Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
would be amazing if you are right... I would love for this stock to go on a run. Already own a bunch but would buy more if they came out with an additional information about progress.
Cant believe this stock went to $6 when it was a pipedream and now is at .075 when it gets closer and closer to being a real game changer
First, read this part ( multiple diseases including cancer)PV 10 is a cancer destroyer what else does PV 10 do, for multiple diseases? And second, how is PVCT going to bring back balance to the public health care system? if it is not helping with corvid 19.
no, I think he is referring more to the issue that with the attention being so focused on COVID, dont forget how important successful cancer treatments still are.
MY take at least
Could they have a corvid 19 link to PV 10 for a cure?
Study the red highlighted parts of PVCT press release
Mr. Bailey said, “I am gratified to join Provectus’ board of directors at an exciting time in the Company’s history and look forward to helping advance its goal of developing safe, effective, and affordable immunotherapy medicines for multiple diseases including cancer. I feel particularly honored to represent the Clover Fork Trust and our extended family, which stretches from New Jersey to Alabama. Many of our family members have been ravaged by cancer, and the Clover Fork Trust recently committed to do something about this terrible disease. Helping Provectus make cancer care more accessible and affordable closely aligns with our family’s goal and dream.”
Ed Pershing, CPA, Chair of Provectus’ Board, said “We are pleased to receive Clover Fork Trust’s support and obtain Webster’s participation on our board of directors. The coronavirus pandemic has exposed vulnerabilities in societies around the world and in the United States in particular. Part of Provectus’ mission, and ingrained in our company culture, is the development of drugs that bring balance back to our public health care system and provide greater access to affordable health care.”
I think breaking through 0.075 can happen every minute. But I have no idea what led to this week's strong and strange action.
would love a catalyst to come out and really drive this through .075 to a new 52 week high
Someone wants to get some 0.0747 (Perhaps an omen - Boeing's 747 will also not be for sale anymore soon. ![]() )
)
Seriously: The only news is that Form 3 for Mr. Websters son and step son. I can't imagine he did this in anticipation of a sale of the company.
yesterday was a very weird trading day
Why it dropped to .04 on the open to only rally back to .074 was atypical of this stock.
But I agree with you, it is waking up for some reason.
Something is brewing. Hopefully some rose bengal
It is a hidden Gem, yes a buy out would be nice, 5.00 a share would be 2 billion for the company I would be happy with 2.50 a share .
finally broke over .06....
close to making a 52 week high at .075.
Something cooking?
Wouldnt it be nice if Merck just bought them out and they could have the combination drugs to fight multiple forms of cancer
Would have no problem being bought out at $5/share in an all stock deal with MRK.
NEWS -- Provectus Biopharmaceuticals Announces Acceptance of Two PV-10® Melanoma Cancer Abstracts at European Society for Medical Oncology (ESMO) Virtual Congress 2020
KNOXVILLE, TN, July 22, 2020 (GLOBE NEWSWIRE) -- Provectus (OTCQB: PVCT) today is pleased to announce that data from an ongoing clinical trial of investigational autolytic cancer immunotherapy PV-10 (rose bengal disodium) in combination with immune checkpoint blockade for the treatment of advanced cutaneous melanoma (NCT02557321) will be presented at the European Society for Medical Oncology (ESMO) Virtual Congress, to be held online from September 19-21, 2020.
The abstracts accepted for electronic poster presentations are entitled:
$PVCT Mr. Bailey said, “I am gratified to join Provectus’ board of directors at an exciting time in the Company’s history and look forward to helping advance its goal of developing safe, effective, and affordable immunotherapy medicines for multiple diseases including cancer.
Now, this is interesting the stock is holding at around .05 cents , board members think this is an exciting time could they have found out that this has a corvid 19 cure, a dream, or am I onto something.
Mr. Bailey said, “I am gratified to join Provectus’ board of directors at an exciting time in the Company’s history and look forward to helping advance its goal of developing safe, effective, and affordable immunotherapy medicines for multiple diseases including cancer.
NEWS -- Provectus Biopharmaceuticals Announces Change to Board of Directors
KNOXVILLE, TN, July 16, 2020 (GLOBE NEWSWIRE) -- Provectus (OTCQB: PVCT) today is pleased to announce the addition of Webster Bailey to the Company's Board of Directors (the “Board”), effective as of July 20, 2020. Mr. Bailey is a member of the board of directors of the Clover Fork Coal Company investment trust (the “Clover Fork Trust”), which had invested in the Company.
As contemplated by the Definitive Financing Commitment Term Sheet entered into on March 19, 2017 between the Company and a group of its stockholders (the “PRH Group”), which set forth the terms on which the PRH Group would provide financing to the Company (the “2017 Definitive Financing”), Jan Koe resigned from the Board on July 11, 2020.
Mr. Bailey currently serves as the Executive Director of Business Development and Marketing at Cornerstone of Recovery (“Cornerstone”), an East Tennessee substance abuse rehabilitation center. Since 2007, he has directed all of Cornerstone’s marketing, business development, and outreach efforts. Mr. Bailey also serves on Cornerstone’s Continuous Quality Improvement and Executive Committees. He is involved in several East Tennessee substance abuse prevention and recovery-related initiatives, including having served multiple terms as President of the boards of directors of the Metro Drug Coalition and the Blount County Recovery Court Foundation. For these and other community leadership efforts, Mr. Bailey received the Community Service Award from the Tennessee Licensed Professional Counselors Association (2013), the Recovery Services Award from the Metropolitan Drug Commission (2014), and the Prevention Champion Award from the Blount County Community Health Initiative (2015), and was named Professional of the Year by the East Tennessee Association of Alcoholism and Drug Abuse Counselors (2016). He received a Bachelor's Degree in Communications and Public Relations from the University of Tennessee.
Mr. Bailey said, “I am gratified to join Provectus’ board of directors at an exciting time in the Company’s history and look forward to helping advance its goal of developing safe, effective, and affordable immunotherapy medicines for multiple diseases including cancer. I feel particularly honored to represent the Clover Fork Trust and our extended family, which stretches from New Jersey to Alabama. Many of our family members have been ravaged by cancer, and the Clover Fork Trust recently committed to do something about this terrible disease. Helping Provectus make cancer care more accessible and affordable closely aligns with our family’s goal and dream.”
Ed Pershing, CPA, Chair of Provectus’ Board, said “We are pleased to receive Clover Fork Trust’s support and obtain Webster’s participation on our board of directors. The coronavirus pandemic has exposed vulnerabilities in societies around the world and in the United States in particular. Part of Provectus’ mission, and ingrained in our company culture, is the development of drugs that bring balance back to our public health care system and provide greater access to affordable health care.”
Mr. Pershing added, “On behalf of our shareholders, the board of directors thanks Jan Koe for his time, effort, and board contributions. He worked collaboratively with the PRH Group as it entered into the 2017 Definitive Financing with Provectus, introduced a new corporate strategy for the Company, and executed an initial business plan. I want to personally express my appreciation for Jan’s service on our board.”
About Provectus
Provectus Biopharmaceuticals, Inc. (Provectus or the Company) is a clinical-stage biotechnology company developing a new class of drugs based on an entirely- and wholly-owned family of chemical small molecules called halogenated xanthenes. Information about the Company’s clinical trials can be found at the NIH registry, http://www.clinicaltrials.gov. For additional information about Provectus, please visit the Company's website at http://www.provectusbio.com.
FORWARD-LOOKING STATEMENTS: The information in this press release may include “forward-looking statements,” within the meaning of U.S. securities legislation, relating to the business of the Company and its affiliates, which are based on the opinions and estimates of Company management and are subject to a variety of risks and uncertainties and other factors that could cause actual events or results to differ materially from those projected in the forward-looking statements. Forward-looking statements are often, but not always, identified by the use of words such as “seek,” “anticipate,” “budget,” “plan,” “continue,” “estimate,” “expect,” “forecast,” “may,” “will,” “project,” “predict,” “potential,” “targeting,” “intend,” “could,” “might,” “should,” “believe,” and similar words suggesting future outcomes or statements regarding an outlook.
The safety and efficacy of the agents and/or uses under investigation have not been established. There is no guarantee that the agents will receive health authority approval or become commercially available in any country for the uses being investigated or that such agents as products will achieve any particular revenue levels.
Due to the risks, uncertainties, and assumptions inherent in forward-looking statements, readers should not place undue reliance on these forward-looking statements. The forward-looking statements contained in this press release are made as of the date hereof or as of the date specifically specified herein, and the Company undertakes no obligation to update or revise any forward-looking statements, whether as a result of new information, future events or otherwise, except in accordance with applicable securities laws. The forward-looking statements are expressly qualified by this cautionary statement.
Risks, uncertainties, and assumptions include those discussed in the Company’s filings with the U.S. Securities and Exchange Commission, including those described in Item 1A of the Company’s Annual Report on Form 10-K for the year ended December 31, 2019 and the Company’s Quarterly Report on Form 10-Q for the quarter ended March 31, 2020.
###
Contact:
Provectus Biopharmaceuticals, Inc.
Heather Raines, CPA
Chief Financial Officer
Phone: (866) 594-5999
pvctw?.. delisted??. what now? warrant holders loose all or is there any value to it?
Yes, its time to perform!!
NEWS -- Provectus Biopharmaceuticals Announces Discovery of Unique PV-10®-Induced STING Pathway Mechanism for Presentation at American Association for Cancer Research (AACR) 2020 Virtual Annual Meeting II
Provectus Biopharmaceuticals Announces Discovery of Unique PV-10®-Induced STING Pathway Mechanism for Presentation at American Association for Cancer Research (AACR) 2020 Virtual Annual Meeting II
PV-10 treatment leads to STING dimerization
Researchers identify potential association of heat shock proteins with STING in PV-10-treated models
Monday June 22, 2020
KNOXVILLE, TN, June 22, 2020 (GLOBE NEWSWIRE) - Provectus (OTCQB: PVCT) today is pleased to announce that data from ongoing preclinical study of investigational autolytic cancer immunotherapy PV-10 (rose bengal disodium) is being presented at the American Association for Cancer Research (AACR) 2020 Virtual Annual Meeting II, held online June 22-24, 2020. This PV-10 research has been led by Aru Narendran, MD, PhD and his team of researchers at the University of Calgary in Alberta, Canada (UCalgary).
Dr. Narendran and his colleagues studied the effects of PV-10 treatment on primary cells and cell lines derived from pediatric leukemia patients. UCalgary showed that PV-10 treatment led to STING dimerization and the release of interferon gamma (IFNγ), indicating a potential immune activation mechanism of PV-10. UClalgary further showed that heat shock proteins (HSPs), which chaperone misfolded or abnormally folded proteins, associated with STING dimerization in PV-10-treated cells, indicating a mechanism that may lead to enhanced STING activation following PV-10 treatment.
A copy of the AACR poster presentation is available on Provectus' website at https://www.provectusbio.com/aacr-2020-poster.
"The essence of cancer is the struggle for survival of these abnormal cells in the body. Over the course of their existence, cancer cells acquire multiple cellular pathways that become active or inactive in order for cancer to have a survival advantage against our immune system. This struggle changes the biology of cancer cells, which may have a direct impact on the activity of anticancer drugs within these cells," Dr. Narendran said. "We observed these very dynamics from our research on PV-10 and pediatric leukemia cells. Classic STING activation does not occur through PV-10 treatment. Rather, STING forms a dimer complex following PV-10 treatment, which may potentially lead to effective immune activation and anticancer activity."
Dr. Narendran added, "Our PV-10 research has enabled us to show, we believe for the first time, that heat shock proteins, which play important roles in the survival of cancer cells, are involved in STING activation. We also believe the involvement of heat shock proteins with STING is an important observation that requires further study."
Dominic Rodrigues, Vice Chair of Provectus' Board of Directors, said "We are grateful to Dr. Narendran, his team, and the University of Calgary for their consequential research on PV-10 to better understand the basic biology of cancer. This seminal discovery of PV-10-induced alterations of the STING pathway, which plays a pivotal role in innate immunity, contributes to an increasing body of knowledge about how and why PV-10 may function as an immunotherapy across a growing number of cancer types."
About PV-10
By targeting tumor cell lysosomes, investigational new drug PV-10 treatment may yield immunogenic cell death in solid tumor cancers that results in tumor-specific reactivity in circulating T cells and a T cell mediated immune response against treatment refractory and immunologically cold tumors.1-3 Adaptive immunity can be enhanced by combining checkpoint blockade (CB) with PV-10.4
PV-10 is undergoing clinical study for adult solid tumor cancers, such as relapsed and refractory cancers metastatic to the liver and metastatic melanoma. PV-10 is also undergoing preclinical study for relapsed and refractory pediatric solid tumor cancers (e.g., neuroblastoma, Ewing sarcoma, rhabdomyosarcoma, and osteosarcoma5,6 and relapsed and refractory pediatric blood cancers (such as acute lymphocytic leukemia and acute myelomonocytic leukemia)7,8.
Tumor Cell Lysosomes as the Seminal Drug Target
Lysosomes are the central organelles for intracellular degradation of biological materials, and nearly all types of eukaryotic cells have them. Discovered by Christian de Duve, MD in 1955, lysosomes are linked to several biological processes, including cell death and immune response. In 1959, de Duve described them as 'suicide bags' because their rupture causes cell death and tissue autolysis. He was awarded the Nobel Prize in 1974 for discovering and characterizing lysosomes, which are also linked to each of the three primary cell death pathways: apoptosis, autophagy, and necrosis.
Building on the Discovery, Exploration, and Characterization of Lysosomes
Cancer cells, particularly advanced cancer cells, are very dependent on effective lysosomal functioning9. Cancer progression and metastasis are associated with lysosomal compartment changes10,11, which are closely correlated with (among other things) invasive growth, angiogenesis, and drug resistance12.
PV-10 selectively accumulates in the lysosomes of cancer cells upon contact, disrupting the lysosomes and causing the cells to die. Provectus1,13, external collaborators6, and other researchers14,15,16 have independently shown that PV-10 (RB) triggers each of the three primary cell death pathways: apoptosis, autophagy, and necrosis.
Cancer Cell Autolytic Death via PV-10: PV-10 induced autolytic cell death, or death by self-digestion, in Hepa1-6 murine HCC cells can be viewed in this Provectus video of this event (ethidium homodimer [ED-1] stains DNA, but is excluded from intact nuclei; lysosensor green [LSG] stains intact lysosomes; the video is provided in 30-second frames; the event has a duration of approximately one hour). Exposure to PV-10 triggers the disruption of lysosomes, followed by nucleus failure and autolytic cell death. Identical responses have been shown by the Company in HTB-133 human breast carcinoma (which can be viewed in this Provectus video; this event has a duration of approximately two hours) and H69Ar human multidrug-resistant small cell lung carcinoma. Cancer cell autolytic cell death was reproduced by research collaborators from POETIC using relapsed and refractory human pediatric neuroblastoma cells to show that lysosomes are disrupted upon exposure to PV-10.5
PV-10 causes acute autolytic destruction of injected tumors (i.e., cell death), mediating the release of danger-associated molecular pattern molecules (DAMPs) and tumor antigens that may initiate an immunologic cascade where local response by the innate immune system may facilitate systemic anti-tumor immunity by the adaptive immune system. The DAMP release-mediated adaptive immune response activates lymphocytes, including CD8+ T cells, CD4+ T cells, and NKT cells, based on clinical and preclinical experience in multiple tumor types. Mediated immune signaling pathways may include an effect on STING, which plays an important role in innate immunity8.
Orphan Drug Designations (ODDs)
ODD status has been granted to PV-10 by the U.S. Food and Drug Administration for the treatments of metastatic melanoma in 2006, hepatocellular carcinoma in 2011, neuroblastoma in 2018, and ocular melanoma (including uveal melanoma) in 2019.
Drug Product
Rose bengal disodium (RB) (4,5,6,7-tetrachloro-2',4',5',7'-tetraiodofluorescein disodium salt) is a small molecule halogenated xanthene and PV-10's active pharmaceutical ingredient. The Company manufactures RB using a patented process designed to meet strict modern global quality requirements for pharmaceuticals and pharmaceutical ingredients (Good Manufacturing Practice, or GMP). PV-10 drug product is an injectable formulation of 10% w/v GMP RB in 0.9% saline, supplied in single-use glass vials containing 5 mL (to deliver) of solution, and administered without dilution to solid tumors via IT injection.
Intellectual Property (IP)
Provectus' IP includes a family of US and international (a number of countries in Asia, Europe, and North America) patents that protect the process by which GMP RB and related halogenated xanthenes are produced, avoiding the formation of previously unknown impurities that exist in commercial grade RB in uncontrolled amounts. The requirement to control these impurities is in accordance with International Council on Harmonisation (ICH) guidelines for the manufacturing of an injectable pharmaceutical. US patent numbers are 8,530,675, 9,273,022, and 9,422,260, with expirations ranging from 2030 to 2031.
The Company's IP also includes a family of US and international (a number of countries in Asia, Europe, and North America) patents that protect the combination of PV-10 and systemic immunomodulatory therapy (e.g., anti-CTLA-4, anti-PD-1, and anti-PD-L1 agents) for the treatment of a range of solid tumor cancers. US patent numbers are 9,107,887, 9,808,524, 9,839,688, and 10,471,144, with expirations ranging from 2032 to 2035; US patent application numbers include 20200138942.
About Provectus
Provectus Biopharmaceuticals, Inc. (Provectus or the Company) is a clinical-stage biotechnology company developing a new class of drugs based on an entirely-and-wholly-owned family of chemical small molecules called halogenated xanthenes. Information about the Company's clinical trials can be found at the NIH registry, www.clinicaltrials.gov. For additional information about Provectus, please visit the Company's website at www.provectusbio.com.
References
Wachter et al. Functional Imaging of Photosensitizers using Multiphoton Microscopy. Proceedings of SPIE 4620, 143, 2002.
Liu et al. Intralesional rose bengal in melanoma elicits tumor immunity via activation of dendritic cells by the release of high mobility group box 1. Oncotarget 7, 37893, 2016.
Qin et al. Colon cancer cell treatment with rose bengal generates a protective immune response via immunogenic cell death. Cell Death and Disease 8, e2584, 2017.
Liu et al. T cell mediated immunity after combination therapy with intralesional PV-10 and blockade of the PD-1/PD-L1 pathway in a murine melanoma model. PLoS One 13, e0196033, 2018.
Swift et al. Potent in vitro and xenograft antitumor activity of a novel agent, PV-10, against relapsed and refractory neuroblastoma. OncoTargets and Therapy 12, 1293, 2019.
Swift et al. In vitro and xenograft anti-tumor activity, target modulation and drug synergy studies of PV-10 against refractory pediatric solid tumors. 2018 ASCO Annual Meeting, J Clin Oncol 36, 2018 (suppl; abstr 10557).
Swift et al. In Vitro Activity and Target Modulation of PV-10 Against Relapsed and Refractory Pediatric Leukemia 2018 ASH Annual Meeting, Blood 132 2018 (suppl; abstr 5207).
Narendran et al. Association of heat shock proteins as chaperone for STING: A potential link in a key immune activation mechanism revealed by the novel anti-cancer agent PV-10. 2020 AACR VAM II, pending (abstr 5393).
Piao et al. Targeting the lysosome in cancer. Annals of the New York Academy of Sciences. 2016; 1371(1): 45.
Nishimura et al. Malignant Transformation Alters Intracellular Trafficking of Lysosomal Cathespin D in Human Breast Epithelial Cells. Pathology Oncology Research. 1998; 4(4): 283.
Gocheva et al. Distinct roles for cysteine cathepsin genes in multistage tumorigenesis. Genes & Development. 2006; 20(5): 543.
Fehrenbacher et al. Lysosomes as Targets for Cancer Therapy. Cancer Research. 2005; 65 (8): 2993.
Wachter et al. Imaging Photosensitizer Distribution and Pharmacology using Multiphoton Microscopy. Proceedings of SPIE 4622, 112, 2002..
Koevary Selective toxicity of rose Bengal to ovarian cancer cells in vitro. International Journal of Physiology, Pathophysiology and Pharmacology 4(2), 99, 2012.
Zamani et al. Rose Bengal suppresses gastric cancer cell proliferation via apoptosis and inhibits nitric oxide formation in macrophages. Journal of Immunotoxicology, 11(4), 367, 2014.
Luciana et al. Rose Bengal Acetate photodynamic therapy-induced autophagy. Cancer Biology & Therapy, 10:10, 1048, 2010.
Trademark
PV-10® is a registered trademark of Provectus, Knoxville, Tennessee, U.S.A.
The complete press release is available at https://www.provectusbio.com/news/press-releases/provectus-pr-20200622-1 on the Provectus website.
WTF is this PVCT is .05c, the stock price did not go up but warrant (pvctw) shot up 1000+%, how is this possible??....usually warrants trade 1/1000th of the share price?...
anyone to explain this?..
NEWS -- Provectus Biopharmaceuticals Expands Global Patent Portfolio to India for Cancer Combination Therapy
PVCT- calling breakout!!, why is this stock is trading at few pennies instead of in dollars???...
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=156058048
NEWS -- Provectus Announces Publication of Updated Data from Metastatic Neuroendocrine Phase 1 PV-10® Study at American Society of Clinical Oncology (ASCO) 2020 Virtual Scientific Program
NEWS Provectus Announces Publication of Updated Data from Metastatic Neuroendocrine Phase 1 PV-10® Study at American Society of Clinical Oncology (ASCO) 2020 Virtual Scientific Program
Press Release | 06/01/2020
Treatment refractory, immunologically cold, hepatic tumor type
50% reduction of treated disease; 83% stabilization of overall patient disease (RECIST)
Median progression-free survival not reached (by CT scan; RECIST)
Overall survival >22.5 months; ongoing follow-up for 50% of patients
KNOXVILLE, TN, June 01, 2020 (GLOBE NEWSWIRE) -- Provectus (OTCQB: PVCT) today is pleased to announce that updated data from the first cohort of the Company’s neuroendocrine tumors (NET) metastatic to the liver (mNET) Phase 1 study of investigational autolytic cancer immunotherapy PV-10 (rose bengal disodium) were published as an abstract as part of the American Society of Clinical Oncology (ASCO) 2020 Virtual Scientific Program, held online May 29-31, 2020.
Small molecule-based PV-10 is administered either by cutaneous intratumoral (IT) injection to superficial melanoma and non-melanoma skin cancer tumors (such as basal cell carcinoma, Merkel cell carcinoma, and squamous cell carcinoma) or by percutaneous IT injection to visceral primary and metastatic hepatic tumors (such as hepatocellular carcinoma, metastatic colorectal cancer, mNET, and metastatic uveal melanoma). By targeting tumor cell lysosomes, PV-10 treatment may yield immunogenic cell death in solid tumor cancers that results in tumor-specific reactivity in circulating T cells and a T cell mediated immune response against treatment refractory and immunologically cold tumors.1-3 Adaptive immunity may be enhanced by combining immune checkpoint blockade (CB) with PV-10.4
This single-center Phase 1 study is being conducted at The Queen Elizabeth Hospital in Adelaide, Australia to evaluate the potential safety, tolerability, and preliminary efficacy of single-agent PV-10 treatment in symptomatic mNET patients (NCT02693067) refractory to systemic somatostatin analogues (SSA) and peptide receptor radionuclide therapy (PRRT). The primary endpoint of the trial is safety; secondary endpoints include objective response rate (ORR) of injected target and measurable bystander lesions, target lesion somatostatin receptor expression, and biochemical response. Response assessments were conducted using Response Evaluation Criteria in Solid Tumors (RECIST) criteria and by independent review. Six patients in the first cohort each received one percutaneously-administered IT injection of PV-10 to one target lesion per treatment cycle. Patients in the second cohort can receive PV-10 injection of multiple lesions per cycle; enrollment of this cohort is nearing completion.
Highlights from the mNET Presentation at ASCO:
Baseline characteristics (N=6): 67% male; median age of 65 years (range 47-72).
Disease characteristics
Primary tumor site: small bowel (50%), pancreas (33%), and caecal (17%)
NET grades: Grade 1 (83%) and Grade 2 (17%)
All patients were refractory to SSA and PRRT
PV-10 treatment summary: Median of 1 cycle (mean 1.7, range 1-4)
Safety: Post-procedure pain, carcinoid flare, and nausea
Injected target lesion efficacy (RECIST 1.1): 50% partial response (PR), 50%ORR
Patient-level efficacy (RECIST 1.1)
83%a disease control rate (DCR)
Progression-free survival (PFS): Not reached by computerized tomography (CT) scan (range 2.4-25.3+ months); 6.1 months by positron emission tomography (PET) scan
Overall survival (OS): >22.5 months; 50% ongoing response follow-up (range 18.1-33.8 months OS) at the January 2020 data cut-off
a Typographical error in the abstract
A copy of the abstract, which was first published on May 13th, is available on the ASCO website at https://meetinglibrary.asco.org/record/186339/abstract .
About PV-10
PV-10 is an investigational new drug undergoing clinical study for adult solid tumor cancers, like melanoma and cancers of the liver (including mNET and metastatic uveal melanoma). PV-10 is also undergoing preclinical study for pediatric solid tumor cancers (like neuroblastoma, Ewing sarcoma, rhabdomyosarcoma, and osteosarcoma) and pediatric blood cancers (like leukemia).5,6
Tumor Cell Lysosomes as the Seminal Drug Target
Lysosomes are the central organelles for intracellular degradation of biological materials, and nearly all types of eukaryotic cells have them. Discovered by Christian de Duve, MD in 1955, lysosomes are linked to several biological processes, including cell death and immune response. In 1959, de Duve described them as ‘suicide bags’ because their rupture causes cell death and tissue autolysis. He was awarded the Nobel Prize in 1974 for discovering and characterizing lysosomes, which are also linked to each of the three primary cell death pathways: apoptosis, autophagy, and necrosis.
Building on the Discovery, Exploration, and Characterization of Lysosomes
Cancer cells, particularly advanced cancer cells, are very dependent on effective lysosomal functioning.7 Cancer progression and metastasis are associated with lysosomal compartment changes8,9, which are closely correlated with (among other things) invasive growth, angiogenesis, and drug resistance10.
PV-10 selectively accumulates in the lysosomes of cancer cells upon contact, disrupting the lysosomes and causing the cells to die. Provectus1,11, external collaborators6, and other researchers13,14,16 have independently shown that PV-10 (RB) triggers each of the three primary cell death pathways: apoptosis, autophagy, and necrosis.
Cancer Cell Autolytic Death via PV-10: PV-10 induced autolytic cell death, or death by self-digestion, in Hepa1-6 murine HCC cells can be viewed in this Provectus video of the event (ethidium homodimer 1 [ED-1] stains DNA, but is excluded from intact nuclei; lysosensor green [LSG] stains intact lysosomes; the video is provided in 30-second frames; the event has a duration of approximately one hour). Exposure to PV-10 triggers the disruption of lysosomes, followed by nucleus failure and autolytic cell death. Identical responses have been shown by the Company in HTB-133 human breast carcinoma (which can be viewed in this Provectus video; this event has a duration of approximately two hours) and H69Ar human multidrug-resistant small cell lung carcinoma. Cancer cell autolytic cell death was reproduced by research collaborators from POETIC using relapsed and refractory human pediatric neuroblastoma cells to show that lysosomes are disrupted upon exposure to PV-10.5
Immune Signaling Pathways: PV-10 causes acute autolytic destruction of injected tumors (i.e., cell death), mediating several identified immune signaling pathways studied to date, such as the release of danger-associated molecular pattern molecules (DAMPs) and tumor antigens that initiate an immunologic cascade where local response by the innate immune system facilitates systemic anti-tumor immunity by the adaptive immune system. The DAMP release-mediated adaptive immune response activates lymphocytes, including CD8+ T cells, CD4+ T cells, and NKT cells, based on clinical and preclinical experience in multiple tumor types. Other mediated immune signaling pathways that have been identified include poly (ADP-ribose) polymerase (PARP) cleavage5 and stimulator of interferon genes (STING), which plays an important role in innate immunity15. PV-10 is the first cancer drug that may facilitate multiple, complementary, immune system signaling pathways.16
Orphan Drug Designations (ODDs)
ODD status has been granted to PV-10 by the U.S. Food and Drug Administration for the treatments of metastatic melanoma in 2006, hepatocellular carcinoma in 2011, neuroblastoma in 2018, and ocular melanoma (including uveal melanoma) in 2019.
Drug Product
Rose bengal disodium (RB) (4,5,6,7-tetrachloro-2’,4’,5’,7’-tetraiodofluorescein disodium salt) is a small molecule halogenated xanthene and PV-10’s active pharmaceutical ingredient. The Company manufactures RB using a patented process designed to meet strict modern global quality requirements for pharmaceuticals and pharmaceutical ingredients (Good Manufacturing Practice, or GMP). PV-10 drug product is an injectable formulation of 10% w/v GMP RB in 0.9% saline, supplied in single-use glass vials containing 5 mL (to deliver) of solution, and administered without dilution to solid tumors via IT injection.
Intellectual Property (IP)
Provectus’ IP includes a family of US and international (a number of countries in Asia, Europe, and North America) patents that protect the process by which GMP RB and related halogenated xanthenes are produced, avoiding the formation of previously unknown impurities that exist in commercial grade RB in uncontrolled amounts. The requirement to control these impurities is in accordance with International Council on Harmonisation (ICH) guidelines for the manufacturing of an injectable pharmaceutical. US patent numbers are 8,530,675, 9,273,022, and 9,422,260, with expirations ranging from 2030 to 2031.
The Company's IP also includes a family of US and international (a number of countries in Asia, Europe, and North America) patents that protect the combination of PV-10 and systemic immunomodulatory therapy (e.g., anti-CTLA-4, anti-PD-1, and anti-PD-L1 agents) for the treatment of a range of solid tumor cancers. US patent numbers are 9,107,887, 9,808,524, 9,839,688, and 10,471,144, with expirations ranging from 2032 to 2035; US patent application numbers include 20200138942.
About Provectus
Provectus Biopharmaceuticals, Inc. (Provectus or the Company) is a clinical-stage biotechnology company developing a new class of drugs based on an entirely- and wholly-owned family of chemical small molecules called halogenated xanthenes. Information about the Company’s clinical trials can be found at the NIH registry, www.clinicaltrials.gov. For additional information about Provectus, please visit the Company's website at www.provectusbio.com.
References
1. Wachter et al. Functional Imaging of Photosensitizers using Multiphoton Microscopy. Proceedings of SPIE 4620, 143, 2002.
2. Liu et al. Intralesional rose bengal in melanoma elicits tumor immunity via activation of dendritic cells by the release of high mobility group box 1. Oncotarget 7, 37893, 2016.
3. Qin et al. Colon cancer cell treatment with rose bengal generates a protective immune response via immunogenic cell death. Cell Death and Disease 8, e2584, 2017.
4. Liu et al. T cell mediated immunity after combination therapy with intralesional PV-10 and blockade of the PD-1/PD-L1 pathway in a murine melanoma model. PLoS One 13, e0196033, 2018.
5. Swift et al. Potent in vitro and xenograft antitumor activity of a novel agent, PV-10, against relapsed and refractory neuroblastoma. OncoTargets and Therapy 12, 1293, 2019.
6. Swift et al. In vitro and xenograft anti-tumor activity, target modulation and drug synergy studies of PV-10 against refractory pediatric solid tumors. 2018 ASCO Annual Meeting, J Clin Oncol 36, 2018 (suppl; abstr 10557).
7. Piao et al. Targeting the lysosome in cancer. Annals of the New York Academy of Sciences. 2016; 1371(1): 45.
8. Nishimura et al. Malignant Transformation Alters Intracellular Trafficking of Lysosomal Cathespin D in Human Breast Epithelial Cells. Pathology Oncology Research. 1998; 4(4): 283.
9. Gocheva et al. Distinct roles for cysteine cathepsin genes in multistage tumorigenesis. Genes & Development. 2006; 20(5): 543.
10. Fehrenbacher et al. Lysosomes as Targets for Cancer Therapy. Cancer Research. 2005; 65 (8): 2993.
11. Wachter et al. Imaging Photosensitizer Distribution and Pharmacology using Multiphoton Microscopy. Proceedings of SPIE 4622, 112, 2002.
12. Koevary. Selective toxicity of rose Bengal to ovarian cancer cells in vitro. International Journal of Physiology, Pathophysiology and Pharmacology 4(2), 99, 2012.
13. Zamani et al. Rose Bengal suppresses gastric cancer cell proliferation via apoptosis and inhibits nitric oxide formation in macrophages. Journal of Immunotoxicology, 11(4), 367, 2014.
14. Luciana et al. Rose Bengal Acetate photodynamic therapy-induced autophagy. Cancer Biology & Therapy, 10:10, 1048, 2010.
15. Narendran et al. Association of heat shock proteins as chaperone for STING: A potential link in a key immune activation mechanism revealed by the novel anti-cancer agent PV-10. 2019 AACR Annual Meeting, pending (abstr 8165).
16. Panzarini et al. Timing the multiple cell death pathways initiated by Rose Bengal acetate photodynamic therapy. Cell Death & Disease 2, 169, 2011.
Trademarks
PV-10® is a registered trademark of Provectus, Knoxville, Tennessee, U.S.A.
FORWARD-LOOKING STATEMENTS: This release contains forward-looking statements as defined under U.S. federal securities laws. These statements reflect management's current knowledge, assumptions, beliefs, estimates, and expectations and express management's current views of future performance, results, and trends and may be identified by their use of terms such as “anticipate,” “believe,” “could,” “estimate,” “expect,” “intend,” “may,” “plan,” “predict,” “project,” “will,” and other similar terms. Forward-looking statements are subject to a number of risks and uncertainties that could cause our actual results to materially differ from those described in the forward-looking statements. Readers should not place undue reliance on forward-looking statements. Such statements are made as of the date hereof, and we undertake no obligation to update such statements after this date. No claims with respect to PV-10, Provectus’ investigational drug for oncology, or PH-10, the Company’s investigational drug for dermatology, are intended regarding safety or efficacy in the context of any forward-looking statements made in this press release.
Risks and uncertainties that could cause our actual results to materially differ from those described in forward-looking statements include those discussed in our filings with the Securities and Exchange Commission (including those described in Item 1A of our Annual Report on Form 10-K for the year ended December 31, 2019).
###
NEWS -- Provectus Announces PV-10® STING Agonist Abstract at American Association for Cancer Research (AACR) 2020 Virtual Annual Meeting II
KNOXVILLE, TN, May 18, 2020 (GLOBE NEWSWIRE) -- Provectus (PVCT) today is pleased to announce the publication of an abstract about and data from ongoing preclinical study of investigational autolytic cancer immunotherapy PV-10 (rose bengal disodium) for the American Association for Cancer Research (AACR) 2020 Virtual Annual Meeting II, to be held June 22-24, 2020. The abstract was posted online by AACR on May 15 in advance of the virtual meeting.
Small molecule-based PV-10 is administered either by cutaneous intratumoral (IT) injection to superficial melanoma and non-melanoma skin cancer tumors (such as basal cell carcinoma, Merkel cell carcinoma, and squamous cell carcinoma) or by percutaneous IT injection to visceral primary and metastatic tumors of the liver (such as hepatocellular carcinoma, metastatic colorectal cancer, metastatic neuroendocrine tumors, and metastatic uveal melanoma). By targeting tumor cell lysosomes, PV-10 treatment may yield immunogenic cell death in solid tumor cancers that results in tumor-specific reactivity in circulating T cells.1-4
Abstract:
NEWS -- Provectus Announces Acceptance of PV-10® Liver Cancer Abstracts at American Society of Clinical Oncology (ASCO) 2020 Virtual Scientific Program
KNOXVILLE, TN, April 29, 2020 (GLOBE NEWSWIRE) -- Provectus (PVCT) today is pleased to announce that abstracts about and data from our clinical trials of investigational autolytic cancer immunotherapy PV-10 (rose bengal disodium) as a single-agent for the treatment of neuroendocrine tumors metastatic to the liver (NCT02693067) and in combination with immune checkpoint blockade for the treatment of uveal melanoma metastatic to the liver (NCT00986661) were accepted for the American Society of Clinical Oncology (ASCO) 2020 Virtual Scientific Program, to be held May 29-31, 2020 online.
Small molecule-based PV-10 is administered by percutaneous intratumoral injection to primary or metastatic tumors of the liver, such as hepatocellular carcinoma, metastatic colorectal cancer, metastatic neuroendocrine tumors, and metastatic uveal melanoma. By targeting tumor cell lysosomes, PV-10 treatment may yield immunogenic cell death in solid tumor cancers that results in tumor-specific reactivity in circulating T cells.1-4
Abstract #1:
NEWS -- Provectus Announces PV-10® Liver Cancer Presentation in ePoster Gallery of Society of Interventional Radiology (SIR) 2020 Annual Scientific Meeting
KNOXVILLE, TN, April 16, 2020 (GLOBE NEWSWIRE) -- Provectus (PVCT) today said that data from its clinical trial of autolytic cancer immunotherapy PV-10 (rose bengal disodium) as a single-agent and in combination with immune checkpoint blockade for the treatment of primary or metastatic tumors of the liver (NCT00986661) was uploaded to the SIR 2020 ePoster Gallery of the canceled Society of Interventional Radiology (SIR) 2020 Annual Scientific Meeting, which was originally scheduled to be held March 28-April 2, 2020 in Seattle, Washington.
SARS-CoV-21 note: Provectus’ ongoing clinical trials continue to enroll new patients, and patients who have been enrolled and treated on these trials still receive care.
Small molecule-based PV-10 is administered by percutaneous intratumoral injection to primary or metastatic tumors of the liver, such as hepatocellular carcinoma (HCC), metastatic colorectal cancer (mCRC), metastatic neuroendocrine tumors, and metastatic uveal melanoma (mUM). Intratumoral injection with lysosomal-targeting PV-10 yields immunogenic cell death in solid tumor cancers that results in tumor-specific reactivity in circulating T cells.2-5
This multicenter, open-label, Phase 1 basket study is evaluating PV-10’s safety, tolerability, and preliminary efficacy in unresectable cancers of the liver. PV-10 is administered under image guidance to one to three tumors of 1.0 to 4.9 cm in diameter. Response assessments (Computed Tomography [CT], Magnetic Resonance Imaging [MRI], and/or Positron Emission Tomography [PET]) are performed at day 28, and then every 12 weeks. Patients with additional injectable disease may receive further PV-10 injection after day 28. Modified Response Evaluation Criteria in Solid Tumors (mRECIST) and European Association of the Study of the Liver (2D-EASL) criteria were used to evaluate response of injected tumors; 2D-EASL response assessment measures only viable tumor tissue. PV-10 is radiopaque, which facilitates disease treatment and response follow-up.
Updated Hepatic Tumor Results from the Presentation at SIR 2020:
NEWS -- PV-10® Abstract Previously Accepted for Presentation at Terminated American Association for Cancer Research (AACR) Annual Meeting 2020
KNOXVILLE, TN, March 17, 2020 (GLOBE NEWSWIRE) -- Provectus (PVCT) today said that data from ongoing research into investigational lysosomal-targeting cancer immunotherapy PV-10 (rose bengal disodium) for the treatment of solid tumor and blood cancers had been accepted for presentation at the now terminated AACR Annual Meeting 2020, which was originally scheduled to be held April 24-29 in San Diego, California. Intratumoral injection with PV-10 yields immunogenic cell death in solid tumor cancers that results in tumor-specific reactivity in circulating T cells.1-4
The details of the previously accepted abstract were:
Are you basing this on a story you saw on PVCT?
Well, it had a nice run years ago. Then, the company changed its name which made it difficult to trade. And, a negative article killed any positive progress. While it's been mostly quiet, the company continues to participate in medical presentation.
Over 15 years I’ve been in this pos. Hope something is up
Feels like something big is imminent....
NEWS -- Provectus Announces Presentation of Preliminary Results from Phase 1 Trial of PV-10® (rose bengal disodium) for Treatment of Metastatic Uveal Melanoma at ESMO I-O Congress 2019
NEWS -- Provectus Announces PV-10® Abstract Accepted for Presentation at Society of Interventional Radiology (SIR) 2020 Annual Scientific Meeting
KNOXVILLE, TN, Dec. 02, 2019 (GLOBE NEWSWIRE) -- Provectus (PVCT) today announced that data from an ongoing clinical trial of lysosomal-targeting cancer immunotherapy PV-10 (rose bengal disodium) as a single-agent for the treatment of primary or metastatic tumors of the liver (NCT00986661) will be presented at the upcoming Society of Interventional Radiology (SIR) 2020 Annual Scientific Meeting, to be held March 28-April 2, 2020, in Seattle, Washington. The accepted abstract is:
NEWS -- Provectus Announces Presentation of Initial Results from Phase 1B Trial of PV-10® in Combination with KEYTRUDA® for Treatment of Checkpoint-Refractory Advanced Melanoma at SMR 2019 Congress
KNOXVILLE, TN, Nov. 21, 2019 (GLOBE NEWSWIRE) -- Provectus (PVCT) today announced that data from the Company's ongoing Phase 1b/2 study of lysosomal-targeting cancer immunotherapy PV-10 (rose bengal disodium) in combination with KEYTRUDA (pembrolizumab) for the treatment of checkpoint inhibition-refractory advanced cutaneous melanoma patients were presented at the 16th International Congress of the Society for Melanoma Research (SMR 2019 Congress), held in Salt Lake City, Utah from November 20-24, 2019. Intralesional (aka intratumoral) injection with PV-10 can yield immunogenic cell death in solid tumor cancers that results in tumor-specific reactivity in circulating T cells.1-4
A first expansion cohort of the Phase 1b portion of the study began enrolling patients with metastatic melanoma who were checkpoint inhibition-refractory (the “Checkpoint-Refractory Cohort”) in December 2018 (NCT02557321). The Checkpoint-Refractory Cohort extended an exploratory group of refractory patients enrolled into the main cohort of this study, which primarily enrolled patients naïve to checkpoint inhibition (the “Checkpoint-Naïve Cohort”). Patients with at least one injectable lesion and who were candidates for KEYTRUDA were eligible. Eligible subjects received the combination treatment of PV-10 and KEYTRUDA every three weeks for up to five cycles (i.e., over a period of up to 12 weeks, with no further PV-10 administered after week 12), followed by only KEYTRUDA every three weeks for up to 24 months. The primary endpoint for the Phase 1b trial was safety and tolerability. Objective response rate and progression-free survival were key secondary endpoints (both assessed via RECIST 1.1 after five treatment cycles, and then every 12 weeks thereafter).
Initial Checkpoint-Refractory Results from the Presentation at SMR 2019 Congress:
Sapna Patel at CureOM....
Is this the same conference that is going on in Salt Lake City, or is this something different?
It looks like the CureOM is held "in conjunction" with SMR?
She's talking about Uveal, but our SMR posters aren't for Uveal, right?
Cool.
Sapna Patel @DrSapnaPatel reporting on the use of PV-10 to generate immune responses in uveal melanoma. @MRFCureOM @CureMelanoma pic.twitter.com/rdSAJchtwr
— Keiran Smalley (@LabSmalley) November 20, 2019
NEWS -- Provectus Announces Acceptance of PV-10® Poster Presentations at ESMO Immuno-Oncology Congress 2019
KNOXVILLE, TN, Oct. 24, 2019 (GLOBE NEWSWIRE) -- Provectus (PVCT) today announced that data from an ongoing clinical trial of lysosomal-targeting, cancer immunotherapy PV-10 (rose bengal disodium) as a single-agent and in combination with standard of care checkpoint inhibition for the treatment of uveal melanoma metastatic to the liver (NCT00986661, single-center expansion cohort) will be presented on two poster presentations at the European Society for Medical Oncology (ESMO) Immuno-Oncology Congress 2019, held in Geneva, Switzerland from December 11-14, 2019. The accepted abstracts are:
NEWS -- Provectus Announces Acceptance of PV-10 Poster Presentations at SMR 2019 Congress
KNOXVILLE, TN, Sept. 24, 2019 (GLOBE NEWSWIRE) -- Provectus (PVCT) today announced that data from an ongoing clinical trial of lysosomal-targeting cancer immunotherapy PV-10® in combination with checkpoint inhibition for the treatment of advanced cutaneous melanoma (NCT02557321) will be presented on two poster presentations at the SMR 2019 Congress (the Society for Melanoma Research annual meeting), held in Salt Lake City, Utah from November 20-23, 2019. The accepted abstracts are:
NEWS -- Provectus Names Harold Schmitz, PhD to Company's Scientific Advisory Board
I'm definitely holding. In fact whatever profit I make from flipping stinky pinkie stocks I use to buy more PVCT.
Eyed for acquisition.Very possible here! Insiders are holding.
NEWS -- PV-10-based Cancer Combination Therapy Clinical Trial Design Wins Australasian Gastro-Intestinal Trials Group’s New Concepts Award
It seems that someone needed some beer money today and sold some.
Warrants from the 10Q
What happens to warrants in the event of a reverse split?
Is there an adjustment provision, or not?
I admittedly did only limited research just now, but what I did read said that in the event of a reverse split, warrant prices don't get adjusted based on the split. This was only from one article though, from a while back.
Is it possible the PRH group has a R/S in the back of their minds, and thus whoever bought these $1-2 warrants would be in the money after a split?
Pfizer hits an alert
AS does another big fishy w/ another
They don't have all the money in the world
but what they all do is buy earnings
This is interesting that pfizer is a co-assignee
The USPTO has allowed Provectus' (OTC:PVCT) US patent application 15/804,357 for the combination of intratumoral PV-10 and systemic immunomodulatory therapy for the treatment of a range of solid tumor cancers.
This prospective new patent is the third continuation of USP 9,107,887, Provectus’ first and foundational cancer combination therapy patent granted by the USPTO in 2015.
It is also related to USP 9,808,524 and USP 9,839,688, which are also continuations and the Company’s second and third cancer combination therapy patents granted by the USPTO in 2017.
Pfizer is a co-assignee on all four awarded and allowed patents.
NEWS -- Provectus Names Frank Akers, PhD to Company’s Strategic Advisory Board
7+ days to go up 14% and just 1k to drop it -13%
|
Followers
|
185
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
9952
|
|
Created
|
08/09/02
|
Type
|
Free
|
| Moderators | |||
.png)

Provectus Biopharmaceuticals is investigating new therapies for the treatment of skin cancer and liver cancer. Provectus investigational oncology drug, PV-10, is an ablative immunotherapy under investigation in solid tumor cancers. The Company has received orphan drug designations from the FDA for its melanoma and hepatocellular carcinoma indications. PH-10, its topical investigational drug for dermatology, is undergoing clinical testing for psoriasis and atopic dermatitis. Provectus has completed Phase 2 trials of PV-10 as a therapy for metastatic melanoma, and of PH-10 as a topical treatment for psoriasis. In addition, Provectus has begun a Phase 3 trial as a therapy for metastatic melanoma. Information about these and the Company's other clinical trials can be found at the NIH registry, www.clinicaltrials.gov. For additional information about Provectus, please visit the Company's website at www.provectusbio.com.
The red synthetic dye Rose Bengal is shown in a bottle at Provectus Pharmaceuticals, Inc.
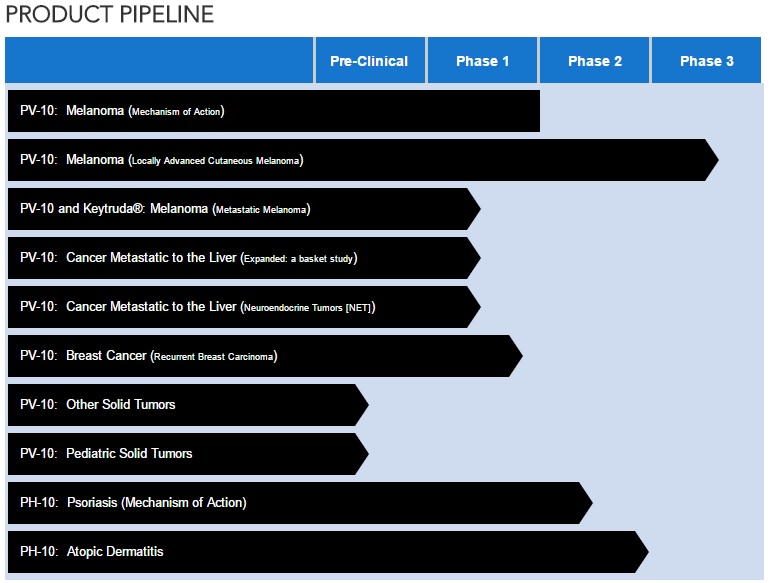
CURRENT PVCT PIPELINE (as of May 23, 2017)
--------------------------------------------------------------
Charts and Technical:
PVCT 6 months chart:
http://stockcharts.com/h-sc/ui?s=PVCT&p=D&yr=0&mn=6&dy=0&id=p03754661229
Technical analysis:
http://www.stockta.com/cgi-bin/analysis.pl?symb=PVCT&num1=7&cobrand=&mode=stock
PVCT News and Analysis:
PVCT News Blog with the latest news and analysis:
http://provectuspharmaceuticalsinc.blogspot.ca/p/news.html
CONNECTING THE DOTS - CURRENT NEWS PAGE
http://provectuspharmaceuticalsinc.blogspot.ca/p/current-news_22.html
CONNECTING THE DOTS - BLOG PAGE
http://provectuspharmaceuticalsinc.blogspot.ca/
PVCT News at OTC
http://www.otcmarkets.com/stock/PVCT/news
CLINICAL TRIALS Updates and Info:
| Short Interest | 167,323 (69.16%) Apr 13, 2017 |
| Significant Failures to Deliver | No |
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
