Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Hahahahahahaha 😂 it is funny to see some in here losing the patience over these drops . Soo many speculation, incredible the human being. Guys relax, it is not the first time the Doc dilute us at these levels AND lower. Relax, we will be back up in a few weeks… The way this 1Q was made leads to understand ViaDerma got a bigger agenda going On
Wow, the pps is getting ugly. Lows keep getting lower. Why would the Doc build in a graduated bonus structure based on revenue milestones along with a 100,000,000 share award if he didn't want the company to succeed? I still am hard pressed to believe that this has all been a ruse just to fleece the shareholders. On the flip side, ruse or not, the Doc gets a big fat "F" for failure to produce. In the six years or more that I have been here, he has not accomplished one thing, not one. In any normal business, he would have been uncerimoneoulsy fired long ago and escorted from the building.
People lose faith but next thing boom off to the races as we have seen before
I thought my last buy at 83 was golden, oh well. It still will be but hold great number of shares. Cookie jar empty of crazy money.
Wow this is a buyers dream easy double
even a broken clock is right twice a day. i'm gambling the slopster is right this time. my buy order is in.
Exactly! Like when you said we would never see .007….you gotta be tired of being wrong all the time
And you are SURELY one of them 😢 😂
Well he decided to stay current, maybe the next big push it cures ED🤣
Watching for "Red Flags" Revealing Unreliable Microcap Press Releases
Given press release misinformation risks, savvy microcap investors watch for certain "red flags" that may reveal a release contains unreliable claims:
- Announcements lacking specifics or relying on vague claims that would not allow objective independent verification.
- Press releases strangely timed late in the day after markets close or late on Fridays, suggesting an aim to avoid scrutiny.
- Material announcements failing to provide customary supporting evidence like verifiable names of referenced customers or vendors.
- Major claims that seem to directly contradict objective data in the company's actual financial disclosures and filings.
Rich has gone back into his predominant mode of ignoring shareholder's emails. For about a month, I have been pursuing explanation for why official public PR information on state licensing does not match revenue posted in official public financial reports, since beginning of 2023 (definitely not insider information). For example, based on 10/24/23 PR, state licensing revenue should have been $210K (14*$5K*3) for 1Q24, not $150K (10*5K*3). No response is the only "response".
😂🤡🤡sure thing Bobo
Good question.
What we all know now... They did not get to 50 states licensing deals as stated. They did not receive a $1.2 million payment as stated. They have not shipped any products overseas. They have had no news in months and Otiko took out $700k and for what?
Anyone ever get in touch with the elusive IR guy to explain this BS?
Welp my 75s are here… and I don’t want the darn things. Based on recent events, I will not purchase until the 55s. You heard it here first… They are coming…
Hahaha ….I can vouch for that. Guy was PM’ing me some gay shit on Christmas Day one year. Total loser
Any explanation what the loan is for?
Wait, hold on everyone. Why the worry. DB had a 1/2 hour long conversation with IR and he is fine with his investment.
Now, what we need is a good PR from him. That will get us from under water. Afterwards, farewell mates
Yeah, keep it to yourself in the low. One thing is reading and the other yelling out everywhere this is a Nigerian vampire and let’s form a guerrilla group and go to the SEC to repot the dancer! I did read the entire document at the second he dropped it and didn’t say anything. I really don’t want this to end up like QNTA. Lesson learned with the scorpion venom
You went from looking great with this filing to let the scam continue pretty quickly. Reading is fundamental
So a little over half of the Receivables was collected then immediately borrowed? What in the world
I said it before and I-will say it AGAIN. Stop being a HOTHEAD and stop calling out The SEC, scams and other bull$hit… Is the man a Crook? A Scam, a Nigerian dancer? If he is all those: He’s is ours, wether you like it or not, the Doc is our Goldigga, period. Please let the “Scam” to continue its course, for the sake of our positions. Remember what happened to QNTA: A hothead went to the SEC and they listened the moron. Who lost thousands? Mckillean? Heck no, he paid a fine and still running QNTA in the private sector… Shareholders got it in hard and without lube. Be smart and leave the bat to suck his blood quietly…
If you are patient and smart you could get spared and GTFO with your money back
Welcome to OTCM, where the dumb money goes
He did break one undesirable trend, this quarter, at least (being late), but that is all, though.
It is legit, you can’t go to the SEC with such. He could repay such in 10-20 years from now and np. He’s the CEO and he did report the loan. Transparency is required, the man did it it right. I got your point, legitimate point on Otiko but there is not grounds for illegal activity in here, yet.
Thanks very much for catching my omission. I did a word search on his new term, not finding it, and scanned looking for explanation but totally missed it. Thus, the $700K is used in the least desirable way. I, too, remember the previous $300K+ forgiven, after years of promised repayment.
I would have given the report a generous C-, but with "On February 14, 2024, the Company’s CEO borrowed $700,000 from the Company. ", along with continued disconnect between reported states and reported revenue; previous granting of 100M shares for $120K+ debt; previous over-the-top contract bonus provision; and no evidence of product for the much ballyhooed overseas shipping that was to occur in March, but did not, the grade will be a generous D--. Only one person remains the focus of this.
This is proving to be nothing more than your standard Nigerian scam.
It’s stated on the 3rd line on the 2nd to last page which means s page 27 of 28.
You MISSED the biggest takeaway. This fucking Crook Otiko BORROWED $700k from the company. He is probably going to have the $700K forgiven again like the last $300K he borrowed from the company and never repaid. Everyone needs to call the SEC and filing a complaint on this piece of shit thief!!!
No bad Otiko, no bad at all. Looking great with this filling. Hats down to him. I guess I will leave the Doc alone for a while. He is working well and company has proven to have revenue. Not doubts ViaDerma is one of the few left as safe heaven to invest long term. I guess we’ll be fine in here
https://www.otcmarkets.com/otcapi/company/financial-report/402037/content
Nothing on Consolidated Income Statements looks like payment for product production.
Quick Takeaways
Cash increased $367K, not around $1.2M, with $700K of Accounts Receivable appearing to have only been renamed "Receivable from related party", versus actual receipt of back licensing payments ($1.2M payment receipt reported on X on 2/1/24).
Revenue, like all of 2023, appears to reflect 10 state licensing agreements, deviating from reported state agreement counts, with latest being 14 states (as reported in 10/24/23 PR). This should not be related to Dr's over-the-top, personal bonus, contract revenue provision, whereas the total revenue would still be reflected ($210K) partially offset by expense ($63K) for the bonus provision.
Trickle of online sales continues.
Convertible notes remains $323K.
At least a quarterly was on time for the first time in over a year, and Cash increased $350K+.
1Q24 Financial Report has been posted.
https://www.otcmarkets.com/otcapi/company/financial-report/402037/content
I think he's beached
I just want to see the dude that has access to the IR and insider info. I forgot his user name but he called out the filling last time in a timely manner because IR told him such. Why isn’t he coming around this time?
Anyways, does anyone know if ORCA made it from the E.R?
$.0088 I'm expecting something very good very soon! Believe it, don't, I don't give a .....!
Not worried one bit, nor am I whining!
Little less than 20.00
Oh wow share price is in the green today!!!
What is 2,105 shares worth? Like $2?
Market really loves this stock!!!
Where I disagree is him hiring assistants/employees/help. I mean, this guy has done it all completely ALONE. Closed the Dubai deal, Nigeria, etc, all completely by himself!
No need to hire help! He will organically grow this $8.5M company to $100M on his own!
He doesn’t even need CPAs because he doesn’t file FINs, pretty damn efficient!!
Ohh yeah, Dubai and Mexico. He is fighting hard for us. We gotta be paytient… Marijuana is legal now folks, use it… Doc O. Must be stoned now with lots of VDRM logs and drawings all over his granny’s basement now
Viva ViaDerma!!! Note: When $hit hits the fan, please don’t panic, keep smoking weee like the Doc and y’all be fine. LMAOOOO
Ohh my gosh, wait, what’s this ? Mates, I got $hit all over, gosh, the $hit already hit the fan!
OTIKO where at you?
Yeah, I agree, it is super deals time. He probably won’t be capable to sleep either of so many international paper work and deals he has landed already. He will need to hire a few assistants, happy ending assistants, massage therapist assistant, etc. very stressful agenda he might be now. International deals in Dubai, serious business there. Viva ViaDerma
Board took a turn hahaha
Homeboy wants the butt cheeks!!!!
Hahahhahahha
This is gold
Make sense , he may have gone overseas to make atleast 1B deal.
|
Followers
|
619
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
74600
|
|
Created
|
10/27/08
|
Type
|
Free
|
| Moderators | |||

ViaDerma's proprietary transdermal delivery system allows for rapid mass transfer of the pharmaceutical active ingredient across the skin and into the body to provide immediate localized therapy.
The technology allows transfer of chemicals through the stratum corneum (outer layer of skin) with a diffusion constant which is 10,000 times higher than the diffusion constant which characterizes water movement through the stratum corneum.
This enables ViaDerma to pair almost any active ingredient with the technology and provide rapid transport of the medicine right to the site of action.
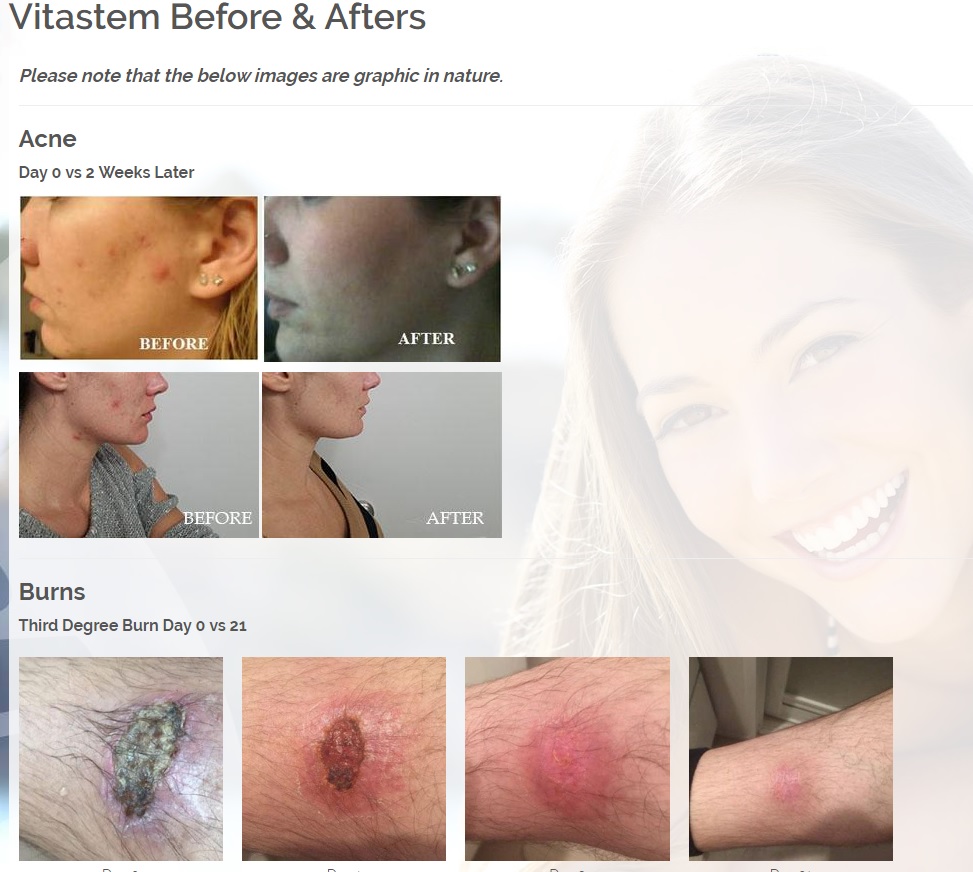
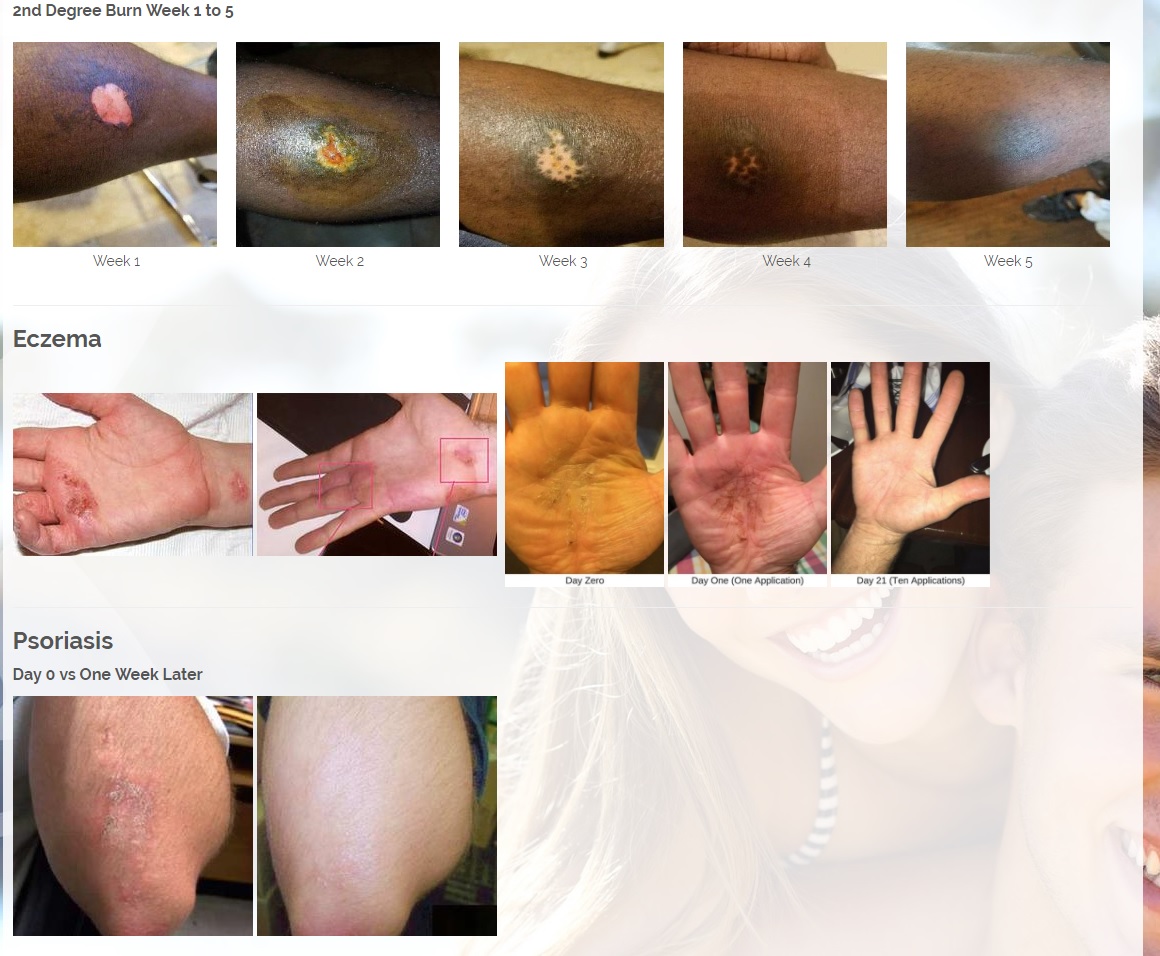
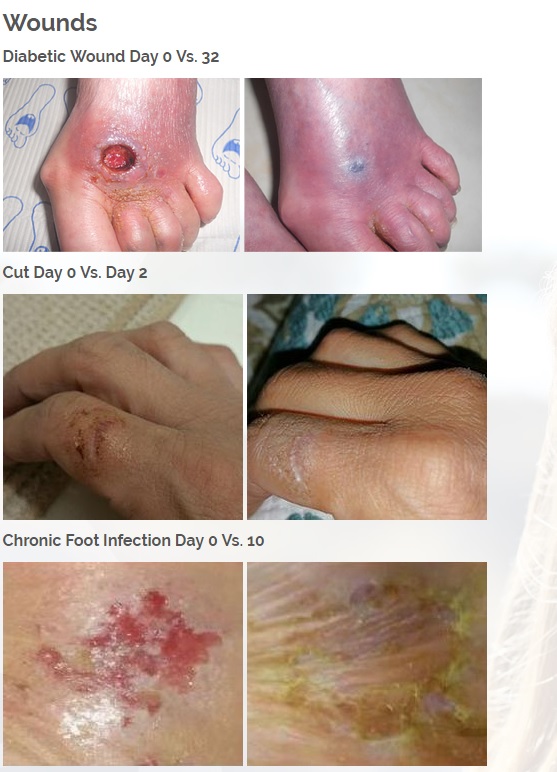
The first product is a broad spectrum tetracycline-based topical antibiotic is the only antibiotic in the world that kills bacteria both a physical and a chemical mechanism. All known antibiotics (other than ours) primarily use only a chemical mechanism of kill. The physical mechanism of kill is a key feature of what we call Rapid Active Ingredient Delivery System (RAIDS). One result of RAIDS is that tetracycline is carried in higher concentrations, more quickly, to and through the cell walls, where the tetracycline can become more effective than if conventional antibiotics were used. Conventional antibiotics require more time (usually prescribed for 5 to 7 days for best results), whereas our tetracycline-based products usually produce desirable results in 24 hours (or less) because of the RAIDS effect.

A second important result of RAIDS is that the topical antibiotic kills all harmful Gram positive and Gram negative bacteria that have been available for testing. We believe this is the world’s strongest broad-spectrum topical antibiotic available.
The potential commercial impact is immense. Drug developers believe it takes much longer for bacteria to develop drug resistance to a physical kill mechanism. This is because it is relatively easy for bacteria to change their response to a chemical threat, but it takes numerous generations for bacteria to grow a new kind of cell wall structure to respond to a physical threat.
Our novel approach to overcome drug resistance of antibiotics is designed to sustain the effectiveness of antibiotics and other topical drugs for many years. This gives new topical drug products a longer useful lifetime and therefore more commercial value. This technology, when licensed to larger pharmaceutical companies, may provide stronger incentive for the discovery and development of new antimicrobial drugs. In recent years, the dearth of new antibiotics has been largely due to the uncertain new-drug commercial lifetime which is diminished when bacteria develop immunity to that drug.
In addition the technology can be licensed to other companies to convert existing oral drugs to transdermal medications extending the profitably of the existing drug.


We are developing products in the following fields of use;
Treats all types of bacteria including MRSA, VRE and other flesh eating bacteria. Fights drug resistant bacteria. $6B/year global market.

Onychomycosis, a fungal infection of the toenails, is a major health problem. It is estimated that there are in excess of 40 million sufferers with this condition in the USA. It is a problem throughout the world. A recent European study showed that the prevalence of Onychomycosis may be as high as 26.9%. Fungal resistance can occur when the oral antifungal agents are used on a long-term basis. Topically applied antifungal drugs may work somewhat better adjunctive to surgical removal or chemical dissolution of the nail plate. Yet, this often ineffective and traumatic procedure leaves the subject without a nail for months at risk for re-infection. Given the limitations of current treatment options in this $3B market, there is a great need for a simple, nontoxic and effective alternative treatment. Estimated Global prevalence rate: 140 million.
Treats symptoms of Influenza which is a$4B/ year global market: Common
Diabetic foot wounds.

Per 3/8/18 PR: ViaDerma’s technology is currently being used in Elixr Cannabis products; Topical Balm, Topical Serum and Topical Spray. Sales have begun in Canada.
Per 12/7/17 PR: The Company has signed an MOU and will start production of its Patent Pending CBD or Cannabinoids product line. The Company would combine its two Patent pending technologies, the MMJ Patent #62466209, a patent for delivering medical marijuana / cannabis to the body by applying the medication onto the skin in an ointment base topical solution and the second Provisional patent # 62433964 for enhanced antibiotic and drug delivery for "Aqueous Topical Applications" for human and veterinary uses. The Company would combine its proprietary solutions with CBD's and other natural products that treat psoriasis, fibromyalgia for pain and other ailments.
Per 1/23/18 PR: The Company has also a licensing and distribution agreement in place with its Canadian partners to produce a CBD Topical Serum, which has a 92% absorption rate powered by ViaDerma's Patent Pending Dual Carrier Technology. The CBD and Terpene profile (enriched with specific essential oils and vitamins C, and D) is crafted to alleviate Chronic Dry Skin, Psoriasis, Eczema, Rosacea, and many other skin conditions. The serum has anti-inflammatory, anti-bacterial, and chronic pain relief properties that are absorbed by the skin and provide overall healing benefits. Sales are expected to begin in the first quarter of 2018 and are projected to generate approximately $2 million annual revenues.
The Patented Pending CBD or Cannabinoids product line. The Company would combine its two Patent pending technologies, the MMJ Patent, a patent for delivering medical marijuana / cannabis to the body by applying the medication onto the skin in an ointment base topical solution and the second Provisional patent for enhanced antibiotic and drug delivery for "Aqueous Topical Applications" for human and veterinarian uses. The Company would combine its proprietary solutions with CBD's and other natural products that treat psoriasis, fibromyalgia for pain and other ailments. The Company plans to continue to expand its (IP) "Intellectual Property Portfolio" in 2018.

CBD is a compound found in the Cannabis family of plants such as hemp. CBD is combined into our topical body care products like Elixr's Topical Balm, Serum, and Spray.
CBD is an antioxidant. Antioxidants protect our skin from damaging exposure to smoke, UV rays, and environmental pollutants. Cannabinoid receptors are located all throughout the skin, Topical CBD products interact with those receptors resulting homeostasis and healing. Studies show that CBD can help treat an array of skin conditions such as eczema, psoriasis, rosacea, and acne.
| "What is Intellectual Property?" - A Publication by the World Intellectual Property Organization -
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
