Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
It looks like another one Otikos back door companies besides wound pros is called national institutes of health (NIH)
Ya … the “doc’s” new product is “tetracyte” not Vitastem. Wonder who is producing it and who is receiving all the proceeds from the sale of the “new” INNOVATIVE product. It sounds like the EXACT same description of Vitastem Ultra but labeled tetracyte for wound pro’s exclusively. WTF is this sh!t. Are we the share holders getting back door swindled ??? Why does the same product need to be labeled differently ??? Hey doc … Why doesn’t wound pros use and sell Vitastem Ultra so both of YOUR companies benefit (especially VDRM) for you AND the shareholders sake ???
Even a blind hog finds an acorn now & then
You can ask the editor:
Editor In Chief IKENNA EMEWU For adverts, please call +234-812-099-7006 This site is owned and operated by the AFRI-CHINA MEDIA CENTRE LIMITED
Why no mention of VDRM or Viaderma in the first article? Almost as if the Doc's privately held company is competing against VDRM.
Thanks for pointing that out. I only checked the quarterly report, Unfortunately, it is of the same time period (2017/2018). With ONLY <$0.7K of product sales for each of last 6 quarters, something needs to break the product sales revenue into 4 digits (5 digits, 6 digits, etc.). Maybe, it will be that after all this time, if the asserted information is ultimately documented in financial report as spot on.

The point would be, with no anywhere recent, up to date information in the 1Q24 financial report, or any other "official" source, how would anyone know, legitimately, about sales in Canada, even if true?
Old news. Please get to a point. What point are you trying to make? Chuckles!!!
Tell us something we don’t know
For what it is worth, since Section 4) Issuer's Business, Products and Services is 3-7 years out of date, the only mention of Canada (via word search), in 1Q24 financial report, is:
On January 1, 2018, the Company entered into a licensing agreement with SSP Asset Management Corp., an organization incorporated under the laws of Alberta, Canada (“SSP”). The license granted SSP the right to use the Company’s transdermal technology for use within certain products. The Company will receive Fifty Percent (50%) of the net profits of any products sold by SSP. The term of the license is 25 years and is exclusive for a minimum of two years on any new products developed by SSP. The Company also issued 20 million restricted common shares to the shareholders of SSP to assist in the development of the new products.
Are you talking about the CBD Cream which they PR’d years ago????
1,000 units or $1,000 worth of units??
How do you know this?
They got the product, right?
Excellent choice.
Hopefully sooner!
I understand. It comes from a place of feeling sorry for our colleagues. I’ve actually made money on this and feel bad because the majority has not…
I smoked KWBT back in 2020-21
I’ve been quite lucky on these gambles. But most people are not!
One would hope the OTC folks are okay with the risk
I sympathize with what you said but frankly you sound a little naïve when you said: he doesn’t realize if there are people in here that has lost serious money. Who cares? It is the mere reason why many of these OTCM CEOs come around here because the easy access to dumb $$$. And the ones in here are gamblers, nobody is that naïve to not even consider the risks in OTC. Don’t get me wrong, I ain’t trying to justify our Dancer but I couldn’t resist when you said such.
Bud I’m playing golf with a couple of idiots right now having a hell of a time,
If company is even a smidget of legit why would you be worried about a negative (truthful) comment or two? Will it really get in the way of the next pump a year from now lol
No just get tired of the hang ons that have said they moved on but still here. They need more friends.
Is he interfering with your bubble?
Get out the rest of the way.
The arrogance of this guy blatantly lying (which in my opinion is defrauding people) is astounding…
Does he not realize there are people that have lost serious money here? He lives in California where criminals are let off the hook every day. Has to drive to and from work… Clinic and even personal residence can be found online… friendly advice here Dr. you might want to be careful stealing from people, even if you did it in a “legal” way…
Warning signs have been here for years with all the “deals” and other nonsense. Think I’m one of the lucky ones that got 90% out a couple months ago. Feel bad for those that “believed”.
I cannot conceive of even a shoe string argument as to how the 95.5M excess shares (6.2% of company, including preferred share impact) could be deemed merited and not insultingly, egregiously unjustified, by non-CEO stockholder, with the reported performance record of CEO, over last nearly a decade.
Note: Licensing is first column. Product sales is second column.

If CEO really wanted to give more complete air of changed ways, ethics, respectability, and responsibility to shareholders, he would return the unwarranted 95.5M excess shares he "gifted" himself, beyond the 4.5M shares, on 03/28/23, necessary to pay off the legitimate $118K principal and interest (on 03/28/23).
It's a scam they obviously know lol
Even though the Company never responds, maybe they do hear things...like rumblings of investigations and SEC noncompliance.
We should all put together a list of empty promises and send it to him and Rich. They won't respond of course, but they'll know that we know that they are self-dealing, BSers, conducting themselves in a (possibly) fraudulent manner and that we are watching.
Right on point. Forgot, weren't they supposed to up list VDRM too? How long ago was that? 1-2 years ago i think.
It's not ignorance. Its borderline fraud, if not outright fraud. IMO.
All that hard work over years? And what the hell have you actually produced otiko? Self dealing accounting swaps to his other company Wound Pros?
No overseas deals signed to date either. No march shipments. But now they are "hiring 3 sales reps"? GTHOH.
What did the 250 mill share 25% dilution get us? Where is the $$$?
Agreed and the icing on the cake was how they thought no one would be pissed off about reading that Dr. Otiko took the only revenue the company has supposedly made as a loan. And then on top of that says how he has worked tirelessly through the years. Does he not even have a clue that it is the investors throughout the years who have carried the company as well by buying shares of the company’s stock with Hard Earned MONEY?????
I mean come on now.
He cannot be that ignorant of a man can he???
And he SHOULD BE WORKING round the clock since he does own over 150 million shares I believe that once the company becomes fully audited and reporting can sell those shares through notifications.
Exactly. Very fishy accounting practices overall with no transparency into business activities and constant BS PRs from the IR guy Richard Inza and Otiko.
IMO, I would not be surprised if they're being investigated already by the SEC. Years of producing nothing but BS PRs that try to hide behind a safe harbor clause.
Huge Dubai deal? Nothing. Nupelo? Nothing. Shipping 200,000 products to India and Egypt in March? Nothing.
Otiko claims Vitastem is the strongest topical antibiotic in the world. If that were the case, then you would think that every doctor and hospital in the US and around the world would be buying all of it up. So why are they not?
Taking into account the volume and price action today, investors have spoken...the PR has done nothing to garner further interest in the stock. The pps is back at around .007, where it's averaged since May 15th. Maybe the pps would be worse without the PR, who knows. In any event, investors need evidence of a transaction of substance, something positive and material to give investors a reason to invest in this stock. Right now, having the "best antibiotic on the planet" is just hyperbole. Investors need corroboration! "Pfizer announces that Pfizer and Viaderma have entered into a marketing and distribution agreement for Viaderma's flagship product Vitastem... The Agreement provides for substantial milestone payments of __________based on sales 1,000,000 units... Pfizer worldwide director of market distribution is excited about its partnership with Viaderma and looks forward to a long prosperous future".
What will not move the needle IMHO is, "Viaderma has entered into marketing and distribution agreement with a national pharmaceutical company to service broad regions of Western Europe, it is our hope that shipping will begin in early Fall." (Note no specific third party mentioned, nonspecific regions, "hope" of future shipping). That may have excited the faithful in the past, but nothing (yes nothing ever) has come to fruition and investors need more than the same old illusory promises.
Well, yes 100% will take it Guess it’s better than saying nothing but in the past news like this from the Dr. would have sent this stock off to the Races $$$$$$ .problem is we’ve just heard this too many times hopefully very very soon the next PR will have a lot more meat to it till then still holding and hoping it is a very good product we just need someone to sell ijt $$$$$$$
Well, it has been clarified and now we do know nothing illegal was done.
Agreed. I believe the only reason that they released this PR today was probably because they were receiving multiple calls from shareholders about calling the SEC and reporting them as performing illegal if not illegitimate events. Just my opinion.
Sell move on to something else.
Some seem to readily discount fiduciary responsibility for accurate disclosure to shareholders and public, through use of Standard Accounting Practices, in financial reports and official PRs. If the below excerpt were true (and not laughable or embarrassingly ignorant), there would be, plain and simple, aged accounting entries affecting the Balance Sheet (e.g., outstanding liability to CEO), backing up the claim. And use of childish, derogatory memes, in lieu of refuting with factual evidence, do not change that.
"Additionally, the report reflected a loan of $700,000 to CEO Dr. Otiko. Dr. Otiko has funded many of the Company’s expenses over the past several years from his own resources in order to keep ViaDerma from getting into costly debt."
No they aren't if run legit. Vitastem is supposedly the strongest topical antibiotic in the world. If that's really the case, they should be selling MILLIONS of products yearly. They haven't done crap in years.
All these PRs for years never materialize. Then the doc and useless IR guy are silent for months.
Notice how they never talk about Nupelo, Dubai deals, India and all the other countries they were shipping to in March?
No inventory on their books either. Instead of being more transparent and answering shareholders questions and concerns, they just ignore you. It will likely take a class action lawsuit to uncover what is really going on at Viaderma. Hunch is its just BS.
Just another BS PR and all the Bobos nod in unison
Bud well said. I'll gladly take this bone 4 now
|
Followers
|
619
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
74597
|
|
Created
|
10/27/08
|
Type
|
Free
|
| Moderators | |||

ViaDerma's proprietary transdermal delivery system allows for rapid mass transfer of the pharmaceutical active ingredient across the skin and into the body to provide immediate localized therapy.
The technology allows transfer of chemicals through the stratum corneum (outer layer of skin) with a diffusion constant which is 10,000 times higher than the diffusion constant which characterizes water movement through the stratum corneum.
This enables ViaDerma to pair almost any active ingredient with the technology and provide rapid transport of the medicine right to the site of action.
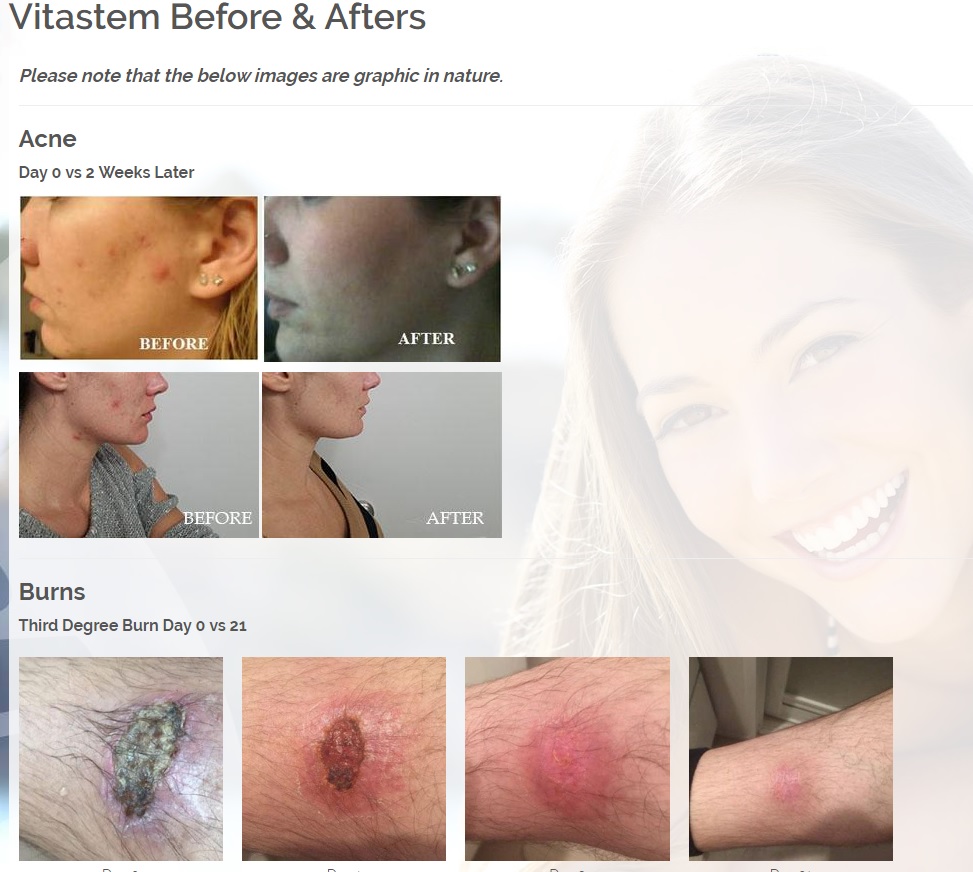
The first product is a broad spectrum tetracycline-based topical antibiotic is the only antibiotic in the world that kills bacteria both a physical and a chemical mechanism. All known antibiotics (other than ours) primarily use only a chemical mechanism of kill. The physical mechanism of kill is a key feature of what we call Rapid Active Ingredient Delivery System (RAIDS). One result of RAIDS is that tetracycline is carried in higher concentrations, more quickly, to and through the cell walls, where the tetracycline can become more effective than if conventional antibiotics were used. Conventional antibiotics require more time (usually prescribed for 5 to 7 days for best results), whereas our tetracycline-based products usually produce desirable results in 24 hours (or less) because of the RAIDS effect.

A second important result of RAIDS is that the topical antibiotic kills all harmful Gram positive and Gram negative bacteria that have been available for testing. We believe this is the world’s strongest broad-spectrum topical antibiotic available.
The potential commercial impact is immense. Drug developers believe it takes much longer for bacteria to develop drug resistance to a physical kill mechanism. This is because it is relatively easy for bacteria to change their response to a chemical threat, but it takes numerous generations for bacteria to grow a new kind of cell wall structure to respond to a physical threat.
Our novel approach to overcome drug resistance of antibiotics is designed to sustain the effectiveness of antibiotics and other topical drugs for many years. This gives new topical drug products a longer useful lifetime and therefore more commercial value. This technology, when licensed to larger pharmaceutical companies, may provide stronger incentive for the discovery and development of new antimicrobial drugs. In recent years, the dearth of new antibiotics has been largely due to the uncertain new-drug commercial lifetime which is diminished when bacteria develop immunity to that drug.
In addition the technology can be licensed to other companies to convert existing oral drugs to transdermal medications extending the profitably of the existing drug.


We are developing products in the following fields of use;
Treats all types of bacteria including MRSA, VRE and other flesh eating bacteria. Fights drug resistant bacteria. $6B/year global market.

Onychomycosis, a fungal infection of the toenails, is a major health problem. It is estimated that there are in excess of 40 million sufferers with this condition in the USA. It is a problem throughout the world. A recent European study showed that the prevalence of Onychomycosis may be as high as 26.9%. Fungal resistance can occur when the oral antifungal agents are used on a long-term basis. Topically applied antifungal drugs may work somewhat better adjunctive to surgical removal or chemical dissolution of the nail plate. Yet, this often ineffective and traumatic procedure leaves the subject without a nail for months at risk for re-infection. Given the limitations of current treatment options in this $3B market, there is a great need for a simple, nontoxic and effective alternative treatment. Estimated Global prevalence rate: 140 million.
Treats symptoms of Influenza which is a$4B/ year global market: Common
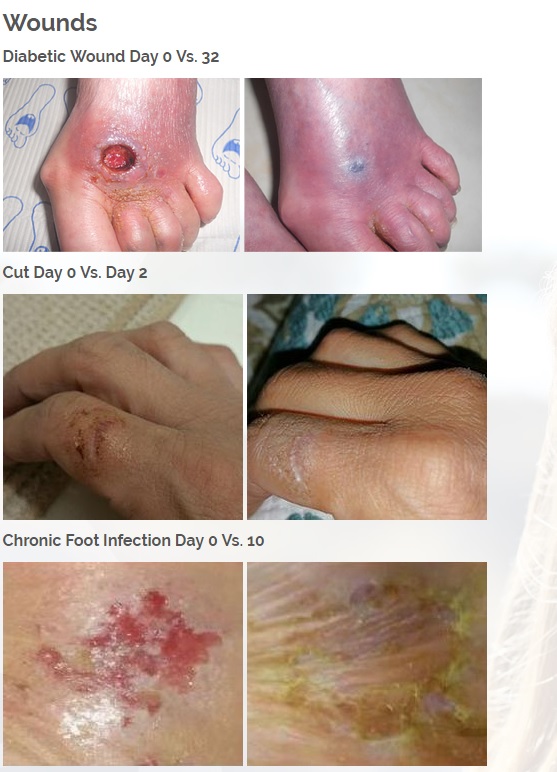
Diabetic foot wounds.

Per 3/8/18 PR: ViaDerma’s technology is currently being used in Elixr Cannabis products; Topical Balm, Topical Serum and Topical Spray. Sales have begun in Canada.
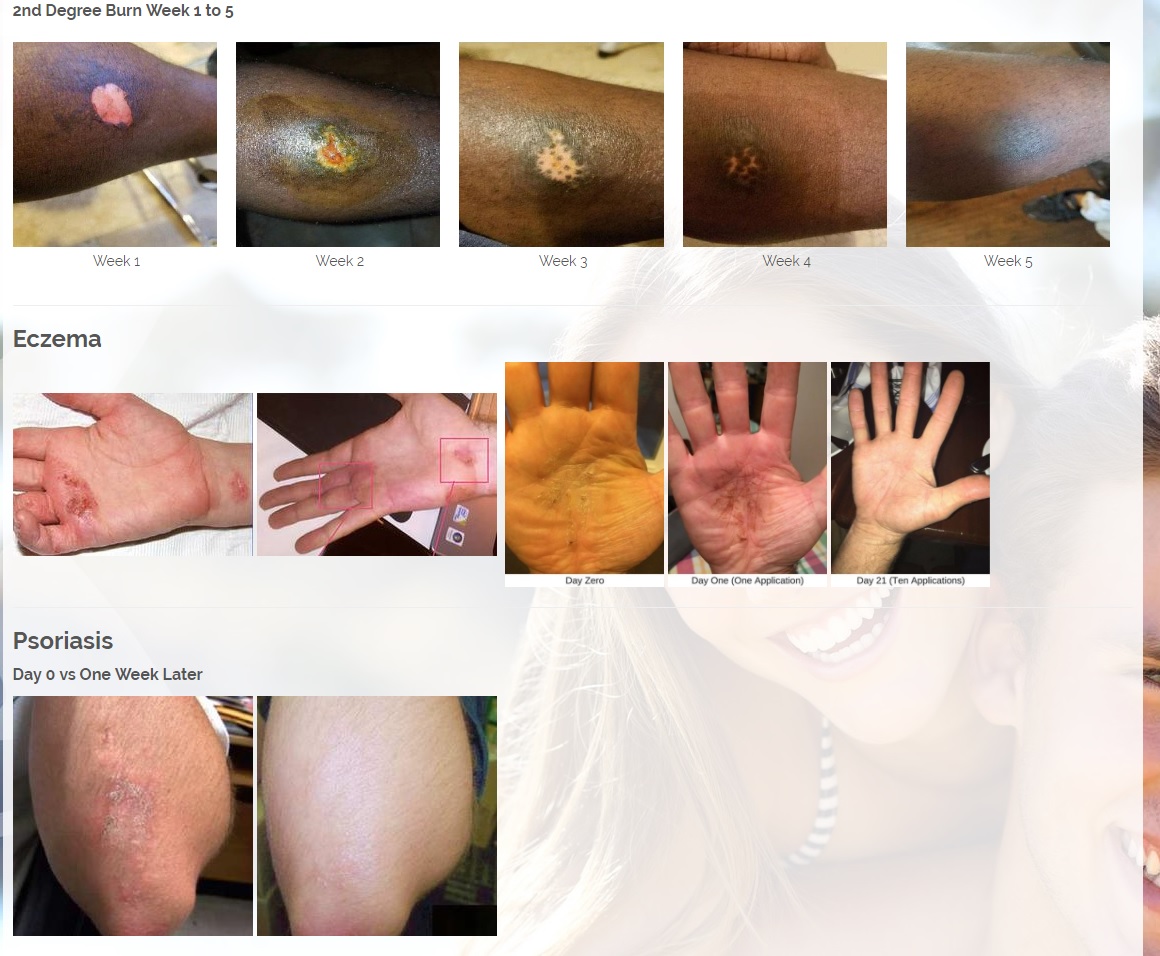
Per 12/7/17 PR: The Company has signed an MOU and will start production of its Patent Pending CBD or Cannabinoids product line. The Company would combine its two Patent pending technologies, the MMJ Patent #62466209, a patent for delivering medical marijuana / cannabis to the body by applying the medication onto the skin in an ointment base topical solution and the second Provisional patent # 62433964 for enhanced antibiotic and drug delivery for "Aqueous Topical Applications" for human and veterinary uses. The Company would combine its proprietary solutions with CBD's and other natural products that treat psoriasis, fibromyalgia for pain and other ailments.
Per 1/23/18 PR: The Company has also a licensing and distribution agreement in place with its Canadian partners to produce a CBD Topical Serum, which has a 92% absorption rate powered by ViaDerma's Patent Pending Dual Carrier Technology. The CBD and Terpene profile (enriched with specific essential oils and vitamins C, and D) is crafted to alleviate Chronic Dry Skin, Psoriasis, Eczema, Rosacea, and many other skin conditions. The serum has anti-inflammatory, anti-bacterial, and chronic pain relief properties that are absorbed by the skin and provide overall healing benefits. Sales are expected to begin in the first quarter of 2018 and are projected to generate approximately $2 million annual revenues.
The Patented Pending CBD or Cannabinoids product line. The Company would combine its two Patent pending technologies, the MMJ Patent, a patent for delivering medical marijuana / cannabis to the body by applying the medication onto the skin in an ointment base topical solution and the second Provisional patent for enhanced antibiotic and drug delivery for "Aqueous Topical Applications" for human and veterinarian uses. The Company would combine its proprietary solutions with CBD's and other natural products that treat psoriasis, fibromyalgia for pain and other ailments. The Company plans to continue to expand its (IP) "Intellectual Property Portfolio" in 2018.

CBD is a compound found in the Cannabis family of plants such as hemp. CBD is combined into our topical body care products like Elixr's Topical Balm, Serum, and Spray.
CBD is an antioxidant. Antioxidants protect our skin from damaging exposure to smoke, UV rays, and environmental pollutants. Cannabinoid receptors are located all throughout the skin, Topical CBD products interact with those receptors resulting homeostasis and healing. Studies show that CBD can help treat an array of skin conditions such as eczema, psoriasis, rosacea, and acne.
| "What is Intellectual Property?" - A Publication by the World Intellectual Property Organization -
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
