Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
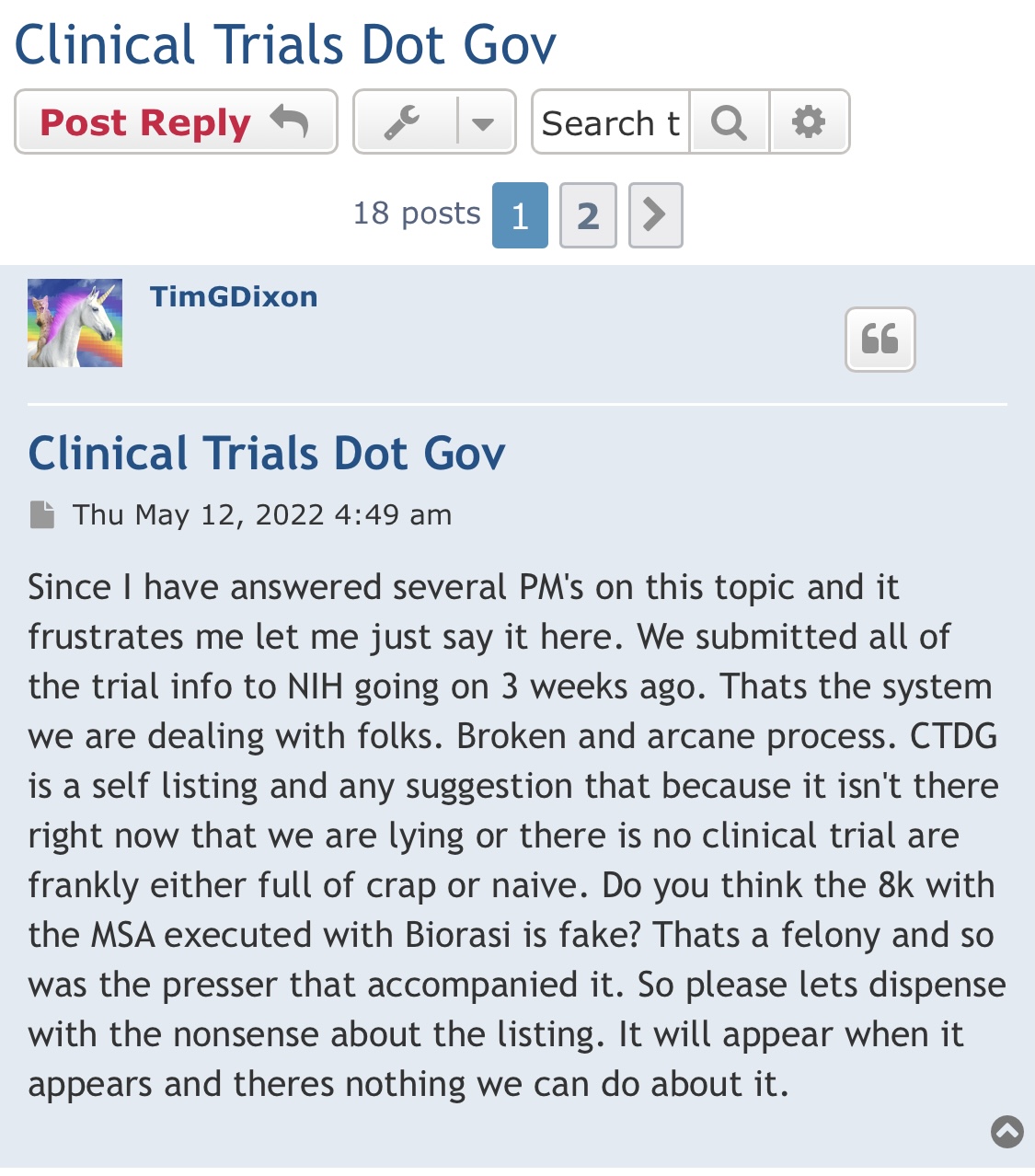
Dr. Phil Thorpe developed bavituximab many years ago for cellular targeting of cancer. He’s since died but bavituximab still being tested.
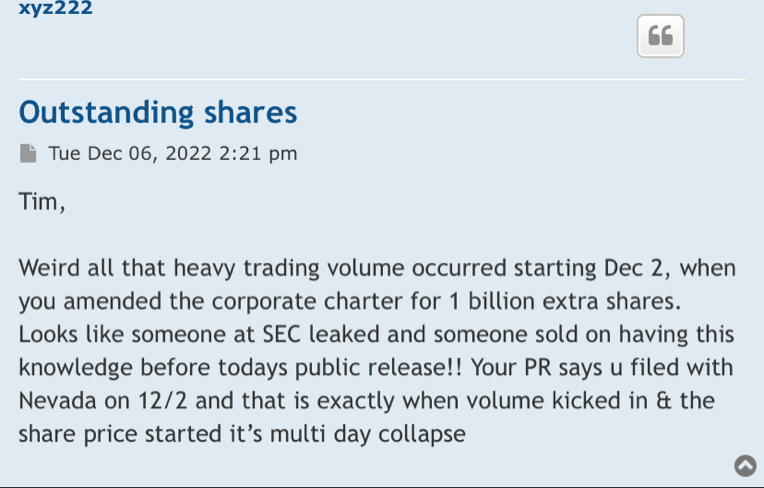
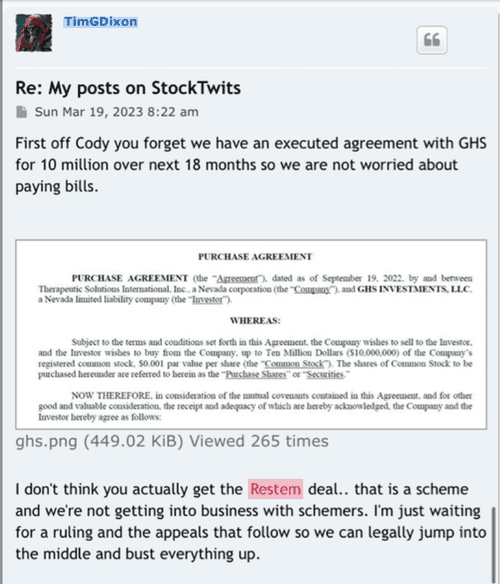
Dilution with a bow on it without any filings to prove it? Hmmmm!!!
It’s just. Dilution with a pretty bow
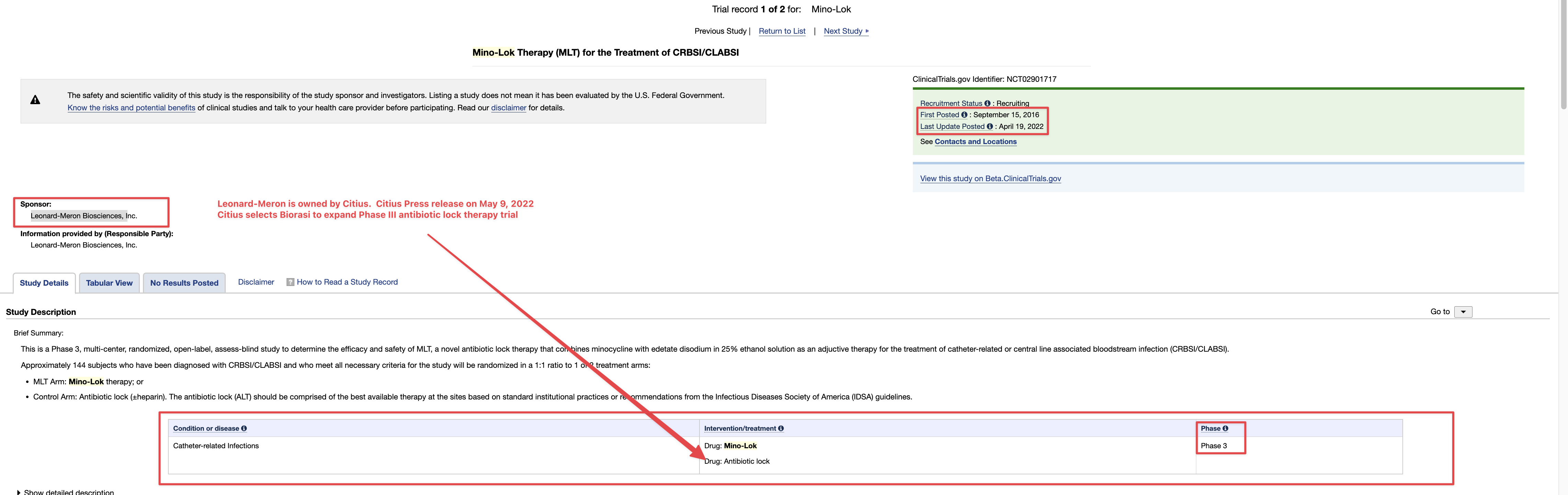
as per GHS Purchase Agreement "Freely" NO Use of Rule 144
GHS Purchased Shares Freely Tradable w/o restrictions on resale
b) Settlement for Purchase Shares
All Purchase Shares issued hereunder will be DWAC Shares.
DWAC Shares means shares of Common Stock that are (i) issued in electronic form. (ii)freely tradable and without restriction on resale and (iii) timely credited by the company to the Investor's or it's designee's specific Deposit Withdrawal at Custodian (DWAC) account with DTC under it's Fast Automated Securities Transfer (FAST) Program, or any similar program hereafter adopted by DTC performing substantially the same function.
(e) Commitment Shares means five million (5,000,000) common stock issued upon the initial closing as an equity incentive
(t)Purchase Price means with respect to a Purchase made pursuant to Section 2(a) hereof 80% of the lowest traded price of Common Stock during the Valuation Period.
(bb) Valuation Period means the ten (10)consecutive Business Days immediately preceding, but not including the Settlement date. [/color]
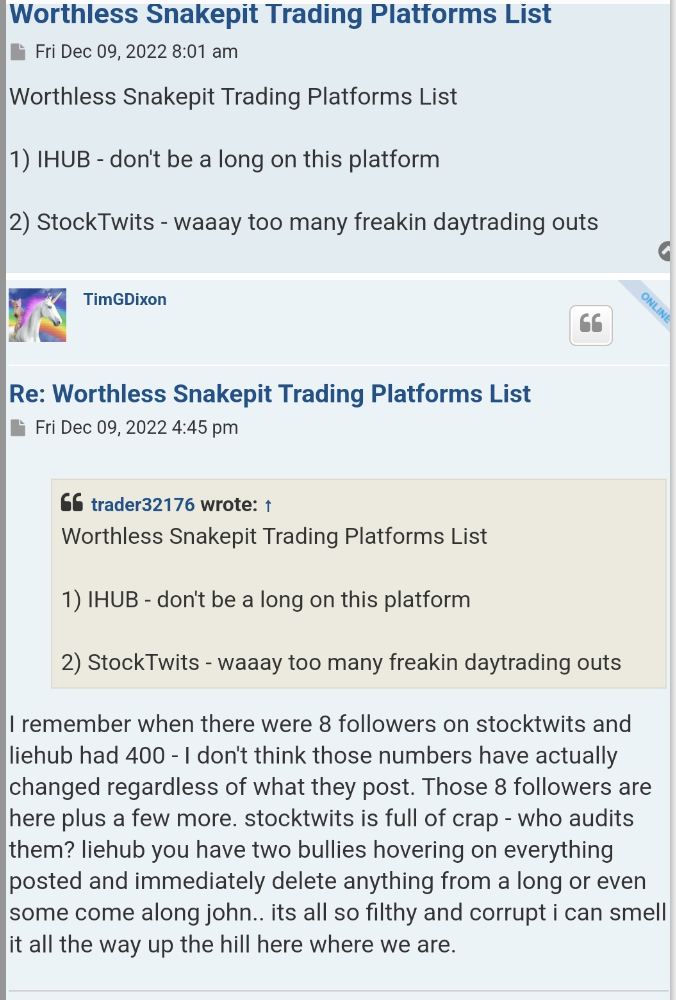
that doesn't really matter squat.
why would
dixon change ? Cmon , really ?
why wouldn't the data from Phase 3 change it ? :)
great stuff Curncman
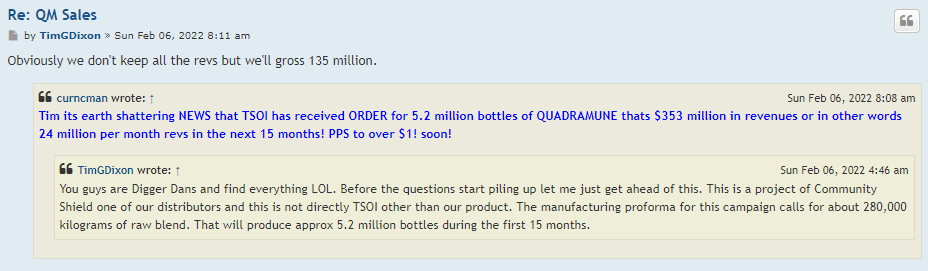
GHS Inc would not be able to buy 10 million worth of shares in the open market without PPS shooting up to 15 cents instead they would be happy to get them at PPS between 5 to 10 cents with direct allotment from the company.
a real discussion of this stock :)
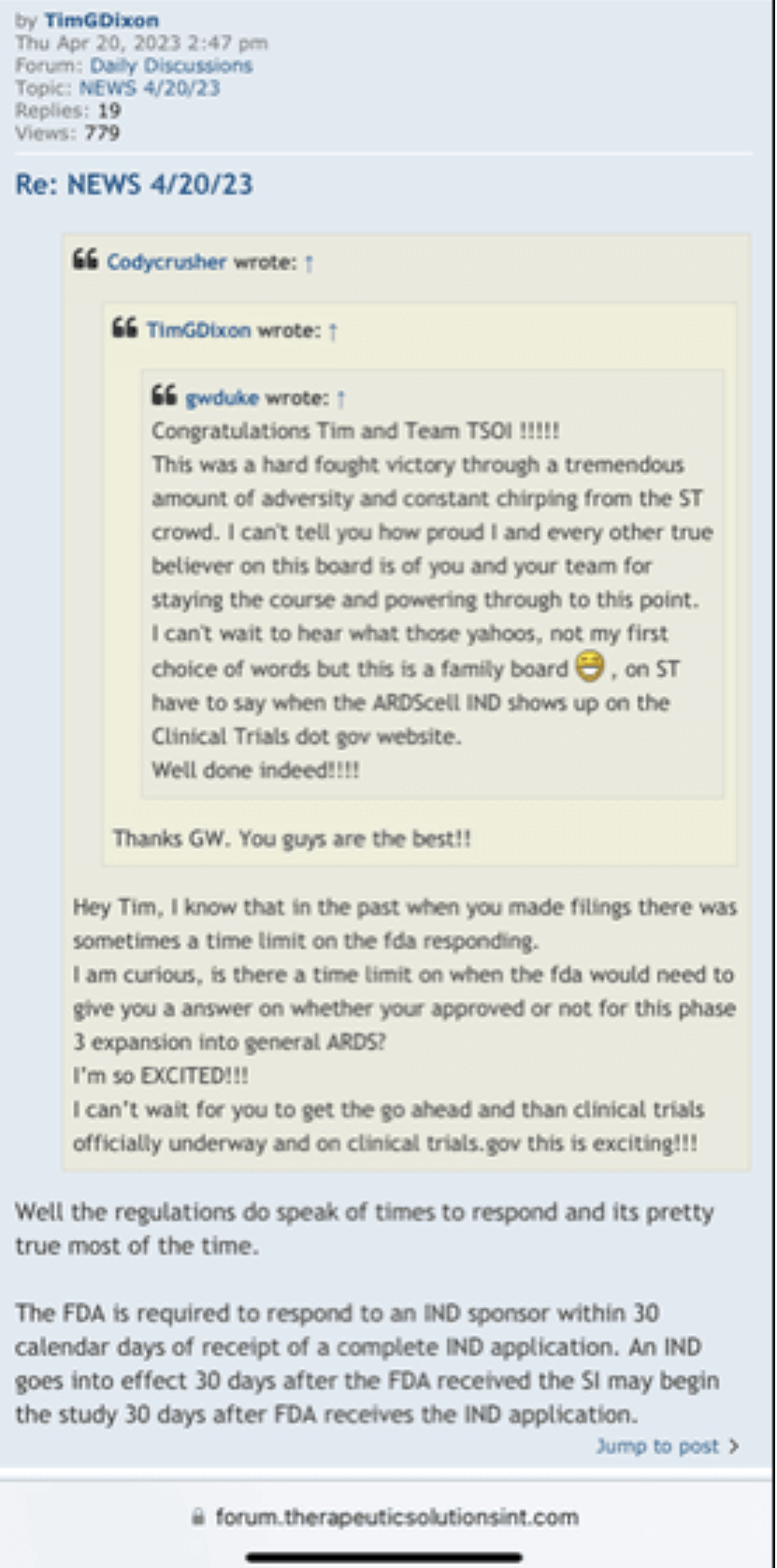
and tim's reply was:
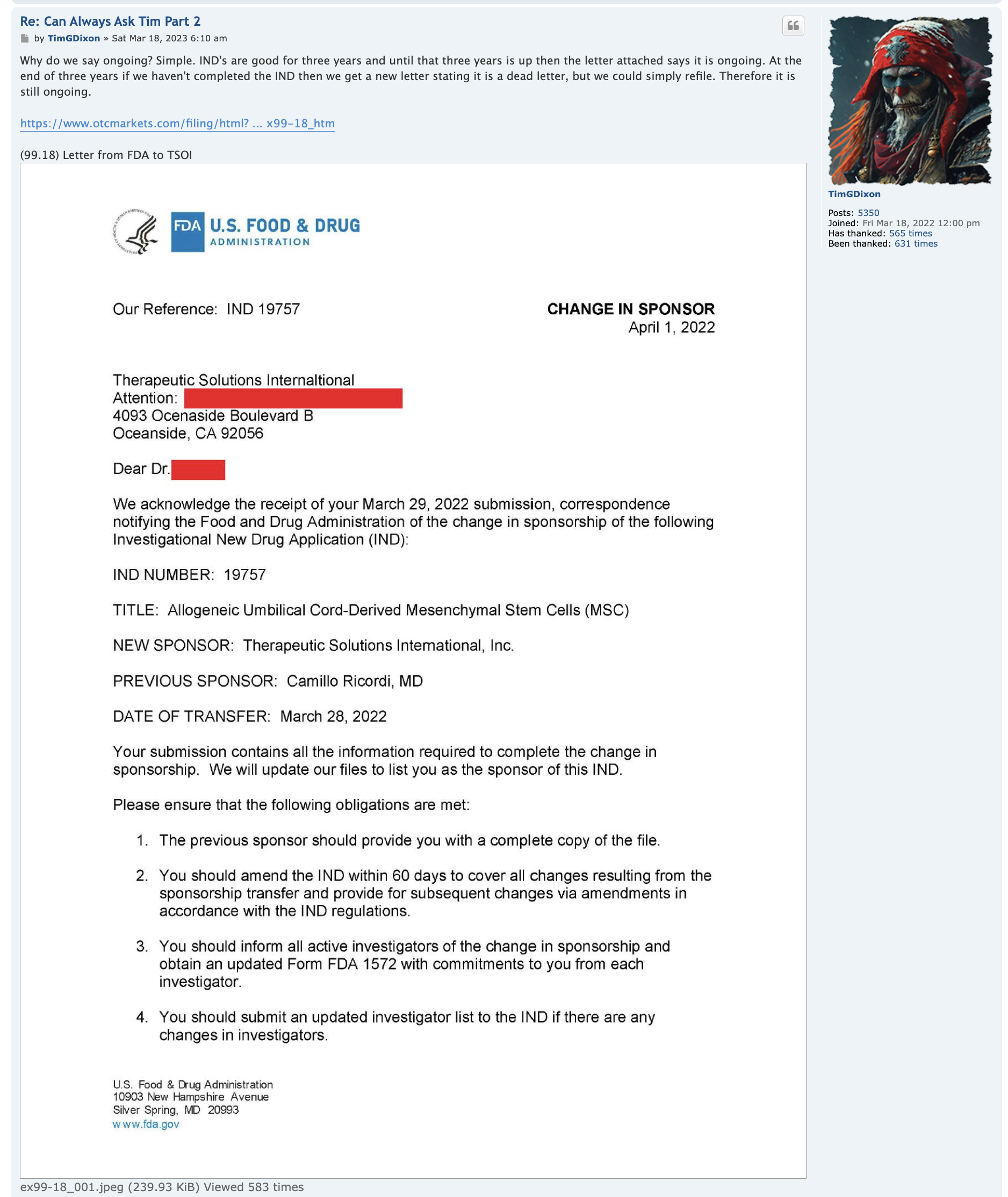
EXHIBIT A
FORM OF PURCHASE NOTICE
________, 202__
To: GHS Investments, LLC
In accordance with Section 2 of the purchase agreement, dated September [ ], 2022 (the “Purchase Agreement”),
between Therapeutic Solutions International, Inc. (the “Company”) and GHS Investments, LLC (the “Investor”), the
Company hereby provides notice to the Investor of a sale by the Company to the Investor of Purchase Shares in the
amount set forth in this Purchase Notice. Capitalized terms used herein have the meanings set forth in the Purchase
Agreement.
Purchase Amount: $___________
Purchase Price per share: $____________
Number of Purchase Shares: __________
Very truly yours,
Therapeutic Solutions International, Inc.
By:_________________________
Name:
Title:
Trading shares Re: GHS :
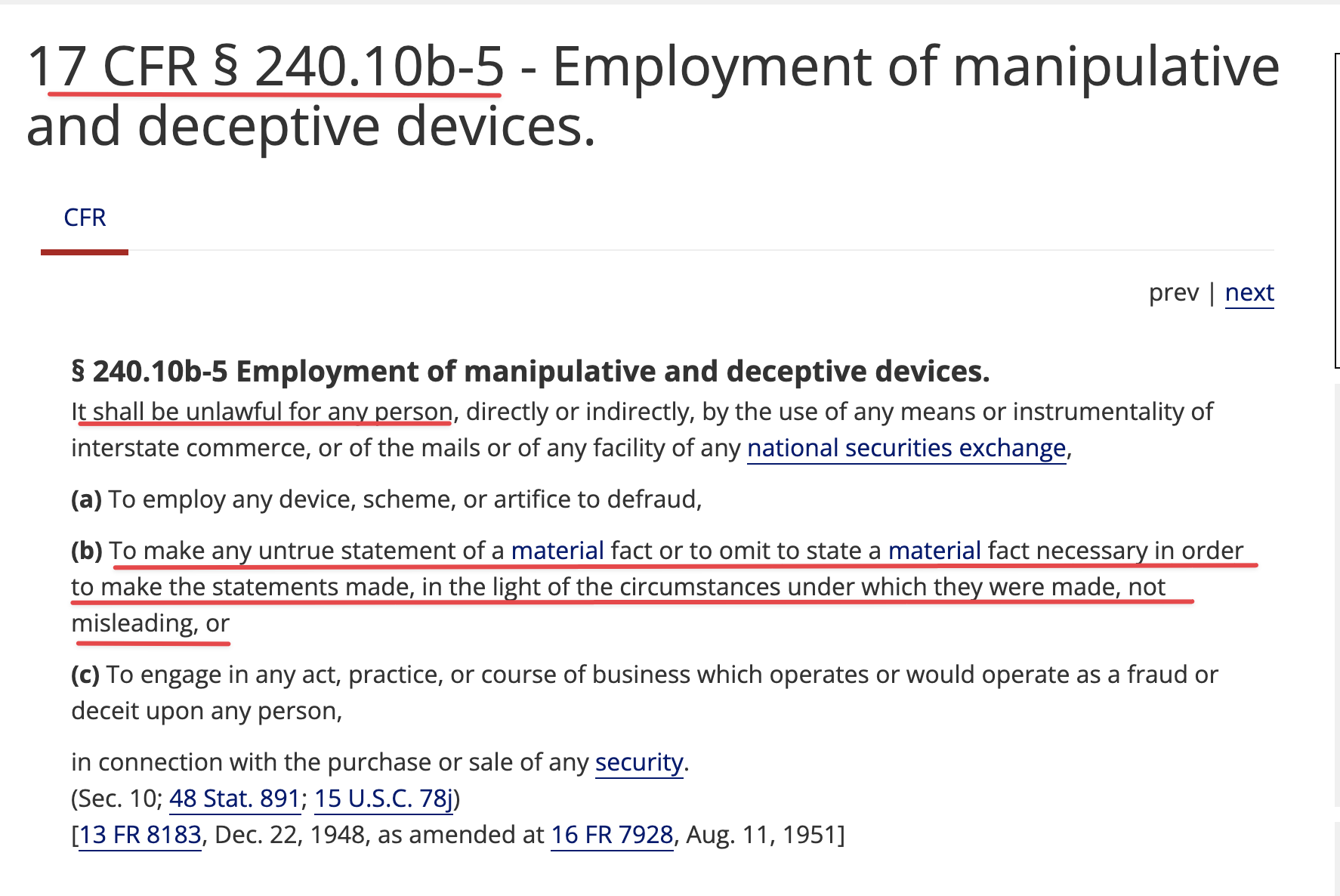
This is a shelf registration that uses Form S-3. Under this shelf registration we may offer and sell, either individually or in combination, in one or more offerings, any of the securities described in the prospectus, for total gross proceeds of up to $10,000,000.
GHS Share Purchases are FREE TRADING (No restrictions) w/ 20% discount to the lowest closing bid price (posted) over the preceding 10 consecutive day evaluation period
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=170075326
Good to see you here as always Rollo
KWYOAOI !
Choking Off Cancer
https://www.technologyreview.com/2002/07/01/234815/choking-off-cancer/
Today’s cancer drugs are notorious for killing healthy cells along with cancerous ones. A new anticancer approach could offer a more precise option: kill just the tumor by choking off its blood supplies. The first drugs based on this approach are now in human trials and, if they work, could provide a virtually side-effect-free means of fighting a host of cancers.
Called vascular targeting agents or antivascular therapies, the new drugs block the blood vessels that carry oxygen and nutrients to tumors. “Basically, it starves the tumor cells to death, so you get a massive amount of tumor-cell kill,” says Dai Chaplin, chief scientific officer at Watertown, MA-based Oxigene. This is in contrast to conventional chemotherapies, which kill cancer cells directly, and to another experimental approach called “anti-angiogenesis,” in which drugs stop new tumor blood vessels from growing.
Oxigene, Aventis and AstraZeneca are each testing vascular targeting agents in humans. These drugs bind to a protein called tubulin, preferentially targeting the tubulin in the cells that line the interior of tumor blood vessels. Long chains of the protein normally form an internal scaffold that helps keep these cells flat. The binding disrupts the scaffolding, and “the cells become fat and podgy and obstruct the blood flow through the tumor,” says vascular-targeting pioneer Phil Thorpe, a professor of pharmacology at the University of Texas Southwestern Medical Center. This effect can be seen within hours after injection of the drug.
So far, researchers have seen very few side effects in human tests, though a number of patients have felt pain in their tumors. And binding to tubulin is short-lived-on the order of hours-long enough to halt tumor blood flow but not long enough to kill normal cells, Thorpe says. “These drugs seem inherently safer than the blanket anticancer drugs that are currently available,” he notes.
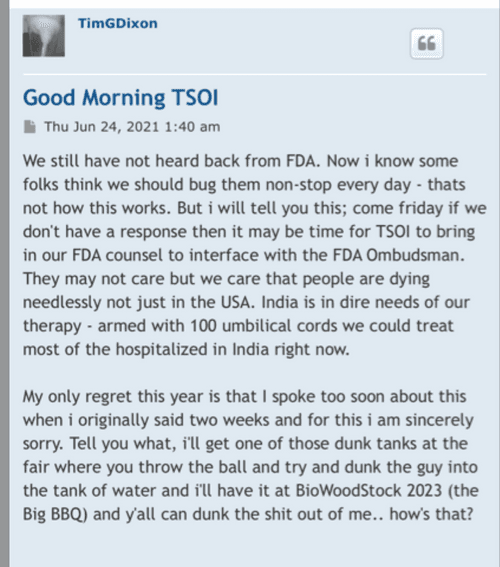
TSOI - Entering that sweet spot.
Fortune favors the bold.
TSOI
Will he? Well that investigation should only take between 2-4 years! Stick around and see…. Tick Tock…
Once again excellent DD Cents!!!!
GO TSOI!!!!
KWYOAOI!!!!
Cheers,
—Rollo
Good try. He will have to answer to the SEC. tick rock
TSOI New Patent published 9/29/22
https://patents.justia.com/patent/20220306994
CHIMERIC CELLS COMPRISING DENDRITIC CELLS AND ENDOTHELIAL CELLS RESEMBLING TUMOR ENDOTHELIUM
Disclosed are means, methods and compositions of matter useful for induction of immunological responses towards tumor endothelial cells. In one embodiment the invention teaches fusion of dendritic cells and cells resembling tumor endothelial cells and administration of such chimeric cells as an immunotherapy for stimulation of tumor endothelial cell destruction. In other embodiments pluripotent stem cells are utilized to generate dendritic cells, wherein said dendritic cells are fused with pluripotent stem cell derived endothelial cells created in a manner to resemble tumor endothelial cells.
1. A hybrid cell comprising of: a) a dendritic or dendritic like cell and b) an endothelial cell generated in a manner to resemble tumor endothelium.
2. The hybrid cell of claim 1, wherein said hybrid cells is generated by fusion of a dendritic or dendritic like cell and an endothelial cell generated in a manner to resemble tumor endothelium.
3. The hybrid cell of claim 3, wherein said fusion is created by placement of both cell types in physical proximity while treating both cells with an agent capable of causing fusion of plasma membrane.
4. The hybrid cell of claim 3, wherein said fusion agent is one or more agents selected from a group comprising of: a) polyethylene glycol; b) ultrasound waves; c) radio waves; and d) phosphatidylcholine.
5. The hybrid cell of claim 1, wherein said dendritic cell is generated from a group of cells comprising of: a) a stem cell; b) a pluripotent stem cell; c) an inducible pluripotent stem cell; d) a parthenogenic stem cell; e) a somatic cell nuclear transfer derived stem cell; f) a pluripotent stem cell is generated by cytoplasmic transfer from an immature cell to a mature cell; and g) embryoid bodies from said pluripotent stem cells.
6. The hybrid cell of claim 5, wherein said embryoid bodies are dissociated and cells are cultured in cytokines capable of expanding dendritic cell progenitors.
7. The hybrid cell of claim 5, wherein said dendritic cell progenitors are cultured in GM-CSF.
8. The hybrid cell of claim 5, wherein said dendritic cell progenitors are cultured in flt-3 ligand.
9. The hybrid cell of claim 5, wherein said dendritic cell progenitors are cultured in IL-4.
10. The hybrid cell of claim 1, wherein said endothelial cells are derived from a pluripotent stem cell.
11. The hybrid cell of claim 10, wherein said pluripotent stem cell is an inducible pluripotent stem cell.
12. The hybrid cell of claim 10, wherein said pluripotent stem cell is a parthenogenic stem cell.
13. The hybrid cell of claim 10, wherein said pluripotent stem cell is a somatic cell nuclear transfer derived stem cell.
14. The hybrid cell of claim 10, wherein said pluripotent stem cell is generated by cytoplasmic transfer from an immature cell to a mature cell.
15. The hybrid cell of claim 1, wherein said hybrid cell is utilized to induce an immune response against tumor endothelial cells.
16. The hybrid cell of claim 10, wherein said endothelial cells are generated by culture of endothelial progenitor cells in a media replicating the tumor microenvironment.
17. The hybrid cell of claim 10, wherein said endothelial cells are generated by culture of endothelial progenitor cells in a media replicating the tumor microenvironment.
18. The hybrid cell of claim 17, wherein said media contains one or more agents selected from a group comprising of: a) prostaglandin E2; b) TGF-beta; c) IL-10; d) VEGF; e) PDGF-BB; f) EGF; g) FGF-1 and h) FGF-2.
19. The hybrid cells of claim 1, wherein antigen presenting activity of said cells is augmented as compared to baseline conditions by treatment with a toll like receptor agonist.
20. The hybrid cells of claim 19, wherein said toll like receptor agonist is HMGB-1 or a peptide derived thereof.
[0001] This application claims priority to U.S. Provisional Application Ser. No. 63/165,056, filed on Mar. 23, 2021, entitled "Chimeric Cells Comprising Dendritic Cells and Endothelial Cells Resembling Tumor Endothelium", which is incorporated herein by reference in its entirety.
FIELD OF THE INVENTION
[0002] The field relates to compositions of matter useful for induction of immunological responses towards tumor endothelial cells, including the fusion of dendritic cells and cells resembling tumor endothelial cells.
BACKGROUND OF THE INVENTION
[0003] The concept of treating cancer by blocking new blood vessel formation, angiogenesis, was pioneered by Judah Folkman who provided convincing arguments that it is not necessary to actively kill the tumor mass, but by suppressing its ability to grow through cutting off blood supply, malignant tumors may be converted into benign masses that eventually regress [1, 2]. Unfortunately, despite discovery of angiostatin, and endostatin, naturally derived inhibitors of angiogenesis, neither of these approaches translated into successful therapies. Nevertheless, the concept of targeting new blood vessel formation led to thousands of publications describing various antiangiogenic agents, of which several eventually proceeded through clinical trials and regulatory approval. Broadly anti-angiogenic agents approved by regulators can be classified into antibodies, such as Bevacizumab (Avastin) which binds VEGF [3], and Ramucirumab (Cyramza) [4], which binds VEGF-R2, as well as small molecules which bind multiple receptor kinases associated with angiogenesis such as Sunitinib [5-7], Cabozantinib [8-11], Pazopanib [12-14], and Regorafenib [15-17].
[0004] These approaches have augmented the standard of care for various tumor types and have achieved some level of progress. Unfortunately, the concept of blocking angiogenesis of cancer was not as simple as originally envisioned. One of the major hurdles in blocking angiogenesis was that even though de novo blood vessels are derived from nonmalignant cells, the malignant cells appear to possess ability to induce mutations in the new blood vessels. One example of the heterogeneity of tumor endothelial cells compared to endothelial cells from low and high metastatic tumors by Ohga et al [18]. The investigators extracted two types of tumor endothelial cells (TEM) from high-metastatic (HM) and low-metastatic (LM) tumors and compared their characteristics. HM tumor-derived TECs (HM-TECs) showed higher proliferative activity and invasive activity than LM tumor-derived TECs (LM-TECs). Moreover, the mRNA expression levels of pro-angiogenic genes, such as vascular endothelial growth factor (VEGF) receptors 1 and 2, VEGF, and hypoxia-inducible factor-1a, were higher in HM-TECs than in LM-TECs. The tumor blood vessels themselves and the surrounding area in HM tumors were exposed to hypoxia. Furthermore, HM-TECs showed higher mRNA expression levels of the stemness-related gene stem cell antigen and the mesenchymal marker CD90 compared with LM-TECs. HM-TECs were spheroid, with a smoother surface and higher circularity in the stem cell spheroid assay. HM-TECs differentiated into osteogenic cells, expressing activated alkaline phosphatase in an osteogenic medium at a higher rate than either LM-TECs or normal ECs. Furthermore, HM-TECs contained more aneuploid cells than LM-TECs. The investigators concluded that the results indicate that TECs from HM tumors have a more pro-angiogenic phenotype than those from LM tumors. It appears that the aggressiveness of the tumor not only can alter endothelial cell function but also drug resistance ability. In another study, Akiyama et al. [19]compared murine TECs and normal ECs. It was found that TECs were more resistant to paclitaxel with the up-regulation of multidrug resistance (MDR) 1 mRNA, which encodes the P-glycoprotein, compared with normal ECs. Normal human microvascular ECs were cultured in tumor-conditioned medium (CM) and became more resistant to paclitaxel through MDR1 mRNA up-regulation and nuclear translocation of Y-box-binding protein 1, which is an MDR1 transcription factor. Vascular endothelial growth factor (VEGF) receptor 2 (VEGFR2) and Akt were activated in human microvascular ECs by tumor CM. The investigators observed that tumor CM contained a significantly high level of VEGF. A VEGFR kinase inhibitor, Ki8751, and a phosphatidylinositol 3-kinase-Akt inhibitor, LY294002, blocked tumor CM-induced MDR1 up-regulation. MDR1 up-regulation, via the VEGF-VEGFR pathway in the tumor microenvironment, is one of the mechanisms of drug resistance acquired by TECs. It was observed that VEGF secreted from tumors up-regulated MDR1 through the activation of VEGFR2 and Akt. This process is a novel mechanism of the acquisition of drug resistance by TECs in the tumor microenvironment. Yet another study demonstrated that tumors can induce a "dedifferentiation" of tumor endothelium. Specifically, compared with NECs, stem cell markers such as Sca-1, CD90, and multidrug resistance 1 are upregulated in TECs, suggesting that stem-like cells exist in tumor blood vessels. TECs and NECs were isolated from melanoma-xenografted nude mice and normal dermis, respectively. The stem cell marker aldehyde dehydrogenase (ALDH) mRNA expression and activity were higher in TECs than those in NECs. Next, ALDHhigh/low TECs were isolated by fluorescence-activated cell sorting to compare their characteristics. Compared with ALDHlow TECs, ALDHhigh TECs formed more tubes on Matrigel-coated plates and sustained the tubular networks longer. Furthermore, VEGFR2 expression was higher in ALDHhigh TECs than that in ALDHlow TECs. In addition, ALDH was expressed in the tumor blood vessels of in vivo mouse models of melanoma and oral carcinoma, but not in normal blood vessels. These findings indicate that ALDHhigh TECs exhibit an angiogenic phenotype. Stem-like TECs may have an essential role in tumor angiogenesis [20].
[0005] What is it that causes the tumor to evoke changes in the endothelium? As suggested above, there is some support for growth factor mediated alterations, additionally, horizontal gene transfer may also play a role [21-29]. Although the field of horizontal gene transfer has historically been controversial one of the strongest evidences supporting this concept is the phenomena of donor-derived relapse in leukemic patients. In these situations patients with leukemia who relapse after bone marrow transplant have the relapsing cells originate from donor cells that transformed into malignant cells [30, 31]. Another issue that affected efficacy of anti-angiogenesis therapies is that in some tumors, the tumor cells themselves transdifferentiate into endothelial-like cells, termed tumor vascular channels, which possess ability to mutate around either antibody or kinase inhibitor drugs [32-37].
[0006] The previously mentioned means by which tumor endothelial cells can protect themselves against anti-angiogenic agents has resulted in relatively low clinical efficacy of these drugs. To understand the general lack of efficacy in the initial registration trial.sup.ii, median progression free survival (PFS) of ovarian cancer patients who received bevacizumab plus chemotherapy was 6.8 months (95 percent CI: 5.6, 7.8) compared with 3.4 months (95 percent CI: 2.1, 3.8) for those who received chemotherapy alone. There was no statistically significant difference in overall survival (OS) for patients treated with bevacizumab plus chemotherapy compared with chemotherapy alone (median OS: 16.6 months versus 13.3 months; HR 0.89; 95 percent CI: 0.69, 1.14). Subset analysis led to identification that the group of patients that received paclitaxel with the antibody had the largest improvement, resulting in a 5.7-month improvement in median PFS (9.6 months versus 3.9 months; HR 0.47; 95 percent CI: 0.31, 0.72), an improvement in the objective response rate (53 percent versus 30 percent), and a 9.2-month improvement in median OS (22.4 months versus 13.2 months, HR 0.64; 95 percent CI: 0.41, 1.01).sup.iii. Multiple other trials where conducted for different indications using bevacizumab, unfortunately, progression free survival and overall survival was not increase more than a year in any of the studies [38-42], and neither in studies with small molecule kinase inhibitors [43-48].
[0007] This clinical translation, although highly beneficial in some patients, overall the effect was mediocre, highlights the disparity between animal studies, in which some studies complete regression was observed in established tumors [49, 50], whereas in clinical trials, relatively minimal effect compared to animal studies was observed [51]. One lesson from these studies is that the large heterogeneity of the patient and of the tumors, which calls for large patient populations in order to achieve an overall survival advantage.
[0008] Innovations in pharmacogenomics and personalized medicine will help identify specific patients and tumors that are likely to respond. Unfortunately, at present, patients with metastatic disease have limited options and a statistically significant extension of survival does equate to large market demand, as seen by the overall sale of angiogenesis inhibitors for cancer being over 20 billion annually.
From the PR announcing the filing of this patent on 3/23/21 :
Therapeutic Solutions International Creates Hybrid Cell Designed to Educate Immune System to Choke Cancer Blood Vessels
Novel Strategy Demonstrates Potent Results in Animal Model of Lung Cancer through Fusing StemVacs-V™ Immunotherapy with In-Vitro Generated Cancer Blood Vessels
https://www.prnewswire.com/news-releases/therapeutic-solutions-international-creates-hybrid-cell-designed-to-educate-immune-system-to-choke-cancer-blood-vessels-301253715.html
ELK CITY, Idaho, March 23, 2021 /PRNewswire/ — Therapeutic Solutions International, Inc., (OTC Markets: TSOI), announced today filing of a patent with new data on a unique cell created by the Company capable of training the immune system to kill blood vessels feeding cancer, but sparing healthy blood vessels. These data are an extension of previous findings from the Company showing that StemVacs is capable of suppressing new blood vessel production.
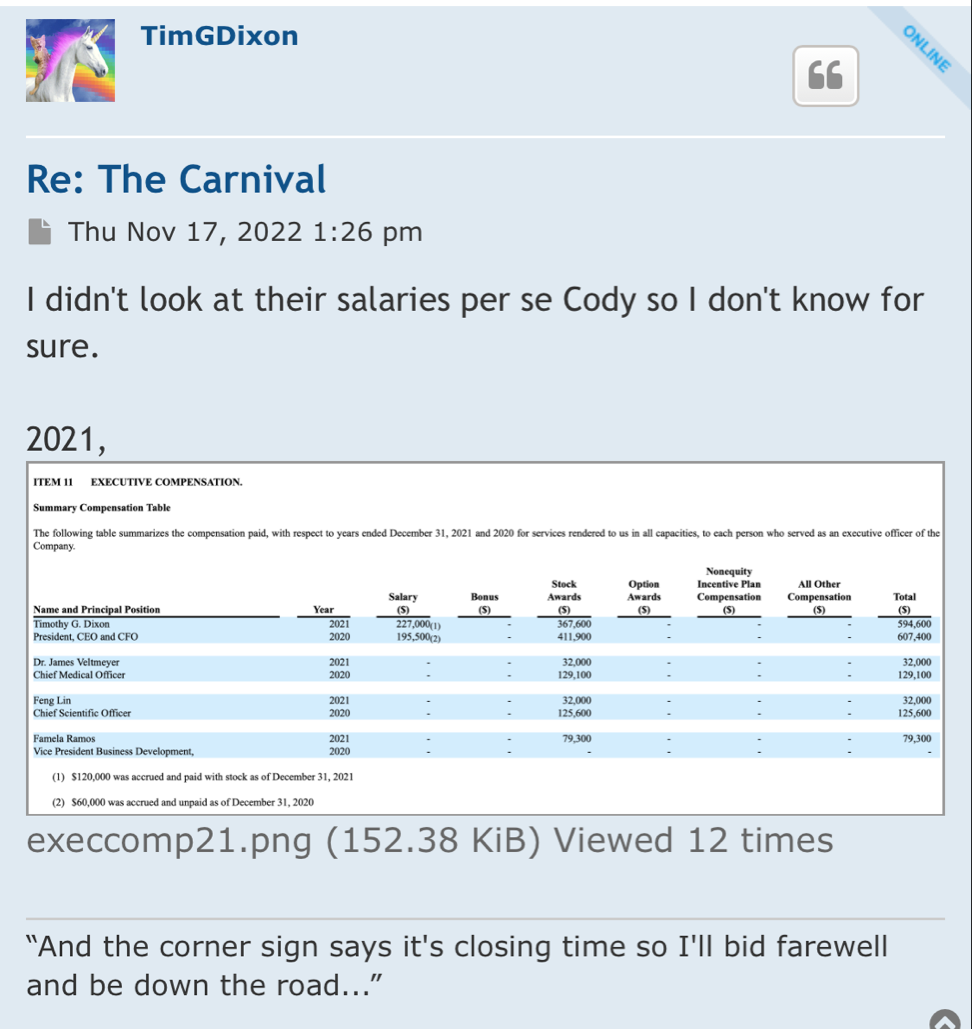
“Killing malignant blood vessels is the basis for Avastin, an antibody-based drug which created 7 billion in revenue last year.1 Another company, Batu Biologics, previously demonstrated vaccination against cancer blood vessels is safe and was cleared by the FDA for clinical trials,” stated Dr. James Veltmeyer, Chief Medical Officer of the Company. “The approach developed by our Company is completely unique because it merges the potent antigen presenting capabilities of StemVacs-V together with the reproducibility and consistency of iPSC generated tumor-like endothelial cells.”
StemVacs-V™ is comprised of “cancer resistant” dendritic cells which are developed from an engineered iPSC cell line. The novel chimeric cell product is generated by fusing StemVacs-V with iPSC derived endothelial cells which are cultured in a manner to make them replicate cancer stem cells. Proof of selective killing of cancer blood vessels was obtained by demonstration of immune response to proteins found only on cancer blood vessels.
“Numerous companies such as Fate Therapeutics, NantKwest, and Century Therapeutics have all achieved significant valuations through pioneering novel uses of iPSC derived immune cells,” said Famela Ramos, Vice President of Business Development. “To our knowledge we are the first company to use iPSC derived cells to stimulate naturally occurring immunity to kill what some call the ‘Achilles Heel’ of cancer.”
“Generation of a new cell that didn’t previously exist is a significant milestone for our Company. We believe the current animal studies in lung cancer can be extrapolated to other cancers, since no cancer can grow more than 2 millimeters without creating new blood vessels,” said Timothy Dixon, President and CEO of the Company and co-inventor of the patent. “By selectively killing cancer blood vessels the problem of mutations is solved since cancer blood vessels are not generated from tumor tissue and they do not mutate. Furthermore, since blood vessels are in direct contact with the blood and immune cells that travel through the blood, it is substantially easier for the immune system to kill malignant vessels as opposed to tumors.”
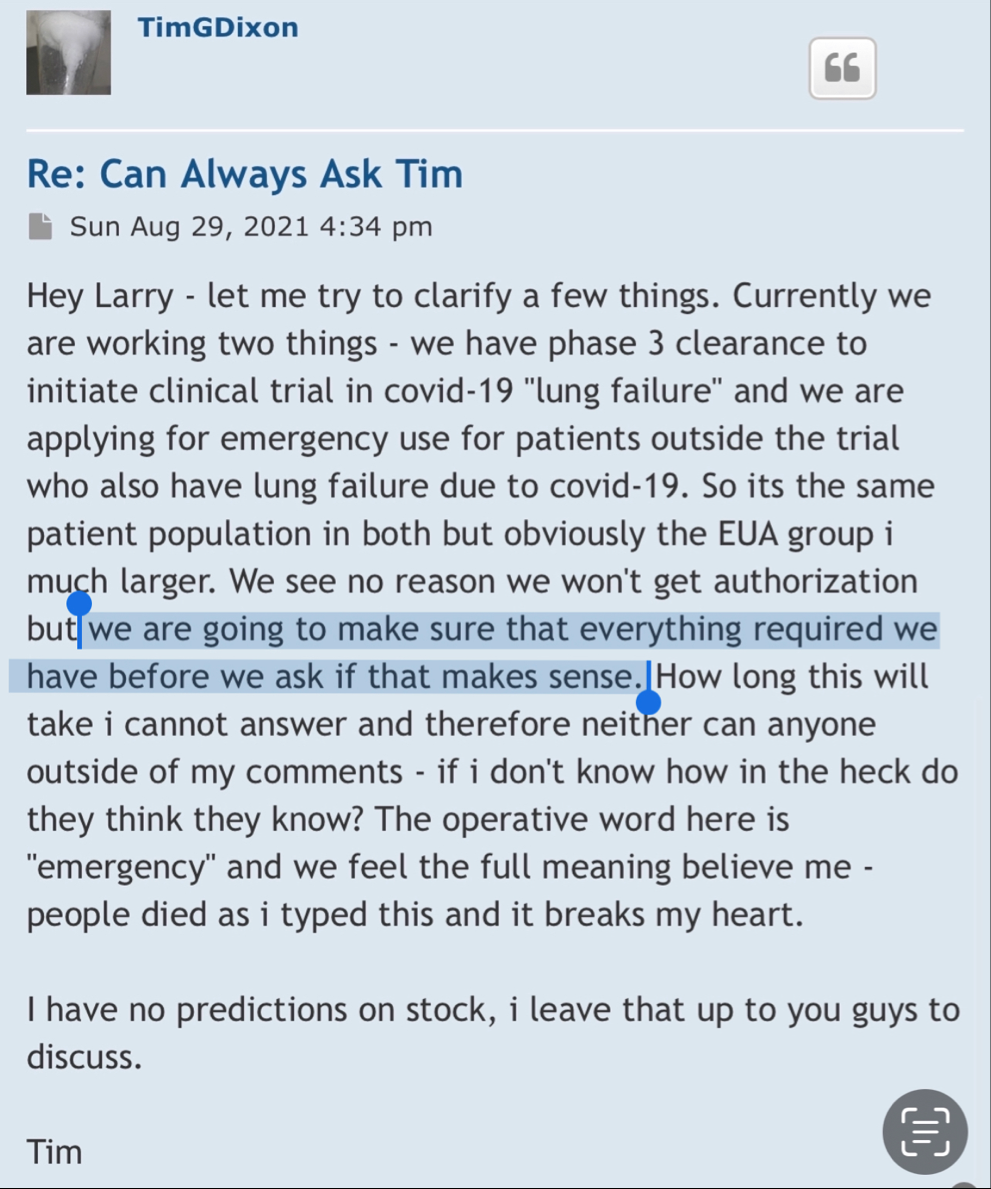
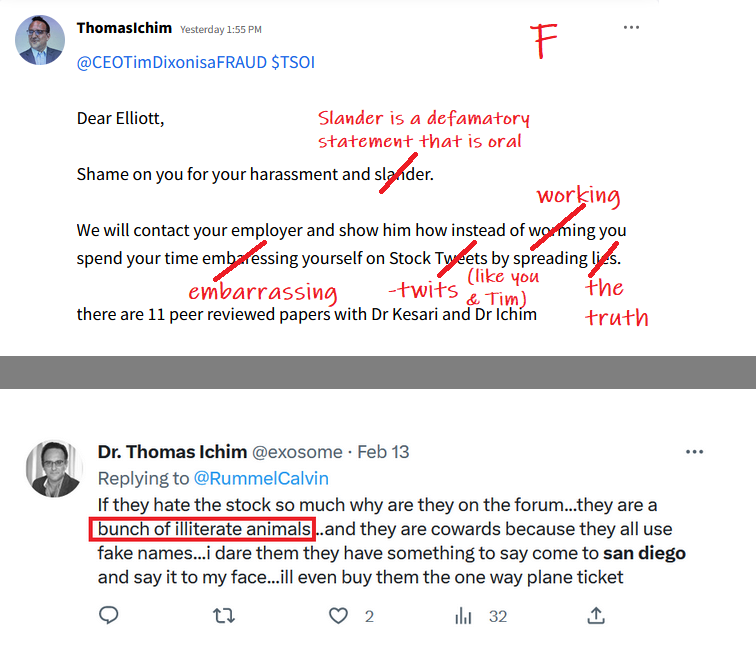
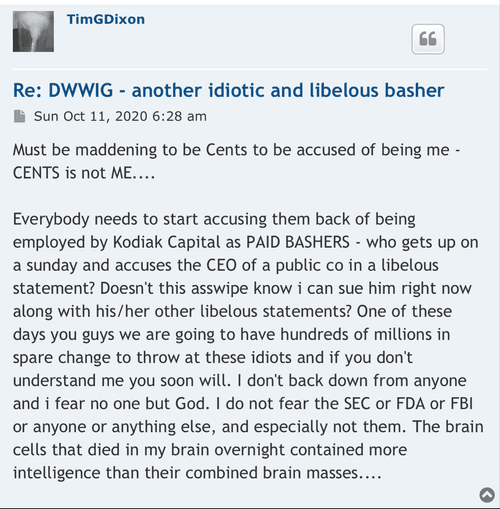
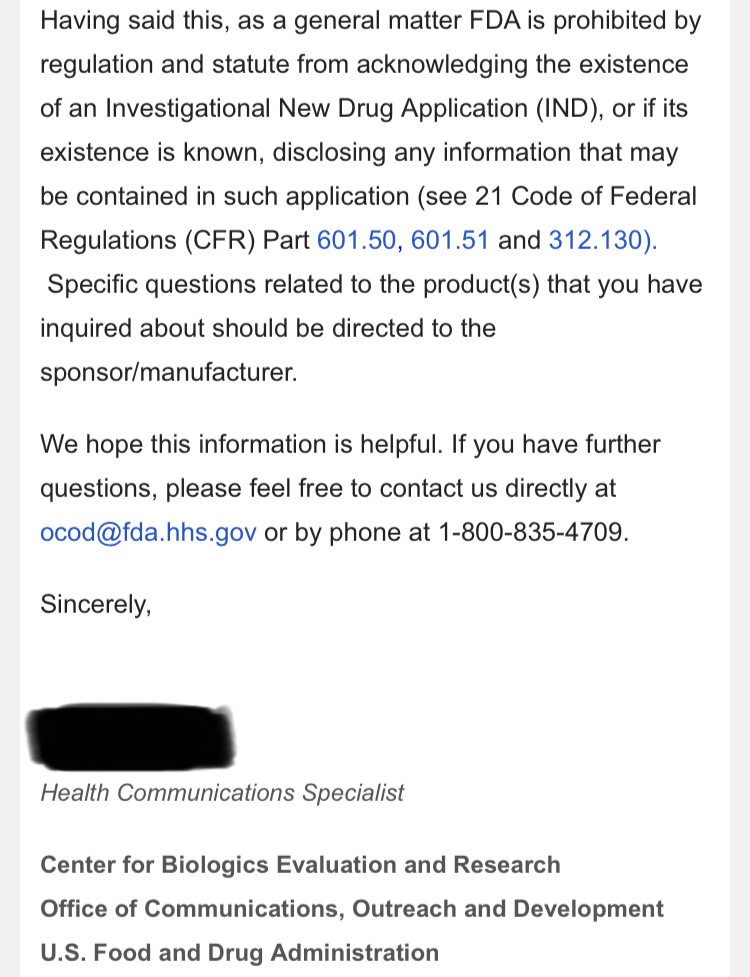
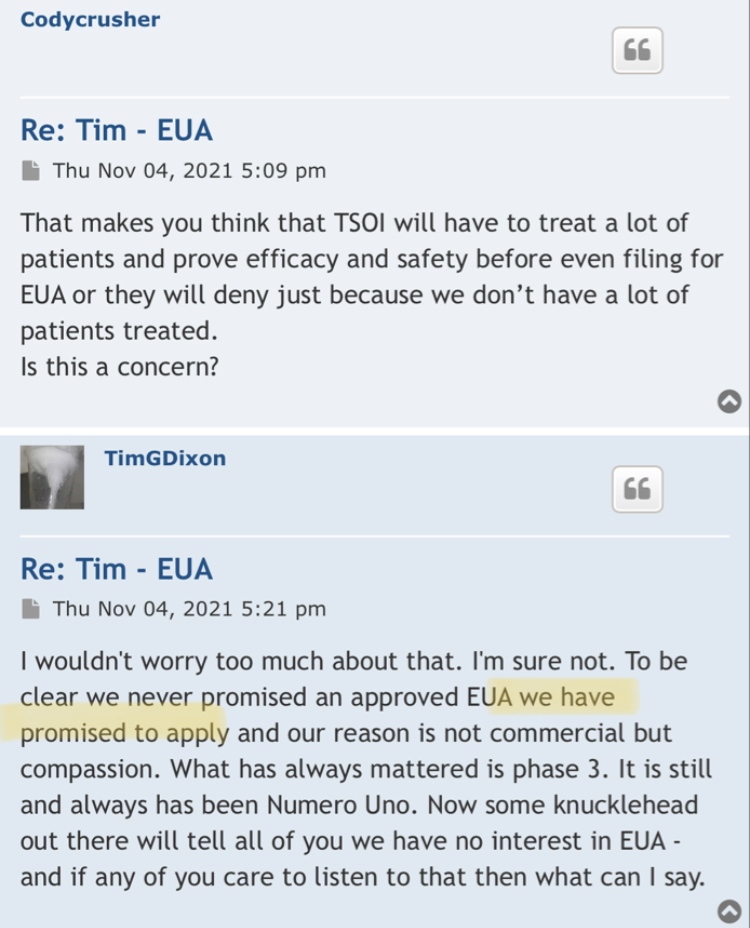
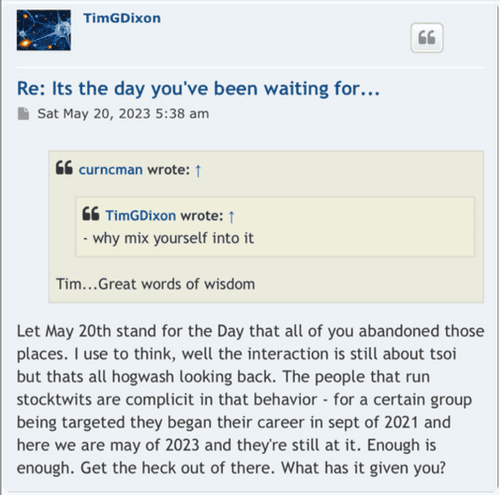
The lies started with the EUA and everyone knows it? That’s just another opinion! Care to enlighten the board what made that EUA press release the perfect opportunity for the CEO to start “supposedly” start lying to shareholders and why he did it? What would have been the benefits of him doing so? Was the CEO lying in press releases to raise the February 2021 share price to .17 compared to the August pump to .13? I hear crickets…
Your going to sit there and tell me that when the ceo of TSOI press released the RUSH to file for EUA he didn’t know he couldn’t even file?
Seriously? Let’s be honest.
Because the lies started with EUA and you and everyone here knows it.
I never said he was a liar! All I mentioned is that the CEO was not considered a liar prior to some becoming shareholders before the Phase 3 and EUA press release! Wouldn’t profiling the CEO as a liar be a red flag? Obviously not for some! So there’s that…
nice GTSM 2 milly bid @ 012
does retail fill the need or
Glad you acknowledge he is in fact a liar
speaking of funding & remembering. Only difference is "Freely" NO Use of Rule 144
GHS Purchased Shares Freely Tradable w/o restrictions on resale
as per Purchase Agreement
(b) Settlement for Purchase Shares
All Purchase Shares issued hereunder will be DWAC Shares.
DWAC Shares means shares of Common Stock that are (i) issued in electronic form. (ii)freely tradable and without restriction on resale and (iii) timely credited by the company to the Investor's or it's designee's specific Deposit Withdrawal at Custodian (DWAC) account with DTC under it's Fast Automated Securities Transfer (FAST) Program, or any similar program hereafter adopted by DTC performing substantially the same function.
remember that post after the results come in
just keeping it real
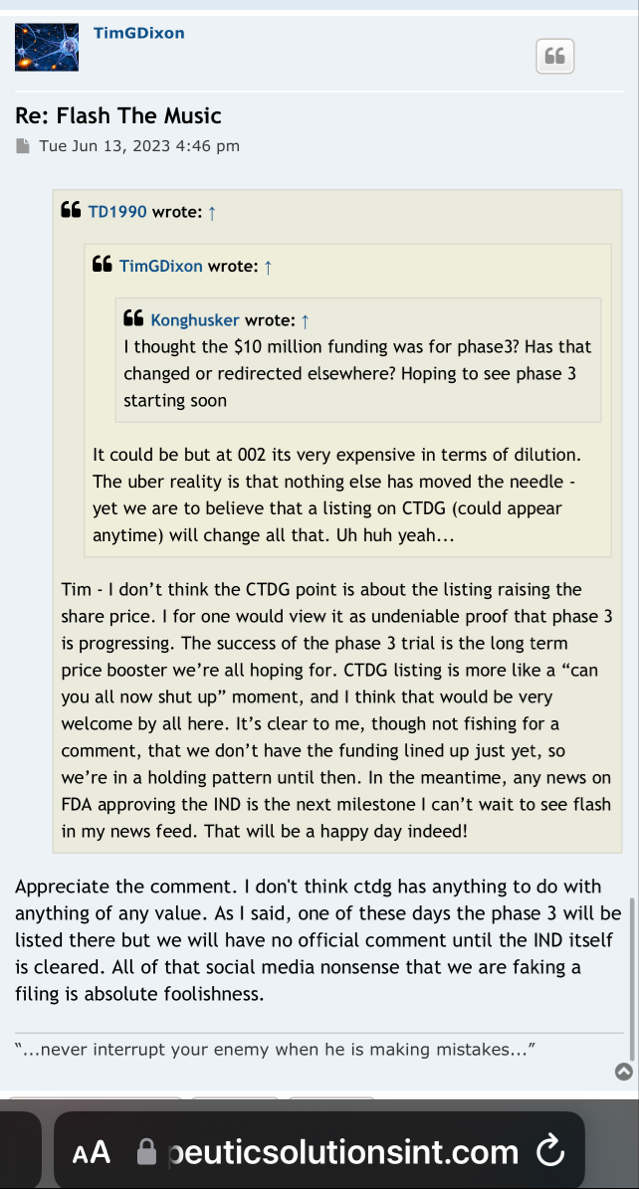
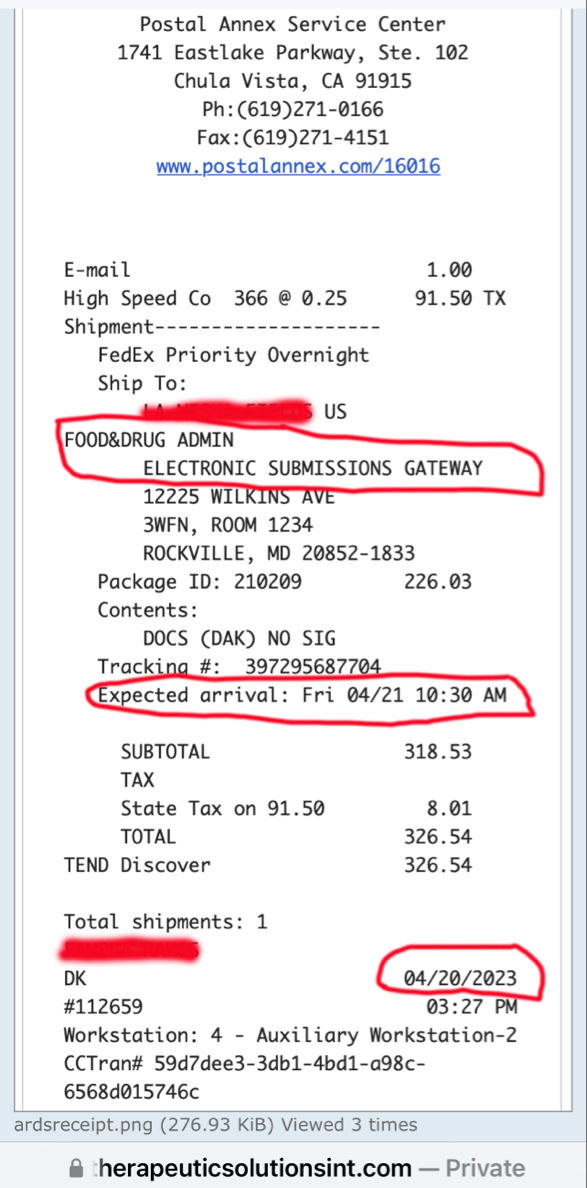
the purpose of securing the 10 Million in funding,
was to fund the Phase 3 clinical trial in ARDS.
(e) Use of Proceeds. The Company will use the net proceeds from the offering for any corporate purpose at the sole discretion of the Company.
that's correct.
they could use it any way where it is needed.
Let's remember that they also have to maintain the cell banks to continue with the clinical trial , as well as for afterwards to keep the products ready for future patients .
It's easy to miss that part of the overall biz plan.
Therapeutic Solutions International Announces Formation of Allogen Biologics Inc.
October 18, 2021, 16:02 GMT
https://www.einnews.com/pr_news/554138453/therapeutic-solutions-international-announces-formation-of-allogen-biologics-incShare This Article
Wholly Owned Subsidiary Created for Manufacturing of JadiCell™ and StemVacs™ Clinical Stage Cell Therapeutics
Therapeutic Solutions International, Inc. (OTCMKTS:TSOI)
OCEANSIDE, CA, USA, October 18, 2021 /EINPresswire.com/ -- Therapeutic Solutions International, Inc., (OTC Markets: TSOI) announced today the formation of Allogen Biologics Inc, a wholly owned subsidiary of the Company.
Last week the Company successfully initiated production of its first pilot batch production of JadiCells ™ under Good Manufacturing Practices in anticipation for uses such as Emergency Use Authorization requests, Right To Try, and our upcoming CTE clinical trial. Allogen Biologics will be led by Dr. Thomas Ichim, currently a board member of Therapeutic Solutions International. Dr. Ichim is an industry veteran with four cleared INDs, over 200 patents and patent applications, and over 120 peer reviewed papers.
Allogen Biologics will house intellectual property and Standard Operating Procedures related to generation of the Company’s existing and anticipated cellular therapeutics.
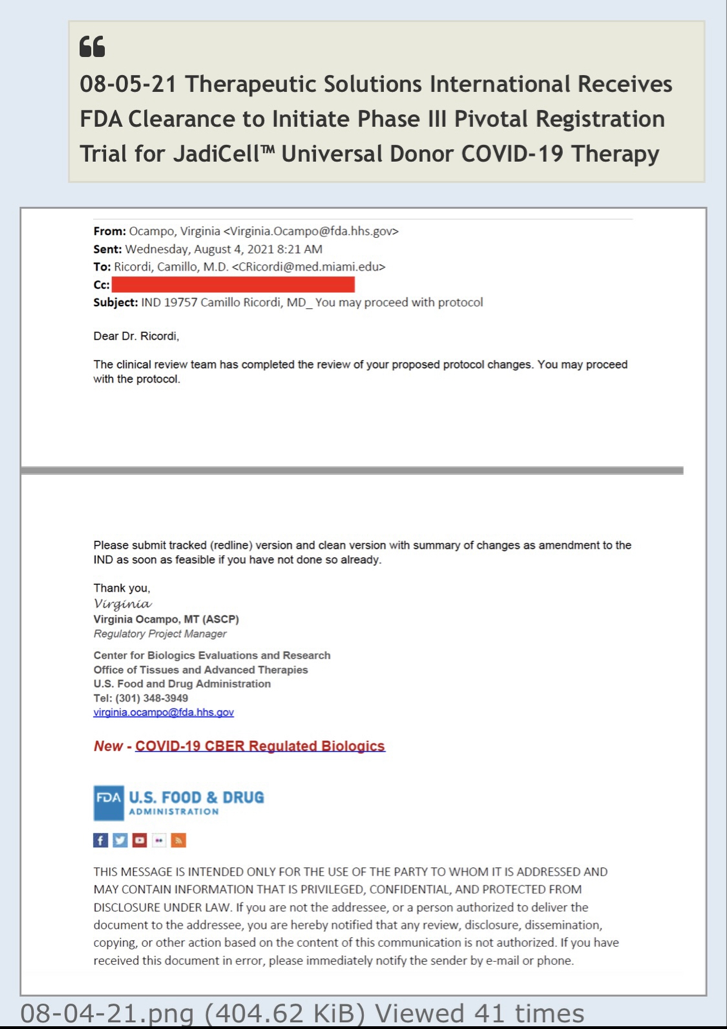
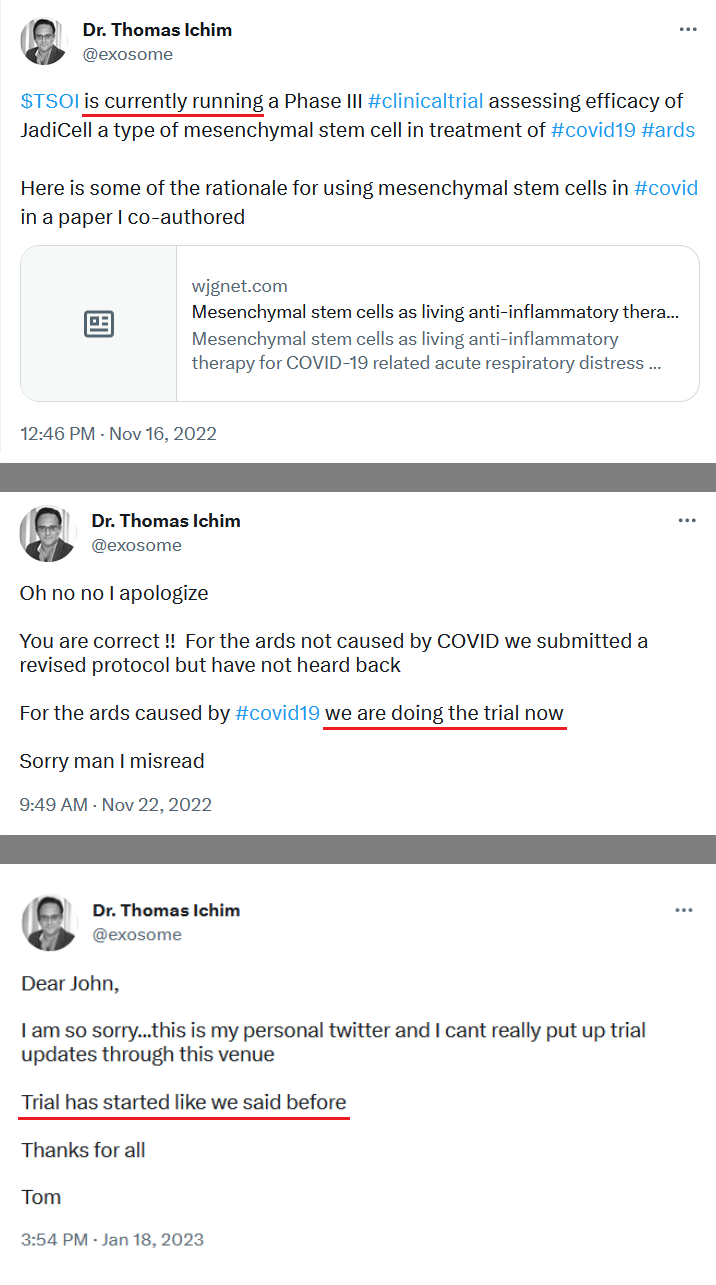
The JadiCell™ is a universal donor stem cell that has recently been cleared by the FDA to begin Phase III clinical trials of COVID-19. Under the Right to Try Law, companies are permitted to administer and charge for experimental therapeutics if no other treatment options exist. The Company has recently treated a Navy SEAL under this exemption.
“As we advance our cellular therapeutic products, the need for flexibility and manufacturing has become more and more apparent. By controlling our own manufacturing, we believe the Company is in a significantly better position to advance its objectives and reduce costs of production” said Timothy Dixon, President and CEO of Therapeutic Solutions International and Chairman of Allogen Biologics. “Dr. Ichim is one of the most accomplish developers of cellular therapies in regenerative medicine and immunology and we are honored for him to lead Allogen Biologics.”
“Timothy Dixon is a true visionary and innovator” said Dr. Thomas Ichim. “Though his efforts, and the work of his advisors such as Francesco Marincola from Gilead, Santosh Kesari from John Wayne Cancer Center, and Boris Reznik from Venvalo, Timothy Dixon has developed multiple drug candidates and nutraceuticals for a variety of therapeutic indications. I am excited to increase my involvement with Timothy and his world-class team.”
they handed the ball off to
other people, and i would like to see more results from that also.
latest patent to come out was PRed on Mar 23, 2021,
and just published today .
18 months for that one.
But, but, but the TSOI CEO wasn’t considered a liar until the Phase 3 and EUA press release in August 2021? I see! Maybe that’s why the DD was considered lovable for the prior decade before that! That also means the February 2021 sell off from .17 down to .03 was lovable! Go figure…
(e) Use of Proceeds. The Company will use the net proceeds from the offering for any corporate purpose at the sole discretion of the Company.
the purpose of securing the 10 Million in funding,
was to fund the Phase 3 clinical trial in ARDS.
10 Million to get to the end results / the data from that trial , and then
it's either a partnership, licensing, a buyout, or treatments in clinics . These are several of the options i can come up with when they get to point B
so remember , it's the funding that was secured to get to point B .
i hope when the right time comes, we see better valuation, after that 10 million infusion.
It's bio, it's risky like everything else. there's always competition to get your products to the market in Bio.
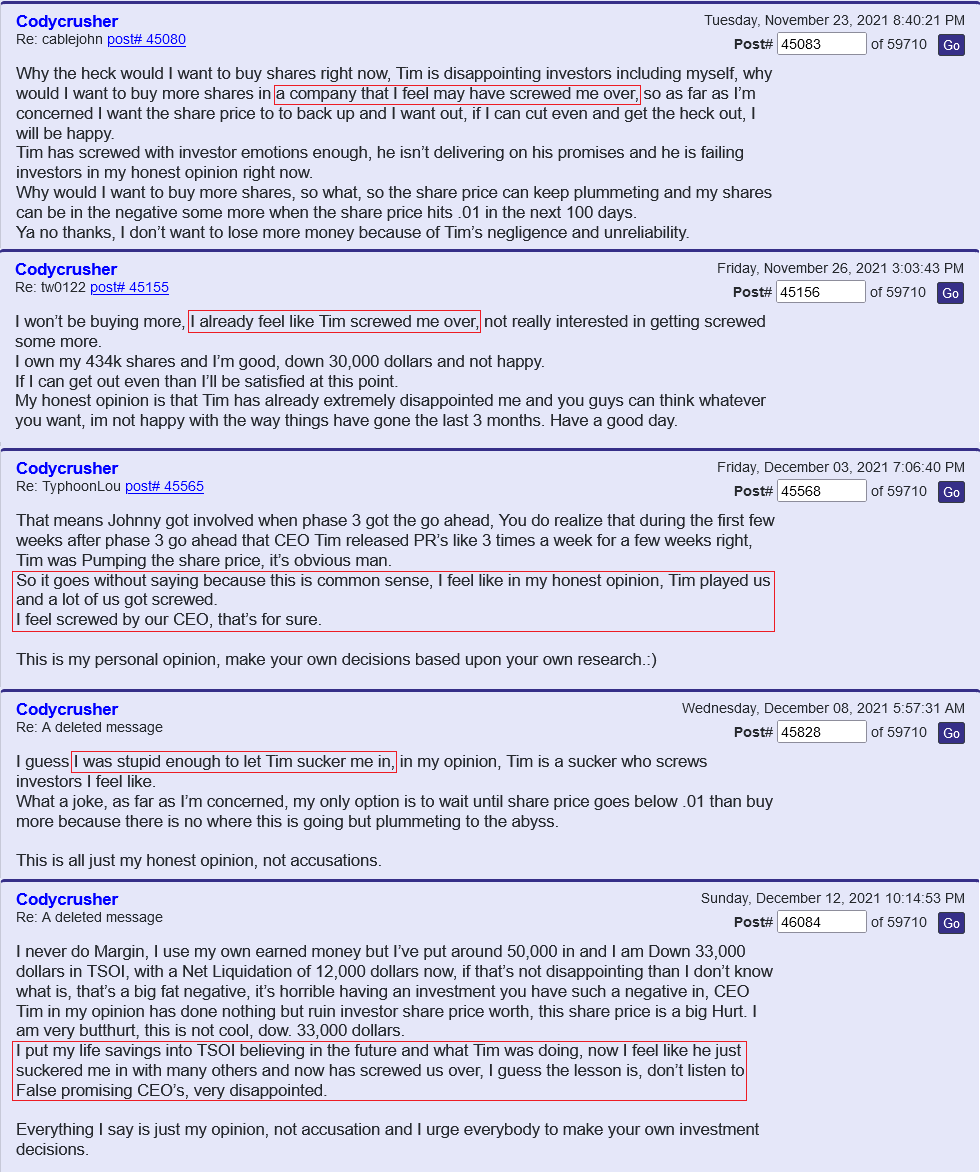
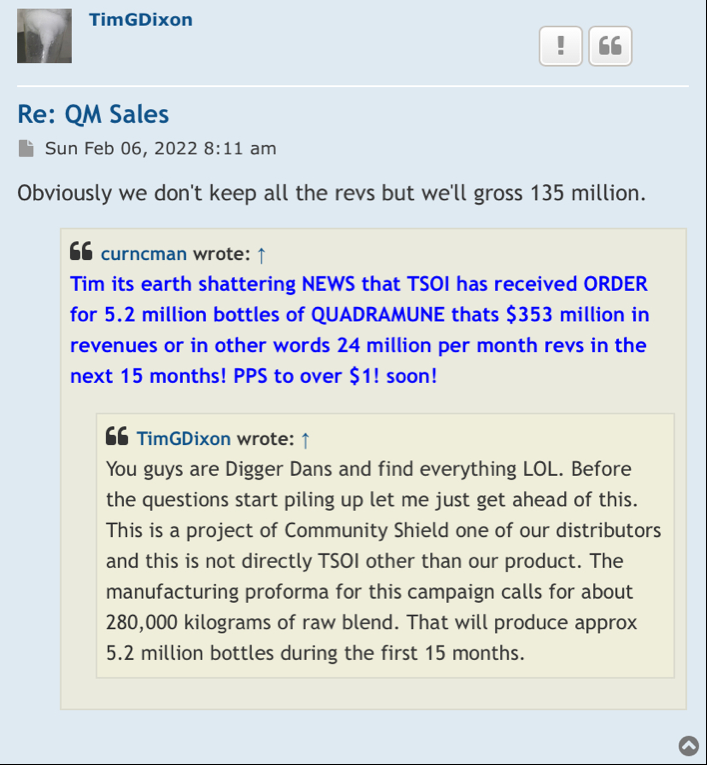
But but but it’s worth trillions of dollars and wait the supplements are suppose to be in 600 stores and that’s millions
Oh wait yeah that was just some more bullshit pump earlier this year from they lying ceo
BBW you are right I absolutely agree with you. We need revenue coming in badly in order for this price to do anything. Course that’s going to be some time off before that happens because this one man show in my opinion are gonna do it their way best or not when I think you could sell some thing you may not get the price you want but get you it will get you a start.
I think Quadra Mune would be a good place to start I don’t know what it would bring I’m curious is why others would think it you could get out of it but whoever buys it Shirley can make or get the price down and really advertise it worldwide to where you could really sell millions of it Right now it’s running around $73 the supplement is and it’s about twice as high as other supplements I take the average middle-class person $73 is tough on them especially if they’re taking others to. But if they could sell millions of bottles every quarter and get the price down 3540 bucks I think the thing would take on off the cell at worldwide.
Do you have an idea of what Quadra moon is worth if you sold it to another company and what it would bring you I’m curious maybe it won’t bring anything I don’t know so maybe I’m barking up the wrong tree but I’d like to know others opinions of what it could bring.
TSOI needs real R&D Revenues & Deposits
Really? So all other penny stock bios besides TSOI are trading green today when all the major stock exchanges are deep in the red? I find that hard to believe! Aren’t they independent from what’s happening with other stock indexes? Can’t blame that one!
that bump wasn't TSOI's doing-imo
It came outta nowhere, and was a surprise surprise - even for Gomer Pyle.
whether it was a bump related to TSOI having the 10Million dollar funding, or not,
that bump came out of nowhere.
Let's see how it plays out , and not jump the gun yet.
Haha right. Tsoi is a penny stock. Anyone who trades penny stocks knows market movement has nothing to do with fly by night wannabe bios. But hey ya found something to blame it on.
Down down she goes.
I wonder why all the major indexes are down significantly today? Would that ever effect TSOI regardless of the patent news? Yawn…
Back to sub penny we go. Great job tsoi
Pretty clear it’s a red day. That’s typical with every damn time tsoi had any patent news. Yawn. Next.
Yippee! What does that mean? A TSOI green day…. Yea, inverted south!
Yippee it’s patent Thursday! What’s that mean kiddos? A red day
We shalll see. It is a great day to be alive though
WOW, today should be a good day, I can feel it in my bones.
Whelp there went them gains haha wow
subs, good statement from TSOI, we are on the move ![]()
maybe what i should have said:
weak hands into stronger hands :)
gotta do that if you want a long train runnin
but for churning .
I been around the rails, waitin for that train .
But i be here cookin some hobo chili w/ my ear to the rail.
:)
Agreed Yoop :)
Most don't want to wait .
taking profits always comes first to those that wanna beat everyone else out the door.
now is a good time for it to move up again, and leave the daytrading mentality behind . it would be kill to see it happen on this long train runnin .
the main structural problem of the OTC / Pinks is exactly this .
Very rarely any startups have any good runs because of it .
I know that no trader wants to get stuck either, but there is no balance .
Looks like the answer is - the sell off and profit taking is happening today
|
Followers
|
526
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
65630
|
|
Created
|
10/04/08
|
Type
|
Free
|
| Moderators BigBadWolf johnnytrader33 JMC$ Yooperman Hogwarts | |||























































Preclinical Data Suggests QuadraMune™ Prevents Stress-Induced Suppression of Neurogenesis More Effectively than Prozac
OCEANSIDE, Calif., Dec. 9, 2020 /PRNewswire/ -- Therapeutics Solution International, Inc., (OTC Markets: TSOI), announced today new data suggesting the possibility that QuadraMune™ may mediate neuroprotective activity through preserving the ability of regenerative brain cells to proliferate subsequent to psychological stress.
The experiments, which involved exposing mice to established stressors, demonstrated that specific areas of the brain associated with production of new brain cells are damaged by stress. In agreement with previously published research, administration of fluoxetine (Prozac™) protected the brain from stress-induced damage. Surprisingly, QuadraMune™ administration appeared superior to Prozac™ at stimulating proliferation of new brain cells.
"QuadraMune™ which is currently in a clinical trial for prevention of COVD-191, has also been demonstrated to possess anti-inflammatory activity in other clinical trials, suppressing cytokines such as IL-62, which are known to be involved in depression3 and suicide4" said Kalina O'Connor, Director of Campbell Neurosciences and co-inventor on the patent. "Given major depressive disorder causes a significant risk for suicide, we are highly interested in exploring the use of QuadraMune™ for preventing suicide."
"Although much enthusiasm has been generated over the planned distribution of the COVID vaccine, at present little is being done to address mental health issues that are being exacerbated by the current pandemic" said Dr. James Veltmeyer, co-inventor of the patent, and Chief Medical Officer of the Company. "If current results are reproducible, the possibility that a nutraceutical would concurrently boost immunity while preserving mental health is highly enticing."
"It has not escaped us that COVID-19 is associated with increased inflammatory cytokines in the blood of patients, cytokines that also predispose to depression" said Famela Ramos, Vice President of Business Development for the Company. "It may be that the recent increase in suicides and suicide attempts is related biologically to activities of the coronavirus. It will be interesting to examine whether QuadraMune™ may modify putative negative mental effects of the virus."
"An estimated 17.3 million adults in the United States had at least one major depressive episode. This number represented 7.1% of all U.S. adults" stated Timothy Dixon, President and CEO of the Company. "We believe the Mission of our Company is not just providing a return on investment to our shareholders, but also increasing the quality of life for Americans. We are extremely pleased to report this unexpected finding with significant potential implications to advancing non-toxic means of helping patients with this terrible condition."
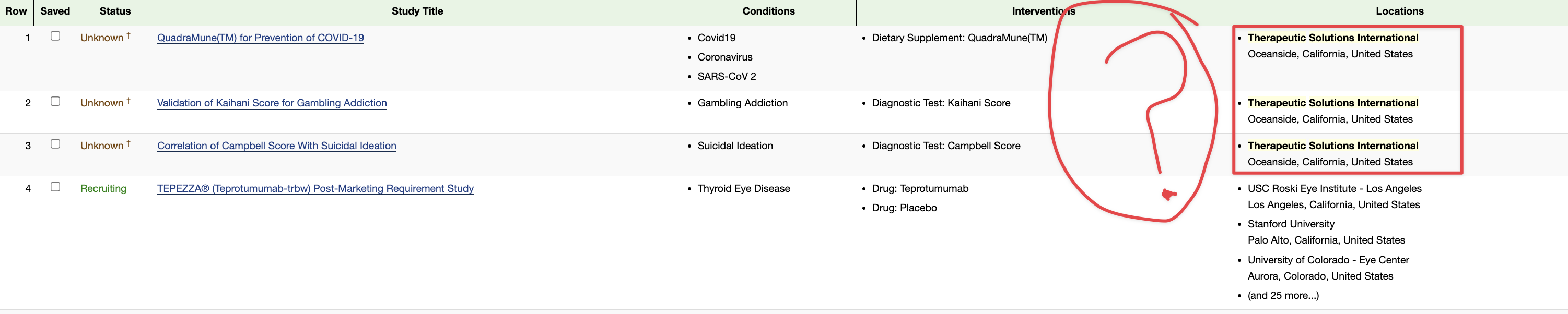
1 QuadraMune(TM) for Prevention of COVID-19 - Full Text View - ClinicalTrials.gov
2 Therapeutic Solutions International Announces Positive Preclinical and Clinical Evaluation of Nutritional Supplement QuadraMune™, Designed to Protect Against COVID-19 | BioSpace
3 Ting et al. Role of Interleukin-6 in Depressive Disorder. Int J Mol Sci. 2020 Mar 22;21(6):2194.
4 O'Donovan et al. Suicidal ideation is associated with elevated inflammation in patients with major depressive disorder. Depress Anxiety. 2013 Apr;30(4):307-14.
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
