Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Yea, the partner left nothing to be desired…, 200k for me as well
I'll 2nd that.. " I lost a lot of money finding out truly how corrupt the fda and other government agencies truly are and it is sad. It makes me sad as an American." ..lost over 200k
PS.. our partners ceo didn't deal from the top of the deck.. that had an effect in many of our decisions...IMO
Great color, thanks!
TM,it seems there is more truth in those words than I like,
even though I will always believe RLF-100 is a safe effective life saving therapeutic that needs approval for numerous health conditions, I remain hopeful the world will open up, even if the USA approvals are last on the list.
Also, I clearly feel that NRXP needs to go much much lower, esp without a settlement w RLF, and or approvals by the FDA. .
Because that is not how our corrupt medical system works my friend….100% safe and effective Zyesami would negatively impact the pockets of their friends waiting to hire them as a consultant down the road… I lost a lot of money finding out truly how corrupt the fda and other government agencies truly are and it is sad. It makes me sad as an American. It needs fixed badly and Trump stirred the feathers and made things tough for ole Joseph our fearless leader….less
New All Time Low Today for Neuro ......
Do You Suppose things are going well with the negotiations ??
Or Not So Well ?
What is the possibility/probability of going under 2 in the near term future ?
https://stockcharts.com/h-sc/ui?s=NRXP&p=D&b=5&g=0&id=p57685788653
Not pretty charts.
https://stockcharts.com/h-sc/ui?s=NRXP&p=D&yr=0&mn=4&dy=0&id=p56160925541
The Department of Defense Just Got Caught Lying About Vaccine Injuries
https://emeralddb3.substack.com/p/the-department-of-defense-just-got?s=r
Why not approve a safe effective therapeutic by RLF / NRXP ???
Pandemic Lessons Learned: CDC Versus Natural Immunity
https://www.zerohedge.com/covid-19/pandemic-lessons-learned-cdc-versus-natural-immunity
"I’m beginning to wonder if CDC stands for Center for Data Control.
Those Recovered From COVID Are Best Protected
On March 1, the scientific journal Clinical Infectious Diseases published a peer-reviewed article titled “Risk of reinfection after seroconversion to SARS-CoV-2: A population-based propensity-score matched cohort study.” This Swiss study “observed a 94% reduction in the hazard of being infected among SARS-CoV-2 seropositive participants, when compared to seronegative controls, >8 months after serology assessment.”
This level of protection (natural immunity) from SARS-CoV-2 infection (94 percent) is comparable to that of the Pfizer vaccine but lasts longer (eight months and counting).
In a peer-reviewed article published in the journal Science Immunology on Jan. 25, scientists from Oregon Health & Science University showed in raw data that antibodies derived from previous COVID-19 infection are at least 10 times more potent than that generated by vaccination alone. "
ITS TIME FOR Approvals of AVIPTADIL and or ZYESAMI and or RLF-100
Fifth COVID-19 Vaccine Shot May Be Needed In Fall: FDA Official
https://www.zerohedge.com/covid-19/fifth-covid-19-vaccine-shot-may-be-needed-fall-fda-official
Is it a case of clot shots until you die from one ???
Or is it approval of a safe effective therapeutic like
AVIPTADIL/ZYESAMI/RLF-100 ?????
Shanghai Authorities Extend COVID Lockdown As Cases Continue To Surge
https://www.zerohedge.com/covid-19/shanghai-authorities-extend-covid-lockdown-cases-continue-surge
https://www.zerohedge.com/markets/shanghai-residents-struggle-obtain-food-medicine-citywide-lockdown-continues
And this is why the FDA needs to Approve a safe effective therapeutic like AVIPTADIL/ZYESAMI/RLF-100 quickly...
Plus, evidently, The Clot Shot is ineffective, and unsafe, or we would not see all the deadly side effects (VAERS), (Military Data Base), and the need to constantly have "just one more shot" of an experimental mRNA drug with unknown and known deadly long term and or short term side effects.
Shanghai Orders Staggered Lockdowns After Reporting Record New COVID Cases
https://www.zerohedge.com/covid-19/shanghai-orders-staggered-lockdowns-after-reporting-record-new-covid-cases
After weeks of panic-buying for fear that the Communist authorities would order another lockdown, President Xi has apparently tossed his "targeted" approach aside and ordered a lockdown in Shanghai, the country's largest city and its financial hub.
The city of 26 million will be locked down "in two stages" over the span of nine days as authorities try to quash surging COVID numbers. The lockdowns were ordered after the city reported a new record number of infections on Saturday.
During the lockdowns, public transit will be suspended, including ride-hailing services, according to an announcement from the city government released on its official WeChat account. Personal vehicles will be barred from the roads unless otherwise approved.
Basically you are screwed if you live in Shanghai.....But...
Where is the FDA approval for a safe effective therapeutic like
............AVIPTADIL....ZYESAME...RLF-100.......??????
China Orders 51 Million Into Lockdown As COVID Numbers Spike
https://www.zerohedge.com/covid-19/china-orders-51-million-lockdown-covid-numbers-spike
Will the FDA approve a safe effective therapeutic like
Aviptadil/zyesami ????
Or will the FDA approve more ineffective unsafe clot shots,
and then pay the MSM a Billion dollars to advertise they are
safe,effective,n free ???
China Locks Down 17.5 Million In Shenzhen As Daily COVID Cases Soar To 2-Year High
https://www.zerohedge.com/covid-19/china-locks-down-175-million-shenzhen-daily-covid-cases-double
USA/ROW needs a safe effective therapeutic like AVIPTADIL/ZYESAMI for Covid, and its approval is needed right now.....
FDA is foot dragging again while people suffer..
Shanghai Shutters Schools, Builds Hospitals As COVID Comes Roaring Back Across China, .....Worst Outbreak Since Wuhan
https://www.zerohedge.com/markets/shanghai-shutters-schools-builds-hospitals-covid-comes-roaring-back-across-china
Yes, Its Time for Approvals on AVIPTADIL and ZYESAMI....!!!
The VAX is NOT safe, NOT effective, and NOT free, and
it was falsely advertized..... as safe, effective, free...all BS.
UNREPORTED TRUTHS:90% of the Dead Received mRNA jabs in Britain...
https://alexberenson.substack.com/p/urgent-covid-infections-in-britain/comments?s=r
URGENT: Covid infections in Britain are rising again, and 90 percent of the dead are vaccinated. Have mRNA jabs ruined our chance at herd immunity?
Alex Berenson Mar 9 930 612
New figures from Britain raise bright red flags about the direction of Covid in wealthy countries that used mRNA and DNA shots to attempt to defeat the coronavirus last year.
Hospitalizations and deaths remain stubbornly high and overwhelmingly occur in vaccinated people. In February, 90 percent of the 1,000 Britons who died each week of Covid were vaccinated.
THE RLF / NRX THERAPEUTIC IS PROVEN SAFE AND EFFECTIVE....
WHY IS THE GOV NOT APPROVING ITS USE ?????????
Go Figure eh
Covid BA.2 Variant in Hong Kong / Australia is ..............
another very important reason for fast AVIPTADIL/ZYESAMI Approval !
https://pubmed.ncbi.nlm.nih.gov/35271728/
https://www.zerohedge.com/covid-19/public-health-experts-have-new-covid-boogeyman-called-stealth-omicron
https://www.wsws.org/en/articles/2022/03/10/auco-m10.html
When will the FDA and those in power wake up to a safe effective therapeutic like Aviptadil and or Zyesami. ???
Yes, PW...thank you again.
https://ir.stockpr.com/neurorxpharma/sec-filings-email/content/0000950142-22-000997/es220233694_4-besthof.html
I guess you are referring to the new acting CEO.
All good so far, but waiting for more details and the certainty
that knuckleheaded surprises remain in the past.
Joe, that attorney has $250K in RLFTF, so he has a lot of skin in the game. So, he is closely watching events as they unfold. I am invested in both RLFTF and NRXP, which some traders refer to as potentially excessive correlation. Although my skin in NRXP is minimal.
PW....thank you for sharing..
Greatly Appreciated.
Per the discord site...
The hearing date for both motions has been set for June 8,2022 at 11am from information I received. My thoughts are that given the length of the delay on the hearing date, coupled with the news of JJ’s retirement from the ceo position, I believe the parties are giving themselves time to continue settlement negotiations and gather/process information. People like JJ who have had success being a certain way don’t usually just suddenly stop their behavior so again to not be a Debbie Downer I’m being cautiously optimistic with an inside smile on my face until I see a judgement signed by the Judge. Just my opinion. Hopefully by June, we will see a lot of great things happening with everything in our pipeline all at once.
Do you suppose perhaps that the newest Consultant for Neuro is
considering this a personal treatment option ???
NRx Pharmaceuticals: Potential For First In Bipolar Depression Suicidal Ideation Treatment, Along As Covid-19 Play
https://seekingalpha.com/article/4489385-nrx-pharmaceuticals-potential-for-first-in-bipolar-depression-along-as-covid-19-play
Good luck and GOD bless,
Additional Memoralizing......of interesting comments from y board.
Zye of Relief 9 hours ago
My personal take on this: JJ is stepping out and NRxP focusing on the U.S. It really says mediation is going well for RT, as evidenced by the firing of Sir Bowtie and the forfieture of its claim to Canadian and Israeli territories. And now we wait,"FOR THE REST OF THE STORY" LONGER AND STRONGER EVERYDAY-1 ?? unnecessarily JJ
Bullish
Serge 1 hour ago
Robert M. Califf and Anthony S. Fauci
March 3, 2022
Page 2
Fauci
touted ZYESAMI as a promising treatment for COVID 19. 8 However, the drug remains
largely unavailable, and we are told FDA refuses to review the data of NRx’s EUA until the
National Ins titute of Health ( completes clinical trials of ZYESAMI later this year. 9
Two
years into a pandemic and with a death toll exceeding a reported 900,000
Americans, it is unacceptable that the FDA and NIAID are needlessly delaying a treatment for
late stage COVID 19 with a remarkable track record of success. This bureaucratic dragging of
your feet appears in stark contrast to the expedited review of other treatments like remdesivir,
Molnupiravir, Paxlovid and the COVID 19 vaccines. The FDA’s disparate review processes for
different treatments that appears to favor large manufacturers is troubling.
To
better understand the FDA’s decisions regarding an EUA for ZYESAMI, we
respectfully request the following information:
1. Please provide a timeline of FDA and NIAID actions to review ZYESAMI as an
emergency treatment for COVID 19.
2. Please provide documentation of any communications between FDA or NIAID and
physicians or hospitals that are utilizing ZYESAMI under Right to Try.
3. Has the FDA ever accepted the data of ongoing clinical trials when issuing an EUA for a
treatment for COVID 19? If so, please provide the treatment and its EUA.
4. Please explain why the FDA refuses to review ZYESAMI data until completion of a
clinical trial.
5. For COVID patients tha t received remdesivir and steroids but did not recover, what is the
FDA and NIAID’s current treatment recommendation?
Please
provide this material as soon as possible but no later than 5:00 p.m. on March 17
2022. Thank you for your attention to this ur gent matter.
Sincerely
Ron Johnson Ted Cruz
United States Senator United States Senator
8 Press
WONG 17 hours ago
It's about time.
In a recent letter co-authored by Sen. Ted Cruz and Reps. Andy Biggs and Chip Roy, Sen. Ron Johnson demands answers from the Food and Drug Administration (FDA) and the National Institute of Allergy and Infectious Diseases (NIAID) as to why they have refused to even entertain the emergency use authorization request or even review the data from ZYESAMI manufacturer NRxPharmaceuticals on its efficacy against COVID.
Bullish
Evidently, it appears, the mediation, and all the lawsuits, has finally put a spotlight on jj that the BOD could not accept !
Gotta wonder,.. how will the rest of the mediation now transpire ???
And what will the RLF / NRX relationship now become...???
But the story today seems to be going from CEO to Consultant.
Will RLF hold a Ticker tape parade ? time and date TBD ?
Is there a buyout offer in the works?
Good luck and GOD bless,
So it is not clear if Jonathan Javitt, MD, MPH is still the Chairman of the Board of Directors?
https://ir.nrxpharma.com/news-events/press-releases/detail/120/nrx-pharmaceuticals-announces-leadership-transition
Good luck and GOD bless,
BigT really ?!?
That was on Friday, February 11, and then Saturday went by with no word from the company.
Mr. Akins then upgraded his LinkedIn account so he could message the manufacturer’s founder and CEO Jonathan C. Javitt, MD, for help directly.
And it worked.
“Within 45 minutes he called on my cell phone and said he would move it forward,” recalled Mr. Akins early this week. “Twenty minutes later it was fully approved.”
The closest location of the drug was four hours away in South Carolina, so off he traveled on Superbowl Sunday, returning eight hours later with twice the dosage of the intravenous medication requested by the hospital.
Once hospital staff completed training themselves on the proper administration of the experimental drug, Ms. Bonville was treated during a three-day period with progressively more infusions of the medication.
“Forty-eight hours after the treatment, her lungs almost looked back to normal,” said Mr. Akins. While she’s still recovering, he said, “she’s out of the woods.”
“A lot of people don’t understand they have the ability to advocate for patients and the right to try experimental medicines,” said Mr. Akins.
“Everything just fell into place. It’s an amazing story. It’s just incredible,” he said.
https://bakercountypress.com/2022/02/lauds-mothers-covid-recovery-after-experimental-drug-infusion/
Good luck and GOD bless.
nice jj...but do you have a date? Or A link ? a source ?
PIT PRESS
A Home >> The Press >> News >> Lauds mother's COVID recovery after experimental drug infusion
LAUDS MOTHER'S COVID RECOVERY AFTER EXPERIMENTAL DRUG INFUSION
Macclenny business owner Mark Akins is calling his mother's recovery from COVID-19 pneumonia this
month nothing short of "incredible."
He credits an experimental drug undergoing clinical trials from NRx Pharmaceuticals called avipdatil with
her recovery. Getting her the medication was no easy feat, however.
Bonnie Bonville's doctors at Piedmont Mountainside Hospital in Jasper, GA had given the 75-year-old
steroids but avoided anti-body treatments due to her history of kidney failure. After an arthritis medication to
suppress her immune system led to only a minor improvement of her condition, her doctors were out of
options, said Mr. Akins.
His sister then began researching online for alternative treatments and uncovered stage 3 clinical trials
ongoing for avipdatil.
The so-called "Right to Try Act," signed into federal law in 2018, gives those facing life-threatening
conditions the right to access treatments unapproved by the Food and Drug Administration. Part of the
process is petitioning the hospital for approval of the treatment. That was started and then contact was
made with the drug manufacturer.
That was on Friday, February 11, and then Saturday went by with no word from the company.
Mr. Akins then upgraded his Linkedin account so he could message the manufacturer's founder and CEO
Jonathan C. Javitt, MD, for help directly.
And it worked.
"Within 45 minutes he called on my cell phone and said he would move it forward," recalled Mr. Akins early
this week. "Twenty minutes later it was fully approved."
The closest location of the drug was four hours away in South Carolina, so off he traveled on Superbowl
Sunday, returning eight hours later with twice the dosage of the intravenous medication requested by the
hospital.
Once hospital staff completed training themselves on the proper administration of the experimental drug,
Ms. Bonville was treated during a three-day period with progressively more infusions of the medication.
"Forty-eight hours after the treatment, her lungs almost looked back to normal," said Mr. Akins. While she's
still recovering, he said, "she's out of the woods.
"A lot of people don't understand they have the ability to advocate for patients and the right to try
experimental medicines." said Mr. Akins.
"Everything just fell into place. It's an amazing story. It's just incredible," he said.
Good luck and GOD bless,
Memorializing an interesting comment or two........from the y board that seems appropriate here. I wish NRX could provide at least one FDA success story of an approval for what they were hired..... Sadly, there is still not any indication that NRX is even willing to acknowledge any wrong doing at all or any compromise ...
G-Money yesterday
This is heading to court and needs to end in divorce. No point in mediating new terms. You can't trust jj/nrxp to act honorably. Court will show that zyesami is fundamentally the Aviptadil formulation used for RLF-100. RLFTF can then use the trademarks of RLF-100 and zyesami interchangeably. That's important because the next move for RLFTF is to get listed as a sponsor on current trials. Next, get RLF -100 listed synonimously with zyesami. Finally, if possible, take over as primary sponsor of the trials and get all reports directly from the source. No more nrxp, no more jj, no more partnership. Upon approvals market globally as RLF-100. That's the best path for moving forward.
woodcrest yesterday
86. In September 2020, the same month the Collaboration Agreement was signed, Relief was forced to write to NeuroRx's counsel in an attempt to resolve Dr. Javitt's outright refusals to share basic information relevant to the parties' collaboration. Information that NeuroRx was wrongfully withholding included, but was not limited to, the following:
a. Information relevant to an audit and a pause in enrollment for intravenous aviptadil trials that had apparently occurred at Houston Methodist – the hospital that had generated tremendous early access treatment results. Relief was surprised to learn about these issues second-hand;
b. Information relevant to allegations by certain research organizations that NeuroRx "faces a growing number of challenges executing the aviptadil" trial and that it may be falling short of "protect[ing] patient safety and meet[ing] [its] intended clinical and business objectives of the study";
c. Information concerning the specific formulations of aviptadil that NeuroRx was using pursuant to the collaboration. As Relief expressed at the time, it was concerned that NeuroRx might be using formulations that "evade or lie outside the scope of Relief's issued intellectual property" – which would be harmful "to the overall commercial value of the drug"; and,
d. Information pertaining to whether Dr. Javitt was potentially diverting Relief's funds to his relatives. In particular, Relief learned that Dr. Javitt was directing funds provided by Relief to PillTracker, a company headed by his son, Zachary Javitt, that has no expertise relevant to aviptadil. PillTracker is a company focused on improving patient compliance with pill-based therapeutics. However, aviptadil cannot be administered via a pill (which is why the parties specifically contracted to develop intravenous and inhaled – and not pill – formulations of aviptadil). Thus, Dr. Javitt's diversion of funds to his son's company alarmed Relief. Likewise, Relief also explained that it was concerned by the possibility that Dr. Javitt was directing funds to Gail Javitt, Dr. Javitt's sister.
Neuro went under $2.50 today....... jj might be having some type of issue or panic attack, with postings again on the net ..... someone sold some Neuro in volume with a big fast drop today.. How about a public release of info for the stcok market and not just stock twits.
https://stocktwits.com/Jonathan_Javitt/message/441379643
COMMENTS FROM ANOTHER Board@yoh@@
yoke 16 minutes ago
?? JJ drunk posting fantasy on social media and the price tanks
woodcrest 21 minutes ago
$RLFTF conversation
(Javitt on Stock Twits 33 minutes ago.. The stress is driving him crazy.)
MERICA 21 minutes ago
Is this drop the dilution? Or people understanding Javitt tried to covered up yet another FDA rejection with BTD??
Is neuro planning on writing a short novel ?
https://iapps.courts.state.ny.us/nyscef/ViewDocument?docIndex=s4c9kbJKs2G12L31_PLUS_ru48w==
At the last minute... Fiction perhaps ?
j junior...Pitifully inadequate at this stage.
telling us what has been known for a long time.
Where are the approvals ??
Where is the willingness to honor signed agreements.??
Earlier treatment leads to improved outcomes. Aviptadil showed a manageable safety profile, with no unexpected serious adverse events.
https://www.nrxpharma.com/wp-content/uploads/2022/03/ZYESAMI-Clinical-Trial-Results.pdf
Good luck and GOD bless,
Joe... We have seen similar results posted.... But isn't this latest a new release and a new addendum to the FDA for EUA evaluation? The report included a statement that this info will reset the 60 day time clock for the FDA evaluation. Thoughts? Thanks....
Unfortunately this info was all known to RLF over a year and a half ago....
Can Neuro get some approvals and be a team player for RLF ???
EVIDENTLY NOT.
Clinical Trial Results!
RECOVERY AND SURVIVAL IN COVID-19 RESPIRATORY FAILURE, WHEN TREATED WITH AVIPTADIL
Aviptadil helps to resolve respiratory failure and
extend survival at tertiary care sites in COVID-19
https://www.nrxpharma.com/wp-content/uploads/2022/03/ZYESAMI-Clinical-Trial-Results.pdf
https://www.nrxpharma.com/ceo-update-zyesami-clinical-trial-results-covid-aiv-presented-at-peer-reviewed-congress-on-retroviruses-and-opportunistic-infections/
Memorializing some interesting Comments from Another board @y
Sylvia 2 days ago
$RLF.SW conversation
$NRXP
JJ wants another extension!?!
https://iapps.courts.state.ny.us/nyscef/ViewDocument?docIndex=HiXvj_PLUS_Vs1wAxQm/PPWIalg==
_____________________________________
REPLIES.............
ted 2 days ago
Yes, Saliva, unlike Ram, he's actually busy.
....................
WONG 2 days ago
@ted Ram has acquired APR. Giving instant revenue in-house R&D, a promising pipeline and existing logistical and manufacturing relationships.
Ram acquired Advita and all of there IP and expertise with inhaled Aviptadil. It has already produced an ODD for Sarcoidosis from the FDA and an upcoming patent to treat CIP for cancer patients undergoing immunotherapy treatment.
Ram has created and is moving forward with an ADR program that will uplist Relief to NASDAQ in 2022.
Ram is running trials with both Sentinox and Aviptadil to combat Covid. In Switzerland and Italy.
Ram has helped with the
development and commercialization for ACER-001 that should receive an NDA this June. The deal is as follows:
The companies will split net profits from Acer’s territories 60:40 in favor of Relief. Relief will also license the rights for the rest of the world, where Acer will receive from Relief a 15% net sales royalty on all revenues received in Relief’s territories.
Ram has filed and recieved a trademark for RLF-100TM with the United States Patent and Trademark Office (“USPTO”). Something JJ failed in doing.
Ram has signed a collaboration agreement with InveniAI a U.S. based company that has pioneered the application of artificial intelligence and machine learning across biopharma and other industries, in order to identify promising drug candidates to treat rare and specialty diseases.
Seems he has been busy.
................
Zye of Relief 2 days ago
@ted jj has accomplished nothing he was hired to do. Do we have adequate trial sizes, locations and qualified investigators? NO! Do we have adequate clear cut positive trial data? NO! Do we have an EUA? NO! Do we have BTD? NO! Do we have an NDA? NO! Do we have an EUA in Georgia, data from Goergia, data from inhaled..... DATA AT ALL? NO, EMPHUCKINPHATICALLY NO! Sue him into oblivion, if your contractor did this to you, there would be no hesitation on your part. His failures, and it pains me say this and take this position as I was an enormous fan of sir Bowtie. We are in this place as a direct result of his failures, his and his alone. GREED, SHORTCUTS and FALSEHOODS! LONGER AND STRONGER EVERYDAY-1 ??
Sad. POS JJ professional scam artist. Sad for GEM. Sad for all those that suffered in need and thru this person's greed/incompetence's. billion$ lost in RLF/NRX. Lives too, IMO.
These kinds of criminals seldom are punished, but a dude with a joint goes to jail.
I hope he gets cancer in his lying mouth and demented brain, then burns in Hell. JMHFO...GLTA somewhere else.
Another group of very informative thoughts and interesting posts over on another board at the_y@h@@ boards by
Brian A 4 hours ago
So there are are lot of comments regarding Neuro’s recent, and successful, requests for an “extension of deadlines.”
These deadlines you’re referring to have NOTHING to do with the Court Ordered attempts at Mediation. NOTHING!!
If you read the documents, it’s pretty clear the deadlines being referred to are with regard to the lawsuit filed by Relief against Neuro, and Dr. Javitt personally. Just because you’re involved in mediation doesn’t mean your legal team stops work around the pending lawsuit(s).
The deadline extensions specifically refer to Motions #1 and #2 before the Supreme Court of New York. So what are those motions? I’m glad you asked!
Motion #1 is a motion to Strike the allegations made by Relief in its Complaint against Dr. Javitt for a) sabotage, b) apparent nepotism, and c) that Dr. Javitt issued a “series of misleading statements to the press to portray himself as the inventor of the idea of using Aviptadil to treat COVID-19.”
Motion #2 is a Notice of Motion to Dismiss the complaint against Dr. Javitt due to a lack of personal jurisdiction, or on the merits.
The Judge ordered documents relating to these Motions to be submitted to the Court by certain dates. Neuro’s attorneys filed deadline extension requests in order to have more time to gather and submit documentation in support of Motions #1 and #2. The Judge agreed, and granted Neuro’s legal team additional time to submit the information. They were given one additional week.
Again…this has NOTHING to do with the Mediation efforts that are purportedly underway. Neither side will reveal ANYTHING about Mediation until BOTH sides have either reconciled one way or another, OR determined that efforts toward Mediation are fruitless.
It’s as simple as that!
Sign in to post a reply.
>>>>>>>>>>>>>>>>>
Replies.......
Brian A 1 hour ago
@G-Money Thanks, and I agree with you. Both motions are designed to sever / relieve Dr. Javitt of all personal liability with regard to his actions and comments. I’m hopeful both motions fail, because like you, I believe if he can’t hide behind LLC, he will clearly not want to face a jury for any number of reasons, most especially due to his credibility issues.
Brian A 1 hour ago
@BBB Thanks very much, and I couldn’t agree more with your assessment.
G-Money 1 hour ago
Nice synopsis. If either motion is granted then jj can hind behind llc if this goes to trial. Hopefully the motions are denied. He is FAR less likely to take his chances with a jury trial if he is personally liable. Sadly he will have to be ordered by a court to behave honorably.
Interesting post from another well read over board @youwh@@......
Kelvin 4 hours ago
If you look at the time when Relief and NeuroRX are still Honey buddy.
JJ indicated RLF-100 (Aviptadil) is a patented formulation of Vasoactive Intestinal Polypeptide (VIP) that was originally developed and is currently marketed in Europe for the treatment of erectile dysfunction.
=======================
On Mar. 25, 2020:
RLF-100 (Aviptadil) is a patented formulation of Vasoactive Intestinal Polypeptide (VIP) that was originally developed and is currently marketed in Europe for the treatment of erectile dysfunction. VIP is known to be highly concentrated in the lung and to inhibit a variety of inflammatory cytokines. Aviptadil was awarded Orphan Drug Designation in 2001 by the US FDA for treatment of Acute Respiratory Distress Syndrome and in 2005 for treatment of Pulmonary Arterial Hypertension. Aviptadil was awarded Orphan Drug Designation by the European Medicines Agency in 2006 for the treatment of Acute Lung Injury and in 2007 for the treatment of Sarcoidosis. Both the US FDA and the EMEA have granted Investigational New Drug licenses for human phase 2 trials of Aviptadil. Aviptadil is Vasoactive Intestinal Polypeptide (VIP), a naturally-occurring peptide hormone that is known to be concentrated in the lungs. VIP has been shown in five species of animal models to have potent effect in models of ARDS and Acute Lung Injury. In these models, Aviptadil has shown potent anti-inflammatory and specifically anti-cytokine activity in the lungs.
https://www.nrxpharma.com/582541-2/
============================
After everything went sour, look what JJ said about RLF-100(Aviptadil) that Relief hold the patent on it
============================
RADNOR, Pa., Jan. 3, 2022
In our previous guidance to investors, we advised that there were no filed patents protecting the manufacture of ZYESAMI. With the filing of this patent and the inventions described within, we at NRx believe we have a path to a long term commercial life for a stable and pharmaceutically active form of VIP as an innovative drug," said Prof Jonathan Javitt, MD, MPH, Chairman and CEO of NRx Pharmaceuticals. "Although, for obvious reasons, our initial focus has been the use of VIP in lung disease caused by COVID-19, we are now considering potential use of ZYESAMI in non COVID-related lung disease, liver disease, eye disease, and organ transplantation. We are deeply grateful to our partners at Nephron Pharmaceuticals for working with us and embracing the painstaking work required to bring a seventy-year-old dormant drug to life."
https://finance.yahoo.com/news/nrx-pharmaceuticals-files-provisional-patent-121600329.html
=============================
YOU ALL CAN JUDGE FOR YOURSELF, MAYBE JUDGE Schecter CAN LOOK INTO IT TO SEE HOW THE MAN LIES
I am thinking that secrecy is the security that will save face. We will probably not be told unless the negotiations are fruitless.
Together they are stronger.... so all we can hope is that pride does not spoil the prize.
TB, please let us know the progress of the fraud
investigations ... TIA
|
Followers
|
52
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
1124
|
|
Created
|
12/15/20
|
Type
|
Free
|
| Moderators ProfitScout jedijazz | |||





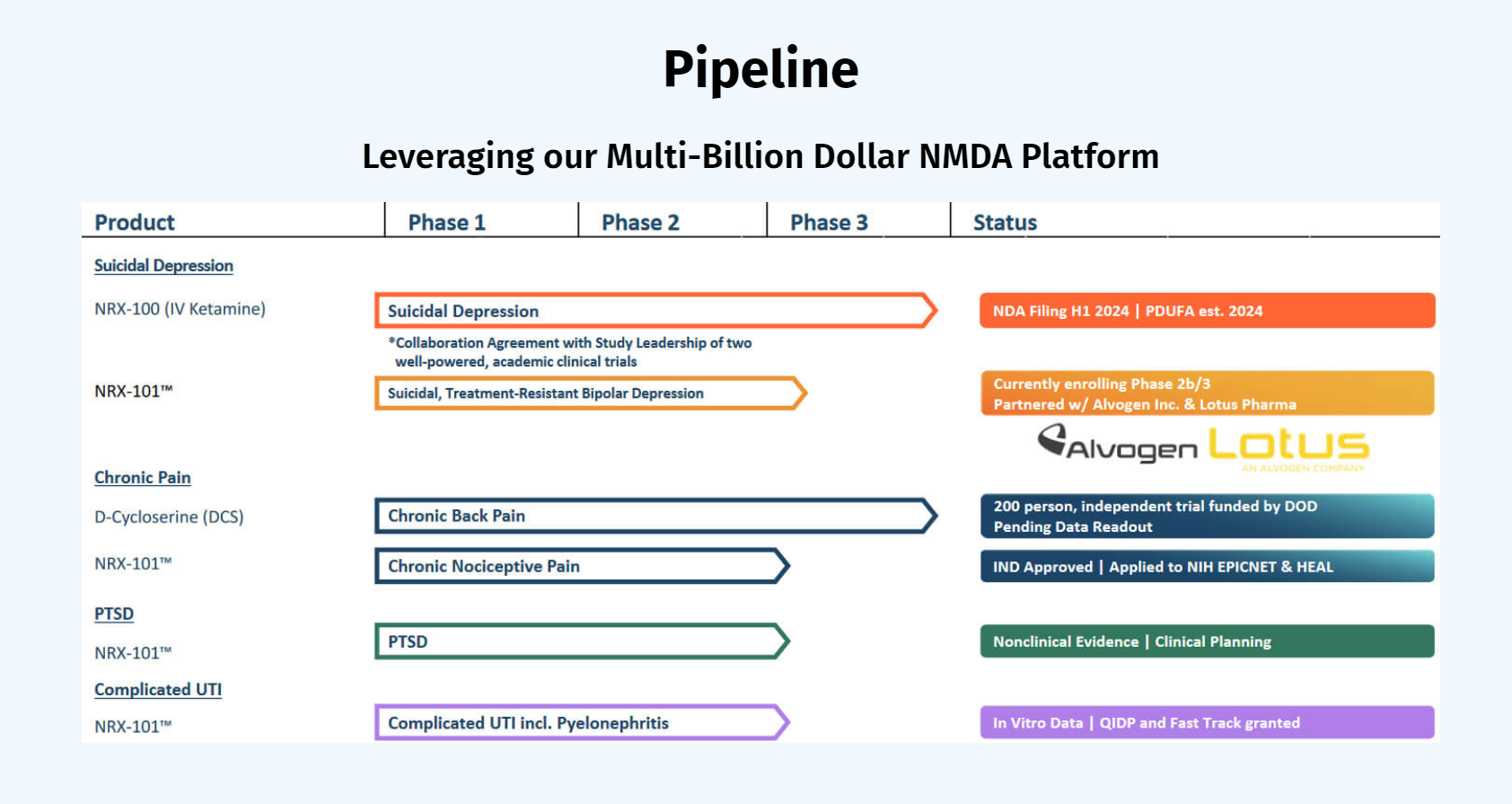
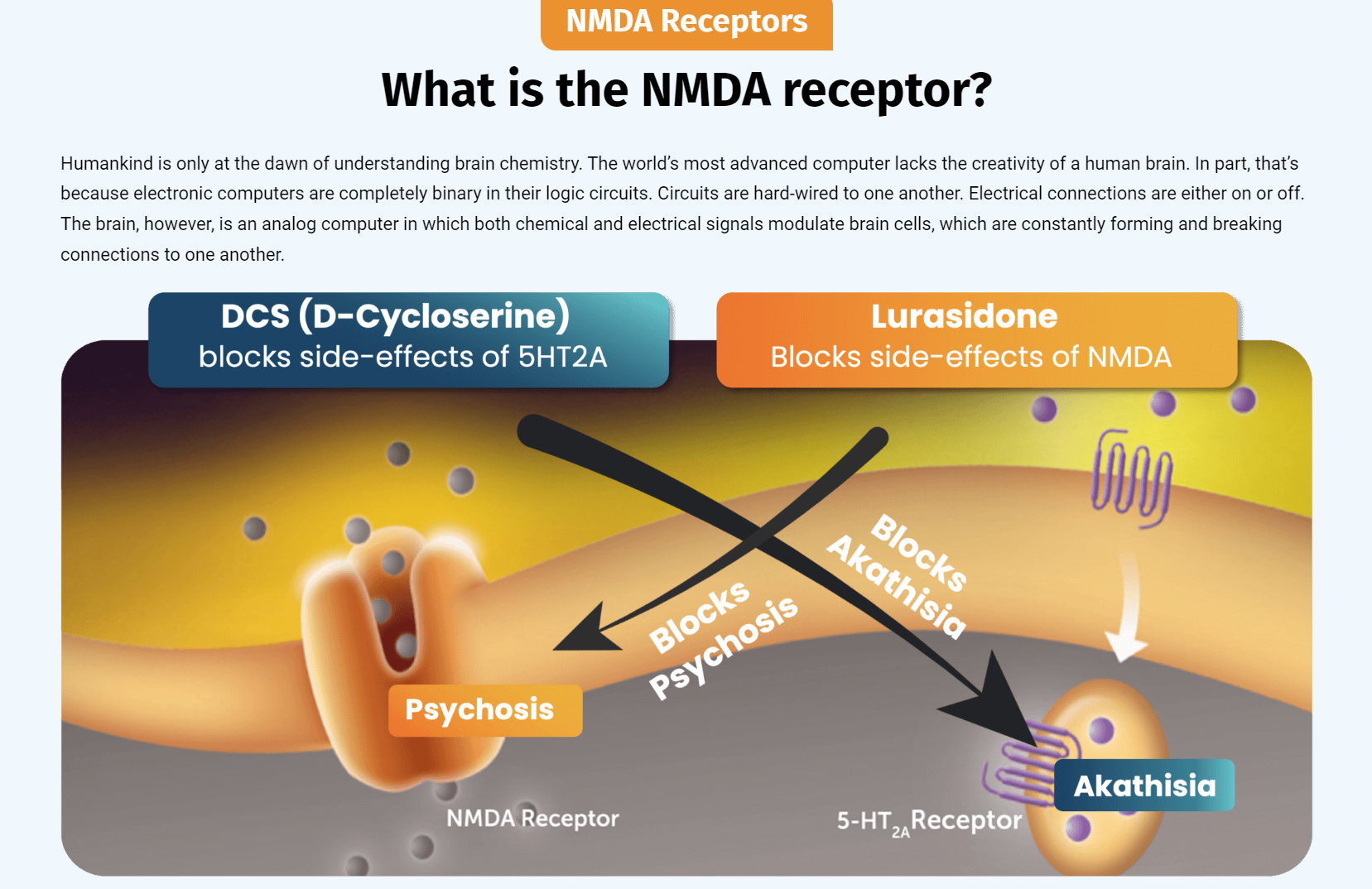







Both drugs demonstrated > 50% response for treating depression. NRX-101 demonstrated a mean 76% reduction in symptoms of akathisia compared to lurasidone that was sustained over 42 days (Effect...

New data demonstrates no impact of NRX-101 on gut or vaginal flora – considered primary causes of pseudomembranous colitis due to C difficile and vaginal yeast infections NRX-101 previously...
Formulation based on prior patents by NRx founder Achieved pH neutral formulation of ketamine, potentially enabling both intravenous (IV) and subcutaneous (SQ) administration Company expects...
Data transferred for independent statistical analysis Top-line data expected in April 2024 RADNOR, Pa., April 8, 2024 /PRNewswire/ -- NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) ("NRx...
Four potential near-term milestones, including data from two clinical trials, an NDA filing and an upcoming share dividend -- 50% reduction in corporate overhead and 25% reduction in overall net...
Four potential near-term milestones, including data from two clinical trials, an NDA filing and a share dividend RADNOR, Pa., March 28, 2024 /PRNewswire/ -- NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) ("
NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) ("NRx Pharmaceuticals", the "Company"), a clinical-stage biopharmaceutical company, today announced that it will release its fourth quarter and full year...
NRx Pharmaceuticals, Inc. (Nasdaq:NRXP) ("NRx Pharmaceuticals", the "Company"), a clinical-stage biopharmaceutical company, today announced that Dr. Jonathan Javitt, Chairman and Chief Scientist...
NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) ("NRx Pharmaceuticals", the "Company"), a clinical-stage biopharmaceutical company, today announced that it will release its fourth quarter and full year...
61.4% (56,781,354) of eligible shares voted 94.4% of votes were cast in favor of the resolution RADNOR, Pa., March 21, 2024 /PRNewswire/ -- NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) ("NRx...
NRx Pharmaceuticals Board of Directors has authorized its Chairman, CEO, and management to take all necessary steps to affect the Dividend and Royalty Rights to NRXP Shareholders and applicable...
Initiative is intended to protect shareholder value through continued compliance with Nasdaq listing rules and elimination of naked short sales positions in the Company's securities The Company is...
Company to receive first allocation of ketamine for sale by month end Partners preparing to ship IV Ketamine to full range of customers via 503a and 503b pharmacies NRx Pharmaceuticals and HOPE...
Marks a major step in the development of what could be the first drug approved for Suicidal Bipolar Depression The study database is being cleaned and locked; statistical analysis and top-line...
NRx Pharmaceuticals received approximately $1.0 million in cash from an existing investor Shares were sold at $0.38, a 26.7% premium the recent share offering, along with one common 5-year warrant...
In the news release, NRx Pharmaceuticals, Inc. Announces Pricing of $1.5 Million Underwritten Public Offering of Common Stock, issued 27-Feb-2024 by NRx Pharmaceuticals, Inc. over PR Newswire, we...
NRx Pharmaceuticals, Inc. (Nasdaq: NRXP), ("NRx Pharmaceuticals" or the "Company"), a clinical-stage biopharmaceutical company, today announced that it intends to offer to sell shares of its...
Presentation available on NRx Pharmaceuticals website at https://www.nrxpharma.com/hope-therapeutics/ KEY UPDATES ARE AS FOLLOWS: NRx management is proposing to award 50% of founding shares in...
DISCLAIMER:
Nothing in the contents transmitted on this board should be construed as an investment advisory, nor should it be used to make investment decisions.
There is no express or implied solicitation to buy or sell securities.
The author(s) may have positions in the stocks or financial relationships with the company or companies discussed and may trade in the stocks mentioned.
Readers are advised to conduct their own due diligence prior to considering buying or selling any stock. All information should be considered for information purposes only.
No stock exchange has approved or disapproved of the information here.
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
