Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
$NRXP $2.81 HOD $2.87 NRx Pharmaceuticals, Inc. (NASDAQ:NRXP) Initiates Strategy to Combat Short Sales https://prn.to/4baoVcG $RENB $BMY
$NRXP Huge article out today. NRx Pharmaceuticals Eyes First Commercial Revenue in 2024, Announces Breakthroughs in Bi-Annual Milestone Update https://finance.yahoo.com/news/nrx-pharmaceuticals-eyes-first-commercial-135500896.html?soc_src=social-sh&soc_trk=tw&tsrc=twtr via @YahooFinance
Very good to see NRXP is making every effort to get the word out on the company's important new developments, especially for suicide prevention treatments. This is a top mental health crisis today. The EF Hutton Conference is a great opportunity to reach high value investors and institutions.
The EF Hutton Annual Global Conference, which will take place on May 15, 2024, at The Plaza Hotel in New York City.
NEWS: NRx Pharmaceuticals, Inc. (NASDAQ:NRXP) to Participate in the EF Hutton Annual Global Conference on May 15, 2024
PR Newswire
Wed, May 8, 2024, 8:30 AM EDT
In This Article: NRXP
RADNOR, Pa., May 8, 2024 /PRNewswire/ -- NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) ("NRx Pharmaceuticals", the "Company"), a clinical-stage biopharmaceutical company, today announced that it will participate in the EF Hutton Annual Global Conference, which will take place on May 15, 2024, at The Plaza Hotel in New York City.
Prof. Jonathan Javitt, MD, MPH, the Company's Chairman and Chief Scientist, and Matthew Duffy, the Company's Chief Business Officer, will hold one-on-one meetings with investors throughout the day. Interested parties can register to attend here.
About NRx Pharmaceuticals
NRx Pharmaceuticals is a clinical-stage biopharmaceutical company developing therapeutics based on its NMDA platform for the treatment of central nervous system disorders, specifically suicidal bipolar depression, chronic pain and PTSD. The Company is developing NRX-101, an FDA-designated investigational Breakthrough Therapy for suicidal treatment-resistant bipolar depression and chronic pain. NRx has partnered with Alvogen and Lotus around the development and marketing of NRX-101 for the treatment of suicidal bipolar depression. NRX-101 additionally has potential to act as a non-opioid treatment for chronic pain, as well as a treatment for complicated UTI.
NRx has recently announced plans to submit a New Drug Application for HTX-100 (IV ketamine), through Hope Therapeutics, in the treatment of suicidal depression, based on results of well-controlled clinical trials conducted under the auspices of the US National Institutes of Health and newly obtained data from French health authorities, licensed under a data sharing agreement. NRx was awarded Fast Track Designation for development of ketamine (NRX-100) by the US FDA as part of a protocol to treat patients with acute suicidality.
About HOPE Therapeutics, Inc.
HOPE Therapeutics, Inc. (www.hopetherapeutics.com) is a Specialty Pharmaceutical Company, wholly-owned by NRX Pharmaceuticals focused on development and marketing of an FDA-approved form of intravenous ketamine for the treatment of acute suicidality and depression together with a digital therapeutic-enabled platform designed to augment and preserve the clinical benefit of NMDA-targeted drug therapy.
View original content to download multimedia:https://www.prnewswire.com/news-releases/nrx-pharmaceuticals-inc-nasdaqnrxp-to-participate-in-the-ef-hutton-annual-global-conference-on-may-15-2024-302139427.html
SOURCE NRx Pharmaceuticals, Inc.
$NRXP More DD: Of the thousands of drugs approved by FDA, only 544 have been designated as “Breakthrough Therapy.” NRX-101 is the first Breakthrough Therapy for Suicidal Depression https://axecapitalusa.com/nrxp/
Be sure to follow NRXP on Twitter (X): Results Show Superior Safety & Efficacy for NRX-101; Accelerated FDA Approval sought for Bipolar Depression
Results Show Superior Safety & Efficacy for NRX-101; Accelerated FDA Approval sought for Bipolar Depression
— NRx Pharmaceuticals (@NRxPharma) May 7, 2024
Read More - https://t.co/VzEAskHG4j#PharmaNews #mentalillness #mentalhealth #mentalhealthmatters #depression #ketamine #stress #PTSD #bipolar #NRX101 $NRXP
NEWS: NRx Pharmaceuticals (Nasdaq:NRXP) Announces Final Clinical Trial Results: Superior Safety Combined with Similar Efficacy in the Trial of NRX-101 Compared to Lurasidone in Suicidal Bipolar Depression
PR Newswire
Mon, May 6, 2024, 8:30 AM EDT
In This Article: NRXP
Both drugs demonstrated > 50% response for treating depression. NRX-101 demonstrated a mean 76% reduction in symptoms of akathisia compared to lurasidone that was sustained over 42 days (Effect Size .37; P=0.025), using prespecified analytic methodology memorialized in FDA Special Protocol Agreement. Levels of akathisia with NRX-101 were essentially zero at day 42
This safety advantage was previously reported in the Company's published STABIL-B trial
Akathisia is identified as a life-threatening side effect of nearly all antidepressants, reported in 10-15% of treated patients and is closely linked to suicide in FDA black box warning
Akathisia was seen in 2% of participants treated with NRX-101 vs. 11% treated with lurasidone
Company plans to seek accelerated approval of NRX-101 for use in patients with bipolar depression at risk of akathisia while continuing to broaden the indication to all patients with bipolar depression and perhaps schizophrenia
Study will be presented at the American Society of Clinical Psychopharmacology (ASCP) meeting May 28-31, 2024 (Miami) together with study investigators, accompanied by a broadcast scientific presentation on akathisia and antidepressant safety, and investor Q&A
RADNOR, Pa., May 6, 2024 /PRNewswire/ -- NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) ("NRx Pharmaceuticals", the "Company"), a clinical stage pharmaceutical company, today announced a statistically significant safety advantage of NRX-101 compared to the standard of care comparator in its recently completed clinical trial in patients with suicidal bipolar depression. Therefore, the Company believes that demonstration of reduced akathisia in the setting of comparable antidepressant efficacy constitutes a basis for Accelerated FDA Approval of NRX-101. The full clinical trial results will be presented at the upcoming meeting to the American Society of Clinical Psychopharmacology held May 28-31, 2024 in Miami. NRx will gather Key Opinion Leaders to educate the public on the importance and potentially life-saving implication of this finding.
Figure 1: Akathisia rating by study day: A consistent effect is seen commencing at the first post-randomization visit and continued throughout the study (Mixed Model Repeated Measures Regression Effect size =.37; P=0.025).
Figure 1: Akathisia rating by study day: A consistent effect is seen commencing at the first post-randomization visit and continued throughout the study (Mixed Model Repeated Measures Regression Effect size =.37; P=0.025).
Last week, the Company released preliminary top-line data as required by SEC disclosure rules. The Company believes that today's findings based on mixed model regression analysis as specified in the Company's Special Protocol Agreement with the FDA, when combined with the prior STABIL-B trial1, demonstrate a basis for seeking accelerated drug approval of NRX-101 based on improved safety related to akathisia and suicidality in the setting of comparable antidepressant efficacy.
Trial participants had identical mean scores on the Barnes Akathisia Rating Scale (BARS) at baseline with subsequent decrease in the NRX-101 treated group versus an increase in the lurasidone-treated group, yielding a 76% relative mean difference between the groups. The difference was apparent at the first post-randomization visit and continued throughout the trial. (Fig 1) Over the 42 days of observation, an effect size of .37 was identified with a statistically significant P value of 0.025 on the Mixed Model for Repeated Measures (MMRM) methodology agreed to with FDA in the 2018 Special Protocol Agreement. Akathisia as ascertained by a 1 point increase in the BARS was seen in 11% of participants randomized to lurasidone (comparable to previous reports in the literature) and seen in only 2% of those treated with NRX-101, an akathisia level that was previously reported for the placebo arm of the lurasidone registration trial.
Akathisia was a prespecified key safety endpoint of the Company's clinical trial. Hence this finding is not a "post-hoc" observation. As previously noted, this clinical trial of 91 participants with suicidal bipolar depression who were not pre-treated with ketamine demonstrated that NRX-101 and lurasidone were comparable in their antidepressant effect. A 33% but statistically non-significant sustained decrease in suicidality was also seen favoring NRX-101. As noted above, improved antidepressant efficacy is not required to seek drug accelerated drug approval based on a statistically-significant safety benefit.
Based on this safety finding, NRx plans to seek Accelerated Approval of NRX-101 for treatment of bipolar depression in patients at risk for akathisia who are at highest risk of suicide, while continuing to develop evidence to support broader indications both in treatment of depression and schizophrenia. Should these data be confirmed in additional large scale trials, the Company believes that physicians and patients will universally prefer antidepressant and antipsychotic drugs with a reduced akathisia risk. The NRx patent portfolio supports the development of a broad range of combined NMDA/serotonergic drugs for treatment of depression and psychosis.
There is a recent regulatory precedent for the approval of psychiatry drugs that demonstrate comparable efficacy with improved safety. A combination of olanzapine and samidorphan (LYBALVI®) was approved based on comparable effect on schizophrenia symptoms with evidence of less weight gain favoring LYBALVI. Thus, public assertions by journalists and short-sellers that NRx has no path to market based on the finding of comparable efficacy in this trial are utterly baseless and may be designed to mislead investors.
"More than 7 million Americans suffer from bipolar depression with a potential market opportunity in excess of $20 billion. No prior drug to treat bipolar depression has demonstrated superiority on side effects most closely linked to suicide. Patients with bipolar depression have a 50% lifetime risk of a suicidal attempt and a 20% lifetime risk of dying from suicide. On this basis of these superiority findings, previously seen in the STABIL-B trial, we plan to seek Accelerated Approval from FDA for treatment of patients with bipolar depression who are at risk for akathisia, as we and our partners continue to broaden the indication to the treatment of all patients with bipolar depression." said Dr. Jonathan Javitt, NRx's Chairman and Chief Scientist. "Patients and key opinion leaders alike have told us clearly that an antidepressant with comparable antidepressant effect and reduced risk of akathisia and other risk factors for suicidality would be unambiguously preferred in the marketplace."
Background on Akathisia
Akathisia is a known extrapyramidal side effect of all serotonin-targeted antidepressants and antipsychotic drugs. It can be demonstrated in laboratory models, using rodent behavioral data.2 Prof. Daniel Javitt, the inventor of NRX-101 first discovered the ability of NMDA antagonist drugs to reduce akathisia in 2009, which forms the basis of the Company's composition of matter patent portfolio. In his 2012 patent application Javitt stated 3
A major limitation in use of antipsychotic and antidepressant medications is the liability to produce behavioral side effects, especially anxiety, agitation, and akathisia, all of which are associated with generating or exacerbating suicidality in psychotic or depressed patients. These behavioral side effects can be differentiated from symptoms of the illness by consideration of both time course and specific patterns of symptoms.
The side effect is clearly recognized in an FDA-mandated black box warning applied to lurasidone and all medications in its class of drugs, with a specific warning about suicidality as follows:
"The following symptoms, anxiety, agitation, panic attacks, insomnia, irritability, hostility, aggressiveness, impulsivity, akathisia (psychomotor restlessness), hypomania, and mania have been reported in adult and pediatric patients being treated with antidepressants for major depressive disorder as well as for other indications, both psychiatric and non-psychiatric. Although a causal link between the emergence of such symptoms and either the worsening of depression and/or the emergence of suicidal impulses has not been established, there is concern that such symptoms may represent precursors to emerging suicidality"4.
In most clinical trials, safety endpoints are not a basis for drug approval, although adverse safety findings may prevent drug approval. Typically, new drugs are compared to placebo in order to prove a difference in efficacy. In this case, the trial was conducted against the standard of care drug, not against placebo and, therefore, safety differences are highly important. Akathisia is a labeled negative side-effect of lurasidone and all similar drugs, and is closely linked to suicidal behavior.56 It is a side effect about which patients must be warned[7] and has figured prominently in class action lawsuits brought against manufacturers of serotonin-targeted antidepressants and antipsychotics. Currently, there is no FDA-approved treatment for akathisia and it is considered a medical emergency by many treating psychiatrists. Akathisia causes extreme anxiety to patients, inability to control motor movement, and impulsive acts, all to often leading to suicide and may be the key driver of the observed increase in suicidal ideation and behavior associated with all serotonin-targeted antidepressants and antipsychotic drugs.
"These studies support our original observation that D-cycloserine reverses the akathisia-like behaviors induced by lurasidone in rodent models. It is exciting to see those findings demonstrated in a second trial with unequivocal statistical significance," said Prof. Daniel Javitt, co-founder of NRx Pharmaceuticals. "Hopefully, this opens a path to bringing a lifesaving drug to millions of patients with suicidal depression whose only approved long-term treatment is electroconvulsive therapy."
About NRx Pharmaceuticals
NRx Pharmaceuticals is a clinical-stage biopharmaceutical company developing therapeutics based on its NMDA platform for the treatment of central nervous system disorders, specifically suicidal bipolar depression, chronic pain and PTSD. The Company is developing NRX-101, an FDA-designated investigational Breakthrough Therapy for suicidal treatment-resistant bipolar depression and chronic pain. NRx has partnered with Alvogen and Lotus around the development and marketing of NRX-101 for the treatment of suicidal bipolar depression. NRX-101 additionally has potential to act as a non-opioid treatment for chronic pain, as well as a treatment for complicated UTI.
NRx has recently announced plans to submit a New Drug Application for HTX-100 (IV ketamine), through Hope Therapeutics, in the treatment of suicidal depression, based on results of well-controlled clinical trials conducted under the auspices of the US National Institutes of Health and newly obtained data from French health authorities, licensed under a data sharing agreement. NRx was awarded Fast Track Designation for development of ketamine (NRX-100) by the US FDA as part of a protocol to treat patients with acute suicidality.
About HOPE Therapeutics, Inc.
HOPE Therapeutics, Inc. (www.hopetherapeutics.com) is a Specialty Pharmaceutical Company, wholly-owned by NRX Pharmaceuticals focused on development and marketing of an FDA-approved form of intravenous ketamine for the treatment of acute suicidality and depression together with a digital therapeutic-enabled platform designed to augment and preserve the clinical benefit of NMDA-targeted drug therapy.
Notice Regarding Forward-Looking Statements
The information contained herein includes forward-looking statements within the meaning of Section 21E of the Securities Exchange Act of 1934, as amended, and Section 27A of the Securities Act of 1933, as amended. These statements include, among others, statements regarding the proposed public offering and the timing and the use of the proceeds from the offering. Forward-looking statements generally include statements that are predictive in nature and depend upon or refer to future events or conditions, and include words such as "may," "will," "should," "would," "expect," "plan," "believe," "intend," "look forward," and other similar expressions among others. These statements relate to future events or to the Company's future financial performance, and involve known and unknown risks, uncertainties and other factors that may cause the Company's actual results to be materially different from any future results, levels of activity, performance or achievements expressed or implied by these forward-looking statements. You should not place undue reliance on forward-looking statements since they involve known and unknown risks, uncertainties and other factors which are, in some cases, beyond the Company's control and which could, and likely will, materially affect actual results, levels of activity, performance or achievements. Any forward-looking statement reflects the Company's current views with respect to future events and is subject to these and other risks, uncertainties and assumptions relating to the Company's operations, results of operations, growth strategy and liquidity. More detailed information about the Company and the risk factors that may affect the realization of forward-looking statements is set forth in the Company's most recent Annual Report on Form 10-K and other filings with the Securities and Exchange Commission. Investors and security holders are urged to read these documents free of charge on the SEC's website at http://www.sec.gov. Except as may be required by applicable law, The Company assumes no obligation to publicly update or revise these forward-looking statements for any reason, or to update the reasons actual results could differ materially from those anticipated in these forward-looking statements, whether as a result of new information, future events or otherwise.
1 Nierenberg A, Lavin P, Javitt DC, et. al. NRX-101 vs lurasidone for the maintenance of initial stabilization after ketamine in patients with severe bipolar depression with acute suicidal ideation and behavior; a randomized prospective phase 2 trial. Int J Bipolar Dis 2023;11:28-38, doi.org/10.1186/s40345-023-00308-5
2 Walf, A. A., & Frye, C. A. (2007). The use of the elevated plus maze as an assay of anxiety-related behavior in rodents. Nature Protocols, 2(2), 322-328. https://doi.org/10.1038/nprot.2007.44
3 Javitt DC. Composition and method for treatment of depression and psychosis in humans. US Patent 10583138B2. Granted March 10, 2020.
4 Trivedi et al., J Clin Psychiatry, 72:765-774, 2011
5 Chow LC, Kahouh NK, Bostwick JR, et. al., Akathisia and newer second-generation antipsychotic drugs: A review of current evidence. Pharmacotherapy 202;40(6):565-574 doi: 10.1002/phar.2404
6 Uwai Y, Nabekura T. Risk factors for suicidal behavior/ideation and hostility/aggression in patients with bipolar disorders: An analysis using the Japanese adverse drug event report database. J Psychiatric Res 2022;153:99-103. https://doi.org/10.1016/j.jpsychires.2022.07.005
7 https://www.webmd.com/schizophrenia/side-effects-of-lurasidon
View original content to download multimedia:https://www.prnewswire.com/news-releases/nrx-pharmaceuticals-nasdaqnrxp-announces-final-clinical-trial-results-superior-safety-combined-with-similar-efficacy-in-the-trial-of-nrx-101-compared-to-lurasidone-in-suicidal-bipolar-depression-302136430.html
SOURCE NRx Pharmaceuticals, Inc.
$NRXP $2.27 +8.10% Premarket- New analyst report from Ed Woo, CFA, NRx Pharmaceuticals publishing analyst at Ascendiant Capital Markets. View Report - https://t.co/XDweCNyr8U #PharmaNews #mentalillness #depression #ketamine #stress #PTSD #suicide #bipolar #NRX101 $NRXP #Bullish
Good morning $NRXP New Report Out!
$NRXP $2.28 +11.00% Strong Q4 Results and Milestones Announced Including Approved Stock Dividend & 8 Million More in Capital: (Nasdaq: NRXP) https://www.einpresswire.com/article/700334263/strong-q4-results-and-milestones-announced-including-approved-stock-dividend-8-million-more-in-capital-nasdaq-nrxp via @ein_news
NRX-101 is the first oral antidepressant to show 33% advantage in sustained remission in suicidality (not statistically significant at this sample size) and 75% advantage in relief from Akathisia relative to lurasidone - never previously shown with an oral antidepressant. Suicidality signal met the study's promising zone criteria and the akathisia signal approached statistical significance (P=0.076)
Study concluded:
Both NRX-101 and lurasidone, an accepted standard of care in Bipolar Depression, demonstrated approximately 50% reduction in symptoms of depression
These data are comparable to previous statistically-significant finding of reduced suicidality and in the published STABIL-B trial and support an approval pathway via a 300-person registrational trial with sustained remission in suicidality as the primary endpoint
Company believes that an oral antidepressant that demonstrates reduction in suicidality has potential to become standard of care for treatment of bipolar depression.
Data from this study expand the potential utility of NRx-101 to treat both patients with suicidal bipolar depression (who will require prior use of ketamine) and those without subacute suicidality (nearly 7 million patients in the US).
https://www.benzinga.com/pressreleases/24/04/n38518066/nrx-pharmaceuticals-nasdaq-nrxp-announces-promising-findings-in-phase-2b3-clinical-trial-of-nrx-10
$NRXP Oral Antidepressants for Reduction in Suicidality Could Deliver New Standard of Care for Bipolar Depression
-US Healthcare Journal
https://ushealthcarejournal.com/article/707706109-oral-antidepressants-for-reduction-in-suicidality-could-deliver-new-standard-of-care-for-bipolar-depression-nasdaq-nrxp
#PharmaNews #mentalillness #mentalhealth #SuicidePrevention #depression #ketamine #stress #PTSD #bipolar #NRX101 $NRXP
Article:
Why Is NRx Pharmaceuticals Stock Falling After Data From Suicidal Bipolar Depression Study
https://www.benzinga.com/trading-ideas/movers/24/04/38525975/why-is-nrx-pharmaceuticals-stock-falling-after-data-from-suicidal-bipolar-depression-study
$NRXP NEWS: NRx Pharmaceuticals (Nasdaq:NRXP) Announces Promising Findings in Phase 2b/3 Clinical Trial of NRX-101 vs. Lurasidone for Treatment of Suicidal Bipolar Depression
PR Newswire
Tue, Apr 30, 2024, 8:30 AM EDT
NRX-101 is first oral antidepressant to show 33% advantage in sustained remission in suicidality (not statistically significant at this sample size) and 75% advantage in relief from Akathisia relative to lurasidone - never previously shown with an oral antidepressant. Suicidality signal met the study's promising zone criteria and the akathisia signal approached statistical significance (P=0.076)
Both NRX-101 and lurasidone, an accepted standard of care in Bipolar Depression, demonstrated approximately 50% reduction in symptoms of depression
These data are comparable to previous statistically-significant finding of reduced suicidality and in the published STABIL-B trial and support an approval pathway via a 300-person registrational trial with sustained remission in suicidality as the primary endpoint
Company believes that an oral antidepressant that demonstrates reduction in suicidality has potential to become standard of care for treatment of bipolar depression.
Data from this study expand the potential utility of NRx-101 to treat both patients with suicidal bipolar depression (who will require prior use of ketamine) and those without subacute suicidality (nearly 7 million patients in the US).
RADNOR, Pa., April 30, 2024 /PRNewswire/ -- NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) ("NRx Pharmaceuticals", the "Company"), a clinical stage pharmaceutical company today that its Breakthrough Therapy designated investigational drug NRX-101 vs lurasidone demonstrated a promising, though not yet statistically significant 33% reduction in suicidality together with a 70% reduction (P=.076) reduction in symptoms of akathisia – a side effect of antidepressants that is closely linked to suicide and considered a medical emergency. Because of the high-risk nature of these patients, a placebo group could not be employed, and NRX-101, a fixed dose combination of D-cycloserine (DCS) and lurasidone, was compared to lurasidone alone (the standard of care). In the Company's previously published STABIL-B trial (STABIL-B), NRX-101 was demonstrated to be superior to lurasidone in reducing both depression and suicidality after ketamine while showing a trend towards reducing akathisia (a side effect involving restlessness and agitation that is considered a warning sign of impending suicide). In this trial, without prior use of ketamine, NRX-101 and lurasidone were comparable in their effect on depression. The trial was a randomized, prospective, double-blind study conducted at multiple sites in the Unites States whose protocol and statistical analysis plan may be viewed on www.clinicaltrials.gov (NCT03395392).
Onset of remission in suicidality
Onset of remission in suicidality
"We are gratified by these results, which extend the findings of the STABIL-B trial in suggesting that NRX-101 has the potential to be the first oral antidepressant to decrease potential for suicide, whereas all currently approved oral antidepressants are known to increase the risk of suicide," said Prof. Jonathan Javitt, MD, MPH, the Company's Chairman and Chief Scientist. "Should these findings be confirmed in a registrational trial of 300 patients, NRX-101 has the potential to represent a paradigm-changing blockbuster drug. The finding of a dramatic difference in akathisia was also seen in the STABIL-B trial and provides important mechanistic support for the difference seen on the Columbia Suicide Severity Rating Scale. Many of the patients who tragically die from suicide in bipolar depression are taking traditional antidepressants at the time of their death, a tragedy we have seen within the families of our investors and board members, as well as the many patients we have known. If today's findings are replicated in a registration-sized trial, we will change the world for patients who currently have a 50% lifetime risk of suicide attempt, a 20% lifetime risk of death by suicide, and whose only approved treatment option today is electroshock therapy."
"These findings are consistent with our original Phase 2 objectives and promising zone methodology in terms of a demonstrable advantage of NRX-101 compared to the standard of care in treating patients with bipolar depression who are known to be at high risk of suicide. We originally proposed to test suicidality, rather than depression as the primary endpoint for this trial and took the advice of senior FDA leadership that demonstrating a difference in suicidality might be too challenging. Today's findings demonstrate that differences in suicidality and akathisia can be demonstrated compared to best available antidepressant therapy in a properly sized registration trial and that superiority over placebo on the depression scale may readily be demonstrated in a less acute patient population where it would be safe to do so," said Dr. Philip Lavin, the study's Lead Methodologist. Dr. Lavin is one of the world's most widely published statisticians who has led the approval/clearance of more than 80 drugs, devices, and biologics.
In the current study, without prior use of ketamine, NRX-101 and lurasidone exhibited comparable antidepressant effects, each reducing depression (the primary endpoint) on the Montgomery Asberg Depression Rating Scale (MADRS) by about 50% from baseline. Lurasidone is known to reduce symptoms of depression by approximately 4 points in multiple registration trials compared to placebo.
Analysis of suicidality using the Columbia Suicide Severity Rating Scale (C-SSRS) demonstrated a sustained 33% advantage in remission from suicidality favoring NRX-101 (see figure). This difference was not statistically significant at the phase 2 sample size but met the study's original promising zone criteria and, if sustained in a registration trial of 300 or more patients, would be powered to yield a statistically significant result. The reduction in suicidality is comparable to that demonstrated after ketamine, both in the Company's STABIL-B trial and in an independently conducted trial comparing DCS to placebo after ketamine (Chen, et. al.). A meaningful remission in suicidality has not been demonstrated with any prior oral antidepressant drug – indeed, antidepressant drugs carry a Black Box warning of increased suicide risk.
Reduction in akathisia was first identified in the laboratory as a distinguishing feature of DCS and is the basis of the approved claims in the Company's Composition of Matter patents. Akathisia is often characterized as a state of agitation and motor restlessness that is associated with particularly impulsive and tragically effective attempts at suicide, such as hanging, shooting, jumping from buildings and in front of vehicles and trains. In this trial, a 75% relative difference was seen on the Barnes Akathisia Rating Scale (BARS), with two-sided P=0.076, which would be expected to achieve significance in a properly powered registration-sized trial. While reduction in akathisia is not proposed as a primary labeled indication, continued finding of a statistically significant reduction in this side effect would be highly supportive of a demonstrated primary endpoint of reduced suicidality and would provide clinical corroboration.
Based on these findings and widespread adoption of ketamine as initial treatment for suicidal depression, the Company believes that NRX-101 may become the drug of choice for potentiating the effect of ketamine in patients with acute and subacute suicidality. The FDA recently affirmed to the Company that the Special Protocol Agreement for this indication remains in place, subject to the Company filing a New Drug Approval for ketamine, which is expected by July 2024. Moreover the Company aims to explore the role of NRX-101 as primary treatment for the much larger population (approximately 7 million in the US) of patients with bipolar depression who do not have active suicidality and, therefore, do not require prior treatment with intravenous ketamine.
About NRx Pharmaceuticals
NRx Pharmaceuticals is a clinical-stage biopharmaceutical company developing therapeutics based on its NMDA platform for the treatment of central nervous system disorders, specifically suicidal bipolar depression, chronic pain and PTSD. The Company is developing NRX-101, an FDA-designated investigational Breakthrough Therapy for suicidal treatment-resistant bipolar depression and chronic pain. NRx has partnered with Alvogen and Lotus around the development and marketing of NRX-101 for the treatment of suicidal bipolar depression. NRX-101 additionally has potential to act as a non-opioid treatment for chronic pain, as well as a treatment for complicated UTI.
NRx has recently announced plans to submit a New Drug Application for HTX-100 (IV ketamine), through Hope Therapeutics, in the treatment of suicidal depression, based on results of well-controlled clinical trials conducted under the auspices of the US National Institutes of Health and newly obtained data from French health authorities, licensed under a data sharing agreement. NRx was awarded Fast Track Designation for development of ketamine (NRX-100) by the US FDA as part of a protocol to treat patients with acute suicidality.
About HOPE Therapeutics, Inc.
HOPE Therapeutics, Inc. (www.hopetherapeutics.com) is a Specialty Pharmaceutical Company, wholly-owned by NRX Pharmaceuticals focused on development and marketing of an FDA-approved form of intravenous ketamine for the treatment of acute suicidality and depression together with a digital therapeutic-enabled platform designed to augment and preserve the clinical benefit of NMDA-targeted drug therapy.
Notice Regarding Forward-Looking Statements
The information contained herein includes forward-looking statements within the meaning of Section 21E of the Securities Exchange Act of 1934, as amended, and Section 27A of the Securities Act of 1933, as amended. These statements include, among others, statements regarding the proposed public offering and the timing and the use of the proceeds from the offering. Forward-looking statements generally include statements that are predictive in nature and depend upon or refer to future events or conditions, and include words such as "may," "will," "should," "would," "expect," "plan," "believe," "intend," "look forward," and other similar expressions among others. These statements relate to future events or to the Company's future financial performance, and involve known and unknown risks, uncertainties and other factors that may cause the Company's actual results to be materially different from any future results, levels of activity, performance or achievements expressed or implied by these forward-looking statements. You should not place undue reliance on forward-looking statements since they involve known and unknown risks, uncertainties and other factors which are, in some cases, beyond the Company's control and which could, and likely will, materially affect actual results, levels of activity, performance or achievements. Any forward-looking statement reflects the Company's current views with respect to future events and is subject to these and other risks, uncertainties and assumptions relating to the Company's operations, results of operations, growth strategy and liquidity. More detailed information about the Company and the risk factors that may affect the realization of forward-looking statements is set forth in the Company's most recent Annual Report on Form 10-K and other filings with the Securities and Exchange Commission. Investors and security holders are urged to read these documents free of charge on the SEC's website at http://www.sec.gov. Except as may be required by applicable law, The Company assumes no obligation to publicly update or revise these forward-looking statements for any reason, or to update the reasons actual results could differ materially from those anticipated in these forward-looking statements, whether as a result of new information, future events or otherwise.
View original content to download multimedia:https://www.prnewswire.com/news-releases/nrx-pharmaceuticals-nasdaqnrxp-announces-promising-findings-in-phase-2b3-clinical-trial-of-nrx-101-vs-lurasidone-for-treatment-of-suicidal-bipolar-depression-302131482.html
SOURCE NRx Pharmaceuticals
$NRXP $3.63 +2.25% "The new national strategy focuses on a "whole of society" approach, which is unique-and critical -because no single entity alone can reduce suicide rates. But together, we have a real opportunity for impact." - NAM Chief Advocacy Officer https://t.co/FKUyFTYbUj
"The new national strategy focuses on a "whole of society" approach, which is unique-and critical -because no single entity alone can reduce suicide rates. But together, we have a real opportunity for impact." - NAM Chief Advocacy Officer @HannahWes https://t.co/FKUyFTYbUj
— NAMI (@NAMICommunicate) April 24, 2024
$NRXP $3.29 +4.78% New Data on Infection Avoidance and Development of pH Neutral Ketamine, for Intravenous & Subcutaneous Use NASDAQ: NRXP https://www.einpresswire.com/article/704132993/new-data-on-infection-avoidance-and-development-of-ph-neutral-ketamine-for-intravenous-subcutaneous-use-nasdaq-nrxp via @ein_news @NRxPharma
$NRXP $3.03 Demonstrating No Damage to Intestinal and Vaginal Flora in Validated Rodent Models Compared to Standard Antibiotics: Potential Implications for Avoidance of C. Difficile infections https://ir.nrxpharma.com/index.php?s=43&item=229 #News
$NRXP NRx Pharmaceutucals is focused on the high unmet need for lifesaving treatments for people with severe bipolar depression & PTSD in the presence of suicidality.
Follow our journey.
https://NRxPharma.com
#mentalhealth #depression #bipolar #PTSD #NRX101 #NRX100 #ketamine $NRXP
$NRXP New Data On Infection Avoidance And Development Of Ph Neutral Ketamine, For Intravenous & Subcutaneous Use
Read More from Today in Finance - https://todayinfinance.com/article/704132993-new-data-on-infection-avoidance-and-development-of-ph-neutral-ketamine-for-intravenous-subcutaneous-use-nasdaq-nrxp
#PharmaNews #mentalillness #mentalhealth #depression #ketamine #stress #PTSD #bipolar #NRX100 #NRX101 $NRXP
$NRXP PR Newswire -NRx Pharmaceuticals (NASDAQ:NRXP) Receives Notice from Nasdaq that the Company has Demonstrated Compliance with the Bid Price Requirement
Thu, April 18, 2024 at 8:32 AM EDT
In this article: NRXP
The Nasdaq Panel has determined to continue the listing of the Company's securities on The Nasdaq Stock Market and is closing this matter
Four upcoming milestones expected: data from trials in Suicidal Bipolar Depression and Chronic Pain, NDA filing for IV Ketamine and distribution of shares of HOPE Therapeutics to existing shareholders
Recent unanticipated achievements related to intravenous ketamine and complicated Urinary Tract Infection/pyelonephritis
RADNOR, Pa., April 18, 2024 /PRNewswire/ -- NRx Pharmaceuticals, Inc. (Nasdaq:NRXP) ("NRx Pharmaceuticals", "NRx", the "Company"), a clinical-stage biopharmaceutical company, today announced that (the "Company") has received confirmation from Nasdaq that they have demonstrated compliance with the Nasdaq bid price requirement in Listing Rule 5550(a)(2) and has determined to continue the listing of the Company's securities on The Nasdaq Stock Market and is closing this matter.
The Company is now in a position to reach four important milestones:
Release of top-line data from its trial of NRX-101 in Treatment Resistant Suicidal Bipolar Depression
Release of top-line data from Northwestern University's Department of Defense funded trial of the main component of NRX-101 in Chronic Pain
NDA filing for IV Ketamine for treatment of Suicidal Depression
Planned distribution of shares of HOPE Therapeutics to existing shareholders
"We greatly appreciate the support of our investors through the process of protecting our Nasdaq listing," said Dr. Jonathan Javitt, Founder, Chairman and Chief Scientist of NRx Pharmaceuticals. "We are now poised to reach an important series of milestones, have the potential to be transformative to the company and dramatically enhance shareholder value. In addition to the near-term readouts and filings that we have identified for investors, recent unanticipated advances in the formulation of new forms of ketamine and potential use of NRX-101 in the treatment of complicated UTI and pyelonephritis have significant potential to create value for shareholders."
About NRx Pharmaceuticals
NRx Pharmaceuticals is a clinical-stage biopharmaceutical company developing therapeutics based on its NMDA platform for the treatment of central nervous system disorders, specifically suicidal bipolar depression, chronic pain and PTSD. The Company is developing NRX-101, an FDA-designated investigational Breakthrough Therapy for suicidal treatment-resistant bipolar depression and chronic pain. NRx has partnered with Alvogen and Lotus around the development and marketing of NRX-101 for the treatment of suicidal bipolar depression. NRX-101 additionally has potential to act as a non-opioid treatment for chronic pain, as well as a treatment for complicated UTI.
NRx has recently announced plans to submit a New Drug Application for HTX-100 (IV ketamine), through Hope Therapeutics, in the treatment of suicidal depression, based on results of well-controlled clinical trials conducted under the auspices of the US National Institutes of Health and newly obtained data from French health authorities, licensed under a data sharing agreement. NRx was awarded Fast Track Designation for development of ketamine (NRX-100) by the US FDA as part of a protocol to treat patients with acute suicidality.
About HOPE Therapeutics, Inc.
HOPE Therapeutics, Inc. (www.hopetherapeutics.com) is a Specialty Pharmaceutical Company, wholly-owned by NRX Pharmaceuticals focused on development and marketing of an FDA-approved form of intravenous ketamine for the treatment of acute suicidality and depression together with a digital therapeutic-enabled platform designed to augment and preserve the clinical benefit of NMDA-targeted drug therapy.
Notice Regarding Forward-Looking Statements
The information contained herein includes forward-looking statements within the meaning of Section 21E of the Securities Exchange Act of 1934, as amended, and Section 27A of the Securities Act of 1933, as amended. These statements include, among others, statements regarding the proposed public offering and the timing and the use of the proceeds from the offering. Forward-looking statements generally include statements that are predictive in nature and depend upon or refer to future events or conditions, and include words such as "may," "will," "should," "would," "expect," "plan," "believe," "intend," "look forward," and other similar expressions among others. These statements relate to future events or to the Company's future financial performance, and involve known and unknown risks, uncertainties and other factors that may cause the Company's actual results to be materially different from any future results, levels of activity, performance or achievements expressed or implied by these forward-looking statements. You should not place undue reliance on forward-looking statements since they involve known and unknown risks, uncertainties and other factors which are, in some cases, beyond the Company's control and which could, and likely will, materially affect actual results, levels of activity, performance or achievements. Any forward-looking statement reflects the Company's current views with respect to future events and is subject to these and other risks, uncertainties and assumptions relating to the Company's operations, results of operations, growth strategy and liquidity. More detailed information about the Company and the risk factors that may affect the realization of forward-looking statements is set forth in the Company's most recent Annual Report on Form 10-K and other filings with the Securities and Exchange Commission. Investors and security holders are urged to read these documents free of charge on the SEC's website at http://www.sec.gov. Except as may be required by applicable law, The Company assumes no obligation to publicly update or revise these forward-looking statements for any reason, or to update the reasons actual results could differ materially from those anticipated in these forward-looking statements, whether as a result of new information, future events or otherwise.
HOPE Therapeutics, Inc. (PRNewsfoto/NRx Pharmaceuticals, Inc.)
HOPE Therapeutics, Inc. (PRNewsfoto/NRx Pharmaceuticals, Inc.)
Cision
Cision
View original content to download multimedia:https://www.prnewswire.com/news-releases/nrx-pharmaceuticals-nasdaqnrxp-receives-notice-from-nasdaq-that-the-company-has-demonstrated-compliance-with-the-bid-price-requirement-302120577.html
SOURCE NRx Pharmaceuticals, Inc.
$NRXP Press Release: NRx Pharmaceuticals, Inc. Announces Pricing of $2.0 Million Underwritten Public Offering of Common Stock
Thu, April 18, 2024 at 8:30 AM EDT
In this article: NRXP
RADNOR, Pa., April 18, 2024 /PRNewswire/ -- NRx Pharmaceuticals, Inc. (Nasdaq: NRXP), ("NRx Pharmaceuticals" or the "Company"), a clinical-stage biopharmaceutical company, today announced the pricing of its underwritten public offering of shares of its common stock at a public offering price of $3.30 per share, for aggregate gross proceeds of approximately $2.0 million, prior to deducting underwriting discounts and other offering expenses. In addition, the Company has granted the underwriters a 45-day option to purchase up to an additional 91,050 shares of common stock at the public offering price per share, less the underwriting discounts to cover over-allotments, if any. The offering is expected to close on April 19, 2024, subject to satisfaction of customary closing conditions.
EF Hutton LLC is acting as the sole book-runner for the offering.
NRx Pharmaceuticals intends to use the net proceeds from the offering for working capital and general corporate purposes, including its plan to initiate a national treatment protocol and safety database. The Company may also use the net proceeds from the offering to repay certain outstanding debt.
The securities are being offered by the Company pursuant to a "shelf" registration statement on Form S-3 (File No. 333-265492), including a base prospectus, filed with the U.S. Securities and Exchange Commission (the "SEC") on June 9, 2022, as amended on June 14, 2022, and declared effective on June 21, 2022.
The offering is being made only by means of a prospectus supplement and accompanying prospectus. A prospectus supplement describing the terms of the public offering will be filed with the SEC and will form a part of the effective registration statement. A preliminary prospectus supplement and accompanying prospectus relating to this offering have been filed with the SEC.
Copies of the prospectus supplement and the accompanying prospectus relating to this offering may be obtained, when available, on the SEC's website at http://www.sec.gov or by contacting EF Hutton LLC Attention: Syndicate Department, 590 Madison Avenue, 39th Floor, New York, NY 10022, by email at syndicate @Dowgal.
This press release shall not constitute an offer to sell or the solicitation of an offer to buy any of the securities described herein, nor shall there be any sale of these securities in any state or jurisdiction in which such offer, solicitation or sale would be unlawful prior to registration or qualification under the securities laws of any such state or jurisdiction. Any offer, if at all, will be made only by means of the prospectus supplement and accompanying prospectus forming a part of the effective registration statement.
About NRx Pharmaceuticals
NRx Pharmaceuticals is a clinical-stage biopharmaceutical company developing therapeutics based on its NMDA platform for the treatment of central nervous system disorders, specifically suicidal bipolar depression, chronic pain and PTSD. The Company is developing NRX-101, an FDA-designated investigational Breakthrough Therapy for suicidal treatment-resistant bipolar depression and chronic pain. NRx has partnered with Alvogen and Lotus around the development and marketing of NRX-101 for the treatment of suicidal bipolar depression. NRX-101 additionally has potential to act as a non-opioid treatment for chronic pain, as well as a treatment for complicated UTI.
NRx has recently announced plans to submit a New Drug Application for HTX-100 (IV ketamine), through Hope Therapeutics, in the treatment of suicidal depression, based on results of well-controlled clinical trials conducted under the auspices of the US National Institutes of Health and newly obtained data from French health authorities, licensed under a data sharing agreement. NRx was awarded Fast Track Designation for development of ketamine (NRX-100) by the US FDA as part of a protocol to treat patients with acute suicidality.
About HOPE Therapeutics, Inc.
HOPE Therapeutics, Inc. (www.hopetherapeutics.com) is a wholly owned subsidiary of NRX Pharmaceuticals focused on development and marketing of an FDA-approved form of intravenous ketamine for the treatment of acute suicidality and depression together with a digital therapeutic-enabled platform designed to augment and preserve the clinical benefit of NMDA-targeted drug therapy.
Notice Regarding Forward-Looking Statements
This press release contains forward-looking statements which involve substantial risks and uncertainties. Forward-looking statements are often identifiable by the words "anticipate," "believe," "continue," "could," "estimate," "expect," "intend," "may," "might," "objective," "ongoing," "plan," "predict," "project," "potential," "should," "will," or "would," or the negative of these terms, or other comparable terminology intended to identify statements about the future. These statements involve known and unknown risks, uncertainties and other factors that may cause the Company's actual results, levels of activity, performance or achievements to be materially different from the information expressed or implied by these forward-looking statements. Although the Company believes that it has a reasonable basis for making each forward-looking statement contained in this press release, the Company cautions that these statements are based on a combination of facts and factors currently known by the Company and its expectations of the future, about which the Company cannot be certain. Forward-looking statements are subject to considerable risks and uncertainties, as well as other factors that may cause the Company's actual results, levels of activity, performance or achievements to be materially different from the information expressed or implied by these forward-looking statements. These risks and uncertainties include, without limitation, risks and uncertainties related to whether or not the Company will be able to raise capital through the sale of its securities; the final terms of the proposed offering; market conditions; satisfaction of customary closing conditions related to the public offering; the Company's ability to maintain adequate liquidity and financing sources; various risks related to the Company's business operations; and other risks and uncertainties, including those described within the section entitled "Risk Factors" in the Company's Annual Report on Form 10-K for the year ended December 31, 2023, and other filings with the Securities and Exchange Commission. Investors and security holders are urged to read these documents free of charge on the SEC's website at http://www.sec.gov. Except as may be required by applicable law, the Company assumes no obligation to publicly update or revise these forward-looking statements for any reason, or to update the reasons actual results could differ materially from those anticipated in these forward-looking statements, whether as a result of new information, future events or otherwise.
SOURCE NRx Pharmaceuticals, Inc.
Cision
Cision
View original content to download multimedia:https://www.prnewswire.com/news-releases/nrx-pharmaceuticals-inc-announces-pricing-of-2-0-million-underwritten-public-offering-of-common-stock-302120498.html
SOURCE NRx Pharmaceuticals, Inc.
$NRXP News: NRx Pharmaceuticals, Inc. (NASDAQ:NRXP) Announces New Data on NRX-101 Demonstrating No Damage to Intestinal and Vaginal Flora in Validated Rodent Models Compared to Standard Antibiotics: Potential Implications for Avoidance of C. Difficile infections
Wed, April 17, 2024 at 8:30 AM EDT
In this article: NRXP
New data demonstrates no impact of NRX-101 on gut or vaginal flora – considered primary causes of pseudomembranous colitis due to C difficile and vaginal yeast infections
NRX-101 previously demonstrated potent activity against resistant urinary pathogens and has been shown to be fully excreted, unmetabolized, in the urine
NRX-101 has received FDA Qualified Infectious Disease Product (QIDP) and Fast Track Designation in Complicated Urinary Tract Infection (cUTI) and Pyelonephritis
RADNOR, Pa., April 17, 2024 /PRNewswire/ -- NRx Pharmaceuticals, Inc. (Nasdaq:NRXP) ("NRx Pharmaceuticals", "NRx", the "Company"), a clinical-stage biopharmaceutical company, today announced new data that demonstrate that in a rodent model NRX-101 shows no measurable damage to either intestinal or vaginal flora, compared to the significant negative effect caused by drugs such as ciprofloxacin. Antibiotics commonly used to treat complicated urinary tract infections (cUTI) are associated with pseudomembranous colitis caused by Clostridium difficile (C diff) and vaginal yeast infections, primarily owing to their impact on normal flora.
C. diff causes an intractable diarrhea in approximately 500,000 Americans each year and kills 1 in 11 Americans over age 65 who contract the infection. Costs of C. diff are estimated at $24,000 per patient and are significantly higher when C. diff occurs as part of a hospital admission. Whereas most antibiotics have substantial effect in the large bowel, the key component of NRX-101 (D-cycloserine) is entirely absorbed in the small intestine and excreted unmetabolized in the urine. If the nonclinical data reported today are replicated in patients, NRX-101 could represent the first antibiotic for cUTI and pyelonephritis that has essentially no risk of causing C. diff infection or vaginal yeast infection. There is an extensive literature surrounding the use of D-cycloserine to treat tuberculosis and cases of C. Diff are unknown.
D-cycloserine's effect as an antibiotic is based on its propensity to substitute for the amino acid alanine in the formation of the bacterial cell wall.
Dr. Michael Manyak, noted Professor of Urology and former Global Medical Director for Urology at Glaxo SmithKline stated "As a Urologist, I'm acutely aware of the importance of avoiding common side effects of current antibiotics. NRX-101's lack of impact on normal flora could potentially confer a distinct advantage for the product in for the treatment of cUTI and pyelonephritis. This profile could change the lives of the half million Americans each year who contract C. diff and save lives among the tens of thousands who die from antibiotic-induced C. diff infections. These potential advantages should make NRX-101 even more attractive to potential partners in this multi-billion-dollar market." Dr. Manyak serves as NRx's Medical Thought Leader for urology.
"While we have primarily focused on NRX-101 as a drug to treat CNS disease, these new and highly provocative findings suggest that NRX-101 could find a home as a first line treatment for cUTI and pyelonephritis, which afflicts more than 3 million Americans each year. Should the rodent model findings prove applicable to the people, the use of NRX-101 to treat cUTI without increasing the risk of C. diff infection could have multibillion dollar potential," said Stephen Willard, JD, Chief Executive Officer of NRx pharmaceuticals.
About D-Cycloserine in cUTI
The active antibiotic ingredient of NRX-101 is D-cycloserine (DCS) that was developed as an antibiotic in the 1950's and used worldwide for the treatment of tuberculosis. However, it fell out of favor with the development of trimethoprim/sulfa and various penicillins, cephalosporins, and tetracyclines, in part because of the CNS effects associated with DCS-induced blockade of the brain's NMDA receptor. In the course of its CNS research, NRx Pharmaceuticals has demonstrated that small doses of lurasidone counteract those CNS effects, potentially providing a new therapeutic life to DCS as an antibiotic. Over the ensuing decades, increased antibiotic resistance has rendered standard treatments for UTI ineffective in many cases and today 3 million Americans suffer from cUTI requiring increasingly toxic antibiotics, increasingly frequent intravenous therapy, and increased need for hospital admission. (Lodise TP, et. al. Open Forum Infectious Diseases https://doi.org/10.1093/ofid/ofac307)
Because DCS has the unique property of being highly concentrated, unmetabolized, in the urine with oral administration, the Company believes, and previous literature has suggested that DCS may effectively treat, and therefore help prevent, the need for intravenous and inpatient treatment of cUTI.
Moreover, because DCS is rapidly absorbed and excreted in the urine, NRX-101 has now been shown to have minimal tendency to disrupt the microbiome of the intestine and vagina and which can lead to secondary Clostridium difficile and/or yeast infections. C. diff associated colitis doubles hospital mortality and costs the American healthcare system up to $1.6 billion each year. (Drozd EM, et. al. Mortality, Hospital Costs, Payments, and Readmissions Associated With Clostridium difficile Infection DOI: 10.1097/IPC.0000000000000299) Additionally, DCS has no known association pulmonary fibrosis, a rare, lethal condition that has been associated with macrolide (tetracycline family) antibiotics.
NRX-101 has received FDA Qualified Infectious Disease Product (QIDP) and Fast Track Designations in Complicated Urinary Tract Infection and Pyelonephritis.
About NRx Pharmaceuticals
NRx Pharmaceuticals is a clinical-stage biopharmaceutical company developing therapeutics based on its NMDA platform for the treatment of central nervous system disorders, specifically suicidal bipolar depression, chronic pain and PTSD. The Company is developing NRX-101, an FDA-designated investigational Breakthrough Therapy for suicidal treatment-resistant bipolar depression and chronic pain. NRx has partnered with Alvogen and Lotus around the development and marketing of NRX-101 for the treatment of suicidal bipolar depression. NRX-101 additionally has potential to act as a non-opioid treatment for chronic pain, as well as a treatment for complicated UTI.
NRx has recently announced plans to submit a New Drug Application for HTX-100 (IV ketamine), through Hope Therapeutics, in the treatment of suicidal depression, based on results of well-controlled clinical trials conducted under the auspices of the US National Institutes of Health and newly obtained data from French health authorities, licensed under a data sharing agreement. NRx was awarded Fast Track Designation for development of ketamine (NRX-100) by the US FDA as part of a protocol to treat patients with acute suicidality.
About HOPE Therapeutics, Inc.
HOPE Therapeutics, Inc. (www.hopetherapeutics.com) is a Specialty Pharmaceutical Company, wholly-owned by NRX Pharmaceuticals focused on development and marketing of an FDA-approved form of intravenous ketamine for the treatment of acute suicidality and depression together with a digital therapeutic-enabled platform designed to augment and preserve the clinical benefit of NMDA-targeted drug therapy.
Notice Regarding Forward-Looking Statements
The information contained herein includes forward-looking statements within the meaning of Section 21E of the Securities Exchange Act of 1934, as amended, and Section 27A of the Securities Act of 1933, as amended. These statements include, among others, statements regarding the proposed public offering and the timing and the use of the proceeds from the offering. Forward-looking statements generally include statements that are predictive in nature and depend upon or refer to future events or conditions, and include words such as "may," "will," "should," "would," "expect," "plan," "believe," "intend," "look forward," and other similar expressions among others. These statements relate to future events or to the Company's future financial performance, and involve known and unknown risks, uncertainties and other factors that may cause the Company's actual results to be materially different from any future results, levels of activity, performance or achievements expressed or implied by these forward-looking statements. You should not place undue reliance on forward-looking statements since they involve known and unknown risks, uncertainties and other factors which are, in some cases, beyond the Company's control and which could, and likely will, materially affect actual results, levels of activity, performance or achievements. Any forward-looking statement reflects the Company's current views with respect to future events and is subject to these and other risks, uncertainties and assumptions relating to the Company's operations, results of operations, growth strategy and liquidity. More detailed information about the Company and the risk factors that may affect the realization of forward-looking statements is set forth in the Company's most recent Annual Report on Form 10-K and other filings with the Securities and Exchange Commission. Investors and security holders are urged to read these documents free of charge on the SEC's website at http://www.sec.gov. Except as may be required by applicable law, The Company assumes no obligation to publicly update or revise these forward-looking statements for any reason, or to update the reasons actual results could differ materially from those anticipated in these forward-looking statements, whether as a result of new information, future events or otherwise.
View original content to download multimedia:https://www.prnewswire.com/news-releases/nrx-pharmaceuticals-inc-nasdaqnrxp-announces-new-data-on-nrx-101-demonstrating-no-damage-to-intestinal-and-vaginal-flora-in-validated-rodent-models-compared-to-standard-antibiotics-potential-implications-for-avoidance-of-c--302119157.html
SOURCE NRx Pharmaceuticals, Inc.
$NRXP Successful Development of pH Neutral #Ketamine, Potentially Enabling Both Intravenous and Subcutaneous Use; Trials Progressing for Suicide Drug Treatment After Strong Q4 Results: $NRXP - @bostonherald
https://markets.financialcontent.com/bostonherald/article/getnews-2024-4-15-successful-development-of-ph-neutral-ketamine-potentially-enabling-both-intravenous-and-subcutaneous-use-trials-progressing-for-suicide-drug-treatment-after-strong-q4-results-nrxp/
#PharmaNews #depression #bipolar #NRX100 #NRX101
$NRXP NRx Pharma Shares Rise, Company to Distribute HOPE Therapeutics Shares as Dividend
https://www.marketwatch.com/story/nrx-pharma-shares-rise-company-to-distribute-hope-therapeutics-shares-as-dividend-9e8695cb?mod=mw_quote_news
$NRXP News: NRx Pharmaceuticals (NASDAQ:NRXP) Announces Development of New, Proprietary Formulation of HTX-100 (IV Ketamine)
Formulation based on prior patents by NRx founder
Achieved pH neutral formulation of ketamine, potentially enabling both intravenous (IV) and subcutaneous (SQ) administration
Company expects Composition of Matter patent protection
Company previously executed joint development agreement with US manufacturer of insulin pumps
RADNOR, Pa., April 15, 2024 /PRNewswire/ -- NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) ("NRx Pharmaceuticals", "NRx", the "Company"), a clinical-stage biopharmaceutical company, today announced that the Company has developed a novel, proprietary formulation of IV Ketamine for use as HTX-100. This new formulation has the key advantage of achieving neutral pH, in contrast to the acidic pH of generic formulations of ketamine. Acidic substances are tolerated when diluted for intravenous use, but cause pain and may cause skin ulcers if administered subcutaneously. This patentable invention may enable the administration of ketamine in insulin pump-like devices in the clinic setting, eliminating the requirement for intravenous infusion personnel. NeuroRx, Inc. previously executed a joint development agreement with a manufacturer of insulin pumps but has been awaiting a suitable, pH neutral formulation of ketamine.
With this proprietary formulation, developed with our partner Nephron Pharmaceuticals, a leading sterile products manufacturer, NRx is expected to generate one or more patents, such as composition of matter or formulation.
HTX-100 is expected to be marketed by HOPE Therapeutics, Inc., a wholly owned subsidiary of NRx.
"The current formulation of IV Ketamine was developed for battlefield use in the 1960s and has remained relatively static since then. With our new formulation, and Nephron's state of the art manufacturing expertise, we are pleased to be approaching the market with an improved formulation ready for use in modern medicine," said Dr. Jonathan Javitt, Founder, Chairman and Chief Scientist of NRx Pharmaceuticals. "Development of a pH-neutral, subcutaneous form of ketamine administration enables a significantly less complex route of administration in the outpatient setting. Moreover, advances in insulin pump technology create the potential to monitor the brain wave activity and other physiologic parameters associated with ketamine administration and modulate ketamine therapy based on the patient's individual physiologic response."
About NRx Pharmaceuticals
NRx Pharmaceuticals is a clinical-stage biopharmaceutical company developing therapeutics based on its NMDA platform for the treatment of central nervous system disorders, specifically suicidal bipolar depression, chronic pain and PTSD. The Company is developing NRX-101, an FDA-designated investigational Breakthrough Therapy for suicidal treatment-resistant bipolar depression and chronic pain. NRx has partnered with Alvogen and Lotus around the development and marketing of NRX-101 for the treatment of suicidal bipolar depression. NRX-101 additionally has potential to act as a non-opioid treatment for chronic pain, as well as a treatment for complicated UTI.
NRx has recently announced plans to submit a New Drug Application for HTX-100 (IV ketamine), through Hope Therapeutics, in the treatment of suicidal depression, based on results of well-controlled clinical trials conducted under the auspices of the US National Institutes of Health and newly obtained data from French health authorities, licensed under a data sharing agreement. NRx was awarded Fast Track Designation for development of ketamine (NRX-100) by the US FDA as part of a protocol to treat patients with acute suicidality.
About HOPE Therapeutics, Inc.
HOPE Therapeutics, Inc. (http://www.hopetherapeutics.com) is a Specialty Pharmaceutical Company, wholly-owned by NRx Pharmaceuticals focused on development and marketing of an FDA-approved form of intravenous ketamine for the treatment of acute suicidality and depression together with a digital therapeutic-enabled platform designed to augment and preserve the clinical benefit of NMDA-targeted drug therapy.
Notice Regarding Forward-Looking Statements
The information contained herein includes forward-looking statements within the meaning of Section 21E of the Securities Exchange Act of 1934, as amended, and Section 27A of the Securities Act of 1933, as amended. These statements include, among others, statements regarding the proposed public offering and the timing and the use of the proceeds from the offering. Forward-looking statements generally include statements that are predictive in nature and depend upon or refer to future events or conditions, and include words such as "may," "will," "should," "would," "expect," "plan," "believe," "intend," "look forward," and other similar expressions among others. These statements relate to future events or to the Company's future financial performance, and involve known and unknown risks, uncertainties and other factors that may cause the Company's actual results to be materially different from any future results, levels of activity, performance or achievements expressed or implied by these forward-looking statements. You should not place undue reliance on forward-looking statements since they involve known and unknown risks, uncertainties and other factors which are, in some cases, beyond the Company's control and which could, and likely will, materially affect actual results, levels of activity, performance or achievements. Any forward-looking statement reflects the Company's current views with respect to future events and is subject to these and other risks, uncertainties and assumptions relating to the Company's operations, results of operations, growth strategy and liquidity. More detailed information about the Company and the risk factors that may affect the realization of forward-looking statements is set forth in the Company's most recent Annual Report on Form 10-K and other filings with the Securities and Exchange Commission. Investors and security holders are urged to read these documents free of charge on the SEC's website at http://www.sec.gov. Except as may be required by applicable law, The Company assumes no obligation to publicly update or revise these forward-looking statements for any reason, or to update the reasons actual results could differ materially from those anticipated in these forward-looking statements, whether as a result of new information, future events or otherwise.
https://c212.net/c/img/favicon.png?sn=CL87341&sd=2024-04-15 View original content to download multimedia:https://www.prnewswire.com/news-releases/nrx-pharmaceuticals-nasdaqnrxp-announces-development-of-new-proprietary-formulation-of-htx-100-iv-ketamine-302116345.html
SOURCE NRx Pharmaceuticals, Inc.
$NRXP - NRX's (NASDAQ: $NRXP) #FactoidFriday #mentalhealth #mentalhealthawareness #mentalillness #mentalhealthmatters #bipolar #NRX101 #NRX100 $NRXP
Learn More - https://nrxpharma.com/nrx-100-nrx-101/#NRX100-101
$NRXP $5.93 7.67% Pre-market just maybe surpass $6 at open #Bullish
$NRXP - NRX's (NASDAQ: $NRXP) NMDA Modification Therapy And Its Multiple Applications From Mental Health To Chronic Pain-
@Benzinga
https://benzinga.com/partner/biotech/24/04/38180971/nrxs-nasdaq-nrxp-nmda-modification-therapy-and-its-multiple-applications-from-mental-health-to-ch
#PharmaNews #mentalillness #mentalhealth #mentalhealthmatters #depression #ketamine #stress #PTSD #bipolar #NRX100 #NRX101
$NRXP $5.51 HOD $5.545 Great chart performance this week.
$NRXP! NRX's (NASDAQ: NRXP) NMDA Modification Therapy And Its Multiple Applications From Mental Health To Chronic Pain $NRXP https://www.benzinga.com/partner/biotech/24/04/38180971/nrxs-nasdaq-nrxp-nmda-modification-therapy-and-its-multiple-applications-from-mental-health-to-ch via @Benzinga
$NRXP We wanted to alert you to a new report from MDPI - NRX-101 (D-Cycloserine + Lurasidone) Is Active against Drug-Resistant Urinary Pathogens In Vitro
#NRxPharmaceuticals Scientific Publications page - https://nrxpharma.com/scientific-publications
#NRxPharmaceuticals #PharmaNews #NRX101 $NRXP
We wanted to alert you to a new report from MDPI - NRX-101 (D-Cycloserine + Lurasidone) Is Active against Drug-Resistant Urinary Pathogens In Vitro#NRxPharmaceuticals Scientific Publications page - https://t.co/v5OFuSwbhg#NRxPharmaceuticals #PharmaNews #NRX101 $NRXP
— NRx Pharmaceuticals (@NRxPharma) April 9, 2024
$NRXP $5.53 +4.34% on Data/Funding News: "With positive data from this study and FDA comment, NRx becomes eligible to receive the balance of its first milestone (an additional $4 million) from partners Alvogen, Inc. and Lotus Pharmaceuticals, Inc. (1745.TW)"
$NRXP News: NRx Pharmaceuticals (NASDAQ:NRXP) Announces Data-Lock of Phase 2b/3 Trial of NRX-101 in Suicidal Treatment Resistant Bipolar Depression
PR Newswire
Mon, Apr 8, 2024, 8:30 AM EDT
In This Article:
NRXP
+1.92%
NRXPW
+17.65%
Data transferred for independent statistical analysis
Top-line data expected in April 2024
RADNOR, Pa., April 8, 2024 /PRNewswire/ -- NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) ("NRx Pharmaceuticals", the "Company"), a clinical-stage biopharmaceutical company, today announced that the Company has achieved data-lock in its Phase 2b/3 Suicidal Treatment Resistant Bipolar Depression Study with NRX-101. With data-lock, as forecast in last week's earnings call, the complete data set passed on for statistical analysis; top-line data release expected in April 2024.
With positive data from this study and FDA comment, NRx becomes eligible to receive the balance of its first milestone (an additional $4 million) from partners Alvogen, Inc. and Lotus Pharmaceuticals, Inc. (1745.TW). These partners would then be responsible for all future development costs in this indication. NRx retains rights for all other indications, including chronic pain and PTSD. NRx is then poised to receive $320 million in further milestones along with mid-teen royalties on Net Sales.
NRX-101 has been awarded Breakthrough Therapy Designation, Fast Track Designation, a Biomarker Letter of Support, and a Special Protocol Agreement by the FDA for treatment of Suicidal Treatment Resistant Bipolar Depression. It is the only oral medication to have demonstrated both reduced symptoms of depression and reduced suicidal ideation in patients with bipolar depression, a lethal disease that claims the lives of one in five who live with it.
The previous STABIL-B trial, (Ref. STABIL-B) resulted in the award of Breakthrough Therapy Designation for the use of NRX-101 following ketamine in hospitalized patients with Severe Bipolar Depression and Acute Suicidal Ideation and NRx was cleared by FDA to conduct a phase III trial in this regard under the Special Protocol Agreement. FDA then suggested that the Company explore whether NRX-101 might be applicable to the much larger population of patients with subacute suicidality who are cared for in the outpatient setting on a chronic basis. The purpose of this trial was to determine whether a signal could be detected that would support a much broader indication for NRX-101. Successful data in this regard would expand the potential market for NRX-101 from several hundred thousand patients per year to several million patients per year.
"Patients with active suicidal ideation have been excluded from the clinical trials of all previously known oral antidepressants. This trial of NRX-101 is the first under an FDA Investigational New Drug application to attempt to develop an oral therapy that offers patients with suicidal bipolar depression an alternative to electroconvulsive therapy, which is currently the only FDA-approved therapy for suicidal depression," said Dr. Jonathan Javitt, Founder, Chairman and Chief Scientist of NRx Pharmaceuticals. "The Company has also announced plans to submit a New Drug Application to FDA this quarter for the use of ketamine as a short-term therapy for suicidal bipolar depression. We believe this is the first trial to demonstrate that suicidal patients can safely be enrolled and treated within the context of a clinical trial, and this was only possible because of the close monitoring of our study investigators and site personnel. We are deeply appreciative of the tremendous work done by our clinical development team, the study sites and, most importantly, the patients who participated in our trial."
The Phase 2b/3 trial (www.clinicaltrials.gov NCT 03395392) is a randomized, prospective, multicenter, double-blind study comparing NRX-101 to lurasidone over six weeks. The Principal Investigator is Prof. Andrew Nierenberg of Harvard Massachusetts General Hospital. The primary efficacy endpoint is reduction in depression as measured on the Montgomery-Asberg Depression Rating Scale (MADRS) and the secondary endpoint is reduction of suicidal ideation as measured by the Clinical Global Impression Suicidality Scale (CGI-SS). As previously disclosed, treatment compliance and concordance of local raters to central raters scores was in excess of 94%, well above the industry standard that is normally seen in CNS trials.
About NRx Pharmaceuticals
NRx Pharmaceuticals is a clinical-stage biopharmaceutical company developing therapeutics based on its NMDA platform for the treatment of central nervous system disorders, specifically suicidal bipolar depression, chronic pain and PTSD. The Company is developing NRX-101, an FDA-designated investigational Breakthrough Therapy for suicidal treatment-resistant bipolar depression and chronic pain. NRx has partnered with Alvogen and Lotus around the development and marketing of NRX-101 for the treatment of suicidal bipolar depression. NRX-101 additionally has potential to act as a non-opioid treatment for chronic pain, as well as a treatment for complicated UTI.
NRx has recently announced plans to submit a New Drug Application for HTX-100 (IV ketamine), through Hope Therapeutics, in the treatment of suicidal depression, based on results of well-controlled clinical trials conducted under the auspices of the US National Institutes of Health and newly obtained data from French health authorities, licensed under a data sharing agreement. NRx was awarded Fast Track Designation for development of ketamine (NRX-100) by the US FDA as part of a protocol to treat patients with acute suicidality.
About HOPE Therapeutics, Inc.
HOPE Therapeutics, Inc. (www.hopetherapeutics.com) is a Specialty Pharmaceutical Company, wholly-owned by NRX Pharmaceuticals focused on development and marketing of an FDA-approved form of intravenous ketamine for the treatment of acute suicidality and depression together with a digital therapeutic-enabled platform designed to augment and preserve the clinical benefit of NMDA-targeted drug therapy.
Notice Regarding Forward-Looking Statements
The information contained herein includes forward-looking statements within the meaning of Section 21E of the Securities Exchange Act of 1934, as amended, and Section 27A of the Securities Act of 1933, as amended. These statements include, among others, statements regarding the proposed public offering and the timing and the use of the proceeds from the offering. Forward-looking statements generally include statements that are predictive in nature and depend upon or refer to future events or conditions, and include words such as "may," "will," "should," "would," "expect," "plan," "believe," "intend," "look forward," and other similar expressions among others. These statements relate to future events or to the Company's future financial performance, and involve known and unknown risks, uncertainties and other factors that may cause the Company's actual results to be materially different from any future results, levels of activity, performance or achievements expressed or implied by these forward-looking statements. You should not place undue reliance on forward-looking statements since they involve known and unknown risks, uncertainties and other factors which are, in some cases, beyond the Company's control and which could, and likely will, materially affect actual results, levels of activity, performance or achievements. Any forward-looking statement reflects the Company's current views with respect to future events and is subject to these and other risks, uncertainties and assumptions relating to the Company's operations, results of operations, growth strategy and liquidity. More detailed information about the Company and the risk factors that may affect the realization of forward-looking statements is set forth in the Company's most recent Annual Report on Form 10-K and other filings with the Securities and Exchange Commission. Investors and security holders are urged to read these documents free of charge on the SEC's website at http://www.sec.gov. Except as may be required by applicable law, The Company assumes no obligation to publicly update or revise these forward-looking statements for any reason, or to update the reasons actual results could differ materially from those anticipated in these forward-looking statements, whether as a result of new information, future events or otherwise.
CisionCision
Cision
View original content to download multimedia:https://www.prnewswire.com/news-releases/nrx-pharmaceuticals-nasdaqnrxp-announces-data-lock-of-phase-2b3-trial-of-nrx-101-in-suicidal-treatment-resistant-bipolar-depression-302110102.html
SOURCE NRx Pharmaceuticals, Inc
$NRXP Huge open at $5.30 Now $5.2991 +1.91% #bullish #shortsqueeze
$NRXP Four New Strong Buy Stocks for April 2024 - Great Article https://finance.yahoo.com/news/strong-buy-stocks-april-4th-104800566.html?soc_src=social-sh&soc_trk=tw&tsrc=twtr via @YahooFinance
$NRXP NRx Pharmaceuticals (NRXP) Upgraded to Strong Buy https://www.zacks.com/stock/news/2250692/nrx-pharmaceuticals-nrxp-upgraded-to-strong-buy-heres-what-you-should-know?art_rec=quote-stock_overview-zacks_news-ID01-txt-2250692
$NRXP $5.21 +9.92% Nice bullish move today. Shorts covering on news this week!
https://ir.nrxpharma.com/2024-04-01-NRx-Pharmaceuticals-Nasdaq-NRXP-Reports-Fourth-Quarter-and-Full-Year-2023-Financial-Results-and-Provides-Business-Update
$NRXP $5.00 0.26 (5.49%) Post split price HOD $5.06 Headed to $6.00+
$NRXP News! Digital Therapeutics make important Clinical and Regulatory steps forward - https://otsuka-us.com/news/rejoyn-fda-authorized
#PharmaNews #mentalillness #mentalhealth #mentalhealthawareness #mentalhealthmatters #depression #ketamine #stress #PTSD #bipolar #NRX100 #NRX101 $NRXP
Digital Therapeutics make important Clinical and Regulatory steps forward - https://t.co/kZAp292xP7#PharmaNews #mentalillness #mentalhealth #mentalhealthawareness #mentalhealthmatters #depression #ketamine #stress #PTSD #bipolar #NRX100 #NRX101 $NRXP
— NRx Pharmaceuticals (@NRxPharma) April 4, 2024
$NRXP NRx Pharmaceuticals (Nasdaq:NRXP) Launches HOPE Therapeutics
HOPE Therapeutics, an NRx Pharma daughter company, is developing NRX-100 (IV Ketamine) for treatment of Acute Suicidality and Depression.
Learn more about how we are bringing hope to 3.5 million lives in the USA. https://www.nrxpharma.com/hope-therapeutics/
$NRXP - Four potential near-term milestones, including data from two clinical trials, an NDA filing and an upcoming share dividend
-- 50% reduction in corporate overhead and 25% reduction in overall net loss in 2023, compared to 2024 with $0.20 per share improvement in negative earnings. Additions to working capital of $8 million in Q1 2024.
-- Company forecasts first commercial revenue in 2024 from sales of ketamine and related technologies. Company received advance of first milestone payments in 2024 for ongoing development of NRX-101 from Alvogen and Lotus Pharmaceuticals, Inc. (1975.TW)
-- Company announces new partnership around the first drug to potentially modify the underlying cause of schizophrenia
-- Data lock this week and top-line data expected this month, after completed enrollment of the Phase 2b/3 trial of NRX-101 in Treatment Resistant Bipolar Depression (TRBD); trial demonstrated 94% rater concordance, far in excess of industry norms and exceeded industry norms in medication compliance
-- Two new Investigational New Drug applications (INDs) accepted by the US Food and Drug Administration (FDA) for NRX-101 in Chronic Pain and Complicated UTI.
-- Data lock expected this week in 200-person DOD-funded trial of D-cycloserine (DCS), the key component of NRX-101, to treat chronic pain, conducted by Northwestern University
-- Grant of Qualified Infectious Disease Product (QIDP), Fast Track and Priority Review designations for NRX-101 in the treatment of Complicated Urinary Tract Infection (cUTI); Publication last week of QIDP-qualifying data in a peer-reviewed journal. NRx is reviewing partnership options
-- Established HOPE Therapeutics to develop and launch IV Ketamine together with related technologies with FDA New Drug Application to be submitted this year. In advance of FDA approval, HOPE is partnered with national 503b and 503a pharmacies to address the ketamine shortage declared by FDA. HOPE is planned to be spun out as a separate company to be owned by NRx, current NRx shareholders via a tax-free dividend, and new investors; Term Sheets received from prospective anchor investors for $60 million of new investment, once publicly listed
-- HOPE is presenting data from four randomized, prospective trials demonstrating safety and efficacy in 800 patients of IV Ketamine in treating severe and suicidal depression as the clinical basis for New Drug Application (NDA) for HTX-100 (IV Ketamine); expecting stability and CMC data sufficient for NDA filing by June 2024.
-- Added over $8 million in working capital, including an advance of a $5.1 million milestone payment from partners Alvogen, Inc. and Lotus Pharmaceuticals
-- Elected nationally recognized attorney in highly regulated industries, and healthcare specialist, Janet Rehnquist, Esq., to the Company's Board of Directors
-- Management has taken actions to address NASDAQ listing compliance and naked shorting of NRx securities
-- Management to host a conference call, April 1, 2024, at 8:30 AM ET
$NRXP $4.71 HOD $5.25 after the reverse split took effect today.
NRX-100/NRX-101: We are studying Suicidal Treatment Resistant Bipolar Depression
NRX-101 is a fixed-dose combination of D-Cycloserine and Lurasidone. It is the first drug in FDA trials for suicidal bipolar depression.
Looks like a 1:10 reverse split. No announcement that I can find but shows up in my Fidelity account at a price of $5.25 and appropriate reduction in share number. Hope it holds above $5. GLTA.
$NRXP News: NRx Pharmaceuticals (Nasdaq:NRXP) Reports Fourth Quarter and Full Year 2023 Financial Results and Provides Business Update
PR Newswire
Mon, Apr 1, 2024, 8:28 AM EDT15 min read
In This Article:
NRXP; NRXPW
Four potential near-term milestones, including data from two clinical trials, an NDA filing and an upcoming share dividend
-- 50% reduction in corporate overhead and 25% reduction in overall net loss in 2023, compared to 2024 with $0.20 per share improvement in negative earnings. Additions to working capital of $8 million in Q1 2024.
-- Company forecasts first commercial revenue in 2024 from sales of ketamine and related technologies. Company received advance of first milestone payments in 2024 for ongoing development of NRX-101 from Alvogen and Lotus Pharmaceuticals, Inc. (1975.TW)
-- Company announces new partnership around the first drug to potentially modify the underlying cause of schizophrenia
-- Data lock this week and top-line data expected this month, after completed enrollment of the Phase 2b/3 trial of NRX-101 in Treatment Resistant Bipolar Depression (TRBD); trial demonstrated 94% rater concordance, far in excess of industry norms and exceeded industry norms in medication compliance
-- Two new Investigational New Drug applications (INDs) accepted by the US Food and Drug Administration (FDA) for NRX-101 in Chronic Pain and Complicated UTI.
-- Data lock expected this week in 200-person DOD-funded trial of D-cycloserine (DCS), the key component of NRX-101, to treat chronic pain, conducted by Northwestern University
-- Grant of Qualified Infectious Disease Product (QIDP), Fast Track and Priority Review designations for NRX-101 in the treatment of Complicated Urinary Tract Infection (cUTI); Publication last week of QIDP-qualifying data in a peer-reviewed journal. NRx is reviewing partnership options
-- Established HOPE Therapeutics to develop and launch IV Ketamine together with related technologies with FDA New Drug Application to be submitted this year. In advance of FDA approval, HOPE is partnered with national 503b and 503a pharmacies to address the ketamine shortage declared by FDA. HOPE is planned to be spun out as a separate company to be owned by NRx, current NRx shareholders via a tax-free dividend, and new investors; Term Sheets received from prospective anchor investors for $60 million of new investment, once publicly listed
-- HOPE is presenting data from four randomized, prospective trials demonstrating safety and efficacy in 800 patients of IV Ketamine in treating severe and suicidal depression as the clinical basis for New Drug Application (NDA) for HTX-100 (IV Ketamine); expecting stability and CMC data sufficient for NDA filing by June 2024.
-- Added over $8 million in working capital, including an advance of a $5.1 million milestone payment from partners Alvogen, Inc. and Lotus Pharmaceuticals
-- Elected nationally recognized attorney in highly regulated industries, and healthcare specialist, Janet Rehnquist, Esq., to the Company's Board of Directors
-- Management has taken actions to address NASDAQ listing compliance and naked shorting of NRx securities
-- Management to host a conference call, April 1, 2024, at 8:30 AM ET
RADNOR, Pa., April 1, 2024 /PRNewswire/ -- NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) ("NRx Pharmaceuticals", the "Company"), a clinical-stage biopharmaceutical company, today announced its financial results for the quarter and year ended December 31, 2023 and provided a business update.
"2023 was a pivotal year for NRx in which we advanced from a single clinical trial in a single indication to a dramatically streamline its operations with a 50% reduction in overhead costs, a 25% reduction in overall costs, and a $0.20 per share improvement in negative earnings, while completing our clinical trial objectives. We expect data from two key trials this month and predict our first commercial revenue by the end of 2024. Over the past year the Company has navigated the most challenging business environment in the history of the biotechnology industry. Despite unprecedented headwinds, we negotiated a critical commercial partnership for our lead compound in bipolar depression, while retaining rights to the far larger indications of chronic pain and PTSD," said Stephen Willard, J.D., Chief Executive Officer and Director of NRx Pharmaceuticals. "We augmented our intellectual property portfolio to include the use of our lead compound in chronic pain and anticipate results of a 200-person efficacy trial that could open a multibillion dollar opportunity in this therapeutic area. We have acquired sufficient data on safety and efficacy of ketamine to support a New Drug Application for IV ketamine in acute suicidality. We established the foundation of a specialty pharmaceutical business around ketamine that we expect to yield positive cash flow by the end of 2024. Finally, we received unanticipated data supporting the use of our lead compound to treat complicated Urinary Tract Infection and Pyelonephritis, a condition that affects 3 million Americans and results in more than 15,000 deaths annually. We believe that NRx is poised for substantial growth in 2024 and look forward sharing further results in our conference call."
Fourth Quarter Clinical, Regulatory and Corporate Highlights
Development of NRX-101 for Treatment-Resistant Suicidal Bipolar Depression
The Company has announced today that it expects data lock this week and release of top line data this month in its Phase 2b/3 trial of NRX-101 in Suicidal Bipolar Depression. In 2023, the Company published results of a phase 2 trial demonstrating that oral NRX-101 extends the effect of IV ketamine in reducing both suicidality and depression in patients presenting to the hospital. This trial is designed to determine whether oral NRX-101 can reduce depression and suicidality in outpatients, which would be a massive broadening of the potential market for the drug. This potential expansion was guided by the Company's January 2023 meeting with the FDA in which NRx was advised to seek approval for NRX-101 as a drug for bipolar depression as a chronic, intermittent disease, a far broader indication.
With the completion of data collection, an unprecedented data integrity standard (94% agreement between site raters and central raters) has been achieved across the completed cohort of patients.
In 2023, the Company completed manufacture and Chemical Manufacturing Controls for NRX-101. This initiative is expected to yield stability data sufficient to support a shelf life in excess of two years at time of potential drug launch (should the clinical trials be successful). The completion of this manufacturing milestone allowed the Company to decrease its ongoing expenditure associated with manufacturing and development of chemical manufacturing controls.
During Q4 and in early 2024, the Company has continued to solidify its working relationship with Alvogen and Lotus, and begun working in unison to plan the final development and commercialization of NRX-101. These partners recently advanced $5 million of the first milestone to the Company. As previously announced, a successful readout from this trial and FDA interaction will trigger an additional $4 million milestone payment together with transfer of future development costs to our partner. The partnership provides for potential milestones of $329 million and a royalty reaching 15% on Net Sales.
NRX-101 for Treatment of Chronic Pain:
The Company has previously detailed the scientific basis for treatment of chronic pain with DCS as outlined in a 2016 scientific paper published by Schnitzer, et. al. and in the White Paper posted by the Company's Scientific Leadership (Sappko, et. al.). In 2023, the Company licensed US Patent 8,653,120 for the use of DCS in chronic pain and filed a now-accepted Investigational New Drug (IND) application with the FDA to initiate commercial drug development of NRX-101 in chronic pain.
Chronic pain affects more than 50 million American adults, compared to the approximately 3 million who report thoughts of suicide on an annual basis. There has been no new non-opioid class of drugs to treat nociceptive pain in the past two decades and NRX-101 has the potential to be the first N-methyl-D-aspartate (NMDA)-antagonist drug to seek approval for this indication. Today, ketamine is used off label to treat nociceptive pain, despite its clear limitations (addiction, neurotoxicity, hallucination, and the need for IV administration.)
The Company is advised that data lock will occur this week in a 200-person randomized prospective trial funded by the US DOD (NCT 03535688) in which patients with chronic pain were randomly assigned to DCS 400mg/day vs. placebo. Top line results will follow. Should these results support efficacy of DCS in the treatment of chronic low back pain, they are expected to provide a Breakthrough Therapy path towards treatment of chronic pain with DCS and DCS-containing medicines.
Spin out of HOPE Therapeutics and progress towards an NDA for NRX-100 (ketamine) in the treatment of suicidal depression.
When NRx met with the FDA in January 2023, the agency strongly encouraged the Company to develop NRX-100 (IV Ketamine) as a labeled drug, rather than rely on prior stabilization of suicidality and depression achieved via the common clinical practice of infusing generic ketamine compounded in licensed pharmacies. Shortly thereafter, the FDA issued the first of two advisory letters warning physicians against using compounded forms of ketamine and began a program of rigorous inspections of such pharmacies. Although there was once an expectation that intranasal administration of ketamine would be effective in treating suicidality, the attempts to demonstrate the clinical efficacy of nasal racemic ketamine for acute suicidality have not succeeded.
Accordingly, in Q3 the Company finalized a scientific collaboration with Prof. Marion Leboyer of Paris, France and Prof. Mocrane Abbar of Lyon, France in order to incorporate the results of a 156-person inpatient trial of intravenous ketamine vs. placebo for the stabilization of patients admitted for acute suicidality (the KETIS trial). The findings of the trial demonstrate a statistically significant reduction in both suicidality (the primary endpoint) and depression (the secondary endpoint) among patients treated with intravenous ketamine compared to those treated with placebo. (Link)
In the fourth quarter, the company similarly licensed data from Columbia University. In this trial, Dr. Michael Grunebaum and colleagues demonstrated a rapid and statistically significant reduction in Suicidal Ideation (SSI) at day 1 (p=0.0003) and in depression (P=0.0234), as measured by the Profile of Mood States (POMS) among patients randomized to IV Ketamine compared to those randomized to midazolam. This trial was published in the American Journal of Psychiatry https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5880701/.
The patient-level deidentified data from both studies have now been received by the Company and are being assembled in the electronic format required by the FDA. The Company believes that these randomized, blinded prospective trials encompassing nearly 240 participants, when submitted for review at a patient level could be sufficient to demonstrate preliminary safety and efficacy of intravenous ketamine in acutely suicidal patients. Data are expected to be transmitted to FDA by the end of 2Q23.
Submission of an NDA for the use of IV Ketamine is dependent upon submission of a manufacturing file documenting the manufacture of a presentation of ketamine suitable for single-patient use in the treatment of suicidal depression. In November 2023, the Company initiated manufacture of ketamine together with Nephron Pharmaceuticals, Inc. (West Columbia, SC) to develop a single patient presentation of ketamine. This formulation is expected to overcome some of the formulation deficiencies of existing forms of ketamine (developed for anesthesia) and is expected to have diversion-resistant and tamper-resistant features.
On March 30, 2024, the Company booked first commercial delivery of ketamine manufactured to 503b pharmacy standards from Nephron. HOPE will be distributing this presentation to qualified ketamine clinics in coordination with Nephron under Nephron's 503b pharmacy license in light of the current FDA-declared drug shortage of ketamine, starting this month (April 2024).
Treatment of Urinary Tract Infection (UTI) and Urosepsis:
Although treatment of UTI is quite different from use of NRX-101 to treat Central Nervous System disorders, D-cycloserine was originally developed as an antibiotic because of its role in disrupting the cell wall of certain pathogens. This is true of a number of drugs used in psychiatry today. D-cycloserine fell out of favor as an antibiotic in the 1970s because of the CNS effects caused by its NMDA-blocking properties and because of the widespread availability of effective first and second-generation antibiotics.
During Q3 2023, NRx tested NRX-101 and its components against resistant pathogens that appear on the Congressionally-mandated Qualified Infectious Disease Product (QIDP) list and proved in vitro effectiveness against antibiotic-resistant E. coli, Pseudomonas, and Acinetobacter. Accordingly, NRx was granted QIDP designation, Fast Track Designation, and Priority Review by the US FDA.
In recent years, increased antibiotic resistance to common pathogens that cause urinary tract infections and urosepsis (i.e., sepsis originating in the urinary tract) has resulted in a marked increase in cUTI, hospitalization, and death from urosepsis. The US Center for Disease Control and Prevention reports that more than 1.7 million Americans contract sepsis each year, of whom at least 350,000 die during their hospitalization or are discharged to hospice (CDC Sepsis Ref.). There are approximately 3 million patients per year who contract cUTI in the US annually (Lodise, et. al.). Additionally, should NRX-101 succeed in clinical trials, the Company will consider developing a follow-on product that is anticipated to achieve another 20 years of patent exclusivity.
Qualification for QIDP affords a sponsor five years of additional market exclusivity from FDA, regardless of patent status.
The Company does not anticipate funding this initiative with core NRx assets and is exploring structures for partnership opportunities. Should the Company or its partners succeed in serving 10% of the cUTI market, the Company believes that the revenue from NRX-101 has the potential to hundreds of million annually, based on 3 million cases per year (Lodise, et. al.) in the US and potential pricing of over $3,500/course of therapy.
Cash runway and financing
The Company continues to believe cash on hand is sufficient to fund operations through potential delivery of the upcoming milestones described herein.
Financial Results for the Quarter and Year Ended December 31, 2023
For the three months ended December 31, 2023, we at NRx Pharmaceuticals reduced our net loss from $10.2 million in the final quarter of 2022 to $4.3 million in 2023, representing nearly a 60% improvement year over year. For that same period, we reduced research and development expenses from $4.5 million in 2022 to $2.5 million in 2023, while substantially improving and finalizing our clinical trial enrollment. The $2.0 million decrease is related primarily to a decrease of $1.1 million in clinical trial expenses, $0.6 million in stock-based compensation, and $0.2 million in consulting and personnel wage costs. Also in that 3 month period we recorded a 67% reduction in general and administrative expenses, from $5.4 million in 2022 to $1.8 million in 2023. The decrease of $3.6 million is related primarily to a decrease of $1.3 million in insurance expenses, $1.3 million in stock-based compensation, $0.5 million in employee expenses, $0.2 million in legal and professional consulting fees, and $0.2 million in franchise tax expenses.
For the year ended December 31, 2023, NRx Pharmaceuticals reduced its net loss to $30.2 million compared to $39.8 million in the prior year. These efficiencies represent an improvement in net loss of nearly $10.0 million year over and a 20 cent, or 34%, improvement in net loss per share year over year. Over that annual period we recorded $13.4 million of research and development expenses compared to $17.0 million for the same period in 2022 representing a 21% decrease year over year. The decrease of $3.6 is related primarily to a decrease of $2.1 million in clinical trial and development expenses, $0.9 million related to fees paid to regulatory and process development consultants, $0.8 million in stock-based compensation while offset by a $0.2 million increase in patent costs as our patent portfolio has expanded.
Please note that the improvement in G&A expenses is even larger than the improvement in other areas. We decreased G&A by $13.1 million, from $27.3 million in 2022 to $14.2 million in 2023, nearly a 50% decrease year over year.
As of December 31, 2023, we had $4.6 million in cash and cash equivalents. Over the first three months of 2024 we improved our access to working capital by $8 million total, representing $2.9 million from equity sales and $5.1 million from the Alvogen milestone advance, while reducing our corporate indebtedness by 50% as shown in our financial statements.
We continue to implement operational efficiencies to extend runway and focus on our path to generating revenue. We believe that the near-term delivery of clinical trial data and the planned the launch of HOPE Therapeutics will be defining events in the second quarter of 2024.
Conference Call and Webcast Details
A live webcast of the conference call will be available on the Company's website at 8:30 a.m. ET today, at https://ir.nrxpharma.com/events. An archive of the webcast will be available on the Company's website for 30 days. Participants that are unable to join the webcast can access the conference call via telephone by dialing domestically 1-877-704-4453 or internationally 1-201-389-0920.
About NRx Pharmaceuticals
NRx Pharmaceuticals is a clinical-stage biopharmaceutical company developing therapeutics based on its NMDA platform for the treatment of central nervous system disorders, specifically suicidal bipolar depression, chronic pain and PTSD. The Company is developing NRX-101, an FDA-designated investigational Breakthrough Therapy for suicidal treatment-resistant bipolar depression and chronic pain. NRx has partnered with Alvogen and Lotus around the development and marketing of NRX-101 for the treatment of suicidal bipolar depression. NRX-101 additionally has potential to act as a non-opioid treatment for chronic pain, as well as a treatment for complicated UTI.
NRx has recently announced plans to submit a New Drug Application for HTX-100 (IV ketamine), through Hope Therapeutics, in the treatment of suicidal depression, based on results of well-controlled clinical trials conducted under the auspices of the US National Institutes of Health and newly obtained data from French health authorities, licensed under a data sharing agreement. NRx was awarded Fast Track Designation for development of ketamine (NRX-100) by the US FDA as part of a protocol to treat patients with acute suicidality.
About HOPE Therapeutics, Inc.
HOPE Therapeutics, Inc. (www.hopetherapeutics.com) is a Specialty Pharmaceutical Company, wholly-owned by NRX Pharmaceuticals focused on development and marketing of an FDA-approved form of intravenous ketamine for the treatment of acute suicidality and depression together with a digital therapeutic-enabled platform designed to augment and preserve the clinical benefit of NMDA-targeted drug therapy.
Notice Regarding Forward-Looking Statements
The information contained herein includes forward-looking statements within the meaning of Section 21E of the Securities Exchange Act of 1934, as amended, and Section 27A of the Securities Act of 1933, as amended. These statements include, among others, statements regarding the proposed public offering and the timing and the use of the proceeds from the offering. Forward-looking statements generally include statements that are predictive in nature and depend upon or refer to future events or conditions, and include words such as "may," "will," "should," "would," "expect," "plan," "believe," "intend," "look forward," and other similar expressions among others. These statements relate to future events or to the Company's future financial performance, and involve known and unknown risks, uncertainties and other factors that may cause the Company's actual results to be materially different from any future results, levels of activity, performance or achievements expressed or implied by these forward-looking statements. You should not place undue reliance on forward-looking statements since they involve known and unknown risks, uncertainties and other factors which are, in some cases, beyond the Company's control and which could, and likely will, materially affect actual results, levels of activity, performance or achievements. Any forward-looking statement reflects the Company's current views with respect to future events and is subject to these and other risks, uncertainties and assumptions relating to the Company's operations, results of operations, growth strategy and liquidity. More detailed information about the Company and the risk factors that may affect the realization of forward-looking statements is set forth in the Company's most recent Annual Report on Form 10-K and other filings with the Securities and Exchange Commission. Investors and security holders are urged to read these documents free of charge on the SEC's website at http://www.sec.gov. Except as may be required by applicable law, The Company assumes no obligation to publicly update or revise these forward-looking statements for any reason, or to update the reasons actual results could differ materially from those anticipated in these forward-looking statements, whether as a result of new information, future events or otherwise.
View original content to download multimedia:https://www.prnewswire.com/news-releases/nrx-pharmaceuticals-nasdaqnrxp-reports-fourth-quarter-and-full-year-2023-financial-results-and-provides-business-update-302104468.html
SOURCE NRx Pharmaceuticals, Inc.
$NRXP company newsletter sign-up, for any new investors. Strong financials reported today!
http://www.ir.nrxpharma.com/email-alerts
NRXP: Effective Tues. April 2,2024 a one for 10 reverse split:
https://hedgefollow.com/upcoming-stock-splits.php
|
Followers
|
52
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
1125
|
|
Created
|
12/15/20
|
Type
|
Free
|
| Moderators ProfitScout jedijazz | |||





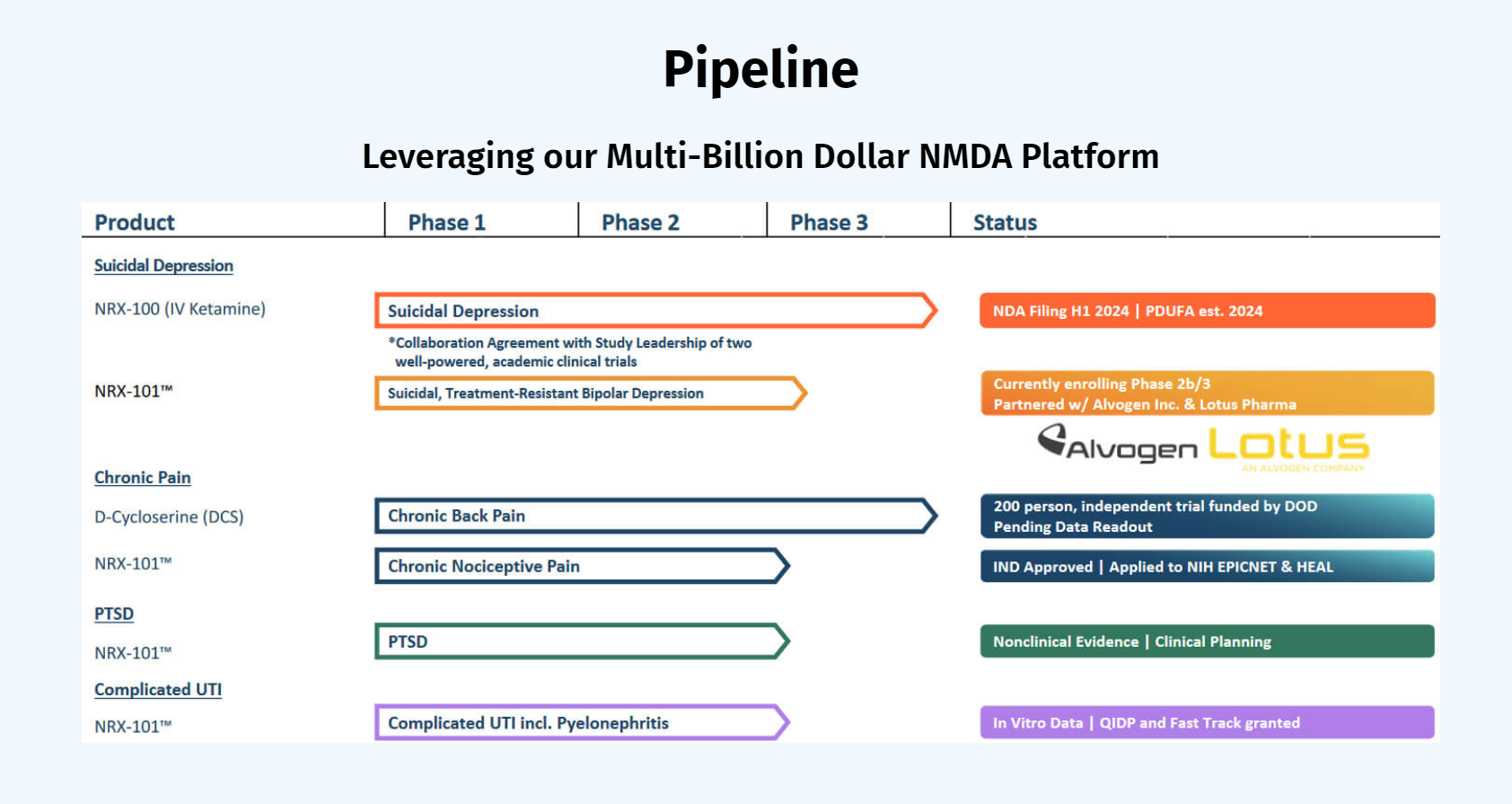
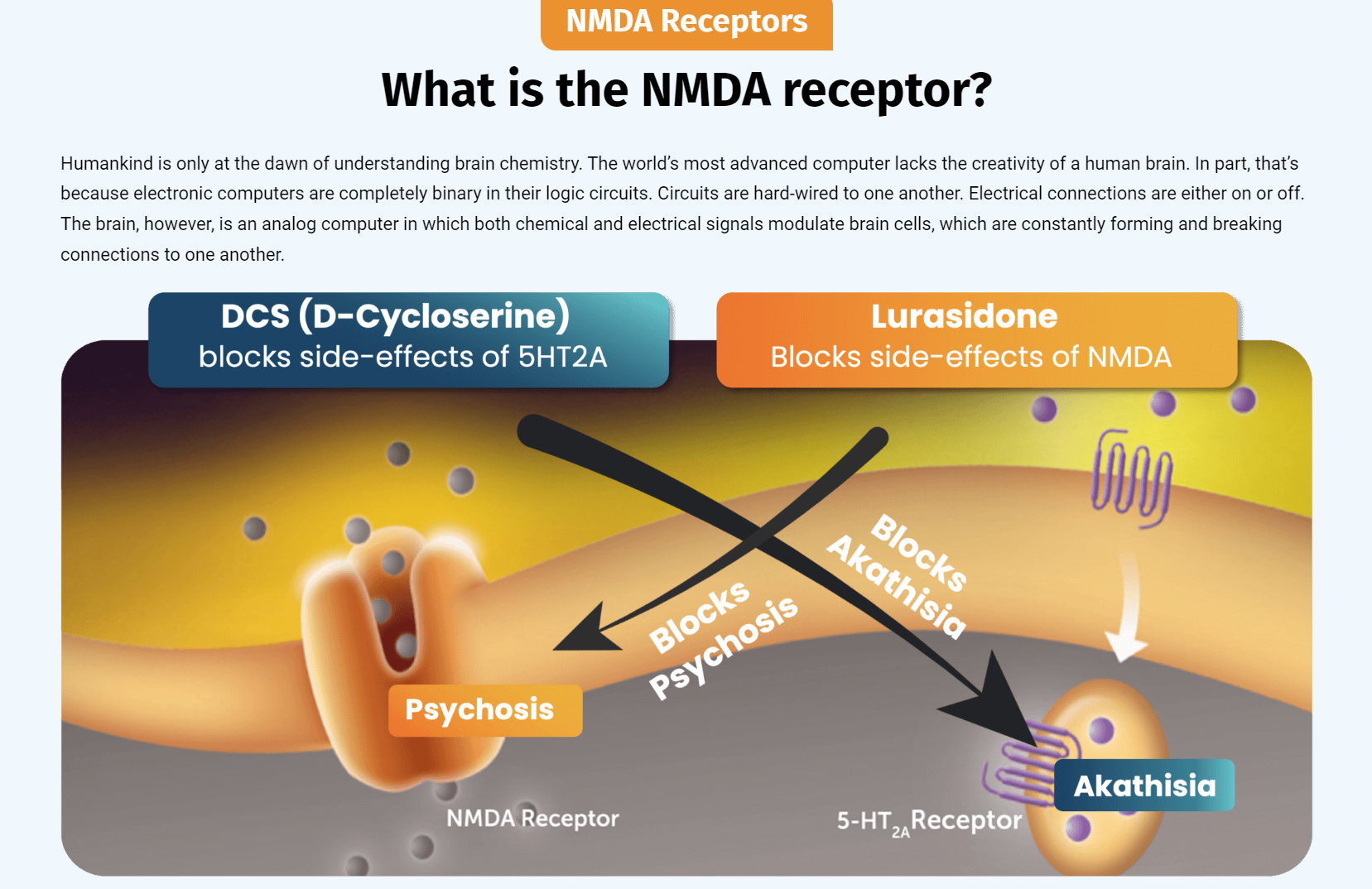







Both drugs demonstrated > 50% response for treating depression. NRX-101 demonstrated a mean 76% reduction in symptoms of akathisia compared to lurasidone that was sustained over 42 days (Effect...

New data demonstrates no impact of NRX-101 on gut or vaginal flora – considered primary causes of pseudomembranous colitis due to C difficile and vaginal yeast infections NRX-101 previously...
Formulation based on prior patents by NRx founder Achieved pH neutral formulation of ketamine, potentially enabling both intravenous (IV) and subcutaneous (SQ) administration Company expects...
Data transferred for independent statistical analysis Top-line data expected in April 2024 RADNOR, Pa., April 8, 2024 /PRNewswire/ -- NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) ("NRx...
Four potential near-term milestones, including data from two clinical trials, an NDA filing and an upcoming share dividend -- 50% reduction in corporate overhead and 25% reduction in overall net...
Four potential near-term milestones, including data from two clinical trials, an NDA filing and a share dividend RADNOR, Pa., March 28, 2024 /PRNewswire/ -- NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) ("
NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) ("NRx Pharmaceuticals", the "Company"), a clinical-stage biopharmaceutical company, today announced that it will release its fourth quarter and full year...
NRx Pharmaceuticals, Inc. (Nasdaq:NRXP) ("NRx Pharmaceuticals", the "Company"), a clinical-stage biopharmaceutical company, today announced that Dr. Jonathan Javitt, Chairman and Chief Scientist...
NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) ("NRx Pharmaceuticals", the "Company"), a clinical-stage biopharmaceutical company, today announced that it will release its fourth quarter and full year...
61.4% (56,781,354) of eligible shares voted 94.4% of votes were cast in favor of the resolution RADNOR, Pa., March 21, 2024 /PRNewswire/ -- NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) ("NRx...
NRx Pharmaceuticals Board of Directors has authorized its Chairman, CEO, and management to take all necessary steps to affect the Dividend and Royalty Rights to NRXP Shareholders and applicable...
Initiative is intended to protect shareholder value through continued compliance with Nasdaq listing rules and elimination of naked short sales positions in the Company's securities The Company is...
Company to receive first allocation of ketamine for sale by month end Partners preparing to ship IV Ketamine to full range of customers via 503a and 503b pharmacies NRx Pharmaceuticals and HOPE...
Marks a major step in the development of what could be the first drug approved for Suicidal Bipolar Depression The study database is being cleaned and locked; statistical analysis and top-line...
NRx Pharmaceuticals received approximately $1.0 million in cash from an existing investor Shares were sold at $0.38, a 26.7% premium the recent share offering, along with one common 5-year warrant...
In the news release, NRx Pharmaceuticals, Inc. Announces Pricing of $1.5 Million Underwritten Public Offering of Common Stock, issued 27-Feb-2024 by NRx Pharmaceuticals, Inc. over PR Newswire, we...
NRx Pharmaceuticals, Inc. (Nasdaq: NRXP), ("NRx Pharmaceuticals" or the "Company"), a clinical-stage biopharmaceutical company, today announced that it intends to offer to sell shares of its...
Presentation available on NRx Pharmaceuticals website at https://www.nrxpharma.com/hope-therapeutics/ KEY UPDATES ARE AS FOLLOWS: NRx management is proposing to award 50% of founding shares in...
DISCLAIMER:
Nothing in the contents transmitted on this board should be construed as an investment advisory, nor should it be used to make investment decisions.
There is no express or implied solicitation to buy or sell securities.
The author(s) may have positions in the stocks or financial relationships with the company or companies discussed and may trade in the stocks mentioned.
Readers are advised to conduct their own due diligence prior to considering buying or selling any stock. All information should be considered for information purposes only.
No stock exchange has approved or disapproved of the information here.
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
