Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
janet.....I'm heading to the mall tonight..... I'm going to sit on Santa's lap and ask him.... I'll put in a good word for you
well well is SANTA coming to rescue ?????
woodsotck... thanks for the info.... I actually emailed Scott today, asking about today's activity, and he answered, "your guess is as good as mine"....which leads me to believe that TO may have found some new investors(instead of news forthcoming)- of course, he may not be at liberty to divulge.... good luck dealing with j***-o** at stockhouse...lol Any activity at this point is good, but today's action definitely means something is coming....imo
Nice reply you got woodstock135 !
Best wishes for a nice week end
janet
This was an answer to an email I sent a few weeks ago. Got lost in my junk folder. Not much to say but at least I received a reply.
Unfortunately the share price is struggling. We are working on new avenues of revenue to help increase sales and the bottom line. The marketplace is overloaded with hand sanitizer right now (left over inventory from H1N1 scare) but fortunately we have numerous other infection control products to sell. We are certainly lacking on the news front over the last couple of months but continue to move forward on the therapeutic side of things.
Thank you for remaining as a shareholder. We continue to work on the share price and hopefully we will see some improvement over the next few months.
Scott Young
over 1 million shares traded now.....(I have to go do some shopping- see ya later)....
Santa is good, but not that good!... lol
Santa whants to see Drumstick smiling......
ALDA starting a run....Early Christmas?... over 475,000 traded all buys.....now back to .11...
over 200,000 shares today, mostly buying....that's something to be thankful for... (but pales in comparison to family and friends).....
It would be a good idea to "STOP" obsessing about Alda! When the Co. will be ready then we can start talking againg.......In the meantime, lets find others stocks to play with
Have a good day
janet
Wonderful to hear, so what about Alda... rermember?
IIROC issues proposal for Single-Stock Circuit Breakers and invites public comment
TORONTO, Nov. 18 /CNW/ - The Investment Industry Regulatory Organization of Canada (IIROC) has issued for public comment a proposal to implement single-stock circuit breakers to address rapid, significant and unexplained price movement in a security that calls into question whether there is a "fair and orderly" market for that security. The proposal builds on the approach followed by IIROC when instituting coordinated regulatory halts across all Canadian marketplaces that trade a particular security.
Under IIROC's proposal, single-stock circuit breakers would apply to all securities listed on an exchange in Canada, including securities inter-listed in Canada and the U.S. When a stock price increases or decreases past a certain threshold, the circuit breaker would trigger a trading halt to provide a pause.
The proposed halt would last five minutes for TSX-listed shares, with the possibility of an extension in the case of a severe order imbalance. The proposed halt for stocks listed on the TSX-Venture and CNSX exchanges is 10 minutes to account for lower levels of liquidity.
Shortly after the May 6 Flash Crash, the Canadian Securities Administrators and IIROC together announced they had begun a comprehensive analysis of the events of the crash, including a review of the "appropriateness of the existing circuit breaker policy." During the crash, markets in the US and Canada experienced extreme volatility, with some stocks quickly experiencing dramatic price declines and rallies.
Under the Universal Market Integrity Rules (UMIR), IIROC has the authority to delay, halt or suspend trading at any time where it is considered appropriate and in the interest of a fair and orderly market. The regulatory halt may apply market-wide or to particular securities. This allows for the imposition of manual halts to address unexplained price movements in a single security. However, current practice is not adequate where there is a need to halt trading in multiple securities across multiple marketplaces, all in a short window of time.
The thresholds under the proposal distinguish between more liquid and less liquid securities. The proposed thresholds are:
•For TSX-listed securities, a halt would be initiated where a stock's price increases or decreases by at least 10 per cent or 10 trading increments, whichever is greater, over a five-minute period.
•For securities listed on the TSX-V or CNSX, a halt would be initiated where a stock's price increases or decreases by at least 20 per cent or 20 trading increments, whichever is greater, over a 10-minute period.
The circuit breakers would be in effect from 9:50 am to 3:40 pm ET.
The Single-Stock Circuit Breaker Proposal is intended to operate as part of a multi-tiered approach for controlling short-term, unexplained price volatility:
I. The first set of controls is at the market participant level where, under UMIR, firms are required to comply with their existing trade supervision obligations.
II. The second set of controls would be effective marketplace volatility controls with consistent objectives across marketplaces.
III. The single-stock circuit breaker would represent the third level of controls.
Both market participant obligations and marketplace level controls should be designed to detect 'erroneous orders' prior to execution. Each of the above-noted elements plays an important role in the overall control framework designed to mitigate the risks associated with unexplained short-term price movement and to promote fair and orderly markets.
The proposal calls for a three-stage process consisting of initial consultation and development, implementation and review/evaluation.
Public Comment Period: The proposal is being issued for a 60-day comment period.
Once comments are received from the public, marketplace participants and the Canadian Securities Administrators, IIROC will review the comments and, as appropriate, revise the Single-Stock Circuit Breaker Proposal. Comments should be directed to:
Timothy Ryan,
Director, Market Regulation Policy,
Investment Industry Regulatory Organization of Canada,
Suite 900 145 King Street West, Toronto, Ontario. M5H 1J8
Fax: 416.646.7265 e-mail: tryan@iiroc.ca
Did you see this "SMKY" ????? Pas Mal ......
XMT.H doing fine today......
News out for SAEI:
Supatcha Announces Stock Repurchase Plan for up to $3 Million
Supatcha Resources Inc. (OTCBB:SAEI)
Intraday Stock Chart
Today : Monday 22 November 2010
Supatcha Resources Inc. (OTC Bulletin Board: SAEI) ("Supatcha" or the "Company") is pleased to announced today that its Board of Directors has authorized a share repurchase program of up to $3 million of the Company's outstanding common shares. The repurchase program will be funded by the Company's available cash. The Board of Directors believes that a share repurchase program would be in the best interests of the Company and its shareholders, and that it will not impact the Company's ability to execute its growth plans.
Commenting, Nikolae Yagodka, CEO and Director of Supatcha said, "We are very pleased to announce this repurchase program to benefit shareholders at a time when we believe our stock is undervalued. This initiative will improve future earnings per share by reducing the number of shares outstanding. The repurchase program reflects the Board of Directors' continued confidence in the Company and the commitment to enhance shareholder value. Our shareholders have been and continue to be our most important asset, so rewarding them is a high priority. We have been astounded by the sharp decline in our share price over the past year. Nevertheless, we believe Supatcha's share price has become significantly undervalued based on the strength of the Gold market's current level and future outlook."
Acquisitions of stock under the repurchase plan will be made from time to time as permitted by securities laws and other legal requirements, and subject to market conditions and other factors. The repurchase program will be funded by the Company's available cash. The plan will continue as long as periodic management reviews determine it to be fiscally feasible and may be discontinued at any time.
In addition, the Company expects the NI 43-101 technical report for the Barlevskoye and Vynohradiv gold projects to be completed and filed by December 05, 2010. The report will provide measured, indicated and inferred resources that will be the basis for the Preliminary Economic Assessment and final feasibility studies that will convert those resources into reserves.
About Supatcha Resources Inc. (OTCBB: SAEI)
Supatcha Resources Inc. is a gold exploration company focused on acquiring and developing mineral properties in Ukraine. Supatcha is in the final stages of acquiring two additional gold mining projects, with world-class potential, in southwestern Ukraine.
Notice Regarding Forward Looking Statements
This news release contains "forward-looking statements", as that term is defined in Section 27A of the United States Securities Act of 1933 and Section 21E of the Securities Exchange Act of 1934. Statements in this press release which are not purely historical are forward-looking statements and include any statements regarding beliefs, plans, expectations or intentions regarding the future. Actual results could differ from those projected in any forward-looking statements due to numerous factors. Such factors include, among others, the inherent uncertainties associated with mineral exploration. We are not in control of metals prices and these could vary to make development uneconomic. These forward-looking statements are made as of the date of this news release, and we assume no obligation to update the forward-looking statements, or to update the reasons why actual results could differ from those projected in the forward-looking statements. Although we believe that the beliefs, plans, expectations and intentions contained in this press release are reasonable, there can be no assurance that such beliefs, plans, expectations or intentions will prove to be accurate. Investors should consult all of the information set forth herein and should also refer to the risk factors disclosure outlined in our annual report on Form 10-KSB for the last reported fiscal year, our quarterly reports on Form 10-QSB and other periodic reports filed from time-to-time with the Securities and Exchange Commission.
SOURCE Supatcha Resources Inc.
OT. WISHING GOOD LUCK!!!
http://zvcapital.com/
Actually I was much better several years ago before the market turn into the "racket" that we know........More and more is like starting "sunny" and end with "rain"....TRDY today is a typical example ?????......
I am keeping a little bottle of Alda with my few shares and a special GRAPPA "home made" to celebrate the Glorious Day when it will happen ????? - In the meantime I am contributing to keep this board going........hope someone else will do it as well.....
Do you have any good pick to share ???? jocking......
ot............posting picks is fine..... at least until we hear something from ALDA......you had a couple good ones lately....must be that clear Canadian air, or, your stock-picking is maturing like a fine wine....
LOLOLOLOLOLLLL Surfin! You really have a way ........
By the way, I hope you no mean that I should stop posting picks???
Take care
janet
ot....thanks janet, I'll check it out..... I am thinking of renaming this board to "janet's penny picks"....
Hi SurfinNj, did you see this Board??????
http://investorshub.advfn.com/boards/board.aspx?board_id=18004
Happy week end,
janet
Hi SurfinNj,thank you for looking after me, appreciate! it is what I intend to do tomorrow unless .......something come up????
Received a tip for X-TAL MINERALs CORPORATION- symbol "XMT.H" Canadian Co. XMT.H is Halted, going to check it out.....
Take care SurfinNj,
janet
OT.....hi janet....trdy made a great continued move up today....congrats...you might consider taking some off the table, lock in some profit, because technically it should pull back soon...... good position to be in, though..... nice call
OT. TRDY looks like is "breaking out".......Keep a very close eye on this one .......
Good luck,
janet
Good luck to us!!
Ron.
some big buying toward the end of day today...around 150,000 at 10.5 I have a good feeling about that.....(especially since it wasn't me that bought it)...
Thanks SurfinNj. Nice close for boths. Looking forward......
Sweet Dreams
janet
OT.....hello "janet"....... just to let you know that AAVG closed higher today, and technically gives a buy signal....
http://stockcharts.com/h-sc/ui?s=AAVG&p=D&yr=0&mn=6&dy=0&id=p73148617900&a=211129785&listNum=1
I would expect TRDY to get hammered back within a day or 2, based on the recent huge run-up(jmo)....
hi paul..........enjoying some of those ALDA profits? (lol)....I still think T36 is better than Purell, but unfortunately, few people know about it!.....maybe they can still rectify that situation..... The flow rider looks neat, never have tried one though....
Just got off Royal Carib.'s "Oasis of the Seas" Largest cruise ship in the world. Everyone there (7000 incl crew) using the auto hand sanitizers throughout the ship. Purell of course. On a brighter note...you would have loved this one Surf... Two flow rider machines meant you never had to wait for a wave LOL - Paul
It was for you! I bought some TRDY friday at 0,009 as well as some SAEI
Hope both will do well...... AAVG takes more times......
Take care,
janet
There's always time to get some....if not, make the time.... by the way, AAVG made a nice little move....be nmice to see that break out to the upside...I will check out TRDY
Good morning SurfinNj, look into TRDY, might be still time to get some.....
Take care
janet
Whatever, it seems that I've been right, unfortunately, all along. Managment has no clue, or no intent. Bunch of useless slackers. They're only interest is in flipping shares. Too bad they won't get anymore spikes since they can't even come up with another BS NR. FU TO.
answer to email:
*** - we moved our offices to Richmond so to put all of warehousing and offices under the same roof - before we were scattered around 3 different locations.(and yes - makes much better economic sense) We continue to move forward with our therapeutic plans however as you are aware it is a long and costly process.As far as a liquidation sale goes, the retail markets still have a lot of hand sanitizer in inventory. Our wholesale pricing is competitive with most of our competition. Our wipes and hard surface disinfectant and cleaners are our sales focus right now.I will have to get back to you on the status of the China contract.
Scott Young
drum............thanks for the positive post..... I'm still not counting ALDA out, although the financials did not give much to cheer about.... With the great products, there is still the hope of finding additional financing, a partnership, or buyout, imo...
It's Sunday morning, and a new week beckons. So I'd like to say something positive about Alda Pharmaceutical for a change. I'm sure most are in agreement that present management is pathetic, and have proved beyond a shadow of a doubt a total inability, or possibly interest in advancing, and growing the core business. So what is there to be positive about?
They have a fantastic line of products. They have patents in place around the world. They have FDA approval. T36 has been labratory tested on many of the the most serious, and evil infective organisms out there, and destroyed everything in it's path.
This is the only reason I continue to hold. An effective, and focused management, with hopefully the finances behind them, could do wonders.
I can only dream...
Don't hold your breath. Financials are out, and looks like besides moving, and playing with their GD pps, they've been doing nothing. Kind of makes you sick. Should have dumped this crap a long time ago.
or one with a bunch of money.....em
A new CEO with the right priorities would be a new lease on life foe Alda.
drum............I wouldn't rule that out, if they got into a partnership.....em
Maybe TO will announce his resignation..Ya Hoooo!
bouch......... Well, I wouldn't want you to feel too bad, being a fellow poster(who sold off 90% of his holdings), however, I would prefer the deal.... but, your hope may be realized, just financials.....
Have a feeling you may be right - only 10% in right now so I hope for my sake that it is only financials & not some T.O. blockbuster deal !!! LOL - Paul
gut feeling, we might(or should) hear something from this comany soon........... anything would be nice, to know they are still kicking.....
I'll try to listen to it.... em
|
Followers
|
26
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
4094
|
|
Created
|
08/07/04
|
Type
|
Free
|
| Moderators | |||
INVESTMENT SUMMARY
- Received a Drug Establishment License ("DEL") and approvals from Health Canada for 30 of its 49 prescription generics products; already has manufacturing approved, labeling and packing secured, and is establishing a sales force 1Q15 salesforc
- Simple business model: Vanc sources drugs that have already obtained FDA equivalent approvals through affiliated companies in China and India, in exchange for manufacturing rights when the drugs are approved by Health Canada
- Potential to scale quickly, comparable to another Canadian company, Paladin Labs, that got bought out for $1.6 billion in 2013 by Endo Health (NASDAQ:ENDP)
- CEO, Arun Nayyar has an extensive track record in the industry - specifically generics - having held executive positions with pharma companies in Latin America, Asia, and Canada and played an instrumental role opening up new markets abroad
- Pre-revenue, albeit with all of the components in place to immediately impact the generics market in Western Canada, and unlock shareholder value through a number of visible, value-unlocking events throughout 2015
Between 2010 and August 2013, brand-name drugs with sales totaling $6 billion a year in Canada lost their patent protection - opening the door to far cheaper generic copies, according to the IMS Brogan market-research company.
Compounding the so-called "patent cliff," a growing list of insurance companies that manage private, workplace drug plans have recently made substituting generics for brands a mandatory policy - a step that most government plans took years ago. The result: more then two thirds of prescriptions in Canada are now filled with generics, while some brand manufacturers lack new drugs in the pipeline to take up the slack.
COMPANY OVERVIEW
Vanc Pharmaceuticals Inc. ("Vancpharma") (OTCQB: NUVPF) is a Canadian company (TSX-V: NPH) focused on providing Canadian health care professionals and consumers with high quality, affordable generics and over-the-counter ("OTC") healthcare products. They are the first Canadian generics company in Western Canada.
GENERICS PORTFOLIO
The company's currently approved in-licensed generics portfolio consists of 30 molecules, comprising of 67 dosage forms across various therapeutic categories: including both chronic (long-term) therapy and acute (short-term) therapy. Management estimates that the aggregate annual Canadian sales of its 30 approved products represents "a $1-billion market opportunity". Furthermore, the company plans to launch "with additional products and we will provide further updates in the coming months," said Arun.
The generics division of the company was only launched last Spring, since then the new management team has reached a number of significant milestones including:
On April 15th, 2014 Vancpharma announced that it had signed Cross Referencing Agreements ("CRA") "for prescription generic products for Canadian markets. These agreements are with three large pharmaceutical companies and cover 48 prescription generic products. The suppliers will handle manufacturing, and Vancpharma will market and sell these new product lines under its own label.
On November 18th, 2014 the company received approval via a Notice of Compliance ("NOC") from Health Canada for 22 generic molecules, comprising 51 dosage forms.
On December 10th, 2014 the company announced that it had been issued a drug establishment licence ("DEL") (licence No. 102220-A) by Health Canada. "The issuance of a drug establishment licence, along with the approval of our partner's GMP manufacturing site, is a key step towards the commercialization of our generic drug portfolio," said Arun Nayyar, CEO. The licence allows Vancpharma to import pharmaceutical products and distribute them within Canada.
This news was particularly important for the company and shareholders because: it positioned Vancpharma to become one of the only 40 companies in Canada that are licensed to manufacture current and future drugs at its GMP facility; allows Vancpharma to import from other manufacturers across the world and faces less barriers to entry; allows for importing both generic and non-generic drugs thus allowing the company to compete with other companies in branded drugs as well as their core generics business; and adds major clout when negotiating for exclusivity rights across Canada. Previously, manufacturers wouldn't commit their exclusively to Vancpharma as they were unsure if it could make good on importing and selling their products.
On December 15th, 2014 the company received an NOC from Health Canada for 7 additional generic molecules, comprising 15 dosage forms.
Lastly, on January 14th, 2015 Vancpharma placed inventory purchase orders for 30 generic molecules and expects to deliver these products 2Q 2015. "We are excited to take this important step towards commercialization and look forward to launching sales in Q2 2015. These 30 molecules represent best-selling generics in the Canadian market and our aim to provide Canadians with quality and cost-effective products is well served by them," said Arun. "Our initial marketing and outreach activities with select pharmacy customers in Western Canada have been positive and we look forward to working with our partners."
Figure 1: Generics Portfolio

Source: Press Release/Company Website
*Note: I'm aware that's only 29, however the 30th hasn't been updated on the website although it's been mentioned as approved (new approvals could come at any time)
Here are the next steps with the generics portfolio:
Figure 2: Generics Through Global Partnership
For those wondering what Vancpharma's margins are, the company has not made that public yet. As you can imagine, they don't want to expose their margins for specific drugs too early. Investors will be able to see them in the financial statements at a later date. However, generics are quite lucrative. Vancpharma will be taking on the risk of inventory, licensing, approvals, sales and marketing, whereas the manufacturer takes and fills an order from Vancpharma as necessary and collects payment. Accordingly, the reward follows the risk and in this case, Vancpharma would pay a fixed price to the manufacturer depending on the size of the manufacturing run. If you look at the financials of another generics company, Biosyent (TSX-V: RX) you will see margins typically ~70%-75%.
The margins generated from strategic cross-reference partnerships, while very enticing, pale in comparison to the financial opportunities presented by exclusive or co-development partnerships which management has indicated they want to pursue in the future.
OTC PRODUCTS
The OTC Products Division is focused on the marketing and sales of novel and proprietary healthcare products and consists of four (4) such products, all of which now have a Natural Product Number.
Figure 3: Vancpharma Pharmaceuticals OTC Products
It can't and won't be the cash cow for Vancpharma like its generics portfolio - the Canadian market for these OTC products is only ~$60-70 million - but it should actually start generating revenue sooner. Manufacturing should commence in January, with the first sales hopefully starting to come sometime during March.
Looking ahead at the future pipeline of OTC products, they include nutraceuticals, vitamin supplements, and skin care products.
MANAGEMENT
The team has extensive experience and expertise that spans across various functions such as research, development, manufacturing, and marketing of generics and OTC health care products in the global pharmaceuticals industry. This understanding of industry best practices and strong insight allows the company to identify emerging trends in medicine and the marketplace.
The secret to being able to license so many drugs within such a short period of time, less than a year after being restructured is CEO, Arun Nayyar.
Arun only joined the company November 25th, 2013, but came with an extensive track record in the industry - specifically generics - having held executive positions with pharmaceutical companies in Latin America, Asia, and Canada. He has been instrumental in opening up new markets abroad, and domestically his accomplishments include Director, Business Development and International Sales for Shoppers Drug Mart ("SDM"), and consulting for Sanis Healthcare, George Weston Ltd. (Loblaws group), and SDM. To have a more extensive look at Arun's resume and job history, you can view his LinkedIn here.
Some information that you won't find on LinkedIn is that he's an owner of a few Shoppers Drug stores in the Vancpharmaouver area, and also has deep-ties, and solid connections in India. This helps to secure licensing of the generics in exchange for manufacturing rights when Health Canada approves the products.
The newest hire, replacing Jamie Lewin as director and CFO and announced December 3rd, 2014 was Aman Parmar. "I look forward to working with the team at Vancpharma Pharmaceuticals and am impressed by how far they have progressed with limited capital," said Mr. Parmar. "Capital efficiency and creating shareholder value will be my primary focus at Vancpharma." Since joining Vancpharma, Aman has purchased 135,000 shares on the open market ranging from 19 to 24 cents - clearly indicative that he believes the company is undervalued at these levels.
Given the fact that the company is ramping up its business, they've already started putting together a sales and marketing team, it wouldn't surprise me if another executive was brought in to help.
You can read more about the entire Vancpharma team by clicking this link (note, this page needs to be updated with Jamie/Aman).
FINANCIALS
Although Vancpharma is a pre-revenue company, I see a multiple number of visible, value-unlocking events through 2015 that can meaningfully impact stock performance.
Figure 4: Summary of Quarterly Results
The company closed an oversubscribed, non-brokered private placement for gross proceeds of $1,141,000 by issuing 7,607,332 units at $0.15/unit on December 11th, 2014. "This round of funding enables us to move our portfolio of generic drugs and OTC products into commercialization," said Arun Nayyar. "Specifically we will be acquiring generic drug inventory and building our sales team to target pharmacy customers."
The company is financed for the time being, but may have to do another round in March depending on how the roll-outs are going, and for general working capital purposes. If so, I'm sure that it would be strategic - I would think at least >$0.20, comprised primarily by sophisticated retailers and brokers, and it would not be a raise of much more than $1 million. Management only wants to raise whatever money they believe is necessary right now because they know as soon as the company starts generating revenue that its valuation has easily be many multiples of where it sits today.
SHARE STRUCTURE
Shares outstanding: 44,374,407
Stock Options: 3,975,000
Warrants: 16,212,252
Fully Diluted: 64,561,659
Major shareholders of the company, and percentage owned include:
It's a tight share structure, the 'effective' float is ~9 million shares - much less than what's been traded the past few months - 42,798,548 shares at an average price of $0.194:
Figure 5: House Positions
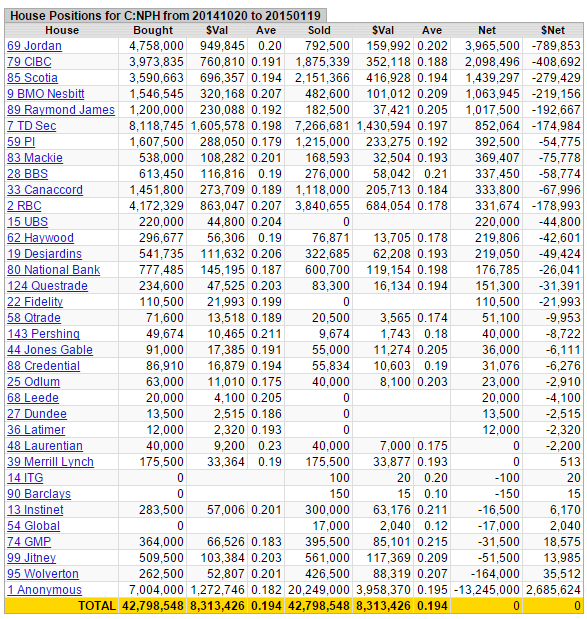
Source: Stockwatch
Figure 6: Management Insider Filings
REVENUE RAMP/VALUATION
It's a little early for me to try and assign a valuation, or forecast revenue and earnings for the company, however I anticipate doing so before year-end. The company hasn't informed investors of the specific margins and obviously we don't know the adoption rate because we don't know how good the sales team will be. Take a look at this article though, "The Top 11 Fastest-Growing Generis Companies", when compared some of the other generics companies, Vancpharma looks very undervalued based solely off of its standing drug portfolio.
The two best examples that come to my mind are: Paladin Labs and BioSyent (OTCPK:BIOYF).
Former pharma sales rep., and Cantech Letter contributor Hogan Mullally wrote that Paladin "became the poster child for Canadian specialty pharma. They built a remarkably successful business by acquiring the Canadian rights to a wide variety of drugs. These drugs were either too small for medium/big pharma, or were developed by a company without a Canadian commercial footprint, whatever the reason, Paladin was able to amass an eclectic and diverse portfolio of prescription and OTC drugs for the Canadian market. Through their "sum of the parts" strategy, Paladin grew to over $200 million in annual sales and in 2013 was acquired by Endo Pharmaceuticals for approximately $1.6 billion.
BioSyent is in between Vancpharma and Paladin, as it has already amassed an impressive portfolio by searching the globe to in-license or acquire innovative pharmaceutical products for the Canadian market. It too focuses on products that are too small for medium/big pharma, and have a competitive angle that can be exploited by a modest sales force. BioSyent's business model is structured to minimize risk, and to produce high growth. For the four years ended December 31, 2013 the company experienced a CAGR of 67% while consistently growing profits:
Figure 7: BioSyent Financials at a Glance
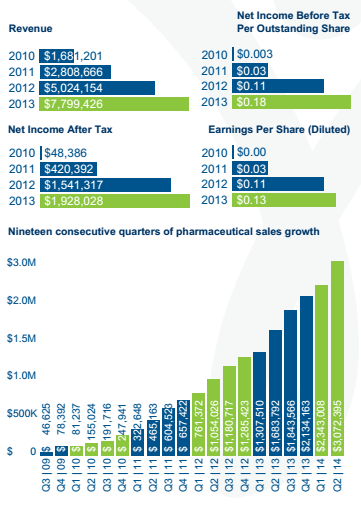
Source: Fact Sheet
There is an exit strategy for the company and shareholders - take-out target for an M&A transaction. It might take getting into a few hundred pharmacies, but the precedent has already been set. The initial adoption rate risk is reduced from the outset because between members of the management team, they own a little more than a dozen pharmacies. Not to mention now that Health Canada has granted the company a Drug Establish License it can pursue additional revenue opportunities and further de-risked the investment.
RISKS
Vancpharma is still considered an early stage company, as such there are a number of risks associated with making an investment at this point in time, including but not limited to:
(1) The company just hired a new CFO, and has a management team has dozens of years of business experience, most dealing with generic pharmaceuticals.
(2) The company plans to manufacture its products at four certified GMP pharmaceuticals factories in Canada, India and China. These U.S. FDA approved plants are capable of manufacturing a wide range of Generic Pharmaceuticals and OTC Health care products at these facilities, under the VANCPHARM label.
(3) Like I mentioned before, management controls a number of pharmacies which will de-risk the rollout process and immediately start generating revenue form the company's generics portfolio.
(4) Arun has built deep, extensive relationships in India and China, and has a lot of ideas to add more products to the portfolio (not to mention the future potential to co-development or development exclusively).
(5) The company sources its products from big products that are already approved by either the U.S. FDA or UPHRA, and also approved by Health Canada. As long as the company is in compliance with Health Canada guidelines then the ANDS application gets expedited, updated and faster tracked. The company hasn't had so much of a hiccup yet, because of the experienced team in place filing all of the paperwork.
(6) The company hasn't had an issue raising money despite the poor market for companies trading on the TSX Venture. The last PP was oversubscribed, and the shares were spread around to strong, strategic hands. Presuming management continues to achieve its milestones, I anticipate lots of eagerness for the next round.
CONCLUSION
Vancpharma set forth some ambitious goals, but has already accomplished so much in such a short amount of time that I really have large aspirations for it (and shareholders).
The company has a tried and tested business model which is simple to understand. Vancpharma enters into Cross-Referencing agreements with affiliated companies whom source products from China and India which are already approved, in exchange for manufacturing rights when the drugs are approved by Health Canada. This is very economical as there is a minimal cross-referencing fee paid to Health Canada, and a very large market opportunity (>$1 billion for current generics portfolio), with margins typically ~70%-75%.
Source: StockCharts.com
Taking a look at recent trading, there has been some healthy consolidation after the initial big run-up, ~33% off its 52-week high. MACD and RSI indicators are now in 'oversold' territory, and the stock is approaching its 200-day moving average. Up until a few months ago this stock traded 'by appointment only'. Since the middle of October, 42,798,548 shares at an average price of $0.194 have traded.
Now that the company has received 30 of its 49 Health Canada approvals (with the rest to be submitted shortly), there is a bit of work that needs to be done in order advance it from being a purely speculative growth biotech with Health Canada approvals, to one that's one being noticed by institutions and an American audience.
In the meantime, investors have two options: (1) buy now, taking the risk knowing that the company may need to finance further, but is extremely close to generating revenue; or (2) sit on the sidelines and wait until the second quarter financials are released, and make a decision then.
The latter is much less risky, however if management successfully executes on its business plan and stays on track with milestones, then I'm sure its share price will reflect it this, and it will be at a significant premium.
Bottom line, I think that there is a very compelling opportunity to invest right now given: (1) the recent pullback in share price; (2) the significance of forthcoming news releases to act as catalysts to unlock shareholder value; (3) management's achievements to date and their track record to deliver results; (4) market opportunity (>$1 billion on current portfolio) and attractive valuations of sector peers; and (5) lack of competition in the Western Canadian generics market and the opportunity to generate imminent revenue.
(For additional liquidity, NUVPF. trades in Canada on the TSX Venture as "NPH". 3-month average volume 637,531 shares/day.)
Please feel free to comment below or send me an inbox message if you have any questions or comments about this article.
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
