Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Guess I shouldn't have posted my sell price. I did get rid of some of it.
Figures. They shut it down right at my cost basis. Dang it!
Does this re-open at $2.50 or .53?
What the heck?? I mention $1.93 on here and suddenly a ton of dumping at $1.93.
OK, I guess it is halted. Now showing 0X0 for price
I don't see a halt.
The stock typically trades under 300k shares per day. Today so far its at 37.5 MILLION!
Nice move today,dang halts,can’t sell,ZERO BORROW,42%fee is promising,profit taking now,sma200 is .80 support bounce
The cost basis on most of my shares is $1.93. I'm hoping to dump when it gets there.
Admittedly I don't have a full grasp on approval for small pharma companies (most still seem worthless). I don't understand how this news means a whole lot to the company as, it's not a phase III approval, but apparently someone does. And they've been loading up all morning.
I don't own enough of this to care much. Stupidly, I bought into it based on Jim Cramer back in November 2020.
I am always on here telling people not to throw out money on stupid stock plays. Occasionally I do it myself and almost without fail, I am reminded not to do it again.
Please do the after lunch BIG run !
GLTA
The moment market makers realized this is for realz!
Geez, I'll be curious to see if all those shares picked up first thing this morning at .56 get dumped later today.
11/30/23 09:51:15 AM D O 2 0.9800 I
11/30/23 09:51:15 AM D O 600 0.9800
11/30/23 09:51:15 AM D O 160 1.0000
11/30/23 09:51:15 AM P 50 1.0000 I
11/30/23 09:51:15 AM D O 941 1.0000
11/30/23 09:51:15 AM P 10 1.0000 I
11/30/23 09:51:15 AM P 46 1.0000 I
11/30/23 09:51:15 AM P 70 1.0000 I
11/30/23 09:51:15 AM P 50 1.0000 I
11/30/23 09:51:15 AM P 1 1.0000 I
11/30/23 09:51:15 AM P 82 1.0000 I
11/30/23 09:51:15 AM D O 100 0.9900
11/30/23 09:51:15 AM O 0.9800 O 1.0000 28 X 465 BBO
11/30/23 09:51:15 AM P 200 0.9800 Y F
11/30/23 09:51:15 AM P 100 0.9800 Y F
11/30/23 09:51:15 AM O 100 0.9800 Y F
11/30/23 09:51:15 AM O 0.9800 O 1.0000 27 X 465 BBO
11/30/23 09:51:15 AM P 200 0.9800 Y F
11/30/23 09:51:15 AM O 37 0.9800 Y F I
Trading a MASSIVE amount of shares today. This may be real. There has been no pre-loading for a pump and dump.
Not sure I trust any of it but....
Soligenix Receives FDA IND Clearance for Phase 2 Clinical Trial of Dusquetide in the Treatment of Aphthous Ulcers in Behçet's Disease
PR Newswire7:30 AM (UTC-05:00) Eastern Time (US & Canada) Nov 30, 2023
Soligenix Receives FDA IND Clearance for Phase 2 Clinical Trial of Dusquetide in the Treatment of Aphthous Ulcers in Behçet's Disease
PR Newswire
PRINCETON, N.J., Nov. 30, 2023
Pipeline Expansion of Novel Innate Defense Regulator Technology
PRINCETON, N.J., Nov. 30, 2023 /PRNewswire/ -- Soligenix, Inc. (Nasdaq: SNGX) (Soligenix or the Company), a late-stage biopharmaceutical company focused on developing and commercializing products to treat rare diseases where there is an unmet medical need, announced today that the U.S. Food and Drug Administration (FDA) has cleared the Investigational New Drug (IND) application for a Phase 2a clinical trial entitled, "Pilot Study of SGX945 (Dusquetide) in the Treatment of Aphthous Ulcers in Behçet's Disease." The study is designed to evaluate the safety and efficacy of SGX945 (dusquetide) and is expected to begin patient enrollment in the second half of 2024.
"We are pleased to have received FDA clearance on our SGX945 Phase 2 pilot trial in aphthous ulcers of Behçet's disease," stated Christopher J. Schaber, PhD, President and Chief Executive Officer of Soligenix. "Our previous studies with dusquetide in oral mucositis have clearly validated the biologic activity in aphthous ulcers induced by chemotherapy and radiation. Given the role of the innate immune system in ulcers associated with Behçet's Disease, and the unmet medical need particularly for more severe ulcers such as genital and leg ulcers, we believe that dusquetide may offer significant relief to patients. We are excited to expand dusquetide's development into different innate immune-related inflammatory conditions such as Behçet's disease, as a component of our long-term strategy to enhance the value of this unique compound. Behçet's disease is an unmet medical need, with up to 18,000 people in the U.S., 80,000 in Europe, 350,000 people in Turkey and as many as 1 million people worldwide affected by this incurable disease. Given our promising results with aphthous ulcers in oral mucositis, we are hopeful dusquetide will have a role to play in helping underserved patients suffering from this difficult to treat and chronic disease."
Under this IND, the pilot clinical trial of SGX945 will be an open-label study that will enroll approximately 25 patients age 18 years or older with mild to moderate Behçet's disease active oral and/or genital ulcers. Patients will receive SGX945 as a twice weekly 4-minute intravenous (IV) infusion for 4 weeks. Efficacy endpoints will include the extent of lesion clearance, timeline to lesion clearance, and patient reported quality of life indices.
About Dusquetide
Dusquetide (the active ingredient in SGX945 and SGX942 in development for oral mucositis) is an innate defense regulator (IDR), a new class of short, synthetic peptides. It has a novel mechanism of action whereby it modulates the body's reaction to both injury and infection towards an anti-inflammatory, anti-infective, and tissue healing response. IDRs have no direct antibiotic activity but, by modulating the host's innate immune system responses, increase survival after infections caused by a broad range of bacterial Gram-negative and Gram-positive pathogens. It also accelerates resolution of tissue damage following exposure to a variety of agents including bacterial pathogens, trauma, and chemo- and/or radiation therapy. Preclinical efficacy and safety have been demonstrated in numerous animal disease models including mucositis, colitis, macrophage activation syndrome (MAS) as well as bacterial infections, including melioidosis. In addition, potential anti-tumor activity has been demonstrated in multiple in vitro and in vivo xenograft studies.
Dusquetide has demonstrated safety and tolerability in a Phase 1 clinical study in 84 healthy human volunteers. In Phase 2 and 3 clinical studies with SGX942 in over 350 subjects with oral mucositis due to chemoradiation therapy for head and neck cancer, positive efficacy results were demonstrated, including potential long-term ancillary benefits.
Soligenix has a strong intellectual property position in the IDR technology platform, including composition of matter for dusquetide and related analogs. Dusquetide was developed pursuant to discoveries made by Professors B. Brett Finlay, PhD and Robert Hancock, PhD of the University of British Columbia, Canada.
About Behçet's Disease
Behçet's Disease (BD) is commonly known as an inflammatory disorder of the blood vessels (vasculitis). Often first diagnosed in young adults, it's effects and severity will wax and wane over time. Major signs and symptoms usually include mouth sores (approximately 95% of patients), skin rashes and lesions (approximately 50% of patients), genital sores (approximately 50% of patients), leg ulcers (approximately 40% of patients) and eye inflammation (approximately 15% of patients). It is a painful disease, directly impacting the patient's quality of life and ability to productively engage in life activities, including work.
BD is thought to be an auto-immune disease with both genetic and environmental factors. It is most common along the "Silk Road" in the Middle East and East Asia, including Turkey, Iran, Japan and China. There are approximately 18,000 known cases of BD in the U.S. and 80,000 in Europe. There are as many as 1,000,000 people worldwide living with BD.
There is no cure for BD, rather treatments are prescribed to manage symptoms. Treatments may include both maintenance therapies and those specifically addressing flares (e.g., mouth ulcers, genital ulcers and leg ulcers). Corticosteroids are generally applied topically to sores and as eyedrops and may also be given systemically to reduce inflammation. Although used frequently, they have limited efficacy over the long-term and have significant side effects that become more concerning with more chronic use. Genital ulcers are often associated with significant genital scarring while leg ulcers can result in a post-thrombotic syndrome. Other treatments for BD flares involve suppressing the immune system with drugs (e.g., cyclosporine or cyclophosphamide). These drugs come with a higher risk of infection, liver and kidney problems, low blood counts and high blood pressure. Finally, anti-inflammatory drugs are also used, including anti-TNF medications. The only approved drug in BD is apremilast, which is used as a maintenance therapy to prevent formation of oral ulcers. Unfortunately, apremilast is associated with both high cost and side effects including diarrhea, nausea, upper respiratory tract infection and headache.
About Soligenix, Inc.
Soligenix is a late-stage biopharmaceutical company focused on developing and commercializing products to treat rare diseases where there is an unmet medical need. Our Specialized BioTherapeutics business segment is developing and moving toward potential commercialization of HyBryte™ (SGX301 or synthetic hypericin sodium) as a novel photodynamic therapy utilizing safe visible light for the treatment of cutaneous T-cell lymphoma (CTCL). With a successful Phase 3 study completed, regulatory approval is being sought and commercialization activities for this product candidate are being advanced initially in the U.S. Development programs in this business segment also include expansion of synthetic hypericin (SGX302) into psoriasis, our first-in-class innate defense regulator (IDR) technology, dusquetide (SGX942) for the treatment of inflammatory diseases, including oral mucositis in head and neck cancer, and (SGX945) in Behçet's Disease. The Company also is developing proprietary formulations of oral beclomethasone 17,21-dipropionate (BDP) for the prevention/treatment of gastrointestinal (GI) disorders characterized by severe inflammation such as pediatric Crohn's disease (SGX203).
Our Public Health Solutions business segment includes active development programs for RiVax®, our ricin toxin vaccine candidate, as well as our vaccine programs targeting filoviruses (such as Marburg and Ebola) and CiVax™, our vaccine candidate for the prevention of COVID-19 (caused by SARS-CoV-2). The development of our vaccine programs incorporates the use of our proprietary heat stabilization platform technology, known as ThermoVax®. To date, this business segment has been supported with government grant and contract funding from the National Institute of Allergy and Infectious Diseases (NIAID), the Defense Threat Reduction Agency (DTRA) and the Biomedical Advanced Research and Development Authority (BARDA).
For further information regarding Soligenix, Inc., please visit the Company's website at https://www.soligeni... and follow us on LinkedIn and Twitter at @Soligenix_Inc.
This press release may contain forward-looking statements that reflect Soligenix, Inc.'s current expectations about its future results, performance, prospects and opportunities, including but not limited to, potential market sizes, patient populations and clinical trial enrollment. Statements that are not historical facts, such as "anticipates," "estimates," "believes," "hopes," "intends," "plans," "expects," "goal," "may," "suggest," "will," "potential," or similar expressions, are forward-looking statements. These statements are subject to a number of risks, uncertainties and other factors that could cause actual events or results in future periods to differ materially from what is expressed in, or implied by, these statements, and include the expected amount and use of proceeds from the offering and the expected closing date of the offering. Soligenix cannot assure you that it will be able to successfully develop, achieve regulatory approval for or commercialize products based on its technologies, particularly in light of the significant uncertainty inherent in developing therapeutics and vaccines against bioterror threats, conducting preclinical and clinical trials of therapeutics and vaccines, obtaining regulatory approvals and manufacturing therapeutics and vaccines, that product development and commercialization efforts will not be reduced or discontinued due to difficulties or delays in clinical trials or due to lack of progress or positive results from research and development efforts, that it will be able to successfully obtain any further funding to support product development and commercialization efforts, including grants and awards, maintain its existing grants which are subject to performance requirements, enter into any biodefense procurement contracts with the U.S. Government or other countries, that it will be able to compete with larger and better financed competitors in the biotechnology industry, that changes in health care practice, third party reimbursement limitations and Federal and/or state health care reform initiatives will not negatively affect its business, or that the U.S. Congress may not pass any legislation that would provide additional funding for the Project BioShield program. In addition, there can be no assurance as to the timing or success of any of its clinical/preclinical trials. Despite the statistically significant result achieved in the HyBryte™ (SGX301) Phase 3 clinical trial for the treatment of cutaneous T-cell lymphoma, there can be no assurance that a marketing authorization from the FDA or EMA will be successful. Notwithstanding the result in the HyBryte™ (SGX301) Phase 3 clinical trial for the treatment of cutaneous T-cell lymphoma and the Phase 2a clinical trial of SGX302 for the treatment of psoriasis, there can be no assurance as to the timing or success of the clinical trials of SGX302 for the treatment of psoriasis. Despite the positive efficacy results demonstrated in the Phase 2 and 3 clinical studies of SGX942 for the treatment of oral mucositis due to chemoradiation therapy for head and neck cancer, there can be no assurance as to the timing or success of the clinical trials of SGX945 for the treatment of Behçet's Disease. Further, there can be no assurance that RiVax® will qualify for a biodefense Priority Review Voucher (PRV) or that the prior sales of PRVs will be indicative of any potential sales price for a PRV for RiVax®. Also, no assurance can be provided that the Company will receive or continue to receive non-dilutive government funding from grants and contracts that have been or may be awarded or for which the Company will apply in the future. These and other risk factors are described from time to time in filings with the Securities and Exchange Commission (the "SEC"), including, but not limited to, the Company's preliminary prospectus (Registration No. 333-271049) filed with the SEC on May 4, 2023, and Soligenix's reports on Forms 10-Q and 10-K. Unless required by law, Soligenix assumes no obligation to update or revise any forward-looking statements as a result of new information or future events.
SOURCE SOLIGENIX, INC.
Copyright (c) 2023 PR Newswire Association,LLC. All Rights Reserved.
SNGX 11/30 confirmation news the real deal?
Or another run up to dilute, r/s, and fail another 2-3 years?
One can only hope..
GLTA
“7:30a ET 11/30/2023 - PR Newswire
Soligenix Receives FDA IND Clearance for Phase 2 Clinical Trial of Dusquetide in the Treatment of Aphthous Ulcers in Beh?et's Disease”
I saw no mention of r/s.
Surprisingly
Read the details of annual meeting,see what is on vote agenda,usually reverse split or other bad dilution when meeting
SNGX low volume sell off? WTH?
No insider dumping,they loading for some reason? Big Accumulation today
Trading natural today,no dilution,accumulation last 5 days
3Xs ave vol,2Xs buy vol,bids/demand growing
The biotech sector is draconian
95 per cent losers
5 per cent long term winners
Crap shoot
Nice work Schaber. Penny for your thoughts.
SNGX now penny stock pre split…
Seemed bit of interest pops this 9m float fast after hours…
Can only hope for good news next week…
GLTA
100% same here!
SNGX Volume very low so fat lady hasn’t sung but definitely warming up…
I've got to quit buying garbage stocks in my IRA. Can't even take the tax loss.
SNGX 50 cent after hours must be leak of ANOTHER reverse split?…
I can’t it’s stuck in 3 of my portfolios !!!
SNGX 7/11/2023: Phase 2a Study in Mild-to-Moderate Psoriasis
Soligenix, Inc., a late-stage biopharmaceutical company focused on developing and commercializing products to treat rare diseases where there is an unmet medical need,announced today the expansion of its Phase 2a trial of SGX302 (synthetic hypericin) for the treatment of mild-to-moderate psoriasis after demonstration of biological effect in the initial five subjects. The study is expected to enroll at least an additional five subjects, exploring the use of SGX302 in the standard of care psoriasis setting, prior to undertaking the larger phase of the study.
https://mma.prnewswire.com/media/1768046/Soligenix_Logo.jpg
Evaluation of the initial five subjects enrolled demonstrated a clear biological signal, with the majority of patients recording an improvement in the PASI (psoriasis area and severity index) score. Enrolling additional patients will enable Soligenix to evaluate the use of SGX302 in the context of accelerated light treatment and additional adjunct treatment.
"Patients with mild-to-moderate disease do not qualify for the use of biologics and have fewer treatment options. The patients enrolled in this study all demonstrated clinical improvement and, with the experience gained in this first portion of the study, we anticipate being able to further improve responses as we move forward," stated Neal Bhatia, MD, Director of Clinical Dermatology at Therapeutics Clinical Research in San Diego and Chair of the Company's Psoriasis MAB. "Having previously participated in the positive Phase 3 clinical trial of HyBryte™ (synthetic hypericin) in the treatment of cutaneous T-cell lymphoma (CTCL), I have had a clinical interest in this novel therapy and am pleased that the Soligenix team is advancing synthetic hypericin in psoriasis where there remains an unmet medical need. Similar to CTCL, psoriasis is a chronic disease where the management of side effects and toxicities is as important as the management of the disease itself. Psoriasis affects more than 7.5 million patients in the U.S. alone, and 60-125 million people worldwide. Having treated psoriasis patients for over 20 years and seen first-hand how they struggle to find good treatment options, access to an additional effective and safe therapy would add significantly to patient care and quality of life for many people that are living with this disease. I look forward to working with my esteemed colleagues to support Soligenix in advancing this important development program forward."
"We are pleased with the initial results from our Phase 2a trial," stated Christopher J. Schaber, PhD, President and Chief Executive Officer of Soligenix. "The expansion of this psoriasis study continues our evaluation of synthetic hypericin into other disease indications, including non-orphan indications, where there remains an unmet medical need. Current estimates show as many as 60-125 million people worldwide living with the condition, with a global treatment market valued at approximately $15 billion in 2020 and projected to reach as much as $40 billion by 2027. The success of HyBryte™ in targeting malignant T-cells during CTCL clinical trials is a promising indicator of the ability of SGX302 to provide a much-needed approach for the treatment of mild-to-moderate psoriasis, also caused by dysregulated T-cells. We anticipate being in a position to report additional SGX302 study data before year-end, as well as the outcome of our ongoing discussions with the FDA regarding a feasible and executable trial design for a confirmatory Phase 3 study with HyBryte™ in the treatment of early stage CTCL."
SNGX sell off 6/20 must be MORE bad news
Wow SNGX now .05 "pre-split"... Schaber trying to kill people or save lives???
Nice work
To think SNGX over $4 few days ago…
GLTA
I only need SNGX 12s !
GLTA
Soligenix Announces Positive Clinical Results from Compatibility Study of HyBryte™ in the Treatment of Cutaneous T-Cell Lymphoma
Results Confirm and Extend Response Results from Phase 3 FLASH Study
PRINCETON, N.J., May 4, 2023 /PRNewswire/ -- Soligenix, Inc. (Nasdaq: SNGX) (Soligenix or the Company), a late-stage biopharmaceutical company focused on developing and commercializing products to treat rare diseases where there is an unmet medical need, announced today positive clinical results from a compatibility study evaluating HyBryte™ (synthetic hypericin sodium) in the treatment of cutaneous T-cell lymphoma (CTCL) using the commercially ready Daavlin Series 7 visible light device, which recently received 510(k) clearance from U.S. Food and Drug Administration (FDA). The open-label study (protocol HPN-CTCL-02) enrolled 9 patients to receive 8 weeks of HyBryte™ treatment of their cancerous lesions, with an assessment of treatment response conducted at week 10 using the Composite Assessment of Index Lesion Severity (CAILS) score. All subjects were enrolled by Brian Poligone, MD, PhD, at the Rochester Skin Lymphoma Medical Group.
The purpose of the study was to establish that any light device capable of producing visible light of an appropriate and consistent wavelength (500 to 650 nm) was suitable for use with HyBryte™ and extend the pharmacokinetic profile using a recently developed, more sensitive hypericin assay. In addition to meeting these objectives, the efficacy demonstrated strongly substantiates the results of the Phase 3 FLASH (Fluorescent Light Activated Synthetic Hypericin) study. The treatment response data of 22% following 8 weeks of twice weekly HyBryte™ therapy recapitulates the results of the FLASH trial, despite the fact that patients in the current study were specifically selected to have more extensive disease consistent with its potential commercial use. Additionally, in this study all patients had improvements in their cumulative CAILS score (average improvement of 36.4%, range 8 to 100%). Results in individual lesions showed that 7 of the 27 index lesions (25.9%) had at least a 50% improvement in their CAILS score and 4 of the 27 index lesions (14.8%) were completely resolved after as little as 8 weeks of treatment. Other key evaluations included measurements of systemic exposure and cardiac output, which yielded extremely low and limited levels of systemic hypericin (plateau concentration of approximately 0.00013 µg/mL) detected in the blood and no observable impact to normal sinus rhythm, reinforcing the safety of HyBryte™.
"We were excited for the opportunity to work with Soligenix and make HyBryte™ available to our patients," stated Brian Poligone, MD, PhD, Director of the Rochester Skin Lymphoma Medical Group, Fairport, NY, and Principal Investigator for the compatibility study and Leading Enrolling Investigator in the FLASH study. "Since the completion of the Phase 3 FLASH study, I have had a number of patients asking about possible access to this promising therapy. Fortunately, we were chosen to conduct this study and the patients' enthusiasm for the product was evident by their willingness to participate in the trial, allowing for its rapid completion. I look forward to continuing to work with Soligenix to further advance the HyBryte™ program so my patients can have this much needed treatment option available to them."
"These results reinforce the positive HyBryte™ data from the FLASH study. Important corporate objectives for the study were to replicate results previously observed in the FLASH study, while using finished drug product manufactured by our proposed commercial contract manufacturer and activated using a commercially viable light device," stated Christopher J. Schaber, PhD, President and Chief Executive Officer of Soligenix. "We look forward to continuing to work with Dr. Poligone and all of our committed clinical investigators to make HyBryte™ available to this underserved orphan patient population."
About HyBryte™
HyBryte™ (research name SGX301) is a novel, first-in-class, photodynamic therapy utilizing safe, visible light for activation. The active ingredient in HyBryte™ is synthetic hypericin, a potent photosensitizer that is topically applied to skin lesions that is taken up by the malignant T-cells, and then activated by visible light approximately 24 hours later. The use of visible light in the red-yellow spectrum has the advantage of penetrating more deeply into the skin (much more so than ultraviolet light) and therefore potentially treating deeper skin disease and thicker plaques and lesions. This treatment approach avoids the risk of secondary malignancies (including melanoma) inherent with the frequently employed DNA-damaging drugs and other phototherapy that are dependent on ultraviolet exposure. Combined with photoactivation, hypericin has demonstrated significant anti-proliferative effects on activated normal human lymphoid cells and inhibited growth of malignant T-cells isolated from CTCL patients. In a published Phase 2 clinical study in CTCL, patients experienced a statistically significant (p=0.04) improvement with topical hypericin treatment whereas the placebo was ineffective. HyBryte™ has received orphan drug and fast track designations from the FDA, as well as orphan designation from the European Medicines Agency (EMA).
The recently published Phase 3 FLASH trial enrolled a total of 169 patients (166 evaluable) with Stage IA, IB or IIA CTCL. The trial consisted of three treatment cycles. Treatments were administered twice weekly for the first 6 weeks and treatment response was determined at the end of the 8th week of each cycle. In the first double-blind treatment cycle, 116 patients received HyBryte™ treatment (0.25% synthetic hypericin) and 50 received placebo treatment of their index lesions. A total of 16% of the patients receiving HyBryte™ achieved at least a 50% reduction in their lesions (graded using a standard measurement of dermatologic lesions, the CAILS score) compared to only 4% of patients in
I think you are the only person in the world who still believes in this company. We might see a rebound to $5, but ....screw it. I may buy enough to get back to even if it hits $5. Talk about adding good money to bad.
OK, I doubled my investment and got 10X as many shares. How ridiculous is this?
Of course I shouldn't have done a market order. I just keep getting dumber and dumber by being here.
SNGX from other board: "BARDA Contract Award"
"BARDA was expected (I thought) to release news on the contract proposals for Sudan-Ebolavirus and Marburg Virus very soon, if not already. Although monovalent vaccines are of primary focus per BARDA, Soligenix's bivalent SuVax and MarVax vaccine have proven to give 100% protection, and relatively quickly, which is ultimately what BARDA claims they're after.
Does anyone have any insight into this decision and if/when contract awards will be announced?"
Any others seen / heard of this potential upcoming news ?
GLTA
Nothing, absolutely nothing.
More delays. And with such a small float now the drama can be overwhelming.
WTH today SNGX ???!!!
I don't think date for that meeting was yet confirmed?
Waiting to see how the meeting goes with the FDA.
When was the reverse split here? Did I miss it?
Now I see it, looks like 1-15. Didn't think this stock could get any worse.
The HUGE news has caused the stock to raise by 2 cents. TWO CENTS!!
Good thing I never put much money here, I'm only down by 93%.
SNGX 4/05/2023 receives approval for HyBryte...
- "Soligenix Receives USAN Approval for "Hypericin Sodium" as Nonproprietary Name for Novel Active Ingredient in HyBryte™ and SGX302. 7:30 am ET April 5, 2023 (PR Newswire).
Soligenix, Inc. (Nasdaq: SNGX) (Soligenix or the Company), a late-stage biopharmaceutical company focused on developing and commercializing products to treat rare diseases where there is an unmet medical need, announced today that the United States Adopted Names (USAN) Council has approved the use of the nonproprietary name of "hypericin sodium" for the novel active ingredient in both HyBryte™ (research name SGX301) for the treatment of cutaneous T-cell lymphoma (CTCL)and SGX302 for the treatment of mild-to-moderate psoriasis.
- "We are pleased that USAN has approved the proposed name," statedChristopher J. Schaber, PhD, President and Chief Executive Officer ofSoligenix. "We look forward to continuing to work with the World Health Organization (WHO) to advance the International Nonproprietary Name (INN) hypericin from a proposed INN to a recommended INN, which is expected to occur later this year."
- Information on hypericin sodium will be posted on the USAN website (www.ama-assn.org/go/usan) before the end of 2023 and will be submitted to the U.S. Pharmacopeial Convention for publication in the U.S. Pharmacopeia Dictionary of USAN and International Drug Names.
About USAN
The USAN Council serves health professionals in the U.S. by selecting simple, informative, and unique nonproprietary names for drugs by establishing logical nomenclature classifications based on pharmacological and/or chemical relationships to ensure that drug information is communicated accurately and unambiguously. The USAN Council aims for global standardization and unification of drug nomenclature by working closely with the International Nonproprietary Name Program of WHO and various national nomenclature groups.
About Synthetic Hypericin Sodium
Visible light-activated synthetic hypericin sodium is a novel, first-in-class, photodynamic therapy (PDT) that is expected to avoid much of the long-term risks associated with other PDT treatments. Synthetic hypericin sodium is a potent photosensitizer that is topically applied to skin lesions and taken up by cutaneous T-cells. With subsequent activation by safe, visible light, T-cell apoptosis is induced, addressing the root cause of both CTCL and psoriasis lesions. Other PDTs have shown efficacy in psoriasis with a similar apoptotic mechanism, albeit using ultraviolet (UV) light associated with more severe potential long-term safety concerns. The use of visible light in the red-yellow spectrum has the advantage of deeper penetration into the skin (much more than UV light) potentially treating deeper skin disease and thicker plaques and lesions, similar to what was observed in previous clinical trials.
- This treatment approach avoids the risk of secondary malignancies (including melanoma) inherent with both the frequently used DNA-damaging drugs and other phototherapies that are dependent on UV A or B exposure. The use of synthetic hypericin sodium coupled with safe, visible light also avoids the risk of serious infections and cancer associated with the systemic immunosuppressive treatments used in CTCL and psoriasis.
- In a published Phase 1/2 proof of concept clinical study using synthetic hypericin sodium, efficacy was demonstrated in patients with CTCL (58.3% response, p=0.04) as well as psoriasis (80% response, p<0.02).
- The recently published Phase 3 FLASH (Fluorescent Light Activated Synthetic Hypericin)studyenrolled a total of 169 patients (166 evaluable) with Stage IA, IB or IIA CTCL. The trial consisted of three treatment cycles. Treatments were administered twice weekly in 6-week cycles. In the first double-blind treatment cycle, 116 patients received HyBryte™ (the tradename used in CTCL) treatment and 50 received placebo treatment of their index lesions. A total of 16% of the patients receiving HyBryte™ achieved at least a 50% reduction in their lesions (using the standard Composite Assessment of Index Lesions Severity [CAILS] score) compared to only 4% of patients in the placebo group after just 6 weeks of treatment (p=0.04). Further treatment with HyBryte™ increased the number of treatment successes to 40% and 49% after 12 and 18 weeks, respectively (p<0.0001 for both). Additional analyses also indicated that HyBryte™ is equally effective in treating both plaque (42% treatment response rate after 12 weeks treatment, p<0.0001 relative to placebo treatment in Cycle 1) and patch (37%, p=0.0009) lesions of CTCL, a particularly relevant finding given the historical difficulty in treating plaque lesions. This is also relevant to psoriasis where the lesions can be thicker than the patches observed in CTCL.
In a subset of patients evaluated during their third treatment cycle, it was demonstrated that HyBryte™ is not systemically available, consistent with the general safety of this topical product observed to date. At the end of Cycle 3, HyBryte™ continued to be well tolerated despite extended and increased use of the product to treat multiple lesions.
- A HyBryte™ new drug application (NDA) has been submitted for the treatment of CTCL with the U.S. Food and Drug Administration (FDA). It is currently the subject of an FDA Refusal to File(RTF) letter, as upon preliminary assessment, the FDA determined that it was not sufficiently complete to permit substantive review. A Type A meeting with the FDA has been scheduled to gain further clarity and to respond to the issues identified in the RTF letter, as well as to seek additional guidance concerning information that the agency would require for a resubmitted NDA to be deemed acceptable.
- SGX302 (synthetic hypericin sodium) is currently being evaluated in a Phase 2a clinical trial targeting enrollment of up to 42 patients ages 18 years or older with mild to moderate, stable psoriasis covering 2 to 30% of their body. Patients will undergo treatments for a total of 18 weeks and, on completion, will be followed for a 4-week follow-up period in which patients will not receive other psoriasis treatments. The study is divided into two parts. In Part A, 5-10 patients will be assigned open-label SGX302 (0.25% hypericin) at the time of enrollment. Once the tolerability and response to SGX302 has been established, Part B of the protocol will commence. In Part B, patients will be randomized to double-blind treatment groups at a ratio 1:1 of active drug to placebo ointment."
GLTA
I never recommend penny stocks as a smart investment, but, if you are a short time trader and realize you're just trying to beat the flippers at their own game (for all I know, you may be one), I can see the stock making a short term spurt to $7+
Someone willing to sell a FEW every day…
SNGX likely trade under 20k shares again today
|
Followers
|
208
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
9747
|
|
Created
|
06/06/06
|
Type
|
Free
|
| Moderators | |||
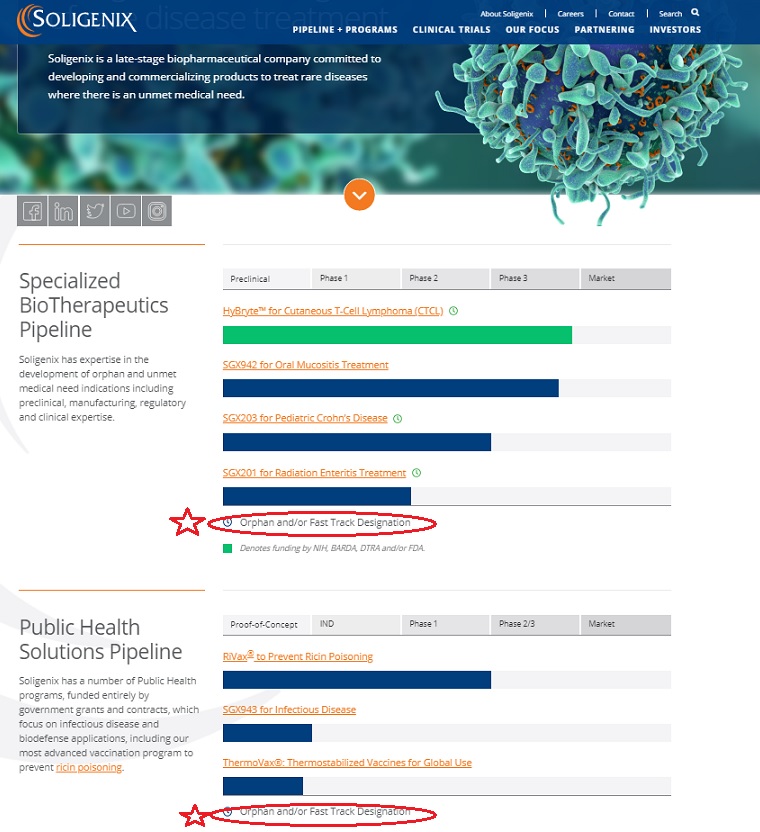
| SNGX [NASD] |
| Soligenix, Inc. |
| Healthcare | Biotechnology | USA |
Soligenix, Inc. (Soligenix) is a late-stage biopharmaceutical company.
The Company is focused on developing and commercializing products to treat rare diseases.
The Company operates through two business segments:
Specialized BioTherapeutics and Public Health Solution. Soligenix’s Specialized BioTherapeutics business segment is developing
a photodynamic therapy (SGX301) utilizing topical synthetic hypericin activated with safe visible fluorescent light for the treatment of cutaneous T-cell lymphoma (CTCL).
The Public Health Solutions business segment includes active development programs for RiVax,
its ricin toxin vaccine candidate and SGX943, its therapeutic candidate for antibiotic-resistant and emerging infectious disease.
CTCL is its innate defense regulator technology, dusquetide (SGX942) for the treatment of oral mucositis in head and neck cancer.
Soligenix has expertise in the development of orphan and unmet medical need indications including preclinical, manufacturing, regulatory and clinical expertise.
Soligenix has expertise in the development of orphan and unmet medical need indications including preclinical, manufacturing, regulatory and clinical expertise.
| Preclinical | Phase 1 | Phase 2 | Phase 3 | Market |
|---|---|---|---|---|
| HyBryte™ for Cutaneous T-Cell Lymphoma (CTCL) | ||||
| | | | | |
| SGX942 for Oral Mucositis Treatment | ||||
| | | | | |
| SGX203 for Pediatric Crohn’s Disease | ||||
| | | | | |
| SGX201 for Radiation Enteritis Treatment | ||||
| | | | | |
Orphan and/or Fast Track Designation
Soligenix has a number of Public Health programs, funded entirely by government grants and contracts, which focus on infectious disease and biodefense applications, including our most advanced vaccination program to prevent ricin poisoning.
| Proof-of-Concept | IND | Phase 1 | Phase 2/3 | Market |
|---|---|---|---|---|
| RiVax® to Prevent Ricin Poisoning | ||||
| | | | | |
| SGX943 for Infectious Disease | ||||
| | | | | |
| ThermoVax®: Thermostabilized Vaccines for Global Use | ||||
| | | | | |
Orphan and/or Fast Track Designation


| Preclinical | Phase 1 | Phase 2 | Phase 3 | Market |
|---|---|---|---|---|
| HyBryte™ for Cutaneous T-Cell Lymphoma (CTCL) almost done [in phase 3] | ||||
| | | | | |
| SGX942 for Oral Mucositis Treatment almost done [in phase 3] | ||||
| | | | | |
| SGX203 for Pediatric Crohn’s Disease [phase 2 almost done] | ||||
| | | | | |
| SGX201 for Radiation Enteritis Treatment [phase 1 going into phase 2] | ||||
| Proof-of-Concept | IND | Phase 1 | Phase 2/3 | Market |
|---|---|---|---|---|
| RiVax® to Prevent Ricin Poisoning | ||||
| | | | | |
| SGX943 for Infectious Disease | ||||
| | | | | |
| ThermoVax®: Thermostabilized Vaccines for Global Use | ||||
| | | | | |
Orphan and/or Fast Track Designation
| PER IHUB MGMT |
02-07-2021
DISCLAIMER: ONLY FOR MICK
https://investorshub.advfn.com/boards/profilea.aspx?user=1012
*The Board Monitor and herewithin , are not licensed brokers and assume NO responsibility for actions,
investments,decisions, or messages posted on this forum.
CONTENT ON THIS FORUM SHOULD NOT BE CONSIDERED ADVISORY NOR SOLICITATION
AUTHORS MAY HAVE BUYS OR SELLS WITH THE COMPANIES MENTIONED IN TRADING POSTERS SHOULD DUE DILIGENT BUYING OR SELLING.
ALL POSTING SHOULD BE CONSIDERED FOR INFORMATION ONLY. WE DO NOT RECOMMEND ANYONE BUY OR SELL ANY SECURITIES POSTED HEREWITHIN.
ANY trade entered into risks the possibility of losing the funds invested.
• There are no guarantees when buying or selling any security.Any
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
