Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
PEOPLE ARE BAILING LIKE I SAID EXPERT MARKET NEXT STOP.OTIKO GOT US.LIKE I PREDICTED.SCAMBAG.
In my opinion, you really mean PR (Public Relations), i.e., Intent Sciences, if the contract is still in place, and, even then, they would need information from management to communicate. On the other hand, IR is quite limited legally in what he can communicate. IR, in my opinion, is still very lax by not answering emails for long periods of time, with, at least "There is no additional information to communicate at this time." or the like.
His previous response was on Jan 31st. He stated "the next 2 Q's are close to being completed." I waited to begin following up until 2/20/24. Sadly, I have found that one must almost be a total nuisance to get a response, thus, I email every couple of business days until I get a response. The excerpt posted was from response received today.
how long ago did you send an email to IR? I can't seem to get a response in a timely manner if at all. funny how IR says he is frustrated at the accountant all well IR is not doing their part with PRs to update its shareholders either.
No, just no interest. Waiting on the Doc to produce....zzzzzzzzzzzzz
Is there a trading halt for VDRM? There are no trades today for the first two hours. Do trading halts get posted somewhere?
Excerpt from today's response, finally, from IR on Overdue Reports:
"My hope is that everything will be caught up to date by the end of this month, which is when the annual is due. I know the accountant is working on it because she's reached out to me about a couple of things. It's very frustrating and I appreciate your patience. I'm right there with you in the frustration department."
I see.....Doc 😉
Great movie by the way 👍
Got that from the movie tombstone.
No thanks. Surely would be a waste of my time. What a random post......
GM I know let's have a spelling contest 😃
Fdc4 Why do you think I mentioned it?????
😢 Already knew this which is why "he mentioned it" yet originally asked why the payment went through Biogenix. Clueless anyone? LMFAOOOOOOOO 🤡 😢
The only issues with the compensation are continuing lack of required information, in a timely manner, to shareholders, and recent past to past's lack of fulfillment of announced objectives, without explanation to shareholders (e.g., Nigeria deal, Medicare/US insurance approvals, and, most grossly, fall through of Dubai deal, after announcing deal and funds in escrow)..
With the above being said though, once $1.2M almost catch up payment of licensing funds is on 1Q24 financial report balance sheet, and 100,000 (or more) units shipments actually begin this month and show up on 1Q24 financial report, all is forgiven and the compensation package will be well worth it.
The below is excerpt from 2023 Annual Report, regarding CEO compensation.
On October 1, 2022, the Company entered into an employment agreement with CEO, the Company’s President and Chief Executive Officer, pursuant to which the Company agreed to issue 20,000,000 shares of common stock as an incentive compensation for each year CEO is employed with the Company. The fair value of these shares
was $0.001 per share and booked the stock-based compensation pro rata within the relative service period. For the year ended December 31, 2022, 5,000,000 shares of common stock are to be issued and the Company recorded $5,000 as stock compensation. The total outstanding accrued compensation as of December 31, 2022 was $30,000.
They are here because they love me.
Ha ha lot of jokers in here. All I see is the same faces allways in here must be reason to be here
Fdc4 Why do you think I mentioned it?????
Sloppy, that intel been in OTCM for several months. Really?
Hey mate! You still got your seatbelt on?😂
I think you are getting into hypothermia stage. You gotta get that checked out again lol.
gameccks1 Thanks for the info.
What are everyone’s thoughts about the Dr.s compensation package?
INTERESTING.SO THEY ARE STILL IN THE BOAT.The agreement was terminate on March 31, 2023 unless extended by both parties. Viaderma Distribution, Inc. (Formerly Biogenx, Inc.) has the right to terminate the agreement early with two month notice if it deems the arrangement to not be financially viable. This agreement was renewed on January 1, 2023.
Excerpt from 1Q23 Financial Report provides explanation:
On January 1, 2017, the Company entered into a licensing and distribution agreement with Biogenx, Inc. for the purpose of commercializing and distributing a topical antibiotic product to be branded VitaStem. On August 1, 2018, Biogenx, Inc. changed its legal entity name as Viaderma Distribution, Inc. The product will carry the Company’s tetracycline-based technology. This product will be separately registered with the FDA. Pursuant to the agreement, the Company will receive 50% of gross profit from sales of Vitastem. For purposes of the agreement, gross profit is defined as total revenues less cost of production, distribution and marketing. In addition ViaDerma will receive an additional 5% of gross sales as a licensing fee. The agreement was terminate on March 31, 2023 unless extended by both parties. Viaderma Distribution, Inc. (Formerly Biogenx, Inc.) has the right to terminate the agreement early with two month notice if it deems the arrangement to not be financially viable. This agreement was renewed on January 1, 2023.
GonjaRelaxation Posted October 19, 2017)
I’m sorry but this whole Biogenix thing seems kinda scammy. Hope I’m wrong
Why? Did you see the dates on what you posted?
Might be about to get hot in here
"ViaDerma's Manufacturer has completed the first run of its topical antibiotic being sold as VitaStem by its Sales, Marketing and Distribution Partner Biogenx, Inc.; Distribution will begin as the product is shipped to Distribution points in Florida and Abroad"
https://www.google.com/url?sa=t&source=web&rct=j&opi=89978449&url=https://finance.yahoo.com/news/viaderma-inc-commences-distribution-vitastem-175713167.html&ved=2ahUKEwjVjOigiNyEAxWhlIkEHSycDsEQFnoECBIQAQ&usg=AOvVaw3_T9Q4YNI2fF1SRwEUB9Lk
"ViaDerma's Manufacturer has completed the first run of its topical antibiotic being sold as VitaStem by its Sales, Marketing and Distribution Partner Biogenx, Inc.; Distribution will begin as the product is shipped to Distribution points in Florida and Abroad"
https://www.google.com/url?sa=t&source=web&rct=j&opi=89978449&url=https://finance.yahoo.com/news/viaderma-inc-commences-distribution-vitastem-175713167.html&ved=2ahUKEwjVjOigiNyEAxWhlIkEHSycDsEQFnoECBIQAQ&usg=AOvVaw3_T9Q4YNI2fF1SRwEUB9Lk
Thanks for sharing, this doesn't really seem good for us.
viaderma office says Biogenx is parent company
Why is Biogenx Inc collecting the $$ 4 ViaDerma?
Read info very interesting when GOOGLE
You authorized a payment of $99.95 USD to Biogenx Inc (info@viaderma.com)
lol Ok 👍 you’re funny that’s why you’re not on the ignore list .
Hold some just in case he’s not a lying piece of shit and it does not go to the expert market then maybe you can even leave a tip after your meal …. Hope we have a run soon we are overdue,
RELOAD TO FLIP AGAIN FOR CHICKEN TRIO:))
HE IS LYING THE DIRT BAG.
Why would you want to reload you’ve been complaining that the CEO is a scumbag piece of $hit and the company is going to the expert market ….. you just like throwing your $ money away 😀😀
amitgupta What report ? where ? post it.
|
Followers
|
619
|
Posters
|
|
|
Posts (Today)
|
3
|
Posts (Total)
|
74600
|
|
Created
|
10/27/08
|
Type
|
Free
|
| Moderators | |||

ViaDerma's proprietary transdermal delivery system allows for rapid mass transfer of the pharmaceutical active ingredient across the skin and into the body to provide immediate localized therapy.
The technology allows transfer of chemicals through the stratum corneum (outer layer of skin) with a diffusion constant which is 10,000 times higher than the diffusion constant which characterizes water movement through the stratum corneum.
This enables ViaDerma to pair almost any active ingredient with the technology and provide rapid transport of the medicine right to the site of action.
The first product is a broad spectrum tetracycline-based topical antibiotic is the only antibiotic in the world that kills bacteria both a physical and a chemical mechanism. All known antibiotics (other than ours) primarily use only a chemical mechanism of kill. The physical mechanism of kill is a key feature of what we call Rapid Active Ingredient Delivery System (RAIDS). One result of RAIDS is that tetracycline is carried in higher concentrations, more quickly, to and through the cell walls, where the tetracycline can become more effective than if conventional antibiotics were used. Conventional antibiotics require more time (usually prescribed for 5 to 7 days for best results), whereas our tetracycline-based products usually produce desirable results in 24 hours (or less) because of the RAIDS effect.

A second important result of RAIDS is that the topical antibiotic kills all harmful Gram positive and Gram negative bacteria that have been available for testing. We believe this is the world’s strongest broad-spectrum topical antibiotic available.
The potential commercial impact is immense. Drug developers believe it takes much longer for bacteria to develop drug resistance to a physical kill mechanism. This is because it is relatively easy for bacteria to change their response to a chemical threat, but it takes numerous generations for bacteria to grow a new kind of cell wall structure to respond to a physical threat.
Our novel approach to overcome drug resistance of antibiotics is designed to sustain the effectiveness of antibiotics and other topical drugs for many years. This gives new topical drug products a longer useful lifetime and therefore more commercial value. This technology, when licensed to larger pharmaceutical companies, may provide stronger incentive for the discovery and development of new antimicrobial drugs. In recent years, the dearth of new antibiotics has been largely due to the uncertain new-drug commercial lifetime which is diminished when bacteria develop immunity to that drug.
In addition the technology can be licensed to other companies to convert existing oral drugs to transdermal medications extending the profitably of the existing drug.

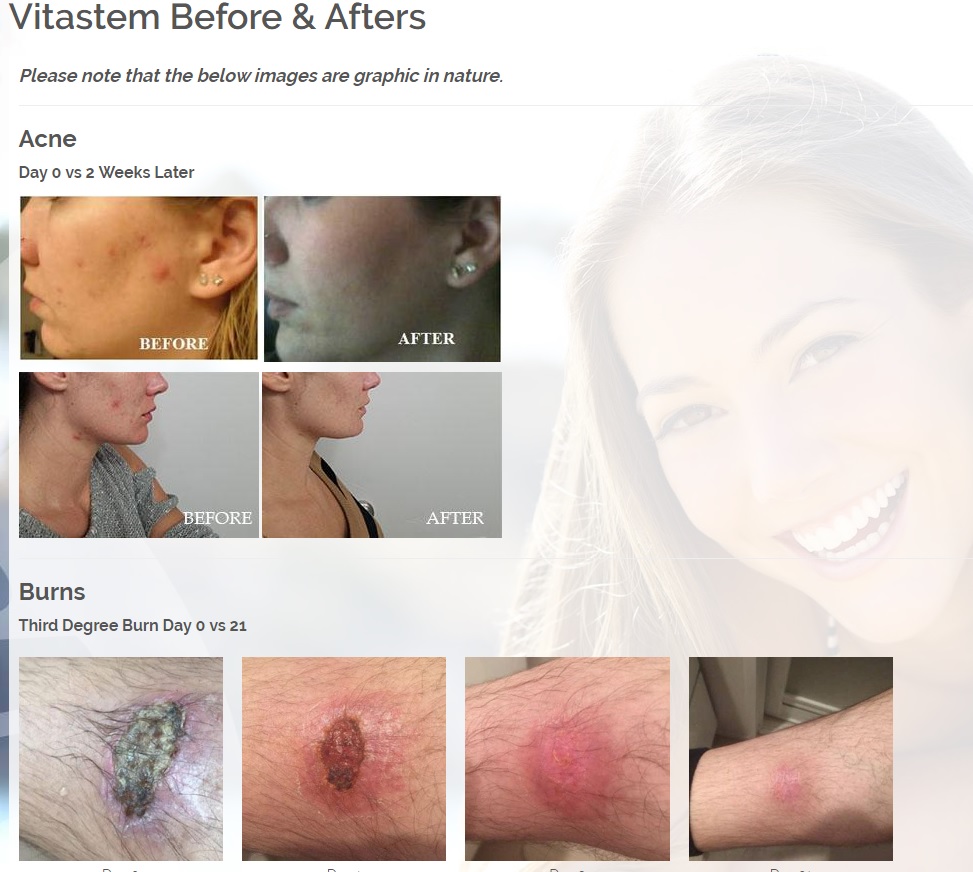
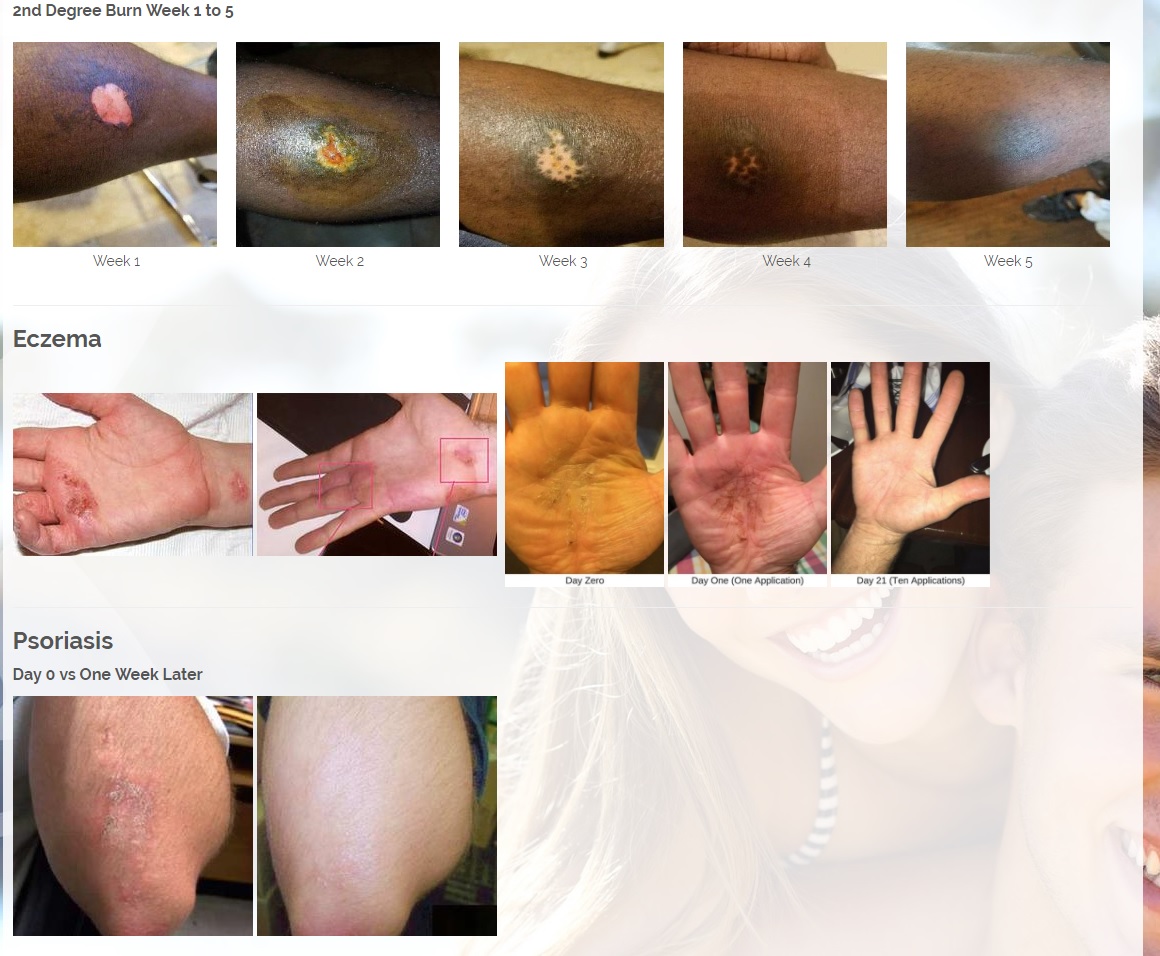
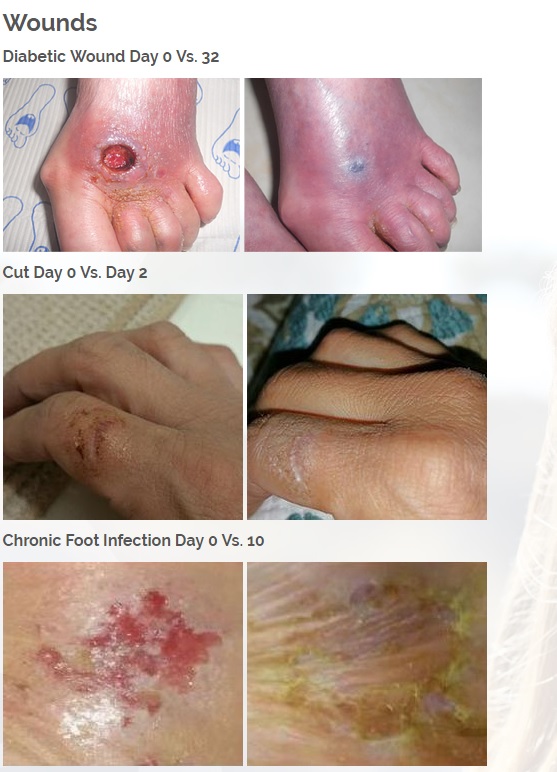

We are developing products in the following fields of use;
Treats all types of bacteria including MRSA, VRE and other flesh eating bacteria. Fights drug resistant bacteria. $6B/year global market.

Onychomycosis, a fungal infection of the toenails, is a major health problem. It is estimated that there are in excess of 40 million sufferers with this condition in the USA. It is a problem throughout the world. A recent European study showed that the prevalence of Onychomycosis may be as high as 26.9%. Fungal resistance can occur when the oral antifungal agents are used on a long-term basis. Topically applied antifungal drugs may work somewhat better adjunctive to surgical removal or chemical dissolution of the nail plate. Yet, this often ineffective and traumatic procedure leaves the subject without a nail for months at risk for re-infection. Given the limitations of current treatment options in this $3B market, there is a great need for a simple, nontoxic and effective alternative treatment. Estimated Global prevalence rate: 140 million.
Treats symptoms of Influenza which is a$4B/ year global market: Common
Diabetic foot wounds.

Per 3/8/18 PR: ViaDerma’s technology is currently being used in Elixr Cannabis products; Topical Balm, Topical Serum and Topical Spray. Sales have begun in Canada.
Per 12/7/17 PR: The Company has signed an MOU and will start production of its Patent Pending CBD or Cannabinoids product line. The Company would combine its two Patent pending technologies, the MMJ Patent #62466209, a patent for delivering medical marijuana / cannabis to the body by applying the medication onto the skin in an ointment base topical solution and the second Provisional patent # 62433964 for enhanced antibiotic and drug delivery for "Aqueous Topical Applications" for human and veterinary uses. The Company would combine its proprietary solutions with CBD's and other natural products that treat psoriasis, fibromyalgia for pain and other ailments.
Per 1/23/18 PR: The Company has also a licensing and distribution agreement in place with its Canadian partners to produce a CBD Topical Serum, which has a 92% absorption rate powered by ViaDerma's Patent Pending Dual Carrier Technology. The CBD and Terpene profile (enriched with specific essential oils and vitamins C, and D) is crafted to alleviate Chronic Dry Skin, Psoriasis, Eczema, Rosacea, and many other skin conditions. The serum has anti-inflammatory, anti-bacterial, and chronic pain relief properties that are absorbed by the skin and provide overall healing benefits. Sales are expected to begin in the first quarter of 2018 and are projected to generate approximately $2 million annual revenues.
The Patented Pending CBD or Cannabinoids product line. The Company would combine its two Patent pending technologies, the MMJ Patent, a patent for delivering medical marijuana / cannabis to the body by applying the medication onto the skin in an ointment base topical solution and the second Provisional patent for enhanced antibiotic and drug delivery for "Aqueous Topical Applications" for human and veterinarian uses. The Company would combine its proprietary solutions with CBD's and other natural products that treat psoriasis, fibromyalgia for pain and other ailments. The Company plans to continue to expand its (IP) "Intellectual Property Portfolio" in 2018.

CBD is a compound found in the Cannabis family of plants such as hemp. CBD is combined into our topical body care products like Elixr's Topical Balm, Serum, and Spray.
CBD is an antioxidant. Antioxidants protect our skin from damaging exposure to smoke, UV rays, and environmental pollutants. Cannabinoid receptors are located all throughout the skin, Topical CBD products interact with those receptors resulting homeostasis and healing. Studies show that CBD can help treat an array of skin conditions such as eczema, psoriasis, rosacea, and acne.
| "What is Intellectual Property?" - A Publication by the World Intellectual Property Organization -
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
