Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Just wanted to come back a sec to your astute observation
I hope you realize that most hear realize the next batch of trial results is what matters,not who is buying or selling at this point,or "dumping" as you so elequently put it.
The heavy lifting has been done here.This horse is well past the 4th turn and headed down the stretch for the line...
My bet is on ZYNERBA.....if I'm wrong......I lose,if I'm right....well...…………aint enough mint for all the julips Ill have.
Watch for news tomorrow on C virus from Emory University.
I know,and it's given me another chance to buy a few more shares.At
what I consider a very discounted rate...OH YEA,
AJIMO.
LOL
Some pretty large dumps/sells last few days..
Back above 4 in ah,I'm done with trying to figure this out.
Before I hang up
I had a call several years ago from some guy named Gaske,or Gatetttes,or something like that,he was telling me about an idea concerning some kind of small home machinehe he was working on in his garage with a friend,a very small computer that he thought could replace machines that took up 1000 of sq ft and make it fit into a size that sat on top of your desk .
Anyone ever hear anything on that ?
Hello
This is Doc. Shuebgtsawcvqda,Im with the universal xyz company,and
was wondering if someone with your company would perhaps be interested in or cbd drug that cures every thing......we just need help with our method of geting or cbd into the blood stream without so many bad side effects.
Please touch base with me if yoU are interested...
This could be a multi billion dollar venture in the short term perhaps,and most of all save millions ,and help millions of peope suffering.
TIA
This thought
and it's certanly not an original one on my part.
The fact that the CBD beast has been released into the field of medicine world wide …(SOME COUNTRIES A TAD SLOW TO REALIZE THIS FACT) ,but I degress
is that what Zenerba may have as a means of transporting cbd into the human body with such minimal side effects that may help so many compounds of cbd delivery into the human body. 'TRANSDERMAL'...
Now I realize the 'PATCH' has been around a while ,but the dose method and other patented aplications Zanerba gives one reason to pause,and consider...
At this point in this companies life I don't really need to be pumping,we are around the fourth turn now,headed for the home stretch.....
MY BET IS IN!
Lumpy, any thoughts on any idea as to the big volume day yesterday,I see we are up again today with vol.,but not as much as yesterdays.
Im wondering if it was a cannabis sector thing as a whole being up,or something else.
I wanted to add my thanks to all the people that has been selling into this run and keeping this stock at a great price this last week or so. I was able to help my Grand Daughter, my wife and a friend load up his Roth. Thanks again and I need just one more small dip back to $4.10 to round off my own bundle...GLTA Bo
OK,NOW...Before I log out,and behave myself
I just want to personally thank all those who sold out here
with the pr of last trial results and the blitz of BS ,IMO that followed,and gave me and others this gift that has been avalible for a while now to buy shares at such a HUGE discount...…….T/Y...T/Y...T/Y KISS KISS BIG HUG!!!!! ;)
BAZARROW WORLD'S ABOUT TO BE FIXED!!!!~
See yah at 15...…….then we will talk about 100
Is there anyone out there who wants to
sell me their 2 or 3 buck shares now over 4 bucks who may have bought at 15?..and filled a law suit or sold out into the BS??????
CHUCKLE
Re: mwhite57 post# 2251
0
Post # of 2305
I'm hopping that's the question
people are asking themselves soon.
Whyyyyyyy,why didn't I buy down here.
Yes. I’m liking it.
Have passed daily vol. avg in less than 1 hour into trading.
Cant find any news...
This train sure looks to be steady on the #4 track,for now anyway.
ALL ABOARD!!!!
This is who I suspect being behind the pull back for such a long period of time.
https://images.search.yahoo.com/yhs/search?p=SKULL+DUGURIE&fr=yhs-omr-001&hspart=omr&hsimp=yhs-001&imgurl=http%3A%2F%2Fimages4.fanpop.com%2Fimage%2Fphotos%2F21100000%2FSkull-skullduggery-pleasant-21177136-720-540.jpg#id=2&iurl=http%3A%2F%2Fimages4.fanpop.com%2Fimage%2Fphotos%2F21100000%2FSkull-skullduggery-pleasant-21177136-720-540.jpg&action=click
OH YEA...JMO
I also did read the data as positive. But I thought that I remembered the company providing additional further explanation that the occurrence of SAE were common and wasn't unusual. I think that the wording of "serious SAE" were key words that caused the confusion IMO.
My bad, you are right - I was thinking of the 10 subjects that reported serious SAE reported for Phase 2 DEE in Sept 2019, not the most recent reporting of data.
Here is the actual PR.Who in their right mind would not consider this as positive???
https://zynerba.com/zynerba-pharmaceuticals-announces-positive-top-line-data-from-believe-1-open-label-phase-2-study-of-zygel-in-developmental-and-epileptic-encephalopathies-dee/
ONLY IN BAZARROW WORLD.
Not sure I buy that explanation.
I'm about as average joe as they get,Im no expert analyst,or a Doctor,but I know this,as soon as I read the pr on that trial result I knew it was positive,if I recall it even said that in the pr.....positive results..
Secondly the very next day this analyst,who I do consider an expert wrote this...https://thefly.com/landingPageNews.php?id=2965581&headline=ZYNE;GWPH-Zynerba-selloff-on-openlabel-data-for-Zygel-unwarranted-says-HC-Wainwright
I read several other write ups that said the same.I find it hard to swallow that the reason this sold off as it did was lack of investors,or analyst understanding the data.it was crystal clear.
I referred to it as the BAZARROW WORLD SYNDROME...Im hopping that soon
no matter what caused this sell of it turns out to have been a huge blessing,and a market bargain of enormous size
I believe that the previous write up of the results of the side effects were too technical To understand and thus it was misinterpreted by the analysts and investors. Even though they tried to explain that it was actually a good result and not abnormal, the damage had been done and some of the investors must have lost interest in the stock and moved on. ZYNE will have to prove itself again and I am sure that this time, they will be more careful how they release their data so the average joe will not misunderstand the results this time. I think that hardly anyone looks for retractions or follow up press releases after the damage has been done. No matter, good results in Late May and late June will have get this stock rolling fast IMO.
"Cautiously hopeful for favorable results since the prior study was so stellar."
Aint that the truth,still shaking my head over last trial results,and how the "market" reacted...
ZYNE is Fairly stable at this point. Thought I heard from the presentation that some Phase 2 data for one of their products will be reported earlier than the Phase 3 FX. Looks like it will bounce between 3.5-4 until the next set of news. Heard that that GWPH got good news for epilepsy product. Hope this doesn't deter from ZYNE product. Cautiously hopeful for favorable results since the prior study was so stellar.
Not sure why shorts won’t stay away. https://fintel.io/ss/us/zyne Especially after such a fantastic company update at Needham. Everyone should listen to it if you can. Here are the slides.
http://ir.zynerba.com/static-files/701d035a-2839-49a8-9251-58438c0b29f6
There is a lot of info out there on this,regarding your question.
Here is just a tad.
https://thefly.com/landingPageNews.php?id=2965581&headline=ZYNE;GWPH-Zynerba-selloff-on-openlabel-data-for-Zygel-unwarranted-says-HC-Wainwright
OH YES,and ZYNERBAS has much fewer side effects,such as liver issues.
Do you think gwph getting the approval helps us here? Different product but that is why I am over here
This is just a hunch,but I think we get news before the month is out on DEES,and FDA meeting date.
Hope so. We should see green days ahead
Just looked at the slide show,everythings looking good,and on schedule.FDA meeting news could come any day on DEES,ballance sheet looks great ,low float ,cash till well into next year,no debt
Wonder if there will be a Q and A today?
ALL SYSTEMS GOOOOO!
Looking forward to tomorrows trading.With the presentation starting at 2:50 ET will give the market plenty of time to digest.
Speculating on the pre market this morning.
I can only attribute it to Wed.up coming company presentation.
Any thoughts?
To all hear,and all envolved with Zynerba,Doctors,patients,family.
Happy Easter
That’s sounds very positive to me!
A very early positive outcome of the CONNECT FX TRIAL is the fact so many have chosen to continue on with open label extension.
This tells me things are looking good,and end results will be good overall.
All IMO
"Robust Enrollment Continues into Open Label Extension Study
During the screening phase of CONNECT-FX, caregivers of patients in the trial were informed that their participating child may have the opportunity to receive Zygel in an open label extension trial following the child’s compliant completion of CONNECT-FX, regardless of their child’s perceived response or actual blinded drug assignment at randomization in CONNECT-FX. As of March 9, 2020, 97% of the 163 patients who have completed the 14-week blinded portion of the CONNECT-FX trial have enrolled in the open label extension trial."
Thanks dhbuzz. Appreciated.
|
Followers
|
91
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
2676
|
|
Created
|
08/06/15
|
Type
|
Free
|
| Moderators | |||

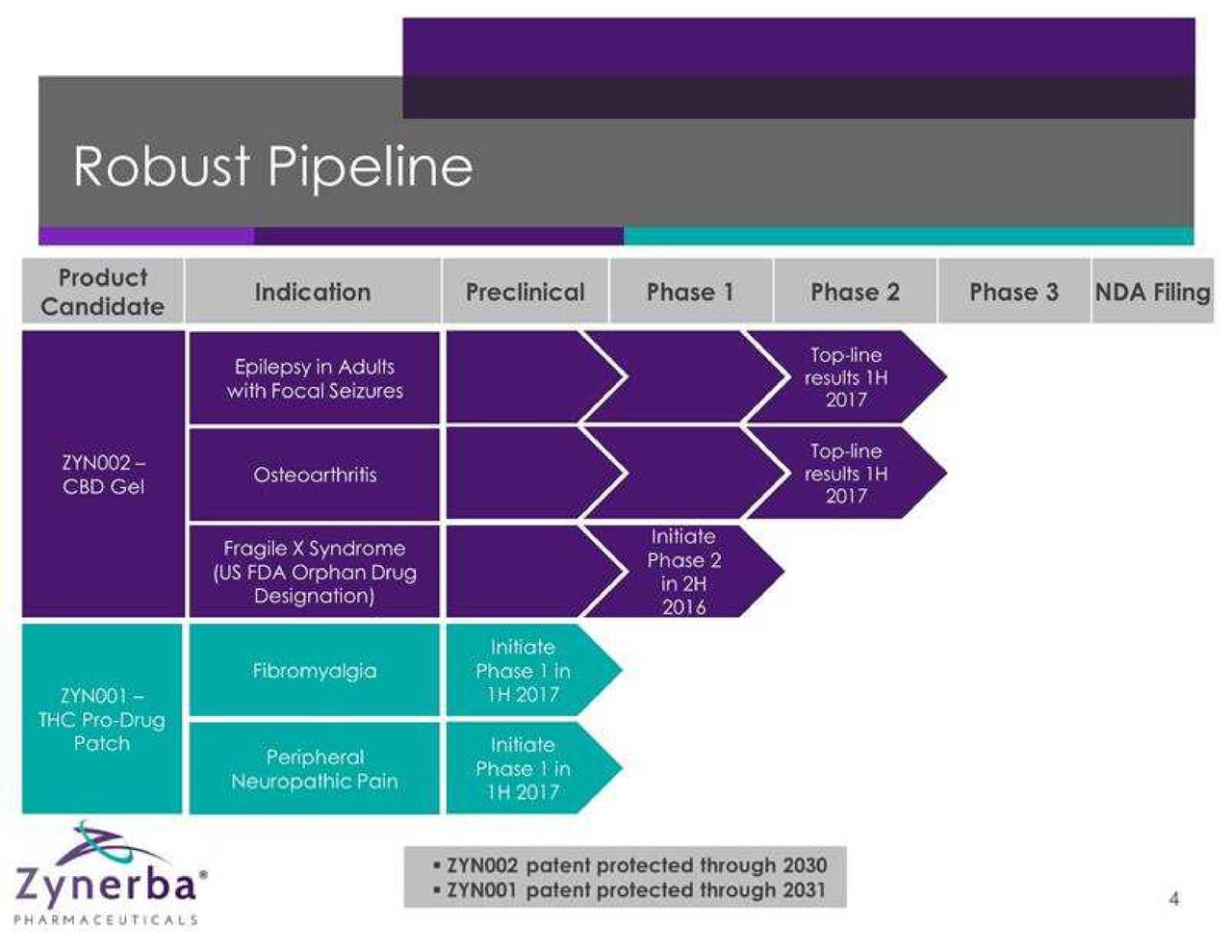
Zynerba Pharmaceuticals, Inc. is a specialty pharmaceutical company focused on developing and commercializing synthetic cannabinoid therapeutics formulated for transdermal delivery. The Company is evaluating approximately two product candidates, ZYN002 and ZYN001, in over five indications. The Company intends to study ZYN002 in patients with refractory epilepsy, osteoarthritis and Fragile X syndrome. The Company's ZYN002 is synthetic cannabidiol (CBD) formulated as a permeation-enhanced gel for transdermal delivery. ZYN002 is being developed as a clear that is designed to provide controlled drug delivery with once- or twice-daily dosing. ZYN001 is a pro-drug of tetrahydrocannabinol (THC) that enables transdermal delivery through a patch. The Company intends to test the ZYN001 patch for application to the arm, back and thigh. The Company intends to study ZYN001 in patients with fibromyalgia and peripheral neuropathic pain.
| Corporate Profile |
Zynerba (NASDAQ: ZYNE) is pioneering the development of patent-protected, next-generation synthetic cannabinoid therapeutics formulated for transdermal delivery. Its two lead product candidates in development include ZYN002 and ZYN001, which are being evaluated in five indications. ZYN002 is the first and only synthetic cannabidiol (CBD) formulated as a permeation-enhanced gel for transdermal delivery. In June 2016, the company initiated the STAR 1 Phase 2 clinical trial in refractory epilepsy patients and in August 2016, initiated the STOP Phase 2 clinical trial in patients with osteoarthritis of the knee. A Phase 2 clinical trial in patients with Fragile X syndrome will be initiated in the second half of 2016.
ZYN001, a prodrug of THC that enables transdermal delivery through the skin and circulatory system via a patch, is in preclinical development. A Phase 1 clinical trial is planned in the first half of 2017.
In August 2015, Zynerba completed an initial public offering, raising net proceeds of $42.1 million. As of June 30, 2016, cash and cash equivalents totaled $32.1 million, which is projected to fund five Phase 2 clinical trials through 2017.
| EPS (TTM) | 9/30/2016 | -2.47 |
|---|---|
| P/E Ratio | 9/30/2016 | -- |
| Market Cap | Micro Cap | 146M |
| Shares Outstanding | 9.95M |
| Float | 7.2M |
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
