Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
This had to hurt all the blow hards NO DOUBT
King Liar wants an apology?😂
Fdc4 HB put you in your place with FACTS
Not 1 of you are man enough to apologize
Still have some bait in the waters just in case it dips. :)
Got the right and reason to do so. This CEO is very disrespectful and doesn’t care about shareholders. Or you think otherwise? You know I am right so shut up
You have all made yourselves like EXPERT FOOLS
All of U that have been disresopecting bashing
He just got lucky
HB told was bashed for posting this FACT yesterday
".......THERE WILL BE NO FILING MONDAY." No filing????? Is it Monday, or April FOOL day? I think both!
ORCA
Sunday, March 31, 2024 9:09:41 PM
Post# of 72699 Go
.......THERE WILL BE NO FILING MONDAY.
Finally: Anual report https://www.otcmarkets.com/otcapi/company/financial-report/396282/content
I will end up with strength shirt and mental breakdown due to OTIKO.
I hear you. Beyond frustrating
Guess I’ll pick some up at the 75s lol
I would rather leave these concerns covered, buried. Like the army says: Don’t ask, don’t tell!
I understand your concern but the logic is we can’t force the doc to do anything. What do you exactly mean by forcing?
Mate!!! You have been doing thousands of daily predictions wrong… You are the least indicated to call me out. KHALI!!!
OR - is the unpaid licensing fees (by his other company) HOW we prove fraud?
Serious question here - is there any way we can force the Dr. to collect the licensing fees? I’m talking about the $1M in A/R that’s just sitting there… unpaid…. months and quarters on end…
Relax U know the Dr. always last minute
WHERE IS THE FILING????ARE WE GOING TO EXPERT MARKET???
ZERO SELF REFLECTION!!!
Fdc4U made dozens of predictions Never once right.
Fdc4 Your imagianry source you claimed all BS.
I called it as it is and you know it. FACTS. Now, please proof me wrong right now. Stop supporting Bs speculations
Yeah boy well said
I would have taken that from someone else but not from you Slops. There is not more bullshitter and speculator in here than you. Proven, facts Lmaoo
YOU HAVE YOUR EAR OR YOUR NOSE IN OTIKOS A$$??THERE WILL BE NO FILING MONDAY.
That was really uncalled for and a little over the top. HB has always provided credible intell; unlike most of the gibberish you spew. Be prepared to issue a retraction. HB, thanks for the DD.
Fdc4 The truth is YOU don't know jack sh*t
Fdc4 U have told more lies than everyone on the board
Nope, noo, hell no, that is not going to happen man. Heck not, relax and wait for normal yield sign update with nothing else in it. OTIKO will go MIA for a few months after that and then the new pump idea will come
So, let me guess. The friend of my friend told you that!!! An insider like I had? You spoke to Otiko himself? Waiting, you work for ViaDerma!!! Crap, you are the one with a crystal ball, right? Shit you know the future!!! Wait, do you have access to the sec system from inside? Do you have someone that pay a fee to check such before anyone else? Come share with me only, nobody will know. Lol
Like slop said, thanks for your DD Lmaoo
Maybe we will have some more states paying or money in the bank??? Either one we will be over .03 fast. Any great news like a contract .10 by July ??
Hummingbird2 Thanks for the DD info.
Hearing 10k is done & will be filed on time on Monday.
Hahahahahahhaah darn ORCA, you are on fire now. Arrest the MF!!! I wonder what would you do to the Doc if someone leaves him to you, wrapped with barb wire, seated on a chair, feet in a cylindrical metal container full of water and 2 wires in it fed from a battery truck IN A DARK, REMOTE, HUMID ROOM. So nasty ORCA.... LMAO While dropping baby powder from the roof AND MONKEYS jumping on the bed
I bet you would tattoo DUBAI VDRM on his chest
FAGI FILED ON TIME,JNSH,AND CMGO FILED TODAY FOR NOTIFICATION OF LATE FILING.WHILE OTIKO FILED NOTHING.ARREST THE MF.
Last annual report was done on Mid-April-2023. Historically, we are on schedule.
|
Followers
|
619
|
Posters
|
|
|
Posts (Today)
|
3
|
Posts (Total)
|
74600
|
|
Created
|
10/27/08
|
Type
|
Free
|
| Moderators | |||

ViaDerma's proprietary transdermal delivery system allows for rapid mass transfer of the pharmaceutical active ingredient across the skin and into the body to provide immediate localized therapy.
The technology allows transfer of chemicals through the stratum corneum (outer layer of skin) with a diffusion constant which is 10,000 times higher than the diffusion constant which characterizes water movement through the stratum corneum.
This enables ViaDerma to pair almost any active ingredient with the technology and provide rapid transport of the medicine right to the site of action.
The first product is a broad spectrum tetracycline-based topical antibiotic is the only antibiotic in the world that kills bacteria both a physical and a chemical mechanism. All known antibiotics (other than ours) primarily use only a chemical mechanism of kill. The physical mechanism of kill is a key feature of what we call Rapid Active Ingredient Delivery System (RAIDS). One result of RAIDS is that tetracycline is carried in higher concentrations, more quickly, to and through the cell walls, where the tetracycline can become more effective than if conventional antibiotics were used. Conventional antibiotics require more time (usually prescribed for 5 to 7 days for best results), whereas our tetracycline-based products usually produce desirable results in 24 hours (or less) because of the RAIDS effect.

A second important result of RAIDS is that the topical antibiotic kills all harmful Gram positive and Gram negative bacteria that have been available for testing. We believe this is the world’s strongest broad-spectrum topical antibiotic available.
The potential commercial impact is immense. Drug developers believe it takes much longer for bacteria to develop drug resistance to a physical kill mechanism. This is because it is relatively easy for bacteria to change their response to a chemical threat, but it takes numerous generations for bacteria to grow a new kind of cell wall structure to respond to a physical threat.
Our novel approach to overcome drug resistance of antibiotics is designed to sustain the effectiveness of antibiotics and other topical drugs for many years. This gives new topical drug products a longer useful lifetime and therefore more commercial value. This technology, when licensed to larger pharmaceutical companies, may provide stronger incentive for the discovery and development of new antimicrobial drugs. In recent years, the dearth of new antibiotics has been largely due to the uncertain new-drug commercial lifetime which is diminished when bacteria develop immunity to that drug.
In addition the technology can be licensed to other companies to convert existing oral drugs to transdermal medications extending the profitably of the existing drug.

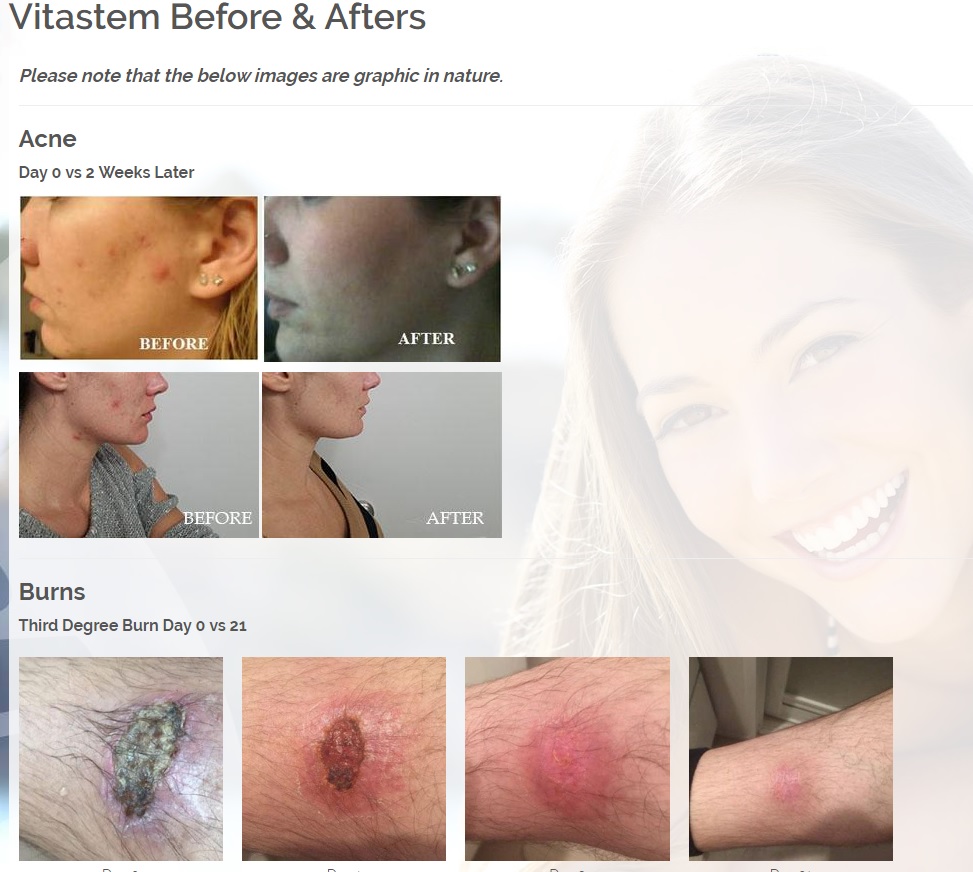
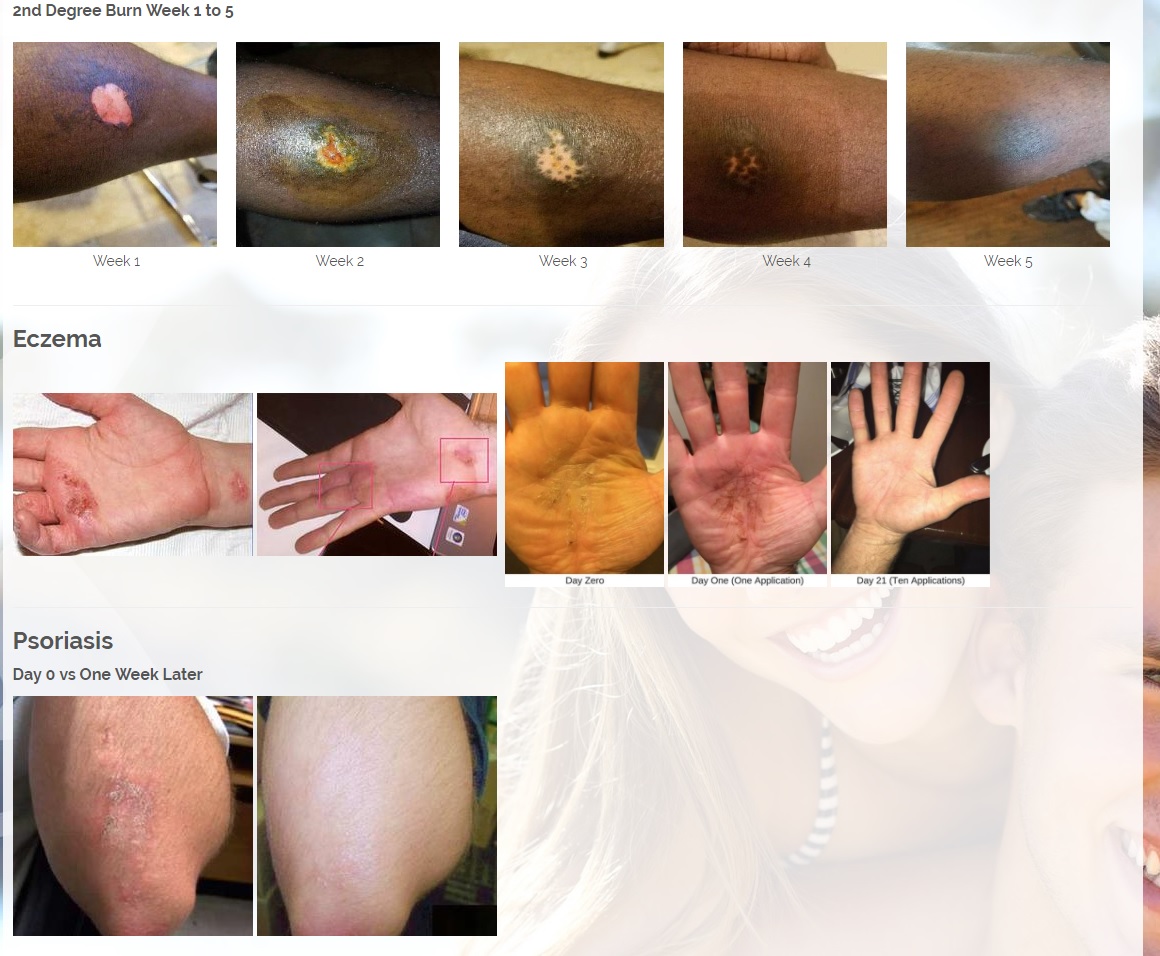
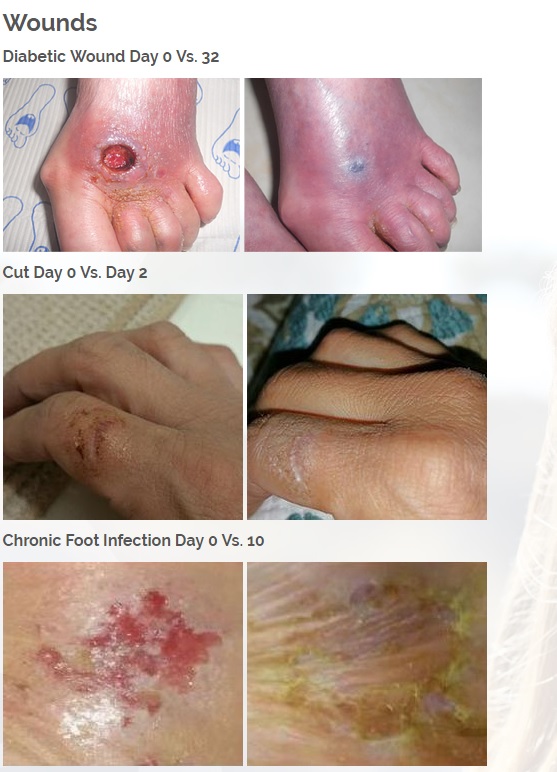

We are developing products in the following fields of use;
Treats all types of bacteria including MRSA, VRE and other flesh eating bacteria. Fights drug resistant bacteria. $6B/year global market.

Onychomycosis, a fungal infection of the toenails, is a major health problem. It is estimated that there are in excess of 40 million sufferers with this condition in the USA. It is a problem throughout the world. A recent European study showed that the prevalence of Onychomycosis may be as high as 26.9%. Fungal resistance can occur when the oral antifungal agents are used on a long-term basis. Topically applied antifungal drugs may work somewhat better adjunctive to surgical removal or chemical dissolution of the nail plate. Yet, this often ineffective and traumatic procedure leaves the subject without a nail for months at risk for re-infection. Given the limitations of current treatment options in this $3B market, there is a great need for a simple, nontoxic and effective alternative treatment. Estimated Global prevalence rate: 140 million.
Treats symptoms of Influenza which is a$4B/ year global market: Common
Diabetic foot wounds.

Per 3/8/18 PR: ViaDerma’s technology is currently being used in Elixr Cannabis products; Topical Balm, Topical Serum and Topical Spray. Sales have begun in Canada.
Per 12/7/17 PR: The Company has signed an MOU and will start production of its Patent Pending CBD or Cannabinoids product line. The Company would combine its two Patent pending technologies, the MMJ Patent #62466209, a patent for delivering medical marijuana / cannabis to the body by applying the medication onto the skin in an ointment base topical solution and the second Provisional patent # 62433964 for enhanced antibiotic and drug delivery for "Aqueous Topical Applications" for human and veterinary uses. The Company would combine its proprietary solutions with CBD's and other natural products that treat psoriasis, fibromyalgia for pain and other ailments.
Per 1/23/18 PR: The Company has also a licensing and distribution agreement in place with its Canadian partners to produce a CBD Topical Serum, which has a 92% absorption rate powered by ViaDerma's Patent Pending Dual Carrier Technology. The CBD and Terpene profile (enriched with specific essential oils and vitamins C, and D) is crafted to alleviate Chronic Dry Skin, Psoriasis, Eczema, Rosacea, and many other skin conditions. The serum has anti-inflammatory, anti-bacterial, and chronic pain relief properties that are absorbed by the skin and provide overall healing benefits. Sales are expected to begin in the first quarter of 2018 and are projected to generate approximately $2 million annual revenues.
The Patented Pending CBD or Cannabinoids product line. The Company would combine its two Patent pending technologies, the MMJ Patent, a patent for delivering medical marijuana / cannabis to the body by applying the medication onto the skin in an ointment base topical solution and the second Provisional patent for enhanced antibiotic and drug delivery for "Aqueous Topical Applications" for human and veterinarian uses. The Company would combine its proprietary solutions with CBD's and other natural products that treat psoriasis, fibromyalgia for pain and other ailments. The Company plans to continue to expand its (IP) "Intellectual Property Portfolio" in 2018.

CBD is a compound found in the Cannabis family of plants such as hemp. CBD is combined into our topical body care products like Elixr's Topical Balm, Serum, and Spray.
CBD is an antioxidant. Antioxidants protect our skin from damaging exposure to smoke, UV rays, and environmental pollutants. Cannabinoid receptors are located all throughout the skin, Topical CBD products interact with those receptors resulting homeostasis and healing. Studies show that CBD can help treat an array of skin conditions such as eczema, psoriasis, rosacea, and acne.
| "What is Intellectual Property?" - A Publication by the World Intellectual Property Organization -
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
