Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Juicy!!! Ready for blast off
$.0086 All evidence? Here is some EVIDENCE:
https://www.barchart.com/stocks/quotes/VDRM/overview
Really? All evidence to the contrary. Fantasyland.
Sure wish someone would harpoon the whale
BRO IS NOT DONE YET ORCA ON THE WHEEL:)))MMs KNOW WHAT'S UP WHEN ORCA ON THE BIDS:)))
THE ORDER WAS IN ALL DAY.ZERO SHARES GOT FILLED
THEY DO NOT EVEN TEST MY BIDS ANYMORE.BECAUSE THEY KNOW BETTER.THAT THEY ARE REAL.NOT ONE SINGLE SHARE.UNREAL.
Expired Buy 500000 VDRM Limit 0.0086 -- 08/18/22 16:07:04 08/18/22
Order No. 9195346723 Viaderma, Inc. Common Sto...
Entered:10:21:02 08/18/22
No, absolutely not! VDRM goes much higher!
It did, I hope you took profits!!!!
$.0086 I love how VDRM is unfolding, with small issues, YET SHARE PRICE ASCENDING! VDRM is going to rock!
VDRM is a tiny company with few resources. Dr. Otiko has it coming and I am great with his leadership!
Relax...PATIENCE!
I would rather hear nothing than lots of lies
Filings coming soon!
Added some $VDRM today.
Something is cooking here.
Mayor move ahead!
WHO IS BUYING EVERY DAY????THIS IS CRAZY.I MEAN NO FILINGS AND THEY ARE BUYING DAILY.
Going to one that no one should miss out on
Especially if yhave been here a while
There aren’t easy or difficult solution in this one but to wait to get even and GTFO
Oh ,,when some were dumping at 55 and 6 I was adding to my already fat position. Hmmm 55 to 96,,big board stocks do not move like that in 6 months, maybe six years. Good with my investment.!!!
$.0096 Bullshit, Dr. Otiko is in charge and will get the filing taken care of. VDRM is fine and news will come soon.
For those who don't like how VDRM is being run, there are easy solutions!
I hear ya. OTC's are frustrating!
Oh I am still very optimistic. Just annoyed the guy in charge who holds almost 50MM shares can’t file paperwork on time lol!
Been saying for a while, Dr. O is in the dancing business only... He has to pass the flag to someone else more capable to run a business and he can go off the radar to do his own research, if he wants to. He is not a business man, he will burn the company to the ground, if not close there yet... I hope he set aside his ego and recognize where his forte is at.... Time to sell the company or step down!!!! Where are the board of shareholders when you need them At?
LMFAO!!!!
There is no "LEGIT" reason on delaying any financials............
Can you please let us know how many were missed in the past?
All "LEGIT" reasons to, eh?
PAAAALLLEASE.
SMFH
Agree on hiring someone that knows what they are doing. The Dr. Seems to be a jack of all trades and master of none. He has too much going on and is clearly not focusing on VDRM
I totally agree with you I was just poking fun at the situation but the doc needs someone who can run this company properly
inexcusable. smh. inexcusable.
Calm down boys he's out of the country on a holiday and just forgot
This is inexcusable!!! No reason not to file the NT-Late Filing…
Lmao. Now that’s funny….
Tried to call IR on his cell Mail box full
Bid $.0093 for 874K...MARKET REALLY WORRIED! LOL
Has anyone reached out to IR?
Very telling. It will be interesting and entertaining reading the excuses that are sure to come.
BS,THE SCAMBAG DID NOT EVEN FILE LATE EXTENTION.
The delay may be legit VDRM news any day
BAD NEWS,VDRM DID NOT FILE THE Q2 REPORT.AND GOT THE YIELD SIGN ON.AND DID NOT FILE FOR AN EXTENTION EITHER.HMMMMM.THAT POS OTIKO.IS UNREAL.
https://www.otcmarkets.com/stock/VDRM/disclosure
Dr. O won't do nothing mate, as usual. Flip this scam
$.0099 earlier....I LIKE! Dr. Otiko will come through on this!
Technicals. Oh my
VDRM is not a tech stock.
Techs looking juicy. If we have some decent news this thing could SOAR
Yes big things coming but that .01 is a huge hurdle currently. To be blown aside . Would like it to be today
$.0094 Looking good, VDRM! There might be another, I doubt it, but folks had their chance!
Before claims are substantiated the pps will be back in the .03 range… People in “my camp” see an opportunity at these levels. Not everyone has to follow or believe/gamble
Large orders of what? I don't remember reading about a market ready product anywhere. Alleged (great) test results have not even been released yet and they are way passed the published due date. Whatever. Always the same unsubstantiated noise and nonsense.
With the great testing results and large orders shipped, 3rd quarter should be very positive!

ViaDerma's proprietary transdermal delivery system allows for rapid mass transfer of the pharmaceutical active ingredient across the skin and into the body to provide immediate localized therapy.
The technology allows transfer of chemicals through the stratum corneum (outer layer of skin) with a diffusion constant which is 10,000 times higher than the diffusion constant which characterizes water movement through the stratum corneum.
This enables ViaDerma to pair almost any active ingredient with the technology and provide rapid transport of the medicine right to the site of action.
The first product is a broad spectrum tetracycline-based topical antibiotic is the only antibiotic in the world that kills bacteria both a physical and a chemical mechanism. All known antibiotics (other than ours) primarily use only a chemical mechanism of kill. The physical mechanism of kill is a key feature of what we call Rapid Active Ingredient Delivery System (RAIDS). One result of RAIDS is that tetracycline is carried in higher concentrations, more quickly, to and through the cell walls, where the tetracycline can become more effective than if conventional antibiotics were used. Conventional antibiotics require more time (usually prescribed for 5 to 7 days for best results), whereas our tetracycline-based products usually produce desirable results in 24 hours (or less) because of the RAIDS effect.

A second important result of RAIDS is that the topical antibiotic kills all harmful Gram positive and Gram negative bacteria that have been available for testing. We believe this is the world’s strongest broad-spectrum topical antibiotic available.
The potential commercial impact is immense. Drug developers believe it takes much longer for bacteria to develop drug resistance to a physical kill mechanism. This is because it is relatively easy for bacteria to change their response to a chemical threat, but it takes numerous generations for bacteria to grow a new kind of cell wall structure to respond to a physical threat.
Our novel approach to overcome drug resistance of antibiotics is designed to sustain the effectiveness of antibiotics and other topical drugs for many years. This gives new topical drug products a longer useful lifetime and therefore more commercial value. This technology, when licensed to larger pharmaceutical companies, may provide stronger incentive for the discovery and development of new antimicrobial drugs. In recent years, the dearth of new antibiotics has been largely due to the uncertain new-drug commercial lifetime which is diminished when bacteria develop immunity to that drug.
In addition the technology can be licensed to other companies to convert existing oral drugs to transdermal medications extending the profitably of the existing drug.

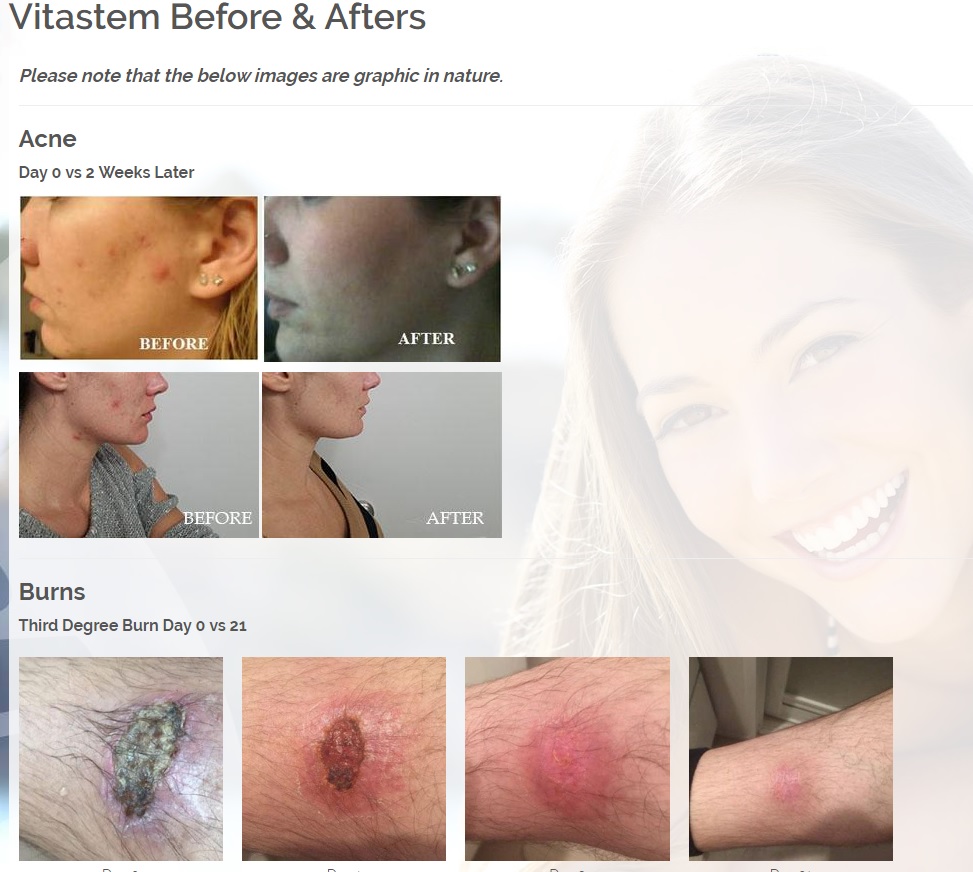
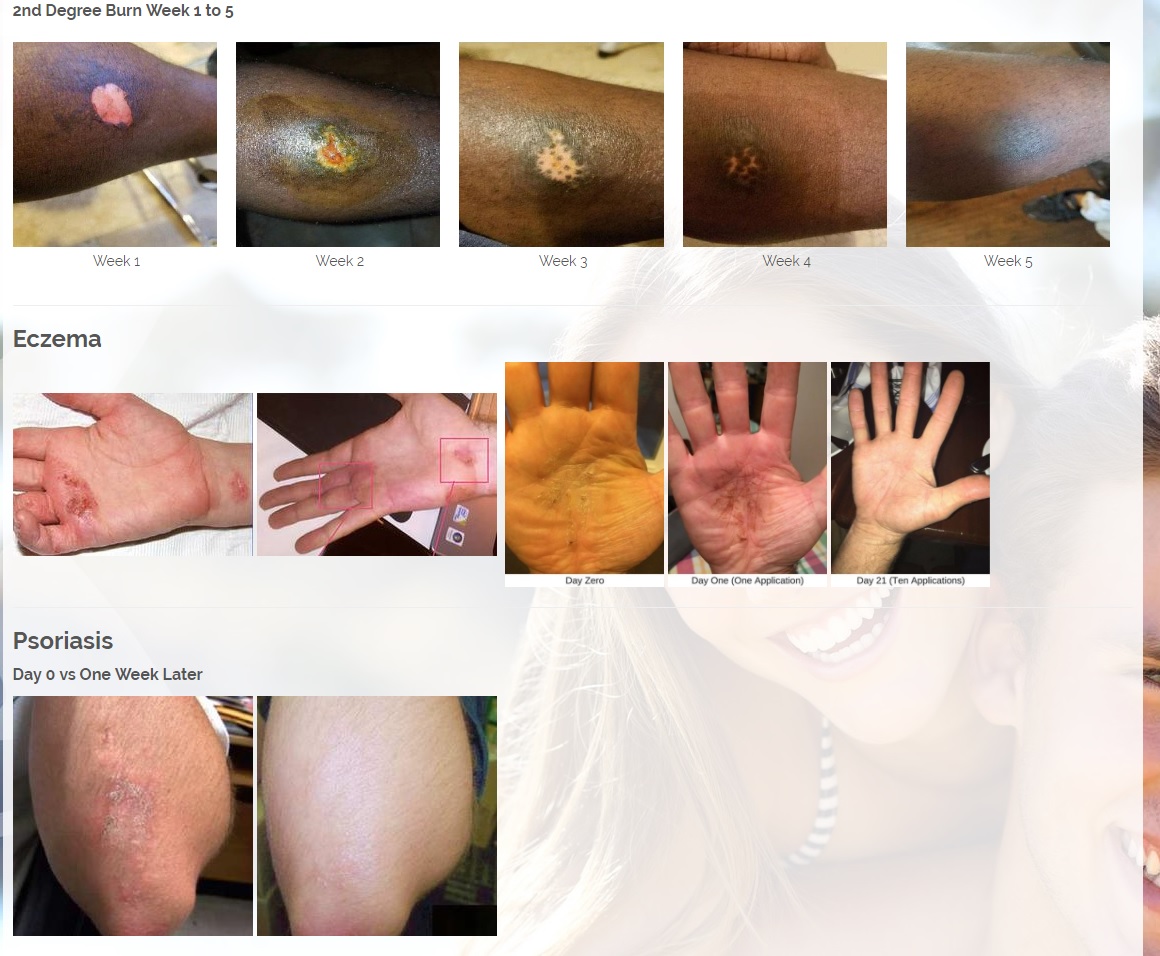
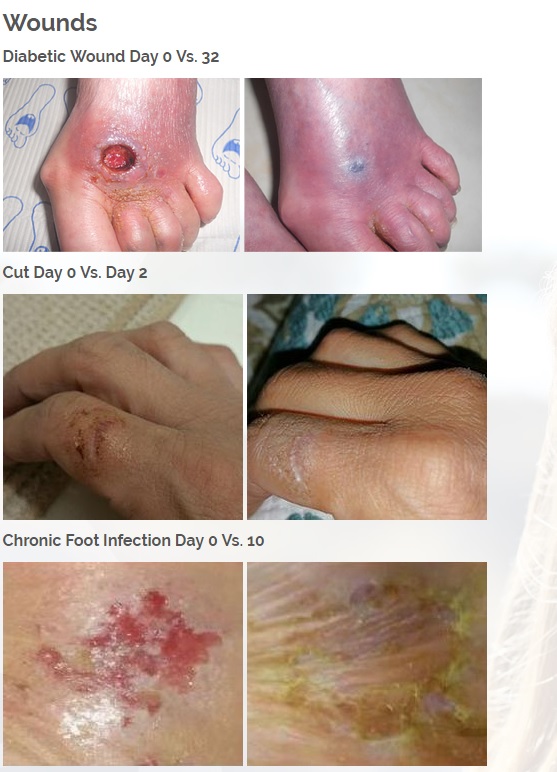

We are developing products in the following fields of use;
Treats all types of bacteria including MRSA, VRE and other flesh eating bacteria. Fights drug resistant bacteria. $6B/year global market.

Onychomycosis, a fungal infection of the toenails, is a major health problem. It is estimated that there are in excess of 40 million sufferers with this condition in the USA. It is a problem throughout the world. A recent European study showed that the prevalence of Onychomycosis may be as high as 26.9%. Fungal resistance can occur when the oral antifungal agents are used on a long-term basis. Topically applied antifungal drugs may work somewhat better adjunctive to surgical removal or chemical dissolution of the nail plate. Yet, this often ineffective and traumatic procedure leaves the subject without a nail for months at risk for re-infection. Given the limitations of current treatment options in this $3B market, there is a great need for a simple, nontoxic and effective alternative treatment. Estimated Global prevalence rate: 140 million.
Treats symptoms of Influenza which is a$4B/ year global market: Common
Diabetic foot wounds.

Per 3/8/18 PR: ViaDerma’s technology is currently being used in Elixr Cannabis products; Topical Balm, Topical Serum and Topical Spray. Sales have begun in Canada.
Per 12/7/17 PR: The Company has signed an MOU and will start production of its Patent Pending CBD or Cannabinoids product line. The Company would combine its two Patent pending technologies, the MMJ Patent #62466209, a patent for delivering medical marijuana / cannabis to the body by applying the medication onto the skin in an ointment base topical solution and the second Provisional patent # 62433964 for enhanced antibiotic and drug delivery for "Aqueous Topical Applications" for human and veterinary uses. The Company would combine its proprietary solutions with CBD's and other natural products that treat psoriasis, fibromyalgia for pain and other ailments.
Per 1/23/18 PR: The Company has also a licensing and distribution agreement in place with its Canadian partners to produce a CBD Topical Serum, which has a 92% absorption rate powered by ViaDerma's Patent Pending Dual Carrier Technology. The CBD and Terpene profile (enriched with specific essential oils and vitamins C, and D) is crafted to alleviate Chronic Dry Skin, Psoriasis, Eczema, Rosacea, and many other skin conditions. The serum has anti-inflammatory, anti-bacterial, and chronic pain relief properties that are absorbed by the skin and provide overall healing benefits. Sales are expected to begin in the first quarter of 2018 and are projected to generate approximately $2 million annual revenues.
The Patented Pending CBD or Cannabinoids product line. The Company would combine its two Patent pending technologies, the MMJ Patent, a patent for delivering medical marijuana / cannabis to the body by applying the medication onto the skin in an ointment base topical solution and the second Provisional patent for enhanced antibiotic and drug delivery for "Aqueous Topical Applications" for human and veterinarian uses. The Company would combine its proprietary solutions with CBD's and other natural products that treat psoriasis, fibromyalgia for pain and other ailments. The Company plans to continue to expand its (IP) "Intellectual Property Portfolio" in 2018.

CBD is a compound found in the Cannabis family of plants such as hemp. CBD is combined into our topical body care products like Elixr's Topical Balm, Serum, and Spray.
CBD is an antioxidant. Antioxidants protect our skin from damaging exposure to smoke, UV rays, and environmental pollutants. Cannabinoid receptors are located all throughout the skin, Topical CBD products interact with those receptors resulting homeostasis and healing. Studies show that CBD can help treat an array of skin conditions such as eczema, psoriasis, rosacea, and acne.
| "What is Intellectual Property?" - A Publication by the World Intellectual Property Organization -
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
