Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
They are just part of the team.....we have others on our group........
Z
every DDAmanda post you've provided shows breakout already occurred.
Looks like you're bluffing. You claim DDAmanda scans show stocks right before they run. yet you consistently and every time show stock scans that just broke out.
So your paying DDAmanda customers is the DDAmanda team?
Hold up. So your paying subscribers is the team you refer to as "we" when you refer to DDAmanda doing something like making a post of a stock pick. So, the "we" is in fact only you making a stock pick post.
So you need paying DDAmanda subscribers to make up the DDAmanda team.
No. The scans show possibilities before they run. Of course you have to do the research on them, but invariably they will run based on the scans.......
Z
Yes......we have a membership group.............
Z
Your software scans only show them after they've run. Your DDAmanda doesn't show before stocks have run.
Your examples of runners are what you did find with #DDAmanda but found only after they ran.
You say "we." There's a team of you at DDAmanda?
They really aren't picks.......just examples of runners you could have found with #DDAmanda........
Z
More........and occasionally we do post picks.........like $IDGR for instance.......
Z
So you're not in-heavy on any DDAmanda board-posted picks.
Just in heavy on some no news pick that you have inside information on, that also you've never posted a DDAmanda chart along with notations that you do with hundreds of other tickers.
What's heavy, $1,000?
Can't buy them all.......currently in heavy on $IDGR ......now you have a pick that hasn't run ........yet.......
Z
You specifically recommended SNGX. Then you re-emphasized your recommendation.
Yet you don't buy your own called-out recommendations.
LOL.
As for your DD Amanda "picks" as found from your software, you list them on ihub only after they breakout.
I bet you're full of misleading claims.
As long as those running the company keep getting their pay check and options this company will continue to do stock offerings until the count is high and the price low.
Then it will r/s...over and over and over.
Rivax was/is a joke
It all is a joke
I miss the days when one of management had a breakdown on the elevator at the FDA meeting, and had all that drama. At least it was entertaining as it was disappointing.
I don't buy every one of my recos.......and a lot of those aren't recos......they're examples of how #DDAmanda finds winners.........
Z
Does SNGX 5/22 meeting doc show potential r/s? Again?
Well, I think your answer speaks for itself about your recommendations.
You are partially correct. SNGX has done many R/S. I know as I lost a lot of money believing in their delusions and went through a whole lot of R/S
This
Company
Will
Never
Enrich
SNGX
Shareholders...
Ever.
Schaber would be a better librarian or city refuse collector than a CEO of anything that employs more than himself and his dog.
SNGX already did 1 reverse split but Schaber doesn’t care about PPS so allows to tank, dilute, reverse, repeat…
Good business. For him
Did you buy? I don't like the financials that I posted the other day, so won't buy, did you?
They have to dilute to raise Shareholder Equity to avoid delisting, correct?
But how will they satisfy the $1 minimum bid price requirement? That seems like a bigger problem. They tried pretty hard to pump it up over $1 but it just didn't work. Dilution won't fix the Minimum Bid Price problem.
From the 10-K:
On June 23, 2023, we received a letter from the Listing Qualifications Department of Nasdaq stating that we were not in compliance with the $1.00 minimum bid price requirement set forth in Nasdaq Listing Rule 5550(a)(2) for continued listing on the Nasdaq Capital Market (the “Minimum Bid Price Rule”) because our common stock failed to maintain a minimum closing bid price of $1.00 for 30 consecutive trading days. In accordance with Nasdaq Listing Rule 5810(c)(3)(A), we were afforded an initial period of 180 calendar days, or until December 20, 2023, to regain compliance with the Minimum Bid Price Rule. We were unable to regain compliance with the Minimum Bid Price Rule prior to the expiration of the 180 calendar day period.
On December 21, 2023, we received written notice from Nasdaq stating that we had not complied with the Minimum Bid Price Rule and were not eligible for a second 180-day period because we did not comply with the $5,000,000 minimum stockholders’ equity initial listing requirement for The Nasdaq Capital Market. In that regard, our Quarterly Report on Form 10-Q for the quarter ended September 30, 2023 reported stockholders’ equity of $4,221,155. As a result, the notice indicated that our common stock would be suspended from trading on Nasdaq unless we requested a hearing before a hearings panel by December 28, 2023. Nasdaq has scheduled a hearing for March 26, 2024, which stayed any trading suspension of our common stock until completion of the Nasdaq hearing process and expiration of any additional extension period granted by the panel following the hearing.
Stay tuned......it's very rare that a stock will go up as soon as you buy........lol.......
Z
Schaber such a skank dilute with .40 shares with supposed millions in cash on hand???
ASTI, a ddamanda software pick down 66% in a week after a massive dilutive $6M offering priced at $0.14 is good stock pick why?
I suppose the point that you post about the software that shows illustrations of tickers only after a stock breaks out, not before, and not before with time to enter. So based upon your near daily ticker posts what your software seems to show is high risk entry points after breakouts.
Only the Shadow knows what lies in the Hearts of Men..................
Z
Definitely orchestrated pump with DUMP hard so far today…
Schaber only looking to keep his salary afloat by adding shares??
GLTA
They're not picks.........they're illustrations of what #DDAmanda can do...........
However if you want a pick: $ASTI
Z
Yeah.........could get a second wind..........
Z
I notice you announce most all your picks after they have broken out.
Thanks. SNGX horribly short pop…
FDA Grants Soligenix Orphan Drug Designation for the Prevention and Post-Exposure Prophylaxis Against Marburg Marburgvirus Infection
Exactly. Playing this more for the volatility with a small float. Figured we'd see the .30's.
And down SNGX goes..
Too many prior “orphan grants” and nothing happens…
GLTA
FDA Grants Soligenix Orphan Drug Designation for the Prevention and Post-Exposure Prophylaxis Against Sudan Ebolavirus Infection
Provides SuVax™ Heat Stable Vaccine Seven Years of U.S. Market Exclusivity Upon FDA Approval
Facebook Twitter Instagram Linkedin Youtube
Princeton, NJ – April 11, 2024 – Soligenix, Inc. (Nasdaq: SNGX) (Soligenix or the Company), a late-stage biopharmaceutical company focused on developing and commercializing products to treat rare diseases where there is an unmet medical need, announced today that the Office of Orphan Products Development of the United States (U.S.) Food and Drug Administration (FDA) has granted orphan drug designation to the active ingredient in SuVax™, the subunit protein vaccine of recombinantly expressed Sudan ebolavirus (SUDV) glycoprotein, for “the prevention and post-exposure prophylaxis against SUDV infection.”
The U.S. Orphan Drug Act is intended to assist and encourage companies to develop safe and effective therapies for the treatment of rare diseases and disorders, defined as one that affects fewer than 200,000 people in the U.S. In addition to providing a seven-year term of market exclusivity upon final FDA approval, orphan drug designation also positions Soligenix to be able to leverage a wide range of financial and regulatory benefits, including government grants for conducting clinical trials, waiver of expensive FDA user fees for the potential submission of a Biologics License Application (BLA), and certain tax credits.
“SuVax™ is based on our novel vaccine platform which includes three major components: a robust protein manufacturing process that has been demonstrated on multiple protein antigens, a novel nano-emulsion adjuvant which induces broad immunity, and a formulation procedure which enables thermostabilization of the combination of adjuvant and antigen in a single vial,” stated Oreola Donini, PhD, Senior Vice President and Chief Scientific Officer of Soligenix. “The SuVax™ vaccine specifically addresses the potentially lethal Sudan Virus Disease, caused by SUDV. SUDV is a type of ebolavirus for which there is no current treatment or vaccine. The SuVax™ vaccine has demonstrated 100% protection of non-human primates infected with a lethal amount of SUDV. Although vaccines exist for Zaire ebolavirus, they are ineffective against SUDV. With recent outbreaks in 2022 and 2023, developing vaccines for all Ebola type diseases remains an important worldwide priority. The FDA’s decision to grant orphan drug designation signifies an important step for Soligenix as we continue to advance the program and adds significantly to the existing patent estate surrounding this novel technology.”
About SuVax™
SuVax™ is a subunit protein vaccine of recombinantly expressed Sudan Ebola virus glycoprotein, developed in partnership with Dr. Axel Lehrer at the University of Hawai?i at Manoa. The vaccine includes a protein found on the surface of Sudan ebolavirus (SUDV), to engender an appropriate immune response without posing a risk of infection, as well as a novel adjuvant which stimulates both humoral and cell mediated immune responses, in combination with Generally Regarded as Safe (GRAS) excipients that enable lyophilization (i.e., freeze-drying) of the vaccine. The resulting product is manufactured as a heat stable powder in a vial which is reconstituted with generically available water for injection immediately prior to use. SuVax™, as a heat stable protein subunit vaccine, has protected 100% of non-human primates exposed to a lethal injection of SUDV. Stability studies have demonstrated that SuVax™ is heat stable for at least 2 years at temperatures of at least 40 degrees Celsius (104 degrees Fahrenheit).
Manufacture of the recombinant protein utilized in SuVax™ utilizes a robust protein manufacturing process, developed and tested in other subunit vaccines advanced through clinical testing. Similarly, the selected adjuvant, while novel, has also been independently tested in Phase 1 and Phase 2 clinical studies. SuVax™ can also be expressed as part of a multivalent vaccine, in combination with antigens against Marburg marburgvirus (MARV) for example.
About Sudan ebolavirus Infection
Ebola-like disease can be caused by one of six species of Ebolavirus, four of which are known to cause disease in humans, including its best-known member, Zaire ebolavirus (EBOV; causing Ebola Virus Disease), with Sudan ebolavirus (SUDV; causing Sudan Virus Disease) being the second-most common cause of human infection. All species of ebolavirus belong to the Filoviridae family, a family that further contains the equally human pathogenic Marburg marburgvirus (MARV). Filoviruses are believed to be harbored in various animal species in Africa, particularly bats, although the specific reservoir host for many of these viruses is still unknown. There have been several known Ebola (both Sudan and Zaire) and Marburg Virus Disease outbreaks since 1967 with the most recent SUDV outbreak occurring in August – October, 2022 in Uganda according to the Centers for Disease Control and Prevention (CDC).
Transmission of filoviruses requires direct contact with bodily fluids from an infected person or contact with infected animals. The mortality rates following filovirus infections are extremely high, and, in the absence of wide availability of effective therapeutics, are affected by the quality of supportive care available with a focus on early initiation of treatment. Resolution of the disease largely depends on the patient’s own immune system. While there are limited treatment options for disease caused by EBOV, there are no available treatments or vaccines available for Sudan Virus Disease. The approved vaccines for EBOV utilize a viral vector approach which has contraindications for some individuals and require stringent, ultra-low cold-chain storage, inhibiting their broad use in challenging conditions where power supply can be uncertain and ambient temperature can be very high.
About Soligenix, Inc.
Soligenix is a late-stage biopharmaceutical company focused on developing and commercializing products to treat rare diseases where there is an unmet medical need. Our Specialized BioTherapeutics business segment is developing and moving toward potential commercialization of HyBryte™ (SGX301 or synthetic hypericin sodium) as a novel photodynamic therapy utilizing safe visible light for the treatment of cutaneous T-cell lymphoma (CTCL). With successful completion of the second Phase 3 study, regulatory approvals will be sought to support potential commercialization worldwide. Development programs in this business segment also include expansion of synthetic hypericin (SGX302) into psoriasis, our first-in-class innate defense regulator (IDR) technology, dusquetide (SGX942) for the treatment of inflammatory diseases, including oral mucositis in head and neck cancer, and (SGX945) in Behçet’s Disease.
?
Our Public Health Solutions business segment includes development programs for RiVax®, our ricin toxin vaccine candidate, as well as our vaccine programs targeting filoviruses (such as Marburg and Ebola) and CiVax™, our vaccine candidate for the prevention of COVID-19 (caused by SARS-CoV-2). The development of our vaccine programs incorporates the use of our proprietary heat stabilization platform technology, known as ThermoVax®. To date, this business segment has been supported with government grant and contract funding from the National Institute of Allergy and Infectious Diseases (NIAID), the Defense Threat Reduction Agency (DTRA) and the Biomedical Advanced Research and Development Authority (BARDA).
?
For further information regarding Soligenix, Inc., please visit the Company's website at https://www.soligenix.com and follow us on LinkedIn and Twitter at @Soligenix_Inc.
This press release may contain forward-looking statements that reflect Soligenix, Inc.’s current expectations about its future results, performance, prospects and opportunities, including but not limited to, potential market sizes, patient populations and clinical trial enrollment. Statements that are not historical facts, such as “anticipates,” “estimates,” “believes,” “hopes,” “intends,” “plans,” “expects,” “goal,” “may,” “suggest,” “will,” “potential,” or similar expressions, are forward-looking statements. These statements are subject to a number of risks, uncertainties and other factors that could cause actual events or results in future periods to differ materially from what is expressed in, or implied by, these statements, and include the expected amount and use of proceeds from the offering and the expected closing date of the offering. Soligenix cannot assure you that it will be able to successfully develop, achieve regulatory approval for or commercialize products based on its technologies, particularly in light of the significant uncertainty inherent in developing therapeutics and vaccines against bioterror threats, conducting preclinical and clinical trials of therapeutics and vaccines, obtaining regulatory approvals and manufacturing therapeutics and vaccines, that product development and commercialization efforts will not be reduced or discontinued due to difficulties or delays in clinical trials or due to lack of progress or positive results from research and development efforts, that it will be able to successfully obtain any further funding to support product development and commercialization efforts, including grants and awards, maintain its existing grants which are subject to performance requirements, enter into any biodefense procurement contracts with the U.S. Government or other countries, that it will be able to compete with larger and better financed competitors in the biotechnology industry, that changes in health care practice, third party reimbursement limitations and Federal and/or state health care reform initiatives will not negatively affect its business, or that the U.S. Congress may not pass any legislation that would provide additional funding for the Project BioShield program. In addition, there can be no assurance as to the timing or success of any of its clinical/preclinical trials. Despite the statistically significant result achieved in the first HyBryte™ (SGX301) Phase 3 clinical trial for the treatment of cutaneous T-cell lymphoma, there can be no assurance that the second HyBryte™ (SGX301) Phase 3 clinical trial will be successful or that a marketing authorization from the FDA or EMA will be granted. Additionally, although the EMA has agreed to the key design components of the second HyBryte™ (SGX301) Phase 3 clinical trial, no assurance can be given that the Company will be able to modify the development path to adequately address the FDA’s concerns or that the FDA will not require a longer duration comparative study. Notwithstanding the result in the first HyBryte™ (SGX301) Phase 3 clinical trial for the treatment of cutaneous T-cell lymphoma and the Phase 2a clinical trial of SGX302 for the treatment of psoriasis, there can be no assurance as to the timing or success of the clinical trials of SGX302 for the treatment of psoriasis. Despite the positive efficacy results demonstrated in the Phase 2 and 3 clinical studies of SGX942 for the treatment of oral mucositis due to chemoradiation therapy for head and neck cancer, there can be no assurance as to the timing or success of the clinical trials of SGX945 for the treatment of Behçet’s Disease. Further, there can be no assurance that RiVax® will qualify for a biodefense Priority Review Voucher (PRV) or that the prior sales of PRVs will be indicative of any potential sales price for a PRV for RiVax®. Also, no assurance can be provided that the Company will receive or continue to receive non-dilutive government funding from grants and contracts that have been or may be awarded or for which the Company will apply in the future. These and other risk factors are described from time to time in filings with the Securities and Exchange Commission (the “SEC”), including, but not limited to, the Company’s preliminary prospectus (Registration No. 333-271049) filed with the SEC on May 4, 2023, and Soligenix’s reports on Forms 10-Q and 10-K. Unless required by law, Soligenix assumes no obligation to update or revise any forward-looking statements as a result of new information or future events.
Past Press Releases
April 3, 2024 - Soligenix Announces Agreement on the Design of a Second Confirmatory Placebo-Controlled Trial for HyBryte™ With the European Medicines Agency
March 15, 2024 - Soligenix Announces Recent Accomplishments and Year End 2023 Financial Results
February 8, 2024 - Soligenix Announces Formation of Behçet's Disease Medical Advisory Board
Company Contact Information
?
Jonathan Guarino
Chief Financial Officer
(609) 538-8200
ir@soligenix.com
29 Emmons Drive
Suite B-10
Princeton, NJ 08540
www.soligenix.com
Lower imo. At this level it's a go for me in the .30's. Anywhere around there into the .40's is worth the risk for me really. This pos just may give a decent run into the summer... sell in may???? lol
We'll see. I put this back on my radar anyway. Been a bit.
https://bigcharts.marketwatch.com/advchart/frames/frames.asp?show=&insttype=&symb=sngx&x=0&y=0&time=3&startdate=1%2F4%2F1999&enddate=4%2F9%2F2024&freq=6&compidx=aaaaa%3A0&comptemptext=&comp=none&ma=6&maval=9&uf=8&lf=65536&lf2=16&lf3=512&type=4&style=320&size=4&timeFrameToggle=false&compareToToggle=false&indicatorsToggle=false&chartStyleToggle=false&state=11
Nice try Dr Shabby with repeat PR: "April 3, 2024 Randomized Study with 18 Weeks Continuous Treatment Expected to Replicate and Extend Results from the First, Statistically Significant Phase 3 Study"...
BS press release after yesterday 4/2 filing for your 16m share dilution AGAIN...
How LOW do you take SNGX ???
Schaber must need few bucks so planning another RS.
Nice business.
Jim Cramer should go down with this ship show as he promoted it….
OK, well good on you for saying so. Now, why are we feeling a $27k investment is going to influence this sucker at all?
Yeah ok. That was a squeeze play and I got it wrong.
ehhhh, not so much....this was it's top of the market at the time. Within a month the stock was at .63....I believe you called me "salty" when I spoke out against your recommendation.
masterofdisaster
Re: None
Monday, August 07, 2023 11:19:15 AM
Post#
10993
of 11238
Buying at $2.75
It went up and then cratered
I think you said the same thing right before Rite Aid cratered.
Someone just bought 38,000 shares. Something big is happening here.
There must be potential here a lot of buying going on
Maybe I lied to myself and will buy some today. Geez I HATE penny stocks!
|
Followers
|
208
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
9772
|
|
Created
|
06/06/06
|
Type
|
Free
|
| Moderators | |||
| SNGX [NASD] |
| Soligenix, Inc. |
| Healthcare | Biotechnology | USA |
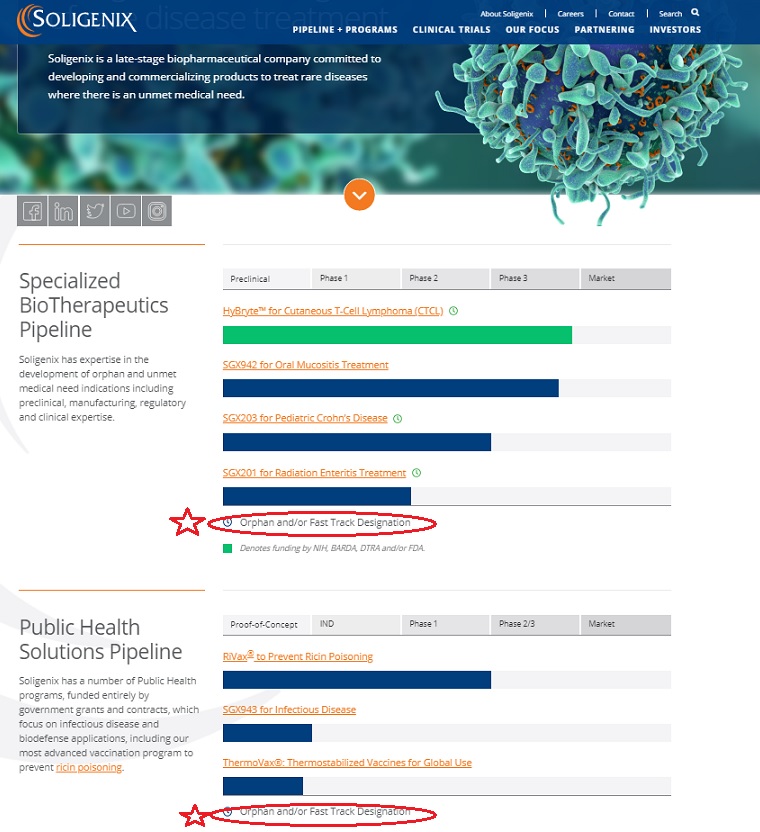
Soligenix, Inc. (Soligenix) is a late-stage biopharmaceutical company.
The Company is focused on developing and commercializing products to treat rare diseases.
The Company operates through two business segments:
Specialized BioTherapeutics and Public Health Solution. Soligenix’s Specialized BioTherapeutics business segment is developing
a photodynamic therapy (SGX301) utilizing topical synthetic hypericin activated with safe visible fluorescent light for the treatment of cutaneous T-cell lymphoma (CTCL).
The Public Health Solutions business segment includes active development programs for RiVax,
its ricin toxin vaccine candidate and SGX943, its therapeutic candidate for antibiotic-resistant and emerging infectious disease.
CTCL is its innate defense regulator technology, dusquetide (SGX942) for the treatment of oral mucositis in head and neck cancer.
Soligenix has expertise in the development of orphan and unmet medical need indications including preclinical, manufacturing, regulatory and clinical expertise.
Soligenix has expertise in the development of orphan and unmet medical need indications including preclinical, manufacturing, regulatory and clinical expertise.
| Preclinical | Phase 1 | Phase 2 | Phase 3 | Market |
|---|---|---|---|---|
| HyBryte™ for Cutaneous T-Cell Lymphoma (CTCL) | ||||
| | | | | |
| SGX942 for Oral Mucositis Treatment | ||||
| | | | | |
| SGX203 for Pediatric Crohn’s Disease | ||||
| | | | | |
| SGX201 for Radiation Enteritis Treatment | ||||
| | | | | |
Orphan and/or Fast Track Designation
Soligenix has a number of Public Health programs, funded entirely by government grants and contracts, which focus on infectious disease and biodefense applications, including our most advanced vaccination program to prevent ricin poisoning.
| Proof-of-Concept | IND | Phase 1 | Phase 2/3 | Market |
|---|---|---|---|---|
| RiVax® to Prevent Ricin Poisoning | ||||
| | | | | |
| SGX943 for Infectious Disease | ||||
| | | | | |
| ThermoVax®: Thermostabilized Vaccines for Global Use | ||||
| | | | | |
Orphan and/or Fast Track Designation


| Preclinical | Phase 1 | Phase 2 | Phase 3 | Market |
|---|---|---|---|---|
| HyBryte™ for Cutaneous T-Cell Lymphoma (CTCL) almost done [in phase 3] | ||||
| | | | | |
| SGX942 for Oral Mucositis Treatment almost done [in phase 3] | ||||
| | | | | |
| SGX203 for Pediatric Crohn’s Disease [phase 2 almost done] | ||||
| | | | | |
| SGX201 for Radiation Enteritis Treatment [phase 1 going into phase 2] | ||||
| Proof-of-Concept | IND | Phase 1 | Phase 2/3 | Market |
|---|---|---|---|---|
| RiVax® to Prevent Ricin Poisoning | ||||
| | | | | |
| SGX943 for Infectious Disease | ||||
| | | | | |
| ThermoVax®: Thermostabilized Vaccines for Global Use | ||||
| | | | | |
Orphan and/or Fast Track Designation
| PER IHUB MGMT |
02-07-2021
DISCLAIMER: ONLY FOR MICK
https://investorshub.advfn.com/boards/profilea.aspx?user=1012
*The Board Monitor and herewithin , are not licensed brokers and assume NO responsibility for actions,
investments,decisions, or messages posted on this forum.
CONTENT ON THIS FORUM SHOULD NOT BE CONSIDERED ADVISORY NOR SOLICITATION
AUTHORS MAY HAVE BUYS OR SELLS WITH THE COMPANIES MENTIONED IN TRADING POSTERS SHOULD DUE DILIGENT BUYING OR SELLING.
ALL POSTING SHOULD BE CONSIDERED FOR INFORMATION ONLY. WE DO NOT RECOMMEND ANYONE BUY OR SELL ANY SECURITIES POSTED HEREWITHIN.
ANY trade entered into risks the possibility of losing the funds invested.
• There are no guarantees when buying or selling any security.Any
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
