Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
FDA reviewing NVS’ 351(k) submission for Neulasta FoB: #msg-118583523.
AVEO 8-K filing today: Novartis AV-380 Inventory Reimbursement
Link:
http://www.otcmarkets.com/edgar/GetFilingHtml?FilingID=11012782
Novartis AV-380 Inventory Reimbursement
As previously disclosed, on August 13, 2015 (the “Effective Date”), AVEO Pharmaceuticals, Inc. (“AVEO”), entered into a License Agreement (the “License Agreement”) with Novartis International Pharmaceutical Ltd., a Bermuda corporation (“Novartis”). Under the License Agreement, AVEO granted to Novartis the exclusive right to develop and commercialize worldwide AVEO’s proprietary antibody AV-380 and related AVEO antibodies that bind to Growth Differentiation Factor 15 (GDF15) for the treatment and prevention of diseases and other conditions in all indications in humans.
Under the License Agreement, Novartis was granted the right to acquire AVEO’s inventory of clinical quality, AV-380 biological drug substance. On November 12, 2015, Novartis informed AVEO that it would exercise this right. Pursuant to the License Agreement, Novartis will reimburse AVEO $3.45 million for this existing inventory.
In addition to the $3.45 million inventory reimbursement payment, AVEO previously disclosed that under the terms of the License Agreement, AVEO received an upfront payment of $15 million and is eligible to receive potential clinical, regulatory and sales-based milestone payments as well as tiered royalties on product sales, each assuming successful advancement of the product. Novartis has responsibility under the License Agreement for the development, manufacture and commercialization of the AVEO antibodies and any resulting approved therapeutic products.
The foregoing information regarding the License Agreement does not purport to be complete and is qualified in its entirety by the full text of the License Agreement, which AVEO filed with the Securities and Exchange Commission as an exhibit to its Quarterly Report on Form 10-Q for the period ended September 30, 2015.
3Q15 results were in-line with prior guidance except at Alcon, which under-performed significantly.
Was it good or bad?
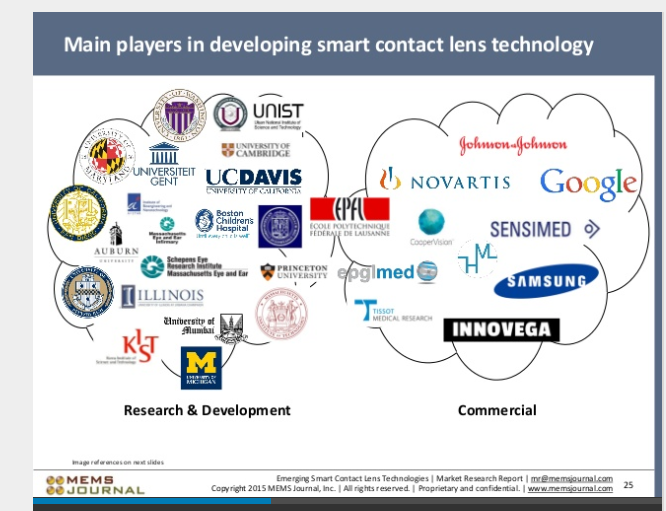
MEMS Journal ranks $NVS as a Major Player in upcoming smart contact revolution.
$COO $EPGL $GOOG $JNJ #SAMSUNG #SENSIMED #Smartcontactlens
http://www.slideshare.net/MikePinelisPhD/smart-contact-lenses-technologies-market-research-report-sample
50 BELOW 200 EMA LONG TERM DOWN
20 BELOW 50 EMS MID TERM DOWN
5 BELOW 2 0 EMA SHORT TERM DOWN
Novartis' inhaler for COPD gets FDA approval
Utibron Neohaler--Courtesy of Novartis
Novartis ($NVS) announced FDA approval last week of its Utibron Neohaler as a long-term maintenance treatment for COPD via the delivery of the bronchodilating agents indacaterol and glycopyrrolate.
In addition, the agency approved the use of glycopyrrolate inhalation powder as a monotherapy, which will be delivered using the Seebri Neohaler. Both combination products will hit the market in early 2016, according to a company release.
The approval comes after a trial of 2,654 patients with COPD that consisted of two 12-week efficacy studies and a 52-week safety study, the release notes. Novartis notes that the approval does not cover asthma, and Utibron is not meant to replace rescue inhalers for sudden attacks.
"Patients have told us about the tremendous impact their COPD can have on everyday activities," said Christi Shaw, Novartis' U.S. country head, in a statement. "With this approval, the COPD community now has a new medicine that can help so many patients by improving not only their symptoms, but also their health-related quality of life. This means the possibility of doing things that matter to them."
Novartis says the Neohaler line of inhalers generates low resistance during delivery, making it easy to use.
Patients are instructed to insert an Utibron capsule inside the inhaler and then pierce it by squeezing buttons on the side of the device. Then, patients must put their mouth on the mouthpiece, inhale and hold their breath for 5 to 10 seconds. Once the capsule is empty of powder (this could take more than one breath), it's time to finish the 13-step process by removing the capsule from the inhaler, according to the product website.
Unfortunately, proper breathing technique and usage of inhalers have been shown to be challenging for patients, making simplification a drug delivery challenge for the future.
Novartis says almost 27 million Americans have COPD, making it the third leading cause of death in the U.S. Novartis hopes to grab a large share of a market expected to reach $14 billion by 2018.
The Utibron will compete with GlaxoSmithKline's ($GSK) Anoro and Breo Ellipta, as well as Boehringer's Stiolto Respimat in the combination drug COPD arena.
NVS launches Zarxio (Neupogen FoB) at ~15% price discount: #msg-116719512.
AVEO Oncology (NASDAQ:AVEO) today announced an exclusive, worldwide license agreement with Novartis for the development and commercialization of AVEO’s first-in-class, potent, humanized inhibitory antibody targeting growth differentiation factor 15 (GDF15), AV-380, and related antibodies, including modified or derivative forms of any such antibody (the “Product”).
nvs will move up. A deal with epgl coming!!!!
Novartis and Google need EPGL. They have known this for some time... EPGL tech is the key to their success. IMO
This is just the beginning...
Huge News for $NVS Novartis, $EPGL is the missing link to get #Smartcontactlens to market for real world everyday use! #TheStreet #BionicLens
EPGL and Novartis to Enter Discussions
IRVINE, Calif., July 30, 2015 /PRNewswire/ -- EP Global Communications, Inc. (Public OTC: EPGL) announced today that Novartis /Alcon executives have recently contacted the Company about discussing EPGL's electronic contact lens technologies. Discussions will begin as early as Monday, August 3, 2015. Novartis CEO Joe Jimenez has been quoted as saying that the coming electronic contact lens market could be worth tens of billions of dollars over the next several years. Novartis partnered with Google in 2014 to create "smart contact lenses" and has been very public about their desire to develop such contact lenses successfully.
As previously disclosed EPGL owns 100% of certain patents pending and jointly owns certain patents pending with CooperVision, Inc. EPGL also remains in negotiations with CooperVision, Inc. as well. "The technology speaks for itself and we intend to license it to all of the top companies, unless someone secures exclusivity," said Michael Hayes. "We expect to announce a deal in the near future with someone who is a leader in the ophthalmic or technology field. We are just at the beginning stages of the coming 'Vision Revolution' and things are beginning to heat up," Hayes added.
In an effort to be more specific about EPGL developments to the investment community, on August 3, 2015, EPGL will announce an innovative accessory device technology which every electronic contact lens wearer in the future could potentially need to own.
About EP Global Communications, Inc.
EP Global Communications, Inc. is an advanced technology research and development company for both medical devices and consumer electronics devices specializing in microelectronic mechanical systems (MEMS).
EPGL and Novartis to Enter Discussions
IRVINE, Calif., July 30, 2015 /PRNewswire/ -- EP Global Communications, Inc. (Public OTC: EPGL) announced today that Novartis /Alcon executives have recently contacted the Company about discussing EPGL's electronic contact lens technologies. Discussions will begin as early as Monday, August 3, 2015. Novartis CEO Joe Jimenez has been quoted as saying that the coming electronic contact lens market could be worth tens of billions of dollars over the next several years. Novartis partnered with Google in 2014 to create "smart contact lenses" and has been very public about their desire to develop such contact lenses successfully.
As previously disclosed EPGL owns 100% of certain patents pending and jointly owns certain patents pending with CooperVision, Inc. EPGL also remains in negotiations with CooperVision, Inc. as well. "The technology speaks for itself and we intend to license it to all of the top companies, unless someone secures exclusivity," said Michael Hayes. "We expect to announce a deal in the near future with someone who is a leader in the ophthalmic or technology field. We are just at the beginning stages of the coming 'Vision Revolution' and things are beginning to heat up," Hayes added.
In an effort to be more specific about EPGL developments to the investment community, on August 3, 2015, EPGL will announce an innovative accessory device technology which every electronic contact lens wearer in the future could potentially need to own.
About EP Global Communications, Inc.
EP Global Communications, Inc. is an advanced technology research and development company for both medical devices and consumer electronics devices specializing in microelectronic mechanical systems (MEMS).
Zarxio (Neupogen FoB) update: #msg-115571193.
NVS either made or is about to make a new 52 week high and I think an all time high again!!! Glad I stayed long with this beauty!!
Nice gains the last 3-4 days.
So much for the Grexit Euro effect? Nice news on the new drug approval too? NVS is like the energizer bunny, it just keeps going higher
NVS rallied and is heading back up ![]()
FDA approves Entresto (f/k/a LCZ696) for heart failure: #msg-115224110.
NVS acquires (private) Spinfex Pharma for $200M plus future considerations:
#msg-115006245
NVS Investor Day slide sets:
https://www.novartis.com/sites/www.novartis.com/files/2015-06-meet-the-management-1-group.pdf (overview)
https://www.novartis.com/sites/www.novartis.com/files/2015-06-meet-the-management-2-pharma.pdf (pharma)
https://www.novartis.com/sites/www.novartis.com/files/2015-06-meet-the-management-3-alcon.pdf (Alcon)
https://www.novartis.com/sites/www.novartis.com/files/2015-06-meet-the-management-4-sandoz.pdf (Sandoz)
https://www.novartis.com/sites/www.novartis.com/files/2015-06-meet-the-management-5-nibr.pdf (R&D)
Press release:
https://www.novartis.com/sites/www.novartis.com/files/2015-06-meet-the-management-media-release.pdf
Barron’s plugs NVS:
http://online.barrons.com/articles/novartis-looks-much-healthier-1430533899
Link to pipeline of NVS that I requested be sent to me from AACR 2015 NVS location:
http://www.novartis.com/innovation/research-development/clinical-pipeline/index.shtm
JUNO, NVS, settle CAR-T patent litigation: #msg-112436395.
Aduro, NVS ink immuno-oncology collaboration: #msg-112231501.
More good news from NVS biotech I am long in
In fact there were two good news items, one about a big move into immuno therapy to treat cancer!!!
In fact there has been a string of great news.
NVS News: New indication for Novartis bone marrow disorder drug cleared in Europe 03/17/2015 07:35:19 AM
NVS News: Wall Street Breakfast: Japan Marches Higher As Nikkei Breaches 19,000 03/13/2015 06:30:41 AM
NVS News: Cellectis: Finding Value In A Crowded CAR-T Space 03/10/2015 09:51:04 PM
NVS News: Ophthotech earns $50M Fovista milestone from Novartis 03/10/2015 07:48:59 AM
NVS News: Arrowhead buys Novartis' RNAi assets 03/05/2015 08:06:10
And now this:
http://seekingalpha.com/news/2373126-new-indication-for-novartis-bone-marrow-disorder-drug-cleared-in-europe
The European Commission approves Novartis' (NYSE:NVS) Jakavi (ruxolitinib) for the treatment of adult patients with polycythemia vera (PV) who are resistant to or intolerant of hydroxyurea. Polycythemia vera is a rare blood cancer associated with the overproduction of blood cells, principally red blood cells. It is caused by a mutation in the Janus kinase 2 (JAK2) gene. It affects ~180K people in the European Union.
The goal of treatment is to reduce the thickness of the blood and prevent bleeding and clotting. If left untreated, the disorder can lead to blood clots, stroke and heart attack. About 25% of PV patients develop resistance to or intolerance of hydroxyurea and are considered to have uncontrolled disease.
Jakavi is an oral inhibitor of the JAK1 and JAK2 tyrosine kinases. It was previously approved for the treatment of myelofibrosis, a disorder of the bone marrow in which the marrow is replaced by scar tissue.
Novartis licensed ruxolitinib from Incyte (NASDAQ:INCY) for development and commercialization ex-U.S. Incyte retains commercialization rights in the U.S.
NVS buying opportunity coming up?
http://www.buyins.net/tools/symbol_stats.php?sym=nvs
And news is out today
http://seekingalpha.com/news/2356756-ophthotech-earns-50m-fovista-milestone-from-novartis
Unless the SF keeps appreciating.(edit) Against the USD, that is.
Should be a 1-off event, IMO.
Thanks, We were beginning to suspect that. Any idea if it will continue?
Down sharply yesterday in Zurich (just like SYT and other Swiss multinationals).
The recent pop in NVS ADRs was due to the currency move by the Swiss National Bank. The same thing happened to SYT ADRs yesterday: #msg-109953954.
No idea, but that could be an interesting question and might have tripped a short covering, or some sort of currency wire or someshing? maybe the euopean price of the stock boosted the US dollar price??
How did it trade in terms of Swiss francs in Switzerland?
http://investorshub.advfn.com/boards/read_msg.aspx?message_id=109965356
As an old bowling coach of mine, circa 1976 told me
"When your good let em know it!!!!"
HELL YES!!!!! I love it when a plan comes together!!!!
I called it, right here, that NVS the huge Biotech Pharma firm would make a new 52 week and all time high this year of over $100/share. It broke out today!!! and made and held over $100/share
My NVS chart (More charts are here in my intro page http://investorshub.advfn.com/Ecomikes-Favorite-stock-picks-and-Stock-DD-topics-26971/ )
I love this one, nice news, thanks!!! Chart is showing an energizer bunny stock, glad I bought and held in 2009 go NVS!!!
I have more charts here
http://investorshub.advfn.com/Ecomikes-Favorite-stock-picks-and-Stock-DD-topics-26971/
45 % rise in net profit .
I wasnt asking if U knew what "market Cap" means in that TERM. What I was writing is that no Company seems to go past that market cap. In other words there are so many up too and close too 250B. That if lets say Roche, Sanfori, and Pfizer all did a merger it would be would 600B which would be twice Google.
For some reason no one can burst past the 250B. In reality, yes, it can go above 250 to whatever. There is some type of market restraint on all of them not allowing it. I think it is none want to get too big, thus they dump the cash in dividend, or spin off. Thois will keep novartis and roche PPS from rising. It cant do anything above a few %
Yes, I know what market cap means. There is no ceiling on the market cap of a pharma company, as you suggest.
Do you get what I mean about maket cap ? Goog is 350B.. But no pharm can get past 260B... None of them and there are 15 - 20 between 180 240B. Novartis throws out a hefty Dividend. Outside of that, you cant invest in them because of the market cap restriction that seem to be put in place by the investors or market makers.
Novartis is BIG behind the scenes in the new cell engineering going on in cancer. When the times comes, like last year when they proved it worked and terminally ill patients all got better, and into reminsion... etc..
Well, the RICH man will stop at nothing to save his life... 50M to do it, go ahead... Novartis, I think could double because of the base, The P/E.. EPS just keep on going There in again is y point 100 a share and CANT go up... 2.5B shares so they ant really split
Actually, NVS is considerably bigger than JNJ is pharmaceuticals. JNJ's market cap is bigger, but only about half of it is attributable to pharma.
|
Followers
|
18
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
430
|
|
Created
|
06/12/09
|
Type
|
Free
|
| Moderators | |||
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
