with minimal capital expenditure.
Well Power has secured the licensing rights to Texas with the first right of refusal on the other U.S. states, such as North Dakota, where gas flaring is a significant and concern. The company plans to be able to provide its technology with full-service engineering, design, construction, modular fabrication, maintenance and construction management services to clients in the upstream areas of exploration and production.
In addition, Well Power will offer consulting services, process assessments, facility appraisals, feasibility studies, technology evaluations, project finance structuring and support, and multi-client subscription services.
Leveraging the potential of the MRUs, Well Power is aligned with the Zero Routine Flaring by 2030 initiative and offers the opportunity to create value from a wasted resource while enabling wider access to energy, improved environmental conditions, and economic development for local populations.
$PMCB PharmaCyte FDA Orphan Drug Designation PMCB
http://www.accessdata.fda.gov/scripts/opdlisting/oopd/detailedIndex.cfm?cfgridkey=457014
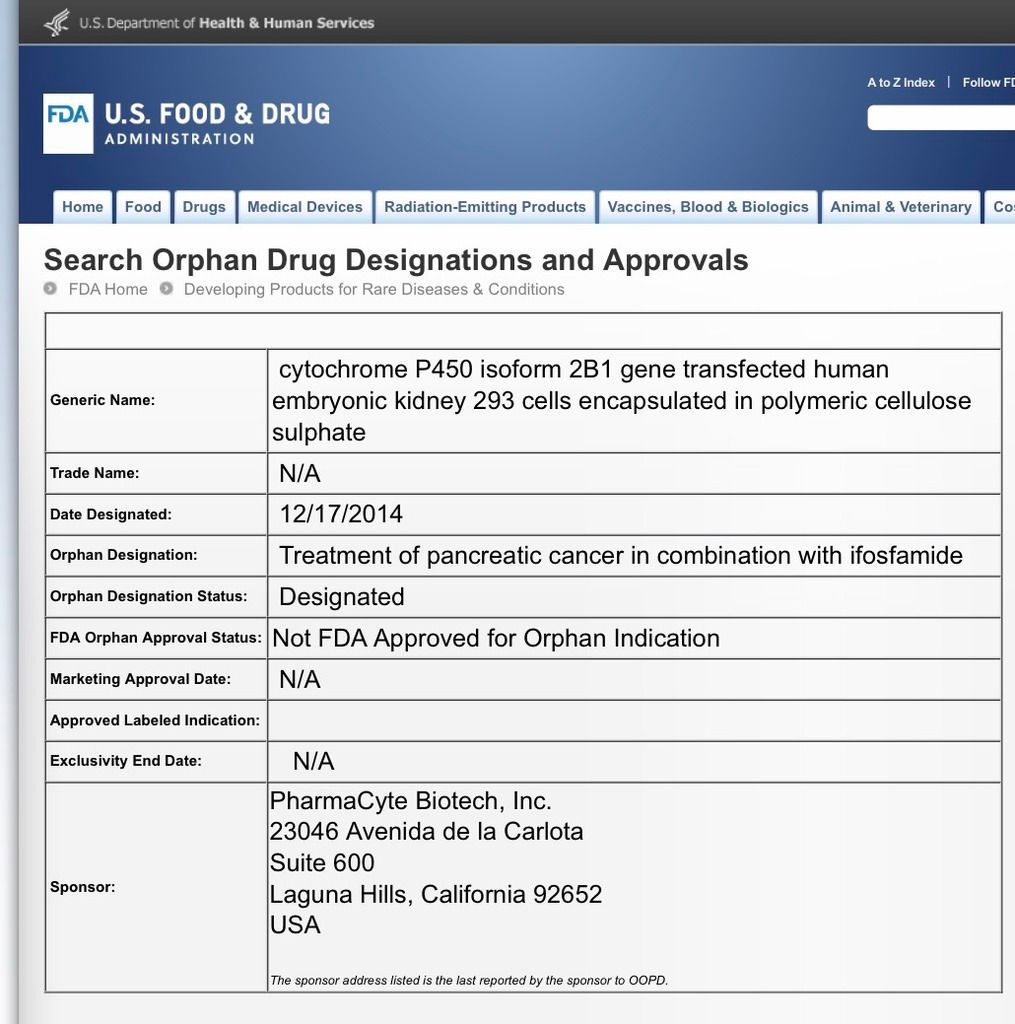
Go $PMCB
Silicon Valley biotech trading at .0065 and heading up, ARYC, updates:
Arrayit Silicon Valley store sales exceed $54 million all-time on 222,269 microarray products and services
http://
shop.arrayit.com
Arrayit receives new financing lead that could generate 6,000 x $100K = $600MM in Arrayit microarray platform sales
http://
arrayit.com
Arrayit signs 3-year $24.4 million patented microarray technology sales and marketing agreement with a leading laboratory services provider.
Arrayit Corporation ?@arrayit
Arrayit quotes $16,000,000 microarray services contract to top US-based research laboratory http://arrayit.com/Services/services.html …
Arrayit backorders soar to record $2.5MM on strong demand for microarray products & services
http://
arrayit.com/index.html
Arrayit Corporation
http://www.arrayit.com/
Check out their Facebook page: Arrayit Corporation
Follow $ARYC on Twitter @Arrayit
Avant Diagnostics, Inc. provide the market’s first large panel biomarker monitoring test for ovarian cancer (OVC). OvaDx® is advanced microarray-based test that measures the activation of the immune system in blood samples in response to early stage ovarian tumor cell development. Research studies with OvaDx® indicate high sensitivity and specificity for all types and stages of ovarian cancer including stage IA-IV borderline serous, clear cell, endometrioid, mixed epithelial, mucinous, serous, and ovarian adenocarcinoma. Upon FDA approval, Avant will offer OvaDx® as an elective test for women seeking greater wellness and for women in the elevated risk category for ovarian cancer. OvaDx® will be used by doctors to advance the forefront of ovarian cancer treatment including improved surgical options, more effective chemotherapies, and to supplement existing tests including CA-125, OVA1®, and transvaginal ultrasound.
This board brings back some good memories!
This is going to be a historical rally at ARYC that may never be rivaled. We've all seen similar but this could dominate those in its capacity. Currently trading at 1/4th of a penny but analysts are giving it a book value of $2.74/share. 500, 000 shares currently costs approximately $1,500. 500,000 at $2.74 is approximately $1.3 million. We've all seen it happen before and it can very easily happen with this extremely undervalued microarray company
http://www.arrayit.com/
Facebook page: Arrayit Corporation
Follow $ARYC on Twitter @Arrayit
Two contracts signed with major Silicon Valley companies totaling between $48 million and $51 million dollars with details to be released soon!!
Please begin by following the company on Twitter for many daily updates. These are free PR's for the company.
Then follow them on Facebook.
If you live in the Bay Area you can visit the company at:
Arrayit Corporation
927 Thompson Place
Sunnyvale, CA 94085
USA
Phone 408-744-1331
FAX 408-744-1711
Email arrayit@arrayit.com
Web www.arrayit.com
Located in the heart of Silicon Valley near Apple, Yahoo, AMD, Juniper, MBZ, LinkedIn, Facebook and many, many other leaders of the tech industry.
Google their Headquarters:
927 Thompson Place
Sunnyvale, CA 94085
A few recent pieces of news that were issued and are causing this group of new followers:
Arrayit signs $27.1 million term sheet for microarray services with US-based research leader
http://
arrayit.com/Services/servi
ces.html
…
Arrayit signs 3-year $24.4 million patented microarray technology sales and marketing agreement with a leading laboratory services provider.
Arrayit receives new financing lead that could generate 6,000 x $100K = $600MM in Arrayit microarray platform sales
http://
arrayit.com
Arrayit Corporation ?@arrayit
Arrayit quotes $16,000,000 microarray services contract to top US-based research laboratory http://arrayit.com/Services/services.html …
Two microarray contracts for a total of $51 million over 3 years signed in the last 2 months
Earnings of $2.3 million last year and $7 million so far this year per company updates.
Growlife,(PHOT),Cannabis/Marijuana. Focused on indoor Marijuana grow equipment. Greners.com
The indoor growing market for cannabis plants is here to stay. Opportunity for it to double for several years, especially after this year when up to 20 more States vote on further Cannabis legalization at the end of the year. GrowLife will continue to establish itself as a national brand to provide the much-needed advisory services and sell lighting, nutrients, mediums and other equipment and supplies to cultivators across the country. The demand for more healthy, safer and economical ways to run indoor growing operations will dramatically increase and require innovative products to intelligently drive down costs without compromising quality. This is our expansion strategy.
Growlife (PHOT) growth plan consists of adding more knowledgeable, talented and committed people, creating greater access to equipment and supplies, and offering more and better choices. This is our strategy: Talented people, greater access, and better choices. Growlife.
$PMCB Extraction Technology Becoming Paramount in Cannabis Industry as Consumers Gravitate Towards Concentrates and Higher Quality of Substance

http://www.prnewswire.co.uk/news-releases/extraction-technology-becoming-paramount-in-cannabis-industry-as-consumers-gravitate-towards-concentrates-and-higher-quality-of-substance-587443771.html
GRNH
The company's websites -- CBDVentures www.greengrotech.com, www.420scan.com, www.growcameras.com, www.verticalhydrogarden.com, and www.bpgardens.com -- offer regular updates including educational videos, projects updates, recipes and nutritional information, and where to find the company's products.
https://greengrotech.com/
https://www.growcameras.com/
https://420scan.com/
https://verticalhydrogarden.com/
https://bpgardens.com/
$PMCB Pharmacyte Biotech Inc (OTCMKTS:PMCB): Upside Potential On A Phase 2 Catalyst

Mid September, Pharmacyte Biotech Inc (OTCMKTS:PMCB) CEO Kenneth L. Waggoner put out a press release addressing to the company’s pancreatic cancer investigation. Well, it was titled as such, but after touching on pancreatic cancer he went on to say he knows that the company’s share price is frustrating for investors, and that even though it looks like there’s very little going on behind the scenes, the company is working hard and fast to get its treatment back into the clinic.
A phase 2 study should have been well under way now, and certain delays have meant this isn’t yet the case. The delays, Waggoner says, are primarily related the the fact that the drug is a biologic and not an NCE. There’s some validity in what he is saying – biologics are more difficult to develop from a manufacturing and storage perspective – but it probably won’t wash with those shareholders that have held stock for the last two or three years waiting for a catalyst.
Anyway, we think that things are finally about to get moving again, and based on the company’s rhetoric, that we could see the initiation of the long awaited phase 2b at some point early next year.
We also really like the science. The company is using what it calls its Cell in a Box technology to go after a number of oncologic indications, of which pancreatic cancer is the primary target. The MOA is simple, but elegant. Currently, a combo treatment of Abraxane and gemcitabine (two SOC chemo agents) is the gold standard in pancreatic cancer. It works to some degree, but has a really severe toxicity profile, and patients are essentially undergoing treatment to get an extra eight weeks of life, each of which they spend in pain and vomiting. It’s the gold standard, but in this space, that doesn’t mean much.
Pharmacyte’s technology is rooted in a type of encapsulated cell that produces a protein that is otherwise produced only in the liver. With traditional chemotherapy, the chemo is introduced systemically, it passes through the liver, and the cell in question activates it. It then carries on through the system to its target (in this instance, the pancreas). Of course, as well as the pancreas, it’s also being washed around other parts of the body, killing healthy cells as it goes.
With Cell in a Box, a physician injects the encapsulated cell into the patient, as close to the cancerous cells (tumor) as possible. They then introduce a chemo agent called ifosfamide systemically, but at one third the dose of a standard treatment. The ifosfamide would normally need to pass through the liver for activation, but instead it travels to the cell capsules, which absorb it, activate it, and pump it back out on location.
So that’s how it works, what do we know about efficacy so far?
Well, from two trials that have been conducted, we know two important things.
One, that it seems to work. In a phase I trial, as compared to gemcitabine (another SOC chemodrugm, with the data points deriving from an old trial), the Cell in a Box ifosfamide combination increased median survival from 28 to 44 weeks, and the percentage of one-year survivors increased from 18% to 36%.
Two, that it isn’t improved with an increased dose. In a phase II trial, patients took a double dose, and efficacy results were weak. Also at the higher dose, the safety profile was poor.
With proof of concept in place, an established maximum tolerable dose and some sound safety data in hand, it can only be the above mentioned manufacturing issues that are holding Pharmacyte back. And it’s not the first company to require a biologic for an expanded trial, so we know the issue will resolve sooner or later.
Which brings us to a final point, cash. The longer the company holds out on initiating the trial, the longer it is going to have to rely on shareholder cash injections to maintain operations. It’s got about $2 million on hand right now, and burns about $1 million every quarter. Even without the added cost of a phase IIb initiation, therefore, cash wouldn’t last too far in to 2017. As such, we’ll almost certainly see a dilutive raise between now and trial initiation.
$TPNI .0002x.0003 huge support - gonna rip
Marijuana stock extraordinaire!!
GRNH getting ready for the next leg up my friends .....load up!
http://www.barchart.com/headlines/story/1143029/greengro-technologies-announces-sales-continue-to-soar-year-to-date-and-quarter-to-quarter-sales-prove-intense-demand-for-greengro-product-line
$ICNV .0005s going ! 500m AS
Iconvapor.com
$PMCB Live-cell encapsulation tech acts as both artificial pancreas and liver

http://www.fiercebiotech.com/medical-devices/pharmacyte-s-live-cell-encapsulation-tech-can-act-as-both-artificial-pancreas-and
This is the beginning of a MASSIVE RUN!
Arrayit Corporation, ARYC
@Arrayit
Market Cap of only $720k !!!!!!!
Featuring $5M credit facility
should be able to grow $20M revenues, $2-3M earnings, $60-90M cap and NASDAQ listing. Revenue of $820K last quarter and $2.3 million last year. Trading at an unprecedented low PPS of .0007
Owning shares of ARYC is a huge opportunity to create wealth. Arrayit should be trading at .10 -.25 based on earnings and growth model.
Just tweeted on Friday @Arrayit: over $1,000,000 in sales in first 2 months of 2016!!!!
Subsidiary of ARYC, AVDX now .22 Puts ARYC 40mm shares at $16mm. Yet market cap is currently at $720k??????...this one will be huge!!!!
A couple updates from this past week @Arrayit:
Arrayit schedules microarray services conference call today with world protein therapeutics leader Philadelphia PA.
Arrayit Store sales exceed $54 million all-time on 222,269 microarray products and services http://shop.arrayit.com pic.twitter.com/HbVEPj4bMX
Arrayit reports $138,478 microarray platform inquiry top medical researcher New Delhi India http://arrayit.com/Products/Microarray_Platforms/CGH_Microarrays/cgh_microarrays.html …
Arrayit reports record blood card sales in Q3-16 on microarray immunoassay research apps http://www.arrayit.com/Produc
Arrayit quotes $160,000 microarray platform to molecular diagnostic leader Akonni Biosystems http://www.akonni.com
Arrayit reports $34500 SpotBot 4 Microarray platform sale, leading European Distributor Grupo Taper Sintra Portugal http://www.arrayit.com/Products/Microarrayers/Microarrayer/microarrayer.html …
Arrayit reports microarray sale to testing leader RDL Reference Laboratory Los Angeles CA http://www.rdlinc.com
Arrayit ships $40,000 microarray platform life sciences leader Daemyung Sciences Seoul Korea http://www.dm4you.com
Arrayit reports $30,000 microarray services sale to a top research laboratory in Texas http://arrayit.com
Arrayit quotes $179,430 microarrays to biomedical leader Progenie Molecular Valencia España http://www.progenie-molecular.com
Arrayit quotes $51,680 microarrays to top leader De La Salle University Manila Philippines http://www.dlsu.edu.ph
Arrayit quotes $102,240 microarray platform to venture think tank Leading Technology Group http://www.leadingtechnology.co
Arrayit quotes $38,485 microarray platform to top Korean distributor Daemyung Science Seoul http://www.dm4you.com
Arrayit Corporation ?@arrayit
Arrayit completes conference call on huge DNA testing opportunity in massive market in India http://arrayit.com/Services/SNP_Genotyping/snp_genotyping.html…
Arrayit reports DNA testing inquiry from top agency in India. Market penetration of 0.1% would produce $48M revenues http://arrayit.com/Services/SNP_Genotyping/snp_genotyping.html…
Arrayit Corporation ?@arrayit
Arrayit quotes $235,000 microarray instruments to top lab University Ottawa Ontario Canada http://www.uottawa.ca
Arrayit reports microarray sale to advanced technology leaders Waki Company Tokyo Japan http://waki-bg.jp
Arrayit reports microarray sale to automated molecular testing leader AutoGenomics Vista CA http://www.autogenomics.com
Arrayit reports microarray sale to life sciences European Biotech Network Dolembreux Belgium http://www.euro-bio-net.com
Arrayit reports microarray sale to top medical researcher at $13.4B endowed MIT Cambridge MA http://web.mit.edu
Arrayit expands marketing on $100K+ TissueMax™ Automated and Personal Tissue Microarrayers
Arrayit reports $400,000 microarray instruments inquiry from top European Union distributor http://arrayit.com/Products/Microarrayers/Microarray_Printers/microarray_printers.html …
Arrayit invited to appear on Business Television BTV hosted by broadcast partner Bloomberg http://www.b-tv.com
Arrayit $17.7MM OvaDx® spin-off and 6,000 diagnostic test pipeline possibility emphasizes the $17.7MM x 6,000 = $106B ARYC market potential.
Arrayit reports $9K chemical sale in specialty trading subsidiary TeleChem International Inc http://www.telecheminternational.com
Arrayit reports $100K microarray inquiry Al Rayyan International Science Center Amman Jordan https://www.linkedin.com/in/nader-ghanayem-3b1306a2?authType=NAME_SEARCH&authToken=ofog&locale=en_US&srchid=2270647621456852532077&srchindex=1&srchtotal=2&trk=vsrp_people_res_photo&trkInfo=VSRPsearchId%3A2270647621456852532077%2CVSRPtargetId%3A365047477%2CVSRPcmpt%3Aprimary%2CVSRPnm%3Atrue%2CauthType%3ANAME_SEARCH …
$PMCB SCN's Wendy Gillette Interviews CEO Kenneth Waggoner of PharmaCyte Biotech, Inc. (OTCQB:PMCB)
ARYC: Arrayit Corporation reports $4,000,000+ purchase orders on surging 2016 sales, ARYC expansion and microarray industry growth http://arrayit.com
Currently trading at .0008!!!!!!!
Market Cap of $820k!!!!!
This is the one my friends:)
@arrayit
A couple updates from this past week @Arrayit:
Arrayit quotes $179,430 microarrays to biomedical leader Progenie Molecular Valencia España http://www.progenie-molecular.com
Arrayit quotes $51,680 microarrays to top leader De La Salle University Manila Philippines http://www.dlsu.edu.ph
Arrayit quotes $102,240 microarray platform to venture think tank Leading Technology Group http://www.leadingtechnology.co
Arrayit quotes $38,485 microarray platform to top Korean distributor Daemyung Science Seoul http://www.dm4you.com
Arrayit Corporation ?@arrayit
Arrayit completes conference call on huge DNA testing opportunity in massive market in India http://arrayit.com/Services/SNP_Genotyping/snp_genotyping.html…
Arrayit reports DNA testing inquiry from top agency in India. Market penetration of 0.1% would produce $48M revenues http://arrayit.com/Services/SNP_Genotyping/snp_genotyping.html…
Arrayit Corporation ?@arrayit
Arrayit quotes $235,000 microarray instruments to top lab University Ottawa Ontario Canada http://www.uottawa.ca
Arrayit reports microarray sale to advanced technology leaders Waki Company Tokyo Japan http://waki-bg.jp
Arrayit reports microarray sale to automated molecular testing leader AutoGenomics Vista CA http://www.autogenomics.com
Arrayit reports microarray sale to life sciences European Biotech Network Dolembreux Belgium http://www.euro-bio-net.com
Arrayit reports microarray sale to top medical researcher at $13.4B endowed MIT Cambridge MA http://web.mit.edu
Arrayit expands marketing on $100K+ TissueMax™ Automated and Personal Tissue Microarrayers
Arrayit reports $400,000 microarray instruments inquiry from top European Union distributor http://arrayit.com/Products/Microarrayers/Microarray_Printers/microarray_printers.html …
Arrayit invited to appear on Business Television BTV hosted by broadcast partner Bloomberg http://www.b-tv.com
Arrayit $17.7MM OvaDx® spin-off and 6,000 diagnostic test pipeline possibility emphasizes the $17.7MM x 6,000 = $106B ARYC market potential.
Arrayit reports $9K chemical sale in specialty trading subsidiary TeleChem International Inc http://www.telecheminternational.com
Arrayit reports $100K microarray inquiry Al Rayyan International Science Center Amman Jordan https://www.linkedin.com/in/nader-ghanayem-3b1306a2?authType=NAME_SEARCH&authToken=ofog&locale=en_US&srchid=2270647621456852532077&srchindex=1&srchtotal=2&trk=vsrp_people_res_photo&trkInfo=VSRPsearchId%3A2270647621456852532077%2CVSRPtargetId%3A365047477%2CVSRPcmpt%3Aprimary%2CVSRPnm%3Atrue%2CauthType%3ANAME_SEARCH …
$PMCB PharmaCyte Biotech’s Live-Cell Encapsulation Facility is Commissioned for GMP Manufacture

SILVER SPRING, Md., April 25, 2016 (GLOBE NEWSWIRE) — PharmaCyte Biotech, Inc. (OTCQB:PMCB), a clinical stage biotechnology company focused on developing targeted treatments for cancer and diabetes using its signature live-cell encapsulation technology, Cell-in-a-Box®, today reported that the encapsulation facility for its pancreatic cancer therapy is ready to manufacture PharmaCyte’s biologic product under current Good Manufacturing Practices (GMP) standards. The facility will be used to encapsulate the live cells used for PharmaCyte’s pancreatic cancer therapy. The assessment was issued by Chamow & Associates, the biopharmaceutical consulting firm that specializes in the inspection of facilities for GMP compliance.
Dr. Steven Chamow of Chamow & Associates stated, “Use of the live-cell encapsulation technology is very complex with many manufacturing steps and processes. GMP readiness of a biologic manufacturing facility such as the Bangkok site is no exception. In a relatively short period of time, with the relentless and determined cooperation we received from Austrianova, the facility is ready for GMP manufacture to produce the encapsulated cells that are a major part of PharmaCyte’s pancreatic cancer therapy.”
PharmaCyte Biotech’s Chief Executive Officer, Kenneth L. Waggoner, commented, “We are delighted to announce this exciting milestone in the development of our therapy for pancreatic cancer. This is a major step in generating the overall Chemistry, Manufacturing and Controls (CMC) information, which makes up a large part of the pre-IND package we will be submitting to the U.S. Food and Drug Administration (FDA). We would like to thank Chamow & Associates and Austrianova for completing this process. With the facility ready for cGMP manufacture, we are now in a position to engage with the FDA to discuss our clinical investigational plan and initiate our first clinical trial.”
Over the last few months, following an initial on-site audit of the facility by Chamow & Associates, numerous detailed documents were required to be prepared, reviewed and approved by Chamow. These documents include the quality management system manual and a variety of facility-related standard operating procedures (SOPs). In addition, all facility-related equipment has successfully completed the necessary Installation Qualifications (IQ) and Operational Qualifications (OQ).
Dr. Brian Salmons, the Chief Executive Officer of Austrianova, said, “We are exceedingly pleased that we were able to have our unique state-of-the-art live-cell encapsulation facility assessed as ready for GMP manufacture by Chamow & Associates. Both of the teams from Austrianova and Chamow worked together seamlessly and virtually nonstop to ensure that the facility is ready for GMP manufacture of PharmaCyte’s biologic product. We did so to enable PharmaCyte to reach the clinic with its novel therapy for pancreatic cancer at the earliest opportunity.”
The Austrianova facility will be used to encapsulate the genetically engineered live human cells that, together with low doses of the cancer prodrug ifosfamide, make up PharmaCyte’s pancreatic cancer therapy. PharmaCyte’s cancer therapy attacks the pancreas tumor with “targeted chemotherapy,” resulting in significant tumor shrinkage, the ability to convert some tumors from inoperable to operable, a reduction in the pain associated with the disease and an improvement to a patient’s overall quality of life.
PharmaCyte’s upcoming clinical trial in advanced inoperable pancreatic cancer involves placing the genetically modified live cells near the blood supply to the pancreas. The cancer prodrug ifosfamide is then given at one-third the normal dose. The prodrug is converted to its active form at the site of the tumor. In an earlier Phase 1/2 clinical trial, this “targeted chemotherapy” demonstrated far greater efficacy than the then “gold-standard” of care with no meaningful side-effects from the chemotherapy. Patients enrolled in PharmaCyte’s clinical trial will have non-metastatic, locally advanced and inoperable pancreatic cancer. They will be eligible for the trial if their tumors are either stable or progressing after 4-6 cycles of treatment with either of the two most commonly used chemotherapies for these cancers – the two-drug combination of Abraxane® plus gemcitabine or the four-drug combination known as FOLFIRINOX. PharmaCyte’s therapy will be compared in such patients with the current “standard of care,” which consists of the combination of the anticancer drug capecitabine plus radiation.
About PharmaCyte Biotech
PharmaCyte Biotech is a clinical stage biotechnology company focused on developing and preparing to commercialize treatments for cancer and diabetes based upon a proprietary cellulose-based live cell encapsulation technology known as “Cell-in-a-Box®”. This unique and patented technology will be used as a platform upon which treatments for several types of cancer and diabetes are being developed. PharmaCyte’s treatment for cancer involves encapsulating genetically modified live cells that convert an inactive chemotherapy drug (ifosfamide) into its active or “cancer-killing” form. The encapsulated live cells are placed as close to a cancerous tumor as possible. Once implanted in a patient, ifosfamide is given intravenously at one-third the normal dose. The ifosfamide is carried by the circulatory system to where the encapsulated live cells have been placed. When the ifosfamide, which is normally activated in the liver, comes in contact with the encapsulated live cells, activation of the chemotherapy drug takes place at the source of the cancer without any side effects from the chemotherapy. This “targeted chemotherapy” has proven remarkably effective and safe to use in past clinical trials.
In addition to developing a novel treatment for cancer, PharmaCyte is developing a treatment for Type 1 diabetes and Type 2 insulin-dependent diabetes. PharmaCyte plans to encapsulate a human cell line that has been genetically engineered to produce, store and release insulin in response to the levels of blood sugar in the human body. The encapsulation will be done using the Cell-in-a-Box® technology.
Safe Harbor
This press release may contain forward-looking statements regarding PharmaCyte and its future events and results that involve inherent risks and uncertainties. The words “anticipate”, “believe”, “estimate”, “expect”, “intend”, “plan” and similar expressions, as they relate to PharmaCyte or its management, are intended to identify forward-looking statements. Important factors, many of which are beyond the control of PharmaCyte, could cause actual results to differ materially from those set forth in the forward-looking statements. They include PharmaCyte’s ability to continue as a going concern, delays or unsuccessful results in preclinical and clinical trials, flaws or defects regarding its product candidates, changes in relevant legislation or regulatory requirements, uncertainty of protection of PharmaCyte’s intellectual property and PharmaCyte’s continued ability to raise capital. PharmaCyte does not assume any obligation to update any of these forward-looking statements.
More information about PharmaCyte Biotech can be found at www.PharmaCyte.com. It can also be obtained by contacting Investor Relations.
Investor Relations:
PharmaCyte Biotech, Inc.
Investor Relations Department
Telephone: 917.595.2856
Email: Info@PharmaCyte.com
http://investingnews.com/daily/life-science-investing/biotech-investing/pharmacyte-biotechs-live-cell-encapsulation-facility-commissioned-gmp-manufacture/
$MCGI Nursing Schools Use Nurses Lounge to Re-Connect with Alumni
ARYC! @Arrayit
Unbelievably low market cap of $540K! Trading at .15 x earnings!
Earnings of over $1 million in first 2 months of 2016.
Located in the heart of Silicon Valley near Apple, Yahoo, AMD, Juniper, MBZ, LinkedIn, Facebook and many other leaders of the tech industry.
Earnings of $2.3 million last year and on track for even higher this year
Trading at unprecedented low of .0005!!!!!!!!
No other sub penny company has the potential of ARYC
Check out their tweets @Arrayit or go to their Facebook page.
RXSF $1.56 going now only 175 left then $2.20 ~ up list coming!!!
$PMCB PharmaCyte Biotech on Schedule for 2016 Cancer Clinical Trial in Pancreatic Cancer

SILVER SPRING, Md., Feb. 24, 2016 (GLOBE NEWSWIRE) -- PharmaCyte Biotech, Inc. (OTCQB:PMCB), a clinical stage biotechnology company focused on developing targeted treatments for cancer and diabetes using its signature live-cell encapsulation technology, Cell-in-a-Box®, issued a further update on its upcoming clinical trial in pancreatic cancer. In this update, PharmaCyte’s Chief Executive Officer, Kenneth L. Waggoner, clarifies statements made earlier this week that reportedly confused numerous shareholders and potential investors.
“Last Monday I tried to address certain issues that shareholders and potential investors have raised repeatedly about our upcoming clinical trial and provide an update on our efforts to complete the process of preparing the Investigational New Drug application (IND) that PharmaCyte must submit to the FDA to start our clinical trial in pancreatic cancer. Given the number of telephone calls and emails the Company has received since that update was published, it has become apparent that the update caused confusion and raised issues of concern that were never intended.
“When it was stated on Monday that the expected ‘start date’ for PharmaCyte’s clinical trial in pancreatic cancer remains difficult to announce with certainty, we were not implying that we aren’t still on schedule to get into the clinic in 2016 or that we don’t have any idea when we will be in the clinic. We plan to be in the clinic well before the end of this year. It was simply a statement that intended to refrain from announcing a specific date or time period for starting the trial because much of what is left to complete is out of our control.
“PharmaCyte is involved in a well-planned, detailed and methodical process to insure that every aspect of the IND is correct before we submit the IND to the FDA. As the “Sponsor” of the IND, it is our responsibility to insure that the Chemistry, Manufacturing and Controls (CMC) section of the IND is complete and accurate in every respect. We are working with Translational Drug Development (TD2), Chamow & Associates (Chamow) and Austrianova in completing everything that is required. It is a process that began last year and is well underway.
“TD2 is fully engaged and working diligently in all aspects of preparing for the clinical trial. With respect to developing the CMC information, the process is complicated, labor intensive and highly technical. But with TD2 taking the lead, working in concert with Chamow and Austrianova, I am very confident that our schedule is on track.”
About PharmaCyte Biotech
PharmaCyte Biotech is a clinical stage biotechnology company focused on developing and preparing to commercialize treatments for cancer and diabetes based upon a proprietary cellulose-based live cell encapsulation technology known as “Cell-in-a-Box®.” This unique and patented technology will be used as a platform upon which treatments for several types of cancer and diabetes are being developed. PharmaCyte’s treatment for cancer involves encapsulating genetically modified live cells that convert an inactive chemotherapy drug into its active or “cancer-killing” form. These encapsulated live cells are placed as close to a cancerous tumor as possible. Once implanted in a patient, a chemotherapy drug which needs to be activated in the body (ifosfamide) is then given intravenously at one-third the normal dose. The ifosfamide is carried by the circulatory system to where the encapsulated cells have been placed. When the ifosfamide, which is normally activated in the liver, comes in contact with the encapsulated live cells, activation of the chemotherapy drug takes place at the source of the cancer without any side effects from the chemotherapy. This “targeted chemotherapy” has proven remarkably effective and safe to use in past clinical trials.
In addition to developing a novel treatment for cancer, PharmaCyte is developing a treatment for Type 1 diabetes and Type 2 insulin-dependent diabetes. PharmaCyte plans to encapsulate a human cell line that has been genetically engineered to produce, store and release insulin in response to the levels of blood sugar in the human body. The encapsulation will be done using the Cell-in-a-Box® technology.
Safe Harbor
This press release may contain forward-looking statements regarding PharmaCyte Biotech and its future events and results that involve inherent risks and uncertainties. The words "anticipate," "believe," "estimate," "expect," "intend," "plan" and similar expressions, as they relate to PharmaCyte or its management, are intended to identify forward-looking statements. Important factors, many of which are beyond the control of PharmaCyte, could cause actual results to differ materially from those set forth in the forward-looking statements. They include PharmaCyte's ability to continue as a going concern, delays or unsuccessful results in preclinical and clinical trials, flaws or defects regarding its product candidates, changes in relevant legislation or regulatory requirements, uncertainty of protection of PharmaCyte’s intellectual property and PharmaCyte’s continued ability to raise capital. PharmaCyte does not assume any obligation to update any of these forward-looking statements.
More information about PharmaCyte Biotech can be found at www.PharmaCyte.com. It can also be obtained by contacting Investor Relations.
Investor Relations:
PharmaCyte Biotech, Inc.
Investor Relations Department
Telephone: 917.595.2856
Email: info@PharmaCyte.com
TRTC! Terra Tech Corp. Closes Acquisition of Blüm Oakland, a Retail Medical Cannabis Dispensary
http://www.otcmarkets.com/stock/TRTC/news/Terra-Tech-Corp--Closes-Acquisition-of-Bl-uuml-m-Oakland--a-Retail-Medical-Cannabis-Dispensary?id=128530&b=y
TRTC Terra Tech Launches New Line of Pre-filled Cannabis Oil Cartridges
http://www.otcmarkets.com/news/otc-market-headline?id=476417
$PMCB PharmaCyte Biotech Addresses Development of Targeted Cannabinoid Chemotherapy

SILVER SPRING, Md., March 01, 2016 (GLOBE NEWSWIRE) -- PharmaCyte Biotech, Inc. (PMCB), a clinical stage biotechnology company focused on developing targeted treatments for cancer and diabetes using its signature live-cell encapsulation technology, Cell-in-a-Box®, announced today that Scientific Advisory Board member Mark L. Rabe, MD, was a featured speaker at a Physicians Roundtable that took place in San Diego this past weekend. The Physicians Roundtable was designed to educate local physicians and health care providers on the topic: “The Endocannabinoid System: Leveraging the Largest Receptor System in the Human Body.”
Dr. Rabe’s presentation, “The Evolution of Medical Cannabis,” traced the history of Cannabis from ancient times to the present day, citing numerous references from the proliferation of medical literature that document the anti-cancer, pain-relieving, anti-inflammatory and neuroprotective effects of the “phyto”-cannabinoid molecules contained in the Cannabis plant. Looking to the future, Dr. Rabe explained how PharmaCyte is seeking to leverage the body’s endocannabinoid system through development of tumor-targeted treatments for serious and deadly cancers by utilizing cannabinoid prodrugs in combination with the versatile Cell-in-a-Box® live-cell encapsulation platform. This research is being conducted for PharmaCyte by researchers at the University of Northern Colorado (UNC) under a Schedule 1 license successfully obtained from the U.S. Drug Enforcement Agency.
"In order to provide better care to their patients, it is very heartening to see health care professionals dedicate precious time on a Saturday morning to become more educated in the recommendation of an herbal medicine that a significant – and growing – number of patients report works better, with far fewer side effects, than the pharmaceutical alternatives,” commented Dr. Rabe. “The work being conducted at UNC to utilize Cell-in-a-Box® live cell encapsulation in combination with a unique bioengineered cell line to activate cannabinoid prodrugs in a targeted fashion to treat cancer, and potentially other diseases, exemplifies where things are headed in the future.”
A copy of Dr. Rabe’s slide deck and references are posted at the PharmaCyte Biotech website: http://www.PharmaCyte.com/media
About PharmaCyte Biotech
PharmaCyte Biotech is a clinical stage biotechnology company focused on developing and preparing to commercialize treatments for cancer and diabetes based upon a proprietary cellulose-based live cell encapsulation technology known as “Cell-in-a-Box®.” This unique and patented technology will be used as a platform upon which treatments for several types of cancer and diabetes are being developed. PharmaCyte’s treatment for cancer involves encapsulating genetically modified live cells that convert an inactive chemotherapy drug into its active or “cancer-killing” form. These encapsulated live cells are placed as close to a cancerous tumor as possible. Once implanted in a patient, a chemotherapy drug which needs to be activated in the body (ifosfamide) is then given intravenously at one-third the normal dose. The ifosfamide is carried by the circulatory system to where the encapsulated cells have been placed. When the ifosfamide, which is normally activated in the liver, comes in contact with the encapsulated live cells, activation of the chemotherapy drug takes place at the source of the cancer without any side effects from the chemotherapy. This “targeted chemotherapy” has proven remarkably effective and safe to use in past clinical trials.
In addition to developing a novel treatment for cancer, PharmaCyte is developing a treatment for Type 1 diabetes and Type 2 insulin-dependent diabetes. PharmaCyte plans to encapsulate a human cell line that has been genetically engineered to produce, store and release insulin in response to the levels of blood sugar in the human body. The encapsulation will be done using the Cell-in-a-Box® technology.
Safe Harbor
This press release may contain forward-looking statements regarding PharmaCyte Biotech and its future events and results that involve inherent risks and uncertainties. The words "anticipate," "believe," "estimate," "expect," "intend," "plan" and similar expressions, as they relate to PharmaCyte or its management, are intended to identify forward-looking statements. Important factors, many of which are beyond the control of PharmaCyte, could cause actual results to differ materially from those set forth in the forward-looking statements. They include PharmaCyte's ability to continue as a going concern, delays or unsuccessful results in preclinical and clinical trials, flaws or defects regarding its product candidates, changes in relevant legislation or regulatory requirements, uncertainty of protection of PharmaCyte’s intellectual property and PharmaCyte’s continued ability to raise capital. PharmaCyte does not assume any obligation to update any of these forward-looking statements.
More information about PharmaCyte Biotech can be found at www.PharmaCyte.com. It can also be obtained by contacting Investor Relations.
Investor Relations:
PharmaCyte Biotech, Inc.
Investor Relations Department
Telephone: 917.595.2856
Email: Info@PharmaCyte.com
ARYC: Arrayit @Arrayit: posted excellent quarterly earnings, over $2.3 million in sales last year, a dividend is going to be paid to shareholders and the stock is at an all time low. We are seeing great volume, one of the primary marketmakers raised it's Ask up and Arrayit's products are selling, in the thousands of dollars every single day. The company is working to increase your share price and has actually stated that is its intention. Do not miss an opportunity, do not get swayed by misinterpreted guidance, buy as many shares as possible, I know the share price for Arrayiti has potential to easily reach $.25 a share, with PPS potential of $1+. This is an opportunity of a lifetime.
U are very welcome
Thanks for that great chart! VCEL $$$$$
VCEL-- solid emerging growth pharma that has all the makings of next AMGN! Check out lasers posts on VCEL board, excellent DD! VERY PROMISING YOUNG STOCK AT A GOOD PRICE NOW! Data release April 4th too!!!
Anyone remember BRWC? 2 8k's should be read
The first is an 8k that secures $250k financing backed by warrants that exercise at .0075 while the current price is .0004.
Current Report Filing (8-k) http://ih.advfn.com/p.php?pid=nmona&article=69990479
On January 5, 2016, Boreal Water Collection, Inc. entered into an agreement with Mr. Abdul Aziz Al Athel (“subscriber”). The Agreement is for $250,000 of Secured Debentures and Common Share Warrants in Units of $1000.00. Each Unit contains one 7.5% interest bearing Secured Debenture in the principal amount of $1000.00 and a Warrant to purchase 1,352,532 common shares at an exercise price of $0.0075 per share.
My client, Boreal Water Collection Inc is exercising its First Right of Refusal as outlined in a certain agreement between Leisure Time Spring Water, Inc., (Leisure Time), A New York Corporation, Alpine Farms, Inc (Alpine), a New York Corporation and Andrew J. Krieger and Suri Levow Krieger dated November 1, 1995, as modified by an Agreement of Modification between Leisure Time and Alpine, dated April 25, 2000, which agreement provides, inter alia, Leisure Time the sole and exclusive right to draw water and use the artesian springs, water supply and existing facility upon Town of Callicoon SBL No: 4-1-14.1 and which further provides, pursuant to paragraph 13 thereof, Leisure Time an option of first refusal in the event that the owner of the aforesaid real property contracts to sell same.
Enclosed please find check number 15437 made payable to you as attorney in the amount of sixty thousand ($60,000.00) dollars which represents required down payment in the contract of sale.
ARYC!!
Arrayit.com website
@Arrayit
Float is low and locked up.
Extremely undervalued with a Market Cap ($620K) that is less than its last quarter earnings.
$2.3 million revenue last year and will be much higher this year!
Up from 52 week low last week and beginning a massive run up.
Arrayit Corporation ?@arrayit
Arrayit reports 24.9% revenues increase and 42.2% gross profit increase for Q3-2015 compared to Q3-2014. Go ARYC!!! http://www.sec.gov/Archive
$CRTC ~ NEWS IS OUT ~ PPS .0004
http://www.otcmarkets.com/stock/CRTC/news/Critic-Clothing-announces-plans-for-massive-growth-in-2016?id=122367&b=y
Critic Clothing announces plans for massive growth in 2016
Jan 04, 2016
OTC Disclosure & News Service
Mount Sinai, NY -
MOUNT SINAI, N.Y., January 4, 2016 – Critic Clothing, Inc. (OTCPink: CRTC) today announced that the company has big plans lined up for 2016, including several patents to be filed, Additions to the board of directors, local celebrity events, and even the creation of a non-profit organization to give back to the community.
Now that the long awaited name change is completed, Critic Clothing can start to push forward and announce the many developments the company has been working on all year. Alec Simione, the company’s Founder and CEO explained, “I will admit that we have been working mostly in secrecy this past year because the majority of what we have been working on is so big that we didn’t want it to come out under a company name we knew would be changing. I want to thank our fans, partners, and shareholders for being so patient with us, and I also want to thank FINRA for finally deciding in favor of the name change. I can tell everyone with confidence that they have only seen the tip of the iceberg in terms of what we are, and I think everyone is in for some big surprises very soon as we uncover our full strategy, partners, and deals.”
The past year did bring successes for Critic Clothing who, as the subsidiary of Evader, Inc. has begun to make a name for itself in the apparel industry. Many professional athletes have been spotted wearing Critic gear and many of them praised the company and gave a shout out as well. In response, the company has created the 24k Club, an area of its website at www.criticclothing.com to feature celebrity shout-out’s and sightings. Some of the additions to the 24K club are players from the Miami Dolphins, New York Giants, ST. Louis Rams, Atlanta Falcons, Buffalo Bills, New England Patriots, Chicago Bears, New York Yankees, and many more. Several of these players have authorized a “Signature Series” shirt with their number, favorite slogans, and printed autograph, making these shirts very popular with fans. The company asserts that there are many other public personalities and celebrities that will soon be gracing the pages of the 24k club as well. Many are the result of the ESPYS awards attended by Critic in 2015, and have just not been promoted as of yet because of the name change that was not completed until recently. Promotions and even autograph signings are in the works so the public is encouraged to watch the Critic website for updates.
Aside from the public exposure, Critic Clothing has been working very hard behind the scenes to line up distribution in retail both online and in brick and mortar stores. The company plans to announce several deals in the early part of 2016 to carry special edition items in well knows retail chains, which could make Critic a household name over night. Perhaps more exciting than any of the developments thus far is the patent that the company is currently in the process of filings, soon to be announced, it is one of many completely unique clothing items that Critic Clothing feels will set the company apart from any company like it in the world. “Our next big evolution is the creation of unique patented, fashionable and functional clothing items that nobody makes but us. That, coupled with our strong message and celebrity following is what makes this company great!” Mr. Simione explained, “But, at the end of the day we do not just sell t-shirts, we sell dreams. That’s what Critic is all about and that is why we have been so successful so far at spreading the word.”
Critics powerful message comes to life in one of the projects being put together by Mr. Simione, which involves the creation of a non-profit organization for the purpose of giving back a percentage of company earnings to the community, specifically geared towards skate parks and similar projects in order to inspire young people to overcome criticism and reach for their dreams no matter how far away they may seem.
About Critic Clothing, Inc. (Formerly Evader, Inc.)
Critic Clothing, Inc. is a manufacturer of clothing geared towards young adults and teens with a passion for extreme sports. The company sells products on its website www.criticclothing.com and has plans in the works to expand from local retail shops to retail stores across America. For more information call: (631) 333-1600 or follow the company on twitter: @criticclothing
DISCLAIMER
This Press Release may contain certain forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. The Company has tried, whenever possible, to identify these forward-looking statements using words such as "anticipates," "believes," "estimates," "expects," "plans," "intends," "potential" and similar expressions. These statements reflect the Company's current beliefs and are based upon information currently available to it. Accordingly, such forward-looking statements involve known and unknown risks, uncertainties and other factors which could cause the Company's actual results, performance or achievements to differ materially from those expressed in or implied by such statements. The Company undertakes no obligation to update or advise in the event of any change, addition or alteration to the information catered in this Press Release including such forward-looking statements.
Copyright © 2016 OTC Markets. All Rights Reserved
The above news release has been provided by the above company via the OTC Disclosure and News Service. Issuers of news releases and not OTC Markets Group Inc. are solely responsible for the accuracy of such news releases.
********$PNOW .0003x.0004 w/ .0002 Bottom *******
ARYC continued it's great earnings with Q3 bringing in almost $620,000. Earnings for 2015 were $2.3 million. ARYC is currently trading at .0008 with 707 mill outstanding shares. Currently trading at .25 X Earnings. Get in before it returns to where it should be trading: .25+
Please DD ARYC, my friends.
ARYC: Arrayit is a leading edge developer, manufacturer and marketer of next-generation life science tools and integrated systems for the large scale analysis of genetic variation, biological function and diagnostics. Using Arrayit’s proprietary and patented technologies, the Company provides a comprehensive line of products and services that currently serve the sequencing, genotyping, gene expression and protein analysis markets, and the Company expects to enter the market for manufacturing molecular diagnostics.
Arrayit has earned respect as a leader in the health care and life sciences industries with its proven expertise in three key areas: the development and support of microarray tools and components, custom printing and analysis of microarrays for research, and the identification and development of diagnostic microarrays and tools for early detection of treatable disease states. As a result, Arrayit has provided tools and services to thousands of the leading genomic research centers, pharmaceutical companies, academic institutions, clinical research organizations, government agencies and biotechnology companies worldwide.
Check out their Facebook page and daily updates on Twitter.
ARYC I'm still in not averaging down unless it hits 0005 average is .0008 spotted it at .0005 but didn't pul the trigger fast enough
ARYC. Not a good call. Pump and dump
ARYC cross trade of last note for the year 24M .00095
Bro we have ourselves a could be promising big hitter coming
|
Followers
|
137
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
3023
|
|
Created
|
06/01/14
|
Type
|
Free
|
| Moderator TheTradingNinja | |||
| Assistants MoneyMaker20 | |||

|
Posts Today
|
0
|
|
Posts (Total)
|
3023
|
|
Posters
|
|
|
Moderator
|
|
|
Assistants
|
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
