Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
For once can you say something that makes sense?
Never underestimate o tiki
Never underestimate o tiki
Slowly slowly back to 0.0016
Slowly slowly back to 0.0016
Don't think he's based in LA anymore.
Does anyone here live near Los Angeles? Reseda CA?
Stop by and talk to him.
I’m in Texas or else I would
The unfortunate sad reality here is that unless the Doc puts out a fluff piece, we won't see any news because he has nothing to report. Let me repeat that again, he has nothing to report. Let that sink in. Don't you think that if he actually had inked a deal with big pharma or a hospital or an insurance carrier, he would shout that from the rooftops?? Who knows what this guy is doing on a daily basis but he's not working on VDRM. Furthermore he doesn't seem to give a shit either as the price continues to trickle downward.
Laundered… Clean accounting stealing. Chinese style cooking books. This CEO is a piece of work. That 700k, watch, it will be cleaned as well in a year or 2
Great dude diligence here. I had no idea about this previous loan. That is absolutely WILD
No kidding! But DB was a broker and has years and years of experience investing. And I flew the Space Shuttle. What a joke. I spoke with Otiko earlier today, he was watching golf. Wanted me to thank everyone for the new pad he is enjoying. Standby for some earth breaking news on the Nupelo front as it was discovered it has an even better use and is going to turn the market upside-down. I'll be happy to share the news with the board once he gives me the final details. LMFAO
What's said at the table stays at the table
Interesting tidbit out of Rich: he said Dr. Otiko.....wait, I don't share here!
You should your last post to Rich and see if he responds.
Thanks for the update but infuriating he's ignoring what matters most.
What is the point of having a head of IR if they're not even going to answer shareholders questions.
50 states licensing by end of 2023? Never happened.
Shipping 250K+ product per country overseas and shipping starts in March? Never happened.
Nupelo will use new natural solution? Never happened.
VA deal? Never happened.
Large HMO deal? Never happened.
Dubai multi-million $$ deal? Never happened.
2023 will be a pivotal year? Never happened.
Now in 2024: No tweets in months. No PRs in months. Otiko gives him $700k with no explanation. Near total silence from IR.
https://www.globenewswire.com/en/news-release/2023/10/24/2765805/33818/en/ViaDerma-Update-on-Stability-Testing-Global-Sales-Distribution-National-Licensing-Nupelo-More.html
Got an answer from Rich, today, regarding he will pass along the information on Section 4) (see https://investorshub.advfn.com/boards/read_msg.aspx?message_id=174436345 ). Of course, he is still ignoring my emails regarding the growing and glaring discrepancy between official, public revenue information from financial reports and official, public press releases, regarding state licensing agreements?
2Q23 $150K vs $165K (per 03/02/23 PR)
3Q23 $150K vs $165K (per 03/02/23 PR)
4Q23 $150K vs $210K (per 10/24/23 PR)
1Q24 $150K vs $210K (per 10/24/23 PR)
The lack of response pretty much points to negative possibility (the "official" PRs are just wrong, and that is not desirable, or enough of a concern, to admit to investors).
It's not just a one man show.
Garrett Adams has been the president of Viaderma since 2016
https://www.linkedin.com/in/garrettradams
Here's an old post from 7 years ago. https://investorshub.advfn.com/boards/read_msg.aspx?message_id=134497161
His contact information is on there. Maybe he'll actually answer people since the IR guy never does. He's also listed as the trademark owner with otiko for vitastem ultra. https://uspto.report/TM/98338274
Adams is also the president of biogenx which doesn't seem like it's in business anymore - http://biogenx.net/ - website is unavailable. Not even sure what this guy actually does at Viaderma. Based on all the VDRM filings he's obviously not selling anything and it looks like he's the one in charge of sales. They only have licensing deals with otiko's other company the wound pros.
Richard Inza has been IR person there for several years too. But not sure if he's still there since no one can ever get a hold of him and IR@viadmera.com never responds to any emails.
Interesting speculation, I thought about such before but this CEO isn’t destabilizing anything. He’s irresponsible and it is natural this outcome. PPS dropped like this or lower before 0.005 once, and lower too. It is not the first time he gets loans. It is not the first time he lies. We just gotta hold and keep it cool. It will bounce up again. He is running the company like a grocery store. Not growth or partnerships or board. It is him alone
I have a thought that it seems as though this ceo is trying to destabilize vdrm to maybe knock down the stock price to buy up cheap shares one day and boom it takes off as it should as with all these patents why put in the effort otherwise 🤔
You never know , he must have bought Vdrm on recent pull back , watch he gonna push towards 0.0018
You never know , he must have bought Vdrm on recent pull back , watch he gonna push towards 0.0018
These accusations in public and reaching the SEC over this will lock this company til complete investigation. Shareholders will lose everything at the end and CEO will only pay a small fine. Please do your search quietly, if it is a scam keep it to yourself and GTFO when you get the chance. Many scams or ponzi do still operate normal in OTCM if left alone and make people money. I know you are frustrated with the dancer CEO we got but it is our dancer and we can’t change such during under water now. Chill all
You can see https://investorshub.advfn.com/boards/read_msg.aspx?message_id=174410521 for conglomerated excerpts regarding previous related party "loan" cancellation.
There was a mention that Dr. Otiko would use a natural ingredient in Nupelo instead of the planned for minoxidil for which the FDA would have required expensive and lengthy clinical testing. A natural product does not require any clinical testing to my knowledge.
Can anybody think of what sort of natural ingredient would be a good hair growth candidate? Better skin penetration by combining with the Viaderma transdermal transporter chemistry could plausibly have a greater effect at the hair follicle and grow hair more effectively. I guess I sound gullible to give this a thought. Hair growth for men is a valuable market.
Should the SEC investigate the $700,000 loan Dr. Otiko gave to himself? He never repaid to the company , to my knowledge , the previous $300,000 amount loaned to himself. The company has no income. How can this be justified or legal?
Are there any laws that regulate this sort of activity? Wouldn’t there need to be some sorts of terms regarding the loan and a stated interest rate? Just wondering if anybody, ideally a lawyer or an MBA , has any opinions about this.
As a shareholder myself I do not want to harm the stock price by asking such questions. I just feel offended by it. I would like to see the shareholders take control of the company.
Another area of exasperation from and complete lack of professionalism in 1Q24 Financial Report is the out of date, nearly in total, nature of key 4) Issuer's Business, Products, and Services section. For the most part, I have only been focusing on the financial tables and new notes in recent financial reports. However, this time, upon reading the current section 4), it is a complete embarrassment. The Biogenx paragraph is loosely up to date, but could be significantly streamlined, due to passage of time; state licensing information is over three (3) years out of date; and everything else is four (4) to seven (7) years out of date, and/or speaking of multiple, previous, if ever really existed, failed and/or never heard from again ventures as if ongoing. The section probably could be an updated state licensing paragraph; streamlined Biogenx paragraph; hold note on Nupelo; and any other truly ongoing activity information, if there actually is any. Infuriating!
Yup....plus it's pretty simple since nothing is different really....financials wise.......
I surmise he is trying to use a seemingly infinitesimal piece of new credibility, by first on-time quarterly in over a year, and previously announced $1.2M cash receipt, to distract from all of the failure/lack of progress, and personal loan.
He's always late getting the financials out, why do you think he was in such a hurry to get this BS posted on time??
I would paid to see that happening. This is the type of man that would rather burn his company before getting kicked out. Certain buttons are better not to be touched sometimes.
But, again, it is a good idea if it can get pulled off on a company that does not have board of shareholders formed
I only have the information in the report, which declares the $700K, as follows: "On February 14, 2024, the Company’s CEO borrowed $700,000 from the Company." In the follow-on post, I have compiled the details for past cancelled "loan" to related party: https://investorshub.advfn.com/boards/read_msg.aspx?message_id=174410521 . The presentation is different. Thus, I assume it was just a ridiculous personal "loan" to himself.
Regarding the licensing, the related party pays VDRM the license fees, when they pay. It does appear that an additional $35K partial 1Q24 cash payment was made.
In my opinion, reporting receipt of $500K alone of real cash January 31st, verified in 1Q24 financial report, would have been notable enough change from year plus with no actual cash payments. That, also in my opinion, makes your surmised, likely very risky move unnecessary.
The collectability of the new "loan" remains to be seen as time goes on.
i want to see 3 year lows, lets see next week
I will not surprise if they close the office and run
GC, I did not think that $700k cash actually left the company to go to the Doc. Rather, the affiliate that collects the license fees does not intend to pay those fees to the company. So the receivable from affiliate got reclassed into a receivable from the Doc...and the company will soon declare it to be uncollectable. Is that correct?
From the text: "On February 14, 2024, the Company’s CEO borrowed $700,000 from the Company.", the "loan", assuming he ever pays it back, would be for his personal use. With the over-the-top bonus clause, <$5M contract, brought to company by him, would "pay" him roughly same amount, including taxes impact. Thus, infuriatingly and typically, that looks like another indicator of no real deal near fruition, if there ever were any, near fruition, in the first place.
How about a shareholder meeting to rescind his self-dealing contracts and vote that MFer out!
Then you'd think they'd move that $700k to a line for "inventory" or AP. But a third-party loan given most likely to himself is not going to sit well with investors without explanation. And most of the time the explanations they send out are absolute BS from the company. 100K+ units to Egypt, Middle East, Caribbean, India were discussed in the past. Now, nothing from them. Always excuses or just total silence. This stinks like horse sh*t and is the last straw for most shareholders IMO. The talk about SEC complaints is growing daily and 100% warranted if this turns out to be one big criminal fraud org they are possibly running. The silence from the IR guy and the company the past few months is not helping their case either.
Your option may be possible. However, I look at it as an either/or. Either there needs to be inventory purchased, which temporarily shows up on the Balance Sheet, until shipment and delivery, or an Accounts Payable that accounts for product order being produced for drop shipment. In the recent hyped, 100000+ units to multiple countries,"on schedule" for March shipments, if they had been real, seems to require one or the other on the company books, whereas no manufacturer is going to produce that (or probably any) quantity of product, without signed, legal commitments, from this company.
Thanks for the recalibration. I had allowed myself to adopt jaded expectations, through track record of multiple otcm CEOs, over the years, and what has passed as so called "business practices". That, along with not desiring a potential, ultimate legal outcome, at this time, due to the likely injurious nature to shareholders, led to my opinion. However, thanks for the reminder that neither of those impact actual, legal responsibilities of CEO.
Borderline?
It is 100% fraudulent if proven to be true, which seems more likely now as each day passes.
The stock seems to be trading more on technicals now and looks headed to .0055 - .005. Typically there's a bounce. But with no PRs in months, No tweets in months, and the CEO taking out $700K with zero explanation is not looking great. No one seems able to get in touch with the IR guy either. Not good. This guy gets paid with millions in shares and won't even reply to anyone. What's the point of having an IR if they don't communicate with shareholders. This whole thing starting to stink like total sh*t now.
If they actually did start shipping products in March as stated, they would have PR'd that by now. 99.9% it never happened and now they'll just post some new bullsh*t excuses or just not say a word. If not, this will just slide back to the .001 range and maybe to 000s.
If anyone ever does get a hold of IR, please share the BS excuses on here. If they keep ignoring shareholders and best interests of shareholders and are just self-dealing and siphoning money out of the company, then theres going to be much bigger problems for them.
Regarding no inventory/ no payables; I wonder if VDRM will ever actually carry inventory? I suppose there could be an arrangement with a manufacturer who sells direct with an appropriate percentage back to the company. No inventory is very concerning though; I think I'm just trying to sugar coat it.
Agreed, Doc actions are clearly self-dealing. He is acting in his own best interest instead of the best interest of the company and the shareholders. It's also a huge conflict of interest and I agree with GC ...highly unethical. Other thoughts come to mind as well, gross negligent use of company assets and borderline fraudulent.
I do agree with you partially. The part you didn’t include is that this is a business and it is shareholders money, he must declare it and there should be limits to CEO to do such. It is the 2nd loan asked, for what? Can the company afford it? What if we did inherit a gambler on this CEO!!! There should be a procedure, this CEO didn’t follow or explained nobody none.
Despite of the ethical part, you know it is wrong what he is doing: The Devil is within the details
Incorrect since his Fiduciary Duty to shareholders is to declare prior why and at what terms I/e interest rate payable to the company!!!
Ooohh, thats why you don't share. Got it. Thanks for clearing that up.
CEO borrowed the funds from the company personally, as deemed credit worthy by himself, basically. Thus, it is more likely falls under ethical concern than legal one, due to his position, in my opinion.
|
Followers
|
615
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
74011
|
|
Created
|
10/27/08
|
Type
|
Free
|
| Moderators | |||

ViaDerma's proprietary transdermal delivery system allows for rapid mass transfer of the pharmaceutical active ingredient across the skin and into the body to provide immediate localized therapy.
The technology allows transfer of chemicals through the stratum corneum (outer layer of skin) with a diffusion constant which is 10,000 times higher than the diffusion constant which characterizes water movement through the stratum corneum.
This enables ViaDerma to pair almost any active ingredient with the technology and provide rapid transport of the medicine right to the site of action.
The first product is a broad spectrum tetracycline-based topical antibiotic is the only antibiotic in the world that kills bacteria both a physical and a chemical mechanism. All known antibiotics (other than ours) primarily use only a chemical mechanism of kill. The physical mechanism of kill is a key feature of what we call Rapid Active Ingredient Delivery System (RAIDS). One result of RAIDS is that tetracycline is carried in higher concentrations, more quickly, to and through the cell walls, where the tetracycline can become more effective than if conventional antibiotics were used. Conventional antibiotics require more time (usually prescribed for 5 to 7 days for best results), whereas our tetracycline-based products usually produce desirable results in 24 hours (or less) because of the RAIDS effect.

A second important result of RAIDS is that the topical antibiotic kills all harmful Gram positive and Gram negative bacteria that have been available for testing. We believe this is the world’s strongest broad-spectrum topical antibiotic available.
The potential commercial impact is immense. Drug developers believe it takes much longer for bacteria to develop drug resistance to a physical kill mechanism. This is because it is relatively easy for bacteria to change their response to a chemical threat, but it takes numerous generations for bacteria to grow a new kind of cell wall structure to respond to a physical threat.
Our novel approach to overcome drug resistance of antibiotics is designed to sustain the effectiveness of antibiotics and other topical drugs for many years. This gives new topical drug products a longer useful lifetime and therefore more commercial value. This technology, when licensed to larger pharmaceutical companies, may provide stronger incentive for the discovery and development of new antimicrobial drugs. In recent years, the dearth of new antibiotics has been largely due to the uncertain new-drug commercial lifetime which is diminished when bacteria develop immunity to that drug.
In addition the technology can be licensed to other companies to convert existing oral drugs to transdermal medications extending the profitably of the existing drug.

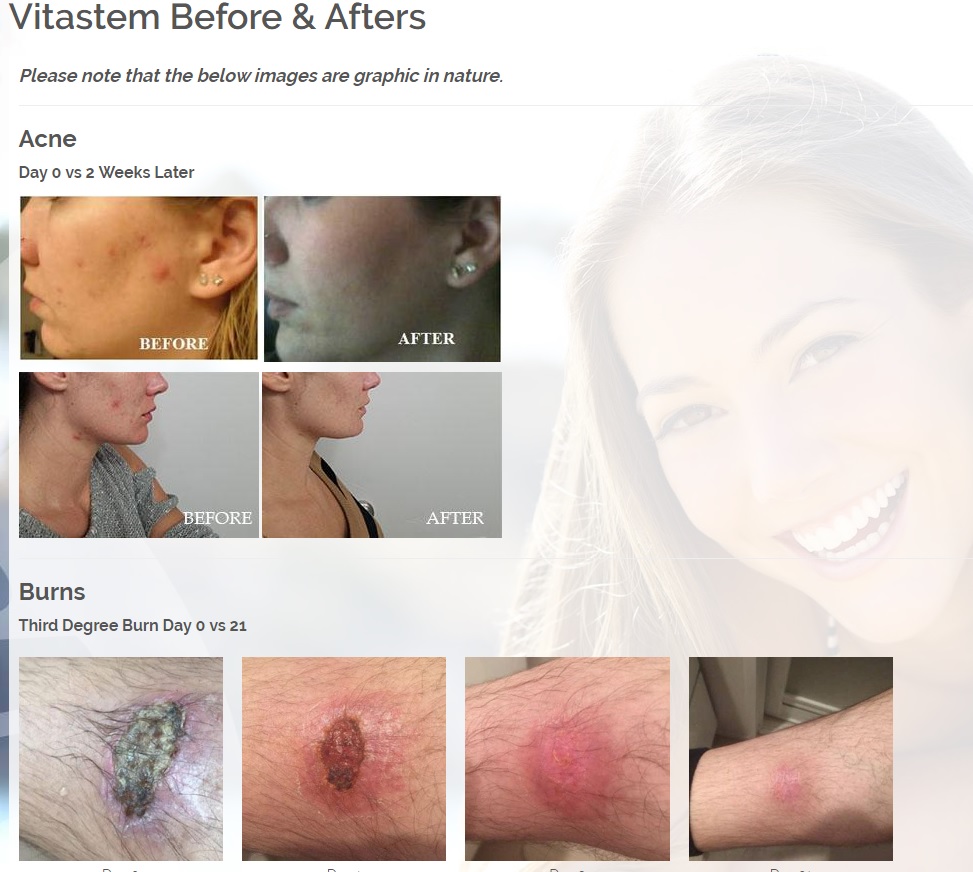
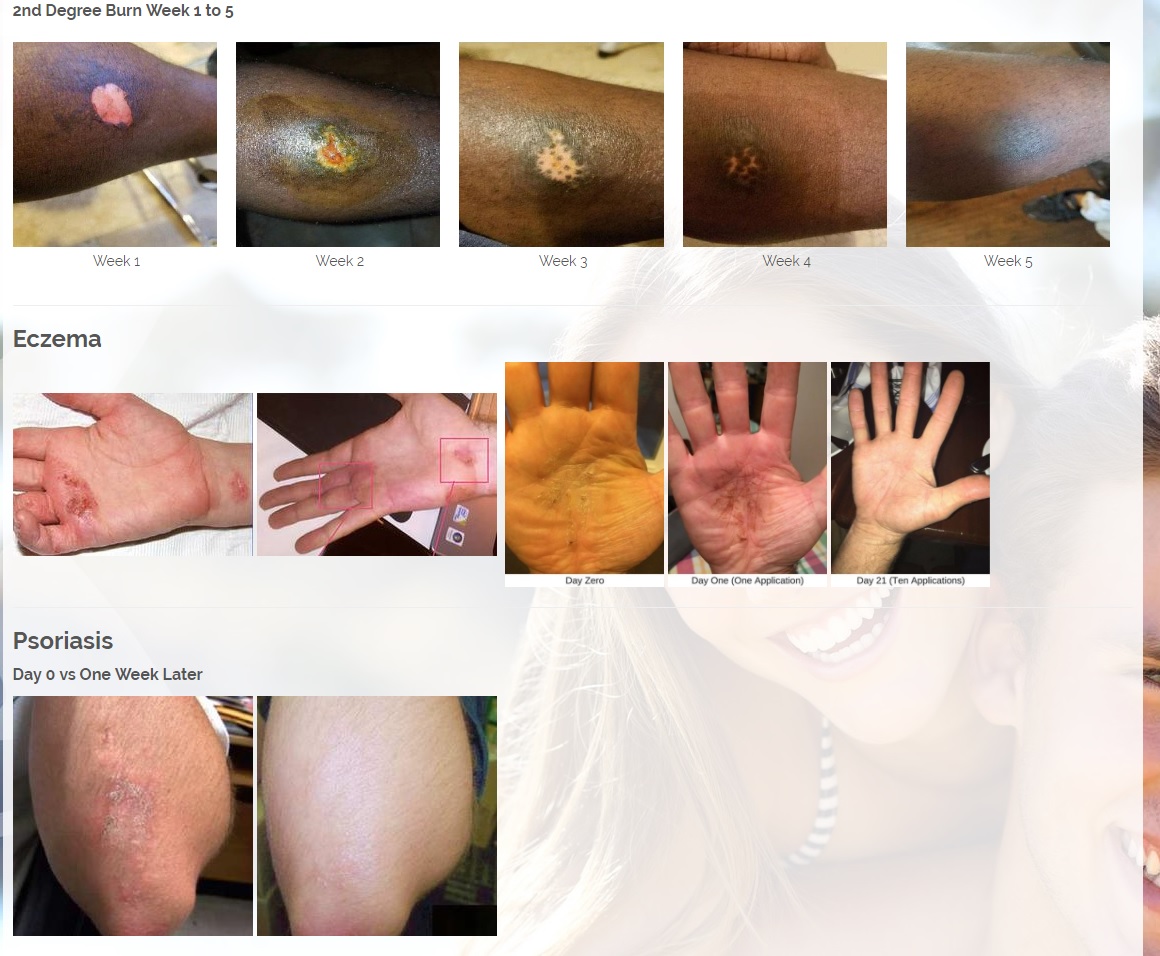
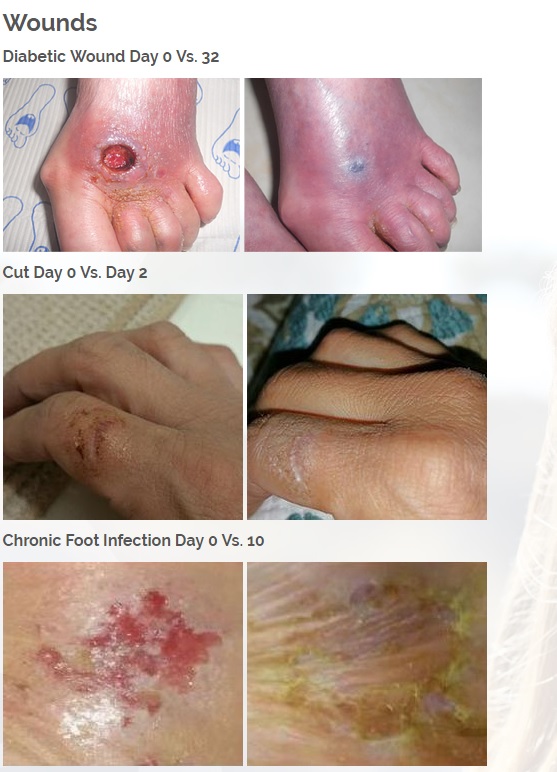

We are developing products in the following fields of use;
Treats all types of bacteria including MRSA, VRE and other flesh eating bacteria. Fights drug resistant bacteria. $6B/year global market.

Onychomycosis, a fungal infection of the toenails, is a major health problem. It is estimated that there are in excess of 40 million sufferers with this condition in the USA. It is a problem throughout the world. A recent European study showed that the prevalence of Onychomycosis may be as high as 26.9%. Fungal resistance can occur when the oral antifungal agents are used on a long-term basis. Topically applied antifungal drugs may work somewhat better adjunctive to surgical removal or chemical dissolution of the nail plate. Yet, this often ineffective and traumatic procedure leaves the subject without a nail for months at risk for re-infection. Given the limitations of current treatment options in this $3B market, there is a great need for a simple, nontoxic and effective alternative treatment. Estimated Global prevalence rate: 140 million.
Treats symptoms of Influenza which is a$4B/ year global market: Common
Diabetic foot wounds.

Per 3/8/18 PR: ViaDerma’s technology is currently being used in Elixr Cannabis products; Topical Balm, Topical Serum and Topical Spray. Sales have begun in Canada.
Per 12/7/17 PR: The Company has signed an MOU and will start production of its Patent Pending CBD or Cannabinoids product line. The Company would combine its two Patent pending technologies, the MMJ Patent #62466209, a patent for delivering medical marijuana / cannabis to the body by applying the medication onto the skin in an ointment base topical solution and the second Provisional patent # 62433964 for enhanced antibiotic and drug delivery for "Aqueous Topical Applications" for human and veterinary uses. The Company would combine its proprietary solutions with CBD's and other natural products that treat psoriasis, fibromyalgia for pain and other ailments.
Per 1/23/18 PR: The Company has also a licensing and distribution agreement in place with its Canadian partners to produce a CBD Topical Serum, which has a 92% absorption rate powered by ViaDerma's Patent Pending Dual Carrier Technology. The CBD and Terpene profile (enriched with specific essential oils and vitamins C, and D) is crafted to alleviate Chronic Dry Skin, Psoriasis, Eczema, Rosacea, and many other skin conditions. The serum has anti-inflammatory, anti-bacterial, and chronic pain relief properties that are absorbed by the skin and provide overall healing benefits. Sales are expected to begin in the first quarter of 2018 and are projected to generate approximately $2 million annual revenues.
The Patented Pending CBD or Cannabinoids product line. The Company would combine its two Patent pending technologies, the MMJ Patent, a patent for delivering medical marijuana / cannabis to the body by applying the medication onto the skin in an ointment base topical solution and the second Provisional patent for enhanced antibiotic and drug delivery for "Aqueous Topical Applications" for human and veterinarian uses. The Company would combine its proprietary solutions with CBD's and other natural products that treat psoriasis, fibromyalgia for pain and other ailments. The Company plans to continue to expand its (IP) "Intellectual Property Portfolio" in 2018.

CBD is a compound found in the Cannabis family of plants such as hemp. CBD is combined into our topical body care products like Elixr's Topical Balm, Serum, and Spray.
CBD is an antioxidant. Antioxidants protect our skin from damaging exposure to smoke, UV rays, and environmental pollutants. Cannabinoid receptors are located all throughout the skin, Topical CBD products interact with those receptors resulting homeostasis and healing. Studies show that CBD can help treat an array of skin conditions such as eczema, psoriasis, rosacea, and acne.
| "What is Intellectual Property?" - A Publication by the World Intellectual Property Organization -
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
