Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
The fact is anything is possible at this point
There are two likely big drags on your analysis potential realization and that is the CEO and the CEO. Infuriatingly, in all of these years and his"proclamations", he has yet to show that he can land any large contracts, that actually come to fruition. I am hoping that maybe he is constrained from announcing the actual overseas shipping start, due to the Yield sign, but I have not been able to confirm that. Separately, the CEO is again the big drawback, if he lands the contracts, by the embarrassing, beyond egregious contract clause (shown below) that he gave to himself.on October 1, 2022. We, the shareholders, need the company to land contracts, but not by the CEO, due to that ridiculous, undeserved clause. In any event, still hoping he somehow becomes professional and effective and makes us shareholders a great deal of money.
"In addition, CEO shall be entitled to a bonus of 30% of any revenue generated from contracts he brings into the company for the first
$1,000,000 in any fiscal year, or a bonus of 40% of any revenue generated from contract he brings into the company
over $2,000,000 in any fiscal year."
.50 cents per share you say? i think you are correct. simply break it down
50 states at 5k each is 250k per month = 3 million annual revenue
300k units per country in only 6 countries =1.8 million units at 50.00 per unit(its much more than that) = 90 million
that's 93 million divide by 1.25 billion shares =.0744 per share then put a 10x multiplier (should be 25% in this industry)
and there you have .744 cents per share value.
DOES NOT COUNT ANY NEW COUNTRES OR NUPELO AR ANYTHING ELSE IN THE PIPELINE
Never.....unless it reverse splits down the road ..
DB, let me know when it'll hit $.50; I'd like to retire soon.
$.01172 VDRM is coming together! It WILL go much higher and my only worry is someone taking it out @ too low a price. On its own, I can see $.50, YES FITTY CENTS! Don't think so? I don't give a F!
Too many know nutt'ns on this....
More like 7 years of dilution and no execution
Seven yrs. of hard work & waiting is behind us
I am on the positive side I use the product
That’s true. This company unlike many others out there has potential and good-real products. I have always had said it: VIADERMA ISSUE IS MANAGEMENT. Nothing else
It’s possible but the question is: Is there really going to be any news? I got the feeling Otiko will sink his big head for a long, long time in his mummy basement. Fillings? Sure, I won’t be surprised if I see attorney letter this month but beyond pinkeye current, Naah, a gamble.
SO much potential. That’s why it’s so damn frustrating lol
I Like the fact nobody was selling at the 8s and very few at 9s. Have a small position not as big as couple years ago. All good, hope everyone has a good weekend
Yes it does and maybe with the news it takes to get there it can HOLD and begin to CLIMB A LITTLE HIGHER.
I think I agree on both. Despite of the Doc’s lies, this spikes once or twice a year to 0.03-0.05 cents. We just have to be alert.
Slops likes drama. Just giving him a little bit of what he likes
Let’s go back to:
“This is going to the moon!”
And
“The Dr. is a lying and scamming con artist”
Of course not. You dropped Kindergarten, remember?. I am sorry but don’t expect me to teach that as well, not the place. Anyways, From which ranch do you come from?
Fdc4Sorry I can't even understand that post
Someone knows something we don’t? VDRM made a small run up.
But you haven’t gotten yours yet and look at you, trying, trying to be Mr. Somebody. Bwahh
Get your GED before giving advice Corky
Nobody is seriously buying in here anymore. I only hope OTIKO realizes shareholders ain’t buying his bull$hit at this point . He better manifest with something concrete work, otherwise, poof, we’re done
ORCA and SLOPS should take some time off in somewhere in Mexico or Mumbai. These folks have hit Rock bottom on wrong predictions. Embarrassing
Give it up lil minnow .007 aint gonna happen
ORCA
Sunday, March 31, 2024 9:09:41 PM
Post# of 72805
YOU HAVE YOUR EAR OR YOUR NOSE IN OTIKOS A$$??THERE WILL BE NO FILING MONDAY.
OTIKO!!! OTIKO!!! Where’s your head sunken at?
As long as it takes. OTIKO may be Natty but VDRM always spikes up once a year. Once it does happen, I will be there
I do still have my sights intact, Slop. What? Don’t tell me you do not know how to read mate!!! Check the old fillings
You still in your car belted up waiting for October?
It is not up to the OTC yet, that was lie. They better move their A$$ up with attorney letter so OTC can move forward. No attorney letter, which it is what legally speaking seals the confirmation of the recent filings submitted. OTC can’t move forward without a legal representative letter, which is an attorney. Delays as usual and not from OTC, these companies always want to blame OTC now that they can’t blame COVID-19 delays anymore. Chuckles. Ottiko is a mess and this IR too. Anyways, at the end, they will deliver
Kinda wouldn’t makes sense to announce a PR until the yield sign comes down
Who cares.....it won't move the stock....
For board's information, I sent email inquiry today to IR about attorney letter, profile update and Pink Limited Information marker removal The response is as follows:
Hello. Yes, we are all up to date finally. It usually takes the o Tc. About ten days to remove the yield sign. The attorney is going to start working on the letter. We have a little bit of time to get that done
Welcome to OTCM mate… The majority in here have those growing concerns. Where you fail to realize is this company has product and they do sale. Also licensed that we benefit from too. OTIKO soon will realize that in order to survive, he will need to partner or be acquired by a bigger pharmaceutical. And trust me, the bid fight will be huge in here, who wouldn’t like to own these products. OTIKO isn’t a businessman, he is a science man or a doctor for gosh sake. I am impressed how far he has gotten with this business. I think he is learning as he goes. I am just waiting when he hits the brick wall and realize that needs help. We will make a banging
That's required by the SEC it's a form letter
Form letter VDRM did NOT compose it.
ViaDerma Inc. and its Subsidiary
Notes to Unaudited Consolidated Financial Statements
December 31, 2023
Management believes that the current available resources will not be sufficient to fund the Company’s planned
expenditures over the next 12 months. Accordingly, the Company will be dependent upon the raising of additional
capital through placement of common shares, and/or debt financing in order to implement its business plan and
generating sufficient revenue in excess of costs. If the Company raises additional capital through the issuance of
equity securities or securities convertible into equity, stockholders will experience dilution, and such securities may
have rights, preferences or privileges senior to those of the holders of common stock or convertible senior notes. If
the Company raises additional funds by issuing debt, the Company may be subject to limitations on its operations,
through debt covenants or other restrictions. If the Company obtains additional funds through arrangements with
collaborators or strategic partners, the Company may be required to relinquish its rights to certain geographical areas,
or techniques that it might otherwise seek to retain. There is no assurance that the Company will be successful with
future financing ventures, and the inability to secure such financing may have a material adverse effect on the
Company’s financial condition. These consolidated financial statements do not include any adjustments to the
amounts and classifications of assets and liabilities that might be necessary should the Company be unable to continue
as a going concern.
VDRM SHOWS READ.SO MUCH ABOUT THE CRAPPY FILINGS.READ THE NEXT POST SOON.
ORCA, you have been wrong sooo much that it hurts now. Mate, it is bad. LMAOOOO
We went to kindergarten together, did you forget? You failed and you mummy didn’t bring you back anymore because she thought you were a waste of time.
Pls buy your shares at ask, Orca.
NOW YOU "PREDICT" A R/S???? THANK YOU, THANK YOU, THANK YOU, as your predictions never seem to work out... THIS ONE WON'T EITHER! Try elf-shoes, shopping, humming, maybe; PREDICTIONS, ACCURACY DON'T SEEM TO WORK FOR YOU!
""".......THERE WILL BE NO FILING MONDAY."""" No filing????? Is it Monday, or April FOOL day? I think both!
ORCA
Sunday, March 31, 2024 9:09:41 PM
Post# of 72699 Go
.......THERE WILL BE NO FILING MONDAY.
|
Followers
|
619
|
Posters
|
|
|
Posts (Today)
|
3
|
Posts (Total)
|
74600
|
|
Created
|
10/27/08
|
Type
|
Free
|
| Moderators | |||

ViaDerma's proprietary transdermal delivery system allows for rapid mass transfer of the pharmaceutical active ingredient across the skin and into the body to provide immediate localized therapy.
The technology allows transfer of chemicals through the stratum corneum (outer layer of skin) with a diffusion constant which is 10,000 times higher than the diffusion constant which characterizes water movement through the stratum corneum.
This enables ViaDerma to pair almost any active ingredient with the technology and provide rapid transport of the medicine right to the site of action.
The first product is a broad spectrum tetracycline-based topical antibiotic is the only antibiotic in the world that kills bacteria both a physical and a chemical mechanism. All known antibiotics (other than ours) primarily use only a chemical mechanism of kill. The physical mechanism of kill is a key feature of what we call Rapid Active Ingredient Delivery System (RAIDS). One result of RAIDS is that tetracycline is carried in higher concentrations, more quickly, to and through the cell walls, where the tetracycline can become more effective than if conventional antibiotics were used. Conventional antibiotics require more time (usually prescribed for 5 to 7 days for best results), whereas our tetracycline-based products usually produce desirable results in 24 hours (or less) because of the RAIDS effect.

A second important result of RAIDS is that the topical antibiotic kills all harmful Gram positive and Gram negative bacteria that have been available for testing. We believe this is the world’s strongest broad-spectrum topical antibiotic available.
The potential commercial impact is immense. Drug developers believe it takes much longer for bacteria to develop drug resistance to a physical kill mechanism. This is because it is relatively easy for bacteria to change their response to a chemical threat, but it takes numerous generations for bacteria to grow a new kind of cell wall structure to respond to a physical threat.
Our novel approach to overcome drug resistance of antibiotics is designed to sustain the effectiveness of antibiotics and other topical drugs for many years. This gives new topical drug products a longer useful lifetime and therefore more commercial value. This technology, when licensed to larger pharmaceutical companies, may provide stronger incentive for the discovery and development of new antimicrobial drugs. In recent years, the dearth of new antibiotics has been largely due to the uncertain new-drug commercial lifetime which is diminished when bacteria develop immunity to that drug.
In addition the technology can be licensed to other companies to convert existing oral drugs to transdermal medications extending the profitably of the existing drug.

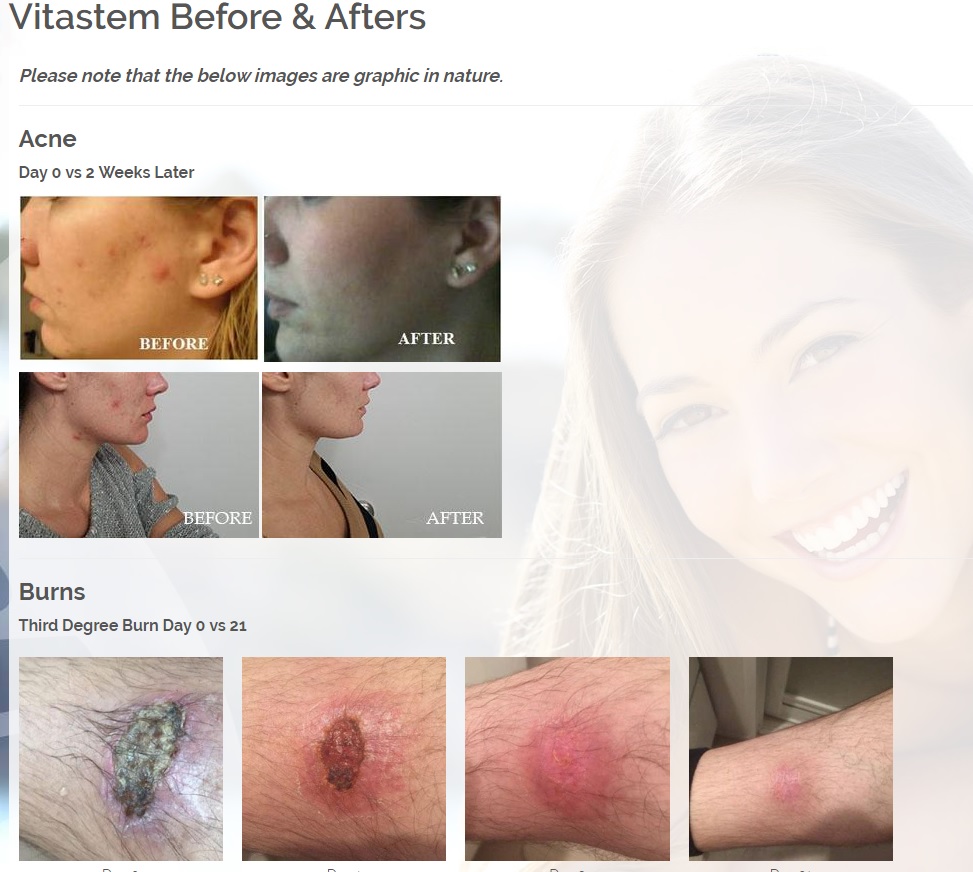
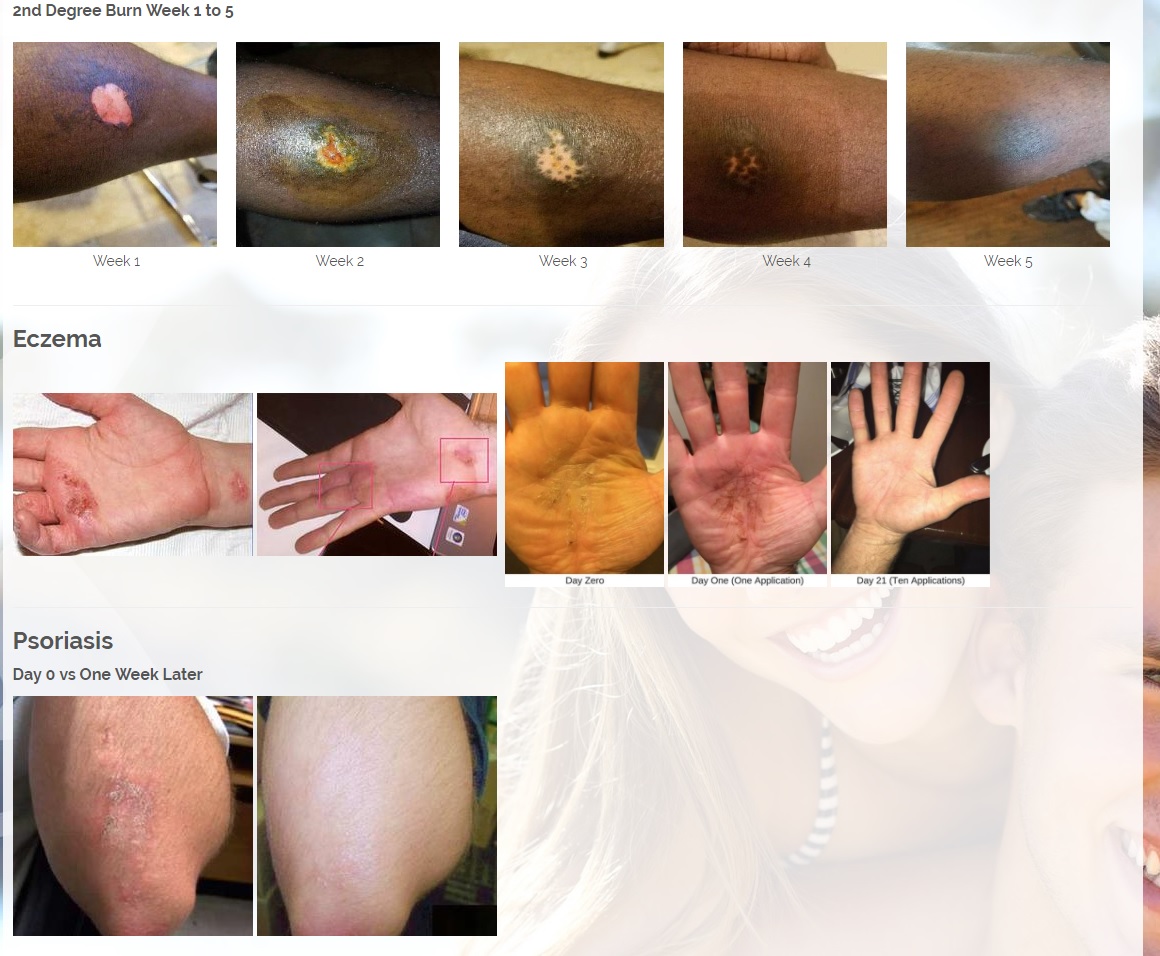
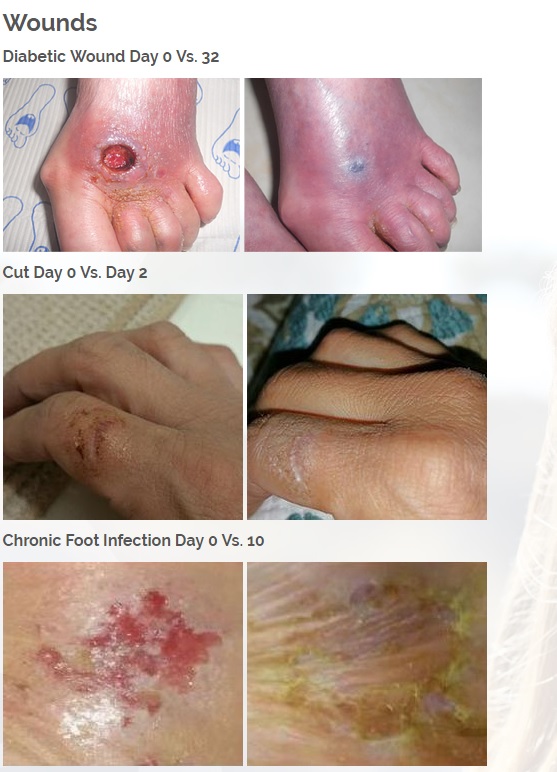

We are developing products in the following fields of use;
Treats all types of bacteria including MRSA, VRE and other flesh eating bacteria. Fights drug resistant bacteria. $6B/year global market.

Onychomycosis, a fungal infection of the toenails, is a major health problem. It is estimated that there are in excess of 40 million sufferers with this condition in the USA. It is a problem throughout the world. A recent European study showed that the prevalence of Onychomycosis may be as high as 26.9%. Fungal resistance can occur when the oral antifungal agents are used on a long-term basis. Topically applied antifungal drugs may work somewhat better adjunctive to surgical removal or chemical dissolution of the nail plate. Yet, this often ineffective and traumatic procedure leaves the subject without a nail for months at risk for re-infection. Given the limitations of current treatment options in this $3B market, there is a great need for a simple, nontoxic and effective alternative treatment. Estimated Global prevalence rate: 140 million.
Treats symptoms of Influenza which is a$4B/ year global market: Common
Diabetic foot wounds.

Per 3/8/18 PR: ViaDerma’s technology is currently being used in Elixr Cannabis products; Topical Balm, Topical Serum and Topical Spray. Sales have begun in Canada.
Per 12/7/17 PR: The Company has signed an MOU and will start production of its Patent Pending CBD or Cannabinoids product line. The Company would combine its two Patent pending technologies, the MMJ Patent #62466209, a patent for delivering medical marijuana / cannabis to the body by applying the medication onto the skin in an ointment base topical solution and the second Provisional patent # 62433964 for enhanced antibiotic and drug delivery for "Aqueous Topical Applications" for human and veterinary uses. The Company would combine its proprietary solutions with CBD's and other natural products that treat psoriasis, fibromyalgia for pain and other ailments.
Per 1/23/18 PR: The Company has also a licensing and distribution agreement in place with its Canadian partners to produce a CBD Topical Serum, which has a 92% absorption rate powered by ViaDerma's Patent Pending Dual Carrier Technology. The CBD and Terpene profile (enriched with specific essential oils and vitamins C, and D) is crafted to alleviate Chronic Dry Skin, Psoriasis, Eczema, Rosacea, and many other skin conditions. The serum has anti-inflammatory, anti-bacterial, and chronic pain relief properties that are absorbed by the skin and provide overall healing benefits. Sales are expected to begin in the first quarter of 2018 and are projected to generate approximately $2 million annual revenues.
The Patented Pending CBD or Cannabinoids product line. The Company would combine its two Patent pending technologies, the MMJ Patent, a patent for delivering medical marijuana / cannabis to the body by applying the medication onto the skin in an ointment base topical solution and the second Provisional patent for enhanced antibiotic and drug delivery for "Aqueous Topical Applications" for human and veterinarian uses. The Company would combine its proprietary solutions with CBD's and other natural products that treat psoriasis, fibromyalgia for pain and other ailments. The Company plans to continue to expand its (IP) "Intellectual Property Portfolio" in 2018.

CBD is a compound found in the Cannabis family of plants such as hemp. CBD is combined into our topical body care products like Elixr's Topical Balm, Serum, and Spray.
CBD is an antioxidant. Antioxidants protect our skin from damaging exposure to smoke, UV rays, and environmental pollutants. Cannabinoid receptors are located all throughout the skin, Topical CBD products interact with those receptors resulting homeostasis and healing. Studies show that CBD can help treat an array of skin conditions such as eczema, psoriasis, rosacea, and acne.
| "What is Intellectual Property?" - A Publication by the World Intellectual Property Organization -
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
