Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Once again:
Annual Report due March 30, 2024
** 16-month deadline is April 30, 2024
** 16-month plus 15-day grace period deadline is May 15, 2024
** These ONLY come into play if overdue 2023 2nd and 3rd Qtr Reports, 2023 Annual Reports, attorney letter, and profile update are not completed by then.
January 31, 2024 email from IR (Rich) excerpt:

Governing rules from otcmarkets:

It is due March 30th from what I can see.
TOMORROW IS MARCH ALREADY.THEY HAVE TO FILE BY END OF MARCH THE YEARLY REPORT.I DO NOT SEE GETTING FILED.
I believe they have 16 months to submit financial statements before the last annual report becomes "stale"...so April 30th.
...and at least ORCA stopped selling..
Well, next month is tomorrow, so TO DA MOON !!!
Your grind is stale
Because it’s next month. Lol
All joking aside, they have until I believe March 30th to submit the Annual and if not by then, Orca unfortunately will start to look correct with his “Expert Market” call…
VDRM SECURITY DETAILS
Share Structure
Market Cap Market Cap
12,337,131
02/28/2024
Authorized Shares
1,250,000,000
02/20/2024
Outstanding Shares
1,233,713,103
02/20/2024
Restricted
176,456,186
02/20/2024
Unrestricted
1,057,256,917
02/20/2024
Held at DTC
1,016,023,836
02/20/2024
Float
911,600,265
07/23/2021
Par Value
0.001
https://www.otcmarkets.com/stock/VDRM/security
sluggercjb U B DA Man Bro Go VDRM
You really showed him🙄
Keep your day job you’re not much of a comedian … at least Orca is funny at times… one more added to ignore list
Looks like only one purchase at .01 or above, MM sure are keeping this thing up lol
How is the new VDRM product Nupelo going???
Hahhahahhahahhahahahah
SCAMBAG OTIKO WHERE THE F ARE YOU???WHERE ARE THE FILINGS????MARCH IS HERE.
I was going to send you to OTCM to verify it yourself but Gameccks1 answered you with concrete info:
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=173933946
3.2 million last .0104,,asked .0109. finally a interesting day
Hopefully the sub penny are also gone after today's trades.
Your just angry that you didn't get back in @ 8's. No beer money for you. :)
Haven’t been here in a week or so. We still are NOT current???
Hahahhaahhaha wow
HEY LYING SCAMBAG YOU STILL HERE TO SUCKER IN PLAYERS WITH YOUR LIES??
VDRM WILL JOIN THE OTHER 5 STOCKS THAT WENT TO EXPERT MARKET.THEY ARE ALL DEAD.ZERO VOLUMES FOR A WHILE NOW.
WSRC,BRAV,MEDH,GLCO,SPSO.ALL DEAD.VDRM IS NEXT IN LINE.
Shouldn't be tough seeing as the licensing deals are through his other business IMVHO
NONE OF THIS MATTERS IF THERE IS NO FILINGS,AND IT GOES TO EXPERT MARKET.
Lets gooo Dr throw us a bone we are wayyy over due for some Fluff news anything for a $ pop $
Slops, as you know, the Doc has to get 2 quarterly reports and an annual report completed and filed along with an attorney letter. I can only hope that those items are already in process. Although preparing financial statements for a company like VDRM can't possibly be too technically difficult since there is hardly any activity to account for. I wish I knew what the hell he is waiting for. If the Doc truly received $1.2 million recently from licensing fees, it would seem like there should be some cash left to pay the accounting firm and the attorney. GET IT DONE ALREADY!!
MY POINT IS WE HAVE PLENTY OF TIME
Supposedly $1.2 million has been collected. We need financial statements to confirm.
How can the licensing be real? We've never been paid for them?
ITS NOT UP TO HIM.ITS UP TO OTC.THE OTC WILL SEND HIM TO EXPERT MARKET.
DR. HAS NO REASON TO END THIS CASH COW
I BELIEVE THAT THIS IS GOING TO EXPERT MARKET.IMO.
Can't fight against history, and further more, cant be changed, below proof that this CEO always been delayed but he ends up always providing the fillings... The licenses are real tho. The product is real. Only fake part is The hospital and Dubai deals... Of course, coming soon, all these deals he claimed to have now, potentially might be Fugazi as well. But everything else, current product is FDA checked, this company is not a Scam. Proven in disclosure and I have bought the product before, it is good as advertised. I speak with proof below, for free:
https://www.otcmarkets.com/stock/VDRM/disclosure
<<<< Thanks for the high post ratings☺️
Good post Plenty of time no reason to panic.
Re-post of my #71877 post: from two weeks ago:
Steps To Continue On Pink Sheets
1) Finish and issue overdue 2Q23 Quarterly Report before 4/30/24 (16-month deadline, not counting 15-day grace period)
2) Finish and issue overdue 3Q23 Quarterly Report before 4/30/24 (16-month deadline, not counting 15-day grace period)
3) Prepare and issue 2023 Annual Report, due 3/30/24, but certainly before 4/30/24 (16-month deadline, not counting 15-day grace period)
4) Provide annual profile update and get verified, when required
Source: https://www.otcmarkets.com/corporate-services/pink-market/pink-current-and-limited-information-disclosure-guidelines
Most likely on April 1st if all the 2023 filings are not submitted.
|
Followers
|
619
|
Posters
|
|
|
Posts (Today)
|
3
|
Posts (Total)
|
74600
|
|
Created
|
10/27/08
|
Type
|
Free
|
| Moderators | |||

ViaDerma's proprietary transdermal delivery system allows for rapid mass transfer of the pharmaceutical active ingredient across the skin and into the body to provide immediate localized therapy.
The technology allows transfer of chemicals through the stratum corneum (outer layer of skin) with a diffusion constant which is 10,000 times higher than the diffusion constant which characterizes water movement through the stratum corneum.
This enables ViaDerma to pair almost any active ingredient with the technology and provide rapid transport of the medicine right to the site of action.
The first product is a broad spectrum tetracycline-based topical antibiotic is the only antibiotic in the world that kills bacteria both a physical and a chemical mechanism. All known antibiotics (other than ours) primarily use only a chemical mechanism of kill. The physical mechanism of kill is a key feature of what we call Rapid Active Ingredient Delivery System (RAIDS). One result of RAIDS is that tetracycline is carried in higher concentrations, more quickly, to and through the cell walls, where the tetracycline can become more effective than if conventional antibiotics were used. Conventional antibiotics require more time (usually prescribed for 5 to 7 days for best results), whereas our tetracycline-based products usually produce desirable results in 24 hours (or less) because of the RAIDS effect.

A second important result of RAIDS is that the topical antibiotic kills all harmful Gram positive and Gram negative bacteria that have been available for testing. We believe this is the world’s strongest broad-spectrum topical antibiotic available.
The potential commercial impact is immense. Drug developers believe it takes much longer for bacteria to develop drug resistance to a physical kill mechanism. This is because it is relatively easy for bacteria to change their response to a chemical threat, but it takes numerous generations for bacteria to grow a new kind of cell wall structure to respond to a physical threat.
Our novel approach to overcome drug resistance of antibiotics is designed to sustain the effectiveness of antibiotics and other topical drugs for many years. This gives new topical drug products a longer useful lifetime and therefore more commercial value. This technology, when licensed to larger pharmaceutical companies, may provide stronger incentive for the discovery and development of new antimicrobial drugs. In recent years, the dearth of new antibiotics has been largely due to the uncertain new-drug commercial lifetime which is diminished when bacteria develop immunity to that drug.
In addition the technology can be licensed to other companies to convert existing oral drugs to transdermal medications extending the profitably of the existing drug.

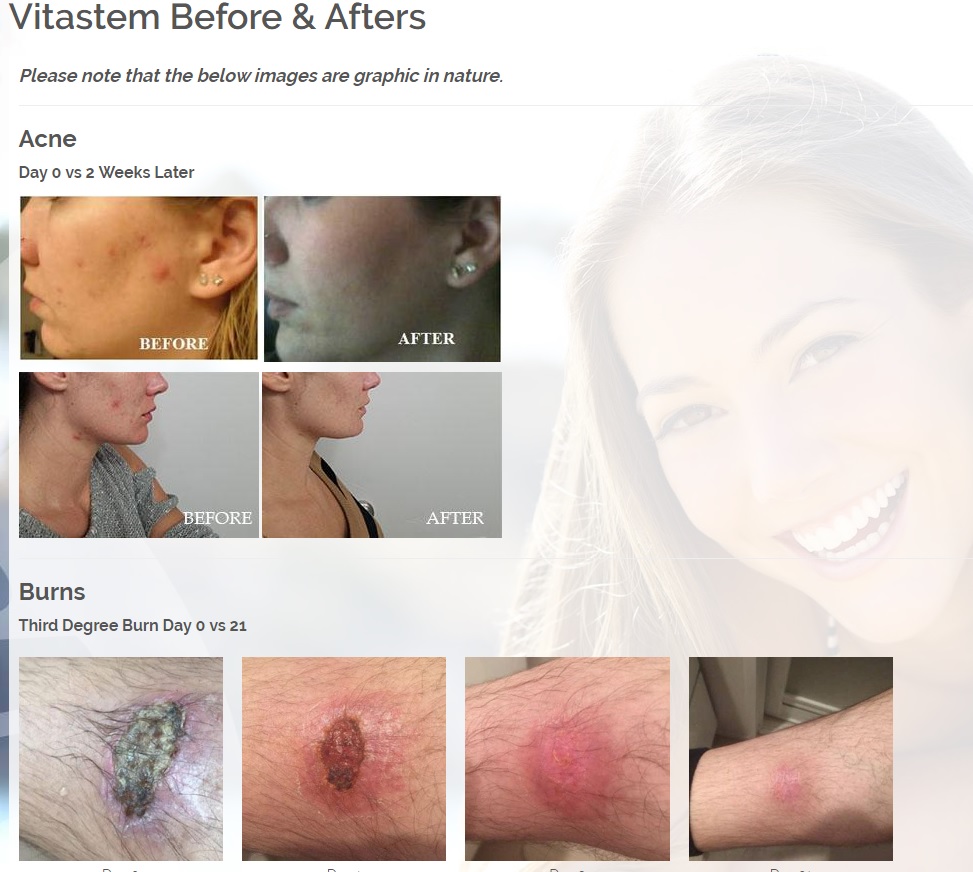
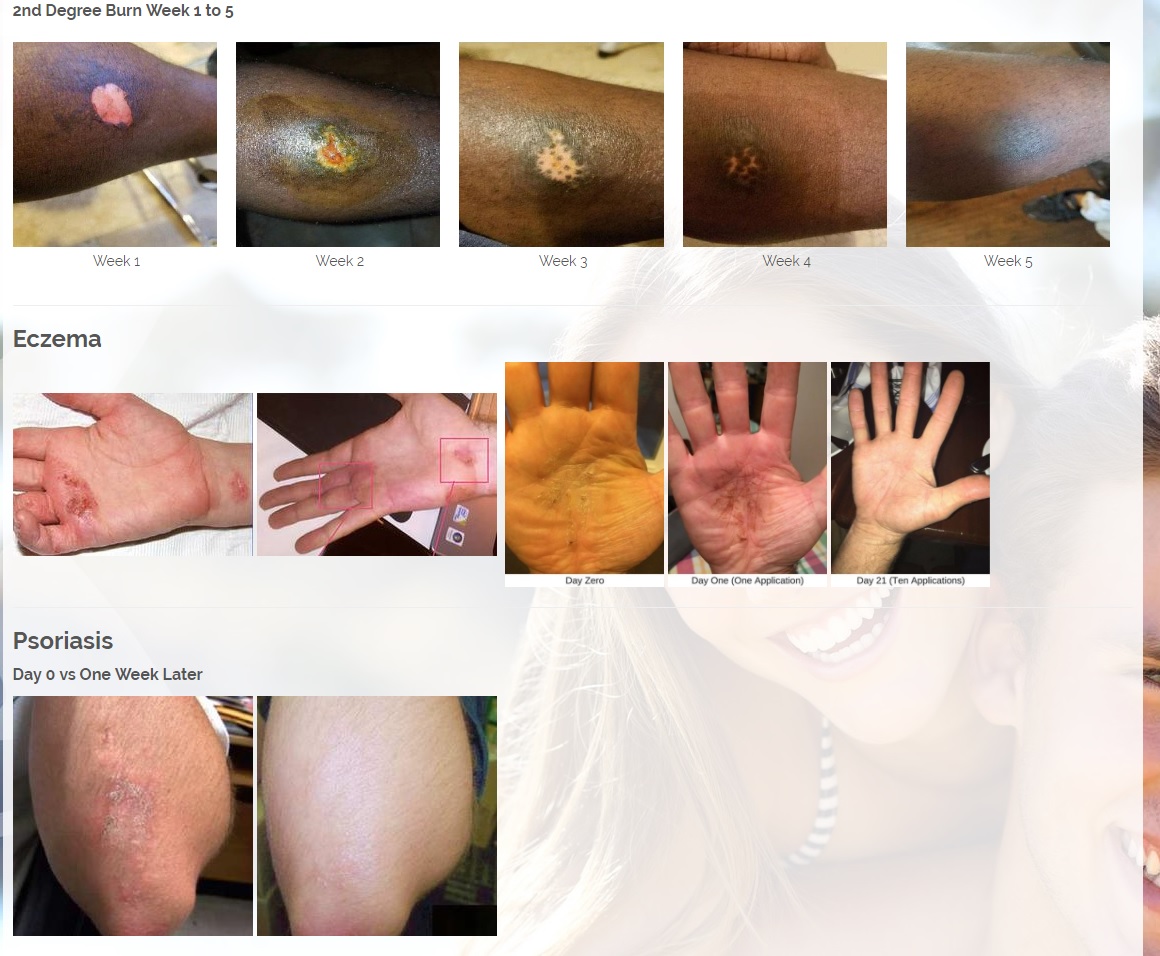
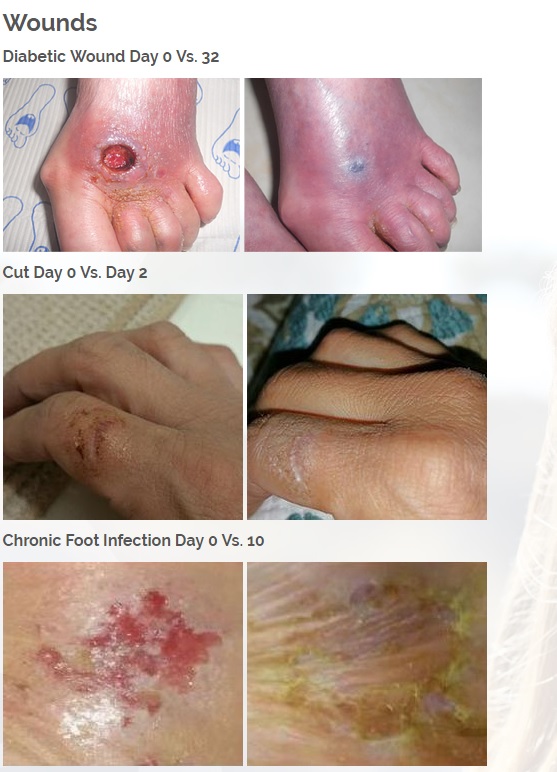

We are developing products in the following fields of use;
Treats all types of bacteria including MRSA, VRE and other flesh eating bacteria. Fights drug resistant bacteria. $6B/year global market.

Onychomycosis, a fungal infection of the toenails, is a major health problem. It is estimated that there are in excess of 40 million sufferers with this condition in the USA. It is a problem throughout the world. A recent European study showed that the prevalence of Onychomycosis may be as high as 26.9%. Fungal resistance can occur when the oral antifungal agents are used on a long-term basis. Topically applied antifungal drugs may work somewhat better adjunctive to surgical removal or chemical dissolution of the nail plate. Yet, this often ineffective and traumatic procedure leaves the subject without a nail for months at risk for re-infection. Given the limitations of current treatment options in this $3B market, there is a great need for a simple, nontoxic and effective alternative treatment. Estimated Global prevalence rate: 140 million.
Treats symptoms of Influenza which is a$4B/ year global market: Common
Diabetic foot wounds.

Per 3/8/18 PR: ViaDerma’s technology is currently being used in Elixr Cannabis products; Topical Balm, Topical Serum and Topical Spray. Sales have begun in Canada.
Per 12/7/17 PR: The Company has signed an MOU and will start production of its Patent Pending CBD or Cannabinoids product line. The Company would combine its two Patent pending technologies, the MMJ Patent #62466209, a patent for delivering medical marijuana / cannabis to the body by applying the medication onto the skin in an ointment base topical solution and the second Provisional patent # 62433964 for enhanced antibiotic and drug delivery for "Aqueous Topical Applications" for human and veterinary uses. The Company would combine its proprietary solutions with CBD's and other natural products that treat psoriasis, fibromyalgia for pain and other ailments.
Per 1/23/18 PR: The Company has also a licensing and distribution agreement in place with its Canadian partners to produce a CBD Topical Serum, which has a 92% absorption rate powered by ViaDerma's Patent Pending Dual Carrier Technology. The CBD and Terpene profile (enriched with specific essential oils and vitamins C, and D) is crafted to alleviate Chronic Dry Skin, Psoriasis, Eczema, Rosacea, and many other skin conditions. The serum has anti-inflammatory, anti-bacterial, and chronic pain relief properties that are absorbed by the skin and provide overall healing benefits. Sales are expected to begin in the first quarter of 2018 and are projected to generate approximately $2 million annual revenues.
The Patented Pending CBD or Cannabinoids product line. The Company would combine its two Patent pending technologies, the MMJ Patent, a patent for delivering medical marijuana / cannabis to the body by applying the medication onto the skin in an ointment base topical solution and the second Provisional patent for enhanced antibiotic and drug delivery for "Aqueous Topical Applications" for human and veterinarian uses. The Company would combine its proprietary solutions with CBD's and other natural products that treat psoriasis, fibromyalgia for pain and other ailments. The Company plans to continue to expand its (IP) "Intellectual Property Portfolio" in 2018.

CBD is a compound found in the Cannabis family of plants such as hemp. CBD is combined into our topical body care products like Elixr's Topical Balm, Serum, and Spray.
CBD is an antioxidant. Antioxidants protect our skin from damaging exposure to smoke, UV rays, and environmental pollutants. Cannabinoid receptors are located all throughout the skin, Topical CBD products interact with those receptors resulting homeostasis and healing. Studies show that CBD can help treat an array of skin conditions such as eczema, psoriasis, rosacea, and acne.
| "What is Intellectual Property?" - A Publication by the World Intellectual Property Organization -
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
