Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
I don't short low priced stocks as the risk to reward isn't there.
I would look for an entry once news is released though.
Rarely worth holding in anticipation these days. I still don't short though.
Be careful if holding a short position,if news hits like rumored, short profits could avaporare in a blink of an eye.
Be careful on these stocks below $1 moving up on no news.
Could be someone generating volume and interest to sell out a position.
Keep it on watch for news but be weary that someone with a substantial position may just be creating a way for them to sell.
Not lookin good for Neuro or .....a guy that is a "consultant" , .....
Time to settle with RLF and pronto ?
https://ir.stockpr.com/neurorxpharma/sec-filings-email/content/0000950142-22-002222/eh220269223_8k.htm
Notice of Delisting or Failure to Satisfy a Continued Listing Rule or Standard; Transfer of Listing.
On July 12, 2022, the Company received a written notification (the “Notice”) from the Nasdaq Stock Market LLC (“Nasdaq”) indicating that the Registrant was not in compliance with Nasdaq Listing Rule 5450(a)(1), as the Registrant’s closing bid price for its common stock was below $1.00 per share for the last thirty (30) consecutive business days.
Another New CEO for Neuro Installed !!!
Call me skeptical at best.
A desperate attempt to re float a sinking ship ??
https://finance.yahoo.com/news/nrx-pharmaceuticals-announces-appointment-stephen-111500208.html
NRx Pharmaceuticals Announces the Appointment of Stephen Willard, Chief Executive Officer and Director
Wed, July 13, 2022 at 6:15 AM
In this article:
NRXP
-8.72%
NRXPW
-3.85%
Extensive experience in Law, Finance, and Management of Public and Private Biotechnology Companies
Proven track record of creating value for shareholders
National Science Board Presidential Appointee, 2018-2024
Former roles at Federal Deposit Insurance Corporation (FDIC) and E*Trade Financial
RADNOR, Pa., July 13, 2022 /PRNewswire/ -- NRx Pharmaceuticals, Inc. (Nasdaq: NRXP): ("NRx Pharmaceuticals", or the "Company"), a clinical-stage biopharmaceutical company, today announced the appointment of Stephen Willard, as its Chief Executive Officer ("CEO") and a member of the Company's Board of Directors. The Company's interim CEO, Robert Besthof, will continue to support the Company and return to his roles as Head of Operations and Chief Commercial Officer.
Stephen Willard, Chief Executive Officer, Director, NRx Pharmaceuticals, Inc.
Stephen Willard, Chief Executive Officer, Director, NRx Pharmaceuticals, Inc.
@NRXP Board Appoints BioPharma Veteran and National Science Board Presidential Appointee to Lead NRx Pharmaceuticals, Inc.
"We are delighted to have attracted a candidate like Steve whose background and experience align with NRx Pharmaceuticals' current needs. Steve's passion to provide breakthrough therapies to address critical unmet needs positions the Company for success," said Patrick Flynn, a member of the Company's Board of Directors. "We look forward to Steve's leadership of the Company in the next stages of growth to benefit patients, our shareholders, and the broader NRx team."
Mr. Willard brings a wealth of experience in the management of publicly traded biotechnology companies, together with his background in law and finance. Most recently, Mr. Willard served as CEO of Cellphire Therapeutics, where he grew the company and shepherded their revolutionary human platelet platform through key clinical trials, growing the company and significantly increasing the share price. Prior to Cellphire, he served as CEO of publicly traded Flamel Technologies now known as Avadel Pharmaceuticals. Mr. Willard is currently serving a six-year term from 2018-2024 as a presidential appointee to the National Science Board.
Mr. Willard's career in financial services includes government service as Associate Director of the Federal Deposit Insurance Corporation (FDIC), where he served in the United States Senior Executive Service (SES) from 1991-1994, and on the board of E*Trade Financial Services from 2000-2014. He has practiced law in New York, London, and Washington, D.C. Mr. Willard earned his undergraduate degree from Williams College and attended Yale University where he earned a JD in law.
I sent an email to the NRX "Right to Try" page to see if they are still allowing its use. We shall see.
All this is so strange and conflicting because right at the beginning of 2022 we were informed of this:
NRx Receives Initial Report of Patient Safety and Survival from Right to Try use of ZYESAMI® (aviptadil) during Omicron Surge
-A Southwestern hospital has reported safety data collected under the Federal Right to Try law indicating that 16 of 19 patients (84%) with COVID-19 respiratory failure treated with ZYESAMI® by Dec 31, 2021 have survived the ICU.
-No Serious Adverse Events were reported
-Patients were treated during the Omicron Surge
-Patients were treated at first onset of respiratory failure after exhausting remdesivir and other approved therapies
-Data are being included provided by NRx to FDA in support of ongoing application to FDA for Emergency Use Authorization
https://www.prnewswire.com/news-releases/nrx-receives-initial-report-of-patient-safety-and-survival-from-right-to-try-use-of-zyesami-aviptadil-during-omicron-surge-301468389.html
So, the question is, what suddenly caused the positive results to flip to negative? Will this be addressed during the shareholders meeting?
This was sent out by NRX: (In part)
You are cordially invited to attend the 2022 Annual Meeting of Stockholders of NRx Pharmaceuticals, Inc. to be held on July 18, 2022, at 11:00 a.m., local time, in virtual format at:
https://www.cstproxy.com/nrxpharma/2022
NRx Pharmaceuticals Provides Update on Breakthrough Therapy Designation (BTD) Request for ZYESAMI® (aviptadil)
JUNE 10, 2022 10:47PM EDT
Download as PDF
RADNOR, Pa., June 10, 2022 /PRNewswire/ -- NRx Pharmaceuticals, Inc. (Nasdaq: NRXP, NRx Pharmaceuticals), ("NRXP" or the "Company"), a clinical-stage biopharmaceutical company, today announced that the U.S. Food and Drug Administration (FDA) denied the Breakthrough Therapy designation (BTD) request for ZYESAMI® (aviptadil) and emphasized its focus on the company's NRX-101 BTD compound for Bipolar Depression associated with Suicidality. The request for BTD was submitted based on the positive finding of a post-hoc subgroup analysis of patients who in addition to Aviptadil or placebo were also treated with Remdesivir and whose respiratory failure due to Critical COVID-19 continued to progress.
(PRNewsfoto/NRx Pharmaceuticals)
"Although we are disappointed about FDA's decision, as recently announced, our strategic focus has already shifted to the advancement of our Breakthrough Therapy designation drug NRX-101. We expect topline data for our ongoing Phase II study of NRX-101 in patients with bipolar depression with sub-acute suicidal ideation (SSIB) by the end of the year. COVID-19, unfortunately, has also created a mental health crisis, including depression and suicides. Our commitment to helping patients with high unmet needs remains at the core of our work," said Robert Besthof, interim CEO of NRx Pharmaceuticals.
"Given ZYESAMI's mechanism and its well characterized safety profile, NRx Pharmaceuticals will further evaluate the options for its use in other high unmet pulmonary as well as other non-pulmonary indications."
Good luck and GOD bless,
I saw that too and I am wondering the same thing…?
Wednesday, JJ bought 100,000 shares (@ 0.58) of NRXP direct in the market. What is that about?
JJ.... how much, if any stock, do you still retain stock in neuro ??
52 weeks at most it was X50 more!!
After receiving the latest NRX news release regarding the cancellation of further trials due to the failure of Aviptadil and remembering that India actually issued an EUA for this very same drug, I knew that something was different somewhere. So I decided to contact one of the several doctors in India who were directly involved with the testing of Aviptadil. The Dr preferred to not be named and I will respect that, but I can say that the Dr was one of many who was at the top of the testing process. The Dr was kind enough to answer my questions although the Dr did not directly address as to why the US recent DSMB testing was labeled as "futile." The Dr did indicate that after a certain length of time if Aviptadil is not helping covid recovery, then "other" health factors could be the causation of death. Or, that is at least my understanding of the Dr's response and I could be misunderstanding some of it. As far as the DSMB statement goes, I am sure that not much can be said because the Dr. has no access to US testing data that was reviewed by the US DSMB.
I will share here and will lay it out in this order: My first introductory email to the Dr and the Dr's first response. Then below that will be my next response email to the Dr and the Dr's final response.
It is apparent to me at least that a different approach to testing as well as the target goals of the testing plus the duration of the testing may be the difference that caused the US DSMB to be so negative and different from the testing and results performed by India's health agencies. We will have to wait and see if NRX studies the US results and provides any comments or explanations to all of us shocked shareholders. I hope so... They owe us that much at the very least.
==============================================================
26 May 2022
Dear Dr. XXXXXXX
I am reaching out to you in regard to the approval of Aviptadil in India. I am a shareholder of NRP and RLFTF. As you may already know, we have been notified that the DSMD has cancelled testing in the US of Aviptadil because they basically said it is worthless.
We shareholders are surprised and shocked. We have no idea as to why they made this decision. Previous studies fully indicated that this drug functioned well and saved lives. We had been waiting for a US EUA.
I know you are probably very busy, but if you would kindly offer your opinion as to why you think this has happened I would really appreciate your thoughts.
I read that India approved your EUA for this drug and I know that you also have stringent requirements. India is a leader in medical research. So I am just wondering what the difference may be.
Thank you so much for any info or opinion on this subject.
Take care!
Bob
======================================================
27 May 2022
In a message dated 5/27/2022 12:38:56 AM Pacific Standard Time, xxxxxxxx writes:
Dear Bob,
Aviptadil is a highly effective drug and it can reverse the ARDS (Acute respiratory distress syndrome) condition promptly. If you look at the USFDA website you'll find that USFDA has approved Vasoactive intestinal peptide (Aviptadil) as an orphan drug for acute respiratory distress syndrome. Aviptadil is also approved as an orphan drug by FDA for Pulmonary hypertension, which is a condition directly linked to ARDS. The same has also been approved by EMA (European Medical agency). Another condition directly linked to ARDS is Acute lung injury, which is also an orphan drug status approved indication for Aviptadil in both the regions.
We have found excellent results of the drug during clinical trials in Indian patients.
Indian Drug regulatory agencies with help of their Subject expert committees scrutinized the clinical trial data for several months and had several review meetings for the scientific discussions with us before granting the EUA permission.
Our clinical trial, is under publication and will come online by first week of June.
Hope the above information is helpful to you.
Kind Regards
Dr. XXXXXXXX
========================================================
27 May 2022
Dear Dr XXXXXXXX,
Thank you so very much for your response to my question. I appreciate you taking the time out of your busy day!
As I mentioned, I am a shareholder with NRXP and RLFTF. They both for some time now have been trying to obtain a US EUA. Then last Wednesday we all received a News Release from NRX announcing the cancellation of further testing because the DMSB said Aviptadil did no better than the placebo results. We are confused and shocked because earlier test results showed, just as you have also indicated that Aviptadil saves lives. We have no idea as to why the test results for this current testing were are they were reported.
I posted the news release and the link below. If you have time, can you review the data and provide your opinion on this please? I am saddened mostly because people are dying when all this time Aviptalil could have prevented much of that loss.
And out of respect, am I free to share your opinion on this subject?
Once again I sincerely thank you for responding!
I wish you all the best and take care!
Bob
=======================================================
28 May 2022
NOTE: The Dr approved release but prefers to remain anonymous.
Dear Mr. XXXXXXX,
Scientifically speaking the effect of Aviptadil needs to be observed for the first week only (Acute phase), which is most critical period for saving life of patient. After that other factors become overwhelming in deciding the outcome of the patient's condition. Immediate improvement in oxygen saturation is the clear cut proof of it. In simpler words, since aviptadil is a short acting drug, we either need to infuse it repeatedly or give through nebulizer for managing a chronic condition. The clinical trials in Covid-19 shouldn't have focused on the clinical outcome longer than 1 week outcome after aviptadil infusion, other wise repeated doses need to be given.
Even in our clinical trial, the quick relief from the symptoms has lead to the long term improvement in clinical condition, since the "Standard of care" was being given along with for long term management.
The PO2:FiO2 ratio starkly improved on day2 onwards and that played a major role in overall clinical improvement. The critical level is 100, once the patient achieves level above 100 for the ratio, generally the survival becomes better, and same was observed in our clinical trial.
Kind Regards
Dr. XXXXXXXXX
Things do NOT add up regarding the stated results and the cancellation of the ongoing testing. As typical, it takes time for questions to be answered. Here is an interesting article that may provide a "peek" into some questionable connections. Could this be about class action suits and sheltering people? Or is it stock manipulation? Is it a part of the ongoing bad blood between NRXP and RLFTF and those negotiations? This may take time to get to the bottom...
https://www.politico.com/news/2020/09/17/andy-harris-coronavirus-neurorx-board-417329
Mask skeptic lawmaker takes on side gig overseeing drug company trial
Several bioethicists question Rep. Andy Harris' lack of experience.
Rep. Andy Harris
Rep. Andy Harris is one of three members of the data and safety monitoring board for the trial run by NeuroRx, a small Delaware company whose CEO and founder has longstanding ties to the congressman. | Al Drago/Pool via AP
By ZACHARY BRENNAN
09/17/2020 06:13 PM EDT
Rep. Andy Harris, a Maryland Republican and medical doctor, has broken with the scientific mainstream during the coronavirus pandemic — decrying stay-at-home orders and warning of a “cult of masks.”
Now the five-term congressman has joined the hunt for coronavirus treatments with an unpaid side gig overseeing a drug company trial. Harris is one of three members of the data and safety monitoring board for the trial run by NeuroRx, a small Delaware company whose CEO and founder has longstanding ties to the congressman.
The members of the board are responsible for monitoring safety and efficacy data from the ongoing trial, which started in May, and have the power to end the study if problems arise. The drug in question, aviptadil, was created in 1970 to treat lung inflammation, but never reached the market. NeuroRx is now investigating whether it can help Covid-19 patients with acute lung failure.
Harris’s role is an unusual one: Former FDA Commissioner Scott Gottlieb told POLITICO that he’d never heard of a sitting member of Congress serving on a data monitoring committee for any drug. The arrangement doesn’t appear to run afoul of House ethics rules because Harris is not being paid by NeuroRx and does not own a stake in the company. But several bioethicists questioned his lack of experience in evaluating data from drug trials, and said that his close ties to NeuroRx CEO Jonathan Javitt could complicate matters if problems arose with the company’s study.
“The concern about bias wouldn’t arise unless the decision-making [on data from the trial] was difficult,” said Susan Ellenberg, a biostatistician at the University of Pennsylvania, whose research focuses on the ethical issues related to clinical trials. “If the data are such that it’s a hard call as to whether to stop the study or not, that’s when the personal connections seep into consciousness,” added Ellenberg, a former FDA staffer who wrote the agency’s guidance document on data monitoring committees.
In a statement to POLITICO, Harris — an anesthesiologist who still practices part-time — brushed aside any suggestion that he is not qualified to serve on the data board.
“I have a Master of Health Science degree from the Hopkins school of public health, which involves training in biostatistics (that’s a matter of public record, which you probably already knew),” he said. “I have been an investigator on numerous NIH grants and published numerous papers that involved complex statistical analysis, and have been involved in clinical research studies as a principal investigator.”
The congressman said that he plans to report the NeuroRx position on his next annual ethics filing, which is due in May, and that he does not interact with FDA as part of his data board service. He did not respond to a question about whether he had sought advice from the House Ethics Committee on the NeuroRx role before he had accepted it.
null“My only hope was to volunteer my expertise to help America find our way out of this pandemic, as every American who has a skill set to offer hopefully should be willing to do,” Harris said in his statement.
Harris met NeuroRx’s CEO, Javitt, more than two decades ago when they worked together at Johns Hopkins University. Javitt was then a surgeon, while Harris specialized in obstetric anesthesiology.
The two have kept in touch over the years. Javitt, who donated $2,000 to Harris’s campaign in 2010, said he’d visited Harris at his House office in recent years to discuss NeuroRx’s other drug in development, for suicidal bipolar depression.
Javitt — who is also vice chairman of the board of directors at Relief Therapeutics, which holds the patent on aviptadil — told POLITICO that he selected Harris for the data committee because of the congressman’s experience in critical care settings. Javitt also said that the FDA signed off on the trial knowing that Harris would be involved with the committee.
The aviptadil trial started in May at four U.S. sites, and aims to enroll more than 100 participants. The FDA has granted the drug “fast track” status, which is designed to speed the availability of drugs to treat serious diseases. NeuroRx is also offering aviptadil through an FDA-approved expanded access program for hospitalized patients at the same sites in Houston, Kentucky, Florida and California that are hosting the current trial.
The data board that Harris is part of has already made at least one crucial decision — recommending in July that the trial continue after analyzing preliminary data from 30 patients, Relief Therapeutics said in press release this week.
FDA’s guidance for data monitoring committees recommends that at least one member is a “biostatistician knowledgeable about statistical methods for clinical trials and sequential analysis of trial data.” But neither Harris nor the other two members of the NeuroRx panel fit that bill.
Alfred Sommer is the dean emeritus of the Johns Hopkins Bloomberg School of Public Health, with training in ophthalmology and epidemiology. Rita Colwell, an aquatic biologist, led the National Science Foundation under presidents Bill Clinton and George W. Bush. She now holds joint appointments at the University of Maryland and Johns Hopkins.
Colwell told POLITICO that she had served on data monitoring committees for other clinical trials. Sommer and Harris had never done so before joining the NeuroRx panel.
But all three of the NeuroRx panel members have professional or personal relationships with Javitt. Sommer, the chair of the data committee, worked with Javitt at Johns Hopkins; the pair have known each other for about 40 years. Javitt met Colwell during her tenure leading NSF, when he was a fellow at the non-profit Potomac Institute for Policy Studies providing support to the agency.
“It seems like a strange collection of people and when you think about the documents this committee would need to review — are they going to understand the adverse event reports? You want them to be informed enough to ask questions on if the adverse events are caused by the drug,” said Leigh Turner, an associate professor at the University of Minnesota Center for Bioethics.
In some cases, the data monitoring board for a trial would be faced with deciding whether to stop that study. “You don’t want three well-intentioned friends being spoon-fed information from the trial and who can’t look at it critically,” Turner added.
Ellenberg -- who wrote the FDA guidelines for data monitoring committees while on staff at the agency -- echoed those concerns. “Neither Harris nor Colwell is known for clinical trials expertise,” she said. “Members of a DMC should all be very familiar with clinical trials.”
Javitt said that an outside biostatistician is working with the committee, but is not a member of it. He also downplayed the panel’s role in the trial. “Unless there are unexpected severe adverse events to take action on, the committee doesn’t have much to do,” he said.
Asked specifically about the unusual decision to include a sitting member of Congress on the data monitoring board, Javitt said it shows Harris’ commitment to fighting the coronavirus and finding effective treatments.
Holly Fernandez-Lynch, an assistant professor of bioethics at the University of Pennsylvania’s Perelman School of Medicine, said there is no shortage of specialists in critical-care medicine who have the biostatistics skills to serve on a Covid-19 drug data monitoring committee.
“Other COVID drugs — and there are many under study — presumably have done so without going to members of Congress,” she said.
Walter Shaub, the former director of the Office of Government Ethics, said “it’s concerning” that Harris is involved with a company regulated by FDA, given his position on the House appropriations subcommittee that controls the agency’s purse. “FDA officials may feel nervous about holding the company to high standards if a member of their appropriations subcommittee is involved in the trial,” he said.
Still, Harris’s involvement with NeuroRx does not appear to violate House ethics rules.
“The only thing that’s prohibited is outside earned income,” said Richard Painter, the former chief ethics lawyer for President George W. Bush who is now a law professor at the University of Minnesota. “House ethics rules are pretty loosey goosey.”
Painter said that Harris should recuse himself from any bills targeted at NeuroRx, but not necessarily the pharmaceutical industry as a whole. He also said that the congressman should recuse himself “from any company involvement with the FDA,” including any interventions on NeuroRx’s behalf.
But Henry Greely, a law professor and chair of the steering committee of the Center for Biomedical Ethics at Stanford University, saw no obvious problems arising from Harris’s work with NeuroRx.
“It’s strange but I don’t think it’s an ethical issue unless Harris tries to use his influence with FDA on behalf of the company,” Greely said. “It would be a closer question for me if he were somehow generally advising the firm, but a [data monitoring board] has a pretty discrete and specific task.”
Lotto play here, mmmm nvm. il wait till it goes down another 50%, then ll buy
Instead Something is up.... additional big cheese are exiting. Lots of speculation.
From HODL 6 hours ago ......Y board.
Resignation of Alessandra Daigneault as Chief Corporate Officer, General Counsel and Corporate Secretary
On May 19, 2022, Ms. Alessandra Daigneault tendered her resignation as Chief Corporate Officer, General Counsel and Corporate Secretary of NRx Pharmaceuticals, Inc. (the “Company”) effective on July 18, 2022 or such earlier date as may be agreed with the Company. Ms. Daigneault is leaving the Company to pursue other personal and professional interests.
The Company’s interim Chief Executive Officer, Mr. Robert Besthof, expressed his thanks to Ms. Daigneault for her outstanding contributions to the Company, including overseeing the merger of NeuroRx, Inc, and Big Rock Partners Acquisition Corp in May 2021, which resulted in the formation of the Company.
Mr. Michael Kunz, the Company’s Deputy General Counsel, is expected to assume the duties of General Counsel and Corporate Secretary.
Resignation of Mr. Ira Strassberg as Chief Financial Officer and Treasurer
On May 19, 2022, Mr. Ira Strassberg tendered his resignation as Chief Financial Officer and Treasurer of the Company effective July 18, 2022. Mr. Strassberg is leaving the Company to pursue other personal and professional interests. Mr. Strassberg will continue to fulfill his duties as Chief Financial Officer and Treasurer of the Company until his departure. The Board will be conducting a search for a successor.
The Chairman of the Company’s Audit Committee, Mr. Patrick Flynn, and Mr. Besthof, expressed their thanks to Mr. Strassberg for his meaningful contributions to the Company, including the establishment of robust internal controls and a strong finance and accounting function at the Company.
At this low low price, insiders sb buying millions of shares ...IF
there is a bright future for Neuro, imho. .
Why are the big insiders not buying in at low prices ???? Where is bowtie ???
Someone told me this stock. I just wondering before covid, this stock traded around $10. Why it is only $1.32? Because depression trial failed? But I thought P3 is continue and not completed yet. Can someone in the board delight me what is going on? Thanks.
No, I suspect that eventually we'll be flirting with sub penny on this one. Dr JJ only has one pot cooking on the stove, and NIH results are 6-12 months away. I think an R/S may be in the cards now, if the SEC permits that. I'm not sure if the ongoing litigation could be tying his hands on things. He really bungled this one! I wonder if he still posts that pic with him and his family/friends toasting wine on his yacht.
New Low $1.17 today. / so far.
Waiting for the great dr jj to buy in really big before considering another purchase.
Where is the bottom ??
That down trend is not over!
Nrx is getting destroyed again today. on very low volume.
Will jj ever decide to buy at any price with his own jack ?
And Side Bets on how low this will go ?
Just a matter of time, IMHO, before this company does not exist as it is today, with perhaps a buyout at a much much lower price, considering all the legal liabilities. ,
The criminal Dr JJ could have joined RLF with their ACER sub and the Nrx drugs to become a Real pharma. But NO, personal greed, incompetent CT design and implementation by this criminal has delayed and then ruined most possibilities.
Sorry for the venting to all still invested. I lost on RLF too. Worst slime ball POS I've ever heard of. GEM must be so happy, they've been filling his pockets for years before Avitadil which is better and safer than Paxlovid Criminal incompetence on so many levels, IMO.
...GLTA...elsewhere...
So big pharma... Pfizer received an emergency use authorization for Paxlovid before any real proof that it actually works! The FDA has some serious corruption and it is affecting the health of Americans. Government run agencies are not exactly full of the brightest and best of people. Political influence, graft, corruption and special future employment favors... all influence their decisions.
Pfizer Inc. slumped after Paxlovid, its pill for treating Covid-19, failed to show benefit as a preventive therapy in a trial.
https://finance.yahoo.com/news/pfizer-falls-covid-pill-fails-130238318.html
"Paxlovid is poised to become one of the fastest-selling drugs of all time, with $24 billion in projected 2022 sales, according to analytics group Airfinity Ltd. Pfizer has also reaped billions in global sales from its Covid-19 vaccine.
The company said late Friday that the drug failed to hit its main goal of reducing the risk that adults exposed to the coronavirus through contact with a household member would become infected. Compared with those who took a placebo, people who received Paxlovid had about a third less risk of infection, which wasn’t statistically significant.
The shares fell 2.2% at 9:36 a.m. in New York. They had declined 17% this year through Friday’s close.
The trial failure, along with reports of rebounding coronavirus levels in some patients who have completed a course of Paxlovid, are unlikely to change the drug’s sales potential, according to Bloomberg Intelligence analysts John Murphy and Sam Fazeli.
“The trial miss at most takes away a small incremental sales opportunity in future years,” they said Monday in a research note. The reports of rebound infections are “also not surprising as viral dynamics during an infection are unlikely to fit perfectly with a rigid 5-day course.”
Morgan Stanley analysts, who had estimated the drug’s sales to reach $48 billion in 2022, said the prophylaxis data might affect their forecast."
GoodStick, Very Much Agreed. Hopefully, but unfortunately, its looking like without FDA approvals and without any meaningful assets ( none I am aware of ) and settlement of all the open lawsuits with various lawfirms and RLF, its difficult to make a case for owning this stock.
FWIW...NRX IS AT ANOTHER NEW LOW IN ITS STOCK PRICE PRICE TODAY OF $1.56
SEEMS LIKE THE BULLS WOULD BE BUYING NOW, EH ?
At this point, simply doing the right and honorable thing, that should have been done all along, is about the only thing that can save this company from going down the drain. Hopefully, it's not too late.
Either you control your ego and greed, or they will control you.
PW, and sadly, NRX owns almost nothing at this point, and any
judgements could be greater than the entire market cap of this miserable
company, and the assets of those that have made terrible NRX mgmt decisions
If it is true that the FDA is waiting for the completion of the NIH trial before making a decision on EUA, then unfortunately we may easily see this drift down well below a buck because for all practical purposes NRXP has nothing else of value (IMO) in their pipeline. JMHO
Another ugly day for NRX ........ on ugly low volume.
https://stockcharts.com/h-sc/ui?s=NRXP&p=D&yr=0&mn=4&dy=0&id=p56160925541
One dollar a share is a real possibility.
Any word on the seller of the 8 million shares ?
Or the buyer if there is one ?
new lows continue for NRXP and insiders are not buying...
go figure, is the last one out a rotten egg ?
What you say is true.
Let's hope this doesn't turn into the rabbit against the turtle race.
GLTA
Headline...I would have preferred a headline stating that EUA was approved. VERU still has a long row to hoe. They need to construct the EUA application from their data and submit to FDA (1-2 months). Then the FDA will review and make a decision (1-3 months). And that assumes no mishaps along the way. So, VERU is behind NRXP regarding EUA processing.
This should have been the headline for NRXP months ago, but greed and double-dealing got in the way. (IMO)
Veru’s Novel COVID-19 Drug Candidate Reduces Deaths by 55% in Hospitalized Patients in Interim Analysis of Phase 3 Study; Independent Data Monitoring Committee Halts Study Early for Overwhelming Efficacy
https://www.wallstreetreporter.com/2022/04/11/verus-novel-covid-19-drug-candidate-reduces-deaths-by-55/
JK...very, very true. However, if mediation prevents the law suit from going to the courts, it does give NRXP a more favorable chance. I suspect that one of the primary reasons why the SP NRXP is in the tank is due to the existence of the law suit. But the market is fickle, so no one knows for sure what the outcome would be if both parties resolved their differences without going to court. NRXP has a new CEO, so they may be starting from a new, clean slate.
PW, please consider that any mediation progress does not necessarily mean that this progress is favorable to Nrx. Common sense is that if it were favorable to Nrx vs RLF, then this major major shareholder would not be selling, ie, "reselling", or filing for "resale" per Seeking Alpha and "Files for resale" is an odd way of saying "sell", IMHO.
https://seekingalpha.com/news/3822137-nrx-pharmaceuticals-files-for-resale-of-up-to-782m-shares-by-selling-shareholders?utm_source=advfn.com&utm_medium=referral
NRx Pharmaceuticals files for resale of up to 7.82M shares by selling shareholders
Apr. 08, 2022 5:09 PM ETNRx Pharmaceuticals, Inc. (NRXP)By: Anuron Mitra, SA News Editor
Biopharmaceutical company NRx Pharmaceuticals (NASDAQ:NRXP) on Friday filed for resale of up to 7.82M shares of common stock by certain selling shareholders.
The company is also registering 391,236 shares of common stock to be issued upon exercise of the placement agent options.
The company will not get any proceeds from the sale of shares by the selling shareholders. It will receive the proceeds from any exercise of the investment options and placement agent options for cash.
choo, Progress with median has been reported!! If RLFTF and NRXP finally come to amicable terms via mediation, we could see a total turn around in SP for both entities. The NIH trial is driving the success/failure of RLF-100/Zyesami.
choo choo, thanks for sharing..
I also sold months ago, luckly with a relatively small loss.
Seems there are only two with that number of shares that I can find,
so my guess is the DUMPER would be a Javitt, as I can not locate
any other share holders with that number of shares, even institutional.
Please feel free to correct me if I am viewing this incorrectly.
https://finance.yahoo.com/quote/NRXP/holders?p=NRXP
https://finance.yahoo.com/quote/NRXP/insider-transactions?p=NRXP
Personally, I would not touch that number of shares right now, even if I was as well off as a MUSK or say a BEZOS.
All good succinct questions Joe, but who knows what's going on with this ticker anymore. I sold months ago and now just watch as this company falls into graveyard territory. It's really hard to believe, especially after having so much hope for NRXP, how this has all played out. I still hope that somehow NRXP can pull a rabbit out of the hat for the sake of those who are still invested here.
And the DUMPER of roughly EIGHT MILLION Shares is ?????
Time to get out before the bottom falls out ?????
Humm....ya think someone would want a big ol discount on the current price and with all the problems at this time ?
Gee, is a bid on some at 7 or 8 cents a share is a fair price ?
Now the million dollar question, will jj jump in and buy ?????
Or is he selling ???
Uh-oh, looks like some heavy hitters are bailing on NRXP. What a travesty this whole mess has been. The corporate and governmental systems in place in our country are full of crooked immoral people. We are living in an upside-down, convoluted, dystopian period of time in our society which if not turned around soon will be the downfall of this once great nation.
https://seekingalpha.com/news/3822137-nrx-pharmaceuticals-files-for-resale-of-up-to-782m-shares-by-selling-shareholders?utm_source=advfn.com&utm_medium=referral
PW, thanks for the colorful and interesting analogy.
JJ is like a rat caught in a corner with no teeth. No telling what he will do.
At this point, I do not see how he has provided any benefit to RLF, and it seems like to me, it been all give by RLF, and all take by Nrx.
It is interesting and ironic that this all started with Relief needing JJ's help to break into the US market.
JJ, driven by greed, made a mess of it and now his only hope is to cooperate with Relief in order to save his bacon. (Pardon the Pun)
I think JJ is in damage control mode across the board. He's sitting with a very week hand (legally). And he's had very bad luck with EUA, so I doubt he wants to be a bag holder IF the FDA rejects the 3rd EUA (no matter the reason). JJ is like a rat caught in a corner with no teeth. No telling what he will do. If zyesami collapses, then so does he and his corporate house of cards (NRXP). If mediation proves to be successful for both sides, then I suspect that we'll see a boost in PPS for both sides.
Very Low Volumes, Very Near A New Low Again Today, not pretty.
https://stockcharts.com/h-sc/ui?s=NRXP&p=D&yr=0&mn=4&dy=0&id=p56160925541
MACD looks ugly with plenty of downside from here without some approvals/agreements/settlements/end to lawsuits, IMHO.
To me, it Looks like, if jj were to sell a large position, there may be few buyers, or, the buyers may want a much lower price, and a sub two dollar
range is not that far away right now.
Do you suppose that if jj was really bullish, he would be buying hand over fist with his own money in big open market positions ??
Yes, this was posted on 4/1/22 under "Show eFiled Documents" at this link. You have to do a "Party Search" first on "Relief Therapeutics". Great to show some progress.
https://iapps.courts.state.ny.us/webcivil/FCASSearch
A friend of mine sent this to me:
New court doc from RLFTF board by IG states that both sides are making progress and request a stay of the proceedings while they continue to work through mediation.
https://iapps.courts.state.ny.us/nyscef/ViewDocument?docIndex=FcZfoGTkugRo6zR3yNzM9w==
|
Followers
|
53
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
1108
|
|
Created
|
12/15/20
|
Type
|
Free
|
| Moderators ProfitScout jedijazz | |||





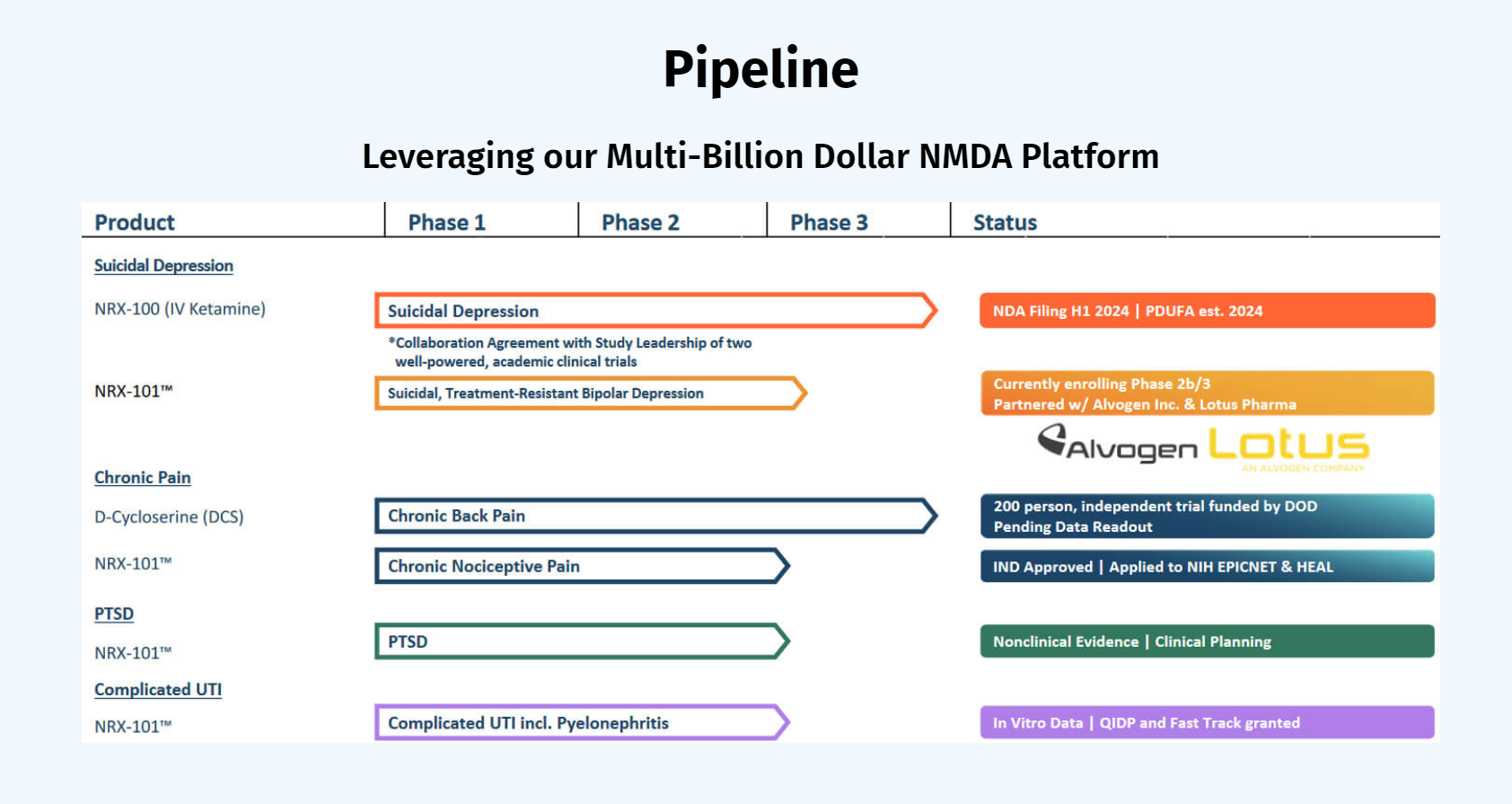
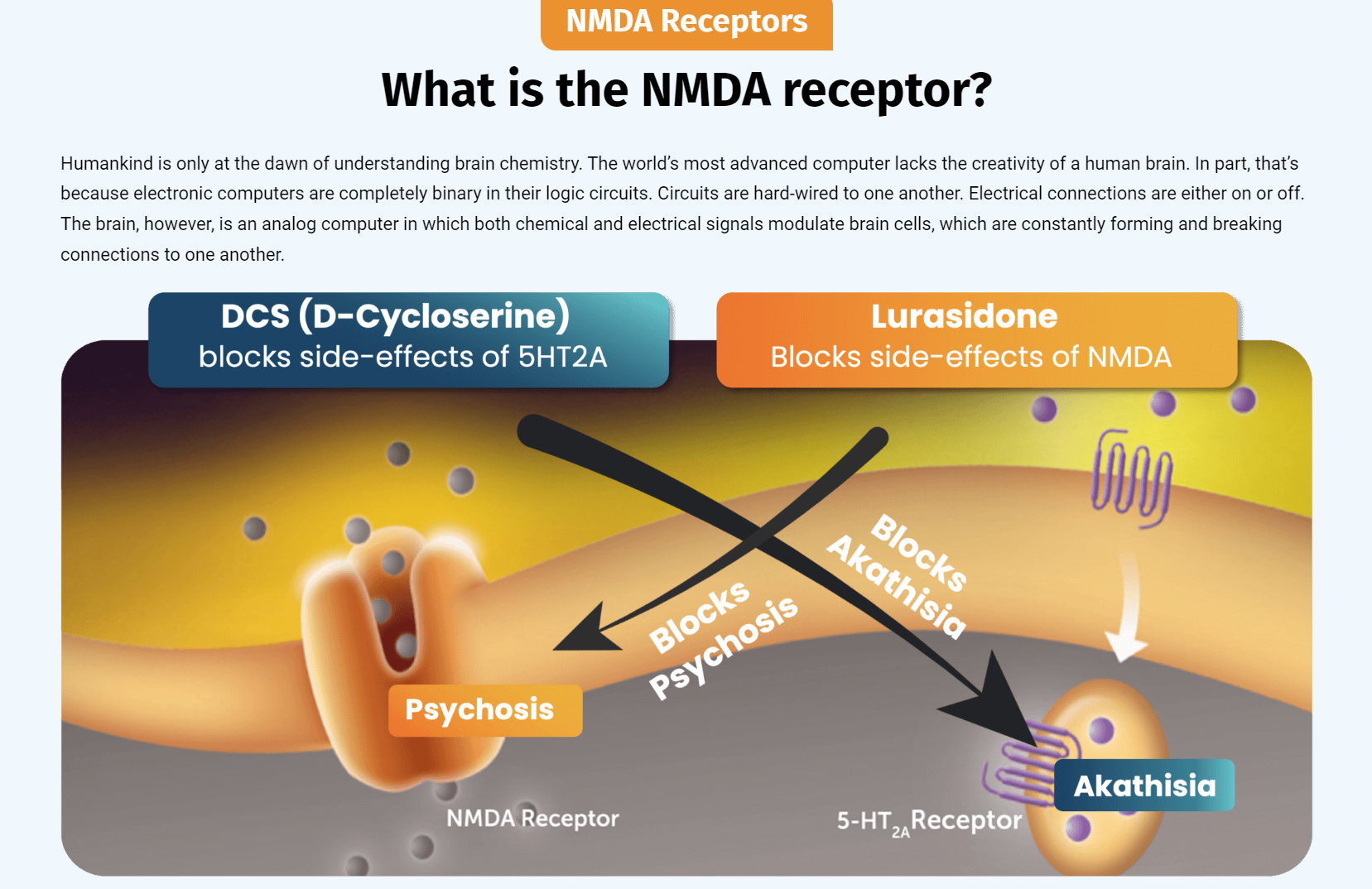







Both drugs demonstrated > 50% response for treating depression. NRX-101 demonstrated a mean 76% reduction in symptoms of akathisia compared to lurasidone that was sustained over 42 days (Effect...

New data demonstrates no impact of NRX-101 on gut or vaginal flora – considered primary causes of pseudomembranous colitis due to C difficile and vaginal yeast infections NRX-101 previously...
Formulation based on prior patents by NRx founder Achieved pH neutral formulation of ketamine, potentially enabling both intravenous (IV) and subcutaneous (SQ) administration Company expects...
Data transferred for independent statistical analysis Top-line data expected in April 2024 RADNOR, Pa., April 8, 2024 /PRNewswire/ -- NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) ("NRx...
Four potential near-term milestones, including data from two clinical trials, an NDA filing and an upcoming share dividend -- 50% reduction in corporate overhead and 25% reduction in overall net...
Four potential near-term milestones, including data from two clinical trials, an NDA filing and a share dividend RADNOR, Pa., March 28, 2024 /PRNewswire/ -- NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) ("
NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) ("NRx Pharmaceuticals", the "Company"), a clinical-stage biopharmaceutical company, today announced that it will release its fourth quarter and full year...
NRx Pharmaceuticals, Inc. (Nasdaq:NRXP) ("NRx Pharmaceuticals", the "Company"), a clinical-stage biopharmaceutical company, today announced that Dr. Jonathan Javitt, Chairman and Chief Scientist...
NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) ("NRx Pharmaceuticals", the "Company"), a clinical-stage biopharmaceutical company, today announced that it will release its fourth quarter and full year...
61.4% (56,781,354) of eligible shares voted 94.4% of votes were cast in favor of the resolution RADNOR, Pa., March 21, 2024 /PRNewswire/ -- NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) ("NRx...
NRx Pharmaceuticals Board of Directors has authorized its Chairman, CEO, and management to take all necessary steps to affect the Dividend and Royalty Rights to NRXP Shareholders and applicable...
Initiative is intended to protect shareholder value through continued compliance with Nasdaq listing rules and elimination of naked short sales positions in the Company's securities The Company is...
Company to receive first allocation of ketamine for sale by month end Partners preparing to ship IV Ketamine to full range of customers via 503a and 503b pharmacies NRx Pharmaceuticals and HOPE...
Marks a major step in the development of what could be the first drug approved for Suicidal Bipolar Depression The study database is being cleaned and locked; statistical analysis and top-line...
NRx Pharmaceuticals received approximately $1.0 million in cash from an existing investor Shares were sold at $0.38, a 26.7% premium the recent share offering, along with one common 5-year warrant...
In the news release, NRx Pharmaceuticals, Inc. Announces Pricing of $1.5 Million Underwritten Public Offering of Common Stock, issued 27-Feb-2024 by NRx Pharmaceuticals, Inc. over PR Newswire, we...
NRx Pharmaceuticals, Inc. (Nasdaq: NRXP), ("NRx Pharmaceuticals" or the "Company"), a clinical-stage biopharmaceutical company, today announced that it intends to offer to sell shares of its...
Presentation available on NRx Pharmaceuticals website at https://www.nrxpharma.com/hope-therapeutics/ KEY UPDATES ARE AS FOLLOWS: NRx management is proposing to award 50% of founding shares in...
DISCLAIMER:
Nothing in the contents transmitted on this board should be construed as an investment advisory, nor should it be used to make investment decisions.
There is no express or implied solicitation to buy or sell securities.
The author(s) may have positions in the stocks or financial relationships with the company or companies discussed and may trade in the stocks mentioned.
Readers are advised to conduct their own due diligence prior to considering buying or selling any stock. All information should be considered for information purposes only.
No stock exchange has approved or disapproved of the information here.
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
