Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Agreed. Invariably, someone here will claim this to be a buying opportunity. Been there done that. But when do we get the selling opportunity?? When does our CEO create shareholder value which increases buying interest in the stock which increases the price so that we can capitalize on these so-called buying opportunities?? When you own large blocks of shares, it is hard to flip for small blips in price.
At least .01 with over 1 million in cash and monthly income, no way we should be at these levels.
The price action is infuriating!!! Does Otiko give a sh** at all??
Correct....not sure what he is trying to debate with you.....
One last attempt:
SEC sets due dates for reporting. CEOs do not always meet them. VDRM has very spotty record, but has been doing better last two issuances.
2023 Annual Report (1/1/23-12/31/23) was issued on time on 4/1/24.
1Q24 Report (1/1/24-3/31/24) was issued two days early on 5/13/24.
(see OTCM screenshot below)
In no way, shape or form is a financial report due on last day of a reporting period, in the upcoming case, June 30th, for 2Q24 (4/1/24-6/30/24), nor will a CEO issue a financial report on the last day of the reporting period, again, in the upcoming case June 30th, as was stated in Post #73,517.

We are talking 2 different things in here. Not sure what you mean with falsely equated!! I do write down every time OTIKO submit such and it does match OTCM. You can open his quarterly fillings, dates also match my data. Facts.
Yes, SEC has their general agenda but that’s far from reality in some real scenarios. ViaDerma is proof of such. His delays and history of submitting such. I am only talking about VDRM. Not general SEC data
End of quarter and filing date are being falsely equated.
The end of the quarter is June 30th.
The filing due date per SEC requirements is, as stated, August 14th, with possible 15 Day Extension with Filing of NT 10-K. This information is from the SEC's EDGAR system, as per following link, that was also included at the end of the original reply:
https://edgarsolutions.com/resources/sec-filing-calendar/
Hopefully, he will not be late filing this quarter.
Not sure where did you get that from… That’s general scenario expected from the SEC, in a perfect word… I do follow the Doc patterns and historical submissions on the filings
https://www.otcmarkets.com/otcapi/company/financial-report/394787/content
For the period ending of June 30 2023
So….. Same period on June 30 2024
He does March, then June, then September and so On, check his history filings please
OTIKO has done in like I explained. It is all in OTCM
2nd Qtr 10-K is not due to August 14, with possible 15 Day Extension with Filing of NT 10-K.

https://edgarsolutions.com/resources/sec-filing-calendar/
2nd Quarter filing Due June 30th, we’ll see what he got by then
I could not agree more. My point is Dr. O is aware of the share holders complaints.
Right or wrong he will do it his way. He is the shot caller not the S/H's.
He will stay within his legal limits as this is not his first rodeo.
Long story short we wait.
Ohh yeah, not doubt about it. He’s a good dancer
Unofficial Board Procedure For A Few:
When you have NOT ONE fact to support your contrary position, childish emojis are the ONLY response alternative, since factual, intelligent debate is not a remotely possible option.
I, on the other hand, can defend my contention with those pesky things called facts.
If, and ONLY if, what he is doing has nothing to do with his actual job
(fiduciary responsibility to shareholders;
growing sales and company;
growing shareholder value;
forthright and timely communications with shareholders through financial reporting and press releases and investor relations,
etc.).
I don’t think he will expose himself in such way. Also I don’t see the benefit of a special meeting at these levels. We are in the sucking boat already, our luck won’t change just because we accomplish to meet up with him. He will probably control the flow of questioning and as a good dancer, he will spin all of us with beautiful words. What I want is more fillings with more Intel and less blah blah blah. PRs and CCs are Nothing, he can say and promise whatever he wants in such. Total waste of time. Don’t believe me? Check RGBP and other OTC companies conferences. The more the CEO talked, the worst it got
It is a good speculation but this OTIKO is a broken butt. I don’t think he is buying
AVGO,ADBE,NVDA,AAPL Money making machine
Dr.Otiko must be buying at low, the way to make money, once he announce the product sales numbers stock will shoot up 0.02
It would be beautiful if he does such as is but you know he will never go for it. He will go south
Hasta la vista, baby!
Agree with you fully on this one. Set up an X space or zoom call with otiko would be great since the pr guy is useless. Think these are the only emails that seem to get through to him that are in old PRs and on this board. drotiko@yahoo.com and chris@viaderma.com
CEO is either in it to win it or just needs to step down and bring in a pro CEO to run this thing. Enough is enough.
Here are the beginning steps of the CEO corrective action plan, for remaining 2Q24, since yet another quarter has been largely squandered, without any improved shareholder value, AGAIN:
1) Now that he returned the ridiculous $700K personal loan (assuming 2Q24 financial report confirms), for the phantom “expenses”, supposedly paid by him, that are not backed up by the Balance Sheet, he should NEVER mention the phantom expenses again.
2) Return the extra 95.5 Million shares he gifted to himself (or grifted for himself) in 03/28/23 transaction. He was legitimately owed 4.5 Million for compensation principal and interest ($118K), but nothing else, especially when his near-total lack of performance and return for shareholders is factored in.
3) Revise the financial report section 4) Issuer’s Business, Products and Services, and any other affected sections, as per regulatory requirements and fiduciary responsibility to shareholders, to document what is ACTUALLY going on in the company, the little that there is, and not be insultingly, infuriatingly, and totally 3-7 years out of date.
4) Announce officially (correctly this time) where the state licensing stands. Is it ONLY 10 states as per the financial reports for the last four quarters or is it 11 states as per 3/2/23 PR, and then 14 states as per 10/24/23 PR? Then re-issue financial reports or issue clarifying PR, as required to correct the record or define current status.
5) Renounce that totally UNDESERVED, INSULTING employment agreement clause he gifted himself with (bonus of 30% of any revenue generated from contracts he brings into the company for the first $1,000,000 in any fiscal year, or a bonus of 40% of any revenue generated from contract he brings into the company over $2,000,000 in any fiscal year (NOT THAT HE CAN CLOSE A DEAL ANYWAY)), for basically just doing his job, which he is already compensated for.
6) Commit to only rewarding himself for actual business results for the company and shareholders, and in an amount commensurate with reasonable and customary (like the following clause: If the stock price reaches $1.00, CEO shall receive an additional one-time bonus of 20,000,000 shares of common stock.)
7) Commit to timely, accurate, complete reporting of information to shareholders, via financial reports and PRs, including close out of failed ventures.
8) Find competent sales people POST HASTE and FINALLY begin to close deals and add SIGNIFICANT value for shareholders, instead of pumping “imminent deals” that never happen, and are never heard from again, as he has been doing for more than half a decade.
9) Do his job for the company and shareholders!
It's time for shareholders to call a special meeting and the shareholders and grill the CEO. If he fails to attend the meeting, I'll make a motion to terminate him.
Naah, this time he got anxiety and bent on his knees, mate. He didn’t bank this time. I know ORCA, he would be showing off his success all over. Wherever he is, his knees shall be shaking
Uh huh.....Orca made bank flipping..
Dude, he probably got a few cousins that just arrived to the US that will be in his payroll. You will not see a strong name added. Just 2 kids that will take notes of whatever he says. IT WAS A PR made on him... If he post such on fillings and approved by attorney letter, then I WILL BELIEVE IT, otherwise, it can go 100 days, it will not cause any impact!!! One thing is certain: He will pay those 2 with more dilution, at our expense
I know of one: The influence of ORCA getting addicted to acid and sending him to E.R.
The Doc landed that on his hands, the poor ORCA health is done because of VDRM
LMAOOOO HAHAHAHAHAHHAHA, ohh gosh, I did turn red of laughing
Not sure but I understand your point. However, business is business... It wont affect the contract they have on the license deals, I would not worry on that side. One thing they are associated and another is getting a contract on a product
Very accurate post! Says it all really. Facts are facts.
Sure sloppy. Show us ONE PR that has ever materialized. Just ONE.
Nupelo?
CBD pain relief product?
OTC Uplisting?
Dubai deal?
VA deal?
HMO deal?
Hospital deals?
Wholesale reseller deals?
Overseas shipments in March?
50 states licensing deals?
Any US sales?
Anything?
Bueller?
Not one to date has ever materialized. Don't hold your breathe sloppy.
We will see if the last PR is true within 30 days.
With new staff working on sales and the Dr. having more time
to focus on the company things should start to happen soon.
Just my thoughts.
They might still be paying for licensing the product, however everything with Viaderma is a big ass TBD. We'll only see in the filings which are almost always late.
$1 million on the books? Any inventory on the books? International sales on the books? New state licensing deals on the books? Anything new?
That's what needs to be addressed and fast!
Well this sucks, wasn't wound pros paying the so called license deals?
Interesting your findings on Dr. Chris Otiko is no longer employed with Wound Pros. ViaDerma took lots of pride with the association. Anyways, I am not surprised either, non-profit organizations don’t take well on the long road people with such questionable reputation as the Dr describes in his own BIO, even if he got exonerated from such federal situation. I am really concerned and if some of you are willing to team up to open an investigation case, count with me as well. I am ready to lawyer myself up and begin to knock the SEC’s doorbell over this possible Ponzi set up.
Sad because he got something good going in here but that is what the greed built in Men drive you to do….
If he is natty, he will soon learn that it is not what you don’t know that get you in trouble but that you think it is but really isn’t!!
I wouldn't call them smoke signals. Shareholders are genuinely concerned about their investments in this company and they really should be.
Now Otiko dilutes another 40 million shares. No reason given by him or the useless PR guy either. We're far past the point of being patient enough to see results or have any inclination to what they are actually doing with the business. Still no mentions about Nupelo, Dubai deal, or all of those overseas shipments in March that they PR'd and tweeted about. No inventory on their books either while Otiko gives himself $700,000 but now claims the company is in its best place financially? No one believes this BS. Where are all the sales for the strongest topical antibiotic in the world?
At this point, it will likely take a class action lawsuit or filing multiple SEC complaints to uncover what they are actually doing with an investigation and discovery.
email cotiko@thewoundpros.com and get this response.
Thank you for your email. Dr. Chris Otiko is no longer employed with Wound Pros. Your email will be directed to the appropriate Wound Pros department for prompt handling. If you need immediate assistance, please contact Dr. Bill Releford at DrBill@thewoundpros.com or Attorney Keith Greer at KGreer@thewoundpros.com.
What is going on here? Isn't he the cofounder? He most likely got fired or maybe, just maybe he's going to fully focus on growing Viaderma now. Doubtful that is the case but a very small chance this might be it.
Dr will increase the A/S next. If he can’t dilute and get free product, this company is worthless to him
"When $99 . . . 50% price increase, is baffling" . . . Stupid things happen when dumb people have an idea . . . someone here in management must have had a thought
When $99 price point is ONLY generating from <$100 to <$700 in gross profit per quarter, per the clause below, over last six quarters, since restart of online sales, and after all of Intent Sciences promotion, even consideration of price increase, much less 50% price increase, is baffling.
On January 1, 2017, the Company entered into a licensing and distribution agreement with Biogenx, Inc. for the purpose of commercializing and distributing a topical antibiotic product to be branded VitaStem. On August 1, 2018, Biogenx, Inc. changed its legal entity name as Viaderma Distribution, Inc. The product will carry the Company’s tetracycline-based technology. This product will be separately registered with the FDA. Pursuant to the agreement, the Company will receive 50% of gross profit from sales of Vitastem. For purposes of the agreement, gross profit is defined as total revenues less cost of production, distribution and marketing. In addition ViaDerma will receive an additional 5% of gross sales as a licensing fee. The agreement was terminate on Marche 31, 2024 unless extended by both parties. Viaderma Distribution, Inc. (Formerly Biogenx, Inc.) has the right to terminate the agreement early with two month notice if it deems the arrangement to not be financially viable. This agreement was renewed on January 1, 2023.
So he can make an extra $50 per bottle, why else
And why all of a sudden did he increase the price per bottle to $149??? He didn’t even mention anything on that front in the last PR!
Only reason In my own opinion that Dr Otiko finally did a PR the other day was because of the “Smoke Signals” he probably was seeing of sick and tired investors who were speaking about going to file a complaint against him and the company for that whole Bullshit loan and definition of it in the quarterly.
Even the PR stinks to the high heavens as Bullshit. He probably came up with this “hiring” employees so that he can burn up the $1 million in cash the company is supposed to have on hand!!!!
I won’t believe anything further until I see concrete proof.
Disappointing, whereas this will likely turn trickle of product sales revenue into a drip.
Ohh my gosh, what is all this $HIT over me? Did the $hit hit the Fan? Yes it did because it spread all over the room. You are all ViaDerma dirty. Hahahahahahaha
Relax folks, ViaDerma been here before, better get these cheapies before it moves
|
Followers
|
617
|
Posters
|
|
|
Posts (Today)
|
4
|
Posts (Total)
|
74426
|
|
Created
|
10/27/08
|
Type
|
Free
|
| Moderators | |||

ViaDerma's proprietary transdermal delivery system allows for rapid mass transfer of the pharmaceutical active ingredient across the skin and into the body to provide immediate localized therapy.
The technology allows transfer of chemicals through the stratum corneum (outer layer of skin) with a diffusion constant which is 10,000 times higher than the diffusion constant which characterizes water movement through the stratum corneum.
This enables ViaDerma to pair almost any active ingredient with the technology and provide rapid transport of the medicine right to the site of action.
The first product is a broad spectrum tetracycline-based topical antibiotic is the only antibiotic in the world that kills bacteria both a physical and a chemical mechanism. All known antibiotics (other than ours) primarily use only a chemical mechanism of kill. The physical mechanism of kill is a key feature of what we call Rapid Active Ingredient Delivery System (RAIDS). One result of RAIDS is that tetracycline is carried in higher concentrations, more quickly, to and through the cell walls, where the tetracycline can become more effective than if conventional antibiotics were used. Conventional antibiotics require more time (usually prescribed for 5 to 7 days for best results), whereas our tetracycline-based products usually produce desirable results in 24 hours (or less) because of the RAIDS effect.

A second important result of RAIDS is that the topical antibiotic kills all harmful Gram positive and Gram negative bacteria that have been available for testing. We believe this is the world’s strongest broad-spectrum topical antibiotic available.
The potential commercial impact is immense. Drug developers believe it takes much longer for bacteria to develop drug resistance to a physical kill mechanism. This is because it is relatively easy for bacteria to change their response to a chemical threat, but it takes numerous generations for bacteria to grow a new kind of cell wall structure to respond to a physical threat.
Our novel approach to overcome drug resistance of antibiotics is designed to sustain the effectiveness of antibiotics and other topical drugs for many years. This gives new topical drug products a longer useful lifetime and therefore more commercial value. This technology, when licensed to larger pharmaceutical companies, may provide stronger incentive for the discovery and development of new antimicrobial drugs. In recent years, the dearth of new antibiotics has been largely due to the uncertain new-drug commercial lifetime which is diminished when bacteria develop immunity to that drug.
In addition the technology can be licensed to other companies to convert existing oral drugs to transdermal medications extending the profitably of the existing drug.


We are developing products in the following fields of use;
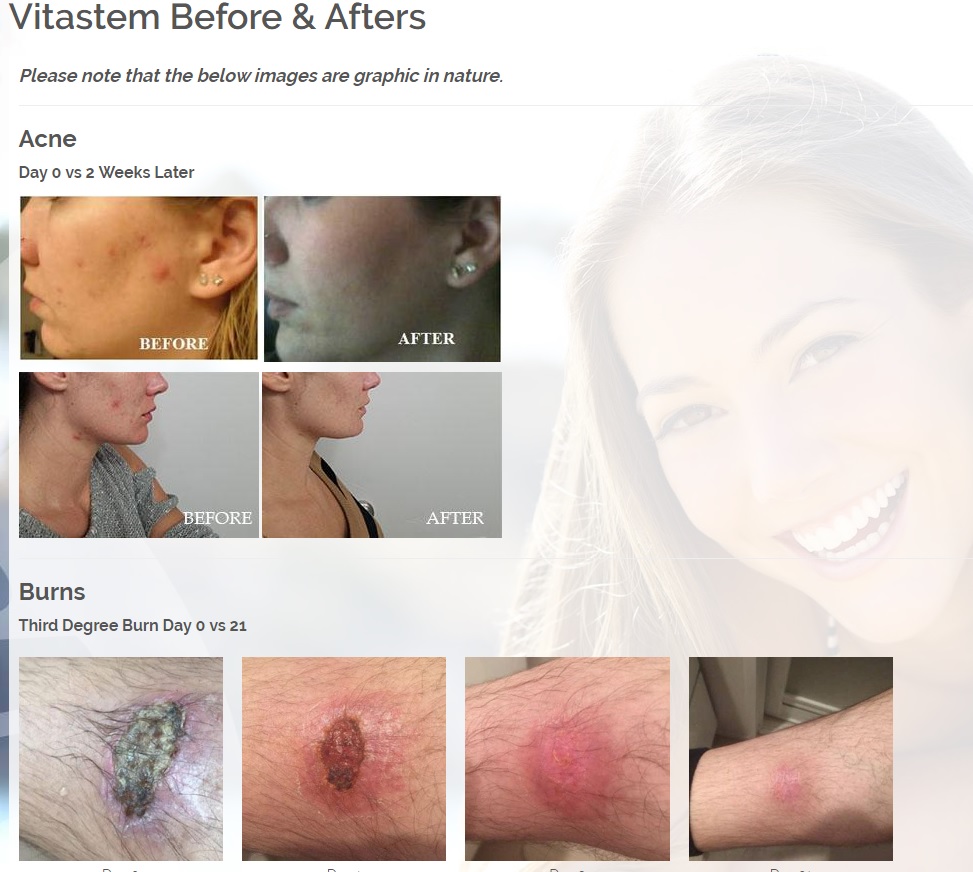
Treats all types of bacteria including MRSA, VRE and other flesh eating bacteria. Fights drug resistant bacteria. $6B/year global market.

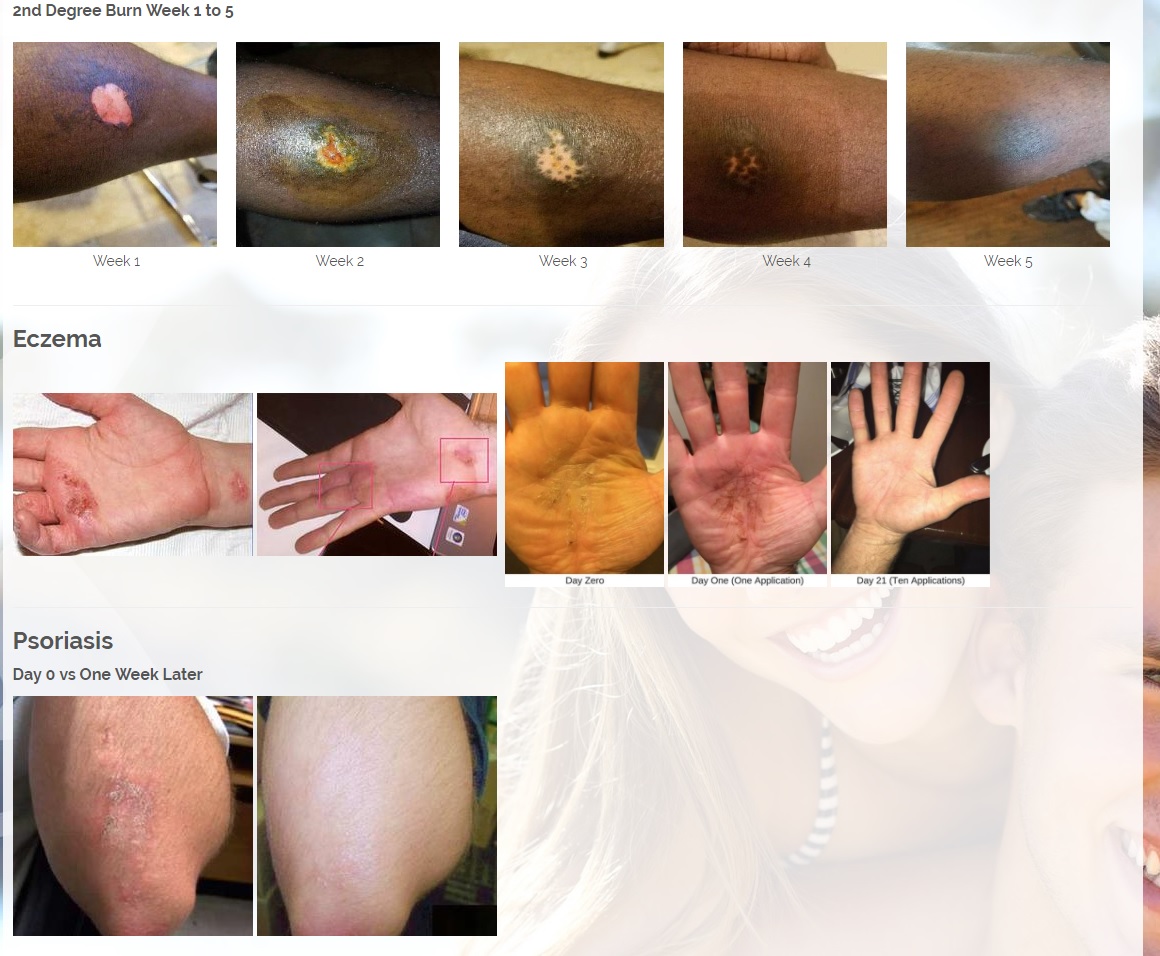
Onychomycosis, a fungal infection of the toenails, is a major health problem. It is estimated that there are in excess of 40 million sufferers with this condition in the USA. It is a problem throughout the world. A recent European study showed that the prevalence of Onychomycosis may be as high as 26.9%. Fungal resistance can occur when the oral antifungal agents are used on a long-term basis. Topically applied antifungal drugs may work somewhat better adjunctive to surgical removal or chemical dissolution of the nail plate. Yet, this often ineffective and traumatic procedure leaves the subject without a nail for months at risk for re-infection. Given the limitations of current treatment options in this $3B market, there is a great need for a simple, nontoxic and effective alternative treatment. Estimated Global prevalence rate: 140 million.
Treats symptoms of Influenza which is a$4B/ year global market: Common
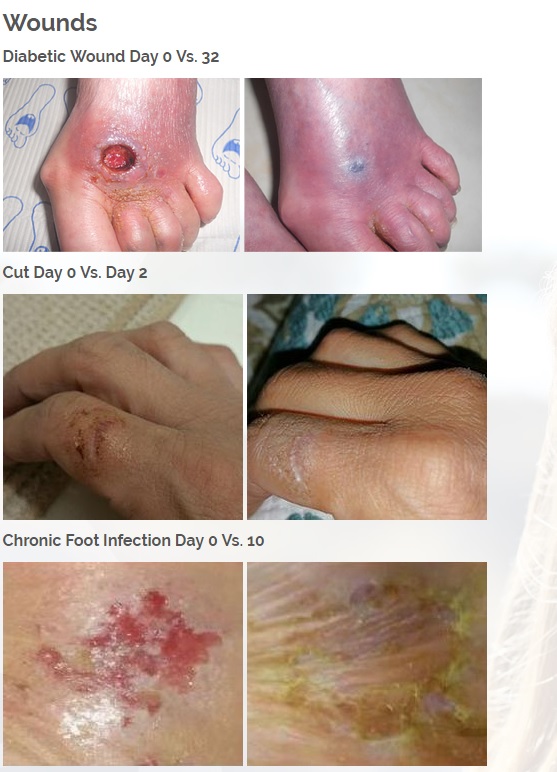
Diabetic foot wounds.

Per 3/8/18 PR: ViaDerma’s technology is currently being used in Elixr Cannabis products; Topical Balm, Topical Serum and Topical Spray. Sales have begun in Canada.
Per 12/7/17 PR: The Company has signed an MOU and will start production of its Patent Pending CBD or Cannabinoids product line. The Company would combine its two Patent pending technologies, the MMJ Patent #62466209, a patent for delivering medical marijuana / cannabis to the body by applying the medication onto the skin in an ointment base topical solution and the second Provisional patent # 62433964 for enhanced antibiotic and drug delivery for "Aqueous Topical Applications" for human and veterinary uses. The Company would combine its proprietary solutions with CBD's and other natural products that treat psoriasis, fibromyalgia for pain and other ailments.
Per 1/23/18 PR: The Company has also a licensing and distribution agreement in place with its Canadian partners to produce a CBD Topical Serum, which has a 92% absorption rate powered by ViaDerma's Patent Pending Dual Carrier Technology. The CBD and Terpene profile (enriched with specific essential oils and vitamins C, and D) is crafted to alleviate Chronic Dry Skin, Psoriasis, Eczema, Rosacea, and many other skin conditions. The serum has anti-inflammatory, anti-bacterial, and chronic pain relief properties that are absorbed by the skin and provide overall healing benefits. Sales are expected to begin in the first quarter of 2018 and are projected to generate approximately $2 million annual revenues.
The Patented Pending CBD or Cannabinoids product line. The Company would combine its two Patent pending technologies, the MMJ Patent, a patent for delivering medical marijuana / cannabis to the body by applying the medication onto the skin in an ointment base topical solution and the second Provisional patent for enhanced antibiotic and drug delivery for "Aqueous Topical Applications" for human and veterinarian uses. The Company would combine its proprietary solutions with CBD's and other natural products that treat psoriasis, fibromyalgia for pain and other ailments. The Company plans to continue to expand its (IP) "Intellectual Property Portfolio" in 2018.

CBD is a compound found in the Cannabis family of plants such as hemp. CBD is combined into our topical body care products like Elixr's Topical Balm, Serum, and Spray.
CBD is an antioxidant. Antioxidants protect our skin from damaging exposure to smoke, UV rays, and environmental pollutants. Cannabinoid receptors are located all throughout the skin, Topical CBD products interact with those receptors resulting homeostasis and healing. Studies show that CBD can help treat an array of skin conditions such as eczema, psoriasis, rosacea, and acne.
| "What is Intellectual Property?" - A Publication by the World Intellectual Property Organization -
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
