Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
What I am certain is that the PPS will fly over $0.07 cents for sure
Yes it’s been a long time but sold some at times for a nice profit hopefully he comes through again, I think he will but will definitely sell enough for a profit and ride free plus have 1 mil locked in at the lottery price $1.00 $$$$$
Sub penny will be history for ever after filings.
And Beyond!!!!
And Beyond!!!!
MLK Alias Born 04/08/2022
Search Member Posts
2023
About
Latest Posts
Boards Posted On
Boards Moderated
My Stocks
FInal pump...he'll be out Monday....on to the next Grift...
I believe he will deliver. Been a slow multi year start
I don’t think that’s the way it works. The fillings does not have to do nothing with the states contracts. The fillings are just documents showing what the company has been doing, nothing else: Transparency to the people… The contracts could be already done without him filling in. Do you get the logic. Relax, the Doc seems to have made the few first steps forward to the right direction. There’s not reason for failure from this point. He will deliver
I got you $weet$$$
Risk equals rewards my brothers, that’s how it works
Great run on Friday, more to come soon!
You can pump all you want but no one running out to buy shares based on your reco
I'm sure ORCA will thank you and go easy
Go easy and watch strong opening at premarket , can not trade premarket but 9:30AM bid and ask will be nasty upside move.They may 🛑 stop or halt for trading if huge buy orders come in.
Do whatever you want I am looking big jackpot on this stock
Go easy and watch strong opening at premarket , can not trade premarket but 9:30AM bid and ask will be nasty upside move.They may 🛑 stop or halt for trading if huge buy orders come in.
Do whatever you want I am looking big jackpot on this stock
Go easy and watch strong opening at premarket , can not trade premarket but 9:30AM bid and ask will be nasty upside move.They may 🛑 stop or halt for trading if huge buy orders come in.
Trust but verify is always necessary, especially with this company. However, they did make the below X post on February 1st. Thus, it will, as a minimum, have to be in the 1Q24 Financial Report issued around May 15, 2024 (hopefully). Admittedly I do not know the applicable rules, but it could possibly be added as supplementary information in 2023 Annual Report, expected on or before March 30, 2024. By the way, $1.2M is all but about $0.2M of the projected Accounts Receivable balance through Jan 2024, which would be significant step in right direction. However, trust but verify still applies and is necessary.

Cannot receive if not due, hmmm.
Nice 2 C the board so busy sharing thoughts
What good is adding the rest of the states if they don’t ever pay for the licensing fee?
Showing a line as Accounts Receivable and then actually showing it as received and showing an increase in Cash On Hand with no debt is what matters!
Those are with the CEO’s other company Wound Pros. And Wound Pros does not pay the bill. Just a little smoke and a lot of mirrors
Yes Bud that would be very attractive 2 buyers
We need the rest of the states added, where are they !???
Relax big fish He has no reason not 2 flie
MONDAY IS THE 25TH.AND THE FOLLOWING MONDAY IS THE 1ST OF APRIL.WE BETTER HAVE FILED BY THEN.IF NOT THEN BY END OF APRIL.OS THERE IS A TON OF RISK.BUT IF THEY DO FILE.BIG REWARD.
India would be a good start 1419,66 million
States can be compared with countries given their seize.
I AM TALKING ABOUT BIG ONES OUTSIDE THE COUNTRY.
There are already 14 contracts with 14 states.
THERE WILL BE NO CONTRACTS UNTIL HE FILES THE REPORTS FIRST.
What is 1 M for a whole country to import a product with many years of R&D?
hans1111 I think the license price is high
I don't dwell on the past . So negative
Estimation. Do you agree or do you have another idea?
Considering your track record over the last 6 years…I’ll go with you being 100% wrong
One license? $ 1,2 M a year ?
They already have licensing agreements within 14 states. More or less a calculation of what could be if this goes worldwide. Think of the Egypt journey of Otiko. Means they are buzzy to concyer the world. Remember this article too.
ViaDerma, Inc.
ViaDerma, Inc.
LOS ANGELES, Calif., Oct. 24, 2023 (GLOBE NEWSWIRE) -- ViaDerma, Inc. (OTC Pink: VDRM) is pleased to announce several new and exciting business updates for current and prospective shareholders, customers, partners, and healthcare providers.
The company is currently in the final phases of completing our stability testing requirements with our third-party testing facility. Based on the feedback we received from our testing partner last week, the company is on pace to have everything finalized by the end of October. The stability testing requirements are a crucial step in order to greenlight the approvals to distribute our lead product Vitastem Ultra throughout multiple countries in the Caribbean and Middle East, along with India - the largest populated nation on Earth with 1.486 billion people, which recently surpassed China earlier this year.
ViaDerma’s management team has been working diligently with our new contract manufacturer to ramp up production capabilities in order to accommodate several large volume orders of 250,000 to 300,000 plus units per country this year. ViaDerma’s Founder & CEO, Dr. Christopher Otiko is expected to travel to Egypt in Q4 to present Vitastem Ultra to their government officials as the company is expecting to fulfill purchase orders of 250,000 units or more in Egypt alone.
Upon the completion of stability testing for Vitastem Ultra, we will be able to begin distributing Vitastem internationally by fulfilling initial purchase orders throughout the Caribbean, Middle East, and India where the demand for Vitastem is highest. Based on the discussions with our sales partners, ViaDerma is planning to initially distribute 300,000 plus units per country yearly and expect to scale the volume of units delivered throughout each country.
Our new and much larger product manufacturer located in the US is crucial for the company to meet the growing demand for new & innovative topical antibiotics like Vitastem Ultra that are required to combat the ever-growing presence of superbugs like staph & MRSA that arise due to HAIs (healthcare-associated infections) throughout healthcare facilities globally. As these types of infections create numerous additional health risks to patients, hospital staff, and relatives of the patients, Vitastem is a proven solution with this serious matter.
Due to several ongoing updates and FDA drug application requirements for Nupelo’s use of the active ingredient, Minoxidil, our FDA-registration status is currently on hold for the time-being. ViaDerma is looking into utilizing a natural active product that can offer the same results as Minoxidil.
According to Dr. Otiko, “Due to the requirements for a new and very lengthy drug application required by the FDA and for sake of bringing Nupelo to market faster, we have been actively looking into incorporating an alternative natural active ingredient that can do the same thing as Minoxidil but will not require such a lengthy process in order to finalize the registration of Nupelo with the FDA.”
The company can either go with a new natural formula or use Minoxidil and sell Nupelo to compound pharmacies. Updates for Nupelo will be forthcoming once a viable solution is met to accelerate distribution and sales nationwide.
Furthermore, ViaDerma is advancing conversations and negotiations with the VA (Veterans Affairs Department), along with a very large HMO about distributing Vitastem Ultra through their pharmacies. Due to confidentiality requirements, we cannot disclose their name until given the okay, however, this tier-1 HMO leader has a large national presence with over 650 healthcare facilities, 90,000 physicians and nurses that service nearly 13 million people throughout the United States. Updates will be forthcoming.
The company is also pleased to announce that we have expanded licensing agreements and are now up & running in 14 states and expect to keep expanding into more states throughout the year.
“2023 is shaping up to be a very pivotal year for ViaDerma and we are extremely excited about the future of our business moving forward. The growing demand for Vitastem is testament to the powerful healing properties this broad-spectrum topical antibiotic offers to healthcare globally. Our team is working around the clock to ensure operations, manufacturing, and product distribution are sound and ready to meet this growing demand around the world and fulfill purchase orders in the hundreds of thousands per country with the expectations to continually compound sales growth throughout 2024 and beyond.” said Dr. Otiko.
The ViaDerma management team deeply appreciates all the ongoing feedback and patience from shareholders, partners, customers, and healthcare providers. We are extremely grateful and excited about the progress we have made this year and for the future of the business over the next year. We look forward to sharing several more updates as soon as possible with everyone as soon as they arise.
About ViaDerma, Inc.
ViaDerma, Inc. (OTC: VDRM) is a publicly traded specialty pharmaceutical company committed to bringing new products to market and licensing its innovative transdermal drug delivery technology solutions to current leaders in the pharmaceutical industry in a wide variety of therapeutic areas. For more information, please visit: https://viaderma.com
Any forecast of future performance is a "forward looking statement" under securities laws. Such statements are included to allow potential investors the opportunity to understand management’s beliefs and opinions with respect to the future so that they may use such beliefs and opinions as one factor among many in evaluating an investment.
Contact information:
Investor Relations
Email: ir@viaderma.com
Phone: 310-734-6111
Follow ViaDerma on Twitter: https://twitter.com/viaderma
Follow ViaDerma on Facebook: https://www.facebook.com/ViaDermaLicensing
hans Where did you get those figures?
If one license for one country is $ 1,2 M a year, how much will that be for 195 countries? Exactly $234.000.000 a year. Without sales, only the licenses!
Budman I agree 100%
I do believe that we are finally on the cusp of a steady year long price improvement. A few good contracts and easily over .25 by years end.
He's a Grifter...he will be gone next week...
3/22. VDRM VWAP=$.0113 VOLUME=17.5mm Offer 4 to 3
3/21. VDRM VWAP=$.0089 VOLUME=204k Bid 7 to 1
3/20. VDRM VWAP=$.0086 VOLUME=680k Bid nearly all
3/19. VDRM VWAP=$.0089 VOLUME=1k
3/18. VDRM VWAP=$.0082 VOLUME=431k Bid-side nearly all
3/15. VDRM VWAP=$.0085 VOLUME=337k Bid-side nearly all
3/14. VDRM VWAP=$.0081 VOLUME=1.2mm Bid-side 8x
3/13. VDRM VWAP=$.0087 VOLUME=1.7mm id-side 5x
GREAT DAY, KNEW IT WAS COMING!
30 yrs. trading My posts are what I believe.
|
Followers
|
619
|
Posters
|
|
|
Posts (Today)
|
3
|
Posts (Total)
|
74600
|
|
Created
|
10/27/08
|
Type
|
Free
|
| Moderators | |||

ViaDerma's proprietary transdermal delivery system allows for rapid mass transfer of the pharmaceutical active ingredient across the skin and into the body to provide immediate localized therapy.
The technology allows transfer of chemicals through the stratum corneum (outer layer of skin) with a diffusion constant which is 10,000 times higher than the diffusion constant which characterizes water movement through the stratum corneum.
This enables ViaDerma to pair almost any active ingredient with the technology and provide rapid transport of the medicine right to the site of action.
The first product is a broad spectrum tetracycline-based topical antibiotic is the only antibiotic in the world that kills bacteria both a physical and a chemical mechanism. All known antibiotics (other than ours) primarily use only a chemical mechanism of kill. The physical mechanism of kill is a key feature of what we call Rapid Active Ingredient Delivery System (RAIDS). One result of RAIDS is that tetracycline is carried in higher concentrations, more quickly, to and through the cell walls, where the tetracycline can become more effective than if conventional antibiotics were used. Conventional antibiotics require more time (usually prescribed for 5 to 7 days for best results), whereas our tetracycline-based products usually produce desirable results in 24 hours (or less) because of the RAIDS effect.

A second important result of RAIDS is that the topical antibiotic kills all harmful Gram positive and Gram negative bacteria that have been available for testing. We believe this is the world’s strongest broad-spectrum topical antibiotic available.
The potential commercial impact is immense. Drug developers believe it takes much longer for bacteria to develop drug resistance to a physical kill mechanism. This is because it is relatively easy for bacteria to change their response to a chemical threat, but it takes numerous generations for bacteria to grow a new kind of cell wall structure to respond to a physical threat.
Our novel approach to overcome drug resistance of antibiotics is designed to sustain the effectiveness of antibiotics and other topical drugs for many years. This gives new topical drug products a longer useful lifetime and therefore more commercial value. This technology, when licensed to larger pharmaceutical companies, may provide stronger incentive for the discovery and development of new antimicrobial drugs. In recent years, the dearth of new antibiotics has been largely due to the uncertain new-drug commercial lifetime which is diminished when bacteria develop immunity to that drug.
In addition the technology can be licensed to other companies to convert existing oral drugs to transdermal medications extending the profitably of the existing drug.


We are developing products in the following fields of use;
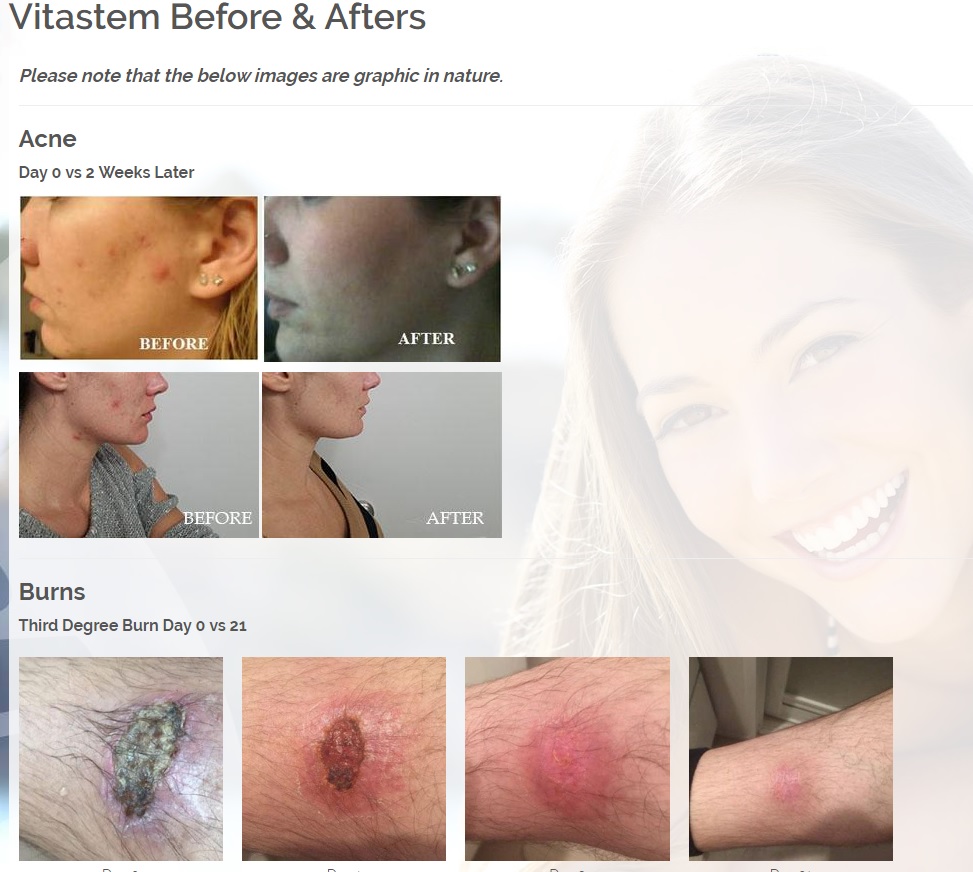
Treats all types of bacteria including MRSA, VRE and other flesh eating bacteria. Fights drug resistant bacteria. $6B/year global market.

Onychomycosis, a fungal infection of the toenails, is a major health problem. It is estimated that there are in excess of 40 million sufferers with this condition in the USA. It is a problem throughout the world. A recent European study showed that the prevalence of Onychomycosis may be as high as 26.9%. Fungal resistance can occur when the oral antifungal agents are used on a long-term basis. Topically applied antifungal drugs may work somewhat better adjunctive to surgical removal or chemical dissolution of the nail plate. Yet, this often ineffective and traumatic procedure leaves the subject without a nail for months at risk for re-infection. Given the limitations of current treatment options in this $3B market, there is a great need for a simple, nontoxic and effective alternative treatment. Estimated Global prevalence rate: 140 million.
Treats symptoms of Influenza which is a$4B/ year global market: Common
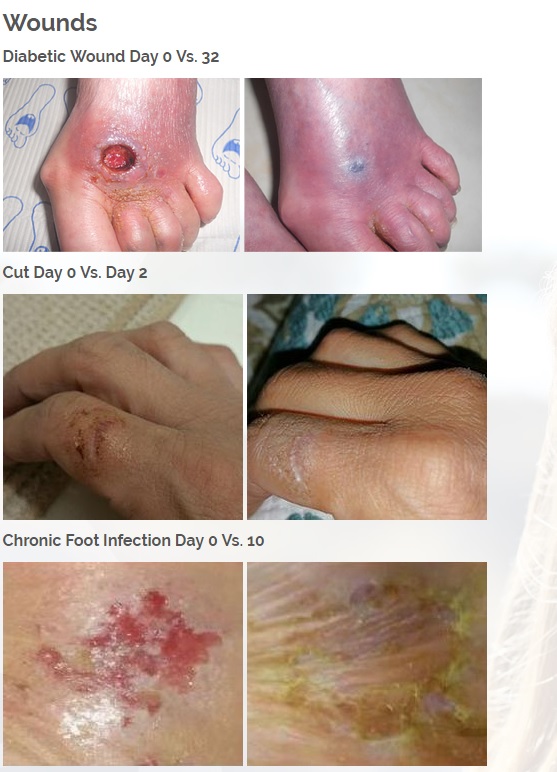
Diabetic foot wounds.

Per 3/8/18 PR: ViaDerma’s technology is currently being used in Elixr Cannabis products; Topical Balm, Topical Serum and Topical Spray. Sales have begun in Canada.
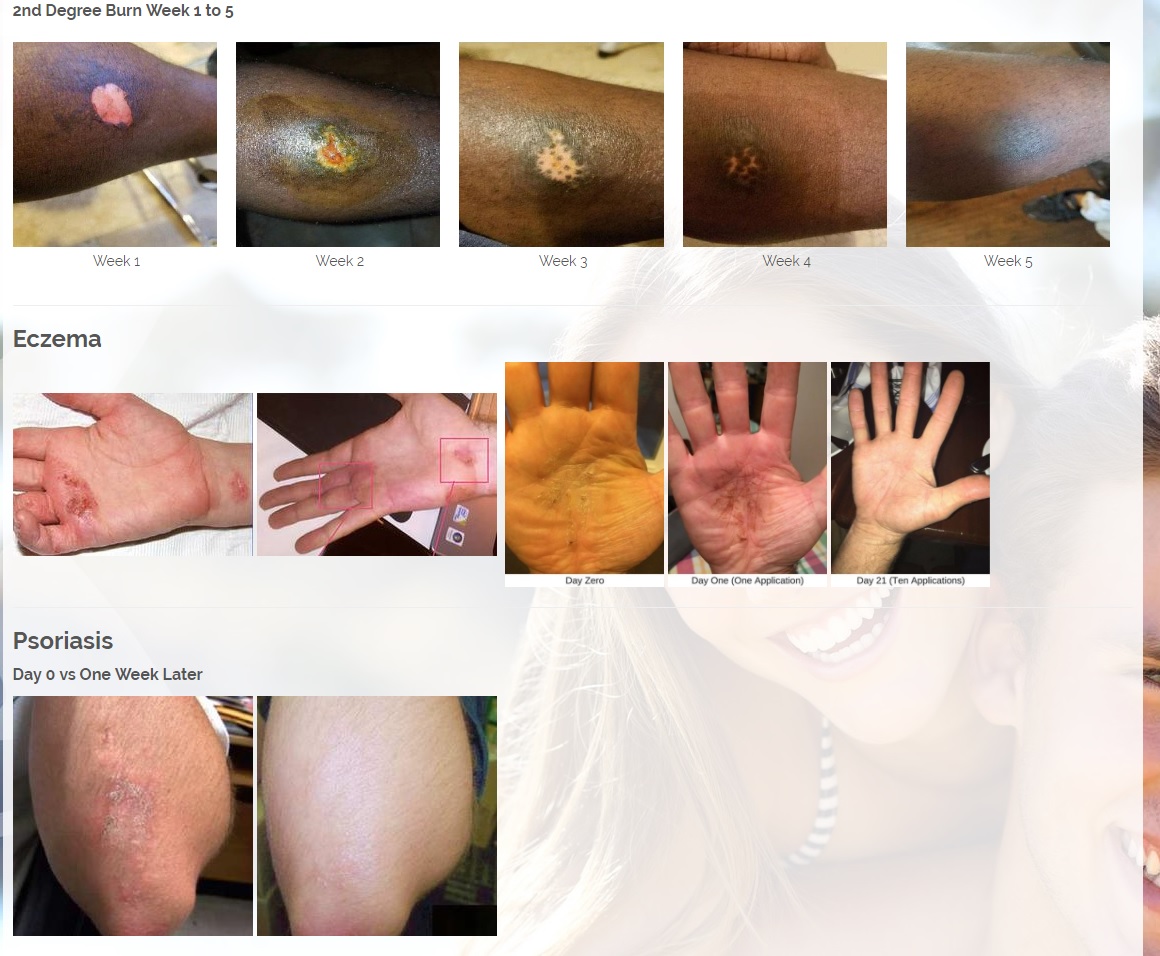
Per 12/7/17 PR: The Company has signed an MOU and will start production of its Patent Pending CBD or Cannabinoids product line. The Company would combine its two Patent pending technologies, the MMJ Patent #62466209, a patent for delivering medical marijuana / cannabis to the body by applying the medication onto the skin in an ointment base topical solution and the second Provisional patent # 62433964 for enhanced antibiotic and drug delivery for "Aqueous Topical Applications" for human and veterinary uses. The Company would combine its proprietary solutions with CBD's and other natural products that treat psoriasis, fibromyalgia for pain and other ailments.
Per 1/23/18 PR: The Company has also a licensing and distribution agreement in place with its Canadian partners to produce a CBD Topical Serum, which has a 92% absorption rate powered by ViaDerma's Patent Pending Dual Carrier Technology. The CBD and Terpene profile (enriched with specific essential oils and vitamins C, and D) is crafted to alleviate Chronic Dry Skin, Psoriasis, Eczema, Rosacea, and many other skin conditions. The serum has anti-inflammatory, anti-bacterial, and chronic pain relief properties that are absorbed by the skin and provide overall healing benefits. Sales are expected to begin in the first quarter of 2018 and are projected to generate approximately $2 million annual revenues.
The Patented Pending CBD or Cannabinoids product line. The Company would combine its two Patent pending technologies, the MMJ Patent, a patent for delivering medical marijuana / cannabis to the body by applying the medication onto the skin in an ointment base topical solution and the second Provisional patent for enhanced antibiotic and drug delivery for "Aqueous Topical Applications" for human and veterinarian uses. The Company would combine its proprietary solutions with CBD's and other natural products that treat psoriasis, fibromyalgia for pain and other ailments. The Company plans to continue to expand its (IP) "Intellectual Property Portfolio" in 2018.

CBD is a compound found in the Cannabis family of plants such as hemp. CBD is combined into our topical body care products like Elixr's Topical Balm, Serum, and Spray.
CBD is an antioxidant. Antioxidants protect our skin from damaging exposure to smoke, UV rays, and environmental pollutants. Cannabinoid receptors are located all throughout the skin, Topical CBD products interact with those receptors resulting homeostasis and healing. Studies show that CBD can help treat an array of skin conditions such as eczema, psoriasis, rosacea, and acne.
| "What is Intellectual Property?" - A Publication by the World Intellectual Property Organization -
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
