Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Lots of talk but no one seems to really know what is going on. I guess things are happening. McCoy has his Consulting firm and they are linked up with Life Sciences in NZ. They are legit and they offer many more things besides the collagen. That's where John Weber is, so he is still involved. Looks like hooked up with Meta Alliance, They are a firm that gets involved in financing as well as other things. So that's the group putting or trying to get funding together.
I guess when he has something definite we will here something but the right people seemed to be involved from various industries. Seems strange that he was not making any announcements all year about the McCoy consulting firm and other discussions. Maybe wants to get it all together no one knows.
As far as a stock pop, I see someone saying $3-$6. That has about a .001% chance. There are 154M shares outstanding, so its not going to be a 500M -$1B company on a little financing. I would like to see $50 - $100M market cap so maybe .30 to .60 cents at first then see if they get going and move forward.
Good luck all....I just wouldn't be short, can make .13 cents but lose much more.
Things are picking up the volume sure is, Maybe a early X mas present from McCoy. I wonder if his red suit still fits.
Good looking day! I love the volume more than the price. The lack of a communication still bothers me a lot. Does anyone think the release is ready and what we are seeing are the crumbs being floated out there? If so it should be good when it hits. Its hard to see a downside coming after the last week...imo
Don't say you weren't warned. Got a cousin in NZ, news for McCoy ain't good down there. Or anywhere else for that matter.
LOL oh ya!?
Im just going by what has been touted by Nicole, she seems to feel we'll get news anyday now BEFORE the end of the year. We are due for some kind of update. The other co. does regular updates on Twitter laying out the game plan for everyone to see.
McCoy must be old school, probably still has a flip phone from the 90's
will pop first then these guys some time next week
© 2017 InvestorsHub.com, Inc.
Ya the company has been pretty quiet while their lining up their ducks. Hush Hush FDA trials stuff. Isolated heard for Collagen stuff, funding etc yada yada yada. Its all suppose to come togther before years end.
I recall a while back they announced receiving some kind of FDA preliminary approvals the sp popped to .64 cents before being shot down because of suggestive wording in the PR.
INTV will pop first then these guys some time next week
Guestimate is $3.00-$6.00
Zero volume? Not the best advertisement.
Guestimate is $3.00-$6.00
What kind of move are you anticipating here?
I don't get some of the folks on this board. One month slamming company as a "Pump & Dump" then the next month the exact opposite.
So when the stock hits big, are we going to have a chorus of "I told you all along" from all the naysayers and flip-floppers?
It's a penny stock... it's a gamble... if you are so unhappy just cash out and take your negative and useless re-hashing of speculation/nonsense with you.
Seems I may need to support this. Deep look checks out fine. Looks as if more diligence to advancing product vs. filing paperwork may be the result.
Still doesn't excuse late filings and lack of shareholder information.
Price holding and activity sequence look to be some type of announcement coming after Jan 1st or the week after.
I'd be looking for mid week announce for heaviest exposure.
We'll see.
Someone knows something, probably the lawyers proof reading McCoys PR's. They always have the inside scoop.
Volume the best its been in a long time...still see the manipulation going on. With this volume one can only assume someone is loading up thinking that some positive news is on the immediate horizon.
we'll see, the daily volume has a little uptick. I'd like to see RGIN's news launch them like INTV's news has, then it will be a Very nice Holiday
RGIN is due news -IND-BOOM..TICK, TICK
I would like to see RGIN explode upward like INTV did today. Does McCoy tweet?
We have minimul volume keeping RGIN in this range
We have minimul volume keeping RGIN in this range. Is it a stall tactic until McCoy announces the goods? Someone has been buying.
What would be the reason to bounce this thing between .086 and .09? Looks to me like someone is buying it at the .086 so it does not tank all the way. Hard to have faith in Mr Mccoy when he has put no news out in a year where his goal was to be in clinical trials. He cant even file his other papers in a timely fashion. I hope Im wrong but this is not looking good imo.
Nicole...we get the whole.."It takes time" concept. Well thought out release on evidence of progress is fundamental to keeping the trust.
It’s coming, have some patience Mr McCoy will get things done very very quickly
2+ weeks till New Years. Is McCoy going to give investors something to celebrate? At this juncture its not looking promising.
Ho Oh NO
Changes to FDA trials protocols are certainly a factor here. Required resources to deal w/FDA must be a factor in any planning. Recent sign of trading life are positive, IMO. Note, all BIO are going through the same thing w/FDA new rules.
Theres been little movement here for over a year now. Randy promised updates last year and hasnt delivered. The filings are late with no NT 10k announcement makes retail wonder whats really going on.
There has been a bid in for .086 for the last couple of days. It was originally for around 150k shares...down to about 83k shares now. It looks like someone is manipulating this to keep it at current price (bounce between .086 and .09) I would think this is a bad thing...without the manipulation where would we be? I am losing faith daily in this stock but dont want to be that guy who sells and it takes off. Until news comes out I know we are stuck but does anyone out there have any positive info that can keep me going? lol
RGIN's 3rd Quarter 10Q is overdue No?
There hasnt bee any updates by RGIN in almost a year. Maybe the touts info is accurate and Randy has accomplished something? The volume indicates something is up.
Noticed different pattern this week after years w/little signs of life. Who knows when this gets good news but it could move fast when it does after sooo long being weak. Good to see.
Huge volume today...237000 traded. Could this be the start of the run? Any info greatly appreciated
Volume thin but I liked the way it started and was OK w/day. We need to see now if new FDA trials protocol will matter here. It should, IMO, if this real.
NEWS must be imminent, LOL look at that Volume, woooo whoooo Regenicin is flying!
Nicole, I am a believer or would not be here. I trust that the technology could be effective. News on IND would help. I already own a lot.
Still awaiting some kind, any kind, of evidence. I'm long anyway but getting bored.
Nidan, we all need to support the stock now or never , if we kept on buying ,keeping positive, believing and have trust in this life saving technology , they are working very very hard in last 4 months to keep the company going board members put there life saving, no we need to support the stock before the news breaks , so there will be no shorts out there and not enough stock for day traders to munipilate the stock , so let’s all stand strong and buy so we can get this stock up to 20 cents before the news comes out, people at his means a lot trust me .
Funding as we speak IND hope for this stock to hit huge
Funding as we speak IND hope for this stock to hit huge
Regenicin is goig to blow out of the sky ??
Santa Clause is Coming very very soon
Maybe it's a good time for SEC to take a peekaboo. FDA pushes paperwork as I said before. And they are among the best at it. That being said it's easy enough for RGIN / Randall McCoy. To generate the correct amount of paperwork and mirrors to just drag this to exactly where it's at. Nowhere.
This guy's entire profile if carefully looked into is as basically a college professor. Which in and of itself is a joke.
NO. Real world experience. These people are born in an aspirin encapsulated world and never leave there.
The entire theory is outdated and being far surpassed by MIT and other solutions that are miles ahead of this
This guy has never been out of tenure and never will be . He's almost 70. It ain't happening.
RGIN is and always has been smoke and mirrors.
I'll get in touch with an old friend at Treasury see what's really going on. Enough benefit of the doubt has been given here I believe.
Simple write off / Loss in Jan 18 forms on 2017 utter failure.
Its been 3 weeks since this post and NOT a WORD from RGIN and all of their spectacular accomplishments. Just like when Randy promised a update last December, Nadda from Regenicin. I bet ya he has no problems feeding his face at Applebee's while shareholders get crumbs.
Like I said before. There just may be nothing to this at all anymore. Simple wisps of smoke after the fire.
Without any substantial statements on the company's stability or recent movement on going forward.
The 2000 trade was just a simple 130 dollar electric bill for Randy.
Lonza was the outcome that was hoped for from the start. Loose ends like that aren't missteps. Calculation.
It's just little Piggy Bank now. At least as long as it stays semi viable.
Ya big news coming soon... Ha ha .
Pretty sad that a 2000 share trade takes RGIN down .02 cents when its struggling to stay afloat. No news is NO NEWS
Old News Is Good News , They are working very hard for this life saving technology, people have some patience don’t be so greedy, IND on the way , Funding , Collegen New Zeland News , this a a very reputable company that will make it all the way through, Regenicin Skin is one of a kind plus 5 other patents that go along with it, News is coming
Old news with NO follow up is OLD news. A lot has happened since those days I just hope its not proven that McCoy like others is a THIEF. Sometimes that isnt proven until the backstabbing is done.
|
Followers
|
71
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
2931
|
|
Created
|
08/06/10
|
Type
|
Free
|
| Moderators | |||
Regenicin, Inc.
10 High Court
Little Falls, NJ 07424
**updated and correct phone number** (973) 557-8914
they filed this on aug 22 2016- http://www.otcmarkets.com/edgar/GetFilingHtml?FilingID=11558959
| Name: | Randall McCoy |
| Title: | Principal Executive Officer and Director |
| Date: | August 22, 2016 |
| Name: | John J. Weber |
| Title: | Principal Financial Officer and Director |
| Date: | August 22, 2016 |


LITTLE FALLS, N.J., June 12, 2012 /PRNewswire/ -- Regenicin, Inc. (OTC Bulletin Board: RGIN) a biotechnology company specializing in the development of and commercialization of regenerative cell therapies to restore the health of damaged tissues and organs, announced today that the Food and Drug Administration (FDA) has granted Orphan Status approval for the PermaDerm® product, the only tissue-engineered skin prepared from autologous (patient's own) skin cells consisting of both epidermal and dermal layers, that is indicated for catastrophic burn patients. The FDA had previously designated PermaDerm® to be a biological/drug (permanent skin replacement) not a medical device (temporary skin replacement). PermaDerm® is the only skin replacement technology to receive this Biological/drug designation.
COMPANY HIGHLIGHTS:
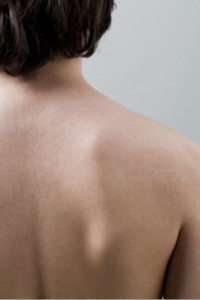
PermaDerm™ is being developed to be the only tissue-engineered skin prepared from autologous (patient's own) skin cells consisting of both epidermal and dermal layers. A small harvested section of the patient's own skin can be grown to graft an area one hundred times its size in as little as thirty days.
These living, self-to-self skin graft tissue are intended to form permanent skin tissue that will not be rejected by the immune system of the patient, a critical possibility in porcine or cadaver skin grafts used today.
The technology has been clinically tested in over 150 pediatric, catastrophic burn patients. Currently Regenicin is working with its contract manufacturer to prepare for pre-market approval of PermaDerm™ from the FDA.
PermaDerm™ is being designed to save lives, reduce healthcare costs by decreasing the patient's stay in the Critical Care Unit and reduce the need for additional surgeries. An insurance company procedural code has been approved for reimbursement of costs to hospitals. The American Medical Association has assigned CPT (Current Procedural Terminology) code for cultured skin substitutes under the dermal substitute category which enables insurance companies to process and hospitals to be reimbursed for cultured skin substitutes once approved by the FDA.
According to the American Burn Association, there are currently over 2,000 cases annually involving burns covering over 50% of the patient's total body surface area.
PermaDerm™ is being developed for the treatment of catastrophic, full-thickness burns covering over 50% of the total body surface area.
PermaDerm™ has been used in clinical trials on pediatric patients with catastrophic burns covering from 50 to 90+% of their body.
PermaDerm™ is being developed to require less donor skin from the patient's own body, thus needing fewer surgeries to harvest skin. The result is expected to be less scarring, with fewer infections and faster healing.
Randall McCoy
Chief Executive Officer
Randall McCoy has more than 37 years of experience in the healthcare industry. His experience includes executive positions and scientific research and development. His previous employment includes George Washington University, Temple University Medical School and RCA/SRI International (Director of Life Sciences and Electronic Displays). Mr. McCoy has assisted both small and major pharmaceutical/device companies address FDA issues. He has also helped over 225 foreign and domestic companies introduce their FDA regulated drug and medical device products into the US and World market. Mr. McCoy currently has over 30 Patents US and International.
Changed to AMBS
Dr. Joseph Rubinfeld Ph.D
Director at CytRX; Initial Co-Founder of Amgen, Inc.
Dr. Joseph Rubinfeld co-founded SuperGen, Inc. in 1991 and has served as its Chief Executive Officer and President, Chief Scientific Officer and as a director. He has been a director of CytRX since July 2002. Dr. Rubinfeld is also a founder of JJ Pharma. Dr. Rubinfeld was one of the four initial founders of Amgen, Inc. in 1980 and served as a Vice President and its Chief of Operations until 1983. From 1987 until 1990, Dr. Rubinfeld was a Senior Director at Cetus Corporation and from 1968 to 1980, Dr. Rubinfeld was employed at Bristol-Myers Company. Dr. Rubinfeld received a B.S. degree in chemistry from C.C.N.Y. and an M.A. and Ph.D in chemistry from Columbia University.
John Weber
Regenicin™'s Interim CFO
John Weber most recently served as Executive Vice President of Fujifilm USA, the highest ranked American corporate officer, from 2006 until his departure in 2009. His responsibilities included overseeing all corporate activity with the exception of R&D. During that time Fujifilm was ranked as the fastest growing medical imaging company, consistently ranking #1 or #2 in customer satisfaction. From 1998 through 2006 he served as Senior Vice President, Operations at Fujifilm USA where he spearheaded the transition of the company from a film distributor to a digital medical informatics company. From 1986 until 1998 he served as CFO where he helped to profitably manage Fuji's growth from an employee base of 75 to over 1,000. Prior to his distinguished career at Fujifilm USA, Mr. Weber served as the CFO for the confectionary and drinks division of Cadbury Schweppes Limited for three years and as Corporate Controller for an additional five years.
Dr. Craig Eagle
Pfizer Oncology
Dr. Craig Eagle joined Pfizer Australia in 2001 as part of the medical group. In Australia, his role involved leading and participating in scientific research, regulatory and pricing & re-imbursement negotiations for compounds in therapeutic areas including oncology, anti-infectives, respiratory, arthritis and pain management. In 2003, Pfizer relocated Dr. Eagle to the United States where he was appointed as the worldwide lead for development of Celecoxib in oncology to oversee the global research program. Since that time he has had increasing responsibility for overseeing the global research plans and teams for Irinotecan and Dalteparin. In 2007, he became head of Medical Affairs and Outcomes Research for Pfizer, including the US oncology business. Dr. Eagle has led, or been directly involved with, teams that resulted in eight new products or indications. As part of his current role at Pfizer, he has led the integration of the Pfizer/Wyeth oncology businesses and portfolio.
The Broadsmoore Group, LLC 9,223,770(8)
560 Lexington Ave., 16 th Fl.
New York, NY 10022
PDA Associates LLC 7,770,000(9)
560 Lexington Ave., 16 th Fl.
New York, NY 10022
Officers, directors and 5 percent shareholders collectively 54,508,099(10) 59.74%
* Less than 1%
(1) Unless otherwise indicated, each person or entity named in the table has sole voting power and investment power (or shares that power with that person's spouse) with respect to all shares of common stock listed as owned by that person or entity.
(2) A total of 83,807,964 shares of the Company's common stock are considered to be outstanding pursuant to Rule 13d-3(d)(1) under the Securities Exchange Act of 1934.
(3) Includes 32,821,641 shares of common stock held in his name and options to purchase 885,672 shares of common stock.
(4) Includes 50,000 shares of common stock held in his name and options to purchase 885,672 shares of common stock,
(5) Includes 1,050,000 shares of common stock held in his name and options to purchase 885,672 shares of common stock
(6) Includes 50,000 shares of common stock held in his name and options to purchase 885,672 shares of common stock,
(7) Includes 33,971,641 shares of common stock, options to purchase 3,542,688 shares of common stock and shares of Series A Convertible Preferred Stock convertible into 3,350,000 shares of common stock,
(8) Includes 5,706,270 shares of common stock held in its name, warrants to purchase 167,500 shares of common stock and shares of Series A Convertible Preferred Stock convertible into 3,350,000 shares of common stock,
(9) Includes 7,400,000 shares of common stock held in its name and warrants to purchase 370,000 shares of common stock,
(10) Includes 47,077,911 shares of common stock, warrants to purchase 537,500 shares of common stock, options to purchase 3,542,688 shares of common stock and shares of Series A Convertible Preferred Stock convertible into 3,350,000 shares of common stock
http://ih.advfn.com/p.php?pid=nmona&article=5144097
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
