Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
http://www.biotuesdays.com/features/2015/6/30/regenicin-readying-clinical-trials-of-novaderm-skin-substitute it all states clearly just believe in this life saving technology it right here , great things happens when people work hard and put all there effort, things don’t happen overnight, now just sit back and wait it’s coming up very very soon be patient
Type in Novaderm, it comes back to this
https://www.accessdata.fda.gov/scripts/opdlisting/oopd/
Todays action for a Friday was 100k in buys the 90k in sells. Most days RGIN doesnt trade anything at all so possibly next week we'll hear what Nicole has been clammering about? I know I havent been able to find any information regarding Regenicin
Big Action Coming
The short answer is, no. Any real, fact based discussion has been hard to come by. RGIN looks like it is real but could be a mirage. Remain hopeful for sake of pts and my interests.
Were you able to source RGIN's FDA calendar?, I saw them on it from one of Nicoles links but havent seen them mentioned there since.
Somethings coming alright, 1260 shares yesterday, 11,000 today!
whooo hooo are we living large yet!?
https://www.fda.gov/downloads/BiologicsBloodVaccines/GuidanceComplianceRegulatoryInformation/Guidances/CellularandGeneTherapy/UCM585403.pdf
The new FDA guidance will certainly help w/RGIN product introduction processes and trials
60,000 shares dumped today after a week of thin trades if any, is just showing no support for McCoys story.
Ive been looking for FDA anything regarding RGIN and Im unable to find a source. I did find a picture of McCoy doing a ceremony of some kind but that isnt going to help anyone suffering from burns.
If RGIN news ever arrives I'll be pleasantly surprised.
3 trades today ALL sells
https://ih.advfn.com/stock-market/USOTC/regenicin-inc-pc-RGIN/trades
Im sure you thought the cheerleading would have a different affect
Even believers need evidence....ASAP would be good.
Road To Riches for all believers that waited so long for this incredible Life Saving Technology
I'm completely open if a 10Q is on time and definitive.
But I would like to have some type of clarification of how the things moved so far away... New Zealand? Really seems much harder and more logistically problematic.
Someone update the website.
Someone give some sense of clarity and direction.
Don't come back and tell me anyone or Randy is far too busy.
Not anymore. Doesn't jive
Based soley on the last 10Q in August, RGIN should have a quaterly report due next week.
https://seekingalpha.com/filing/3638091
This is not Randy land my friend. This is RGIN. It's a traded company. Not a pastime. Unless it's to dilute the company and walk away with the lonza dough and anything else scrapped up from the next few months. Keep selling..... Someone will keep buying . 500-600 a day adds up quick.
Maddof. ...... All day long. This things like a piggy bank.
Where does the FDA appointment stand that was this week supposedly......
Crickets.....,. thanks
Nicole,
Please read me a bedtime story. Something good, like how rich I am going to be when I wake up. Pretty please with money on top!
Its a interesting article one which Novaderm or what ever its being called these days can help with. Regenicin has been fighting a up hill battle since the Lonza litigation days. From what Ive seen McCoy has been working diligently to get his product to market. I would prefer to learn of his accomplishments via company announcements and not through hearsay. I dont know the man or his team but they all appear to be credible people. Im not seeing typical penny stock pumping or hype, no fake news articles, no dilution to speak of, no shorting either. If one believes Nicole1128's loose insights then adding to this position at these levels is the way to go. I would hold pat and see what develops.
Today 6 trades totaling 25,000 shares exchanged hands. Someone other than me is buying. That in itself is a good sign.
Lets see how things in Randy land unfold. Someone seems to think big news is about to arrive.
RGIN - 10:18:03
Bought 5000s @ $0.086
Scottrader. I have a position in Rgin for quite some time and I am continuing to hold and hope. I just saw this article in the WSJ I am a novice investor and Im not sure how to feel about the article. What do you think. TY
https://www.wsj.com/articles/new-skin-for-7-year-old-boy-marks-advance-in-gene-therapy-1510164025
1 more point: from someone who has supposedly done this for the last 37 years.... Per website. To not know what the FDA is going to be requesting before any trails or formal testing can begin is an oversight that can't be ignored. McCoy would be well versed in the obstacles in the system within and how to be prepared for them with 5-7 years starting his career . As any projects he's supposed to have worked on have surely encountered regulations and testing surety. This is being sold as a complete unknown. Which is nonsense. Your not in a field of work or professional occupation without even briefly understanding or compreshension of the clear path. Hiccups very rarely occur. There anticipated and worked through. There is no patent. No product. No business model. No pre sales chain. If this was going to go. For me I would have a far better plan outlayed with people in each expertise level. Ready to go and signed contracts. I mean 10 M upcoming could easily secure top field talent. No
mention of that anywhere at anytime. Just going for more dollars on the tick
and pump. That hopefully goes up for large holders to sell and bail just a bit more of worthless holdings.
Not to mention . There is no longer a functional BOD. I just checked the website. Seems John Webber is CFO of a company that has no positive cash flow. No sales. Involved in a company that was involved in it's own law suit and stripped of it rights. His last claim to fame was for attempting to screw Fuji LTD out of retirement benefits. Every one else has disappeared of the BOD list. But amazingly squeaks out info about FDA this and FDA that seem to have popped up. Super disappointed
Consistent late filings. No news from this company ever. It's a joke. No professional ism on driving the business other than trying to raise money and pump stock price. Not how businesses are built. Dumping dollars onto a non existent business model with no employee base. No corporate structure. I don't see anyone in correct state of mind putting 10M in funding to anything in this state. For this product. 10M doesn't even begin to be able to fund the resources, technical,clinical,legal,office and material resources needed. Pipe dream Nicole
Here is the last 10Q, please, point out EVERYTHING thats been posted about and what page THAT info is located;
http://www.regenicin.com/Investor-Relations/SEC-Filings/documents/2017/0808/rgin10q.htm
the Only reference to "funding" is they dont have any and NO mention at all about any patent study or New Zealand so WHERE is this information coming from if its NOT in the financials? has to be Insider info
and its Collagen, not Collegen according to McCoy
There is no insider information, last 10q states everything what’s happening, so now it’s time for great things to happen, we all no about Regenicin was working for a long time to raise Moneys , New Zeland Collegen , IND and 10 patient study for path to success, everything is written in black and white . It’s common sense Regenicin is going to sell collegen to others so they can bring money in ,plus to use it on burn patients , IND application was put awhile ago ,so it’s time for great news to come , Mr. McCoy is no Bernie my friend like some posted he’s a hard working individual he had 150 approvals in his lifetime and cares about this project,it’s not about money to him , his has to much pride ,he’s a gift from God with lots of intelligence.
Knowing insider information is not a crime.Trading based on insider information is a crime.
If there's material change then company has 5days or so to make it public.I don't remember exact days.If materail change doesn't become public information then company is in trouble with SEC.Never heard of a CEO fined because someone else made insider information public.
Big question is do you believe what's posted on a message board?
Its Alllll insider information that will land McCoy in HOT water once the news that has been posted about is released. Sometimes over excited friends or family close to the company show up to vent their enthusiams. But they dont realize the damage that can be caused to a little struggling company. Breakdown everything that has been posted. There are 4-5 items being announced according to source. If they get announced as stated the messenger is going to JAIL and the CEO will be fined by FINRA and the SEC. Remember Martha Stewart got some inside info then traded on it, she bought a Felony. There are a LOT of regulatory eyes monitoring these boards. Any PR's released by McCoy about the posted subject matter and someone suffering from Big Mouth syndrome will have SEC investigators knocking on the door.
What are you talking about? How do you know this information. Their is no research that will give anyone the data you espouse in your post. I truly want to believe you, however, there is nothing you are presenting that has any truth. How is it you are the only person on the face of the planet can find this information?
New Zeland , Funding, IND , FDA,all great and positive news to come ,you seem not to believe in anything, why would Regenicin go this far with alll the investment McCoy has put his life saving into this project with family friends and investors Randy all along said he will not let this project go I’m sure you will see shortly all the positive and prospective things RGIN has in hand , like I said this is the only life saving technology that FDA said all along they have never see anything like this , once the Skin is approved there are 5 other patents that go along with this , this my friend is a score , Good things happen to those that have be patient . I believe this week is the funding after IND Then the next big thing Regenicin will start selling collegen to major companies like Integra, J&J and some many others,with the company will start bringing money , ten Patient Study for the skin project and FDA approval my friends Do your homework God Bless to all .......
There's simply just no there . There. Somebody's having a bang up time in New Zealand.
What investment relations ? There's no company. Far too much time has past with no comment and or communication. That's not how it works
Contact RGIN to see if your child can become Investor Relations
Kinda like having a dentist appointment. Hasn't there been reports of misleading trials and inappropriate methods with this already? Yes there has. My 10 year old could file forms and forge any of what's being put forth. FDA gives guidelines to meet. Now just fill it out to their delight.
The only verification I could come up with was Regenicin being on the FDA's calendar. That accomplishment isnt performed by a part time anything. Everything else touted remains to be seen. If McCoy comes through, its happy days for everyone involved. Volumes picking up. If not,
Starting to look like a simple Ponzi account. Sure file paperwork with FDA. Aplply for this. Apply for that. All looks above board. Or so you think. Whose selling shares and when? Couple thousand in pocket cash. This is looking like a part time job for Randy. Maddof did much better. But same idea
I hope shes not crying wolf because so far none of whats been touted is coming true. I suppose the FDA thing will take a few business days to be clarified. I did look up Regenicin and they are on the FDA's calendar for last week, so we'll see if the pied piper is pie eyed or factual
Regenicin will definitely hit the $5 dollar range in no time , there is some much news coming back to back it’s a score
Big Big News Coming ??percent money in our pockets
Randy we all believed in you ,especially myself I knew all along you made this happen with all the hard work you have done ,it's now time for a rewarding life saving technology, Get ready everyone God Bless Randy for all the hard work you put into this project
Here we are a year later and Not a Peep from McCoy. What ever happened to the December 2016 Update he promised?
Sounds good to me, Roll em'
Remember the last time RGIN published FDA trials news? RGIN popped to .64 cents! Rinse and repeat?
Nice POP at end of trading today. Hope and pray it is a sign of things to be sustained, soon would be nice. Very much appreciate the recent insights posted here also.
Yea, it does seem a bit random for some cheerleading to come out of nowhere. I'm long and still waiting.
right, just looking to see if/what might be out there. any news and boom, imo
Most of todays action is primarily Buys, so someone is heeding Nicoles advice.
https://ih.advfn.com/stock-market/USOTC/regenicin-inc-pc-RGIN/trades
Im a little leary, someone shows up out of the blue waving the pompoms when not a peep has surfaced from McCoy himself. That gives me pause.
Nicole...waited long time for good news. I own many shares for a long time. Bought b/c I liked what they published ref FDA support/IND work/plans/goals looked to be well done/rational and positive. But, no action for long time.
Can you say what and when will be published/proven to get us going. For example, progress w/FDA or plans for trials or $$ positive support. No splits, no share dilution, just solid results/progress. TIA
|
Followers
|
71
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
2931
|
|
Created
|
08/06/10
|
Type
|
Free
|
| Moderators | |||
Regenicin, Inc.
10 High Court
Little Falls, NJ 07424
**updated and correct phone number** (973) 557-8914
they filed this on aug 22 2016- http://www.otcmarkets.com/edgar/GetFilingHtml?FilingID=11558959
| Name: | Randall McCoy |
| Title: | Principal Executive Officer and Director |
| Date: | August 22, 2016 |
| Name: | John J. Weber |
| Title: | Principal Financial Officer and Director |
| Date: | August 22, 2016 |


LITTLE FALLS, N.J., June 12, 2012 /PRNewswire/ -- Regenicin, Inc. (OTC Bulletin Board: RGIN) a biotechnology company specializing in the development of and commercialization of regenerative cell therapies to restore the health of damaged tissues and organs, announced today that the Food and Drug Administration (FDA) has granted Orphan Status approval for the PermaDerm® product, the only tissue-engineered skin prepared from autologous (patient's own) skin cells consisting of both epidermal and dermal layers, that is indicated for catastrophic burn patients. The FDA had previously designated PermaDerm® to be a biological/drug (permanent skin replacement) not a medical device (temporary skin replacement). PermaDerm® is the only skin replacement technology to receive this Biological/drug designation.
COMPANY HIGHLIGHTS:
PermaDerm™ is being developed to be the only tissue-engineered skin prepared from autologous (patient's own) skin cells consisting of both epidermal and dermal layers. A small harvested section of the patient's own skin can be grown to graft an area one hundred times its size in as little as thirty days.
These living, self-to-self skin graft tissue are intended to form permanent skin tissue that will not be rejected by the immune system of the patient, a critical possibility in porcine or cadaver skin grafts used today.
The technology has been clinically tested in over 150 pediatric, catastrophic burn patients. Currently Regenicin is working with its contract manufacturer to prepare for pre-market approval of PermaDerm™ from the FDA.
PermaDerm™ is being designed to save lives, reduce healthcare costs by decreasing the patient's stay in the Critical Care Unit and reduce the need for additional surgeries. An insurance company procedural code has been approved for reimbursement of costs to hospitals. The American Medical Association has assigned CPT (Current Procedural Terminology) code for cultured skin substitutes under the dermal substitute category which enables insurance companies to process and hospitals to be reimbursed for cultured skin substitutes once approved by the FDA.
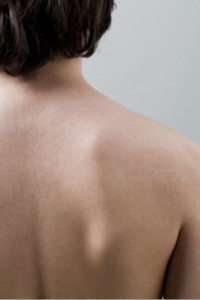
According to the American Burn Association, there are currently over 2,000 cases annually involving burns covering over 50% of the patient's total body surface area.
PermaDerm™ is being developed for the treatment of catastrophic, full-thickness burns covering over 50% of the total body surface area.
PermaDerm™ has been used in clinical trials on pediatric patients with catastrophic burns covering from 50 to 90+% of their body.
PermaDerm™ is being developed to require less donor skin from the patient's own body, thus needing fewer surgeries to harvest skin. The result is expected to be less scarring, with fewer infections and faster healing.
Randall McCoy
Chief Executive Officer
Randall McCoy has more than 37 years of experience in the healthcare industry. His experience includes executive positions and scientific research and development. His previous employment includes George Washington University, Temple University Medical School and RCA/SRI International (Director of Life Sciences and Electronic Displays). Mr. McCoy has assisted both small and major pharmaceutical/device companies address FDA issues. He has also helped over 225 foreign and domestic companies introduce their FDA regulated drug and medical device products into the US and World market. Mr. McCoy currently has over 30 Patents US and International.
Changed to AMBS
Dr. Joseph Rubinfeld Ph.D
Director at CytRX; Initial Co-Founder of Amgen, Inc.
Dr. Joseph Rubinfeld co-founded SuperGen, Inc. in 1991 and has served as its Chief Executive Officer and President, Chief Scientific Officer and as a director. He has been a director of CytRX since July 2002. Dr. Rubinfeld is also a founder of JJ Pharma. Dr. Rubinfeld was one of the four initial founders of Amgen, Inc. in 1980 and served as a Vice President and its Chief of Operations until 1983. From 1987 until 1990, Dr. Rubinfeld was a Senior Director at Cetus Corporation and from 1968 to 1980, Dr. Rubinfeld was employed at Bristol-Myers Company. Dr. Rubinfeld received a B.S. degree in chemistry from C.C.N.Y. and an M.A. and Ph.D in chemistry from Columbia University.
John Weber
Regenicin™'s Interim CFO
John Weber most recently served as Executive Vice President of Fujifilm USA, the highest ranked American corporate officer, from 2006 until his departure in 2009. His responsibilities included overseeing all corporate activity with the exception of R&D. During that time Fujifilm was ranked as the fastest growing medical imaging company, consistently ranking #1 or #2 in customer satisfaction. From 1998 through 2006 he served as Senior Vice President, Operations at Fujifilm USA where he spearheaded the transition of the company from a film distributor to a digital medical informatics company. From 1986 until 1998 he served as CFO where he helped to profitably manage Fuji's growth from an employee base of 75 to over 1,000. Prior to his distinguished career at Fujifilm USA, Mr. Weber served as the CFO for the confectionary and drinks division of Cadbury Schweppes Limited for three years and as Corporate Controller for an additional five years.
Dr. Craig Eagle
Pfizer Oncology
Dr. Craig Eagle joined Pfizer Australia in 2001 as part of the medical group. In Australia, his role involved leading and participating in scientific research, regulatory and pricing & re-imbursement negotiations for compounds in therapeutic areas including oncology, anti-infectives, respiratory, arthritis and pain management. In 2003, Pfizer relocated Dr. Eagle to the United States where he was appointed as the worldwide lead for development of Celecoxib in oncology to oversee the global research program. Since that time he has had increasing responsibility for overseeing the global research plans and teams for Irinotecan and Dalteparin. In 2007, he became head of Medical Affairs and Outcomes Research for Pfizer, including the US oncology business. Dr. Eagle has led, or been directly involved with, teams that resulted in eight new products or indications. As part of his current role at Pfizer, he has led the integration of the Pfizer/Wyeth oncology businesses and portfolio.
The Broadsmoore Group, LLC 9,223,770(8)
560 Lexington Ave., 16 th Fl.
New York, NY 10022
PDA Associates LLC 7,770,000(9)
560 Lexington Ave., 16 th Fl.
New York, NY 10022
Officers, directors and 5 percent shareholders collectively 54,508,099(10) 59.74%
* Less than 1%
(1) Unless otherwise indicated, each person or entity named in the table has sole voting power and investment power (or shares that power with that person's spouse) with respect to all shares of common stock listed as owned by that person or entity.
(2) A total of 83,807,964 shares of the Company's common stock are considered to be outstanding pursuant to Rule 13d-3(d)(1) under the Securities Exchange Act of 1934.
(3) Includes 32,821,641 shares of common stock held in his name and options to purchase 885,672 shares of common stock.
(4) Includes 50,000 shares of common stock held in his name and options to purchase 885,672 shares of common stock,
(5) Includes 1,050,000 shares of common stock held in his name and options to purchase 885,672 shares of common stock
(6) Includes 50,000 shares of common stock held in his name and options to purchase 885,672 shares of common stock,
(7) Includes 33,971,641 shares of common stock, options to purchase 3,542,688 shares of common stock and shares of Series A Convertible Preferred Stock convertible into 3,350,000 shares of common stock,
(8) Includes 5,706,270 shares of common stock held in its name, warrants to purchase 167,500 shares of common stock and shares of Series A Convertible Preferred Stock convertible into 3,350,000 shares of common stock,
(9) Includes 7,400,000 shares of common stock held in its name and warrants to purchase 370,000 shares of common stock,
(10) Includes 47,077,911 shares of common stock, warrants to purchase 537,500 shares of common stock, options to purchase 3,542,688 shares of common stock and shares of Series A Convertible Preferred Stock convertible into 3,350,000 shares of common stock
http://ih.advfn.com/p.php?pid=nmona&article=5144097
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
