Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Talk to us Buzz. What do you think?
s Zynerba Pharmaceuticals (ZYNE) Stock Outpacing Its Medical Peers This Year?
Zacks
Zacks Equity Research
,Zacks•May 25, 2020
Is Zynerba Pharmaceuticals (ZYNE) Stock Outpacing Its Medical Peers This Year?
More
The Medical group has plenty of great stocks, but investors should always be looking for companies that are outperforming their peers. Is Zynerba Pharmaceuticals (ZYNE) one of those stocks right now? By taking a look at the stock's year-to-date performance in comparison to its Medical peers, we might be able to answer that question.
Zynerba Pharmaceuticals is one of 889 individual stocks in the Medical sector. Collectively, these companies sit at #1 in the Zacks Sector Rank. The Zacks Sector Rank considers 16 different sector groups. The average Zacks Rank of the individual stocks within the groups is measured, and the sectors are listed from best to worst.
The Zacks Rank is a proven model that highlights a variety of stocks with the right characteristics to outperform the market over the next one to three months. The system emphasizes earnings estimate revisions and favors companies with improving earnings outlooks. ZYNE is currently sporting a Zacks Rank of #2 (Buy).
Over the past three months, the Zacks Consensus Estimate for ZYNE's full-year earnings has moved 7.90% higher. This means that analyst sentiment is stronger and the stock's earnings outlook is improving.
Based on the most recent data, ZYNE has returned 7.28% so far this year. Meanwhile, the Medical sector has returned an average of -1.66% on a year-to-date basis. As we can see, Zynerba Pharmaceuticals is performing better than its sector in the calendar year.
Breaking things down more, ZYNE is a member of the Medical - Generic Drugs industry, which includes 24 individual companies and currently sits at #15 in the Zacks Industry Rank. On average, stocks in this group have lost 6.85% this year, meaning that ZYNE is performing better in terms of year-to-date returns.
Investors with an interest in Medical stocks should continue to track ZYNE. The stock will be looking to continue its solid performance.
Thanks BrownCornSnake.No doubt, it's going to get real interesting around here,short,and long term.
Looking at those possible (conservative) revenue numbers ,and then looking at the current SP........."Disconnect" is the understatement of the century.
GLTY and all longs.
Good stuff. Thanks for all you do. I’m looking forward to the next few weeks and months.
A glance back to what brought on the fall from 16 to 2 ,and what may be instore.
The benefits in the behavioral components of FXS have led to the development of Zygel for other syndromes with behavioral manifestations. These include ASD and 22q. ASD affects nearly 1 million patients, and can include symptoms such as anxiety, repetitive patterns of behavior, impairment in social communication, and social impairment. There are limited products approved for ASD as of present time. The BRIGHT study is a 14 week trial evaluating Zygel in ASD patients. The trial is a single arm trial that will evaluate the effect of Zygel on several aspects of behavior and anxiety. Results of this trial are expected in the second quarter. Finally, we have 22q, which affects around 81,000 patients in the United States. This disorder results in neuropsychiatric illnesses including anxiety. Presently, there are no approved drugs for the treatment of 22q, which paves the way for Zygel to be a first to market product. The INSPIRE trial is evaluating the use of Zygel for patients with 22q11.2 deletion syndrome, and will look at the neuropsychiatric effect at 14 weeks, similar to the BRIGHT trial. Results of this trial are expected in the third quarter of 2020. So lets look at the market opportunity here. The current WAC price of Epidiolex is around $32,500 annually. We will use a similar price point for our estimates. Using the market opportunities as estimated by Zynerba, we arrive at these conservative annual revenue for Zygel:
FXS (10% market penetration): $210 million annually
ASD (2% market penetration): $585 million annually
22q (10% market penetration): $240 million annually
These estimates do not include any potential revenue from a DEE indication either. As you can see, these potential revenues far exceed the current valuation (market cap of $100 million). So where is the disconnect? Well when Zynerba released their most recent DEE, the stock sold off sharply on what is perceived to be safety concerns. Most people will point to the infection-related adverse events. First, in clinical trials, all adverse events are reported, whether they are related to trial drug or not (runny nose, ingrown toenail, scrapes, etc.). So the absolute rate of adverse events is misleading. Furthermore, what people need to understand is that these patients are medically fragile. Many of these patients have co-morbid conditions such as cerebral palsy, chronic respiratory infections, tracheostomies (breathing tubes), and feeding tubes. Many of these conditions predispose patients to frequent health care visits (risk factor for infection) and infections (breathing tubes). To be honest, it is not surprising these patients had infections, but it is more likely related to their other conditions and not related to treatment with Zygel. All of this concern overshadowed the fact that Zygel resulted in a 44% median reduction in seizures by month 2. Furthermore, only one patient discontinued therapy with Zygel (due to application site reaction).
BIZARROW WORLD aint nearly as bizarrow as it looks some time....there IS a method to the madness...9 out of 10 times.
Got to wonder,how many lawyers that opened the door to law suits
bought shares in the 2 to 6 range? lol
Hey 4in...Thanks for that,glad to bring company info here.
Armando and his team are the real workers here,I've had the chance to speak
with Will Roberts several times over the years,and he is TOP NOTCH.As Armando stated months ago "the heavy lifting has been done"
Harvest time is closer.
Hmm... lol thanks dhbuzz. Your work here is outstanding! Go zyne
What could Armando posibly have to talk about???
"![]()
Older news for longs,but good info for any new
board readers.I'm sure we are going to start seeing more new faces.
https://zynerba.com/zynerba-pharmaceuticals-receives-fast-track-designation-for-zygel-for-the-treatment-of-behavioral-symptoms-associated-with-fragile-x-syndrome-fxs/
A deep look into FX.
https://www.cdc.gov/ncbddd/fxs/index.html
Here is what we are waiting on.BRIGHT Phase 2,and also FDA meeting date anounced on DEES.Could happen any day now.
Then the big one... FX phase 3 results sometime in June,and FDA NDA...meeting.
DEVON, Pa., March 7, 2019 – Zynerba Pharmaceuticals, Inc. (NASDAQ:ZYNE), the leader in innovative pharmaceutically-produced transdermal cannabinoid therapies for rare and near-rare neuropsychiatric disorders, today announced that it has initiated the Phase 2 BRIGHT (An Open-Label Tolerability and Efficacy Study of ZYN002 Administered as a Transdermal Gel to Children and Adolescents with Autism Spectrum Disorder) trial. The trial will assess the safety, tolerability and efficacy of Zygel (previously referred to as ZYN002) for the treatment of child and adolescent patients with Autism Spectrum Disorder (ASD). The Company expects to present topline data from this study in the first half of 2020.
“Autism spectrum disorder can have a devastating impact on a child and their family,” said Armando Anido, Chairman and Chief Executive Officer of Zynerba. “The medical need is significant and unmet despite high awareness and advocacy efforts. Though there has been an accelerating rate of diagnosis, to date there are only two FDA approved products indicated for the treatment of ASD symptoms. Both have significant side effect profiles, and neither have been approved to address the key symptoms of social impairment and anxiety. We are excited to initiate the BRIGHT trial evaluating the role of Zygel in ASD and are hopeful that Zygel may improve some of the core social and behavioral symptoms of ASD. We look forward to presenting topline data in the first half of 2020.”
The 14-week BRIGHT trial is an open-label multi-dose Phase 2 clinical trial designed to evaluate the efficacy and safety of Zygel in approximately 36 children and adolescents (ages four through 17) with ASD as confirmed by DSM-5 diagnostic criteria for ASD. Enrolled patients will receive weight-based initial doses of 250 mg daily or 500 mg daily of Zygel. The efficacy assessments include the Aberrant Behavior Checklist, Parent Rated Anxiety Scale – Autism Spectrum Disorder, Autism Impact Measure, and Clinical Global Impression – Severity and Improvement.
Z looks like Headway in the gate.Bouncing around and ready to run this morning. lol
Love that last paragraph
*****************
Final Thoughts
Zynerba has the potential to provide a therapy for many patients with an unmet need. The upside of Zynerba is massive after considering the potential revenue opportunity compared to its current valuation (market cap of $100 million). I believe that the misunderstanding on the safety of Zygel has created a severe disconnect between the current valuation and potential valuation. This disconnect will likely be rectified in the coming weeks to months with several data readouts due. I anticipate these data readouts will be positive, resulting in price appreciation. At some point, I anticipate they will raise capital, however in the short and long term, I am very bullish on ZYNE. For an outline of expected catalysts, please see Figure 2, and best of luck to all investors!
Thanks BC Snake.
Keep that volume cranked.At this point I'd say its safe to assume Z is getting some attention.
Good luck to you too. Let’s see one more run to end the day!!
Exactly. GLTY GO Z!!!
Nice. I’ve been adding each week for a while. Not much but it all adds up in the end.
I've been a tiny part of the vol. today
100 shrs. added
Here we go. Good volume again today
Zynerba...AKA HEAD WAY in the horse racing circles...
https://video.search.yahoo.com/yhs/search?fr=yhs-omr-001&hsimp=yhs-001&hspart=omr&p=u+tube+hhose+race+headed+for+home&_guc_consent_skip=1589757789#id=42&vid=54f657cbc8e79ad818700a113ce2037e&action=view
We are also seeing more days with above avg. volume.We saw recently how quickly it moved to 5.20
Clock is ticking
That gives me even more confidence when you see a number like that.
47% of stock owned by insitutions.That's a pretty darn good number.
Always like to see the big players get in.
Looks like they did.
So,am I reading this right. ETF Management Group bought over a million shares yesterday?
https://ih.advfn.com/stock-market/NASDAQ/zynerba-pharmaceuticals-ZYNE/stock-news/82427221/amended-statement-of-ownership-sc-13g-a
Another strong volume day.Sounds like all trials are moving along on schedule,and next FDA scheduled meeting update could come any day now.
|
Followers
|
91
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
2676
|
|
Created
|
08/06/15
|
Type
|
Free
|
| Moderators | |||

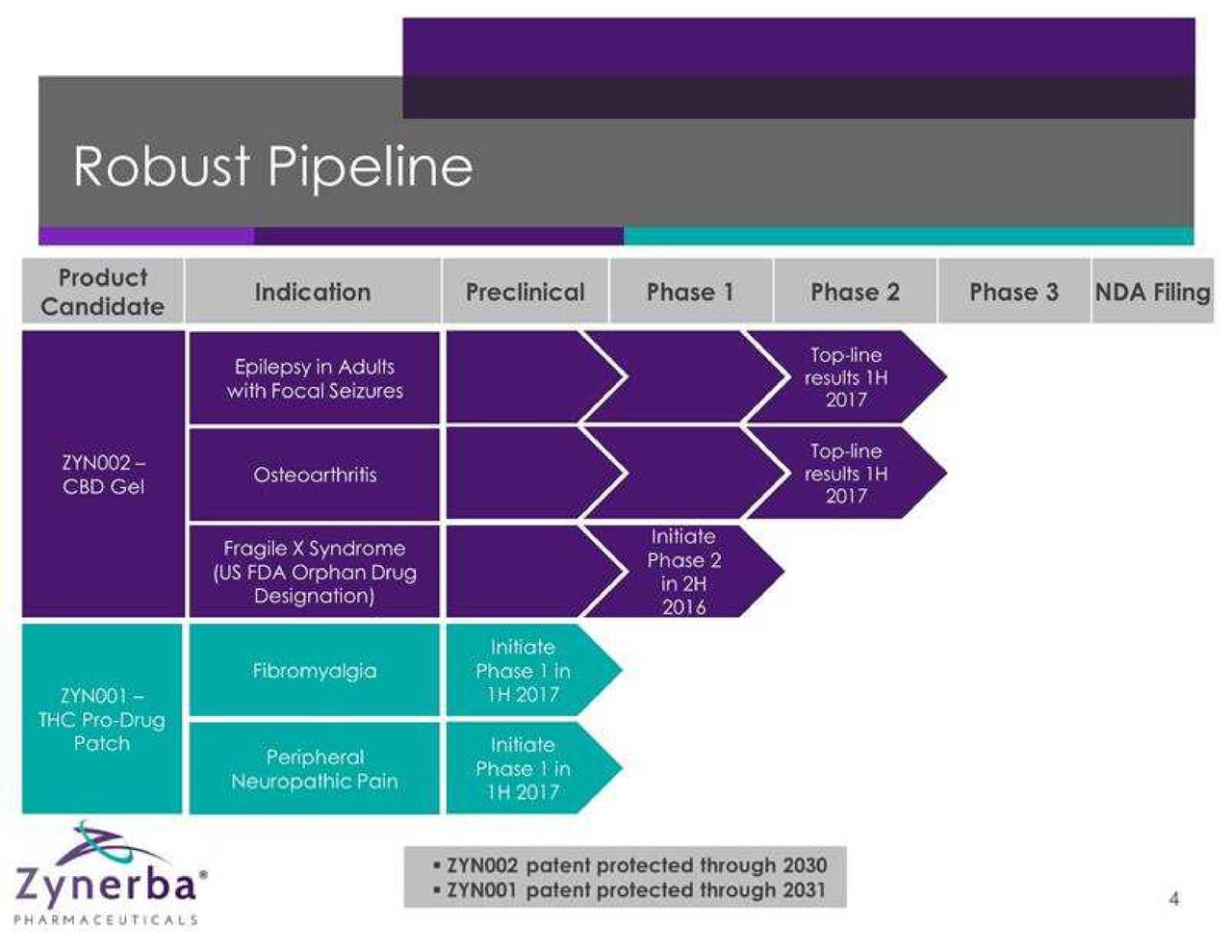
Zynerba Pharmaceuticals, Inc. is a specialty pharmaceutical company focused on developing and commercializing synthetic cannabinoid therapeutics formulated for transdermal delivery. The Company is evaluating approximately two product candidates, ZYN002 and ZYN001, in over five indications. The Company intends to study ZYN002 in patients with refractory epilepsy, osteoarthritis and Fragile X syndrome. The Company's ZYN002 is synthetic cannabidiol (CBD) formulated as a permeation-enhanced gel for transdermal delivery. ZYN002 is being developed as a clear that is designed to provide controlled drug delivery with once- or twice-daily dosing. ZYN001 is a pro-drug of tetrahydrocannabinol (THC) that enables transdermal delivery through a patch. The Company intends to test the ZYN001 patch for application to the arm, back and thigh. The Company intends to study ZYN001 in patients with fibromyalgia and peripheral neuropathic pain.
| Corporate Profile |
Zynerba (NASDAQ: ZYNE) is pioneering the development of patent-protected, next-generation synthetic cannabinoid therapeutics formulated for transdermal delivery. Its two lead product candidates in development include ZYN002 and ZYN001, which are being evaluated in five indications. ZYN002 is the first and only synthetic cannabidiol (CBD) formulated as a permeation-enhanced gel for transdermal delivery. In June 2016, the company initiated the STAR 1 Phase 2 clinical trial in refractory epilepsy patients and in August 2016, initiated the STOP Phase 2 clinical trial in patients with osteoarthritis of the knee. A Phase 2 clinical trial in patients with Fragile X syndrome will be initiated in the second half of 2016.
ZYN001, a prodrug of THC that enables transdermal delivery through the skin and circulatory system via a patch, is in preclinical development. A Phase 1 clinical trial is planned in the first half of 2017.
In August 2015, Zynerba completed an initial public offering, raising net proceeds of $42.1 million. As of June 30, 2016, cash and cash equivalents totaled $32.1 million, which is projected to fund five Phase 2 clinical trials through 2017.
| EPS (TTM) | 9/30/2016 | -2.47 |
|---|---|
| P/E Ratio | 9/30/2016 | -- |
| Market Cap | Micro Cap | 146M |
| Shares Outstanding | 9.95M |
| Float | 7.2M |
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
