Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
BLDV will Acquire 30% of CannSan Cannabis Sanitation......
"CannSan Platform will be Global effort that will help Operators with Sanitation Compliance
DENVER, Jan. 23, 2018 (GLOBE NEWSWIRE) -- Blue Diamond Ventures Inc. (OTCPK:BLDV) today, Clean 1st LLC, the parent company of Cannabis Clean and a wholly owned subsidiary of BLDV announces it will own 30% of the newly organized CannSan Cannabis Sanitation Technology Platform.
Cannabis Clean has been acting as a Building Services Contractor (BSC) in Denver, Colorado for the last three years and has struggled with a business plan that would allow the Company to expand to other markets in USA, Canada, and beyond. “The business model of a BSC is highly dependent on labor and overlooks the potential of being a Technology Resource to the Industry,” said Josh Alper CEO of BLDV. “Our work in Canada has revealed the challenges that these operators face, a comprehensive approach that includes, Consulting, Training, Equipment Supply, and Certification is what the Industry needs.”
Cannabis Clean will still operate as a BSC in Denver and will license the Cannabis Clean name to CannSan Certified organizations across North America. “CannSan Certification is at the heart of this effort,” says Steve McMorrow, President of Clean 1st LLC. “Certification is being offered for Facilities, Equipment, Workers and will create a verified end-to-end supply chain for Sanitation Compliance.”
The CannSan Technology Platform and CannSan Certification is being driven by Tyson Bernthal, a well-known Sanitation Industry Expert. “Tyson is working as a consultant to CannSan to integrate the latest equipment and technology in the platform,” continued Mr. McMorrow. “This is an exciting time at Cannabis Clean, this Technology Platform, combined with e-learning and the CannSan Certification process will allow us to build on the work done to date, and have a scalable business model.”
Listen to BLDV interview today on the Uptick Newswire. https://upticknewswire.com/featured-interview-ceo-joshua-alper-of-blue-diamond-ventures-inc-otcpink-bldv-4
About BLDV: Blue Diamond Ventures, Inc. seeks to partner with individuals and companies that share a common synergy, mission and vision to enable products/services that are produced, delivered and consumed utilizing fewer natural resources, providing a sustainable alternative to traditional products on the market today. As a diversified customer, centric Management / Holding Company; Blue Diamond Ventures, Inc. seeks opportunities in various markets and is driven by critical thinking and the scientific method.
Safe Harbor: This press release contains forward-looking statements. Such forward-looking statements are subject to several risks, assumptions, and uncertainties that could cause the Company's actual results to differ materially from those projected in such statements. Forward-looking statements speak only as of the date made and are not guarantees of future performance. We undertake no obligation to publicly revise any forward-looking statements."
Contact:
Blue Diamond Ventures Inc.
535 N. Michigan Avenue, Suite 3001, Chicago, Illinois, 60611
Joshua B. Alper Chief Executive Officer - (844) 637-6377 info@bldvinc.com
www.bldvinc.com
https://www.otcmarkets.com/stock/BLDV/news/Blue-Diamond-Ventures-Inc-Cannabis-Clean-Finds-Path-Forward?id=181156
Thanks for $$BLDV & all the charts Pete!
OGRMF~"Canadian Cannabis Producer With $100,000,000 For International Expansion"......
"Canadian Cannabis Producer With $100 Million For International Expansion" https://seekingalpha.com/article/4149238-canadian-cannabis-producer-100-million-international-expansion
$$OGRMF
$PMCB Institutional Stock Ownership and Shareholders PMCB
https://fintel.io/so/us/pmcb
PharmaCyte Biotech, Inc. (OTC:PMCB) has 4 institutional investors and shareholders that have filed 13D/G or 13F forms with the Securities Exchange Commission (SEC). These institutions hold a total of 2,054,073 shares. Largest shareholders include Panagora Asset Management Inc, Fisher Asset Management, LLC, Adell Harriman & Carpenter Inc, and Moloney Securities Asset Management, LLC.
PharmaCyte Biotech, Inc. (OTC:PMCB) ownership summary shows current positions in the company by institutions and funds, as well as latest changes in position size. Major shareholders can include individual investors, mutual funds, hedge funds, or institutions. The Schedule 13D indicates that the investor holds more than 5% of the company and intends to actively pursue a change in business strategy. Schedule 13G indicates a passive investment of over 5%. Green rows indicate new positions. Red rows indicate closed positions. Click the link icon to see the full transaction history.
$PMCB The "Financial Times”, the world’s leading global business publication, has just published that PharmaCyte has completed the preparation of the Master Cell Bank of the cells destined to be part of the treatment for pancreatic cancer and the cells will now enter the testing phase. Both of these activities were performed by Eurofins as a subcontractor. Austrianova will receive the cells, once testing is complete, and commence the manufacture of the the encapsulated cell clinical trial product
https://markets.ft.com/data/announce/detail?dockey=600-201802060900BIZWIRE_USPRX____BW5609-1
$PMCB PharmaCyte Discusses Phase 2b Clinical Trial in Pancreatic Cancer PMCB
LAGUNA HILLS, Calif.--(BUSINESS WIRE)-- PharmaCyte Biotech, Inc. (OTCQB: PMCB), a clinical stage biotechnology company focused on developing targeted cellular therapies for cancer and diabetes using its signature live-cell encapsulation technology, Cell-in-a-Box®, today explains PharmaCyte’s plans to conduct a Phase 2b clinical trial.
When PharmaCyte met with the FDA in January of 2017, PharmaCyte’s pre-IND meeting submission was predicated on PharmaCyte conducting a Phase 2b trial. During discussions with the U.S. Food and Drug Administration (FDA), PharmaCyte asked whether the data from that planned trial could be considered “pivotal” and thus support registration for marketing purposes. The FDA indicated that this was a possibility, but that the trial would have to be much larger than PharmaCyte was planning and the data would have to be markedly superior to the data seen with the comparator treatment.
PharmaCyte’s decision to conduct a Phase 2b trial rather than a pivotal trial was made relatively recently based on advice from PharmaCyte’s consulting oncologists, Chief Medical Officer and information obtained from PharmaCyte’s Advisory Board. PharmaCyte must rely on data from two European trials from 27 patients that were conducted about 20 years ago. The data from these trials are incomplete when compared to what is now required by the FDA to support a pivotal trial.
The Phase 2b trial is designed to determine how effective and safe multiple courses of ifosfamide will be in patients with locally advanced, non-metastatic, inoperable pancreatic cancer (LAPC) and how PharmaCyte’s treatment compares to a commonly used treatment for LAPC after patients’ tumors no longer respond following 4 to 6 months of the combination therapy of gemcitabine and Abraxane®. PharmaCyte is designing a Phase 2b clinical trial that, if successful, it believes will give the company a much more solid foundation for dealing with the FDA with the goal of bringing its pancreatic cancer therapy to market.
The planned Phase 2b trial will be significantly larger than the original Phase 2b trial PharmaCyte previously discussed with the FDA and will include multiple courses of low dose ifosfamide (the earlier trials used only two courses). This trial will also provide better statistical analyses of PharmaCyte’s therapy for LAPC and the comparator arm in terms of survival and safety. Also, the possibility exists that if the data from PharmaCyte’s therapy are significantly better than the data from the comparator arm, this may allow PharmaCyte to apply to the FDA for accelerated approval.
About PharmaCyte Biotech
PharmaCyte Biotech is a clinical stage biotechnology company developing cellular therapies for cancer and diabetes based upon a proprietary cellulose-based live cell encapsulation technology known as “Cell-in-a-Box®.” This technology will be used as a platform upon which therapies for several types of cancer and diabetes are being developed.
PharmaCyte’s therapy for cancer involves encapsulating genetically engineered human cells that convert an inactive chemotherapy drug into its active or “cancer-killing” form. For pancreatic cancer, these encapsulated cells are implanted in the blood supply to the patient’s tumor as close as possible to the site of the tumor. Once implanted, a chemotherapy drug that is normally activated in the liver (ifosfamide) is given intravenously at one-third the normal dose. The ifosfamide is carried by the circulatory system to where the encapsulated cells have been implanted. When the ifosfamide flows through pores in the capsules, the live cells inside act as a “bio-artificial liver” and activate the chemotherapy drug at the site of the cancer. This “targeted chemotherapy” has proven effective and safe to use in past clinical trials and results in no treatment related side effects. PharmaCyte’s therapy for Type 1 diabetes and insulin-dependent Type 2 diabetes involves encapsulating a human cell line that has been genetically engineered to produce, store and release insulin in response to the levels of blood sugar in the human body. The encapsulation will be done using the Cell-in-a-Box® technology. Once the encapsulated cells are implanted in a diabetic patient, they will function as a “bio-artificial pancreas” for purposes of insulin production.
Safe Harbor
This press release contains forward-looking statements, which are generally statements that are not historical facts. Forward-looking statements can be identified by the words "expects," "anticipates," "believes," "intends," "estimates," "plans," "will," "outlook" and similar expressions. Forward-looking statements are based on management's current plans, estimates, assumptions and projections, and speak only as of the date they are made. We undertake no obligation to update any forward-looking statement because of new information or future events, except as otherwise required by law. Forward-looking statements involve inherent risks and uncertainties, most of which are difficult to predict and are generally beyond our control. Actual results or outcomes may differ materially from those implied by the forward-looking statements due to the impact of numerous risk factors, many of which are discussed in more detail in our Annual Report on Form 10-K and our other reports filed with the Securities and Exchange Commission.
More information about PharmaCyte Biotech can be found at www.PharmaCyte.com. Information may also be obtained by contacting PharmaCyte’s Investor Relations Department.
View source version on businesswire.com: http://www.businesswire.com/news/home/20180228005518/en/
Investor Relations:
PharmaCyte Biotech, Inc.
Dr. Gerald W. Crabtree
Investor Relations Department
917.595.2856
Info@PharmaCyte.com
Source: PharmaCyte Biotech, Inc.
Released February 28, 2018
$PMCB PharmaCyte Announces Successful Completion of 6-Month Stability Study on Encapsulated Cells for Clinical Trial PMCB
https://finance.yahoo.com/news/pharmacyte-announces-successful-completion-6-140000667.html
Go $PMCB
$RAD Zacks Industry Outlook Highlights: Amazon, Wal-Mart, Target and Rite Aid RAD
https://www.nasdaq.com/article/zacks-industry-outlook-highlights-amazon-wal-mart-target-and-rite-aid-cm930047
$RAD Starbucks Chief to Help Fuse Albertsons, Rite Aid RAD
https://www.wsj.com/articles/former-starbucks-chief-to-help-fuse-albertsons-rite-aid-1520226061Former
$RAD State of Wisconsin Investment Board Has $7.08 Million Stake in Rite Aid Co. (RAD)
https://weekherald.com/2018/03/01/state-of-wisconsin-investment-board-has-7-08-million-stake-in-rite-aid-co-rad.html
State of Wisconsin Investment Board raised its holdings in shares of Rite Aid Co. (NYSE:RAD) by 93.3% during the 4th quarter, according to its most recent filing with the Securities and Exchange Commission (SEC). The fund owned 3,593,475 shares of the company’s stock after acquiring an additional 1,734,500 shares during the quarter. State of Wisconsin Investment Board’s holdings in Rite Aid were worth $7,079,000 as of its most recent filing with the Securities and Exchange Commission (SEC).
Other institutional investors have also modified their holdings of the company. Parametrica Management Ltd purchased a new position in shares of Rite Aid during the 4th quarter worth $182,000. State of Alaska Department of Revenue purchased a new position in shares of Rite Aid during the 4th quarter valued at $195,000. Investment Centers of America Inc. lifted its stake in shares of Rite Aid by 53.3% during the 3rd quarter. Investment Centers of America Inc. now owns 184,991 shares of the company’s stock valued at $365,000 after buying an additional 64,300 shares in the last quarter. Russell Investments Group Ltd. lifted its stake in shares of Rite Aid by 75.3% during the 3rd quarter. Russell Investments Group Ltd. now owns 219,886 shares of the company’s stock valued at $431,000 after buying an additional 94,436 shares in the last quarter. Finally, Ladenburg Thalmann Financial Services Inc. lifted its stake in shares of Rite Aid by 35.5% during the 3rd quarter. Ladenburg Thalmann Financial Services Inc. now owns 225,131 shares of the company’s stock valued at $441,000 after buying an additional 59,000 shares in the last quarter. Institutional investors and hedge funds own 51.68% of the company’s stock.
$RAD Aid Co. (RAD) Receives $2.75 Average Price Target from Brokerages RAD
https://stocknewstimes.com/2018/03/02/rite-aid-co-rad-receives-2-75-average-price-target-from-brokerages.htmlRite
$RAD Merger with Albertsons Provides “Transformative Opportunity” For Rite Aid RAD
https://www.loyalty360.org/content-gallery/daily-news/merger-with-albertsons-provides-transformative-op
$RAD Rite Aid Commences Offer to Purchase Certain of its Outstanding Series of Notes RAD
https://finance.yahoo.com/news/rite-aid-commences-offer-purchase-211500044.html
$RAD Albertsons-Rite Aid merger announced RAD
By Dee Camp As of Tuesday, February 27, 2018 Sign in to favorite this Discuss
Comment, Blog about
Share this
Email, Facebook, Twitter
BOISE, Idaho – Albertsons Companies and Rite Aid Corp. announced plans last week for a merger, but there’s no word yet on whether the Omak Rite Aid store will be part of the deal.
Boise-based Albertsons is one of the nation's largest grocery retailers and Rite Aid is one of the nation's leading drugstore chains.
“We don’t yet have a store list,” said Chris Wilcox, vice president of communications, public affairs and education for Albertsons. “It’s still fairly early on, so we have a lot of work to do yet.
“We will likely update the investor (Web) page with new info as it becomes available, but I don’t know that we would publish a complete store list. Store lists aren’t something that we typically share even under standard operating conditions.”
Rite Aid officials did not respond to a Chronicle request for comment.
Under the terms of the agreement, in exchange for every 10 shares of Rite Aid common stock, Rite Aid shareholders can receive either one share of Albertsons common stock plus approximately $1.83 in cash 1.079 shares of Albertsons stock.
Current Rite Aid Chairman and Chief Executive Officer John Standley will become CEO of the combined company, with current Albertsons Chairman and CEO Bob Miller serving as chairman. The combined company is expected to be comprised of leadership from both companies and will be dual headquartered in Boise, Idaho, and Camp Hill, Penn.
The integrated company will operate approximately 4,900 locations, 4,350 pharmacy counters and 320 clinics across 38 states and Washington, D.C.
The transaction has been approved by boards of both companies. The merger is expected to close early in the second half of calendar year 2018, subject to the approval of Rite Aid's shareholders, regulatory approvals and other customary closing conditions, said Albertsons.
In January, Rite Aid said it transferred 625 of its stores to Walgreens Boots Alliance and that Walgreen planned to buy 1,932 stores and three distribution centers from Rite Aid, according to Forbes.
$RAD Today’s Hot Stock Under Review – Rite Aid Corporation (NYSE: RAD)
http://alphabetastock.com/2018/02/26/todays-hot-stock-under-review-rite-aid-corporation-nyse-rad/
$RAD Kimco Sees Further Opportunity from Albertsons Companies Investment RAD
"Today's announced agreement between Albertsons and Rite Aid will result in a stronger, more diversified company that combines health, wellness and convenience to deliver an enhanced customer experience, demonstrating what we believe is the future of retail," stated Conor Flynn, Kimco's Chief Executive Officer. "Additionally, this merger underscores how we continue to benefit from our highly successful investment in Albertsons. Upon completion, we will have a clear indication of the resulting market value of our stake." Flynn continued, "Beyond the strong returns we have already generated, we will have greater flexibility and liquidity with our investment via the public markets."
https://www.nasdaq.com/press-release/kimco-sees-further-opportunity-from-albertsons-companies-investment-20180220-00465
$RAD Albertsons Sees Room In Supermarkets To Expand Rite Aid Clinics RAD
https://www.forbes.com/sites/brucejapsen/2018/02/21/albertsons-sees-room-in-supermarkets-to-expand-rite-aid-clinics/#76736ab963d2
$RAD Rite Aid's PBM Becomes More Attractive Under Albertsons RAD
https://www.forbes.com/sites/brucejapsen/2018/02/22/rite-aids-pbm-becomes-more-attractive-under-albertsons/#acfc45e7b060
$RAD Institutional Ownership 47.10%
Total Shares Outstanding (millions) 1,068
Total Value of Holdings (millions) $1,106
https://www.nasdaq.com/symbol/rad/institutional-holdings
$RAD Analysts think these stock’s can be a game changer: Rite Aid Corporation (RAD), Akers Biosciences, Inc. (AKER)
https://stocknewsjournal.com/2018/02/07/analysts-think-these-stocks-can-be-a-game-changer-rite-aid-corporation-rad-akers-biosciences-inc-aker/
$RAD Grocery retailer Albertsons to buy drugstore chain Rite Aid RAD
http://www.wpxi.com/news/top-stories/grocery-retailer-albertsons-to-buy-drugstore-chain-rite-aid-1/703568437
$RAD Albertsons, Rite Aid deal creates U.S. supermarket and health care giant RAD
https://www.usatoday.com/story/money/2018/02/20/albertsons-and-rite-aid-plan-merger-creates-u-s-supermarket-and-healthcare-giant/353958002/
$RAD Is Rite Aid Corporation Stock Being Bought Out at $6 or $2.50? RAD
https://finance.yahoo.com/news/rite-aid-corporation-stock-being-170823321.html
$RAD Safeway parent Albertsons buying what's left of Rite Aid RAD
https://finance.yahoo.com/m/13f75c08-ffcf-3ce0-b854-050c076606ae/safeway-parent-albertsons.html
$RAD Albertsons Scoops Up Remainder of Rite Aid in Face of Online Threat RAD
https://finance.yahoo.com/m/bbe372b2-8805-3c10-b5f2-7c0b03b03e02/%5B%24%24%5D-albertsons-scoops-up.html
$RAD Here’s why Albertsons is buying Rite Aid RAD
https://finance.yahoo.com/video/why-albertsons-buying-rite-aid-161512718.html
$DFFN PHASE 3 GBM TRIAL UNDERWAY; PLANNING FOR PHASE 2 TRIAL IN STROKE DFFN
http://scr.zacks.com/News/Press-Releases/Press-Release-Details/2018/DFFN-Phase-3-GBM-Trial-Underway-Planning-for-Phase-2-Trial-in-Stroke-article/default.aspx
$DFFN Biotech, Breaking News, InvestingDiffusion Pharmaceuticals (DFFN) Stock: Gaining Big On Patent News DFFN
https://cnafinance.com/diffusion-pharmaceuticals-dffn-stock-gaining-big-on-patent-news/17904
Go $DFFN
$DFFN Diffusion Pharmaceuticals to Present at the 2018 BIO CEO & Investor Conference DFFN
https://finance.yahoo.com/news/diffusion-pharmaceuticals-present-2018-bio-210000601.html
Go $DFFN
$DFFN Diffusion Pharmaceuticals Announces Allowances of Two U.S. Patent Applications DFFN
https://finance.yahoo.com/news/diffusion-pharmaceuticals-announces-allowances-two-131500164.html
CHARLOTTESVILLE, Va., Feb. 01, 2018 (GLOBE NEWSWIRE) -- Diffusion Pharmaceuticals Inc. (DFFN) (“Diffusion” or “the Company”), a clinical-stage biotechnology company focused on extending the life expectancy of cancer patients, today announced receipt of two patent application allowances relating to its lead compound trans sodium crocetinate (“TSC”) in the U.S. The allowances include claims for both method of use and composition of matter.
U.S. patent application number 14/993,047 includes claims relating to treating a number of cancers including brain cancers such as glioblastoma, and cancer of the pancreas, using bipolar trans carotenoids, including TSC, along with chemotherapy and radiation therapy.
U.S. patent application number 14/642,703, includes claims relating to novel compositions of bipolar trans carotenoids, including TSC, for oral delivery.
“We have worked hard to ensure our discoveries are protected and are grateful to receive these patent application allowances. We look forward to these patents being issued in the coming months,” said David Kalergis, Chief Executive Officer of Diffusion Pharmaceuticals. “We believe strong intellectual property protections are vital to Diffusion’s ability to compete in the marketplace and to attract potential strategic partners. As we progress our pivotal Phase 3 study in inoperable glioblastoma patients who are administered TSC along with their standard therapies, we feel that it is imperative that our proprietary position be protected to add increased value to the Company.”
About Diffusion Pharmaceuticals Inc.
Diffusion Pharmaceuticals Inc. is a clinical-stage biotechnology company focused on improving patient outcomes in unmet medical needs using its novel small molecule trans sodium crocetinate (TSC). Diffusion is developing TSC for use in conditions where hypoxia (oxygen deprivation) is known to diminish the effectiveness of standard of care (SOC) treatments. In oncology, TSC targets the cancer's hypoxic micro-environment, re-oxygenating treatment-resistant tissue and making the cancer cells more vulnerable to the therapeutic effects of SOC treatments without the apparent occurrence of any serious side effects. In non-oncology indications, therapeutic benefit would be achieved directly through re-oxygenation of the tissue threatened with cell death from hypoxia.
A Phase 3 randomized, controlled registration trial with TSC and SOC chemotherapy and radiation, compared with SOC alone in 236 patients newly diagnosed with inoperable glioblastoma multiforme (GBM), a type of brain cancer, is underway. A Phase 2 clinical program was completed in the second quarter of 2015 and evaluated 59 patients with newly diagnosed GBM. This open-label, historically controlled study demonstrated a favorable safety and efficacy profile for TSC combined with SOC, including a 37% improvement in overall survival compared with the control group at two years. A particularly strong efficacy signal was seen in the subset of inoperable patients where survival of TSC-treated patients at two years was nearly four-fold higher compared with the controls. Due to its novel mechanism of action, TSC has safely re-oxygenated a range of tumor types in preclinical and clinical studies. Diffusion believes the therapeutic potential of TSC is not limited to specific tumors, thereby making it potentially useful to improve SOC treatments of other life-threatening cancers. Additional studies under consideration include Phase 2 trials in pancreatic cancer and brain metastases, with study initiation subject to receipt of additional funding or collaborative partnering. The Company also believes that TSC has potential application in other indications involving hypoxia including stroke, where the Company recently announced its PHAST-TSC study which will be conducted in co-operation with the University of California Los Angeles (UCLA) and the University of Virginia (UVA) to test TSC in stroke patients in an in-ambulance clinical trial setting.
Forward-Looking Statements
To the extent any statements made in this news release deal with information that is not historical, these are forward-looking statements under the Private Securities Litigation Reform Act of 1995. Such statements include, but are not limited to, statements about the company's plans, objectives, expectations and intentions with respect to future operations and products, the potential of the company's technology and product candidates, the anticipated timing of future clinical trials and protocol review, and other statements that are not historical in nature, particularly those that utilize terminology such as "would," "will," "plans," "possibility," "potential," "future," "expects," "anticipates," "believes," "intends," "continue," "expects," other words of similar meaning, derivations of such words and the use of future dates. Forward-looking statements by their nature address matters that are, to different degrees, uncertain. Uncertainties and risks may cause the company's actual results to be materially different than those expressed in or implied by such forward-looking statements. Particular uncertainties and risks include: general business and economic conditions; the company's need for and ability to obtain additional financing; and the difficulty of developing pharmaceutical products, obtaining regulatory and other approvals and achieving market acceptance, and the various risk factors (many of which are beyond Diffusion’s control) as described under the heading “Risk Factors” in Diffusion’s filings with the United States Securities and Exchange Commission. All forward-looking statements in this news release speak only as of the date of this news release and are based on management's current beliefs and expectations. Diffusion undertakes no obligation to update or revise any forward-looking statement, whether as a result of new information, future events or otherwise.
$DFFN Breaking News DFFN

“We are pleased to be dosing patients so soon following the opening of the INTACT trial just a few weeks ago,” said David Kalergis, Chief Executive Officer of Diffusion Pharmaceuticals. “We believe that this Phase 3 study will offer new hope for inoperable GBM patients who are administered TSC along with their standard therapies.”
http://investors.diffusionpharma.com/file/Index?KeyFile=391934667
$DFFN TECHNOLOGY OVERVIEW DFFN
http://www.diffusionpharma.com/technology/technology-overview/
Oxygen deprivation at the cellular level (“hypoxia”) is the result of rapid tumor growth, causing the tumor to outgrow its blood supply. Cancerous tumor cells actually thrive on hypoxia, as the resultant changes in the tumor microenvironment confer “treatment resistance” to radiation and chemotherapy treatments within the hypoxic cancerous cell.
Mechanism of Action: TSC’s unique re-oxygenation capabilities derive from its novel mechanism of action, promoting enhanced diffusion of oxygen through the blood plasma and into the hypoxic tumor microenvironment.

Oxygen levels of normal tissue remain unaffected, thereby avoiding the introduction of additional harmful side effects. Disruption of the treatment-resistance syndrome by re-oxygenation promotes enhanced cancer killing power from standard radiation and chemotherapy treatments, thereby safely extending survival.

$DFFN Diffusion Pharmaceuticals Announces First Patient Dosed in Phase 3 Trial with TSC in Inoperable Glioblastoma Multiforme DFFN
https://www.marketwatch.com/story/diffusion-pharmaceuticals-announces-first-patient-dosed-in-phase-3-trial-with-tsc-in-inoperable-glioblastoma-multiforme-2018-01-29
CHARLOTTESVILLE, Va., Jan 29, 2018 (GLOBE NEWSWIRE via COMTEX) --
Diffusion Pharmaceuticals Inc. DFFN, +6.53% ("Diffusion" or "the Company"), a clinical-stage biotechnology company focused on extending the life expectancy of cancer patients, today announced that the first patient has been dosed in the Company's Phase 3 clinical trial with trans sodium crocetinate ("TSC") in patients with newly-diagnosed inoperable glioblastoma multiforme ("GBM"), a type of brain cancer. The INTACT (INvestigating Tsc Against Cancerous Tumors) trial will compare standard of care ("SOC") radiation therapy ("RT") and chemotherapy plus TSC against SOC alone.
"We are pleased to be dosing patients so soon following the opening of the INTACT trial just a few weeks ago," said David Kalergis, Chief Executive Officer of Diffusion Pharmaceuticals. "We believe that this Phase 3 study will offer new hope for inoperable GBM patients who are administered TSC along with their standard therapies."
The INTACT Phase 3 study follows a previous Phase 2 GBM study in which the inoperable patient subgroup showed a nearly four-fold increase in survival compared with historical controls when TSC was added to their treatment regimen (40.0% alive at two years vs. 10.4%). TSC's unique mechanism of action affects the tumor micro-environment, making treatment-resistant cancer cells more susceptible to the tumor-killing power of conventional RT and chemotherapy (temozolomide) by re-oxygenating the hypoxic portion of the tumor. The Company believes that a largely intact GBM tumor vasculature with limited surgical resection is conducive to TSC's tumor re-oxygenation properties, and that this contributed to the survival increase in the Phase 2 GBM inoperable patient subgroup.
The trial will screen 300 patients and enroll 264 with the expectation that results from 236 patients will be available for analysis. Enrolled patients will be randomized in a 1:1 ratio into treatment and control groups. Patients in the treatment group will receive SOC temozolomide and RT plus an intravenous bolus of TSC administered shortly before their SOC treatments. Patients in the control group will receive SOC alone. The study will compare overall survival at two years between patients in the two groups. Up to 100 clinical sites in the U.S. and Europe are expected to participate.
About the GBM Phase 3 INTACT Trial
The INTACT clinical trial is an open-label, randomized, controlled, Phase 3 safety and efficacy registration trial. Subjects will be randomized at baseline to SOC for first-line treatment of GBM plus TSC, or to SOC alone. The SOC for GBM is temozolomide plus RT for 6 weeks followed by 28 days of rest, followed by 6 cycles of post-radiation temozolomide treatment.
For patients randomized to the TSC group, TSC will be administered during both the RT and post-radiation temozolomide treatment periods.
During the RT treatment period subjects will receive:
Focal RT delivered as 60Gy/30 fractions scheduled at 2Gy/day for 5 days each week (Monday through Friday) for 6 weeks.
Temozolomide 75 mg/m [2 ] orally once daily (usually administered the night preceding each RT session) starting the evening before the first RT session over a period of 42 calendar days with a maximum of 49 days.
TSC 0.25 mg/kg IV for 3 days each week (e.g., Monday, Wednesday, Friday, or other schedule that supplies a minimum 3 TSC doses per week) administered between 45 to 60 minutes prior to each RT session.
During the 28-day rest period all subjects will receive no treatment.
During the post-radiation 6-cycle temozolomide treatment period:
All subjects will receive 28-day oral temozolomide (150 mg/m [2] first cycle and 200 mg/m [2] all subsequent cycles as tolerated) administered on Day 1-5 (Monday through Friday) of each 28-day cycle.
Controls will receive oral temozolomide at night at home per the SOC and are not required to attend clinic visits during this period.
Subjects randomized to TSC will receive TSC 1.5 mg/kg (or another dose if recommended by the Data Safety Monitoring Board ("DSMB") 1.5 to 2 hours before their temozolomide dose during the daytime for 3 days during the first week of each 28-day cycle (Days 1, 3, and 5; e.g., Monday, Wednesday, Friday or other schedule that supplies at minimum 3 TSC doses per week). The Tuesday, Thursday doses will be given at night at home. Long-acting antiemetics may be administered prior to daytime temozolomide dosing on Days 1, 3, 5.
The safety, tolerability and pharmacokinetics ("PK") of TSC at higher doses than 0.25 mg/kg with temozolomide will be assessed during adjuvant therapy. TSC at doses between 0.25 mg/kg and up to 1.5 mg/kg in combination with concomitant temozolomide will be assigned (not randomized) in the first 8 subjects enrolled in the INTACT trial. These patients will undergo RT plus temozolomide plus TSC treatment (0.25 mg/kg) for 6 weekly cycles followed by 4 weeks of rest in standard fashion. At the Week 10 clinic visit the same 8 subjects will be assigned to treatment, with 2 subjects each assigned to TSC at doses of 0.25, 0.50, 1.0, and 1.5 mg/kg. These subjects will be studied in parallel for 2 28-day cycles with inclusion of appropriate blood sampling collection for TSC and temozolomide PK. The DSMB will examine the resultant safety data after 2 cycles (Weeks 11 through 18 of post-radiation temozolomide treatment period; Days 1 to 56). The DSMB may recommend continued use of the 1.5 mg/kg TSC dose for the post-radiation temozolomide treatment period, or may prescribe another dose based on their observations. Subjects then entering into the INTACT trial will be randomized at baseline between TSC plus SOC, or SOC alone.
Further details about the trial protocol are available at www.clinicaltrials.gov.
The baseline assessment for determining progression-free survival overall response rate and to rule out pseudo-progression will be at 10 weeks via MRI using the modified Response Assessment in Neuro-Oncology scale. The hazard ratio for the trial will be 0.67, which corresponds to 22% two-year survival in the TSC arm, the lower limit of the 95% confidence interval for the biopsy-only subjects in Diffusion's Phase 2 trial, and 10% survival in the SOC arm. The estimated median survival is therefore 10 months for the SOC arm vs. 14.9 months for the TSC plus SOC arm. In order to achieve 80% power, the trial requires 118 subjects in each arm.
The study will achieve the designed 80% statistical power at 198 events, where an event is defined as death. The first analysis will occur at the earlier of two years follow-up for all subjects or 198 events. If the first analysis is at 198 events, the analysis will be a standard 2-sided stratified log-rank test at the ?'?=0.05 significance level. If the first analysis is at two years, the Company will perform the analysis using the O'Brien-Fleming Method.
About Treatment-Resistant Cancers and TSC
Oxygen deprivation at the cellular level (hypoxia) is the result of rapid tumor growth, causing the tumor to outgrow its blood supply. Cancerous tumor cells thrive with hypoxia and the resultant changes in the tumor microenvironment cause the tumor to become resistant to RT and chemotherapy. Using a novel, proprietary mechanism of action, Diffusion's lead drug TSC appears to counteract tumor hypoxia - and therefore treatment resistance - by safely re-oxygenating tumor tissue, thus enhancing tumor kill and potentially prolonging patient life expectancy. Oxygen levels of normal tissue appear to remain unaffected upon administration of TSC.
About Diffusion Pharmaceuticals Inc.
Diffusion Pharmaceuticals Inc. is a clinical-stage biotechnology company focused on improving patient outcomes in unmet medical needs using its novel small molecule trans sodium crocetinate (TSC). Diffusion is developing TSC for use in conditions where hypoxia (oxygen deprivation) is known to diminish the effectiveness of SOC treatments. In oncology, TSC targets the cancer's hypoxic micro-environment, re-oxygenating treatment-resistant tissue and making the cancer cells more vulnerable to the therapeutic effects of SOC treatments without the apparent occurrence of any serious side effects. In non-oncology indications, therapeutic benefit would be achieved directly through re-oxygenation of the tissue threatened with cell death from hypoxia.
A Phase 3 randomized, controlled registration trial with TSC and SOC chemotherapy and radiation, compared with SOC alone in 236 patients newly diagnosed with inoperable glioblastoma multiforme (GBM), a type of brain cancer, is underway. A Phase 2 clinical program was completed in the second quarter of 2015 and evaluated 59 patients with newly diagnosed GBM. This open-label, historically controlled study demonstrated a favorable safety and efficacy profile for TSC combined with SOC, including a 37% improvement in overall survival compared with the control group at two years. A particularly strong efficacy signal was seen in the subset of inoperable patients where survival of TSC-treated patients at two years was nearly four-fold higher compared with the controls. Due to its novel mechanism of action, TSC has safely re-oxygenated a range of tumor types in preclinical and clinical studies. Diffusion believes the therapeutic potential of TSC is not limited to specific tumors, thereby making it potentially useful to improve SOC treatments of other life-threatening cancers. Additional studies under consideration include Phase 2 trials in pancreatic cancer and brain metastases, with study initiation subject to receipt of additional funding or collaborative partnering. The Company also believes that TSC has potential application in other indications involving hypoxiaincluding stroke, where the Company recently announced its PHAST-TSC study which will be conducted in co-operation with the University of California Los Angeles (UCLA) and the University of Virginia (UVA) to test TSC in stroke patients in an in-ambulance clinical trial setting.
Forward-Looking Statements
To the extent any statements made in this news release deal with information that is not historical, these are forward-looking statements under the Private Securities Litigation Reform Act of 1995. Such statements include, but are not limited to, statements about the company's plans, objectives, expectations and intentions with respect to future operations and products, the potential of the company's technology and product candidates, the anticipated timing of future clinical trials and protocol review, and other statements that are not historical in nature, particularly those that utilize terminology such as "would," "will," "plans," "possibility," "potential," "future," "expects," "anticipates," "believes," "intends," "continue," "expects," other words of similar meaning, derivations of such words and the use of future dates. Forward-looking statements by their nature address matters that are, to different degrees, uncertain. Uncertainties and risks may cause the company's actual results to be materially different than those expressed in or implied by such forward-looking statements. Particular uncertainties and risks include: general business and economic conditions; the company's need for and ability to obtain additional financing; and the difficulty of developing pharmaceutical products, obtaining regulatory and other approvals and achieving market acceptance, and the various risk factors (many of which are beyond Diffusion's control) as described under the heading "Risk Factors" in Diffusion's filings with the United States Securities and Exchange Commission. All forward-looking statements in this news release speak only as of the date of this news release and are based on management's current beliefs and expectations. Diffusion undertakes no obligation to update or revise any forward-looking statement, whether as a result of new information, future events or otherwise.
Contacts:
David Kalergis, CEO
Diffusion Pharmaceuticals Inc.
(434) 220-0718
dkalergis@diffusionpharma.com
or
LHA Investor Relations
Kim Sutton Golodetz
(212) 838-3777
kgolodetz@lhai.com
Copyright (C) 2018 GlobeNewswire, Inc. All rights reserved.
$AZFL Amazonas Florestal Ltd. Announces That Its Subsidiary Amazon Hemp Ltd. Has Completed Delivery of Its First Hemp Product Covering Initial Advanced Sales; Product is Now Officially On Sale to the Public AZFL
https://finance.yahoo.com/news/amazonas-florestal-ltd-announces-subsidiary-123000975.html




$AZFL Hemp full plant extract AZFL
https://azhempusa.com/en/details?lang=en&new=03
Amazonas Florestal Ltd. Announced Today That The Company's Subsidiary Amazon Hemp will be selling their new products on Bonsai MedSpa located in Miami. Coming soon in our website and in the best online sites!
Amazon Hemp Team

$AVEW AVEW HOLDINGS Announces Status of Central Texas Plant Sites for Precious Metal Recovery Processing Plant AVEW
https://www.otcmarkets.com/stock/AVEW/news/AVEW-HOLDINGS-Announces-Status-of-Central-Texas-Plant-Sites-for-Precious-Metal-Recovery-Processing-Plant?id=159533
$ZMRK ~ Welcome to Zalemark Holding Co.; Inc. (ZMRK) DD






$ZMRK Security Details
Share Structure
Market Value1 $920,112
a/o Mar 15, 2016
Authorized Shares 150,000,000 a/o Sep 30, 2015
Outstanding Shares 80,009,742 a/o Sep 30, 2015
-Restricted Not Available
-Unrestricted Not Available
Held at DTC Not Available
Float 27,507,181
a/o Sep 30, 2015
Par Value 0.0001
$MCGI Security Details MCGI
Share Structure
Market Value1 $228,631 a/o Sep 22, 2017
Authorized Shares 4,000,000,000 a/o Feb 24, 2016
Outstanding Shares 571,577,017 a/o Jul 31, 2016
-Restricted Not Available
-Unrestricted Not Available
Held at DTC Not Available
Float 347,979,442 a/o Jun 30, 2016
Par Value 0.001
Transfer Agent(s)
Island Stock Transfer
Shareholders
Shareholders of Record 80 a/o Jun 27, 2016
Security Notes
Capital Change=shs increased by 10 for 1 split, payable upon surrender.. Pay date=01/07/2010.
Short Selling Data
Short Interest 8,400 (100%)
May 31, 2017
Significant Failures to Deliver No
$MCGI Looking to be a Nurse Practitioner in L.A.? Check out Cedars-Sinai for jobs, learn about company culture, or connect with a recruiter for questions about employment. MCGI
Looking to be a Nurse Practitioner in L.A.? Check out Cedars-Sinai for jobs, learn about company culture, or connect with a recruiter for questions about employment. https://t.co/eBznIc6eGD pic.twitter.com/nMPXTybYIT
— Nurses Lounge (@TheNursesLounge) November 14, 2017
|
Followers
|
157
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
20724
|
|
Created
|
04/07/17
|
Type
|
Free
|
| Moderator $Pistol Pete$ | |||
| Assistants mick | |||
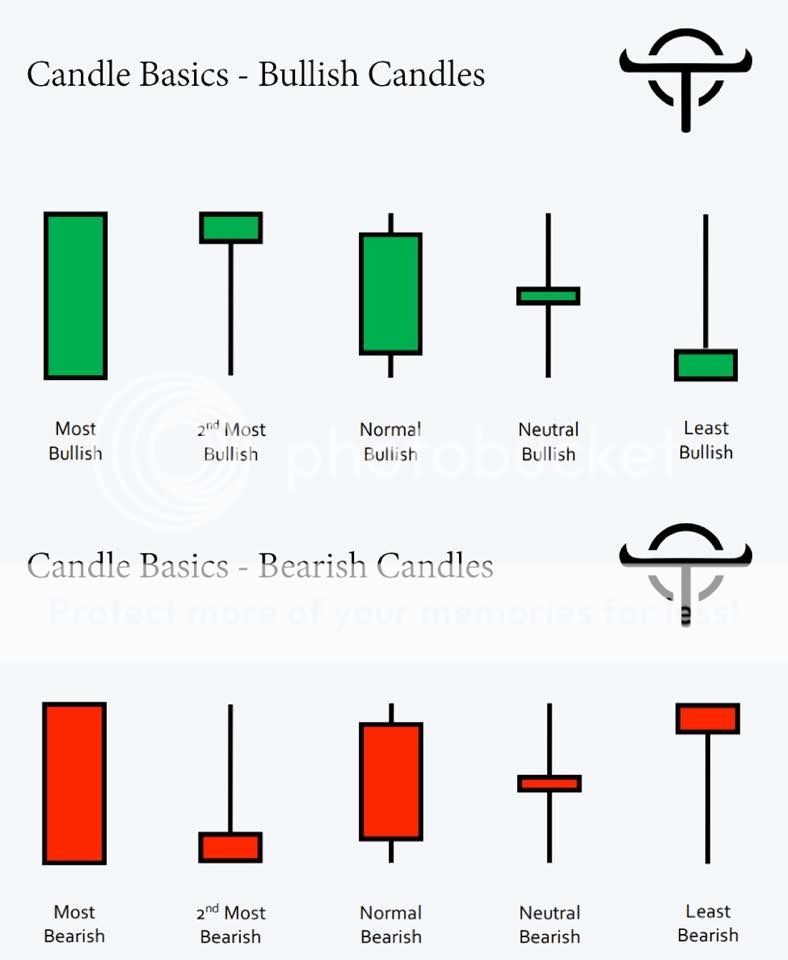
PER MGMT DAVE -
| IH Geek [Dave] DISCLAIMER |
01-31-2021
DISCLAIMER: ONLY FOR MICK
https://investorshub.advfn.com/boards/profilea.aspx?user=1012
*The Board Monitor and Board Assistants herewithin , are not licensed brokers and assume NO responsibility for actions,
investments,decisions, or messages posted on this forum.
CONTENT ON THIS FORUM SHOULD NOT BE CONSIDERED ADVISORY NOR SOLICITATION
AUTHORS MAY HAVE BUYS OR SELLS WITH THE COMPANIES MENTIONED IN TRADING POSTERS SHOULD DUE DILIGENT BUYING OR SELLING.
ALL POSTING SHOULD BE CONSIDERED FOR INFORMATION ONLY. WE DO NOT RECOMMEND ANYONE BUY OR SELL ANY SECURITIES POSTED HEREWITHIN.
ANY trade entered into risks the possibility of losing the funds invested.
• There are no guarantees when buying or selling any security.Any
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
