Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Insomnia Treatment Study using MediPharm Labs CBD50 Product to Proceed to Phase 2 with Health Canada Consent
August 31, 2021
• McMaster University researchers using MediPharm Labs’ CBD50 formula and a CBD:THC 10:2 formulation, which has the same cannabinoid ratio as MediPharm Labs’ CBD25:, have received a No Objection Letter from Health Canada to begin recruiting human subjects for a placebo-controlled trial using cannabis- oil for treatment of insomnia in major depressive disorder.
• Among those with depression, 75% have difficulty falling or staying asleep - according to John Hopkins Medicine. MediPharm Labs and the McMaster Research Team hope CBD50 and/or CBD10:2 provides these patients with relief from this condition.
BARRIE, Ontario, Aug. 31, 2021 (GLOBE NEWSWIRE) -- MediPharm Labs Corp. (TSX: LABS) (OTCQX: MEDIF) (FSE: MLZ) (“MediPharm Labs” or the “Company”) a pharmaceutical company specialized in cannabis, is pleased to announce that the Company’s research partner McMaster University (the “Researcher”) has received a no objection letter from Health Canada to proceed with research with MediPharm Labs CBD50 and CBD10:2. This is a key milestone in moving forward with the Company’s strategy to be the go-to cannabis partner for pharmaceuticals containing cannabis.
This phase 2 trial is a pilot, double-blind, randomized, placebo-controlled trial that will be evaluating the efficacy and safety of a cannabis-oil for the treatment of insomnia in major depressive disorder. The study is being conducted by a clinical research group led by Dr. Benicio Frey, Professor, McMaster University and Psychiatrist, St. Joseph’s Healthcare Hamilton. As North America has made progress in diagnosing and eliminating the stigma around mental health, more patients are looking for therapeutic treatment options.
The Researchers believe that cannabidiol (“CBD”) with or without THC could be effective in the treatment of sleep disorders related to depression. In order to prove this efficacy, a high quality, dosable and standardize formulation is needed for clinical trial material. MediPharm Labs will use its Cannabis Drug Licence and Drug Establishment Licence to provide access to clinical trial material that meets these pharmaceutical quality standards and Good Manufacturing Practices (“GMP”), as well as provide investigative protocol and regulatory approval support.
“As a pharmaceutical company specialized in cannabis, MediPharm Labs is encouraged by the ongoing research and development of drugs containing cannabis. This study will create opportunities to show efficacy in our already commercialized cannabis oil while also making progress on our long-term goal to manufacture pharmaceutical drugs containing cannabinoids as an active ingredient,” said Keith Strachan, President and Interim CEO, MediPharm Labs.
“Many individuals around the world are using cannabinoids to treat their sleep problems without robust clinical evidence from placebo-controlled trials. This study is, therefore, a major step towards evidence-based research investigating the efficacy and safety of CBD and CBD/THC in the treatment of insomnia in those who suffer from depression,” said Dr. Benicio Frey.
The is the second approval of its kind for a clinical trial with McMaster. A trial lead by a separate clinician group received a letter of no objection in June 2021 to proceed with using MediPharm Labs CBD50 for a randomized trial studying the effect of cannabidiol vs. placebo on persistent post-surgical pain following total knee arthroplasty.
The Company looks forward to progressing both these active trials with the respective researchers while using its GMP Drug Establishment License and Health Canada Cannabis Drug License to supply API and finished goods to others development pharmaceutical drugs with marketing authorization.
About MediPharm Labs
Founded in 2015, MediPharm Labs is a pharmaceutical company that specializes in the development and manufacture of purified, pharmaceutical-quality cannabis concentrates, active pharmaceutical ingredients and advanced derivative products utilizing a Good Manufacturing Practices certified facility with ISO standard-built clean rooms. MediPharm Labs has invested in an expert, research driven team, state-of-the-art technology, downstream purification methodologies and purpose-built facilities with four primary extraction lines for delivery of pure, trusted and precision-dosed cannabis products for its customers. Through its wholesale and white label platforms, MediPharm Labs formulates, develops (including through sensory testing), processes, packages and distributes cannabis extracts and advanced cannabinoid-based products to domestic and international markets. As a global leader, MediPharm Labs has completed commercial exports to Australia and completed commercialization of its Australian extraction facility which generated its first revenues in H1 2020. MediPharm Labs Australia was established in 2017.
Cautionary Note Regarding Forward-Looking Information:
This news release contains “forward-looking information” and “forward-looking statements” (collectively, “forward-looking statements”) within the meaning of the applicable Canadian securities legislation. All statements, other than statements of historical fact, are forward-looking statements and are based on expectations, estimates and projections as at the date of this news release. Any statement that involves discussions with respect to predictions, expectations, beliefs, plans, projections, objectives, assumptions, future events or performance (often but not always using phrases such as “expects”, or “does not expect”, “is expected”, “anticipates” or “does not anticipate”, “plans”, “budget”, “scheduled”, “forecasts”, “estimates”, “believes” or “intends” or variations of such words and phrases or stating that certain actions, events or results “may” or “could”, “would”, “might” or “will” be taken to occur or be achieved) are not statements of historical fact and may be forward-looking statements. In this news release, forward-looking statements relate to, among other things, the successful performance of the clinical trials as planned; being the go-to cannabis partner for pharmaceuticals containing cannabis; the creation of opportunities to show efficacy in the Company’s already commercialized cannabis oil; progress on the Company’s long-term goal to manufacture pharmaceutical drugs containing cannabinoids as an active ingredient; and the development of pharmaceutical drugs containing cannabis. Forward-looking statements are necessarily based upon a number of estimates and assumptions that, while considered reasonable, are subject to known and unknown risks, uncertainties, and other factors which may cause the actual results and future events to differ materially from those expressed or implied by such forward-looking statements. Such factors include, but are not limited to: general business, economic, competitive, political and social uncertainties; the inability of MediPharm Labs to obtain adequate financing; the delay or failure to receive regulatory approvals; and other factors discussed in MediPharm Labs’ filings, available on the SEDAR website at www.sedar.com. There can be no assurance that such statements will prove to be accurate, as actual results and future events could differ materially from those anticipated in such statements. Accordingly, readers should not place undue reliance on the forward-looking statements and information contained in this news release. Except as required by law, MediPharm Labs assumes no obligation to update the forward-looking statements of beliefs, opinions, projections, or other factors, should they change.
For further information, please contact:
MediPharm Labs Investor Relations
Telephone: +1 416.913.7425 ext. 1525
Email: investors@medipharmlabs.com
Website: www.medipharmlabs.com
Source: MediPharm Labs Corp.
Released August 31, 2021
I own CRLBF, ALEAF, VLNCF, CBDD, OGI, MJ and GDNSF. They're all stuck in the mud....and, have been for a long time. The tide will eventually turn, though, for cannabis stocks. It always does. And, MEDIF will be part of the upswing. Disclaimer....I no longer own shares of MEDIF.
I am stubborningly stuck in the MediPharm mud
Since I am stubborningly stuck in the MediPharm mud, I'm just hoping that USA legalization sympathetically lifts Medi's PPS so I can get out.
mauiguy2 Thursday, 08/19/21 09:56:08 AM
Re: FUNMAN post# 6648
Post # 6650 of 6650
Seriously, Funman, the cannabis sector is so out of favor these days that if MEDIF suddenly announced earnings of $1/share, the PPS would probably sink.
The Australia news sounds good......BUT, see the above paragraph.
Seriously, Funman, the cannabis sector is so out of favor these days that if MEDIF suddenly announced earnings of $1/share, the PPS would probably sink.
The Australia news sounds good......BUT, see the above paragraph.
No good news shall go unpunished ... ![]()
Stay safe >> M
MediPharm Labs Australia Receives Government Grant to Assist with International Expansion
https://www.medipharmlabs.com/news/press-releases/detail/190/medipharm-labs-australia-receives-government-grant-to
August 19, 2021
* Received first instalment of a government grant for $330,000AUD as part of the Australian government’s Manufacturing Modernisation program aimed at supporting manufacturers scaling their businesses.
* Grant to be used for automation of downstream manufacturing and scaling purification abilities.
* Government grant as part of broader manufacturing sector funding demonstrates the backing of the medical cannabis industry by the Australian federal government and recognition of MediPharm Labs Australia’s role in this industry.
BARRIE, Ontario, Aug. 19, 2021 (GLOBE NEWSWIRE) -- MediPharm Labs Corp. (TSX: LABS) (OTCQX: MEDIF) (FSE: MLZ) (“MediPharm Labs” or the “Company”) a pharmaceutical company that specializes in cannabis, is pleased to announce it’s wholly owned subsidiary MediPharm Labs Australia Pty Ltd (“MediPharm Labs Australia”) has been awarded $330,000 as part of the Australian Government’s $50 million Manufacturing Modernisation Fund. This funding is part of a key initiative to support businesses transforming manufacturing and supporting job growth.
MediPharm Labs Australia has now received the first instalment of this grant.
The Manufacturing Modernisation Fund is focused on investing in small and medium sized Australian enterprises, with the intended outcomes of the program being:
* increased innovation and enhanced productivity in Australian manufacturing operations; and
* increased job opportunities and upskilling in the Australian manufacturing sector.
Medipharm Labs Australia intends to use the funds to increase the efficiency of its cannabis purification system and enhance the automation of its primary packaging manufacturing process.
The enhanced automation of these production processes will allow MediPharm Labs Australia to service its growing domestic and international client base along with the anticipated increase in demand for its products.
In Q1 2021, MediPharm Labs Australia made it’s first shipments to STADA and AdrexPharma in Germany. In Q2 2021, MediPharm Labs Australia, made shipments to a further two customers in Germany.
“Through the awarding of this grant, the Australian Government has recognised MediPharm Labs as a key player in the Australian cannabis industry and the manufacturing sector. This grant will allow MediPharm Labs to invest in innovation which will boost productivity to accelerate its expansion plans to execute on its global strategy - a great outcome for our Company’s growth trajectory,” said Warren Everitt, CEO Asia Pacific, MediPharm Labs.
The Company looks forward to further announcing executed progress on this project and how it will lead to revenue growth opportunities.
About MediPharm Labs
Founded in 2015, MediPharm is a pharmaceutical company that specializes in the development and manufacture of purified, pharmaceutical-quality cannabis concentrates, active pharmaceutical ingredients and advanced derivative products utilizing a Good Manufacturing Practices certified facility with ISO standard-built clean rooms. MediPharm has invested in an expert, research driven team, state-of-the-art technology, downstream purification methodologies and purpose-built facilities with four primary extraction lines for delivery of pure, trusted and precision-dosed cannabis products for its customers. Through its wholesale and white label platforms, MediPharm formulates, develops (including through sensory testing), processes, packages and distributes cannabis extracts and advanced cannabinoid-based products to domestic and international markets. As a global leader, MediPharm has completed commercial exports to Australia and completed commercialization of its Australian extraction facility which generated its first revenues in H1 2020. MediPharm Labs Australia was established in 2017.
Cautionary Note Regarding Forward-Looking Information:
This news release contains “forward-looking information” and “forward-looking statements” (collectively, “forward-looking statements”) within the meaning of the applicable Canadian securities legislation. All statements, other than statements of historical fact, are forward-looking statements and are based on expectations, estimates and projections as at the date of this news release. Any statement that involves discussions with respect to predictions, expectations, beliefs, plans, projections, objectives, assumptions, future events or performance (often but not always using phrases such as “expects”, or “does not expect”, “is expected”, “anticipates” or “does not anticipate”, “plans”, “budget”, “scheduled”, “forecasts”, “estimates”, “believes” or “intends” or variations of such words and phrases or stating that certain actions, events or results “may” or “could”, “would”, “might” or “will” be taken to occur or be achieved) are not statements of historical fact and may be forward-looking statements. In this news release, forward-looking statements relate to, among other things, statement regarding: increased efficiency; enhanced automation; growing domestic and international client base; anticipated increase in demand for products; boosted productivity; accelerated expansion plans; and execution on the Company’s global strategy. Forward-looking statements are necessarily based upon a number of estimates and assumptions that, while considered reasonable, are subject to known and unknown risks, uncertainties, and other factors which may cause the actual results and future events to differ materially from those expressed or implied by such forward-looking statements. Such factors include, but are not limited to: general business, economic, competitive, political and social uncertainties; the inability of MediPharm Labs to obtain adequate financing; the delay or failure to receive regulatory approvals; and other factors discussed in MediPharm Labs’ filings, available on the SEDAR website at www.sedar.com. There can be no assurance that such statements will prove to be accurate, as actual results and future events could differ materially from those anticipated in such statements. Accordingly, readers should not place undue reliance on the forward-looking statements and information contained in this news release. Except as required by law, MediPharm Labs assumes no obligation to update the forward-looking statements of beliefs, opinions, projections, or other factors, should they change.
For further information, please contact:
MediPharm Labs Investor Relations
Telephone: +1 416.913.7425 ext. 1525
Email: investors@medipharmlabs.com
Website: www.medipharmlabs.com
MediPharm Labs Receives Export Authorization to Ship Medical Cannabis to Brazil
https://www.medipharmlabs.com/news/press-releases/detail/189/medipharm-labs-receives-export-authorization-to-ship
August 18, 2021
Completed the regulatory process required to ship medical cannabis to Brazil with import permits issued from ANVISA and export permits issued from Health Canada. Initial deliveries will be under the compassionate care program.
First shipments to Brazil to be used for patient trials to collect data that may be used in future product marketing authorizations under Brazil’s medical cannabis program. A program that saw over 20,000 patients in 2020 and is growing by 1,500 patients a month.
BARRIE, Ontario, Aug. 18, 2021 (GLOBE NEWSWIRE) -- MediPharm Labs Corp. (TSX: LABS) (OTCQX: MEDIF) (FSE: MLZ) (“MediPharm Labs” or the “Company”) a pharmaceutical company that specializes in cannabis, is pleased to announce that it has received approval to ship medical cannabis oil to Brazil. This approval, issued by both ANVISA and Health Canada, allows initial shipments to patients for the Company’s partner XLR8. The shipment is a crucial milestone for the Company in using its pharmaceutical Good Manufacturing Practices (“GMP”) platform for international manufacturing and distribution.
The Company and XLR8, a Curitiba based value-added distributor serving Brazil, entered into an agreement in September 2020. The two-year agreement is to commence from the time of ANVISA Sanitary Product Authorization, which is expected later this year. The registration will be for a mix of medical oil SKUs such as balanced and high CBD formulas.
This initially approved delivery goes directly to patients under the compassionate care program, allowing XLR8 to begin its medical cannabis research. This first delivery opens a regulatory pathway for future opportunities for authorized product distribution. Brazil is one of the most significant medical cannabis market opportunities in LATAM.
“MediPharm Labs continues to execute our on plan to provide pharmaceutical-grade medical cannabis products to patients around the world. It is with great partners, like XLR8, with local country expertise that we can effectively achieve this goal,” said Keith Strachan, President and Interim CEO, MediPharm Labs.
”Based on our business pillars, clinical research development is a strategic path to provide substantial clinical evidence to support physicians in making appropriate decisions in patient care and cannabis is a real alternative. Our clinical research follows all national and international ethical standards, demonstrating our responsibility to provide all necessary resources to our patients” said Thiago Callado, CEO and Founder, XLR8 BRAZIL.
The Company looks forward to announcing further executed deliveries to Brazil in the coming months.
Earnings Call Transcript: Labs Corp. (MEDIF) CEO Keith Strachan on Q2 2021 Results - Earnings Call Transcript
Aug. 16, 2021 1:49 PM ET
MediPharm Labs Corp. (OTCQX:MEDIF) Q2 2021 Earnings Conference Call August 13, 2021 8:30 AM ET
Company Participants
Keith Strachan – President and Interim CEO
Greg Hunter – Chief Financial Officer
Chris Taves – Chairman
Conference Call Participants
Scott Fortune – ROTH Capital Partners
Aaron Grey – Alliance Global Partners
Natalia Cochran – Scotiabank
Tamy Chen – BMO Capital Markets
Operator
Good morning, ladies and gentlemen. Welcome to the MediPharm Labs Second Quarter 2021 Conference Call and Webcast. I will now hand the call over to Keith Strachan, MediPharm Labs, President, and Interim CEO.
Keith Strachan
Thanks, Operator. And good morning, everyone. With me on the call, today are Greg Hunter, our CFO, and Chris Taves, our new Chairman. Before we begin, please note the following caution respecting forward-looking statements which are made on behalf of MediPharm Labs and all of its representatives on this call.
The statements made on this call will contain forward-looking information that involves risks and uncertainties including those introduced by the COVID -19 pandemic. Actual results could differ materially from the conclusion, forecast, or projection in the forward-looking information. But certain material factors or assumptions were applied in drawing a conclusion or making a forecast or projection as reflected in the forward-looking information.
Additional information about the material factors that could cause actual results to differ materially from the conclusions, forecasts, or projections in the forward-looking information and the material factors or assumptions that were applied in drawing a conclusion or making a forecast, or a projection as reflected in the forward-looking information are contained in MediPharm Lab's filings with the Canadian and Provincial Securities Regulators, which are available on SEDAR, after the website sedar.com.
Our second quarter was one of transformational growth in establishing ourselves as a true pharmaceutical Company with expertise in Canada. Today, I will speak to the advancements made in Q2, which were achieved in the context of ongoing Canadian domestic challenges, offset by international growth. We will then discuss Q2 results, including areas where we saw growth, segments where we have the opportunity to improve, and continued progress with rightsizing our inventory.
We will then introduce our new Board Chair, Chris Taves, and I will close with final observations on executing our business strategy. To start, I would like to provide more in-depth detail on our recent pharmaceutical licensing achievement, the way it grows our business, and our plans to capitalize on its unique status in the industry.
From the outset, MediPharm 's objective was to become a leading pharmaceutical Company specializing in Canada, taking charge in an emerging multi-billion-dollar global pharmaceutical and medical Cannabis market by providing multiple products in turnkey solutions to a broader customer base across multiple jurisdictions. A key part of becoming a pharmaceutical Company is pharmaceutical licensing.
And in Canada, that means a drug establishment license. A drug establishment license is a certification issued by Health Canada that a manufacturer maintains pharmaceutical good manufacturing practices, commonly referred to as GMP. It is done through an extensive review of the Company's quality management system over several months and then an in-person or virtual inspection spanning multiple weeks.
A massive undertaking in comparison to our Cannabis processing license issued in 2018. This license is awarded by the Health Canada pharmaceutical branch and is the exact same license as the one held by Fortune 500 multinational drug companies with operations in Canada. Pharmaceutical leaders like Eli Lilly, Abdi, and Merck just to name a few.
Our drug establishment license allows MediPharm to manufacture any non-sterile drug in finished goods or active pharmaceutical ingredient, API format, leaving many options as we start to see more complex formulas in drugs where cannabinoids are the API. We are the first and only Company in North America to receive a pharmaceutical GMP certification, which includes commercial scale extraction of natural cannabinoids.
Other pharma companies working with Cannabis with GMP licenses from Health Canada or the U.S. FDA are only held by those doing final product formulation or working with synthetic cannabinoids. Based on the current therapeutic evidence in natural cannabinoids in products such as FDA -approved Epidiolex, there's a large demand for naturally derived and pharmaceutically approved cannabis API and finished goods.
So, what does this drug establishment license means for MediPharm and its shareholders? To summarize, with this unique license, we can now access more markets globally where special access for OTC policies for cannabis are in place. We can now distribute cannabis API to pharmaceutical companies around the world, including the U.S. for use in both branded and generic drugs with marketing authorizations.
We can now provide finished dose manufacturing to pharmaceutical companies seeking to outsource the productions of their Cannabis-based drugs. We can now be a service provider to other large Cannabis companies with aspirations to enter the pharmaceutical cannabis space or expand their international reach.
And we can now support new clinical trials with GMP clinical trial material to further advance research of the benefits of cannabinoids and give many Pharm future manufacturing rights. These drug establishment license business attributes create near-term opportunities while also preparing in the long-term opportunity to produce future Canopy states, clinical proven FDA registered, and approved drugs.
This is a great development for MediPharm and our shareholders and it means our outlook for growth has a specialist pharma [Indiscernable]. Now turning to our second-quarter results, beyond the great advancements in our Pharmaceutical strategy, Q2 saw growth in other areas. The most promising in being international distribution. As a testament to the execution of our international contracts.
We saw quarter-over-quarter growth of 24% in our international revenue. Now with regulatory channels open, we expect to continue to be the leading private label medical Cannabis concentrate provider in new global markets. Like our industry's unique supply agreement with STADA, one of the EU's largest generic drug companies MediPharm has many contracts and a full pipeline of future contracts for private label medical Cannabis products around the globe.
Many of these being concentrated in the EU [Indiscernable]. There is no shortage of demand for quality medical Cannabis products from established companies with no interest in doing manufacturing themselves. The difficulty comes in the regulatory execution of delivering on these contracts. For example, in Brazil, you must register your product with ANVISA, the Brazilian FDA for sanitary authorization.
This process can only be done with a lengthy product dossier to show the quality, safety, and stability of the products. This process can take up to 3 months for approval following your manufacturing of pilot batches and six months for stability testing. Narcotic registration in places like Germany and New Zealand follow similar processes, with each country being unique. This is on top of the country's specific import and export process, which in the past year and a half has been subject to longer-than-normal processing times, as global health regulators focused efforts on their COVID-19 pandemic response. In 2021, we have seen Health Canada export permits to take anywhere from 30 to 60 calendar days after the receiving country institutes an import permit.
MediPharm masters this regulatory process with our staff who come with decades of experience in the international registration of pharmaceuticals. Now, having worked with international partners for over 18 months, many of Pharm's hard works are starting to result in the steady growth of material, international revenue. We are also establishing a cadence in permits ahead of the international customer forecast so that as we go into the back half of 2021 and beyond, we can have more repeatable monthly international revenue. A great example of these advancements is in Germany.
In March, we committed our first delivery to 2 extract customers in the region, STADA, and ADREXpharma. In Q2, not only did we deliver to those customers again, but we also delivered to 2 new customers in Cantaraj (ph) and Demi can (ph). Of course, our reach goes beyond Germany, having already delivered to Peru in March and we expect to deliver to Denmark, Brazil, and New Zealand before the end of the year.
However, I do want to highlight that Germany alone is an extremely attractive market. It stands as the world's leading country for medical Cannabis with more than 320,000 Cannabis prescriptions approved in 2020. This is a more advanced medical market than any other and is said to be growing at around 30% annually according to Forbes.
Our strategy of targeted international expansion to medical and wellness customers is gaining traction and that will lead to higher sales in the back half of this year. Our strategy is clearly global in nature, but we are also committed to driving growth in Canada's medical and adult-use markets as part of our priority of building a profitable and sustainable business. In Canada, we continue to launch new and innovative products. Our oil portfolio continues to grow and is stable for many adult-use consumers looking for Cannabis wellness options.
On the innovation front, in early Q2, we launched a capable CBN product, which is the only product of its kind in the market and gives users the ability to inhale CBN for faster onset, which is easier to titrate. In late Q2, we released a capable CBD product where we saw a gap in the market, as other CBD vapes were either heavily diluted or subject to user difficulty as CBD crystallized in the competitor's purchase.
These quality and innovative products will be accessed by more consumers as MediPharm officially launched in Quebec in May. Quebec is one of the leading provinces in cannabis sales and with a more complex listing process, the product categories are not crowded as we sometimes see in other provinces. The growth in Quebec should be better reflected in Q3 and onwards, as we add more skews and fulfill weekly shipments.
We continue to see opportunities to utilize capacity as our CMO partners grow their brands, such as the expansion of avocados gels and topicals and the growth in Ace Valley vape products, which we now produce for cannabis growth following the successful acquisition of that brand. Our sales in the domestic market are not where we want them to be. In Q2, we continue to be strained by COVID-19 restrictions at the retail level, which resulted in eventual distributors lowering inventory on hand.
For the majority of Q2, the province of Ontario, which is our biggest domestic customer, still has significant COVID-19 restrictions placed on retail stores. As MediPharm is still less than 12 months into domestic retail sales, I see some immediate improvements we can make in managing provincial listings and fulfillment. This coupled with our high-quality products and relaxed COVID-19 restrictions can result in near-term improvements in this idle market.
I will touch on our strategy to increase sales later in the call. Finally, we continue to add to innovation in both products and manufacturing automation. Our team of research experts have developed other rare cannabinoids formulations beyond our recently launched CBN and have innovative consumer delivery methods such as tasteless and odorless water-soluble drops.
We will work with provincial distributors to sell these new products as the various provincial listing schedules permit. In automation, our engineers continued to deploy equipment already purchased to reduce the direct cost of our manufacturing. This will help improve the gross margin on our high volume skews such as our Cannabis oils.
Currently, MediPharm branded oil ranks fourth in Cannabis oil sales in Ontario. This is with double the retail price of the average oil skew. This proves Cannabis consumers are starting to recognize and are willing to pay more for high-quality products. And also presents a great margin opportunity as we implement our fully funded automation. Overall, our Domestic presence is still growing revenue, but it serves as a proof-of-concept for our ability or about end-to-end development, manufacturing, and distribution solutions for multi-national Pharma, CPG, and innovative health and wellness brand companies. I will now turn the call over to Greg to discuss our Financial results.
Greg Hunter
Thanks, Keith. And good morning, everyone. I'm pleased to report, we continued to make progress with our international expansion, with international revenues increasing 24% sequentially in Q2 versus Q1. This is the second consecutive quarter with double-digit international revenue growth. In addition, we added 2 new customers in Germany, bringing our customer count with successful German deliveries to 4.
Germany is the largest international medical market with a market value estimated to be PS7.7 billion by 2028 according to Forbes and continues to be a strategic priority for MediPharm. As Keith mentioned in Q2, we continue to experience Headwinds in our domestic business driven by restricted COVID -19 lockdown and further channel inventory reductions with provincial distributors, which caused our Canadian and overall revenue to decline sequentially.
Despite COVID-19 making the first half of 2021 challenging, we continue to make progress and are optimistic that revenue will rebound post-COVID-19 lockdowns, with our recent expansion into Quebec and the launch of new innovative products such as CBN oil and CBD and CBN vapes. As I said last quarter, as a management team, we are committed to growing our top-line and adjusting our cost structure to return MediPharm to profitability. While we made progress in the first half of 2021 there is still work to be done. Turning to the P&L performance for the second quarter.
Q2 revenue decreased 7.7% sequentially from 5.5 million in Q1 to 5,100,000 in Q2. Internationally revenues increased 24% sequentially to 2.5 million with German revenues increasing 24% sequentially to 1.5 million and Australian revenues increasing 30% sequentially to 0.9 million. Domestic Canadian revenue decreased 26% sequentially to 2.6 million largely as a result of restricted COVID-19 locked down and further channel inventory reductions with provincial distributors, as mentioned previously.
Gross profit for the Quarter of negative $7.7 million was impacted by a $5.7 million inventory write-down and 0.6 million of accelerated depreciation for assets no longer in use. Adjusted for these items, gross profit of $-1.4 million declined sequentially from $-0.7 million in Q1. Q2 gross profit was negative and declined sequentially due to unabsorbed overhead with lower production volumes and product mix with more flour being sold to German customers with lower margins.
General and administrative expenses in the quarter increased sequentially from $4.0 million in Q1 to 5.2 million in Q2, largely driven by bad debt expense for one customer, higher insurance costs, and higher freight expense for our international customers. Marketing and selling expenses in the quarter decreased sequentially from 1.3 million in Q1 to 1.1 million in Q2, driven by lower promotional activity. R&D expenses decreased sequentially from 350,000 in Q1 to 140,000 thousand in Q2.
These expenses will vary as we selectively invest to advance our capabilities and product portfolio. Other operating income increased sequentially from a 0.7 million expense in Q1 to an income of 3.2 million. Q2 included 3.7 million of income from the Canadian emergency wage and rent subsidies while Q1 did not. Finance expense decreased sequentially from 9.7 million in Q1 to 0.6 million in Q2 as a result of accelerated conversions on the convertible debenture. Adjusted EBITDA for Q2 was -3.7 million and improved sequentially from -6.2 million in Q2, primarily driven by income from the Canadian emergency wage and rent subsidy. Moving to a few notable items on the balance sheet. Inventory decreased from $24.2 million in Q1 to $13.7 million in Q2.
This includes the inventory write-down of $5.7 million mentioned earlier. Trade and other receivables increased from 27.8 million in Q1 to 32.6 million in Q2, largely driven by the Canadian emergency wage and rent subsidy. As discussed in previous quarters, there are two customers owing to a total of approximately 19 million, including 8.5 million, which is subject to legal proceedings that we have previously disclosed and remain confident in its collection. The remainder of the 19 million is due from a second customer, and we are confident in its collectability.
Adjusting for these 2 customers, and the wage and rent subsidy trade and other receivables are 10.2 million. The current tax receivable of 4.3 million is a refund from 2020 that we expect to collect in Q3 and will further improve our cash position. Finally, our cash balance on June 30th was 38.9 million, which decreased from 42.1 million on March, 31. The cash balance decreased 3.2 million, largely driven by operating activities. Capital expenditures were modest and approximately 180,000 for the quarter.
Year-to-date capital expenditures are 460,000 as we continue to manage and prioritize select capital investments to expand the business. The cash balance on the convertible debenture stood at approximately 2 million at the end of June, which is due to be repaid in September and October. While we made progress in the quarter by expanding our international presence and revenue and managing our cash consumption, we still have work to do to return the business to profitability and drive positive cash flow. With that, I'll turn it back to Keith.
Keith Strachan
Thanks, Greg. (ph). This morning, we were also very excited to announce the appointment of Chris Taves (ph) as Chair of our Board effective immediately. Chris (ph) joined our Board in July of 2020 and he's applied his leadership in many areas, including as Chair of our audit committee and providing guidance to our successful March 2021 financing.
Chris is a financial industry expert, serving as Chief Operating Officer of BMO Capital Markets, one of the largest banks in North America. He also serves as a board member of BMO, China CO, and First Mortgage General Partnership. I would like to pass the call to Chris to introduce himself and discuss our Board priorities.
Chris Taves
Thanks, Keith. I appreciate the Board's confidence to oversee the leadership of MediPharm as it executes on a strategy, to be a leader in the supply of Cannabis-based drugs and API to pharmaceutical companies around the world. In my career at BMO Capital Markets and prior, I have worked with many public companies, including those with Cannabis and Pharma specialties.
With MediPharm, I see an incredible growth opportunity, as the pharmaceutical industry is just scratching the surface when it comes to accessing the therapeutic benefits of cannabinoids
in drugs with marketing authorization. MediPharm's Recent drug establishment license is a huge endorsement that it will be a go-to supplier for big Pharma Cannabis market entrants. One of the Board's current initiatives is the appointment of a permanent CEO. To date, we have had significant interest in candidates across North America and have been interviewing strong candidates, meeting with these individuals has assisted us in perfectly defining the right person for the job.
Since we started this task, MediPharm has made great progress in international sales and specialized pharmaceutical licensing. Developments like these in a fast-moving industry have helped us refine the experience and expertise in the criteria for our permanent CEO. Our selection process continues to progress and we're confident we will appoint the perfect candidate for this role and the exciting future of MediPharm. And then, in the interim, the current management team has the full confidence on the Board to execute on the Company vision and to drive growth. Keith, back to you.
Keith Strachan
Thanks, Chris. I want to thank you for taking on this role and providing your experienced leadership at such a pivotal point in our business. I will now provide some final thoughts on our outlook and strategic plan to increase revenue. There's a lot of opportunities for increased sales in Canada and internationally. Based on our fully built and funded manufacturing platform, we can increase revenue significantly without further capital investments. Activities are already underway to achieve this goal. These strategic revenue goals have three main focuses. One, investment into more sales resources.
Domestically, we have increased our sales team by almost doubling our retail sales reps subsequent to quarter close. With the more ongoing ramp-up of the innovative products we offer to the market, we can make great use of this newly scaled salesforce. Internationally, we are putting more boots on the ground in growing markets.
In August, we had one of our top international business development associates relocate to Frankfurt, Germany. And in September we have our first personnel in Mexico, starting with us, to expand our reach in Latam. As our area of highest growth so far this year, having in-country sales and business development representation will add to our growing international revenue opportunity. Two, expanding contract manufacturing:
Providing partners with access to the highest quality cannabis manufacturing platform to deliver them cost-savings and international reach. For example, here in Canada, just last week, we started a GMP tolling contract for one of the world's biggest cannabis companies. On the international front, we have continued to add to the private label services provided out of MediPharm Labs Australia. Three continued innovations. Internationally, there is a lot of white space when it comes to innovation.
Just like in the established adult-use markets such as Canada or legal U.S. states. Many international patients are seeking new formulation and delivery methods. The barrier is usually making one of these popular formats in a GMP-compliant way. MediPharm has just the expert team to assess and meet that challenge. Domestically, as Cannabis categories become crowded, and distributors scale back product listings, innovation is the sure way to get the product on the shelf and increased sales. We saw this recently with baseball, CBN, and CBD products, and we'll use the same approach going forward to get more products on the shelf with retailers.
In the pharmaceutical space, our R&D is more complex as Pharma customers will seek API suppliers who have borrowed the active ingredients from the FDA. These filings take in-depth characterization of substances with very tight specifications. This is an area where we want Chris ' efforts to remain ahead of the competition.
Overall, Q2 was a productive quarter with the accomplishment of our drug establishment license and further global market penetration for our products. 2021 is a year where progress will occur as we gain traction with our priorities and create a MediPharm that I know will deliver tangible value to our customers and shareholders. In my opinion, our current market Cap does not reflect the value of our very unique pharmaceutical licenses and strategies, making MediPharm a great investment opportunity.
As one of the founders of MediPharm, I can tell you that since day one, we had the vision to be a pharmaceutical Companies specialized in Cannabis, providing Companies in patients with API and drugs through a traditional marketing authorization, as opposed to being just another Cannabis Company who distributes to non-federally regulated or special-access medical programs. Having achieved our Drug Establishment License in Q2, I can confidently say we have reached that goal. This will result in many positive business outcomes for MediPharm and its shareholders. I cannot wait to show you what we will do next. Now, Operator, can you please open the lines for questions from our callers?
Question-and-Answer Session
Operator
[Operator Instructions]. Our first question comes from the line of Aaron Grey with Alliance Global Partners.
Aaron Grey
Hi, good morning. Thank you for the questions and congrats. Congratulations on the drug establishment license. So the first question for me, I'd like to just talk about the license and maybe in terms of the timing of where you see the opportunities. So certainly seems that you guys have a differentiated strategy there than maybe some of the other Cannabis plays, but we talk a lot about potential API that allows for you guys in terms of Big Pharma, could you maybe elaborate maybe on some of the conversations that you've had with pharmaceutical companies to provide that type of API.
Right now, there are such drugs like have dialects and Marinol out there. So how do you see in terms of the timing of that license eventually translating into revenue? Is it more of a near-term or maybe 2,3 years out there? Thank you.
Keith Strachan
Thanks, Aaron. Appreciate you calling in this morning. Great question. Our drug establishment license does truly differentiate us in many ways and does bring both near-term and long-term opportunities. I think in the near term, what we're looking at is more international sales. So we're resolved at 24% growth quarter over quarter in the international sales. This helps us, as part of the complex industrial processes and export processes around special access programs that are opening up and it seems like every couple of weeks you see a new country that's going to try something.
I think those health authorities are looking for a GMP solution and sometimes just didn't have one because they didn't exist. So, there are some near-term opportunities there for sure that we'll keep seeing that international growth. I think you nailed it on what the long-term opportunity is. Really, that's an API provider for drugs that have marketing authorizations. So, we've seen the great success of the effects of Epidiolex. If you look at their annual run rate in the U.S. of Epidiolex sales being over $600 million U.S. a year. You could imagine that most generic Companies are looking at that as an opportunity. And with those, they're already starting the development process. So there are multiple chair Companies that are looking at it, the development process, but that long term.
Just to create a timeline on that, there's a lot of patents on that product, it could take up to 2025, 2026 to really enter the market with the generic. But when they do, generic sometimes make up 30%, 40% of the market share. So there's a massive opportunity. These other drugs like, as you mentioned, Marinol in Germany, it's referred to as [Indiscernable] is still one of the main concentrates sold in Germany. So we can participate that right away as those products are often right up, has not been an office API opportunity for some really generic drug Company, but there is an opportunity for ourselves. To summarize, there is near-term material growth that does make a difference to our revenue today. And then the long term is limitless in where we can go.
Aaron Grey
I appreciate that color, Keith, that's really helpful. The second question for me, turning to the domestic market in Canada, you talked about finding gaps in the marketplace certainly being some potential buyers sure really are taking a focus on that in terms of new products that they'll take in. But just an overall question in terms of, as you're looking for these gaps in the market, how do you look at potential ROI in terms of the effort and CapEx needed is maybe ramp up production of that product versus how big a market share of maybe that gap in a market might be? How many other interests might come in? I will just like if some further commentary from you guys in terms of the ROI and different things you're looking at as you look to find gaps to mark and then decide whether or not you guys would be an opportune position to try and fill that gap. Thank you.
Keith Strachan
Yeah. For sure, Canada continues to be the focus that we're looking to increase in that retail space. As I mentioned, there's a big opportunity there. Any product that we do, we're obviously looking at it from an ROI perspective to make sure that it is accretive to the bottom line, but really what we're looking at in innovation is the Canadian space, there are some large players in there that have spent a lot of money on their retail whether they've owned, there's a retail platform, or they have like a mass of retail sales group. I don't think that we're looking to go head-to-head with a lot of those in some of the big categories.
But what we can do is we can own niches where there is innovation because we have such a strong team. So really for us, it's things like that CBN base, where we are the only ones who make it for the CBD based that doesn't crystallize, that's where we see that -- so in the future, we're looking at other formulations that would be minor cannabinoid base something like a CBG which you can find in the U.S. today, but not yet in Canada is probably a good spot where we could be and then, you know what different formats we can do.
So looking at there are some water-soluble products in the market today, but most of them are low quality. So how could we maybe make a higher potency water-soluble product for consumers? So there is a lot of -- there's a lot of opportunities there and a lot of ROI. And we're really focused on those niches of innovation and what really also helps us. And what's important is we are using it as a platform, as a proof-of-concept for our Big Pharma and CBG customers current and in the future.
So as we say start, I'd like to launch, like a CBN or CBG is their patient base. We're able to show that not only through the reformulated market and some small-scale responses from consumers and patients. So it really gives us back a proof-of-concept platform that really does build into that more future opportunities.
Aaron Grey
Great, thanks for that call and then just last one from me, if I could. Just for Germany then, Right. So as we look potentially see a bigger ramp in the overall market, once some of these COVID bought on that ease could shock on whether or not on your dice and there are any types of bottlenecks in terms of getting supplier or getting the product out there for a starter or any of your partners or if it's more of just like a demand and current -- current COVID dynamics there, right? So if the demand comes up and you see more prescriptions being issued in Germany. Do you guys have all the capabilities to fulfill that demand in the back half or through 2022, thank you.
Keith Strachan
Yes, thanks, Aaron. No bottlenecks on our side on, let's say, the delivery and fulfillment. We have no problem making those products and really, now that we have this drug establishment license, it does help with a protect, as far as EBITDA supply chain, whether we make it in Canada or we make it in Australia, it starts to give us a lot of options. Where is the bottleneck?
And you know, we saw in Q1, we had 2 customers; in Q2, we had 4 customers. And then we'll see that grow at that kind of rate. Where the bottleneck is is more on the registration site, as I mentioned in the call, when there are new narcotic products and when we onboard a German customer, they have to be on a B Farm, which is like the FDA of Germany. And then they register that product through [Indiscernable] .
That process can take anywhere from 3 to 6 months. And then once it's registered, we're able to fly for the next for authorization. So there is a bottleneck on new products. But once the channels open, we're able to get a good cadence. I don't think that we have any problem with demand. The demand keeps going up and really right now, we're the only really private label option in a state like Germany. So our competitors in Germany are with their own branded products like, let's say Tilray.
We have Tilray Oil in Germany [Indiscernable] we see more and more entrants like STADA and our partner that adjuncts or even [Indiscernable] we're looking to outsource it, we're really one of the only options and now we're probably the best as far as quality, license, and experience in getting to the market. So, I think we'll continue to see demand and MediPharm's prepared and well-capitalized, and well resourced to meet that demand.
Aaron Grey
Great, thanks for the call and I'll jump back in the queue.
Operator
Your next question will come from the line of Scott Fortune with ROTH Capital Partners.
Scott Fortune
Good morning and thanks for the questions. Real quick. First, housekeeping call where are the inventories sitting at now at 13.7 billion after the inventory write-off, and what type of cost does this inventory include. I just want to get a sense as we look at gross margins going forward from this point as far as that's concerned
Keith Strachan
Yeah. Thanks for the question. So we have the inventory being more right-sized now with some of these gaps. So we may be sitting at 13 million, there's five million exchange in -- as we've said, in raw material, which primarily flowers that we use for extraction And then the bulk of the rest, it's in the finished or semi-finished product, which again, some of that -- what's in there, which is new in the last couple of quarters is finished goods, flour that we sell for sale purposes or end consumption within Germany.
Obviously, this inventory, where we're tracking very closely as you can see, we are trying to manage it down as we improve our cash position and get better tendency issues with demand, both from the provinces and internationally and we'll continue to focus on managing our inventory to help improve cash.
Scott Fortune
Okay. I appreciate the color. And then, focusing on the EU or international side, Keith, you called out new product formats on the GMP compliant side of things and the timing is maybe new derivative products being getting approved for use in Germany, and then from your skews in the product portfolio, what's being in demand and the opportunity to increase different skews our products into Germany, as we look out to the second half and into 2022 here?
Keith Strachan
Yeah, Thank you. It's a great question I think. As you look at like U.S. date in Canada, popular format, things like a vape pen or like a water-soluble that you can put into a drink easier or something like that. As far as patient use goes for patients is really a great opportunity even well-developed medical programs such as the one in Australia right now really drive flower oil the only choices for them.
The reason why this has happened today is unique to do it as a GMP product. So doing like an oral solution, as a GMP product is something that there are some systems are in place because there are oral solutions for other drugs. But when you look at something like a vape pen, GMP vape pen has become very complex as you have to take into consideration the hardware and the stability of something in the vape pen and like nothing and we've got these new recreational markets that are so recent I don't think a lot of those studies have been done, so we're doing those now.
That registration process will probably take another three months to six months. So it's probably more of a 2022 opportunity as far as let's say something like a GMP-based friend. But We might be able to achieve that late this year, or early next year. But the development is starting today and we've just -- in the first part of this year engaging customer interest and patient interest and the ability to source all those GMP suppliers for that supply chain.
Scott Fortune
Okay, appreciate. And in focusing back on Canada, the growth in the second half here, as we open full-day, are you seeing more consolidation here or where is the growth going to come from? More pick up in the 2.0 expansion in that category. And then what are you seeing from some of the LPs front side of things or are they looking to outsource now more potentially in this environment, just thoughts on how we are stronger, second-half growth in Canada.
Keith Strachan
For sure, I think, you know, we'll continue to sell our quality and innovative products, branded products at the store level. And I think as I mentioned, subsequent to quarter close, we've increased our sales team there and some of our resources on the retail efforts. So we could see some incremental growth there.
But since we do participate in some of the smaller categories, we're back to more of incremental growth part of the back year. A back half of this year, I think where you mentioned where we see also a lot of opportunities is the B2B services coming back. So 2019, 2020 a lot of our business was B2B and now are seeing some of that come back as far as some of the big licensed producers that have a great brand, great marketing, really need some help there.
And then what's really great is now that we have this drug establishment license and we're the only one in North America who have this drug establishment license for the extraction of cannabinoids, large license producers are coming to us to do GMP -type services, whether that's totaling or using material that we source, we can make them like a GMP API or a GMP concentrate so that they can put that into their pipeline internationally.
And we've already started doing some of those activities just as recently as August, so we'll see a good pickup in some of our B2B totaling services with that.
Scott Fortune
I appreciate the color. Thanks, guys. I'll jump back into the queue.
Keith Strachan
Thanks, Scott.
Operator
Your next question will come from the line of Natalia Cochran (ph) with Scotiabank.
Natalia Cochran
Hi there. Thanks for the color on international and I have some questions on the Canadian side. So from the perspective of the provinces, can you give some color on what you're seeing now, are things improving compared to the restrictions that we've had so far? Are things opening up? And for instance, is that or doing more? And the second part of the question is the margin side. So can you give some color on are margins expected to improve or at what sales-velocity will you break even on the profit -- gross margins? So if you can give some color on that. Thank you.
Keith Strachan
Sure. Thanks, Natalia (ph). Thanks for joining us this morning. I'll provide a little bit of color on the promises and I'll hand it over to Greg (ph) to talk about the plans for their margin improvement. On the provincial side, we are seeing some encouraging demand signals, the province of Ontario being our biggest domestic buyer continues to open more retail stores. With restrictions being lifted, on June, July as far as people being able to shop in-stores, again, because we do have that high-quality wellness brand.
It is something that helps for sales in Brick-and-mortar as some of them might go in for flowers and then see a CBD or CBN oil that they want to try. So that doesn't really help us as far as the demand signals go. We're waiting to see if provinces will pick up inventory again like the distributors will pick up inventory as we saw in the last bit of 2020. So what we've seen to date of 2021 is distributors lower their inventory on hand to more of like a just-in-time ordering to get it out to retailers probably because of the uncertainty around COVID-19.
Now that uncertainty is away, we hope to see them pick up that inventory again, which will advert smoother and more deliveries on our end, but we are still waiting to see how that plays out. And I think that the provinces have done a good job now, looking at consumer demand. I think in our carrier specifically, they are changing the product core system and how they work with licensed producers on, what products they take in and what product they list, and when they list them. And I think given our long relationship with the provinces, that will bode well for MediPharm as well. So I'll hand over Greg to also talk about the margin.
Greg Hunter
Yeah. Thanks, Keith. With gross margin, as said prior quarters, we do see a path back here to not only just profitable on the gross margin side but EBITDA profitability. And there's a couple of different aspects where we're focused on that are going to drive that. One, as Keith mentioned in his prepared remarks is around automation, which we continue to bring into our production facilities, which will obviously enhance our cost positions.
The other one with the international expansion, which is why we're so pleased to see that sequential growth, there's obviously higher pricing and better profitability in the international markets, so that will continue. The other one is, as I mentioned, with some of our flower sales into the international markets where we did have to lower margin s this quarter, impacting us. The team is actively working on improving that market and we expect to see some improvements there in the future and then as well the international market, with some mix changes as we move into different products and potentially away from flour and oil should help.
And then the last 2 I would mention is one, the volume as I said in my remarks was challenging in the domestic market within Canada. So as Keith talked about it, and we expect to see that improvement and volume go up which helps us significantly in our manufacturing. And then finally, with some of the new products that Keith had talked about. With some of the new innovative products where we can get a higher margin on those as well. So those are all the different factors that are going to contribute in the future as we see getting back to a positive gross margin.
Natalia Cochran
Sounds great. Thanks. That's all from my side. I'll pass on.
Greg Hunter
Thank you.
Operator
Your next question will come from the line of Tamy Chen with BMO Capital Markets.
Tammy Chen
Good morning. Thanks for the question. Wanted to ask about Germany. Can you speak a bit about the evolving competitive landscape there? Because I think, as noticed, a couple of the other Canadian licensed producers that participate in that market have recently mentioned that the competitive level has increased in that market. So if you could talk a bit about that and how the relationship with STADA has been involving with respect to your penetration of Germany.
Keith Strachan
Yeah. Thanks for joining us. I think Everyone, sees that -- that great opportunity in Germany and we'll continue to see new entrants into that space. I think to date, we haven't been really hindered by co-profession, especially on the concentrated side as there are still products to be registered there. So I think that there's still opportunity. I think if we hear some more companies going into that phase and some more investment in that space, but really, I think it's also being faced with new patients.
So every month if you look at some of the data, I mean out of Germany as far as coverage by the national insurer, I think we're getting more and more patients that are looking to [Indiscernable] cannabis. So, as more people come in, there's also more patients there, so there's nothing there that we really -- that we see is a near-term, really competitive, but we continue to work with our partners there, especially STADA to look at that landscape and look at our pricing to make sure that we don't get priced out of the market and things like that.
So, it is something that we're looking at, but it's encouraging to see everyone else wanting to come to Germany because obviously, they see what we see in the great opportunity there. The STADA relationship is progressing nicely as we mentioned, we made our second delivery to them on the oil side in Australia in Q2. We continue to provide them with flowers of value-add service.
And as Greg (ph) mentioned, we think that we can improve our margin on that value-add service. But we've made multiple deliveries from Canada to Germany for that flower supply. So it really is ramping up I think as a large multinational Company. They are pretty cautiously moving forward with our product launches, so their trajectory is one that's a bit more conservative, as they add in more patients.
But what they are adding in on the patient side is probably more sticky than you would see in some of their competitors. So, their focus, and I believe is a great one, is that they're going to the patient level for education to make sure that they get those patients and then they keep the patients, no matter what cannabis solution they are looking for, for that therapeutic benefit, whereas some of the other companies in Germany are really focused on, let's say, pharmacist s that have a high sell-through of Cannabis, so then they're the only men in those pharmacies.
So I think that the status approach, while a little bit slower start, has a longer scale power. And we'll see more and more great results and the ability to put patients into new formulations or product delivery mechanisms in the future as well. So we're really happy with the progress there.
Tammy Chen
That's interesting ongoing through their patient level. Okay. And then the last question for me is a bit of clarification. The drug establishment license, Sorry. Did you say that did you say you are working on over the next several months, some sort of registrations and filings with the FDA for APIs, and that only once you have that, then the long-term vision of supplying APIs to Pharma Company can happen. Is that's the next step that you have to do to finish up on top of the drug establishment license? I wasn't clear if you could clarify that. Thank you.
Keith Strachan
Yeah, for sure. The drug establishment license is very unique in our industry, but not unique in the pharmaceutical industry. And there is a number of other steps that we could take to work with it. So the long-term strategy of working with a major pharmaceutical Company on the launch of a new drug containing Cannabis or a launch of a generic drug containing Cannabis is well underway and there's a lot of steps that we could do today that we are doing today to achieve that.
There is a Fortune, one of the Trex. If you register a new drug, you'd go, to the U.S., through a new drug application process. And the natural that you don't need to have that say the API registered as the registry in the entire drug. We could provide our customer with the API in that case. well, preferably doing the FDA filings. If you're doing the abbreviated drug application, which is a generic drug application with the FDA, they would be looking for you to be using an API that does have a drug master file already on file with the FDA.
So it's an important step for us to do and is something that MediPharm is actively working on. Going on well with the process of registering our API we're not only the FDA but Health Canada and the European Medicines Agency. So that way we weren't at our customer's needs. An API in the pharmaceutical space could go to those Vegas basis to validate that though EGI will work in there, I'm hundreds.
Tammy Chen
Okay, I understand. Thank you.
Operator
At this time, there are no further questions. Do you have any closing remarks?
Keith Strachan
Yeah, thanks. I really appreciate everyone joining the call. It was great to speak with everyone this morning. As I mentioned, we're really excited about the future and I appreciate your time. I hope everyone has a great day.
Operator
Ladies and gentlemen, thank you for participating. You may now disconnect.
A failed Co? See red below.-->>MediPharm Reports Second Quarter 2021 Results and Advances Pharmaceutical Strategy; Announces new Board Chair
Their active clients are well positioned for EU legalization but it's not on the near term horizon. South American announced customers are going no where.
https://www.medipharmlabs.com/news/press-releases/detail/188/medipharm-reports-second-quarter-2021-results-and-advances
thanks fun , for the much needed and greatly appreciated POSITIVE news......a little light at the end of a long tunnel .
Barrie's MediPharm Labs becoming world player in cannabis market
https://www.barrietoday.com/following-up/barries-medipharm-labs-becoming-world-player-in-cannabis-market-3981490
'We’re actually the first in North America to have a pharmaceutical production licence for the extraction of cannabinoid,' says Keith Strachan
about 2 hours ago By: Ian McInroy
Keith Strachan, president and interim CEO of Barrie-based MediPharm Labs, holds containers of CBD 100, the company'??s maximum CBD (cannabidiol) formulation. The company has just received a pharmaceutical certification from Health Canada.
MediPharm Labs employees pack product for shipments in the John Street facility.
MediPharm Labs vape-pen cartridges are prepared for shipment. Vaporizing accounts for anywhere from 20 to 30 per cent of cannabis users in Canada, according to company president and interim CEO Keith Strachan.
PreviousNext1 / 3 Keith Strachan, president and interim CEO of Barrie-based MediPharm Labs, holds containers of CBD 100, the company'??s maximum CBD (cannabidiol) formulation. The company has just received a pharmaceutical certification from Health Canada. Ian McInroy for BarrieToday
Keith Strachan, president and interim CEO of Barrie-based MediPharm Labs, holds containers of CBD 100, the company'??s maximum CBD (cannabidiol) formulation. The company has just received a pharmaceutical certification from Health Canada. MediPharm Labs employees pack product for shipments in the John Street facility. MediPharm Labs vape-pen cartridges are prepared for shipment. Vaporizing accounts for anywhere from 20 to 30 per cent of cannabis users in Canada, according to company president and interim CEO Keith Strachan.
The next act on the world business stage is playing out for a Barrie-based company.
MediPharm Labs has just achieved a major pharmaceutical manufacturing milestone since starting up about six years ago.
“The latest news for us is we just received our Health Canada licence for GMP (Good Manufacturing Practices), a pharmaceutical designation that’s recognized around the world,” MediPharm Labs president and interim CEO Keith Strachan tells BarrieToday.
The company, which produces concentrate cannabis products, is part of a pharmaceutical inspection co-op that has more than 50 countries in it, meaning it’s an internationally recognized standard.
Here in Canada, the industry is governed under the Cannabis Act and a general cannabis production facility would have what’s called a good production practices designation.
“So, we have taken it to the next level by getting this pharmaceutical licence,” he says. “We’re actually the first in North America to have a pharmaceutical production licence for the extraction of cannabinoid, a very unique licence not only for Canada, but for Canada and the U.S.”
Strachan says the new certification will make some major changes in what the company does.
“We can now provide cannabis for pharmaceutical drugs,” he says. “Being a traditional pharmaceutical drug means access is easier, so you can pick it up from the pharmacist as well as doctors being able to prescribe it easier.
“There is some data behind it, so it changes our business a lot in the products we can make.”
MediPharm Labs, headquartered and started in Barrie, has about 120 employees based here with another 25 or so working from home.
“But we also have a factory just outside of Melbourne, Australia, with about 45 staff there today. We supply the Australian market and European market from there,” Strachan says. “What this new certification does for us here in Barrie is now we can make more products internationally.
“Today, we ship products to Australia from our facility, as well as places like Peru, but this will open the doors for us to sell products all across Europe as well eventually getting into places which have medical programs, such as Mexico and Brazil.”
What’s important about MediPharm is that it's “pharmaceutical and medically focused,” Strachan says.
“It was founded in 2015 on the basis of it being a pharmaceutical company,” he says. “We just entered into a joint research agreement with McMaster University where they’re using our CBD (cannabidiol) oil to do studies on post-surgery pain.
“That’s research that’s been approved by Health Canada and being done by experienced researchers. It could be a different option for pain management,” Strachan says.
The company’s nondescript John Street building houses pristine high-tech labs and security, spotless sorting and shipping areas and, of course, boxes and boxes of various strains of raw herb waiting to be transformed for various purposes.
“It’s an old building that we completely renovated and purpose-built for our use,” says Strachan. “It contains multiple ISO (International Organization for Standardization) clean rooms that you would see in any pharmaceutical facility.”
COVID-19 has been challenging for MediPharm, he adds.
“As an essential service, we never closed so our staff did different things like shift segregation so the night shift would never meet the day shift,” he says. “We put a number of protocols in place here to ensure it was a safe environment.
“It did cause a slowdown in sales as we saw some of our regulatory pathways come to a bit of a halt while international health authorities dealt with the pandemic at hand. But we’re seeing a lot of that get better now.
“There’s a lot of promise coming through the other end of COVID, but we worked really hard to make sure we got through it.”
This stock is such a "ball and chain" in my portfolio ...
When was the last time this had a bullish move up ?
Endless shares being sold it seems .
Stay safe >> M
Agreed, it may take years if it even happens.
Thanks Funman, interesting read. For MEDIF to make money, trials need to bear fruit, owners of those trials need to engage and partner with MEDIF to produce and distribute product. It's potentially a very compelling long term strategy, but there's a long row to hoe before this bears the fruit expected.
Medipharm Labs Is One Of The Most Misunderstood Companies In The Canadian Cannabis Sector
EDITORIAL Jul 21, 2021
8:01 AM EDT
3 MIN READ
BY MICHAEL BERGER
https://www.technical420.com/cannabis-article/medipharm-labs-is-one-of-the-most-misunderstood-companies-in-the-canadian-cannabis-sector/#
We consider MediPharm Labs Corp. (TSX: LABS) (OTCQX: MEDIF) (FSE: MLZ) to be one of the most misunderstood growth stories in the cannabis industry and are favorable on how the business has advanced since 2020.
From entering new international markets to advancing initiatives that are focused on the biotech side of the cannabis industry, MediPharm has been nothing short of an execution story and is an opportunity to be aware of. The company represents a multi-faceted growth story and is a pharmaceutical firm that specializes in the development and manufacturing of cannabis active pharmaceutical ingredients (API) and finished products.
Following some major developments, MediPharm has evolved into a full-service biotech firm that can manufacture and sell cannabis APIs and pharmaceutical drug products. Due to this, the company can capitalize on the increasing demand for APIs at the global level and is positioned to record strong growth.
Granting of the Drug Establishment License is a Major Milestone
Earlier this week, MediPharm reported to have received a Drug Establishment License (DEL) from Health Canada (in accordance with the Food and Drugs Act and Regulations). We consider this to be an important milestone for the company as the DEL shows that it is following Good Manufacturing Practice (GMP) standards.
Another reason for the significance of the license is that it’s a first of its kind license for cannabis manufacturing in North America. A Canadian GMP DEL complements MediPharm Labs’ existing Australian TGA GMP certification. The license can be used for the manufacturing, testing and sale of API and pharmaceutical drug products that contain cannabis. The DEL includes drugs that have marketing authorizations as either novel or generic pharmaceutical drug products containing cannabis.
The DEL allows MediPharm to leverage mutual recognition agreements (MRA) that were established between Canada and other global regulatory authorities including the European Economic Area (consists of all EU member states as well as the three countries of the European Free Trade Association, Australia, the UK and Switzerland).
The granting of the DEL enhances MediPharm’s supply chain capabilities by allowing it to ship products from Canada to its global customer base. The development further strengthens the company’s global reach, including potential US pharmaceutical customers, and we are bullish on this aspect of the story.
A Undervalued Growth Story?
Physician prescribed pharmaceutical drug products that contain cannabis are being used to treat an increasing number of indications around the world and this number is expected to continue to grow with additional new drugs and abbreviated new drug applications. According to Grandview Research, predicts this segment of the pharmaceutical industry to be a $5.8 billion market by 2027.
When paired with the Cannabis Drug license that was granted to MediPharm earlier this year, the DEL will allow for the commercial distribution of drugs that contain cannabis. With many global pharmaceutical trials underway in which cannabis is an active ingredient, many drug producers will need a manufacturing partner. MediPharm is strategically positioned to provide that service to all global pharma partners.
The granting of the license further differentiates MediPharm from other Canadian cannabis manufacturers and we are bullish on the growth prospects that are associated with it. At current levels, we find the valuation to be compelling and the risk-reward profile to be favorable.
We believe that MediPharm is executing on a multi-national and multi-faceted growth strategy and are bullish on how the story has advanced so far this year.
Agree. Let's hope that they aren't just being academically adept at filing paperwork, and that they are already targeting potential new customers with never available before solutions.
The time to act is now. They have to generate new revenues.
I am very afraid that the next ER will be largely unchanged, and the next round of dilutive equity raising may be just around the corner.
lazur Wednesday, 07/14/21 10:11:55 AM
Re: FUNMAN post# 6635
Post # 6636 of 6636
agreed, medif needs to take advantage of the current illegality of pot to develop IP, products, customers to at least be worthy of a buyout rather than just be squashed. For them it's a foot race against legalization in the US.
agreed, medif needs to take advantage of the current illegality of pot to develop IP, products, customers to at least be worthy of a buyout rather than just be squashed. For them it's a foot race against legalization in the US.
I partially agree. Big Pharma isn't in cannabis because it is federally illegal. Once the barrier goes away you can bet they will be leaders in R&D, manufacturing, and distribution of cannabis drugs.
They won't cede ground to anyone. They will be direct competitors of MediPharm. They don't need to acquire MediPharm because they have the wherewithal to do it all.
I'm not sure where MediPharm goes with this.
Something good may come of it.
We'll only know that if we see new customer announcements that leverage the license.
I'm not buying, but I am not selling either.
lazur Wednesday, 07/14/21 09:57:17 AM
Re: FUNMAN post# 6631
Post # 6634 of 6634
Isn't the good news on this is that a cannabis friendly company is now a pharmaceutical company, where historically big Pharma has been a bit wary of cannabis due to its impact on other drug sales?
This news sets up an interesting challenge of a David v Goliath in regard to MJ pharmaceutical drugs. I usually bet on where the big money is, but I'll keep my holding here for now since this is worth watching over the short term...
Isn't the good news on this is that a cannabis friendly company is now a pharmaceutical company, where historically big Pharma has been a bit wary of cannabis due to its impact on other drug sales?
This news sets up an interesting challenge of a David v Goliath in regard to MJ pharmaceutical drugs. I usually bet on where the big money is, but I'll keep my holding here for now since this is worth watching over the short term...
At this point buy out target ?
'Pisd', for contracts/revenues; so "manufacture"? or trial?, Labs losing my confidence atm... gl
W/Blue highlights--->>> MediPharm Labs Achieves Major Pharmaceutical Manufacturing Milestone:
Receives Canadian GMP Pharmaceutical Drug Establishment License
This achievement may turn out to be extremely important. Instead of MediPharm's original extractor business model to support cannabis manufacturing of oils and derivatives, MediPharm has positioned themselves as a manufacturer of cannabis drugs. Moreover, this qualifies them to receive USA FDA reviews and potentially opens up the USA medical market.
Upon USA cannabis legalization I think this is going to put them in direct competition with the USA pharma industry.
USA Pharma will not cede cannabis drugs to MediPharm. Currently, they just aren't operating in an "Federally illegal business", and manufacturing products that will compete with some of their own.
MediPharm's new business plan still needs time to unfold. I hope they aren't reliant upon waiting for USA legalization.
We have to wait on new customer announcements from MediPharm that leverages this new first of its kind license.
Regarding is this investible news, let's see what they do with it. - FUNMAN
https://ir.medipharmlabs.com/press-releases/detail/186/medipharm-labs-achieves-major-pharmaceutical-manufacturing
July 14, 2021
* Health Canada Drug Establishment License allows MediPharm Labs to conduct pharmaceutical manufacturing and sale of Active Pharmaceutical Ingredients and Pharmaceutical Drug Products
* Makes MediPharm Labs a full-service pharmaceutical company that can support the global pharma entry into the drugs containing cannabis market, including in the US
* First domestic GMP License in North America for cannabis-related pharmaceutical manufacturing that includes the extraction of the cannabis plant
* Permits the manufacturing of all cannabinoids including MediPharm Labs GMP CBD isolate which is >98% pure
* Enhances supply chain capabilities by allowing MediPharm Labs to ship products from Canada to its global B2B customer base. In conjunction with MediPharm Labs’ Australian subsidiary, this further strengthens the Company’s global reach, including potential US pharmaceutical customers
BARRIE, Ontario, July 14, 2021 (GLOBE NEWSWIRE) -- MediPharm Labs Corp. (TSX: LABS) (OTCQX: MEDIF) (FSE: MLZ) (“MediPharm Labs” or the “Company”), a pharmaceutical company specialized in research-driven development and manufacturing of cannabis API and finished products, is pleased to announce it has received a Drug Establishment License (the “DEL”) issued by Health Canada in accordance with the Food and Drugs Act and Regulations. The DEL serves to confirm compliance to Good Manufacturing Practice (“GMP”) standards.
The DEL is a first of its kind license for cannabis manufacturing in North America. A Canadian GMP DEL complements MediPharm Labs’ existing Australian TGA GMP certification. The license can be used for the manufacturing, testing and sale of Active Pharmaceutical Ingredients (“API”) and pharmaceutical drug products containing cannabis. This includes drugs that have marketing authorizations as either novel or generic pharmaceutical drug products containing cannabis.
The DEL allows MediPharm Labs to leverage the Mutual Recognition Agreements (“MRA”) established between Canada and other global regulatory authorities including the European Economic Area consisting of all EU member states as well as the three countries of the European Free Trade Association, Australia, the UK and Switzerland. This differentiates MediPharm Labs from other Canadian cannabis manufacturers.
In addition, as a member of the International Pharmaceutical Inspection Co-operation Scheme, the Health Canada DEL can be recognized in over 50 different countries including the US and the majority of the EU. With the DEL, MediPharm Labs would be eligible for a US Food and Drug Administration (“FDA”) foreign inspection if a US or other global pharmaceutical customer files a registration application for a pharmaceutical drug product containing MediPharm Labs’ API. In 2020, 64% of drugs exported from Canadian Drug Establishment License holders were exported to the US with FDA approval.(1) MediPharm Labs will use the DEL in conjunction with its Australian facility TGA GMP to optimize the supply chain via MRAs for export of its products to over five countries in 2021.
When paired with MediPharm Labs’ existing Cannabis Drug License received earlier this year, the DEL will allow for the commercial distribution of drugs containing cannabis. With many global pharmaceutical trials underway in which cannabis is an active ingredient, many drug producers will need a manufacturing partner. MediPharm Labs is now strategically positioned to provide that service to all global pharma partners. This includes clinical trial material, which MediPharm Labs already supplies to researchers, and pharmaceutical drug products. Physician prescribed pharmaceutical drug products containing cannabis are being used to treat various indications around the world and will continue to grow with additional new drugs and abbreviated new drug applications. Grandview Research predicts this segment of the pharmaceutical industry will be a $5.8 billion (USD) market by 2027.(2)
“MediPharm Labs has achieved a great milestone with the issuance of our Drug Establishment License. This truly makes us a full-service pharmaceutical company that can support global pharma’s entry into the drugs containing cannabis space,” explained Keith Strachan, President and Interim CEO, MediPharm Labs. “This adds to our growing portfolio of advanced regulatory licenses globally. MediPharm Labs now becomes one of a small number of companies who can take cannabis plant material and produce an API or a pharmaceutical drug product that could have marketing authorization by regulatory bodies such as the European Medicines Agency and FDA.”
This completes MediPharm Labs pharmaceutical licensing requirements. Future specialized licencing achievements are still being pursued in conjunction with specific product registrations and jurisdictions.
(1) https://www.ic.gc.ca/eic/site/lsg-pdsv.nsf/eng/h_hn01703.html
(2) https://www.grandviewresearch.com/industry-analysis/cannabis-pharmaceuticals-market
About MediPharm Labs
Founded in 2015, MediPharm Labs is a pharmaceutical company that specializes in the development and manufacture of purified, pharmaceutical-quality cannabis concentrates, active pharmaceutical ingredients and advanced derivative products utilizing a Good Manufacturing Practices certified facility with ISO standard-built clean rooms. MediPharm Labs has invested in an expert, research driven team, state-of-the-art technology, downstream purification methodologies and purpose-built facilities with four primary extraction lines for delivery of pure, trusted and precision-dosed cannabis products for its customers. Through its wholesale and white label platforms, MediPharm Labs formulates, develops (including through sensory testing), processes, packages and distributes cannabis extracts and advanced cannabinoid-based products to domestic and international markets. As a global leader, MediPharm Labs has completed commercial exports to Australia and completed commercialization of its Australian extraction facility which generated its first revenues in H1 2020. MediPharm Labs Australia was established in 2017.
For further information, please contact:
MediPharm Labs Investor Relations
Telephone: +1 416.913.7425 ext. 1525
Email: investors@medipharmlabs.com
Website: www.medipharmlabs.com
Cautionary Note Regarding Forward-Looking Information:
This news release contains “forward-looking information” and “forward-looking statements” (collectively, “forward-looking statements”) within the meaning of the applicable Canadian securities legislation. All statements, other than statements of historical fact, are forward-looking statements and are based on expectations, estimates and projections as at the date of this news release. Any statement that involves discussions with respect to predictions, expectations, beliefs, plans, projections, objectives, assumptions, future events or performance (often but not always using phrases such as “expects”, or “does not expect”, “is expected”, “anticipates” or “does not anticipate”, “plans”, “budget”, “scheduled”, “forecasts”, “estimates”, “believes” or “intends” or variations of such words and phrases or stating that certain actions, events or results “may” or “could”, “would”, “might” or “will” be taken to occur or be achieved) are not statements of historical fact and may be forward-looking statements. In this news release, forward-looking statements relate to, among other things, conducting pharmaceutical manufacturing; sale of active pharmaceutical ingredients and pharmaceutical drug products; global pharma’s entry into the drugs containing cannabis market; the manufacturing, testing and sale of API and pharmaceutical drug products containing cannabis; enhanced supply chain capabilities; strengthened global reach; commercial distribution of drugs containing cannabis; serving global pharma partners; growth of physician prescribed pharmaceutical drug products containing cannabis and this segment of the pharmaceutical industry; additional new drugs and abbreviated new drug applications; producing a cannabis pharmaceutical drug product that would have marketing authorization by regulatory bodies such as the European Medicines Agency and FDA; and future specialized licencing achievements. Forward-looking statements are necessarily based upon a number of estimates and assumptions that, while considered reasonable, are subject to known and unknown risks, uncertainties, and other factors which may cause the actual results and future events to differ materially from those expressed or implied by such forward-looking statements. Such factors include, but are not limited to: general business, economic, competitive, political and social uncertainties; the inability of MediPharm Labs to obtain adequate financing; the delay or failure to receive regulatory approvals; and other factors discussed in MediPharm Labs’ filings, available on the SEDAR website at www.sedar.com. There can be no assurance that such statements will prove to be accurate, as actual results and future events could differ materially from those anticipated in such statements. Accordingly, readers should not place undue reliance on the forward-looking statements and information contained in this news release. Except as required by law, MediPharm Labs assumes no obligation to update the forward-looking statements of beliefs, opinions, projections, or other factors, should they change.
Primary Logo
Source: MediPharm Labs Corp.
Released July 14, 2021
Haven't given up here, gl.. MEDIF$...
Nice news out this morning
Again..wanting for contracts/revenues; so "manufacture"? or trial?, Labs losing my confidence atm..
gl
Medipharm Labs Corp (OTCMKTS: MEDIF) Partners WithMcmaster University To Manufacture Drugs Containing Cannabis Candidates
Published 21 mins ago on July 8, 2021
By Ward Sport
https://www.mmjreporter.com/medipharm-labs-corp-otcmkts-medif-partners-withmcmaster-university-to-manufacture-drugs-containing-cannabis-candidates-41747
Medipharm Labs Corp (OTCMKTS: MEDIF) has spoken about its newly-formed partnership with McMaster University. The two parties want to engage in the development of drugs that contain cannabis candidates. This company has, over the years, operated as a manufacturer of cannabis API and derivative products.
The essence of the new collaboration
MediPharm Labs have a lot to gain from the new partnership, according to analysts. For example, it is an excellent opportunity to collaborate with McMaster University’s massive and diversified roster of clinicians. According to the analysts, such collaboration is what it takes to achieve massive cannabis research and drug development success.
The terms of the deal
The terms of the deal provide the various responsibilities MediPharm Labs will be tasked with moving forward. For instance. MediPharm Labs will sign a separate Statement of Work with every single clinician group. It is a rather transformative period as MediPharm Labs considers how it will take advantage of its Cannabis Drug Licence to ensure the delivery of top-notch clinical trial material that will guarantee patients Good Manufacturing Practice (GMP). It will also be about meeting the various pharmaceutical quality standards and the conformity to investigative protocol. Seeking out regulatory approval also happens to be a part of the plan in the latest deal.
The President and Interim CEO of MediPharm Labs, Keith Strachan, opines, “As a pharmaceutical company specialized in cannabis, MediPharm Labs is excited to participate in the development of drugs containing cannabis. This will further our initiative to fulfill the global need for pharmaceutically approved drugs containing cannabis.”
Strachan believes that it is such a great honor for them to get the opportunity to collaborate with an outstanding partner like McMaster University. He is quick to refer to it as a global leader in medical cannabis research. He looks forward to great times ahead as they prepare to make special announcements regarding specific projects under the Agreement.
See Blue Tilray Celebrates Germany Subsidiary’s Cannabis Success
By Terry Lassitenaz -July 9, 2021
https://hempgazette.com/news/tilray-aphria-germany-hg1478/
Tilray, Inc. has announced its wholly-owned subsidiary in Germany, Aphria RX GmbH, has successfully completed its first harvest of medical cannabis cultivated in Germany for local distribution.
Germany’s medical cannabis market is a particularly lucrative one, representing an estimated 75% of the current EU market. Any doctor in Germany has been able to prescribe medical cannabis since 2017, and depending on the circumstances, health insurance companies pick up the tab.
The brisk uptake of medical cannabis in Germany has resulted in issues in ramping up local production of medicines to meet growing demand, leading to heavy dependence on imports. The final quarter of 2020 saw medical cannabis flower imports into Germany reach more than 3.2 tonnes.
But it’s an ill wind that blows nobody any good – Australia’s MediPharm Labs exported its first shipment of cannabis oil products to Germany back in April and WA’s Little Green Pharma have also done well from the situation.
Germany’s domestic supply issue was expected to ease this year. Tilray/Aphria’s efforts are helping, via its 6,000 square meter indoor growing facility in Neumünster.
Tilray’s Head of International and Chief Strategy Officer, Denise Faltischek, said the achievement was an important milestone.
“It is a testament to the professionalism and dedication of our team that, despite the challenges of a global pandemic, we remained on track as the first licensed producer to cultivate medical cannabis in Germany,” said Ms. Faltischek. “Our achievement would not have been possible without the trust and cooperation of the Federal Institute for Drugs and Medical Devices (BfArM) and the Germany Cannabis Agency, for which we are sincerely grateful.”
Tilray says it was the first company to import medical cannabis products into the EU and that the EU is central to its worldwide distribution network.
In other recent news from the company, late last month it launched a new medical cannabis brand – Symbios. Tilray said the new brand, targeting Canada, was developed to provide a broader spectrum of products and cannabinoid ratios at a better price point – and that’s what the fans want to hear. The product line includes flower, oils, and pre-rolls.
MediPharm gets Australian government grant for cannabis project
Published 10 mins ago
https://mjbizdaily.com/medipharm-receives-australian-government-grant-for-cannabis-project/
The Australian subsidiary of Barrie, Ontario-headquartered MediPharm Labs has received a government grant worth 466,577 Australian dollars ($352,000) to help fund a capacity optimization project at its cannabis extraction facility in Wonthaggi, Victoria.
The grant is part of Australia’s AU$50 million Manufacturing Modernization Fund, which offers funding to help small- and medium-sized manufacturing businesses scale up and compete internationally.
MediPharm Labs Australia Pty Ltd, the recipient of the grant, will use the funding for its Manufacturing Innovation and Capacity Optimization Project, according to the government announcement.
The project carries a total price tag of AU$1.9 million.
“Current operations have been focused on purified cannabis API development, which includes raw material extraction and cannabis oil purification. With increasing number of customers both domestically and internationally MediPharm Labs needs to increase its throughput of finished goods manufacturing,” a spokesperson for MediPharm in Australia told MJBizDaily via email.
“On top of the current orders for finished products, MediPharm Labs is about to bring online its soft gel cap line.”
The spokesperson said MediPharm is exploring vapes and topicals produced under good manufacturing practice conditions.
“This investment helps realize these initiatives quicker.”
The Manufacturing Modernization Fund is currently closed to applications.
MediPharm took possession of 100% of its Australian subsidiary last September, increasing its stake from 80%.
The 10,000 sq. ft. facility received its Australian Office of Drug Control manufacturing license in May 2019, according to a regulatory filing.
Shares of MediPharm are traded on the Toronto Stock Exchange as LABS.
There are silver linings I suppose to Medipharm's PPS. My shares are stuck. Can't inject any more to further reduce my cost basis without "signs-of-life".
Medipharm just looks dead in the water until they get more EU approvals. That could take another 6 - 12 months, if ever.
Expect another capital raise before then to keep the lights on. That's the definition of "bad dilution".
New money on dips at the other extractor is the better play.
It's just a little sad that Medipharm's big news is executing a research agreement while Valens is rocking and rolling with new 3rd party product deals and accretive acquisitions.
I fear Medipharm is a bit lost in the weeds.
Hopefully they can find their way.
Medipharm Labs Has Been Executing Flawlessly On Its Pharma Strategy
EDITORIAL Jun 25, 2021
7:33 AM EDT
BY MICHAEL BERGER
https://technical420.com/cannabis-article/medipharm-labs-has-been-executing-flawlessly-on-its-pharma-strategy/#
A few months ago, Jazz Pharmaceuticals plc (Nasdaq: JAZZ) acquired GW Pharmaceuticals for more than $7 billion and the transaction represented a major milestone for the cannabis industry.
GW was the first company to bring a cannabis-based therapy through US Food and Drug Administration (FDA) clinical trials and proved there are medical benefits associated with cannabis.
The size of the acquisition was eye-popping, and we have been focused on identifying companies that are focused on the biotech side of the cannabis industry. MediPharm Labs Corp. (TSX: LABS) (OTCQX: MEDIF) (FSE: MLZ) is a Canadian cannabis company that has expanded the business and we are favorable on how the management team has been executing.
MediPharm is Positioned to Record Strong Growth
Although MediPharm’s trend has been to the downside, we believe the business is positioned to record strong growth and are favorable on its leverage to the biotech industry. Earlier this week, MediPharm announced a research partnership master agreement with McMaster University. The relationship is expected to play a key role in the cannabis research and drug development that is being conducted by MediPharm and believe the market does not appreciate the growth prospects that are associated with it.
MediPharm’s cannabis drug license and expertise positions it to supply clinical trial material, assist in investigation protocol and provide regulatory support for multiple trials. Three initial proposed clinical trials will evaluate the effectiveness of proprietary tetrahydrocannabinol (THC) and cannabidiol (CBD) drug candidates for indications that include pain, insomnia associated with major depression, and uremic pruritus.
MediPharm is considered to be a global leader in specialized, research-driven pharmaceutical-quality development and manufacturing of cannabis active pharmaceutical ingredients (API) and derivative products. We are favorable on the amount of value that can be generated through the relationship and will monitor how the story advances from here.
A Growth Story that is Flying Under the Radar
Over the next few months, MediPharm is expected to make additional announcements in regards to specific projects that fall under the agreement as well as the projects that receive support from Health Canada.
At current levels, we believe that MediPharm has a compelling valuation and a favorable risk-reward profile. The company has been quietly executing on the biotech side of the cannabis industry and we are bullish on this aspect of the story.
Yes, seems like noise/static. Show me the money!
Pisd Monday, 06/21/21 09:01:33 AM
Re: FUNMAN post# 6613
Post # 6614 of 6617
similar to this one?.. https://www.medipharmlabs.com/news/press-releases/detail/154/medipharm-labs-initiates-clinical-trial-for-use-of-cannabis
Mr. Taves is responsible for setting and overseeing implementation of BMOCM’s strategies and for all balance sheet and risk taking activity as well as regulatory, compliance and operational functions.
Chris Taves was appointed Chief Operating Officer, BMO Capital Markets in January 2018. In this role, he has oversight of all balance sheet and risk taking activity, as well as regulatory, compliance, and operational functions to ensure they are optimized to satisfy client needs and BMO FG requirements.
Mr. Taves brings over 20 years of customer facing experience covering corporate and institutional clients across a wide range of products, including derivatives, investments, and corporate finance, working closely with I&CB and all areas within Trading Products. Since joining BMO in 2009, he has taken on broader mandates within the Trading Products team, most recently as Co-Head of Global Trading Products.
Mr. Taves is part of the BMO Capital Markets Operating and Executive Committees, the board of BMO Capital Market’s US broker-dealer – BMO Capital Markets Corp., the board of BMO China, and various other leadership committees within BMO Capital Markets and BMO Financial Group.
Mr. Taves holds an MBA from the Ivey School of Business at the University of Western Ontario, a Mathematics degree from the University of Waterloo, and is a Chartered Professional Accountant. He is a member of the United Way Toronto and York Region Major Individual Giving Cabinet and a Director on the board of Armagh, a second-stage housing program for abused women, Operating and Executive committees.
Deep Connections ? Also serves as board member BMO China Co.
https://www.bmo.com/asia/about_BMO_China.html
Use MEDIF$ products as a standard or baseline, awesome. Now let's see Keith promoted to CEO, or, once when bringing in some big name BOD's was said we would be benefitting from the "deep connections" etc. So let's see it, bring it. Acting CEO for months now.
MediPharm Labs Appoints BMO Capital Markets Executive to Board of Directors
TORONTO, July 14, 2020...appointment of Chris Taves – Chief Operating Officer (“COO”) of BMO Capital Markets – to its Board of Directors effective July 13, 2020. This key appointment follows an extensive planning and search process.
As COO of BMO Capital Markets (“BMOCM”), a leading full-service financial services provider and member of BMO Financial Group, one of the largest banks in North America, Mr. Taves is responsible for setting and overseeing implementation of BMOCM’s strategies and for all balance sheet and risk taking activity as well as regulatory, compliance and operational functions. He also serves as a Board Member of BMO China Co. and BMO Capital Markets Corp. Prior to assuming his current role in 2018, he served as Head of Global Markets responsible for BMO’s global trading businesses and in various other roles over an 11-year career at BMO...
...Mr. Taves began his career at KPMG in 1989 and joined Citigroup in 1997 where he became Head of Corporate Canada Team, Derivatives & Structured Product before moving to BMOCM. He has an MBA from the Ivey Business School at Western University, and a Bachelor of Mathematics from the University of Waterloo. He is a CA CPA and a member of the National Association of Corporate Directors.
MediPharm Labs Appoints Former Nestlé Canada President and CEO, Shelley Martin, to Board of Directors
TORONTO, June 22, 2020...appointment of Shelley Martin – former President and CEO of Nestlé Canada and a consumer packaged goods industry top executive with an outstanding track record of success – to its Board of Directors effective immediately. This key appointment follows an extensive planning and search process.
Ms. Martin served in a variety of senior executive roles at Nestlé Canada Inc. from 1990 until she retired after five years as President and Chief Executive Officer in 2018. During her time leading Nestlé Canada, she drove a substantial increase in revenue, market share and profitability and transformed core business units and brands by introducing new formulas, packaging, pricing, global sources of supply and Lean (Six Sigma) tools. In 2018, Nestlé Canada’s annual sales were approximately $2.6 billion. She began her career at General Mills Canada in 1985 and was named one of Canada’s Most Powerful Women by the Women’s Executive Network (WXN) in 2015, 2016 and 2018...
...Ms. Martin is a member of the Advisory Board of Moosehead Breweries as well as Crosby Molasses, and is a Director of Vineland Research and Innovation Centre, a leader in horticultural research and innovation. From 2016 to 2018, she served as Board Chair of Food & Consumer Products of Canada (FCPC), which represents more than 100 food, beverage, and consumer product manufacturers of all sizes. From 2013 to 2018, she was a Director of The Grocery Foundation, a not-for-profit organization that has raised over $90 million for student nutrition programs. Ms. Martin is a graduate of Wilfrid Laurier University (Bachelor of Business Administration) and earned the Institute of Corporate Directors ICD. D designation in 2016...
MediPharm Labs Appoints Former President of Janssen Inc., one of the Janssen Pharmaceutical Companies of Johnson & Johnson, to Board of Directors
TORONTO, Aug. 04, 2020...announce the appointment of Chris Halyk – former President of Janssen Inc. (Canada), one of the Janssen Pharmaceutical Companies of Johnson & Johnson – to its Board of Directors, effective immediately. This is the third appointment that the Company’s Board has made this year to prepare MediPharm Labs for its next stage of international growth and development.
Mr. Halyk has 35 years of pharmaceutical industry experience including 33 years with Johnson & Johnson. Among his many career highlights, he served as: Vice-President, Sales and Marketing and a member of the Janssen Management Board with responsibility for innovations in direct-to-consumer advertising, patient education and sales force automation; Managing Director of Ortho Biotech, the biopharmaceutical division of Janssen Inc.; and from 2006 until his retirement in 2019 as the President of Janssen Inc. (Canada) where under his leadership, the company became the largest pharmaceutical company in Canada. During his tenure, his accomplishments included, bringing innovative products, services and solutions to market along with new technologies and treatments in oncology, immunology, neuroscience, infectious diseases and vaccines, cardiovascular and metabolism and pulmonary hypertension...
..."I look forward to participating actively in its (Medipharm's) development.”
...Mr. Halyk holds an Honours Business Administration degree from Ivey Business School at Western University. He served as the Chair of the Board of Directors for Rx&D, the industry association for Canada’s Research-Based Pharmaceutical Companies (now named Innovative Medicines Canada) from 2012 until 2013. Before joining Johnson & Johnson, Mr. Halyk worked for pharmaceutical company Warner-Lambert in Toronto...
MediPharm Labs Enters Research Agreement with McMaster University to Develop Drugs Containing Cannabis Candidates
https://www.medipharmlabs.com/news/press-releases/detail/185/medipharm-labs-enters-research-agreement-with-mcmaster
June 21, 2021
MediPharm Labs’ Cannabis Drug Licence and expertise qualifies and positions the Company to supply clinical trial material, assist in investigation protocol and provide regulatory support for multiple trials
Three initial proposed clinical trials will evaluate the effectiveness of proprietary THC and CBD drug candidates for multiple indications including Pain, Insomnia associated with Major Depression, and Uremic Pruritus.
BARRIE, Ontario, June 21, 2021 (GLOBE NEWSWIRE) -- MediPharm Labs Corp. (TSX: LABS) (OTCQX: MEDIF) (FSE: MLZ) (“MediPharm Labs” or the “Company”) a global leader in specialized, research-driven pharmaceutical-quality development and manufacturing of cannabis API and derivative products, is pleased to announce it has entered into a research partnership Master Agreement (the “Agreement”) with McMaster University. The Agreement is a key step that enables MediPharm Labs to work with McMaster University’s large and diversified roster of clinicians leading the way in cannabis research and drug development.
Under the terms of the Agreement, MediPharm Labs will enter into a separate Statement of Work with each clinician group. MediPharm Labs will use its Cannabis Drug Licence to provide access to clinical trial material that meets pharmaceutical quality standards and Good Manufacturing Practice (GMP), as well as investigative protocol and regulatory approval support.
MediPharm Labs and McMaster University researchers have proposed three distinct clinical trials, each led by separate clinician groups looking to develop novel cannabis-based drugs to treat different indications including pain, insomnia associated with major depression, and uremic pruritus.
MediPharm Labs will assist in the development of the study drugs that will be evaluated, and each will name a Principal Investigator physician. MediPharm Labs’ expertise in formulation and delivery methods will be critical to the execution of these trials. Before each study begins, all necessary approvals from relevant regulatory authorities, including Health Canada, will be obtained.
“As a pharmaceutical company specialized in cannabis, MediPharm Labs is excited to participate in the development of drugs containing cannabis. This will further our initiative to fulfill the global need for pharmaceutically approved drugs containing cannabis. We are honoured to partner with McMaster University, a global leader in medical cannabis research.” said Keith Strachan, President and Interim CEO, MediPharm Labs.
The Company looks forward to further announcements regarding specific projects under the Agreement as the projects receive initial Health Canada support.
About MediPharm Labs
Founded in 2015, MediPharm Labs specializes in the development and manufacture of purified, pharmaceutical-quality cannabis concentrates, active pharmaceutical ingredients (API) and advanced derivative products utilizing a Good Manufacturing Practices certified facility with ISO standard-built clean rooms. MediPharm Labs has invested in an expert, research driven team, state-of-the-art technology, downstream purification methodologies and purpose-built facilities with five primary extraction lines for delivery of pure, trusted and precision-dosed cannabis products for its customers. Through its wholesale and white label platforms, MediPharm Labs formulates, develops (including through sensory testing), processes, packages and distributes cannabis extracts and advanced cannabinoid-based products to domestic and international markets. As a global leader, MediPharm Labs has completed commercial exports to Australia and completed commercialization of its Australian extraction facility which generated its first revenues in H1 2020. MediPharm Labs Australia was established in 2017.
Cautionary Note Regarding Forward-Looking Information:
This news release contains “forward-looking information” and “forward-looking statements” (collectively, “forward-looking statements”) within the meaning of the applicable Canadian securities legislation. All statements, other than statements of historical fact, are forward-looking statements and are based on expectations, estimates and projections as at the date of this news release. Any statement that involves discussions with respect to predictions, expectations, beliefs, plans, projections, objectives, assumptions, future events or performance (often but not always using phrases such as “expects”, or “does not expect”, “is expected”, “anticipates” or “does not anticipate”, “plans”, “budget”, “scheduled”, “forecasts”, “estimates”, “believes” or “intends” or variations of such words and phrases or stating that certain actions, events or results “may” or “could”, “would”, “might” or “will” be taken to occur or be achieved) are not statements of historical fact and may be forward-looking statements. In this news release, forward-looking statements relate to, among other things, the successful performance of the Agreement; the entering into separate Statements of Work; the occurrence of three distinct clinical trials; the development of novel cannabis-based drugs to treat different indications including pain, insomnia associated with major depression, and uremic pruritus; obtaining all necessary approvals from relevant regulatory authorities, including Health Canada; and the development of pharmaceutical drugs containing cannabis. Forward-looking statements are necessarily based upon a number of estimates and assumptions that, while considered reasonable, are subject to known and unknown risks, uncertainties, and other factors which may cause the actual results and future events to differ materially from those expressed or implied by such forward-looking statements. Such factors include, but are not limited to: general business, economic, competitive, political and social uncertainties; the inability of MediPharm Labs to obtain adequate financing; the delay or failure to receive regulatory approvals; and other factors discussed in MediPharm Labs’ filings, available on the SEDAR website at www.sedar.com. There can be no assurance that such statements will prove to be accurate, as actual results and future events could differ materially from those anticipated in such statements. Accordingly, readers should not place undue reliance on the forward-looking statements and information contained in this news release. Except as required by law, MediPharm Labs assumes no obligation to update the forward-looking statements of beliefs, opinions, projections, or other factors, should they change.
For further information, please contact:
MediPharm Labs Investor Relations
Telephone: +1 416.913.7425 ext. 1525
Email: investors@medipharmlabs.com
Website: www.medipharmlabs.com
Primary Logo
Source: MediPharm Labs Corp.
Released June 21, 2021
..too little too late then quits. The turnaround plan is my play Australia operations/licensing, Germany but Pat won't be getting any credit, too bad..
No kidding..Pat can move on..make Keith permanent CEO.. MEDIF$...
MediPharm Labs Announces Voting Results From the 2021 Annual Meeting of Shareholders
Notice how Patrick McCutcheon basically got an angry vote of no confidence.
For: 27,289,324
Votes withheld 19,689,547
MediPharm Labs Corp.
Thu, June 10, 2021, 7:01 AM·4 min read
https://finance.yahoo.com/news/medipharm-labs-announces-voting-results-110100760.html
BARRIE, Ontario, June 10, 2021 (GLOBE NEWSWIRE) -- MediPharm Labs Corp. (TSX: LABS) (OTCQX: MEDIF) (FSE: MLZ) (“MediPharm Labs” or the “Company”) a global leader in specialized, research-driven pharmaceutical-quality cannabis extraction, distillation and derivative products, today announced the results of matters voted on at its annual meeting of holders of common shares held on Wednesday, June 9, 2021 (the “Meeting”). The voting results for each of the matters presented at the Meeting are outlined below.
There were 361 shareholders represented virtually or by proxy at the Meeting holding 65,230,720 common shares, representing 25.29% of MediPharm Labs total issued and outstanding common shares as at the record date for the Meeting. As the Meeting was held virtually, all resolutions were passed by a ballot vote.
1. Election of Directors
- ADVERTISEMENT -
Each of the nominees for election as directors were elected as directors of MediPharm Labs for the ensuing year or until their successors are elected or appointed. Voting results for the election of the individual directors are as set out below:
Nominee
Votes For
Votes Withheld
#
#
Warren Everitt
44,825,123
2,153,748
Chris Halyk
44,759,019
2,219,852
Shelley Martin
44,775,094
2,203,777
Patrick McCutcheon
27,289,324
19,689,547
Miriam McDonald
44,443,162
2,535,709
Keith Strachan
44,606,498
2,372,373
Dr. Paul Tam
42,048,386
4,930,485
Chris Taves
44,932,867
2,046,004
2. Appointment of Auditor
KPMG LLP, Chartered Professional Accountants, was appointed auditor of MediPharm Labs until the next annual meeting of the holders of the Shareholders at remuneration to be fixed by the directors. Voting results are as set out below:
Votes For
Votes Withheld
#
#
62,195,505
2,975,447
About MediPharm Labs
Founded in 2015, MediPharm specializes in the development and manufacture of purified, pharmaceutical-quality cannabis concentrates, active pharmaceutical ingredients (API) and advanced derivative products utilizing a Good Manufacturing Practices certified facility with ISO standard-built clean rooms. MediPharm has invested in an expert, research driven team, state-of-the-art technology, downstream purification methodologies and purpose-built facilities with five primary extraction lines for delivery of pure, trusted and precision-dosed cannabis products for its customers. Through its wholesale and white label platforms, MediPharm formulates, develops (including through sensory testing), processes, packages and distributes cannabis extracts and advanced cannabinoid-based products to domestic and international markets. As a global leader, MediPharm has completed commercial exports to Australia and completed commercialization of its Australian extraction facility which generated its first revenues in H1 2020. MediPharm Australia was established in 2017.
CAUTIONARY NOTE REGARDING FORWARD-LOOKING INFORMATION:
This news release contains “forward-looking information” and “forward-looking statements” (collectively, “forward-looking statements”) within the meaning of the applicable Canadian securities legislation. All statements, other than statements of historical fact, are forward-looking statements and are based on expectations, estimates and projections as at the date of this news release. Any statement that involves discussions with respect to predictions, expectations, beliefs, plans, projections, objectives, assumptions, future events or performance (often but not always using phrases such as “expects”, or “does not expect”, “is expected”, “anticipates” or “does not anticipate”, “plans”, “budget”, “scheduled”, “forecasts”, “estimates”, “believes” or “intends” or variations of such words and phrases or stating that certain actions, events or results “may” or “could”, “would”, “might” or “will” be taken to occur or be achieved) are not statements of historical fact and may be forward-looking statements. Forward-looking statements are necessarily based upon a number of estimates and assumptions that, while considered reasonable, are subject to known and unknown risks, uncertainties, and other factors which may cause the actual results and future events to differ materially from those expressed or implied by such forward-looking statements. Such factors include, but are not limited to: general business, economic, competitive, political and social uncertainties; the inability of MediPharm Labs to obtain adequate financing; the delay or failure to receive regulatory approvals; and other factors discussed in MediPharm Labs’ filings, available on the SEDAR website at www.sedar.com. There can be no assurance that such statements will prove to be accurate, as actual results and future events could differ materially from those anticipated in such statements. Accordingly, readers should not place undue reliance on the forward-looking statements and information contained in this news release. Except as required by law, MediPharm Labs assumes no obligation to update the forward-looking statements of beliefs, opinions, projections, or other factors, should they change.
CONTACT: For further information, please contact: Laura Lepore, VP, Investor Relations and Communications Telephone: 416-913-7425 ext. 1525 Email: investors@medipharmlabs.com Website: www.medipharmlabs.com
Gosh, let's hope so.
Pisd Tuesday, 06/08/21 01:22:26 PM
Re: FUNMAN post# 6607
Post # 6608 of 6608
Dredging lows here imo.. MEDIF$...
Dredging lows here imo.. MEDIF$...
MediPharm Labs Extends Strategic Partnership with ADREXpharma and continues to Build its Presence in Germany
https://www.medipharmlabs.com/news/press-releases/detail/183/medipharm-labs-extends-strategic-partnership-with
June 08, 2021
MediPharm and ADREXpharma continue to lead the way addressing rising patient demand in the burgeoning German Medical Cannabis Market that saw sales increase 34% last year
128,000 active patients at end of 2020; German patients spent an average of $500EUR per month, finished products and extract sales continue to gain market share
MediPharm and ADREXpharma on track to deliver subsequent shipments to Germany in Q2 2021, completed first shipments in Q1 2021
ADREXpharma is Germany’s largest independent privately owned Medical Cannabis company with deep pharma market expertise.
TORONTO, June 08, 2021 (GLOBE NEWSWIRE) -- MediPharm Labs Corp. (TSX: LABS) (OTCQX: MEDIF) (FSE: MLZ) (“MediPharm” or the “Company”), a global leader in specialized, research-driven pharmaceutical-quality development and manufacture of cannabis API and derivative products, is pleased to announce that it has extended its supply agreement (the Agreement) with ADREXpharma® GmbH (“ADREX”), a leading developer and distributor of medicinal cannabis in the European market. The Agreement was renewed for 5 years, to June 2026, with a mutual option to extend further.
MediPharm supplies ADREX with high quality, purity assured, THC and CBD cannabis products for sale and distribution in Germany to approximately 19,000 pharmacies that could provide access to cannabis products over time.
Over the past year, with the support of ADREX, MediPharm has firmly established its presence and foothold in Germany as the go-to-supplier of cannabis products and regulatory leader in the field of cannabis for pharmaceutical companies. MediPharm and ADREX pioneered and progressed German regulatory acceptance of EU GMP cannabis API and finished dosed products from Canada and Australia.
As a result, MediPharm successfully completed its first shipment of cannabis product to ADREX in Q1 2021 and is on track to deliver its next shipments to Germany in Q2 2021.
“With our global platform purpose built to good manufacturing practices, MediPharm has established a strong foothold in the nascent, but burgeoning, German cannabis market,” said Keith Strachan, President and Interim CEO, MediPharm. “Our collaboration with ADREX has been an important and strategic step in our growth in this attractive market. We will continue to work together, combining ADREX’s experience and local networks with our licences, production and supply chain capabilities, to ensure German patients gain access to the high-quality cannabis products they can trust.”
“We are excited having launched the first products produced by MediPharm in the growing German market and to expand our international footprint with this strategic collaboration,” said Mario Eimuth, Founding Partner, ADREXpharma. “Our mission is to serve and offer the best therapeutical options to all patients in need in Germany. MediPharm is a leading global manufacturer of medical cannabis products and we are looking forward to expanding our partnership even further in the next coming months in order to meet the increasing demand in the German market.”
Germany has become Europe’s largest market with a patient base that has grown to 128,000. This helped drive Germany’s medical cannabis market sales up 34% in 2020. Also, according to Prohibition Partners latest European report, Germany’s patient base is expected to continue to increase as development of the medical cannabis system is still ongoing, with the streamlining of wholesale pricing regulations being an important next step. The market is also expected to continue to move towards more extract-based medicine instead of flower-based products.
About ADREXpharma
ADREXpharma® GmbH is an independent German pharmaceutical company which is specialized in the development and distribution of medicinal cannabis in the European market. ADREXpharma developed several dosages (extracts, powder, resin, flowers) for the growing pharma market in Germany. Due to its unique supply network, ADREXpharma serves all of the approximate 19,000 pharmacies in Germany and is market-leader in pharmacies for several product categories. ADREXpharma is committed to quality, authenticity and innovation, as well as to contributing to the growing body of evidence-based research regarding the benefits and efficacy of cannabis to validate new therapy options for patients. ADREXpharma aims to provide the best possible treatment for medical cannabis patients – today and always.
About MediPharm
Founded in 2015, MediPharm specializes in the development and manufacture of purified, pharmaceutical-quality cannabis concentrates, active pharmaceutical ingredients (API) and advanced derivative products utilizing a Good Manufacturing Practices certified facility with ISO standard-built clean rooms. MediPharm has invested in an expert, research driven team, state-of-the-art technology, downstream purification methodologies and purpose-built facilities with five primary extraction lines for delivery of pure, trusted and precision-dosed cannabis products for its customers. Through its wholesale and white label platforms, MediPharm formulates, develops (including through sensory testing), processes, packages and distributes cannabis extracts and advanced cannabinoid-based products to domestic and international markets. As a global leader, MediPharm has completed commercial exports to Australia and completed commercialization of its Australian extraction facility which generated its first revenues in H1 2020. MediPharm Australia was established in 2017.
Cautionary Note Regarding Forward-Looking Information:
This news release contains “forward-looking information” and “forward-looking statements” (collectively, “forward-looking statements”) within the meaning of the applicable Canadian securities legislation. All statements, other than statements of historical fact, are forward-looking statements and are based on expectations, estimates and projections as at the date of this news release. Any statement that involves discussions with respect to predictions, expectations, beliefs, plans, projections, objectives, assumptions, future events or performance (often but not always using phrases such as “expects”, or “does not expect”, “is expected”, “anticipates” or “does not anticipate”, “plans”, “budget”, “scheduled”, “forecasts”, “estimates”, “believes” or “intends” or variations of such words and phrases or stating that certain actions, events or results “may” or “could”, “would”, “might” or “will” be taken to occur or be achieved) are not statements of historical fact and may be forward-looking statements. In this news release, forward-looking statements relate to, among other things, the successful performance of the agreement as planned; delivering subsequent shipments to Germany in Q2 2021; having a strong foothold in the nascent, but burgeoning, German cannabis market; having a foothold in Germany as the go-to-supplier of cannabis products and regulatory leader in the field of cannabis for pharmaceutical companies; growth in the German market; and growth of the German cannabis market. Forward-looking statements are necessarily based upon a number of estimates and assumptions that, while considered reasonable, are subject to known and unknown risks, uncertainties, and other factors which may cause the actual results and future events to differ materially from those expressed or implied by such forward-looking statements. Such factors include, but are not limited to: general business, economic, competitive, political and social uncertainties; the inability of MediPharm to obtain adequate financing; the delay or failure to receive regulatory approvals; and other factors discussed in MediPharm’s filings, available on the SEDAR website at www.sedar.com. There can be no assurance that such statements will prove to be accurate, as actual results and future events could differ materially from those anticipated in such statements. Accordingly, readers should not place undue reliance on the forward-looking statements and information contained in this news release. Except as required by law, MediPharm assumes no obligation to update the forward-looking statements of beliefs, opinions, projections, or other factors, should they change.
All information contained in this press release with respect to ADRES was supplied by ADREX for inclusion herein.
For further information, please contact:
Laura Lepore, VP, Investor Relations
Telephone: +1 416.913.7425 ext. 1525
Email: investors@medipharmlabs.com
Website: www.medipharmlabs.com
"...The recreational cannabis operator opted to expand its product offering in the German market by launching a range of specially formulated high-THC, balanced-THC and CBD oil extracts as part of a two-year supply agreement between its German subsidiary Adjupharm GmbH and MediPharm Labs (OTC: MEDIF)..."
The Flowr Corp. To Export Up To 500KG Of Medical Cannabis To Israel
1:35 pm ET June 7, 2021 (Benzinga) Print
The Flowr Corporation (OTCQX: FLWPF) will export up to 500 kilos of premium dry cannabis flower in bulk into Israel.
The move follows the company's recent announcement that it had raised $15.9 million by selling units.
Deal Details: Under the terms of the supply agreement with Focus Medical Herbs Ltd., a company with which IM Cannabis Corp. (NASDAQ: IMCC) (CSE: IMCC) has an exclusive commercial agreement within Israel, Flowr's wholly-owned subsidiary will export medical cannabis into the Middle Eastern country.
The Toronto-based company said Monday that it expects the first shipment of BC Pink Kush to hit the Israeli market in the second half of this year.
Flowr confirmed Monday that the collaboration with Focus Medical will last three years.
Darryl Brooker, CEO of Flowr, said he is delighter to team up with IMC and Focus Medical to introduce the company's product to the Israeli market.
"Coupled with our ability to grow high THC premium dry flower from our EU GMP facilities in Portugal, we view this partnership as an important next step to becoming a significant international producer of cannabis with a globally recognized brand," Brooker continued.
Oren Shuster, CEO of IMC, looks forward to working with the Flowr team.
"We believe that the Israeli market will receive Flowr's renowned BC Pink Kush and BC Black Cherry strains with open arms," Shuster added.
IMC's Recent Moves: IMC debuted on the NASDAQ Capital Market last year.
In April, the company confirmed the acquisition of MYM Nutraceuticals and its Licensed Producer subsidiary Highland Growth, which is scheduled to close by the end of 2021.
The recreational cannabis operator opted to expand its product offering in the German market by launching a range of specially formulated high-THC, balanced-THC and CBD oil extracts as part of a two-year supply agreement between its German subsidiary Adjupharm GmbH and MediPharm Labs (OTC: MEDIF).
...oops, meant to say a $, MEDIF$, INTERNATIONAL peeps...
Future news will be positive news..go over .50 imo...
buyer .42..seller .50, too easy, maybe more, .60's?.. MEDIF$...
|
Followers
|
73
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
6727
|
|
Created
|
10/05/18
|
Type
|
Free
|
| Moderators | |||
TORONTO, Jan. 29, 2021 (GLOBE NEWSWIRE) -- MediPharm Labs Corp. (TSX: LABS) (OTCQX: MEDIF) (FSE:MLZ) (“MediPharm Labs” or the “Company”) a global leader in specialized, research-driven pharmaceutical-quality cannabis extraction, distillation and derivative products, is pleased to announce Greg Hunter will join MediPharm Labs as Chief Financial Officer (CFO), effective February 8, 2021.
Greg Hunter brings over 20 years of experience as a business executive holding various senior finance and leadership roles across multiple industries including healthcare distribution, telecommunications, pharmaceuticals, biotechnology, medical device and consumer packaged goods. Greg also brings a track record and deep expertise in capital management, audit, compliance, tax, treasury, ERP, manufacturing, contract management and pricing strategy.
“As MediPharm continues to focus on accelerating the growth and execution of our global pharmaceutical, health and wellness business, Greg’s background in growing large pharmaceutical operations combined with a track record in strategic capital allocation and cost management is a strong addition to our Company,” said Keith Strachan, President and Interim CEO, MediPharm Labs. “As we look to seize new growth opportunities and expand our product offerings, his addition will ensure that we are well positioned to execute on our plans and deliver profitable growth in the future.”
Most recently, Greg was Chief Financial Officer of Medical Pharmacies Group Limited, a leading pharmacy and medical equipment manufacturer and distributor in Canada. Previously in the pharmaceuticals industry, Greg held various senior management roles with Baxter International Inc. including serving as CFO of Baxter’s Canadian subsidiary. Greg also previously held various senior operational and finance roles at Janssen-Ortho Inc., a Johnson and Johnson company.
Keith Strachan added, “I would also like to thank Olga Utkutug for her many contributions and leadership as Interim CFO. We look forward to her continued support as a key member of our finance team.”
Greg holds an MBA from McMaster University, an Honors B.Sc. in Microbiology and Immunology from University of Western Ontario and is a Chartered Professional Accountant.
In connection with Greg’s appointment as CFO, the Company granted 600,000 stock options to him with an exercise price set at the close of business on January 28, 2021. Each grant has a five-year term expiring January 28, 2026, and vests in five equal instalments, the first of which vests immediately with the four other instalments vesting on the dates which are six, twelve, eighteen and twenty-four months from the grant date. The stock options are subject to any necessary regulatory approvals.
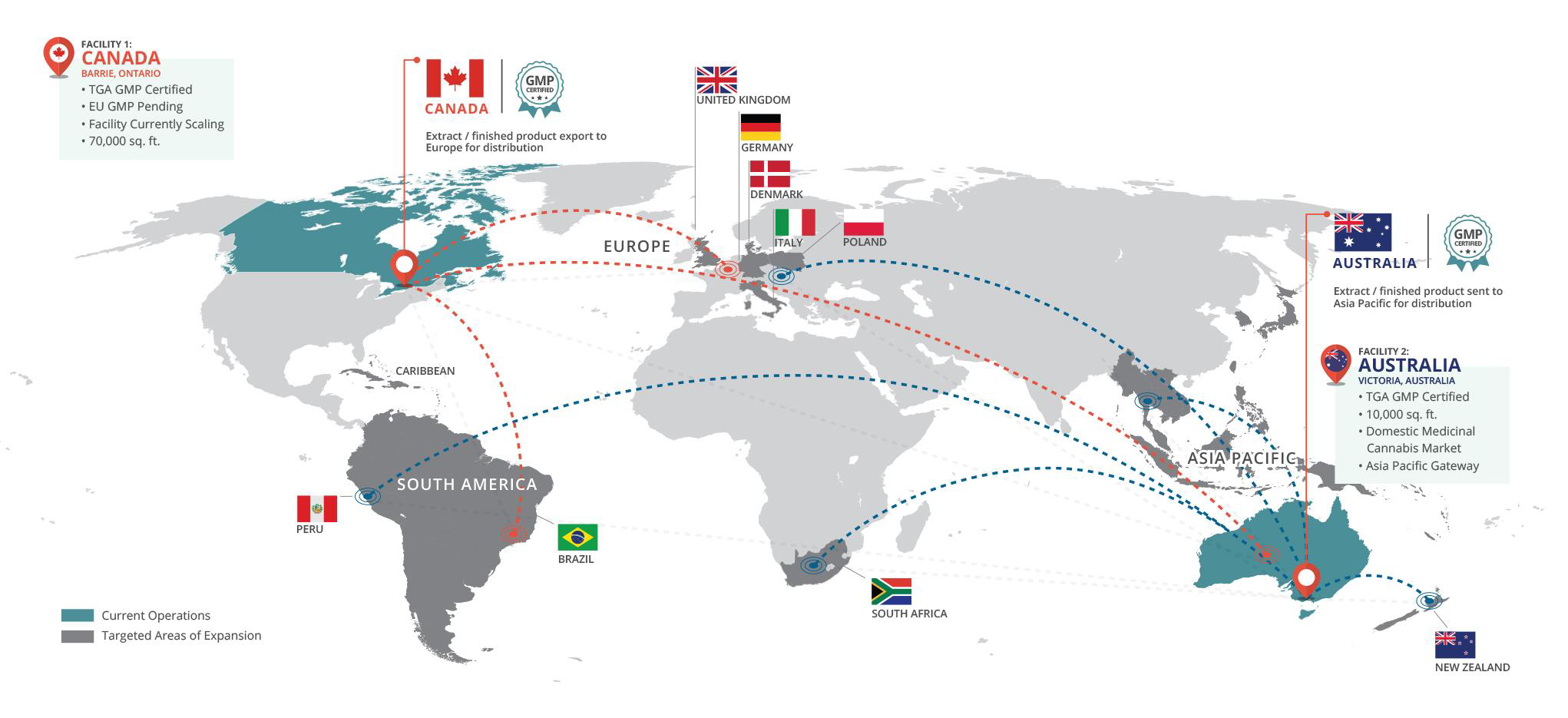
BAD VILBEL, Germany and TORONTO, Oct. 05, 2020 (GLOBE NEWSWIRE) -- As one of the leading European Consumer Healthcare and Generics companies, STADA, with a product presence in 120 countries, has signed an exclusive supply agreement with MediPharm Labs Inc. (“MediPharm”), a wholly owned subsidiary of MediPharm Labs Corp. (TSX: LABS) (OTCQX: MEDIF) (FSE:MLZ) (“MediPharm Labs” or the “Company”) a global leader in specialized, research-driven pharmaceutical-quality development and manufacture of cannabis API and derivative products to provide medical cannabis products for the European pharma sector.
“Working with MediPharm will meet the needs of pharmacists and patients and deliver on STADA’s purpose of caring for people’s health as a trusted partner,” commented STADA CEO Peter Goldschmidt. “This partnership with MediPharm demonstrates STADA’s ambition to be the go-to-partner for Generics, Consumer Health and Specialty Products.”
Under the terms of the exclusive partnership, MediPharm will supply GMP certified medical cannabis products to STADA, as well as manufacturing, logistics, and regulatory support. STADA will be responsible for commercializing the cannabis products, initially in Germany as well as marketing and medical education utilizing a pharmaceutically experienced field force.
“This partnership is exactly the type of business MediPharm has been pursuing since its inception and a validation of our strategy to deliver GMP-certified cannabis products that can be used in multiple new products in multiple markets around the world,” said Pat McCutcheon, CEO, MediPharm Labs. “This mandate has meaningful revenue potential for MediPharm Labs and we couldn’t be more excited to partner with a company that has the reputation and resources that STADA commands. As a powerhouse in sales and distribution of pharmaceuticals and non-prescription consumer health products, STADA has the ability to become a transformative force in European medical cannabis markets and we are thrilled to be their exclusive partner.”
The partners will initially focus on Germany, before potentially expanding to other European countries and territories. The partnership also signals MediPharm’s entry into the global pharmaceutical industry within a major European market.
With over 83 million inhabitants benefitting from broad access to healthcare services, Germany currently represents an estimated three-quarters of the current EU medical cannabis market.
The Medical Cannabis Network reports the medical cannabis market in Germany is currently valued at between €150m and €175m, despite only around 10% of the 20,000 pharmacies in Germany selling medical cannabis products today. Furthermore, only a limited number of doctors in Germany currently prescribe medical cannabis. With greater awareness and education around the benefits of medical cannabis, the Medical Cannabis Network estimates that Germany’s dominance in the European cannabis market could expand to €1.5bn by 2025.(1)
This is a further milestone for the global cannabis industry as STADA, in partnership with MediPharm, forges the way as a large pharmaceutical company to commercialize medical cannabis products.
Details of the products that STADA will bring to market through this partnership with MediPharm Labs will be unveiled in the coming months in the respective relevant communication channels.
June 25th, 2020
TORONTO, June 25, 2020 (GLOBE NEWSWIRE) -- MediPharm Labs Corp. (TSX: LABS) (OTCQX: MEDIF) (FSE:MLZ) (“MediPharm Labs” or the “Company”) a global leader in specialized, research-driven pharmaceutical-quality cannabis extraction, distillation and derivative products, today announced a further expansion of its innovative, pharma-quality family of branded products with the retail introduction of CBD25:5 Release Formula (“CBD25:5”).
MediPharm Labs announces the launch of CBD25:5 Release Formula, a 25:5 CBD:THC formulated oil to harness the properties of these two cannabinoids for patients and adult consumers.

“We’ve positioned ourselves to lead the market in well-defined Cannabis 2.0 categories on our own and through our growing white-label supply business,’” said Pat McCutcheon, Chief Executive Officer, MediPharm Labs. “Today’s announced product launch, coupled with other recently formed partnerships, demonstrate just how far we’ve come in realizing our ambition to transition our focus to commercializing and distributing a robust portfolio of pharma-quality products.”
Cannabis 2.0 Update
In late December 2019, the Company commenced shipments of Cannabis 2.0 products into three of Canada’s provincial jurisdictions – British Columbia, Manitoba and Saskatchewan.
Since then, MediPharm Labs has received multiple reorders and added several notable new customers:
-MediPharm Labs is able to supply and provide tailored product formulations and solutions to its Licensed Producer and CPG customers who serve the four product categories recognized under Schedule 4 of the Cannabis Act: cannabis extracts (for ingestion and inhalation), cannabis topicals, edible cannabis (beverages and food products) and cannabis oils. This broad exposure to Cannabis 2.0 categories is a key business strength.
WATCH THESE VIDEOS:
extraction:
https://youtu.be/HAzs2xhbHgM
soft gel manufacturing:
https://www.youtube.com/watch?v=t9tKO_cFLnY&feature=youtu.be
December 30, 2019
"...MediPharm Labs Inc. (“MediPharm”) has received a licence amendment from Health Canada allowing for production to begin in the recently expanded area of its specialized manufacturing facility in Barrie, Ontario.
This amendment increases MediPharm’s licensed facility footprint by around three times, or 16,746 sq. ft., to a total of approximately 25,000 sq. ft., allowing it to productively use more of its manufacturing space for cannabis activities – including automated downstream production and packaging, cannabis quality control and testing, research and development and secure storage to support the fulfillment and distribution of new product format formulations and orders.
MediPharm Labs Canadian Facility Expansion
In 2019, MediPharm Labs commenced a phased plan to expand its licensed space and scale its operations in Canada within its 70,000 sq.ft. facility in Barrie, Ontario. Under new regulations, the Company was required to complete construction prior to applying for an expansion licence. Today’s announcement means MediPharm received its Health Canada licence amendment just four months after completing construction.
The recently completed first phase of expansion added 16,746 square feet, all of it now licensed. This represents a three-fold increase in total licensed footprint suitable for cannabis activities to a total of approximately 25,000 sq. ft.
The now-completed first phase of the expansion includes:

July 16 update: MediPharm Labs Provides Operational Update and Increases Average Weekly Production of Active Cannabinoid Component to 75 Million Milligrams
Growing Private Label Production and White Label Platform: MediPharm Labs has increased production of private label cannabis concentrate products and distillate supply – including CBD or THC dominant as well as balanced formulations – to meet the growing demand for specialty concentrate based consumer end products ahead of legalization of vapeables, edibles and topicals in the fall of 2019. Production and quality operations teams ramped up weekly production output to approximately 75 million milligrams of active cannabinoid component concentrate at the end of Q2 2019.
To date, MediPharm Labs has signed four significant contracts for the sale of private label cannabis concentrate including to Canopy Growth, Cronos Group and AusCann. The Company also launched its White Label platform to service direct-to-consumer brands, CPG companies and large LPs in Canada. In June 2019, MediPharm announced its first white label deal to produce a minimum of approximately two million vape pens with Ace Valley, subject to purchase orders from provincial distributors.
Increasing Production Volumes: The number of dry cannabis suppliers and concentrate purchasers in Canada continues to increase. In turn, this has resulted in higher demand that supports MediPharm Labs’ differentiated, private label business model.
The Company announced it successfully acquired 9,000 KG of dried cannabis in the last three weeks of June 2019 expected to be processed and sold in Q3 2019, and 5,000 KG in the last two weeks of March 2019 that were processed and sold in Q2 2019.
Enhanced Governance and Quality: To date, the Company has purchased dried cannabis from over 23 suppliers across the country. MediPharm Labs has conducted in-depth qualifications of all of its dry cannabis and product suppliers.
Potential suppliers to MediPharm Labs are subjected to rigorous scrutiny before entering into agreements which require an enhanced Quality Agreement beyond Health Canada standards. This enhanced supplier qualification aligns with our pharmaceutical-like standards for production of cannabis oil and derivative products. All suppliers and customers remain in good standing with Health Canada Licensing and Inspection division.
Broadening Domestic and Global Distribution: During the quarter, MediPharm Labs became the only third-party concentrate manufacturer to begin shipping white label products, including bottled oil, to Provincial cannabis distributors. MediPharm Labs’ white label products can be found in three provinces including Ontario, British Columbia and Alberta. The Company expects to rollout distribution of Health Canada approved products to all remaining provinces during H2 2019.
MediPharm Labs successfully completed its first international export of commercial volume cannabis concentrate to AusCann in Australia in June 2019. The Company continues to focus on accessing and distributing its private label cannabis concentrate to global markets, including across Europe, and is working on various commercial terms.
Expanding Footprint in Canada: At MediPharm Labs’ facility in Barrie, Ontario, the Company is adding over 25,000 square feet of purpose-built production space for filling and packing automation, new product manufacturing, cannabinoid isolation activities and specialized R&D projects. Taking a phased approach, operations in this new space are expected to come as each phase becomes approved by Health Canada during the second half of 2019.
Building Scale and Increasing Capacity: MediPharm Labs has successfully completed several equipment automation and innovation projects. The success of these optimization projects has resulted in an increased in capacity of dry cannabis processing to 300,000 KG annually.
As a global leader in extraction with a focus on providing high quality cannabinoid concentrates, MediPharm Labs recently completed a 14-month project for an additional, fully customized large-scale extraction line. The extractor has completed its factory testing in Europe and is being shipped to the Company’s Barrie, Ontario facility. Upon completion of installation, operator training, EU GMP qualification and regulatory prestart activities, operational capacity of specialized and automated dry cannabis processing will exceed 500,000 KG annually.
New Product Development: MediPharm Labs is continuing to evolve its product mix. The bulk of its production and revenue to date has been in high quality winterized resin. This critical input ingredient will become the key building block for future high-growth and high-margin products:
Soft Gel Caps: MediPharm Labs has focused on building scale across operations and product lines including bottled oil and gel caps. An industrial scale soft gel capsule project is underway. Equipment has been deployed, equipment training has started, and formulation work is being conducted.
Vapourizers: The Company is building the foundation to be one of the largest vaporizer cartridge manufacturers in Canada. With new regulations finalized and set to come into effect on October 17, 2019 MediPharm Labs has finalized various formulations and started vapourizer pen cartridge filling research and development trials to ensure readiness to serve current and future white-label partners.
Australia Gaining Momentum: MediPharm Labs Australia received its manufacturing licence in May from the Australian Office of Drug Control. Construction of the facility is well underway and nearing completion with equipment ordered for installation in H2 2019. MediPharm Labs Australia is expected to have 75,000 KG of annual capacity.
Financing: The Company completed a $75 million bought deal financing to fund capital expenditures at its Canadian and Australian facilities, for domestic and international expansions, research and development and general corporate purposes. Signed a $20 million debt facility term sheet with a Schedule 1 Bank.
MediPharm Labs Announces Addition to $1.2B MJ ETFMG Alternative Harvest ETF (USA)
Tuesday 21 May 2019

TORONTO, May 22, 2019 (GLOBE NEWSWIRE) -- MediPharm Labs Corp. (TSXV: LABS) (OTCQX: MEDIF) (FSE:MLZ) (“MediPharm Labs”) a leader in specialized, research-driven cannabis extraction and cannabinoid isolation, is pleased to announce it has been selected by The Mount Sinai Hospital, New York City (“Mount Sinai”), to participate in a clinical trial dedicated to developing a non-addictive oral gelcap medication for the treatment of opioid use disorder through anti-anxiety intervention utilizing hemp-derived CBD combined with a proprietary formula (the “Formula”). This will be a U.S. and international large-scale, multi-site clinical trial that will include at least 500 patients spanning the United States, Canada, Australia, Europe and Jamaica.
The clinical trial will be led by renowned principal researcher, Dr. Yasmin L. Hurd, PhD, the Ward-Coleman Chair of Translational Neuroscience at the Icahn School of Medicine at Mount Sinai, New York City, and Director of the Center for Addictive Disorders for the Mount Sinai Behavioral Health System...
...MediPharm Labs will be the exclusive manufacturer of a proprietary hemp-derived CBD oral gelcap medication utilizing the Formula provided by Timeless Herbal for all phases of the clinical trials related to this study that will allow researchers to test an investigational product containing CBD active ingredient.
MAY 17, 2019
Emerald Health Therapeutics, Inc. (“Emerald”) (TSXV: EMH; OTCQX: EMHTF) has shipped 6,000 40 ml units of its SYNC 25 CBD oil to the British Columbia Liquor Distribution Branch (BCLDB). The Emerald-branded pure CBD oil offers consumers a non-THC, smoke-free product alternative.
“We are pleased to offer our new Emerald-branded wellness product SYNC 25 to the recreational marketplace in British Columbia,” said Dr. Avtar Dhillon, President and Executive Chairman of Emerald. “The introduction of SYNC 25 marks the next step in our product growth strategy to develop innovative and quality consumer products that respond to a growing demand while providing higher profit margins.”
The SYNC 25 CBD oil product was extracted by MediPharm Labs Inc...
May 15, 2019
MediPharm Labs Corp. (TSXV: LABS) (OTCQX: MEDIF) (FSE:MLZ) (“MediPharm Labs”) a leader in specialized, research-driven cannabis extraction and cannabinoid isolation, is pleased to announce that its wholly-owned subsidiary, MediPharm Labs Inc. (“MediPharm”), has signed a committed term sheet for a $20 million senior secured revolving credit facility ("Credit Facility") from a Canadian Schedule 1 Bank. The Credit Facility is intended to provide MediPharm Labs access to non-dilutive capital to fund planned growth, as well as for general corporate and working capital purposes.
The Credit Facility consists of a $15 million operating loan with a one-year term and a $5 million non-revolving equipment term loan with a three-year term. The Credit Facility, once closed, will bear interest at the Schedule 1 Bank’s prime lending rate plus 1.85% per annum.
“This commitment marks yet another important milestone that will provide access to non-dilutive capital to support our plans for accelerated growth as we continue to ramp up operations ahead of expected legalization of broader concentrate-based products this fall,” said, Patrick McCutcheon, CEO, MediPharm Labs. “We are pleased to have a leading financial institution support MediPharm Labs as we continue to innovate and expand our private and white label offerings to serve a growing demand for high-quality, adult-use and medical cannabis products.”

14 MAY 2019
Cronos Group and MediPharm Labs Enter Additional Multi-Year Tolling Arrangement
TORONTO, May 14, 2019 (GLOBE NEWSWIRE) -- Cronos Group Inc. (NASDAQ: CRON) (TSX: CRON) (“Cronos Group” or the “Company”), is pleased to announce that it has entered into a multi-year supply agreement with MediPharm Labs Corp. (TSXV: LABS) (OTCQX: MEDIF) (“MediPharm Labs”).
MediPharm Labs will supply Cronos Group with approximately $30 million of high-quality private label cannabis concentrate over 18-months, and, subject to certain renewal and purchase options, potentially up to $60 million over 24-months. In addition, Cronos Group and MediPharm Labs have entered into a multi-year tolling agreement, where Cronos Group will supply bulk cannabis to MediPharm Labs’ state of the art extraction facility in Barrie, Ontario, to fulfill certain additional processing needs of the Company.




2019 Year-to-Date Highlights
(Mr. McCutcheon continued)... “Looking ahead, we are now working on an ambitious, well-planned agenda for 2019 that will enable MediPharm Labs to extend our first-mover advantage. We are ramping up production, adding capacity, targeting EU GMP certification, expanding product offerings, developing R&D and IP, signing new sales agreements, and executing on our M&A and international growth pipeline.”
“Most importantly, we see all of this as a starting point. We expect to accelerate our growth globally as the size of our addressable market increases and we strengthen our foothold domestically with the expected legalization of vapeables, edibles, beverages and topicals providing a strong growth trajectory in Canada. We will continue to capitalize on the numerous opportunities available to us through effective capital deployment and continued expert execution to create shareholder value for the long term.”
2019 Strategic Priorities

For further information, please contact:
Laura Lepore, VP, Investor Relations
Telephone: 705-719-7425 ext 216
Email: investors@medipharmlabs.com
Website: www.medipharmlabs.com
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
