Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
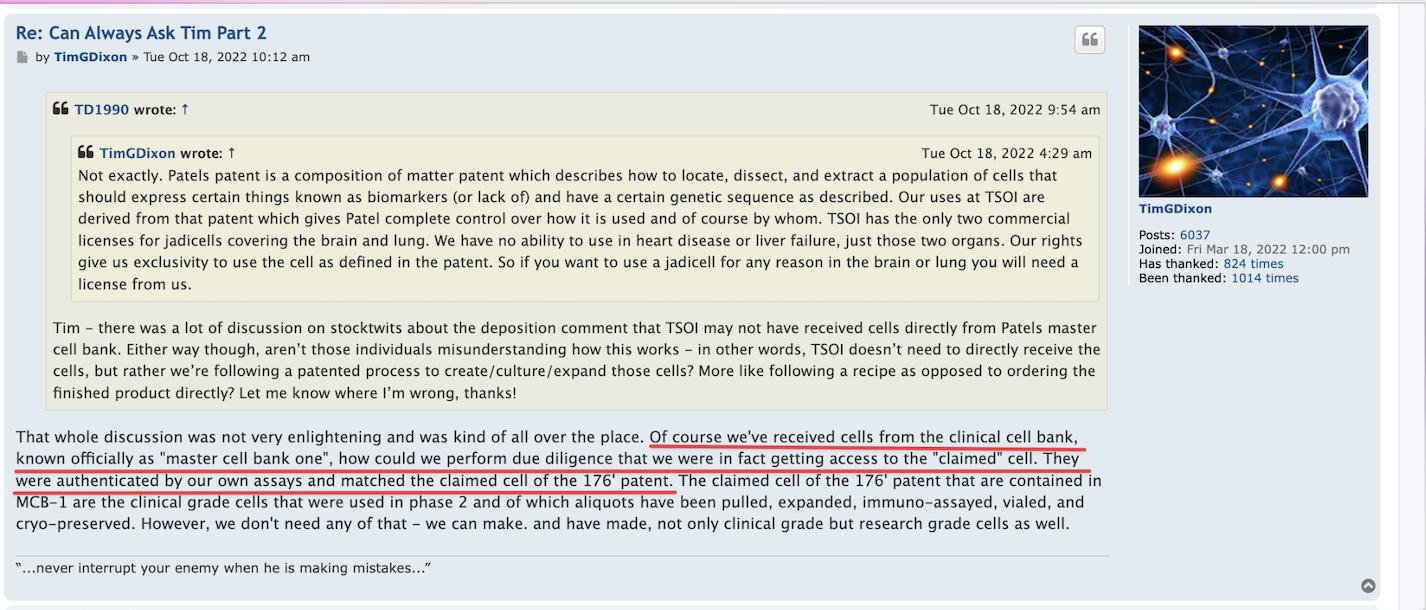
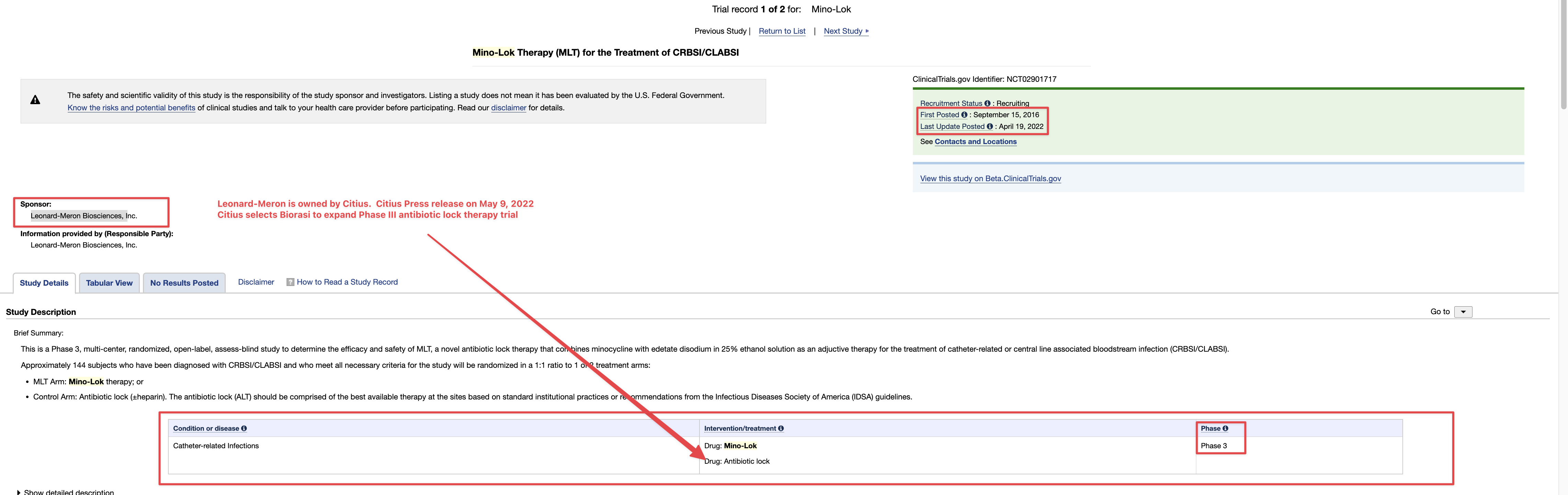
there was a new TSOI patent published today
by TimGDixon » Thu Jun 09, 2022 6:56 am
Not on Justia yet.
https://appft.uspto.gov/netacgi/nph-Parser?Sect1=PTO2&Sect2=HITOFF&u=%2Fnetahtml%2FPTO%2Fsearch-adv.html&r=13&f=G&l=50&d=PG01&p=1&S1=dixon&OS=dixon&RS=dixon
TREATMENT OF MAJOR DEPRESSIVE DISORDER AND SUICIDAL IDEATIONS THROUGH STIMULATION OF HIPPOCAMPAL NEUROGENESIS UTILIZING PLANT-BASED APPROACHES
Disclosed are means and methods of treating major depressive disorder and/or other disorders that predispose to suicide by administration of nutraceutical means, wherein said nutraceuticals are administered at a frequency and/or concentration sufficient to induce proliferation of endogenous neural progenitor cells. In one embodiment said nutraceuticals are comprised of green tea extract, and/or Nigella sativa, and/or pterostilbene, and/or sulforaphane. In some embodiment's nutraceutical compositions are utilized to overcome treatment resistant of currently used antidepressants.
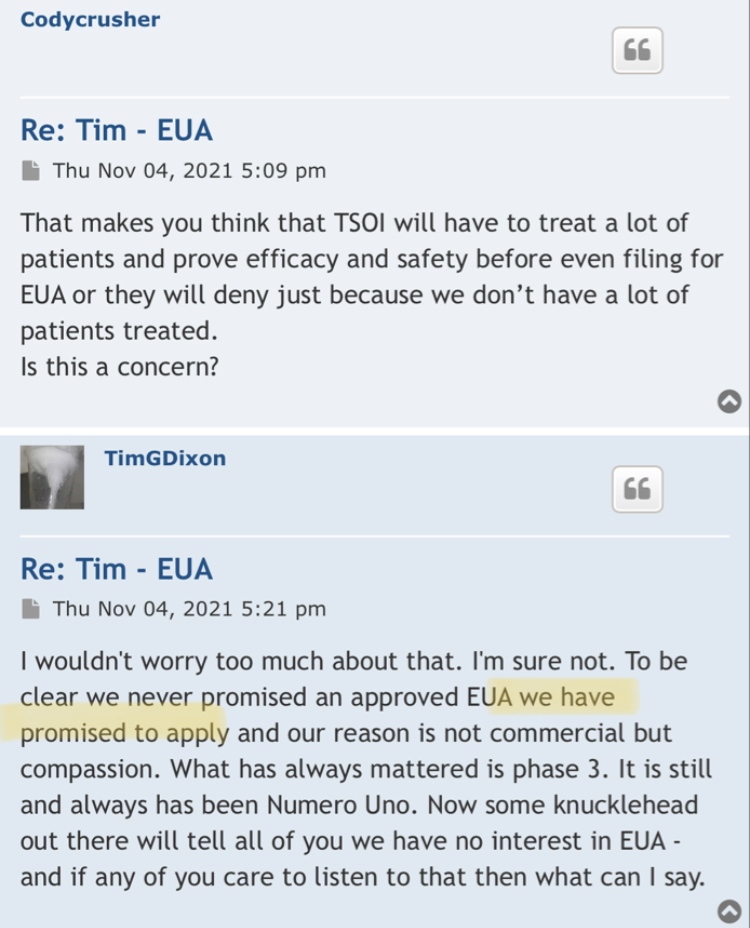
how many QM patents are in the mix?
I have to do a review .
Lay em on me TR TR , and i will too . Let's compare notes :)
-Cents
Here is a PR on patent # 2
March 10, 2022 at 6:35 AM PST
Share this article
Therapeutic Solutions International Granted Second United States Patent on
QuadraMune® for Prevention, Inhibition, and Treatment of COVID-19
https://www.bloomberg.com/press-releases/2022-03-10/therapeutic-solutions-international-granted-second-united-states-patent-on-quadramune-for-prevention-inhibition-and
Therapeutic Solutions International announced today issuance of United States
Patent #11,266,707 entitled “Nutraceuticals for the prevention, inhibition,
and treatment of SARS-CoV-2 and associated COVID-19.”
and in the future TR TR, are more QM - related patents .
from the Leonard A. Farber interview:
Medically speaking, why is Quadramune such a powerful natural supplement that people should consider taking?
"QuadraMune consists of 4 main ingredients that have been proven in clinical trials to decrease the inflammatory response in the body as well as modulate the susceptibility by inhibiting immune suppressive enzymes. In other words and can be immune stimulating or boosting and preventative of an unhealthy immune response. This allows the body a better chance not only fight the onset of an infection or inflammatory reaction but also to lessen the severity of certain illnesses, especially those that are inflammatory-based in nature."
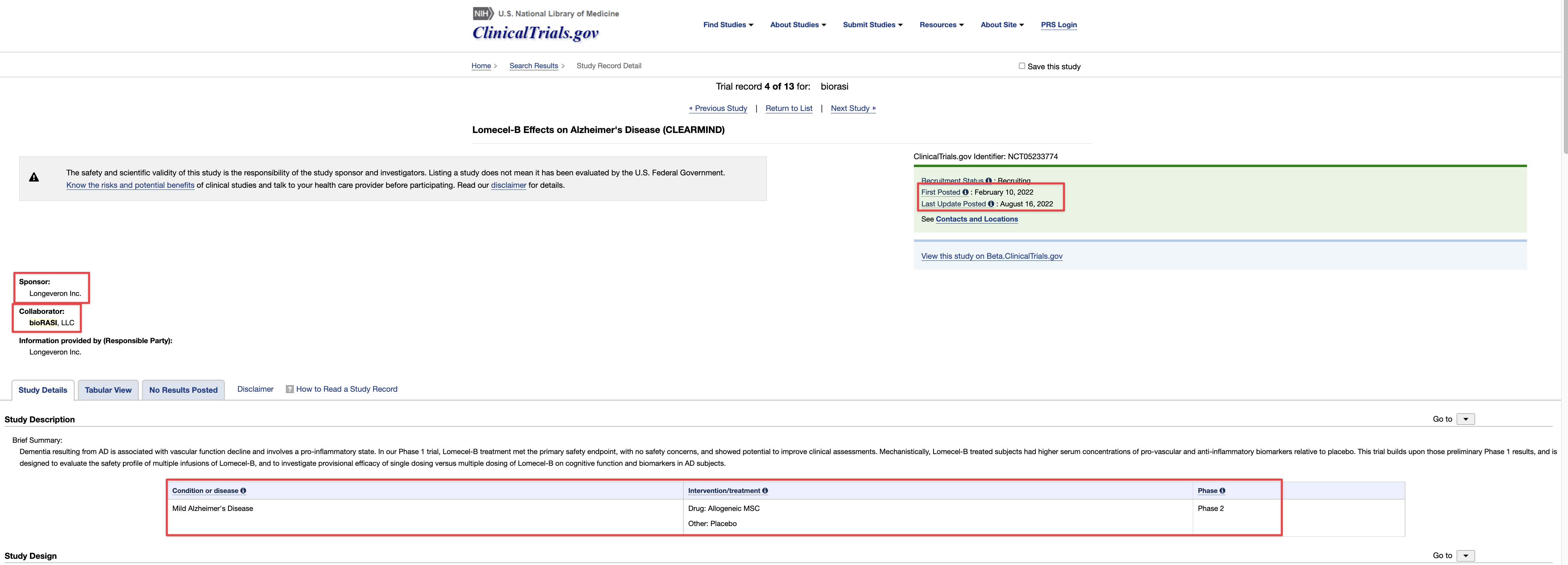
https://theorg.com/org/curescience/wires/dr-boris-reznik-of-bioras-d0e841f6
$TSOI QUADRAMUNE NUBRAINER NANOSTILBENE are need of the hour to whole world post pandemic
https://www.brainzmagazine.com/post/q-a-with-dr-leonard-a-farber-boosting-immunity-and-reducing-inflammation-with-supplements
they put this up on their website, so it aint a rumor any more - imo
Venvalo Group and Dr. Boris Reznik to Present TSOI at BIO International Convention
in One on One Partnership Meetings June 13-16, 2022·San Diego Convention Center
https://therapeuticsolutionsint.com/venvalo-group/
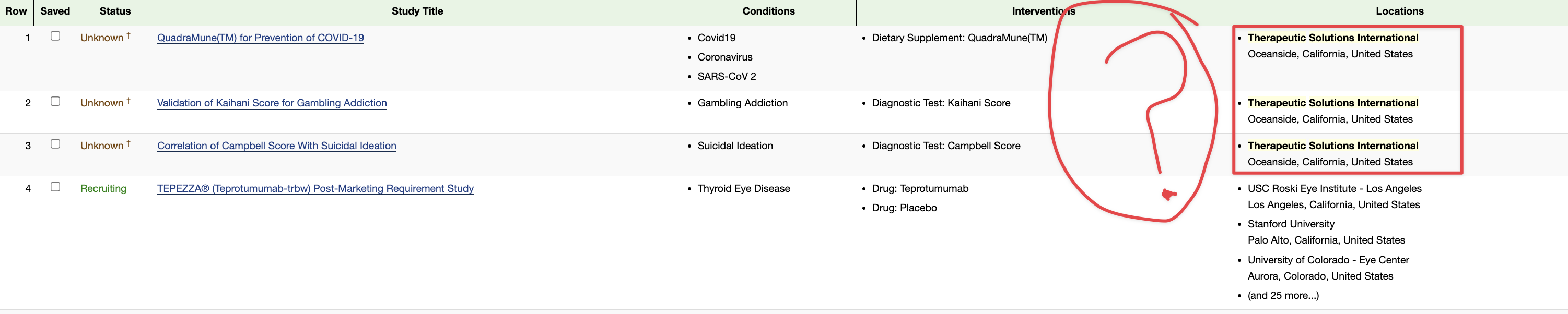
TSOI is a clinical stage regenerative medicine company. Below is our product pipeline. We would love to discuss with you these various areas of potential out-licensing and/or collaboration:
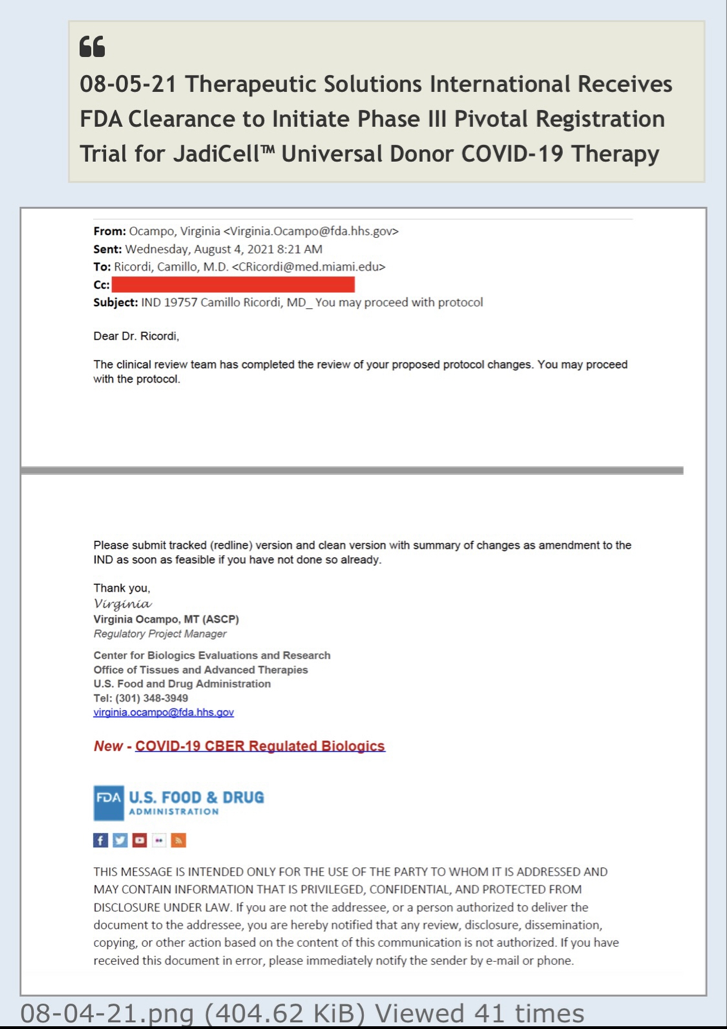
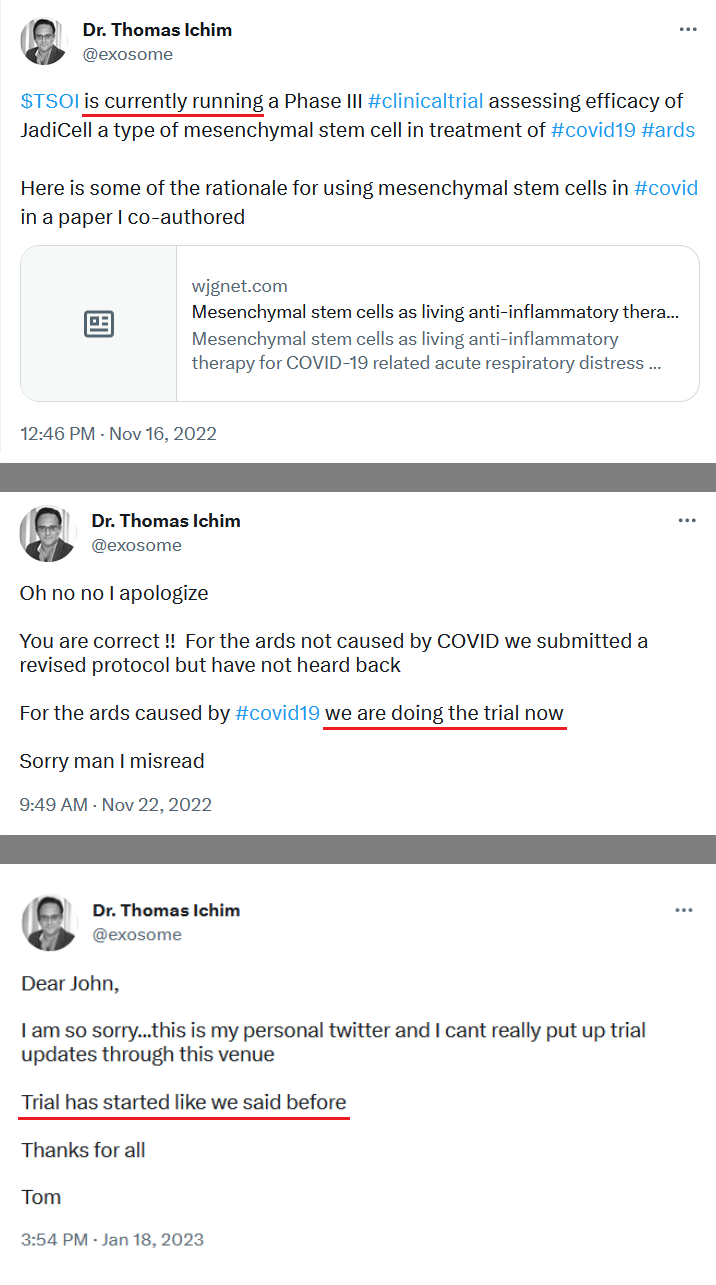
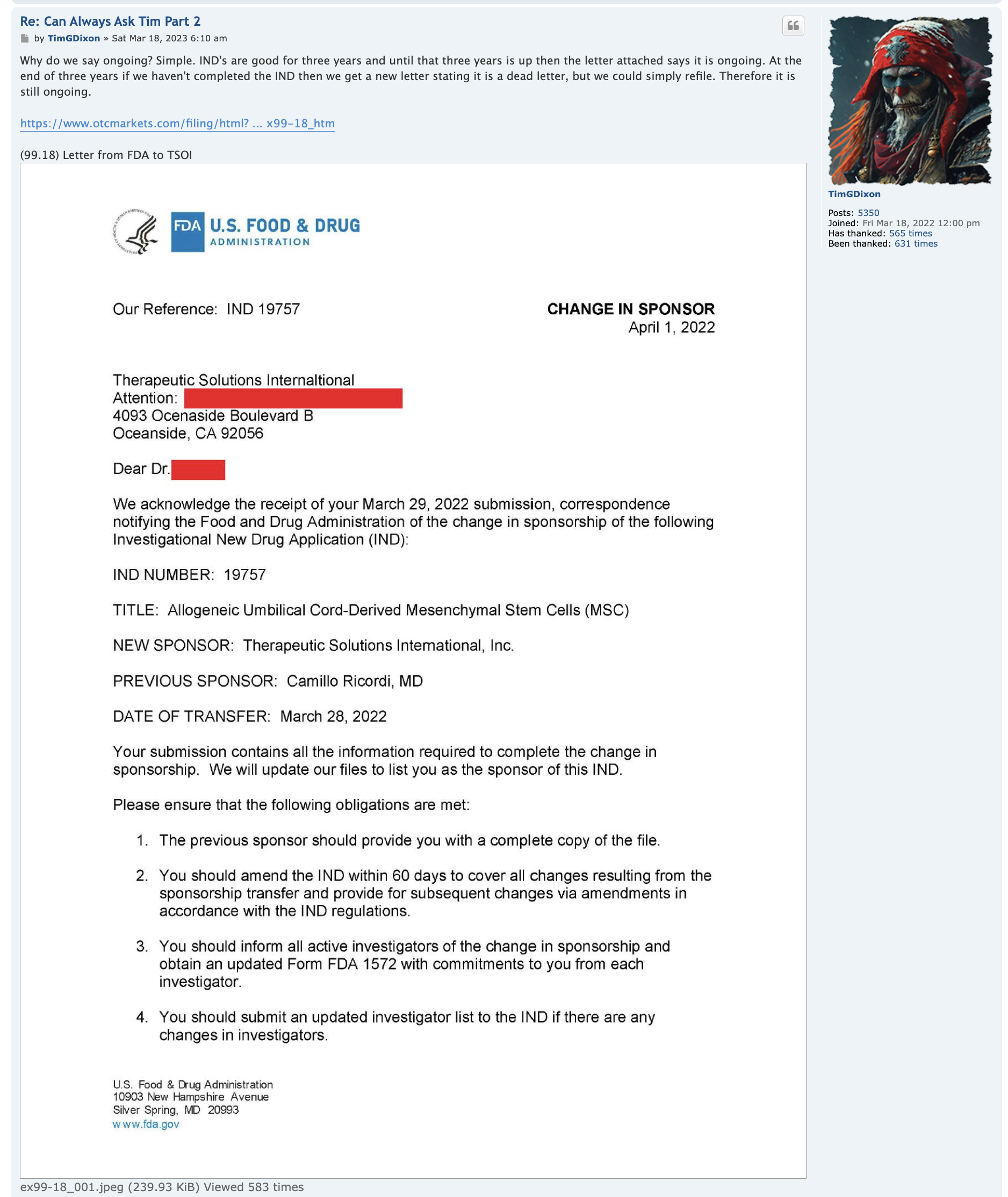
JadiCell for COVID-19 (Phase III ongoing #19757)
JadiCell for COPD (IND #28508)
JadiCell for Chronic Traumatic Encephalopathy (IND #27377)
JadiCell for Idiopathic Pulmonary Fibrosis (Late Preclinical)
StemVacs Dendritic Cells for Breast Cancer (IND #28508)
StemVacs-V iPSC Derived Tumor Endothelial Targeting Vaccine (Late Preclinical)
QuadraMune Nutraceutical for COVID-19 Prevention (Commercialized and Validated by Trials and John Hopkins Publication) John Hopkin’s University Publishes Efficacy of QuadraMune™ Ingredient Sulforaphane at Inhibiting Coronavirus in Laboratory Studies | BioSpace
I noticed the one on one meetings mentioned, as you scroll down.
Looked to see if they were " members ", but they were not listed.
(Should have been on page 69.)
that news by itself
should have drawn attention now.
but wait ,
you can have two partners or more from Bio-Pharma , while you watch it tank -
This is by far the funniest anomaly in the OTC .
lololol
even rumors can't fix it.
Even when it's presented at the bio,
this stock would not move up.
what is seriously wrong with this picture?
Who are the oppressors ?
Please drag all of them out from under their rocks, CUZ they have some serious 'Splainin to do on how they wrecked this stock
Hat tip to TR TR on ST !
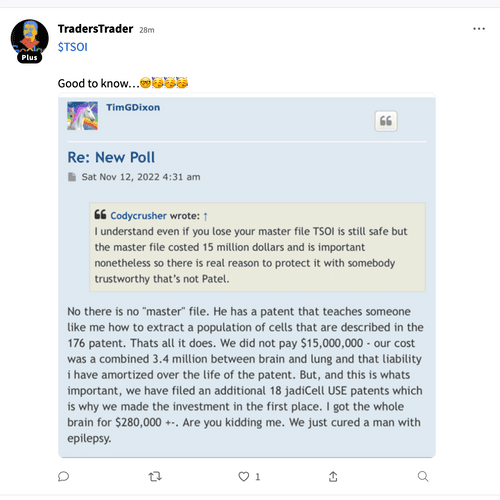
If this is real , then put Boris in coach !
Venvalo Group and Dr. Boris Resnik to present TSOI @ Bio-International Convention
June 13-16 2022 San Diego Convention Center
https://stocktwits.com/symbol/TSOI
Venvalo Group and Dr. Boris Reznik to Present TSOI at BIO International Convention
in One on One Partnership Meetings June 13-16, 2022·San Diego Convention Center
I'm excited TR TR
thanks for the excellent DD.
-Cents
Lost my father to lung disease. This would be huge
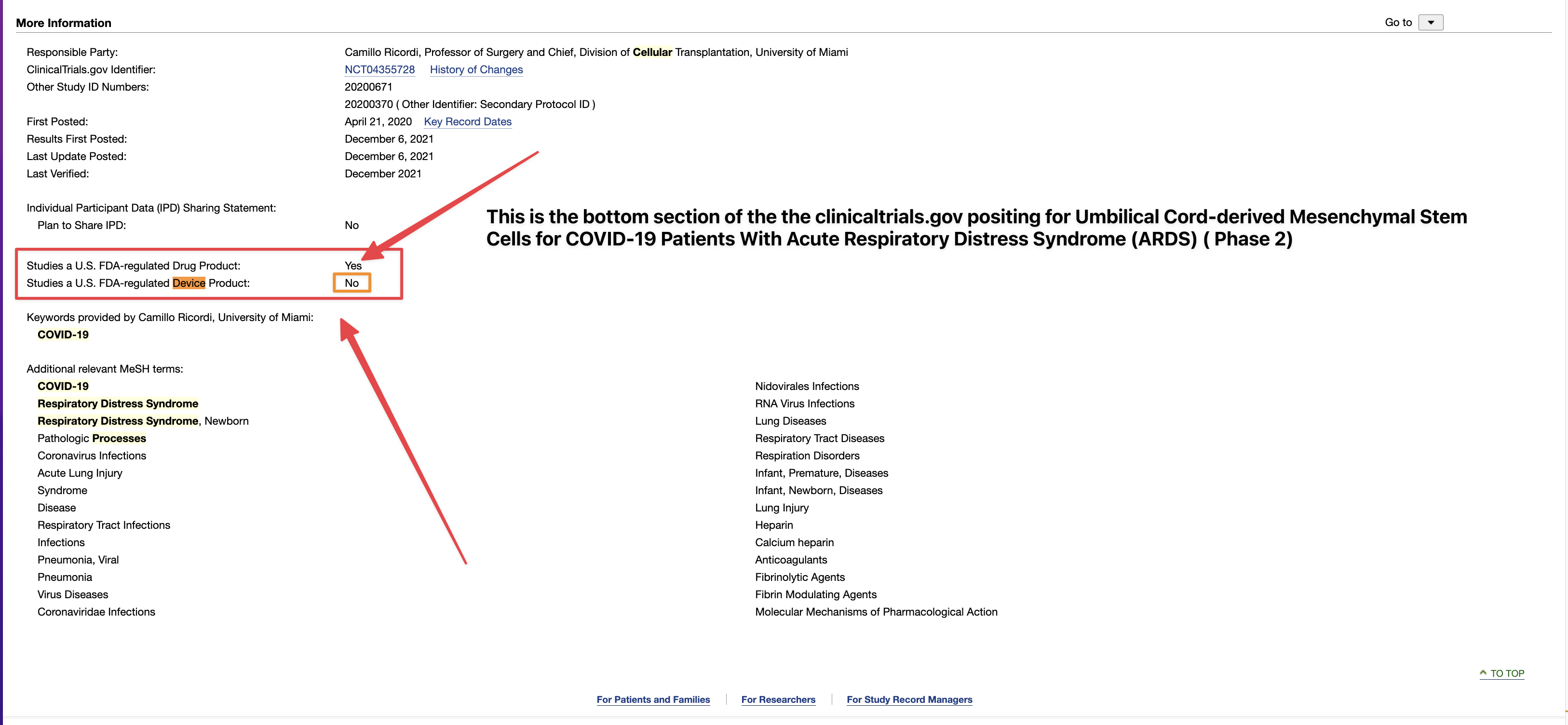
TSOI Nets IND Number Ahead of Proposed JadiCell Clinical Trial
https://copdnewstoday.com/2022/06/02/study-evaluate-jadicells-copd-given-go-ahead-fda/
Note: This story was updated June 6, 2022, to note that the U.S. Food and Drug Administration has assigned an Investigational New Drug application (IND) number to Therapeutic Solutions International experimental JadiCell therapy. The FDA has not yet approved the company’s IND application.
The U.S. Food and Drug Administration’s review of JadiCell as a treatment for chronic obstructive pulmonary disorder (COPD) has taken a step forward with the assignment of an Investigational New Drug (IND) application number ahead of a proposed clinical trial to evaluate the therapy.
The company asked the agency for permission in the form of an IND application following TSOI’s announcement that the therapy preserved lung function in mice with inflammation-induced lung damage.
JadiCell is currently being tested in a Phase 3 trial for COVID-19, and TSOI was recently granted an IND for chronic traumatic encephalopathy (CTE), or brain degeneration caused by repeated head trauma.
“COPD is a devastating disease, which in my opinion, is ripe for stem cell intervention,” James Veltmeyer, MD, chief medical officer of TSOI, said in a press release. “Based on the striking animal data in COPD models, as well as clinical recoveries in other pulmonary conditions that I have witnessed, I am eager to begin formal clinical investigations of this cell.”
Recommended Reading
COPD heart disease | COPD News Today | illustration of lungs
May 26, 2022 News by Patricia Inacio PhD
COPD Patients Have a Higher Risk of Heart Disease, Study Shows
With the IND, the FDA assigned the stem cell therapy the new name of “Allogeneic Umbilical Cord Mesenchymal Stem Cells, JadiCell; Intravenous injection.” In addition, the company has already begun discussions with the FDA regarding clinical trial details and participant population.
COPD is marked by lung inflammation and damage to alveoli — the tiny air sacs in the lungs responsible for gas exchange. The precursor cells that line the alveoli, called alveolar epithelial progenitor cells, cannot adequately respond to this damage by generating enough new alveoli.
JadiCells are mesenchymal stem cells (MSCs) — a type of adult stem cell found in several tissues that can give rise to many different cell types. MSCs possess anti-inflammatory, neuroprotective, and regenerative properties that have made them increasingly popular as potential therapeutic avenues for some conditions.
According to TSOI, JadiCells, derived from the lining of the umbilical cord, may outperform other types of stem cells in producing certain therapeutically relevant factors and be stimulated in alternate ways to enhance their regenerative effects.
In a mouse model of emphysema, a severe form of COPD, JadiCells reduced lung injury and surpassed stem cells from other sources, including those from adipose (fatty) tissue or bone marrow, in their ability to do so. Preclinical data also indicated JadiCells may reduce levels of specific pro-inflammatory proteins, while increasing those of anti-inflammatory markers.
Supporting JadiCells’ use in COPD, data from the Phase 3 COVID-19 study demonstrated 100% effectiveness in preventing mortality among patients under the age of 85 who were on a ventilator in the intensive care unit. In patients older than 85, the survival rate was 91%.
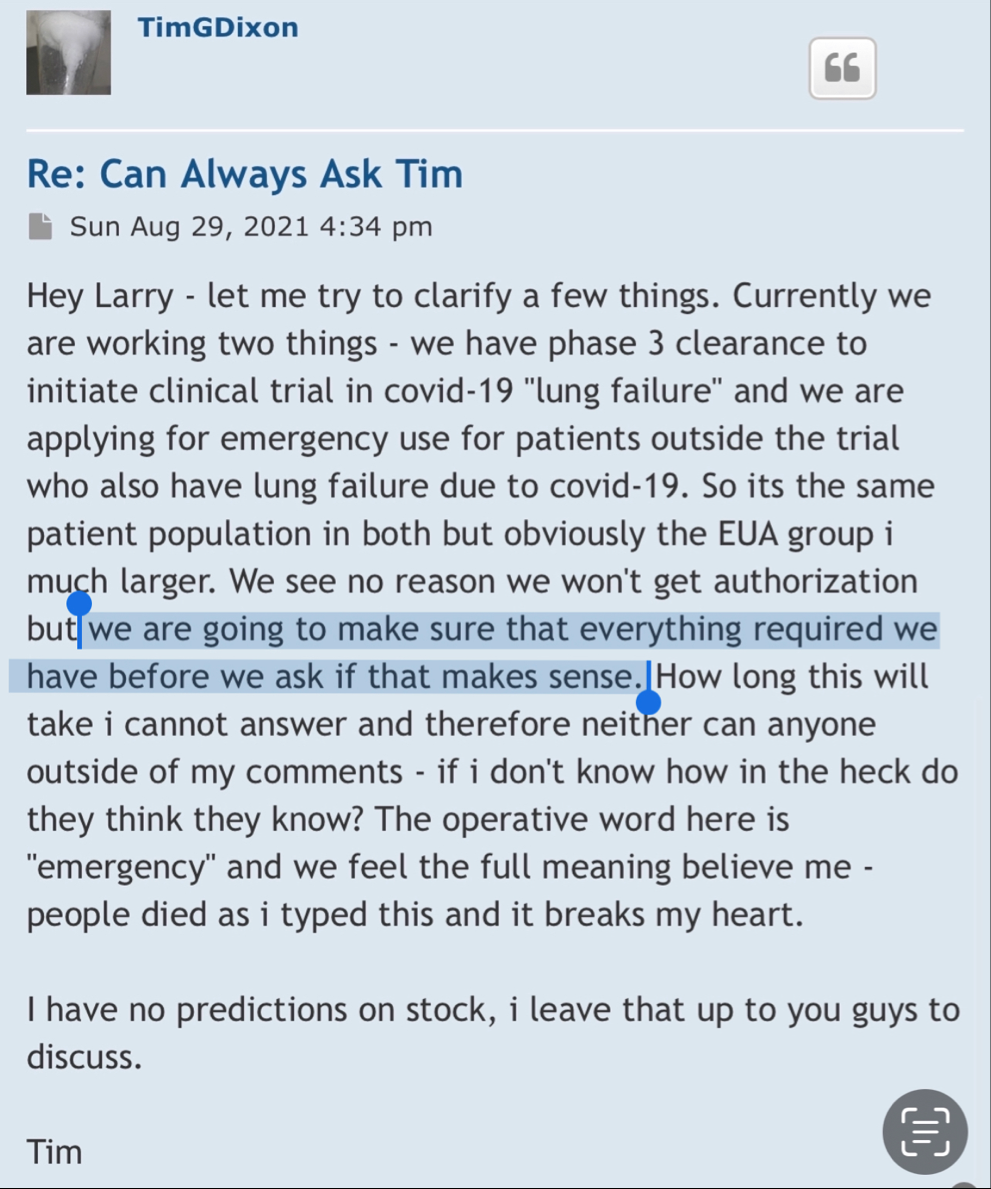
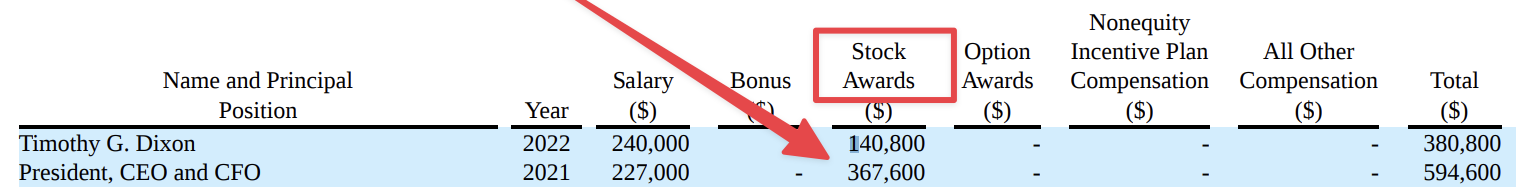
“We are thankful to the FDA for rapidly initiating the review of our work and we are confident that through this interaction we will create a trial that is beneficial to patients suffering from this condition for which to date, no curative options exist,” Timothy Dixon, president and CEO of TSOI, said.
CRP- (C reactive protein) level is one of the things that will show the inflammation level .
Mine has been normal , after taking QM .
The second time i got covid , the recovery time was cut in half , as i knew the beginning symptoms from the first time i got covid, and doubled my QM to 2 per day .
QM shows great promise as an immune booster for the immuno-compromised.
The first thing to go is teeth , and those expensive root canals .
so i always keep taking QM , to avoid the endo (endodontist).
-imho
ThanX... would luv to hear if you think of your next blood work results... thought about it before and put it off but think I will order some...
My plan is to see if there’s any impact on blood work in a few months.
Inflammation is being identified as such a problem I’m willing to continue to use.
Sounds good... Thank you for the update...
I can't say that Quadramune is the reason why I haven't been sick while others near and dear to me have been. But generally, I have been feeling better since I started taking QM.
To be completely transparent, I did recover from Covid last June, so I don't know if my natural immunity is helping or if it's QM.
Hope you don’t mind the input? But next time you’re at the doctors, it might be worth the trouble, to have a blood test, to see if you have developed natural immunity for COVID. Wondering if you had it and didn’t even know it. Seems unusual to live in the same house, and you didn’t get it. ( Quadramune???)
I should say I take 2/day.
Bought my first bottle in Dec., 2019, and have been taken it ever since. It was 3 months before it helped with my breathing. After that It got better and better. I never had any bad lung issues, but I did smoke cigs and pot for many years, cigars too. Noticed that my beathing wasn't as good when I played sports, or ran before taking Quad. Don't put anything into my lungs now for a while. But I noticed a pretty good improvement after taking Quad, especially when I exerted myself. I still take it as a precautionary measure. My wife got Covid, I didn't. Could be me, or maybe Quad helped, don't know. Also, I don't take the usual dose, I switch between 1 and two a day.
That’s pretty cool. Thinking of giving it a try
Using QM for a yr. No colds or sinus infections or Covid. Nothing! all of them happened all the time. Plus C and D3 and zinc. Your guess is as good as mine. Stay well
I’ve been using for 4 months. I haven’t really noticed any impact. I haven’t been ill but I don’t know if that’s been aided by the product or not. In general I don’t get sick that often. My plan is to see if there’s any impact on blood work in a few months.
Inflammation is being identified as such a problem I’m willing to continue to use.
I've been using QM for 6 months and will continue to do so.
If you open the link below & scroll down to line #66 you will see as of 5-26-2022 THERAPEUTIC SOLUTIONS INT INC [US] was added to OEPM for Stimulation of NK Cell Activity by QuadraMune Alone and together with Metformin
https://www.oepm.es/es/informacion_tecnologica/informacion_gratuita/Alertas_Tecnologicas/getXls.xls?id=68400&n=CORONAVIRUS:%20DIAGNOSTICO%20Y%20TERAPIA%20EN%20HUMANOS
OEPM is “The Spanish Patent and Trademark Office is an autonomous agency of the Ministry of Industry, Trade and Tourism of Spain”
This could possibly be a sign of further product fulfillment in more countries to come.
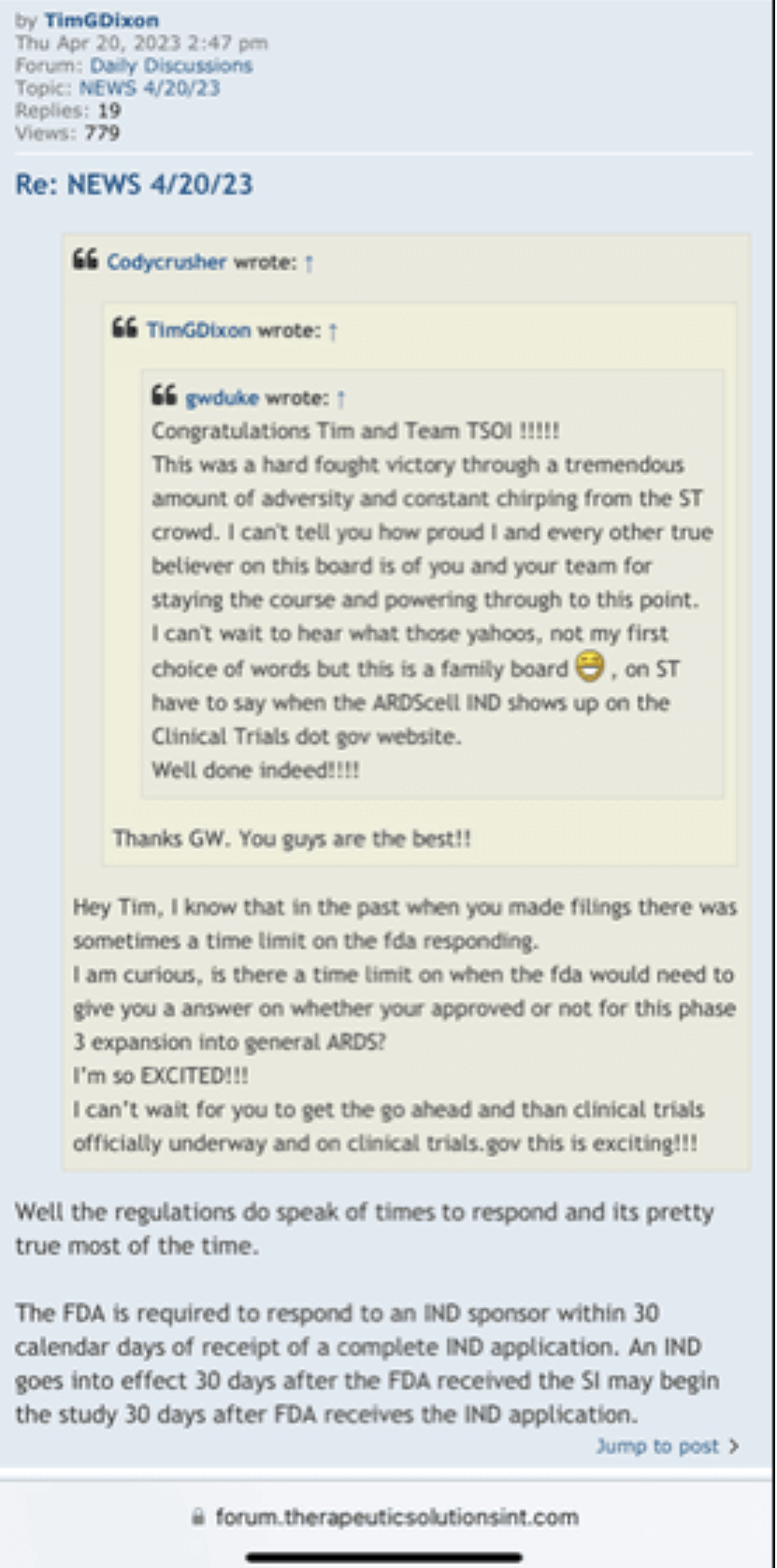
Post by TimGDixon » Sat Jun 04, 2022 9:21 am
Here's a positive vibe we were just approved for remote fulfillment in Canada & Mexico on Amazon. So if you have friends in those countries they will soon be able to obtain our products on Amazon.
https://forum.therapeuticsolutionsint.com/viewtopic.php?t=73&start=210
That being said I could have worded it better so I will request fit removal. I see your point
Hold on let me get that information for you, on it….
What is not true
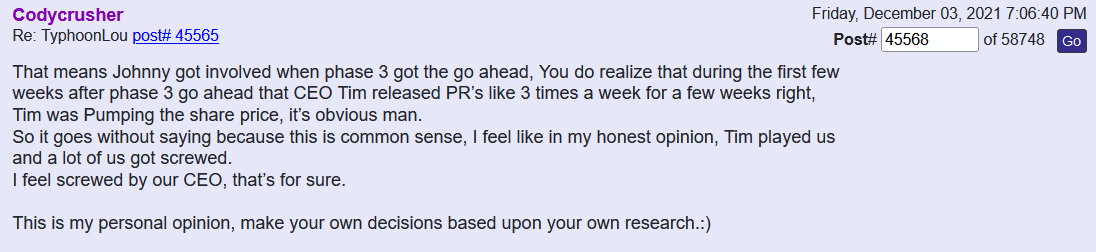
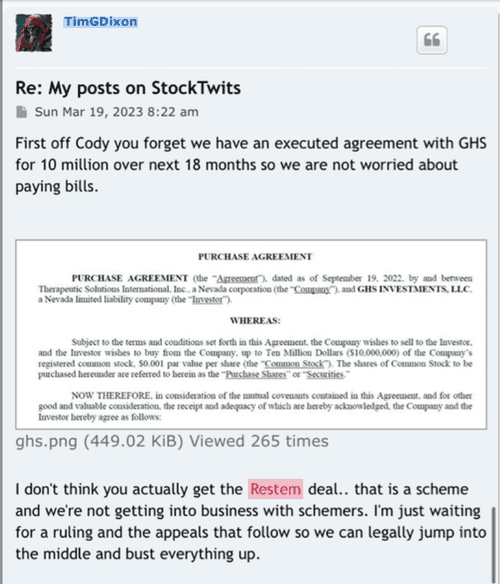
I was one of the ones who was very impatient here, not anymore. I needed to think about how someone would accomplish building a successful company that Tim and team/us/humanity wants/needs so badly. They have targeted a few organs that have minimal successful treatments. And to do so, you need to patent/claim every possible idea/process/angle to prevent parasites like China, Restem, Big Pharma leaches, etc. to swoop in and steal it. Yes, those PR's alone won't move the needle because each one by themselves is just protection and a small piece of the puzzle...but when they are all put together...bingo! The Golden Egg. And their products will be on all the shelves everywhere just in time for when the trial is successful. When people are finally treated after commercialization, they and everyone else in their lives (knowing that their loved one's life was just saved) will be buying like the products like crazy. Tim and team are building a mansion. The foundation needs to be laid first. Is the foundation worth more than the entire mansion? No. And this is just one out of a few mansions being built.
by TimGDixon » Thu Jun 02, 2022 12:13 pm
That is not true at all and I can't believe they said that. Ugh.
https://forum.therapeuticsolutionsint.com/viewtopic.php?p=3917#p3917
If that’s the case, what made TSOI RUN to .17 in February 2021? Tell us about the “trust” in Tim there? Then what’s the logical explanation why it sold off down to .03? This has been discussed numerous times…
Good thing this one isn’t a lie!!!
Post by TimGDixon » Wed May 25, 2022 7:01 pm
Now for sure I am happy you have all been able to load the boat as they say so I won't be too sad about that part in fact I am clapping. There are many ways to skin a cat without diluting everyone into oblivion or reversing them to a fraction. For me both scenarios leaves me with a stench I could never wash off this company or myself. So, if the powers to be think I am backed into a corner, they have thought wrong.
I’ll keep an open mind here. This may explode
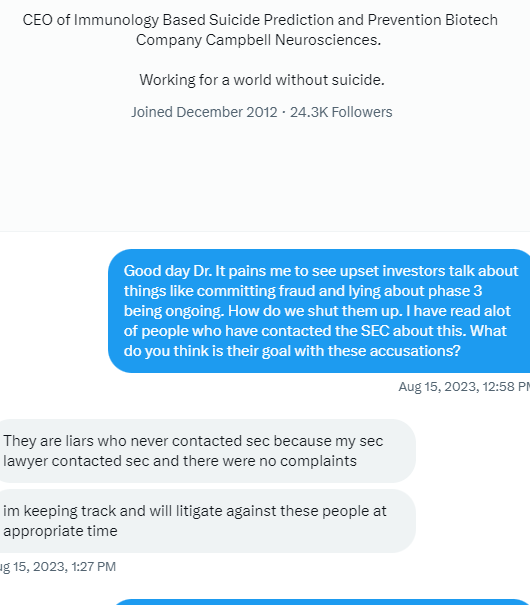
Facts people do not trust Tim Little Dixon which is why I believe this stock cannot gain any traction. I mean why trust a liar
No the EUA press release was a frickin lie. Next
Didn’t the August 16, 2021 Phase 3 and EUA press release get the the stock price moving backwards? Check out the TSOI price history! That had nothing to do with Tim or the last 5 press releases…
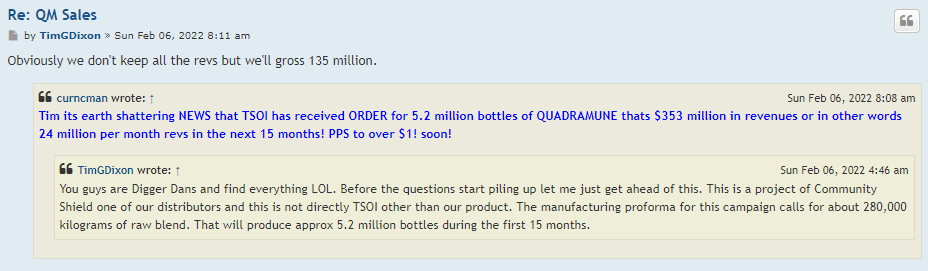
I like that Quadramune is starting to bring in sales revenues even before phase 3 results for Jadicell trials. I've been using QM for 6 months and will continue to do so. It's now available on Amazon.com, Walmart.com, and hopefully at 600 Costco stores in the near future. The stock price should be in the mid 30 cent range just on QM profits alone.
(These are my own projections and valuations; do your own DD)
Not in pennies. But ok
You do know a press release is just news it’s not always designed or intended to “move” the needle ….it’s just information …..
I think you're one of the few people that think this way. Most people can see the parh.
And I know what I own which right now is a pile of shit
Haven’t the past 5 press releases been great? Not one has gotten the stock moving. People don’t trust Tim Little Dixon. Can you blame them
I've been sitting here thinking how glad I am to have invested in this bio stock. I believe that we'll hear some really good news soon, and it will start to take off. If you know what you own, you'll agree. Go TSOI
Hope so brother. I need to fill up my gas tank soon:) -Pier.
Could go ballistic. Faster than the speed of light!
They update when the wind blows. Doesn’t move the stock in any way. We need hard verifiable news
$TSOI Looking forward new update hitting up here soon
|
Followers
|
528
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
64379
|
|
Created
|
10/04/08
|
Type
|
Free
|
| Moderators BigBadWolf johnnytrader33 JMC$ Yooperman Hogwarts | |||












































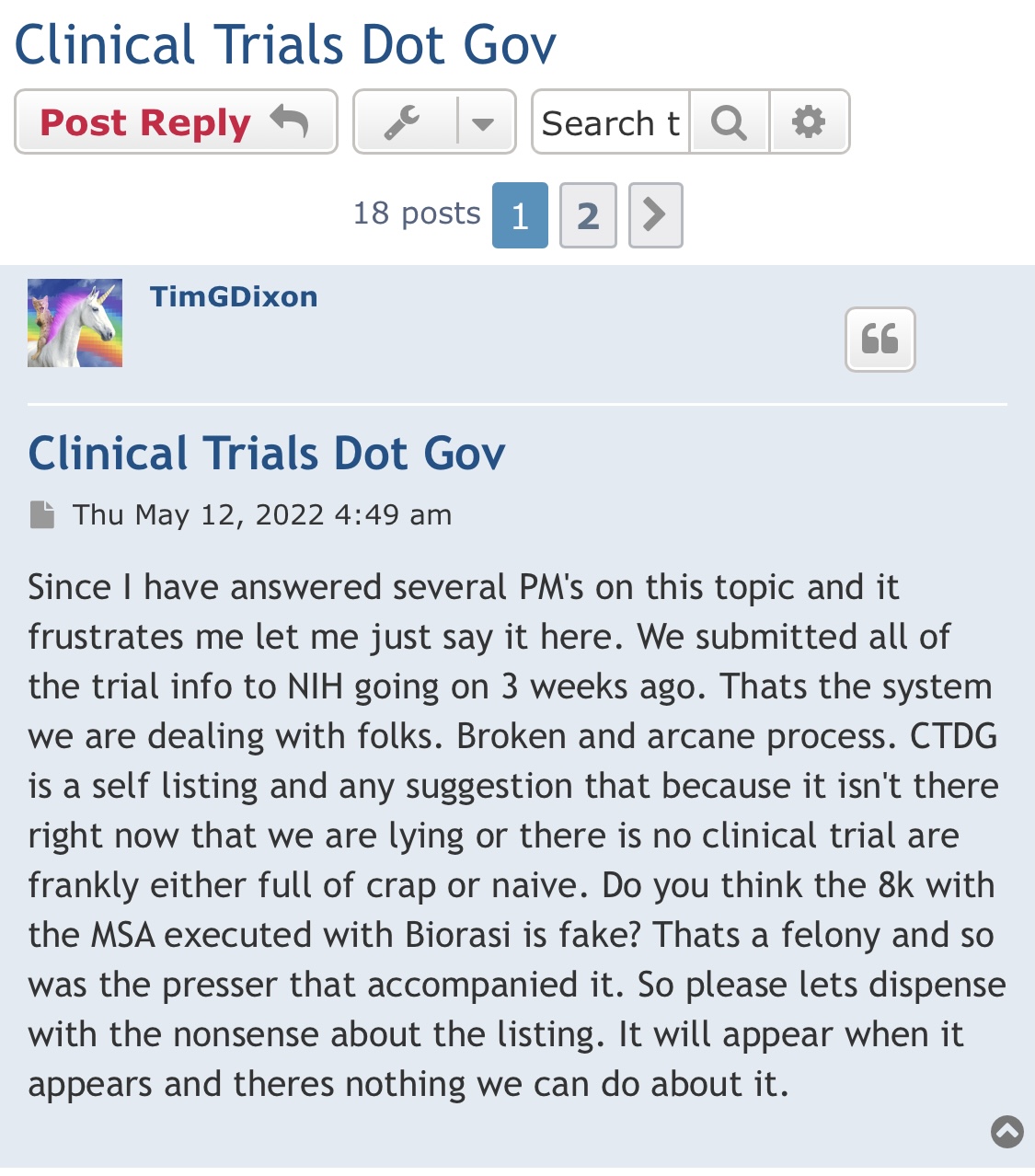

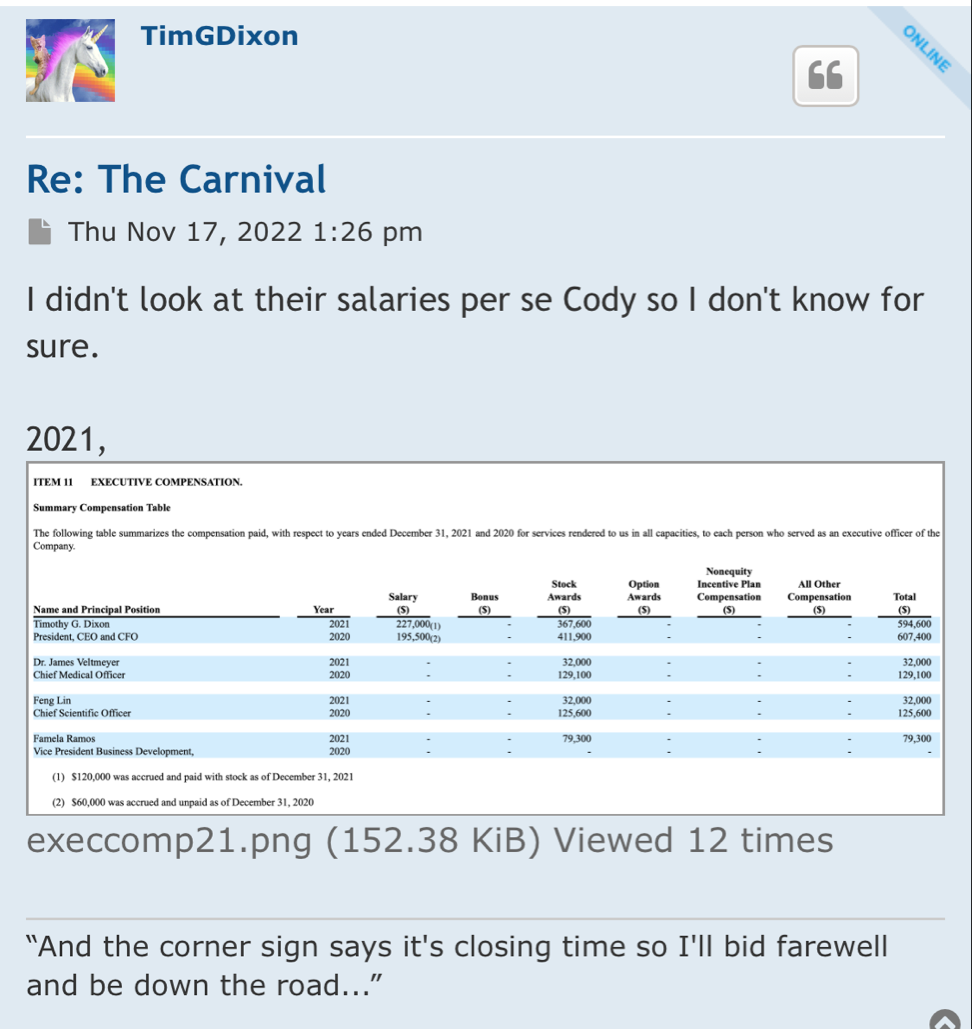










Preclinical Data Suggests QuadraMune™ Prevents Stress-Induced Suppression of Neurogenesis More Effectively than Prozac
OCEANSIDE, Calif., Dec. 9, 2020 /PRNewswire/ -- Therapeutics Solution International, Inc., (OTC Markets: TSOI), announced today new data suggesting the possibility that QuadraMune™ may mediate neuroprotective activity through preserving the ability of regenerative brain cells to proliferate subsequent to psychological stress.
The experiments, which involved exposing mice to established stressors, demonstrated that specific areas of the brain associated with production of new brain cells are damaged by stress. In agreement with previously published research, administration of fluoxetine (Prozac™) protected the brain from stress-induced damage. Surprisingly, QuadraMune™ administration appeared superior to Prozac™ at stimulating proliferation of new brain cells.
"QuadraMune™ which is currently in a clinical trial for prevention of COVD-191, has also been demonstrated to possess anti-inflammatory activity in other clinical trials, suppressing cytokines such as IL-62, which are known to be involved in depression3 and suicide4" said Kalina O'Connor, Director of Campbell Neurosciences and co-inventor on the patent. "Given major depressive disorder causes a significant risk for suicide, we are highly interested in exploring the use of QuadraMune™ for preventing suicide."
"Although much enthusiasm has been generated over the planned distribution of the COVID vaccine, at present little is being done to address mental health issues that are being exacerbated by the current pandemic" said Dr. James Veltmeyer, co-inventor of the patent, and Chief Medical Officer of the Company. "If current results are reproducible, the possibility that a nutraceutical would concurrently boost immunity while preserving mental health is highly enticing."
"It has not escaped us that COVID-19 is associated with increased inflammatory cytokines in the blood of patients, cytokines that also predispose to depression" said Famela Ramos, Vice President of Business Development for the Company. "It may be that the recent increase in suicides and suicide attempts is related biologically to activities of the coronavirus. It will be interesting to examine whether QuadraMune™ may modify putative negative mental effects of the virus."
"An estimated 17.3 million adults in the United States had at least one major depressive episode. This number represented 7.1% of all U.S. adults" stated Timothy Dixon, President and CEO of the Company. "We believe the Mission of our Company is not just providing a return on investment to our shareholders, but also increasing the quality of life for Americans. We are extremely pleased to report this unexpected finding with significant potential implications to advancing non-toxic means of helping patients with this terrible condition."
1 QuadraMune(TM) for Prevention of COVID-19 - Full Text View - ClinicalTrials.gov
2 Therapeutic Solutions International Announces Positive Preclinical and Clinical Evaluation of Nutritional Supplement QuadraMune™, Designed to Protect Against COVID-19 | BioSpace
3 Ting et al. Role of Interleukin-6 in Depressive Disorder. Int J Mol Sci. 2020 Mar 22;21(6):2194.
4 O'Donovan et al. Suicidal ideation is associated with elevated inflammation in patients with major depressive disorder. Depress Anxiety. 2013 Apr;30(4):307-14.
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
