Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
My first response from Investor Relations in over a year was received overnight. It is to an email that I sent 7/25/22. I had inquired:
"Is it expected that the 2nd quarter report will be issued on time (on or before 8/15/22)."
Response received:
"Yes, probably before. We are usually early on our filings."
Note: It was sent to info@viadermalicensing.com.
I have NOT yet received response to emails sent on 7/26/22 and yesterday. I will pass along responses if, and when received.
It's going to the Expert Market.
They don't understand the new Regulation.
Has NOTHING to do with the upcoming Quarterly Report LMFAO.....
Then will immediately display no quotes and become illiquid.
DeerBalls good post I agree 100%
Relax everything is moving at a slow pace
And the hits just keep on coming. Wow! Again, I feel so much better. NOT! All, and I mean ALL evidence to the contrary. Great to talk about what used to be and how great the product was but what good is the memory of a product the public can't buy or find anywhere. Generating revenue? Really where? Except for maybe some minor runs this company seems to be circling the drain. Just excuses after excuses after more excuses. But what remains humorous is the nonsense through the prism of smoking mirrors. As always just my opinion but at least it's honest and from someone with a sizable position here.
THERE IS NO EXCUSE.THAT FPOS OTIKO SHOULD UPDATE SHAREHOLDERS OF WHAT THE F IS GOING ON.GOOD OR BAD.1)WHAT HAPPENED WITH THE TESTING???
2)UPDATE US ABOUT THE OTC PROBLEM.LIKE ALL HE HAS TO SAY IS:DEAR SHAREHOLDERS WE ARE AWARE WITH THE PROBLEM,AND WE ARE ON IT,AT THE MOMENT TO FIX IT,AND GET BACK TO CURRENT STATUS.
$.0067 Just a suggestion from someone who was a broker for most of my career and have been around thousands of stocks/situations: Investors contacting the company/IR expecting it to be productive in the sense of, "this is a problem/fix this/are you aware that" is TOTAL FOLLY! It may make some feel they are doing something, but it is as productive as moving papers around on a messy desk.
VDRM will be fine, things will get fixed, in my view. VDRM is tiny, underfunded and has had some bumps, ok. VDRM is aware of the OTC Markets screwup, OTC Markets fixes it when they do. IT WILL GET FIXED!
This all reminds me of a buddy(broker I worked with) who passed a few years back. Nicest, low key guy you'd ever meet, but would stand up for himself when pushed. His wife was Type-A and would pick/pick/pick...my buddy would take it for days, until he had enough and BOOM, the "SHUT-THE-F-UP" would come. The wife would for a few days. My point, don't be that wife as a shareholder, it is counterproductive.
VDRM has a great product and we will be happy in the relative near-term! CHILL!
No or, hopefully, not yet.
Did you get a reply?
$.006635 BULLSHIT! There are revenues, when product is available. It will be soon. Because a pink sheet, VDRM, doesn't have the resources of pfe, and misses being spot-on time-wise means little.
I know folks who have used the product and they rave about it.
Licensing revenues, are they for NOTHING? I don't think so.
Hey, no one is forced to buy/hold... I'M DEFINITELY HOLDING!
4 wks has come and gone and people are surprised her has not come out with a statement.? Do your research, this has been going on for years. Fluff PRs and then you never hear about it again. Otiko major scammer
Thanks for that key piece of information. I had been using the address on the ViaDerma Licensing webpage. I have now sent email to him at that address.
Email investor Relations Richard Inza.
Richardinza@gmail.com
No panic. I am just trying to figure out how to facilitate the required communication from the company to otcmarkets. As reported in 2nd paragraph of #60963, otcmarkets is sitting on their hands until official representative contacts them. It would be nice to have some acknowledged contact from the company, regarding awareness and near-term plan of action.
It may not be tomorrow but soon.
Agree. No concern over here.
Last current idea: If someone has access to FAX capability, one could try the FAX number from https://www.viadermalicensing.com/ ( 310-943-1457 ). I am posting the text of my email that could be modified for a FAX, if possible.
Below is the text of my email, mentioned in previous post:
Since you are not responding to info@viadermalicensing,com, I will try this address.
Urgent action is required from Dr Otiko or other authorized representative regarding communication with otcmarkets to get substantive otcmarkets errors corrected.
Otcmarkets ( https://www.otcmarkets.com/stock/VDRM/disclosure ) has mistakenly assumed VDRM is Accelerated Filer and thus, 2nd Qtr Report due 7/31/22. Further, otcmarkets has massively misinterpreted Rule 15c2-11:
https://www.wilmerhale.com/insights/client-alerts/20201029-sec-amends-rule-15c2-11-to-enhance-publicly-available-information-for-securities-quoted-in-the-over-the-counter-markets
Instead of applying the Grace Period after the due date is passed, they are applying it before the due date (and the wrong grace period and due date at that). They have then erroneously applied the Grace Period, and Dark or Defunct labels. These are wrong on so many levels. VDRM is Non-Accelerated Filer and thus, 2nd Quarter Report is not due until 8/15/22. The four-day Grace Period should then be applied beyond that.
I realize this is the wrong email address for this, but we shareholders do not seem to have any other means to identify this to Dr Otiko. Please pass it along, with URGENCY.
Thanks, in advance, for any and all help in the above.
$.0065 The mistake was not that of VDRM, it was OTC MARKETS and we have to wait for them to fix it. The way it is and if that is to much, there is a bid.
It is amazing what will be attributed to VDRM/ Dr. Otiko.
No one is forcing anyone to buy/hold/sell VDRM.
CHILL!
I tried the sales email address to see if I can get the concern to Dr Otiko that way. I will post if I hear anything.
Thanks to ORCA for sending the tweet earlier.
i realize it's a clerical mistake gameccks1, but errors like this don't get fixed expeditiously. this will go dark by monday. my question is......will vdrm have 18 months to file the necessary reporting to get this back on the pink sheets?
Absolutely. Only reason to freak out here is if you were planning on selling at .008 in the next week or two. If you are holding this puppy for several months I would just ignore all of this.
As others have stated VDRM is up to date.
I ain’t worried. Picked up 500K shares this morning at .006
Good luck everyone.
There is actually nothing to submit (see #60971). There needs to be communication with otcmarkets, getting them to understand, admit and correct their mistake. The 2nd Quarterly Report is not due until 8/15/22.
starting to freak out. if vdrm does go to the 'expert mkt' next week, can they get back to the pink if they submit the necessary documents?
I don’t disagree with anything you’ve said here. The reason this company is a 3-5X opportunity/gamble is the issues that you’ve stated.
I own plenty (I’m sure you do as well) of companies that have product, zero filing issues, pay dividends, etc etc. With those companies I am happy to earn 10-15% per year.
I came over to the micro side looking for explosive opportunities. Not just happy with double digit… long response here, but the moral of the story is these micro companies have large potential because they are risky AF. Comes with the territory “shrug”
Sure. Buying opportunity. Really? People can do what they want with their money. Can just burn it if they want. But everything is going to be all right. I know I feel better now. This company is turning into a joke with excuses more available than their product no one can find. But according to some, orders pending are huge and retailers are too numerous to count. Close your eyes. What do you see? NOTHING!
Just my opinion of course.
THE COMPANY AND OTIKO IS A F POS.BECAUSE IF HE WAS A GOOD CEO WOULD HAVE ADDRESSED IT RIGHT AWAY VIA PR TO HIS SHAREHOLDERS.BUT INSTEAD DID NOTHING.SO PEOPLE PANIC AND TAKING LOSSES.F YOU OTIKO.
The ongoing drop in share price has been triggered by an unbelievably careless error by otcmarkets. If regulation worked, WHICH IT DOES NOT, especially for small companies, all of the trades during the time period of the error would be cancelled, without mutual re-agreement of both parties.
If VDRM were an Accelerated Filer, meaning 2nd Qtr Report due 7/31/22. The four-day Grace Period, per Rule 15c2-11, will be between 8/1/22 - 8/4/22, i.e., NOT YET STARTED. However, VDRM is a non-Accelerated Filer, meaning 2nd Qtr Report due 8/15/22. Thus, the four-day Grace Period will be between 8/16/22 - 8/19/22. This is a significant mistake by otcmarkets. One can only wonder if it is systemwide and thus affecting more companies. May otcmarkets receive appropriate blowback.
https://www.wilmerhale.com/insights/client-alerts/20201029-sec-amends-rule-15c2-11-to-enhance-publicly-available-information-for-securities-quoted-in-the-over-the-counter-markets
$.00664 The VDRM Yield Sign/dark/defunct BS is a screw up on OTC Markets part. Being corrected!
$.0065 Just another VDRM buying opportunity. Things will be fine!
I JUST TWEETED THE PROBLEM FROM OTC TO OTIKO.
YOU NEED TO ADDRESS THIS ASAP. TO YOUR SHAREHOLDERS VIA PR.SERIOUS PROBLEM ON OTC.
— ORCA (@NBBLegend) July 27, 2022
CHECK LINK.SHAREHOLDERS ON IHUB ARE PISSED OFF ABOUT THIS PROBLEM.PLEASE ADDRESS IT.ASAP.
Grace Period IconGrace Period
Dark or Defunct IconDark or Defuncthttps://t.co/MbH4u1MScg
Yep.....News is once the Grace Period is up we hit the Expert Market where there will be no liquidity as it will not have any MM's.
Thanks for taking the time to do that. You never know...the Company may actually take notice.
For the board's information, I have emailed investor relations and otcmarkets about the issue. I do not expect response from investor relations since I have not received any response on over a year. Interestingly, I got a boilerplate response from otcmarkets:
As a market operator, our policy is not to comment on any specific company's status unless you are an officer or their legal counsel. You can contact the company directly regarding their Grace Period status.
My original message:
You are incorrectly tagging VDRM as Accelerated Filer which it is not. It is a Non-Accelerated Filer. Thus, your website is misleading in the Pink Limited Information.
The 2nd Quarter Report is not due to 8/15/22. Lastly, a grace period would be properly applied after a due date to get into compliance. Even if the nutty label is correct, the Grace Period label, being attached incorrectly since 8/15/22 -15 days is July 31, 2022.
I believe the OTC Market should be held liable for their mistake if they admit to making the mistake!!!
Rule 15c2-11 - It seems otcmarkets has carelessly messed up the whole thing relative to VDRM and that rule.
VDRM is Non-Accelerated Filer. Thus, 2nd Quarter Report is due 8/15/22, and not 7/31/22 for Accelerated Filer.
The Grace Period is 4 business days, not 15, and it is like every other grace period that I have ever heard of it, meaning that the grace period is AFTER a delinquency.
VDRM, and any other companies wrongly tagged, should issue letters to otcmarkets regarding the mistake, or take more aggressive action.
https://www.wilmerhale.com/insights/client-alerts/20201029-sec-amends-rule-15c2-11-to-enhance-publicly-available-information-for-securities-quoted-in-the-over-the-counter-markets
This company has revenues, profits, a proven and approved product and large orders to be filled. Based on comps with similar companies, many of whom have few or none of above, sp should be, at least, in the .40's, but probably won't be soon because of the quick flippers.
2022 has been setback after setback. Price has largely remained in the .006-.008 range despite all the negativity.
I’m here for the long haul. Not too concerned about this week or next, the expected news or filing status.
Sure does suck for the quick flippers though
Lol. Pretty sure there is no way out of the grace period. After 15 days.....expert market.....no liquidity etc
$.0076 Dr. Otiko will come through! VDRM will be fine, shortly!
Boom - thank you.
It appears to relate to the 2nd Quarter Report. It would appear that OTC does not have VDRM set correctly as Non-Accelerated Filer (8/15/22 due date (8/15/22 - 15 days = 7/31/22) versus 8/9/22 for Accelerated Filers (8/9/22 - 15 days = 7/25/22)). If you hover over Grace Period, it will give more information. However, in any event, this is still the first regulatory/financial type milestone, that I have encountered, that has a Grace Period leading up to the due date and not as attached additional period, after the due date.
The 2021 annual report was submitted on 3/29/2022. What was due/big event was around 180days ago?
Or wonder if VDRM notified OTCM of an amendment and that triggered the status. Hopefully find out soon
Thanks for the information.
I had never noticed those markings before on the OTC page or the additional information from hovering over them. I will say it is unexpected to have a grace period before a regulatory due date and not as additional time thereafter.
LMFAO.
15 Day Grace period BEFORE....
EXPERT MARKET
The amended rule adds a 15-day conditional grace period from the date of a publicly available determination that a company no longer has current information within the 180-day specified period as set forth in the chart below, for a broker-dealer to continue to quote the particular security. The purpose of the grace period is to give the markets notice that the company is in danger of no longer being quoted and provide investors with an opportunity to liquidate positions
I totally understand. Hopefully, it will become apparent soon and not be yet another setback!!!!
I am just posting as it shown. Maybe some of the previous filings are wrong…. Who knows/// It seems a very grave error by OTCM to make then. Got me off base to be honest tho
|
Followers
|
615
|
Posters
|
|
|
Posts (Today)
|
15
|
Posts (Total)
|
73875
|
|
Created
|
10/27/08
|
Type
|
Free
|
| Moderators | |||

ViaDerma's proprietary transdermal delivery system allows for rapid mass transfer of the pharmaceutical active ingredient across the skin and into the body to provide immediate localized therapy.
The technology allows transfer of chemicals through the stratum corneum (outer layer of skin) with a diffusion constant which is 10,000 times higher than the diffusion constant which characterizes water movement through the stratum corneum.
This enables ViaDerma to pair almost any active ingredient with the technology and provide rapid transport of the medicine right to the site of action.
The first product is a broad spectrum tetracycline-based topical antibiotic is the only antibiotic in the world that kills bacteria both a physical and a chemical mechanism. All known antibiotics (other than ours) primarily use only a chemical mechanism of kill. The physical mechanism of kill is a key feature of what we call Rapid Active Ingredient Delivery System (RAIDS). One result of RAIDS is that tetracycline is carried in higher concentrations, more quickly, to and through the cell walls, where the tetracycline can become more effective than if conventional antibiotics were used. Conventional antibiotics require more time (usually prescribed for 5 to 7 days for best results), whereas our tetracycline-based products usually produce desirable results in 24 hours (or less) because of the RAIDS effect.

A second important result of RAIDS is that the topical antibiotic kills all harmful Gram positive and Gram negative bacteria that have been available for testing. We believe this is the world’s strongest broad-spectrum topical antibiotic available.
The potential commercial impact is immense. Drug developers believe it takes much longer for bacteria to develop drug resistance to a physical kill mechanism. This is because it is relatively easy for bacteria to change their response to a chemical threat, but it takes numerous generations for bacteria to grow a new kind of cell wall structure to respond to a physical threat.
Our novel approach to overcome drug resistance of antibiotics is designed to sustain the effectiveness of antibiotics and other topical drugs for many years. This gives new topical drug products a longer useful lifetime and therefore more commercial value. This technology, when licensed to larger pharmaceutical companies, may provide stronger incentive for the discovery and development of new antimicrobial drugs. In recent years, the dearth of new antibiotics has been largely due to the uncertain new-drug commercial lifetime which is diminished when bacteria develop immunity to that drug.
In addition the technology can be licensed to other companies to convert existing oral drugs to transdermal medications extending the profitably of the existing drug.

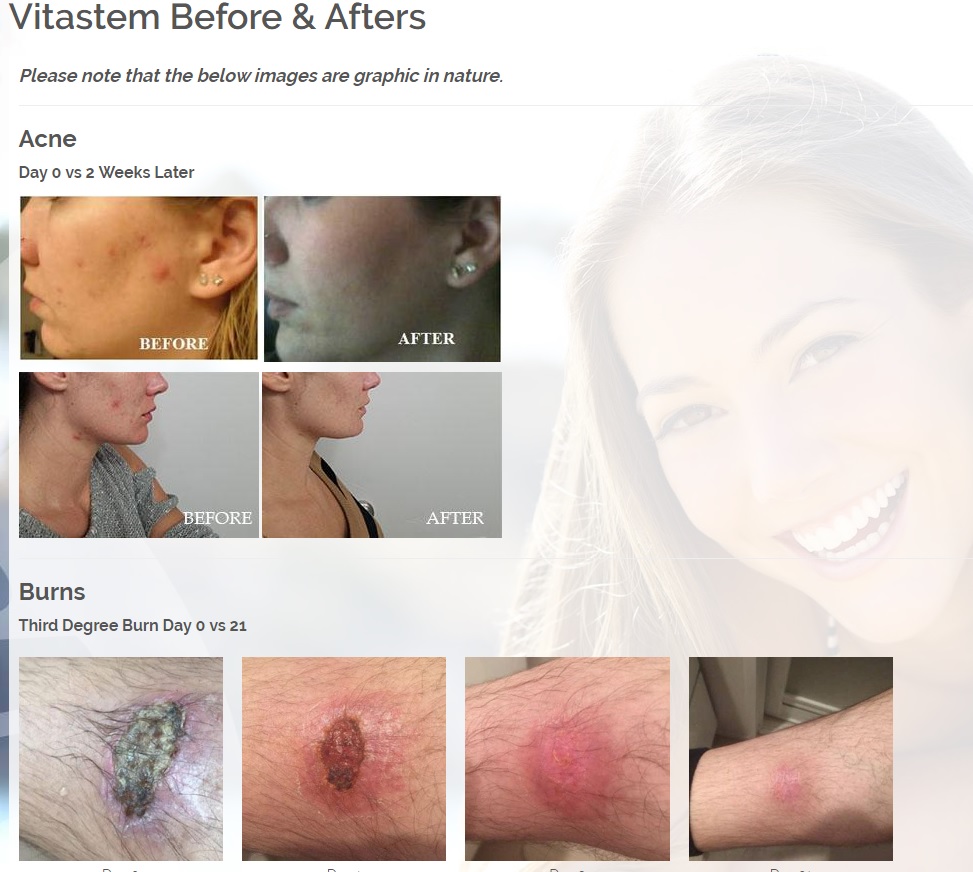
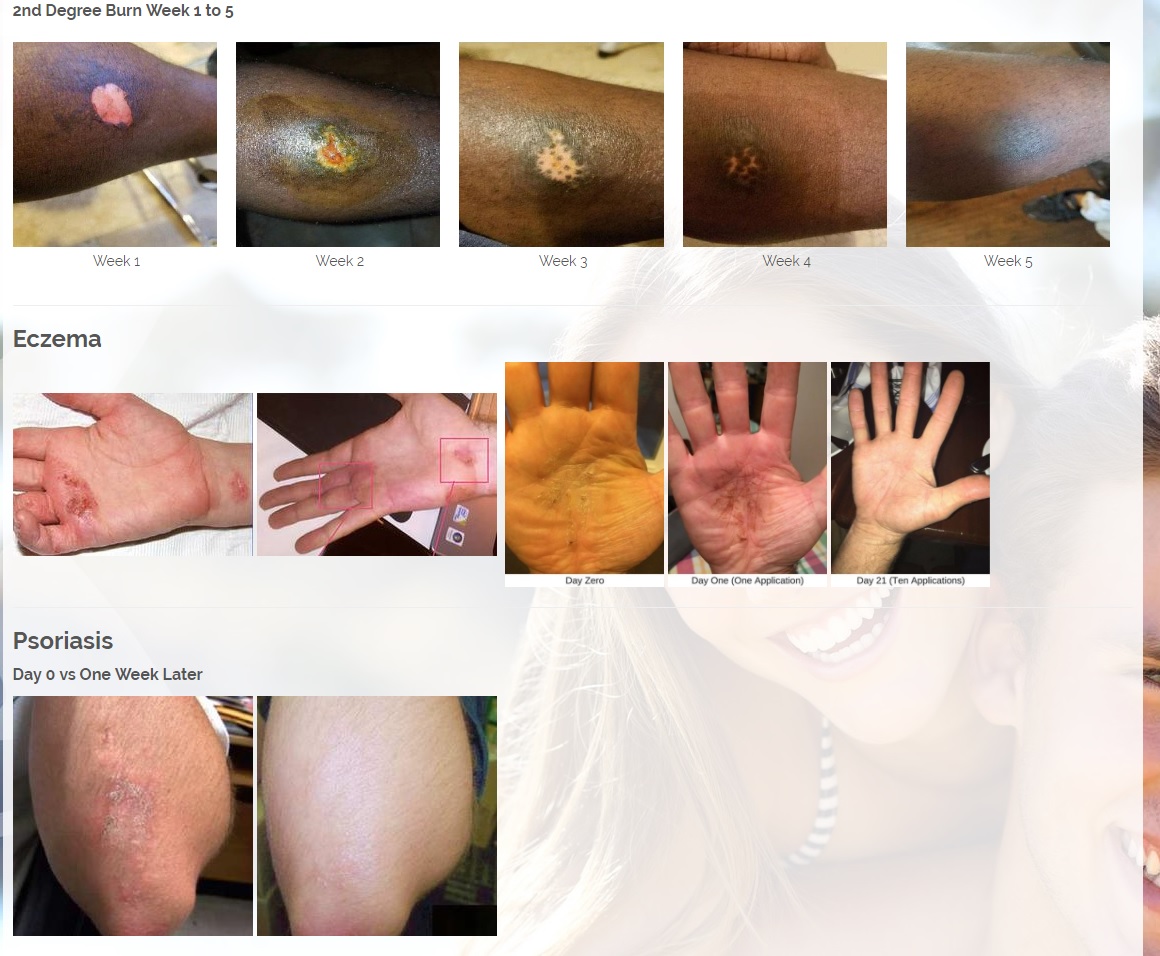
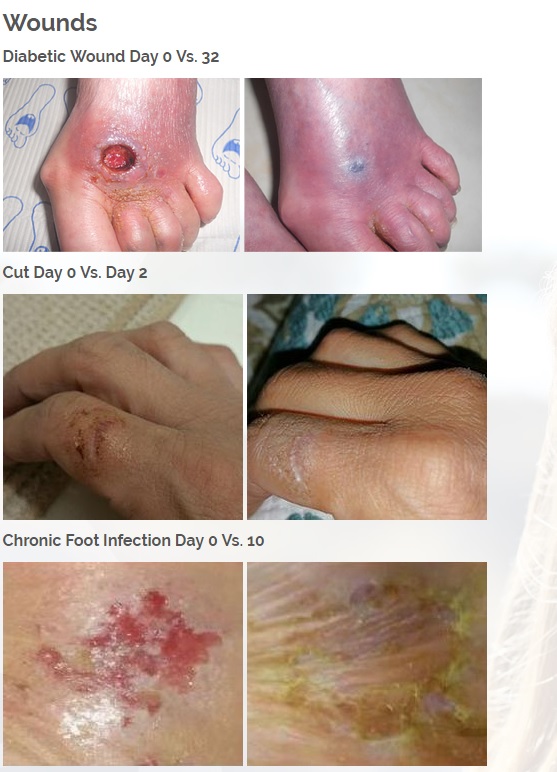

We are developing products in the following fields of use;
Treats all types of bacteria including MRSA, VRE and other flesh eating bacteria. Fights drug resistant bacteria. $6B/year global market.

Onychomycosis, a fungal infection of the toenails, is a major health problem. It is estimated that there are in excess of 40 million sufferers with this condition in the USA. It is a problem throughout the world. A recent European study showed that the prevalence of Onychomycosis may be as high as 26.9%. Fungal resistance can occur when the oral antifungal agents are used on a long-term basis. Topically applied antifungal drugs may work somewhat better adjunctive to surgical removal or chemical dissolution of the nail plate. Yet, this often ineffective and traumatic procedure leaves the subject without a nail for months at risk for re-infection. Given the limitations of current treatment options in this $3B market, there is a great need for a simple, nontoxic and effective alternative treatment. Estimated Global prevalence rate: 140 million.
Treats symptoms of Influenza which is a$4B/ year global market: Common
Diabetic foot wounds.

Per 3/8/18 PR: ViaDerma’s technology is currently being used in Elixr Cannabis products; Topical Balm, Topical Serum and Topical Spray. Sales have begun in Canada.
Per 12/7/17 PR: The Company has signed an MOU and will start production of its Patent Pending CBD or Cannabinoids product line. The Company would combine its two Patent pending technologies, the MMJ Patent #62466209, a patent for delivering medical marijuana / cannabis to the body by applying the medication onto the skin in an ointment base topical solution and the second Provisional patent # 62433964 for enhanced antibiotic and drug delivery for "Aqueous Topical Applications" for human and veterinary uses. The Company would combine its proprietary solutions with CBD's and other natural products that treat psoriasis, fibromyalgia for pain and other ailments.
Per 1/23/18 PR: The Company has also a licensing and distribution agreement in place with its Canadian partners to produce a CBD Topical Serum, which has a 92% absorption rate powered by ViaDerma's Patent Pending Dual Carrier Technology. The CBD and Terpene profile (enriched with specific essential oils and vitamins C, and D) is crafted to alleviate Chronic Dry Skin, Psoriasis, Eczema, Rosacea, and many other skin conditions. The serum has anti-inflammatory, anti-bacterial, and chronic pain relief properties that are absorbed by the skin and provide overall healing benefits. Sales are expected to begin in the first quarter of 2018 and are projected to generate approximately $2 million annual revenues.
The Patented Pending CBD or Cannabinoids product line. The Company would combine its two Patent pending technologies, the MMJ Patent, a patent for delivering medical marijuana / cannabis to the body by applying the medication onto the skin in an ointment base topical solution and the second Provisional patent for enhanced antibiotic and drug delivery for "Aqueous Topical Applications" for human and veterinarian uses. The Company would combine its proprietary solutions with CBD's and other natural products that treat psoriasis, fibromyalgia for pain and other ailments. The Company plans to continue to expand its (IP) "Intellectual Property Portfolio" in 2018.

CBD is a compound found in the Cannabis family of plants such as hemp. CBD is combined into our topical body care products like Elixr's Topical Balm, Serum, and Spray.
CBD is an antioxidant. Antioxidants protect our skin from damaging exposure to smoke, UV rays, and environmental pollutants. Cannabinoid receptors are located all throughout the skin, Topical CBD products interact with those receptors resulting homeostasis and healing. Studies show that CBD can help treat an array of skin conditions such as eczema, psoriasis, rosacea, and acne.
| "What is Intellectual Property?" - A Publication by the World Intellectual Property Organization -
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
