Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
What's your point everything is hear say including your lmao. Unless you got insider information. We all out here guessing. But if you ain't positive about possibility of this company why frequent the board of one smallest companys on whole market. Make it make sense why spend so much of your time here. But never have anything of substance to say?
Define "positive post". Anything positive posted on this board is always hearsay, conjecture, or unsubstantiated nonsense. Folks caught up with mostly excuses and articles that contain nothing new and are the same claims over and over. Stability requirements met, ready for export, signed contracts and on and on and on. Then it was all about the filings, why are they always late, what is the Doc thinking, why this and why that. Then more excuses. The company has a proven product, but the availability is confined to a single website that a sixth grader could have created. Then let's spend days and days posting about market manipulation as if that has a single thing to do about product availability. Yea that's it, it's the Money Managers that are responsible for product distribution. The only thing unfortunately that is consistent here are the excuses.
Lol all you want. I've seen for yrs you never post anything meaningful. Only post making derogatory remarks on any positive post. Always wonder why your even posting. Waste of words
I been here awhile, probly 4 plus yrs. Untill today I only read this board. So I signed up today and this will be my first post.
I would speculate that Dubai and other foreign deals where nixed cause of the staining issues. Which has been the obvious cause of all forward progress halting. I would assume Including payments for state licensing payments . Where these clinics able to get any product?
Fast forward several yrs now new formula. Non staining and have already went through the testing and further stability testing. Also apparently have a new manufacturer that can give huge supply ability.
This just might be vdrm time to level up we shall see soon I think
Indeed!!! Product is real but those expectations from that article with the CEO we do have on this company are totally unrealistic!!! FUGAZI mate. If you do not believe it, please investigate what happened with the hospital deals and Dubai set up? :)
New York Weekly releasted Jan. 5 2024
indyterp TY very informative link
1.5 month old rehashed garbage
...yawn.....
I hope you are right my friend It's time
@any day @anytime soon
What make you believe Dr. O will use that cash to pay for such? The man is hard from the elbows!! He will do a small dilution to cover such at shareholder's expense... It is the way of the OTC. I am not uncertain that he did screw up badly and this will end up exactly as the hospital and Dubai deals he offered long time ago. Only thing I am at peace is that the product is real and VDRM is here to stay, regardless of how good of a dancer the Doc is. Soo natty
Thanks funny. He owns interest in the licensing company. He can rob Peter to pay Paul. I never mentioned the "F" word but since it was brought up. . , It's most likely not New Money.
IMHO
Boom - thank you :)
Post #71760 has screenshot of 2/1/24 twitter post, regarding receipt of $1.2M of the licensing fees Accounts Receivable. Of course, this cannot be absolutely verified by shareholders until issue of 1Q24 Financial Report, hopefully on time (~5/15/24).
My previous post #71768 has basic status analysis, assuming verification of payment.
ViaDerma still Way under valued
Last post I promise - if the licensing fees are not actually being paid, and he has ownership in the other company licensing, would that be considered fraud? Maybe Money laundering?
Do we as stake holders (minority) have any claim to the accounts receivables?
Intent sciences 40M shares
Green tree financial group 49M shares
These shares were issued at the same time we had $700K in accounts receivables
Hahahhaha
Not to mention the 100M shares to the Dr, 20M to Jack brewer group, AT THE SAME TIME we had $700K in accounts receivables
Hahahahhaha
Please be careful here folks. Treat this “stock” like the blackjack table
Why do we want VDRM to collect the cash?
-pay cash for CPA to audit FINs (maybe he’s scared of this)
-pay cash for Intent Sciences or whatever joke of a marketing company he finds
It is VERY telling that he gives shares instead of collecting and paying with cash….
What are everyone’s thoughts?
What % of the accounts receivable is his other companies bills? Is his other company actually going to pay the (surely past due) bills?
As VDRM runs out of cash, predictably, why is he issuing more shares versus chasing down accounts receivables?
another week of no news. IR dept is terrible here!
All of them. accounts receivables always has a high number and cash minimal.
12/31/2022 accts receivable $700K
3/31/2023. Accts receivable $850K
12/31/2022 cash $10K
3/31/2023 cash $9.5K
How did we go 3months without collecting the prior 700K and increasing 150K to receivables? My direct question - are the licensing fees actually being paid?
Specifically looking at page 14, I am happy to be corrected. I don’t pour hours upon hours on FINs
https://www.otcmarkets.com/otcapi/company/financial-report/391590/content
What state are you talking about?
Market cap say this company worth $12.4M
Hahahahhahahahahhahahhahahahahha
The state license deals are through his other company. I’m not certain his other company is actually paying the license fees
Ok. Apologies Fdc. Holding this stock for so long is maddening. Our leader thinks that we should all be ecstatic because he files a long overdue report or because he sends out a tweet every now and then. I don't know what it's going to take for this guy to actually produce anything but bullsh*t. The Doc is completely unchecked and unaccountable. The saving grace (maybe) is that he has 100 million shares and he should want to increase the stock price. Sometime in the near future would nice!
LETS GO DOC!! DO YOUR JOB!!!!!
Fasctrack English is his second language don't knit pick
English must be your second language. Grammatically your sentence was written like a 4th grader and added nothing of substance toward the discussion of VDRM. Sorry mate!!!!
Do I have to teach you such as well? Holy Molly... Really? This is not the section for such mate, sorry!!! LMAFO. Can't make this up really.
And for the record, I did not get this from the friend of my friend... Just saying!!!
People, OTTIKO is in MIA mode now. Smh
"The shit did hit the fan and spreaded all over the room and on his face, not water to clean that mess, he is trapped."
What does any of that mean??
HE STILL HAS SOMETIME.WE WILL SEE WHAT HAPPENS.GLAD I OWN SEVERAL HUNDRED Ks AND NOT MILLIONS.
You are not the only one who thinks like that, I am finding people in stocktwits and Twitter questioning the same way. OTTIKO can't justify or lie anymore from this point. It is obvious he is stalling and lying. The shit did hit the fan and spreaded all over the room and on his face, not water to clean that mess, he is trapped. Good thing is the product is real and he is profitable with the states licenses but all that BS with international deals and Dubai and MExico and NUPELO.... FUGAZI
OTIKALOGY NEEDS TO FILE ASAP.
Hopefully, one day, in the not too distant future, the use of "very soon" or "soon" will not be either pumper/basher flipper attempts to manipulate.
Funny how on a day when the major indexes are crashing with Nasdaq down 200pts and Dow down over 400pts, we have all the Bears screaming “The bridge is falling down”…..
My reports are late because the dog eat them. LOL
Steps To Continue On Pink Sheets
1) Finish and issue overdue 2Q23 Quarterly Report before 4/30/24 (16-month deadline, not counting 15-day grace period)
2) Finish and issue overdue 3Q23 Quarterly Report before 4/30/24 (16-month deadline, not counting 15-day grace period)
3) Prepare and issue 2023 Annual Report, due 3/30/24, certainly before 4/30/24 (16-month deadline, not counting 15-day grace period)
4) Provide annual profile update and get verified, when required
Source: https://www.otcmarkets.com/corporate-services/pink-market/pink-current-and-limited-information-disclosure-guidelines
IT IS NOT A STEAL IF IT GOES TO EXPERT MARKET.VERY SOON.I WARNED PEOPLE ON WSRC AND BRAV.THEY BOTH WENT EXPERT MARKET RECENTLY.AND HAVE ZERO VOLUMES.DEAD.AND TO THINK THEY BOTH HAD FILED MORE FILINGS THAN VDRM.
Budman33 I agree present PPS is a steal
.07 a little late, what a steal here !
YEA BUT THE EXPERT MARKET WILL SAY HELLO WITHIN A MONTH.IN CASE YOU DID NOT KNOW.CHECK WSRC,AND BRAV.
DEAD EXPERT MARKET ZERO VOLUMES.VDRM IS NEXT.
Or maybe after the first Quarter 24 But not before
30 days from now I may agree. Not Yet.
|
Followers
|
617
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
74429
|
|
Created
|
10/27/08
|
Type
|
Free
|
| Moderators | |||

ViaDerma's proprietary transdermal delivery system allows for rapid mass transfer of the pharmaceutical active ingredient across the skin and into the body to provide immediate localized therapy.
The technology allows transfer of chemicals through the stratum corneum (outer layer of skin) with a diffusion constant which is 10,000 times higher than the diffusion constant which characterizes water movement through the stratum corneum.
This enables ViaDerma to pair almost any active ingredient with the technology and provide rapid transport of the medicine right to the site of action.
The first product is a broad spectrum tetracycline-based topical antibiotic is the only antibiotic in the world that kills bacteria both a physical and a chemical mechanism. All known antibiotics (other than ours) primarily use only a chemical mechanism of kill. The physical mechanism of kill is a key feature of what we call Rapid Active Ingredient Delivery System (RAIDS). One result of RAIDS is that tetracycline is carried in higher concentrations, more quickly, to and through the cell walls, where the tetracycline can become more effective than if conventional antibiotics were used. Conventional antibiotics require more time (usually prescribed for 5 to 7 days for best results), whereas our tetracycline-based products usually produce desirable results in 24 hours (or less) because of the RAIDS effect.

A second important result of RAIDS is that the topical antibiotic kills all harmful Gram positive and Gram negative bacteria that have been available for testing. We believe this is the world’s strongest broad-spectrum topical antibiotic available.
The potential commercial impact is immense. Drug developers believe it takes much longer for bacteria to develop drug resistance to a physical kill mechanism. This is because it is relatively easy for bacteria to change their response to a chemical threat, but it takes numerous generations for bacteria to grow a new kind of cell wall structure to respond to a physical threat.
Our novel approach to overcome drug resistance of antibiotics is designed to sustain the effectiveness of antibiotics and other topical drugs for many years. This gives new topical drug products a longer useful lifetime and therefore more commercial value. This technology, when licensed to larger pharmaceutical companies, may provide stronger incentive for the discovery and development of new antimicrobial drugs. In recent years, the dearth of new antibiotics has been largely due to the uncertain new-drug commercial lifetime which is diminished when bacteria develop immunity to that drug.
In addition the technology can be licensed to other companies to convert existing oral drugs to transdermal medications extending the profitably of the existing drug.

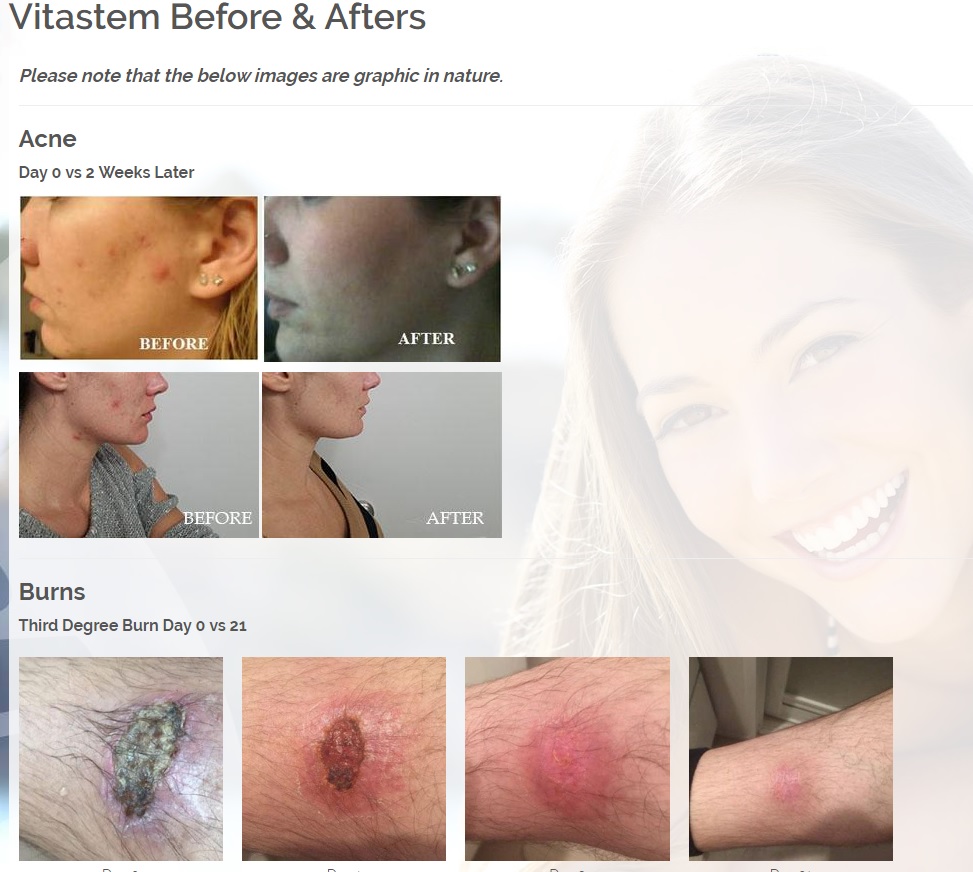
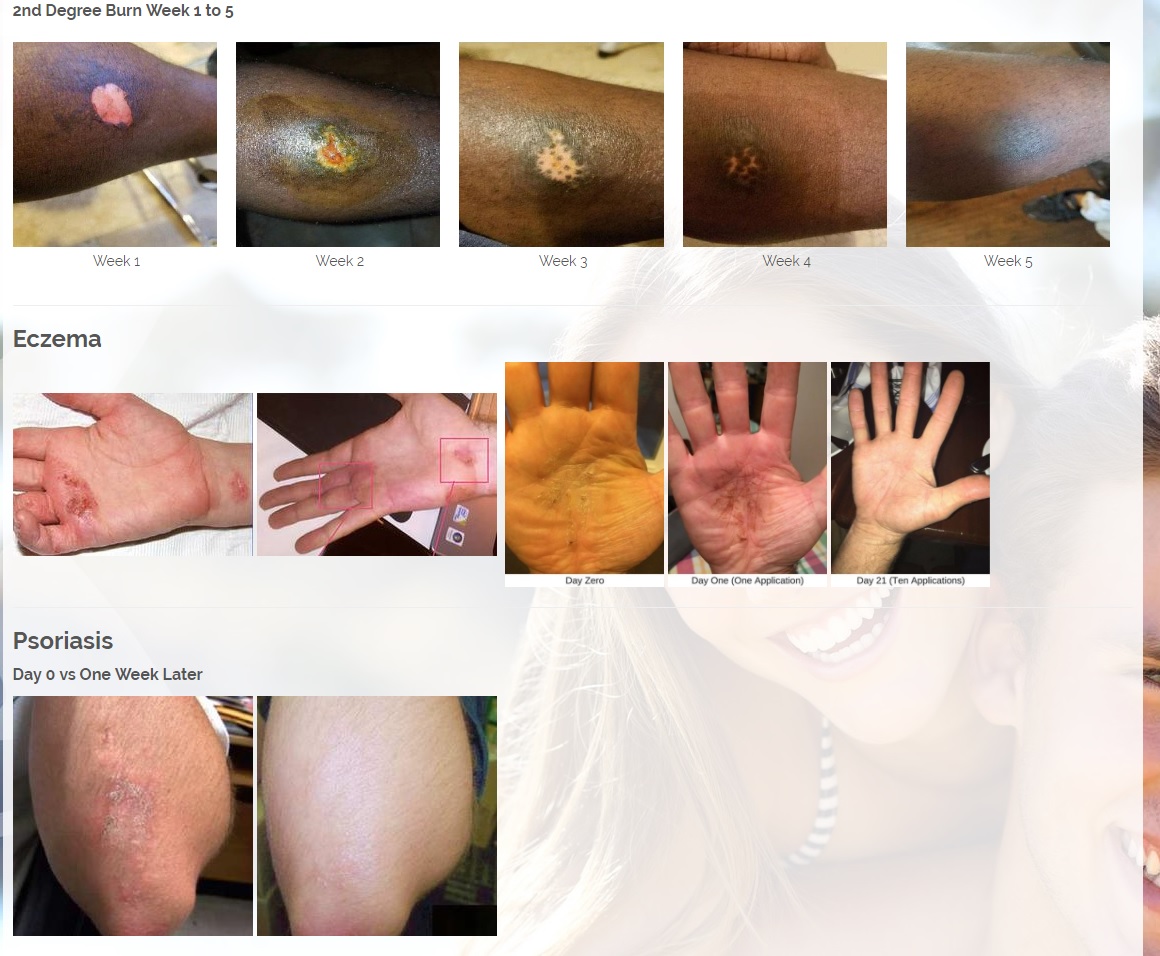
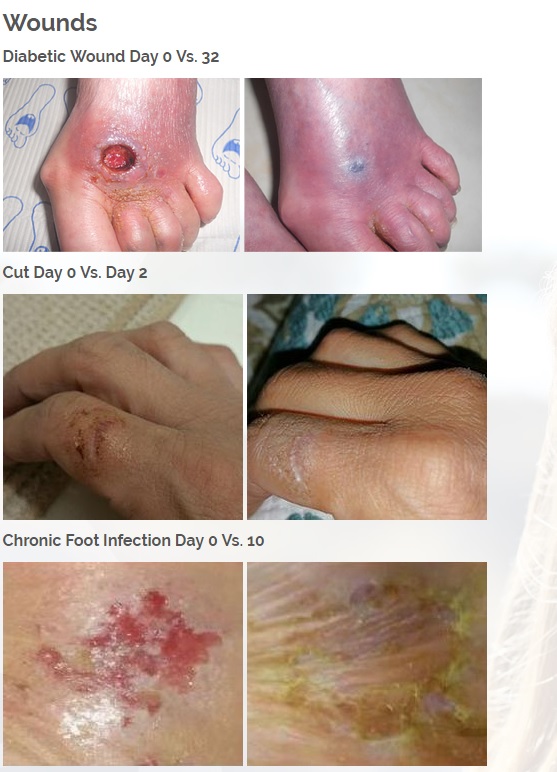

We are developing products in the following fields of use;
Treats all types of bacteria including MRSA, VRE and other flesh eating bacteria. Fights drug resistant bacteria. $6B/year global market.

Onychomycosis, a fungal infection of the toenails, is a major health problem. It is estimated that there are in excess of 40 million sufferers with this condition in the USA. It is a problem throughout the world. A recent European study showed that the prevalence of Onychomycosis may be as high as 26.9%. Fungal resistance can occur when the oral antifungal agents are used on a long-term basis. Topically applied antifungal drugs may work somewhat better adjunctive to surgical removal or chemical dissolution of the nail plate. Yet, this often ineffective and traumatic procedure leaves the subject without a nail for months at risk for re-infection. Given the limitations of current treatment options in this $3B market, there is a great need for a simple, nontoxic and effective alternative treatment. Estimated Global prevalence rate: 140 million.
Treats symptoms of Influenza which is a$4B/ year global market: Common
Diabetic foot wounds.

Per 3/8/18 PR: ViaDerma’s technology is currently being used in Elixr Cannabis products; Topical Balm, Topical Serum and Topical Spray. Sales have begun in Canada.
Per 12/7/17 PR: The Company has signed an MOU and will start production of its Patent Pending CBD or Cannabinoids product line. The Company would combine its two Patent pending technologies, the MMJ Patent #62466209, a patent for delivering medical marijuana / cannabis to the body by applying the medication onto the skin in an ointment base topical solution and the second Provisional patent # 62433964 for enhanced antibiotic and drug delivery for "Aqueous Topical Applications" for human and veterinary uses. The Company would combine its proprietary solutions with CBD's and other natural products that treat psoriasis, fibromyalgia for pain and other ailments.
Per 1/23/18 PR: The Company has also a licensing and distribution agreement in place with its Canadian partners to produce a CBD Topical Serum, which has a 92% absorption rate powered by ViaDerma's Patent Pending Dual Carrier Technology. The CBD and Terpene profile (enriched with specific essential oils and vitamins C, and D) is crafted to alleviate Chronic Dry Skin, Psoriasis, Eczema, Rosacea, and many other skin conditions. The serum has anti-inflammatory, anti-bacterial, and chronic pain relief properties that are absorbed by the skin and provide overall healing benefits. Sales are expected to begin in the first quarter of 2018 and are projected to generate approximately $2 million annual revenues.
The Patented Pending CBD or Cannabinoids product line. The Company would combine its two Patent pending technologies, the MMJ Patent, a patent for delivering medical marijuana / cannabis to the body by applying the medication onto the skin in an ointment base topical solution and the second Provisional patent for enhanced antibiotic and drug delivery for "Aqueous Topical Applications" for human and veterinarian uses. The Company would combine its proprietary solutions with CBD's and other natural products that treat psoriasis, fibromyalgia for pain and other ailments. The Company plans to continue to expand its (IP) "Intellectual Property Portfolio" in 2018.

CBD is a compound found in the Cannabis family of plants such as hemp. CBD is combined into our topical body care products like Elixr's Topical Balm, Serum, and Spray.
CBD is an antioxidant. Antioxidants protect our skin from damaging exposure to smoke, UV rays, and environmental pollutants. Cannabinoid receptors are located all throughout the skin, Topical CBD products interact with those receptors resulting homeostasis and healing. Studies show that CBD can help treat an array of skin conditions such as eczema, psoriasis, rosacea, and acne.
| "What is Intellectual Property?" - A Publication by the World Intellectual Property Organization -
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
